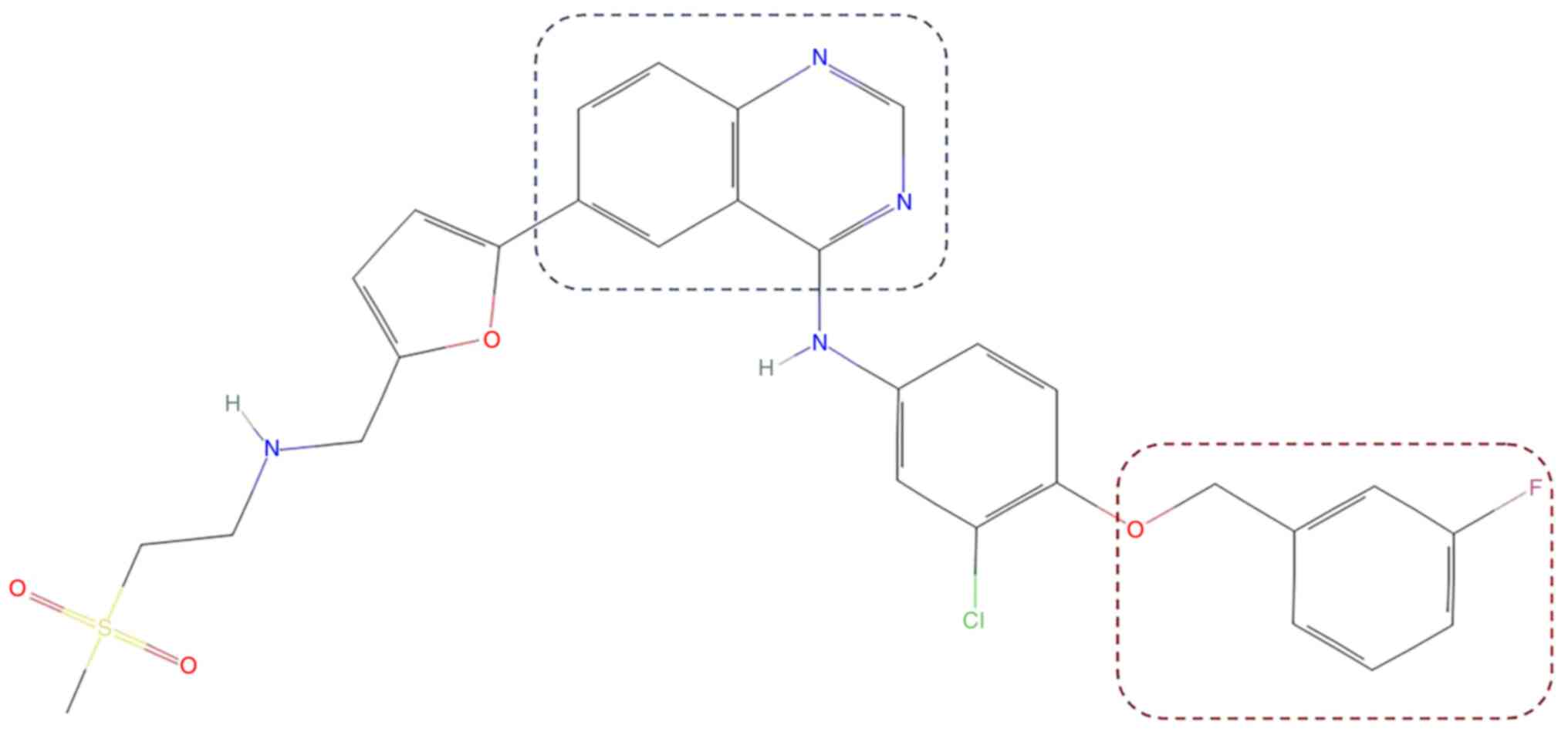
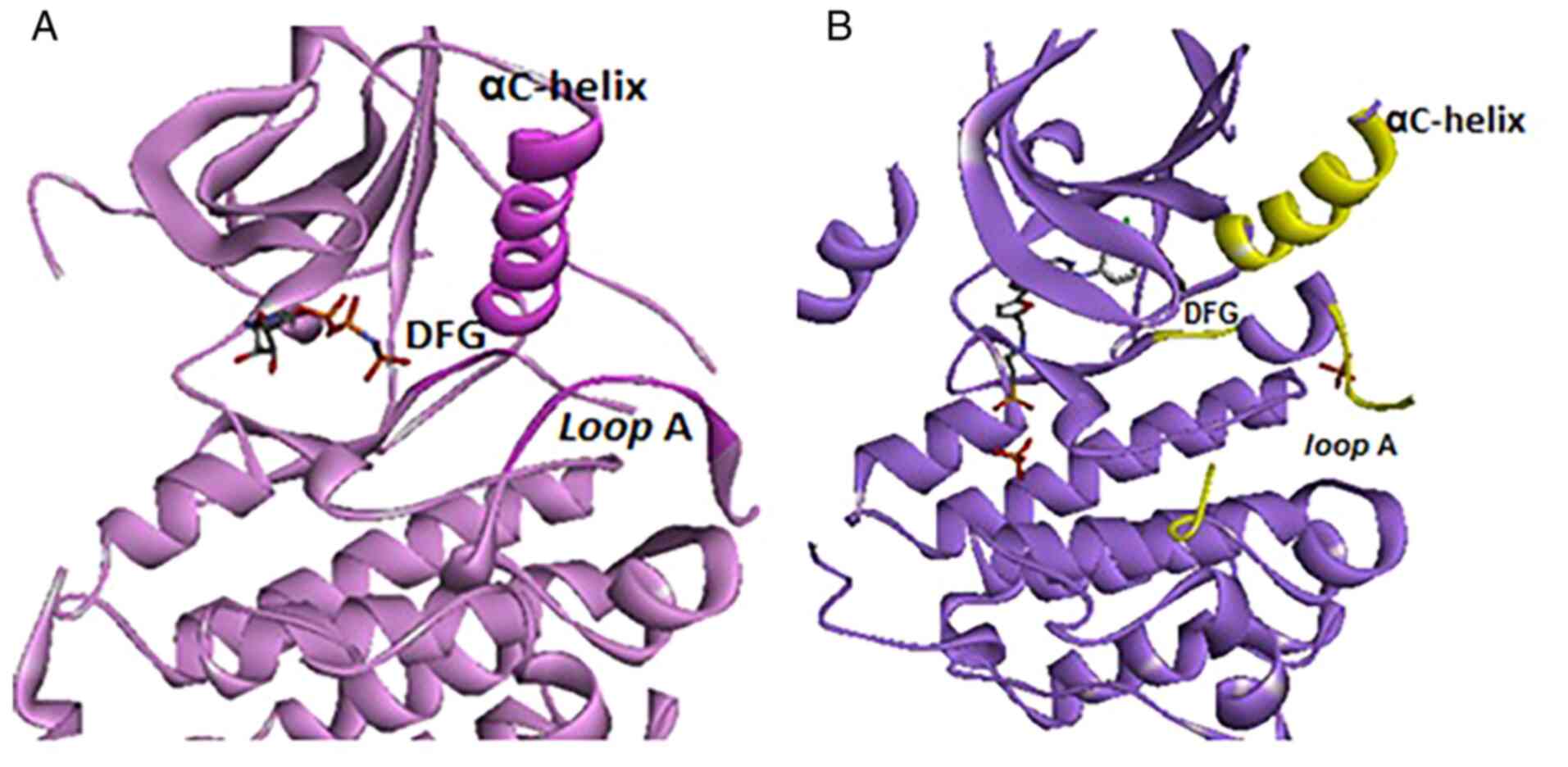
|
1
|
Johnson DE, Burtness B, Leemans CR, Lui
VWY, Bauman JE and Grandis JR: Head and neck squamous cell
carcinoma. Nat Rev Dis Primers. 6(92)2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Sung H, Ferlay J, Siegel RL, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global cancer statistics 2020:
GLOBOCAN estimates of incidence and mortality worldwide for 36
cancers in 185 countries. CA Cancer J Clin. 71:209–249.
2021.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Ghiani L and Chiocca S: High risk-human
papillomavirus in HNSCC: Present and future challenges for
epigenetic therapies. Int J Mol Sci. 23(3483)2022.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Blaszczak W, Barczak W, Wegner A,
Golusinski W and Suchorska WM: Clinical value of monoclonal
antibodies and tyrosine kinase inhibitors in the treatment of head
and neck squamous cell carcinoma. Med Oncol. 34(60)2017.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Denaro N and Merlano MC: Profile of
afatinib and its potential in the treatment of head and neck
cancer: Current evidence. J Oncol Transl Res. 1(101)2015.
|
|
6
|
Aung KL and Siu LL: Genomically
personalized therapy in head and neck cancer. Cancers Head Neck.
