The present review summarizes the current
understanding of frailty in elderly patients with multiple myeloma
(MM), focusing on mechanisms, assessment tools, clinical
implications and recent advances. A critical overview of the
literature is provided, controversies and research gaps are
highlighted, and future directions for research and clinical
practice are discussed.
MM is a malignant disorder marked by clonal plasma
cell proliferation, which leads mainly to immunodeficiency and bone
destruction (1,2). MM is the second most common
hematological malignancy (3),
affecting mostly middle-aged and elderly individuals, with a median
onset age of 55–59 years, and is rare in individuals aged <40
years (4). Despite advances in
therapy, MM remains incurable (5,6).
Recent treatment optimization has led to the use of immunotherapy
for MM (7,8), markedly improving survival rates. The
IMROZ trial conducted by Manier et al (9), confirmed that the isatuximab +
bortezomib/lenalidomide/dexamethasone (VRd) regimen notably
improved progression-free survival (PFS) with a manageable safety
profile in frail, transplant-ineligible patients with newly
diagnosed MM (NDMM). In addition, the IFM2017-03 trial completed by
Manier et al (10) showed
that the dexamethasone-sparing regimen (daratumumab + lenalidomide)
could extend PFS to 53.4 months compared with traditional regimens,
providing a safer option for elderly frail patients. The
GEM-2017FIT trial led by Mateos et al (11) demonstrated that the
carfilzomib-lenalidomide-dexamethasone and
daratumumab-carfilzomib-lenalidomide-dexamethasone regimens were
superior compared with bortezomib-melphalan-prednisone (VMP) and
lenalidomide-dexamethasone in terms of minimal residual disease
(MRD) negativity rate, thus providing evidence for regimens adapted
to different frailty statuses. However, drug resistance continues
to drive disease progression (12–14).
Conventional prognostic methods, such as the Second
Revised International Staging System (R2-ISS) (15) and the Mayo additive staging system
(16), do not fully capture
prognosis due to high interpatient variability, particularly among
elderly patients (15–17). In this context, it is important to
note that age alone is not the most important prognostic factor in
elderly patients with MM (18,19).
For example, individuals aged >65 years with good organ function
and physical status may undergo intensive chemotherapy and
autologous transplantation, whereas some individuals aged <65
years with comorbidities and limited psychological resilience might
not tolerate such treatment. As a result, frailty assessment has
become an important and objective tool to guide treatment selection
and predict prognosis in MM (20–23).
Frailty is a nonspecific clinical syndrome that
impairs homeostasis across several physiological systems, including
neurological, metabolic-endocrine and immune domains, and
sarcopenia forms a core element (24). Frailty lowers physiological
reserves, reduces resilience to stress and limits recovery, and is
considered the most clinically notable geriatric syndrome (25,26).
Notably, frailty increases the risk of poor outcomes, including
longer hospital stays, increased treatment toxicity and mortality
(27). Clinicians most widely use
the Fried Frailty Phenotype criteria (19,28) as
clinical diagnostic standards, and the criteria are defined as
follows: i) Unexplained weight loss: ≥5% weight loss or >4.5 kg
weight loss within 1 year (excluding deliberate dieting or other
disease-related causes); ii) fatigue: Feeling exhausted even after
simple activities (such as walking and household chores); iii)
decreased muscle strength: Reduced handgrip strength (such as
difficulty wringing out a towel); iv) reduced activity level:
Avoiding going out and decreased physical activity; and v) slow
walking speed: A walking speed of ≤0.8 m/sec (for example, being
unable to cross the street before the green light changes). Meeting
three or more of these criteria indicates frailty, meeting one or
two indicates pre-frailty and meeting none indicates a healthy
status.
Approximately two-thirds of elderly patients
(patients aged >70 years at diagnosis) with MM have frailty,
with ≥40% experiencing severe forms (29). This condition greatly reduces
quality of life (21,22,30–32),
and can lead to longer hospitalization, increased treatment
toxicity and a higher risk of mortality. Frailty is a dynamic and
possibly reversible process (33,34);
early identification allows interventions such as exercise,
nutrition and disease management to delay its progression and
reduce associated risks. In MM, frailty typically includes physical
decline, fatigue and decreased activity, and is often complicated
by comorbidities or reduced daily functioning. Assessment of
myeloma-related frailty can be performed by the methods described
in this section.
Frail patients often experience fatigue, pain and
reduced quality of life, which are associated with disease stage,
anemia and poor physical performance. Frailty influences treatment
tolerance and prognosis, and is associated with reduced survival
(36,37).
Frailty status may change over time or with
treatment progress, requiring regular assessment (38). Some patients improve following
therapy, whereas others may experience worsening due to disease
progression or treatment side effects (38).
Frailty in the elderly is an age-related syndrome
associated with MM. The influencing factors are complex and diverse
(39), and have been categorized in
the present review as uncontrollable, controllable and other.
The uncontrollable factors that influence frailty
include: i) Genetic factors: Genetic polymorphisms [such as
interleukin (IL)-6 and angiotensinogen genes] may influence
susceptibility to frailty (40,41);
ii) age: With advancing age, organ function declines and
physiological reserves decrease, notably increasing the risk of
frailty (42); and iii) sex: Women
have higher frailty rates than men, mostly due to postmenopausal
estrogen decline that affects muscle and nerve function (43).
The controllable factors that influence frailty
include: i) Lifestyle: Unhealthy habits such as smoking, excessive
alcohol consumption and physical inactivity increase frailty risk
(44); ii) chronic conditions,
including hypertension, diabetes, coronary heart disease, chronic
obstructive pulmonary disease and arthritis, as well as the
presence of multiple coexisting conditions (five or more
comorbidities) are major contributors (45–47);
iii) medications: Inappropriate drug use (such as anticholinergic
agents or antipsychotics) or polypharmacy may contribute to the
onset of frailty (48); iv)
psychological factors, such as anxiety, depression and sleep
disorders interact with frailty (49,50);
and v) socioeconomic factors: Inadequate social support, being
unmarried, living alone, financial hardship or low educational
attainment may elevate frailty risk (51).
Malnutrition can influence frailty as deficiencies
in nutrients, such as protein, vitamin D and calcium, or inadequate
intake caused by decreased appetite or dysphagia, may accelerate
muscle wasting and functional decline (52). In addition, immune dysfunction,
including abnormal inflammatory responses [such as elevated IL-6
and C-reactive protein (CRP) levels] are closely associated with
frailty (53). MM and frailty also
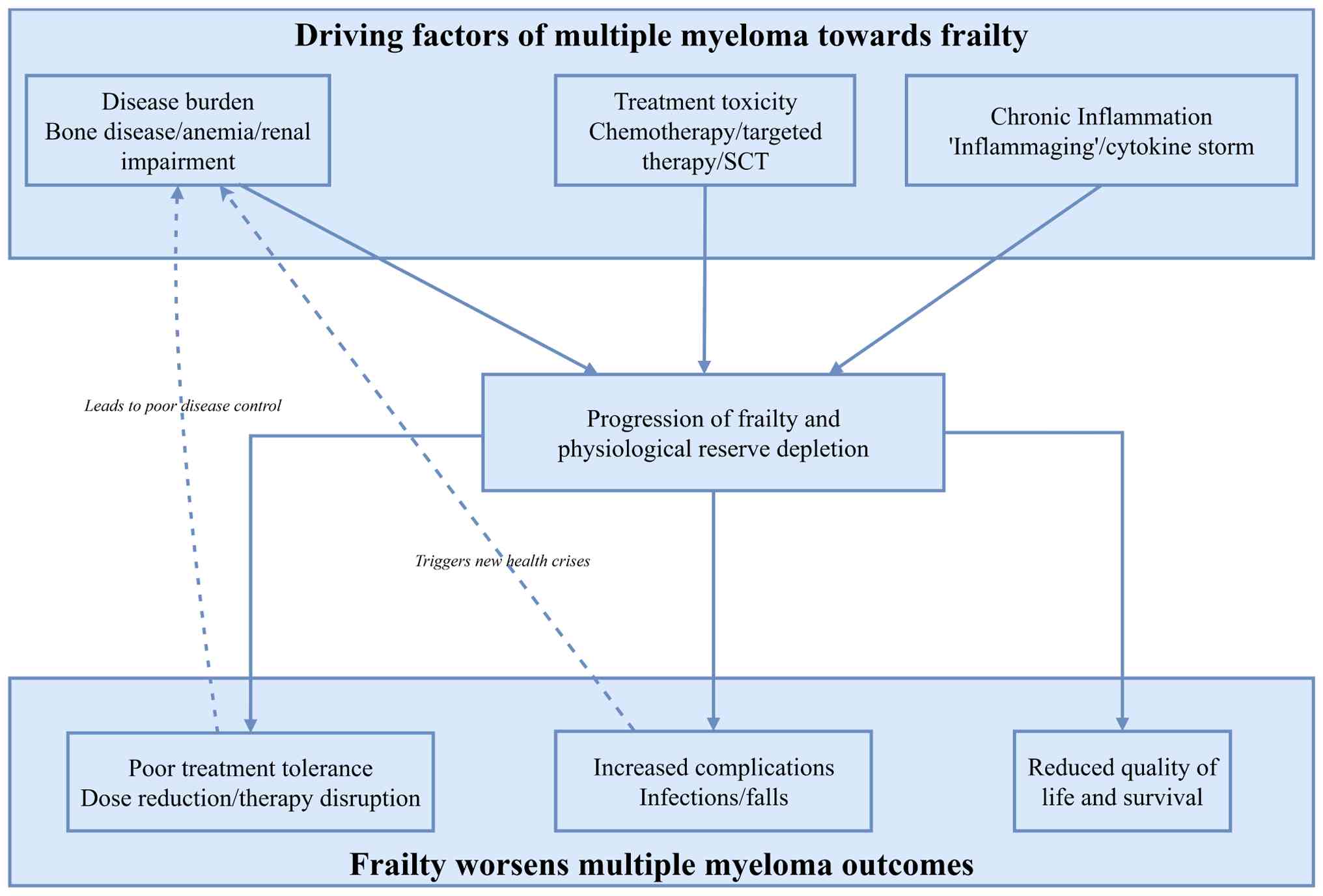
influence each other (Fig. 1). The
disease and its treatment reactions can cause frailty, which in
turn can worsen disease severity (54). MM can often lead to other conditions
such as anemia, osteolytic lesions and renal issues (55); in turn, anemia can lead to fatigue
and dizziness, whereas osteolytic lesions contribute to pain and
fractures, increasing activity avoidance and muscle loss (56). Renal failure may exacerbate anemia,
bone damage and metabolic problems, further increasing frailty
(24). A balanced diet, regular
exercise, health checkups, chronic disease management, appropriate
medication and attention to mental health can help reduce frailty
risk.
Elderly patients with MM display wide variability in
physiological and functional status, necessitating individualized
treatment. Transplant-eligible patients should receive intensive
therapies, whereas frail patients require regimens with adjusted
doses or schedules (57). MM may
impair physical function due to disease complications, although
effective initial therapy can reverse these effects, and additional
interventions may be introduced as needed (34,58).
The Comprehensive Geriatric Assessment (CGA) is the
most sensitive tool used to identify frailty (59); however, clinical practice is limited
by lack of time and resources (60). The CGA is time-intensive, complex
and requires a multidisciplinary team (a group of healthcare
professionals such as doctors, nurses, social workers and
therapists working together). Recognizing that MM has unique
characteristics and the need for a special frailty assessment model
has led to the development of the MM frailty model (60,61).
