Introduction
Migraine is a chronic neurovascular disorder
potentially influenced by the activation of the trigeminovascular
system, neuropeptide release and systemic inflammatory
sensitization. Calcitonin gene-related peptide (CGRP) mediates
neurogenic inflammation and vasodilation, while interleukin (IL)-1β
is released from activated glia and immune cells, and is associated
with central sensitization and chronicity (1). Pentraxin-3 (PTX-3), an acute-phase
protein produced by endothelial and immune cells, reflects vascular
inflammation and endothelial dysfunction, linking systemic
inflammation to migraine pathophysiology (2). Emerging evidence indicates that
mucosal inflammatory sources, particularly gut barrier disruption,
may elevate systemic pro-inflammatory mediators, such as IL-6 and
lipopolysaccharide (LPS), lowering the threshold for trigeminal
activation and promoting vascular dysregulation (3). Together, these mechanisms support an
integrated model in which neurogenic, systemic and mucosal
inflammation converge to be associated with recurrent attacks and
the chronification of migraines (4,5).
Individuals with both migraines and periodontitis exhibit elevated
salivary IL-1β, (linked to periodontal severity) and high serum
levels of PTX-3 and CGRP (linked to migraine chronicity),
highlighting shared inflammatory mechanisms (6-8).
Periodontitis, characterized by a microbial shift
towards Gram-negative anaerobes, induces a persistent
immune-inflammatory response, evidenced by elevated levels of
IL-1β, IL-6, CRP and PTX-3 (9-11).
LPS from bacteria activate Toll-like receptor 4 (TLR4) and the
NLRP3 inflammasome, potentially related to IL-1β production,
neutrophil infiltration and MMP-9-mediated connective tissue
breakdown. PTX-3, the expression of which is upregulated in
response to IL-1β and LPS, plays a role in both local periodontal
and systemic inflammatory processes (12,13).
This systemic inflammatory milieu may potentiate migraine
pathophysiology through the activation of the trigeminovascular
system (14). Elevations in the
levels of IL-1β, CGRP and PTX-3 associated with periodontitis can
sensitize trigeminal neurons, promoting neurogenic inflammation and
vascular alterations characteristic of chronic migraines.
Convergent inflammatory pathways and TLR-mediated immune
dysregulation create a bidirectional feedback loop that may be
associated with both conditions (15,16).
While periodontitis is commonly observed in patients with chronic
migraines and is associated with increased levels of biomarkers,
such as CGRP, PTX-3 and soluble tumor necrosis factor-like weak
inducer of apoptosis (sTWEAK) in blood, saliva and gingival
crevicular fluid, to the best of our knowledge, no studies have yet
examined the simultaneous association of serum and salivary IL-1β,
CGRP and PTX-3 levels with periodontal status in individuals with
periodontitis, chronic migraines, or both. Thus, the mechanistic
link between periodontitis and migraine chronification remains
incompletely understood.
Therefore, the present study aimed to evaluate and
correlate salivary and serum IL-1β, CGRP and PTX-3 levels among
individuals with periodontitis, chronic migraines, or both
conditions. By integrating periodontal clinical parameters with
systemic and mucosal biomarker profiles, the authors sought to
elucidate whether shared inflammatory mediators could reflect a
potential inflammatory axis linking the two conditions. The present
study aimed to correlate the interrelationship between periodontal
clinical parameters [probing pocket depth (PPD), clinical
attachment level (CAL) and modified sulcus bleeding index (mSBI)]
and the serum and salivary values of IL-1β, CGRP and PTX-3.
Patients and methods
Ethical statement
The present study intended to evaluate the serum and
salivary levels of IL-1β, CGRP and PTX-3 in healthy, periodontally
healthy patients with chronic migraines, systemically healthy
subjects with periodontitis and in subjects with periodontitis with
chronic migraines. All methodologies and experimental protocols
received approval and were executed in compliance with the ethical
standards and protocols of the SRM Dental College Institutional
Review Board (IRB no SRMDC/IRB/2022/MDS/No. 507) in Chennai, India.
The initial ethical approval for the commencement of the study was
granted in December, 2022, which was formally renewed through a
continuing review in June, 2023. Participant recruitment was
initiated from February, 2023 till May, 2023, and biological sample
collection (saliva and serum) was performed from June, 2023 to
September, 2023 after obtaining the ethical renewal. Written
informed consent was obtained from all participants prior to
enrollment and sample collection.
Study population and design
A total of 96 subjects aged 18-70 years were
recruited from the Outpatient Department of SRM Dental College and
SRM General Hospital, Chennai, India. The sample size was
calculated using G power version 3.1.9.2 with 90% power and 5% α
error. A total number of 96 subjects was included, who were further
categorized on their systemic and periodontal status with 24
members in each group. The groups were as follows: Group I,
systemically and periodontally healthy subjects; group II,
individuals with chronic migraine but without periodontitis; group
III, subjects with only periodontitis and no migraines; and group
IV, subjects with co-existing chronic migraines and periodontitis.
Eligibility criteria included the presence of at least 15 teeth, a
clinical diagnosis of chronic migraine, and/or periodontitis with a
pocket depth ≥6 mm and CAL ≤5 mm in >30% of sites, corresponding
to stage II or stage III periodontitis, as per the 2017
classification by the American Academy of Periodontology (17). Subjects were excluded if they had
any systemic diseases (other than migraines) or were under
medication, had undergone periodontal therapy in the prior 6
months, used tobacco (chewing or smoking), or were pregnant or
lactating. A self-reported migraine-specific medication use within
3 months prior to sampling was recorded from the subjects with
migraines; this is presented in Table
I. Additionally, information on the body mass index, diet and
lifestyle variables of the patients was recorded for all subjects
(Table II).
 | Table IMigraine-specific medication use
within 3 months prior to sampling. |
Table I
Migraine-specific medication use
within 3 months prior to sampling.
| Medication
class | Group II (n=24)
(%) | Group IV (n=24)
(%) | Total (n=48)
(%) |
|---|
| Triptans | 5 (20.8) | 6(25) | 11 (22.9) |
| Gepants | 1 (4.2) | 1 (4.2) | 2 (4.2) |
| NSAIDs
(≥2x/month) | 8 (33.3) | 9 (37.5) | 17 (35.4) |
| Acetaminophen | 4 (16.7) | 6(25) | 10 (20.8) |
| CGRP-mAbs | 2 (8.3) | 3 (12.5) | 5 (10.4) |
| Preventive
gepants | 1 (4.2) | 1 (4.2) | 2 (4.2) |
| Beta-blockers | 3 (12.5) | 5 (20.8) | 8 (16.7) |
| Topiramate | 3 (12.5) | 3 (12.5) | 6 (12.5) |
|
Antidepressants | 2 (8.3) | 3 (12.5) | 5 (10.4) |
| Onabotulinumtoxin
A | 1 (4.2) | 2 (8.3) | 3 (6.3) |
| Acute medication
≥10 days/month | 3 (12.5) | 4 (16.7) | 7 (14.6) |
| No migraine
medication | 4 (16.7) | 3 (12.5) | 7 (14.6) |
 | Table IIBMI, diet and lifestyle variables of
the patients. |
Table II
BMI, diet and lifestyle variables of
the patients.
| Variable | Group I (n=24) | Group II
(n=24) | Group III
(n=24) | Group IV
(n=24) | Comment |
|---|
| BMI
(kg/m2), mean ± SD | 24.9±3.0 | 25.4±3.1 | 25.8±3.2 | 26.1±3.4 | Groups matched |
| Diet score
(Mediterranean 0-14), mean ± SD | 6.1±1.8 | 5.8±2.0 | 5.7±1.9 | 5.6±1.9 | No major
differences |
| Vegetarian diet, n
(%) | 4 (16.7%) | 3 (12.5%) | 5 (20.8%) | 4 (16.7%) | Self-reported |
| Alcohol
(occasional), n (%) | 5 (20.8%) | 6 (25.0%) | 6 (25.0 %) | 7 (29.2%) | Not statistically
different |
| Smoking status | 0 smokers | 0 smokers | 0 smokers | 0 smokers | Smoking excluded
per protocol |
Sample collection
Saliva and venous blood samples were collected from
eligible participants, immediately following a clinical examination
and prior to any periodontal or medical intervention.
