Introduction
Esophageal cancer (EC) is the seventh leading cause
of cancer-related deaths worldwide and is mainly classified as
esophageal squamous cell carcinoma (ESCC) and adenocarcinoma
(1). Advances in endoscopic
techniques have improved the accuracy of tumor staging; however,
even when tumors are confined to the mucosa, lymph node metastasis
occurs in 10–15% of patients with tumor invasion of the muscularis
mucosae (2). Consequently, EC is
associated with a poor prognosis, with a 5-year survival rate
<20% (2). Despite advances in
therapy, patient outcomes remain unsatisfactory, and EC continues
to pose a significant challenge to global health. This highlights
the need for improved diagnostic methods and treatment strategies
for EC.
EC treatment primarily involves surgery, including
endoscopic or surgical resection, often combined with chemotherapy.
For ESCC, the Japan Clinical Oncology Group (JCOG) trials have
shown improved overall survival (OS) with neoadjuvant chemotherapy
(NAC) using cisplatin and 5-fluorouracil (5-FU) (CF) in locally
advanced ESCC (JCOG9907) (3) and
further OS benefit when docetaxel is added to CF (DCF) (JCOG1109)
(4). However, key agents such as
5-FU can cause severe toxicities, and prolonged 5-FU administration
can promote resistance through complex, multifactorial mechanisms
involving altered DNA damage responses and signaling pathway
activities (5–7), resulting in reduced efficacy against
ESCC. Moreover, the high cost of treatment further complicates the
balance between efficacy, safety and cost.
Given their low toxicity and cost-effectiveness,
complementary and alternative medicines, especially dietary
compounds, have emerged as compelling areas of study. Andrographis,
a C20-diterpenoid lactone derived from Andrographis
paniculata and a component of traditional Indian medicine, has
been used historically to treat fever, infections and
gastrointestinal disorders (8–10).
Because Andrographis is structurally stable in circulation
(11–13), it exhibits anti-inflammatory,
immunomodulatory and antitumor activities. Its antitumor effects
have been demonstrated in gastric, colorectal and pancreatic
adenocarcinomas, suggesting broad efficacy across gastrointestinal
cancers (14–18). Importantly, the authors' previous
study focused on ferroptosis. Ferroptosis, an iron-dependent, lipid
peroxidation-driven regulated cell death pathway (19–21),
has emerged as a potential mechanism, suggesting that Andrographis
may exert antitumor effects through ferroptosis in adenocarcinomas.
However, evidence in squamous cell carcinomas (SCCs) is scarce
(22,23), and whether Andrographis activates
ferroptosis in ESCC remains unclear.
Although the anticancer effects of andrographolide
in ESCC have been described previously, the potential modulation of
ferroptosis-related transcriptional programs in ESCC, particularly
under combination treatment with 5-FU, has not been investigated.
Therefore, the present study aimed to evaluate the antitumor effect
of Andrographis in ESCC and to determine whether combination
treatment with 5-FU modulates ferroptosis-associated gene
expression and redox-related pathways.
Materials and methods
Cell culture and materials
The EC cell lines KYSE410 [poorly differentiated
SCC, Microsatellite Stable (MSS) and TE1 (well-differentiated SCC,
MSS)] were supplied by the Cell Resource Center of Biomedical
Research, Institute of Development, Aging, and Cancer (Tohoku
University). The identity of the cell line was confirmed by
assessing the genetic and epigenetic markers. The cultures were
periodically tested for mycoplasma contamination. Cells were
cultured in Roswell Park Memorial Institute (RPMI)-1640 medium
(Nacalai Tesque, Inc.) enriched with 10% fetal bovine serum (FBS;
Biowest) and an antibiotic-antimycotic cocktail (Nacalai Tesque,
Inc.). The cells were incubated at 37°C in a humidified incubator
with 5% CO2. Andrographis was kindly provided by
Professor Ajay Goel (City of Hope Comprehensive Cancer Center). The
compound was manufactured by EuroPharma USA. According to the
manufacturer's certificate of analysis, the preparation is a
purified extract of Andrographis paniculata and contains 80%
Andrographolide as the principal active constituent. Chemical
characterization, including the standardization of Andrographolide
content and purity confirmation, was based on the manufacturer's
certificate of analysis. No additional extraction or purification
was performed outside EuroPharma USA for the present study. For all
assays, the compound was dissolved in DMSO to prepare a stock
solution, which was subsequently diluted with the culture medium to
the indicated final concentrations.
Cell viability and proliferation
assay
For Water Soluble Tetrazolium (WST) assay, 4,000
cells/well were seeded in 96-well tissue culture plates (TPP Techno
Plastic Products AG). The cells were cultured in RPMI-1640 medium
supplemented with 10% FBS and antibiotics and allowed to adhere
overnight. Subsequently, ESCC cells were exposed to various
concentrations of 5-FU and Andrographis for 72 h. The tested doses
were 3, 3.5, 4, 4.5 and 5 µM for 5-FU, and 36, 42, 48, 54 and 60
µg/ml for Andrographis, respectively. This procedure was performed
to determine the cytotoxic effects of these compounds. Cell
proliferation was measured using WST 8 (Dojindo Laboratories, Inc.)
according to the manufacturer's instructions. Based on the
inhibitory concentration at 50% (IC50) (24), a combination of 5-FU and
Andrographis (3, 3.5, 4, 4.5 and 5 µM and 36, 42, 48, 54, and 60
µg/ml) was used to treat each cell line for 72 h. Absorbance values
were measured for each well at a wavelength of 450 nm using SoftMax
Pro microplate reader (Molecular Devices, LLC.). To maintain
consistency across the experimental conditions, an identical final
concentration of DMSO was applied to all treatment groups,
including the control group. The interaction between 5-FU and
Andrographis was quantitatively assessed by calculating the
combination index (C.I.) at the IC50 using the
Chou-Talalay method (24). C.I.
values were generated using GraphPad Prism ver. 10.0 (GraphPad
Software Inc.; Dotmatics) and a C.I. <1.0 was regarded as
suggestive of additive effects (24). Each experiment was independently
repeated three times, with technical triplicates included in each
of the biological replicates.
Cell colony formation assay
Colony formation assays were performed based on
established protocols (25) with
minor procedural adaptations. Specifically, KYSE410 and TE1 cells
were seeded in 6-well tissue culture plates (TPP Techno Plastic
Products AG) at a density of 500 cells/well, using an identical
culture medium. The plates were incubated for 24 h. A total of 1
ml/well of 5-FU (3.5 µM), Andrographis (42 µg/ml), and a
combination of 5-FU and Andrographis (3.5 µM, 42 µg/ml) were then
added for 72 h. Following the initial incubation, the medium was
replaced with fresh drug-free culture medium. The cells were then
cultured at 37°C with 5% CO2 in a humidified environment
for 7–9 days. Colonies consisting of at least 50 cells were counted
as a colony. Colony numbers were quantified using ImageJ software
ver.1.54p (National Institutes of Health) (26). These counts were then compared
between the control and drug treatment groups.
Cell apoptosis assay
Apoptosis was assessed using propidium
iodide/annexin V double staining and flow cytometry. Cells were
inoculated into a 6-well tissue culture plate at a density of
1.6×105 cells/well and pre-incubated for 24 h.
Subsequently, the cultures were treated with one of the following
treatments for 72 h: 5-FU (3.5 µM), Andrographis (42 µg/ml), or a
combination of 5-FU and Andrographis (3.5 µM, 42 µg/ml) for 72 h.
Apoptotic cells were measured using Muse® Annexin V and
Dead Cell Assay (Luminex Corporation) on a Muse™ Cell Analyzer
(Merck KGaA), following the manufacturer's instructions. Data
acquisition and analysis were performed using Muse Cell Analyzer
software (version 1.4; Merck KGaA).
