Introduction
Multiple myeloma (MM) is a malignant hematological
disease characterized by the proliferation of monoclonal plasma
cells in bone marrow with the accumulation of genetic changes, and
the interaction between abnormal plasma cells and bone marrow
microenvironment (1-3).
The interaction with the bone marrow environment and the
accumulation of genetic alterations allows these cells to evade
immune surveillance, leading to long-term persistence of malignant
cells in the bone marrow microenvironment and resistance to
conventional chemotherapeutics. Although numerous patients may
achieve long-term remission with the introduction of new regimens,
relapsed/refractory MM remains challenging, and is therefore
considered an incurable disease (4-6).
However, due to recent improvements in treatment strategies and the
application of new drugs, the overall survival (OS) of patients has
significantly increased, leading to requirements for novel
prognostic factors and the measurement of progression-free survival
(PFS) rather than OS when evaluating the survival rates of patients
(7-9).
To date, the International Staging System (ISS) and
the Revised International Staging System (R-ISS) have been
considered the two most critical and widely used systems in the
prognosis of MM. However, ISS was developed from the results of
treatment prior to the use of the current regimen, and it was not
as strongly associated with PFS as it was with OS (10-12).
Despite its suitability for current treatment strategies, R-ISS
requires fluorescence in situ hybridization (FISH)
techniques that are time consuming and costly (13,14).
Moreover, the second revision (R2-ISS) that includes
gain/amplification of 1q21 is also time consuming and costly
(15). Thus, numerous studies have
focused on determining novel prognostic factors that are suitable
for updated treatment strategies, ensuring both convenience and
cost. Notably, the high neutrophil to lymphocyte ratio (NLR) has
been proposed as a poor prognostic factor. Over the past 10 years,
numerous previous studies have evaluated NLR; however, an accurate
cut-off value is yet to be established, while numerous other
prognostic factors have exhibited its use in clinical practice,
with well-established cut-off values; for example, hemoglobin
(<100 g/l) and platelet count (<150x109/l)
(16-27).
Thus, the prognostic value of NLR has yet to be widely used in
clinical practice. Moreover, recent studies with large datasets did
not include NLR in the prognosis of MM (28-31),
and two studies even demonstrated that NLR did not play a key role
in predicting the short-term survival of patients diagnosed with MM
(32,33). It appears that the prognostic role
of NLR in multiple myeloma warrants further investigation. Thus,
the present study aimed to determine the value of NLR in the
prognosis of patients with diagnosed MM, compared with other
prognostic indices.
Patients and methods
Patients
The present study was a retrospective cohort study
conducted at the Hematology and Blood Transfusion Center, Bach Mai
Hospital, Hanoi, Vietnam. In total, 109 patients newly diagnosed
with MM who underwent chemotherapy from January, 2019 to June, 2023
were recruited in in the present study. Patients received specific
treatment regimens, such as bortezomib, cyclophosphamide and
dexamethasone (VCD), bortezomib, thalidomide and dexamethasone
(VTD) and bortezomib, lenalidomide and dexamethasone (VRD), and the
response to chemotherapy was evaluated after four to eight cycles.
Moreover, patient follow-up was carried out for a further 4 years
to assess OS and PFS. In the present study, patients who received
autologous stem cell transplantation were excluded. The study
protocol was approved by the Institutional Review Board of Bach Mai
Hospital (approval no. 7219/QĐ-BM, dated December 31, 2024).
Patients, or their guardians and family members were informed and
consented to participate in the study. They were called to request
consent.
Data collection
Pre-treatment laboratory indices included peripheral
blood cell indices, such as hemoglobin level, white blood cell
(WBC) count, platelet count and NLR, bone marrow cell indices, such
as bone marrow count (BMC) and bone marrow plasma cell percentage,
and biochemical blood indices, such as albumin, creatinine,
calcium, lactate dehydrogenase (LDH) and β 2 microglobulin (β2M)
levels. Prior to treatment, the performance status (PS) of the
patients was recorded using the Eastern Cooperative Oncology Group
(ECOG) criteria (34), and stage
was recorded according to the ISS criteria (10). The data were collected from the
medical records of the patients, which are stored at the hospital.
Data collection began after the study received ethics approval
(after December 31, 2024).
Definitions
Patients were diagnosed with MM according to the
criteria of the International Myeloma Working Group (IMWG)
2014(14). Patients were evaluated
on response to chemotherapy, according to IMWG 2016(35). OS was calculated from the time of
diagnosis to death or the last follow-up. PFS was calculated from
the beginning of treatment to relapse, death or the last
follow-up.
Statistical analysis
Differences in pre-treatment laboratory indices,
including hemoglobin level, WBC count, platelet count, NLR, BMC,
bone marrow plasma cell percentage, albumin, creatinine, calcium,
LDH and β2M levels among ISS groups (ISS I, II and III) were
analyzed using one-way ANOVA or the Kruskal-Wallis test, depending
on whether variables followed a normal or non-normal distribution.
Bonferroni post-hoc tests were applied for parametric data, whereas
Dunn's post-hoc tests were used for non-parametric data. The
Kolmogorov-Smirnov test was used to assess data normality.