Clinical examination
Periodontal parameters, including PPD, CAL, mSBI,
and plaque index (PI) were recorded using a UNC-15 probe. All
measurements were performed by a calibrated examiner
(intra-examiner reliability κ=0.87).
Migraine screening questionnaire
(MS-Q)
Patients with chronic migraines, diagnosed by
neurologists, were included in the present study. The MS-Q was used
for the subjects with migraines (groups II and IV) using the MS-Q,
a self-reported questionnaire (18) (Fig.
1). Each subject answered the following questions with a yes/no
for each parameter. The intensity of the migraine was graded as
‘moderate, severe and very severe’.
Saliva sample collection
Care was taken to avoid the collection of saliva
contaminated by blood; ~5 ml unstimulated whole saliva was
collected in a sterile container by asking the patient to spit in
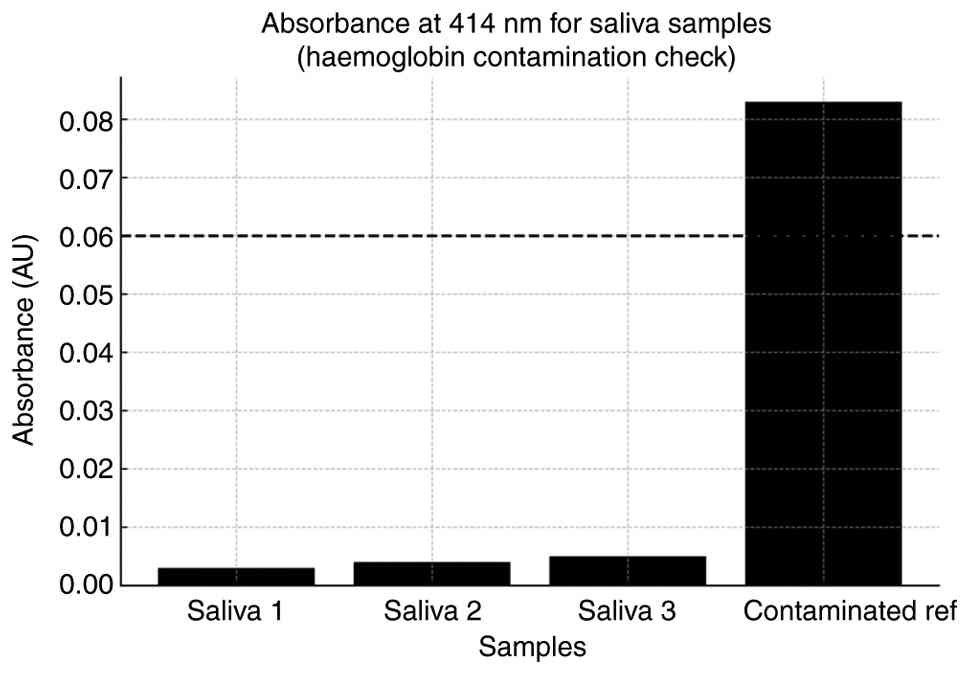
it. To objectively exclude blood contamination, the hemoglobin
absorbance of all saliva samples was measured at 414 nm (Soret
peak) using a microplate spectrophotometer (BioTek Epoch 2; BioTek;
Agilent Technologies, Inc.); recorded absorbance values ranged from
0.002 to 0.008 AU, well below the predefined exclusion threshold
(A414 >0.06), and therefore no samples were excluded
(Fig. 2). All samples were
centrifuged at 3,000 x g for 10 min at room temperature (22-25˚C),
and the clarified supernatant was transferred into UV-transparent
plates. The collected samples were then transferred to Tarsons
conical end centrifuge tubes (Tarsons Products Limited) and
centrifuged for 10 min at 2,700 x g at room temperature (22-25˚C).
Following centrifugation, supernatants were collected and aliquoted
into 0.5 ml Eppendorf tubes which were then preserved at -80˚C
until ELISA was performed.
Serum sample collection
Using the venipuncture technique, 5 ml venous blood
were withdrawn using a 5-ml syringe from the antecubital vein. The
blood was then transferred into non-EDTA tubes and allowed to stand
in a slanting position for 45 min following which the blood was
allowed to coagulate naturally. The tube was then placed in a
centrifuge and spun at 3,000 x g for 10 min at room temperature
(22-25˚C). Serum was separated using a micropipette and stored in
0.5-ml Eppendorf tubes -80˚C until ELISA was performed.
Blinding and randomization
procedures
All saliva and serum tubes were coded using
non-identifiable numeric codes by a researcher independent of
clinical data collection. Laboratory personnel performing ELISA
were blinded to group allocation. Data entry and statistical
analyses were performed using concealed codes until the analysis
was complete.
ELISA and validation
Commercial sandwich ELISA kits (Abbkine Scientific
Co., Ltd.) were used IL-1β (cat. no. KTE6013), PTX-3 (cat. no.
KTE61036), and CGRP (Human CGRP ELISA Kit, cat. no. EH22808) for
IL-1β, CGRP and PTX-3. Validation parameters were included as
manufacturer-reported sensitivity and detection ranges. Standard
curves (R2 >0.98 for all assays) were reported.
Duplicate measurements were performed for each sample. The
intra-assay CV was <8% and the inter-assay CV was <12%. The
analysis was performed with the use of internal controls,
consisting of measured positive control samples with known
concentrations of the target analyte (IL-1β, CGRP and PTX-3) and a
zero-analyte (negative) control, along with blank wells, to ensure
assay accuracy and reliability. Calibration was performed with
known concentration controls and only assays meeting QC thresholds
were included. Spike-recovery experiments were conducted for IL-1β,
CGRP and PTX-3, and recovery rates between 92 and 106% were
demonstrated in the saliva and serum samples, indicating good assay
accuracy and minimal matrix interference. All saliva and serum
samples for CGRP assessment were processed within 30 min of
collection, supplemented with a broad protease-inhibitor cocktail
(Abbkine PI-01; Abbkine Scientific Co., Ltd.), centrifuged
immediately, at 3,000 x g for 10 min at 4˚C, aliquoted and stored
at -80˚C. Samples underwent no more than one freeze-thaw cycle.
Detection ranges were: IL-1β, 7.8--500 pg/ml; CGRP, 3.75-60 ng/l;
PTX-3, 0.5-8 µg/l.
Statistical analysis
The Kolmogorov-Smirnov and Shapiro-Wilks tests were
used to assess the normality of the data and parametric methods
were applied as all variables exhibited a normal distribution.
One-way ANOVA with Tukey's HSD post hoc tests were used to compare
the mean values between groups. An independent samples t-test was
used to compared the means, whereas the Chi-squared test assessed
proportions between the study and control groups, with Fisher's
exact test applied for expected cell frequencies <5. Karl
Pearson correlations evaluated the linear correlations between
variables. All tests were performed at a significance level of
α=0.05. Data were analyzed using SPSS v26 software (Dotmatics).
Effect size reporting (η2 or ANOVA, Cohen's d), α error
was adjusted to 0.05; two-tailed analyses were performed. Post hoc
power analysis was performed for the primary outcome (serum IL-1β:
Group IV vs. group II). Using the observed mean difference
(45.67±10.97 vs. 11.84± 2.72 pg/ml), the calculated effect size was
d=4.07, yielding a power of 1.00 at α=0.05. The Benjamini-Hochberg
FDR correction was applied to ANOVA-derived biomarker comparisons;
all remained significant (adjusted P<0.05). Correlation analyses
were exploratory; therefore, effect sizes (r) and unadjusted
P-values are presented.
Results
Study subjects
A total of 96 subjects were recruited for the study,
including 38 males and 58 females, with a mean age of 44.84 years.
All participants met the established inclusion criteria and
completed the clinical and biochemical evaluations. The four study
groups demonstrated comparable demographic characteristics,
ensuring that observed differences in biomarkers and periodontal
parameters were not attributable to age or sex distribution.
