1. Introduction
The human intestinal microbiota is a topic of great
interest for its fundamental role in several aspects of host
homeostasis, such as nutrition, immune development, metabolism and
defense against pathogens (1). The
gut microbiota (GM) is estimated to be composed of
>1014 microorganisms: It is represented by a complex
system of bacteria, archaea, viruses and fungi who live in
symbiosis with the host (2). The
individual pattern changes over the years and it is subjected to
strong variations due to environmental changes, diet, exposure to
antigens, infections, drugs, hygiene factors and lifestyle
(3). Several studies have assessed
the association between environmental stressors and GM integrity.
The interaction between the host and microbiota determines the
difference between eubiosis and dysbiosis. This balance can be
altered by air pollution, exposure to pesticides and heavy metals,
and can promote the development of diseases, such as obesity, type
2 diabetes mellitus, metabolic diseases and cancer (4-8).
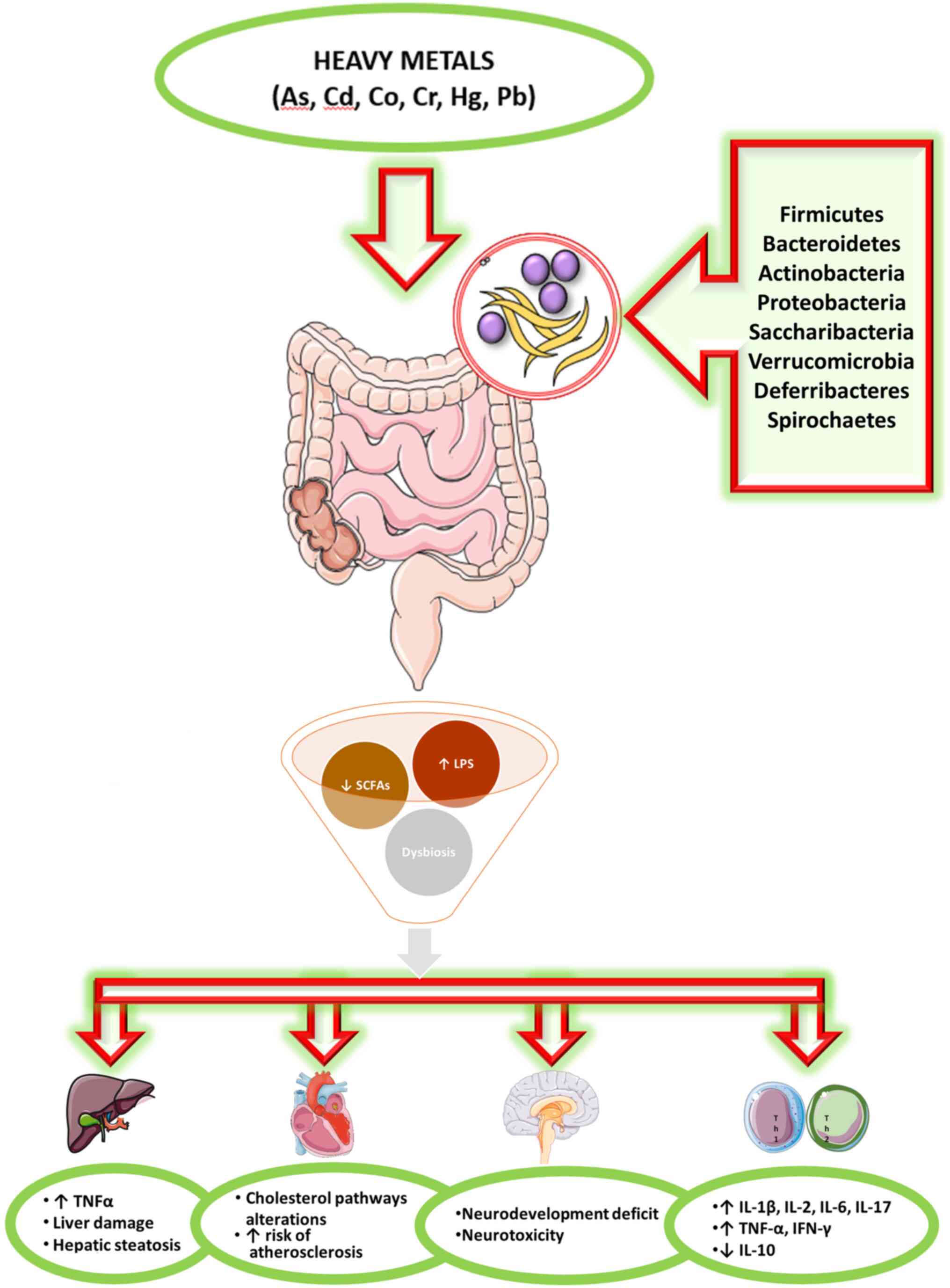
Toxicants in the environment can directly harm the
components of the microbiota, but can also be modified by the
microbiota to become more or less toxic to the host and/or the
microbiota itself (3). The effects
of heavy metals on the intestinal microbiota are not still largely
defined (Fig. 1).
The aim of the present review was to provide an
update of the available evidence in the literature related to the
interaction between the GM and heavy metals, focusing on the
compounds most widely distributed in the environment or considered
biopersistent.
2. Data collection and analysis
A literature search was performed on the PubMed
database to detect full-text articles published over the past 5
years using the following keywords: ‘Gut microbiota AND heavy
metals’ [All fields], ‘gut microbiota AND arsenic’ [All fields],
‘gut microbiota AND cadmium’ [All fields], ‘gut microbiota AND
copper’ [All fields], ‘gut microbiota AND manganese’ [All fields],
‘gut microbiota AND chromium’ [All fields], ‘gut microbiota AND
nickel’ [All fields], ‘gut microbiota AND mercury’ [All fields],
‘gut microbiota AND zinc’ [All fields], ‘gut microbiota AND lead
exposure’ [All fields]. Articles published in languages other than
English, reviews or conference abstracts or letters to editors were
not included.
The search produced a total of 610 results. As much
as 455 articles were not pertinent with the aims of the present
review and after excluding 58 repeated results, a final number of
97 articles was selected for the review. Additionally,
supplementary articles were identified among the reference list of
the screened articles.
3. Overview
Human GM
The human microbiota is the set of symbiotic
microorganisms that coexist in the human body; it is composed of
>100 trillion different organisms in a complex system of
bacteria and viruses, as well as fungi, archaea and protists
(2). In the majority of studies,
the bacterial constituents of a microbial population were
identified by sequencing the 16 sRNA-encoding gene. GM varies among
individuals during development, and is dependent on the host and
environmental factors (9). This
gut microbial community has evolved with its host over a timespan
of millions of years and offers benefits to its host through a
number of mechanisms, including digestion, detoxification, the
production of nutrients, protection against pathogens and the
regulation of the immune system (10). The intestinal tract is exposed to a
plethora of food-borne and bacterial antigens. The epithelial cell
layer prevents a ‘too close contact’ of these antigens with the
immune cells, and thereby also protects the gut from unwanted
immune reactions. This is achieved by the sophisticated
organization of the intestinal epithelium, which establishes a
tightly regulated barrier (11).
In adults, the GM is composed of 500 species of bacteria divided
into 45 genera and 14 families; however, the individual pattern
changes over the years and it is subjected to strong variations due
to environmental changes, diet, exposure to antigens, infections,
drugs, hygiene factors and lifestyle. The most frequent
microorganisms are commensal bacteria that are beneficial for the
host.
The analysis of normal bacterial communities has
indicated that the enteric microbiota in healthy subjects belongs
mainly to 4 phyla: Firmicutes and Bacteroidetes that are the most
representative (together they represent 90% of total enteric
microbiota), Proteobacteria and Actinobacteria (including
Bifidobacterium), which are mainly driven by dietary intake,
but are independent of age or body mass index (12). Fusobacteria, Cyanobacteria and
Verrucomicrobia are less represented.
Heavy metals
Heavy metals are naturally occurring elements that
have a high atomic weight and density. Both prokaryotes and
eukaryotes have developed strategies to benefit their action and to
defend themselves from undue exposure, particularly against those
metals showing high reactivity. These metals are commonly spread
and many of them are used in human activities, such as
manufacturing, mining, agricultural fields, configuring exposure as
environmental and occupational risks. Conversely, other metals can
be found in water and the food supply chain, posing a safety
concern for human health, particularly in developing countries.
Arsenic (As), cadmium (Cd), chromium (Cr) and nickel (Ni) are
classified as group 1 carcinogens by the International Agency for
Research on Cancer (13).
Heavy metals can affect target organs (liver,
kidneys, lungs) through different mechanisms and pathways (14). Moreover, these metals are difficult
to degrade and their bioaccumulation is dependent not only on the
exposure (concentration and frequency), but also on the organism
capability to eliminate the metals. The residuals in the
gastrointestinal tract can affect the human GM that interacts with
heavy metals, and is in part, responsible for the intestinal
barrier functioning involved in the control of absorption of toxic
metals (15).
All studies on humans or different animal models or
in vitro, concerning investigations of ≥2 heavy metals, are
concordant in concluding that heavy metal exposure, even if for a
short period of time, can cause perturbations in the composition,
structure and diversity of the GM. The majority of the studies used
the amplification and sequencing of the 16S rRNA gene for the
microbial community analysis; only 2 studies (16,17),
proceeded with metabolomics approach demonstrating that As, Cd and
selenate also influenced several metabolic pathways. When
investigated together, different toxicants may interact and their
effects, on GM or on health status, could be potentiated.
Human studies
Laue et al conducted a study on 179
6-week-old infants (18), the GM
was assessed using 16S rRNA sequencing. They found that postnatal
exposure to toxic elements was differentially associated with the
infant microbiota. Exposure was negatively associated with
microbial diversity, particularly in infants exposed to peripartum
antibiotics. As, Cd, manganese (Mn), copper (Cu), iron (Fe), lead
(Pb), Ni, selenium, tin, and zinc (Zn) were each differentially
associated with at least one taxon, in particular with
Bacteroides and Lactobacillales, while mercury (Hg)
was associated with specific taxa.
