Introduction
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection can generate a systemic disease known as coronavirus disease 2019 (COVID-19) (1). COVID-19 has led to the deaths of millions of individuals worldwide, and was thus declared a pandemic in early 2020(2). The most commonly reported symptoms are fever and dyspnea/respiratory distress (3). A number of different risk factors are associated with disease severity, such as smoking, chronic obstructive pulmonary disease (4-6), alcohol consumption (7), co-infections (8,9), metabolic diseases (10-12) and old age (13,14).
Nevertheless, to date, at least to the best of our knowledge, only a limited number of studies have identified SARS-CoV-2 infection in patients with sickle cell disease (SCD) (15,16). SCD is a common genetic disease (17-19) and previous research has failed to identify SCD as a risk factor for COVID-19(15). As has been previously demonstrated, COVID-19 may generate anemia (20) and hypercoagulation (21,22).
Patients with severe COVID-19 infection develop severe lymphopenia (23), increasing the susceptibility of patients for co-infection (24) and also develop multi-organ damage. Nevertheless, following SARS-CoV-2 clearance, the long-term effects of COVID-19 on the hematological and immune response remain unclear.
Due to the lack of knowledge on the immune response of patients with SCD during COVID-19 and the long-term effects of COVID-19(25), the present study describes the hematological alterations of a patient with SCD during hospitalization and at two posterior analyses, namely at 60 and 180 days following hospitalization.
Case report
The present study describes the case of a male patient, 42 years of age, previously diagnosed with SCD, homozygous for hemoglobin S, who regularly used folic acid at 5 mg/day. The patient reported a sore throat, body pain and fever (38,5˚C) at 2 days prior to hospitalization. At 1 day prior to hospitalization, he presented with mild dyspnea, a frontal headache and two diarrheal episodes. The patient was diagnosed with COVID-19 on the first hospitalization day, and a chest radiography did not reveal pulmonary ground-glass opacities, with only a small diffuse mosaic attenuation of the pulmonary parenchyma.
The patient was hospitalized in a special ward for COVID-19 patients at the University Hospital (Hospital das Clínicas, Faculty of Medicine, University of São Paulo-HCFMUSP) due to SARS-CoV-2 infection, diagnosed by the nasopharyngeal detection of SARS-CoV-2 RNA (E gene and N gene, with endogenous control with RNAseP), using reverse transcription-polymerase chain reaction (RT-PCR), with a detection limit of 40 copies of viral RNA/reaction (26). The equipment used for RT-PCR was the Abbott m2000sp nucleic acid extractors and the thermal cycler, Abbott m2000rt from Abbott Laboratories Inc. Importantly, the patient did not present any other parasitological, or viral, or bacterial infection during or post-hospitalization, at least to the best of our knowledge. The patient also did not present any comorbidities, such as chronic respiratory diseases, metabolic diseases, or cancer. The laboratory analysis was performed at the Central Laboratory of University Hospital (Central Laboratory Division-HC FMUSP), certified by the College of American Pathologists (CAP), and included the following: Complete blood counts (CBC), liver enzyme levels (alanine aminotransferase and aspartate aminotransferase), bilirubin and fractions (direct and indirect bilirubin), creatinine, C-reactive protein, urea and D-dimer levels, lactate dehydrogenase, total proteins and fractions (albumin and immunoglobulin), erythrocyte sedimentation rate and levels of iron, total iron-binding capacity and iron saturation, in arterial blood collected in K2EDTA collection tubes. Analyses were performed using a Cobas 8100 Automated Workflow Series with a post-analytical unit (Roche Diagnostics).
SARS-CoV-2 S1 IgG was performed by ELISA using the LIAISON® SARS-CoV-2 S1/S2 IgG kit with proper blank and standard concentration curve according to manufacturer's instructions (DiaSorin). The present study was approved by the Ethics Committee of HCFMUSP on 22/04/2020 (no. 30800520.7.0000.0068-2020). The patient provided written informed consent to participate in the study and for his clinical data to be published.
