Introduction
Schwannoma is a benign, encapsulated peripheral
nerve sheath tumor arising from Schwann cells, most commonly
presenting as a solitary, slow-growing lesion (1,2).
Although these tumors may occur anywhere along cranial, peripheral,
or autonomic nerves, extracranial schwannomas of the head and neck
account for approximately one-quarter to almost half of all cases,
with only a small proportion originating from the cervical vagus
nerve (1,3). Vagal schwannomas typically affect
adults in the third to fifth decades of life and are generally
benign; although malignant transformation is rare, it has been
documented (4).
Despite their benign nature, schwannomas in the
cervical region pose a diagnostic challenge due to their rarity and
their tendency to mimic several parapharyngeal or lateral neck
masses (3). Their deep anatomic
location limits early detection and renders pre-operative suspicion
difficult, particularly as patients may remain asymptomatic for
prolonged periods of time (1).
This diagnostic ambiguity contributes to delayed identification and
misclassification.
Failure to recognize cervical vagal schwannoma (CVS)
can complicate clinical decision-making, may contribute to
inappropriate differential diagnoses and hinder optimal treatment
planning. Moreover, although typically indolent, these tumors may
progressively become enlarged, causing functional impairment or
displacement of adjacent neurovascular structures (4). Misinterpretation may also result in
unnecessary or inappropriate interventions.
Various strategies exist for the evaluation and
management of schwannomas; however, their application relies
heavily on correct preoperative identification. The rarity of CVS
limits clinician familiarity with its epidemiological behavior and
typical presentation. Furthermore, available reports highlight that
a number of cases continue to be diagnosed only after surgical
excision (3).
The case presented herein is uncommon due to the
young age of the patient, the imaging features that closely
mimicked a paraganglioma leading to an initial misdiagnosis, and
the successful nerve-preserving excision with an uncomplicated
postoperative course.
Case report
Patient information. On October, 2025, a
26-year-old woman presented to Tikrit Teaching Hospital (Tikrit,
Iraq) with a newly developed swelling on the left side of her lower
neck. The mass was first noted following a recent flu-like illness.
She had initially attended an Ear, Nose, and Throat (ENT) clinic
for evaluation.
Clinical findings
Upon a physical examination, the neck mass was found
to be non-tender, non-pulsatile, smooth and firm. It was mobile
laterally, but not vertically. No associated lymphadenopathy or
respiratory symptoms were present.
Diagnostic assessment
An ultrasonography revealed a well-defined
heterogeneous mass (21-22x17-24 mm) located between the carotid
artery and internal jugular vein, with internal vascularity and
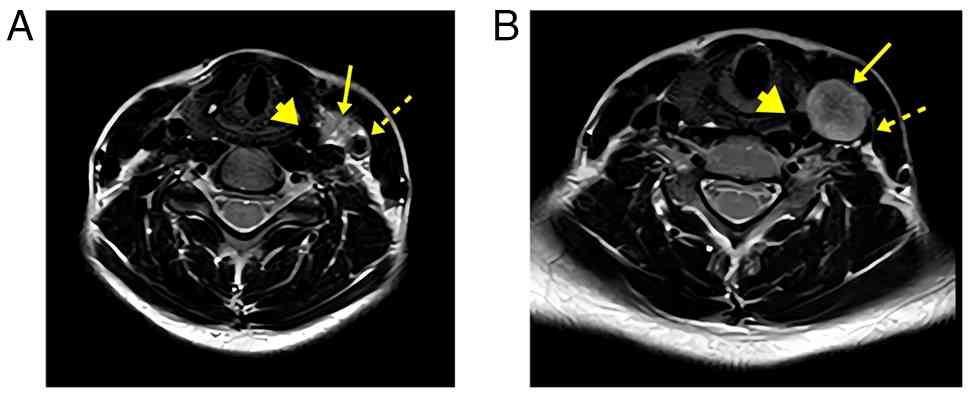
posterior venous displacement. Magnetic resonance imaging (MRI)
demonstrated a rounded mass with intermediate-to-high T2 signal
intensity anteromedial to the carotid bifurcation, causing
characteristic vascular displacement and carotid bifurcation
splaying (Fig. 1). These imaging
features closely mimicked those of a paraganglioma and resulted in
an initial radiological misdiagnosis, highlighting the diagnostic
challenge associated with CVS. A laboratory evaluation revealed
mild leukocytosis (white blood cells, 13.2x109/l),
neutrophilia (74%), thrombocytosis (520x109/l) and
elevated levels of inflammatory markers (erythrocyte sedimentation
rate, 65 mm/h; C-reactive protein, 44 mg/l). Routine biochemical
parameters, including liver and renal profiles, thyroid function
(thyroid stimulating hormone, 2.1 µIU/ml) and metabolic markers,
were all within normal limits (Table
I). The case was reviewed in a Multidisciplinary Oncology Team
(MDT) meeting, where surgical excision was recommended as the
definitive management approach. A chronological summary of the
clinical course of the patient, as well as the investigations
performed and the progression of her condition is presented in
Table II.