To identify frail patients more accurately, the
International Myeloma Working Group (IMWG) established the IMWG
Frailty Score (IMWG-FS) in 2015, based on three prospective
international multicenter clinical studies (60). This system assigns patients to three
groups, including fit (score, 0), intermediate-fit (score, 1) and
frail (score, ≥2), based on a composite score derived from age,
activities of daily living (ADL), instrumental ADL (IADL) and the
Charlson Comorbidity Index (CCI) (62). This scoring system can predict the
overall survival (OS) and PFS of patients with MM, and can predict
the incidence of adverse events (AEs) and drug discontinuation
(62). Multiple studies (36,63–67)
have confirmed that patients assessed as frail using the IMWG-FS
exhibit shorter OS, higher rates of severe AEs and increased risk
of treatment discontinuation. Given differing priorities in frailty
assessment across clinical contexts (such as emphasis on
comorbidities or need for rapid evaluation), a single assessment
tool cannot meet all requirements. Consequently, multiple
alternative frailty assessment systems have been developed and
applied clinically. Beyond the IMWG-FS, the Revised MM Comorbidity
Index (R-MCI) (68,69), the UK Myeloma Research Alliance Risk
Profile (MRP) (70), the Mayo
Clinic fraity index (Mayo-FI) (71), the Freiburger Comorbidity Index
(72) and the Geriatric Assessment
in Hematology (73) are also
commonly used frailty assessment systems in clinical practice.
These systems incorporate combinations of age, comorbidities,
physical performance status and laboratory parameters.
Additionally, the Timed Up and Go test and Mini Nutritional
Assessment-Short Form (TM) Frailty Score (74–76),
Carolina FI (77), Electronic
Frailty Score (36) and cumulative
deficit FI (35,78) are also commonly used frailty scoring
systems for elderly patients.
The Chinese TM Frailty Score system categorizes
elderly patients with MM into three groups, including good, fair
and frail, using the Timed Up and Go (TUG) test and the Mini
Nutritional Assessment Short Form (MNA-SF), demonstrating good
assessment efficacy (79). The
model was developed and internally validated in 167 consecutive
Chinese patients with MM (June 2019-September 2021; First
Affiliated Hospital of Sun Yat-sen University, Guangzhou, China),
with 135 completing an eight-domain CGA for core analysis. The
cohort (median age, 68 years; range, 60–85 years) aligns with the
target elderly MM population and key predictive metrics demonstrate
its robustness, including the TM score (combining TUG and MNA-SF),
which achieved C-indices of 0.741 (grade ≥3 AEs; P<0.001), 0.690
(treatment discontinuation, P<0.001) and 0.702 (OS; P=0.001),
outperforming the IMWG Geriatric Assessment (IMWG GA) (0.662, 0.636
and 0.631) and IMWG GA Plus (0.701, 0.656 and 0.618). Kaplan-Meier
analysis confirmed significant stratification of grade ≥3 AE and OS
risks between fit and frail groups (P<0.05). Regarding
consistency with the IMWG GA, the TM score enhanced risk
discrimination within the IMWG GA intermediate fit group,
identifying subgroups with higher grade ≥3 AE risk (P<0.05),
while retaining consistent prognostic alignment for frail patients
(poorer outcomes vs. fit groups, P<0.05). Although external
validation was not included in the initial abstract due to length
constraints, internal validation in a real-world Chinese MM cohort
provides solid preliminary evidence (79). A multicenter external validation
study is ongoing to further confirm its generalizability,
strengthening the credibility of the TM score as a locally tailored
tool for Chinese patients with MM (79).
Given that the parameters included in each
assessment system are not entirely consistent, the expected
survival of identified frail patients varies. To explore the
consistency among frailty assessment systems, Li et al
(80,81) retrospectively analyzed the clinical
data of 84 patients aged ≥60 years with NDMM. The authors
systematically evaluated the patients using the IMWG-FS, Mayo-FI,
Intergroupe Francophone du Myélome (IFM) simplified score and MRP
score, and compared the PFS and OS of the patients based on
different assessment tools. The results showed low consistency
among the four frailty assessment tools, with 64 patients (76.2%)
defined as frail by at least one tool. Among these, 48 patients
(75.0%) were identified as frail by at least two tools, whereas
only 14 patients (21.9%) were classified as frail by all four
tools. Compared with the Mayo-FI and IFM simplified score, the MRP
score and IMWG-FS demonstrated higher consistency and superior
prognostic stratification efficacy. The IMWG-FS assessed a median
OS of 15.5 months (P=0.01) vs. not reached for the non-frail and
frail groups, respectively, with a median PFS of 42.5 vs. 10.0
months (P=0.011). The MRP score showed a median OS of not reached
and 15.0 months (P<0.001) for the non-frail and frail groups,
respectively, with a median PFS of 42.5 and 9.0 months
(P<0.001). Therefore, combining MRP scoring with IMWG-FS may
effectively enhance the identification of frail elderly patients
with NDMM (82,83). For elderly patients with MM, dynamic
frailty assessment holds greater prognostic value than static
frailty assessment, necessitating treatment adjustments based on
such evaluations (33,63).
With the advent of novel therapies for MM, current
frailty assessment systems are increasingly limited in their
ability to address the complexities of patient care (60,70,71,88).
There is a need to develop more comprehensive frailty assessment
tools to support personalized treatment strategies for patients
with MM.
Due to multiple comorbidities and poor chemotherapy
tolerance, elderly patients with MM are generally considered
unsuitable recipients for transplantation if aged ≥75 years
(70). Treatment selection requires
consideration of multiple factors, including disease-specific
factors such as disease stage and cytogenetic abnormalities, and
patient-specific factors including age, comorbidities, organ
function status and frailty status (97). Initial treatment regimens should be
selected based on shared decision-making between physicians and
patients.
Additionally, initial treatment dosages for elderly
patients should be individualized (98). For example, elderly patients or
those with multiple comorbidities should receive low-dose anti-MM
agents as initial therapy (97).
For patients aged ≥75 years, the starting dose of dexamethasone is
20 mg weekly. For frail patients, a further dose reduction (8–20 mg
weekly) may be considered, with subsequent adjustments based on
response and treatment tolerance. In addition (98), renal impairment is common in the
elderly, necessitating dose adjustments for lenalidomide (99).
Research indicates that frailty is highly associated
with mortality risk, serving as an independent predictor of
all-cause mortality in patients with MM. The severity of frailty is
directly associated with increased mortality risk (102,103). In terms of accuracy and
sensitivity for assessing mortality risk, frailty assessment tools
demonstrate notable superiority over measures evaluating cognition,
function or comorbidities (104).
Frailty assessment tools can predict 3-year OS rates for patients
with MM with varying degrees of frailty. Using the R-MCI, the
3-year survival rates for patients without frailty, and those in
pre-frailty and frailty states were 91, 77 and 47%, respectively,
indicating the notable discriminatory power of the scale (105). It can also provide evidence for
establishing novel clinical or biological prognostic factors
(106). Similarly, the IMWG-FS can
effectively predict survival outcomes in patients with MM with
varying frailty levels. The 3-year survival rates were 84% for
non-frail patients, 76% for pre-frail patients and 57% for frail
patients (60). Thus, frailty
serves as a sensitive predictor of survival in patients with
MM.
In drug therapy for patients with MM, frail patients
experience greater drug-related toxicity during treatment, higher
discontinuation rates and poorer efficacy (63,107).
Using frailty as a screening indicator to predict drug toxicity
reactions, and as a basis for adjusting medication choices or
dosages can maximize patient benefits regarding both efficacy and
quality of life (99).
For patients ineligible for transplantation, the
focus should be on achieving deeper remission, prolonging OS and
PFS, and improving quality of life (116,117). In a frail state, elderly patients
with MM experience reduced muscle strength and immunity, leading to
increased infections and other complications (such as organ damage,
renal impairment and anemia), longer hospital stays and higher
costs (118). This not only
impacts hospital bed turnover rates and constrains the full
utilization of public health resources, but also diminishes social
and economic benefits (118).
Therefore, frailty assessment in elderly patients with MM should
guide the balancing of efficacy and toxicity to deliver appropriate
medical care, maximizing therapeutic benefits while minimizing
treatment risks to improve patient outcomes (119). Concurrently, strengthening family
and social support, implementing standardized management for
outpatients, ensuring timely healthcare responses and fostering
clinician-patient collaboration are essential to optimize resource
utilization efficiency and equity.
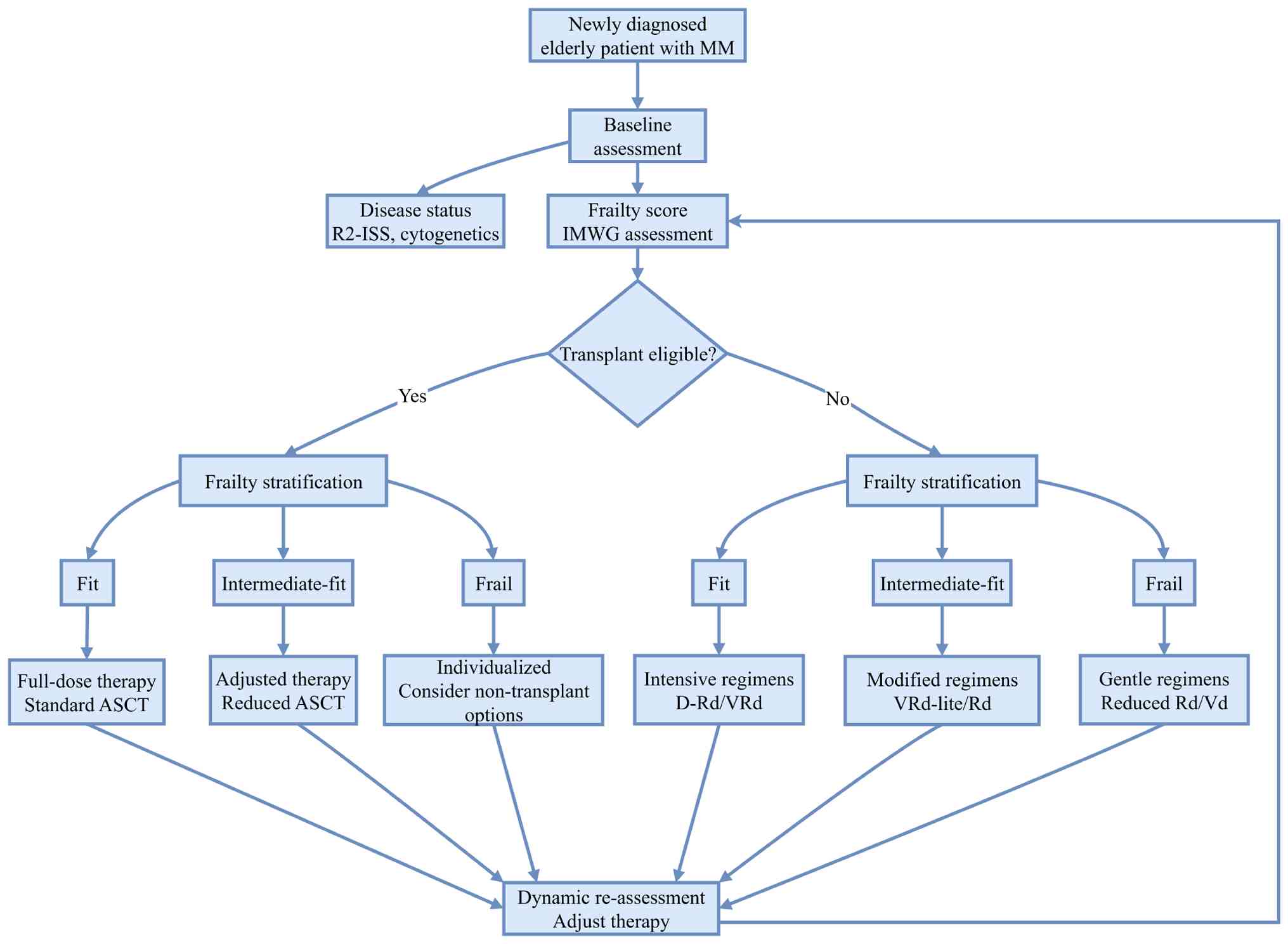
The specific treatment plan is recommended as
follows: For induction therapy in patients with good performance
status, triple or even quadruple combination regimens are
recommended, including daratumumab-lenalidomide-dexamethasone
(107), VRd (120), VCd and daratumumab-VMP (121). For patients with moderate
performance status, treatment should balance efficacy and safety,
aiming for a deep response while maintaining good safety (122). Dose-reduced triple or dual
regimens are recommended (123),
including: Dose-reduced VRd-Lite (124) VMP/VCd (once weekly) (125), induction-lenalidomide maintenance
(126), bortezomib-dexamethasone
(Vd) and lenalidomide-dexamethasone (Rd) (101,123). For frail patients, priority should
be given to enhancing independence and improving
quality-of-life-related aspects. The primary goal is to alleviate
symptoms as much as possible without worsening the disease and to
prolong survival. Reduced-dose, low-toxicity two-drug combinations
(Rd and Vd) are recommended (Fig.