The day the patient was hospitalized was counted as day 0, the patient cleared SARS-CoV2 infection on day 14, and was discharged on day 16. Laboratory analyses then verified that the patient had a reduced number of erythrocytes, hemoglobin and hematocrit counts during and the post-SARS-CoV-2 clearance (Fig. 1A-C). Importantly, the patient was hospitalized with a low platelet count; however, during the COVID-19 infection, the platelet count normalized and remained within reference levels on day 60 (Fig. 1D). Nevertheless, post-discharge on day 180, the patient presented again with thrombocytopenia (Fig. 1D). At hospitalization, the patient presented a high leukocyte and neutrophil count with lymphopenia (Fig. 1E-G), with a high neutrophil-to-lymphocyte ratio (Fig. 1H). Of note, the eosinophil count peaked on days 60 and 180, and the monocyte count peaked on day 6 (Fig. 1I and J). Urea levels were high at hospitalization, but remained low thereafter (Fig. 1K). The levels of C-reactive protein peaked during hospitalization, and were high upon hospital release, but were low on days 60 and 180 (Fig. 1L). It is noteworthy that the creatinine and D-dimer levels peaked during the COVID-19 infection, but remained high even on days 180 and 60, respectively (Fig. 1M and N). The alanine aminotransferase, aspartate aminotransferase, albumin, globulin and total protein levels were within reference values during and following COVID-19 infection (Fig. 1O-S).
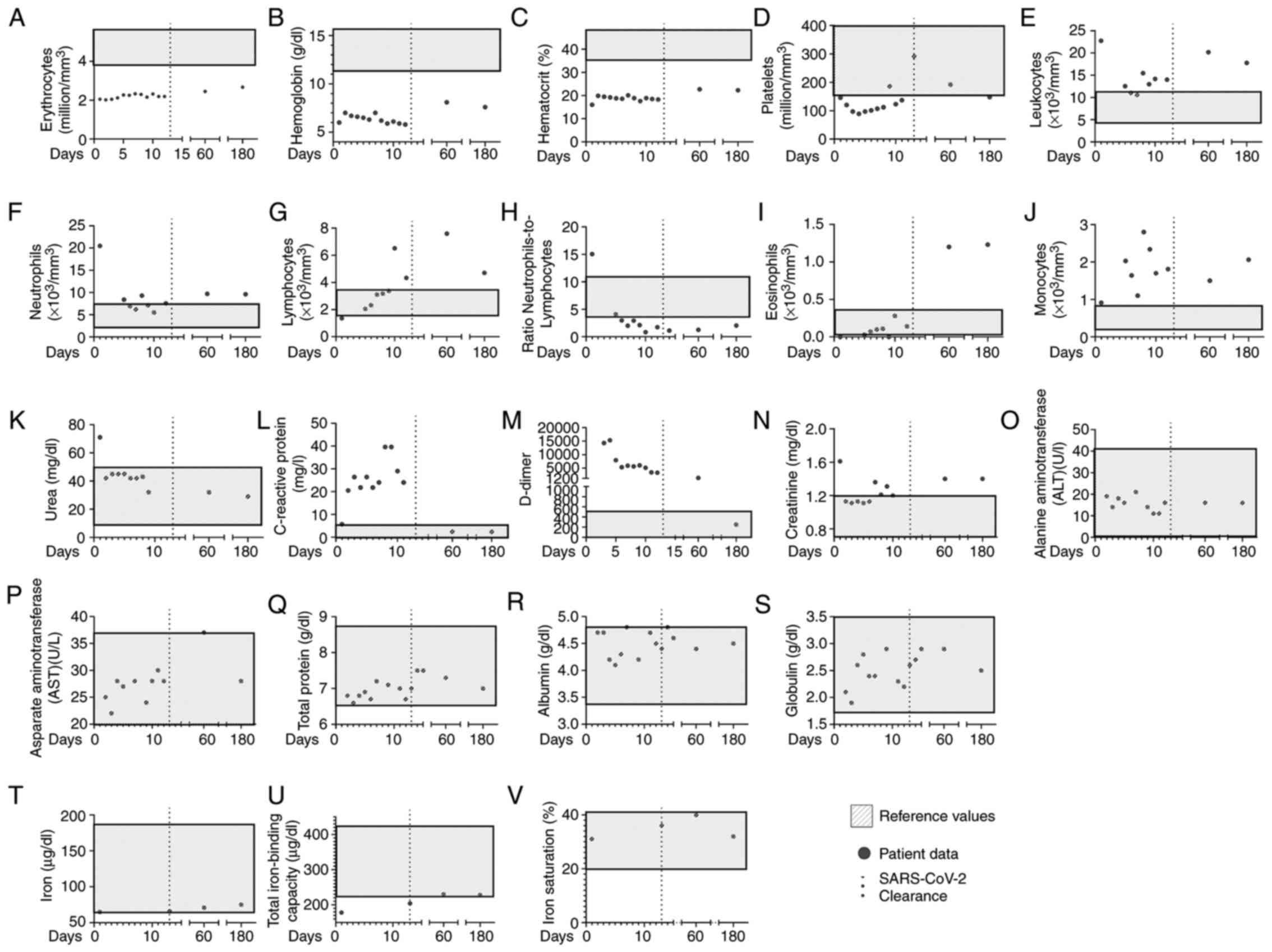 |
Figure 1
Clinical features of the patient, from the first day of hospitalization to 6 months following the onset of COVID-19 infection. Grey dots represent the following patient data: Blood levels of (A) erythrocytes, (B) hemoglobin, (C) hematrocrit, (D) platelets, (E) leukocytes, (F) neutrophils, (G) lymphocytes, (H) neutrophil-to-lymphocyte ratio, (I) eosinophils (J) monocytes, (K) urea, (L) C-reactive protein, (M) D-dimer, (N) creatinine levels, (O) alanine aminotransferase, (P) aspartate aminotransferase, (Q) total protein, (R) globulin and (S) total protein in peripheral blood, (T) iron levels, (U) total iron-binding capacity, and (V) iron saturation. The continuous dotted lines represent patient discharge from the hospital. The grey boxes represent reference values.
|
