1. Introduction
Microplastics are types of plastic pollution that
have piqued increased academic curiosity due to their widespread
presence across different environmental landscapes and their
possible adverse effects on human health. Microplastic particles
have a diameter of <5 mm (1).
These entities can be intentionally manufactured for specific
applications in the cosmetics and textile industries, or they may
be generated inadvertently through the degradation of larger
plastic items (2). The detection
of microplastics has been noted across a range of environmental
locations, comprising water systems, soil landscapes and air
conditioners. It has been reported that they accumulate in
ecological frameworks, seeping into the food web and creating
possible hazards to human health from consuming unsafe food and
water sources (1,3). These tiny plastic fragments can carry
hazardous substances, such as bisphenol A and phthalates, which can
affect the endocrine system. Microplastic identification can be
carried out through a physical approach based on characteristics,
size and color. Furthermore, identification can be combined with
chemical methods to distinguish them from natural materials.
Spectroscopy is one method often used for comprehensive analysis
(4).
Apart from microplastics, there are other small
plastic particles known as nanoplastics. Nanoplastics are particles
measuring between 1 nm and 1 µm (2). Nanoplastics have smaller dimensions,
higher reactivity and mobility compared to microplastics.
Nanoplastics more easily pass through cell membranes than
microplastics, which can affect cellular processes (5). Nanoplastics exhibit unique
interactions with light and natural colloidal substances, thus
affecting their environmental durability and bioavailability
(6). Some potential effects of
nanoplastics on humans are the possibility of triggering
inflammation, oxidative stress and various other health issues
(3). In detecting and identifying
nanoplastics, sophisticated methodologies are still required due to
their very large size (6). The
Microplastics and Nanoplastics dataset is one of the innovations
that has been created for the automatic identification and
classification of microplastics and nanoplastics through the
application of deep learning algorithms (7). Although both forms constitute plastic
pollution, the present review primarily focuses on microplastics.
Although nanoplastics may exhibit similar mechanisms, current
detection evidence is predominantly for microplastics in human
tissues.
Several studies have detected the presence of
microplastics in the human body, including the respiratory system.
Small particles are easily absorbed through inhalation. The
discovery of microplastics in lung tissue has also revealed that
microfibers are more common in neoplastic lesions than in
non-neoplastic tissue (8). This
observation suggests a link between microplastic inhalation and the
development of ground glass nodules (9). The detection of microplastics in
ambient air and their possible role in triggering inflammatory
responses and tissue injury in the lungs has been documented,
although research into the mechanisms of their impact on health
remains limited (10).
Previously considered to be free of microbial
inhabitants, microbial communities have been found in respiratory
organs. The lung microbiota, as a complex and adaptive ecosystem,
plays a role in maintaining respiratory health. The respiratory
microbiota originates from the upper respiratory tract. The lung
microbiome consists of bacteria, fungi and viruses, with
Firmicutes, Bacteroidetes and Proteobacteria
being particularly abundant in the lungs (11). The microbial population in the
lungs is lower than that found in other anatomical locations, such
as the gastrointestinal tract, due to the unique lung environment,
which includes a lipid-rich surfactant that inhibits microbial
proliferation. The lung microbiome is generally transient and is
influenced by microaspiration from the oropharynx, which then
undergoes a clearance process facilitated by mechanisms such as
coughing and ciliary movement (12).
The lung microbiome plays a role in the adjustment
of innate and adaptive immune mechanisms. The microbiome functions
to enhance immune tolerance and reduce excessive inflammatory
responses (13). The microbiome
contributes to maintaining lung homeostasis by preventing
colonization by pathogenic microbes and supporting tissue repair
processes (14). Metabolites
produced by microbes have the capacity to influence human host
metabolism and immune responses, but further research is needed
(15). The lung microbiome also
plays a role related to the gut and oropharyngeal microbiomes. One
such function is the influence of changes in the composition of the
lung microbiome on the immune response and the potential for health
problems such as asthma and chronic obstructive pulmonary disease
(COPD) (11).
The growing concern about the disruption of the lung
microbiome by microplastics and their contribution to respiratory
health risks is attracting increasing academic attention due to
their potential consequences for public health. Evidence suggests
that microplastics may contribute to the increased incidence of
respiratory conditions, including asthma, by affecting the lung
microbiome and triggering inflammation (16). Exposure to fibrous microplastics
has been shown to significantly alter the bacterial composition of
the lungs (16). Research on
ovalbumin-induced asthma in mice has demonstrated that exposure to
microplastics causes significant changes in the relative prevalence
of various bacterial genera, characterized by a decrease in the
amount of beneficial bacteria, such as
Escherichia-Shigella and unidentified strains,
alongside an increase in potentially harmful bacteria, including
Prevotella (16). It has
been shown that changes in the lung microbiome coincide with
increased airway inflammation, mucus buildup and fibrosis in
asthmatic mice, suggesting that microplastics may compromise
respiratory health by affecting the lung microbiome (16).
While the detrimental implications of microplastics
on the lung microbiome and respiratory health are increasingly
being elucidated, it remains critical to consider the broader scope
of environmental pollution and its complex impacts on health. Data
suggest that air pollution, particularly fine particulate matter,
can alter the lung microbiota and increase the rates of respiratory
infections, highlighting the need for comprehensive strategies to
mitigate environmental health hazards (17). The present review aimed to
integrate research on the effects of inhaled microplastics on the
lung microbiome, resulting in changes in microbial composition and
function, and to assess the potential implications of these
modifications for inflammation, immune dysfunction and respiratory
disease. Furthermore, the present review aimed to describe the
broader health consequences of microplastic exposure and outline
key priorities for future research.
