Introduction
Cancer-associated fibroblasts (CAFs) exhibit
fibroblast-like morphology and are known to exert both
tumor-promoting and -suppressing effects (1). CAFs are located in the peritumoral
area and constitute one of the most crucial components of the tumor
microenvironment (TME) (2). They
play vital roles in tumor growth, invasion and metastasis (3,4). In
addition to the significant impact of CAFs on cancer cells, cancer
cells also influence CAFs, highlighting the importance of their
mutual crosstalk in understanding tumor biology (5).
The desmoplastic reaction (DR) classification has
been reported as a stromal evaluation index associated with
prognosis in resected colorectal cancer (CRC) samples (6). DR refers to the fibrotic response
generated by fibroblasts surrounding the tumor, and this stromal
component is known to express CAF markers (7). In CRC, DR is classified into three
categories: i) Immature; ii) intermediate; and iii) mature; and
serves as a prognostic predictor (8).
Since CAFs form cancer stroma and interact closely
with cancer cells, evaluating CAF activity and cancer stromal
status through DR classification appears crucial. The immature
classification in DR is known to indicate poor prognosis in CRC
(9). Periostin is a key gene in the
fibrogenic response of the stroma in CRC, and periostin is reported
to be positively associated with the immature classification in DR
(10). DR and periostin expression
are considered to play an important role in the evaluation of CAFs
in clinical specimens.
The hypoxic environment within tumors affects key
areas of cancer biology, including cellular invasion, metastasis
and the regulation of cell death (11). Reported evidence indicated that gene
expression patterns associated with hypoxia are linked to
unfavorable outcomes in human malignancies (12). A previous study (13) by the authors revealed that CRC liver
metastases show a progressive reduction in vascular density and
become increasingly hypoxic toward the center of the metastases.
Using microarray analysis of cells from the central region of the
metastases, several novel hypoxia-inducible genes including
adrenomedullin (14),
procollagen-lysine, 2-oxoglutarate 5-dioxygenase 2 (15), ephrin-A1 (16,17),
secretoglobin family 2A member 1 (18) and aldolase A (19) were identified to be associated with
CRC prognosis (13). In pancreatic
cancer (PCa), hypoxia in the TME has been reported to enhance
cytokine-induced inflammatory CAF phenotype and promote tumor
growth (20).
While hypoxia is known to enhance malignant tumor
behavior, the specific roles and functional characteristics of CAFs
under hypoxic conditions in CRC remain largely unclear. The aim of
the current study is to elucidate the effects of hypoxia, a
fundamental condition in solid tumors, on CAFs and to identify key
factors involved in hypoxic CAFs. The findings will contribute to a
deeper understanding of the interaction between CAFs and cancer
cells.
Materials and methods
Patient selection criteria and
specimen collection
Patients who underwent surgical procedures at The
University of Osaka Hospital between October 2023 and May 2024 and
were diagnosed with CRC according to the guidelines of the Japanese
Society for Cancer of the Colon and Rectum (21) were considered for inclusion.
Exclusion criteria included inflammatory bowel disease and familial
adenomatous polyposis. No exclusions were made based on age or
disease stage. All cases that underwent surgery during the
specified period and were evaluated by DR classification were
included in the study. A total of 59 CRC tissue samples were
included using opportunistic sampling based on the availability of
cases meeting the inclusion criteria; the sample size was not
determined by formal power calculation. The study cohort consisted
of 26 males and 33 females, with a median age of 73 years (range:
40–88 years). All patients received a definitive diagnosis of CRC.
Clinicopathological classification was determined using the
criteria of the Japanese Society for Cancer of the Colon and
Rectum. After surgery, patients with lymph node metastasis
generally received adjuvant chemotherapy. Collected samples were
immersed in 10% buffered formalin and fixed overnight at 4°C, then
dehydrated through a graded ethanol series and embedded in
paraffin.
Ethics approval and patient
consent
The current study was approved by the ethics
committee of the Graduate School of Medicine, University of Osaka
(approval nos. 15144 and 19020; Suita, Japan). Written informed
consent was obtained from all participants prior to inclusion in
the study. The study protocol conformed to the ethical guidelines
of the 1975 Declaration of Helsinki.
Clinical sample collection and
histological evaluation
Between October 2023 and May 2024, 59 CRC tissue
samples were collected during surgeries performed at the Department
of Gastroenterological Surgery, University of Osaka. Samples were
formalin-fixed at 4°C overnight, processed through graded ethanol
and embedded in paraffin. A pathologist blinded to the clinical
outcomes classified 16 cases as having immature DR and 43 as mature
based on established histological criteria.
Colon fibroblasts
CAFs and normal fibroblasts (NFs) were isolated from
tumor and adjacent normal tissues, respectively, immediately after
CRC resection. To prevent cross-contamination, separate surgical
blades were used for each tissue type. Tissue fragments underwent
enzymatic digestion with collagenase, and the resulting cell pellet
was suspended in Dulbecco's Modified Eagle Medium (DMEM;
Sigma-Aldrich; Merck KGaA; RRID:SCR_001905) with antibiotics. Cells
were seeded in 6-well plates with DMEM supplemented with 10% fetal
bovine serum (FBS; Gibco; Thermo Fisher Scientific, Inc.;
RRID:SCR_008452) and cultured at 37°C with 5% CO2.
Cell culture conditions
CAFs and NFs were cultured in DMEM supplemented with
10% FBS under normoxic (37°C, 5% CO2) and hypoxic (37°C,
1% O2) conditions. The human CRC cell lines HT29 (RRID:
CVCL_0320), HCT116 (RRID: CVCL_0291) and RKO (RRID: CVCL_0504) were
obtained from the American Type Culture Collection and
authenticated using short tandem repeat profiling within 6 months
of experimental use. Cell lines were routinely tested for
mycoplasma contamination using polymerase chain reaction and were
maintained in DMEM supplemented with 10% FBS under the same
conditions as fibroblasts.
Immunofluorescence staining
Cells were cultured in 96-well plates for 24 h,
fixed with 4% paraformaldehyde at room temperature (RT) for 15 min
and treated with Triton X-100 and 5% bovine serum albumin
(MilliporeSigma) at RT for 10 min. Subsequently, cells were washed
twice with phosphate-buffered saline (PBS), and primary antibodies
including anti-α-smooth muscle actin (α-SMA; 1:400; cat. no. 19245;
Cell Signaling Technology, Inc.; RRID: AB_2734735),
anti-hypoxia-inducible factor-1α (HIF-1α; 1:500; cat. no. ab51608;
Abcam; RRID: AB_880418) and anti-epithelial cell adhesion molecule
(EpCAM; 1:500; cat. no. 2929; Cell Signaling Technology, Inc.;
RRID: AB_2098834) were applied. Cells were incubated at RT for 1 h,
washed twice with PBS and incubated with Alexa Fluor 647
anti-rabbit IgG secondary antibody (1:500; cat. no. 4414; Cell
Signaling Technology, Inc.; RRID: AB_10694544) for 30 min. After
washing with PBS, cells were counterstained with
4′,6-diamidino-2-phenylindole (1 µg/ml) and visualized using a
confocal microscope.
Immunohistochemical staining
Immunohistochemistry was performed as previously
described (10,11). Primary antibodies used included
anti-HIF-1α (1:200), anti-periostin (1:400; cat. no. 20302; Cell
Signaling Technology, Inc.; RRID: AB_2798819), anti-parathyroid
hormone-related protein (PTHrP; 1:200; cat. no. 10817-1-AP;
Proteintech Group, Inc.; RRID: AB_2174535) and anti-vitamin D
receptor (VDR; 1:200, cat. no. BS-2987R; Bioss Antibodies; RRID:
AB_11058910). VECTASTAIN® Elite® ABC Rabbit
IgG Kit (Vector Laboratories, Inc.; RRID: AB_2336817) was used
according to manufacturer's instructions. Primary antibodies were
incubated overnight at 4°C. Staining intensity was evaluated by two
pathologists independently and scored as +2 (equivalent to positive
control), +1 (weaker than positive control), or 0 (unstained) for
cytoplasmic and peritumoral stroma. A score of +2 was considered
positive.
Co-culture experiments
For indirect co-culture experiments, supernatants
from CAFs cultured under hypoxic and normoxic conditions were
collected. HT29 cells were then cultured in a medium containing a
1:1 ratio of DMEM supplemented with FBS and the respective
CAF-conditioned supernatant.
Treatment with PTHrP and vitamin
D
NFs and CAFs were cultured in DMEM supplemented with
PBS and PTHrP (1 µg/ml) at 37°C and 5% CO2. For vitamin
D experiments, CAFs were cultured in DMEM supplemented with
1α,25-dihydroxyvitamin D3 (10 µg/ml) dissolved in dimethyl
sulfoxide (DMSO; final DMSO concentration ≤0.5%) at 37°C and 5%
CO2.
