1. Introduction
The most prevalent cause of tooth loss worldwide and
the sixth most common disease affecting individuals globally is
periodontitis, a chronic inflammatory disease that damages the
supporting tissues of the periodontium (1). The onset of periodontitis can be
influenced by several factors, including genetic predisposition,
the composition of the oral microbiota and lifestyle elements, such
as smoking, nutrition, stress and hormones (2). The dysbiotic environment in the
genetically predisposed patient promotes the growth of pathogenic
bacterial populations, which in turn stimulates the host
immunological response. Reactive oxygen species (ROS), produced by
polymorphonuclear leukocytes (PMNs) during the initial
immunological phase, eliminate pathogens (3). In periodontitis, PMN hyperactivity
creates a milieu of oxidative stress, further increasing ROS
generation (4). Strong evidence
has surfaced in recent years linking oxidative stress to the
pathophysiology of periodontitis (5). The body contains various species,
referred to as antioxidants, to defend against the increase of ROS,
which maintains the level of ROS under control even though they are
created in a healthy system (6).
Disease occurs because of a shift in the ratio of antioxidants to
reactive oxygen species. Secondary factors such as diabetes,
cardiovascular diseases, tobacco use and stress all contribute to
the development of periodontitis by aggravating the oxidative
stress environment in patients with periodontal species (7).
The aim of the present review was to discuss the
association between ROS and periodontal disease, highlighting
current biomarkers and their diagnostic value while identifying
gaps and opportunities for future research in early detection and
treatment.
A structured literature search was performed across
PubMed, Google Scholar, Scopus and Web of Science identified ~68
articles related to ROS and periodontal disease. Following title
and abstract screening, 13 studies were excluded due to irrelevance
to periodontal pathology, duplication, non-clinical focus, or
insufficient methodological quality. Full-text assessment was then
performed, and studies specifically evaluating oxidative stress
biomarkers, antioxidant status and their clinical relevance in
periodontitis were included. The final set of articles was
qualitatively synthesized to address the objectives of the present
narrative review.
2. Reactive oxygen species: Origin, types
and physiological role
ROS are chemically reactive molecules containing
oxygen and include free radicals with unpaired electrons and
non-radical species (8). Certain
ROS function as signaling molecules at physiological levels,
whereas others primarily contribute to oxidative damage when
produced in excess. Both oxygen and nitrogen free radicals are part
of the broader category of reactive species, which includes ROS and
reactive nitrogen species. In healthy individuals, these are
produced in trace amounts during biological processes (9). These reactive species originate
endogenously (within the body) or from exogenous factors. In the
event that a bacterial infection occurs, ROS can also function as
second messengers, signaling macrophages to combat invading
pathogens (10,11).
ROS are highly reactive oxygen-containing molecules,
which include free radicals, such as superoxide anion
(O2-) and hydroxyl radical (-OH), as well as
non-radical derivatives involved in oxidative processes, such as
hydrogen peroxide (H2O2), hypochlorous acid
(HOCl), and singlet oxygen (1O2) (12). ROS are constantly generated by
tissue cells and are essential for maintaining normal cellular
metabolism and physiological processes. However, when ROS
accumulate excessively or persist over time, they can trigger
oxidative stress. This stress exacerbates systemic inflammation and
induces substantial tissue injury via mechanisms, such as lipid
peroxidation, DNA and protein modifications, and the oxidation of
critical enzymes, ultimately leading to cell death. Under
physiological conditions, cellular antioxidants neutralize ROS and
prevent oxidative damage. This antioxidant defense system maintains
tissue integrity by effectively mitigating ROS-induced harm.
Research over the past decade has increasingly linked oxidative
stress to the initiation, development and the advancement of
several chronic inflammatory diseases, including periodontitis
(13).
The destructive processes in the periodontium
observed in inflammatory periodontal diseases are considered to
stem from complex interactions arising from a disruption of the
balance between enzymatic and non-enzymatic breakdown pathways.
ROS, produced predominantly by polymorphonuclear leukocytes during
inflammation, are particularly damaging in this context. Several
lines of evidence support the involvement of ROS in the breakdown
of periodontal tissue. Some examples include the presence of
oxidative byproducts, elevated levels of iron and copper ions,
promoting the formation of highly reactive radicals, and an
imbalance between oxidants and antioxidants in the periodontal
pockets. In vitro studies have reported that ROS can degrade
various components of the extracellular matrix (ECM), such as
proteoglycans, by altering amino acid side chains and fragmenting
core proteins, with only minor depolymerization of
glycosaminoglycan chains (14,15).
Moreover, the detection of connective tissue metabolites in
gingival crevicular fluid (GCF), originating from the breakdown of
periodontal tissues, particularly alveolar bone, supports the key
role of ROS in the tissue damage seen in inflammatory periodontal
diseases (16) (Fig. 1). The commonly investigated
oxidative stress and antioxidant biomarkers in periodontitis, their
biological relevance, sampling matrices and methodological
considerations are summarized in Table
I.