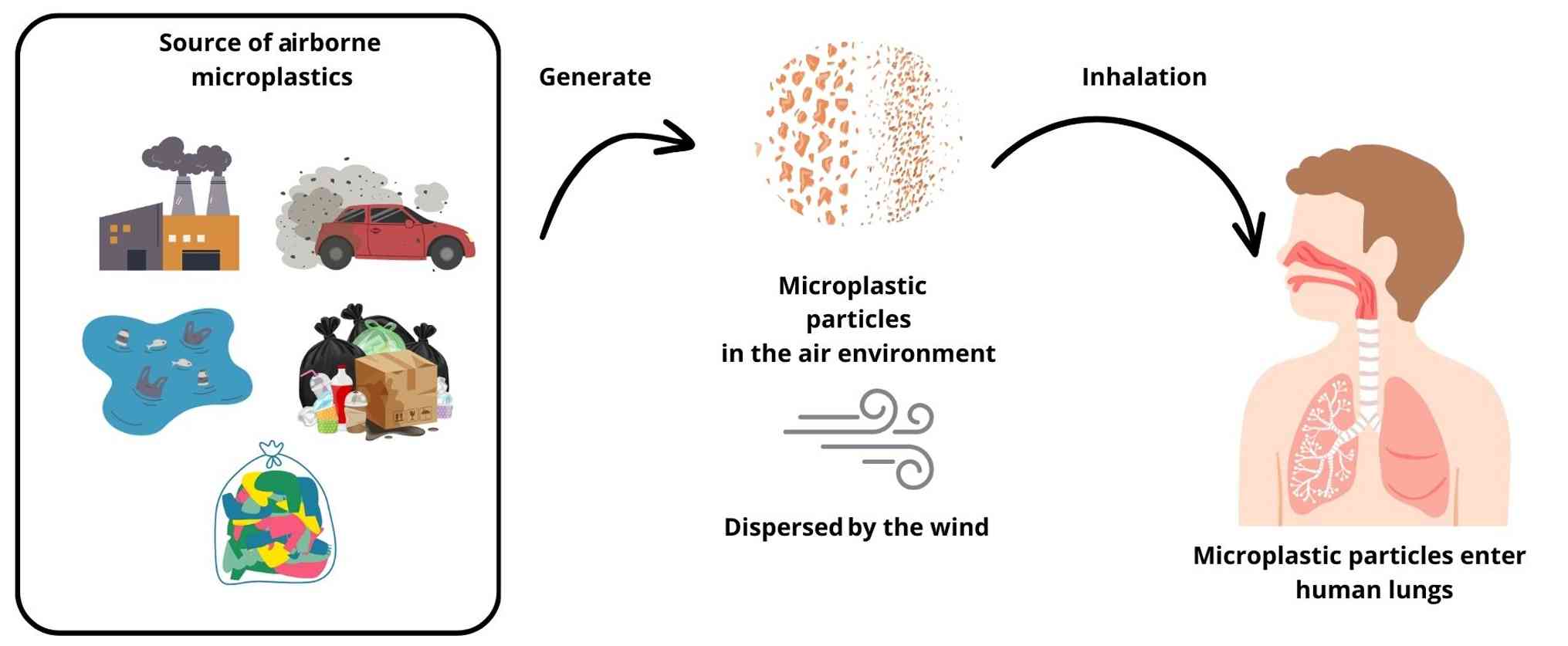
2. Sources and characteristics of airborne
microplastics
Microplastics have been detected in both indoor and
outdoor environments, originating from a variety of sources and
exhibiting varying characteristics. The main contributors to indoor
airborne microplastics are synthetic fabric residues, degradation
of plastic items and the use of plastic polyphenylene ether.
Furthermore, activities such as drying clothes and the presence of
household furniture substantially increase indoor microplastic
concentrations (18). Outdoor
sources of microplastics are diverse, including urban material
particles, emissions associated with vehicle traffic, and
industrial operations. Furthermore, wastewater treatment facilities
and the use of biosolids in agricultural methods further accelerate
the release of microplastics into the atmosphere (18). In coastal areas, maritime transport
can increase airborne microplastic concentrations, although
land-based sources of pollutants also have a more pronounced impact
(19). Airborne microplastics
possess the capacity for long-distance transport, thereby reaching
isolated locales and exacerbating global pollution levels.
Airborne microplastics are primarily characterized
by their fibrous morphology, with fibers constituting the
predominant manifestation observed in both indoor and outdoor
settings (20). The dimensions of
these particulates are heterogeneous; however, they are frequently
aggregated within diminutive size categories, typically measuring
<1 mm (19). Common polymers
found in airborne microplastics include polyethylene terephthalate,
polyethylene, polypropylene and polystyrene (20). The chemical composition is subject
to variations contingent upon the source, with disparate
environments displaying unique polymeric profiles (19). Airborne microplastics are
manifested in a spectrum of colors, with transparent and white hues
being particularly prevalent across numerous studies (18). Morphological forms include
spherules, films, fragments and granules, with fibers representing
the most dominant morphological category (21).
Airborne microplastics are increasingly acknowledged
as a critical environmental and public health issue, with numerous
sources and pathways contributing to their infiltration into the
human pulmonary system (Fig. 1).
Urbanization and industrial activities serve as primary
determinants of airborne microplastics. Particulates associated
with vehicular traffic, synthetic textiles and emissions from
industrial operations are significant contributors within urban
locales (21). Indoor air is
frequently characterized by elevated concentrations of
microplastics relative to outdoor air. Textiles, including garments
and domestic fabrics, represent principal sources of indoor
microplastics, with fibers constituting the predominant form
(22). Roadside dust and household
refuse further augment the prevalence of microplastics within the
atmosphere. These particulates can be reintroduced into the air via
aeolian processes (23).
Agricultural methodologies, such as the application of plastic
mulch, in conjunction with maritime activities, also facilitate the
release of microplastics into the atmosphere, thereby exacerbating
outdoor pollution levels (24).
The leading method through which microplastics reach human lungs is
by means of inhalation. Tiny plastic particles may be lifted into
the air and are subsequently inhaled, reaching the lungs where they
could possibly move into the alveolar space and enter the
bloodstream (22). The
distribution and transport dynamics of microplastics within the
atmosphere are influenced by environmental parameters, including
wind velocity, directional flow and precipitation, which can
promote their long-range transport and eventual deposition
(24).
Microplastics have been identified within human
pulmonary tissues, with one investigation reporting the presence of
108 microplastic particles in samples derived from non-smoking
individuals. The major types observed were polypropylene,
polyethylene terephthalate, and polystyrene, presenting a median
level of 2.19 particles/g (25).
In a previous study, the presence of microplastics was confirmed in
bronchoalveolar lavage fluid (BAL). There were 13 unique forms of
microplastics in samples from those who had not smoked. The
predominant substance identified was polyethylene, succeeded by
polyethylene terephthalate and polypropylene (26). This body of evidence implies that
microplastics can penetrate deeply into the respiratory apparatus,
potentially resulting in detrimental health consequences. A
comprehensive overview of the evidence pertaining to human exposure
to microplastics via inhalation and their identification in
respiratory tissues is presented in Table I (25-33).
 | Table IReports of detection microplastics in
human lung tissues. |
Table I
Reports of detection microplastics in
human lung tissues.
| Microplastics and
nanoplastic materials | Reported
samples | (Refs.) |
|---|
| Plant and plastic
fibers | Detected in 13
histopathology slides of lung tissue from 17 slides that were
examined | (27) |
| Microplastics sized
between 20-100 µm consisted of PP (34.26%), PET (21.30%), and PS
(8.33%) | Lung tissue samples
from 12 non-smoking patients | (25) |
| 12 polymer types
such as PP (23%), PET (18%) and resin (15%) | Detected in 11 of
the 13 lung tissue samples with an average of 1.42±1.50
microplastics/g of tissue | (28) |
| PP (35.1%), PET
(24.3%); cotton (16.2%); polyvinyl chloride and cellulose acetate
(5.4%); and polyamide, polyethylene co-polypropylene, polystyrene,
polystyrene- co-polyvinyl chloride and polyurethane (2.7%) | Observed in 13 of
the 20 autopsied decedents | (29) |
| Various
microplastics/nanoplastics in the interval of 0.14-12.8 particles
per 100 ml of BALF | BALF samples of 10
patients undergoing diagnostic bronchoscopy | (30) |
| PE (86.1%), PET
(7.5%) and PP (1.9%) | A total of 18
never-smokers aged 32-74 years who underwent fiberoptic
bronchoscopy with BALF | (26) |
| Microplastics
(25.86 particles/g), PE (11.34 particles/g), and silicone (1.15
particles/g) | Smokers | (31) |
| Microfibers,
polyester fiber, acrylic fiber, rayon fiber, wool fiber, cotton
fiber and cellulose fiber. | BALF of 44 adult
European citizens. | (32) |
| Microplastics
composed of PP (41.9%), PE (19.4%) and PS (13.6%) | BALF of children
with pulmonary diseases | (33) |
There are several methods that can be used to
identify microplastics in human respiratory tissues. FTIR/Raman
spectroscopy, the gold standard for microplastic identification,
has limited efficacy due to diffraction-limited resolution (~1-5
µm), systematically underdetecting nanoplastics (<1 µm) due to
poor signal-to-noise ratios. Complementary SEM/TEM provides
nanoscale visualization, but requires EDX coupling for chemical
confirmation, as tissue debris frequently generates false positives
(34).