Univariate and multivariate analyses were performed
to identify prognostic factors for OS and PFS. Prognostic variables
included established clinical factors; namely, ISS stage, ECOG PS
score ≥2, low hemoglobin level (<100 g/l), low platelet count
(<150x109/l), high bone marrow plasma cell percentage
(≥40%), elevated creatinine level (≥177 µmol/l), elevated calcium
level (≥2.75 mmol/l), increased LDH level (upper normal limit
>240 U/l according to institutional criteria) and high β2M level
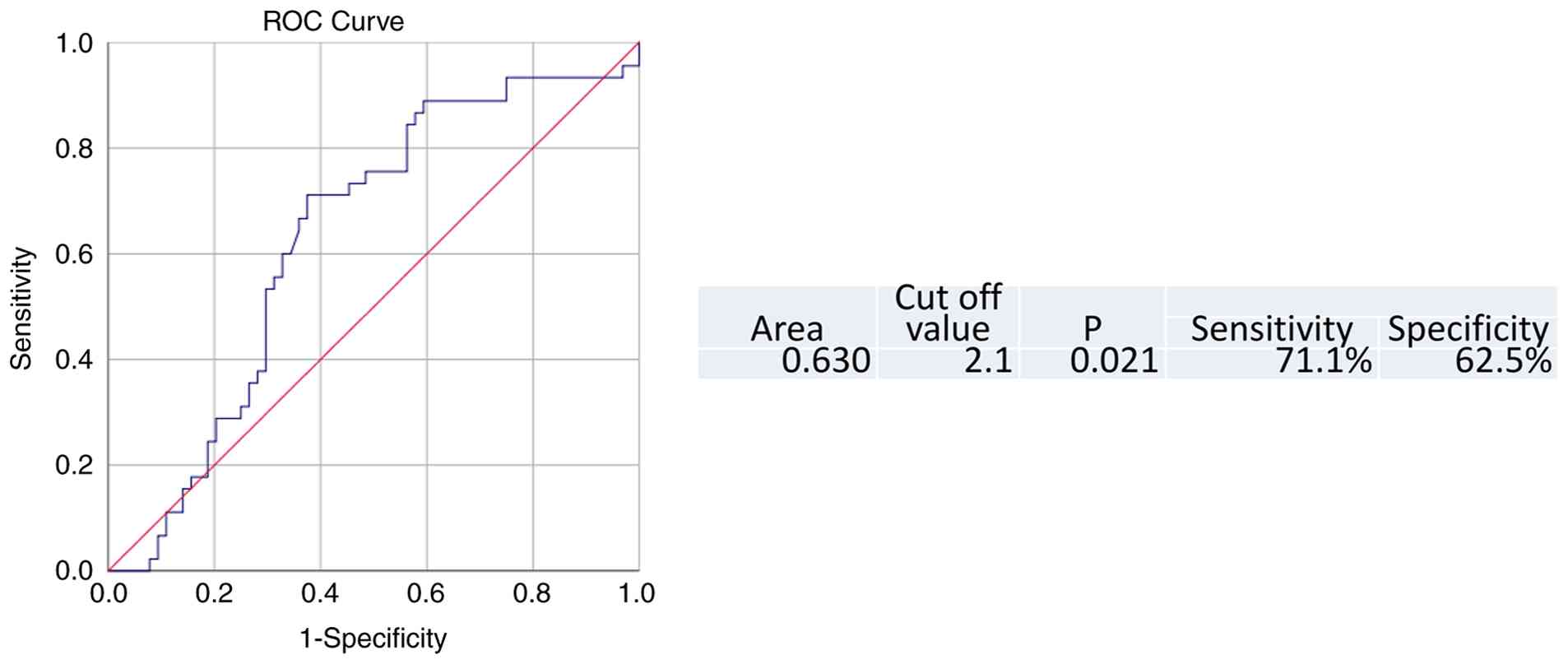
(≥5.5 mg/l). To determine the optimal cut-off value for NLR,
receiver operating characteristic curve analysis was performed to
identify the value associated with a significant difference in OS
beyond 4 years. The area under the curve was 0.63 (P=0.021). Based
on the highest Youden index [J=max (Se + Sp-1)], the optimal NLR
cut-off value was determined to be 2.1, with a sensitivity of 71.1%
and a specificity of 62.5% (Fig.
1).
The authors used a single NLR threshold to evaluate
both OS and PFS. This was also for consistency across survival
endpoints and ease of use, therefore this NLR cut-off value (≥2.1)
was applied in both univariate and multivariate analyses for OS and
PFS. Kaplan-Meier survival curves and log-rank tests were used to
identify factors associated with OS and PFS. Variables that were
statistically significant in univariate analysis were subsequently
included in multivariate analysis using the Cox proportional
hazards model. The aim was to find an index that is meaningful for
both OS and PFS. Therefore, multivariate analysis were only
performed on those metrics that were significant for both OS and
PFS. The relative effects of prognostic factors on OS and PFS were
compared based on hazard ratio (HR) and the absolute values of
standardized regression coefficients (β). Statistical analyses were
performed using SPSS (version, 25; IBM Corp.). P<0.05 was
considered to indicate a statistically significant difference.
Results
Patient data
A total of 109 patients newly diagnosed with MM were
enrolled in the present study, including 54 males and 55 females.
The median age of the patients was 61 years (range, 35-84 years),
and the median follow-up duration was 26 months (range, 5-55
months). Patient distributions according to ECOG PS, type, ISS
stage, treatment regimen and response to chemotherapy are
summarized in Table I. Patients
with ISS stage III accounted for the largest proportion of the
cohort (67%). The proportion of patients achieving a treatment
response of very good partial response or better was 44.9%. The
estimated median OS was 37.3 months, with a 4-year OS rate of 36%.
The number of events for OS was 45. The number of events for PFS
was 60. The estimated median PFS was 30.6 months, with a 4-year PFS
rate of 25.7% (Table I). The
differences in laboratory indices among the ISS groups are shown in
Table II. Statistically
significant differences were observed in hemoglobin, albumin,
creatinine, calcium, β2M levels and bone marrow plasma cell
percentage.
 | Table ICharacteristics of the patients in
the present study. |
Table I
Characteristics of the patients in
the present study.