In addition, on children aged from 3-7 years, other
authors investigated potential interactions between toxic metals,
the gut microbiome and the development of autism spectrum disorders
(ASDs). The levels of Pb, Cd, As, Cu, Zn, Fe, Hg, calcium and
magnesium in hair samples were analyzed. Microbial DNA was
extracted from fecal samples and 16S rRNA was sequenced. Levels of
Pb, As, Cu, Zn, Hg, calcium and magnesium were significantly higher
in the ASD group. Different metals were associated with dissimilar
perturbations in GM composition and diversity, suggesting the
possible pathogenesis of ASD depending on metal absorption and gut
microbial community (19).
Animal studies
Liu et al conducted a study on rats (20), poisoned first with methylmercury
(MeHg) and then treated with sodium selenite (NaSe). Microbial DNA
was extracted from fecal samples and 16S rRNA was sequenced. The
results suggested that MeHg damaged the composition of microbiota.
Following Se treatment, the richness of gut microbial community was
partially reestablished and a similar mechanism can be hypothesized
also for human beings. A most recent study performed on rats
focused on neuro-developmental effects (21). Offspring rats were exposed to
inorganic As (iAs) and fluoride (F-). DNA was extracted from fecal
samples and 16S rRNA gene was sequenced. Noticeable
neuro-developmental effects were observed in rats concurrently
exposed to iAs and F-so microbiome-based biomarkers of iAs and/or
F-may be suggested as early indicators of neuro-developmental
deficits. Richardson et al exposed rats to 3 different doses
of sodium arsenite, Cd chloride, sodium dichromate, cobalt chloride
or Ni chloride by oral gavage for 5 consecutive days (14). DNA was extracted from fecal samples
and 16S rRNA gene was sequenced. Significant variations in GM
composition were observed in high doses of Cr and cobalt, but only
in As, Cd and Ni exposure in a significant dose-dependent manner.
Following As and Ni exposure, bacteria with a huge number of
iron-importing gene were excessively represented. The results
underline the convenience of the microbiome as an early tool for
recognizing specific heavy metal exposures (14). Recently, in a study on mice
(22), animals were exposed to Cu,
Hg or both. DNA was extracted from cecal contents and 16S rRNA was
sequenced. All treatment groups showed intestinal histopathological
damages and alterations in the diversity of gut microbiota in the
cecum of female mice, which can provide new insight into the risk
assessment in intestinal disorders caused by Cu and Hg. Gaulke
et al fed mice with diet containing either different Zn
concentrations and exposed to As for 6 weeks (23). Zn deficiency and As exposure
autonomously changed microbiota composition but, when in
combination, the outcomes in microbial community were intensified,
in particular marginal Zn deficiency resulted in a higher
sensitiveness to As exposure of the GM (23).
Li et al treated mice with Cd chloride and As
in their drinking water for 2 weeks. Colon and caecum content were
collected and 16S rRNA gene was sequenced (16). Both As and Cd significantly
perturbed the metabolome and lowered the GM diversity; however,
only Cd significantly decreased the microbial diversity. The number
of metabolite interactions diminished in a number of genera.
Moreover, As and Cd exposure affected pathways involved in
metabolic health (16).
Previously, other authors performed a research on cows. Animals
were fed supplemental Cu, Zn, and Mn from sulfate minerals,
glycinate minerals or Cu and Mn sulfate with glycinate Zn.
Treponema Operational Taxonomic Units (OTU) in GM, frequently
related to bovine digital dermatitis, were less abundance in cows
fed with Cu and Mn sulfate with glycinate Zn. These results suggest
a further connection between organic Zn supplementation and better
animal health (24). Rothman et
al carried out a study on honey bees administering solutions of
50% sucrose spiked with Cd chloride, pollen patties spiked with
selenium or Cd. Cd and selenate exposure perturbed the composition
of the GM altering the metabolite pathways, particularly those
concerning detoxification, proteolysis, and lipolysis, resulting in
bioaccumulation of these toxicants (17).
In a study conducted on black soldier fly larvae,
the authors administered Cu or Cd. Gut DNA samples were analyzed.
Cu and Cd exposure noticeably perturbed the GM. The highest levels
of exposure significantly reduced the richness of the majority of
dominant families; however, in the meantime, other families were
enriched, resulting in a total decrease of the GM diversity
(25). Zhang et al examined
the GM of Bufo raddei from a polluted and relatively
unpolluted area (15). The
Firmicutes/Bacteroidetes ratio and the number of probiotics in the
gut microbiota from the polluted area were decreased when compared
to those from the unpolluted are. The results suggest that
long-term heavy metal exposure perturbed the structure and reduced
the diversity of thee GM in Bufo raddei (15).
In vitro studies
By using the Simulator of the Human Intestinal
Microbial Ecosystem (SHIME) some authors have simulated an exposure
to As and Fe in different concentrations suggesting that GM had a
significant role in As species modification, and Fe can influence
As transformation through alterations in GM. In the evaluation of
the health hazards of As in drinking water, the residual Fe should
be taken into account (26).
Recently, always through the use of the SHIME, fresh fecal
microorganisms obtained from volunteers were mixed with Fe minerals
(goethite and jarosite). Total and bioaccessible As and Fe
concentrations were determined. As bioavailability in human
intestine was mainly due to FeIII dissolution in
jarosite, and to bacterial community reduction of FeIII
and AsV in goethite, underlining the importance of Fe
minerals in human health risk evaluation associated with
environmental As exposure (27).
4. Arsenic
As, an ubiquitous nonmetal in nature, is a human
carcinogen. While it can be found as a metal in its pure form, it
is typically found as a component in both inorganic and organic
compartments (28). As is found in
water and food worldwide, as both organic and inorganic species in
dietary essentials, such as rice, and exclusively as inorganic As
in water (29). As soil and water
contamination has been associated with human activities, such as
smelting, mining, coal and ash disposal, pesticide use (30). As exposure is a public health
concern worldwide; in fact, >100 million individuals are exposed
to As from drinking water and it has been associated to a number of
human diseases, particularly diabetes, cancer and cardiovascular
illnesses (31). The consumption
of contaminated drinking water and/or food is the principal pathway
of As exposure in humans; thus, the GM can be the most vulnerable
to As exposure (32).
Human studies
Hoen et al analyzed, on 204 infants in the
USA, As levels in urine and microbiota composition from fecal
samples (29). With higher As
exposure, 8 genera were enriched. Urinary As levels were negatively
associated with 15 genera, including Bacteroides and
Bifidobacterium. Following stratification by sex and feeding
method, associations were found in formula-fed males, but not in
the other groups. In infant development, sex-specific effects on
the GM can be observed even with moderate As exposure (29).
A longitudinal study on the health effects of As
conducted in Bangladesh among 250 participants, aimed to find
associations between As exposure, microbiota composition and
carotid intima-media thickness. Significant associations between GM
composition and As exposure were not found. Intima-media thickness
(IMT) was significantly associated with Aeromonadaceae and the
genus Citrobacter. A significant reciprocity between water
As and Citrobacter was observed in IMT. The authors
concluded that in the development of atherosclerosis, GM may have
an important impact, in particular among subjects exposed to higher
As levels (33).
Other authors evaluated 16S rRNA gene sequencing
from fecal samples and urine As concentration in 2 southern Nepal
communities. Pathogenic bacteria were positively associated with
higher As concentrations in the subjects. Lower levels of As were
associated with intestinal commensal bacteria. The authors stated
that As plays a role in the GM shaping through the enrichment of
pathogens and the weakening of commensal bacteria (34).
Previously, a study conducted among children
(32) made a comparison between
high and low As-exposed groups. Children exposed to higher As
concentrations exhibited an abundance of Proteobacteria in their
feces. In the high exposed group, a positive association between
genes involved in virulence and multidrug resistance and As
concentration was observed. The qPCR quantification of 2 As
resistant genes (ArsB, ArsC) revealed a higher expression of these
2 operons in the children with high As exposure.
These studies conducted on humans evaluated the gut
microbiome composition through the 16S rRNA gene sequencing from
fecal samples. All the studies, except one (33), found an association between As
exposure and significant variations in the gut microbiome, with an
increase in the number of pathogens, underlining an important role,
played by As exposure.
Animal studies
Coryell et al exposed mice to As. As levels
in feces and in organs were measured and the GM composition was
analyzed through the 16S rRNA gene sequencing from fecal samples
(28). The lack of As
detoxification enzyme (As3mt) rendered the mice hypersensitive to
As. The protection deriving from humane microbiome transplant
depended on microbiota stability and the presence of certain
bacteria, including Faecalibacterium. In mouse model As3mt
and certain microbiota bacteria are fundamental for protection
against acute As toxicity (28).
Previously, other authors divided mice in cases and controls,
treated with sodium arsenite in their drinking water for 13 weeks.
In the microbiota of mice treated with As, the genes involved in
the biosynthesis of lipopolysaccharides (LPS), in DNA repair
mechanisms and in multidrug resistance were increased and the genes
involved in the production of vitamins (B6 and B12) were enriched
(35). The same authors recently
conducted a study on same animals to compare GM-disrupted animals
(treated with antibiotics for 72 h prior to As exposure) and
non-antibiotic-treated mice. In the GM-disrupted mice, total As
levels were lower in feces and much higher in urine than the levels
in control subgroup mice. The disruption of the microbiota
significantly altered As biotransformation by affecting one-carbon
metabolism and also increased the As toxic effects (36).