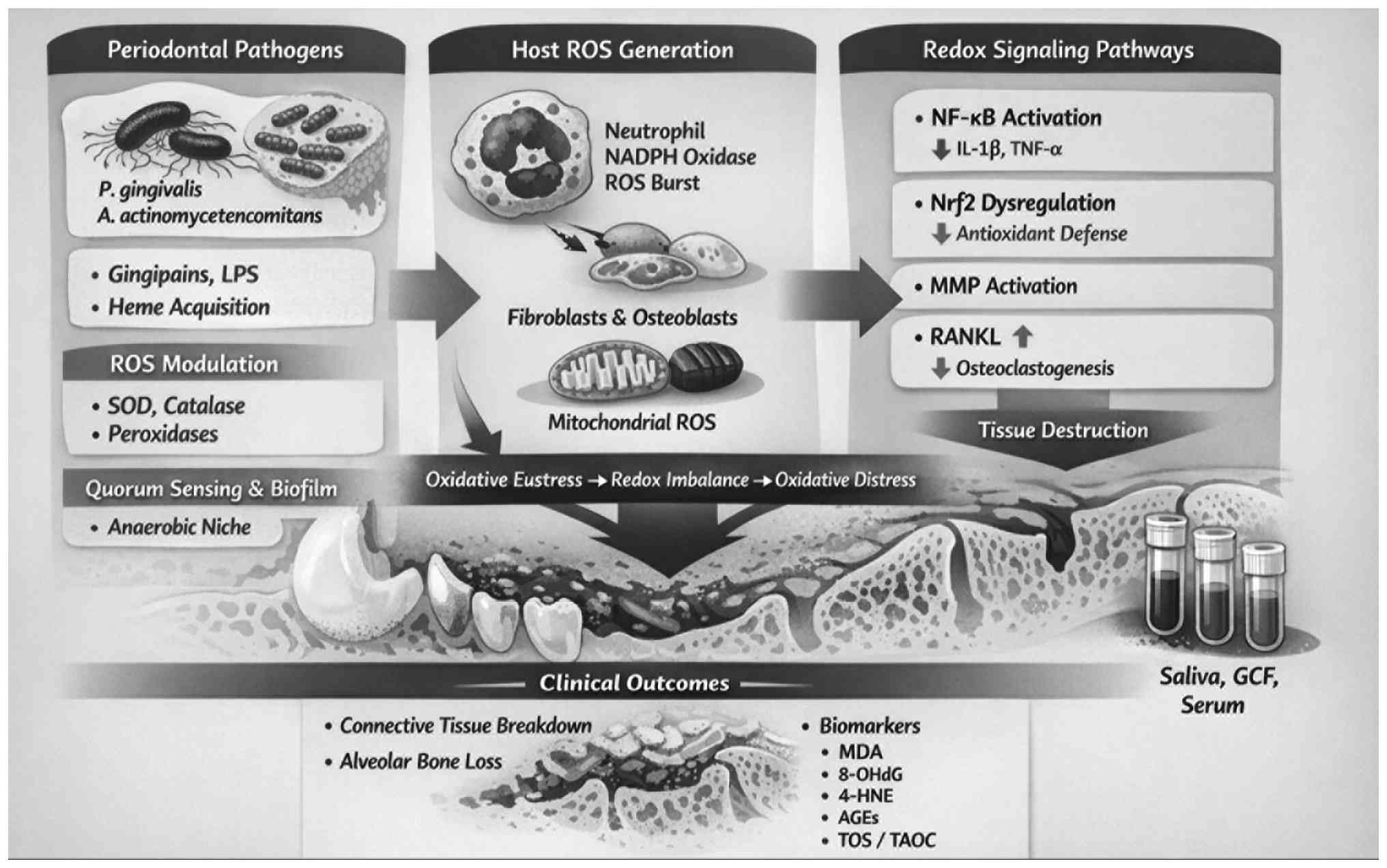 | Figure 1ROS-mediated host–microbial redox
interactions in periodontal disease pathogenesis. ROC, reactive
oxygen species; LPS, lipopolysaccharide; SOD, superoxide dismutase;
MMP, matrix metalloproteinase; MDA, malondialdehyde; 8-OHdG,
8-hydroxydeoxyguanosine; 4-HNE, 4-hydroxy-2-nonenal; AGEs, advanced
glycation end products; TOS, total oxidant status; TAOC, total
antioxidant capacity; GCF, gingival crevicular fluid. |
 | Table IComparative overview of oxidative
stress and antioxidant biomarkers in periodontitis: Biological
relevance, sampling matrices and methodological considerations. |
Table I
Comparative overview of oxidative
stress and antioxidant biomarkers in periodontitis: Biological
relevance, sampling matrices and methodological considerations.