Cell proliferation assay
Cells were seeded in 96-well plates at a density of
1.0×103 cells/well. Cell proliferation was assessed at
24, 48 and 72 h after treatment using 10 µl of Cell Counting Kit-8
(cat. no. CK04; Dojindo Molecular Technologies, Inc.) at 37°C for 2
h according to the manufacturer's protocol. The association between
absorbance measurements and manual cell counting was verified
before the experiment. Absorbance was measured at 450 nm.
Cell migration assay
The wound healing assay was used to assess cell
migration. Cells were plated in 6-well dishes at 2.0×105 cells/well
and cultured to 60–80% confluency. A 200-µl pipette tip was used to
create a linear scratch in the cell monolayer. Culture medium was
switched to DMEM containing 1% FBS to inhibit cell proliferation.
Images were captured using a BZ-X710 microscope (Keyence
Corporation) at 0, 24 and 48 h post-scratch and analyzed using
ImageJ (version 1.54f; National Institutes of Health)
(RRID:SCR_003070). Cell migration was quantified by measuring the
mean area between wound edges at three randomly selected locations
by an investigator blinded to the experimental conditions.
Drug sensitivity assay
HT29 cells co-cultured with supernatant from
normoxia- or hypoxia-cultured CAFs were seeded in 96-well plates at
1.0×104 cells/well for 24 h. Cells were then exposed to
varying concentrations of oxaliplatin. Cell viability was evaluated
using Cell Counting Kit-8 according to the manufacturer's
protocol.
RNA sequencing and analysis
Sample preparation and library construction were
performed using the TruSeq stranded mRNA sample preparation kit
(Illumina, Inc.) following the manufacturer's protocols. HCT116,
HT29 and RKO cell lines were cultured under normoxic (37°C, 5%
CO2) and hypoxic (37°C, 1% O2) conditions.
Sequencing was performed on a DNBSEQ-G400 sequencer (MGI Tech Co.,
Ltd.) in 100-base single-read mode. Adapter sequences were removed
using Trimmomatic (version 0.38; RRID:SCR_011848). Cleaned reads
were aligned to the hg19 human reference genome using TopHat2
(version 2.1.1; RRID:SCR_013035). Fragments per kilobase of exons
per million mapped fragments were calculated using Cufflinks
(version 2.2.1; RRID:SCR_014597). Gene Set Variation Analysis was
performed using R (version 4.3.2; RRID:SCR_001905). RNA quality and
integrity were assessed using the Agilent 2100 Bioanalyzer system.
RNA integrity was evaluated by electrophoresis to measure the
degree of degradation. Sequencing was performed using the NovaSeq
6000 S1 Reagent Kit v1.5 (200 cycles; cat. no. 20028318; Illumina,
Inc.). Final library concentrations were quantified using the KAPA
Library Quantification Kit (cat. no. KK4824/D; Roche Diagnostics)
by real-time PCR. Libraries were normalized to 2 nM before loading
onto the sequencer.
Proteomics analysis
Supernatants from CAFs cultured under hypoxic and
normoxic conditions were collected. Total protein was extracted
using radioimmunoprecipitation assay buffer containing protease and
phosphatase inhibitors (Thermo Fisher Scientific, Inc.). Proteins
were processed using nano liquid chromatography-tandem mass
spectrometry configured with an Ultimate 3000 Nano LC system,
Q-Exactive (Thermo Fisher Scientific, Inc.). Raw data were analyzed
using Scaffold 5 (Proteome Software Inc.; RRID:SCR_014345).
Ionization mode was positive (nano-ESI with capillary voltage of
1.8 kV). Flow rate was 300 nl/min.
Single-cell RNA sequencing
CAFs cultivated under both hypoxic and normoxic
conditions were prepared for single-cell RNA sequencing. Libraries
were prepared according to the Chromium Next GEM Single Cell
3′Reagent Kits (version 3.1; 10X Genomics, Inc.) protocols.
Sequencing was performed on an Illumina HiSeq X platform. The Cell
Ranger pipeline (version 6.1.1; RRID:SCR_017344) was used to
generate the data matrix. Raw sequencing reads were mapped to the
human reference genome (GRCh 38) using the STAR aligner
(RRID:SCR_015899). Data visualization and analysis were performed
using Loupe Browser (version 6.0.1; 10X Genomics, Inc.).
Bioinformatics analysis
For Gene Ontology (GO) analysis, the top 100
differentially expressed genes (DEGs) ranked by P-value were
analyzed using the Metascape platform (https://metascape.org; RRID: SCR_016620). Volcano
plots and correlation analyses (Pearson's correlation coefficient)
were performed using RStudio. Copy Number Variation (CNV) data were
obtained from The Cancer Genome Atlas (TCGA) dataset through the
cBioPortal platform (RRID:SCR_014555). Gene information and
chromosomal locations were obtained from the National Center for
Biotechnology Information (NCBI) Gene database
(RRID:SCR_002472).
Statistical analysis
Statistical analyses were performed using GraphPad
Prism (Dotmatics; RRID: SCR_002798). Data are presented as the mean
± standard deviation from at least three independent experiments.
Comparisons between two groups were performed using two-tailed
unpaired Student's t-test. For multiple comparisons, one-way
analysis of variance followed by Tukey's post-hoc test was used.
Correlation analyses were performed using Pearson's correlation
coefficient and Spearman's rank correlation coefficient. P<0.05
was considered to indicate a statistically significant
difference.
Data availability
All RNA and single-cell RNA sequencing data
generated in the present study have been deposited in the Gene
Expression Omnibus. CNV data from TCGA were accessed through the
cBioPortal platform (https://www.cbioportal.org/). Gene information was
obtained from the NCBI database (https://www.ncbi.nlm.nih.gov/).
Results
Morphology and immunofluorescence
staining of CAFs and NFs
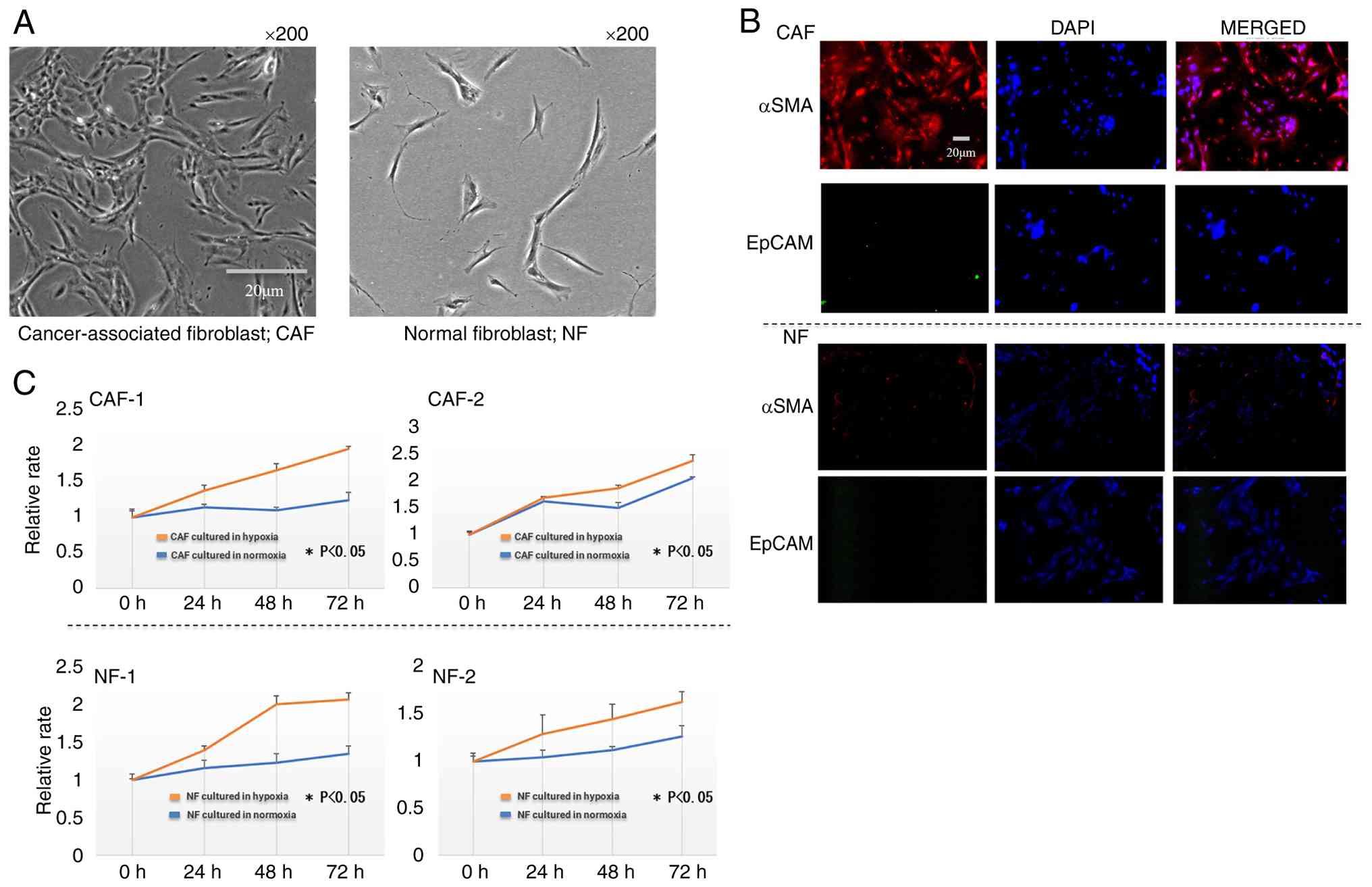
Both CAFs and NFs exhibited a spindle-shaped
morphology, but NFs appeared slightly more flattened (Fig. 1A). Immunofluorescence staining
revealed that CAFs were positive for α-SMA, while NFs were
negative. EpCAM, an epithelial marker, was not detected in either
cell type, confirming the successful establishment of CAFs and NFs
culture systems (Fig. 1B).