Other authors compared Helicobacter-free mice
and Helicobacter trogontum-treated animals (by oral gavage)
successively treated with As. As exposure caused GM perturbation
inducing variations of several metabolite pathways, involving the
metabolism of fatty acid, phospholipids, cholesterols, and
tryptophan. The authors proposed that GM perturbation could worsen
or cause metabolic disturbances induced by As exposure (37). Chi et al treated mice with
As for 4 weeks and evaluated variations in microbiota composition
(31). At the end of the 4-week
treatment, it changed in both males and females, but more
significantly in females than in the respective controls. The host
sex determines the microbiota phenotypes, whose sensitivity to As
differs between male and female mice. Moreover, same authors
compared antibiotic and non-antibiotic treated mice. As exposure
suppressed gene expression in cholesterol metabolism,
transportation and synthesis only in control mice. In
antibiotic-treated mice, a smaller perturbation was observed on
liver and serum lipid homeostasis caused by As exposure. The
As-induced effects on lipid metabolism may be buffered by
regulating the GM (38).
In another study, mice were divided into the control
and As-treated groups. In As-exposed mice, the α-diversity of the
GM was significantly lower than that of the controls. Following As
exposure, carbohydrate metabolism was significantly perturbed due
to the increase of several proteins involved in this pathway. As
exposure may increase the LPS synthesis in the GM through the
upregulation of UDP-N-acetylglucosamine acyltransferase (39).
Chiocchetti et al administered various doses
of AsIII through drinking water to mice (40), evaluating the expression of genes
encoding inflammatory cytokines (TNFα, IL-1β, IL-2 and IL-6) and
claudins (Cldn-1, Cldn-2 and Cldn-4). At As concentrations ≥50
mg/l, an increase in gene and protein expression of
pro-inflammatory cytokines (IL-1β, IL-2 and IL-6) was showed.
Moreover, the diversity and global composition of GM were
accompanied by mucosa and submucosa moderate inflammation. These
effects are suggestive of an increased intestinal permeability,
resulting in a barrier function loss, particularly at the highest
As concentrations.
A study on the adult and developmental stages of CD1
mice (41) evaluated the effects
of a single oral exposure and of repeated exposure. In adults after
a single gavage, responses in microbial composition and bacterial
recovery were observed in a dose and time-dependent manner.
Repeated exposure affected intestinal bacteria with a decrease in
recovery and an abundance of As resistance genes. High levels of
intestine-secreted chemokines were revealed in adult animals
exposed to As. Both in juvenile and adult animals, it was possible
to observe the gut-associated immune status and intestinal
microbiota changes.
Tikka et al compared mice in 2 different
exposure periods (3 and 6 months) (42). As exposure not only altered the GM
composition, but also increased inflammatory cytokines (TNF-α,
IFN-γ and IL-17) and depleted anti-inflammatory cytokines (IL-10).
Moreover, the level of β-catenin, a colon cancer marker, increased
in the animals exposed to As for both 3 and 6 months. These results
suggest that variations in the GM can alter inflammatory cytokine
pathways, resulting in an immune system dysregulation, affecting
colon cancer marker expression (42).
The majority of the studies used C57BL/6 male mice
as a study population; other models used were female BALB/c mice,
adult and developmental stages of CD1-mice and Kunming mice. All
the experiments were designed as control/case studies, exposing
animals to various As concentrations in drinking water, in food or
oral gavage. The majority of the studies evaluated GM composition
through 16S rRNA sequencing from DNA extracted from fecal samples,
only two using metagenomic (31,35)
and one metaproteomic approaches (39). Data on GM composition, gene
expression, metabolic pathways and tissue damage were all
concordant on the negative effects elicited by As exposure on
animal health status.
A study on the earthworm Metaphire
californica (43) evaluated
the interactions between microplastics (MPs), As exposure and GM.
Following As exposure, significant changes in gut bacterial
composition were observed, while MPs did not significantly impact
the gut bacterial communities. MPs not only decreased the As
accumulation, but also the transformation of AsV to
AsIII. MPs may have played a role in lowering As
bioavailability, reducing its effects on the GM, resulting in a
lower toxicity on earthworms.
Another study on the earthworm (Metaphire
sieboldin) compared various As concentrations in the controls
and cases for 28 days. The analysis of gene expression revealed
that As redox and efflux genes were abundant, whereas As
biotransformation (methylation and demethylation) genes were very
low. These results suggest that the earthworm GM has a marked
capability in reducing AsV even if As exposure disturbs
the GM composition (44).
Studies conducted on worms have demonstrated the
negative effects of As exposure on the gut microbiome, resulting in
bioaccumulation, reducing capability and gene expression
alterations.
In vitro studies
By using the in vitro SHIME, Yin et al
aimed to investigate the variability in the As bioaccessibility in
gastric, small intestine and colon phases in fecal microorganisms
(45). The results suggested that
the As bioaccessibility varied in the colon phase, due to microbial
reduction activity. In order to assess hazards to human health
correlated with oral exposure to soil As, joining in vitro
methods and SHIME could increase the accuracy in risk assessment
(45).
The same authors using SHIME, analyzed the
interactions between four soils with different As concentrations
and fecal microorganisms, demonstrating that human GM can release
soil-bound As. In humans subjected to soil As exposure, an precise
risk assessment could be carried out by concurrently defining As
transformation and intestinal absorption (46).
Li et al demonstrated that the bacterium
Bacteroides vulgatus possesses resistance genes to inorganic
As, which can be decisive in the ability of the microbe to maintain
its prevalence in the human intestine even after exposure to
food-related As (47).
In vitro studies, evaluating As
bioaccessibility, transformation and absorption, mediated by the
human GM, particularly in the colon, play an important role in
providing insights on mechanisms involved in As metabolism.
5. Cadmium
Cd, one of the most widespread toxic heavy metal
pollutants, appears in soils and drinking water supplying, as a
side product of human activities, such as mining, agricultural use
(in fertilizers) through air deposition or industrial divisions
(batteries, pigment and plastics) (48,49).
Cd can also be found in aquatic systems. Cd exposure has become a
prevalent health concern due to the access to polluted food and
water. As with other heavy metals, it is associated to several
toxic effects in living organisms, particularly in oxidative stress
induction, DNA damage, carcinogenesis, dysregulation in immune
responses and energy metabolism (50).
Animal studies
Rodent studies have been designed as control/case
experiments, exposing animals to various Cd concentrations. All
studies revealed GM perturbations caused by Cd exposure and several
studies suggested the important role played by Cd in the
alterations of both metabolic and energetic pathways.
Previously, authors demonstrated that the Cd
exposure induced noticeable perturbations of GM in mice, with a
significant decrease in gut microbial richness and reduction in
short-chain fatty acids (SCFAs) production. Moreover, Cd exposure
caused changes in the expression of those genes involved in
metabolic pathways, associated with amino acid and carbohydrate
(48).
In mice, Cd exposure significantly altered the GM
composition and richness, not only at the phylum level, but also at
the family and genus levels. These alterations result in an
increase in serum LPS levels accompanied by hepatic inflammation,
as a result of dysregulation in energy metabolism (50).
Ba et al evaluated metabolic effects in mice
in a sex-dependent perspective, observing that early low-dose Cd
exposure (100 nM) altered the GM in composition and diversity.
Furthermore, Cd exposure caused metabolic effects only in males
(51).
The only experiment concerning the evaluation of a
potential protective probiotic to decrease Cd toxicity, revealed
the failure of Akkermansia muciniphila (AKK). GM alterations
were observed both in acute and chronic Cd exposure; however, the
oral administration of AKK influenced GM composition, particularly
following acute Cd exposure (52).
Following Cd exposure, a previous study observed
both in the liver and GM, noticeable functional and structural
modifications. Moreover, Cd exposure induced variations of several
metabolites, suggesting that Cd may play an important role in
energy metabolism in these animals (53).
Studies on fish and crustaceans underlined the
negative effects of Cd on microbial community diversity. In
particular, dietary supplementation with probiotics revealed the
protective role of certain microorganisms against Cd
bioaccumulation and its toxic effects, providing new insight into
aquaculture and food safety. In a study conducted on common carp,
it was observed that Cd exposure significantly perturbed the
composition of GM in the fish, resulting in a decrease in microbial
community diversity. Conversely, the abundance of AKK,
decreased after Cd exposure (54).
Cd exposure caused, in Procambarus clarkii,
intestinal histological damage and variations in the richness,
diversity and composition of the GM, not only at the phylum level,
but also at the genus level. Moreover, Cd exposure may suggestively
perturb metabolic pathways related to diseases and cellular
processes (55).
Wang et al highlighted the protective effect
of Bacillus cereus against Cd exposure (56). Cd caused manifest variations in the
GM composition of Carassius auratus gibelio; however,
Bacillus cereus succeeded to inverse these effects,
inhibiting perturbations in Cd bioaccumulation and antioxidant
enzyme levels (56). Cd exposure
was also shown to cause a marked decay in gut microbial diversity
and composition in Nile tilapia. Dietary supplementation
with the probiotic Lactobacillus plantarum, CCFM8610,
inverted the variations and reduced Cd accumulation (57).
Studies on amphibians have also disclosed Cd toxic
effects, reducing GM diversity and composition. Ya et al
observed severe gut histopathological modifications and microbial
alterations at the Cd highest doses (100 and 200 µg/l), whilst
small intestine damage was observed at the 5 µg/l Cd concentration
(58,59). Even in Rana chensinensis, Cd
exposure transformed the composition and reduced the microbial
diversity in GM both at phylum and genus levels (60).
In addition, 2 studies on worms confirmed the same
negative effects of Cd exposure on GM composition. Notably,
variations in intestinal microbial communities in earthworm could
be considered as a biological indicator of soil pollution. Šrut
et al revealed a perturbation in GM composition and
increased levels of heavy metal resistant bacteria following Cd
exposure in the earthworm (49).
Lee et al compared the effects of Cd exposure between
Caenorhabditis elegans fed with either soil microbial
community (SMB) or Escherichia Coli strain OP50; in the
OP50-fed worms, microbial community alterations mediated were
particularly severe (61).
6. Mercury
Mercury is an ubiquitous and highly biopersistent
pollutant. For this reason, environmental and occupational exposure
has been subjected to restriction by the European Community.
Generally, individuals are exposed via contaminated food
consumption, particularly fish. Bioavailable organo-metallic
compounds (e.g., MeHg) can cross the blood-brain and placental
barriers, developing neurotoxicity (62,63).
