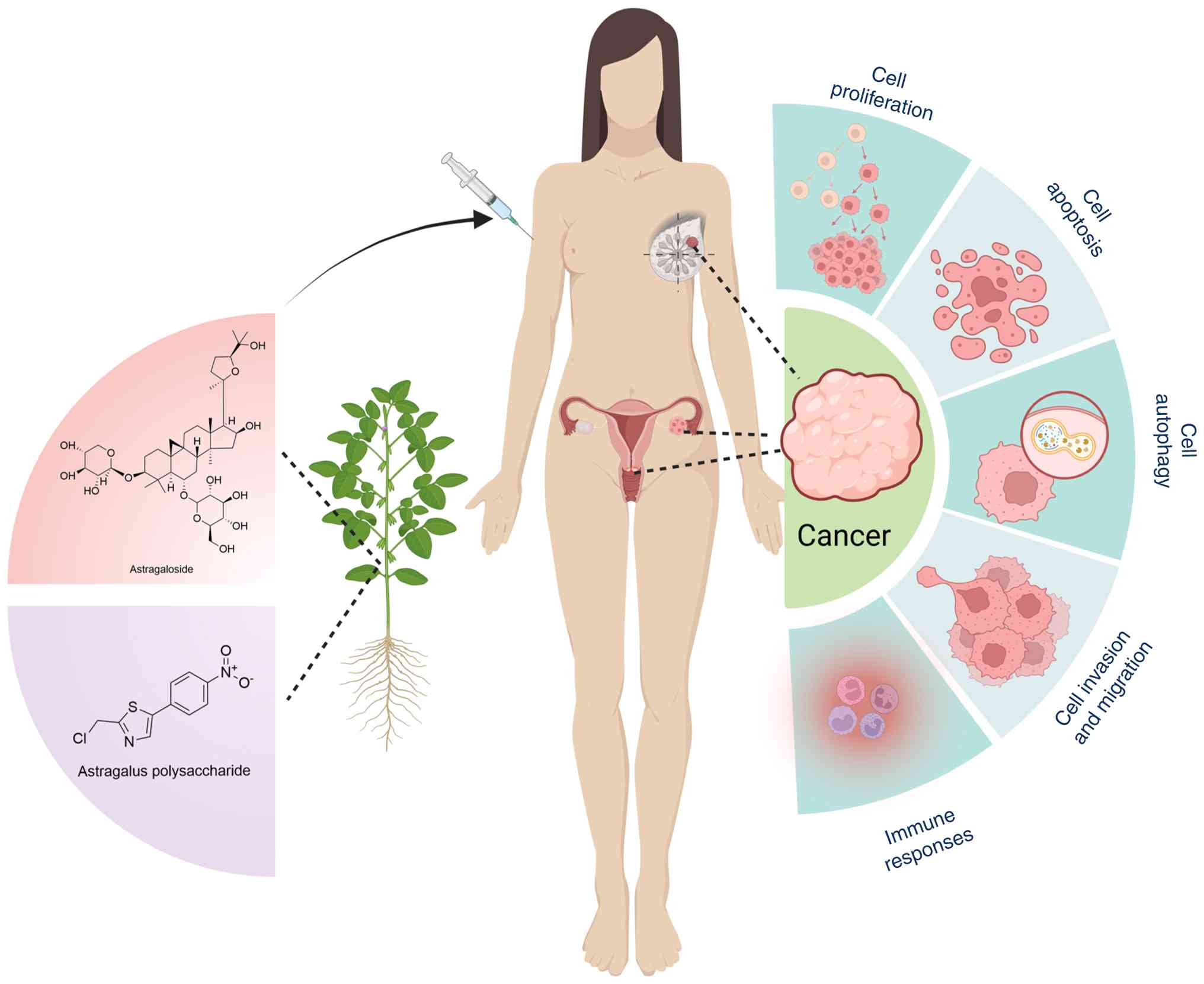
Introduction
Gynecological malignancies, particularly cervical
and ovarian cancers, remain a major threat to women's health and
contribute substantially to the global disease burden. Cervical
cancer is the fourth most common cancer in women worldwide and
accounts for ~6.5% of all female cancer cases (1). It is often highly invasive and
frequently diagnosed at an advanced stage, resulting in
considerable mortality and morbidity (2). Ovarian cancer carries an estimated
lifetime risk of ~2% in women, and both incidence and mortality
increase with age (3). Each year,
~239,000 new ovarian cancer cases are reported globally, with an
estimated 152,000 associated deaths (4). Because ovarian cancer is typically
insidious in onset and effective screening strategies remain
limited, the 5-year survival rate remains <20%, underscoring the
urgency for improved prevention and treatment approaches (5).
Although breast cancer is not traditionally
categorized as a gynecological malignancy, it is closely linked to
women's health and remains a leading cause of cancer-related
mortalities among women. Its complex pathogenesis and heterogeneous
clinical presentation continue to pose major challenges for
effective prevention and management (6). Breast cancer is the most commonly
diagnosed malignancy in women, with ~1.7 million new cases
annually, and both incidence and mortality are projected to rise
further over the next decade (7).
Beyond disease-specific symptoms such as abnormal bleeding, pain
and reproductive dysfunction (8),
breast cancer, ovarian cancer and cervical cancer can necessitate
mastectomy or resection of reproductive organs, and may lead to
premature menopause and infertility, with profound consequences for
physical and psychological well-being. They also impose substantial
psychosocial burdens, affecting family relationships and overall
quality of life, and have therefore attracted increasing clinical
and public attention (9).
Astragalus membranaceus, known as Huangqi, is
a perennial herb in the Fabaceae family. It typically reaches a
height of 50 to 100 cm and bears a thick cylindrical root that is
light brown on the exterior and yellow white internally, with a
firm and fibrous texture. The main and lateral roots are used
medicinally, and the most commonly utilized sources are
Astragalus membranaceus (Fisch.) Bge. var.
mongholicus and Astragalus membranaceus (Fisch.) Bge.
(10). Astragalus contains
diverse chemical constituents, including saponins, polysaccharides,
flavonoids, amino acids, as well as trace elements, such as zinc
and iron.. Among these, saponins and polysaccharides are regarded
as the principal bioactive components (11). The saponin fraction consists
predominantly of triterpenoid saponins, with astragalosides (AST)
I, II and IV and isoastragalosides I and II accounting for >80%
of total saponins (12).
Astragalus polysaccharides (APS) are water-soluble
macromolecules extracted mainly from the stems or roots and
represent one of the most important naturally derived active
ingredients of Astragalus (13). Wild Astragalus species are
primarily distributed across northern China, Kazakhstan, Mongolia
and the Russian Far East, with the largest cultivation area located
in northern China (14). Increasing
evidence has identified Astragalus as a rich source of
bioactive compounds with potent antitumor potential (15–17).
Previous reviews have largely focused on the
anticancer mechanisms of individual Astragalus constituents
in single tumor types. By contrast, evidence regarding the effect
of AST and APS in gynecological and breast cancers remains
scattered and has not been systematically integrated. To address
this gap, the present review provided a comprehensive summary of
the molecular mechanisms and signaling pathways through which AST
and APS exert therapeutic effects in breast, ovarian, and cervical
cancers. This study aims to establish an integrated pharmacological
framework and to support the development of integrative and
precision treatment strategies for women's cancers worldwide, as
illustrated in Fig. 1.
Clinical applications and pharmacological
basis of Astragalus membranaceus
Major bioactive components of
Astragalus membranaceus
Astragalus membranaceus produces a broad
spectrum of biologically active secondary metabolites, with
triterpenoid saponins and polysaccharides representing the major
classes. Triterpenoid saponins are formed through conjugation of
triterpene aglycones with sugars or other substituent groups, and
their biosynthesis proceeds via multiple enzyme-mediated steps that
include precursor generation and carbon ring assembly.
Polysaccharides derived from Astragalus comprise
heteropolysaccharides, neutral polysaccharides, glucans and acidic
polysaccharides, with β- (1→4)-glucosidic linkages as the
predominant glycosidic bonds (18).
Due to the amphiphilic properties of saponins, a variety of
extraction approaches using aqueous or alcoholic solvents have been
developed. Among these, n-butanol and methanol are considered
particularly efficient for isolating triterpenoid saponins
(19). Conventional preparation of
APS typically relies on solvent or aqueous extraction followed by
ethanol precipitation, which remains a widely used and technically
straightforward method (20).
Pharmacological evidence indicates that AST can downregulate p21
and Bcl-xL, and upregulate Caspase-3 and cleaved PARP. It mainly
exerts its antitumor effects by inhibiting tumor cell proliferation
and inducing cell apoptosis (21).
APS also displays a broad range of biological activities, including
antitumor, anti-aging and antiviral effects (22–24).
These actions are mediated through multiple mechanisms, such as
modulation of immune responses, promotion of apoptotic signaling,
and inhibition of tumor invasion and metastasis (25).