MS-Q assessment
The analysis of migraine intensity based on the MS-Q
revealed marked differences among the groups. Group IV,
representing individuals with chronic migraines and periodontitis,
exhibited the greatest symptom burden. Notably, 37.5% of
participants in this group reported ‘very severe’ migraine
episodes, a proportion significantly higher than that observed in
group II (those chronic migraines without periodontitis) (P=0.002;
Table III). This trend
highlights the potential amplifying effect of periodontal
inflammation on migraine severity.
 | Table IIIMigraine intensity based on the
migraine screening questionnaire comparison in Groups II and
IV. |
Table III
Migraine intensity based on the
migraine screening questionnaire comparison in Groups II and
IV.
| Migraine
Intensity | Group II
(n=24) | Group IV
(n=24) | Total (n=48) | P-value |
|---|
| Moderate | 7 (29.2%) | 6 (25.0%) | 13 (27.1%) | 0.0016 |
| Severe | 17 (70.8%) | 9 (37.5%) | 26 (54.2%) | |
| Very severe | 0 (0.0%) | 9 (37.5%) | 9 (18.8%) | |
| Total | 24 (100.0%) | 24 (100.0%) | 48 (100.0%) | |
Groupwise differences in biomarker
levels
Highly significant differences were observed across
all groups for the salivary and serum levels of IL-1β, CGRP and
PTX-3 (P<0.001), indicating distinct inflammatory and
neurovascular profiles among the study categories. Group IV
consistently demonstrated the highest concentrations of all
biomarkers. For example, the serum IL-1β level increased from
5.51±1.19 pg/ml in group I to 11.84±2.72 pg/ml in group II and
45.67±10.97 pg/ml in group IV, while the salivary IL-1β level
increased from 79.06±30.49 to 199.60±36.92 pg/ml in group II and
425.58±46.93 pg/ml in group IV. Similar graded increases were
observed for CGRP and PTX-3 in both serum and saliva (Table IV).
 | Table IVGroupwise comparisons of clinical
parameters and biomarker levels in serum and saliva. |
Table IV
Groupwise comparisons of clinical