BAL sampling introduces critical contamination risks
from airborne microplastics shed by laboratory plastics, synthetic
clothing and polyethylene bronchoscopes. Jenner et al
(28) detected 1.42±1.50
microplastics/g in 11/13 uncontaminated lung tissues, underscoring
procedural blanks as mandatory controls. Best practices mitigating
>90% contamination include: ISO Class 5+ cleanroom processing;
silicone (not polyethylene) bronchoscopy tubing; ≥3 procedural
blanks per batch; 100% cotton lab attire excluding synthetic
fabrics; and HEPA H14-filtered air during dissection. These
protocols described by Jenner et al (28) enable the reliable quantification of
lung burdens reported separately in the studies presented in
Table I (25-33),
such as 2.19 particles/g tissue and 0.14-12.8 particles/100 ml
BAL.
While the identification of microplastics within
human respiratory tissues and environmental air samples provides
substantial evidence of inhalation exposure, the comprehensive
ramifications of such exposure on health remain ambiguous.
Contemporary investigations underscore the necessity for the
establishment of standardized protocols to detect smaller-sized
microplastics and the implementation of longitudinal studies to
evaluate their prospective health hazards. Comprehending the routes
through which microplastics impact human well-being, particularly
regarding the respiratory system, is essential for formulating
successful approaches to lessen exposure and prevent possible
health effects.
3. The respiratory microbiome: Composition,
functions and vulnerabilities
The respiratory microbiome functions as a vital and
multifaceted ecosystem that is key to upholding respiratory health
and supporting disease development. A recent bibliometric analysis
confirmed microplastics as an emerging respiratory research
priority (35). The lung
microbiome predominantly originates from the upper respiratory
tract yet possesses a distinct microbial flora. It is defined by a
transient and mobile characteristic, attributable to mechanisms,
such as coughing, pulmonary macrophages and alveolar surfactant
(11). The collection of
microorganisms in the respiratory system includes a wide variety of
beneficial, cooperative and harmful entities that play a crucial
role in supporting human wellness. Its composition is modulated by
variables, such as age and dietary habits, which in turn influence
its stability and functionality. The microbiome engages with the
host immune system and external pathogens, thus underscoring its
inherent vulnerabilities (36).
The microbiome composition of the respiratory tract and related
organs as documented in prior studies is presented in Table II (11,36-42).
 | Table IIMicrobiome composition of respiratory
organs. |
Table II
Microbiome composition of respiratory
organs.
| Respiratory
organ | Microbiome
composition | (Refs.) |
|---|
| Healthy lung | Firmicutes,
Proteobacteria and Bacteroidetes. | (11) |
| Healthy lung | Bacteria:
Firmicutes, Proteobacteria, Bacteroidetes, Actinobacteria,
Fusobacteria | (36) |
| | Fungi:
Ascomycota, Basidiomycota | |
| | Virus:
Bacteriophages | |
| Healthy lung | Bacteroidetes,
Firmicutes, Proteobacteria and Fusobacteria | (37) |
| Nasopharyngeal
samples (healthy) | The anterior nare
and nostril included Staphylococcaceae,
Propionbibacteriaceae and Corynebacteriaceae. Also found
some oral cavity microorganism, Streptococcaceae,
Veillonellaceae, and Prevotellaceae | (38) |
| Oropharynx and oral
wash (healthy) |
Streptococcaceae, Veillonellaceae,
Fusobacteriaceae and Neisseriaceae | (38) |
| Bronchoalveolar
lavage (healthy) |
Sphingomonadaceae,
Pseudomonaceae, Burkholderiaceae, Staphylococcaceae,
Propionibacteriaceae and Staphylococcaceae | (38) |
| Lungs of patients
with COPD | Haemophilus,
Moraxella, Klebsiella and Pseudomonas.
Streptococcus and Pseudomonas is associated with
different immune responses and exacerbation frequencies in
COPD | (39,40) |
| Lungs of patients
with cystic fibrosis | Staphylococcus
aureus and Prevotella shahii | (41) |
| Lungs of
smokers |
Ralstonia | (42) |
Geographic location exerts a profound influence on
the foundational microbial architecture of healthy pulmonary
systems. In addition, variables such as chronological age, external
factors and sequencing methodology also influence the results of
microbial composition analysis (38). The lung microbiome exhibits a
dynamic nature, initiated by the influx of microbes from the upper
respiratory tract and environmental exposures (43). The lung microbiome plays a crucial
role in immune balance and provides resistance to pathogen
colonization (39). Microbiome
stability is a phenomenon regulated by factors such as host
characteristics, environmental context, and microbial
interrelationships. Such stability is essential for maintaining
health, as disruptions can trigger dysbiosis and associated
diseases. Determinants affecting microbiome stability can be
broadly classified into host-related factors, environmental
variables, microbial interrelationships and external interventions
(44-48).
Host-related factors include biological sex,
chronological age, health conditions and the immune system, in
addition to host genetic predisposition. Biological sex has emerged
as an key determinant of microbiome composition, with females
typically exhibiting greater stability. Chronological age also
influences microbiome dynamics, as changes in bacterial populations
are evident across life stages (45). Health conditions such as metabolic
liver disease and diabetes mellitus are associated with microbiome
instability, often resulting in reduced diversity and increased
abundance of facultative pathogens, which can exacerbate dysbiosis
(46). The immune system of the
host and genetic makeup can also shape microbiome stability by
influencing microbial interactions and responses to environmental
stimuli (47).
The diversity of microbial taxa present in the
environment can drive temporal fluctuations in the host microbiome.
Environments with high microbial diversity can foster a more
dynamic but potentially less stable microbiome (49). Increased biodiversity and complex
microbial networks can enhance microbiome resilience. However,
excessive complexity can also lead to instability; thus, a balance
between diversity and stability is necessary (50). Microbial competition has the
potential to enhance stability by limiting positive feedback
mechanisms and weakening ecological interactions. Conversely,
cooperative networks, while exhibiting efficiency, may
paradoxically possess reduced stability (47). Antibiotic interventions can induce
substantial disturbances within the microbiome, with the recovery
trajectory being contingent upon prior exposures and the presence
of restorative dynamics within the microbial consortium (48). The strategic design of microbial
communities, characterized by specific initial parameters and
inoculation ratios, can optimize metabolic stability, particularly
within regulated environments such as fermentation processes
(51).