Clinical indications of Astragalus
membranaceus
Astragalus membranaceus has traditionally
been used to enhance immune function and support overall
physiological homeostasis. In contemporary oncology, it is
increasingly applied as an adjuvant therapy, with particular
relevance to the management of common malignancies in women, such
as breast, cervical and ovarian cancers (26). A pharmacological study indicates
that bioactive constituents of Astragalus, such as
astragaloside III, exert bidirectional immunomodulatory actions by
activating immune effector cells, including lymphocytes and natural
killer (NK) cells, thereby strengthening antitumor immunity. These
constituents may also mitigate immunosuppressant-associated
myelosuppression, which can improve therapeutic efficacy and reduce
treatment-related adverse effects (27). In addition, APS has been reported to
upregulate IL-12, TNF-α, Bax and Caspase-3, and downregulate Bcl-2,
thereby exerting antitumor activities through immune regulation,
induction of tumor cell apoptosis, and inhibition of invasion and
migration (28,29).
Clinical evidence further suggests that APS can
enhance treatment responses and improve immune status in patients
with cancer. Compared with radiotherapy alone, combined
radiotherapy and APS has been associated with an increased
CD4+ to CD8+ ratio in breast cancer and other
malignancies, which may contribute to improved clinical outcomes
(30). However, clinical studies
specifically evaluating AST and APS remain limited. Most available
evidence derives from investigations of Astragalus as a
whole herb or from traditional Chinese medicine formulas in which
Astragalus is a principal component. This pattern reflects
current clinical practice, where compound prescriptions are
commonly used, and findings from formula-based studies are more
readily translatable to real-world application. For example,
clinical data indicate that Astragalus-based formulas
combined with chemotherapy can reduce post-chemotherapy adverse
reactions and chemotherapy-associated hepatorenal toxicity in
cervical cancer while improving overall efficacy (31). Additional clinical studies have
reported that Astragalus may alleviate common cancer-related
complications and improve patient quality of life (32).
Beyond oncology, Astragalus has shown
beneficial effects in anti-inflammatory, antioxidant and
anti-infective settings (33–35).
Accordingly, its use in patients with cancer is primarily supported
by its roles in immunoregulation and functional support. In this
population, Astragalus may help relieve frequent symptoms
such as fatigue (36), anorexia
(37) and vomiting (38), while enhancing immune competence and
improving quality of life (39).
Therefore, in the clinical management of female malignancies,
Astragalus and its preparations are widely used as
adjunctive interventions to improve treatment tolerance, support
therapeutic response and potentially enhance prognosis.
Mechanisms of Astragalus active constituents
in female malignancies
Mechanisms of Astragalus constituents
in breast cancer
Mechanisms of APS in breast cancer
Disruption of cellular homeostasis is a fundamental
driver of tumor initiation and progression. Uncontrolled tumor cell
proliferation frequently results from impaired apoptosis, a form of
programmed cell death that is essential for maintaining tissue
integrity. Programmed cell death not only shapes the
immunosuppressive tumor microenvironment but also markedly
influences therapeutic responses to anticancer treatments (40). Li et al (41) reported that APS activates RAW264.7
macrophages and increases the production of nitric oxide (NO) and
tumor necrosis factor alpha (TNF-α), thereby inducing apoptosis and
inhibiting the proliferation of breast cancer cells. In a related
study, Li et al (42) showed
that APS increased ANXA1 mRNA and protein expression while reducing
epidermal growth factor receptor (EGFR) mRNA levels in GFP-4T1
cells, suggesting that APS may promote programmed cell death
through coordinated upregulation of ANXA1 and suppression of EGFR
signaling.
Advances in drug delivery have further expanded the
therapeutic potential of APS. A dual-targeted nanodelivery system
based on quercetin 3 prime,3 prime dithiodipropionic acid, APS, and
folate, termed Nano Pomegranate, effectively induced programmed
cell death in breast cancer cells and demonstrated improved
antitumor efficacy with fewer adverse effects compared with free
curcumin, highlighting its translational promise (43). In triple negative breast cancer
(TNBC) 4T1 cells, Sun et al (44) found that 24 h co-treatment with APS
and cisplatin significantly enhanced apoptosis by downregulating
Bcl-2 and Bcl-xL and upregulating PUMA, cleaved caspase 3, and
cleaved PARP. The combination also increased E-cadherin expression.
Because E-cadherin is central to cell-cell adhesion, its loss is
closely associated with epithelial mesenchymal transition (EMT),
enhanced invasiveness, and poor prognosis across multiple cancers
(45). Consistently, Yang et
al (46) reported that APS
treatment for 24 h reduced Snail and vimentin expression, decreased
Wnt1 and β-catenin levels, and increased E-cadherin expression in
MCF 7 and MDA MB 231 cells, supporting inhibition of Wnt/β-catenin
signaling and suppression of breast cancer cell proliferation,
invasion, and migration.
Beyond direct cytotoxicity, APS also exerts
immunomodulatory and synergistic anticancer effects. Zhou et
al (47) showed that APS
stimulates RAW264.7 macrophages to secrete immunoregulatory
mediators including NO, TNF-α, IL-1β, and IL-6 through activation
of the Toll-like receptor 4- and MyD88-dependent pathways, thereby
enhancing antitumor immunity. Moreover, Bao et al (48) demonstrated that APS attenuates
paclitaxel-induced cytotoxicity in RAW264.7 cells by downregulating
phospho-histone H2A (P-HA), PARP, checkpoint kinase 1, p53 and p21
while upregulating Bcl-xL and Mcl-1. This selective protection of
immune cells may improve the therapeutic index of paclitaxel in
breast cancer treatment.
Mechanisms of AST in breast
cancer
Accumulating evidence indicates that ASTs,
particularly astragaloside IV (AS-IV), exert robust antitumor
effects through multiple mechanisms (49). Because chemoresistance remains a
major barrier to effective cancer therapy (50), a study has investigated the
potential of AS-IV to restore treatment sensitivity. Lou et
al (51) reported that AS-IV
upregulates Nrf2 and increases intracellular ATP levels, thereby
enhancing the efflux function of breast cancer resistance protein
(BCRP). Huang et al (52)
showed that AS-IV markedly reduces the stemness of breast cancer
stem cells and reverses paclitaxel resistance in MCF7-derived
cancer stem cells by modulating the (hypoxia-inducible factor)
HIF-1α/HIF-2α axis and inhibiting Notch, PI3K/Akt/mTOR and
JAK/STAT3 signaling. Caveolin-1, a membrane protein linked to
metastasis and drug resistance (53), has also been reported to be
downregulated by AS-IV in a dose-dependent manner. This change
activates the endothelial nitric oxide (NO) synthase
(eNOS)/NO/peroxynitrite (ONOO−) pathway, promotes
oxidative damage, and increases paclitaxel sensitivity in MCF 7 and
MDA MB 231 cells (54). In
addition, a liposomal co-delivery system incorporating oxymatrine
and AS-IV suppresses cancer-associated fibroblast activation and
increases tumor-infiltrating lymphocytes, thereby potentiating the
efficacy of programmed cell death protein 1 inhibitors and
supporting the potential utility of AS-IV-based strategies in
breast cancer immunotherapy (55).
Beyond chemosensitization, AS-IV also inhibits
invasion and metastasis through modulation of tumor and
microenvironmental signaling. Yu et al (56) found that AS-IV downregulated TGF-β
and suppressed Akt/forkhead box protein (FOX)O1 signaling in
THP-1-derived macrophages. This effect reduced M2 polarization and
decreased the expression of IL-10, MMP-9, CD206, CD163 and TGF-β,
which in turn limited macrophage-mediated breast cancer cell
proliferation, invasion and migration. Vav3, a guanine nucleotide
exchange factor that promotes tumor proliferation and invasion when
overexpressed (57), has been
identified as another target of AS-IV. Jiang et al (58) reported that AS-IV downregulated Vav3
and its downstream effectors, ERK1/2, JNK, MMP-2 and MMP-9, thereby
reducing Rac1 activation and inhibiting breast cancer cell
viability and motility. Similarly, Hu et al (59) demonstrated that AS-IV induced the
long non-coding RNA TRHDE-AS1 in MCF-7 and MDA-MB-231 cells in a
dose- and time-dependent manner, leading to reduced proliferation
and invasion in vitro and in vivo. Moreover, Yang
et al (60) demonstrated
that AS-IV activated the transcription factor FOXA1 and upregulates
galactose-3-O-sulfotransferase 1, thereby suppressing proliferation
and viability in obesity-associated triple negative breast cancer
by remodeling sphingolipid metabolism.
Collectively, APS related studies in breast cancer
have primarily employed RAW264.7 macrophages and TNBC 4T1 cells,
with an emphasis on inhibiting proliferation, inducing apoptosis,
modulating immunity and enhancing chemotherapeutic efficacy
(41–44,46–48).