2; Table III) (98,105,107,120,121,125,127,128).
In terms of the efficacy of novel treatment
regimens, multiple studies have confirmed that various therapies
exhibit clinical value in frail patients with RRMM. Real-world data
on B cell maturation antigen (BCMA)-directed chimeric antigen
receptor T cell (CAR-T) therapy showed that 61% of frail patients
achieved a median PFS of 6.9 months and a median OS of 14 months
after treatment. Although these outcomes are inferior to those in
non-frail patients, no excessively severe toxicities were detected,
indicating a controllable risk-benefit ratio (133,134). Among chemo-targeted combinations,
the pomalidomide, bortezomib and dexamethasone (PVd) regimen has
been shown to achieve an objective response rate (ORR) of 79.6% and
a median PFS of 9.7 months in frail patients, which were
significantly superior compared with those in patients treated with
the traditional Vd regimen (135).
Furthermore, dose adjustment of bortezomib can further prolong
treatment duration and PFS (135).
Post-marketing surveillance data from Japan show that the
isatuximab plus pomalidomide and dexamethasone (Isa-Pd) regimen
achieved an ORR of 38.5% in frail patients, with a treatment
discontinuation rate due to disease progression similar to that of
fit/intermediate-fit patients, confirming its real-world
effectiveness (136).
In terms of toxicity management, the toxicity
profiles of different regimens provide clear directions for
individualized interventions in frail patients. For CAR-T therapy,
the incidence of immune effector cell-associated neurotoxicity
syndrome (ICANS) in frail patients (39%) was higher than that in
non-frail patients (17%), but there was no difference in the
incidence of all-grade or high-grade cytokine release syndrome
(CRS) between the two groups. This suggests that close monitoring
of ICANS is crucial rather than excluding the treatment (133). Although the PVd regimen increased
the incidence of grade ≥3 treatment-emergent AEs in frail patients,
with a treatment discontinuation rate (30.1%) higher than that in
non-frail patients (19.2%), ~70% of patients could complete the
treatment without clustering of fatal toxicities, and dose
adjustment is a key safety guarantee (135). In response to the Isa-Pd regimen,
frail patients have been reported to exhibit higher incidences of
myelosuppression and infectious diseases, but no new serious safety
signals were observed (136). By
contrast, the bispecific antibodies regimen has a milder toxicity
profile, and no notable differences in CRS, ICANS or
treatment-related mortality have been reported between frail and
non-frail patients, thus indicating that it is a safer option for
frail patients (134).
In summary, tools such as the IMWG-FS and PRFP
provide reliable support for frailty assessment in patients with
RRMM. Novel regimens including BCMA-directed immunotherapies, PVd
and Isa-Pd have demonstrated acceptable safety and reasonable
efficacy in frail populations.
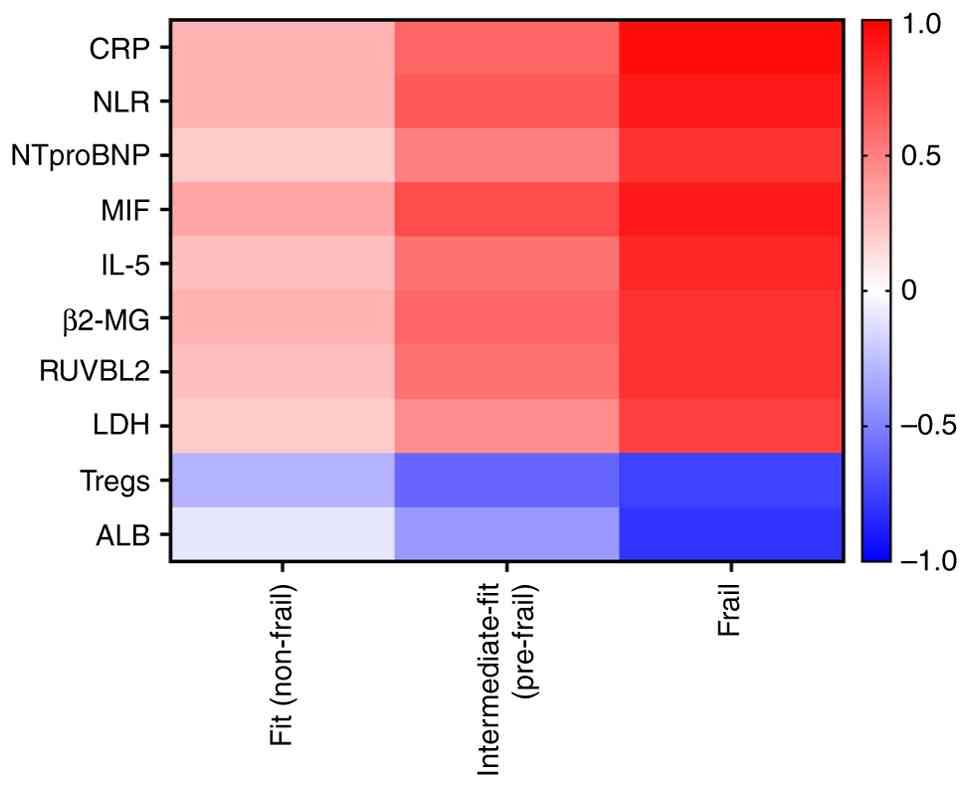
Additionally, studies have indicated that the NLR
combines inflammation with cell cycle alterations, and is thus
associated with frailty and OS in patients with MM (142,143). As a readily obtainable clinical
laboratory biomarker, it serves as a reference indicator for
identifying frailty in patients with MM. NLR links frailty to two
foundational aging-related processes: i) Neutrophilia (marking
chronic inflammation, which drives tissue dysfunction and reduced
stress resilience) (142); and ii)
lymphopenia (indicating immunosenescence and impaired
infection/tumor defense) (142).
In elderly patients with MM, high NLR (top quartile) is associated
with frailty-related phenotypes (such as ECOG-PS ≥2 and renal
dysfunction) and predicts poor survival (142). By integrating inflammation and
immunosenescence, intertwined pathways that exacerbate frailty, NLR
serves as a simple, readily available biomarker that complements
clinical frailty assessments, validating its use in identifying
high-risk patients (142).
Chronic inflammatory responses represent
characteristic alterations in organ aging (144,145). CRP is an inflammatory marker, and
elevated levels of CRP have been reported to be associated with
multiple age-related diseases, such as atherosclerosis, diabetes
and sarcopenia, and to predict mortality risk (146–151). Studies have found that CRP can
activate myeloma cells in the body, thereby promoting
osteoclastogenesis and bone destruction, and suggest that CRP may
serve as a therapeutic target for the prevention or treatment of
myeloma-related bone diseases (152,153). Based on CRP, age, World Health
Organization-PS and ISS staging, the UK Myeloma Research Consortium
established a risk prediction model (MRP) in 2019 (70) capable of forecasting PFS and early
mortality (EM).
While R2-ISS staging, which reflects MM biology,
does not predict EM, frailty and organ function markers such as
ECOG-PS, estimated glomerular filtration rate and NT-pro-BNP do
predict EM risk, with NT-pro-BNP potentially being the most
important independent factor (154). Therefore, incorporating these
frailty biomarkers into the R2-ISS staging system holds promise for
more precise prognostic stratification and EM prediction in elderly
patients with MM. Frailty serves as a risk predictor in patients
with MM. Studies have demonstrated that frailty is closely
associated with mortality risk, acting as an independent factor for
all-cause mortality in patients with MM, with increased frailty
severity associated with higher mortality risk (155,156).
Notably, research on frailty-related biomarkers in
MM has continued to expand. Beyond traditional indicators, a
variety of emerging biomarkers have become the focus of research
due to their clear mechanistic associations and clinical value,
providing new dimensions for disease assessment.
IL-5 is a key cytokine for distinguishing
engraftment syndrome (ES) after autologous hematopoietic stem cell
transplantation (ASCT). ES occurs in 24.0% of patients with NDMM
post-ASCT. The IL-5 level on day 6, combined with the proportion of
CD8+ T cells and daratumumab treatment history, can
accurately predict ES risk in 70.8% of patients (157). Its mechanism is related to
IL-5-mediated immune-inflammatory imbalance, which is a core
pathological basis for frailty (157).
A reduced proportion of Tregs at diagnosis is an
important predictor of early relapse (within 18 months) and
shortened PFS in patients with MM, and synergizes with adverse
factors such as extramedullary disease to predispose patients to a
functionally high-risk state (158). This may be due to the insufficient
regulatory capacity caused by reduced Treg proportions, which fails
to effectively inhibit abnormal inflammatory responses, thereby
exacerbating systemic metabolic disorders and frailty (158).
MM-related specific metabolites identified by
high-throughput analysis can reflect frailty-related abnormalities
in energy metabolism and inflammatory intensity, enabling
non-invasive assessment of the systemic reserve function of
patients (159). Combined with
advances in blood testing technology, their integration with
monoclonal protein structure analysis can improve the accuracy of
frailty risk stratification (160).
Although the IMWG-FS categorizes patients with MM
into fit, intermediate-fit and frail groups and predicts OS, PFS,
treatment discontinuation and non-hematologic toxicity, patient
outcomes exhibit significant heterogeneity even within the same
frailty subgroup (65).
Frailty is a dynamic state, however most existing
studies lack detailed dynamic frailty scoring for specific
subgroups, overlooking the impact of dynamic frailty assessment on
clinical outcomes. Smits et al (38) conducted a prospective study on the
dynamic changes of frailty and its impact on clinical outcomes in
the HOVON 143 study, which involved patients with NDMM (NTE-NDMM)
who were assessed as intermediate-fit and frail and not eligible
for transplantation by the IMWG-FS. The HOVON 123 study confirmed
that improvements in frailty scores are associated with longer OS,
longer PFS and reduced 100-day mortality. During treatment, nearly
half of frail patients showed improvement in IMWG-FSs, and
integrating dynamic frailty assessment significantly enhances
survival prediction. In most clinical settings, treatment
modifications should not be based solely on a single frailty
assessment. Relying solely on one assessment risks inadequate or
excessive treatment, particularly due to underestimating frailty
dynamics.
Early detection of frailty and timely intervention
are key to slowing its progression (77). The International Conference on
Frailty and Sarcopenia Research working group recommends a
patient-centered, multidisciplinary team approach for CGA and
management (169). In China, due
to differences in medical and social environments, the primary
approach is a multidisciplinary collaboration model. Teams
comprising healthcare professionals from oncology, geriatrics,
cardiovascular medicine, nutrition and rehabilitation jointly
develop treatment plans to ensure medical safety and maximize
patient benefit (170). Regular
dynamic assessments of frailty are conducted during this process to
promptly adjust treatment regimens, reduce medication-related
complications, improve patient compliance and reduce financial
burden (170). Interventions and
management strategies for frailty in elderly patients with MM
remain in the exploratory phase domestically, with specific
operational workflows, referral protocols and follow-up procedures
requiring further investigation.