 | Table ISummary of the findings of the
laboratory tests performed for the patient. |
Table I
Summary of the findings of the
laboratory tests performed for the patient.
| Test category | Result | Normal range |
|---|
| White blood cell
count (WBC) |
13.2x109/l |
4.0-11.0x109/l |
| Neutrophils | 9.8x109/l
(74%) |
2.0-7.0x109/l (40-70%) |
| Platelets |
520x109/l |
150-450x109/l |
| ESR | 65 mm/h | <20 mm/h |
| CRP | 44 mg/l | <5 mg/l |
| AST | 28 U/l | 10-40 U/l |
| ALT | 32 U/l | 7-56 U/l |
| ALP | 110 U/l | 44-147 U/l |
| Blood urea | 28 mg/dl | 15-48 mg/dl |
| Serum creatinine | 0.9 mg/dl | 0.7-1.3 mg/dl |
| TSH | 2.1 µIU/ml | 0.8-6.0 µIU/ml |
| HbA1c | 5.3% | 4.0-5.6% |
| Anti-tTG IgA | 3 U/ml | <4 U/ml |
| Anti-tTG IgG | 2 U/ml | <6 U/ml |
 | Table IITimeline of the clinical course of the
patient. |
Table II
Timeline of the clinical course of the
patient.
| Date/timepoint | Event/clinical
course |
|---|
| Week 0 | Patient noticed
painless swelling on the left side of the neck after flu-like
illness. |
| Week 1 | Initial visit to ENT
clinic; physical exam revealed firm, mobile neck mass. |
| Week 2 | Neck ultrasound
showed a well-defined, vascular solid mass. |
| Week 3 | MRI suggested
paraganglioma due to carotid bifurcation splaying. |
| Week 4 | Case reviewed in
Oncology MDT; surgical excision recommended. |
| Week 5 | Underwent surgery;
mass identified as arising from vagus nerve. |
| Post-operative day
2 | Discharged without
complications. |
| 2 Weeks
post-surgery | Follow-up: no
neurological deficits or wound issues. |
| 3 Months
post-surgery | Continued stability;
no evidence of recurrence. |
Therapeutic intervention
The patient underwent surgical resection under
general anesthesia. A transverse cervical approach was used to
access the mass. Careful dissection was performed to separate the
lesion from the surrounding vascular structures and adjacent
nerves. The tumor was well-encapsulated, allowing for safe
mobilization. The complete excision of the mass was achieved
without intraoperative complications (Fig. 2). The lesion was identified as
arising from the vagus nerve and was excised with preservation of
nerve integrity. Given the close association of cervical vagal
schwannomas with vagal nerve fascicles, which often renders
nerve-sparing excision technically challenging and is associated
with a risk of postoperative vocal cord dysfunction, successful
nerve preservation in patient in the present case report is
noteworthy.