Proliferation of CAFs and NFs under
normoxic and hypoxic conditions
Both CAFs and NFs exhibited a significantly enhanced
proliferative capacity under hypoxic conditions compared with
normoxic conditions (P<0.05; Fig.
1C).
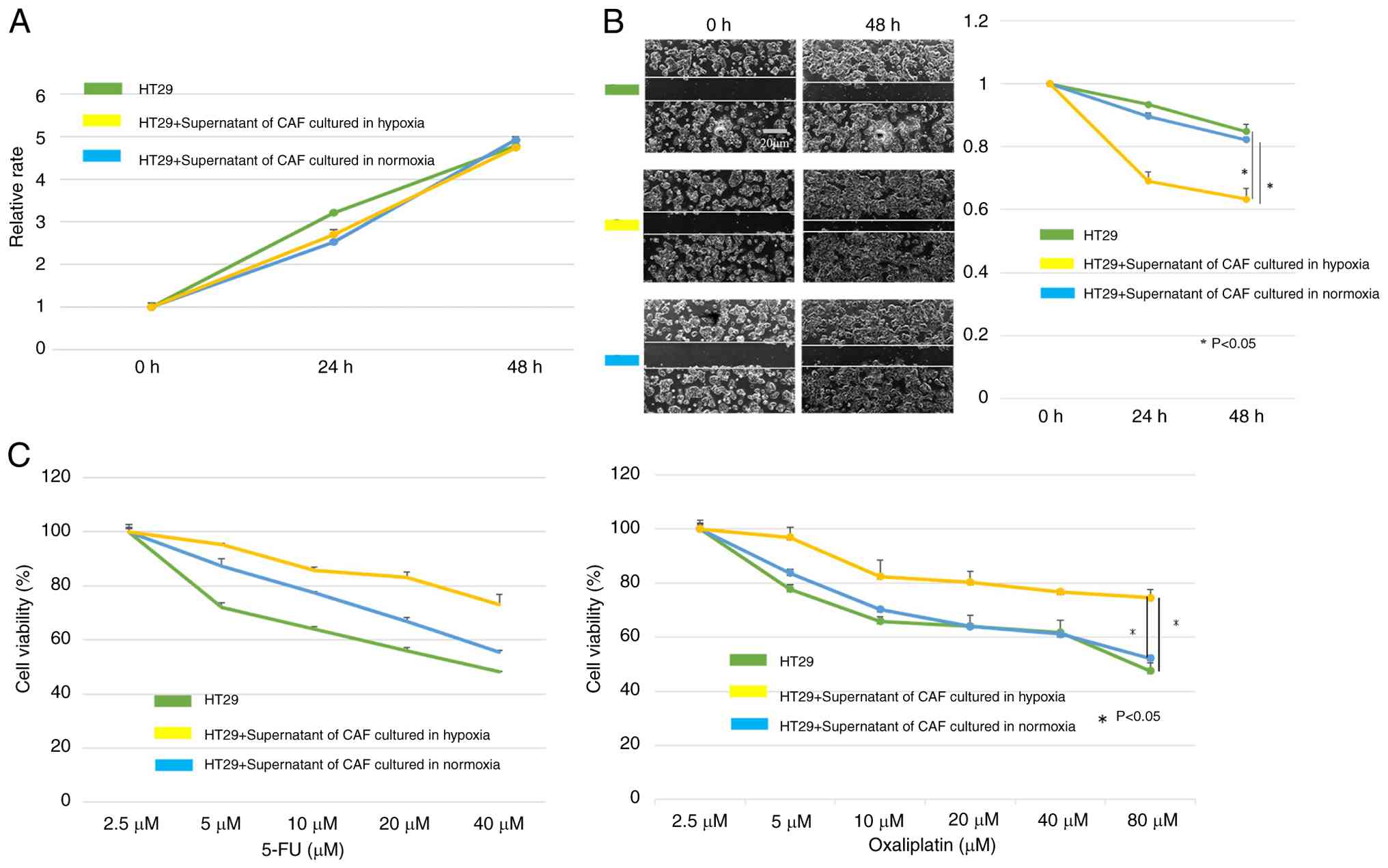
Co-culture of CAFs and HT29
HT29 cells cultured with the supernatant from CAFs
cultured under both normoxic and hypoxic conditions showed no
significant difference in proliferative capacity (Fig. 2A). However, HT29 cells treated with
supernatants from hypoxia-conditioned CAFs exhibited a
significantly enhanced migratory ability (P<0.05; Fig. 2B) and increased resistance to
oxaliplatin (cat. no. S1224; Selleck Chemicals) and 5-Fluorouracil
(5-FU; cat. no. 16220-14; Nacalai Tesque, Inc.) (P<0.05;
Fig. 2C), a key chemotherapeutic
drug for CRC.
Vitamin D-binding protein (DBP) was
significantly increased in the supernatant of hypoxic CAFs
Shotgun proteomic analysis identified 59 proteins in
the supernatant of CAFs. Furthermore, a t-test comparing protein
levels in the supernatant of CAFs cultured under hypoxic and
normoxic conditions identified six proteins with P<0.01 and the
average value fold change greater than two (Table I). Among these, DBP exhibited the
most significant increase.
 | Table I.A total of six proteins with
aP<0.05 shown by
Student's t-test between the two groups and a ratio of the mean
greater than 2-fold. |
Table I.
A total of six proteins with
aP<0.05 shown by
Student's t-test between the two groups and a ratio of the mean
greater than 2-fold.
| MS/MS View:
Identified Proteins (CAF) | n1 | n2 | n3 | h1 | h2 | h3 | P-value | n/h ratio |
|---|
| Vitamin D-binding
protein | 5 | 6 | 5 | 9 | 12 | 13 | 0.0152 | 0.47 |
| Delta-globin B2
variant (Fragment) | 2 | 2 | 2 | 0 | 1 | 1 | 0.0286 | 3 |
| Mutant hemoglobin
alpha 2 globin chain | 6 | 5 | 5 | 4 | 1 | 0 | 0.0417 | 3.2 |
| Collagen
alpha-1(I) | 3 | 2 | 4 | 1 | 1 | 0 | 0.0178 | 4.5 |
| Collagen alpha-2(I)
chain | 3 | 3 | 4 | 1 | 0 | 0 | 0.0016 | 10 |
| Fibronectin 1,
isoform CRA_n | 3 | 7 | 6 | 0 | 1 | 0 | 0.0224 | 16 |
In a single-cell analysis of CAFs cultured under
hypoxic and normoxic conditions, the cells were classified into 15
clusters (Fig. 3A). Clusters
exhibiting upregulation of CAF markers were identified. Clusters 2,
5, 6 and 7, characterized by high expression of α-SMA, periostin
and HIF-1α, were selected for further analysis (Fig. 3B and C). In addition, examination of
our dataset for the expression of CAF marker genes reported by
Elyada et al (22) revealed
that several markers such as FAP and MYL9 were expressed at high
levels in clusters 2, 5, 6 and 7 (Fig.
S1). These clusters were then re-clustered, and DEGs between
hypoxic and normoxic CAFs were identified within each cluster
(Fig. 3D). The proportions of
hypoxic and normoxic cells in each cluster can be confirmed in
Table SI.