|
Characteristics | No. of patients
(n=109) | Percentage |
|---|
| Sex | | |
|
Male | 54 | 49.5 |
|
Female | 55 | 50.5 |
| ECOG PS | | |
|
<2 | 27 | 24.8 |
|
≥2 | 82 | 75.2 |
| Type | | |
|
IgG | 58 | 53.2 |
|
IgA | 21 | 19.1 |
|
Lambda light
chain | 17 | 15.6 |
|
Kappa light
chain | 13 | 11.9 |
| ISS | | |
|
ISS I | 8 | 7.3 |
|
ISS II | 28 | 25.7 |
|
ISS III | 73 | 67.0 |
| Treatment
regimen | | |
|
VTD | 50 | 45.9 |
|
VRD | 23 | 21.1 |
|
VCD | 35 | 33.0 |
| Response to
chemotherapy | | |
|
≥VGPR (after
four cycles) | 49 | 44.9 |
|
≥VGPR (after
eight cycles) | 49 | 44.9 |
| Survival rate | | |
| | Estimate
(months) | Rate for 4 years
(%) | No. of events
(n) |
| OS | 37.3 | 36.0 | 45 |
| PFS | 30.6 | 25.7 | 60 |
 | Table IILaboratory indices of the patients
according to ISS. |
Table II
Laboratory indices of the patients
according to ISS.
| Indices | ISS I (n-8) | ISS II (n=28) | ISS III (n=73) | Total (n=109) |
P-valuea |
|---|
| Hemoglobin (g/l),
mean ± SD | 114.75±21.10 | 95.11±18.15 | 81.57±20.35 | 87.48±22.10 | <0.001 |
| WBC
(x109/l), median | 6.80 | 6.10 | 7.10 | 6.83 | >0.05 |
| Platelet count
(x109/l), median | 262.50 | 199.50 | 191.00 | 199.50 | >0.05 |
| NLR, median | 1.37 | 1.99 | 2.06 | 1.98 | >0.05 |
| Creatinine
(µmol/l), median | 81.00 | 77.50 | 133.50 | 99.00 | <0.001 |
| Albumin (g/l),
median | 36.30 | 29.45 | 32.35 | 33.50 | 0.023 |