By contrast, studies of AS-IV have largely relied on MCF-7 and
MDA-MB-231 models and have highlighted regulation of Akt/FOXO1,
eNOS/NO/ONOO and Rac1/MAPK signaling, together with their
associated molecular targets (51,52,54–56,58–60),
as summarized in Fig. 2.
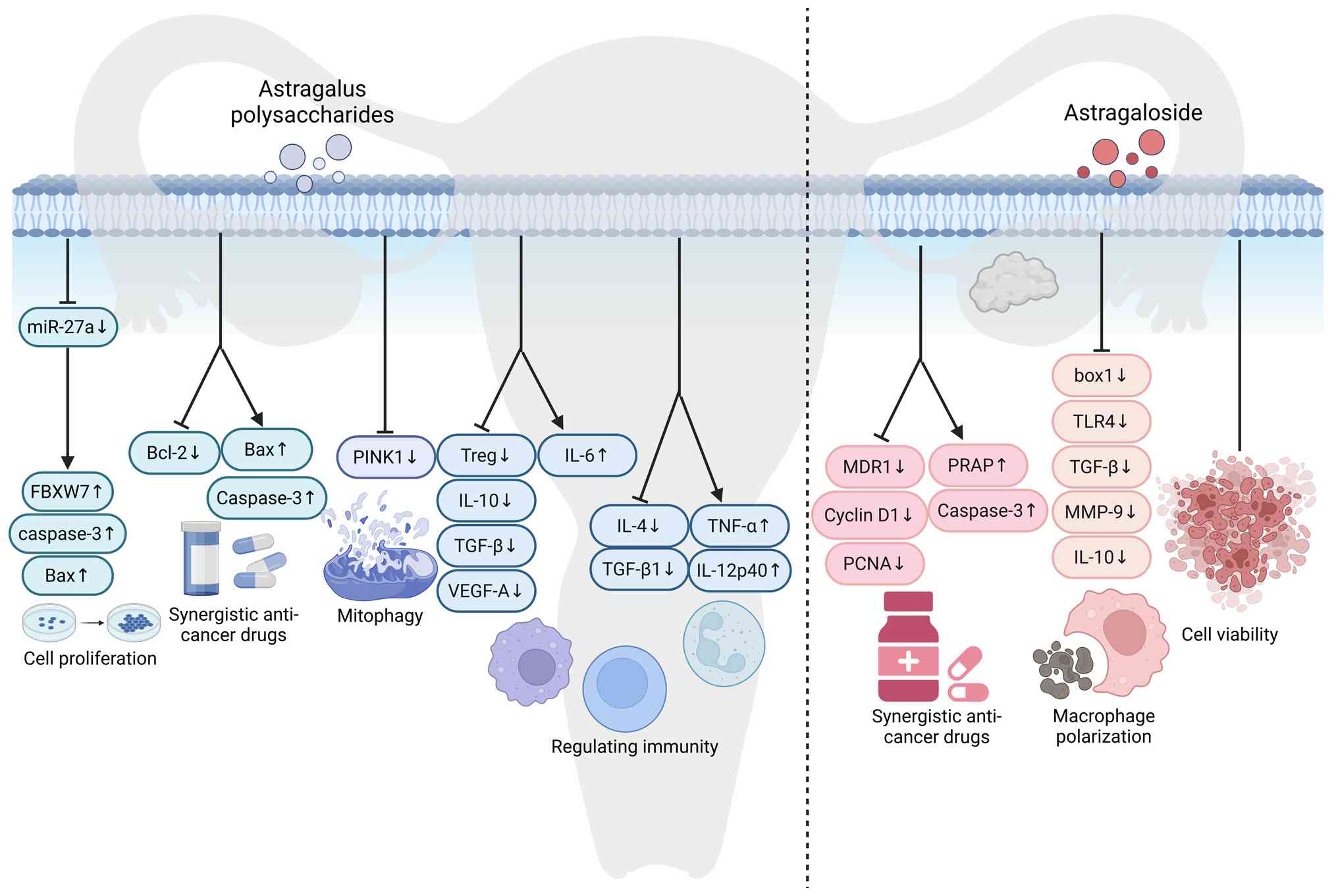
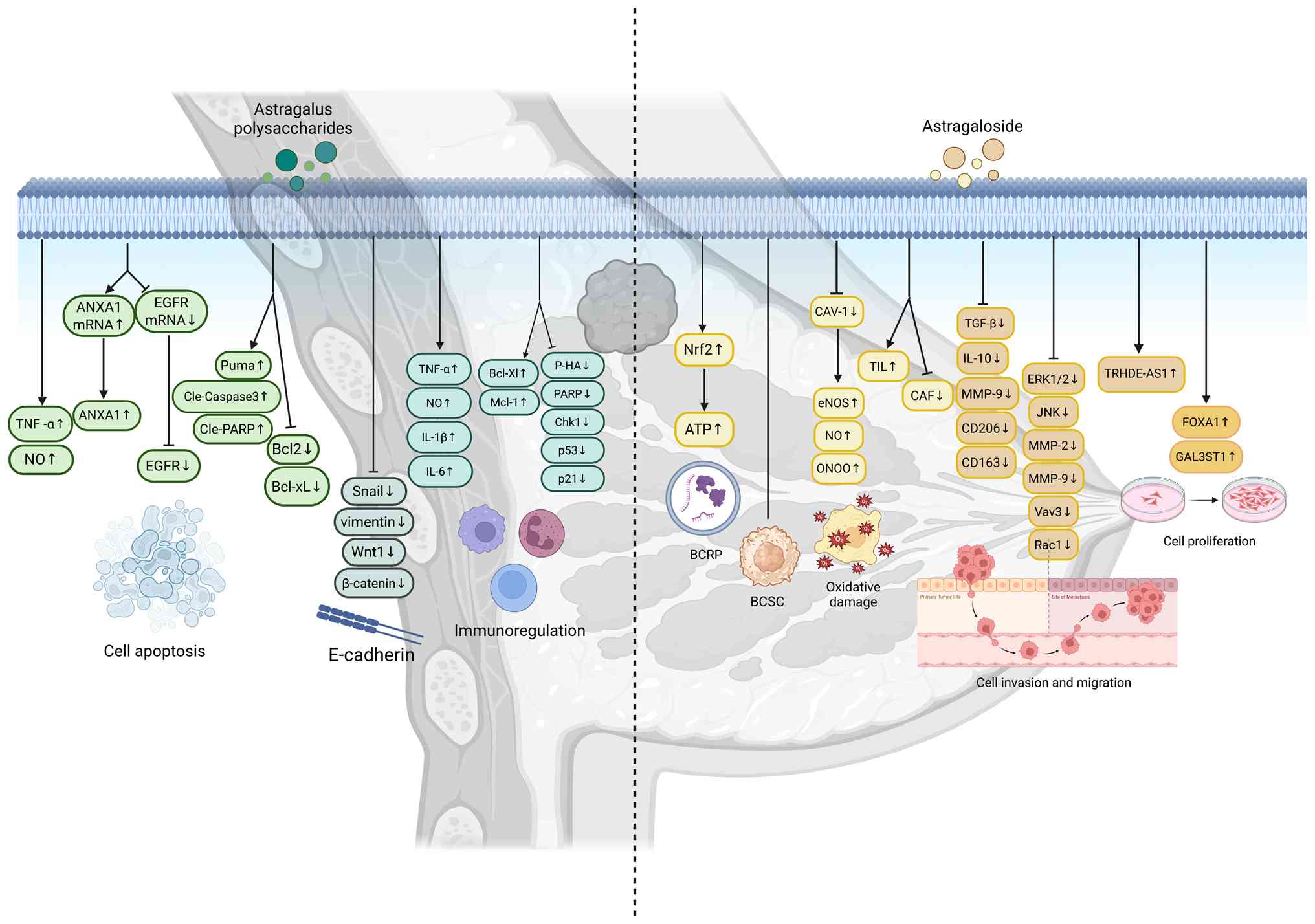 | Figure 2.Mechanism of action of
Astragalus polysaccharides and astragalosides in treating
breast cancer. ANXA, annexin A1; NO, nitric oxide; P-HA,
phospho-histone H2A; Chk1, checkpoint kinase 1; BCRP, breast cancer
resistance protein; BCSC, breast cancer stem cells; ONOO,
peroxynitrite; eNOS, endothelial nitric oxide synthase; CAV-1,
caveolin-1; TIL, tumor-infiltrating lymphocytes; CAF,
cancer-associated fibroblasts; TRHDE-AS1, the lncRNA of TRHDE
antisense RNA 1; FOXA1, forkhead box A1; GAL3ST1,
galactose-3-o-sulfotransferase 1. |
Mechanisms of Astragalus active
constituents in ovarian cancer
Mechanisms of APS in ovarian cancer
APS are natural compounds with strong
immunomodulatory and antitumor properties (61) and have gained attention as potential
therapeutic agents for ovarian cancer. Growing evidence indicates
that APS suppresses tumor growth by inhibiting cancer cell
proliferation and promoting apoptosis. Guo et al (62) reported that APS reduced microRNA
(miR)-27a expression in a dose-dependent manner, which increased
the expression of the tumor suppressor FBXW7 and consequently
inhibited proliferation while inducing apoptosis in the human
ovarian cancer cell line OV-90. Consistently, Li et al
(63) showed that APS combined with
cisplatin markedly decreased SKOV3 cell viability and enhanced
apoptosis by downregulating Bcl-2, upregulating Bax and caspase-3,
and activating JNK1/2 signaling. This synergistic interaction
increased cisplatin sensitivity and strengthened its antitumor
activity in ovarian cancer.