| Biomarker | Biological
class | Sample type(s) | Typical direction
in periodontitis | Common assays | Key
confounders/caveats | Clinical utility
notes | (Refs.) |
|---|
| MDA | Lipid
peroxidation | Saliva, GCF,
serum | ↓ decreases after
therapy |
TBARS/ELISA/LC-MS | Smoking, diabetes,
diet | Good for oxidative
burden; limited specificity | (11) |
| 8-OHdG | DNA oxidation | Saliva, GCF,
serum | ↑; decreases after
therapy | ELISA/LC-MS/MS | Assay
cross-reactivity; renal clearance (serum/urine) | Promising for
activity/response; needs standardization | (17) |
| 4-HNE | Reactive
aldehyde | Saliva, serum | Variable; often ↑
in smokers | ELISA/LC-MS | Short half-life;
protein adducts; smoking | May indicate
high-risk phenotypes | (18) |
| AGEs/RAGE | Glycoxidation
axis | Serum, saliva,
tissue | ↑ (particularly
diabetes/smoking) | ELISA/IHC | Strongly
comorbidity-driven | Links
systemic-local; prognostic in diabetics | (18) |
| TOS | Total oxidant
load | Saliva, GCF | ↑; decreases after
therapy | Colorimetric
(Erel) | Non-specific;
affected by other inflammatory diseases | Useful adjunct
marker, not standalone | (18) |
| TAOC/TAC | Total antioxidant
capacity | Saliva, GCF,
serum | Often ↓ (but
variable) |
Colorimetric/FRAP/ABTS | Uric acid dominates
saliva; diet and stimulation effects | Best interpreted
with TOS and clinical data | (18) |
| SOD/GPx/CAT | Enzymatic
antioxidants | Saliva, GCF,
serum | Variable (↑
compensatory or ↓ depletion) | Enzyme activity
assays | Matrix and timing;
medications; systemic status | More informative in
panels | (18) |
3. Role of oxidative stress in the
pathogenesis of periodontal disease
The presence of oxidative stress indicates an
imbalance between the generation of oxidants, particularly ROS, and
the antioxidant defenses of the body (11). ROS are produced by redox reactions
involving oxygen and can arise from internal cellular sources, such
as NADPH oxidase, cyclooxygenases, peroxisomes, xanthine oxidase
and the microsomal electron transport system. Furthermore, external
factors, including environmental pollution, tobacco smoke,
pesticides, certain drugs, ultraviolet radiation and exposure to
heavy metals, can stimulate ROS generation. The major
oxygen-derived free radicals are O2- and -OH
(17,18).
ROS can function as signaling molecules in a healthy
system, facilitating processes, such as regulating protein function
and activating the immune response; however, excess levels lead to
the development of oxidative stress (17). At low levels, ROS are part of
normal physiological processes, a state known as ‘oxidative
eustress’, in which they regulate protein function and immune
responses. For instance, they can oxidize thiol groups in proteins,
altering their structure and activating their functions. However,
at high concentrations, ROS lead to ‘oxidative distress’, causing
damage to cellular structures, such as DNA, RNA, lipids and
proteins (9,19). To prevent this damage, cells
utilize antioxidants, including enzymes and small molecules, to
neutralize excess ROS and maintain homeostasis.
In periodontal disease, ROS originate from both
host-derived inflammatory responses and pathogen-induced microbial
mechanisms, jointly shaping the periodontal redox microenvironment.
Neutrophils, gingival fibroblasts and osteoblasts generate ROS via
NADPH oxidase and mitochondrial pathways as part of their
antimicrobial defense. By contrast, periodontal pathogens, such as
Porphyromonas gingivalis and Aggregatibacter
actinomycetemcomitans express antioxidant enzymes, including
superoxide dismutase (SOD), catalase (CAT) and peroxidase, thereby
evading oxidative killing within biofilms. Furthermore, microbial
virulence factors, heme acquisition systems and quorum
sensing-regulated biofilm organization modulate host-microbiome
redox interactions and sustain dysbiosis. Excessive ROS activate
redox-sensitive signaling pathways, such as NF-κB, promoting the
release of pro-inflammatory cytokines and the expression of matrix
metalloproteinases (MMPs). The dysregulation of compensatory
antioxidant signaling via the Nrf2/ARE pathway may occur during
chronic inflammation. In addition, mitochondrial ROS signaling
amplifies inflammatory responses and apoptosis. The disruption of
redox feedback loops in gingival fibroblasts, osteoblasts and
neutrophils leads to progressive connective tissue degradation and
alveolar bone loss (12).
4. Effects of oxidative stress in
periodontal disease
Periodontal disease is an inflammatory condition
that destroys tooth-supporting tissues (20). Oxidative stress plays a pivotal
role in its pathogenesis. Elevated ROS levels can damage cellular
structures, including DNA, RNA, lipids and proteins, leading to the
development of diseases, such as cancer, diabetes, rheumatoid
arthritis, neurodegeneration, asthma, reproductive disorders and
periodontitis (13). Specifically,
in periodontal disease, exposure to lipopolysaccharide from
Porphyromonas gingivalis aggravates oxidative stress in
human gingival and periodontal ligament fibroblasts. The
accumulation of oxidative stress markers in the saliva, GCF and
plasma of patients with periodontitis provides additional evidence
linking oxidative stress to periodontal inflammation (21).
5. Reactive oxygen metabolites in
periodontal disease
Alteration of the homeostatic
axis
ROS plays a dual role in periodontal health. Under
normal conditions, they function as cellular messengers, aiding in
the regulation of immune responses and in maintaining homeostasis
in periodontal tissues. However, when the ROS-antioxidant balance
is disrupted, often due to persistent bacterial infections in the
periodontal pockets, this homeostatic axis is altered (22). Excessive ROS production leads to
oxidative stress, promoting inflammation and shifting the
microbiota balance toward a dysbiotic state, exacerbating
periodontal disease (23).