parameters and biomarker levels in serum and saliva.
| Parameter | Group I (n=24) | Group II
(n=24) | Group III
(n=24) | Group IV
(n=24) | P-value |
|---|
| PPD (mm) | 2.16±0.25 | 2.62±0.18 | 5.24±0.68 | 6.85±0.24 |
<0.001a |
| CAL (mm) | 0.00±0.00 | 0.00±0.00 | 3.88±0.66 | 5.2±0.59 |
<0.001a |
| mSBI | 1.37±0.20 | 1.63±0.33 | 2.41±0.23 | 2.77±0.11 |
<0.001a |
| PI | 0.48±0.20 | 2.04±0.40 | 2.14±0.38 | 2.53±0.30 |
<0.001a |
| IL-1β (pg/ml) | | | | | |
|
Serum | 5.51±1.19 | 11.84±2.72 | 23.00±6.24 |
<0.001a |
<0.001a |
|
Saliva | 79.06±30.49 | 199.60±36.92 | 349.58±58.67 | 425.58±46.93 |
<0.001a |
| CGRP (pg/ml) | | | | | |
|
Serum | 27.14±3.75 | 100.21±9.03 | 47.26±3.96 | 106.11±9.12 |
<0.001a |
|
Saliva | 27.08±1.74 | 42.05±5.85 | 32.15±1.60 | 73.85±4.33 |
<0.001a |
| PTX-3 (ng/ml) | | | | | |
|
Serum | 4.57±2.65 | 12.39±1.58 | 4.69±0.61 | 14.00±1.88 |
<0.001a |
|
Saliva | 0.80±0.26 | 10.09±2.76 | 1.37±0.22 | 12.38±3.03 |
<0.001a |
Serum CGRP levels were higher than the salivary
levels in all groups (P<0.001). Serum CGRP concentrations
increased from 27.14±3.75 pg/ml in group I to 100.21±9.03 pg/ml in
group II and 106.11±9.12 pg/ml in group IV, whereas the salivary
levels increased from 27.08±1.74 to 42.05±5.85 pg/ml in group II
and 73.85±4.33 pg/ml in group IV (Table IV). The elevation in the serum
CGRP levels among the chronic migraine groups supports its
established involvement in the sensitization of the
trigeminovascular system and migraine-related neurovascular
activation.
The PTX-3 concentrations exhibited a similar
pattern, with higher serum than salivary values across all groups
(P<0.001). Serum PTX-3 levels increased from 4.57±2.65 ng/ml in
group I to 12.39±1.58 ng/ml in group II and 14.00±1.88 ng/ml in
group IV. Salivary PTX-3 values also increased from 0.80±0.26 ng/ml
in group I to 10.09±2.76 ng/ml in group II and 12.38±3.03 ng/ml in
