Introduction
Takayasu arteritis (TA, also known as ‘pulseless
disease’) was first described in 1908 by the Japanese
ophthalmologist, Mikito Takayasu, in the retinal vessels of a
patient. Currently, it is known that this rare type of vasculitis
(incidence, 0.3-4/million/year) predominantly affects young females
and is commonly associated with tenderness of the carotid artery,
arm claudication, ocular disturbances, weakening of pulses,
differences in blood pressure between arms, syncope and other
central nervous system symptoms (1). The diagnostic criteria for TA,
published in 1990 by the American College of Rheumatology (2), have a sensitivity of 91% and
specificity of 95% and suggest the presence of at least 3 of the
following: i) An age of <40 years; ii) claudication of the
extremities; iii) decreased brachial artery pulse; iv) differences
of >10 mmHg in systolic blood pressure between arms; v) bruit
over subclavian/aorta; and vi) arteriographic abnormalities
(3).
The underlying pathological processes in TA consist
of vascular wall inflammation, immune dysfunction via the
expression of human leukocyte antigen (HLA) antigens on vascular
endothelial cells (4),
particularly in patients with HLA-B*52, and the reduced number of
regulatory T (Treg) cells (5),
conjunctive tissue deterioration [activation of matrix
metalloproteinases (MMP)2, 3 and 9(6), the increased expression of vascular
cell adhesion molecule-1 (VCAM-1), e-Selectin and intercellular
cell adhesion molecule-1 (ICAM-1)] (7), and the stimulation of platelet
aggregation. The activation of leukocyte nuclear factor (NF)-κB is
followed by the secretion of pro-inflammatory cytokines, such as
interferon-α (IFN-α), tumor necrosis factor-α (TNF-α), and
interleukin (IL)-6, IL-8, IL-17A and IL-18; the serum levels of
these cytokines (IL-6, IL-12 and IL-18) are associated with disease
activity (1). The histopathology
of TA-affected blood vessels, more often observed in specimens
obtained by post-surgical excision and autopsies than biopsy, is
characterized by granulomatous inflammation, particularly in the
vasa vasorum and the medio-adventitial junction, edema, the
infiltration of mononuclear cells, laminar necrosis, and the
deterioration of elastic fibers and smooth muscle cells (3).
The trigger for vascular inflammation seems to be
the presence of circulating bacterial antigens and the subsequent
immune cross-reaction involving vascular endothelial cells
(4,8). There is a known molecular mimicry
between the human and mycobacterial 65-kDa heat shock protein (HSP)
and this molecule has been found in aortic biopsies (vasa vasorum
and media) from patients with TA. Moreover, patients with TA have
serum IgG antibodies against aortic endothelial cells directed
against mycobacterial and human 60-65 kDa HSP (9), and T cells are also active against
mycobacterial and human 65-kDa HSP (1). Apart from mycobacterium tuberculosis,
other pathogens may be involved in TA; elevated plasma levels of
pentraxin-related protein (PTX-3) are observed both in TA and in
infections with Aspergillus fumigatus, Neisseria meningitidis
and Pseudomonas aeruginosa (10).
The treatment of patients with TA aims to suppress
both systemic and vascular inflammation with either
corticosteroids, methotrexate, azathioprine, mycophenolate mofetil,
cyclophosphamide or leflunomide (11,12).
In refractory cases there are good results with biological agents,
antagonists of TNF-α and the Il-6 receptor (IL-6R), such as
ustekinumab, infliximab, etanercept, adalimumab, rituximab,
tocilizumab, and in pregnant patients treated with certolizumab
pegol (13). However, TA has also
been observed after the initiation of treatment with anti-TNF-α
agents, and thus far, two patients with spondyloarthritis, one
patient with rheumatoid arthritis, and one patient with Crohn's
disease who developed TA following treatment with anti-TNF-α agents
have been reported (13). Notably,
the natural stilbenoid, curcumin (14), and resveratrol (15) have been shown to exert favorable
effects on TA, probably due to their anti-TNF-α actions and their
promoting effects on circulatory Treg functions. Finally,
minocycline, a MMP inhibitor, in combination with steroids, has
also been shown to be capable of inducing remission (16).