Resorption of the alveolar bone:
Bone-related inflammatory illness
In periodontitis, excessive ROS production disrupts
the RANKL/osteoprotegerin axis, a critical pathway for maintaining
bone homeostasis, and contributes to alveolar bone resorption.
Typically, this axis balances bone resorption and formation;
however, oxidative stress induces the overproduction of
pro-inflammatory cytokines, tipping the balance toward bone
resorption. This mechanism not only leads to the destruction of
alveolar bone in periodontitis, but has also been implicated in
other bone-related inflammatory conditions, such as rheumatoid
arthritis, osteoarthritis and osteoporosis (24).
Loss of the periodontal ligament
(PDL): Clinical attachment loss (CAL)
The PDL is crucial for tooth support, and its loss
is a key indicator of periodontal disease progression. ROS
contribute to this process by activating MMPs, particularly under
conditions of oxidative stress. These MMPs break down essential
components of the PDL, including collagen, elastin, proteoglycans,
and glycosaminoglycans. The breakdown of these ECM components leads
to CAL, destabilizing the tooth-supporting structures (9,25).
6. Antioxidant defense systems in oral and
periodontal health
Saliva contains a comprehensive array of enzymatic
and non-enzymatic antioxidants that collectively protect the oral
cavity from oxidative damage (9).
The enzymatic components include SOD, which converts O2-
into hydrogen peroxide and oxygen; CAT, which decomposes
H2O2 into water and oxygen using
manganese/iron cofactors; glutathione peroxidase (GPx), which
reduces H2O2 to water while oxidizing
glutathione; salivary lactoperoxidase, which generates
antibacterial hypothiocyanite from thiocyanate and
H2O2; myeloperoxidase, which produces
germicidal hypochlorous acid from chloride and
H2O2; and thioredoxin/peroxiredoxin systems,
which facilitate redox cycling to neutralize peroxides (26). These enzymes are complemented by
non-enzymatic antioxidants, which include uric acid (constituting
>70% of the antioxidant capacity of saliva), melatonin (with
antiaging, anti-inflammatory and osteoblast-supporting properties),
glutathione (a critical GPx cofactor and direct ROS scavenger),
vitamins C/A/E, albumin, lactoferrin and bilirubin. Together, these
components form a synergistic defense network that neutralizes ROS,
mitigates oxidative stress and maintains oral tissue homeostasis
(27).
7. Biomarkers of oxidative stress in
periodontitis
Malondialdehyde (MDA)
MDA arises from ROS-triggered lipid peroxidation,
particularly via the attack of free radicals on polyunsaturated
fatty acids (PUFAs). This reaction disrupts cell membrane integrity
and may induce DNA damage (28).
The studies by Trivedi et al (29) and Veljovic et al (30) demonstrated that MDA levels are
associated with the severity of periodontal inflammation, and
decrease following scaling and root planning therapy. Furthermore,
the study Veljovic et al (30) demonstrated that smoking increases
MDA levels in patients with periodontal disease. In addition,
patients with diabetes who have periodontitis exhibit elevated MDA
levels, associating it with systemic oxidative stress (31).
8-Hydroxydeoxyguanosine (8-OHdG)
ROS, such as hydroxyl radicals, oxidize guanine
bases in DNA, forming 8-OHdG, a biomarker of oxidative DNA damage
and mutagenesis. Exogenous 8-OHdG can suppress ROS production and
inflammation by inhibiting Rac1-GTP binding, suggesting a feedback
loop in oxidative stress regulation (32). The studies by Chen et al
(33) and Altıngöz et al
(31) demonstrated that 8-OHdG
levels were significantly elevated in patients with periodontitis,
particularly in those with increased CAL. The level of 8-OHdG
decreases following non-surgical periodontal therapy, as reported
by Önder et al (34). The
strong association between 8-OHdG and CAL, particularly in patients
with diabetes, renders it a promising marker for the diagnosis and
monitoring of periodontal disease.
4-Hydroxy-2-nonenal (4-HNE)
Generated as a result of the ROS-induced lipid
peroxidation of ω-6 PUFAs, 4-HNE is a cytotoxic
α,β-unsaturated aldehyde that promotes apoptosis and protein
damage. In infections, 4-HNE functions as an antimicrobial agent by
damaging bacterial cells, although pathogens, such as
Listeria evolve reductases (e.g., Rha1/Rha2) to neutralize
it (35). While certain
researchers, such as Altıngöz et al (31), found a positive association between
4-HNE levels and periodontal disease indicators [bleeding on
probing (BOP) and CAL], others, such as Onder et al
(34), did not observe significant
changes in 4-HNE levels in patients with periodontopathies. The
concentration of 4-HNE is considerably high in smokers with
periodontitis, suggesting its potential use in identifying patients
at heightened risk owing to smoking-related oxidative stress.
Advanced glycation end products
(AGEs)
AGEs, formed via non-enzymatic glycation, amplify
ROS production via receptor-mediated NADPH oxidase activation
(29,36). The studies by Altıngöz et al
(31) and Katz et al
(37) explored the association
between AGE levels and receptors for advanced glycation end
products (RAGE), revealing that smoking significantly increases
RAGE expression. AGEs and their receptors are linked to increased
cytokine release, bone resorption and collagen degradation,
particularly in patients with diabetes, emphasizing their critical
role in the systemic effects of periodontitis.