Dynamic frailty assessment during induction therapy
requires frequent monitoring to capture rapid changes induced by
treatment toxicity or disease response (33,38,171).
The DynaFiT study (63) implemented
IMWG-FS assessment at the start of each 21-day induction cycle
(eight cycles total), and demonstrated that cycle-specific
adjustments to treatment intensity improved outcomes. Specifically,
58% of frail patients achieved frailty category improvement, with
27% becoming fit. Complementary evidence has been provided by the
HOVON 143 trial (38), which
performed assessments after three and nine cycles of induction,
with 78% of frail patients who improved showing changes within the
first three cycles, highlighting early monitoring as critical for
timely intervention. The SEER-Medicare cohort study (33) further supported that the first year
post-diagnosis (encompassing induction) is a high-risk period for
frailty fluctuation, with 93% of patients experiencing changes,
justifying cycle-level surveillance.
Maintenance therapy demands sustained but pragmatic
monitoring, as frailty changes are less acute but clinically
impactful (86). Zhang et al
(63) recommend frailty
reassessment at maintenance initiation (post-induction) to guide Rd
maintenance dosing, with 40% of patients transitioning to
maintenance showing frailty trajectory shifts that informed
personalized intensity. The Hemo-IMWG GA model validation (89) supported 3–6-month intervals during
maintenance, as HS and IMWG-FS integration identified late
hematological toxicity risks that static assessments missed.
Additionally, the GETH-TC multicenter study (86) emphasized a day +100 post-autologous
HCT assessment (a key maintenance transition point) to optimize
long-term care, as frailty at this timepoint strongly predicted
quality of life and readmission rates.
A total of three non-negotiable cross-phase nodes
ensure continuity: i) Baseline (pre-treatment); ii)
induction-maintenance transition; and iii) disease
relapse/progression. Baseline assessment using Hematopoietic Cell
Transplantation Frailty Scale (86)
establishes a reference for longitudinal comparison. The
induction-maintenance transition assessment (63) integrates frailty changes with MRD
status to avoid undertreatment/overtreatment. At relapse,
reassessment via cumulative deficit FI (77) identifies frailty deterioration
(observed in 30% of patients at 12 months) that guides salvage
therapy adjustments. For implementation, Smits et al
(38) noted that assessments take
<10 min using electronic health record-integrated tools, and
Chen et al (89) recommended
prioritizing higher-risk groups (frail, ≥80 years) for shortened
2-month intervals during early maintenance. Recommended assessment
frequency during treatment is shown in Table IV (33,38,60,63,78,86,89,171–174). Frailty in MM is a dynamic
condition that fluctuates with treatment response and disease
status, requiring systematic reassessment at diagnosis, during
therapy, upon relapse and post-treatment to inform timely
adjustments in treatment intensity (33,38,60,63,78,86,89,171–174).
MM primarily affects elderly individuals, among
whom frailty is highly prevalent due to physiological decline,
disease progression and medication effects. Frailty is a key
predictive indicator in MM, closely associated with survival
prognosis, mortality risk, transplant risk, hospitalization
duration and AE incidence. During drug therapy, frail patients with
MM experience increased drug toxicity, higher discontinuation rates
and poorer treatment outcomes. Employing frailty as a screening
tool to predict drug toxicity, and guide medication selection or
dosage adjustments can optimize both efficacy and quality of life.
Future research should focus on refining frailty assessment tools
for MM by integrating disease-specific molecular markers, such as
BCMA expression, and imaging metrics, such as muscle mass measured
by CT scans. Incorporating these parameters into treatment
decisions and AE evaluations may improve the accuracy of frailty
identification and risk prediction.
Elderly patients with MM have not demonstrated
substantial benefit from novel therapeutic agents, highlighting the
need for robust risk assessment models. Frailty scores facilitate
the identification of patients at a higher risk of treatment
toxicity and reduced survival, thereby supporting the selection of
appropriate therapeutic regimens. In certain cases, prioritizing
supportive care and psychological counseling over curative therapy
may still result in prolonged survival. Although various frailty
assessment tools are available, there is no consensus on
standardized measurement instruments. Future approaches should
incorporate dynamic frailty scoring methods, such as gait speed and
grip strength, to enhance treatment precision. Additionally,
assessment tools specifically designed for the characteristics of
the Chinese elderly population are needed.
The IMWG-FS remains the gold standard for frailty
assessment in MM. However, its subjective and time-intensive nature
limits widespread clinical adoption. Although alternative
assessment models have been introduced, they are not yet widely
implemented in practice. Frailty is inherently dynamic, yet
longitudinal research remains limited. Future research should
prioritize the development of time-series indicator systems to
monitor frailty dynamics. Recent prospective studies are expected
to provide further evidence-based guidance for the management of
elderly patients with MM. In summary, frailty scoring is essential
for treatment selection, prognosis assessment and safety management
in elderly patients with MM. Identifying additional prognostic
parameters, such as immune function and metabolic markers, will
support the creation of novel prognostic models. Standardizing
dynamic frailty assessment will facilitate the transition to
precision medicine in geriatric MM, ultimately improving quality of
life and survival outcomes.
Not applicable.
Funding: Not applicable.
Not applicable.
JLL and DYL were involved in conceptualization.
JLL, DYL, CL and ZRC performed the literature search and data
collection, and wrote the review. CL and ZRC reviewed and edited
the manuscript. Data authentication is not applicable. All authors
read and approved the final manuscript.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
|
1
|
Diaz-delCastillo M, Chantry AD, Lawson MA
and Heegaard AM: Multiple myeloma-A painful disease of the bone
marrow. Semin Cell Dev Biol. 112:49–58. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Michaud TL, Shih YH, Ji M, Huber J, Zhang
W, Wang M, Schoen MW, Thomas TS, Colditz GA, Li JS and Chang SH:
Differential life expectancies and life years lost associated with
multiple myeloma in the United States: A simulation modelling
study. Oncologist. Feb 6–2026.(Epub ahead of print). View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Dimopoulos MA, Moreau P, Terpos E, Mateos
MV, Zweegman S, Cook G, Hájek R, Schjesvold F, Cavo M, Goldschmidt
H, et al: Multiple myeloma: EHA-ESMO clinical practice guidelines
for diagnosis, treatment and follow-up(†). Ann Oncol. 32:309–322.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wang S, Xu L, Feng J, Liu Y, Liu L, Wang
J, Liu J, Huang X, Gao P, Lu J and Zhan S: Prevalence and incidence
of multiple myeloma in Urban area in China: A national
population-based analysis. Front Oncol. 9:15132020. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Rodriguez-Otero P, Paiva B and San-Miguel
JF: Roadmap to cure multiple myeloma. Cancer Treat Rev.
100:1022842021. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Cowan AJ, Green DJ, Kwok M, Lee S, Coffey
DG, Holmberg LA, Tuazon S, Gopal AK and Libby EN: Diagnosis and
management of multiple myeloma: A review. JAMA. 327:464–477. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kauer J, Weinhold N and Raab MS: Acquired
and selected: tracking antigen escape during T cell-redirecting
therapies in multiple myeloma. Blood Cancer Discov. 6:532–534.
2025. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sudha P, Pham P, Niu W, Liu E, Wang L,
Truong G, Ligocki C, Al-Azzawi R, Surapenini M, Bray SM, et al:
Diagnostic sequencing identifies high-risk markers and mechanisms
of resistance to guide immunotherapy selection. Blood Adv.
10:95–105. 2026. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Manier S, Dimopoulos MA, Leleu XP, Moreau
P, Cavo M, Goldschmidt H, Orlowski RZ, Tron M, Tekle C, Brégeault
MF, et al: Isatuximab plus bortezomib, lenalidomide, and
dexamethasone for transplant-ineligible newly diagnosed multiple
myeloma patients: A frailty subgroup analysis of the IMROZ trial.
Haematologica. 110:2139–2150. 2025.PubMed/NCBI
|
|
10
|
Manier S, Lambert J, Hulin C, Macro M,
Laribi K, Araujo C, Pica GM, Touzeau C, Godmer P, Slama B, et al:
Safety and efficacy of a dexamethasone-sparing regimen with
daratumumab and lenalidomide in patients with frailty and newly
diagnosed multiple myeloma (IFM2017-03): A phase 3, open-label,
multicentre, randomised, controlled trial. Lancet Oncol.
26:1323–1333. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Mateos MV, Paiva B, Cedena MT, Puig N,
Sureda-Balari AM, de la Calle VG, Oriol A, Ocio EM, Rosiñol L,
Montes YG, et al: Induction therapy with bortezomib, melphalan, and
prednisone followed by lenalidomide and dexamethasone versus
carfilzomib, lenalidomide, and dexamethasone with or without
daratumumab in older, fit patients with newly diagnosed multiple
myeloma (GEM-2017FIT): A phase 3, open-label, multicentre,
randomised clinical trial. Lancet Haematol. 12:e588–e598. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Tu C, Van der Vreken A, Faict S, Ates G,
Massie A, De Veirman K, De Bruyne E, Vanderkerken K and Menu E: The
clinically available supplement pyruvate enhances the therapeutic
effect of bortezomib in Multiple Myeloma by modulating
mitochondrial metabolism. Cancer Lett. 640:2182452026. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hu S, Xu J, Cui W, Jin H, Wang X and
Maimaitiyiming Y: Post-translational modifications in multiple
myeloma: Mechanisms of drug resistance and therapeutic
opportunities. Biomolecules. 15:7022025. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chen B, Liu J, Zhang Y, Shi C, Zhu D,
Zhang G, Xiao F, Zhong L, Zhang M, Ng LG, et al: Enhancer
extrachromosomal circular DNA ANKRD28 elicits drug resistance via
POU2F2-Mediated transcriptional network in multiple myeloma. Adv
Sci (Weinh). 12:e24156952025. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
D'Agostino M, Cairns DA, Lahuerta JJ,
Wester R, Bertsch U, Waage A, Zamagni E, Mateos MV, Dall'Olio D,
van de Donk NWCJ, et al: Second revision of the International
staging system (R2-ISS) for overall survival in multiple myeloma: A
European myeloma network (EMN) report within the HARMONY project. J
Clin Oncol. 40:3406–3418. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Abdallah NH, Binder M, Rajkumar SV, Greipp
PT, Kapoor P, Dispenzieri A, Gertz MA, Baughn LB, Lacy MQ, Hayman
SR, et al: A simple additive staging system for newly diagnosed
multiple myeloma. Blood Cancer J. 12:212022. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Brieghel C, Slørdahl TS, Andersen MN,
Szabo AG, Niemann CU and Thorsteinsdóttir S: A Real-World
International Staging System (RW-ISS) for patients with newly
diagnosed multiple myeloma. Blood Cancer J. 15:592025. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Pawlyn C, Cairns D, Kaiser M, Striha A,
Jones J, Shah V, Jenner M, Drayson M, Owen R, Gregory W, et al: The
relative importance of factors predicting outcome for myeloma
patients at different ages: results from 3894 patients in the
Myeloma XI trial. Leukemia. 34:604–612. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Bonello F, Boccadoro M and Larocca A:
Diagnostic and therapeutic challenges in the management of
intermediate and frail elderly multiple myeloma patients. Cancers
(Basel). 12:31062020. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Stege CAM, van der Holt B, Dinmohamed AG,
Sonneveld P, Levin MD, van de Donk NWCJ, Mellqvist UH, Waage A and
Zweegman S: Validation of the FIRST simplified frailty scale using
the ECOG performance status instead of patient-reported activities.