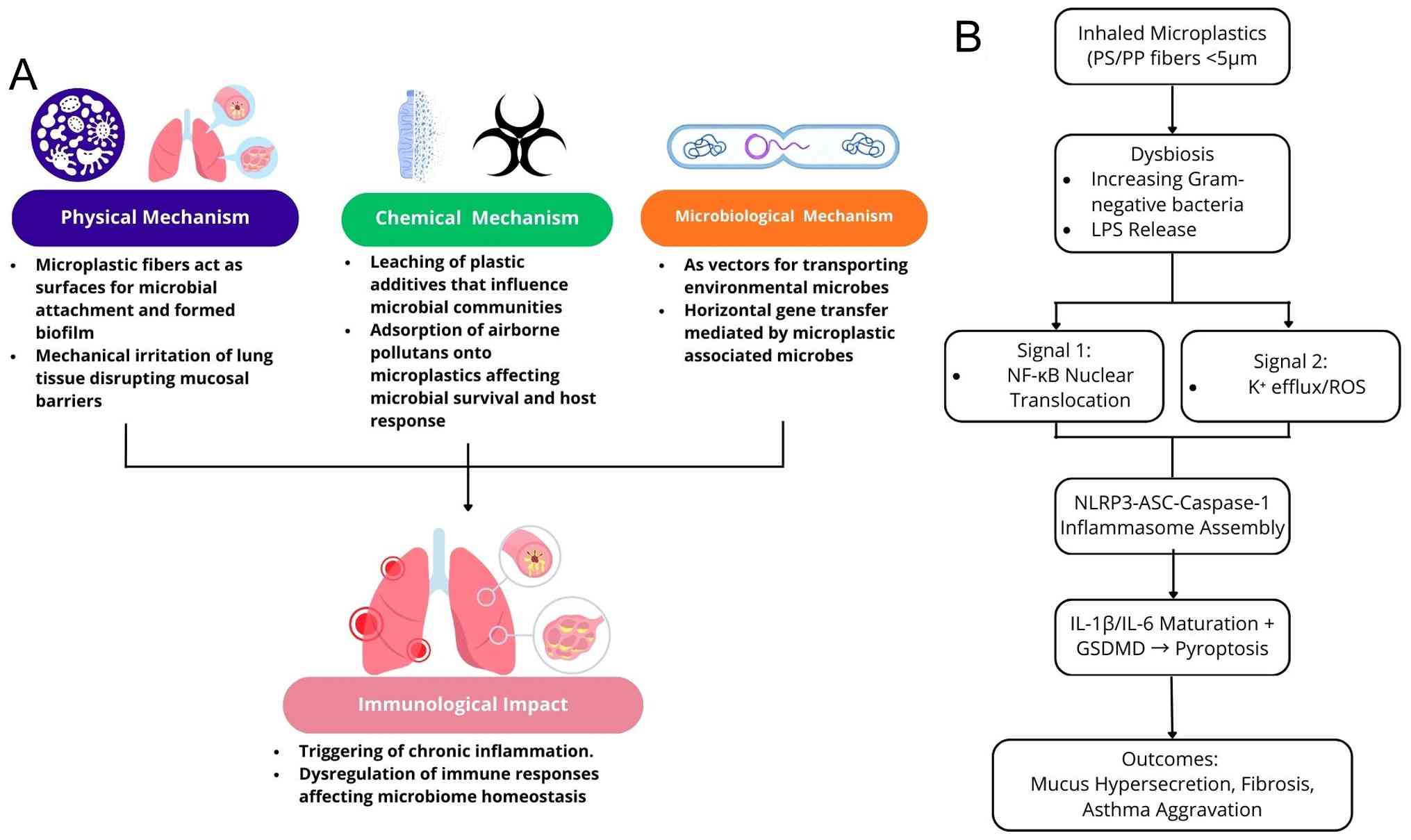
4. Mechanisms: How airborne microplastics
affect the respiratory microbiome
Microplastics, characterized as diminutive
particulates, possess the capability to be inhaled and subsequently
accumulate within the respiratory tract, where they may engage with
the pulmonary microbiota, potentially giving rise to deleterious
health implications. The pathways through which microplastics
influence the respiratory microbiome are illustrated in Fig. 2.
Microplastics exhibit considerable variability in
dimensions, morphology and chemical makeup, which significantly
affects their interactions with pulmonary tissues and associated
microbiota. Smaller particulates, particularly those exhibiting
positive charges, possess an elevated propensity for translocation
and engagement with systemic circulation, thereby impacting the
equilibrium of the respiratory microbiome (52). The surfaces of microplastics
provide an ecological niche for a variety of microbial communities,
encompassing bacteria and fungi, which collectively form biofilms
referred to as the ‘plastisphere’. This microbial niche is enriched
with bacteria, fungi and protists via extracellular polymeric
substances (EPS) that enhance adhesion and horizontal gene
transfer. For example, Laboratory and field studies confirm rapid
colonization on polyethylene, polypropylene and polystyrene in
marine/freshwater systems, with biofilms altering microplastic
buoyancy, sorption and pathogen harboring within days to weeks
(53). Microplastics of smaller
dimensions stimulate the development of microbes, where resilient
forms such as polyvinyl chloride and polystyrene display greater
biomass and diversity within biofilms (54). Microplastics, mainly those
measuring <5 µm, can cross epithelial barriers and gather in
lung tissues, possibly leading to irritation and a disruption of
mucosal integrity (55).
Pulmonary plastisphere formation, however, lacks
direct human evidence and requires validation beyond environmental
analogs. In murine models of ovalbumin-induced asthma, fibrous
microplastics (e.g., polyester) exposure significantly enriched
Prevotella spp. in BALF, which is associated with airway
inflammation, mucus hypersecretion and fibrosis; Prevotella
was found to increase 2-5-fold vs. the controls, alongside reduced
levels of Lactobacillus (16). This suggests fiber surfaces may
selectively promote anaerobic Gram-negatives in asthmatic lungs,
although true biofilm (EPS-matrix) confirmation requires scanning
electron microscopy/transmission electron microscopy analysis.
The process of microplastic formation can instigate
the leaching of plastic additives. Phthalates and bisphenol A (BPA)
are expelled from plastics via several environmental methods, like
photodegradation and mechanical fragmentation. This leaching
phenomenon is intensified by the aging of plastics, which augments
the release of dissolved organic carbon (DOC) into the ecological
milieu, with aged plastics emitting substantially greater
quantities of DOC compared to their virgin counterparts (56). The movement of plastic derivatives
such as phthalates and BPA extensively modifies microbial
environments. The leaching of these additives can precipitate a
decline in microbial diversity and induce a shift in community
composition. For example, exposure to leachates from plastics has
been demonstrated to adversely affect marine picophytoplankton and
modify the taxonomic and functional diversity of marine microbial
assemblages (57).
As explained in the previous section, microplastics
potentially release phthalates and BPA. BPA exposure has been shown
to affect fetal lung development by altering the expression of
genes and proteins involved in steroid synthesis and metabolism
(58). It increases the expression
of GPR30 and ERβ, and activates the NF-κB signaling pathway, which
leads to the decreased release of immune modulators such as IL-6
and ET-1, and increases oxidative stress and DNA damage in lung
fibroblasts. Additionally, BPA suppresses glucocorticoid receptor
(GR) activity through ERβ/NF-κB signaling, leading to the reduced
expression of ENaCγ, a GR target gene crucial for lung function
(58). In vitro research
suggests that BPA affects lung fibroblasts by altering gene
expression and immune signaling pathways, which could affect lung
development and increase susceptibility to respiratory diseases
(58). BPA exposure has been
linked to changes in gut microbial composition, increasing certain
bacterial species that may influence systemic immune responses and
respiratory health via the gut-lung axis (59).