1(2)2016.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Sacco AG and Worden FP: Molecularly
targeted therapy for the treatment of head and neck cancer: A
review of the ErbB family inhibitors. Onco Targets Ther.
9:1927–1943. 2016.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Chapman CH, Saba NF and Yom SS: Targeting
epidermal growth factor receptor for head and neck squamous cell
carcinoma: Still lost in translation? Ann Transl Med.
4(80)2016.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Bossi P, Resteghini C, Paielli N, Licitra
L, Pilotti S and Perrone F: Prognostic and predictive value of EGFR
in head and neck squamous cell carcinoma. Oncotarget.
7:74362–74379. 2016.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Cassell A and Grandis JR: Investigational
EGFR-targeted therapy in head and neck squamous cell carcinoma.
Expert Opin Investig Drugs. 19:709–722. 2010.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Sacco AG and Cohen EE: Current treatment
options for recurrent or metastatic head and neck squamous cell
carcinoma. J Clin Oncol. 33:3305–3315. 2015.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Vermorken JB, Trigo J, Hitt R, Koralewski
P, Diaz-Rubio E, Rolland F, Knecht R, Amellal N, Schueler A and
Baselga J: Open-label, uncontrolled, multicenter phase II study to
evaluate the efficacy and toxicity of cetuximab as a single agent
in patients with recurrent and/or metastatic squamous cell
carcinoma of the head and neck who failed to respond to
platinum-based therapy. J Clin Oncol. 25:2171–2177. 2007.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Iida M, Brand TM, Starr MM, Huppert EJ,
Luthar N, Bahrar H, Coan JP, Pearson HE, Salgia R and Wheeler DL:
Overcoming acquired resistance to cetuximab by dual targeting HER
family receptors with antibody-based therapy. Mol Cancer.
13(242)2014.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Pollock NI and Grandis JR: HER2 as a
therapeutic target in head and neck squamous cell carcinoma. Clin
Cancer Res. 21:526–533. 2015.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Appert-Collin A, Hubert P, Crémel G and
Bennasroune A: Role of ErbB receptors in cancer cell migration and
invasion. Front Pharmacol. 6(283)2015.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Cierpikowski P, Lis-Nawara A, Gajdzis P
and Bar J: PDGFRα/HER2 and PDGFRα/p53 co-expression in oral
squamous cell carcinoma. Anticancer Res. 38:795–802.
2018.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Birkeland AC, Yanik M, Tillman BN, Scott
MV, Foltin SK, Mann JE, Michmerhuizen NL, Ludwig ML, Sandelski MM,
Komarck CM, et al: Identification of targetable HER2 aberrations in
head and neck squamous cell carcinoma. JAMA Otolaryngol Head Neck
Surg. 142:559–567. 2016.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Hynes NE: ErbB2: From an EGFR relative to
a central target for cancer therapy. Cancer Res. 76:3659–3662.
2016.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Arkhipov A, Shan Y, Kim ET, Dror RO and
Shaw DE: Her2 activation mechanism reflects evolutionary
preservation of asymmetric ectodomain dimers in the human EGFR
family. Elife. 2(e00708)2013.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Wang D, Qian G, Zhang H, Magliocca KR,
Nannapaneni S, Amin AR, Rossi M, Patel M, El-Deiry M, Wadsworth JT,
et al: HER3 targeting sensitizes HNSCC to cetuximab by reducing
HER3 Activity and HER2/HER3 dimerization: Evidence from cell line
and patient-derived xenograft models. Clin Cancer Res. 23:677–686.
2017.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Strüder D, Momper T, Irmscher N, Krause M,
Liese J, Schraven S, Zimpfer A, Zonnur S, Burmeister AS, Schneider
B, et al: Establishment and characterization of patient-derived
head and neck cancer models from surgical specimens and endoscopic
biopsies. J Exp Clin Cancer Res. 40(246)2021.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Facompre ND, Rajagopalan P, Sahu V,
Pearson AT, Montone KT, James CD, Gleber-Netto FO, Weinstein GS,
Jalaly J, Lin A, et al: Identifying predictors of HPV-related head
and neck squamous cell carcinoma progression and survival through
patient-derived models. Int J Cancer. 147:3236–3249.
2020.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Tebbutt N, Pedersen MW and Johns TG:
Targeting the ERBB family in cancer: Couples therapy. Nat Rev
Cancer. 13:663–673. 2013.PubMed/NCBI View
Article : Google Scholar
|
|
24
|
Martinez-Useros J and Garcia-Foncillas J:
The challenge of blocking a wider family members of EGFR against
head and neck squamous cell carcinomas. Oral Oncol. 51:423–430.