Leukemia. 34:1964–1966. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Nathwani N, Kurtin SE, Lipe B, Mohile SG,
Catamero DD, Wujcik D, Birchard K, Davis A, Dudley W, Stricker CT
and Wildes TM: Integrating touchscreen-based geriatric assessment
and frailty screening for adults with multiple myeloma to drive
personalized treatment decisions. JCO Oncol Pract. 16:e92–e99.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Sim S, Kalff A, Tuch G, Mollee P, Ho PJ,
Harrison S, Gibbs S, Prince HM, Spencer A, Joshua D, et al: The
importance of frailty assessment in multiple myeloma: A position
statement from the myeloma scientific advisory group to myeloma
Australia. Intern Med J. 53:819–824. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zamagni E, Barbato S and Cavo M: How I
treat high-risk multiple myeloma. Blood. 139:2889–2903. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Möller MD, Gengenbach L, Graziani G, Greil
C, Wäsch R and Engelhardt M: Geriatric assessments and frailty
scores in multiple myeloma patients: A needed tool for
individualized treatment? Curr Opin Oncol. 33:648–657. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kylies J, Brauneck E, Priemel M, Kylies D,
Weisel K, Leonhardt LG and Viezens L: Sarcopenia and fat loss from
serial CT predict survival in multiple myeloma patients undergoing
stem cell transplantation. World J Surg Oncol. 23:3362025.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Sobrini-Morillo P, Corral-Tuesta C,
Sánchez-Castellano C, Gutiérrez-Blanco T, Blanchard-Rodríguez MJ,
Escudero-González B, Nieto-Góngora C, Moujir López J, Serra-Rexach
JA and Cruz-Jentoft AJ: Sarcopenia in older patients with newly
diagnosed multiple myeloma. Gerontology. 72:19–28. 2026. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
João C, Geraldes C, Neves M, Mariz M and
Trigo F: Management of older and frail patients with multiple
myeloma in the Portuguese routine clinical practice: Deliberations
and recommendations from an expert panel of hematologists. J
Geriatr Oncol. 11:1210–1216. 2020. View Article : Google Scholar
|
|
28
|
Fried LP, Tangen CM, Walston J, Newman AB,
Hirsch C, Gottdiener J, Seeman T, Tracy R, Kop WJ, Burke G, et al:
Frailty in older adults: Evidence for a phenotype. J Gerontol A
Biol Sci Med Sci. 56:M146–M156. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Cook G, Larocca A, Facon T, Zweegman S and
Engelhardt M: Defining the vulnerable patient with myeloma-a
frailty position paper of the European Myeloma Network. Leukemia.
34:2285–2294. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Brioli A, Manz K, Pfirrmann M, Hänel M,
Schwarzer AC, Prange-Krex G, Fabisch C, Knop S, Illmer T,
Krammer-Steiner B, et al: Frailty impairs the feasibility of
induction therapy but not of maintenance therapy in elderly myeloma
patients: Final results of the German Maintenance Study (GERMAIN).
J Cancer Res Clin Oncol. 146:749–759. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Bao Y, Ren J, Chen L, Guo W, Feng K, Huang
T and Cai YD: Identification of gene signatures associated with
COVID-19 across children, adolescents, and adults in the
nasopharynx and peripheral blood by using a machine learning
approach. Curr Gene Ther. 25:703–717. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Lu J, Zhen S and Li X: Characteristics of
oxidative phosphorylation-related subtypes and construction of a
prognostic signature in ovarian cancer. Curr Gene Ther. 25:327–344.
2025. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Mian H, Wildes TM, Vij R, Pianko MJ, Major
A and Fiala MA: Dynamic frailty risk assessment among older adults
with multiple myeloma: A population-based cohort study. Blood
Cancer J. 13:762023. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Engelhardt M, Ihorst G, Duque-Afonso J,
Wedding U, Spät-Schwalbe E, Goede V, Kolb G, Stauder R and Wäsch R:
Structured assessment of frailty in multiple myeloma as a paradigm
of individualized treatment algorithms in cancer patients at
advanced age. Haematologica. 105:1183–1188. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Rockwood K and Mitnitski A: Frailty in
relation to the accumulation of deficits. J Gerontol A Biol Sci Med
Sci. 62:722–727. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Chan H, Chong YH, Seow MY, Li J, Garg P,
Kelly M, Neylon A, McDiarmid B, Tan S and Jackson S: Electronic
FRAIL score may predict treatment outcomes in older adults with
myeloma. J Geriatr Oncol. 12:515–520. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Dreyling E, Räder J, Möller MD, Ihorst G,
Wenger S, Pahl A, Arends J, Herget G, Deibert P, Wäsch R and
Engelhardt M: A Randomized Controlled ‘REAL-FITNESS’ trial to
evaluate physical activity in patients with newly diagnosed
multiple myeloma. J Cachexia Sarcopenia Muscle. 16:e137932025.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Smits F, Groen K, Levin MD, Stege CAM, van
Kampen R, van der Spek E, Bilgin YM, Thielen N, Nijhof I, Ludwig I,
et al: Beyond static measurements: Dynamic frailty improves
survival prediction in multiple myeloma. Blood. 145:543–546. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Deng Y, Zhang K, Zhu J, Hu X and Liao R:
Healthy aging, early screening, and interventions for frailty in
the elderly. Biosci Trends. 17:252–261. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Fell G, Rosko AE, Abel GA, Dumontier C,
Higby KJ, Murillo A, Neuberg DS, Burd CE and Lane AA: Peripheral
blood CD3(+) T-cell gene expression biomarkers correlate with
clinical frailty in patients with haematological malignancies. Br J
Haematol. 199:100–105. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Elbahoty MH, Papineni B and Samant RS:
Multiple myeloma: Clinical characteristics, current therapies and
emerging innovative treatments targeting ribosome biogenesis
dynamics. Clin Exp Metastasis. 41:829–842. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Urban VS, Cegledi A and Mikala G: Multiple
myeloma, a quintessential malignant disease of aging: A geroscience
perspective on pathogenesis and treatment. Geroscience. 45:727–746.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Ran T, Medhekar R, Fu AZ, Patel S and
Kaila S: Patient characteristics associated with dose modifications
for VRd among newly diagnosed multiple myeloma patients. Future
Oncol. 18:3983–3991. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Tang RQ, Zheng W, Li HL, Hu XO and Xiang
YB: Lifestyle-related factors and cancer incidence in men: A public
health assessment based on a ‘healthy lifestyle score’. Zhonghua
Liu Xing Bing Xue Za Zhi. 34:109–113. 2013.(In Chinese). PubMed/NCBI
|
|
45
|
Tamura Y, Omura T, Toyoshima K and Araki
A: Nutrition management in older adults with diabetes: A review on
the importance of shifting prevention strategies from metabolic
syndrome to frailty. Nutrients. 12:33672020. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Hayakawa M, Motokawa K, Mikami Y, Yamamoto
K, Shirobe M, Edahiro A, Iwasaki M, Ohara Y, Watanabe Y, Kawai H,
et al: Low dietary variety and diabetes mellitus are associated
with frailty among community-dwelling older Japanese adults: A
cross-sectional study. Nutrients. 13:6412021. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Wildevuur SE and Simonse LW: Information
and communication technology-enabled person-centered care for the
‘big five’ chronic conditions: scoping review. J Med Internet Res.
17:e772015. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Ma W, Wang H, Wen Z, Liu L and Zhang X:
Potentially inappropriate medication and frailty in older adults: A
systematic review and meta-analysis. Arch Gerontol Geriatr.
114:1050872023. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Ye B, Li Y, Bao Z and Gao J: Psychological
resilience and frailty progression in older adults. JAMA Netw Open.
7:e24476052024. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Lameirinhas J, Gorostiaga A and Etxeberria
I: Definition and assessment of psychological frailty in older
adults: A scoping review. Ageing Res Rev. 100:1024422024.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Hanlon P, Politis M, Wightman H,
Kirkpatrick S, Jones C, Khan M, Bezzina C, Mackinnon S, Rennison H,
Wei L, et al: Frailty and socioeconomic position: A systematic
review of observational studies. Ageing Res Rev. 100:1024202024.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Shah UA, Parikh R, Castro F, Bellone M and
Lesokhin AM: Dietary and microbiome evidence in multiple myeloma
and other plasma cell disorders. Leukemia. 37:964–980. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Calder PC: Nutrition and immunity: Lessons
from coronavirus disease-2019. Proc Nutr Soc. 84:8–23. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Murugappan MN, King-Kallimanis BL,
Bhatnagar V, Kanapuru B, Farley JF, Seifert RD, Stenehjem DD, Chen
TY, Horodniceanu EG and Kluetz PG: Measuring frailty using
patient-reported outcomes (PRO) Data: A feasibility study in
patients with multiple myeloma. Qual Life Res. 32:2281–2292. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Nielsen LK, Larsen RF, Jarlbaek L, Möller
S and Jespersen E: Health-related quality of life in patients with
multiple myeloma participating in a multidisciplinary
rehabilitation program. Ann Hematol. 100:2311–2323. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
56
|
O'Neill DE and Graham MM: Anemia,
cardiovascular disease, and frailty in the older adult. Can J
Cardiol. 38:715–717. 2022.PubMed/NCBI
|
|
57
|
Lipof JJ, Abdallah N and Lipe B:
Personalized treatment of multiple myeloma in frail patients. Curr
Oncol Rep. 26:744–753. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Oerlemans S, De Rooij BH, Bennink C,
Bullinger L, Broijl A, D'Agostino M, Laane E, Lupo-Stanghellini MT,
Perrot A, Wester R, et al: Update and European consensus on a
patient-centered core outcome set for multiple myeloma in clinical
practice and research. Haematologica. 109:1956–1959.
2024.PubMed/NCBI
|
|
59
|
Sobrini-Morillo P, Corral-Tuesta C,
Sánchez-Castellano C, Gutiérrez-Blanco T, Palomo-Rumschisky P,
Álvarez-Pinheiro CG, Blanchard-Rodríguez MJ, Serra-Rexach JA and
Cruz-Jentoft AJ: Comprehensive geriatric assessment of older
patients with multiple myeloma: A prospective observational study.
Cancers (Basel). 17:29042025. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Palumbo A, Bringhen S, Mateos MV, Larocca
A, Facon T, Kumar SK, Offidani M, McCarthy P, Evangelista A, Lonial
S, et al: Geriatric assessment predicts survival and toxicities in
elderly myeloma patients: An International myeloma working group
report. Blood. 125:2068–2074. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Yao Y, Sui WW, Liao AJ, Wang W, Chen LJ,
Chu XX, Bao L, Cen XN, Fu R, Liu H, et al: Comprehensive geriatric
assessment in newly diagnosed older myeloma patients: A
multicentre, prospective, non-interventional study. Age Ageing.
51:afab2112022. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Cook G, Pawlyn C, Royle KL, Senior ER,
Everritt D, Bird J, Bowcock S, Dawkins B, Drayson M, Gillson S, et
al: IMWG frailty score-adjusted therapy delivery reduces the early
mortality risk in newly diagnosed tne multiple myeloma: Results of
the UK myeloma research alliance (UK-MRA) Myeloma XIV fitness
trial. Blood. 144 (Suppl 1):6732024. View Article : Google Scholar
|
|
63
|
Zhang Y, Liang X, Xu W, Yi X, Hu R, Ma X,
Yan Y, Zhang N, Wang J, Sun X, et al: Individualized dynamic
frailty-tailored therapy (DynaFiT) in elderly patients with newly
diagnosed multiple myeloma: A prospective study. J Hematol Oncol.