In the advanced stages of TA, with significant
stenosis or the occlusion of blood vessels, invasive treatment is
performed as needed; angioplasty and stent placement are employed
on specific arterial sites with stenosis of <5 cm, while more
severe cases are more effectively treated with open surgery, which
has lower re-stenosis rates and improves long-term survival; pre-
and post-operative immunosuppression is crucial (17).
Case report
The patient presented herein was a 40-year-old
female with a history of tuberculosis and treatment for pleural
effusion 8 years prior, and a urinary tract infection with
Klebsiella, presented at the local county hospital
(Ramnicu-Valcea County Hospital, Ramnicu-Valcea, Romania) with left
hemiplegia and left central facial paresis. Computer tomography
revealed cortico-subcortical ischemia in the territory of the right
medial cerebral artery (MCA).
Upon admission to the Neurology Ward at Colentina
Hospital (Bucharest, Romania), the patient was alert, oriented,
unable to ambulate, in no acute distress, with a blood pressure
(BP) of 130/80 mmHg in the left arm, 70/50 mmHg in the right arm,
pulse of 50 bpm, O2 saturation of 96% in room air;
facial asymmetry with left facial paresis, no motor function of the
left arm, left leg exhibited dorsiflexion of the foot, could
maintain a seated position, deglutition was intact but with Foley
catheter for neurological bladder, positive Babinski sign and
Hoffman reflex on the left side, sensory deficit on the left side;
and a Barthel index of 5. Echocardiography revealed pericardial
effusion, normal systolic and diastolic function with an ejection
fraction of 60%.
An AngioCT revealed the occlusion of the right
subclavian and common carotid arteries, and stenosis of the left
common carotid artery. Temporal artery biopsy confirmed the
diagnosis of TA. Bloodwork analysis revealed the following: Normal
levels of electrolytes, lipid profiles, bilirubin, creatine kinase,
uric acid, creatinine, amylase, alkaline phosphatase, fibrinogen,
anti-pANCA, anti-Ro, anti-double cDNA, cardiolipin IgG and IgM, β-2
glycoprotein IgG, lupus anticoagulant and protein C; increase
levels of liver function tests-ALT 112 UI [upper limit of normal
(ULN) 31]; AST 42 (ULN 32); ESR 29-62 mm/h (ULN 20) leucocytosis
13x106/ml (ULN 10), anti-nuclear antibodies (ANA) 33
(ULN 20) UI/ml, INR 1,31 (ULN 1,14); and decreased levels of
hematocrit 11.3 g/dl [lower limit of normal (LLN) 11.7] protein S
(46%, LLN 60%).
The patient was placed on Medrol 16 mg bid, statin
and a proton pump inhibitor, fraxiparine 0.6 UI bid. After
approximately 1 week on this treatment, during which the patient
regained bladder control and the Foley catheter was taken out, the
patient was complaining about left-sided chest pains, referring to
the left arm, and she was transferred for urgent coronary
angiography.
Upon admission to the Angiomedica Hospital
(Bucharest, Romania) the patient exhibited diminished subclavicular
pulse on the right side, pulses present in both the left and right
radio-ulnar arteries, as well as both feet.
The carotid Doppler revealed the occlusion of common
carotid arteries bilaterally, as well as the occlusion of the right
external and internal carotid arteries, as well as the right
vertebral artery; the left internal and external carotid arteries
were patent, probably through the Willis circle.