Cancer stem cells are closely linked to recurrence
and chemoresistance (64). Peng
et al (65) demonstrated
that APS downregulates PTEN-induced kinase 1 (PINK1) and suppresses
mitochondrial autophagy through the PINK1/Parkin pathway, thereby
increasing the sensitivity of ovarian cancer stem cells to poly
(ADP-ribose) polymerase inhibitors. This combination promoted
apoptosis and reduced both viability and self-renewal capacity of
ovarian cancer stem cells.
APS also modulates immune responses relevant to
ovarian cancer. Shokati et al (66) found that 7 day exposure of
peripheral blood mononuclear cells to APS significantly increased
cell proliferation, reduced the proportion of regulatory T cells in
the T cell population, decreased expression levels of
anti-inflammatory mediators including IL-10, TGF-β and VEGF-A, and
increased the levels of proinflammatory cytokine IL-6, collectively
supporting enhanced antitumor immunity. In addition, Liu et
al (67) developed an APS-based
micellar delivery system modified with methotrexate and
podophyllotoxin. This formulation increased TNF-α and IL-12p40
levels while reducing IL-4 and TGF-β1 expression levels, and it
promoted repolarization of tumor associated macrophages from an
M2-like state toward an M1-like phenotype, supporting immune
mediated targeting of ovarian cancer.
Mechanisms of AST in ovarian
cancer
Given the high rates of chemoresistance and
recurrence in ovarian cancer, natural products with intrinsic
antitumor activity, including AST, are increasingly investigated as
potential adjunctive therapies. Zhang et al (68) demonstrated in both in vitro
and in vivo models that astragaloside II enhances cisplatin
efficacy by downregulating multidrug resistance protein 1 (MDR1),
the cell cycle regulator cyclin D1 and proliferating cell nuclear
antigen, while upregulating apoptosis-associated proteins PARP and
caspase-3. This coordinated regulation promoted apoptosis and
inhibited ovarian cancer cell proliferation, thereby increasing
cisplatin sensitivity. AS-IV has also been reported to influence
ovarian cancer progression through immune microenvironment
modulation. Wang et al (69)
treated IL-4/IL-13-induced THP-1 derived macrophages with AS-IV and
found that it suppressed the overexpression of high mobility group
box 1 (HMGB1) and Toll-like receptor 4 (TLR4), reduced the
secretion of M2-associated factors including TGF-β, MMP-9 and
IL-10, and inhibited ovarian cancer cell proliferation, invasion
and migration by promoting macrophage repolarization. In addition,
Dekinash et al (70)
profiled methanolic Astragalus extracts using HPLC-MS/MS and
identified six saponins and five flavonoids with moderate cytotoxic
activity against SKOV3 ovarian cancer cells, as reflected by
reduced cell viability, which supports a direct antiproliferative
potential of the extract.
Collectively, APS-related studies in ovarian cancer,
which primarily use human cell lines such as OV-90 and SKOV3, have
focused on mechanistic axes including miR-27a/FBXW7, JNK1/2 and
PINK1/Parkin. These findings suggest that APS inhibits
proliferation, induces programmed cell death, regulates immune
responses, modulates macrophage polarization and can synergize with
chemotherapeutic agents to enhance efficacy while reducing
toxicity. The development of APS-based delivery systems further
supports translational potential (62,63,65–67).
By contrast, studies on AST have mainly examined their roles in
suppressing proliferation, promoting apoptosis, improving
chemosensitivity and reshaping macrophage polarization, with
particular emphasis on HMGB1-TLR4 signaling (68–70),
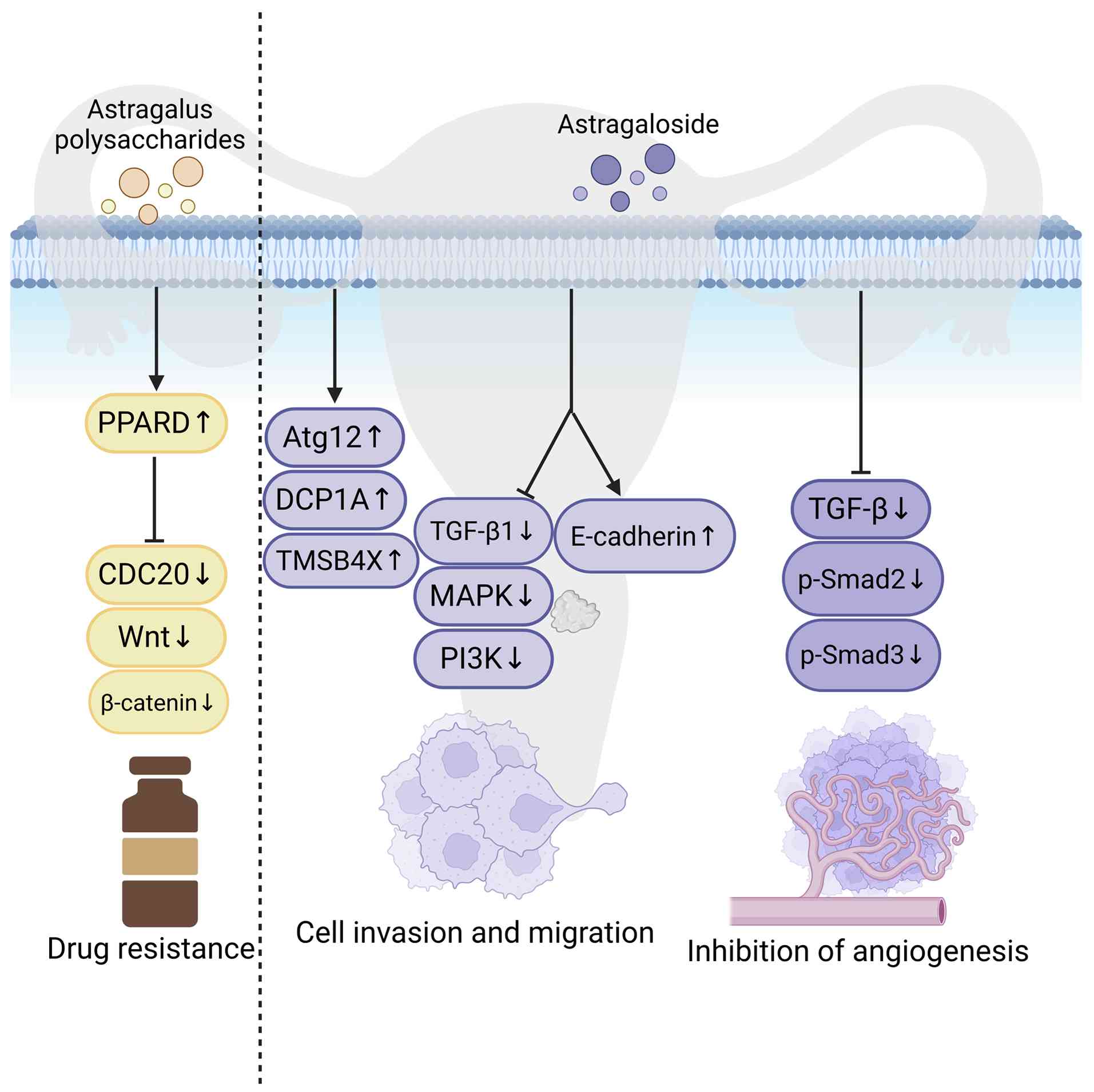
as summarized in Fig. 3.
Mechanisms of Astragalus bioactive
constituents in cervical cancer
Mechanisms of APS in cervical cancer
As principal bioactive constituents of Astragalus
membranaceus, APS have potential therapeutic value in cervical
cancer. Liu et al (71)
reported that 24 h treatment of cisplatin-resistant cervical cancer
cells with APS increased peroxisome proliferator-activated receptor
δ (PPARD) expression, which in turn suppressed CDC20 transcription
and inhibited Wnt/β-catenin signaling. This regulatory effect
markedly enhanced cisplatin sensitivity, reduced proliferative
capacity, and promoted apoptosis. These findings suggest that APS
may exert antitumor activity in cervical cancer by modulating cell
cycle regulation and reversing chemoresistance.