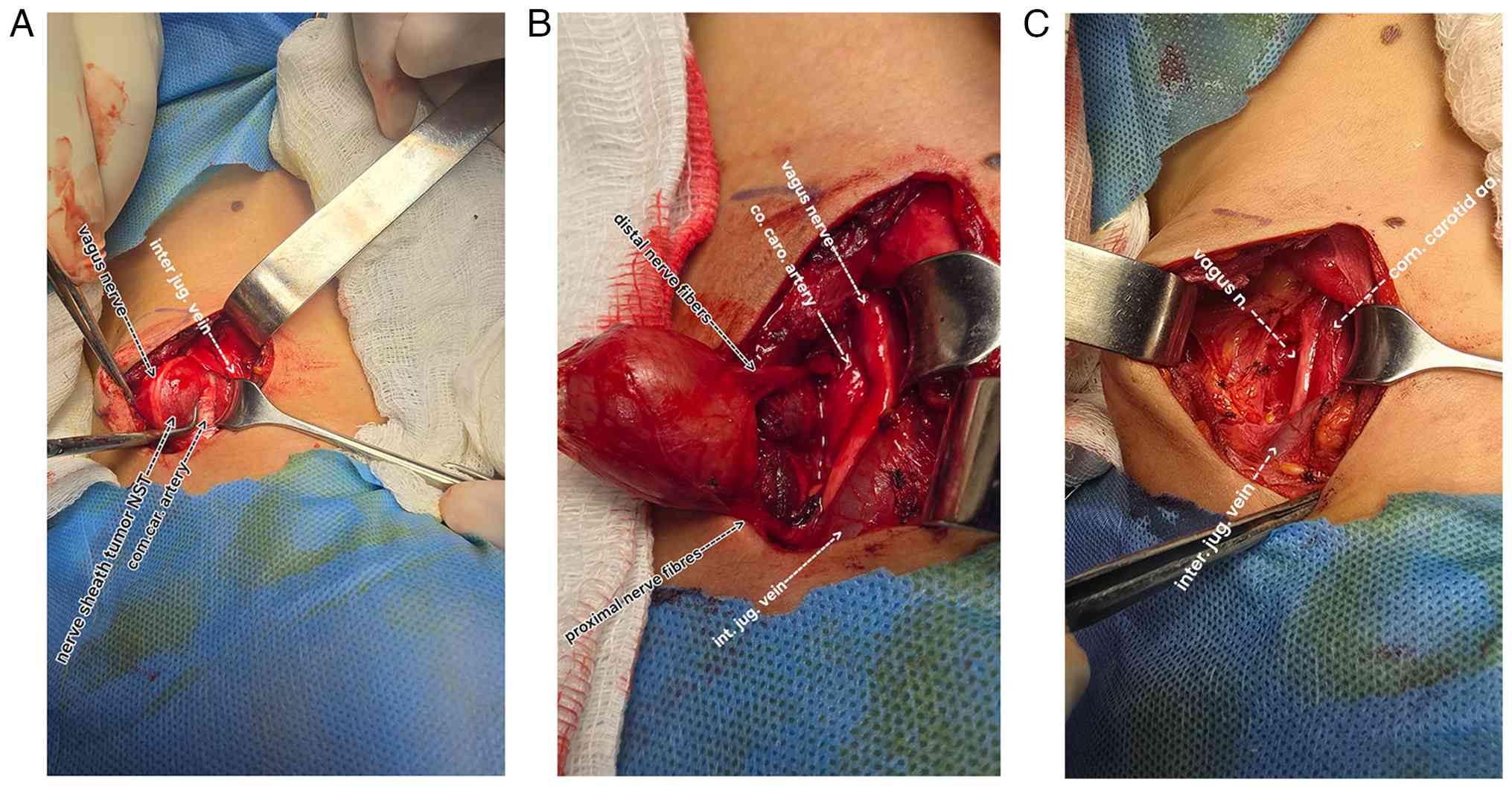 | Figure 2Intraoperative images demonstrating a
carotid sheath nerve sheath tumor adherent to the vagus nerve. (A)
Identification and exposure of the tumor with exploration of
adjacent vital structures, including the common carotid artery,
internal jugular vein, and vagus nerve. (B) Careful dissection of
the tumor from surrounding structures while preserving the proximal
and distal vagal nerve fibers. (C) Surgical field after complete
tumor excision, showing an intact vagus nerve, internal jugular
vein, and common carotid artery, with a small residual cavity at
the tumor bed. inter jug. vein, internal jugular vein; NST, nerve
sheath tumor; com.car.artery, common carotid artery; co. caro.