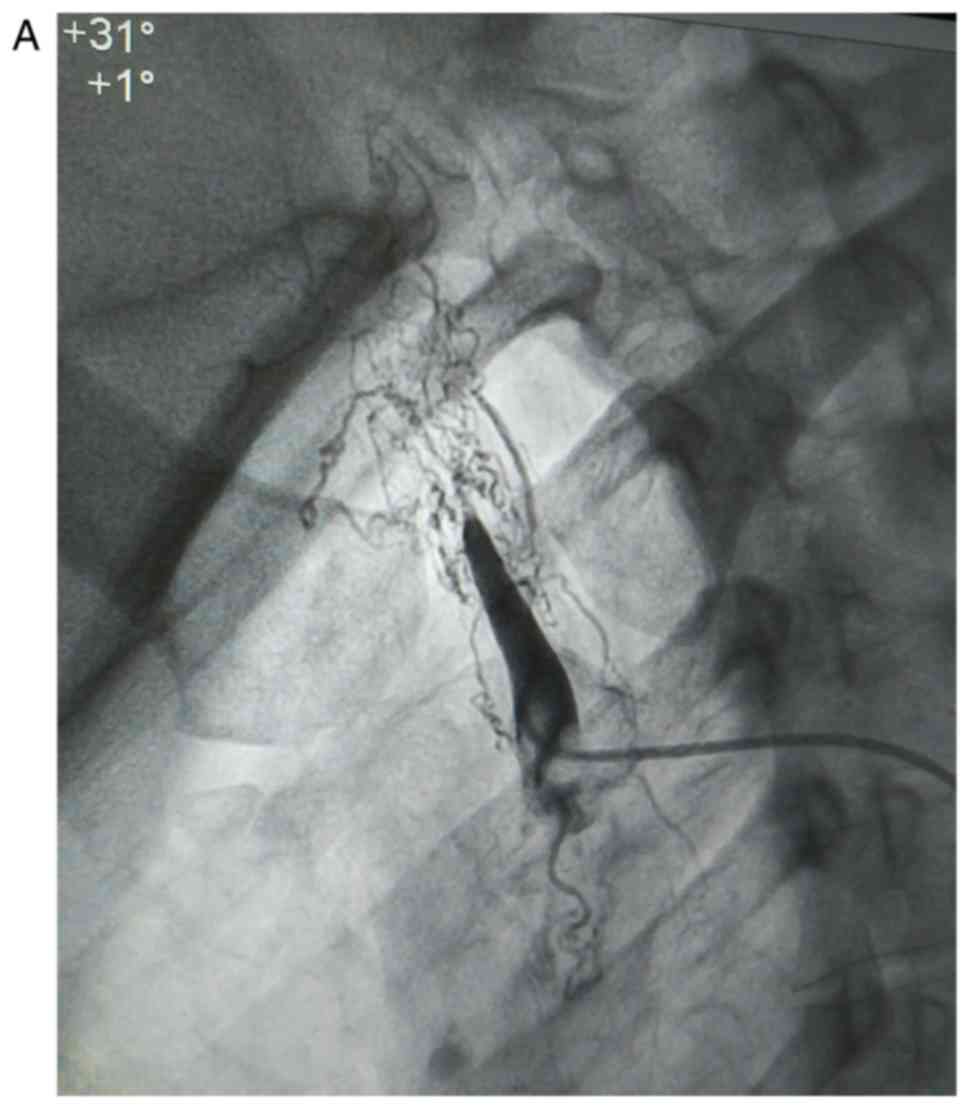
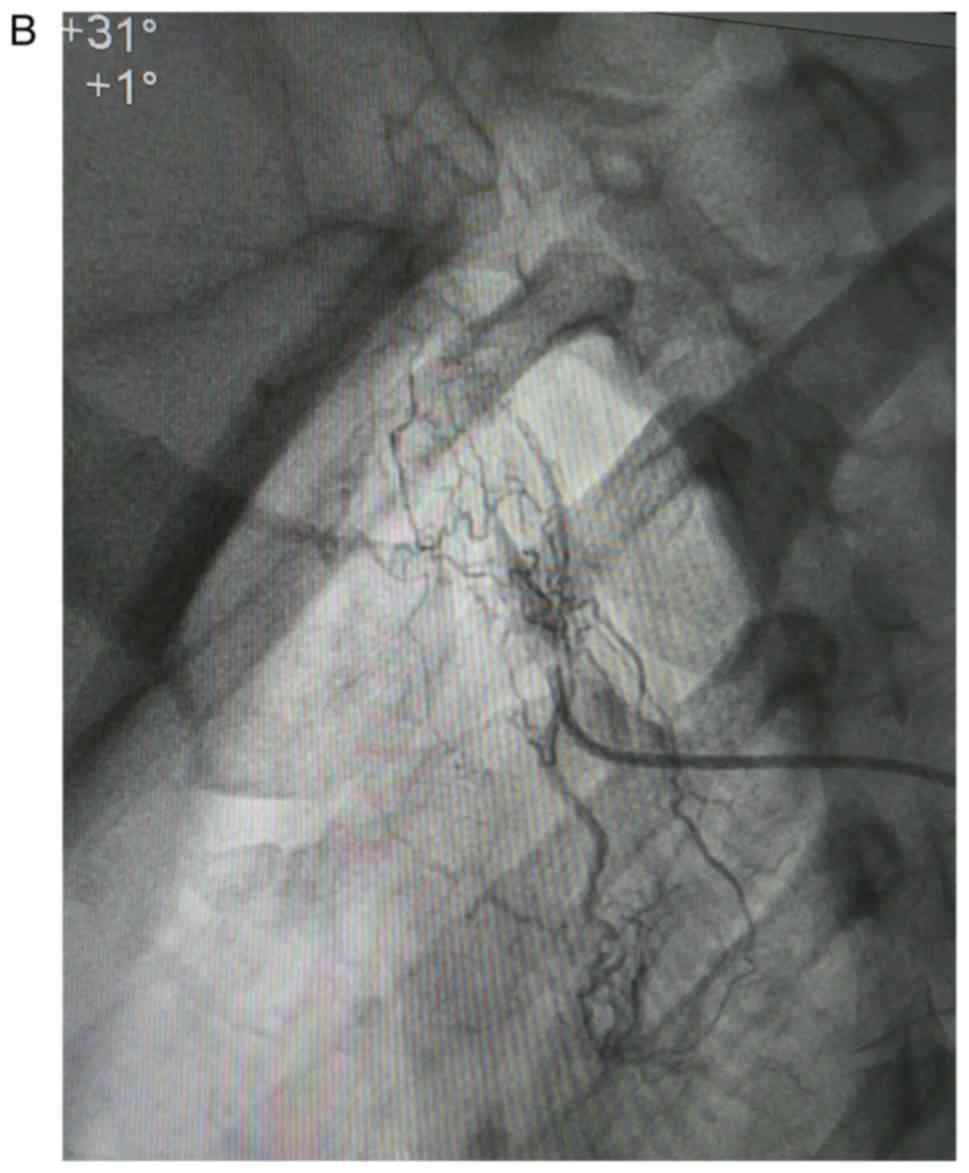
The coronary angiography revealed no coronary
stenosis and confirmed the occlusion of the common carotid arteries
(Fig. 1A-C; angiographs
illustrating the occlusion of the carotids).
Since endovascular treatment was excluded, the
patient was administered intravenous medication consisting of an
original combination of medicines which included dexamethasone 8 mg
(18) and the patient was planned
to be discharged the following morning in a long-term neurological
recovery institution.
That morning the patient was serendipitously
presented to the cardiovascular surgeon, Z.G., who was present by
chance in the angiography suite, and after discussing the case with
the interventional cardiologist and performing another vascular
Doppler on the carotids, patient and family were informed on the
surgical treatment and a decision was made to perform a
left-to-right vascular bypass.
Informed consent was obtained from both the patient
and the family prior to surgery; for the duration of the procedure
(approximately 8 h); conscious intravenous sedation was
administered by the anesthesiologist, M.F.; approximately 2 h into
the procedure, there was an episode of low blood pressure with the
patient responding well to administration of Voluven solution.
The vascular bypass used venous grafts harvested by
E.Q. and M.C. from the patient's both external saphenous veins and
it involved performing a bypass between the left external carotid
artery and the left subclavian artery in the inverted UZI
technique, and a vascular bypass was performed as well to the right
vertebral artery in the suboccipital triangle (via tunneling). An
image of the dissected suboccipital triangle was acquired (Fig. 2; intra-operative visualization of
the right vertebral artery in the suboccipital triangle).
The left-right shunt patency was examined intra- and
post-operatively with the presence of a pulse in the shunt, which
can be palpated superficially, under the skin, in the anterior neck
above the thyroid. Post-operatively, there were no new neurological
deficits, no complications or adverse reactions.
Another intravenous neurotrophic combination, which
included dexamethasone at 8 mg was administered post-surgery, after
which the patient presented with emesis as an adverse reaction; the
patient then remained stable and was not in any distress, and was
discharged in a neurologically stationary condition.
Notably, after approximately 1 week, the patient's
family reported that the patient had improved motor skills on the
left side, and at 3 months post-surgery the patient, was able to
ambulate with a cane and was filmed doing so during a follow-up
visit to the hospital. The motor improvement on the left arm was
minimal.