Quantitative mRNA expression
analysis
To quantify mRNA expression, cells were seeded in
6-well tissue culture plates at a density of 3.0×105
cells/well. After a 24 h adherence period, the cultures were
subjected to drug treatment. Specifically, the cells were exposed
to 5-FU (3.5 µM), Andrographis (42 µg/ml), or a combination of 5-FU
and Andrographis (3.5 µM, 42 µg/ml). Total RNA was extracted from
the cells using an RNA extraction miRNeasy Mini kit (Qiagen GmbH).
Complementary DNA (cDNA) was synthesized from 5.0 ng of total RNA
using a Reverse Transcription kit (Toyobo Co., Ltd.) according to
the manufacturer's instructions. Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR) was performed using cDNA and
the Power SYBR® Green PCR Master Mix (Thermo Fisher
Scientific Inc.). RT-qPCR analysis was conducted using a StepOne™
Real-Time PCR System (Thermo Fisher Scientific, Inc.). The
thermocycling conditions were as follows: Initial denaturation at
95°C for 10 min, followed by 40 cycles of denaturation at 95°C for
15 sec and annealing/extension at 60°C for 60 sec. The primer
sequences were as follows: heme oxygenase 1 (HMOX1) forward,
5′-AAGACTGCGTTCCTGCTCAAC-3′ and reverse,
5′-AAAGCCCTACAGCAACTGTCG-3′; glutamate cysteine ligase catalytic
(GCLC) forward, 5′-AGGCCAACATGCGAAAAC-3′ and reverse,
5′-CGGATATTTCTTGTTAAGGTACTGG-3′; glutamate cysteine ligase modifier
(GCLM) forward, 5′-TTGGAGTTGCACAGCTGGAT-3′ and reverse,
5′-GGTTTTACCTGTGCCCACTGA-3′; glutathione peroxidase 4 (GPX4)
forward, 5′-GAGGCAAGACCGAAGTAAACTAC-3′ and reverse,
5′-CCGAACTGGTTACACGGGAA-3′; solute carrier family 7 member 11
(SLC7A11) forward, 5′-TCTCCAAAGGAGGTTACCTGC-3′ and reverse,
5′-AGACTCCCCTCAGTAAAGTGAC-3′; nuclear factor kappa B subunit 1
(NFKB1) forward, 5′-AACAGAGAGGATTTCGTTTCCG-3′ and reverse,
5′-TTTGACCTGAGGGTAAGACTTCT-3′; signal transducer and activator of
transcription 3 (STAT3) forward, 5′-ACCAGCAGTATAGCCGCTTC-3′ and
reverse, 5′-GCCACAATCCGGGCAATCT-3′; NFE2 like bZIP transcription
factor 2 (NFE2L2) forward, 5′-TCCAGTCAGAAACCAGTGGAT-3′ and reverse,
5′-GAATGTCTGCGCCAAAAGCTG-3′; acyl-CoA synthetase long chain family
member 4 (ACSL4) forward, 5′-AACCCAGAAAACTTGGGCATT-3′ and reverse,
5′-GTCGGCCAGTAGAACCACT-3′; and glyceraldehyde 3-phosphate
dehydrogenase (GAPDH) forward, 5′-GGAAGGTGAAGGTCGGAGTC-3′ and
reverse, 5′-AATGAAGGGGTCATTGATGG-3′. Target gene expression levels
were quantified using the 2−ΔΔCq method (27). The resulting values were
subsequently standardized to the housekeeping gene, GAPDH.
Western blotting
ESCC cells (7.5×105 cells/well) were
treated with 5-FU (3.5 µM), Andrographis (42 µg/ml), and a
combination of 5-FU and Andrographis (3.5 µM, 42 µg/ml) for 72 h.
The control group received RPMI-1640 medium enriched with 10% FBS
and an antibiotic-antimycotic cocktail. Cells were lysed in
Radioimmunoprecipitation Assay buffer (BioDynamics Laboratory,
Inc.) supplemented with a proteinase inhibitor cocktail (Sigma
Aldrich; Merck KGaA). Protein quantification was performed using a
BCA Protein Assay Kit (Thermo Fisher Scientific, Inc.).
Subsequently, the samples were combined with the loading buffer and
denatured by boiling for 5 min. Equal amounts of protein (20 µg per
lane) were loaded. The samples were subjected to electrophoresis on
4–15% gradient Mini-PROTEAN®TGX™ (Bio-Rad Laboratories,
Inc.) for 50 min and transferred to PVDF membranes using
Trans-Blot®Turbo™ (Bio-Rad Laboratories, Inc.) with EB
RAPID for 5 min. The membranes were initially incubated with 5%
milk for 60 min at room temperature for blocking purposes. The
membranes were then exposed to the indicated primary antibodies for
60 min at room temperature. mouse monoclonal anti HMOX1 (1:1,000;
cat. no. sc-136960), mouse monoclonal anti-γ-GCLC (1:2,000; cat.
no. sc-390811) and mouse monoclonal anti-γ-GCLM (1:5,000; cat. no.
sc-55586; all from Santa Cruz Biotechnology, Inc.). The membranes
were then washed three times with Tris buffer saline containing
0.05% Tween 20 at room temperature. Following washing, the
membranes were probed with anti-mouse IgG (cat. no. W4028; Promega
Corporation) as the secondary antibody for 30 min at room
temperature. The secondary antibody was applied at the following
dilutions: 1:10,000 for HMOX1, and 1:20,000 for γ-GCLC, and
1:20,000 for γ-GCLM. Mouse monoclonal β-actin antibody (691001; MP
Biomedicals, LLC) was used as the loading control. Protein signals
were visualized using a chemiluminescent imaging system (ATTO
Corporation) after incubation with Immobilon® Western
(MilliporeSigma).
Statistical analysis
Statistical analyses were performed for each
experiment as follows: Cell viability and proliferation assay: For
comparisons across multiple treatment groups, one-way ANOVA
followed by Tukey's post hoc test was performed. Differences among
multiple treatment conditions were analyzed using one-way ANOVA
with Tukey's post hoc test regarding ell colony formation assays.
Percentages of apoptotic cells in cell apoptosis assay were
compared using one-way ANOVA followed by Tukey's post hoc test.
Relative mRNA expression levels were compared using one-way ANOVA
with Tukey's post hoc test. Data are presented as the mean ±
standard error of the mean (SEM). Statistically significant
difference was defined as P<0.05. Analyses were conducted using
JMP® Pro ver. 18.0.2 (SAS Institute Inc.) and GraphPad
Prism ver. 10.5.0 (Dotmatics).