artery, common carotid artery; vagus n., vagus nerve. |
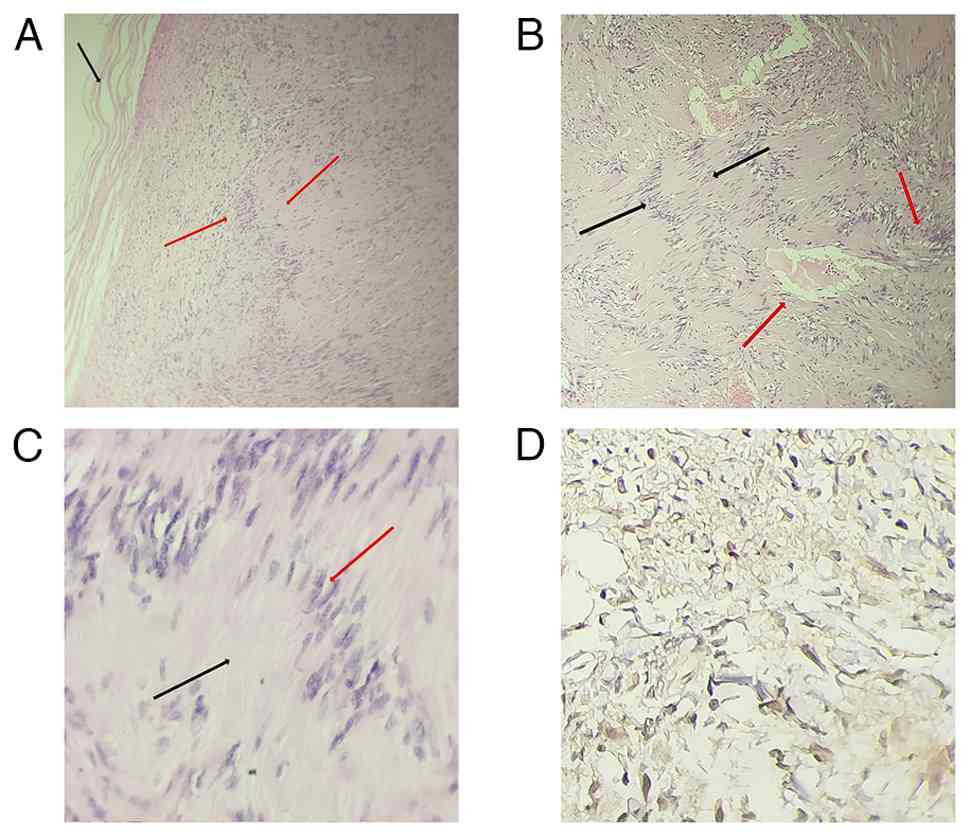
A histopathological evaluation of the excised
specimen revealed a well-encapsulated spindle-cell tumor composed
of both Antoni A and Antoni B areas, with characteristic Verocay
bodies, confirming the diagnosis of schwannoma. For
histopathological examination, tissue sections were cut at a
thickness of 4 µm from formalin-fixed, paraffin-embedded (FFPE)
specimens. The tissues were fixed in 10% neutral buffered formalin
for 24-48 h at room temperature (20-25˚C). Routine staining was
performed using hematoxylin and eosin (H&E); the stains were
obtained from Bio-Optica Co. and supplied locally by Al-Razi
Medical & Laboratory Supplies Company, Baghdad, Iraq. Staining
was carried out manually at room temperature, with sections
immersed in hematoxylin for 5-7 min, followed by differentiation
and bluing, and eosin counterstaining for 1-2 min. The sections
were then dehydrated, cleared, and mounted according to standard
protocols. A histological evaluation was performed using a light
microscope, specifically an Olympus CX23 microscope (Olympus
Corporation), supplied locally by Scientific Bureau/Olympus Iraq,
Baghdad. Immunohistochemical analysis revealed diffuse S-100
positivity, further supporting the diagnosis of a benign peripheral
nerve sheath tumor (Fig. 3). For
immunohistochemical evaluation, 4-µm-thick sections were prepared
from formalin-fixed, paraffin-embedded (FFPE) tissue blocks.