The mechanisms involved in the complex
bio-physico-chemical process of Hg methylation and demethylation
remain to be fully understood. However, MeHg metabolism is the core
research in experiments focused on methylation/demethylation
processes in GM, that is considered to be involved in the
biochemical interplay responsible for its demethylation to
inorganic mercury (64).
Human studies
Rothenberg et al analyzed, in pregnant women,
total Hg and MeHg levels in maternal blood and stool samples,
aiming to investigate whether associations between biomarkers for
prenatal MeHg exposure and maternal GM differed between early and
late gestation (64). The GM
differed, not only between subjects, but also between early and
late gestation in the same individual. Even low MeHg concentrations
(both in blood and stool) were associated with perturbations in
microbiota composition only during early gestation. The results
suggested that mothers ingesting a fiber-rich diet may have
significantly improve MeHg elimination (64).
Animal studies
All in vivo studies were concordant in
stating that mercury exposure is accountable for intestinal damage,
dysbiosis, metabolic disorders, an increase in the expression of
genes involved in apoptosis processes and neurotoxic effects. Some
authors also hypothesized that different chemical forms of Hg are
responsible for its accumulation in gut and dissimilar microbiota
structure alterations (62,65,66).
The GM influences brain activity through the gut-brain axis, by
producing neurotransmitters and their precursors. MeHg can
perturbations in GM structure in rodents, resulting in alterations
in intestinal neurotransmitters responsible to regulate neuron
activity. These results suggest a possible mechanism concerning
MeHg neurotoxicity (63).
7. Lead
Pb has been an ubiquitous environmental pollutant
for decades and has been causally associated with well-known
occupational diseases and innumerable damaging health effects,
including neurological and kidney disorders, anemia and altered
immune system responses (67,68).
There are a number of sources of lead exposure; apart from water
and food, the majority derives from human activities and industrial
productions, such as paint and electronic waste (69).
Human studies. Eggers et al found a
significant association between the adult urinary Pb concentration
and GM composition, particularly as regards the extent of
Proteobacteria colonization (67).
Animal studies. All in vivo studies
were concordant in stating that Pb exposure may play a role in
dysbiosis, inducing structural intestinal damage. GM structure and
diversity were always perturbed by lead exposure (70). In experimental studies performed on
mice, metabolomics profiling and metagenomics sequencing revealed
that a number of metabolic pathways, including energy metabolism,
oxidative stress and detoxification mechanisms, were meaningfully
altered by Pb exposure (69). Pb
exposure caused memory impairment, perturbations in GM composition
and release of oxidative stress biomarkers in serum (71). Prenatal Pb exposure may also
contribute to increase adult bodyweight (72) and can cause large intestine immune
disorders (73). The same effects
were confirmed in zebrafish (74).
In vitro studies. By SHIME simulation, Pb
bioaccessibility was evaluated in soil samples through the
Physiologically Based Extraction Test (PBET). The mean Pb
bioaccessibility changed in gastric, small intestine and colon
phases, depending on soil pH and types. The action of the human GM
was important in the colon phase, when mean Pb bioaccessibility was
compared between farming soils (higher) and mining soils (lower)
(75).
8. Zinc
Zn is a fundamental nutrient for almost all
organisms. From a clinical point of view, even mild deficiencies
can reduce cellular differentiation, influence immune system
developing and deeply affect growth and maturation (76). Infants, also when healthy, are
given additional Zn with food, beverages and nutritional
supplements; consequently, it often exceeds established nutritional
necessities (77). Furthermore,
the microbial community has its own requirements for minerals,
including Zn (78).
Animal studies
The excessive intake of dietary Zn in mice, can
cause oxidative stress in the intestine, resulting in deep shifts
in GM. These perturbations are associated with a microbial
community enriched in pathogenic taxa. Moreover, variations in the
expression of genes involved in metabolic processes, oxidative
stress and protein glycosylation were observed (77). Excess dietary Zn not only
significantly perturbs the GM, but also worsens C.
difficile-associated disease, by varying the host immune
response (79). In a study
conducted on chicken hatchlings (Gallus gallus), animals
receiving a Zn-biofortified diet exhibited a significant increase
in β-but not α-microbial diversity; in particular, associated
intensification in Zn-dependent bacterial metabolic pathways was
detected (76). A research
conducted on Targhee yearling rams revealed that a quantity of
bacterial genera was perturbed, but only the phylum Tenericutes was
significantly decreased by Zn supplementation, suggesting that Zn
formulations can be employed without producing a high-level
modification in the rumen gut microbiome composition, which could
have adverse effects on metabolism and health status (78). These studies demonstrated how an
excess in dietary Zn, not only significantly perturbs microbiota,
but also affects different metabolic pathways and gene expression,
resulting in negative effects on animal health status.
9. Copper
Cu is an essential microelement (80); however, excessive Cu quantities can
affect the ecosystem resulting in soil and water pollution.
Disproportionate Cu exposure may cause negative effects on animal
organisms, inducing respiratory, nervous and gastrointestinal
diseases (81). The effects of
overexposure to Cu on the GM are not yet well described in the
literature (82).
Animal studies
High Cu concentrations can cause liver damage in
rats. Cu exposure can exert dose-dependent effects on the structure
of the GM. Moreover, metabolomic analysis confirmed the effects of
Cu exposure on metabolic pathways involved in hepatic injury and
intestinal inflammation. These alterations may induce an
inflammatory response, in which TNF-α could be the main respondent
in Cu overexposure (82,83). Furthermore, various dietary doses
of Cu and fructose can interact with host microbial metabolic
activities by various mechanisms, resulting in alterations in GM
composition, liver damage and hepatic steatosis (84).
In mice, some authors have demonstrated that high
concentrations of Cu can cause intestinal histopathological lesions
and perturb GM composition (81).
In piglets, a low-Cu diet can alter the structure of the microbiota
and modify bacterial metabolic pathways, including proteins,
carbohydrates, the urea cycle, gluconeogenesis and amino acids,
which may play a role in affecting animal health (80). In a previous study, the GM of
Cu-treated fish (Takifugu fasciatus) exhibited perturbations
in composition at the phylum level, revealing the role of
intestinal microbial community in maintaining the health status of
Takifugu fasciatus for aquaculture (85). In addition, in the common carp
(Cyprinus carpio L.), Cu exposure altered the α- and
β-diversity of the GM (86). The
authors observed that the presence of short-chain fatty acid
(SCFA)-producing bacteria was significantly reduced, as well as
probiotics, with an increase in pathogens, resulting in an
increasing risk of pathogen invasion (86). Moreover, in Rana
chensinensis larvae, the Cu-altered microbiota composition was
observed at the phylum level, resulting in a reduction of
Fusobacteria abundance, while Bacteroidetes did not exhibit
significant differences (87).
These studies suggest that Cu exposure exerts
dose-dependent effects on the structure of the GM. Changes in the
composition of the microbial community may be related to
alterations in several metabolic pathways, inflammatory responses
(through cytokine modulation) and liver damage (88).
10. Nanoparticles
Advances in nanotechnology introduced over the last
decade have allowed for the production of metals on a nanoscale
(1±100 nm). Due to their particular characteristics, the
applications of nanoparticles (NPs) have been extended in different
fields, such as cosmetology, agriculture, textile manufacture,
construction, food and medical area, particularly in diagnostic
imaging, drug technology and tumor therapies. This widespread
introduction of NPs in a number of human activities has unavoidably
lead to NP diffusion in the environment (89-92).
Nanoparticles, including metals, due to their small
dimensions, are able to cross biological membranes, perturb ion
channels transportation, alter enzymatic activities and react with
genomic material. These mechanisms could induce tissue injuries,
oxidative stress (92) and can
modulate immune responses (93).
The majority of studies demonstrate that metal NPs,
through different biochemical mechanisms, can perturb the
microbiota with negative consequences, particularly when high
concentrations in dietary regimens are administered to experimental
animals. Nonetheless, there is also evidence of a possible benefit
in dietary supplementation with NP.
Animal studies
Cholewińska et al in 2018, revealed that Cu
NPs were better absorbed in the intestines of Wistar rats, but
bioaccumulated in the brain, and at a higher dosage induced severe
liver injury. As regards GM, Cu NPs blocked its enzymatic activity
and consequently significantly reduced SCFA production (92).
Cu oxide (CuO) NP exposure can not only induce
perturbations in microbiota composition, but can also affect the
expression of genes involved in immune response in worms, such as
lysozyme and coelomic cytolytic factor. Nonetheless, these
alterations in the microbiome did not increase susceptibility to
bacterial infection (93). In
another study, in poultry, CuO NP added to the feed, did not affect
probiotics or the main representatives of normal microbial
community. The Cu active ions, due to the low dissociation degree
from NP, are released more slowly and therefore have more prolonged
positive effects (94).
In a study conducted on Eisenia fetida
cultured with zinc NP in soil, 35% mortality and perturbations in
GM composition were observed, and in particular, a decrease in
microbial diversity was registered (89). Wang et al exposed mice to
nano-ZnO suspension, which raised Zn concentrations in the serum,
liver and kidneys, but did not perturb the gut microbiome
composition (95). Some authors
have hypothesized an increment of anti-inflammatory activity in ZnO
NP-exposed mice. The effects of ZnO NP varied in the GM, increasing
Firmicutes and decreasing Bacteroidetes in female mice. SCFA were
also increased, indicating that ZnO NP could have possible
utilizations as safe regulators of GM (96). In crossbred weaned piglets, fed a
diet supplemented with nano-Zn, positive effects on gut morphology
and microbiota were observed, similar to those of conventional ZnO
(97). Kolba et al
demonstrated that different metal NPs (TiO2,
SiO2 and ZnO) could perturb the composition of the GM,
negatively influencing intestinal functions and health in chicken
(Gallus gallus) (98).