8. Antioxidant levels in periodontal
disease
Two key antioxidant enzymes involved in redox
reactions are SOD and GPx. Chen et al (33) examined their levels in the saliva
and GCF of patients with periodontitis and healthy controls. Their
findings indicated no significant differences in enzyme levels
between the two groups. However, other studies have documented
inconsistent findings, with some observing variations in the levels
of these enzymes among individuals with periodontal disease. For
example, Trivedi et al (29) reported decreased activities of SOD
and GPx in patients with periodontitis, suggesting that this
reduction may result from antioxidant depletion during ROS
neutralization. By contrast, Villa-Corea et al (38) observed elevated GPx activity in
patients with both aggressive and chronic periodontitis compared
with healthy controls. Similarly, Yang et al (39) noted an increased SOD activity in
patients with periodontitis, with SOD levels positively being
associated with clinical characteristics, such as CAL, BOP, probing
depth, gingival index and plaque index.
Glutathione, another crucial antioxidant, functions
as a scavenger for ROS. Kluknavska et al (40) observed increased glutathione levels
in patients with aggressive and chronic periodontitis, consistent
with earlier studies (38,41). In addition, Kluknavska et al
(40) noted that glutathione
levels were lower in patients with gingivitis than in those with
periodontitis, but higher than in the healthy control group,
implying that glutathione levels surge with increasing inflammation
severity. Conversely, Oktay et al (42) reported lower glutathione levels in
patients with periodontitis than in healthy individuals.
Melatonin, another antioxidant present in saliva,
was studied by Balaji and Rao (43). They found lower melatonin levels in
the GCF of smokers with periodontopathies and non-smokers with
periodontitis compared with healthy controls. The lowest levels
were detected in smokers, likely due to tobacco-induced increase in
ROS levels. Purrahmani et al (44) demonstrated that salivary melatonin
levels, initially low in patients with periodontitis, increased
following non-surgical treatment, indicating that melatonin levels
may be a reliable biomarker for assessing response to periodontal
therapy.
9. Total oxidant status and total
antioxidant capacity
The total oxidant status (TOS) denotes the
cumulative concentration of oxidants, including ROS and other free
radicals, in biological samples, such as saliva or GCF. Elevated
TOS levels in patients with periodontitis reflect increased
oxidative stress resulting from heightened inflammatory activity
and ROS production (45). The
automated colorimetric assay for TOS, developed by Erel (46) in 2005, has since become a standard
method in periodontitis research. Several investigations have
reported high TOS values in individuals with periodontitis. For
example, Zalewska et al (47) established that stimulated saliva
exhibits higher TOS levels than non-stimulated saliva or GCF, which
they attributed to the parotid glands being a significant source of
oral ROS. In another study, Toczewska et al (48) observed a decrease in TOS levels
following non-surgical periodontal treatment, suggesting that it
may be a valuable indicator of oxidative stress changes associated
with periodontal therapy.
Total antioxidant capacity (TAOC/TAC), also referred
to as the total antioxidant status, measures the overall ability of
antioxidants in a sample to neutralize oxidants, providing a
comprehensive evaluation of the antioxidant defense system. In
periodontitis, TAOC levels are decreased, indicating that
antioxidant defenses are inadequate to counteract elevated ROS and
oxidative stress (45). Numerous
studies, including those by Miricescu et al (49) and Nguyen et al (50), have reported significantly lower
TAOC levels in patients with periodontitis compared with healthy
controls, which may be attributed to increased antioxidant
consumption in response to heightened ROS.
Although TOS and TAOC have been proposed as
potential biomarkers of oxidative stress in periodontitis, their
diagnostic utility remains controversial. For instance, Senouci
et al (51) reported an
inverse association between TAC levels and the severity of
periodontitis, whereas Behfarnia et al (52) observed increased TAOC following
nonsurgical periodontal therapy. Toczewska et al (48) cautioned that various inflammatory
conditions can influence TOS and TAOC levels and may not reliably
distinguish between different stages of periodontitis.
Consequently, while TOS and TAOC provide valuable insight into the
oxidative status of the oral environment, they are not yet
established as definitive biomarkers for diagnosing or staging
periodontitis.
10. Challenges and future perspective of ROS
biomarkers in periodontal disease
Despite considerable progress in elucidating the
role of ROS in the pathogenesis of periodontal disease, several
challenges remain in the clinical application of ROS biomarkers. A
key limitation is the variability in the sensitivity of current
measurement techniques for oxidative stress markers, such as TOS,
TAC, MDA, and 8-OHdG in biological fluids. The lack of
standardized, universally accepted protocols hampers
reproducibility and comparability across studies. Furthermore, the
levels of these biomarkers may be affected by various confounding
factors, such as age, sex, smoking, systemic health conditions,
medication use and circadian rhythms, complicating their
interpretation and limiting their disease specificity. Owing to the
dynamic nature of ROS production, which fluctuates in response to
inflammation, treatment and daily oral hygiene, single-time-point
measurements may not accurately reflect chronic oxidative status or
the progression of periodontal disease. In addition, the complex
interplay among ROS, inflammatory mediators, host immune responses
and microbial factors in periodontal disease poses difficulties in
delineating the precise mechanistic pathways via which ROS
contribute to tissue destruction. Although antioxidant-based
adjunctive treatments have demonstrated potential to lower
oxidative stress markers and enhance periodontal health, their
clinical efficacy is limited by the lack of extensive, long-term
randomized controlled trials.