The blood glucose, glycated hemoglobin, potassium, magnesium, sodium, phosphorus, calcium, total bilirubin levels and fractions were maintained within normal levels during and post-hospitalization (data not shown) and to the best of our knowledge, patient was not infected by any bacterial or viral infection during and following COVID-19 hospitalization. On day 180, the platelet count was performed twice, due to excessive platelet aggregation. In addition, on day 180, the patient presented with a high anti-SARS-CoV-2 IgG level >200 UA/ml (data not shown).
The levels of the cardiac function marker, N-terminal pro b-type natriuretic peptide (NT pro-BNP), were 292 pg/ml (reference value, <125 pg/ml) on day 180 (data not shown); however, the pre-COVID-19 levels could not be confirmed. The ferritin levels were high at hospitalization (1,599 ng/ml), and on days 60 (884 ng/ml) and 180 (823 ng/ml), thus suggestive of a severe overload; however, the patient presented with 65 µg/dl of iron at hospitalization, 71 µg/dl on day 60 and 75 µg/dl on day 180 (Fig. 1T). The total iron-binding capacity was below reference values at hospitalization (178 µg/dl), but within the normal range on day 60 (230 µg/dl) and 180 (228 µg/dl) (Fig. 1U). In addition, iron saturation was 31% during COVID-19 infection, 40% on day 60 and 32% on day 180 post-COVID-19 infection (Fig. 1V).
Howell-Jolly corpuscles were found in the patient's blood samples during and post-COVID-19 resolution (days 60 and 180). Reticulocyte frequency was also high during hospitalization and on days 60 and 180 (>11%) (data now shown).
Discussion
Several risk factors have been associated with COVID-19 prognoses, such as chronic respiratory and inflammatory diseases, metabolic disorders and old age (4,10,27,28). However, to date, little is known about the clinical response of patients with SCD during and post-COVID-19 infection.
The literature presents conflicting results involving SCD and COVID-19. A few reports have identified an association between SCD and an increase in severity or COVID-19-mediated mortality (15,16), particularly in younger individuals (29). Previous studies on COVID-19 in patients with SCD have focused on clinical features (16) and laboratory values at a single time point (15). Several investigations on COVID-19 have also evaluated patients at a single time point (7,30,31). Nevertheless, the longitudinal analysis of clinical data may provide a better overview of the anti-SARS-CoV-2 response of patients (4).
Other reports did not identify SCD as a risk factor for COVID-19 (32-34), or COVID-19 as a risk factor for SCD-mediated vaso-oclusive crisis (35). Nevertheless, patients with SCD have a predisposition to the development of pulmonary hypertension and chronic kidney disease (36).