Results
Andrographis demonstrates
anti-proliferative effects and synergizes with 5-FU in ESCC
cells
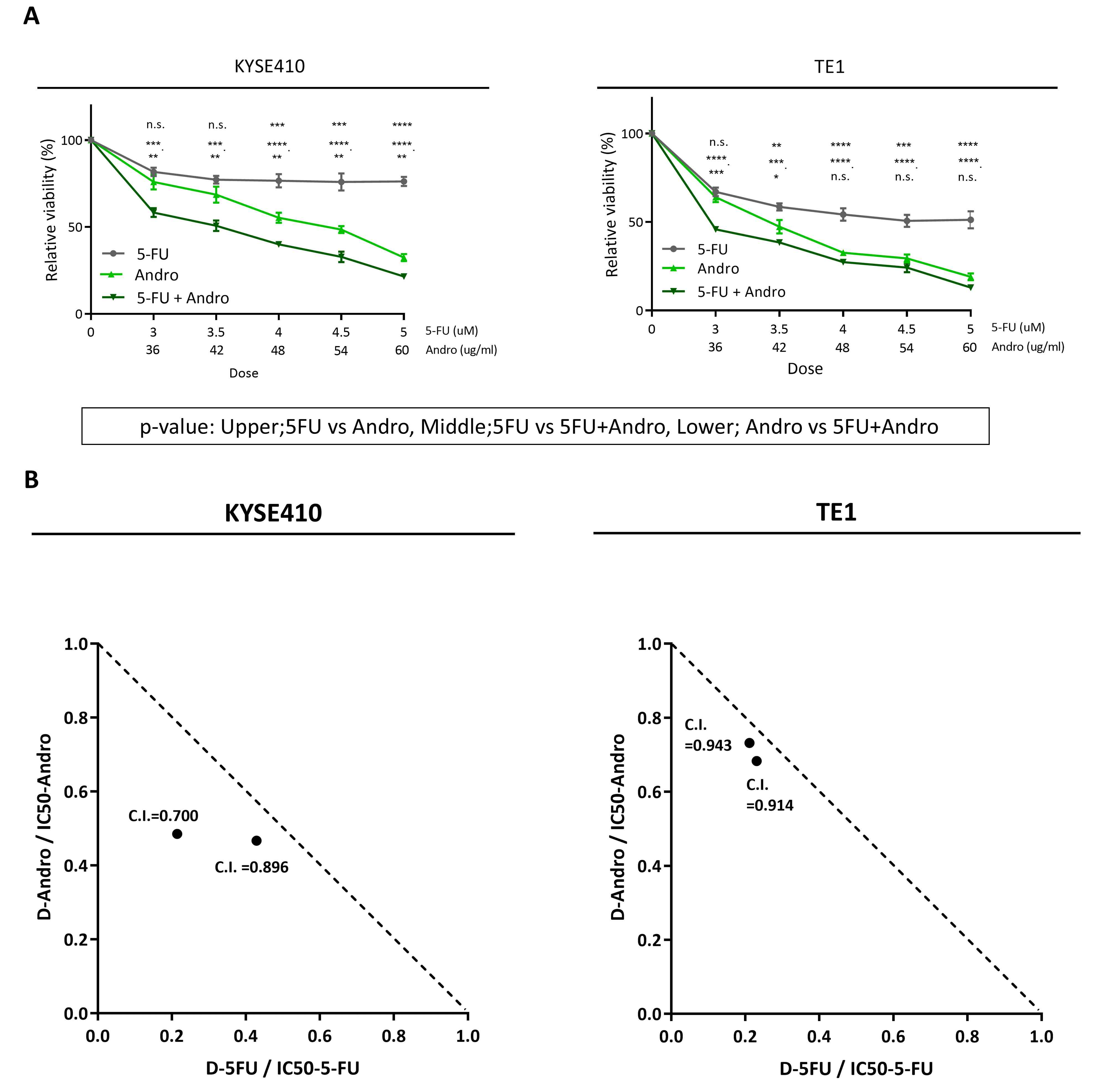
To assess whether Andrographis has
proliferation-inhibitory effects on ESCC cells and whether it
additively enhances the cell proliferation inhibitory effect of
5-FU on ESCC cells, KYSE410 and TE1 were used to examine the
effects of 5-FU (3, 3.5, 4, 4.5 and 5 µM) and Andrographis (36, 42,
48, 54 and 60 µg/ml) individually and in combination on cell
proliferation. WST assay results indicated that Andrographis
exhibited significantly greater proliferation inhibitory activity
than 5-FU in both cell lines (P<0.05). Moreover, the combination
of 5-FU and Andrographis significantly inhibited cell proliferation
compared with individual treatments in both cell lines (P<0.05)
(Fig. 1A).
Notably, both 5-FU and Andrographis inhibited the
proliferation of KYSE410 and TE1 cells in a dose-dependent manner,
with IC50 values of 14.0 and 4.5 µM for 5-FU and 51.7
and 40.9 µg/ml for Andrographis, respectively. Moreover, when used
in combination, the IC50 value was further reduced, and
the evaluation of the C.I. demonstrated an additive effect between
5-FU and Andrographis in both cell lines (Fig. 1B). These findings suggest that
Andrographis inhibits ESCC cell proliferation, and the combination
with 5-FU may enhance this inhibitory effect, following the defined
criteria (28). Based on these
findings, to avoid excessive cytotoxicity caused by 5-FU while
enabling evaluation of whether Andrographis has an additive effect,
subsequent assays were conducted using a sub-IC50
concentration of 5-FU (3.5 µM).
Andrographis serves as a sensitizer or
adjunct to 5-FU in suppressing colony formation in ESCC cells
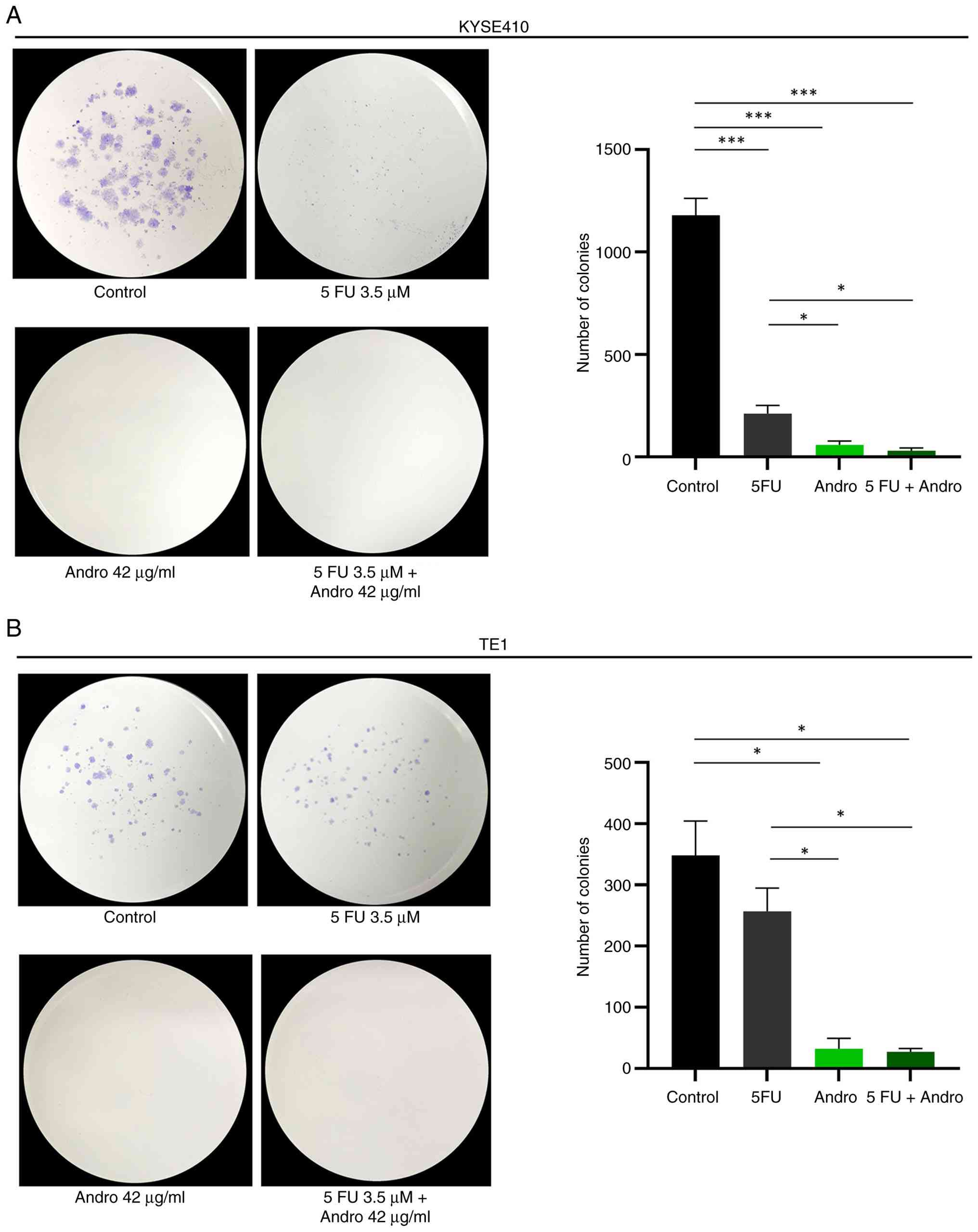
Next, the effects of 5-FU, Andrographis, and their
combination on cell viability were investigated using colony
formation assays. In KYSE410 cells, both 5-FU and Andrographis
significantly reduced colony formation compared with the control
(P<0.001) (Fig. 2A). In TE1
cells, 5-FU did not significantly alter colony formation, whereas
Andrographis significantly decreased the colony number (P<0.05)
(Fig. 2B). In both cell lines,
combination treatment showed a greater reduction in colony numbers
than 5-FU alone. However, the extent of reduction was comparable to
that observed with Andrographis alone, and no significant
difference was detected between the two groups. Thus, while the
combination treatment did not show a clear additional reduction
compared with treatment with Andrographis alone, it exhibited a
more noticeable inhibitory effect relative to 5-FU alone.