Phthalate exposure is associated with lung
microbiota dysbiosis, which can mediate adverse respiratory
effects. This dysbiosis is linked to conditions, such as asthma and
COPD. Phthalates, such as di-(2-ethylhexyl) phthalate (DEHP), have
been shown to alter the gut microbiome. DEHP exposure increases the
diversity of gut microbiota and alters the abundance of specific
bacterial families, which may influence immune responses and
respiratory health (59).
Phthalates have been linked to an increased risk of developing
asthma and respiratory symptoms. The potential mechanism involves
the activation of peroxisome proliferator-activated receptors,
which are implicated in inflammatory responses in the lungs
(60). Additionally, phthalate
exposure is associated with changes in the nasal microbiome, such
as an increased abundance of Moraxella, which is linked to
the development of asthma (61).
The extensive surface area and natural chemical
features of microplastics enhance their ability to capture
pollutants. Various living organisms can ingest these pollutants
that are rapidly taken up by microplastics, leading to their entry
into the food chain. Microplastics containing pollutants can
trigger an imbalance in microbial health, mainly within the
respiratory and gastrointestinal systems, by altering the stability
of microbial communities. This dysbiosis has been shown to be
associated with the enhanced generation of reactive oxygen species
and systemic inflammation, which may jeopardize microbial viability
and host health (62).
Microplastics establish a uniform, water-resistant
base that aids in the attachment and increase of microorganisms,
encompassing bacteria, fungi and viruses. The microbial composition
of these biofilms is modulated by the type of plastic utilized,
prevailing environmental conditions and the ‘weathering age’ of
microplastics (63). Microplastics
found in aquatic environments can promote the growth and
transmission of diseases, thus raising the chances of human
exposure, notably in scenarios such as wastewater management
facilities and aquaculture practices (64). The introduction of microplastics
into the food system raises alarming issues regarding food safety,
since these particles can hold foodborne bacteria and toxic agents.
In addition, the existence of microplastics enhances the lateral
transfer of genes among bacterial communities, possibly aiding in
the spread of genes related to antibiotic resistance. The role of
the plastisphere in disseminating antibiotic-resistant bacteria
represents a novel challenge for mitigating antibiotic resistance
within the environmental context (65).
5. Health implications of respiratory
microbiome dysbiosis induced by microplastics
The impact on health linked to respiratory
microbiome disturbances, resulting from microplastic contact, is
becoming more widely recognized as a critical issue, given how
widespread microplastics are in the ecosystem and their ability to
harm human health. Microplastics possess the capability to be
inhaled and subsequently deposited within the respiratory tract,
penetrating deeply into the pulmonary system, including the
alveolar regions where gas exchange transpires. Such deposition may
contribute to respiratory microbiome dysbiosis by triggering
oxidative stress and inflammatory reactions (66,67).
The presence of microplastics in the respiratory
system may affect the composition of the lung microbiota, leading
to an increased prevalence of Gram-negative bacterial species. This
disruption can trigger immunological responses, such as the release
of lipopolysaccharides (LPS), which trigger Toll-like receptor 4
(TLR4) signaling pathways, leading to increased inflammation and
potential lung damage (67). LPS
binds TLR4/MD-2 complexes on alveolar macrophages and epithelial
cells, inducing receptor dimerization and the recruitment of
adaptor proteins such as MyD88. This activates downstream NF-κB
translocation to the nucleus, promoting the transcription of
pro-inflammatory genes, while simultaneously triggering NLRP3
inflammasome assembly via K+ efflux and the generation
of reactive oxygen species. NLRP3 activation leads to caspase-1
autocleavage, which processes pro-IL-1β and pro-IL-18 into mature
cytokines, alongside gasdermin D cleavage driving pyroptotic cell
death and IL-6 release (68).
In polystyrene microplastic inhalation models, the
upregulation of TLR4 is associated with lung ferroptosis and
fibrosis, with burdens (e.g., 2.19 particles/g tissue) matching
inflammatory thresholds. This cascade exacerbates mucus
hypersecretion and airway remodeling in mice with ovalbumin-induced
asthma. Furthermore, the capacity of microplastics to accumulate
toxic chemicals and heavy metals may increase their toxicological
effects on the respiratory microbiome. These types of interactions
have the potential to perturb microbial ecosystems and exacerbate
dysbiosis (55).
Current evidence indicates inflammatory responses to
inhaled microplastics with dose in experimental animals/ cell
culture. A number of studies have demonstrated dose-dependent
increases in oxidative stress, pro-inflammatory cytokines (e.g.,
IL-6 and TNF-α), epithelial barrier disruption and an aggravated
pathology with greater exposure to microplastics (66,67).
Examples include controlled inhalation or instillation studies
where higher doses or concentrations produced greater inflammatory
readouts and functional impairment (67). Human studies have quantified
internal microplastics burdens, but were only observational and did
not provide controlled dose-response data (25-33).
Mechanistically, immune/inflammatory systems often display graded
responses rather than a single hard threshold; low exposures may
prime immune signaling, whereas greater exposures provoke overt
inflammation (67). Apparently, a
study quantitative human threshold has not yet been
established.
Chronic respiratory conditions such as asthma and
COPD may result from the effects of microplastics on the gut flora.
Microplastic-induced inflammation and oxidative stress are key
contributors to the development and course of these disorders
(69). Systemic health depends on
the respiratory microbiome, and a dysbiosis there can have
far-reaching effects. According to the gut-lung association,
hypothetical changes in the respiratory microbiome may have effects
on gut health and vice versa, which may lead to chronic health
issues and widespread inflammation (70). A microplastic-induced immune
response can lead to chronic inflammation, a known risk factor for
a number of diseases, including metabolic and cardiovascular
conditions (69).
6. Research gaps and future directions
The possible health risks linked to airborne
microplastics are clarified by current research, particularly as
regards their effects on lung structure, function and the
respiratory microbiome (16,25-26,67).
However, further research is urgently required to fully elucidate
these effects and develop effective mitigation measures. It is
clear that there are no established procedures for assessing the
exact harmful effects of microplastics on pulmonary tissues. To
facilitate comparisons between studies and fully determine the
extent of health effects attributed to microplastics, this
standardization is essential (52). There is a gap in the current
knowledge of the long-term health effects and possible chronic
conditions that may arise from prolonged exposure to airborne
microplastics, as the majority of studies focus on acute exposure
(16,33,66-67).
The interactions of the human respiratory microbiome with airborne
microplastics are not yet fully understood. Further investigations
are warranted to assess the mechanisms through which microplastics
affect microbial diversity and the risk of dysbiosis, which can
lead to respiratory illnesses. The health hazards that airborne
microplastics pose to human populations have not been extensively
studied. In vitro and animal studies provide the majority of
the evidence, highlighting the need for human epidemiological
research to assess effects in the real world (71).
The development of novel techniques is essential for
measuring exposure to microplastics. This includes creating
effective, low-cost instruments for airborne particle sampling and
analysis. To establish a clear link between exposure to airborne
microplastics and the consequences for respiratory health,
comprehensive epidemiological studies are necessary. Quantifying
exposure levels and relating them to related health effects should
be the goals of such studies (72). Future studies are required to focus
on elucidating the ways in which airborne microplastics impact
respiratory health, with a particular emphasis on pathways
associated with oxidative stress, inflammatory reactions and immune
regulation. Targeted therapeutic interventions may be made easier
with a thorough understanding of these mechanisms (67).