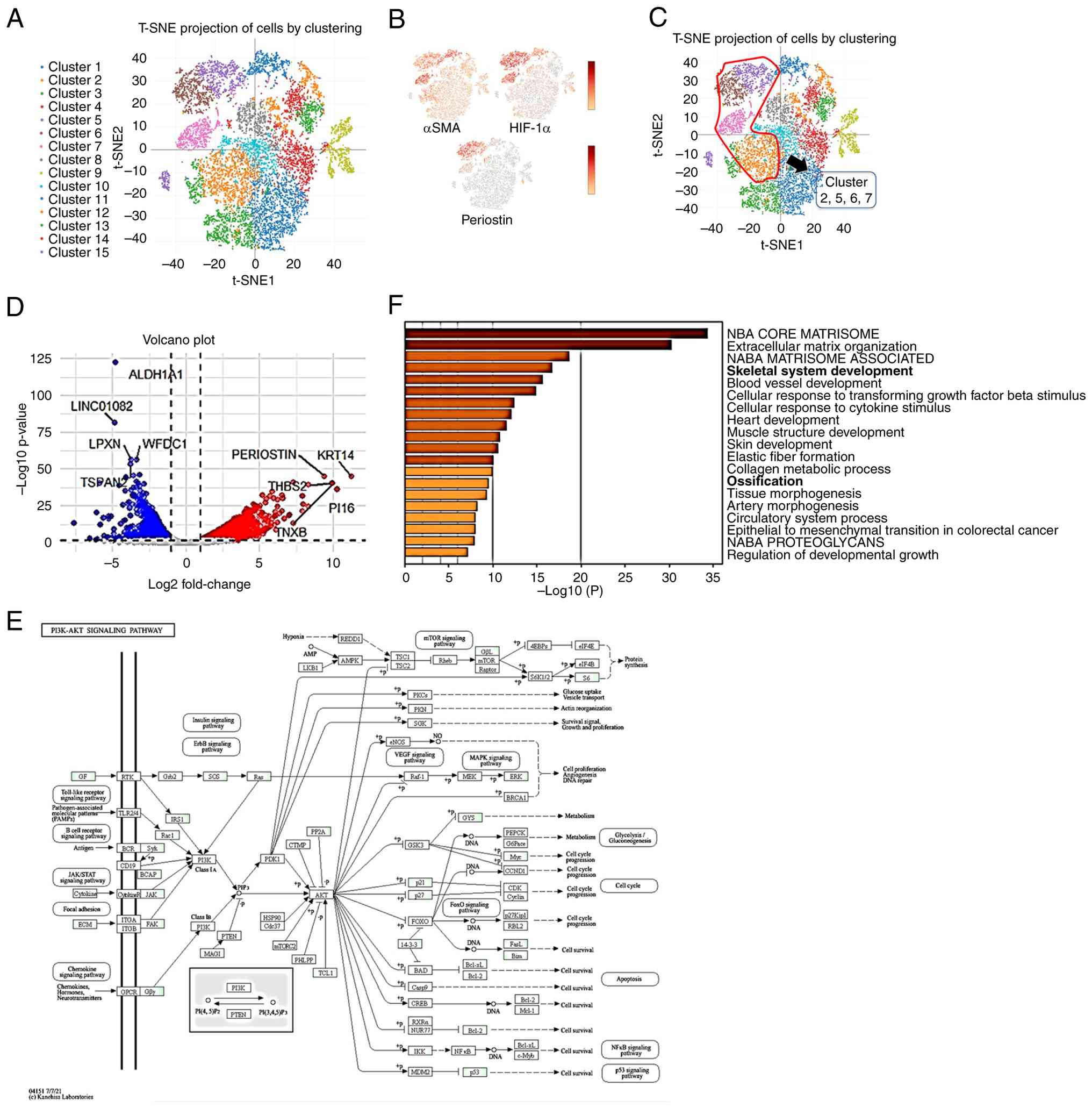 | Figure 3.Single-cell RNA sequencing of human
CAF and generation of data matrix. (A) CAF divided into 15
clusters. (B) CAF gene expression of α-SMA, HIF-1α and periostin.
(C) Selected clusters with high expression of α-SMA, HIF-1α and
periostin. (D) Volcano plots created from DEGs of cluster 2, 5, 6
and 7. (E) Gene list was generated from DEGs and KEGG pathway
analysis was performed. (F) Gene list was generated from DEGs and
GO analysis was performed. CAF, cancer-associated fibroblast;
α-SMA, α-smooth muscle actin; HIF-1α, hypoxia-inducible factor-1α;
DEG, differentially expressed gene; GO, Gene Ontology; KEGG, Kyoto
Encyclopedia of Genes and Genomes. |
GO and Kyoto Encyclopedia of Genes and Genomes
pathway analyses revealed that genes upregulated in hypoxic CAFs
were markedly enriched in pathways related to ossification,
skeletal development and the phosphoinositide 3-kinase-AKT
signaling pathway (PI3K-AKT) signaling pathway (Fig. 3E and F).
Identification of PTHrP as a key
factor in hypoxic CAFs
Given the critical role of crosstalk between CAFs
and tumor cells in the TME, it was aimed to elucidate common
hypoxic response mechanisms across the entire TME. By identifying
genes whose expression was altered not only in hypoxic CAFs but
also in hypoxic CRC cells, the aim was to identify key factors
involved in CAF-cancer cell interactions that contribute to tumor
progression and drug resistance. Among the upregulated genes in
hypoxic CAFs, 59 genes were identified that were also upregulated
in hypoxic CRC cell lines (Table
SII). From these 59 genes, those strongly associated with
vitamin D, bone metabolism and the PI3K-AKT signaling pathway were
selected, factors previously identified as crucial through hypoxic
CAF secretome analysis and pathway analysis, as candidates for
further investigation. Based on these findings, PTHrP was
identified as a key factor in hypoxic CAFs and designated as the
primary candidate for further analysis.
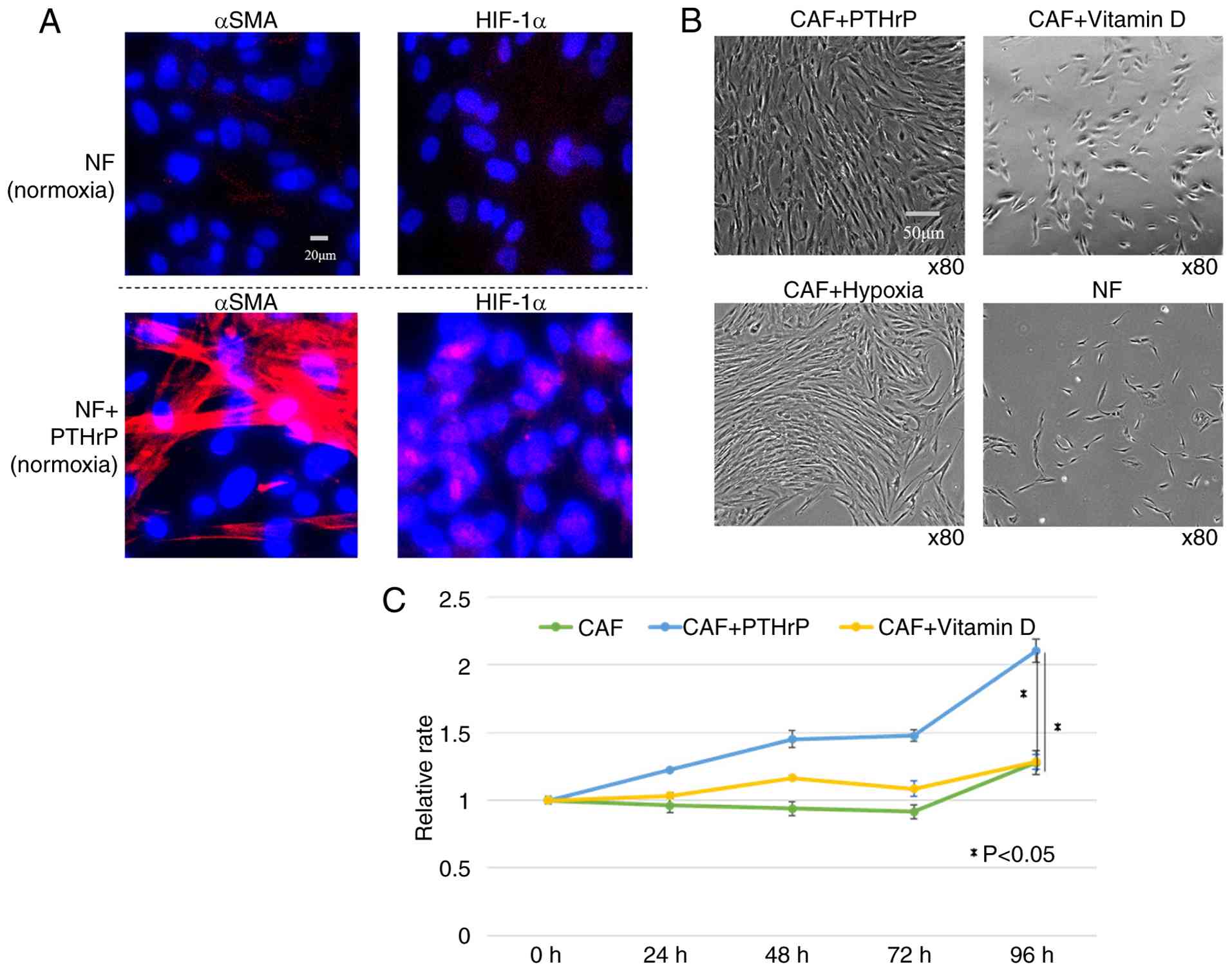
Effect of PTHrP on NF and effect of
PTHrP and vitamin D on CAF
When PTHrP was added to NFs under normoxic
conditions, both α-SMA and HIF-1α expression increased (Fig. 4A). In CAFs, PTHrP treatment induced
morphological changes similar to those observed in hypoxic CAF,
with cells transitioning from a spindle-shaped to a more flattened
morphology and forming layered structures (Fig. 4B), along with enhanced proliferation
capacity (Fig. 4C). DBP, identified
through shotgun analysis of the conditioned medium from hypoxic
CAFs, has been reported to reduce tissue vitamin D levels (23), which are known to exert anticancer
effects (24). Given that vitamin D
has been reported to downregulate PTHrP expression (25), vitamin D was added to the CAF
culture medium to investigate possible attenuation of their
CAF-like features. Vitamin D treatment led to morphological
reversion of CAFs to an NF-like appearance (Fig. 4B), although no significant
suppression of proliferation was observed (Fig. 4C).