In vitro studies
The antimicrobial and antioxidant effects of green
silver nanoparticles (Ag NP) on the GM in healthy individuals have
been simulated by an in vitro one-chamber GIS1 colon
simulator. Ag NP preserved their antioxidant properties, as shown
by a decrease in ammonium compounds; a decrease in
Bacteroides suggested an antimicrobial effect. Moreover, the
NP spherical shape was significantly associated with their
biological outcomes in vitro (99).
11. Other heavy metals
Cr is an essential trace element with positive
health outcomes, which are well demonstrated in humans and animals.
Feeding mice with supplementary Cr-enriched B. subtilis
combined the benefits of Cr and probiotics and positively
influenced caecal microbiota, tissue Cr concentrations, insulin
receptor expression and plasma biochemical profile (100).
However, Cr is also one of the major inorganic
heavy metals and its detrimental action in the environment is well
known. Cr in aquatic environments, deriving from human activities
(textiles, electroplating and printing), is biologically toxic,
particularly in the hexavalent form CrVI. There is
limited evidence on the negative effects of Cr exposure on the GM;
according to Yao et al, CrVI can significantly
perturb GM composition and can cause intestinal tissue damage when
administrated in Bufo gargarizans (101). In mice, it perturbed microbiota
composition and structure. Still, researchers have observed that
Lactobacillus plantarum TW1-1 partially inversed Cr
exposure-linked effects (102).
Mn, an essential element, is fundamental for
physiological brain functioning and main metabolic pathways,
including enzymes. In spite of this, an excessive Mn intake is
related to hepatic damage, cardiovascular disorders and
neurodegenerative diseases (103), as a result of chronic or acute
exposure, also deriving from professional sources (104). The majority of Mn intake is
derived from water and food; however, the interactions between GM
and this element are not straightforward. Chi et al
demonstrated that Mn exposure can alter the GM structure in a
sex-specific manner in mice, can change metabolic profiles and the
expression of bacterial genes involved in neurotransmitter
biosynthesis and inflammatory cytokines (103). Moreover, Mn exposure can induce
hippocampal deterioration and death in rats, also perturbing GM
composition and changing the metabolism of tryptamine. Fecal
microbiota transplantation (FMT) could lessen the neurological
toxic effect of Mn through influencing the gut microbiome (104).
In addition, molybdenum (Mo) is an essential
element and a crucial constituent of enzymes in living organisms.
Mo pollution derives from human activities, such as in mining and
incinerators, especially when regulatory standards are not
respected. Zhou et al orally administered ammonium molybdate
to Boer goats (105). The
contents of Mo, Cu, Zn and Fe in the serum, liver, kidneys, lungs,
cardiac muscle, hair and muscle were then analyzed. The data
displayed a decrease in ciliate and protozoa protein levels. Mo
bioaccumulated in serum and tissues. The excessive Mo intake
perturbed the GM balance and interfered with Cu absorption
(105).
Vanadium, a transitional metal and essential
element, is a natural constituent of approximately 65 minerals.
Although this metal has not shown negative outcomes for human
health, several vanadium mixtures can irritate the mucosae and can
cause damage to the lungs (106).
Recently, toxic effects resulting in GM perturbation have raised a
growing interest on this metal in the breeding field, especially
poultry (107).
Ni is a metal commonly found in the environment. It
is a well-known causative agent for allergic contact dermatitis
deriving from belts, coins, jewelry. Skin contact, inhalation and
ingestion from food and stainless steel pots are the main paths
through which Ni exposure can occur (108). In a previous study, when absorbed
via oral ingestion in mice, Ni did not exert toxic effects, but
acted on the GM, resulting in alterations in metabolic balance and
microbial composition (109).
12. Conclusion
The majority of the reviewed studies demonstrated
that exposure to metals can alter the composition, diversity,
homogeneity and structure of the GM. However, it was clearly
indicated that the specific modifications reported are not
homogeneous (Table I). A number of
factors may explain this variability, including differences in
metal compound, exposure modalities (e.g. food, water, in
vitro), the duration of exposure, qualitative and quantitative
diversity of bacterial species in basal microbiota, analytical
issues. As regards the dose, it must be specified that the
sensitivity or resistance of the whole GM or of an individual
microbe to a given toxicant is strongly dependent on its role in
the exposed organism; i.e., essential metals become toxic at very
high doses, whereas metals which do not play a biological role
cause adverse effects even in very small concentrations. Moreover,
the majority of animal studies reported in the present review
addressed a single metal, while real-life exposure commonly implies
introduction of multiple metals.
 | Table ISummary of the main studies reviewed
categorized by metal and type of study. |
Table I
Summary of the main studies reviewed
categorized by metal and type of study.
| Metal | Study type | Outcomes | GM phyla | Author, year | (Refs.) |
|---|
| As | Human | ↑ Expression of
ArsB and ArsC genes | Proteobacteria | Dong et al,
2017 | (32) |
| | | ↑ Risk of
atherosclerosis | Tenericutes,
Proteobacteria, Firmicutes | Wu et al,
2019 | (33) |
| | Animal | Fatty acid,
phospholipids, cholesterols, and | Proteobacteria | Xue et al,
2019 | (37) |
| | | tryptophan pathways
alterations | | | |
| | | ↓ Gene expression
in cholesterol metabolism | | Chi et al,
2019 | (38) |
| | | LPS synthesis | Firmicutes,
Bacteroidetes | Liu et al
2019 | (39) |
| | | ↑ IL-1β, IL-2,
IL-6 | Bacteroidetes,
Proteobacteria, Firmicutes | Chiocchetti et
al, 2019 | (40) |
| | | ↑
Intestine-secreted chemokines | Firmicutes,
Bacteroidetes, Actinobacteria, Proteobacteria | Gokulan et
al, 2018 | (41) |
| | | | Saccharibacteria,
Verrucomicrobia, Deferribacteres, Spirochaetes | | |
| | | ↑ TNF-α, IFN-γ,
IL-17 | Anthophyta | Tikka et al,
2020 | (42) |
| | | ↓ IL-10 | Aphelidiomycota,
Ascomycota, Basidiomycota, Chytridiomycota | | |
| | | | Mortierellomycota,
Mucoromycota | | |
| Cd | Animal | ↓ Short-chain fatty
acids (SCFAs) production | Cyanobacteria,
Saccharibacteria, Verrucomicrobia, Tenericutes | He et al,
2020 | (48) |
| | | | Actinobacteria,
Deferribacteres, Proteobacteria, Firmicutes, Bacteroidetes | | |
| | | ↑ Serum
lipopolysaccharide | Firmicutes,
Proteobacteria | Zhang et al,
2015 | (50) |
| Cr | Animal | ↑ Insulin receptor
expression | Fusobacteria,
Bacteroidetes | Yang et al,
2016 | (100) |
| | | Intestinal tissue
damage | Fusobacteria,
Saccharibacteria | Yao et al,
2019 | (101) |
| Cu | Animal | ↑ TNFα | Firmicutes,
Bacteroidetes | Dai et al,
2020 | (82) |
| | | Liver damage | Spirochaetes,
Proteobacteria | Zhang et al,
2017 | (83) |
| | | Hepatic
steatosis | Fibrobacteres,
Euryarchaeota | Song et al,
2018 | (84) |
| | | | Cyanobacteria,
Verrucomicrobia, Tenericutes | | |
| | | | Actinobacteria,
Deferribacteres, Fusobacteria | | |
| Pb | Animal | ↑ Oxidative
stress | Actinobacteria | Cheng et al,
2019 | (71) |
| Hg | Animal | ↑ Gene apoptotic
processes | Firmicutes,
Bacteroidetes | Zhao et al,
2020 | (62) |
| | | | Proteobacteria,
Spirochaetes, Actinobacteria, Verrucomicrobia | | |
| Mn | Animal | Hepatic damage,
cardiovascular disorders | Firmicutes,
Bacteroidetes | Chi et al,
2017 | (103) |
| | | and
neurodegenerative diseases | | Wang et al,
2020 | (104) |
| Ni | Animal | Allergic contact
dermatitis | Firmicutes,
Bacteroidetes, Proteobacteria, Deferribacteres | Zhou et al,
2019 | (109) |
Investigations into the effects of nanoparticles
did not lead to straightforward conclusions: Some foresee the
premises for a safe preventive and therapeutic use, while others
have shown harmful effects on both gut microbiome and health. The
majority of the reported studies used 16S rRNA sequencing to
explore gut microbiome composition and diversity. Only a few
studies utilized modern approaches, such as metagenomic sequencing
and metabolomics, including the investigation of potential
interactions between heavy metals, gut microbiome and metabolic
pathways.
Even though further investigations are required,
these findings seem to indicate structural and functional analysis
of GM as an early biomarker of detrimental effects from exposure to
metals.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
CF and CC conceived the study. RC, NF and GB
designed and prepared the review. MT, SI and FG contributed to the
study design, and to the writing and editing of the manuscript. All
authors reviewed and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Hill DA and Artis D: Intestinal bacteria
and the regulation of immune cell homeostasis. Annu Rev Immunol.
28:623–667. 2010.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Cho I and Blaser MJ: The human microbiome:
At the interface of health and disease. Nat Rev Genet. 13:260–270.
2012.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Breton J, Massart S, Vandamme P, De Brandt
E, Pot B and Foligné B: Ecotoxicology inside the gut: Impact of
heavy metals on the mouse microbiome. BMC Pharmacol Toxicol.
14(62)2013.PubMed/NCBI View Article : Google Scholar
|
|
4
|
World Health Organization (WHO): Trace
elements in human nutrition and health. WHO, Geneva, 1996.
|
|
5
|
Rapisarda V, Miozzi E, Loreto C, Matera S,
Fenga C, Avola R and Ledda C: Cadmium exposure and prostate cancer:
Insights, mechanisms and perspectives. Front Biosci (Landmark Ed).
23:1687–1700. 2018.PubMed/NCBI View
Article : Google Scholar
|
|
6
|
Fenga C, Gangemi S, Di Salvatore V,
Falzone L and Libra M: Immunological effects of occupational
exposure to lead. Mol Med Rep. 15:3355–3360. 2017.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Planchart A, Green A, Hoyo C and Mattingly
CJ: Heavy metal exposure and metabolic syndrome: Evidence from
human and model system studies. Curr Environ Health Rep. 5:110–124.