group III. The higher concentrations in observed in groups II and
IV align with the association of PTX-3 with systemic endothelial
dysfunction and inflammatory activity relevant to migraines.
Although group III exhibited elevated inflammatory markers due to
periodontitis, groups II and IV demonstrated substantially higher
serum CGRP and PTX-3 levels, suggesting that periodontal
inflammation alone does not account for the systemic neurovascular
elevations observed in subjects with chronic migraines.
Serum-saliva correlations of
biomarkers
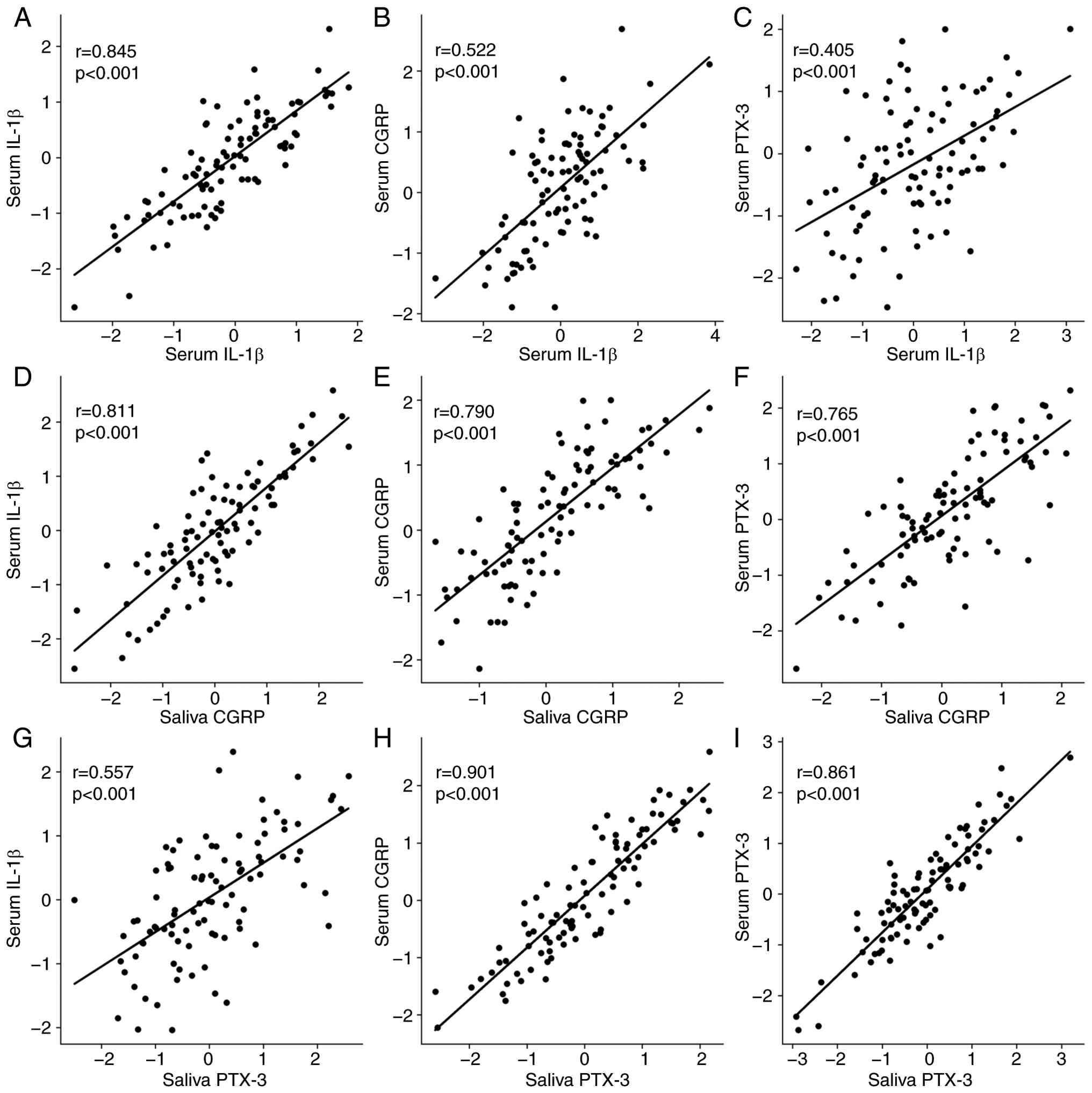
Karl Pearson's correlation analysis revealed
significant positive correlations between the serum and salivary
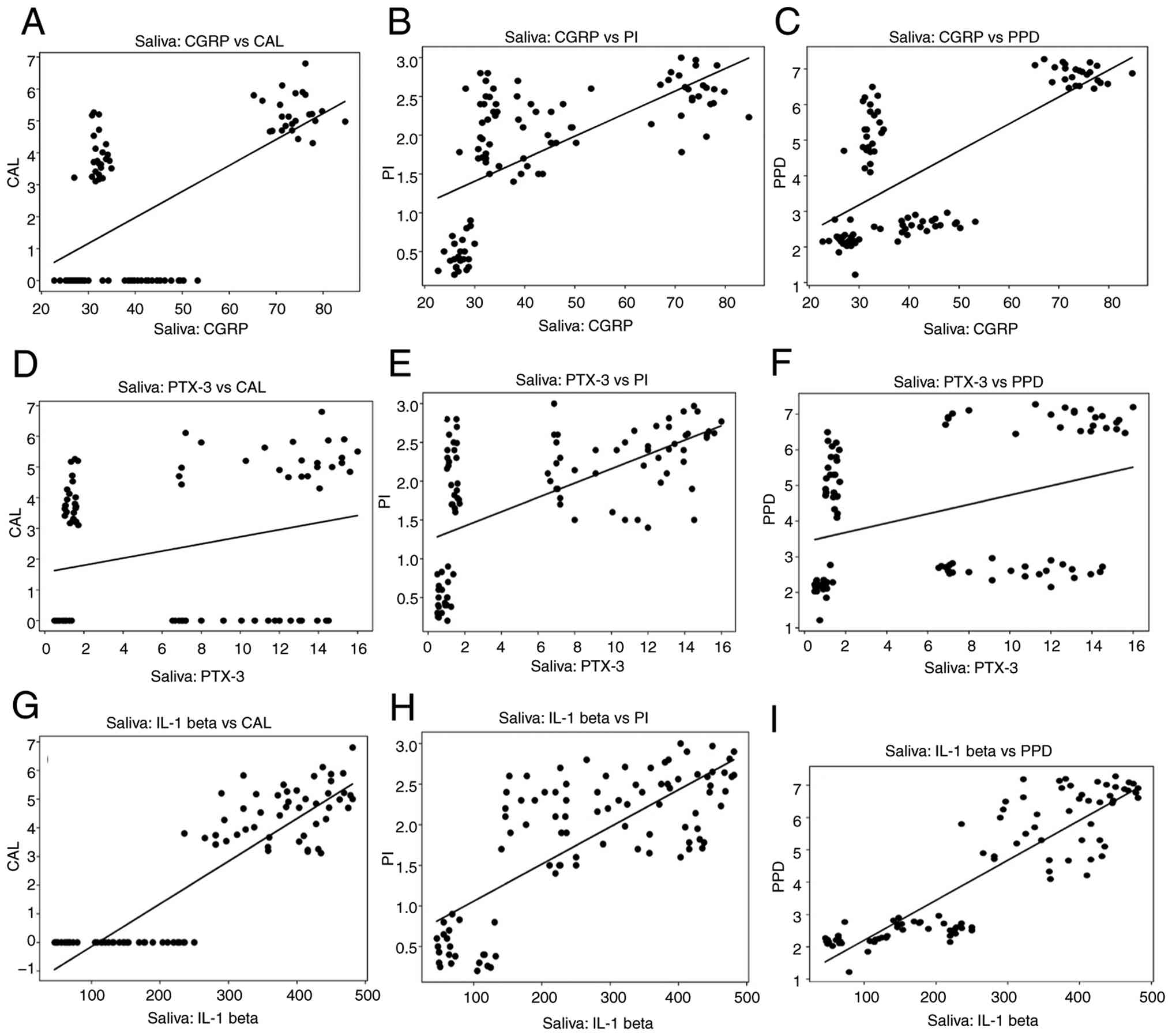
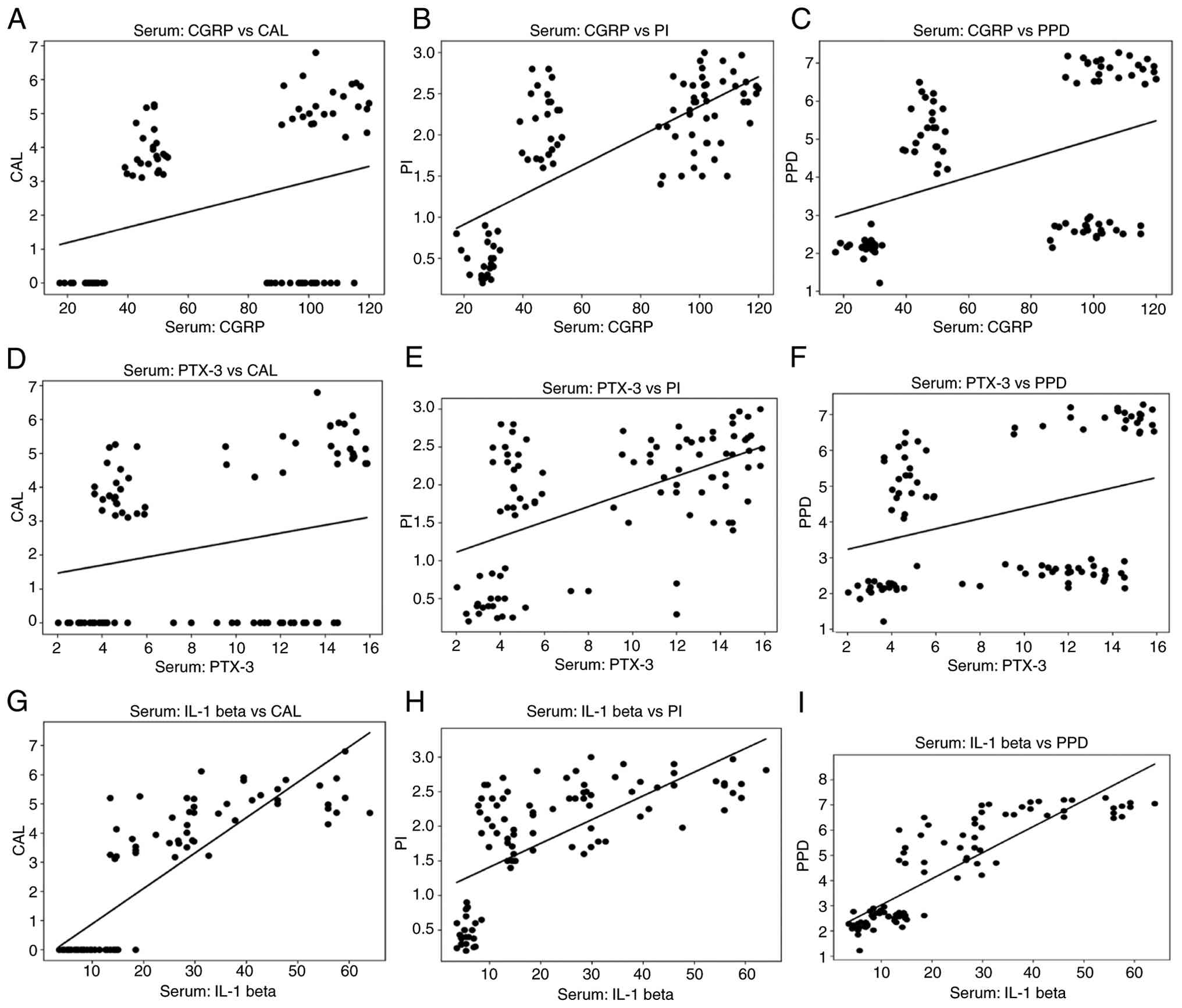
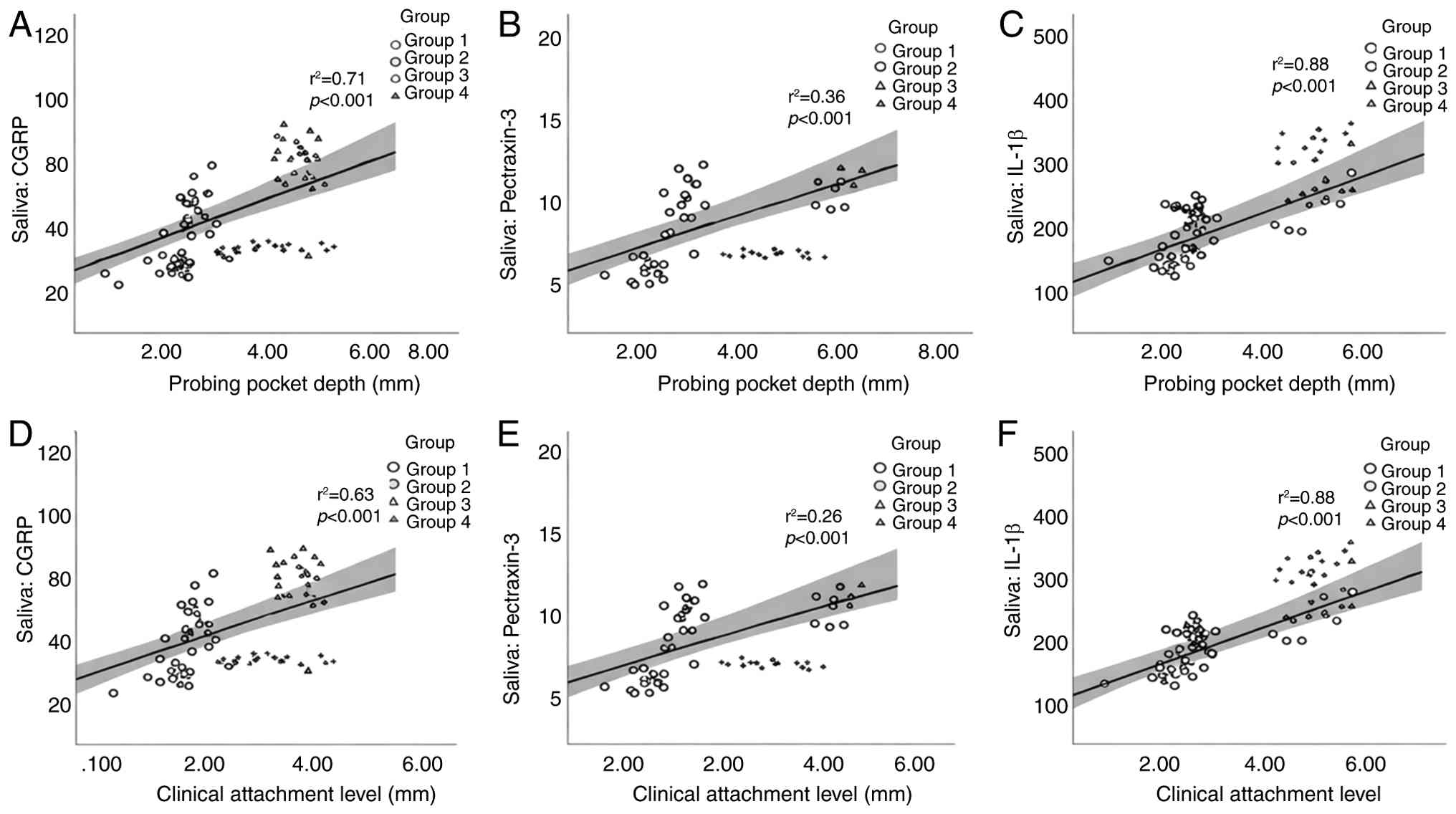
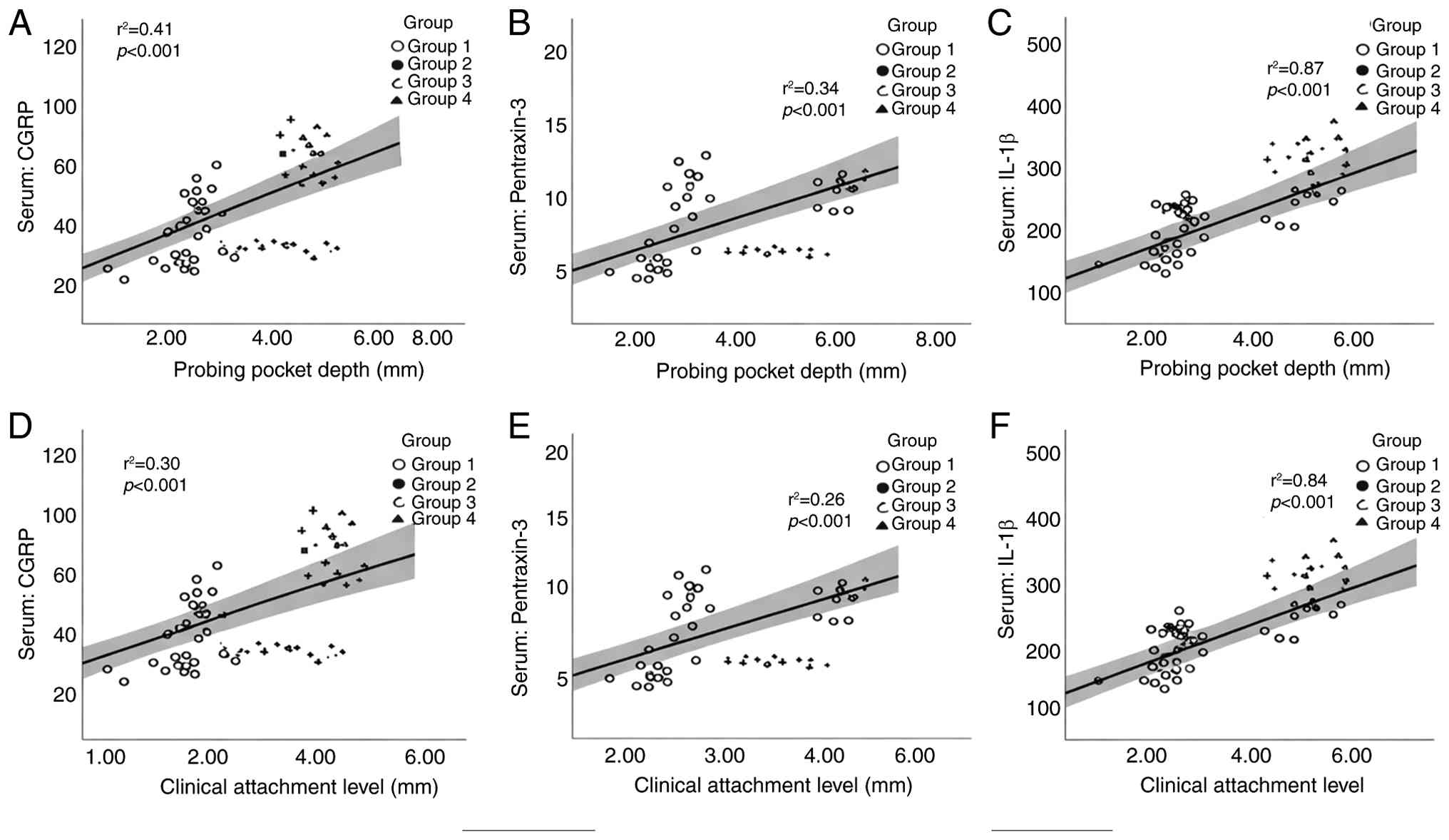
concentrations of IL-1β, CGRP and PTX-3 (P<0.001) (Table V). The correlation scatter plots
for each of these biomarkers in saliva and serum are shown in
Figs. 3 and 4, respectively. These associations
indicate that local periodontal inflammatory activity parallels
systemic inflammatory and neurovascular responses. Salivary IL-1β
demonstrated the strongest correlations, consistent with the oral
cavity being a major source of IL-1β production in periodontitis,
whereas serum CGRP and PTX-3 reflected broader systemic
neurovascular involvement in migraine. The multivariate correlation
matrix (Table VI) reinforced
these findings, with significant positive correlations across all
biomarker pairs. The scatter plots representing these correlations
are depicted in Fig. 5. There was
a sex-dependent variation in the biomarker levels and it was noted
that salivary IL-1β and serum CGRP levels were higher in females
than in males, although this was not statistically significant,
while the other biomarker levels in serum and saliva were equally
expressed in both males and females (Table VII).