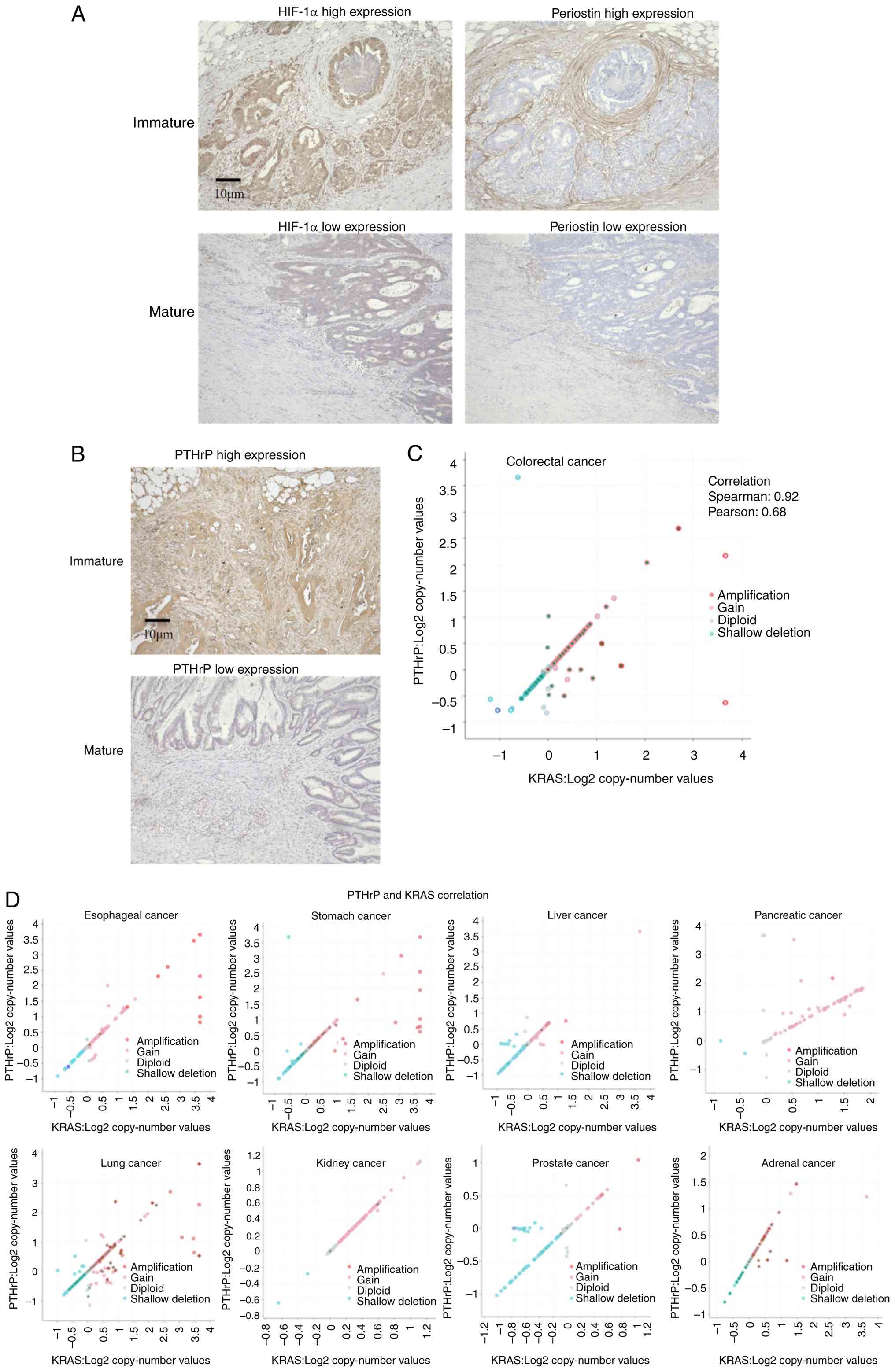
Immunostaining analysis of DR patterns
in patients with CRC
Immunohistochemical analysis of DR patterns (16
mature, nine immature) revealed significantly higher expression of
HIF-1α and periostin, one of the reported CAF markers, in the
immature group, indicating that these tumors were exposed to a more
hypoxic microenvironment (Fig. 5A;
Table II). To perform a more
detailed analysis, an expanded cohort of 59 cases (43 mature, 16
immature) was examined. In this cohort, the immature group showed
significantly higher PTHrP expression in both cytoplasm of tumor
cells at the invasive front and the surrounding stromal areas.
Since RAS mutation has been reported as one of the upstream
regulators of PTHrP (25), the
results of genetic testing performed on tumor tissue samples were
also analyzed. Among the 59 cases, 43 had undergone testing for RAS
and B-Raf proto-oncogene mutations and microsatellite instability
status, with KRAS mutations found in 20 cases and neuroblastoma RAS
viral oncogene homolog mutation in one case (Table SIII) and MSI testing results showed
0% (0/13) MSI-H cases in the immature DR group and 10.0% (3/30) in
the mature DR group, comparable to the reported 5–10% prevalence in
general CRC populations. This suggests that the current cohort is
not uniquely biased toward MSS cases.
 | Table II.Correlation between desmoplastic
reaction and clinicopathological factors including HIF-1α and
periostin expression in CRC samples. |
Table II.
Correlation between desmoplastic
reaction and clinicopathological factors including HIF-1α and
periostin expression in CRC samples.
|
| Desmoplastic
reaction |
|
|---|
|
|
|
|
|---|
| Variable | Immature (n=9) | Mature (n=16) | P-value |
|---|
| Patient
characteristics |
|
|
|
| Age at
surgery (years), [IQR] | 71.2 [46-88] | 67.9 [56-84] | 0.394 |
| Male
sex | 5 (55.6%) | 7 (43.8%) | 0.691 |
| Tumor
characteristics |
|
|
|
|
Location (right/left) | 3/6 | 5/11 | 0.915 |
| T stage
(1–2/3–4) | 0/9 | 2/14 | 0.520 |
| N stage
(0/1–3) | 3/6 | 8/8 | 0.677 |
| Venous
invasion | 5 (55.6%) | 10 (62.5%) | 0.734 |
|
Lymphatic invasion | 4 (44.4%) | 8 (50.0%) | 0.789 |
|
Perineural invasion | 6 (66.7%) | 7 (43.8%) | 0.411 |
| HIF-1α
high expression | 6 (66.7%) | 3 (18.8%) | 0.0308 |
|
Periostin high expression | 8 (88.9%) | 7 (43.8%) | 0.0405 |
RAS mutations were found to be significantly more
frequent in the immature group (Fig.
5B; Table III). Subgroup
analysis based on PTHrP expression levels in both the cancer cells
at the invasive front and the surrounding stroma showed a positive
correlation between PTHrP expression and RAS mutation status
(Tables IV and SII).
 | Table III.Correlation between desmoplastic
reaction and clinicopathological factors including PTHrP expression
or genetic testing in CRC samples. |
Table III.
Correlation between desmoplastic
reaction and clinicopathological factors including PTHrP expression
or genetic testing in CRC samples.
|
| Desmoplastic
reaction |
|
|---|
|
|
|
|
|---|
| Variable | Immature
(n=16) | Mature (n=43) | P-value |
|---|
| Patiet
characteristics |
|
|
|
| Age at
surgery (years), [IQR] | 69.9 [46-88] | 71.7 [40-87] | 0.578 |
| Male
sex | 5 (45.5%) | 21 (48.8%) | 0.255 |
| Tumor
characteristics |
|
|
|
|
Location (right/left) | 8/8 | 17/26 | 0.559 |
| T stage
(1–2/3–4) | 0/16 | 2/41 | 0.256 |
| N stage
(0/1–3) | 6/10 | 25/18 | 0.241 |
| Venous
invasion | 11 (68.8%) | 23 (53.5%) | 0.380 |
|
Lymphatic invasion | 7 (43.8%) | 18 (41.9%) | 0.976 |
|
Perineural invasion | 8 (50.0%) | 12 (27.9%) | 0.132 |
| PTHrP
high expression | 12 (75.0%) | 13 (30.2%) | 0.0029 |
| Genetic testing
performed | 13 (81.3%) | 30 (69.8%) | - |
| RAS
mutation | 10/13 (76.9%) | 11/30 (36.7%) | 0.0217 |
| BRAF
mutation status | 0/13 (0%) | 3/30 (10.0%) | 0.542 |
| MSI-H
cases, n (%) | 0/13 (0%) | 3/30 (10.0%) | 0.542 |
 | Table IV.A subgroup analysis examining the
correlation between clinicopathological factors and two groups
divided based on high and low expression of PTHrP. |
Table IV.