2018.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Rehman K, Fatima F, Waheed I and Akash
MSH: Prevalence of exposure of heavy metals and their impact on
health consequences. J Cell Biochem. 119:157–184. 2018.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Sommer F and Bäckhed F: The gut
microbiota-masters of host development and physiology. Nat Rev
Microbiol. 11:227–238. 2013.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Forbes JD, Van Domselaar G and Bernstein
CN: The gut microbiota in immune-mediated inflammatory diseases.
Front Microbiol. 7(1081)2016.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Mandel LJ, Bacallao R and Zampighi G:
Uncoupling of the molecular ‘fence’ and paracellular ‘gate’
functions in epithelial tight junctions. Nature. 361:552–555.
1993.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Arumugam M, Raes J, Pelletier E, Le
Paslier D, Yamada T, Mende DR, Fernandes GR, Tap J, Bruls T, Batto
JM, et al: Enterotypes of the human gut microbiome. Nature.
473:174–180. 2011.PubMed/NCBI View Article : Google Scholar
|
|
13
|
IARC Monographs: Arsenic, Metals, Fibres,
and Dusts. IARC monographs on the evaluation of carcinogenic risk
to human. Vol 100C. IARC, Lyon, 2012.
|
|
14
|
Richardson JB Jr, Dancy BCR, Horton CL,
Lee YS, Madejczyk MS, Xu ZZ, Ackermann G, Humphrey G, Palacios G,
Knight R and Lewis JA: Exposure to toxic metals triggers unique
responses from the rat gut microbiota. Sci Rep.
8(6578)2018.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Zhang W, Guo R, Yang Y, Ding J and Zhang
Y: Long-term effect of heavy-metal pollution on diversity of
gastrointestinal microbial community of Bufo raddei. Toxicol
Lett. 258:192–197. 2016.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Li X, Brejnrod AD, Ernst M, Rykær M,
Herschend J, Olsen NMC, Dorrestein PC, Rensing C and Sørensen SJ:
Heavy metal exposure causes changes in the metabolic
health-associated gut microbiome and metabolites. Environ Int.
126:454–467. 2019.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Rothman JA, Leger L, Kirkwood JS and
McFrederick QS: Cadmium and selenate exposure affects the honey bee
microbiome and metabolome, and bee-associated bacteria show
potential for bioaccumulation. Appl Environ Microbiol.
85:e01411–19. 2019.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Laue HE, Moroishi Y, Jackson BP, Palys TJ,
Madan JC and Karagas MR: Nutrient-toxic element mixtures and the
early postnatal gut microbiome and in a United States longitudinal
birth cohort. Environ Int. 138(105613)2020.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Zhai Q, Cen S, Jiang J, Zhao J, Zhang H
and Chen W: Disturbance of trace element and gut microbiota
profiles as indicators of autism spectrum disorder: A pilot study
of Chinese children. Environ Res. 171:501–509. 2019.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Liu Y, Ji J, Zhang W, Suo Y, Zhao J, Lin
X, Cui L, Li B, Hu H, Chen C and Li YF: Selenium modulated gut
flora and promoted decomposition of methylmercury in
methylmercury-poisoned rats. Ecotoxicol Environ Saf.
185(109720)2019.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Qiu Y, Chen X, Yan X, Wang J, Yu G, Ma W,
Xiao B, Quinones S, Tian X and Ren X: Gut microbiota perturbations
and neurodevelopmental impacts in offspring rats concurrently
exposure to inorganic arsenic and fluoride. Environ Int.
140(105763)2020.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Ruan Y, Wu C, Guo X, Xu Z, Xing C, Cao H,
Zhang C, Hu G and Liu P: High doses of copper and mercury changed
cecal microbiota in female mice. Biol Trace Elem Res. 189:134–144.
2019.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Gaulke CA, Rolshoven J, Wong CP, Hudson
LG, Ho E and Sharpton TJ: Marginal zinc deficiency and
environmentally relevant concentrations of arsenic elicit combined
effects on the gut microbiome. mSphere. 3:e00521–18.
2018.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Faulkner MJ, Wenner BA, Solden LM and
Weiss WP: Source of supplemental dietary copper, zinc, and
manganese affects fecal microbial relative abundance in lactating
dairy Cows. J Dairy Sci. 100:1037–1044. 2017.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Wu N, Wang X, Xu X, Cai R and Xie S:
Effects of heavy metals on the bioaccumulation, excretion and gut
microbiome of black soldier fly larvae (Hermetia illucens).
Ecotoxicol Environ Saf. 192(110323)2020.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Yu H, Wu B, Zhang XX, Liu S, Yu J, Cheng
S, Ren HQ and Ye L: Arsenic metabolism and toxicity influenced by
ferric iron in simulated gastrointestinal tract and the roles of
gut microbiota. Environ Sci Technol. 50:7189–7197. 2016.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Yin N, Cai X, Zheng L, Du H, Wang P, Sun G
and Cui Y: In vitro assessment of arsenic release and
transformation from As(V)-sorbed goethite and jarosite: The
influence of human gut microbiota. Environ Sci Technol.
54:4432–4442. 2020.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Coryell M, McAlpine M, Pinkham N,
McDermott TR and Walk ST: The gut microbiome is required for full
protection against acute arsenic toxicity in mouse models. Nat
Commun. 9(5424)2018.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Hoen AG, Madan JC, Li Z, Coker M, Lundgren
SN, Morrison HG, Palys T, Jackson BP, Sogin ML, Cottingham KL and
Karagas MR: Sex-specific associations of infants' gut microbiome
with arsenic exposure in a US population. Sci Rep.
8(12627)2018.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Baker BA, Cassano VA, Murray C and Dreger
M: Arsenic exposure, assessment, toxicity, diagnosis, and
management: Guidance for occupational and environmental physicians.
J Occup Environ Med. 60:e634–e639. 2018.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Chi L, Bian X, Gao B, Ru H, Tu P and Lu K:
Sex-specific effects of arsenic exposure on the trajectory and
function of the gut microbiome. Chem Res Toxicol. 29:949–951.
2016.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Dong X, Shulzhenko N, Lemaitre J, Greer
RL, Peremyslova K, Quamruzzaman Q, Rahman M, Hasan OS, Joya SA,
Golam M, et al: Arsenic exposure and intestinal microbiota in
children from Sirajdikhan, Bangladesh. PLoS One.
12(e0188487)2017.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Wu F, Yang L, Islam MT, Jasmine F, Kibriya
MG, Nahar J, Barmon B, Parvez F, Sarwar G, Ahmed A, et al: The role
of gut microbiome and its interaction with arsenic exposure in
carotid intima-media thickness in a Bangladesh population. Environ
Int. 123:104–113. 2019.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Brabec JL, Wright J, Ly T, Wong HT,
McClimans CJ, Tokarev V, Lamendella R, Sherchand S, Shrestha D,
Uprety S, et al: Arsenic disturbs the gut microbiome of individuals
in a disadvantaged community in Nepal. Heliyon.
6(e03313)2020.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Chi L, Bian X, Gao B, Tu P, Ru H and Lu K:
The effects of an environmentally relevant level of arsenic on the
gut microbiome and its functional metagenome. Toxicol Sci.
160:193–204. 2017.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Chi L, Xue J, Tu P, Lai Y, Ru H and Lu K:
Gut microbiome disruption altered the biotransformation and liver
toxicity of arsenic in mice. Arch Toxicol. 93:25–35. 2019.
|
|
37
|
Xue J, Lai Y, Chi L, Tu P, Leng J, Liu CW,
Ru H and Lu K: Serum metabolomics reveals that gut microbiome
perturbation mediates metabolic disruption induced by arsenic
exposure in mice. J Proteome Res. 18:1006–1018. 2019.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Chi L, Lai Y, Tu P, Liu CW, Xue J, Ru H
and Lu K: Lipid and cholesterol homeostasis after arsenic exposure
and antibiotic treatment in mice: Potential role of the microbiota.
Environ Health Perspect. 127(97002)2019.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Liu CW, Chi L, Tu P, Xue J, Ru H and Lu K:
Isobaric Labeling quantitative metaproteomics for the study of gut
microbiome response to arsenic. J Proteome Res. 18:970–981.
2019.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Chiocchetti GM, Domene A, Kühl AA, Zúñiga
M, Vélez D, Devesa V and Monedero V: In vivo evaluation of the
effect of arsenite on the intestinal epithelium and associated
microbiota in mice. Arch Toxicol. 93:2127–2139. 2019.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Gokulan K, Arnold MG, Jensen J,
Vanlandingham M, Twaddle NC, Doerge DR, Cerniglia CE and Khare S:
Exposure to arsenite in CD-1 mice during juvenile and adult stages:
Effects on intestinal microbiota and Gut-associated immune status.
mBio. 9:e01418–18. 2018.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Tikka C, Manthari RK, Ommati MM, Niu R,
Sun Z, Zhang J and Wang J: Immune disruption occurs through altered
gut microbiome and NOD2 in arsenic induced mice: Correlation with
colon cancer markers. Chemosphere. 246(125791)2020.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Wang HT, Ding J, Xiong C, Zhu D, Li G, Jia
XY, Zhu YG and Xue XM: Exposure to microplastics lowers arsenic
accumulation and alters gut bacterial communities of earthworm
Metaphire californica. Environ Pollut. 251:110–116.
2019.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Wang HT, Zhu D, Li G, Zheng F, Ding J,
O'Connor PJ, Zhu YG and Xue XM: Effects of arsenic on gut
microbiota and its biotransformation genes in earthworm metaphire
sieboldi. Environ Sci Technol. 53:3841–3849. 2019.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Yin N, Du H, Zhang Z, Cai X, Li Z, Sun G
and Cui Y: Variability of arsenic bioaccessibility and metabolism
in soils by human gut microbiota using different in vitro methods
combined with SHIME. Sci Total Environ. 566-567:1670–1677.