At 18 months follow-up, the patient has maintained
the motor abilities on the left leg and was able to perform
independently the activities of daily living (Barthel index of 60).
The patient is on Medrol 16 mg, and the persistent bradycardia is
being evaluated for pacemaker stimulation.
Discussion
Even though this patient was not tested for the
presence of 60-65 kDa HSP, nor for vascular endothelial antibodies,
given the patient's history and the sequence of events, it is very
likely that in this patient, the vasculitis was linked to the
presence of mycobacterial infection. The criteria for TA present in
this patient were age, pulse differential between left and right
arms, decreased pulse and ultrasound and angiographic arterial
stenosis. The left-sided chest pain for which the patient was
referred for coronary angiograph was most likely due to ischemic
conditions, either locally in the chest or referred neurological
pain; other possible causes include presence of pericardial
effusion and inflammation.
Due to the patient presenting with stroke, extensive
laboratory workup (TNF-α, IL-6, endothelial antibodies) was
superseded by the need for initiation of corticoid therapy and
anticoagulation, which was followed by a decrease of erythrocyte
sedimentation rate from 62 to 29 mmHg and improved bladder control
during initial care at the referring hospital.
Treatment options
Even though the majority of patients with TA are
treated with medication only, in the case of stenosed or occluded
arteries, vascular intervention offers the most effective long-term
treatment option, particularly for organ ischemia, hypertension and
aneurysmal disease (11,17,19,20).
Endovascular procedures (angioplasty and stents) are
less invasive and have lower complication rates than open surgical
procedures (vascular bypass). Moreover, newly developed
eprolimus-covered stents have proven to be similar in therapeutic
efficacy to coronary bypass surgery [EXCEL trial (21)], and they may also prove to be a
good therapeutic option in the earlier stages of TA, where there is
limited vascular damage.
However, in the long term, the restenosis rates for
endovascular procedures are higher, approximately 50% at 5 years,
and in some studies are as high as 80% (19,22)
compared to surgical revascularization, after which vascular
patency is preserved in approximately 80% of patients and the mean
graft patency is preserved for 9.4 years.
A specific feature of TA is that it occurs at a
younger age (<40 years) and for this reason, long-term vascular
patency becomes a critical issue when compared to atherosclerotic
disease; another strong argument in favor of vascular surgery is
the possibility of vessel dissection and/or rupture when baloon
angioplasty is employed (19).
Considering the issue of severe early complications
observed with vascular surgery (intracerebral hemorrhage,
hyperperfusion syndrome and cardiac tamponade), as well as its
superior long-term patency compared to endovascular procedures
(23), endovascular intervention
is recommended in the case of stenotic lesions with a length of
<5 cm, and respectively for longer occlusive lesions, bypass
surgery; these authors observed at 39 months a mean restenosis rate
of 53% for endovascular procedures and 12.5% for vascular bypass;
the most frequent symptom in these patients was brain ischemia
(23).
To identify the risk factors for complications of
vascular procedures, a clinical study on 66 patients with TA who
had 119 vascular procedures (arm claudication was the most frequent
symptom) was performed (24); in
these patients, there was a higher risk of complications and
restenosis observed in patients with hypertension, dyslipidemia and
patients treated with higher doses of steroid medications.
Active vs. inactive disease
For this patient presenting with stroke, prompt
revascularization proved to be an effective therapeutic option
irrespective of the TA activity status; however, it was observed
that vascular procedures, surgical or endovascular, have optimal
results when performed during inactive disease (11); the administration of steroids or
immunosuppressants before the procedures improved vascular
inflammation, deterioration of elastic fibers and aneurysm
formation; during active disease, the risk of restenosis,
dissection and complications is increased, and in commonly,
interventions are performed after active inflammation is controlled
(19).
Disease activity in TA can be inferred from
increased levels of serum IL-6 and TNFα and a higher uptake of
18F-Fluorodeoxyglucose [(18)F-FDG] in the arterial wall (25,22);
even so, distinguishing between systemic and vascular inflammation
is difficult and various evaluations have been suggested towards
this end: The Indian Takayasu Clinical Activity Score (12), while other authors have recommended
direct imaging of vessel wall thickening (ultrasound, CT,
angiogram) or positron emission tomography computerized tomography
(PET CT) (11).