A subgroup analysis examining the
correlation between clinicopathological factors and two groups
divided based on high and low expression of PTHrP.
|
| PTHrP
expression |
|
|---|
|
|
|
|
|---|
| Variable | Low (n=34) | High (n=25) | P-value |
|---|
| Tumor
characteristics |
|
|
|
|
Location (right/left) | 14/20 | 11/14 | 0.828 |
| T stage
(1–2/3–4) | 2/32 | 0/25 | 0.503 |
| N stage
(0/1–3) | 18/16 | 13/12 | 0.943 |
| Venous
invasion | 18 (52.9%) | 16 (64.0%) | 0.435 |
|
Lymphatic invasion | 15 (44.1%) | 11 (44.0%) | 0.993 |
|
Perineural invasion | 11 (32.4%) | 9 (36.0%) | 0.788 |
| Genetic testing
performed | 22 (64.7%) | 21 (84.0%) | - |
| RAS
mutation | 7/22 (31.8%) | 14/21 (66.7%) | 0.0337 |
Correlation between copy number
variations of PTHrP and KRAS
To further investigate the association between PTHrP
expression and RAS mutation observed in tumor tissue, the
relationship between PTHrP and KRAS was analyzed using public
datasets. Both PTHrP and KRAS are located on chromosome 12.
Analysis of CNVs in patients with CRC using TCGA data revealed a
significant positive correlation between KRAS and PTHrP copy number
(R=0.92; P<0.001; Fig. 5C).
Similar trends were observed across multiple cancer types (Fig. 5D).
Furthermore, copy number correlations were analyzed
for other known driver genes located on chromosome 12, CDK4 and
MDM2, and a very strong positive correlation was found between them
(Spearman's r=0.91; P=5.17×10−196; Fig. S2A). Strong copy number correlations
were also observed between VDR, which is located on the same
chromosome, and both PTHrP (Fig.
S2B) and KRAS (Fig. S2C;
PTHrP, Spearman's r=0.77; P=7.13×10−106; KRAS,
Spearman's r=0.78; P=5.90×10−111). These results support
the possibility that the co-amplification of PTHrP and KRAS is a
passenger event associated with regional chromosomal gains on
chromosome 12, and also suggest potential involvement of other
functional relationships, such as vitamin D signaling through
VDR.
Discussion
In the present study, the focus was on the hypoxic
microenvironment, which is known to promote cancer progression, and
the aim was to elucidate the phenotypic changes of CAFs under
hypoxia and their underlying mechanisms. The results demonstrated
that hypoxia significantly enhanced the proliferative capacity of
both CAFs and NFs. Furthermore, the conditioned medium from
hypoxia-cultured CAFs significantly promoted chemoresistance and
migratory ability in CRC cell lines.
CAFs are a major component of the stromal cell
population within the TME (1), and
their characteristics are notably influenced by crosstalk with
tumor cells (5). In CRC, the DR
classification, categorized as immature, intermediate, or mature,
has been used as a prognostic indicator (8). Since CAFs constitute the tumor stroma
and interact closely with cancer cells, evaluating CAF activity and
the stromal status using the DR classification is considered
important. CAFs release various factors that modify the TME, and
these changes are likely reflected in the DR classification.
Hypoxia, a common feature observed across numerous
solid tumors (10), influences
critical aspects of cancer biology, including cellular invasion,
distant metastasis and regulation of cell death processes (11). In the present study, a marked
increase in DBP was observed in the conditioned medium of
hypoxia-cultured CAFs, whereas collagen and fibronectin levels were
reduced. The decreased levels of these extracellular matrix (ECM)
components may be explained by HIF-1α upregulation promoting ECM
degradation, consistent with the current finding that HIF-1α-high
CAF clusters exhibited increased MMP2 expression. DBP binds to
vitamin D metabolites and transports them to organs such as the
liver, thereby reducing free vitamin D concentrations (23). As a result, increased DBP levels in
tissue can decrease local vitamin D availability.
Single-cell analysis revealed enrichment of bone
metabolism-related genes in hypoxic CAFs. Bone metabolism-related
factors have garnered attention as therapeutic targets in solid
tumors (26,27), and vitamin D, in particular, is
known to exert suppressive effects on cancer. Increased total
vitamin D intake has been reported to be associated with a reduced
risk of early-onset CRC and its precursors (24). These findings suggest that the
vitamin D-related network is one of the critical factors directly
associated with cancer aggressiveness.
In the current study, the focus was on PTHrP as a
factor through which hypoxic CAFs may promote tumor malignancy.
Transcription of the PTHrP gene is suppressed by 1,25(OH)2D
and hypocalcemic vitamin D analogs, and this suppression may help
alleviate hypercalcemia caused by tumor-derived overproduction of
PTHrP (25). Moreover, PTHrP
gene activation is associated with malignant transformation in
normal mammalian cells, with Ras and p53 identified as key upstream
regulators of PTHrP transcription (28).
In CRC, frequent abnormalities have been reported in
major intracellular signaling pathways, including those involving
Ras and p53 (29). In some patients
with gastrointestinal cancers showing p53 immunoreactivity, vitamin
D supplementation has been reported to reduce the risk of
recurrence or death (30).
PTHrP was selected as a key factor in CAFs under
hypoxic conditions. PTHrP has also been reported as one of the
major causes of hypercalcemia in cancer-bearing states (31). Since vitamin D and PTHrP are closely
involved in cancer physiology in vivo, it is strongly
suggested that they may also influence CAFs in the TME.
Immunohistochemical analysis of DR-classified
samples showed that the positivity rates of HIF-1α, a hypoxia
marker (32), and periostin, which
is also considered a CAF marker (33), were higher in the immature group,
suggesting that this group is exposed to a more severe hypoxic
environment. PTHrP expression was positively correlated with the
prognostically poor immature type, confirming its association with
hypoxia.
RAS mutations in tumor cells are important factors
that influence the biological behavior of CRC (34), and RAS-mutant CRCs exhibit distinct
characteristics, making the development of novel therapies highly
anticipated (35). The RAS mutation
rate was elevated in both the immature type of the DR
classification and the high-PTHrP expression group. These findings
suggest a potential association between KRAS-mutant CRC and both
the immature stromal type and high PTHrP expression.
In PCa, where KRAS mutations are frequently
observed, DR caused by CAFs is prominent, and the resulting dense
stroma inhibits angiogenesis, leading to hypoxic regions within the
tumor (36). In addition to KRAS
amplification, amplification of the PTHLH gene, which
encodes PTHrP, has also been observed in patients with PCa, and
PTHrP has been reported to promote tumor growth and metastasis
(37).
In the present study, correlation analysis across
various cancer types revealed a strong association between the copy
numbers of KRAS and PTHrP. Both KRAS and PTHrP are located on
chromosome 12.