Importantly, SCD can generate a vascular-endothelial dysfunction, with increased oxidative stress, coagulation and inflammation (37). The pro-inflammatory status in SCD increases neutrophilic and platelet activation (38), and can be further increased during SARS-CoV-2 infection (39). Patients with SCD also present a partially dysfunction in regulatory T-cells (40), which can favor the hyperinflammatory status during COVID-19 infection (41). Other comorbidities, such as tobacco use and diabetes mellitus may also affect the outcome of patients with SCD (15,42). Recently, long COVID-19(43), or long-term COVID-19 sequelae have been extensively investigated (44-46).
The present study described the first report of a COVID-19 disease course in a patient with SCD and the long-term effects of COVID-19, at 2 and 6 months following SARS-CoV-2 infection and hospitalization. The patient presented with classical symptomatic COVID-19, without the need for mechanically assisted ventilation, and was released from the hospital upon SARS-CoV-2 clearance. During hospitalization, the anemia of the patient was further enhanced, probably by the effect of anemia of inflammation (47). COVID-19 patients can develop anemia during the disease course (20) and short-term immune alterations post-COVID-19 have been described (48). The patient described herein fully recovered after 2 weeks, with complete viral clearance, without mechanical ventilation or intensive care. During COVID-19 infection, the patient presented an elevated neutrophil-to-lymphocyte ratio, creatinine levels and C-reactive protein levels (15,28,49). A previous investigation on patients with SCD with COVID-19 infection identified alterations on alanine aminotransferase and direct bilirubin levels (15); however, the present study failed to observe these alterations in the patient described. Nevertheless, to date, to the best of our knowledge, no long-term effects have been investigated. Most importantly at 2 and 6 months following hospital discharge, the patient still presented alterations in certain inflammatory markers, mainly creatinine and D-dimer levels, and an elevated reticulocyte frequency (>11%). Elevated D-dimer levels have been described after COVID-19 in up to 25% of recovered patients and may predispose individuals post-COVID-19 to coagulatory disorders (45). D-dimer levels are usually elevated during a SCD crisis (50); however, the patient described herein did not report any health issue post-COVID-19 infection. A dysregulation in serum creatinine levels has also been described in patients following COVID-19 infection (44). Upon investigation, it was confirmed that during a previous hospitalization in 2017, patient reticulocyte frequency was much lower (<6%), indicating that this further elevation may be a result of COVID-19 infection. A previous alteration in red blood cells or an increased reticulocyte frequency during COVID-19 have been reported; however, recovered patients did not present long-term alteration (51).
In conclusion, the present study described the case of a patient with SCD with COVID-19. The patient presented a complete viral clearance at 2 weeks following hospitalization in the general ward. To the best of our knowledge, this is the first report demonstrating the long-term effect of COVID-19 in a patient with SCD. The patient presented alterations in reticulocyte count, creatinine and D-dimer levels. Due to the nature of case reports, further in-depth investigations on the long-term effects of COVID-19 sequelae are necessary.
Acknowledgements
Not applicable.
Funding
The present study was funded by the Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP; grant nos. 19/02679‑7 and 17/18199‑9), and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior‑CAPES: 88887.503842/2020‑00.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
RWA, RLO, POR, AJDSD and VA conceived and designed the study, and performed the data analysis and interpretation. MNS performed the data analysis and interpretation. All the authors contributed to the writing and final approval of the manuscript and agree to be accountable for all aspects of the research in ensuring that the accuracy or integrity of any part of the work are appropriately investigated and resolved. All authors confirm the authenticity of all the raw data, and have read and approved the final manuscript.
Ethics approval and consent to participate
The present study was approved by the Ethics Committee of Hospital das Clínicas from the University of São Paulo (HCFMUSP; no. 30800520.7.0000.0068-2020), and was performed out in conformity with the 2013 revision of the Declaration of Helsinki. Informed consent was obtained from the patient.
Patient consent for publication
Informed consent was obtained from the patient for the publication of his clinical data.
Competing interests
The authors declare that they have no competing interests.