Mechanisms of AST in cervical
cancer
Clarifying the mechanisms through which AST regulate
cervical cancer progression is essential for defining their
clinical potential. Available evidence indicates that AST can
inhibit cervical cancer cell invasion and migration through
multiple signaling pathways. Xia et al (72), using integrated in vitro and
in vivo assays together with quantitative proteomics,
reported that AS-IV increased Atg12 expression and upregulated
mRNA-decapping enzyme 1A and Thymosin β-4, thereby inducing
autophagy and suppressing cervical cancer cell proliferation and
invasion. EMT is a critical process that promotes tumor cell
invasion and migration (73). Zhang
et al (74) found that 24 h
AS-IV treatment reduced TGF-β1 expression and increased E-cadherin
levels, while inhibiting phosphorylation of MAPK and PI3K. These
effects were associated with EMT suppression and reduced invasive
and migratory capacity of cervical cancer cells. Macrophages, which
are abundant stromal components within the tumor microenvironment,
also contribute to cancer progression through
polarization-dependent functions (75). Shen et al (76) demonstrated that AS-IV downregulated
TGF-β and decreased phosphorylated Smad2 and Smad3 in cervical
cancer cells. Inhibition of TGF-β/Smad2/3 signaling reduced
macrophage M2 polarization and consequently restrained EMT
progression, migration and angiogenesis.
Overall, the study of APS in cervical cancer has
focused primarily on enhancing chemosensitivity, suppressing
proliferation and inducing programmed cell death through pathways
involving PPARD, CDC20 and Wnt/β-catenin signaling (71). Research on AST, largely conducted in
SiHa cervical cancer cells and macrophage models, has emphasized
regulation of MAPK/PI3K signaling and the TGF-β/Smad2/3 axis
(72,74,76).
Reported mechanisms include inhibition of EMT, suppression of
invasion and migration, and induction of autophagy (72,74,76),
as summarized in Fig. 4.
In summary, the antitumor mechanisms of APS across
breast, ovarian and cervical cancers are most consistently linked
to immunomodulation and apoptosis induction, with frequent
involvement of JNK1/2, Wnt/β-catenin and PINK1/Parkin signaling, as
well as downstream effectors including TNF-α, IL-6, IL-1β, NO,
Bcl-2 and Bax. By contrast, the predominant actions of AST involve
reversal of drug resistance, enhancement of chemosensitivity, and
inhibition of invasion and metastasis. These effects are largely
mediated through the PI3K/Akt/mTOR, TGF-β/Smad, eNOS/NO/ONOO and
Rac1/MAPK pathways, highlighting the multitarget antitumor
potential of Astragalus bioactive constituents in
gynecological malignancies, as summarized in Table I.
 | Table I.Mechanism of Astragalus
polysaccharides and astragalosides in gynecological tumors and
breast cancer. |
Table I.
Mechanism of Astragalus
polysaccharides and astragalosides in gynecological tumors and
breast cancer.
| A, Breast cancer
(treated with APS) |
|---|
|
|---|
| Experimental models
(animal/cell) | Possible
mechanisms | Targets | Doses | (Refs.) |
|---|
| Cell: MCF-7,
RAW264.7 | Induce apoptosis in
tumor cells and inhibit tumor cells proliferation | NO↑, TNF-α↑ | 1,000 µg/ml | (41) |
| Animal: BALB/c mice
Cell: GFP-4T1 | Induce programmed
cell death in tumor cells and inhibit tumor cells
proliferation | ANXA1 mRNA↑,
ANXA1↑, EGFR mRNA↓, EGFR↓ | 200 mg/kg | (42) |
| Cell: MCF-7 | Induce programmed
cell death in tumor cells | NA | 20 mg/ml | (43) |
| Cell: TNBC 4T1
Animal: BALB/c mice | Synergistic
cisplatin, induce apoptosis in tumor cells | Bcl2↓, Bcl-xL↓,
Puma↑, Cle-Caspase3↑, Cle-PARP↑ | 2.5 mM, 100
mg/kg | (44) |
| Cell: MCF7,
Mda-MB-231 | Inhibit tumor cells
proliferation, invasion, and migration | Snail↓, vimentin↓,
Wnt1↓, β-catenin↓ | 800 µg/ml | (46) |
| Cell: RAW264.7 | Regulate
immunity | NO↑, TNF-α↑,
IL-1β↑, IL-6↑ | 400 µg/ml | (47) |
| Cell: 4T1 mouse
breast cancer cell lines, RAW 264.7 Animal: BALB/c mice | Synergistic
paclitaxel, regulate immunity | P-HA↓, PARP↓,
Checkpoint kinase 1↓, p53↓, p21↓, Bcl-Xl↑, Mcl-1↑ | 50 µg/ml, 40
mg/kg | (48) |
|
| B, Breast cancer
(treated with AST) |
|
| Experimental
models (animal/cell) | Possible
mechanisms | Targets | Doses | (Refs.) |
|
| Animal: Male
C57BL/6 mice, Male Nrf2-/- C57BL/6 mice | Enhance the efflux
activity of breast cancer resistance protein | Nrf2↑, ATP↑ | 150 mg/kg, 25
µM | (51) |
| Cell: HepG2 |
|
|
|
|
| Cell:
MCF7-CSCs48 | Synergistic
paclitaxel, reduce the | NA | 60 nM, | (52) |
| Animal:
5-week-old | stemness of breast
cancer stem |
| 40 mg/kg |
|
| Balb/c-nu/nu female
mice | cells |
|
|
|
| Cell: MCF-7,
MDA-MB-231 | Enhance sensitivity
to the | CAV-1↓, eNOS↑,
NO↑, | 30 µM | (54) |
|
| chemotherapy drug
paclitaxel | ONOO↑ |
|
|
| Animal: BALB/c
mice | Synergistically
enhance the | CAF↓, TIL↑ | 1.5 mg/kg | (55) |
| Cell: 4T1-luc,
CTLL-2 | antitumor efficacy
of programmed |
|
|
|
|
| cell death
protein-1 inhibitors |
|
|
|
| Cell: THP-1 | Inhibit tumor cells
proliferation, | TGF-β↓, IL-10↓,
MMP-9↓, | 50 µM | (56) |
|
| invasion, and
migration | CD206↓, CD163↓ |
|
|
| Cell:
MDA-MB-231 | Inhibit tumor cells
invasion and | ERK1/2↓, JNK↓,
MMP-2↓, | 40 µg/ml | (58) |
|
| migration | MMP-9↓, Vav3↓,
Rac1↓ |
|
|
| Cell: MCF-7,
MDAMB-231 | Inhibit tumor cells
proliferation, | TRHDE-AS1↑ | 80 µg/ml, | (59) |
| Animal: BALB/c
mice | invasion, and
migration |
| 20 mg/kg |
|
| Cell:
MDA-MB-231, | Inhibit tumor cells
proliferation | FOXA1↑,
GAL3ST1↑ | 60 µM | (60) |
| MDA-MB-468,
MCF-10A | and viability |
|
|
|
|
| C, Ovarian
cancer (treated with APS) |
|
| Experimental
models (animal/cell) | Possible
mechanisms | Targets | Doses | (Refs.) |
|
| Cell: OV-90,
SKOV-3, HEK 293T | Induce apoptosis in
tumor cells and inhibit tumor cells proliferation | miR-27a↓, FBXW7↑,
caspase-3↑, Bax↑ | 1 mg/ml | (62) |
| Cell: SKOV3 | Inhibit tumor cells
viability, promote tumor cells apoptosis, and enhance sensitivity
to the chemotherapy drug cisplatin | Bcl-2↓, Bax↑,
Caspase-3↑ | 800 µg/ml | (63) |
| Cell: 3AO,
SKOV3 | Induce tumor cells
apoptosis and enhance sensitivity to PARPi | PINK1↓ | 200 mg/l | (65) |
| Cell: PBMC,
A2780 | Regulate
immunity | Treg↓, IL-10↓,
TGF-β↓, VEGF-A↓, IL-6↑ | 1,000 µg/ml | (66) |
| Cell: ID8,
RAW264.7, SKOV3 | Regulate
immunity | TNF-α↑, IL-12p40↑,
IL-4↓, | 30 µM, | (67) |
| Animal: Female
C57BL/6 mice |
| TGF-β1↓ | 80 mg/kg |
|
|
| D, Ovarian
cancer (treated with AST) |
|
| Experimental
models (animal/cell) | Possible
mechanisms | Targets | Doses | (Refs.) |
|
| Cell: A2780,
SKOV3 | Promote tumor cells
apoptosis, inhibit tumor cells proliferation, and enhance
sensitivity to the chemotherapy drug cisplatin | MDR1↓, Cyclin D1↓,
PCNA↓, PARP↑, Caspase-3↑ | 160 µM, 320 µM | (68) |
| Cell: THP-1,
SKOV3 | Inhibit tumor cells
proliferation, invasion and migration | Box1↓, TLR4↓,
TGF-β↓, MMP-9↓, IL-10↓ | 100 µg/ml | (69) |
| Cell: SKOV-3 | Reduce tumor cells
viability | NA | 100 µg/ml | (70) |
|
| E, Cervical
cancer (treated with APS) |
|
| Experimental
models (animal/cell) | Possible
mechanisms | Targets | Doses | (Refs.) |
|
| Cell: Hela, SiHa,
HCerEpics | Promote tumor cells
apoptosis, inhibit tumor cells proliferation, and enhance
sensitivity to the chemotherapy drug cisplatin. | PPARD↑, CDC20↓,
Wnt↓, β-catenin↓ | 3 mg/ml | (71) |
|
| F, Cervical
cancer (treated with AST) |
|
| Experimental
models (animal/cell) | Possible
mechanisms | Targets | Doses | (Refs.) |
|
| Cell: SiHa,
HeLa | Inhibit tumor cells
proliferation, invasion and migration | Atg12↑, DCP1A↑,
TMSB4X↑ | 25 µM | (72) |
| Cell: SiHa | Inhibit tumor cells
invasion and migration | TGF-β1↓, E-cadherin
↑, MAPK↓, PI3K↓ | 800 µg/ml | (74) |
| Cell: THP-1, HeLa,
SiHa, HUVECs | Inhibit macrophage
M2 polarization, inhibit tumor cells migration, inhibit
angiogenesis | TGF-β↓, p-Smad2↓,
p-Smad3↓ | 20 µmol/l, 40
µmol/l | (76) |
Discussion
APS and AST show therapeutic potential against
breast, ovarian and cervical cancers by acting on diverse molecular
targets and signaling pathways. The present review systematically
synthesizes the antitumor mechanisms of APS and AST, the major
bioactive constituents of Astragalus membranaceus, in
gynecological malignancies and delineates their key targets and
regulatory networks. These insights provide a mechanistic rationale
for the clinical use of Astragalus as an adjuvant therapy in
breast, ovarian and cervical cancers. Notably, because intratumoral
heterogeneity is substantial across malignancies, the biological
effects of Astragalus constituents are likely to vary among
tumor subtypes. In clinical practice, breast cancer is commonly
classified according to molecular features and biological behavior
(77) into Luminal A, Luminal B,
HER2-enriched and TNBC (78). The
studies included in the current review placed particular emphasis
on TNBC, where APS and AST most consistently suppress tumor cell
proliferation and promote apoptosis, thereby offering mechanistic
support and new directions for therapeutic development in this
refractory subtype. By contrast, subtype-specific evidence in
ovarian and cervical cancers remains limited. A number of studies
have treated these malignancies as single disease entities in
mechanistic investigations, and the present review reflects the
current evidence base. This gap highlights the need for future
research integrating refined pathological and molecular subtyping
to better define subtype dependent responses and therapeutic
potential.