In summary, CAFs under hypoxic conditions exhibit
tumor-promoting properties, and single-cell analysis revealed that
PTHrP is one of the key factors involved. In addition, hypoxic CAFs
increase the secretion of DBP, leading to a reduction in the local
concentration of vitamin D around the tumor. Similar to the known
relationship between PTH and vitamin D, a potential interaction
between PTHrP and vitamin D was also suggested. Immunohistochemical
analysis indicated that CRC tissues with high PTHrP expression are
exposed to a more hypoxic environment, and these features were also
strongly correlated with KRAS mutations. Furthermore, analysis of
public datasets showed a strong correlation between the copy
numbers of PTHrP and KRAS, suggesting that the PTHrP-vitamin D-RAS
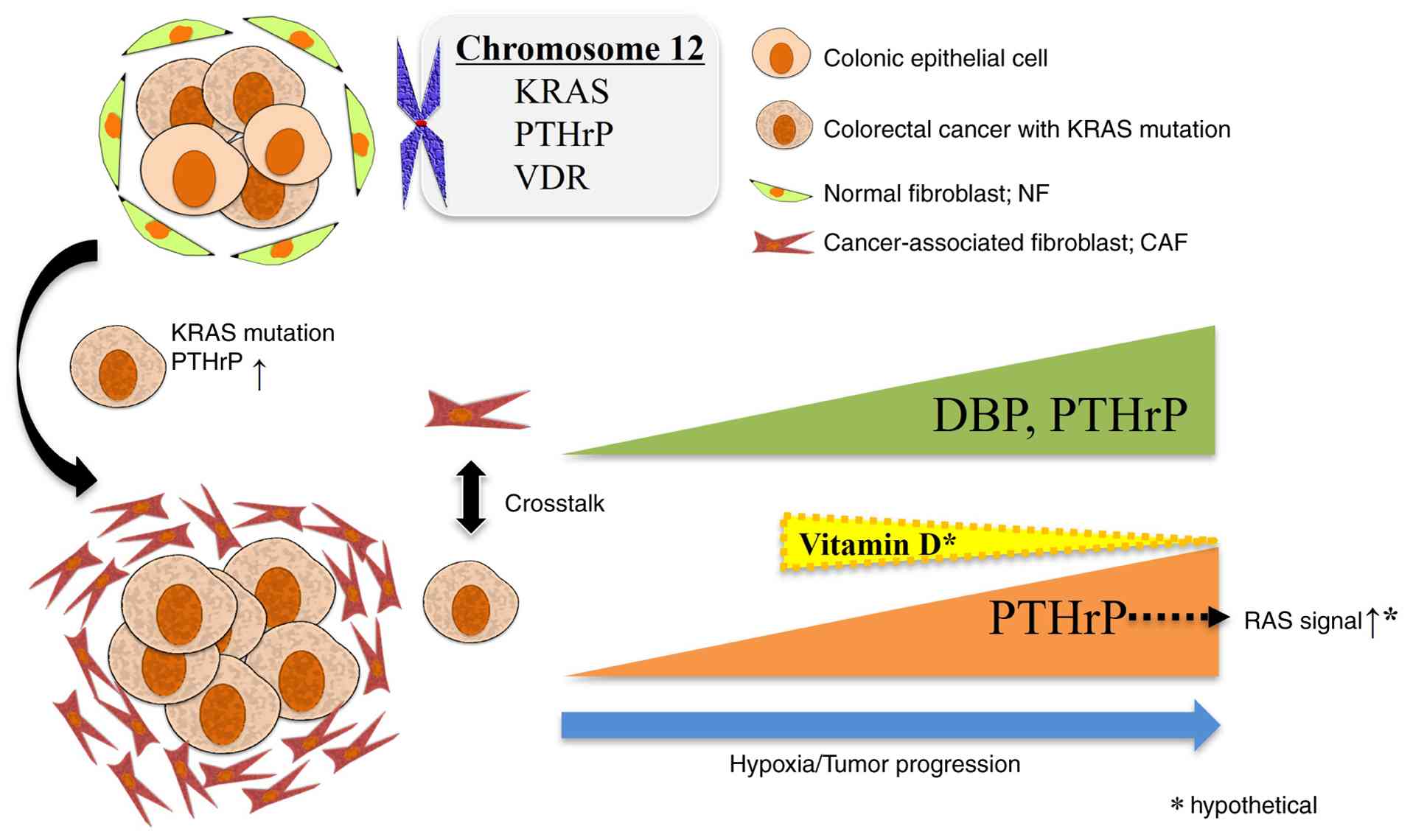
axis may be a key regulatory pathway involving CAFs.
One limitation of the present study is that vitamin
D levels were not evaluated, as quantification of vitamin D within
tissue samples is technically challenging. The concentration of
vitamin D in resected CRC specimens is likely influenced by various
factors, including the absorption status of intestinal epithelium
at the time of surgery and the circulating levels of vitamin D.
Therefore, it is challenging to accurately assess tissue vitamin D
levels and their impact on CAFs.
These findings suggest a potential PTHrP-vitamin
D-RAS axis in the function of CAFs, in which hypoxia-induced PTHrP
expression in tumor cells is associated with enhanced RAS
signaling. This axis may provide new insights into CAF biology and
TME interactions (Fig. 6).
The present study has several limitations that
should be considered. Fluorescence images were evaluated based on
qualitative comparisons rather than quantitative measurements. This
approach was chosen because the cultured CAFs and NFs were not 100%
pure, and contaminating cell populations could limit the
reliability of quantitative analysis. Additionally, structural
differences between tissue samples and uneven antibody staining
made quantitative fluorescence measurements technically challenging
and potentially misleading.
Due to technical constraints, an indirect co-culture
system was employed in the present study, which limited the
assessment of direct CAF-tumor cell interaction pathways. This may
explain why our proteomic analysis did not reveal clear increases
in proliferation-related proteins despite observing enhanced
migration. Future studies employing direct co-culture systems and
pathway blockade assays using specific inhibitors would provide
valuable insights into the cytokine profiling and proliferation
signaling cascades involved in CAF-tumor cell interactions.
While focus was addressed on HIF-1α as the primary
hypoxic response indicator, other common canonical HIF targets such
as VEGF, CA9 and GLUT1 were not comprehensively assessed.
Single-cell analysis showed that clusters with high HIF-1α
expression also exhibited increased VEGF and GLUT1 expression,
while CA9 showed no marked increase. However, functional validation
of these targets and their roles in the hypoxic CAF phenotype was
not performed in the present study. Additionally, whether PTHrP is
a direct transcriptional target of HIF-1α in CRC remains unclear
and requires further investigation through chromatin
immunoprecipitation-sequencing or promoter assays. Future studies
should comprehensively evaluate the expression and functional
significance of canonical hypoxia regulators in CAFs and validate
the direct transcriptional regulation of PTHrP by HIF-1α.
Supplementary Material
Supporting Data
Supporting Data
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be found
in the Japan ProteOme STandard Repository under accession number
JPST004147 or at the following URL: https://repository.jpostdb.org/entry/JPST004147; in
the DDBJ Sequence Read Archive (DRA) under accession numbers
DRA023624 and DRA023649 or at the following URL (https://ddbj.nig.ac.jp/search); in the NCBI SRA
database under accession numbers DRA023624 and DRA023649 or at the
following URL: (https://www.ncbi.nlm.nih.gov/sra/?term=DRA023624
and http://www.ncbi.nlm.nih.gov/sra/?term=DRA023649).
Authors' contributions
SN and MU substantially contributed to the study
conceptualization and design. HK, MO, CK, NT and MTa contributed to
data acquisition. SN significantly contributed to data analysis and
interpretation, and manuscript drafting. SN and MU critically
revised the manuscript for important intellectual content. SN and
MU confirm the authenticity of all the raw data. All authors read
and approved the final version of the manuscript and agreed to be
accountable for all aspects of the work.
Ethics approval and consent to
participate
The present study was approved by the Human Ethics
Review Committee of the Graduate School of Medicine, University of
Osaka (approval nos. 15144 and 19020; Suita, Japan). Written
informed consent was obtained from all patients for the use of
their tissue samples for research purposes, including primary cell
isolation.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
AKT
|
protein kinase B
|
|
CAF
|
cancer-associated fibroblast
|
|
CNV
|
copy number variation
|
|
CRC
|
colorectal cancer
|
|
DBP
|
vitamin D-binding protein
|
|
DEG
|
differentially expressed gene
|
|
DMEM
|
Dulbecco's Modified Eagle Medium
|
|
DMSO
|
dimethyl sulfoxide
|
|
DR
|
desmoplastic reaction
|
|
EpCAM
|
epithelial cell adhesion molecule
|
|
FBS
|
fetal bovine serum
|
|
GO
|
Gene Ontology
|
|
HIF-1α
|
hypoxia-inducible factor 1-alpha
|
|
KRAS
|
Kirsten rat sarcoma viral oncogene
homolog
|
|
NCBI
|
National Center for Biotechnology
Information
|
|
NF
|
normal fibroblast
|
|
PBS
|
phosphate-buffered saline
|
|
PI3K-AKT
|
phosphoinositide 3-kinase-AKT
signaling pathway
|
|
PTH
|
parathyroid hormone
|
|
PTHrP
|
PTH-related protein
|
|
RAS
|
rat sarcoma oncogene
|
|
RRID
|
research resource identifier
|
|
TCGA
|
The Cancer Genome Atlas
|
|
TME
|
tumor microenvironment
|
|
VDR
|
vitamin D receptor
|
|
αSMA
|
alpha-smooth muscle actin
|
References
|
1
|
Luo H, Xia X, Huang LB, An H, Cao M, Kim
GD, Chen HN, Zhang WH, Shu Y, Kong X, et al: Pan-cancer single-cell
analysis reveals the heterogeneity and plasticity of
cancer-associated fibroblasts in the tumor microenvironment. Nat
Commun. 13:66192022. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Mhaidly R and Mechta-Grigoriou F: Role of
cancer-associated fibroblast subpopulations in immune infiltration,
as a new means of treatment in cancer. Immunol Rev. 302:259–272.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chen Y, McAndrews KM and Kalluri R:
Clinical and therapeutic relevance of cancer-associated
fibroblasts. Nat Rev Clin Oncol. 18:792–804. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Broz MT, Ko EY, Ishaya K, Xiao J, De
Simone M, Hoi XP, Piras R, Gala B, Tessaro FHG, Karlstaedt A, et
al: Metabolic targeting of cancer associated fibroblasts overcomes
T-cell exclusion and chemoresistance in soft-tissue sarcomas. Nat
Commun. 15:24982024. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wu F, Yang J, Liu J, Wang Y, Mu J, Zeng Q,
Deng S and Zhou H: Signaling pathways in cancer-associated
fibroblasts and targeted therapy for cancer. Signal Transduct
Target Ther. 6:2182021. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ueno H, Kajiwara Y, Ajioka Y, Sugai T,
Sekine S, Ishiguro M, Takashima A and Kanemitsu Y:
Histopathological atlas of desmoplastic reaction characterization
in colorectal cancer. Jpn J Clin Oncol. 51:1004–1012. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sandberg TP, Stuart MPME, Oosting J,
Tollenaar RAEM, Sier CFM and Mesker WE: Increased expression of
cancer-associated fibroblast markers at the invasive front and its
association with tumor-stroma ratio in colorectal cancer. BMC
Cancer. 19:2842019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Hu Q, Wang Y, Yao S, Mao Y, Liu L, Li Z,
Chen Y, Zhang S, Li Q, Zhao Y, et al: Desmoplastic reaction
associates with prognosis and adjuvant chemotherapy response in
colorectal cancer: A multicenter retrospective study. Cancer Res
Commun. 3:1057–1066. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Akimoto N, Väyrynen JP, Zhao M, Ugai T,
Fujiyoshi K, Borowsky J, Zhong R, Haruki K, Arima K, Lau MC, et al:
Desmoplastic reaction, immune cell response, and prognosis in
colorectal cancer. Front Immunol. 13:8401982022. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sueyama T, Kajiwara Y, Mochizuki S,
Shimazaki H, Shinto E, Hase K and Ueno H: Periostin as a key
molecule defining desmoplastic environment in colorectal cancer.