 | Table VOverall correlations between salivary
and serum biomarker levels and clinical parameters. using Karl
Pearson Correlation |
Table V
Overall correlations between salivary
and serum biomarker levels and clinical parameters. using Karl
Pearson Correlation
| | | PI (n=96) | PPD (n=96) | CAL (n=96) |
|---|
| Parameters | | Correlation
value | P-value | Correlation
value | P-value | Correlation
value | P-value |
|---|
| IL-1β (pg/ml) | Saliva | 0.757 |
<0.001a | 0.889 |
<0.001a | 0.885 |
<0.001a |
| | Serum | 0.669 |
<0.001a | 0.873 |
<0.001a | 0.846 |
<0.001a |
| CGRP (pg/ml) | Saliva | 0.635 |
<0.001a | 0.717 |
<0.001a | 0.637 |
<0.001a |
| | Serum | 0.717 |
<0.001a | 0.411 |
<0.001a | 0.305 | 0.003b |
| PTX-3 (ng/ml) | Saliva | 0.600 |
<0.001a | 0.369 |
<0.001a | 0.269 | 0.008b |
| | Serum | 0.551 |
<0.001a | 0.341 |
<0.001a | 0.233 | 0.22 |
 | Table VIOverall correlations between salivary
and serum biomarkers. |
Table VI
Overall correlations between salivary
and serum biomarkers.
| Parameters | Correlation
value | P-value |
|---|
| Saliva IL-1β vs.
serum IL-1β | 0.845 | 0.001a |
| Saliva IL-1β vs.
serum CGRP | 0.522 | 0.001a |
| Saliva IL-1β vs.
serum PTX-3 | 0.405 | 0.001a |
| Saliva CGRP vs.
serum IL-1β | 0.811 | 0.001a |
| Saliva CGRP vs.
serum CGRP | 0.790 | 0.001a |
| Saliva CGRP vs.
serum PTX-3 | 0.765 | 0.001a |
| Saliva PTX-3 vs.
serum IL-1β | 0.557 | 0.001a |
| Saliva PTX-3 vs.
serum CGRP | 0.901 | 0.001a |
| Saliva PTX-3 vs.
serum PTX-3 | 0.861 | 0.001a |
 | Table VIISex-stratified biomarker levels
(serum and saliva). |
Table VII
Sex-stratified biomarker levels
(serum and saliva).
| Biomarker | Compartment | Males (n=38), mean
± SD | Females (n=58),
mean ± SD | P-value | Comment |
|---|
| IL-1β | Serum | 21.8±14.9 | 24.5±15.7 | 0.42 | No significant sex
difference |
| IL-1β | Saliva | 260±140 | 290±150 | 0.38 | Higher variance in
females |
| CGRP | Serum | 63.4±37.9 | 69.8±42.3 | 0.47 | Trending higher in
females |
| CGRP | Saliva | 41.2±18.4 | 46.5±21.1 | 0.33 | Comparable
overall |
| PTX-3 | Serum | 9.10±4.85 | 9.65±5.23 | 0.55 | No sex effect |
| PTX-3 | Saliva | 6.85±4.32 | 7.40±4.67 | 0.51 | No sex effect |
Correlation between biomarkers and
periodontal parameters
The levels of periodontal parameters in group IV
were significantly elevated, reflecting more severe periodontal
disease. The mean PPD was 6.85±0.24 mm in group IV, compared to
5.24±0.68 mm in group III, 2.62±0.18 mm in group II and 2.16±0.25
mm in the healthy controls (P<0.001). Similarly, CAL in group IV
was 5.2±0.59 mm, significantly higher than that in group III
(3.88±0.66 mm) and absent in groups I and II (0.00±0.00 mm)
(P<0.001). The m SBI and PI were also elevated in group IV (m
SBI, 2.77±0.11; PI, 2.53±0.30), indicating an increased local
inflammatory burden. Group-level analyses revealed significant
positive correlations between both serum and salivary IL-1β, CGRP
and PTX-3 levels, and clinical periodontal measures, specifically
PPD and CAL. Individuals with higher PPD and CAL values exhibited
higher concentrations of all biomarkers. These correlations were
strongest in group IV, where both periodontal inflammation and
chronic migraine were present. These correlations are illustrated
in Figs. 6 and 7, revealing clear upward trends between a
worsening periodontal status and increasing biomarker
concentrations in both saliva and serum.
Discussion
The present study provides novel insight into the
inter-relationship between chronic migraines and periodontitis,
highlighting the interplay of neurogenic and systemic inflammatory
pathways. The findings indicated that individuals with co-existing
chronic migraines and periodontitis (group IV) demonstrated a
markedly higher migraine intensity, as assessed by the MS-Q, than
those with chronic migraines but without periodontitis (group II),
periodontitis alone (group III), or the healthy controls (group I).
This observation supports the concept of a synergistic effect of
systemic and local inflammation, potentially lowering the threshold
for migraine initiation and exacerbating chronification (19-21).
The levels of periodontal parameters in group IV
were significantly elevated, reflecting more severe periodontal
disease, indicating an increased local inflammatory burden. These
findings corroborate those of earlier studies by Leira et al
(22) and Camps-Plomer et
al (23) reporting heightened
periodontal indices in patients with migraines and periodontitis.
This suggests that chronic neurogenic inflammation may be
associated with periodontal tissue breakdown or vice versa.
In the present study, the levels of IL-1β, a key
pro-inflammatory cytokine, were significantly elevated in both the
serum and saliva samples of patients in group IV. IL-1β values were
elevated in group IV compared to the other groups and salivary
IL-1β concentrations were substantially higher than serum values (P
0.001). The pronounced elevation in saliva reflects the predominant
local production of IL-1β within periodontal tissues and gingival
crevicular fluid. Micro-bleeding or subclinical gingival
inflammation may also increase salivary concentrations, consistent
with the role of IL-1β as a locally acting pro-inflammatory
cytokine in periodontal disease (24).
The elevated salivary concentrations of IL-1β likely
reflect localized periodontal inflammation, whereas increased serum
levels may indicate the systemic dissemination of inflammatory
mediators, potentially via bacterial endotoxins entering the
circulation. These findings have been reported by Sachelarie et
al (24); Relvas et al
(25) and Neurath and Kesting
(26). Furthermore, Musubire et
al (27) similarly reported
elevated levels of IL-1β in patients with migraines, although the
present study uniquely evaluated the concurrent effects of
periodontal disease and migraine on both salivary and serum IL-1β
levels.
In the present study, the leels of CGRP, a central
mediator of the activation of the trigeminovascular system, were
significantly higher in the migraine groups, with group IV
exhibiting peak values. Elevated salivary levels of CGRP are
indicative of local trigeminal activation and peripheral
nociceptive signaling, while elevations in serum suggest systemic
neurogenic inflammation that may perpetuate recurrent migraine
episodes. The studies by Alpuente et al (28); Guo et al (29), Dholakia et al (30) and Oliveira et al (31) reported similar findings. These
findings support the notion of a bidirectional
neurovascular-inflammatory loop, where periodontal inflammation may
amplify trigeminal activation and neuropeptide release,
intensifying migraine severity.
Herein, the levels of PTX-3, an acute-phase protein
upregulated by IL-1β and TNF-α, were highest in group IV, while
PTX-3 mediates endothelial dysfunction, complement activation and
neutrophil recruitment, linking vascular inflammation to
periodontal tissue injury (32-34).
The concurrent elevation of PTX-3, IL-1β and CGRP underscores a
shared neurovascular-inflammatory axis between migraine and
periodontitis.
Correlation analyses further reinforced the
connection between biomarkers and clinical parameters. Salivary and
serum CGRP, as well as PTX-3, demonstrated significant positive
correlations with all clinical indices (P<0.001), highlighting
the clinical relevance of these biomarkers in reflecting
periodontal and neurogenic inflammation. Yaghobee et al
(10), Relvas et al
(35) and Leira et al
(22,36) similarly observed the same trends in
these biomarker values. Notably, serum PTX-3 exhibited a positive,
but non-significant correlation with CAL, suggesting
tissue-specific activity and potential compartmentalization of
inflammatory signaling.