| LDH (U/l),
median | 175.00 | 167.00 | 171.00 | 167.00 | >0.05 |
| β2M (mg/l),
median | 2.21 | 4.02 | 9.15 | 6.86 | <0.001 |
| Calcium (mmol/l),
median | 2.07 | 2.13 | 2.31 | 2.24 | 0.045 |
| Bone marrow count
(x109/l), median | 32.94 | 33.94 | 43.07 | 40.15 | >0.05 |
| Bone marrow plasma
cell percentage, median | 19.00 | 12.00 | 32.00 | 24.50 | 0.003 |
Prognostic factors
The results of the univariate analysis demonstrated
that a low hemoglobin level (<100 g/l), low albumin level
(<35 g/l) and high creatinine level (≥177 μmol/l) were not
associated with survival outcomes in the present study cohort. This
analysis indicated that a high β2M level (≥5.5 mg/l), PS score ≥2,
elevated LDH level and high ISS stage were associated with a poor
OS; however, these were not significantly associated with PFS.
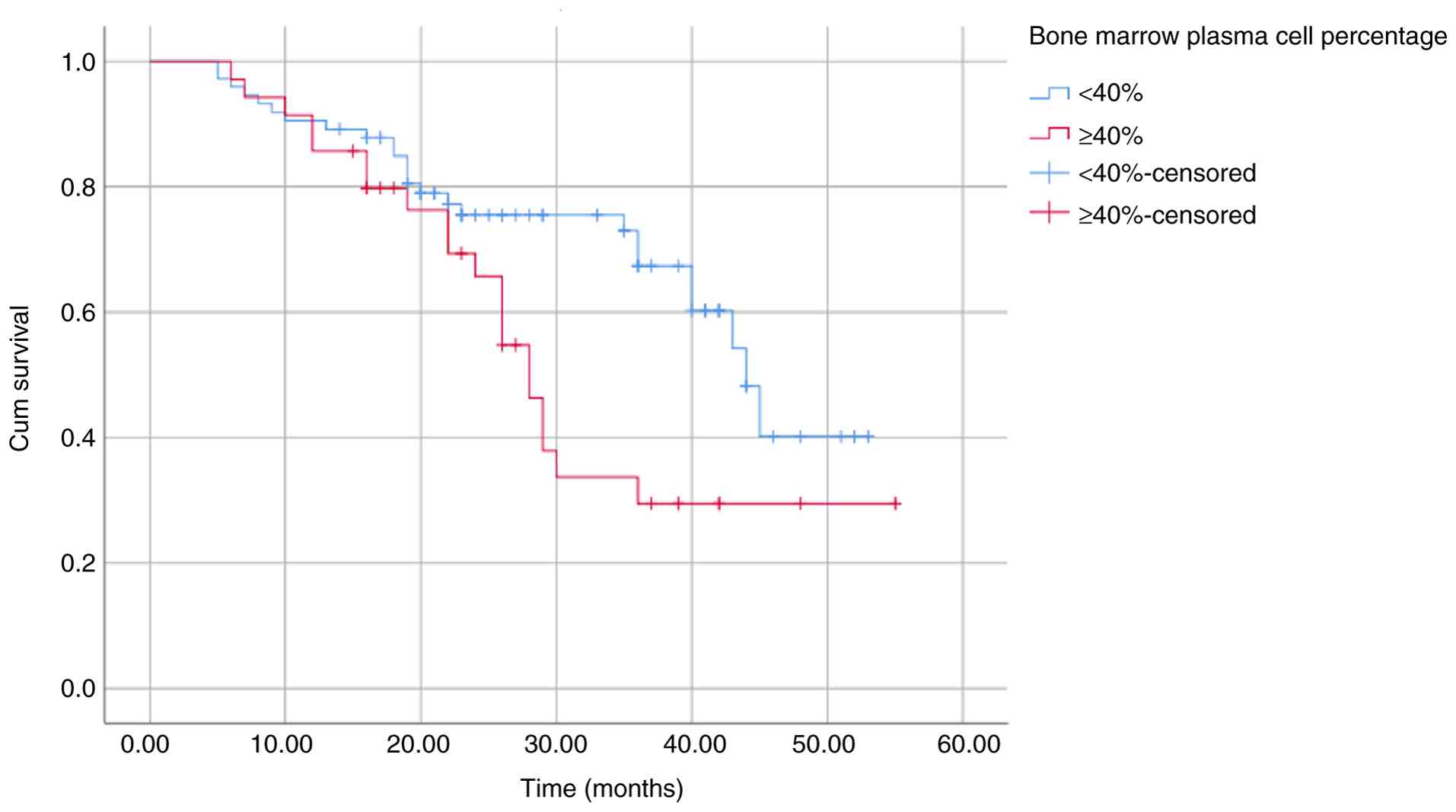
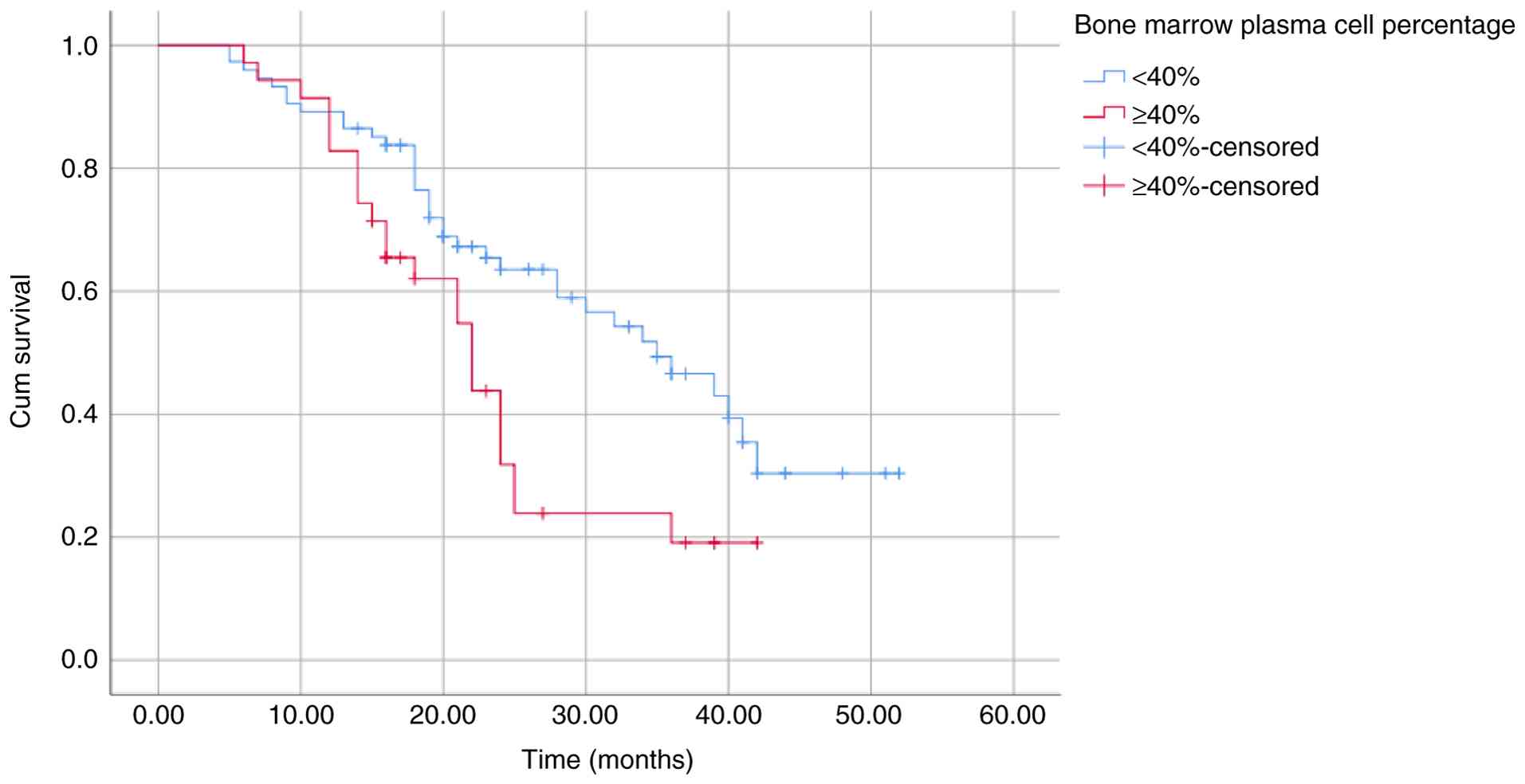
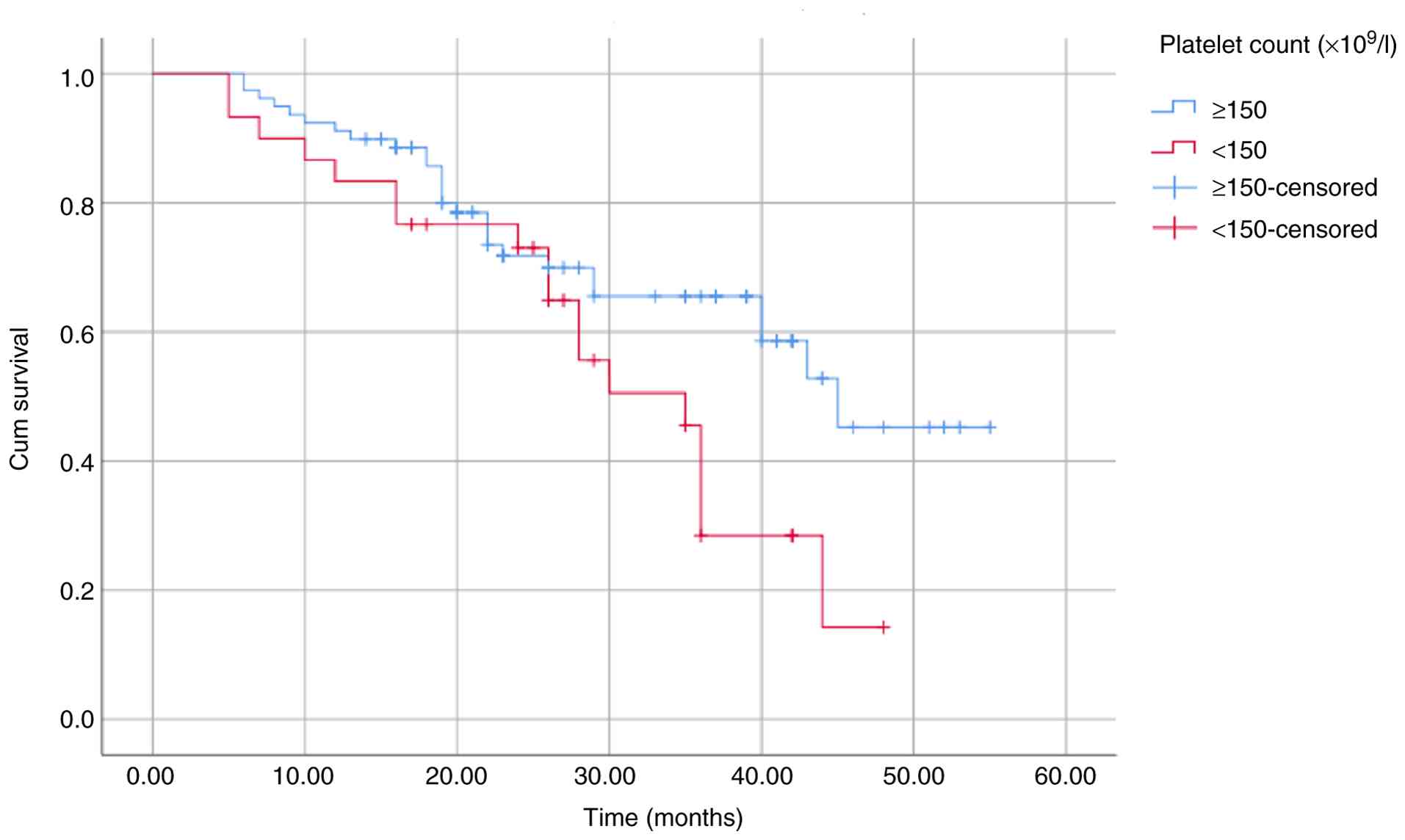
Among all variables examined, only a high bone marrow plasma cell
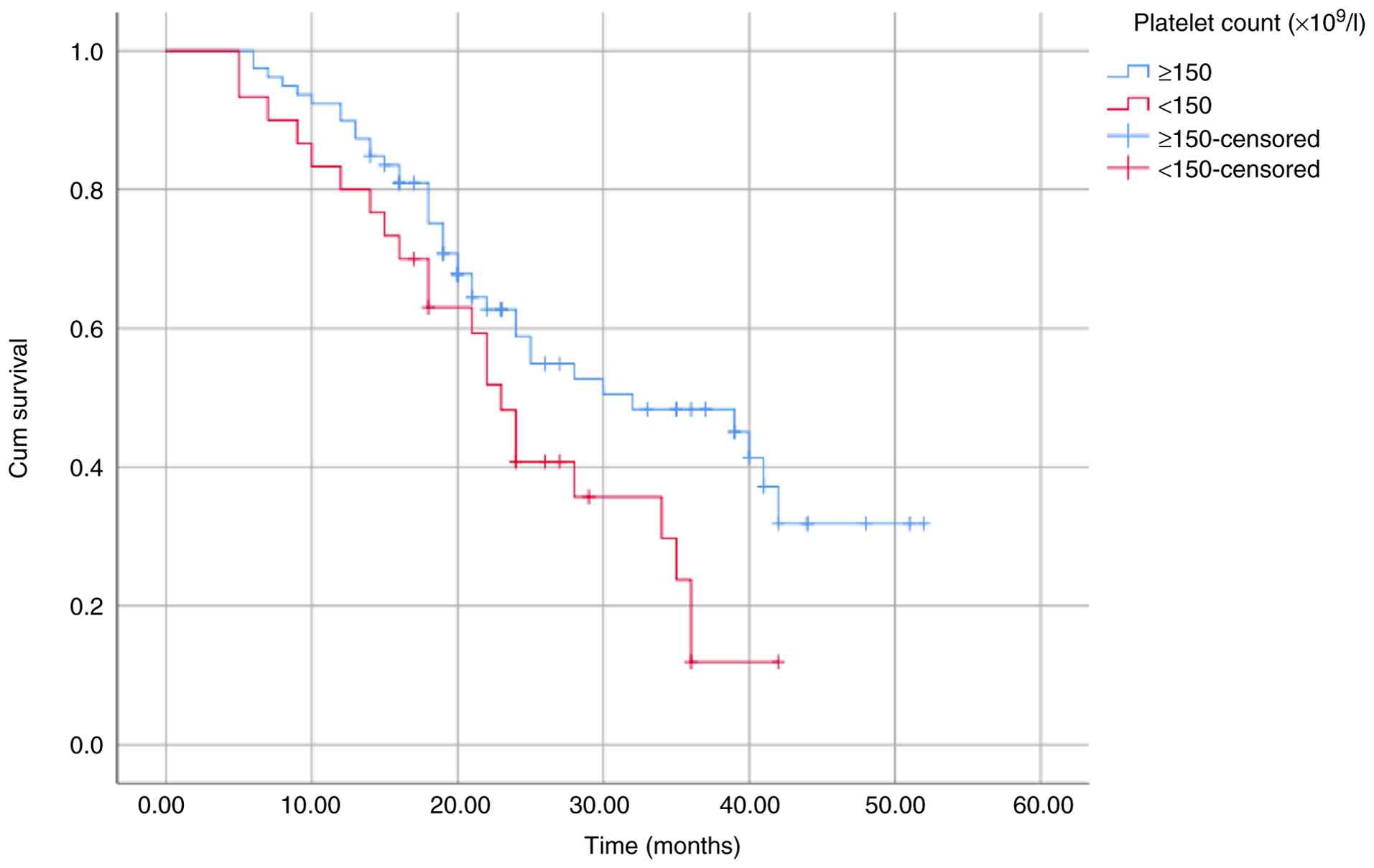
percentage (≥40%), low platelet count (<150x109/l),
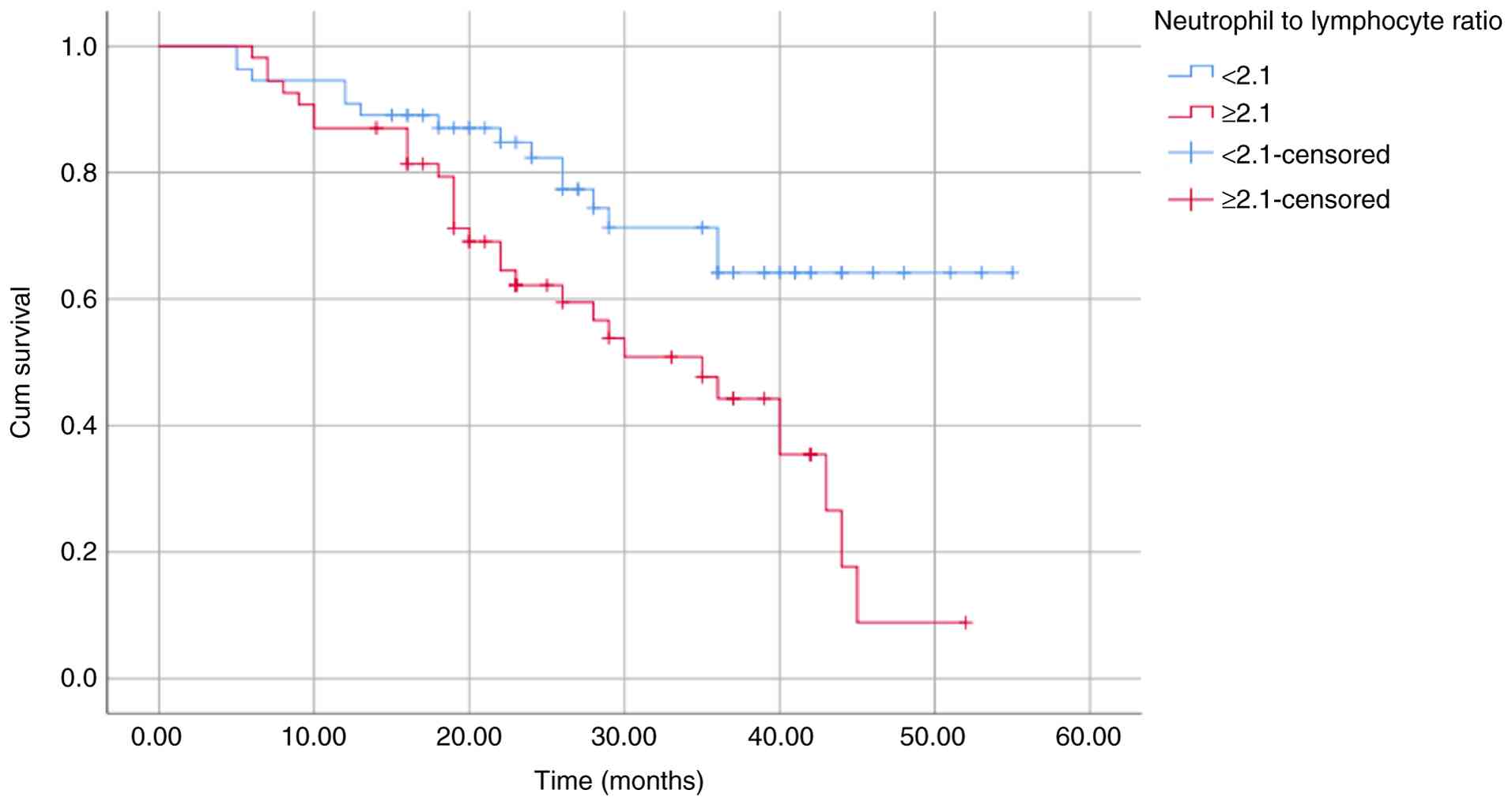
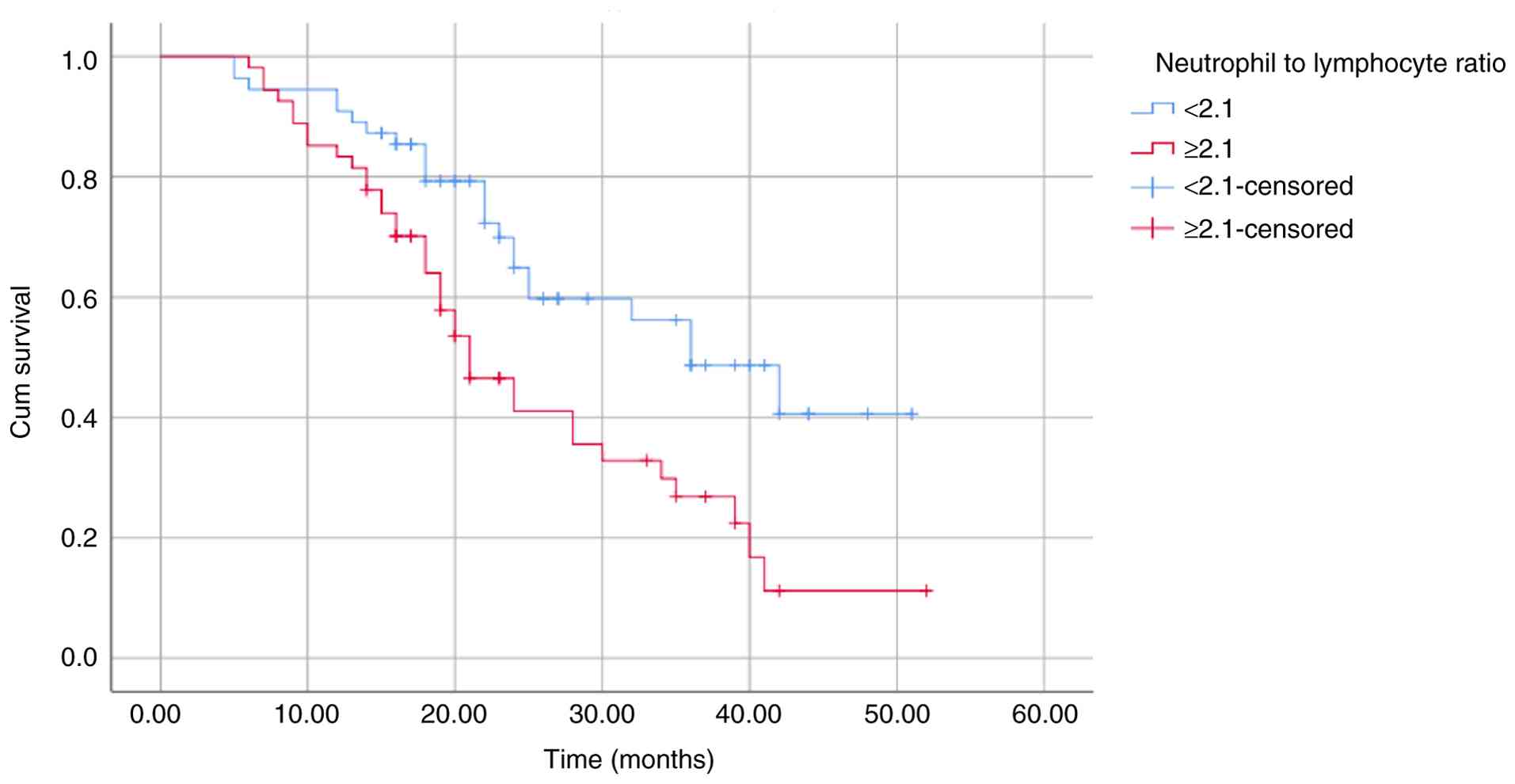
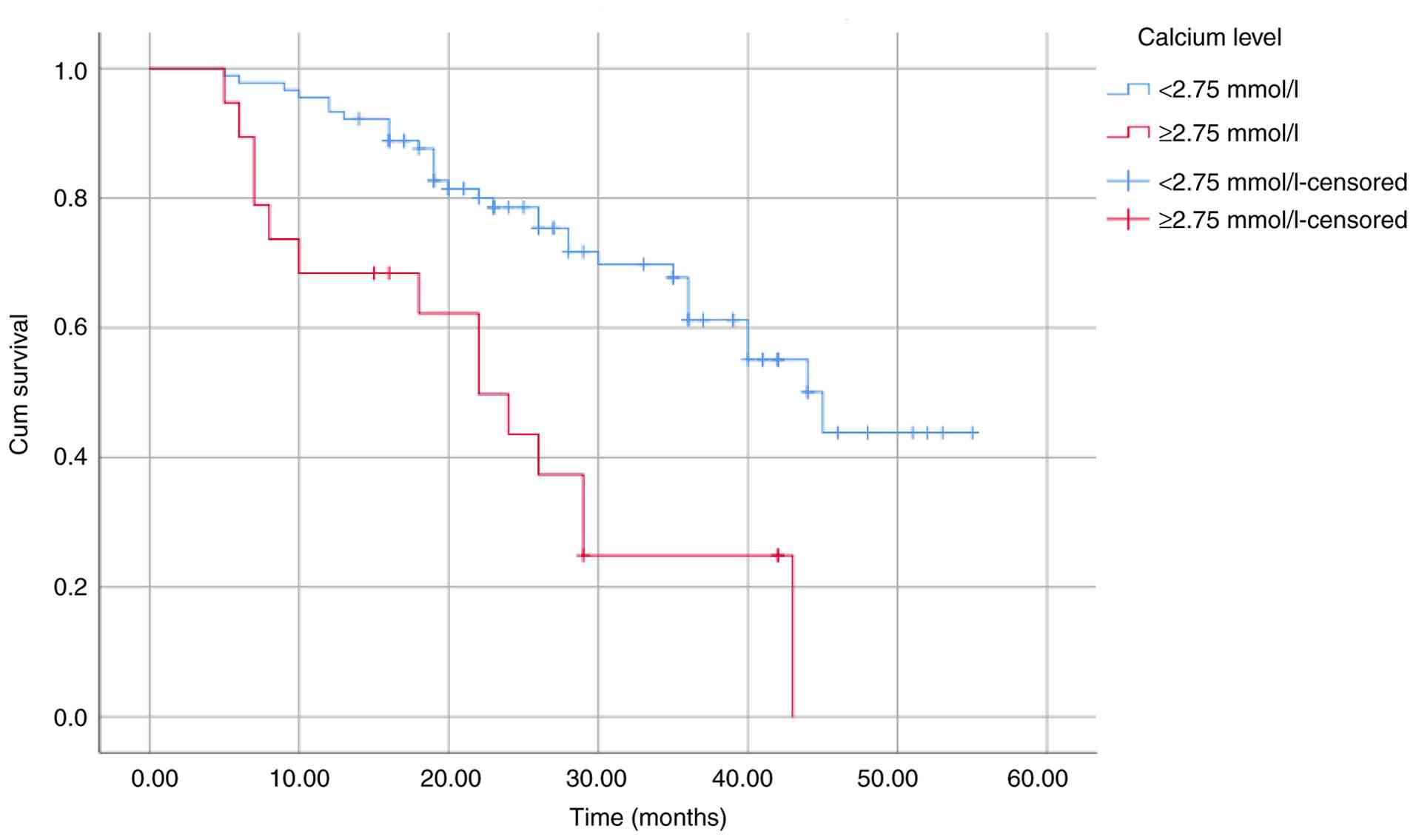
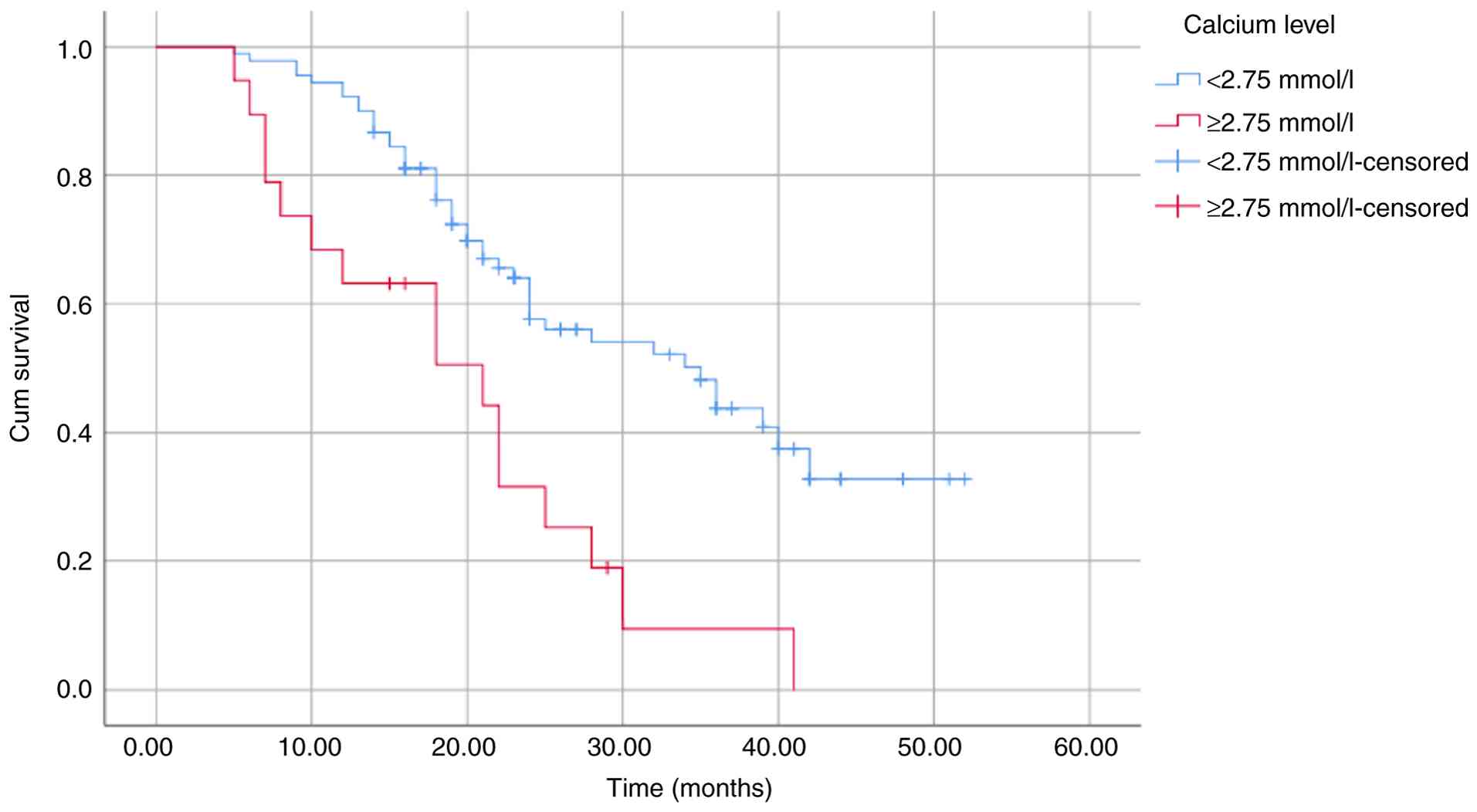
high NLR (≥2.1) and a high calcium level (≥2.75 mmol/l) were
factors for a poor prognosis and significantly associated with both