Emerging evidence also indicates that the
pharmacological actions of Astragalus extend beyond
gynecological oncology to common non-neoplastic disorders of the
female reproductive system. For example, AS-IV has been reported to
activate peroxisome proliferator-activated receptor-γ (PPARγ),
induce autophagy, suppress granulosa cell proliferation and promote
apoptosis, suggesting potential relevance to polycystic ovary
syndrome (PCOS) (79). Similarly,
APS ameliorates PCOS-like phenotypes in mice by improving serum
metabolic profiles, increasing gut microbiome diversity, and
reducing insulin resistance and oxidative stress (80). In addition, aqueous extracts of
Astragalus roots, in which AST are major constituents,
upregulated uterine and ovarian PPARα mRNA and increased
mitochondrial 2,4-dienoyl-CoA reductase expression, thereby
suppressing estrogen-dependent uterine hyperplasia (81). APS has also been shown to alleviate
bovine mammary fibrosis through reduction of reactive oxygen
species, inhibition of NLRP3 and modulation of EMT (82). Collectively, these findings suggest
that Astragalus bioactive compounds exert broad regulatory
effects across female reproductive disorders and may help maintain
reproductive axis homeostasis through multi-target and multi-level
mechanisms.
Despite encouraging preclinical evidence, important
challenges remain for the clinical translation of APS and AST in
gynecological oncology. Current knowledge of their in vivo
metabolism, bioavailability and interactions with the tumor
microenvironment remains incomplete, and the mechanisms that
determine efficacy across distinct subtypes of female malignancies
require further clarification. Although available studies suggest
that APS and AST can influence drug disposition and therapeutic
response by regulating metabolic enzymes and transporters such as
BCRP and MDR1, systematic characterization of absorption,
distribution, metabolism and excretion profiles remains
insufficient. Clinical development is further constrained by poor
water solubility, low oral bioavailability and suboptimal tissue
distribution, which limit accumulation at tumor sites. Advanced
delivery platforms including lipid based carriers, polymeric
micelles and nanoparticle formulations may improve bioavailability,
enable controlled release and reduce adverse effects, yet barriers
related to formulation complexity, large scale manufacturing and
safety evaluation continue to impede translation (83). In addition, chemotherapy remains a
cornerstone of treatment for breast, ovarian and cervical cancers,
but its effectiveness is frequently limited by resistance and
dose-limiting toxicities that reduce adherence and quality of life.
Building on the immunomodulatory and cytoprotective properties of
APS and AST, future studies should develop natural immunomodulatory
adjuvants with improved bioactivity and tumor targeting capacity to
mitigate chemotherapy-related toxicities and enhance therapeutic
outcomes. Notably, most existing evidence is derived from cellular
and animal models, and high quality clinical data remain scarce
regarding long-term efficacy, optimal dosing and rational
combination strategies. Future work should prioritize
multidisciplinary collaboration to clarify molecular mechanisms,
optimize delivery technologies, conduct rigorous clinical trials
and establish standardized quality control systems, thereby
accelerating clinical translation and maximizing the therapeutic
potential of Astragalus in gynecological malignancies.
Acknowledgements
Not applicable.
Funding
This work was supported by the Shandong Province Collaborative
Innovation Center for Classic Traditional Chinese Medicine Formulas
(grant no. 2019KFY12) and the National College Students' Innovation
and Entrepreneurship Training Program (grant no. 202510441006).
Availability of data and materials
Not applicable.
Authors' contributions
RL wrote the manuscript and drew the pictures. ZL
and YL collected and organized literature and participated in the
revision of the manuscript. YC and ZG proofread the manuscript. ZX
is responsible for the study designing, research fields, drafting
and finalizing the manuscript. All authors read and approved the
final version of the manuscript. Data authentication is not
applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Jha AK, Mithun S, Sherkhane UB, Jaiswar V,
Osong B, Purandare N, Kannan S, Prabhash K, Gupta S, Vanneste B, et
al: Systematic review and meta-analysis of prediction models used
in cervical cancer. Artif Intell Med. 139:1025492023. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Olusola P, Banerjee HN, Philley JV and
Dasgupta S: Human papilloma virus-associated cervical cancer and
health disparities. Cells. 8:6222019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Gupta KK, Gupta VK and Naumann RW: Ovarian
cancer: Screening and future directions. Int J Gynecol Cancer.
29:195–200. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kossaï M, Leary A, Scoazec JY and Genestie
C: Ovarian cancer: A heterogeneous disease. Pathobiology. 85:41–49.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Menon U, Karpinskyj C and Gentry-Maharaj
A: Ovarian cancer prevention and screening. Obstet Gynecol.
131:909–927. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Xiong X, Zheng LW, Ding Y, Chen YF, Cai
YW, Wang LP, Huang L, Liu CC, Shao ZM and Yu KD: Breast cancer:
Pathogenesis and treatments. Signal Transduct Target Ther.
10:492025. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Anastasiadi Z, Lianos GD, Ignatiadou E,
Harissis HV and Mitsis M: Breast cancer in young women: An
overview. Updates Surg. 69:313–317. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Jabbour HN, Sales KJ, Catalano RD and
Norman JE: Inflammatory pathways in female reproductive health and
disease. Reproduction. 138:903–919. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Rayburn WF: Gynecologic cancer care:
Innovative progress. Obstet Gynecol Clin North Am. 46:xiii–xiv.
2019. View Article : Google Scholar
|
|
10
|
Li X, Qu L, Dong Y, Han L, Liu E, Fang S,
Zhang Y and Wang T: A review of recent research progress on the
astragalus genus. Molecules. 19:18850–18880. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ma XQ, Shi Q, Duan JA, Dong TT and Tsim
KW: Chemical analysis of radix astragali (Huangqi) in China: A
comparison with its adulterants and seasonal variations. J Agric
Food Chem. 50:4861–4866. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Auyeung KK, Han QB and Ko JK: Astragalus
membranaceus: A review of its protection against inflammation and
gastrointestinal cancers. Am J Chin Med. 44:1–22. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tian J, Huo R, Wang Y, Wang J, Fang F and
Fang C: Astragalus polysaccharide alleviates cognitive decline in
D-galactose-induced aging. Biol Pharm Bull. 48:523–536. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wang X, Liu Y, Peng N, Yu H, Ma Y, Zhang
M, Wang Y, Wang Y and Gao W: Allelopathy and identification of
volatile components from the roots and aerial parts of astragalus
mongholicus Bunge. Plants (Basel). 13:3172024.PubMed/NCBI
|
|
15
|
Li J, Niu Y, Yuan L, Jiang W, Jiao T, Dou
H and Nan Y: Research progress in the medicine-food dual use of
astragalus for gastrointestinal tumors. J Med Food. 27:1145–1157.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Li S, Sun Y, Huang J, Wang B, Gong Y, Fang
Y, Liu Y, Wang S, Guo Y, Wang H, et al: Anti-tumor effects and
mechanisms of Astragalus membranaceus (AM) and its specific
immunopotentiation: Status and prospect. J Ethnopharmacol.