The most up-to-date data are derived from in
vitro or short-term animal experiments, leaving marked
uncertainty as regards the long-term and dose-specific health
consequences following the inhalation of microplastics. To address
this knowledge gap, prospective cohort studies of occupational
exposure workers (e.g., textile producers and waste handlers) and
general population cohorts are urgently required. Such studies
would integrate quantitative exposure quantifications of inhaled
microplastics and nanoplastics with 16S rRNA sequencing or shotgun
metagenomics of respiratory microbiota together with clinical and
immunological characterization. Such an integrative approach would
enable correlation analyses between microplastics burdens,
microbial dysbiosis and disease symptoms, such as asthma, COPD, or
subclinical inflammation. Furthermore, follow-up studies need to
firmly control for confounding environmental and lifestyle
variables, particularly fine particulate matter exposure, cigarette
smoking, occupational pollutant exposures and the level of
urbanization, to assess the independent impact of microplastics on
alterations in respiratory microbiome and lung disease.
Examining the health effects of changes in the
respiratory microbiome brought on by exposure to airborne
microplastics may lead to the development of novel treatment
strategies. Developing methods to increase beneficial microbial
populations and investigating the possible protective roles of a
diverse microbiome are two examples of this (73).
7. Conclusion
The evidence provided in the present review
demonstrates that airborne microplastics are omnipresent
environmental contaminants capable of entering the human
respiratory system through inhalation. There are numerous studies
that have confirmed the presence of microplastics in pulmonary
tissues and BAL fluid, establishing inhalation as the primary route
of exposure (25-33).
Apart from their physical presence, microplastics have been shown
to invade and transform the respiratory microbiome, an essential
regulation of pulmonary immune homeostasis and resistance to
disease. By physical contact with microbial communities, induction
of inflammatory reactions and the carriage of chemical additives or
environmental pollutants, such particles have the potential to
contribute to microbial dysbiosis, oxidative stress, immune
dysregulation and increased susceptibility to chronic respiratory
illness, such as asthma and COPD. Their potential use as vehicles
for pathogenic microorganisms and antibiotic resistance genes
merely serves to enhance their broader public health risk.
In contrast to previous reviews that have had
centrally a focus on exposure quantification, the present synthesis
provides a mechanistic, microbiome-oriented perspective, timing
microplastic exposure with the specific biological pathways
underlying inflammation and microbial dysbiosis (8,29,32,35).
The resolution of this complex issue requires an array of
interdisciplinary synergies that extend from environmental science,
molecular microbiology, through pulmonary medicine, to toxicology.
Key future areas of research include standardization of exposure
assessment methods, long-term cohort studies correlating inhaled
microplastic burdens with microbiome alterations and clinical
outcomes and mechanistic explorations of host-microbe-plastic
interactions at the molecular level.
Finally, effective mitigation strategies are
necessary to reduce exposure to humans and its corresponding health
effect. These include practical interventions such as indoor air
filtration, increased ventilation systems, and policy measures to
reduce synthetic fiber pollution from clothing and manufacturing
units. Together, these scientific and policy efforts can further
elucidate the respiratory effects of microplastics while advocating
evidence-based solutions to safeguard public health.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
JTW and RHH contributed equally to the conception,
design and writing of the review. JTW conducted the literature
search, organized the references and prepared the initial draft of
the manuscript. RHH refined the thematic structure, contributed to
the interpretation and synthesis of the literature, and revised the
manuscript critically for intellectual content. Both authors
approved the final version of the manuscript and agreed to be
accountable for all aspects of the work. Data authentication is not
applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Cherian E, Mohan L and Manoj SE:
Microplastics and nanoplastics and related chemicals. Toxic Effects
of Micro- and Nanoplastics: 73-86, 2024.
|
|
2
|
Koirala N, Sharma A, Gautam S, Chaulagain
N, Charalampos P and Xiao JB: Health and environmental impact of
microplastics: A closer view. Microplastic pollution: Occurrence,
Health Risk and Challenges: 206-227, 2025.
|
|
3
|
Kushbu R, Malleshappa M, Mishra A and
Menon S: Influence of Micro and Nanoplastics in Modern Food Chain:
An Inevitable Intervention. In: Futuristic Trends in Agriculture
Engineering & Food Sciences. Volume 3, Book. 6:32–41. 2024.
|
|
4
|
Llorca M and Farré M: Microplastics. In:
Analytical Methods for Environmental Contaminants of Emerging
Concern. Chapter 10: John Wiley & Sons Ltd, 353-374, 2022.
|
|
5
|
Sharma VK, Ma X, Lichtfouse E and Robert
D: Nanoplastics are potentially more dangerous than microplastics.
Environ Chem Lett. 21:1933–1936. 2023.
|
|
6
|
Gigault J, El Hadri H, Nguyen B, Grassl B,
Rowenczyk L, Tufenkji N, Feng S and Wiesner M: Nanoplastics are
neither microplastics nor engineered nanoparticles. Nat
Nanotechnol. 16:501–507. 2021.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Rezvani H, Zarrabi N, Mehta I, Kolios C,
Jaafar HA, Kao CH, Saeedi S and Yousefi N: Morphological detection
and classification of microplastics and nanoplastics emerged from
consumer products by deep learning arXiv:2409.13688v1, 2024.
|
|
8
|
Vasse GF and Melgert BN: Microplastic and
plastic pollution: Impact on respiratory disease and health. Eur
Respir Rev. 33(230226)2024.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Chen Q, Gao J, Yu H, Su H, Yang Y, Cao Y,
Zhang Q, Ren Y, Hollert H, Shi H, et al: An emerging role of
microplastics in the etiology of lung ground glass nodules. Environ
Sci Eur. 34:1–15. 2022.
|
|
10
|
Marszalek A, Marzec WZ, Łakoma A, Marzec
MT, Choiński M, Wasiewicz-Ciach P, Kuczyński P, Wydra-Rojek A,
Kutyła K and Mokot WJ: Impact of microplastics on human health:
exposure mechanisms and potential health implications. Quality in
Sport. 19(54024)2024.
|
|
11
|
Li R, Li J and Zhou X: Lung microbiome:
New insights into the pathogenesis of respiratory diseases. Signal
Transduct Target Ther. 9(19)2024.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Marimón JM: The lung microbiome in health
and respiratory diseases. Clin Pulm Med. 25:131–137. 2018.
|
|
13
|
Lira-Lucio JA, Falfán-Valencia R,
Ramírez-Venegas A, Buendía-Roldán I, Rojas-Serrano J, Mejía M and
Pérez-Rubio G: Lung microbiome participation in local immune
response regulation in respiratory diseases. Microorganisms.
8(1059)2020.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Lynch SV: The lung microbiome and airway
disease. Ann Am Thorac Soc. 13:S462–S465. 2016.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Garaci E, Pariano M, Nunzi E, Costantini
C, Bellet MM, Antognelli C, Russo MA and Romani L: Bacteria and
fungi of the lung: Allies or enemies? Front Pharmacol.
15(1497173)2024.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Wu Q, Li R, You Y, Cheng W, Li Y, Feng Y,
Fan Y and Wang Y: Lung microbiota participated in fibrous
microplastics (MPs) aggravating OVA-induced asthma disease in mice.