Future research is required to focus on developing
multiplex biomarker panels and integrating omics technologies, such
as proteomics and metabolomics, to identify more specific and
sensitive markers of oxidative stress. Moreover, ROS may be tested
in combination with other adjunctive therapies, such as ozone,
probiotics and photobiomodulation, to assess their potential
effects on oxidative stress and periodontal tissues (53,54).
Rapid, point-of-care diagnostic assays should be
developed, and longitudinal and interventional studies should be
conducted. These measures will aid in validating the clinical
relevance of ROS biomarkers and translating these findings into
effective diagnostic and therapeutic strategies for periodontal
disease.
The present review has certain limitations, which
should be mentioned. At the study level, heterogeneity in study
design, sample size and biomarker assessment methods, as well as
inadequate control of confounding factors, may contribute to
variability and potential bias. At the review level, as this is a
narrative review, the incomplete retrieval of relevant literature
and reporting bias cannot be entirely excluded. Furthermore, the
lack of a quantitative synthesis limits the ability to draw
definitive conclusions about the diagnostic utility of ROS
biomarkers.
11. Conclusion
The evidence provided herein highlights the pivotal
role of ROS in the pathogenesis and progression of periodontal
disease, as demonstrated by increased levels of oxidative stress
markers such as TOS, MDA and 8-OHdG, alongside decreased TAOC.
While these biomarkers hold promise for disease monitoring and
therapeutic assessment, their clinical utility is limited by
methodological variability, the lack of standardization and
confounding factors, such as systemic health and lifestyle habits.
The multifactorial nature of periodontal disease, involving complex
interactions among ROS, inflammation, immune responses and
microbial factors, further complicates the interpretation and
specificity of these markers. Although antioxidant-based therapies
have shown potential benefits, robust evidence from large-scale
clinical trials is still needed. Future directions should focus on
standardizing biomarker assessment, developing multiplex panels,
integrating omics technologies, and advancing point-of-care
diagnostics to validate and enhance the clinical relevance of ROS
biomarkers in the management of periodontal disease.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
AS and NS were involved in designing the concept of
the present review followed by conducting the literature search and
drafting the initial manuscript. AR, BAK and KSC were involved in
revising and editing the manuscript. All the authors have read,
reviewed and approved the final manuscript. Data authentication is
not applicable.
Ethics approval and consent for
publication
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors have declared they have no competing
interests.
References
|
1
|
GBD 2017 Oral Disorders Collaborators.
Bernabe E, Marcenes W, Hernandez C, Bailey J, Abreu L, Alipour V,
Amini S, Arabloo J, Arefi Z, et al: Global, regional, and national
levels and trends in the burden of oral conditions from 1990 to
2017: A systematic analysis for the global burden of disease 2017
study. J Dent Res. 99:362–373. 2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Papapanou PN, Sanz M, Buduneli N, Dietrich
T, Feres M, Fine DH, Flemmig TF, Garcia R, Giannobile WV, Graziani
F, et al: Periodontitis: Consensus report of workgroup 2 of the
2017 world workshop on the classification of periodontal and
Peri-implant diseases and conditions. J Periodontol. 89 (Suppl
1):S173–S182. 2018.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Hajishengallis G and Lamont RJ: Beyond the
red complex and into more complexity: The polymicrobial synergy and
dysbiosis (PSD) model of periodontal disease etiology. Mol Oral
Microbiol. 27:409–419. 2012.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Martu MA, Surlin P, Lazar L, Maftei GA,
Luchian I, Gheorghe DN, Rezus E, Toma V and Foia LG: Evaluation of
oxidative stress before and after using laser and photoactivation
therapy as adjuvant of non-surgical periodontal treatment in
patients with rheumatoid arthritis. Antioxidants (Basel).
10(226)2021.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Dahiya P, Kamal R, Gupta R, Bhardwaj R,
Chaudhary K and Kaur S: Reactive oxygen species in periodontitis. J
Indian Soc Periodontol. 17:411–416. 2013.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Sharifi-Rad M, Anil Kumar NV, Zucca P,
Varoni EM, Dini L, Panzarini E, Rajkovic J, Tsouh Fokou PV, Azzini
E, Peluso I, et al: Lifestyle, oxidative stress, and antioxidants:
Back and forth in the pathophysiology of chronic diseases. Front
Physiol. 11(694)2020.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Beukers NG, van der Heijden GJ, van Wijk
AJ and Loos BG: Periodontitis is an independent risk indicator for
atherosclerotic cardiovascular diseases among 60,174 participants
in a large dental school in the Netherlands. J Epidemiol Community
Health. 71:37–42. 2017.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Li R, Jia Z and Trush MA: Defining
reactive oxygen species in biology and medicine. React Oxyg Species
(Apex). 1:9–21. 2016.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Jakubczyk K, Dec K, Kałduńska J, Kawczuga
D, Kochman J and Janda K: Reactive oxygen species-sources,
functions, oxidative damage. Pol Merkur Lekarski. 48:124–127.