Specimens had been fixed in 10% neutral buffered formalin for 24-48
h at room temperature (20-25˚C). Sections were deparaffinized in
xylene and rehydrated through graded ethanol solutions (100 to 70%)
to distilled water. Heat-induced epitope retrieval was performed
using citrate buffer (pH 6.0) or Tris-EDTA buffer (pH 9.0) at
95-98˚C for 20-30 min, followed by cooling at room temperature. No
additional permeabilization step was applied, as routine FFPE
processing and antigen retrieval provided adequate antigen
exposure.
Endogenous peroxidase activity was blocked using 3%
hydrogen peroxide for 10 min at room temperature. Non-specific
protein binding was blocked with 5-10% normal goat serum for 20 min
at room temperature (Dako; Agilent Technologies, Inc.). Primary
antibodies were applied at manufacturer-recommended dilutions and
incubated for 30-60 min at room temperature, including polyclonal
rabbit anti-human S-100 (1:200; cat. no. Z0311; Dako; Agilent
Technologies, Inc.), mouse monoclonal anti-SOX10 (clone BC34,
1:100; cat. no. CM385; Biocare Medical), mouse monoclonal anti-EMA
(clone E29, 1:100; cat. no. M0613; Dako; Agilent Technologies,
Inc.), mouse monoclonal anti-CD34 (clone QBEnd/10, 1:100; cat. no.
M7165; Dako; Agilent Technologies, Inc.), and mouse monoclonal
anti-Ki-67 (clone MIB-1, 1:100; cat. no. M7240; Dako; Agilent
Technologies, Inc.).
Immunodetection was performed using a polymer-based
horseradish peroxidase (HRP) detection system (EnVision™ FLEX
Detection System, HRP; cat. no. K8002; Dako; Agilent Technologies,
Inc.) with an incubation time of 20 min at room temperature.
Sections were counterstained with Mayer's hematoxylin for 1 min at
room temperature, followed by bluing in tap water, dehydration,
clearing, and mounting.
Immunohistochemical slides were examined using a
light microscope, and photomicrographs were obtained with an
Olympus CX23 light microscope (Olympus Corporation).
Follow-up and outcomes
The post-operative course was smooth and uneventful.
The patient did not experience any neurological deficits, vascular
injury, or wound-related complications. Follow-up was conducted
over a total period of 3 months, comprising two follow-up visits:
the first at 1 month post-operatively and the second at 2 months
thereafter (following manuscript submission). During these visits,
the patient remained clinically stable, demonstrated satisfactory
wound healing, and showed no evidence of tumor recurrence.
Discussion
CVS is a rare benign peripheral nerve sheath tumor
derived from Schwann cells and represents one of the least common
neurogenic masses in the neck (3,5).
Although head and neck schwannomas account for ~25-45% of all
extracranial schwannoma cases, only a small proportion originate
from the vagus nerve, rendering diagnosis particularly challenging.
The clinical significance of these cases lies in their variable
presentations, frequent potential for misdiagnosis and the
implications for preserving nerve function during surgical
intervention. These tumors most commonly occur in adults between
the third and fifth decades of life, with no clear sex predilection
(6,7).
In order to better contextualize the present case,
the authors reviewed previously reported cases of CVS focusing on
clinical presentation, imaging features, management strategies and
outcomes. A total of eight reported cases of cervical vagal nerve
schwannoma were identified as representative samples in the
literature, with the ages of the patients ranging from 13 to 42
years. Tumor sizes varied considerably, from 4 to 13 cm, and the
majority of lesions arose from the right cervical vagus nerve, with
only one case involving the left side. The diagnostic evaluation
most commonly included an ultrasonography, computed tomography
scan, fine needle aspiration cytology and MRI, whereas some reports
additionally used digital subtraction angiography (DSA) or
intraoperative transcranial motor-evoked potential (TcMEP)
monitoring. Surgical management in all cases involved tumor
excision, performed as intracapsular or subcapsular enucleation, or
as total resection, with nerve preservation achieved in the
majority of procedures. Post-operative outcomes were generally
favorable; several patients demonstrated complete recovery or
clinical improvement, no recurrence and was reported during
follow-up of up to 5 years, although some cases experienced
complications, such as hoarseness, dysphonia, laryngeal paralysis
or persistent facial pain. Overall, surgical excision remains the
primary therapeutic modality, with the majority of patients
maintaining satisfactory postoperative function and recovery
(1,4,8-13)
(Table III).