References
|
1
|
Tsai SC, Lu CC, Bau DT, Chiu YJ, Yen YT, Hsu YM, Fu CW, Kuo SC, Lo YS, Chiu HY, et al: Approaches towards fighting the COVID-19 pandemic (review). Int J Mol Med. 47:3–22. 2021.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Pandey V and Singh A, Siddiqui S, Raikwar A, Gond AK, Ali S, Yadav M, Datta A and Singh A: COVID-19: An update of current knowledge (Review). World Acad Sci J. 3(1)2021.
|
|
3
|
Chen C, Guan Y, Wei J, Zhou Y, Yao W, Li X and Hei Z: Restricting activity and earlier oxygen supplementation may reduce adverse outcomes in patients with COVID-19 (Review). World Acad Sci J. 3(1)2021.
|
|
4
|
Alberca RW, Lima JC, de Oliveira EA, Gozzi-Silva SC, Ramos YÁ, Andrade MM, Beserra DR, Oliveira LM, Branco AC, Pietrobon AJ, et al: COVID-19 disease course in former smokers, smokers and COPD patients. Front Physiol. 11(637627)2021.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Leung JM, Yang CX, Tam A, Shaipanich T, Hackett TL, Singhera GK, Dorscheid DR and Sin DD: ACE-2 expression in the small airway epithelia of smokers and COPD patients: Implications for COVID-19. Eur Respir J. 55(2000688)2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Vardavas CI and Nikitara K: COVID-19 and smoking: A systematic review of the evidence. Tob Induc Dis. 18(20)2020.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Alberca RW, Rigato PO, Ramos YÁL, Teixeira FME, Castelo Branco ACC, Fernandes IG, Pietrobon AJ, Duarte AJDS, Aoki V, Orfali RL and Sato MN: Clinical characteristics and survival analysis in frequent alcohol consumers with COVID-19. Front Nutr. 8(689296)2021.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Lai CC, Wang CY and Hsueh PR: Co-infections among patients with COVID-19: The need for combination therapy with non-anti-SARS-CoV-2 agents? J Microbiol Immunol Infect. 53:505–512. 2020.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Alberca R, Aoki V and Sato M: COVID-19 and HIV: Case reports of 2 co-infected patients with different disease courses. World Acad Sci J. 3(4)2020.
|
|
10
|
Barron E, Bakhai C, Kar P, Weaver A, Bradley D, Ismail H, Knighton P, Holman N, Khunti K, Sattar N, et al: Associations of type 1 and type 2 diabetes with COVID-19-related mortality in England: A whole-population study. Lancet Diabetes Endocrinol. 8:813–822. 2020.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Codo AC, Davanzo GG, Monteiro LB, de Souza GF, Muraro SP, Virgilio-da-Silva JV, Prodonoff JS, Carregari VC, de Biagi Junior CAO, Crunfli F, et al: Elevated glucose levels favor SARS-CoV-2 infection and monocyte response through a HIF-1α/glycolysis-dependent axis. Cell Metab. 32:437–446.e5. 2020.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Alberca RW, Oliveira LM, Branco AC, Pereira NZ and Sato MN: Obesity as a risk factor for COVID-19: An overview. Crit Rev Food Sci Nutr. 61:2262–2276. 2021.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Liu K, Chen Y, Lin R and Han K: Clinical features of COVID-19 in elderly patients: A comparison with young and middle-aged patients. J Infect. 80:e14–e18. 2020.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Perrotta F, Corbi G, Mazzeo G, Boccia M, Aronne L, D'Agnano V, Komici K, Mazzarella G, Parrella R and Bianco A: COVID-19 and the elderly: Insights into pathogenesis and clinical decision-making. Aging Clin Exp Res. 32:1599–1608. 2020.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Minniti CP, Zaidi AU, Nouraie M, Manwani D, Crouch GD, Crouch AS, Callaghan MU, Carpenter S, Jacobs C, Han J, et al: Clinical predictors of poor outcomes in patients with sickle cell disease and COVID-19 infection. Blood Adv. 5:207–215. 2021.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Arlet JB, de Luna G, Khimoud D, Odièvre MH, de Montalembert M, Joseph L, Chantalat-Auger C, Flamarion E, Bartolucci P, Lionnet F, et al: Prognosis of patients with sickle cell disease and COVID-19: A French experience. Lancet Haematol. 7:e632–e634. 2020.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Ware RE, de Montalembert M, Tshilolo L and Abboud MR: Sickle cell disease. Lancet. 