2016.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Yin N, Cai X, Du H, Zhang Z, Li Z, Chen X,
Sun G and Cui Y: In vitro study of soil arsenic release by human
gut microbiota and its intestinal absorption by Caco-2 cells.
Chemosphere. 168:358–364. 2017.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Li J, Mandal G and Rosen BP: Expression of
arsenic resistance genes in the obligate anaerobe Bacteroides
vulgatus ATCC 8482, a gut microbiome bacterium. Anaerobe.
39:117–123. 2016.PubMed/NCBI View Article : Google Scholar
|
|
48
|
He X, Qi Z, Hou H, Qian L, Gao J and Zhang
XX: Structural and functional alterations of gut microbiome in mice
induced by chronic cadmium exposure. Chemosphere.
246(125747)2020.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Šrut M, Menke S, Höckner M and Sommer S:
Earthworms and cadmium-Heavy metal resistant gut bacteria as
indicators for heavy metal pollution in soils? Ecotoxicol Environ
Saf. 171:843–853. 2019.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Zhang S, Jin Y, Zeng Z, Liu Z and Fu Z:
Subchronic exposure of mice to cadmium perturbs their hepatic
energy metabolism and gut microbiome. Chem Res Toxicol.
28:2000–2009. 2015.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Ba Q, Li M, Chen P, Huang C, Duan X, Lu L,
Li J, Chu R, Xie D, Song H, et al: Sex-dependent effects of cadmium
exposure in early life on gut microbiota and fat accumulation in
mice. Environ Health Perspect. 125:437–446. 2017.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Feng S, Liu Y, Huang Y, Zhao J, Zhang H,
Zhai Q and Chen W: Influence of oral administration of
Akkermansia muciniphila on the tissue distribution and gut
microbiota composition of acute and chronic cadmium exposure mice.
FEMS Microbiol Lett. 366(fnz160)2019.PubMed/NCBI View Article : Google Scholar
|
|
53
|
He X, Qi Z, Hou H, Gao J and Zhang XX:
Effects of chronic cadmium exposure at food limitation-relevant
levels on energy metabolism in mice. J Hazard Mater.
388(121791)2020.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Chang X, Li H, Feng J, Chen Y, Nie G and
Zhang J: Effects of cadmium exposure on the composition and
diversity of the intestinal microbial community of common carp
(Cyprinus carpio L.). Ecotoxicol Environ Saf. 171:92–98.
2019.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Zhang Y, Li Z, Kholodkevich S, Sharov A,
Chen C, Feng Y, Ren N and Sun K: Effects of cadmium on intestinal
histology and microbiota in freshwater crayfish (Procambarus
clarkii). Chemosphere. 242(125105)2020.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Wang N, Jiang M, Zhang P, Shu H and Li Y,
Guo Z and Li Y: Amelioration of Cd-induced bioaccumulation,
oxidative stress and intestinal microbiota by Bacillus
cereus in Carassius auratus gibelio. Chemosphere.
245(125613)2020.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Zhai Q, Yu L, Li T, Zhu J, Zhang C, Zhao
J, Zhang H and Chen W: Effect of dietary probiotic supplementation
on intestinal microbiota and physiological conditions of Nile
tilapia (Oreochromis niloticus) under waterborne cadmium
exposure. Antonie Van Leeuwenhoek. 110:501–513. 2017.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Ya J, Ju Z, Wang H and Zhao H: Exposure to
cadmium induced gut histopathological damages and microbiota
alterations of Chinese toad (Bufo gargarizans) larvae.
Ecotoxicol Environ Saf. 180:449–456. 2019.PubMed/NCBI View Article : Google Scholar : Exposure to
cadmium induced gut histopathological damages and microbiota
alterations of Chinese toad (Bufo gargarizans) larvae.
|
|
59
|
Ya J, Li X, Wang L, Kou H, Wang H and Zhao
H: The effects of chronic cadmium exposure on the gut of Bufo
gargarizans larvae at metamorphic climax: Histopathological
impairments, microbiota changes and intestinal remodeling
disruption. Ecotoxicol Environ Saf. 195(110523)2020.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Mu D, Meng J, Bo X, Wu M, Xiao H and Wang
H: The effect of cadmium exposure on diversity of intestinal
microbial community of Rana chensinensis tadpoles.
Ecotoxicol Environ Saf. 154:6–12. 2018.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Lee S, Kim Y and Choi J: Effect of soil
microbial feeding on gut microbiome and cadmium toxicity in
Caenorhabditis elegans. Ecotoxicol Environ Saf.
187(109777)2020.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Zhao Y, Zhou C, Wu C, Guo X, Hu G, Wu Q,
Xu Z, Li G, Cao H, Li L, et al: Subchronic oral mercury caused
intestinal injury and changed gut microbiota in mice. Sci Total
Environ. 721(137639)2020.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Lin X, Zhao J, Zhang W, He L, Wang L,
Chang D, Cui L, Gao Y, Li B, Chen C and Li YF: Acute oral
methylmercury exposure perturbs the gut microbiome and alters
gut-brain axis related metabolites in rats. Ecotoxicol Environ Saf.
190(110130)2020.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Rothenberg SE, Wagner CL, Hamidi B,
Alekseyenko AV and Andrea Azcarate-Peril M: Longitudinal changes
during pregnancy in gut microbiota and methylmercury biomarkers,
and reversal of microbe-exposure correlations. Environ Res.
172:700–712. 2019.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Zhang BB, Liu YM, Hu AL, Xu SF, Fan LD,
Cheng ML, Li C, Wei LX and Liu J: HgS and Zuotai differ from HgCl2
and methyl mercury in intestinal Hg absorption, transporter
expression and gut microbiome in mice. Toxicol Appl Pharmacol.
379(114615)2019.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Zhou C, Xu P, Huang C, Liu G, Chen S, Hu
G, Li G, Liu P and Guo X: Effects of subchronic exposure of
mercuric chloride on intestinal histology and microbiota in the
cecum of chicken. Ecotoxicol Environ Saf.
188(109920)2020.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Eggers S, Safdar N, Sethi AK, Suen G,
Peppard PE, Kates AE, Skarlupka JH, Kanarek M and Malecki KMC:
Urinary lead concentration and composition of the adult gut
microbiota in a cross-sectional population-based sample. Environ
Int. 133(105122)2019.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Fenga C, Gangemi S, Alibrandi A, Costa C
and Micali E: Relationship between lead exposure and mild cognitive
impairment. J Prev Med Hyg. 57:E205–E210. 2016.PubMed/NCBI
|
|
69
|
Gao B, Chi L, Mahbub R, Bian X, Tu P, Ru H
and Lu K: Multi-omics reveals that lead exposure disturbs gut
microbiome development, key metabolites, and metabolic pathways.
Chem Res Toxicol. 30:996–1005. 2017.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Yu L, Yu Y, Yin R, Duan H, Qu D, Tian F,
Narbad A, Chen W and Zhai Q: Dose-dependent effects of lead induced
gut injuries: An in vitro and in vivo study. Chemosphere.
266(129130)2021.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Cheng D, Li H, Zhou J and Wang S:
Chlorogenic acid relieves lead-induced cognitive impairments and
hepato-renal damage: Via regulating the dysbiosis of the gut
microbiota in mice. Food Funct. 10:681–690. 2019.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Wu J, Wen XW, Faulk C, Boehnke K, Zhang H,
Dolinoy DC and Xi C: Perinatal lead exposure alters gut microbiota
composition and results in sex-specific bodyweight increases in
adult mice. Toxicol Sci. 151:324–333. 2016.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Kou H, Fu Y, He Y, Jiang J, Gao X and Zhao
H: Chronic lead exposure induces histopathological damage,
microbiota dysbiosis and immune disorder in the cecum of female
Japanese quails (Coturnix japonica). Ecotoxicol Environ Saf.
183(109588)2019.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Xia J, Lu L, Jin C, Wang S, Zhou J, Ni Y,
Fu Z and Jin Y: Effects of short term lead exposure on gut
microbiota and hepatic metabolism in adult zebrafish. Comp Biochem
Physiol C Toxicol Pharmacol. 209:1–8. 2018.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Du H, Yin N, Cai X, Wang P, Li Y, Fu Y,
Sultana MS, Sun G and Cui Y: Lead bioaccessibility in farming and
mining soils: The influence of soil properties, types and human gut
microbiota. Sci Total Environ. 708(135227)2020.PubMed/NCBI View Article : Google Scholar
|
|
76
|
Reed S, Knez M, Uzan A, Stangoulis JCR,
Glahn RP, Koren O and Tako E: Alterations in the Gut (Gallus
gallus) microbiota following the consumption of Zinc
biofortified wheat (Triticum aestivum)-based diet. J Agric
Food Chem. 66:6291–6299. 2018.PubMed/NCBI View Article : Google Scholar
|
|
77
|
Podany A, Rauchut J, Wu T, Kawasawa YI,
Wright J, Lamendella R, Soybel DI and Kelleher SL: Excess dietary
Zinc intake in neonatal mice causes oxidative stress and alters
intestinal host-microbe interactions. Mol Nutr Food Res.
63(e1800947)2019.PubMed/NCBI View Article : Google Scholar
|
|
78
|
Ishaq SL, Page CM, Yeoman CJ, Murphy TW,
Van Emon ML and Stewart WC: Zinc AA supplementation alters yearling
ram rumen bacterial communities but zinc sulfate supplementation
does not. J Anim Sci. 97:687–697. 2019.PubMed/NCBI View Article : Google Scholar
|
|
79
|
Zackular JP, Moore JL, Jordan AT,
Juttukonda LJ, Noto MJ, Nicholson MR, Crews JD, Semler MW, Zhang Y,
Ware LB, et al: Dietary zinc alters the microbiota and decreases
resistance to Clostridium difficile infection. Nat Med.
22:1330–1334. 2016.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Zhang F, Zheng W, Xue Y and Yao W: Suhuai
suckling piglet hindgut microbiome-metabolome responses to
different dietary copper levels. Appl Microbiol Biotechnol.