Prognosis and survival
Based on the literature cited and due to the lack of
early complications and at 18 months, the medium- and long-term
prognostic for this patient was good. Supporting this assessment,
is a retrospective review (26)
which cites a 92-year-old patient who survived 23 years after
vascular bypass without immunotherapy, as well as an analysis of
106 consecutive patients with TA (20), in whom there was a 73.5% survival
rate at 20 years (congestive heart failure was most common
problem), and anastomotic aneurysm had a cumulative incidence of
13.8% at the 20-year follow-up, for which regular follow-up imaging
was recommended and concluded that for patients with occluded
arteries, surgery is the optimal long-term option.
Acknowledgements
The authors would like to thank Dr Roxana
Zmarandescu from Colentina Hospital for referring the patient
presented in this case report.
Funding
No funding was received.
Availability of data and materials
Data sharing is not applicable to this article, as
no datasets were generated or analyzed during the current
study.
Authors' contributions
ZG was involved in the conception and design of the
study, and in the planning and performing of the surgical
procedures, data collection and obtaining surgical images. FS was
involved in the conception and design of the study, and in the
writing of the manuscript, data collection and image processing;
DAS was involved in the conception and design of the study, and in
data analysis and reviewing/revisions; GL was involved in the
conception and design of the study, and in data analysis and
reviewing/revisions. MC was involved in the conception and design
of the study, and in data collection from surgical procedures. EQ
was involved in the conception and design of the study, and in data
collection from surgery, including images. MF was involved in the
conception and design of the study, and in data collection from
surgery. SK was involved in the conception and design of the study,
and in data analysis and reviewing/revisions. All authors have read
and approved the final manuscript.
Ethics approval and consent to
participate
Informed consent was obtained from both the patient
and the family prior to surgery.
Patient consent for publication
The patient provided consent prior to publication
for her data to become public.
Competing interests
DAS is the Managing Editor of the journal, but had
no personal involvement in the reviewing process, or any influence
in terms of adjudicating on the final decision, for this article.
The other authors declare that they have not competing
interests.
References
|
1
|
Russo RAG and Katsicas MM: Takayasu
arteritis. Front Pediatr. 6(265)2018.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Arend WP, Michel BA, Bloch DA, Hunder GG,
Calabrese LH, Edworthy SM, Fauci AS, Leavitt RY, Lie JT, Lightfoot
RW Jr, et al: The American college of rheumatology 1990 criteria
for the classification of Takayasu arteritis. Arthritis Rheum.
33:1129–1134. 1990.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Vaideeswar P and Deshpande JR: Pathology
of Takayasu arteritis: A brief review. Ann Pediatr Cardiol.
6:52–58. 2013.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Arnaud L, Haroche J, Mathian A, Gorochov G
and Amoura Z: Pathogenesis of Takayasu's arteritis: A 2011 update.
Autoimmun Rev. 11:61–67. 2011.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Kong X, Sun Y, Ma L, Chen H, Wei L, Wu W,
Ji Z, Ma L, Zhang Z, Zhang Z, et al: The critical role of IL-6 in
the pathogenesis of Takayasu arteritis. Clin Exp Rheumatol. 34
(Suppl):S21–S27. 2016.PubMed/NCBI
|
|
6
|
Wu G, Mahajan N and Dhawan V: Acknowledged
signatures of matrix metalloproteinases in Takayasu's. arteritis.
Biomed Res Int. 2014(827105)2014.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Tripathy NK, Chandran V, Garg NK, Sinha N
and Nityanand S: Soluble endothelial cell adhesion molecules and
their relationship to disease activity in Takayasu's arteritis. J
Rheumatol. 35:1842–1845. 2008.PubMed/NCBI
|
|
8
|
Kumar Chauhan S, Kumar Tripathy N, Sinha
N, Singh M and Nityanand S: Cellular and humoral immune responses
to mycobacterial heat shock protein-65 and its human homologue in
Takayasu's arteritis. Clin Exp Immunol. 138:547–553.
2004.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Chauhan SK, Tripathy NK and Nityanand S:
Antigenic targets and pathogenicity of anti-aortic endothelial cell
antibodies in Takayasu arteritis. Arthritis Rheum. 54:2326–2333.