17:482024. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Groen K, Stege CAM, Nasserinejad K, de
Heer K, van Kampen RJW, Leys RBL, Thielen N, Westerman M, Wu KL,
Ludwig I, et al: Ixazomib, daratumumab and low-dose dexamethasone
in intermediate-fit patients with newly diagnosed multiple myeloma:
an open-label phase 2 trial. EClinicalMedicine. 63:1021672023.
View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Bringhen S, Pour L, Benjamin R, Grosicki
S, Min CK, C de Farias DL, Vorog A, Labotka RJ, Wang B, Cherepanov
D, et al: Ixazomib versus placebo as postinduction maintenance
therapy in newly diagnosed multiple myeloma patients: An analysis
by age and frailty status of the TOURMALINE-MM4 study. Clin
Lymphoma Myeloma Leuk. 23:491–504. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Seefat MR, Stege CAM, Lissenberg-Witte BI,
Levin MD, Timmers GJ, Hoogendoorn M, Ypma PF, Klein SK, Velders GA,
Westerman M, et al: Quality of life gains in frail and
intermediate-fit patients with multiple Myeloma: Findings from the
prospective HOVON123 clinical trial. Eur J Cancer. 207:1141532024.
View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Efficace F, Gaidano G, Petrucci MT,
Niscola P, Cottone F, Codeluppi K, Antonioli E, Tafuri A, Larocca
A, Potenza L, et al: Association of IMWG frailty score with
health-related quality of life profile of patients with relapsed
refractory multiple myeloma in Italy and the UK: A GIMEMA,
multicentre, cross-sectional study. Lancet Healthy Longev.
3:e628–e635. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Candan O, Bayar A, Naghizada N, Demirtas
D, Yanik AM, Yilmaz AF, Tuglular AT, Toptas T and Atagunduz I:
Determinants of eligibility and timing of autologous
transplantation in multiple myeloma: The role of R-MCI and
diagnostic plasma cell Burden. Diagnostics (Basel). 15:30382025.
View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Holler M, Ihorst G, Reinhardt H, Rösner A,
Braun M, Möller MD, Dreyling E, Schoeller K, Scheubeck S, Wäsch R
and Engelhardt M: An objective assessment in newly diagnosed
multiple myeloma to avoid treatment complications and strengthen
therapy adherence. Haematologica. 108:1115–1126. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Cook G, Royle KL, Pawlyn C, Hockaday A,
Shah V, Kaiser MF, Brown SR, Gregory WM, Child JA, Davies FE, et
al: A clinical prediction model for outcome and therapy delivery in
transplant-ineligible patients with myeloma (UK Myeloma Research
Alliance Risk Profile): A development and validation study. Lancet
Haematol. 6:e154–e166. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Milani P, Vincent Rajkumar S, Merlini G,
Kumar S, Gertz MA, Palladini G, Lacy MQ, Buadi FK, Hayman SR, Leung
N, et al: N-terminal fragment of the type-B natriuretic peptide
(NT-proBNP) contributes to a simple new frailty score in patients
with newly diagnosed multiple myeloma. Am J Hematol. 91:1129–1134.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Kleber M, Ihorst G, Terhorst M, Koch B,
Deschler B, Wäsch R and Engelhardt M: Comorbidity as a prognostic
variable in multiple myeloma: comparative evaluation of common
comorbidity scores and use of a novel MM-comorbidity score. Blood
Cancer J. 1:e352011. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Bonanad S, De la Rubia J, Gironella M,
Pérez Persona E, González B, Fernández Lago C, Arnan M, Zudaire M,
Hernández Rivas JA, Soler A, et al: Development and psychometric
validation of a brief comprehensive health status assessment scale
in older patients with hematological malignancies: The GAH Scale. J
Geriatr Oncol. 6:353–361. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Chen Y, Gu J, Huang B, Liu J, Li X and Li
J: Development and validation of a chemotherapy tolerance
prediction model for Chinese multiple myeloma patients: The TM
frailty score. Front Oncol. 13:11036872023. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Wang DXM, Yao J, Zirek Y, Reijnierse EM
and Maier AB: Muscle mass, strength, and physical performance
predicting activities of daily living: A meta-analysis. J Cachexia
Sarcopenia Muscle. 11:3–25. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Kaiser MJ, Bauer JM, Ramsch C, Uter W,
Guigoz Y, Cederholm T, Thomas DR, Anthony P, Charlton KE, Maggio M,
et al: Validation of the Mini Nutritional Assessment short-form
(MNA-SF): A practical tool for identification of nutritional
status. J Nutr Health Aging. 13:782–788. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Guerard EJ, Deal AM, Chang Y, Williams GR,
Nyrop KA, Pergolotti M, Muss HB, Sanoff HK and Lund JL: Frailty
index developed from a cancer-specific geriatric assessment and the
association with mortality among older adults with cancer. J Natl
Compr Canc Netw. 15:894–902. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Abdallah N, Dizona P, Kumar A, LaPlant B,
Menser T, Schaeferle G, Aug S, Weivoda M, Dispenzieri A, Buadi FK,
et al: Cumulative deficits frailty index and relationship status
predict survival in multiple myeloma. Blood Adv. 9:1137–1146. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Chen Y, Gu J, Huang B, Liu J, Li X and Li
J: P896: Development and validation of a frailty prediction model
more suitable for multiple myeloma in Chinese patients: The TM
frailty score. Hemasphere. 6 (Suppl):S787–S788. 2022. View Article : Google Scholar
|
|
80
|
Li Y, Zhang J, Xu J, Guo H, Wang J, Zhao
S, Liu L, Xue H and Lou Y: Retrospective Analysis of Four Frailty
Assessment Tools in Elderly Patients with Multiple Myeloma. Blood.
142 (Suppl 1):19752023. View Article : Google Scholar
|
|
81
|
Li Y, Zhao S, Xu J, Zhang J, Wang J, Guo
H, Liu L and Xue H: Selection determines therapeutic effects: A
retrospective analysis of the application of different frailty
tools in elderly patients with multiple myeloma. Discov Oncol.
15:5462024. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Xu J, Xue H, Zhao S, Wang J, Guo H, Zhang
J, Liu J, Tian M, Hua L and Zhao M: Evaluating the effectiveness of
risk adjustment using dynamic frailty assessment in treating newly
diagnosed multiple myeloma in elderly patients. Blood. 142
(Sup1):S66442023. View Article : Google Scholar
|
|
83
|
Xu J, Xue H, Zhao S, Wang J, Guo H, Zhang
J, Liu J, Hua L, Tian M and Zhao M: Efficacy analysis of
ixazomib-based regimens based on dynamic frailty assessment in
elderly and frail patients with newly diagnosed multiple myeloma.
Blood. 142 (Sup1):S66902023. View Article : Google Scholar
|
|
84
|
Haider I, Leong DP, Shahid O, Louzada M,
McCurdy A, Pond GR, Cameron R, Balitsky AK, Britto J, Aljama M, et
al: Changes in frailty categorization over one year among
Real-World patients with multiple Myeloma: A Prospective Cohort
Study (MFRAIL). Clin Lymphoma Myeloma Leuk. 26:e164–e171.e1. 2026.
View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Smits F, Groen K, Levin MD, Stege CAM, van
Kampen R, van der Spek E, Nijhof I, Bilgin YM, Thielen N, Ludwig I,
et al: Frail subgroups determine heterogeneous outcomes in older
patients with NDMM: Long-term follow-up of the HOVON 143 trial.
Blood Adv. 9:5828–5836. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Tolosa-Ridao C, González MB, Gómez MA, Fox
L, Pérez Artigas MDM, Santamaría A, Quintela González MDC, Salinas
AS, Salmerón Camacho JM, Illana Álvaro V, et al: Frailty dynamics
and their impact on QoL in patients undergoing autologous HCT for
multiple myeloma: Results from a multicentre GETH-TC study. Br J
Haematol. 207:1504–1516. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Engelhardt M, Domm AS, Dold SM, Ihorst G,
Reinhardt H, Zober A, Hieke S, Baayen C, Müller SJ, Einsele H, et
al: A concise revised Myeloma comorbidity index as a valid
prognostic instrument in a large cohort of 801 multiple myeloma
patients. Haematologica. 102:910–921. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Ma K, Ye J, Wang L, Sun C and Zhou X:
Evaluation of the UK myeloma research alliance risk profile in
Chinese patients with newly diagnosed multiple myeloma without
autologous stem cell transplantation. Onco Targets Ther.
14:2349–2361. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Chen Y, He F, Chen Y, He Z, Wu D, An H,
Liu Q and Zheng Y: Combining IMWG GA and hematopoietic score to
optimize the evaluation of dynamic chemotherapy tolerance in
multiple myeloma. Ann Med. 57:25581272025. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Tian B, Xu L, Jia S, Sun W, Zhang C, Feng
J, Hui J, Li M, Xiao W, Wang L, et al: Development and validation
of a novel frailty model for the patients with newly diagnosed
multiple myeloma. Eur J Med Res. 31:892025. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Lee HS, Lee J, Jo JC, Jung SH, Lee JJ, Kim
D, Lee S and Song K: Development of a new clinical index to easily
assess frailty of elderly patients with multiple myeloma in Asian
population. Sci Rep. 11:229072021. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Palumbo A, Avet-Loiseau H, Oliva S,
Lokhorst HM, Goldschmidt H, Rosinol L, Richardson P, Caltagirone S,
Lahuerta JJ, Facon T, et al: Revised International staging system
for multiple myeloma: A report from International myeloma working
group. J Clin Oncol. 33:2863–2869. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Facon T, Dimopoulos MA, Meuleman N, Belch
A, Mohty M, Chen WM, Kim K, Zamagni E, Rodriguez-Otero P, Renwick
W, et al: A simplified frailty scale predicts outcomes in
transplant-ineligible patients with newly diagnosed multiple
myeloma treated in the FIRST (MM-020) trial. Leukemia. 34:224–233.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Lee JY, Kim SA, Koh Y, Yhim HY, Lee GW,
Min CK, Do YR, Kim HJ, Bae SH, Eom HS, et al: Validating the Korean
geriatric assessment tool in elderly multiple myeloma patients: A
multicenter study. Cancer Res Treat. 58:311–319. 2026. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Krepper D, Sparano F, Giesinger JM,
Gaidano G, Niscola P, Codeluppi K, Antonioli E, Califano C, Floro
L, Annibali O, et al: Assessing frailty in patients with
relapsed/refractory multiple myeloma: A comparison between the
patient-reported frailty phenotype and the International myeloma
working group frailty index. J Geriatr Oncol. 16:1022662025.
View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Muzyka M, Ottaviani S, Caffa I, Bonfiglio
T, Parisi E, Guijarro A, Tagliafico L, Lemoli RM, Ponzano M,
Marelli C, et al: Prognostic frailty-based determinants of
long-term mortality in older patients with newly diagnosed multiple
myeloma. Cancers (Basel). 17:7892025. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Tyczyńska A, Krzempek MK, Cortez AJ,
Jurczyszyn A, Godlewska K, Ciepłuch H, Subocz E, Hałka J,
Kulikowska de Nałęcz A, Wiśniewska A, et al: The real-world
evidence on the fragility and its impact on the choice of treatment
regimen in newly diagnosed patients with multiple myeloma over 75
years of age. Cancers (Basel). 15:34692023. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Abdallah N and Kumar SK: Up-front
treatment of elderly (Age ≥75 Years) and frail patients with
multiple myeloma. J Natl Compr Canc Netw. 22:e2470392024.