Food Chem Toxicol. 190(114776)2024.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Chen YW, Li SW, Lin CD, Huang MZ, Lin HJ,
Chin CY, Lai YR, Chiu CH, Yang CY and Lai CH: Fine particulate
matter exposure alters pulmonary microbiota composition and
aggravates pneumococcus-induced lung pathogenesis. Front Cell Dev
Biol. 8(570484)2020.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Muisa-Zikali N and Mpeta M: Occurrence,
behaviour and fate of airborne microplastics. Emerging contaminants
in the terrestrial-aquatic-atmosphere continuum: Occurrence, Health
Risks and Mitigation: 151-167, 2022.
|
|
19
|
Liu P, Shao L, Guo Z, Zhang Y, Cao Y, Ma X
and Morawska L: Physicochemical characteristics of airborne
microplastics of a typical coastal city in the Yangtze River Delta
Region, China. J Environ Sci. 148:602–613. 2025.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Din KS, Khokhar MF, Parvez S and Niazi
MBK: Emerging environmental challenge: A critical review of
airborne microplastics. Environ Res Commun. 6(092003)2024.
|
|
21
|
Mbachu O, Jenkins G, Pratt C and Kaparaju
P: A new contaminant superhighway? A review of sources, measurement
techniques and fate of atmospheric microplastics. Water Air Soil
Pollut. 231:1–27. 2020.
|
|
22
|
Gaylarde CC, Neto JA and da Fonseca EM:
Indoor airborne microplastics: Human health importance and effects
of air filtration and turbulence. Microplastics. 3:653–670.
2024.
|
|
23
|
Sathyamohan G, Sewwandi M, Ambade B and
Vithanage M: Sources and circulation of microplastics in the
aerosphere-atmospheric transport of microplastics. Microplastics in
the Ecosphere: Air, Water, Soil,and Food: 125-146, 2023.
|
|
24
|
Zhang B, Zhu R and Wang Y: A review of the
sources, environmental behaviours and human health of atmospheric
microplastics. Johnson Matthey Technology Review. 68:232–246.
2024.
|
|
25
|
Wang S, Lu W, Cao Q, Tu C, Zhong C, Qiu L,
Li S, Zhang H, Lan M, Qiu L, et al: Microplastics in the lung
tissues associated with blood test index. Toxics.
11(759)2023.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Qiu L, Lu W, Tu C, Li X, Zhang H, Wang S,
Chen M, Zheng X, Wang Z, Lin M, et al: Evidence of microplastics in
bronchoalveolar lavage fluid among never-smokers: A prospective
case series. Environ Sci Technol. 57:2435–2444. 2023.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Pauly JL, Stegmeier SJ, Allaart HA, Cheney
RT, Zhang PJ, Mayer AG and Streck RJ: Inhaled cellulosic and
plastic fibers found in human lung tissue. Cancer Epidemiol
Biomarkers Prev. 7:419–428. 1998.PubMed/NCBI
|
|
28
|
Jenner LC, Rotchell JM, Bennett RT, Cowen
M, Tentzeris V and Sadofsky LR: Detection of microplastics in human
lung tissue using µFTIR spectroscopy. Sci Total Environ.
831(154907)2022.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Amato-Lourenço LF, Carvalho-Oliveira R,
Júnior GR, dos Santos Galvão L, Ando RA and Mauad T: Presence of
airborne microplastics in human lung tissue. J Hazard Mater.
416(126124)2021.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Uogintė I, Vailionytė A, Skapas M, Bolanos
D, Bagurskienė E, Gruslys V, Aldonytė R and Byčenkienė S: New
evidence of the presence of micro- and nanoplastic particles in
bronchioalveolar lavage samples of clinical trial subjects.
Heliyon. 9(e19665)2023.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Lu W, Li X, Wang S, Tu C, Qiu L, Zhang H,
Zhong C, Li S, Liu Y, Liu J and Zhou Y: New evidence of
microplastics in the lower respiratory tract: Inhalation through
smoking. Environ Sci Technol. 57:8496–8505. 2023.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Baeza-Martínez C, Olmos S,
González-Pleiter M, López-Castellanos J, García-Pachón E,
Masiá-Canuto M, Hernández-Blasco L and Bayo J: First evidence of
microplastics isolated in European citizens' lower airway. J Hazard
Mater. 438(129439)2022.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Chen C, Liu F, Quan S, Chen L, Shen A,
Jiao A, Qi H and Yu G: Microplastics in the bronchoalveolar lavage
fluid of Chinese children: Associations with age, city development,
and disease features. Environ Sci Technol. 57:12594–12601.
2023.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Wan J, Shan Y, Fan Y, Fan C, Chen S, Sun
J, Zhu L, Qin L, Yu M and Lin Z: NFB inhibition attenuates
LPS-induced TLR4 activation in monocyte cells. Mol Med Rep.
14:4505–4510. 2016.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Du Z, Yu X, Li X, Zhang L, Lin Y, He Y and
Wu Y: Mechanism of microplastics in respiratory disease from 2020
to 2024: Visualization and bibliometric analysis. Front Med
(Lausanne). 12(1586772)2025.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Santacroce L, Charitos IA, Ballini A,
Inchingolo F, Luperto P, De Nitto E and Topi S: The human
respiratory system and its microbiome at a glimpse. Biology
(Basel). 9(318)2020.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Yadav M: Microbiome therapeutics in
respiratory illnesses. Microbiome Therapeutics: Personalized
Therapy Beyond Conventional Approaches: 401-419, 2023.
|
|
38
|
Tang Z, Yang S and He Z: Defining the
baseline of pulmonary microbiota in healthy populations and
influencing factors. Highlights in Science, Engineering and
Technology. 11:38–48. 2022.
|
|
39
|
Buslaev VY, Minina VI and Matskova LV: .:
Microbiota: Contribution to carcinogenesis and functioning of the
lung immune system. Bulletin of Siberian Medicine. 22:103–112.
2023.
|
|
40
|
Charlson ES, Bittinger K, Haas AR,
Fitzgerald AS, Frank I, Yadav A, Bushman FD and Collman RG:
Topographical continuity of bacterial populations in the healthy
human respiratory tract. Am J Respir Crit Care Med. 184:957–963.
2011.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Ren L, Zhang R, Rao J, Xiao Y, Zhang Z,
Yang B, Cao D, Zhong H, Ning P, Shang Y, et al: Transcriptionally
active lung microbiome and its association with bacterial biomass
and host inflammatory status. mSystems. 3:199–217. 2018.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Xue Q, Xie Y, He Y, Yu Y, Fang G, Yu W, Wu
J, Li J, Zhao L, Deng X, et al: Lung microbiome and cytokine
profiles in different disease states of COPD: A cohort study. Sci
Rep. 13:1–11. 2023.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Fabbrizzi A, Amedei A, Lavorini F, Renda T
and Fontana G: The lung microbiome: Clinical and therapeutic
implications. Intern Emerg Med. 14:1241–1250. 2019.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Motta H, Reuwsaat JCV, Lopes FC, Viezzer
G, Volpato FCZ, Barth AL, de Tarso Roth Dalcin P, Staats CC,
Vainstein MH and Kmetzsch L: Comparative microbiome analysis in
cystic fibrosis and non-cystic fibrosis bronchiectasis. Respir Res.