2015.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Slobbe P, Windhorst AD, Stigter-van Walsum
M, Smit EF, Niessen HG, Solca F, Stehle G, van Dongen GA and Poot
AJ: A comparative PET imaging study with the reversible and
irreversible EGFR tyrosine kinase inhibitors [(11)C]erlotinib and
[(18)F]afatinib in lung cancer-bearing mice. EJNMMI Res.
5(14)2015.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Fabbro D, Cowan-Jacob SW and Moebitz H:
Ten things you should know about protein kinases: IUPHAR review 14.
Br J Pharmacol. 172:2675–2700. 2015.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Carmi C, Mor M, Petronini PG and Alfieri
RR: Clinical perspectives for irreversible tyrosine kinase
inhibitors in cancer. Biochem Pharmacol. 84:1388–1399.
2012.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Schwartz PA, Kuzmic P, Solowiej J,
Bergqvist S, Bolanos B, Almaden C, Nagata A, Ryan K, Feng J, Dalvie
D, et al: Covalent EGFR inhibitor analysis reveals importance of
reversible interactions to potency and mechanisms of drug
resistance. Proc Natl Acad Sci USA. 111:173–178. 2014.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Solca F, Dahl G, Zoephel A, Bader G,
Sanderson M, Klein C, Kraemer O, Himmelsbach F, Haaksma E and Adolf
GR: Target binding properties and cellular activity of afatinib
(BIBW 2992), an irreversible ErbB family blocker. J Pharmacol Exp
Ther. 343:342–350. 2012.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Hoelder S, Clarke PA and Workman P:
Discovery of small molecule cancer drugs: Successes, challenges and
opportunities. Mol Oncol. 6:155–176. 2012.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Weiss JM, Bagley S, Hwang WT, Bauml J,
Olson JG, Cohen RB, Hayes DN and Langer C: Capecitabine and
lapatinib for the first-line treatment of metastatic/recurrent head
and neck squamous cell carcinoma. Cancer. 122:2350–2355.
2016.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Chen L, Zhang Y, Liu J, Wang W, Li X, Zhao
L, Wang W and Li B: Novel 4-arylaminoquinazoline derivatives with
(E)-propen-1-yl moiety as potent EGFR inhibitors with enhanced
antiproliferative activities against tumor cells. Eur J Med Chem.
138:689–697. 2017.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Dhuguru J, Liu W, Gonzalez WG, Babinchak
WM, Miksovska J, Landgraf R and Wilson JN: Emission tuning of
fluorescent kinase inhibitors: Conjugation length and substituent
effects. J Org Chem. 79:4940–4947. 2014.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Segovia-Mendoza M, González-González ME,
Barrera D, Díaz L and García-Becerra R: Efficacy and mechanism of
action of the tyrosine kinase inhibitors gefitinib, lapatinib and
neratinib in the treatment of HER2-positive breast cancer:
Preclinical and clinical evidence. Am J Cancer Res. 5:2531–2561.
2015.PubMed/NCBI
|
|
35
|
Bernard-Gauthier V, Bailey JJ, Berke S and
Schirrmacher R: Recent advances in the development and application
of radiolabeled kinase inhibitors for PET imaging. Molecules.
20:22000–22027. 2015.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Harrington K, Temam S, Mehanna H, D'Cruz
A, Jain M, D'Onofrio I, Manikhas G, Horvath Z, Sun Y, Dietzsch S,
et al: Postoperative adjuvant lapatinib and concurrent
chemoradiotherapy followed by maintenance lapatinib monotherapy in
high-risk patients with resected squamous cell carcinoma of the
head and neck: A phase III, randomized, double-blind,
placebo-controlled study. J Clin Oncol. 33:4202–4209.
2015.PubMed/NCBI View Article : Google Scholar
|
|
37
|
de Souza JA, Davis DW, Zhang Y, Khattri A,
Seiwert TY, Aktolga S, Wong SJ, Kozloff MF, Nattam S, Lingen MW, et
al: A phase II study of lapatinib in recurrent/metastatic squamous
cell carcinoma of the head and neck. Clin Cancer Res. 18:2336–2343.
2012.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Worsham MJ, Ali H, Dragovic J and
Schweitzer VP: Molecular characterization of head and neck cancer:
How close to personalized targeted therapy. Mol Diagn Ther.