Virchows Arch. 478:865–874. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wilson WR and Hay MP: Targeting hypoxia in
cancer therapy. Nat Rev Cancer. 11:393–410. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Chi JT, Wang Z, Nuyten DS, Rodriguez EH,
Schaner ME, Salim A, Wang Y, Kristensen GB, Helland A,
Børresen-Dale AL, et al: Gene expression programs in response to
hypoxia: Cell type specificity and prognostic significance in human
cancers. PLoS Med. 3:e472006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Uemura M, Yamamoto H, Takemasa I, Mimori
K, Hemmi H, Mizushima T, Ikeda M, Sekimoto M, Matsuura N, Doki Y
and Mori M: Jumonji domain containing 1A is a novel prognostic
marker for colorectal cancer: In vivo identification from hypoxic
tumor cells. Clin Cancer Res. 16:4636–446. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Uemura M, Yamamoto H, Takemasa I, Mimori
K, Mizushima T, Ikeda M, Sekimoto M, Doki Y and Mori M:
Hypoxia-inducible adrenomedullin in colorectal cancer. Anticancer
Res. 31:507–514. 2011.PubMed/NCBI
|
|
15
|
Noda T, Yamamoto H, Takemasa I, Yamada D,
Uemura M, Wada H, Kobayashi S, Marubashi S, Eguchi H, Tanemura M,
et al: PLOD2 induced under hypoxia is a novel prognostic factor for
hepatocellular carcinoma after curative resection. Liver Int.
32:110–118. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yamamoto H, Tei M, Uemura M, Takemasa I,
Uemura Y, Murata K, Fukunaga M, Ohue M, Ohnishi T, Ikeda K, et al:
Ephrin-A1 mRNA is associated with poor prognosis of colorectal
cancer. Int J Oncol. 42:549–555. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wada H, Yamamoto H, Kim C, Uemura M, Akita
H, Tomimaru Y, Hama N, Kawamoto K, Kobayashi S, Eguchi H, et al:
Association between ephrin-A1 mRNA expression and poor prognosis
after hepatectomy to treat hepatocellular carcinoma. Int J Oncol.
45:1051–1058. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Munakata K, Uemura M, Takemasa I, Ozaki M,
Konno M, Nishimura J, Hata T, Mizushima T, Haraguchi N, Noura S, et
al: SCGB2A1 is a novel prognostic marker for colorectal cancer
associated with chemoresistance and radioresistance. Int J Oncol.
44:1521–1528. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kawai K, Uemura M, Munakata K, Takahashi
H, Haraguchi N, Nishimura J, Hata T, Matsuda C, Ikenaga M, Murata
K, et al: Fructose-bisphosphate aldolase A is a key regulator of
hypoxic adaptation in colorectal cancer cells and involved in
treatment resistance and poor prognosis. Int J Oncol. 50:525–534.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Schwörer S, Cimino FV, Ros M, Tsanov KM,
Ng C, Lowe SW, Carmona-Fontaine C and Thompson CB: Hypoxia
potentiates the inflammatory fibroblast phenotype promoted by
pancreatic cancer Cell-derived cytokines. Cancer Res. 83:1596–1610.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hashiguchi Y, Muro K, Saito Y, Ito Y,
Ajioka Y, Hamaguchi T, Hasegawa K, Hotta K, Ishida H, Ishiguro M,
et al: Japanese society for cancer of the colon and rectum (JSCCR)
guidelines 2019 for the treatment of colorectal cancer. Int J Clin
Oncol. 25:1–42. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Elyada E, Bolisetty M, Laise P, Flynn WF,
Courtois ET, Burkhart RA, Teinor JA, Belleau P, Biffi G, Lucito MS,
et al: Cross-species single-cell analysis of pancreatic ductal
adenocarcinoma reveals antigen-presenting cancer-associated
fibroblasts. Cancer Discov. 9:1102–1123. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Bouillon R, Schuit F, Antonio L and
Rastinejad F: Vitamin D binding protein: A historic overview. Front
Endocrinol (Lausanne). 10:9102019. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kim H, Lipsyc-Sharf M, Zong X, Wang X, Hur
J, Song M, Wang M, Smith-Warner SA, Fuchs C, Ogino S, et al: Total
vitamin d intake and risks of Early-onset colorectal cancer and
precursors. Gastroenterology. 161:1208–1217.e9. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Goltzman D, White J and Kremer R: Studies
of the effects of 1, 25-dihydroxyvitamin D on skeletal and calcium
homeostasis and on inhibition of tumor cell growth. J Steroid
Biochem Mol Biol. 76:43–47. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yang C, Tian Y, Zhao F, Chen Z, Su P, Li Y
and Qian A: Bone microenvironment and osteosarcoma metastasis. Int
J Mol Sci. 21:69852020. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Clézardin P, Coleman R, Puppo M, Ottewell
P, Bonnelye E, Paycha F, Confavreux CB and Holen I: Bone
metastasis: Mechanisms, therapies, and biomarkers. Physiol Rev.
101:797–855. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Motokura T, Endo K, Kumaki K, Ogata E and
Ikeda K: Neoplastic transformation of normal rat embryo fibroblasts
by a mutated p53 and an activated ras oncogene induces parathyroid
hormone-related peptide gene expression and causes hypercalcemia in
nude mice. J Biol Chem. 270:30857–30861. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Li XL, Zhou J, Chen ZR and Chng WJ: P53
mutations in colorectal cancer-molecular pathogenesis and
pharmacological reactivation. World J Gastroenterol. 21:84–93.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kanno K, Akutsu T, Ohdaira H, Suzuki Y and
Urashima M: Effect of Vitamin D supplements on relapse or death in
a p53-Immunoreactive subgroup with digestive tract cancer: Post hoc
analysis of the AMATERASU randomized clinical trial. JAMA Netw
Open. 6:e23288862023. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Mundy GR and Edwards JR: PTH-related
peptide (PTHrP) in hypercalcemia. J Am Soc Nephrol. 19:672–675.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zhao M, Wang S, Zuo A, Zhang J, Wen W,
Jiang W, Chen H, Liang D, Sun J and Wang M: HIF-1α/JMJD1A signaling
regulates inflammation and oxidative stress following hyperglycemia
and Hypoxia-induced vascular cell injury. Cell Mol Biol Lett.
26:402021. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Yang Y, Wu J, Zhu H, Shi X, Liu J, Li Y
and Wang M: Effect of hypoxia-HIF-1α-periostin axis in thyroid
cancer. Oncol Rep. 51:572024. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kerk SA, Papagiannakopoulos T, Shah YM and
Lyssiotis CA: Metabolic networks in mutant KRAS-driven tumours:
Tissue specificities and the microenvironment. Nat Rev Cancer.
21:510–525. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhu G, Pei L, Xia H, Tang Q and Bi F: Role
of oncogenic KRAS in the prognosis, diagnosis and treatment of
colorectal cancer. Mol Cancer. 20:1432021. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Brown BA, Myers PJ, Adair SJ, Pitarresi
JR, Sah-Teli SK, Campbell LA, Hart WS, Barbeau MC, Leong K, Seyler
N, et al: A Histone Methylation-MAPK signaling axis drives durable
Epithelial-mesenchymal transition in hypoxic pancreatic cancer.
Cancer Res. 84:1764–180. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Pitarresi JR, Norgard RJ, Chiarella AM,
Suzuki K, Bakir B, Sahu V, Li J, Zhao J, Marchand B, Wengyn MD, et
al: PTHrP drives pancreatic cancer growth and metastasis and
reveals a new therapeutic vulnerability. Cancer Discov.
11:1774–1791. 2021. View Article : Google Scholar : PubMed/NCBI
|