390:311–323. 2017.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Yue L, Lin M, Chen JT, Zhan XF, Zhong DS, Monte-Nguba SM, Liu PF, Pan XF, Huang JH, Wang X, et al: Rapid screening for sickle cell disease by polymerase chain reaction-high resolution melting analysis. Mol Med Rep. 9:2479–2484. 2014.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Zhang X, Li C and Li Q: Magnetic resonance imaging in pediatric sickle cell anemia. Exp Ther Med. 12:555–558. 2016.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Berzuini A, Bianco C, Migliorini AC, Maggioni M, Valenti L and Prati D: Red blood cell morphology in patients with COVID-19-related anaemia. Blood Transfus. 19:34–36. 2021.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Terpos E, Ntanasis-Stathopoulos I, Elalamy I, Kastritis E, Sergentanis TN, Politou M, Psaltopoulou T, Gerotziafas G and Dimopoulos MA: Hematological findings and complications of COVID-19. Am J Hematol. 95:834–847. 2020.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Ye J, Jiao Y, Zhang Y, Li Z, Zeng X, Deng H and Yang M: Hematological changes in patients with COVID-19 (review). Mol Med Rep. 22:4485–4491. 2020.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Alberca RW, Yendo TM, Leuzzi Ramos YÁ, Fernandes IG, Oliveira LM, Teixeira FM, Beserra DR, de Oliveira EA, Gozzi-Silva SC, Andrade MM, et al: Case Report: COVID-19 and chagas disease in two coinfected patients. Am J Trop Med Hyg. 103:2353–2356. 2020.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Netea MG, Giamarellos-Bourboulis EJ, Domínguez-Andrés J, Curtis N, van Crevel R, van de Veerdonk FL and Bonten M: Trained immunity: A tool for reducing susceptibility to and the severity of SARS-CoV-2 infection. Cell. 181:969–977. 2020.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Vasarmidi E, Tsitoura E, Spandidos D, Tzanakis N and Antoniou K: Pulmonary fibrosis in the aftermath of the Covid-19 era (review). Exp Ther Med. 20:2557–2560. 2020.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Corman VM, Landt O, Kaiser M, Molenkamp R, Meijer A, Chu DK, Bleicker T, Brünink S, Schneider J, Schmidt ML, et al: Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Euro Surveill. 25(2000045)2020.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Singh AK, Jena A, Kumar-M P, Sharma V and Sebastian S: Risk and outcomes of coronavirus disease in patients with inflammatory bowel disease: A systematic review and meta-analysis. United European Gastroenterol J. 9:159–176. 2021.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Zhang JJ, Dong X, Cao YY, Yuan YD, Yang YB, Yan YQ, Akdis CA and Gao YD: Clinical characteristics of 140 patients infected with SARS-CoV-2 in Wuhan, China. Allergy. 75:1730–1741. 2020.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Panepintoa JA, Brandow A, Mucalo L, Yusuf F, Singh A, Taylor B, Woods K, Payne AB, Peacock G and Schieve LA: Coronavirus disease among persons with sickle cell disease, United States, March 20-May 21, 2020. Emerg Infect Dis. 26:2473–2476. 2020.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, Zhang L, Fan G, Xu J, Gu X, et al: Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 395:497–506. 2020.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Guan W, Ni Z, Hu Y, Liang W, Ou C, He J, Liu L, Shan H, Lei C, Hui DSC, et al: Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 382:1708–1720. 2020.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Abdulrahman A, Wael M, AlAmmadi F, Almosawi Z, Alsherooqi R, Abduljalil M, Kumar N and AlQahtani M: Is sickle cell disease a risk factor for severe COVID-19 outcomes in hospitalized patients? A multicenter national retrospective cohort study. eJHaem. 2:167–174. 2021.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Azerad MA, Bayoudh F, Weber T, Minon JM, Ketelslegers O, Hoyoux M, Ren X, Azerad MA, Bayoudh F, Weber T, et al: Sickle cell disease and COVID-19: Atypical presentations and favorable outcomes. eJHaem. 1:338–341. 2020.