258:1127972020. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Tan B, Li Z, Chen S, Wan Q, Jiang C, Fu Y,
Su W and Wan X: Advances in molecular mechanisms of anti-tumor
bioactive constituents from Astragalus membranaceus: A
comprehensive review. J Ethnopharmacol. 356:1207442026. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Dong M, Li J, Yang D, Li M and Wei J:
Biosynthesis and pharmacological activities of flavonoids,
triterpene saponins and polysaccharides derived from astragalus
membranaceus. Molecules. 28:50182023. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Nafti K, Giacinti G, Marghali S and
Raynaud CD: Screening for astragalus hamosus triterpenoid saponins
using HPTLC methods: Prior identification of Azukisaponin isomers.
Molecules. 27:53762022. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tian H, An L, Wang P, Zhang X, Gao W and
Li X: Review of Astragalus membranaceus polysaccharides: Extraction
process, structural features, bioactivities and applications. Chin
Herb Med. 17:56–69. 2025.PubMed/NCBI
|
|
21
|
Tin MMY, Cho CH, Chan K, James AE and Ko
JK: Astragalus saponins induce growth inhibition and apoptosis in
human colon cancer cells and tumor xenograft. Carcinogenesis.
28:1347–1355. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Li W, Hu X, Wang S, Jiao Z, Sun T, Liu T
and Song K: Characterization and anti-tumor bioactivity of
astragalus polysaccharides by immunomodulation. Int J Biol
Macromol. 145:985–997. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Yan X, Miao J, Zhang B, Liu H, Ma H, Sun
Y, Liu P, Zhang X, Wang R, Kan J, et al: Study on semi-bionic
extraction of Astragalus polysaccharide and its anti-aging activity
in vivo. Front Nutr. 10:12019192023. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zeng P, Li J, Chen Y and Zhang L: The
structures and biological functions of polysaccharides from
traditional Chinese herbs. Prog Mol Biol Transl Sci. 163:423–444.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zheng Y, Ren W, Zhang L, Zhang Y, Liu D
and Liu Y: A review of the pharmacological action of astragalus
polysaccharide. Front Pharmacol. 11:3492020. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Tang Z and Tian X: Astragalus
membranaceus: A Traditional Chinese medicine with multifaceted
impacts on breast cancer treatment. Biomolecules. 14:13392024.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang S, Long S, Deng Z and Wu W: Positive
role of Chinese herbal medicine in cancer immune regulation. Am J
Chin Med. 48:1577–1592. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Qin ZT, Wu ZH, Wang CM, Xie XC and Wang
YH: Astragalus polysaccharide as a potential antitumor
immunomodulatory drug (Review). Mol Med Rep. 32:3412025. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Duya PA, Chen Y, Bai L, Li Z, Li J, Chai
R, Bian Y and Zhao S: Nature products of traditional Chinese
medicine provide new ideas in γδT cell for tumor immunotherapy.
Acupuncture and Herbal Medicine. 2:78–83. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Li Q, Li J, Wang Y, Wu F and Li T:
Efficacy and safety of astragalus polysaccharides in patients with
malignant tumors: A systematic review and meta-analysis. Naunyn
Schmiedebergs Arch Pharmacol. 398:11705–11732. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Shen L, Gwak SR, Cui ZY, Joo JC and Park
SJ: Astragalus-Containing Chinese herbal medicine combined with
chemotherapy for cervical cancer: A systematic review and
meta-analysis. Front Pharmacol. 12:5870212021. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Sheng X, Yang L, Huang B, Lin G, Wang Y,
Wu X and Lin R: Efficacy of Astragalus Membranaceus (Huang Qi) for
cancer-related fatigue: A systematic review and meta-analysis of
randomized controlled studies. Integr Cancer Ther.
24:153473542413133442025. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Chen G, Jiang N, Zheng J, Hu H, Yang H,
Lin A, Hu B and Liu H: Structural characterization and
anti-inflammatory activity of polysaccharides from Astragalus
membranaceus. Int J Biol Macromol. 241:1243862023. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Shahzad M, Shabbir A, Wojcikowski K,
Wohlmuth H and Gobe GC: The antioxidant effects of Radix Astragali
(Astragalus membranaceus and Related Species) in protecting tissues
from injury and disease. Curr Drug Targets. 17:1331–1340. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Li CX, Liu Y, Zhang YZ, Li JC and Lai J:
Astragalus polysaccharide: A review of its immunomodulatory effect.
Arch Pharm Res. 45:367–389. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zhang C, Guo W, Yao X, Xia J, Zhang Z, Li
J, Chen H and Lin L: Database mining and animal experiment-based
validation of the efficacy and mechanism of Radix Astragali
(Huangqi) and Rhizoma Atractylodis Macrocephalae (Baizhu) as core
drugs of Traditional Chinese medicine in cancer-related fatigue. J
Ethnopharmacol. 285:1148922022. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Huang WC, Kuo KT, Bamodu OA, Lin YK, Wang
CH, Lee KY, Wang LS, Yeh CT and Tsai JT: Astragalus polysaccharide
(PG2) ameliorates cancer symptom clusters, as well as improves
quality of life in patients with metastatic disease, through
modulation of the inflammatory cascade. Cancers (Basel).
11:10542019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Chen MH, May BH, Zhou IW, Zhang AL and Xue
CC: Integrative medicine for relief of nausea and vomiting in the
treatment of colorectal cancer using oxaliplatin-based
chemotherapy: A systematic review and meta-analysis. Phytother Res.
30:741–753. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Zhang X, Qiu H, Li C, Cai P and Qi F: The
positive role of traditional Chinese medicine as an adjunctive
therapy for cancer. Biosci Trends. 15:283–298. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Liu J, Hong M, Li Y, Chen D, Wu Y and Hu
Y: Programmed cell death tunes tumor immunity. Front Immunol.
13:8473452022. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Li W, Song K, Wang S, Zhang C, Zhuang M,
Wang Y and Liu T: Anti-tumor potential of astragalus
polysaccharides on breast cancer cell line mediated by macrophage
activation. Mater Sci Eng C Mater Biol Appl. 98:685–695. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Li W, Hu X, Li Y and Song K: Cytotoxicity
and growth-inhibiting activity of Astragalus polysaccharides
against breast cancer via the regulation of EGFR and ANXA1. J Nat
Med. 75:854–870. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Wang B, Guo C, Liu Y, Han G, Li Y, Zhang
Y, Xu H and Chen D: Novel nano-pomegranates based on astragalus
polysaccharides for targeting ERα-positive breast cancer and
multidrug resistance. Drug Deliv. 27:607–621. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Sun LI, Zhuo S, Li X, Kong H, DU W, Zhou C
and Huang J: Astragalus polysaccharide enhances the therapeutic
efficacy of cisplatin in triple-negative breast cancer through
multiple mechanisms. Oncol Res. 33:641–651. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Al Kassam D, Marcos CA, Blanco I, de Los
Toyos JR and Llorente JL: Diagnostic value of E-cadherin, MMP-9,
activated MMP-13 and anti-p53 antibodies in squamous cell
carcinomas of head and neck. Med Clín (Barc). 129:761–765. 2007.(In
Spanish). View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Yang S, Sun S, Xu W, Yu B, Wang G and Wang
H: Astragalus polysaccharide inhibits breast cancer cell migration
and invasion by regulating epithelial-mesenchymal transition via
the Wnt/β-catenin signaling pathway. Mol Med Rep. 21:1819–1832.
2020.PubMed/NCBI
|
|
47
|
Zhou L, Liu Z, Wang Z, Yu S, Long T, Zhou
X and Bao Y: Astragalus polysaccharides exerts immunomodulatory
effects via TLR4-mediated MyD88-dependent signaling pathway in
vitro and in vivo. Sci Rep. 7:448222017. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Bao WR, Li ZP, Zhang QW, Li LF, Liu HB, Ma
DL, Leung CH, Lu AP, Bian ZX and Han Q: Astragalus Polysaccharide
RAP selectively attenuates paclitaxel-induced cytotoxicity toward
RAW 264.7 cells by reversing cell cycle arrest and apoptosis. Front
Pharmacol. 9:15802019. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Sheng F, Yang S, Li M, Wang J, Liu L and
Zhang L: Research progress on the anti-cancer effects of astragalus
membranaceus saponins and their mechanisms of action. Molecules.