Andrographis treatment potentiates the
effects of 5-FU through increased apoptosis in ESCC cells
Previous studies have indicated that Andrographis
induces apoptosis in various gastrointestinal adenocarcinoma cells
(14–18). Next, it was assessed whether the
observed suppression of cell viability following treatment with
5-FU, Andrographis, and the combination was correlated with an
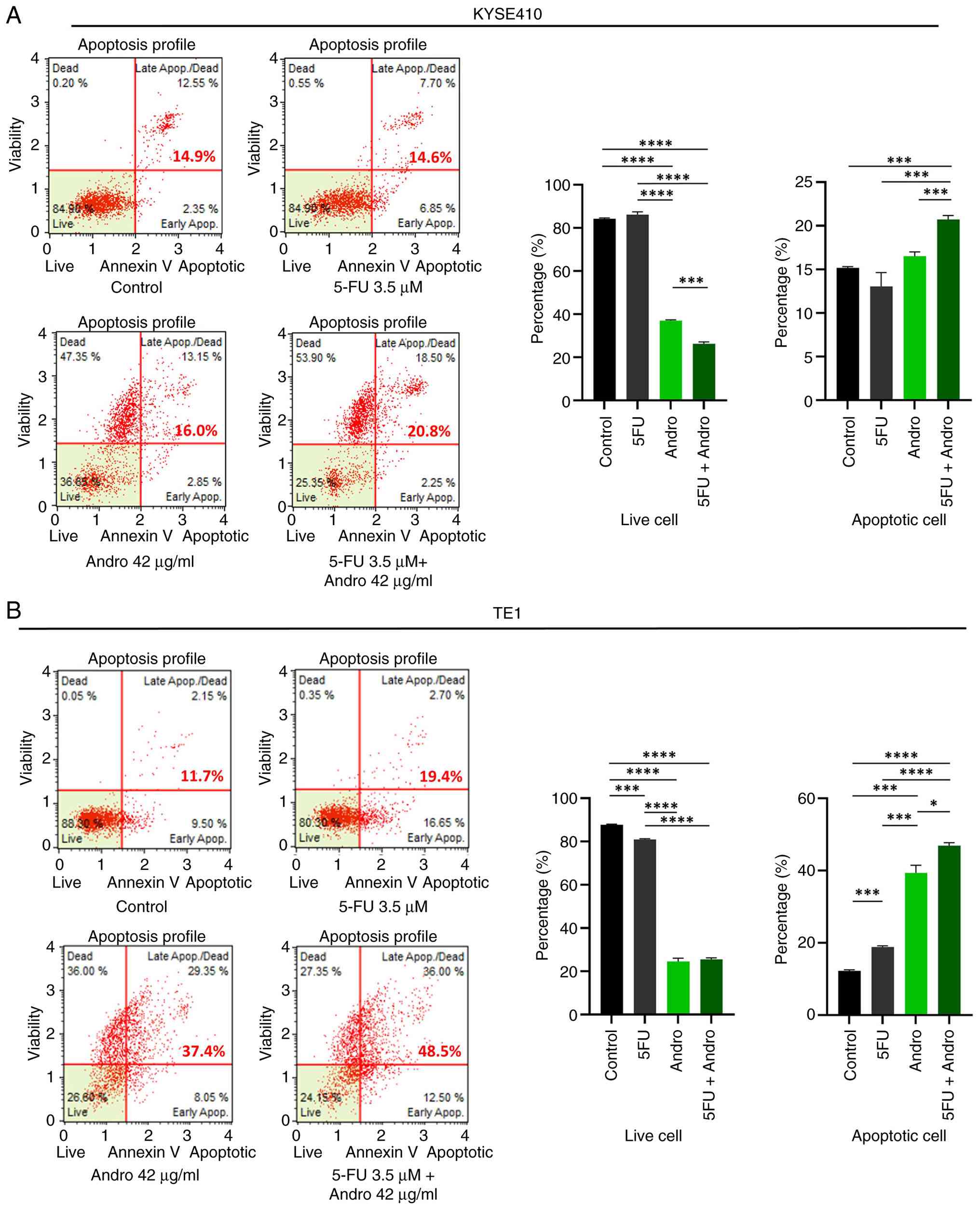
increase in apoptosis in ESCC cells. An Annexin V binding assay was
performed to quantify apoptotic cells after individual and
combination treatments. As shown in Fig. 3, Andrographis significantly elevated
the apoptotic rate compared with the control, particularly in TE1
cells (P<0.05). Furthermore, the combination treatment led to a
further increase in the percentage of apoptotic cells compared with
the control: 20.8% vs. 14.9% in KYSE410 cells (P<0.001)
(Fig. 3A) and 48.5% vs. 11.7% in
TE1 cells (P<0.0001) (Fig. 3B).
Consistent with the enhanced suppression of cell viability observed
with combination treatment, a significantly higher percentage of
apoptotic cells was observed compared with that observed with
individual treatments (P<0.05) (Fig.
3A and B). These findings suggest that Andrographis not only
induces apoptosis in ESCC cells but also enhances 5-FU-mediated
apoptosis, potentially acting as a sensitizer or adjunct agent for
chemotherapy.
Andrographis exerts its antitumor
effect via upregulation of ferroptosis-associated genes
Previously, it was reported that Andrographis
upregulates ferroptosis-related genes, such as HMOX1, GCLC and
GCLM, in gastric and colorectal adenocarcinoma cells (14,16,17).
Other studies have shown that Andrographis activates ferroptosis in
several malignant tumors, such as non-small cell lung cancer
(29) and multiple myeloma
(30) cells. To determine whether
this finding is applicable to ESCC cells, RT-qPCR and western
blotting assays were performed, focusing on HMOX1, GCLC and GCLM as
the key target genes in ferroptosis.
As shown in Fig. 4A,
HMOX1 (KYSE410 P<0.01; TE1 P<0.01), GCLC (KYSE410 P<0.001;
TE1 P<0.05) and GCLM (KYSE410 P<0.001; TE1 P<0.0001) were
significantly upregulated in ESCC cells after treatment with
Andrographis compared with the control. Combination treatment also
upregulated HMOX1 (KYSE410 P<0.0001; TE1 P<0.01), GCLC
(KYSE410 P<0.001; TE1 P<0.05) and GCLM (KYSE410 P<0.001;
TE1 P<0.001) at the mRNA level compared with the control.