2006.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Espinoza JL, Ai S and Matsumura I: New
insights on the pathogenesis of Takayasu arteritis: Revisiting the
microbial theory. Pathogens. 7(73)2018.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Misra DP, Wakhlu A, Agarwal V and Danda D:
Recent advances in the management of Takayasu arteritis. Int J
Rheum Dis. 22 (Suppl):60–68. 2019.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Keser G, Aksu K and Direskeneli H:
Takayasu arteritis: An update. Turk J Med Sci. 48:681–697.
2018.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Ataş N, Varan Ö, Babaoğlu H, Satiş H,
Bilici Salman R and Tufan A: Certolizumab pegol treatment in three
patients with Takayasu arteritis. Arch Rheumatol. 34:357–362.
2019.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Shao N, Jia H, Li Y and Li J: Curcumin
improves treatment outcome of Takayasu arteritis patients by
reducing TNF-α: A randomized placebo-controlled double-blind
clinical trial. Immunol Res. 65:969–974. 2017.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Shi G, Hua M, Xu Q and Ren T: Resveratrol
improves treatment outcome and laboratory parameters in patients
with Takayasu arteritis: A randomized double-blind and
placebo-controlled trial. Immunobiology. 222:164–168.
2017.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Matsuyama A, Sakai N, Ishigami M, Hiraoka
H and Yamashita S: Minocycline for the treatment of Takayasu
arteritis. Ann Intern Med. 143:394–395. 2005.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Mason JC: Surgical intervention and its
role in Takayasu arteritis. Best Pract Res Clin Rheumatol.
32:112–124. 2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Stancioiu F and Makk R: Post-stroke
recovery of motor function with a new combination of medicines-A
pilot study. EJMO. 3:167–181. 2019.
|
|
19
|
Jeong HS, Jung JH, Song GG, Choi SJ and
Hong SJ: Endovascular balloon angioplasty versus stenting in
patients with Takayasu arteritis: A meta-analysis. Medicine
(Baltimore). 96(e7558)2017.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Miyata T, Sato O, Koyama H Shigematsu H
and Tada Y: Long-term survival after surgical treatment of patients
with Takayasu's arteritis. Circulation. 108:1474–1480.
2003.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Stone GW, Sabik JF, Serruys PW, Simonton
CA, Généreux P, Puskas J, Kandzari DE, Morice MC, Lembo N, Brown WM
III, et al: Everolimus-eluting stents or bypass surgery for left
main coronary artery disease. N Engl J Med. 375:2223–2235.
2016.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Perera AH, Youngstein T, Gibbs RG, Jackson
JE, Wolfe JH and Mason JC: Optimizing the outcome of vascular
intervention for Takayasu arteritis. Br J Surg. 101:43–50.
2014.PubMed/NCBI View
Article : Google Scholar
|
|
23
|
Kim YW, Kim DI, Park YJ, Yang SS, Lee GY,
Kim DK, Kim K and Sung K: Surgical bypass vs endovascular treatment
for patients with supra-aortic arterial occlusive disease due to
Takayasu arteritis. J Vasc Surg. 55:693–700. 2012.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Labarca C, Makol A, Crowson CS, Kermani
TA, Matteson EL and Warrington KJ: Retrospective comparison of open
versus endovascular procedures for Takayasu arteritis. J Rheumatol.
43:427–432. 2016.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Arraes AE, de Souza AW, Mariz HA, Silva
NP, Torres IC, Pinto PN, Lima EN and Sato EI:
(18)F-Fluorodeoxyglucose positron emission tomography and serum
cytokines and matrix metalloproteinases in the assessment of
disease activity in Takayasu's arteritis. Rev Bras Reumatol Engl
Ed. 56:299–308. 2016.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Yoshida M, Zoshima T, Hara S, Mizushima I,
Fujii H, Yamada K, Sato Y, Harada K and Kawano M: A long-term
survival after surgical treatment for atypical aortic coarctation
complicating Takayasu arteritis with inactive disease at the
diagnosis: An appropriately treated autopsy case. Intern Med.
58:2241–2246. 2019.PubMed/NCBI View Article : Google Scholar
|