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Singh A, Brandow AM and Panepinto JA: COVID-19 in individuals with sickle cell disease/trait compared with other Black individuals. Blood Adv. 5:1915–1921. 2021.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Alkindi S, Elsadek RA, Al-Madhani A, Al-Musalhi M, AlKindi SY, Al-Khadouri G, Al Rawahi B, Al-Ruqeishi S, Al-Yazeedi J, Wali YA, et al: Impact of COVID-19 on vasooclusive crisis in patients with sickle cell anaemia. Int J Infect Dis. 106:128–133. 2021.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Gladwin MT: Cardiovascular complications in patients with sickle cell disease. Hematology Am Soc Hematol Educ Program. 2017:423–430. 2017.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Rees DC, Williams TN and Gladwin MT: Sickle-cell disease. Lancet. 376:2018–2031. 2010.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Piel FB, Steinberg MH and Rees DC: Sickle cell disease. N Engl J Med. 376:1561–1573. 2017.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Petito E, Falcinelli E, Paliani U, Cesari E, Vaudo G, Sebastiano M, Cerotto V, Guglielmini G, Gori F, Malvestiti M, et al: Association of neutrophil activation, more than platelet activation, with thrombotic complications in coronavirus disease 2019. J Infect Dis. 223:933–944. 2021.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Vingert B, Tamagne M, Desmarets M, Pakdaman S, Elayeb R, Habibi A, Bernaudin F, Galacteros F, Bierling P, Noizat-Pirenne F and Cohen JL: Partial dysfunction of Treg activation in sickle cell disease. Am J Hematol. 89:261–266. 2014.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Qin C, Zhou L, Hu Z, Zhang S, Yang S, Tao Y, Xie C, Ma K, Shang K, Wang W and Tian DS: Dysregulation of immune response in patients with coronavirus 2019 (COVID-19) in Wuhan, China. Clin Infect Dis. 71:762–768. 2020.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Chakravorty S, Padmore-Payne G, Ike F, Tshibangu V, Graham C, Rees D and Stuart-Smith S: COVID-19 in patients with sickle cell disease-a case series from a UK tertiary hospital. Haematologica. 105:2691–2693. 2020.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Raveendran AV, Jayadevan R and Sashidharan S: Long COVID: An overview. Diabetes Metab Syndr. 15:869–875. 2021.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Zhang NH, Cheng YC, Luo R, Zhang CX, Ge SW and Xu G: Recovery of new-onset kidney disease in COVID-19 patients discharged from hospital. BMC Infect Dis. 21(397)2021.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Townsend L, Fogarty H, Dyer A, Martin-Loeches I, Bannan C, Nadarajan P, Bergin C, O'Farrelly C, Conlon N, Bourke NM, et al: Prolonged elevation of D-dimer levels in convalescent COVID-19 patients is independent of the acute phase response. J Thromb Haemost. 19:1064–1070. 2021.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Galeotti C and Bayry J: Autoimmune and inflammatory diseases following COVID-19. Nat Rev Rheumatol. 16:413–414. 2020.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Jurado RL: Iron, infections, and anemia of inflammation. Clin Infect Dis. 25:888–895. 1997.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Demir NA, Basturk A, Ural O, Sumer S, Erdogdu B, Kiratli HE, Celik JB, Koplay M and Vatansev H: A case of Evans syndrome secondary to COVID-19. Blood Transfus. 19:85–88. 2021.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Chen G, Wu D, Guo W, Cao Y, Huang D, Wang H, Wang T, Zhang X, Chen H, Yu H, et al: Clinical and immunologic features in severe and moderate forms of coronavirus disease 2019. medRxiv: Feb 19, 2020 (Epub ahead of print). doi: 10.1101/2020.02.16.20023903.
|
|
50
|
Dar J, Mughal I, Hassan H, Al Mekki TE, Chapunduka Z and Hassan ISA: Raised D-dimer levels in acute sickle cell crisis and their correlation with chest X-ray abnormalities. Ger Med Sci. 8(Doc25)2010.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Kubánková M, Hohberger B, Hoffmanns J, Fürst J, Herrmann M, Guck J and Kräter M: Physical phenotype of blood cells is altered in COVID-19. bioRxiv: 2021.02.12.429482, 2021.
|