103:853–868. 2019.PubMed/NCBI View Article : Google Scholar
|
|
81
|
Cheng S, Mao H, Ruan Y, Wu C, Xu Z, Hu G,
Guo X, Zhang C, Cao H and Liu P: Copper changes intestinal
microbiota of the cecum and rectum in female mice by 16S rRNA gene
sequencing. Biol Trace Elem Res. 193:445–455. 2020.PubMed/NCBI View Article : Google Scholar
|
|
82
|
Dai J, Yang X, Yuan Y, Jia Y, Liu G, Lin
N, Xiao H, Zhang L and Chen J: Toxicity, gut microbiota and
metabolome effects after copper exposure during early life in SD
rats. Toxicology. 433–434. 2020.PubMed/NCBI View Article : Google Scholar
|
|
83
|
Zhang F, Zheng W, Guo R and Yao W: Effect
of dietary copper level on the gut microbiota and its correlation
with serum inflammatory cytokines in Sprague-Dawley rats. J
Microbiol. 55:694–702. 2017.PubMed/NCBI View Article : Google Scholar
|
|
84
|
Song M, Li X, Zhang X, Shi H, Vos MB, Wei
X, Wang Y, Gao H, Rouchka EC, Yin X, et al: Dietary copper-fructose
interactions alter gut microbial activity in male rats. Am J
Physiol Gastrointest Liver Physiol. 314:G119–G130. 2018.PubMed/NCBI View Article : Google Scholar
|
|
85
|
Wang T, Wei X, Chen T, Wang W, Xia X, Miao
J and Yin S: Studies of the mechanism of fatty liver formation in
Takifugu fasciatus following copper exposure. Ecotoxicol
Environ Saf. 181:353–361. 2019.PubMed/NCBI View Article : Google Scholar
|
|
86
|
Meng XL, Li S, Qin CB, Zhu ZX, Hu WP, Yang
LP, Lu RH, Li WJ and Nie GX: Intestinal microbiota and lipid
metabolism responses in the common carp (Cyprinus carpio L.)
following copper exposure. Ecotoxicol Environ Saf. 160:257–264.
2018.PubMed/NCBI View Article : Google Scholar
|
|
87
|
Yang Y, Song X, Chen A, Wang H and Chai L:
Exposure to copper altered the intestinal microbiota in Chinese
brown frog (Rana chensinensis). Environ Sci Pollut Res.
27:13855–13865. 2020.PubMed/NCBI View Article : Google Scholar
|
|
88
|
Hemarajata P and Versalovic J: Effects of
probiotics on gut microbiota: Mechanisms of intestinal
immunomodulation and neuromodulation. Therap Adv Gastroenterol.
6:39–51. 2013.PubMed/NCBI View Article : Google Scholar
|
|
89
|
Yausheva Е, Sizova Е, Lebedev S, Skalny A,
Miroshnikov S, Plotnikov A, Khlopko Y, Gogoleva N and Cherkasov S:
Influence of zinc nanoparticles on survival of worms Eisenia fetida
and taxonomic diversity of the gut microflora. Environ Sci Pollut
Res. 23:13245–13254. 2016.PubMed/NCBI View Article : Google Scholar
|
|
90
|
Costa C, Miozzi E, Teodoro M, Briguglio G,
Rapisarda V and Fenga C: New insights on ‘old’ toxicants in
occupational toxicology (Review). Mol Med Rep. 15:3317–3322.
2017.PubMed/NCBI View Article : Google Scholar
|
|
91
|
Patsiou D, Del Rio-Cubilledo C, Catarino
AI, Summers S, Mohd Fahmi A, Boyle D, Fernandes TF and Henry TB:
Exposure to Pb-halide perovskite nanoparticles can deliver
bioavailable Pb but does not alter endogenous gut microbiota in
zebrafish. Sci Total Environ. 715(136941)2020.PubMed/NCBI View Article : Google Scholar
|
|
92
|
Cholewińska E, Ognik K, Fotschki B,
Zduńczyk Z and Juśkiewicz J: Comparison of the effect of dietary
copper nanoparticles and one copper (II) salt on the copper
biodistribution and gastrointestinal and hepatic morphology and
function in a rat model. PLoS One. 13(e0197083)2018.PubMed/NCBI View Article : Google Scholar
|
|
93
|
Swart E, Dvorak J, Hernádi S, Goodall T,
Kille P, Spurgeon D, Svendsen C and Prochazkova P: The effects of
in vivo exposure to copper oxide nanoparticles on the gut
microbiome, host immunity, and susceptibility to a bacterial
infection in earthworms. Nanomaterials. 10(1337)2020.PubMed/NCBI View Article : Google Scholar
|
|
94
|
Sizentsov AN, Kvan OV, Miroshnikova EP,
Gavrish IA, Serdaeva VA and Bykov AV: Assessment of biotoxicity of
Cu nanoparticles with respect to probiotic strains of
microorganisms and representatives of the normal flora of the
intestine of broiler chickens. Environ Sci Pollut Res.
25:15765–15773. 2018.PubMed/NCBI View Article : Google Scholar
|
|
95
|
Wang C, Cheng K, Zhou L, He J, Zheng X,
Zhang L, Zhong X and Wang T: Evaluation of long-term toxicity of
oral zinc oxide nanoparticles and zinc sulfate in mice. Biol Trace
Elem Res. 178:276–282. 2017.PubMed/NCBI View Article : Google Scholar
|
|
96
|
Song R, Yao J, Shi Q and Wei R:
Nanocomposite of Half-Fin anchovy Hydrolysates/Zinc oxide
nanoparticles exhibits actual non-toxicity and regulates intestinal
microbiota, Short-Chain fatty acids production and oxidative status
in mice. Mar Drugs. 16(23)2018.PubMed/NCBI View Article : Google Scholar
|
|
97
|
Pei X, Xiao Z, Liu L, Wang G, Tao W, Wang
M, Zou J and Leng D: Effects of dietary zinc oxide nanoparticles
supplementation on growth performance, zinc status, intestinal
morphology, microflora population, and immune response in weaned
pigs. J Sci Food Agric. 99:1366–1374. 2019.PubMed/NCBI View Article : Google Scholar
|
|
98
|
Kolba N, Guo Z, Olivas FM, Mahler GJ and
Tako E: Intra-amniotic administration (Gallus gallus) of TiO
2, SiO 2, and ZnO nanoparticles affect brush
border membrane functionality and alters gut microflora
populations. Food Chem Toxicol. 135(110896)2020.PubMed/NCBI View Article : Google Scholar
|
|
99
|
Vamanu E, Ene M, Biță B, Ionescu C,
Crăciun L and Sârbu I: In vitro human microbiota response to
exposure to silver nanoparticles biosynthesized with mushroom
extract. Nutrients. 10(607)2018.PubMed/NCBI View Article : Google Scholar
|
|
100
|
Yang J, Xu Y, Qian K, Zhang W, Wu D and
Wang C: Effects of chromium-enriched Bacillus subtilis KT260179
supplementation on growth performance, caecal microbiology, tissue
chromium level, insulin receptor expression and plasma biochemical
profile of mice under heat stress. Br J Nutr. 115:774–781.
2016.PubMed/NCBI View Article : Google Scholar
|
|
101
|
Yao Q, Yang H, Wang X and Wang H: Effects
of hexavalent chromium on intestinal histology and microbiota in
Bufo gargarizans tadpoles. Chemosphere. 216:313–323.
2019.PubMed/NCBI View Article : Google Scholar
|
|
102
|
Wu G, Xiao X, Feng P, Xie F, Yu Z, Yuan W,
Liu P and Li X: Gut remediation: A potential approach to reducing
chromium accumulation using Lactobacillus plantarum TW1-1.
Sci Rep. 7(15000)2017.PubMed/NCBI View Article : Google Scholar
|
|
103
|
Chi L, Gao B, Bian X, Tu P, Ru H and Lu K:
Manganese-induced sex-specific gut microbiome perturbations in
C57BL/6 mice. Toxicol Appl Pharmacol. 331:142–153. 2017.PubMed/NCBI View Article : Google Scholar
|
|
104
|
Wang H, Zhang S, Yang F, Xin R, Wang S,
Cui D and Sun Y: The gut microbiota confers protection in the CNS
against neurodegeneration induced by manganism. Biomed
Pharmacother. 127(110150)2020.PubMed/NCBI View Article : Google Scholar
|
|
105
|
Zhou S, Zhang C, Xiao Q, Zhuang Y, Gu X,
Yang F, Xing C, Hu G and Cao H: Effects of different levels of
molybdenum on rumen microbiota and trace elements changes in
tissues from goats. Biol Trace Elem Res. 174:85–92. 2016.PubMed/NCBI View Article : Google Scholar
|
|
106
|
Del Carpio E, Hernández L, Ciangherotti C,
Villalobos Coa V, Jiménez L, Lubes V and Lubes G: Vanadium:
History, chemistry, interactions with α-amino acids and potential
therapeutic applications. Coord Chem Rev. 372:117–140.
2018.PubMed/NCBI View Article : Google Scholar
|
|
107
|
Yuan ZH, Wang JP, Zhang KY, Ding XM, Bai
SP, Zeng QF, Xuan Y and Su ZW: Effect of vanadium and tea
polyphenols on intestinal morphology, microflora and Short-Chain
fatty acid profile of laying hens. Biol Trace Elem Res.
174:419–427. 2016.PubMed/NCBI View Article : Google Scholar
|
|
108
|
Guarneri F, Costa C, Cannavò SP, Catania
S, Bua GD, Fenga C and Dugo G: Release of nickel and chromium in
common foods during cooking in 18/10 (grade 316) stainless steel
pots. Contact Dermatitis. 76:40–48. 2017.PubMed/NCBI View Article : Google Scholar
|
|
109
|
Zhou X, Li J and Sun JL: Oral nickel
changes of intestinal microflora in mice. Curr Microbiol.
76:590–596. 2019.PubMed/NCBI View Article : Google Scholar
|