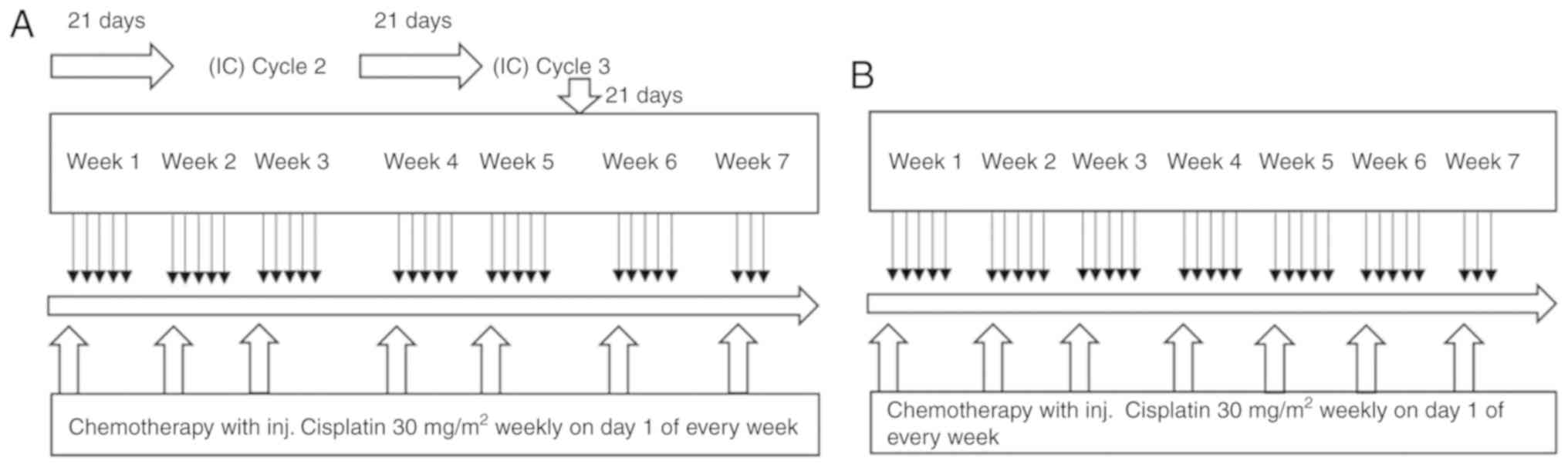
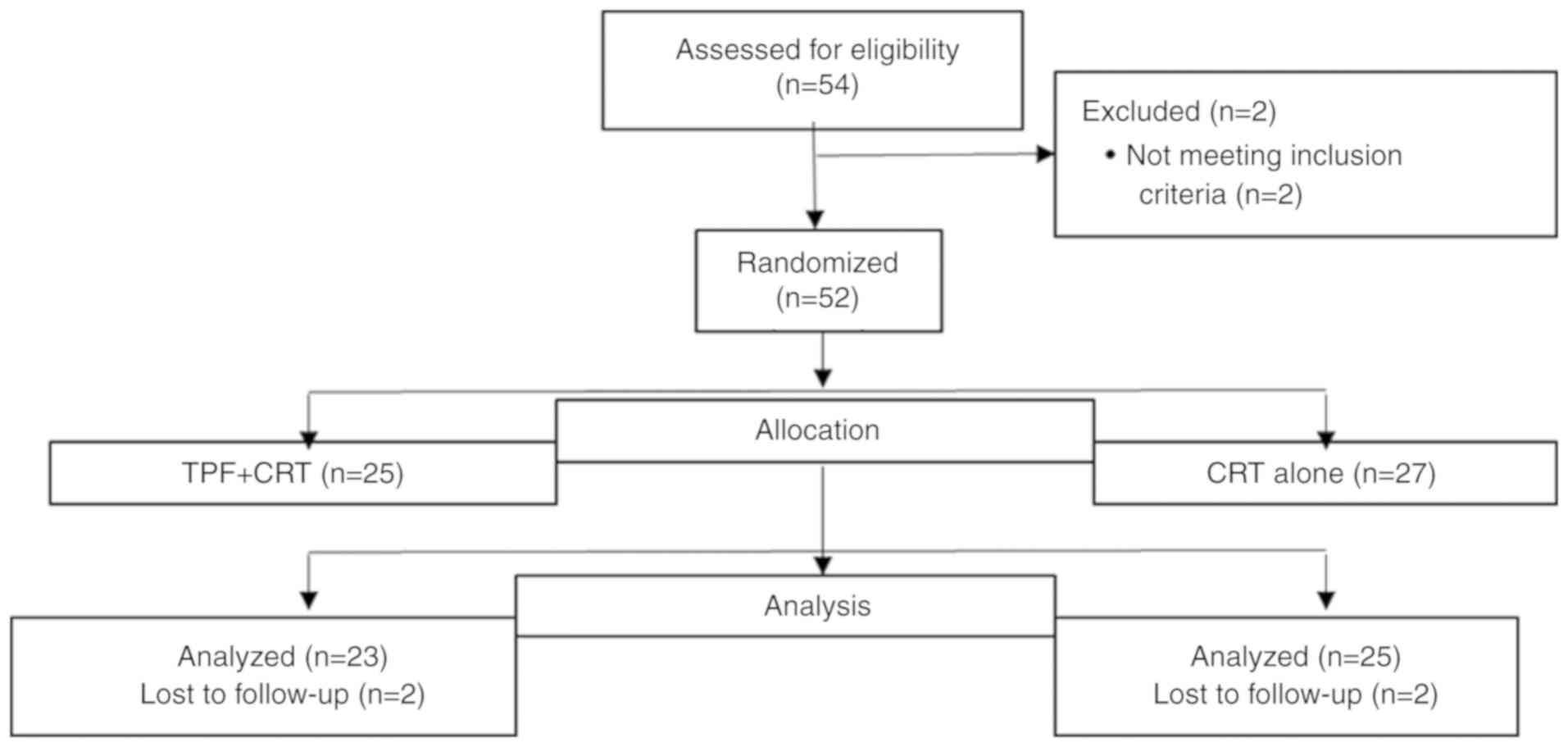
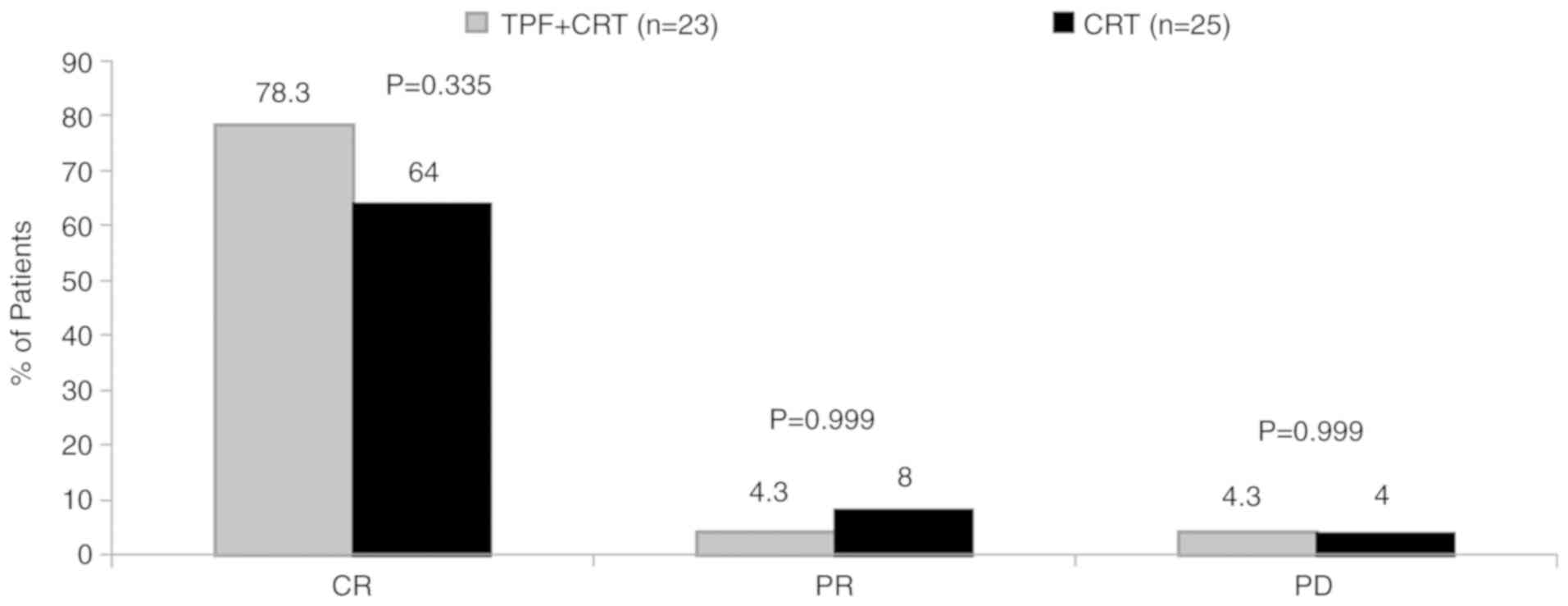
|
1
|
Vigneswaran N and Williams MD:
Epidemiologic trends in head and neck cancer and aids in diagnosis.
Oral Maxillofac Surg Clin North Am. 26:123–141. 2014.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Indian Council of Medical Research.
National Cancer Registry Programme: Ten year consolidated report of
the hospital based cancer registries 1984-1993. New Delhi.
https://ncdirindia.org/NCRP/Old_Reports/HBCR_TEN/inside_pages.pdf.
Accessed July 10, 2014.
|
|
3
|
Dhull AK, Atri R, Dhankhar R, Chauhan AK
and Kaushal V: Major risk factors in head and neck cancer: A
retrospective analysis of 12-year experiences. World J Oncol.
9:80–84. 2018.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Warnakulasuriya S: Global epidemiology of
oral and oropharyngeal cancer. Oral Oncol. 45:309–316.