 | Table IIISummary of cases of cervical vagal
nerve schwannoma reported in the literature. |
Table III
Summary of cases of cervical vagal
nerve schwannoma reported in the literature.
| Authors | Year of
publication | N | Mean age (years) | Tumor size (cm) | Sex | Location | Diagnostic
method | Surgical
techniques |
Outcome/follow-up | (Refs.) |
|---|
| Gaikwad et
al | 2013 | 1 | 13 | 6 | Male | Right cervical
VN | Ultrasound, FNAC,
MRI | Total excision (nerve
sacrificed) | Complete recovery; 10
days | (8) |
| Martins et
al | 2025 | 1 | 38 | 4.5 | Female | Right cervical
VN | MRI | Intracapsular
excision (transcervical) | Paresis, resolved
completely, and 12 months after surgery, the patient was
asymptomatic | (4) |
| Baker et
al | 2018 | 1 | 36 | 4 | Female | Right cervical
VN | CT and MRI | Surgical excision
(nerve resected) | 2-year: Permanent
Horner's and facial pain | (9) |
| Majeed and Ahmed | 2008 | 1 | 24 | 13 | Female | Left cervical VN | X-ray, Ultrasound,
FNAC, CT | Sub-capsular excision
(preserved) | No hoarseness;
successful preservation | (10) |
| de Souza et
al | 2020 | 2 | 34-35 | 7.7, 4 | Male &
Female | Right cervical VN
(both) | FNAC, MRI, CT | Intracapsular
enucleation (both) | 5-year: No
recurrence; dysphonia resolved 6 months | (11) |
| Singh and
Pinjala | 2007 | 1 | 14 | 6 | Male | Right cervical
VN | Ultrasound, FNAC,
CT, DSA | Complete excision
(nerve sacrificed) | 10 days:
Hoarseness, weakness, dysphagia | (12) |
| Tanaka et
al | 2022 | 1 | 42 | 5 | Male | Right cervical
VN | MRI, TcMEP
monitoring | Intracapsular
enucleation + monitoring | 1-year: Laryngeal
paralysis and hoarseness | (13) |
| Aregawi | 2023 | 1 | 30 | 13 | Male | Head and neck | FNAC, CT, MRI | Complete
excision | NA | (1) |
Typically, slow-growing and asymptomatic, vagal
schwannomas may be mistaken for other parapharyngeal or carotid
space lesions, such as paragangliomas, lymphadenopathy,
neurofibromas, or salivary gland tumors due to overlapping clinical
and radiological features (6,14,15).
An MRI plays a central role in the evaluation process; however,
differentiating schwannomas from paragangliomas remains difficult
due to shared radiographic characteristics, including carotid
bifurcation splaying and heterogeneous T2 signal intensity.
Therefore, a definitive diagnosis often relies on a
histopathological confirmation (16). In the case described herein, a
26-year-old patient presented with a newly developed left cervical
mass following a flu-like illness. Although the temporal
association suggested a possible inflammatory process, the clinical
behavior and radiological appearance of the lesion indicated a
neoplastic process. The mass was non-tender, mobile laterally and
located between major vascular structures, which are features
consistent with vagal schwannoma (3).
An ultrasonography and MRI demonstrated carotid
bifurcation splaying and internal vascularity, initially leading to
a radiological impression of paraganglioma. This finding is
consistent with the observations reported in the study by Tzortzis
et al (16), who noted that
vagal schwannomas can mimic carotid body tumors or sympathetic
chain schwannomas due to similar vascular displacement patterns. As
with several other cases, the schwannoma in the patient described
in the present case report remained radiologically
indistinguishable from a paraganglioma until definitive
histopathological confirmation was obtained.
Although the majority of reported cases describe
progressive symptoms, such as hoarseness, dysphagia, or paroxysmal
cough triggered by palpation a sign often considered pathognomonic,
the patient in the present case report was entirely asymptomatic,
apart from localized swelling. This clinical course is consistent
with previously reported cases of asymptomatic or incidentally
discovered cervical vagal schwannomas, in which patients present
with minimal or no neurological symptoms despite the tumor's close
proximity to vital neurovascular structures (3,7,14,16).