25:1–18. 2024.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Byrd AL, Liu M, Fujimura KE, Lyalina S,
Nagarkar DR, Charbit B, Bergstedt J, Patin E, Harrison OJ,
Quintana-Murci L, et al: Gut microbiome stability and dynamics in
healthy donors and patients with non-gastrointestinal cancers. J
Exp Med. 218(e20200606)2020.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Frost F, Kacprowski T, Rühlemann M,
Pietzner M, Bang C, Franke A, Nauck M, Völker U, Völzke H, Dörr M,
et al: Long-term instability of the intestinal microbiome is
associated with metabolic liver disease, low microbiota diversity,
diabetes mellitus and impaired exocrine pancreatic function. Gut.
70:522–530. 2021.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Coyte KZ, Schluter J and Foster KR: The
ecology of the microbiome: Networks, competition, and stability.
Science (1979). 350:663–666. 2015.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Relman DA: The human microbiome: Ecosystem
resilience and health. Nutr Rev. 70:S2–S9. 2012.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Harrison XA, Price SJ, Hopkins K, Leung
WTM, Sergeant C and Garner TWJ: Diversity-Stability dynamics of the
amphibian skin microbiome and susceptibility to a lethal viral
pathogen. Front Microbiol. 10(492347)2019.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Kajihara KT, Yuan M, Amend AS, Cetraro N,
Darcy JL, Fraiola KMS, Frank K, McFall-Ngai M, Medeiros MCI,
Nakayama KK, et al: Diversity, connectivity and negative
interactions define robust microbiome networks across land, stream,
and sea. bioRxiv 10: 01.07.631746, 2025.
|
|
51
|
Tan Y, Zhu Y, Wijffels R, Zhang H, Scott
W, Xu Y and Santos VM: Controlling metabolic stability of food
microbiome for stable indigenous liquor fermentation. NPJ Biofilms
and Microbiomes. 11(124)2024.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Rybalchenko YV: The effect of nanoplastics
and microplastics on lung morphology and physiology: A systematic
review. The Medical and Ecological Problems. 28:42–60. 2024.
|
|
53
|
Jia J, Liu Q, Zhao E, Li X, Xiong X and Wu
C: Biofilm formation on microplastics and interactions with
antibiotics, antibiotic resistance genes and pathogens in aquatic
environment. Eco Environ Health. 3:516–528. 2024.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Jin Z, Chen K, Zhu Q, Hu X, Tian S, Xiang
A, Sun Y, Yuan M and Yao H: Non-degradable microplastic promote
microbial colonization: A meta-analysis comparing the effects of
microplastic properties and environmental factors. Environ Res.
270(121053)2025.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Fröhlich E: Local and systemic effects of
microplastic particles through cell damage, release of chemicals
and drugs, dysbiosis, and interference with the absorption of
nutrients. J Toxicol Environ Health B Crit Rev. 27:315–344.
2024.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Romera-Castillo C, Birnstiel S,
Álvarez-Salgado XA and Sebastián M: Aged Plastic leaching of
dissolved organic matter is two orders of magnitude higher than
virgin plastic leading to a strong uplift in marine microbial
activity. Front Mar Sci. 9(861557)2022.
|
|
57
|
Focardi A, Moore LR, Raina JB, Seymour JR,
Paulsen IT and Tetu SG: Plastic leachates impair picophytoplankton
and dramatically reshape the marine microbiome. Microbiome.
10:1–20. 2022.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Hijazi A, Guan H and Yang K: Bisphenol A
suppresses glucocorticoid target gene (ENaCγ) expression via a
novel ERβ/NF-κB/GR signalling pathway in lung epithelial cells.
Arch Toxicol. 91:1727–1737. 2017.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Chiu K, Bashir ST, Gao L, Gutierrez J, de
Godoy MRC, Drnevich J, Fields CJ, Cann I, Flaws JA and Nowak RA:
Subacute exposure to an environmentally relevant dose of
di-(2-ethylhexyl) phthalate during gestation alters the cecal
microbiome, but not pregnancy outcomes in mice. Toxics.
9(215)2021.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Bølling AK, Holme JA, Bornehag CG, Nygaard
UC, Bertelsen RJ, Nånberg E, Bodin J, Sakhi AK, Thomsen C and
Becher R: Pulmonary phthalate exposure and asthma-is PPAR a
plausible mechanistic link? EXCLI J. 12:733–759. 2013.PubMed/NCBI
|
|
61
|
Dalton KR, Fandiño-Del-Rio M, Louis LM,
Garza MA, Quirós-Alcalá L and Davis MF: Microbiome alterations
associated with phthalate exposures in a US-based sample of Latino
workers. Environ Res. 214(114126)2022.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Mazumder H and Hussain S:
Air-pollution-mediated microbial dysbiosis in health and disease:
Lung-gut axis and beyond. J Xenobiot. 14:1595–1612. 2024.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Bolan NS, Kirkham MB, Ravindran B, Kumar A
and Ding W: Microbial plastisphere: Microbial habitation of
particulate plastics in terrestrial and aquatic environments. In:
Particulate Plastics in Terrestrial and Aquatic Environments. 1st
Edition. CRC Press, Boca Raton, FL: 135-145, 2020.
|
|
64
|
Zhong H, Wu M, Sonne C, Lam SS, Kwong RWM,
Jiang Y, Zhao X, Sun X, Zhang X, Li C, et al: The hidden risk of
microplastic-associated pathogens in aquatic environments. Eco
Environ Health. 2:142–151. 2023.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Zagui GS, Andrade LN, Sierra J, Rovira J,
Darini ALC and Segura-Muñoz S: Plastisphere as a pathway for
antimicrobial-resistant bacteria spread to the environment: New
challenge and open questions. Environ Res.
214(114156)2022.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Saha SC and Saha G: Effect of
microplastics deposition on human lung airways: A review with
computational benefits and challenges. Heliyon.
10(e24355)2024.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Kang H, Huang D, Zhang W, Wang J, Liu Z,
Wang Z, Jiang G and Gao A: Inhaled polystyrene microplastics
impaired lung function through pulmonary flora/TLR4-mediated iron
homeostasis imbalance. Sci Total Environ.
946(174300)2024.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Fu Y, Kim H, Lee DS, Han AR, Heine H,
Zamyatina A and Kim HM: Structural insight into TLR4/MD-2
activation by synthetic LPS mimetics with distinct binding modes.
Nat Communications. 16(4164)2025.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Stoian AI: The impact of microplastics and
nanoplastics on human digestive, respiratory and cardiovascular
systems. International conference KNOWLEDGE-BASED ORGANIZATION.
30:146–152. 2024.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Pang LY, Sonagara S, Oduwole O, Gibbins C
and Nee TK: Microplastics-an emerging silent menace to public
health. Life Sciences, Medicine and Biomedicine 5: doi.org/10.28916/lsmb.5.1.2021.72, 2021.
|
|
71
|
Ningrum PT, Keman S, Sulistyorini L,
Sudiana IK, Hidayat A, Negoro AHS, Junaidi H and Kustin K: A
systematic review of the effects of airborne microplastic
contamination on human lungs. Afr J Reprod Health. 28:430–448.
2024.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Damaj S, Trad F, Goevert D and Wilkesmann
J: Bridging the gaps between microplastics and human health.
Microplastics. 3:46–66. 2024.
|
|
73
|
Yi X, Cai H, Gao J and Wang Z:
Environmental exposure, airway microbiome and respiratory health:
You are what you breathe. Clin Transl Med. 13(e1394)2023.PubMed/NCBI View Article : Google Scholar
|