Furthermore, HMOX1 (KYSE410 P<0.0001; TE1 P<0.001), GCLC
(KYSE410 P<0.001; TE1 P<0.01) and GCLM (KYSE410 P<0.001;
TE1 P<0.001) were significantly upregulated by the combination
treatment compared with those treated with 5-FU in both ESCC cell
lines.
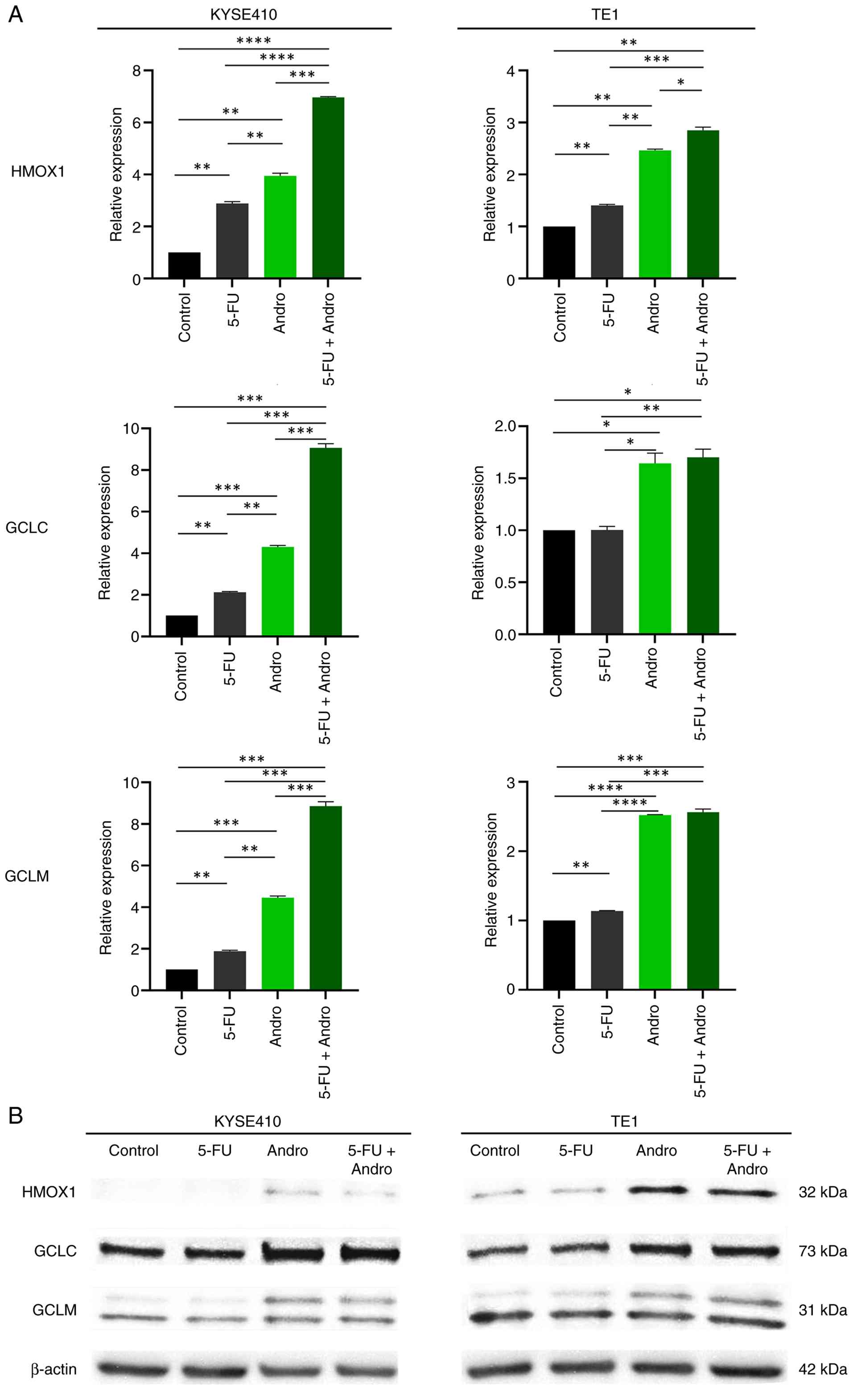 | Figure 4.Altered mRNA and protein expression
levels of the ferroptosis-associated targets HMOX1, GCLC and GCLM
after 5-FU, Andrographis, and combination treatment in esophageal
squamous cell carcinoma cells. (A) Changes in mRNA expression
(HMOX1, GCLC and GCLM) after 5-FU, Andrographis and combination
treatment in KYSE410 and TE1 cells. *P<0.05, **P<0.01,
***P<0.001 and ****P<0.0001 by one-way ANOVA with Tukey's
post hoc test. (B) Representative images of western blotting assay
of KYSE410 and TE1 cells treated as indicated. β-actin was used as
the loading control. HMOX1, heme oxygenase-1; GCLC,
glutamate-cysteine ligase catalytic; GCLM, glutamate-cysteine
ligase modifier; 5-FU, 5-fluorouracil. |
Western blotting was performed to validate the
expression of HMOX1, GCLC and GCLM at the protein level. As shown
in Fig. 4B, the expression levels
of HMOX1, GCLC and GCLM at the protein level were increased in both
the Andrographis and combination treatment groups compared with
those in the control and 5-FU groups in both ESCC cell lines.
Regarding mRNA expressional alteration of other
ferroptosis related genes such as NF-kB (NFKB1), STAT3, Nrf2
(NFE2L2) and ACSL4, these targets did not show consistent trends
between untreated/individual/combination treatment groups in two
ESCC cell-lines (Fig. S1). In case
of GPX4, mRNA expression of GPX4 was downregulated and showed
similar trends after Andrographis or combination treatment in two
ESCC cell-lines (Fig. S1). On the
other hand, mRNA expression of SLC7A11 were upregulated and showed
similar trends after Andrographis or combination treatment
(Fig. S1).
Discussion
In the present study, the antitumor effects of
Andrographis against ESCC cells (KYSE410 and TE1) were examined and
its additive effects with 5-FU, a key agent of systemic
chemotherapy for ESCC, were evaluated. It was demonstrated that
Andrographis inhibited cell proliferation and colony formation and
induced cell death through the activation of apoptosis and
ferroptosis. Using a sub-IC50 dose of 5-FU minimized
cytotoxicity and enabled proper evaluation of its single-agent
effects. Even at this low dose, combining 5-FU with Andrographis
further enhanced the response. These findings align with prior
reports of Andrographis activity in ESCC and suggest its potential
as a 5-FU-based adjunct therapy.
The standard treatment for locally advanced EC is
NAC followed by curative surgery. The JCOG9907 and JCOG1109 trials
demonstrated the efficacy of CF and DCF therapies, respectively, as
promising NAC regimens (3,4). However, both regimens frequently cause
severe toxicities, including febrile neutropenia, weight loss, and
5-FU-related gastrointestinal, hematologic, neurologic, and
cardiovascular adverse events, which can limit dosing and worsen
outcomes (31). These limitations
highlight the need for adjunctive agents that can enhance 5-FU
efficacy while reducing toxicity.
In the present study, Andrographis, a dietary
compound with anti-inflammatory, antioxidant and immunomodulatory
effects (8–13), was investigated as a potential
therapeutic agent for ESCC. While its antitumor effects have been
reported mainly in gastrointestinal adenocarcinomas (14–18),
studies in SCC remain limited (22,23).