View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Salama E, Lam S, Gonsalves WI, Tzachanis
D, Momper JD and M Saunders I: Estimation of kidney function in
patients with multiple myeloma: Implications for lenalidomide
dosing. Ann Pharmacother. 57:29–35. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Kumar SK, Callander NS, Adekola K,
Anderson LD, Baljevic M, Baz R, Campagnaro E, Costello C, D'Angelo
C, Derman B, et al: NCCN Guidelines® Insights: multiple
myeloma, version 1.2025. J Natl Compr Canc Netw. 23:132–140. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Mina R, Bringhen S, Wildes TM, Zweegman S
and Rosko AE: Approach to the older adult with multiple myeloma. Am
Soc Clin Oncol Educ Book. 39:500–518. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
DuMontier C, La J, Bihn J, Corrigan J,
Yildirim C, Dharne M, Hassan H, Yellapragada S, Abel GA, Gaziano
JM, et al: More intensive therapy as more effective treatment for
frail patients with multiple myeloma [corrected]. Blood Adv.
7:6275–6284. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Aureli A, Marziani B, Sconocchia T,
Pasqualone G, Franceschini L, Spagnoli GC, Venditti A and
Sconocchia G: Challenges in multiple myeloma therapy in older and
frail patients. Cancers (Basel). 17:9442025. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Mian H, McCurdy A, Giri S, Grant S,
Rochwerg B, Winks E, Rosko AE, Engelhardt M, Pawlyn C, Cook G, et
al: The prevalence and outcomes of frail older adults in clinical
trials in multiple myeloma: A systematic review. Blood Cancer J.
13:62023. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Engelhardt M, Dold SM, Ihorst G, Zober A,
Möller M, Reinhardt H, Hieke S, Schumacher M and Wäsch R: Geriatric
assessment in multiple myeloma patients: Validation of the
International Myeloma Working Group (IMWG) score and comparison
with other common comorbidity scores. Haematologica. 101:1110–1119.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Dold SM, Möller MD, Ihorst G, Langer C,
Pönisch W, Mügge LO, Knop S, Jung J, Greil C, Wäsch R and
Engelhardt M: Validation of the revised myeloma comorbidity index
and other comorbidity scores in a multicenter German study group
multiple myeloma trial. Haematologica. 106:875–880. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Facon T, Cook G, Usmani SZ, Hulin C, Kumar
S, Plesner T, Touzeau C, Bahlis NJ, Basu S, Nahi H, et al:
Daratumumab plus lenalidomide and dexamethasone in
transplant-ineligible newly diagnosed multiple myeloma: Frailty
subgroup analysis of MAIA. Leukemia. 36:1066–1077. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Bruins WSC, Smits F, Duetz C, Nasserinejad
K, Groen K, Korst CLBM, de Jonge AV, Rentenaar R, Hageman T,
Cosovic M, et al: Immune signatures in older patients with newly
diagnosed multiple myeloma are associated with survival outcomes of
first-line therapy irrespective of frailty levels. Hemasphere.
9:e702102025. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Palumbo A, Bringhen S, Bruno B, Falcone
AP, Liberati AM, Grasso M, Ria R, Pisani F, Cangialosi C, Caravita
T, et al: Melphalan 200 mg/m(2) versus melphalan 100 mg/m(2) in
newly diagnosed myeloma patients: A prospective, multicenter phase
3 study. Blood. 115:1873–1879. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Rodriguez TE, Hari P, Stiff PJ, Smith SE,
Sterrenberg D and Vesole DH: Busulfan, melphalan, and bortezomib
versus high-dose melphalan as a conditioning regimen for autologous
hematopoietic stem cell transplantation in multiple myeloma. Biol
Blood Marrow Transplant. 22:1391–1396. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Brioli A, vom Hofe F, Rucci P, Ernst T,
Yomade O, Hilgendorf I, Scholl S, Sayer H, Mügge LO, Hochhaus A and
von Lilienfeld-Toal M: Melphalan 200 mg/m2 does not increase
toxicity and improves survival in comparison to reduced doses of
melphalan in multiple myeloma patients. Bone Marrow
Transplantation. 56:1209–1212. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Bostankolu Değirmenci B, Yegin ZA, Akdemir
ÜO, Dede A, Gündem GG, Özkurt ZN, Atay LÖ and Yağcı M: Clinical
factors associated with autologous stem cell transplantation
outcomes in multiple myeloma: Upfront transplant with MEL200
remains the standard of care. Ann Hematol. 103:269–283. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Straka C, Salwender H, Knop S, Vogel M,
Müller J, Metzner B, Langer C, Sayer H, Jung W, Dürk HA, et al:
Full or intensity-reduced high-dose melphalan and single or double
autologous stem cell transplant with or without bortezomib
consolidation in patients with newly diagnosed multiple myeloma.
Eur J Haematol. 107:529–542. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Badros A, Barlogie B, Siegel E, Morris C,
Desikan R, Zangari M, Fassas A, Anaissie E, Munshi N and Tricot G:
Autologous stem cell transplantation in elderly multiple myeloma
patients over the age of 70 years. Br J Haematol. 114:600–607.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Dimopoulos MA, Sonneveld P, Leung N,
Merlini G, Ludwig H, Kastritis E, Goldschmidt H, Joshua D, Orlowski
RZ, Powles R, et al: International myeloma working group
recommendations for the diagnosis and management of myeloma-related
renal impairment. J Clin Oncol. 34:1544–1557. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Malard F, Neri P, Bahlis NJ, Terpos E,
Moukalled N, Hungria VTM, Manier S and Mohty M: Multiple myeloma.
Nat Rev Dis Primers. 10:452024. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Ocio EM, Perrot A, Bories P, San-Miguel
JF, Blau IW, Karlin L, Martinez-Lopez J, Wang SY, Bringhen S,
Marcatti M, et al: Efficacy and safety of isatuximab plus
bortezomib, lenalidomide, and dexamethasone in patients with newly
diagnosed multiple myeloma ineligible/with no immediate intent for
autologous stem cell transplantation. Leukemia. 37:1521–1529. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Mian HS, Fiala MA and Wildes TM: Burden of
treatment among older adults with newly diagnosed multiple myeloma.
Clin Lymphoma Myeloma Leuk. 21:e152–e159. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Askeland FB, Haukås E, Slørdahl TS,
Klostergaard A, Alexandersen T, Lysén A, Abdollahi P, Nielsen LK,
Hermansen E and Schjesvold F: Isatuximab, bortezomib, lenalidomide,
and limited dexamethasone in patients with transplant-ineligible
multiple myeloma (REST): A multicentre, single-arm, phase 2 trial.
Lancet Haematol. 12:e120–e127. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Durie BGM, Hoering A, Sexton R, Abidi MH,
Epstein J, Rajkumar SV, Dispenzieri A, Kahanic SP, Thakuri MC, Reu
FJ, et al: Longer term follow-up of the randomized phase III trial
SWOG S0777: bortezomib, lenalidomide and dexamethasone vs.
lenalidomide and dexamethasone in patients (Pts) with previously
untreated multiple myeloma without an intent for immediate
autologous stem cell transplant (ASCT). Blood Cancer J. 10:532020.
View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Mateos MV, San-Miguel J, Cavo M, Suzuki K,
Jakubowiak A, Knop S, Doyen C, Lucio P, Nagy Z, Pour L, et al:
Bortezomib, melphalan, and prednisone with or without daratumumab
in transplant-ineligible patients with newly diagnosed multiple
myeloma (ALCYONE): Final analysis of an open-label, randomised,
multicentre, phase 3 trial. Lancet Oncol. 26:596–608. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Yu C, Cai T, Zhou T, Zeng N, Liang X, Pan
G, Ouyang W and Yuan C: Classification of symptom subtypes in
patients with multiple myeloma during treatment: A cross-sectional
survey study in China. BMJ Open. 13:e0664672023. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Kaweme NM, Changwe GJ and Zhou F:
Approaches and challenges in the management of multiple myeloma in
the very old: Future treatment prospects. Front Med (Lausanne).
8:6126962021. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Okazuka K, Ishida T, Nashimoto J, Uto Y,
Sato K, Miyazaki K, Ogura M, Yoshiki Y, Abe Y, Tsukada N and Suzuki
K: The efficacy and safety of modified
bortezomib-lenalidomide-dexamethasone in transplant-eligible
patients with newly diagnosed multiple myeloma. Eur J Haematol.
104:110–115. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Mateos MV, Cavo M, Blade J, Dimopoulos MA,
Suzuki K, Jakubowiak A, Knop S, Doyen C, Lucio P, Nagy Z, et al:
Overall survival with daratumumab, bortezomib, melphalan, and
prednisone in newly diagnosed multiple myeloma (ALCYONE): A
randomised, open-label, phase 3 trial. Lancet. 395:132–141. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Larocca A, Bonello F, Gaidano G,
D'Agostino M, Offidani M, Cascavilla N, Capra A, Benevolo G, Tosi
P, Galli M, et al: Dose/schedule-adjusted Rd-R vs continuous Rd for
elderly, intermediate-fit patients with newly diagnosed multiple
myeloma. Blood. 137:3027–3036. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Larocca A, Dold SM, Zweegman S, Terpos E,
Wäsch R, D'Agostino M, Scheubeck S, Goldschmidt H, Gay F, Cavo M,
et al: Patient-centered practice in elderly myeloma patients: An
overview and consensus from the European Myeloma Network (EMN).
Leukemia. 32:1697–1712. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Hajek A, Bock JO, Saum KU, Matschinger H,
Brenner H, Holleczek B, Haefeli WE, Heider D and König HH: Frailty
and healthcare costs-longitudinal results of a prospective cohort
study. Age Ageing. 47:233–241. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Jeryczynski G, Bolomsky A, Agis H and
Krauth MT: Stratification for RRMM and risk-adapted therapy:
Sequencing of therapies in RRMM. Cancers (Basel). 13:58862021.
View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Chen W, Cai Z, Chim CS, Chng WJ, Du J, Fu
C, Gao W, Hanamura I, Hou J, Huang JS, et al: Consensus guidelines
and recommendations for the anti-CD38-based therapy in clinical
practice for relapsed/refractory multiple myeloma: From the
Pan-Pacific multiple myeloma working group. Clin Hematol Int.
7:36–59. 2025. View Article : Google Scholar
|
|
131
|
Sánta H, Regáli L, Váróczy L, Szita V,
Wiedemann Á, Varju L, Rejtő L, Bartha NS, Máté D, Masszi A, et al:
Ixazomib-lenalidomide-dexamethasone for the treatment of
relapsed/refractory multiple myeloma: A hungarian real-world
analysis. J Clin Med. 15:2862025. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Murugappan MN, King-Kallimanis BL,
Bhatnagar V, Kanapuru B, Farley JF, Seifert RD, Stenehjem DD, Chen
TY, Horodniceanu EG and Kluetz PG: Patient-reported frailty
phenotype (PRFP) vs. International Myeloma Working Group frailty
index (IMWG FI) proxy: A comparison between two approaches to
measuring frailty. J Geriatr Oncol. 15:1016812024. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Davis JA, Dima D, Ahmed N, DeJarnette S,
McGuirk J, Jia X, Raza S, Khouri J, Valent J, Anwer F, et al:
Impact of frailty on outcomes after chimeric antigen receptor T
cell therapy for patients with relapsed/refractory multiple
myeloma. Transplant Cell Ther. 30:298–305. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Adegbite B, Tan CR, Shekarkhand T,
Firestone RS, Jurgens EM, Miller K, Lesokhin AM, Shah GL, Korde N,
Rajeeve S, et al: Outcomes in frail patients receiving
BCMA-directed bispecific antibodies for relapsed/refractory
multiple myeloma. Blood Adv. 9:4016–4022. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Oriol A, Dimopoulos M, Schjesvold F,
Beksac M, Facon T, Dhanasiri S, Guo S, Mu Y, Hong K, Gentili C, et
al: Pomalidomide, bortezomib, and dexamethasone in
lenalidomide-pretreated multiple myeloma: A Subanalysis of
OPTIMISMM by frailty and bortezomib dose adjustment. Clin Lymphoma
Myeloma Leuk. 24:165–176.e4. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Tagami N, Uchiyama M, Suzuki K, Shirai H,
Seto T and Iida S: Isatuximab plus pomalidomide and dexamethasone
in frail individuals with relapsed/refractory multiple myeloma in
Japan. Int J Hematol. 121:476–482. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Ríos-Tamayo R, Lecumberri R, Cibeira MT,
González-Calle V, Alonso R, Domingo-González A, Landete E, Encinas
C, Iñigo B, Blanchard MJ, et al: A simple frailty score predicts
survival and early mortality in systemic AL amyloidosis. Cancers
(Basel). 16:16892024. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Bayes-Genis A, Docherty KF, Petrie MC,
Januzzi JL, Mueller C, Anderson L, Bozkurt B, Butler J, Chioncel O,
Cleland JGF, et al: Practical algorithms for early diagnosis of
heart failure and heart stress using NT-proBNP: A clinical
consensus statement from the Heart Failure Association of the ESC.