2009.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Gupta N, Gupta R, Acharya AK, Patthi B,
Goud V, Reddy S, Garg A and Singla A: Changing trends in oral
cancer-a global scenario. Nepal J Epidemiol. 6:613–619.
2016.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Szturz P and Vermorken JB: Treatment of
elderly patients with squamous cell carcinoma of the head and neck.
Front Oncol. 6(199)2016.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Marur S and Forastiere AA: Head and neck
squamous cell carcinoma: Update on epidemiology, diagnosis, and
treatment. Mayo Clin Proc. 91:386–396. 2016.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Forastiere A, Koch W, Trotti A and
Sidransky D: Head and neck cancer. N Engl J Med. 345:1890–1900.
2001.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Posner MR, Haddad RI, Wirth L, Norris CM,
Goguen LA, Mahadevan A, Sullivan C and Tishler RB: Induction
chemotherapy in locally advanced squamous cell cancer of the head
and neck: Evolution of the sequential treatment approach. Semin
Oncol. 31:778–785. 2004.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Pignon JP, Bourhis J, Domenge C and
Designe L: Chemotherapy added to locoregional treatment for head
and neck squamous-cell carcinoma: Three meta-analyses of updated
individual data. MACH-NC Collaborative Group. meta-analysis of
chemotherapy on head and neck cancer. Lancet. 355:949–955.
2000.PubMed/NCBI
|
|
11
|
Dietz A, Wiegand S, Kuhnt T and Wichmann
G: Laryngeal preservation approaches: Considerations for new
selection criteria based on the DeLOS-II trial. Front Oncol.
9(625)2019.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Vermorken JB, Remenar E, van Herpen C,
Gorlia T, Mesia R, Degardin M, Stewart JS, Jelic S, Betka J, Preiss
JH, et al: Cisplatin, fluorouracil, and docetaxel in unresectable
head and neck cancer. N Engl J Med. 357:1695–1704. 2007.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Posner MR, Hershock DM, Blajman CR,
Mickiewicz E, Winquist E, Gorbounova V, Tjulandin S, Shin DM,
Cullen K, Ervin TJ, et al: Cisplatin and fluorouracil alone or with
docetaxel in head and neck cancer. N Engl J Med. 357:1705–1715.
2007.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Cox JD, Stetz J and Pajak TF: Toxicity
criteria of the Radiation therapy oncology group (RTOG) and the
European organization for research and treatment of cancer (EORTC).
Int J Radiat Oncol Biol Phys. 31:1341–1346. 1995.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Browman GP, Hodson DI, Mackenzie RJ,
Bestic N and Zuraw L: Cancer Care Ontario Practice Guideline
Initiative Head and Neck Cancer Disease Site Group. Choosing a
concomitant chemotherapy and radiotherapy regimen for squamous cell
head and neck cancer: A systematic review of the published
literature with subgroup analysis. Head Neck. 23:579–589.
2001.PubMed/NCBI View
Article : Google Scholar
|
|
16
|
Vermorken JB, Remenar E, ven Herpen C,
Lluch JG, Stewart S, Gorlia T, Degardin TG, Schollen K and Bernier
J: Standard cisplatin/infusional 5-fluorouracil (PF) vs. docetaxel
(T) plus PF (TPF) as neoadjuvant chemotherapy for nonresectable
locally advanced squamous cell carcinoma of the head andneck
(LA-SCCHN): A phase III trial of the EORTC head and neck cancer
group (EORTC #24971). J Clin Oncol. 22 (14 Suppl)(S5508)2004.
|
|
17
|
Ghi MG, Paccagnella A, Ferrari D, Foa P,
Alterio D, Codeca C, Nole F, Verri E, Orecchia R, Morelli F, et al:
Induction TPF followed by concomitant treatment versus concomitant
treatment alone in locally advanced head and neck cancer. A phase
II-III trial. Ann Oncol. 28:2206–2212. 2017.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Paccagnella A, Ghi MG, Loreggian L,
Buffoli A, Koussis H, Mione CA, Bonetti A, Campostrini F, Gardani
G, Ardizzoia A, et al: Concomitant chemoradiotherapy versus
induction docetaxel, cisplatin and 5 fluorouracil (TPF) followed by
concomitant chemoradiotherapy in locally advanced head and neck
cancer: A phase II randomized study. Ann Oncol. 21:1515–1522.
2010.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Cohen EE, Karrison TG, Kocherginsky M,
Mueller J, Egan R, Huang CH, Brockstein BE, Agulnik MB, Mittal BB,
Yunus F, et al: Phase III randomized trial of induction
chemotherapy in patients with N2 or N3 locally advanced head and
neck cancer. J Clin Oncol. 32:2735–2743. 2014.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Haddad R, O'Neill A, Rabinowits G, Tishler
R, Khuri F, Adkins D, Clark J, Sarlis N, Lorch J, Beitler JJ, et
al: Induction chemotherapy followed by concurrent chemoradiotherapy
(sequential chemoradiotherapy) versus concurrent chemoradiotherapy
alone in locally advanced head and neck cancer (PARADIGM): A
randomised phase 3 trial. Lancet Oncol. 14:257–264. 2013.PubMed/NCBI View Article : Google Scholar
|