29:33882024. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Lin-Rahardja K, Scarborough J and Scott
JG: Evolution-informed strategies for combating drug resistance in
cancer. Int J Mol Sci. 24:67382023. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Lou Y, Guo Z, Zhu Y, Zhang G, Wang Y, Qi
X, Lu L, Liu Z and Wu J: Astragali radix and its main bioactive
compounds activate the Nrf2-mediated signaling pathway to induce
P-glycoprotein and breast cancer resistance protein. J
Ethnopharmacol. 228:82–91. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Huang P, Li H, Ren L, Xie H, Chen L, Liang
Y, Hu Y, Selistre-de-Araujo HS, Boussios S, Jhawar SR, et al:
Astragaloside IV enhances the sensitivity of breast cancer stem
cells to paclitaxel by inhibiting stemness. Transl Cancer Res.
12:3703–3717. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Wang Y, Li Y, Zhong J, Li M, Zhou Y, Lin
Q, Zong S, Luo W, Wang J, Wang K, et al: Tumor-derived Cav-1
promotes pre-metastatic niche formation and lung metastasis in
breast cancer. Theranostics. 13:1684–1697. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Zheng Y, Dai Y, Liu W, Wang N, Cai Y, Wang
S, Zhang F, Liu P, Chen Q and Wang Z: Astragaloside IV enhances
taxol chemosensitivity of breast cancer via caveolin-1-targeting
oxidant damage. J Cell Physiol. 234:4277–4290. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Wei L, Wang H, Ye X, Yue J, Guo H, Mao D,
Li X, Sun Y, Liu C, Liu Y and Chen Y: Oxymatrine and astragaloside
IV co-loaded liposomes: Scale-up purposes and their enhancement of
anti-PD-1 efficacy against breast cancer. Mater Today Bio.
32:1016342025. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Yu Y, Hao J, Wang L, Zheng X, Xie C, Liu
H, Wu J, Qiao S and Shi J: Astragaloside IV antagonizes the
malignant progression of breast cancer induced by macrophage M2
polarization through the TGF-β-regulated Akt/Foxo1 pathway. Pathol
Res Pract. 249:1547662023. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Al-Hawary SIS, Altalbawy FMA, Jasim SA,
Jyothi SR, Jamal A, Naiyer MM, Mahajan S, Kalra H, Jawad MA and
Zwamel AH: VAV3 in human cancers: Mechanism and clinical
implication. Pathol Res Pract. 248:1546812023. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Jiang K, Lu Q, Li Q, Ji Y, Chen W and Xue
X: Astragaloside IV inhibits breast cancer cell invasion by
suppressing Vav3 mediated Rac1/MAPK signaling. Int Immunopharmacol.
42:195–202. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Hu S, Zheng W and Jin L: Astragaloside IV
inhibits cell proliferation and metastasis of breast cancer via
promoting the long noncoding RNA TRHDE-AS1. J Nat Med. 75:156–166.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Yang Y, Lu J, Zhu Y, Chen D, Tang J, Zhang
M, Lu J, Yang Y, Tian S and Zhao H: Astragaloside IV inhibits the
growth of obesity-associated triple-negative breast cancer by
activating FOXA1 transcription factor to regulate GAL3ST1-GalCer
signaling and remodel sphingolipid metabolism. Phytomedicine.
144:1569072025. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Li X, Gong C, Naeem A, Liu J, Yang M,
Zhang J and Shang H: Priming immunity via herbal components and
their nanomedicines for the treatment of cancer. Acupuncture and
Herbal Medicine. 4:436–462. 2024. View Article : Google Scholar
|
|
62
|
Guo Y, Zhang Z, Wang Z, Liu G, Liu Y and
Wang H: Astragalus polysaccharides inhibit ovarian cancer cell
growth via microRNA-27a/FBXW7 signaling pathway. Biosci Rep.
40:BSR201933962020. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Li C, Hong L, Liu C, Min J, Hu M and Guo
W: Astragalus polysaccharides increase the sensitivity of SKOV3
cells to cisplatin. Arch Gynecol Obstet. 297:381–386. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Najafi M, Mortezaee K and Majidpoor J:
Cancer stem cell (CSC) resistance drivers. Life Sci.
234:1167812019. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Peng Q, Yu Y, Ye L, Zhang S, Li Y, Hua X,
Shen S, Hu D and Lu W: Astragalus polysaccharides sensitize ovarian
cancer stem cells to PARPi by inhibiting mitophagy via PINK1/Parkin
signaling. iScience. 27:1103762024. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Shokati E, Motalebnezhad M and Safari E:
Astragalus polysaccharide mediates immunomodulatory effects on
crosstalk between human peripheral blood mononuclear cells and
ovarian cancer cell line. Iran J Allergy Asthma Immunol.
22:172–182. 2023.PubMed/NCBI
|
|
67
|
Liu M, Zhang ZX, Wang JH, Guo RB, Zhang L,
Kong L, Yu Y, Zang J, Liu Y and Li XT: Immunomodulatory and
anti-ovarian cancer effects of novel astragalus polysaccharide
micelles loaded with podophyllotoxin. Int J Biol Macromol.
290:1389602025. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Zhang L, Liu Y, Lei X, Liu X, Sun H and
Liu S: Astragaloside II enhanced sensitivity of ovarian cancer
cells to cisplatin via triggering apoptosis and autophagy. Cell
Biol Int. 47:1600–1613. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Wang X, Gao S, Song L, Liu M, Sun Z and
Liu J: Astragaloside IV antagonizes M2 phenotype macrophage
polarization-evoked ovarian cancer cell malignant progression by
suppressing the HMGB1-TLR4 axis. Mol Immunol. 130:113–121. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Dekinash MF, Okda TM, Elmahallawy EK,
El-Fiky FK, Omran GAEH, Svajdlenka E, Dahran N, El-Khadragy MF,
Al-Megrin WA and El Naggar EMBA: Insights into HPLC-MS/MS analysis,
antioxidant and cytotoxic activity of astragalus fruticosus against
different types of cancer cell lines. Pharmaceuticals (Basel).
15:14062022. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Liu W, Fu Y, Jiang X, Tan J, Zhang Y and
Zhang L: Astragalus polysaccharide regulates the cervical cancer
cell cycle and inhibits cisplatin resistance by blocking the
Wnt/β-catenin pathway through the PPARD/CDC20 axis. Cytotechnology.
77:1262025. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Xia C, He Z and Cai Y: Quantitative
proteomics analysis of differentially expressed proteins induced by
astragaloside IV in cervical cancer cell invasion. Cell Mol Biol
Lett. 25:252020. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Lu W and Kang Y: Epithelial-mesenchymal
plasticity in cancer progression and metastasis. Dev Cell.
49:361–374. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Zhang L, Zhou J, Qin X, Huang H and Nie C:
Astragaloside IV inhibits the invasion and metastasis of SiHa
cervical cancer cells via the TGF-β1-mediated PI3K and MAPK
pathways. Oncol Rep. 41:2975–2986. 2019.PubMed/NCBI
|
|
75
|
Boutilier AJ and Elsawa SF: Macrophage
polarization states in the tumor microenvironment. Int J Mol Sci.
22:69952021. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Shen L, Li Y, Hu G, Song X, Wang X, Li X
and Xu X: Astragaloside IV suppresses the migration and EMT
progression of cervical cancer cells by inhibiting macrophage M2
polarization through TGFβ/Smad2/3 signaling. Funct Integr Genomics.
23:1332023. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Waks AG and Winer EP: Breast cancer
treatment: A review. JAMA. 321:288–300. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Holliday DL and Speirs V: Choosing the
right cell line for breast cancer research. Breast Cancer Res.
13:2152011. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Wen M, Chen W, Zhou Q and Dou X:
Astragaloside IV regulates autophagy-mediated proliferation and
apoptosis in a rat model of PCOS by activating the PPARγ pathway.
Iran J Basic Med Sci. 25:882–889. 2022.PubMed/NCBI
|
|
80
|
Li R, Hu R, Huang Y, Li D, Ma X and Yang
Y: Astragalus polysaccharide alleviates polycystic ovary syndrome
by reducing insulin resistance and oxidative stress and increasing
the diversity of gut microbiota. Endocrine. 83:783–797. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Orkhon B, Kobayashi K, Javzan B and Sasaki
K: Astragalus root induces ovarian β-oxidation and suppresses
estrogen-dependent uterine proliferation. Mol Med Rep.
18:5198–5206. 2018.PubMed/NCBI
|
|
82
|
Zhang J, Liu K, Yang T, Duan H, Xiao L,
Zhang Q, Zhang Y, Dong W and Zhao X: Mechanism of Astragalus
Polysaccharide in alleviating bovine mammary fibrosis through
ROS/NLRP3 inhibition and EMT regulation. Antioxidants (Basel).
14:5032025. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Prakash S: Nano-based drug delivery system
for therapeutics: A comprehensive review. Biomed Phys Eng Express.
9:10.1088/2057–1976/acedb2. 2023. View Article : Google Scholar : PubMed/NCBI
|