2020.PubMed/NCBI
|
|
10
|
Viglianisi G, Tartaglia GM, Santonocito S,
Amato M, Polizzi A, Mascitti M and Isola G: The emerging role of
salivary oxidative stress biomarkers as prognostic markers of
periodontitis: New insights for a personalized approach in
dentistry. J Pers Med. 13(166)2023.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Tretter V, Hochreiter B, Zach ML, Krenn K
and Klein KU: Understanding cellular redox homeostasis: A challenge
for precision medicine. Int J Mol Sci. 23(106)2021.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Chapple IL and Matthews JB: The role of
reactive oxygen and antioxidant species in periodontal tissue
destruction. Periodontol 2000. 43:160–232. 2007.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Buranasin P, Kominato H, Mizutani K,
Mikami R, Saito N, Takeda K and Iwata T: Influence of reactive
oxygen species on wound healing and tissue regeneration in
periodontal and peri-implant tissues in diabetic patients.
Antioxidants. 12(1787)2023.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Panasyuk A, Frati E, Ribault D and
Mitrovic D: Effect of reactive oxygen species on the biosynthesis
and structure of newly synthesized proteoglycans. Free Radic Biol
Med. 16:157–167. 1994.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Moseley R and Waddington RJ: Modification
of gingival proteoglycans by reactive oxygen species: Potential
mechanism of proteoglycan degradation during periodontal diseases.
Free Radic Res. 55:970–981. 2021.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Waddington RJ, Moseley R and Embery G:
Reactive oxygen species: A potential role in the pathogenesis of
periodontal diseases. Oral Dis. 6:138–151. 2000.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Cizmarova B, Tomeckova V, Hubkova B,
Hurajtova A, Ohlasova J and Birkova A: Salivary redox homeostasis
in human health and disease. Int J Mol Sci.
23(10076)2022.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Reczek CR and Chandel NS: ROS-dependent
signal transduction. Curr Opin Cell Biol. 33:8–13. 2015.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Yan LL and Zaher HS: How do cells cope
with RNA damage and its consequences? J Biol Chem. 294:15158–15171.
2019.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Könönen E, Gursoy M and Gursoy UK: .
Periodontitis: A multifaceted disease of tooth-supporting tissues.
J Clin Med. 8(1135)2019.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Liu J, Wang X, Zheng M and Luan Q:
Oxidative stress in human gingival fibroblasts from periodontitis
versus healthy counterparts. Oral Dis. 29:1214–1225.
2023.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Magacz M, Kedziora K, Sapa J and Kryściak
W: The significance of lactoperoxidase system in oral health:
Application and efficacy in oral hygiene products. Int J Mol Sci.
20(1443)2019.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Minic I: Antioxidant role of saliva. J
Otolaryngol Res. 2(124)2019.
|
|
24
|
Shinohara I, Morita M, Chow SKH, Murayama
M, Sususki Y, Gao Q and Goodman SB: Pathophysiology of the effects
of oxidative stress on the skeletal system. J Orthop Res.
43:1059–1072. 2025.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Sam CH and Lu HK: The role of hypochlorous
acid as one of the reactive oxygen species in periodontal disease.
J Dent Sci. 4:45–54. 2009.
|
|
26
|
Cochran DL: Inflammation and bone loss in
periodontal disease. J Periodontol. 79:1569–1576. 2008.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Nelson A, Santosh ABR and Jones T: Review
of salivary antioxidants and their barriers. Dent J (Majalah
Kedokteran Gigi). 57:74–79. 2024.
|
|
28
|
Dharmajaya R and Sari DK: Malondialdehyde
value as radical oxidative marker and endogenous antioxidant value
analysis in brain tumor. Ann Med Surg (Lond).
77(103231)2022.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Trivedi S, Lal N, Mahdi AA, Mittal M,
Singh B and Pandey S: Evaluation of antioxidant enzymes activity
and malondialdehyde levels in patients with chronic periodontitis
and diabetes mellitus. J Periodontol. 85:713–720. 2014.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Veljovic T, Djuric M, Mirnic J, Gusic I,
Maletin A, Ramic B, Neskovic I, Vukoje K and Brkic S: Lipid
peroxidation levels in saliva and plasma of patients suffering from
periodontitis. J Clin Med. 11(3617)2022.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Altıngöz SM, Kurgan Ş, Önder C, Serdar MA,
Ünlütürk U, Uyanık M, Başkal N, Tatakis DN and Günhan M: Salivary
and serum oxidative stress biomarkers and advanced glycation end
products in periodontitis patients with or without diabetes: A
cross-sectional study. J Periodontol. 92:1274–1285. 2021.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Kulasekaran C, Devi M, Dhivya K,
Vijayalakshmi D, Sathishkumar M and Madhanmohan A:
Immunohistochemical detection of 8-hydroxydeoxyguanosine: A
biomarker of oxidative DNA damage in oral submucous fibrosis. J
Oral Maxillofac Pathol. 24:536–541. 2020.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Chen Y, Ji Y, Jin X, Sun X, Zhang X, Chen
Y, Shi L, Cheng H, Mao Y, Li X, et al: Mitochondrial abnormalities
are involved in periodontal ligament fibroblast apoptosis induced
by oxidative stress. Biochem Biophys Res Commun. 509:483–490.