The current in vitro experiments demonstrated that
Andrographis significantly inhibited ESCC cell proliferation and
colony formation and induced apoptosis, suggesting its efficacy
extends to SCC. Notably, based on the patterns observed in the
present data, TE1 and KYSE410 cells appear to differ in their
responsiveness to cytotoxic stress. TE1 cells exhibited strong
apoptotic responses to each single agent, suggesting that apoptosis
pathways may be readily activated, which limits the remaining
dynamic range for further enhancement by the combination
treatment.
By contrast, KYSE410 cells demonstrated only modest
apoptosis induction with both monotherapies, indicating a more
refractory basal phenotype. Consequently, a wider dynamic range
remains available, and the combination of 5-FU and Andrographis
produced additive effect that was suggested to exceed the effect of
5-FU alone.
As one of the underlying mechanisms for its
antitumor effect, focus was addressed on ferroptosis. Ferroptosis
is a regulated cell death distinct from apoptosis and necrosis,
characterized by lipid peroxide accumulation, mitochondrial
dysfunction and hypoxia-inducible factor-1α activation (19–21).
Dietary compounds have been shown to induce ferroptosis via diverse
mechanisms. In colorectal adenocarcinoma, ginsenoside Rh3
suppresses SLC7A11 and activates Stat3/p53/NRF2 (32). Curcumin inhibits glutathione
peroxidase 4 (GPX4) and SLC7A11 while modulating JNK and
PI3K/Akt/mTOR (33,34) in gastric adenocarcinoma,
arenobufagin upregulates Rev-erbα (35), and in ESCC, berbamine destabilizes
GPX4 by downregulating USP51 (36).
Based on previous studies, HMOX1, GCLC and GCLM were identified as
the most consistently and reproducibly regulated
ferroptosis-related genes in microarray analyses of
gastrointestinal adenocarcinoma models treated with Andrographis
(14–17). HMOX1 regulates iron metabolism and
reactive oxygen species (ROS), thereby promoting ferroptosis
(37–39), whereas GCLC and GCLM are essential
for glutathione (GSH) synthesis and antioxidant defense via the
antioxidant response element (ARE)-Nrf2 pathway (40–42).
Because ferroptosis is driven by iron-dependent lipid peroxidation
and disruption of cellular redox homeostasis, regulation of HMOX1
and glutathione synthesis pathways may reflect mechanistic
processes linked to ferroptosis. In the present study, however,
these transcriptional changes are interpreted as evidence of
redox-related pathway modulation rather than definitive ferroptotic
cell death, given the absence of direct lipid peroxidation or
iron-dependent rescue assays. In the present study, redox-related
pathways refer to cellular mechanisms that regulate oxidative
stress, including iron metabolism and glutathione-dependent
antioxidant systems. These observations suggest that such genes may
have translational relevance as therapeutic targets not only in
adenocarcinomas but also in SCCs. Therefore, HMOX1, GCLC and GCLM
were selected as representative ferroptosis-associated genes in the
present study. In KYSE410 cells, the combination markedly
upregulated HMOX1, GCLC and GCLM, suggesting enhanced antioxidant
responses that promote ferroptosis. By contrast, in TE1 cells, only
HMOX1 was increased, whereas GCLC and GCLM changes were limited,
indicating that the effect of 5-FU on GSH synthesis may depend on
ESCC histological subtype. These molecular changes suggest that
Andrographis may sensitize ESCC cells to alter redox homeostasis by
inducing ferroptosis, consistent with previous findings in
gastrointestinal adenocarcinoma cells. Regarding the alteration of
mRNA-level for other ferroptosis regulators by RT-qPCR using cDNA
derived from un-treated or post-treated ESCC cell-lines, GPX4
levels are downregulated after treatment by Andrographis in both
ESCC cell-lines. GPX4 is a remarkable negative regulator of
ferroptosis, which converts reduced GSH to oxidized glutathione and
reduces lipid hydroperoxides, and it has been reported that GPX4
enhances ferroptosis in a MAPK/ERK kinase-, iron-, and
ROS-dependent manner (43). Thus,
in the present study, downregulation of GPX4 after
Andrographis-treatment is expected hypothesis in consistency with
the upregulation of HMOX1/GCLC/GCLM. On the other hand,
upregulation of SLC7A11 after Andrographis-treatment was also shown
in both ESCC cell-lines. SLC7A11 is a cystine/glutamate
transporter, and SLC7A11-mediated cystine transportation plays an
important role in suppressing ferroptosis by unchecked lipid
peroxidation in cellular membranes (44), thus SLC7A11 is also known as a
well-established negative regulator ferroptosis. It is suggested
that the qPCR result of the present study for SLC7A11 which is
opposite from hypothesis is induced because the ferroptosis-related
pathway consists of not a simple but quite a complicated process
including multiple genes (45), and
positive- or negative-regulation of individual ferroptosis-related
genes may occur by Andrographis-treatment. In future studies,
further identification of down-stream ferroptosis-related target
altered by Andrographis treatment is warranted.
Even at sub-IC50 doses of 5-FU, combining it with
Andrographis enhanced antitumor effects, as shown by proliferation
and apoptosis assays. Apoptosis and ferroptosis are not strictly
independent modes of cell death; rather, they may potentially
interact through several shared regulatory hubs associated with
oxidative stress, such as intracellular iron homeostasis,
mitochondrial ROS and lipid peroxidation (46,47).
The ferroptosis-related gene alterations observed in the present
study appear to be compatible with these previous observations and
may indicate stress-response pathways that partially overlap with
apoptosis. The current findings may provide preliminary insight
into how these two pathways might interact.
Regarding clinical exposure, continuous infusion of
5-FU at 500–550 mg/m2 over 24 h yielded a steady-state
plasma concentration of ~120 ng/ml (0.923 µM) (48). While JCOG9907 and JCOG1109 did not
report plasma concentrations of 5-FU (continuous infusion at 800
and 750 mg/m2 over 24 h, respectively) (3), the current findings indicate that
clinically achievable 5-FU levels are lower than the concentrations
used in the present in vitro assays, where the
IC50 values of 5-FU were 14.0 µM in KYSE410 cells and
4.5 µM in TE1 cells. The observed synergy suggests that combining
Andrographis with 5-FU may allow for a reduction in the 5-FU dose
while maintaining comparable cytotoxic effects.
The results of the present study indicate that
Andrographis exerts antitumor effects by activating multiple cell
death pathways, and its combination with 5-FU potentiates these
effects. While the current in vitro findings require careful
preclinical and clinical validation, they highlight Andrographis as
a potential adjunctive strategy for ESCC treatment.
The present study has several limitations. First, a
significant limitation is that the concentrations of 5-FU used in
the in vitro experiments exceeded clinically achievable
steady-state plasma levels (~0.9 µM), including the
sub-IC50 doses. Thus, the observed synergy was obtained
under supra-physiological conditions that may not fully reflect
in vivo pharmacokinetics. The observed synergistic effects
should not be interpreted as direct evidence for clinical
application, but rather as preliminary mechanistic insights derived
from in vitro experiments. Second, the clinical relevance of
combining Andrographis with 5-FU requires rigorous in vivo
validation under pharmacokinetically relevant conditions.
Evaluation in appropriate animal or organoid models, together with
assessment of toxicity and therapeutic window, will be essential to
determine whether a clinically meaningful benefit can be achieved.
These issues represent important objectives for future
investigation. Third, it was confirmed that Andrographis treatment
increased the expression of HMOX1, GCLC and GCLM; however, the
detailed molecular mechanisms responsible for ferroptosis induction
were not examined. Clarifying these mechanisms in future studies
may help bridge basic research and clinical translation. Finally,
the effects of Andrographis in 5-FU-resistant ESCC cells were not
assessed in the present study. Because 5-FU resistance is
multifactorial and often involves DNA damage-response alterations,
determining whether Andrographis induces ferroptosis in resistant
cells may provide insights for refining chemotherapy selection in
ESCC.