Mechanistically, IL-1β generated by periodontal
inflammation can trigger the release of CGRP and PTX-3 synthesis,
enhancing reactive oxygen species-mediated endothelial dysfunction,
trigeminovascular sensitization, and systemic immune activation, as
observed by Pradeep et al (37) Fujita et al (38) and Wang et al (39). Herein, IL-1β, CGRP and PTX-3
elevations in group IV suggested converging inflammatory pathways;
however, these observations do not establish directional causality
between periodontitis and migraine. The proposed IL-1β-CGRP/PTX-3
axis should therefore be considered hypothesis-generating.
Alternative explanations include shared genetic predisposition to
inflammatory hyper-responsiveness, systemic low-grade inflammation,
hormonal influences, or behavioral confounders. Longitudinal and
interventional studies are required to clarify temporal
associations. CGRP, despite its inability to cross the blood-brain
barrier, exerts potent peripheral nociceptive and vasodilatory
effects, lowering the migraine threshold (40,41).
PTX-3 amplification further augments vascular inflammation and
chronicity risk, establishing a vicious cycle of bidirectional
pathology, where periodontitis may intensify migraine attacks, and
chronic migraine-associated neurogenic inflammation could
reciprocally may be associated with periodontal disease.
When contextualized against previously published
migraine-only and periodontitis-only cohorts, the magnitude of
biomarker elevations observed in the present study, particularly in
individuals with co-existing chronic migraines and periodontitis,
appears greater than those typically reported. Prior
migraine-focused studies have documented modest to moderate
increases in serum CGRP (28,29,41)
and PTX-3 (10,34,36,37),
often within a narrower range and primarily during ictal phases or
in highly selected chronic migraine populations. Similarly,
periodontal studies generally report elevated levels of salivary
IL-1β and PTX-3 (10,36,37)
in periodontitis, although with lower absolute concentrations than
those observed herein, particularly in saliva. The comparatively
larger effect sizes in the present study cohort may reflect the
cumulative and synergistic inflammatory burden imposed by the
coexistence of two chronic inflammatory conditions, rather than
either disease in isolation. Periodontitis represents a persistent
peripheral inflammatory source capable of amplifying systemic
cytokine spillover, endothelial activation and neurogenic
sensitization, which may in turn augment CGRP release and PTX-3
expression in individuals already predisposed to migraine
chronification. Additional factors that may contribute to the
observed magnitude include the inclusion of patients with
established chronic migraine, the assessment of both serum and
saliva (capturing compartment-specific inflammatory activity), and
the exclusion of confounding systemic diseases that could dilute
disease-specific effects in broader cohorts. Differences in assay
platforms, sample matrices, disease severity thresholds and the
timing of sample collection relative to migraine activity may
further explain variability across studies. Collectively, these
considerations suggest that the larger observed biomarker changes
likely reflect a biologically meaningful amplification of shared
neurovascular-inflammatory pathways when migraine and periodontitis
coexist, rather than methodological inflation alone.
Despite these compelling findings, certain
limitations of the present study should be acknowledged, such as:
The cross-sectional design precludes causal inference;
single-center recruitment may limit generalizability;
migraine-specific medication use, although recorded, may still
exert residual confounding; absence of a low-frequency migraine
group with healthy periodontium; the lack of an interventional
periodontal-therapy arm; the absence of hormonal and menopausal
stratification; in addition, correlations, although statistically
significant, require validation in larger multicenter cohorts.
Thus, further longitudinal, multicenter studies with interventional
components are warranted to validate these findings and explore
therapeutic targeting of shared inflammatory pathways.
In conclusion, chronic migraines with co-existing
periodontitis are associated with higher serum and salivary IL-1β,
CGRP and PTX-3 levels and a more severe periodontal destruction.
However, although these findings support an association between
periodontal status and neurovascular-inflammatory burden, they do
not establish causality. The present study demonstrates that
patients with co-existing chronic migraine and periodontitis
exhibit markedly elevated serum and salivary levels of IL-1β, CGRP,
and PTX-3, accompanied by worsened clinical periodontal parameters
(PPD, 6.85±0.24 mm; CAL, 5.2±0.59 mm; PI, 2.53±0.30; mSBI,
2.77±0.11). These findings suggest a bidirectional,
neurovascular-inflammatory link between the two conditions, with
IL-1β functioning as a central mediator potentially related to CGRP
release and PTX-3 expression. This inflammatory axis may be
associated with the sensitization of the trigeminovascular system,
vascular dysfunction, and migraine chronification. Salivary
biomarkers, in particular, represent non-invasive surrogates for
systemic inflammation. Further prospective and interventional
studies are required to determine whether modifying periodontal
inflammation influences migraine outcomes.
Acknowledgements
The authors acknowledge SRM Dental College, Chennai,
India, for providing the necessary clinical space and equipment for
conducting the research work.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
SKG contributed to the conception and design of the
study, investigation, data acquisition and in the writing of the
original draft of the manuscript. DP played a central role in the
conception and design of the study; developed and refined the study
methodology; oversaw data acquisition, quality control, and data
curation; supervised the overall conduct of the study; coordinated
and figure preparation, contributed to data interpretation; and was
responsible for writing, critical review, and substantive editing
of the manuscript. CMA was involved in project administration, data
curation, and in the writing, reviewing and editing of the
manuscript. PSGP contributed to the conception and design of the
study, provided critical intellectual input during study planning,
oversaw study execution, guided data interpretation, and
contributed to figure preparation and critical revision of the
manuscript for important intellectual content. DJV contributed to
the conception and design of the study, involved in study
supervision and in the formal analysis. NV played a significant
role in the preparation of the manuscript, including drafting. In
addition, NV provided essential resources, including access to
laboratory equipment, the provision of biological samples, and
reagents necessary for conducting the assays used in this study.
All authors have read and approved the final manuscript.
Ethics approval and consent to
participate
Each experimental protocol and methodology was
approved and carried out in accordance with the ethical guidelines
of the Institutional Review Board of SRM Dental College, Chennai,
India (IRB no: SRMDC/IRB/2022/MDS/No. 507). All individuals gave
their written informed consent to participate in the study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Zhang P and Berk T: Network analysis of
headache diagnoses using international classification of headache
disorders, 3rd edition. Front Neurol. 16(1526037)2025.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Carneiro-Nascimento S and Levy D: Cortical
spreading depression and meningeal nociception. Neurobiol Pain.
11(100091)2022.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Visentin D, Gobin I and Maglica Ž:
Periodontal pathogens and their links to neuroinflammation and
neurodegeneration. Microorganisms. 11(1832)2023.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Mohammed MMA, Almayeef D, Abbas D, Ali M,
Haissam M, Mabrook R, Nizar R, Eldoahji T and Al-Rawi NH: The
association between periodontal disease and chronic migraine: A
systematic review. Int Dent J. 73:481–488. 2023.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Domínguez-Vivero C, Leira Y,
López-Ferreiro A, Saavedra M, Rodríguez-Osorio X, Sobrino T, Campos
F, Castillo J and Leira R: Pentraxin 3 (PTX3): a molecular marker
of endothelial dysfunction in chronic migraine. J Clin Med.
9(849)2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Teles FRF, Chandrasekaran G, Martin L,
Patel M, Kallan MJ, Furquim C, Hamza T, Cucchiara AJ, Kantarci A,
Urquhart O, et al: Salivary and serum inflammatory biomarkers
during periodontitis progression and after treatment. J Clin
Periodontol. 51:1619–1631. 2024.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Stuchfield-Denby E, Pereira B, Bouvier D,
Dallel R and Moisset X: Distinct inflammatory profiles across
migraine states: A systematic review and meta-analysis. J Headache
Pain. 26(219)2025.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Vural S and Albayrak L: Can calcitonin
gene-related peptide (CGRP) and pentraxin-3 (PTX-3) be useful in
diagnosing acute migraine attack? J Recept Signal Transduct Res.