16:209–222. 2012.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Roskoski R Jr: Classification of small
molecule protein kinase inhibitors based upon the structures of
their drug-enzyme complexes. Pharmacol Res. 103:26–48.
2016.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Pereira M, Verma CS and Fuentes G:
Differences in the binding affinities of ErbB family: Heterogeneity
in the prediction of resistance mutants. PLoS One.
8(e77054)2013.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Novotny CJ, Pollari S, Park JH, Lemmon MA,
Shen W and Shokat KM: Overcoming resistance to HER2 inhibitors
through state-specific kinase binding. Nat Chem Biol. 12:923–930.
2016.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Wang Z and Cole PA: Catalytic mechanisms
and regulation of protein kinases. Methods Enzymol. 548:1–21.
2014.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Dixit A and Verkhivker GM:
Structure-functional prediction and analysis of cancer mutation
effects in protein kinases. Comput Math Methods Med.
2014(653487)2014.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Bose R and Zhang X: The ErbB kinase
domain: Structural perspective into kinase activation and
inhibition. Exp Cell Res. 315:649–658. 2010.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Sierra JR, Cepero V and Giordano S:
Molecular mechanisms of acquired resistance to tyrosine kinase
targeted therapy. Mol Cancer. 9(75)2010.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Schroeder RL, Stevens CL and Sridhar J:
Small molecule tyrosine kinase inhibitors of ErbB2/HER2/Neu in the
treatment of aggressive breast cancer. Molecules. 19:15196–15212.
2014.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Müller S, Chaikuad A, Gray NS and Knapp S:
The ins and outs of selective kinase inhibitor development. Nat
Chem Biol. 11:818–821. 2015.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Vijayan RS, He P, Modi V, Duong-Ly KC, Ma
H, Peterson JR, Dunbrack RL Jr and Levy RM: Conformational analysis
of the DFG-out kinase motif and biochemical profiling of
structurally validated type II inhibitors. J Med Chem. 58:466–479.
2015.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Songtawee N, Bevan DR and Choowongkomon K:
Molecular dynamics of the asymmetric dimers of EGFR: Simulations on
the active and inactive conformations of the kinase domain. J Mol
Graph Model. 58:16–29. 2015.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Liu W, Ning JF, Meng QW, Hu J, Zhao YB,
Liu C and Cai L: Navigating into the binding pockets of the HER
family protein kinases: Discovery of novel EGFR inhibitor as
antitumor agent. Drug Des Devel Ther. 9:3837–3851. 2015.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Rudolph J, Xiao Y, Pardi A and Ahn NG:
Slow inhibition and conformation selective properties of
extracellular signal-regulated kinase 1 and 2 inhibitors.
Biochemistry. 54:22–31. 2015.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Tonge PJ: Drug-target kinetics in drug
discovery. ACS Chem Neurosci. 9:29–39. 2018.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Copeland RA: The drug-target residence
time model: A 10-year retrospective. Nat Rev Drug Discov. 15:87–95.
2016.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Fumagalli I, Dugue D, Bibault JE,
Clémenson C, Vozenin MC, Mondini M and Deutsch E: Cytotoxic effect
of lapatinib is restricted to human papillomavirus-positive head
and neck squamous cell carcinoma cell lines. Onco Targets Ther.
8:335–345. 2015.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Hama T, Yuza Y, Saito Y, O-uchi J, Kondo
S, Okabe M, Yamada H, Kato T, Moriyama H, Kurihara S and Urashima
M: Prognostic significance of epidermal growth factor receptor
phosphorylation and mutation in head and neck squamous cell
carcinoma. Oncologist. 14:900–908. 2009.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Del Campo JM, Hitt R, Sebastian P,
Carracedo C, Lokanatha D, Bourhis J, Temam S, Cupissol D, De
Raucourt D, Maroudias N, et al: Effects of lapatinib monotherapy:
Results of a randomised phase II study in therapy-naive patients
with locally advanced squamous cell carcinoma of the head and neck.
Br J Cancer. 105:618–627. 2011.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Harrington K, Berrier A, Robinson M,
Remenar E, Housset M, de Mendoza FH, Fayette J, Mehanna H,
El-Hariry I, Compton N, et al: Randomised Phase II study of oral
lapatinib combined with chemoradiotherapy in patients with advanced
squamous cell carcinoma of the head and neck: Rationale for future
randomised trials in human papilloma virus-negative disease. Eur J
Cancer. 49:1609–1618. 2013.PubMed/NCBI View Article : Google Scholar
|