Surgical excision remains the definitive treatment
for CVS, as is consistently supported in the literature (3,16).
While earlier studies have suggested that nerve preservation is
challenging due to fascicular involvement (17), recent evidence suggests that
extracapsular dissection or enucleation are effective methods for
the preservation of nerve integrity and minimizing post-operative
morbidity (16). In the case
presented herein, complete tumor excision was achieved with
preservation of the vagus nerve and without post-operative
complications, consistent with the favorable outcomes reported in
more recent surgical series (16,18).
However, as demonstrated by Tanaka et al
(13), vagal dysfunction may still
occur post-operatively due to traction or ischemia during surgery,
even when anatomical preservation appears to be achieved. The
absence of neurological impairment in the patient in the present
case report highlights the importance of meticulous microsurgical
technique, particularly in smaller tumors where the risk may be
lower. The present case report further underscores the ongoing
diagnostic ambiguity associated with vagal schwannomas. The initial
misclassification as a paraganglioma illustrates the limitations of
radiological assessment and emphasizes the importance of
histopathological confirmation. Moreover, the lack of pre-operative
neurological symptoms and the uncomplicated postoperative course
suggest that early detection and smaller tumor size may contribute
to more favorable outcomes.
Complete recovery without vocal cord dysfunction or
wound-related complications is in contrast to the findings of other
published cases reporting post-operative hoarseness or vocal fold
palsy (3,18). This difference may reflect
variations in tumor size, the extent of nerve involvement and
surgical expertise.
The present case report has several key limitations
that should be mentioned. First, germline genetic testing for
schwannomatosis-associated genes (NF2, SMARCB1 and
LZTR1) was not performed. Given the young age of the
patient, such testing would have been valuable to exclude an
underlying hereditary tumor predisposition syndrome and to inform
long-term surveillance strategies. However, comprehensive germline
genetic testing is not locally available in Iraq and requires
referral to international laboratories, which limits accessibility
in the authors' setting.
Second, tumor molecular analysis for 22q11 deletion
and somatic NF2 mutations was not conducted. These
alterations are frequently reported in sporadic schwannomas and may
provide insight into tumor pathogenesis and recurrence risk.
Nevertheless, the authors' institution (Tikrit Teaching Hospital,
Tikrit, Iraq) lacks the necessary molecular pathology
infrastructure, including fluorescence in situ
hybridization, chromosomal microarray analysis, and next-generation
sequencing, and international referral poses additional logistical
challenges. Ultrasound images were unavailable for inclusion in
this report because image archiving is not fully integrated between
the hospital information system and the ultrasound unit at the
authors' institution.
Third, comprehensive brain and spine MRI screening
for additional schwannomas was not performed. This decision was
based on the isolated nature of the lesion, the absence of
syndromic features, a negative family history, and a normal
neurological examination. While such screening is recommended in
patients with confirmed hereditary syndromes, it is not routinely
indicated for solitary sporadic schwannomas. The patient remains
under clinical surveillance with ongoing neurological
follow-up.
In conclusion, CVS is a rare diagnostic challenge
that often mimics other carotid-space lesions. Definitive diagnosis
and nerve-preserving excision can result in an uncomplicated
recovery with full functional preservation.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
SHM and RQS were major contributors to the
conception of the study, as well as to the literature search for
related studies. ZDH, BAA and HAA contributed to the clinical
management of the patient, assisted with data acquisition and
interpretation, and participated in the literature review and
manuscript preparation. OMS was the radiologist who assessed the
case. WNH was the pathologist who performed the diagnosis of the
case. BAA and ZDH confirm the authenticity of all the raw data. All
authors have read and approved the final manuscript.
Ethics approval and consent to
participate
Written informed consent was obtained from the
patient for her participation in the present study.