In conclusion, the results of the present study
indicate that Andrographis exerts antitumor effects on ESCC cells
by inducing apoptosis and modulating ferroptosis-related
transcriptional programs. When combined with low-dose 5-FU, these
effects are additively enhanced, suggesting the potential of
Andrographis as an adjunctive therapeutic strategy to improve
efficacy while potentially reducing 5-FU dosage. Further studies
are warranted to clarify the precise molecular mechanisms and to
evaluate its efficacy in 5-FU-resistant ESCC models.
Supplementary Material
Supporting Data
Acknowledgements
The authors would like to acknowledge Ms Amphone
Okada and Ms Yuki Orito (Department of Gastrointestinal and
Pediatric Surgery, Institute of Life Sciences, Mie University
Graduate School of Medicine, Japan) for various technical
suggestions, and Dr Gabrielle White Wolf (https://jp.edanz.com/ac) for editing a draft of this
manuscript.
Funding
The present study was supported by Grants-in-Aid for Scientific
Research (KAKENHI) (grant nos. 24K11846 and 25K11904).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
KY, TS, YOku, RM, HY and YT conceived and designed
the present study. KY and RM performed the experiments and analyzed
the data. KY, TS and RM drafted the manuscript and prepared the
figures. YH, TK, SY, YS, KH, MKa, YK, YOki, SY, MKo, MO, HO, ST and
AG contributed to data acquisition and interpretation of the data.
YOku and YT reviewed and revised the manuscript. All authors read
and approved the final version of the manuscript. KY and TS confirm
the authenticity of all the raw data.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
5-FU
|
5-fluorouracil
|
|
C.I.
|
combination index
|
|
ESCC
|
esophageal squamous cell carcinoma
|
|
FBS
|
fetal bovine serum
|
|
HMOX1
|
heme oxygenase-1
|
|
GAPDH
|
glyceraldehyde 3-phosphate
dehydrogenase
|
|
GCLC
|
glutamate-cysteine ligase
catalytic
|
|
GCLM
|
glutamate-cysteine ligase modifier
|
|
GPX4
|
glutathione peroxidase 4
|
|
GSH
|
glutathione
|
|
MSS
|
microsatellite stable
|
|
ROS
|
reactive oxygen species
|
References
|
1
|
Bray F, Laversanne M, Sung H, Ferlay J,
Siegel RL, Soerjomataram I and Jemal A: Global cancer statistics
2022: GLOBOCAN estimates of incidence and mortality worldwide for
36 cancers in 185 countries. CA Cancer J Clin. 74:229–263.
2024.PubMed/NCBI
|
|
2
|
Lewis S and Lukovic J: Neoadjuvant therapy
in esophageal cancer. Thorac Surg Clin. 32:447–456. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ando N, Kato H, Igaki H, Shinoda M, Ozawa
S, Shimizu H, Nakamura T, Yabusaki H, Aoyama N, Kurita A, et al: A
randomized trial comparing postoperative adjuvant chemotherapy with
cisplatin and 5-fluorouracil versus preoperative chemotherapy for
localized advanced squamous cell carcinoma of the thoracic
esophagus (JCOG9907). Ann Surg Oncol. 19:68–74. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kato K, Machida R, Ito Y, Daiko H, Ozawa
S, Ogata T, Hara H, Kojima T, Abe T, Bamba T, et al: Doublet
chemotherapy, triplet chemotherapy, or doublet chemotherapy
combined with radiotherapy as neoadjuvant treatment for locally
advanced oesophageal cancer (JCOG1109 NExT): A randomised,
controlled, open-label, phase 3 trial. Lancet. 404:55–66. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Hong P, Xu T, Xu J, Chen W, Hu H, Chen J,
Li L, Zheng C, Li B, Liu J, et al: CD24 promotes metastasis and
chemoresistance by directly targeting Arf6-ERK pathway in
esophageal squamous cell carcinoma. Cancer Lett. 594:2169942024.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Yang X, Jiao Y, Zhang Y, Sun M, Gao Y,
Zhou Y, Xiao H, Ren J, Zhou Z, Zhai Y, et al: Oseltamivir enhances
5-FU sensitivity in esophageal squamous carcinoma with high SPNS1.
Biomed Pharmacother. 173:1163672024. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Li Z, Chen X, Li Y, Xu Y and Zhou Y:
Aberrant DNA methylation in esophageal squamous cell carcinoma and
its clinical implications in systemic chemotherapy. Int J Med Sci.
22:1002–1014. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Singh S, Pandey P, Ghosh S and Banerjee S:
Anti-cancer labdane diterpenoids from adventitious roots of
Andrographis paniculata: Augmentation of production prospect
endowed with pathway gene expression. Protoplasma. 255:1387–1400.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Islam MT, Ali ES, Uddin SJ, Islam MA, Shaw
S, Khan IN, Saravi SSS, Ahmad S, Rehman S, Gupta VK, et al:
Andrographolide, a diterpene lactone from Andrographis
paniculata and its therapeutic promises in cancer. Cancer Lett.
420:129–145. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Liu G and Chu H: Andrographolide inhibits
proliferation and induces cell cycle arrest and apoptosis in human
melanoma cells. Oncol Lett. 15:5301–5305. 2018.PubMed/NCBI
|
|
11
|
Jayakumar T, Hsieh CY, Lee JJ and Sheu JR:
Experimental and clinical pharmacology of Andrographis
paniculata and its major bioactive phytoconstituent
andrographolide. Evid Based Complement Alternat Med.
2013:8467402013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Poolsup N, Suthisisang C, Prathanturarug
S, Asawamekin A and Chanchareon U: Andrographis paniculata
in the symptomatic treatment of uncomplicated upper respiratory
tract infection: Systematic review of randomized controlled trials.
J Clin Pharm Ther. 29:37–45. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Dai Y, Chen SR, Chai L, Zhao J and Wang Y
and Wang Y: Overview of pharmacological activities of
Andrographis paniculata and its major compound
andrographolide. Crit Rev Food Sci Nutr. 59 (Suppl 1):S17–S29.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Shimura T, Sharma P, Sharma GG, Banwait JK
and Goel A: Enhanced anti-cancer activity of andrographis with
oligomeric proanthocyanidins through activation of metabolic and
ferroptosis pathways in colorectal cancer. Sci Rep. 11:75482021.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Miyazaki K, Xu C, Shimada M and Goel A:
Curcumin and andrographis exhibit Anti-tumor effects in colorectal
cancer via activation of ferroptosis and dual suppression of
glutathione Peroxidase-4 and ferroptosis suppressor Protein-1.
Pharmaceuticals (Basel). 16:3832023. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ma R, Shimura T, Yin C, Okugawa Y,
Kitajima T, Koike Y, Okita Y, Ohi M, Uchida K, Goel A, et al:
Antitumor effects of andrographis via ferroptosis-associated genes
in gastric cancer. Oncol Lett. 22:5232021. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Sharma P, Shimura T, Banwait JK and Goel
A: Andrographis-mediated chemosensitization through activation of
ferroptosis and suppression of beta-catenin/Wnt-signaling pathways
in colorectal cancer. Carcinogenesis. 41:1385–1394. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Bao GQ, Shen BY, Pan CP, Zhang YJ, Shi MM
and Peng CH: Andrographolide causes apoptosis via inactivation of
STAT3 and Akt and potentiates antitumor activity of gemcitabine in
pancreatic cancer. Toxicol Lett. 222:23–35. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zhang Q, Deng T, Zhang H, Zuo D, Zhu Q,
Bai M, Liu R, Ning T, Zhang L, Yu Z, et al: Adipocyte-Derived
exosomal MTTP suppresses ferroptosis and promotes chemoresistance
in colorectal cancer. Adv Sci (Weinh). 9:e22033572022. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Li H, Yu K, Hu H, Zhang X, Zeng S, Li J,
Dong X, Deng X, Zhang J and Zhang Y: METTL17 coordinates
ferroptosis and tumorigenesis by regulating mitochondrial
translation in colorectal cancer. Redox Biol. 71:1030872024.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Singhal R, Mitta SR, Das NK, Kerk SA,
Sajjakulnukit P, Solanki S, Andren A, Kumar R, Olive KP, Banerjee
R, et al: HIF-2α activation potentiates oxidative cell death in
colorectal cancers by increasing cellular iron. J Clin Invest.