2019.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Önder C, Kurgan S, Altingoz SM, Bagis N,
Uyanik M, Serdar MA, Kantarci A and Gunhan M: Impact of
non-surgical periodontal therapy on saliva and serum levels of
markers of oxidative stress. Clin Oral Investig. 21:1961–1969.
2017.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Tabakh H, McFarland AP, Thomason MK,
Pollock AJ, Glover RC, Zaver SA and Woodward JJ:
4-Hydroxy-2-nonenal antimicrobial toxicity is neutralized by an
intracellular pathogen. Elife. 10(e59295)2021.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Zhang Y, Zhang Z, Tu C, Chen X and He R:
Advanced glycation end products in disease development and
potential interventions. Antioxidants. 14(492)2025.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Katz J, Caudle RM, Bhattacharyya I,
Stewart CM and Cohen DM: Receptor for advanced glycation end
product (RAGE) upregulation in human gingival fibroblasts incubated
with nornicotine. J Periodontol. 76:1171–1174. 2005.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Villa-Correa YA, Isaza-Guzmán DM and
Tobón-Arroyave SI: Influence of periodontal clinical status on
salivary levels of glutathione reductase. J Periodontol.
87:716–724. 2016.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Yang PS, Huang WC, Chen SY, Chen CH, Lee
CY, Lin CT and Huang YK: Scaling-stimulated salivary antioxidant
changes and oral-health behavior in an evaluation of periodontal
treatment outcomes. Sci World J. 2014(814671)2014.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Kluknavska J, Krajcikova K, Bolerazska B,
Maslankova J, Ohlasova J, Timkova S, Drotarova Z and Vaskova J:
Possible prognostic biomarkers of periodontitis in saliva. Eur Rev
Med Pharmacol Sci. 25:3154–3161. 2021.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Grant MM, Brock GR, Matthews JB and
Chapple IL: Crevicular fluid glutathione levels in periodontitis
and the effect of non-surgical therapy. J Clin Periodontol.
37:17–23. 2010.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Oktay S, Ozoner O, Alturfan EE and Noyan
U: Determination of oxidative stress parameters and tissue factor
activity in the saliva of patients with periodontitis. Eur J Biol.
78:63–68. 2019.
|
|
43
|
Balaji TM and Rao SR: Salivary, plasma,
and gingival levels of melatonin and TNF-α in nonsmokers and
current smokers with and without periodontal disease. J Contemp
Dent Pract. 21:897–904. 2020.PubMed/NCBI
|
|
44
|
Purrahmani A, Soghli N, Qujeq D, Kiakojori
A and Gholinia H: Changes in salivary melatonin levels by scaling
and root planing in patients with chronic periodontitis. J
Kermanshah Univ Med Sci. 25(e113275)2021.
|
|
45
|
Mohideen K, Chandrasekaran K,
Veeraraghavan H, Faizee SH, Dhungel S and Ghosh S: Meta-analysis of
assessment of total oxidative stress and total antioxidant capacity
in patients with periodontitis. Dis Markers.
2023(9949047)2023.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Erel O: A new automated colorimetric
method for measuring total oxidant status. Clin Biochem.
38:1103–1111. 2005.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Zalewska A, Maciejczyk M, Szulimowska J,
Imierska M and Blachnio-Zabielska A: High-fat diet affects ceramide
content, disturbs mitochondrial redox balance, and induces
apoptosis in the submandibular glands of mice. Biomolecules.
9(877)2019.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Toczewska J, Maciejczyk M, Konopka T and
Zalewska A: Total oxidant and antioxidant capacity of gingival
crevicular fluid and saliva in patients with periodontitis: Review
and clinical study. Antioxidants (Basel). 9(450)2020.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Miricescu D, Totan A, Calenic B, Mocanu B,
Didilescu A, Mohora M, Spinu T and Greabu M: Salivary biomarkers:
Relationship between oxidative stress and alveolar bone loss in
chronic periodontitis. Acta Odontol Scand. 72:42–47.
2014.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Nguyen TT, Ngo LQ, Promsudthi A and
Surarit R: Salivary lipid peroxidation in patients with generalized
chronic periodontitis and acute coronary syndrome. J Periodontol.
87:134–141. 2016.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Senouci S, Ait Yahia D, Bouziane D, Mehadj
M and Malaisse WJ: The diagnostic role of salivary biomarkers of
oxidative stress and inflammatory status and their relationship in
periodontitis stage III and grade C. Biologia. 76:1617–1625.
2021.
|
|
52
|
Behfarnia P, Dadmehr M, Hosseini SN and
Mirghaderi SA: The effect of vitamin E supplementation on treatment
of chronic periodontitis. Dent Res J (Isfahan).
18(62)2021.PubMed/NCBI
|
|
53
|
Colombo M, Gallo S, Garofoli A, Poggio C,
Arciola CR and Scribante A: Ozone gel in chronic periodontal
disease: A randomized clinical trial on the anti-inflammatory
effects of ozone application. Biology. 10(625)2021.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Meng N, Liu Q, Dong Q, Gu J and Yang Y:
Effects of probiotics on preventing caries in preschool children: A
systematic review and meta-analysis. J Clin Pediatr Dent.
47:85–100. 2023.PubMed/NCBI View Article : Google Scholar
|