42:562–566. 2022.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Martínez-García M and Hernández-Lemus E:
Pro-Inflammatory and anti-inflammatory interleukins in
periodontitis: Molecular roles, immune crosstalk, and therapeutic
perspectives. Int J Mol Sci. 26(10094)2025.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Yaghobee S, Hasannia S, Hamidzadeh F,
Tahbaz SV, Shahmohammadi R and Poursafar F: Evaluation of
pentraxin-3 and interleukin-6 levels in serum and gingival
crevicular fluid in patients with generalized periodontitis and
periodontal health controls before and after scaling and root
planing. Front Dent. 22(1)2025.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Haro A, Saxlin T, Suominen AL, Jula A and
Ylöstalo P: Association of periodontal condition with serum
C-Reactive protein levels: The role of serum apolipoproteins'
concentration. J Clin Periodontol. 52:1454–1465. 2025.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Huynh TN, Toperzer J, Scherer A, Gumina A,
Brunetti T, Mansour MK, Markovitz DM and Russo BC: Vimentin
regulates mitochondrial ROS production and inflammatory responses
of neutrophils. Front Immunol. 15(1416275)2024.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Wu Y, Li J, Liu M, Gao R, Zhou H, Hu Q,
Zhao L and Xie Y: Extracellular vesicles from LPS-Treated PDLSCs
Induce NLRP3 inflammasome activation in periodontitis. Oral Dis.
31:1277–1289. 2025.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Abdulkareem AA, Al-Taweel FB, Al-Sharqi
AJB, Gul SS, Sha A and Chapple ILC: Current concepts in the
pathogenesis of periodontitis: from symbiosis to dysbiosis. J Oral
Microbiol. 15(2197779)2023.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Huang YK, Yang LC, Wang YH and Chang YC:
Increased risk of migraine in patients with chronic periodontitis:
A population-based cohort study. Int J Environ Res Public Health.
18(1921)2021.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Song JH, Rim H, Chang IB, Choi HG, Wee JH,
Kwon MJ, Kang HS and Kim JH: Association of chronic periodontitis
with migraine in a Korean adult population: A nationwide nested
case-control study. Healthcare (Basel). 13(2123)2025.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Caton JG, Armitage G, Berglundh T, Chapple
IL, Jepsen S, Kornman KS, Mealey BL, Papapanou PN, Sanz M and
Tonetti MS: A new classification scheme for periodontal and
peri-implant diseases and conditions-Introduction and key changes
from the 1999 classification. J Periodontol. 89 (Suppl 1):S1–S8.
2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Gonzalez-Martinez A, Muro I, Quintas S,
Chaparro M, Gisbert JP, Sanz-García A, Casanova MJ, Rubín de Célix
C, Vivancos J and Gago-Veiga AB: Headache in patients with
inflammatory bowel disease: Migraine prevalence according to the
Migraine Screening-Questionnaire (MS-Q) and headache
characteristics. Gastroenterol Hepatol. 47:63–71. 2024.PubMed/NCBI View Article : Google Scholar : (In English,
Spanish).
|
|
19
|
Fey JMH, Bikker FJ and Hesse D: Saliva
collection methods among children and adolescents: A scoping
review. Mol Diagn Ther. 28:15–26. 2024.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Ramasundaram V, Ponnaiyan D, Anitha CM,
Prakash PSG, Victor DJ and Singh A: Evaluation of soluble tumor
necrosis factor-like weak inducer of apoptosis, omentin, and tumor
necrosis factor-α in subjects with periodontitis and type 2
diabetes mellitus. Genet Test Mol Biomarkers. 29:1–6.
2025.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Cavestro C: Metabolic dysfunction and
dietary interventions in migraine management: The role of insulin
resistance and neuroinflammation-a narrative and scoping review.
Brain Sci. 15(474)2025.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Leira Y, Ameijeira P, Domínguez C,
López-Arias E, Ávila-Gómez P, Pérez-Mato M, Sobrino T, Campos F,
D'Aiuto F, Leira R and Blanco J: Periodontal inflammation is
related to increased serum calcitonin gene-related peptide levels
in patients with chronic migraine. J Periodontol. 90:1088–1095.
2019.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Camps-Plomer GB, Márquez-Arrico CF,
Iranzo-Cortés JE and Montiel-Company JM: Association between
migraine and periodontitis: A systematic review and meta-analysis.
Eur J Neurol. 32(e70391)2025.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Sachelarie L, Stefanescu CL, Murineanu RM,
Grigorian M, Zaharia A, Scrobota I and Hurjui LL: Role of salivary
biomarkers IL-1β and MMP-8 in early detection and staging of
periodontal disease. Medicina (Kaunas). 61(760)2025.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Relvas M, Silvestre R, Gonçalves M, Cabral
C, Mendes-Frias A, Monteiro L and Viana da Costa A: Analysis of
salivary levels of IL-1β, IL17A, OPG and RANK-L in periodontitis
using the 2017 classification of periodontal diseases-an
exploratory observational study. J Clin Med.
12(1003)2023.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Neurath N and Kesting M: Cytokines in
gingivitis and periodontitis: From pathogenesis to therapeutic
targets. Front Immunol. 15(1435054)2024.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Musubire AK, Cheema S, Ray JC, Hutton EJ
and Matharu M: Cytokines in primary headache disorders: A
systematic review and meta-analysis. J Headache Pain.
24(36)2023.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Alpuente A, Gallardo VJ, Asskour L,
Caronna E, Torres-Ferrus M and Pozo-Rosich P: Dynamic fluctuations
of salivary CGRP levels during migraine attacks: Association with
clinical variables and phenotypic characterization. J Headache
Pain. 25(58)2024.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Guo S, Jansen-Olesen I, Olesen J and
Christensen SL: Role of PACAP in migraine: An alternative to CGRP?
Neurobiol Dis. 176(105946)2023.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Dholakia SB, Rao P, Talluri S and Khan J:
The association between migraines and periodontal disease: A
systematic review of clinical studies. J Oral Biosci. 65:137–145.
2023.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Oliveira R, Gil-Gouveia R and Puledda F:
CGRP-targeted medication in chronic migraine-systematic review. J
Headache Pain. 25(51)2024.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Cicek G, Ozcan O, Akyol P, Isik O, Novak D
and Küçük H: The effect of aerobic and high-intensity interval
training on plasma pentraxin 3 and lipid parameters in overweight
and obese women. Peer J. 12(e18123)2024.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Zhu T, Ding Y, Wu X, Li Y, Cheng G, Wang
N, Yang Q, Zhang W, Chen X and Liu X: Pentraxin 3 may reflects the
expression of pro-inflammatory cytokines and the migration of
macrophages in myocarditis. BMC Cardiovasc Disord.
25(354)2025.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Chen FW, Wu YL, Cheng CC, Hsiao YW, Chi
JY, Hung LY, Chang CP, Lai MD and Wang JM: Inactivation of
pentraxin 3 suppresses M2-like macrophage activity and
immunosuppression in colon cancer. J Biomed Sci.
31(10)2024.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Relvas M, Mendes-Frias A, Gonçalves M,
Salazar F, López-Jarana P, Silvestre R and Viana da Costa A:
Salivary IL-1β, IL-6, and IL-10 are key biomarkers of periodontitis
severity. Int J Mol Sci. 25(8401)2024.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Leira Y, Ameijeira P, Domínguez C,
López-Arias E, Ávila-Gómez P, Pérez-Mato M, Sobrino T, Campos F,
D'Aiuto F, Leira R and Blanco J: Severe periodontitis is linked
with increased peripheral levels of sTWEAK and PTX3 in chronic
migraineurs. Clin Oral Investig. 24:597–606. 2020.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Pradeep AR, Kathariya R, Raghavendra NM
and Sharma A: Levels of pentraxin-3 in gingival crevicular fluid
and plasma in periodontal health and disease. J Periodontol.
82:734–741. 2011.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Fujita D, Matsuoka Y, Yamakita S, Horii Y,
Ishikawa D, Kushimoto K, Amino H and Amaya F: Rapid cleavage of
IL-1β in DRG neurons produces tissue injury-induced pain
hypersensitivity. Mol Pain. 20(17448069241285357)2024.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Wang X, Zhang J and Ji J: IL-1β-induced
pentraxin 3 inhibits the proliferation, invasion and cell cycle of
trophoblasts in preeclampsia and is suppressed by IL-1β
antagonists. Mol Med Rep. 25(115)2022.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Al-Khazali HM, Ashina H, Wiggers A, Rose
K, Iljazi A, Christensen RH, Schytz HW, Amin FM and Ashina M:
Calcitonin gene-related peptide causes migraine aura. J Headache
Pain. 24(124)2023.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Iyengar S, Johnson KW, Ossipov MH and
Aurora SK: CGRP and the trigeminal system in migraine. Headache.
59:659–681. 2019.PubMed/NCBI View Article : Google Scholar
|