131:e1436912021. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Yue GG, Lee JK, Li L, Chan KM, Wong EC,
Chan JY, Fung KP, Lui VW, Chiu PW and Lau CB: Andrographis
paniculata elicits anti-invasion activities by suppressing
TM4SF3 gene expression and by anoikis-sensitization in esophageal
cancer cells. Am J Cancer Res. 5:3570–3587. 2015.PubMed/NCBI
|
|
23
|
Wang ZM, Kang YH, Yang X, Wang JF, Zhang
Q, Yang BX, Zhao KL, Xu LP, Yang LP, Ma J, et al: Andrographolide
radiosensitizes human esophageal cancer cell line ECA109 to
radiation in vitro. Dis Esophagus. 29:54–61. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Stewart MJ and Watson ID: Standard units
for expressing drug concentrations in biological fluids. Br J Clin
Pharmacol. 16:3–7. 1983. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Takahashi M, Sung B, Shen Y, Hur K, Link
A, Boland CR, Aggarwal BB and Goel A: Boswellic acid exerts
antitumor effects in colorectal cancer cells by modulating
expression of the let-7 and miR-200 microRNA family.
Carcinogenesis. 33:2441–2449. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Schneider CA, Rasband WS and Eliceiri KW:
NIH Image to ImageJ: 25 years of image analysis. Nat Methods.
9:671–675. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Chou TC: Drug combination studies and
their synergy quantification using the Chou-Talalay method. Cancer
Res. 70:440–446. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Jiaqi L, Siqing H, Qin W, di Z, Bei Z and
Jialin Y: Andrographolide promoted ferroptosis to repress the
development of non-small cell lung cancer through activation of the
mitochondrial dysfunction. Phytomedicine. 109:1546012023.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Li W, Fu H, Fang L, Chai H, Ding B and
Qian S: Andrographolide induced ferroptosis in multiple myeloma
cells by regulating the P38/Nrf2/HO-1 pathway. Arch Biochem
Biophys. 742:1096222023. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Katada C, Sugawara M, Hara H, Fujii H,
Nakajima TE, Ando T, Kojima T, Watanabe A, Sakamoto Y, Ishikawa H,
et al: A management of neutropenia using granulocyte colony
stimulating factor support for chemotherapy consisted of docetaxel,
cisplatin and 5-fluorouracil in patients with oesophageal squamous
cell carcinoma. Jpn J Clin Oncol. 51:199–204. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wu Y, Pi D, Zhou S, Yi Z, Dong Y, Wang W,
Ye H, Chen Y, Zuo Q and Ouyang M: Ginsenoside Rh3 induces
pyroptosis and ferroptosis through the Stat3/p53/NRF2 axis in
colorectal cancer cells. Acta Biochim Biophys Sin (Shanghai).
55:587–600. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Xin W and Zhang Y: Curcumin activates the
JNK signaling pathway to promote ferroptosis in colon cancer cells.
Chem Biol Drug Des. 103:e144682024. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Chen M, Tan AH and Li J: Curcumin
represses colorectal cancer cell proliferation by triggering
ferroptosis via PI3K/Akt/mTOR signaling. Nutr Cancer. 75:726–733.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Chen K, Li A, Wang J, Li D, Wang X, Liu C
and Wang Z: Arenobufagin causes ferroptosis in human gastric cancer
cells by increasing rev-erbalpha expression. J Tradit Complement
Med. 13:72–80. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Peng H, He Y, Hu Y, Sheng S, Maitiyasen M,
Li J, Liu Y, Hou X, Song H and Yi J: Berbamine promotes ferroptosis
of esophageal squamous cell carcinoma by facilitating
USP51-mediated GPX4 ubiquitination and degradation. Biomed
Pharmacother. 179:1173092024. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Hassannia B, Wiernicki B, Ingold I, Qu F,
Van Herck S, Tyurina YY, Bayır H, Abhari BA, Angeli JPF, Choi SM,
et al: Nano-targeted induction of dual ferroptotic mechanisms
eradicates high-risk neuroblastoma. J Clin Invest. 128:3341–3355.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Chen X, Yu C, Kang R and Tang D: Iron
Metabolism in ferroptosis. Front Cell Dev Biol. 8:5902262020.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Tang D, Chen X, Kang R and Kroemer G:
Ferroptosis: Molecular mechanisms and health implications. Cell
Res. 31:107–125. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Lu CY, Yang YC, Li CC, Liu KL, Lii CK and
Chen HW: Andrographolide inhibits TNFalpha-induced ICAM-1
expression via suppression of NADPH oxidase activation and
induction of HO-1 and GCLM expression through the PI3K/Akt/Nrf2 and
PI3K/Akt/AP-1 pathways in human endothelial cells. Biochem
Pharmacol. 91:40–50. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Nishizawa H, Matsumoto M, Shindo T,
Saigusa D, Kato H, Suzuki K, Sato M, Ishii Y, Shimokawa H and
Igarashi K: Ferroptosis is controlled by the coordinated
transcriptional regulation of glutathione and labile iron
metabolism by the transcription factor BACH1. J Biol Chem.
295:69–82. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Huang CS, Lii CK, Lin AH, Yeh YW, Yao HT,
Li CC, Wang TS and Chen HW: Protection by chrysin, apigenin, and
luteolin against oxidative stress is mediated by the Nrf2-dependent
up-regulation of heme oxygenase 1 and glutamate cysteine ligase in
rat primary hepatocytes. Arch Toxicol. 87:167–178. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Yang WS, SriRamaratnam R, Welsch ME,
Shimada K, Skouta R, Viswanathan VS, Cheah JH, Clemons PA, Shamji
AF, Clish CB, et al: Regulation of ferroptotic cancer cell death by
GPX4. Cell. 156:317–331. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Koppula P, Zhuang L and Gan B: Cystine
transporter SLC7A11/xCT in cancer: Ferroptosis, nutrient
dependency, and cancer therapy. Protein Cell. 12:599–620. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Zhou Q, Meng Y, Li D, Yao L, Le J, Liu Y,
Sun Y, Zeng F, Chen X and Deng G: Ferroptosis in cancer: From
molecular mechanisms to therapeutic strategies. Signal Transduct
Target Ther. 9:552024. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Endale HT, Tesfaye W and Mengstie TA: ROS
induced lipid peroxidation and their role in ferroptosis. Front
Cell Dev Biol. 11:12260442023. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Neitemeier S, Jelinek A, Laino V, Hoffmann
L, Eisenbach I, Eying R, Ganjam GK, Dolga AM, Oppermann S and
Culmsee C: BID links ferroptosis to mitochondrial cell death
pathways. Redox Biol. 12:558–570. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Kuwahara A, Yamamori M, Nishiguchi K,
Okuno T, Chayahara N, Miki I, Tamura T, Kadoyama K, Inokuma T,
Takemoto Y, et al: Effect of dose-escalation of 5-fluorouracil on
circadian variability of its pharmacokinetics in Japanese patients
with Stage III/IVa esophageal squamous cell carcinoma. Int J Med
Sci. 7:48–54. 2010. View Article : Google Scholar : PubMed/NCBI
|