Eur J Heart Fail. 25:1891–1898. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Adamo M, Pagnesi M, Mebazaa A, Davison B,
Edwards C, Tomasoni D, Arrigo M, Barros M, Biegus J, Celutkiene J,
et al: NT-proBNP and high intensity care for acute heart failure:
the STRONG-HF trial. Eur Heart J. 44:2947–2962. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Teramoto K, Tay WT, Tromp J, Ouwerkerk W,
Teng TK, Chandramouli C, Liew OW, Chong J, Poppe KK, Lund M, et al:
Longitudinal NT-proBNP: Associations with echocardiographic changes
and outcomes in heart failure. J Am Heart Assoc. 13:e0322542024.
View Article : Google Scholar : PubMed/NCBI
|
|
141
|
de Terwangne C, Vaes B, Pasquet A, Boland
B, Pouleur AC and Degryse JM: Untangling the prognostic value of
diastolic dysfunction, NT-proBNP, and frailty in older patients
with preserved left ventricular ejection fraction without valvular
disease. BMC Geriatr. 25:9892025. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Giri S, Dahal S, Bal S, Godby KN, Richman
J, Olszewski AJ, Williams GR, Brown C, Buford TW, Costa LJ and
Bhatia S: Pre-treatment neutrophil to lymphocyte ratio as a
biomarker of frailty and predictor of survival among older adults
with multiple myeloma. J Geriatr Oncol. 13:486–492. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Mikulski D, Kościelny K, Nowicki M,
Wawrzyniak E, Kalwas M, Kowalik M, Pryt M, Sęczkowska E, Świątek A,
Wierzbowska A and Fendler W: Neutrophil to lymphocyte ratio (NLR)
impact on the progression-free survival and overall survival of
multiple myeloma patients treated with high-dose chemotherapy and
autologous stem cell transplantation. Leuk Lymphoma. 64:98–106.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Levinson T and Wasserman A: C-Reactive
Protein Velocity (CRPv) as a new biomarker for the early detection
of acute infection/inflammation. Int J Mol Sci. 23:81002022.
View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Roy A, Zeller J, Nero TL, Klepetko J,
Eisenhardt SU, Parker MW, McFadyen JD and Peter K: C-reactive
protein: The nexus between inflammation and protein misfolding
diseases. Front Immunol. 16:16127032025. View Article : Google Scholar : PubMed/NCBI
|
|
146
|
Sibianu M and Slevin M: The pathogenic
role of C-Reactive protein in diabetes-linked unstable
atherosclerosis. Int J Mol Sci. 26:68552025. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Tang M, Cao H, Wei XH, Zhen Q, Liu F, Wang
YF, Fan NG and Peng YD: Association between high-sensitivity
C-reactive protein and diabetic kidney disease in patients with
type 2 diabetes mellitus. Front Endocrinol (Lausanne).
13:8855162022. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Ehteram H, Raji S, Rahmati M, Teymoori H,
Safarpour S, Poursharifi N, Hashem Zadeh M, Pakzad R, Habibi H and
Mobarra N: Association between Pro-oxidant-Antioxidant balance and
high-sensitivity C-reactive protein in type 2 diabetes mellitus: A
Study on Postmenopausal Women. Endocrinol Diabetes Metab.
6:e4002023. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Guo L, Lv H, Wang J, Zhang B, Zhu Y, Zhang
X, Zhu H, Zhou X and Xia Y: Predictive value of high sensitivity
C-reactive protein in three-vessel disease patients with and
without type 2 diabetes. Cardiovasc Diabetol. 22:912023. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Lin S, Chen X, Cheng Y, Huang H, Yang F,
Bao Z and Fan Y: C-Reactive protein level as a novel serum
biomarker in sarcopenia. Mediators Inflamm. 2024:33623362024.
View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Wang A, Wang M, Li G, Zhang X and Wu M:
Impact of C-Reactive protein and liver frailty index on sarcopenia
and poor outcome in liver transplant patients. Clin Lab. doi:
10.7754/Clin.Lab.2024.240332.
|
|
152
|
Yang J, Liu Z, Liu H, He J, Yang J, Lin P,
Wang Q, Du J, Ma W, Yin Z, et al: C-reactive protein promotes bone
destruction in human myeloma through the CD32-p38 MAPK-Twist axis.
Sci Signal. 10:eaan62822017. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Radnay Z, Illés Á, Udvardy M, Prohászka Z,
Sinkovits G, Csányi MC, Kellermayer M, Kiss A and Hársfalvi J: Von
willebrand factor and platelet levels before conditioning
chemotherapy indicate bone marrow regeneration following autologous
hematopoietic stem cell transplantation. Transplant Cell Ther.
28:830.e1–830.e7. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Jimenez-Zepeda VH, Lee H, Tay J, Duggan P,
McCulloch S, Neri P and Bahlis NJ: N-Terminal pro-brain natriuretic
peptide (NTproBNP) in patients with symptomatic multiple myeloma:
report from a single institution. Ann Hematol. 100:2521–2527. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Patel BG, Luo S, Wildes TM and Sanfilippo
KM: Frailty in older adults with multiple myeloma: A study of US
veterans. JCO Clin Cancer Inform. 4:117–127. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
156
|
Ipek Y, Karademir N, Yilmazer O and Yilmaz
G: The effects of second primary malignancies and frailty on
overall survival and mortality in geriatric Turkish patients with
multiple myeloma. Curr Oncol. 30:5615–5630. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Zhang XL, Pan MM, Kaminskiy Y, Jin SW, Mi
JQ, Zhang WP and Xu J: Interleukin-5 is an adjunctive biomarker for
engraftment syndrome in multiple myeloma patients undergoing
autologous hematopoietic stem cell transplantation. Clin Immunol.
279:1105752025. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Zhou Q, Xu F, Wen J, Yue J, Zhang Y, Du L,
Kou K, Su J, Liu Y and Liang X: Tregs at diagnosis as a potential
biomarker for predicting high-risk functionality in newly diagnosed
multiple myeloma. Cancer Med. 14:e709802025. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Varzieva VG, Mesonzhnik NV, Ilgisonis IS,
Belenkov YN, Kozhevnikova MV and Appolonova SA: Metabolomic
biomarkers of multiple myeloma: A systematic review. Biochim
Biophys Acta Rev Cancer. 1879:1891512024. View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Tzasta A, Wijnands C, Baalman K, van Gool
AJ, Zweegman S and Jacobs JFM: Advances in multiple myeloma
blood-based monitoring and its clinical applications. Crit Rev Clin
Lab Sci. 62:491–509. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
161
|
Xu J, Yu N, Zhao P, Wang F, Huang J, Cui
Y, Ding H, Yang Y, Gao Y, Pan L, et al: Intratumor heterogeneity of
MIF expression correlates with extramedullary involvement of
multiple myeloma. Front Oncol. 11:6943312021. View Article : Google Scholar : PubMed/NCBI
|
|
162
|
Li D, Qiao B, Cheng Q, Chen L, Zhao F, Zuo
L, Li Q and Sun C: Macrophage migration inhibitory factor as a
prognostic biomarker in multiple myeloma: Clinical significance and
in vitro effects. Carcinogenesis. 46:bgaf0332025. View Article : Google Scholar : PubMed/NCBI
|
|
163
|
Zhang Z, Zhou X, Guo J, Zhang F, Qian Y,
Wang G, Duan M, Wang Y, Zhao H, Yang Z, et al: TA-MSCs,
TA-MSCs-EVs, MIF: Their crosstalk in immunosuppressive tumor
microenvironment. J Transl Med. 20:3202022. View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Kraus S, Klassen P, Kircher M, Dierks A,
Habringer S, Gäble A, Kortüm KM, Weinhold N, Ademaj-Kospiri V,
Werner RA, et al: Reduced splenic uptake on
(68)Ga-Pentixafor-PET/CT imaging in multiple myeloma-a potential
imaging biomarker for disease prognosis. Theranostics.
12:5986–5994. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
165
|
Soysal P, Stubbs B, Lucato P, Luchini C,
Solmi M, Peluso R, Sergi G, Isik AT, Manzato E, Maggi S, et al:
Inflammation and frailty in the elderly: A systematic review and
meta-analysis. Ageing Res Rev. 31:1–8. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Mohan Lal B, van Rhee F and Al Hadidi S:
Current state of evidence on definitions and management of
high-risk multiple myeloma. Curr Oncol Rep. 27:258–277. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Tang S, Long X, Li F, Jiang S, Fu Y and
Liu J: Identification of RUVBL2 as a novel biomarker to predict the
prognosis and drug sensitivity in multiple myeloma based on
ferroptosis genes. Hematology. 30:24674992025. View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Groen K, Smits F, Nasserinejad K, Levin
MD, Regelink JC, Timmers GJ, de Waal EGM, Westerman M, Velders GA,
de Heer K, et al: Uncovering the ultra-frail: A distinct subgroup
in non-transplant eligible newly diagnosed patients with multiple
myeloma, with an inferior clinical outcome. Hemasphere.
9:e702682025. View Article : Google Scholar : PubMed/NCBI
|
|
169
|
Ruiz JG, Dent E, Morley JE, Merchant RA,
Beilby J, Beard J, Tripathy C, Sorin M, Andrieu S, Aprahamian I, et
al: Screening for and managing the person with frailty in primary
care: ICFSR consensus guidelines. J Nutr Health Aging. 24:920–927.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
170
|
Chinese Hematology Association; Chinese
Society of Hematology, : Guidelines for the diagnosis and
management of multiple myeloma in China (2024 revision). Zhonghua
Nei Ke Za Zhi. 63:1186–1195. 2024.(In Chinese). PubMed/NCBI
|
|
171
|
Dai Y and Jin F: Dynamic frailty-tailored
therapy (DynaFiT): A proof-of-concept study in elderly patients
with newly diagnosed multiple myeloma. Blood Sci. 6:e002082024.
View Article : Google Scholar : PubMed/NCBI
|
|
172
|
Bringhen S, D'Agostino M, Paris L,
Ballanti S, Pescosta N, Spada S, Pezzatti S, Grasso M,
Rota-Scalabrini D, De Rosa L, et al: Lenalidomide-based induction
and maintenance in elderly newly diagnosed multiple myeloma
patients: Updated results of the EMN01 randomized trial.
Haematologica. 105:1937–1947. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
173
|
Szmitko PE, Wang CH, Weisel RD, de Almeida
JR, Anderson TJ and Verma S: New markers of inflammation and
endothelial cell activation: Part I. Circulation. 108:1917–1923.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
174
|
Mohty M, Facon T, Malard F and Harousseau
JL: A roadmap towards improving outcomes in multiple myeloma. Blood
Cancer J. 14:1352024. View Article : Google Scholar : PubMed/NCBI
|