Patient consent for publication
Written informed consent was obtained from the
patient for the publication of the present and any accompanying
images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Aregawi AB: A rare case of cervical vagal
nerve schwannoma in a 30-year-old Ethiopian man. Int Med Case Rep
J. 16:141–151. 2023.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Hammood ZD, Baba HO, Saeed YA, Salih AM,
Noori SS, Abdullah HO, Tahir SH and Kakamad FH: Cellular schwannoma
of the posterior tongue: A rare case report with a literature
review. IJS Short Rep. 7(e54)2022.
|
|
3
|
Thatal S, Karki S, Parajuli A, Agrawal Y
and Paudel D: Cervical vagal schwannoma: A case report and
literature review. Clin Case Rep. 13(e70320)2025.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Martins RS, Oliveira AJ, Lucas E and
Siqueira MG: Unusual surgical resection of asymptomatic schwannoma
of the cervical vagus nerve with risk of stroke: Case report. Case
Rep Surg. 2025(9443139)2025.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Moond V, Diwaker P, Golamari R and Jain R:
Intramuscular ancient schwannoma of the axillary nerve. BMJ Case
Rep. 14(e239445)2021.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Behuria S, Rout TK and Pattanayak S:
Diagnosis and management of schwannomas originating from the
cervical vagus nerve. Ann R Coll Surg Engl. 97:92–97.
2015.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Malone JP, Lee WJ and Levin RJ: Clinical
characteristics and treatment outcome for nonvestibular schwannomas
of the head and neck. Am J Otolaryngol. 26:108–112. 2005.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Gaikwad N, Sathe NU, Wadkar G and
Chiplunkar D: Schwannoma of the cervical vagus nerve in a child: A
case report. Indian J Otolaryngol Head Neck Surg. 65 (Suppl
1):S188–S191. 2013.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Baker AT, Homewood TJ and Baker TR:
Cervical sympathetic chain schwannoma masquerading as a vagus nerve
schwannoma complicated by postoperative Horner's syndrome and
facial pain: A case report. Int J Surg Case Rep. 49:4–7.
2018.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Majeed FA and Ahmed M: Schwannoma of the
cervical vagus nerve. Pakistan Armed Forces Med J. 58:107–111.
2008.
|
|
11
|
De Souza FHM, Bernardino SN, Martins RS,
de Araújo MBL, Malheiros RA, de Souza INB and de Souza RNB:
Cervical schwannoma of the vagus nerve: Two illustrative cases. Am
J Otolaryngol Head Neck Surg. 3(1114)2020.
|
|
12
|
Singh D and Pinjala RK: Schwannoma of the
cervical vagus nerve. Pediatr Neurosurg. 43:403–405. 2007.
|
|
13
|
Tanaka A, Uemura H, Takatani T, Kawaguchi
M, Hayashi H and Kitahara T: Intraoperative transcranial
motor-evoked potential monitoring during head and neck surgeries: A
case of cervical vagus nerve schwannoma with laryngeal paralysis.
Cureus. 14(e30526)2022.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Gilmer-Hill HS and Kline DG: Neurogenic
tumors of the cervical vagus nerve: Report of four cases and review
of the literature. Neurosurgery. 46:1498–1503. 2000.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Cavallaro G, Pattaro G, Iorio O, Avallone
M and Silecchia G: A literature review on surgery for cervical
vagal schwannomas. World J Surg Oncol. 13(130)2015.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Tzortzis AS, Dogantzis P, Koliakos N,
Tsintzos S and Tzortzis G: A rare case of cervical vagus nerve
schwannoma in an adult patient. Cureus. 14(e25211)2022.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Colreavy MP, Lacy PD, Hughes J,
Bouchier-Hayes D, Brennan P, O'Dwyer AJ, Donnelly MJ, Gaffney R,
Maguire A, O'Dwyer TP, et al: Head and neck schwannomas-a 10 year
review. J Laryngol Otol. 114:119–124. 2000.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Sandler ML, Sims JR, Sinclair C, Sharif
KF, Ho R, Yue LE, Téllez MJ, Ulkatan S, Khorsandi AS,
Brandwein-Weber M and Urken ML: Vagal schwannomas of the head and
neck: A comprehensive review and a novel approach to preserving
vocal cord innervation and function. Head Neck. 41:2450–2466.
2019.PubMed/NCBI View Article : Google Scholar
|