OS and PFS (Table III). These
differences were further illustrated using Kaplan-Meier survival
curves (Fig. 2, Fig. 3, Fig.
4, Fig. 5, Fig. 6, Fig.
7, Fig. 8 and Fig. 9).
 | Table IIIPrognostic factors used in univariate
survival analysis. |
Table III
Prognostic factors used in univariate
survival analysis.
| | OS | PFS |
|---|
| Factor | Months | P-value (log-rank
test) | Months | P-value (log-rank
test) |
|---|
| Bone marrow plasma
cell percentage | | | | |
|
<40% | 39.41 | 0.016 | 33.38 | 0.011 |
|
≥40% | 32.01 | | 23.43 | |
| Platelet count
(x109/l) | | | | |
|
≥150 | 39.81 | 0.035 | 32.84 | 0.028 |
|
<150 | 30.47 | | 23.86 | |
| NLR | | | | |
|
<2.1 | 43.30 | 0.003 | 35.14 | 0.003 |
|
≥2.1 | 31.21 | | 25.83 | |
| Calcium level
(mmol/l) | | | | |
|
<2.75 | 40.41 | <0.001 | 33.26 | <0.001 |
|
≥2.75 | 23.54 | | 19.35 | |
| β2M level
(mg/l) | | | | |
|
<5.5 | 43.73 | 0.005 | 34.55 | >0.05 |
|
≥5.5 | 33.85 | | 28.54 | |
| LDH level
(U/l) | | | | |
|
Normal | 40.19 | 0.021 | 31.42 | >0.05 |
|
Elevated | 30.51 | | 26.10 | |
| ECOG PS | | | | |
|
≤2 | 45.62 | 0.006 | 35.13 | >0.05 |
|
>2 | 32.89 | | 28.45 | |
| ISS | | | | |
|
ISS I | 49.75 | 0.014 | 34.19 | >0.05 |
|
ISS II | 41.18 | | 34.33 | |
|
ISS III | 33.85 | | 28.54 | |
| Hemoglobin level
(g/l) | | | | |
|
≥100 | 39.63 | >0.05 | 29.21 | >0.05 |
|
<100 | 36.31 | | 30.14 | |
| Creatinine level
(µmol/l) | | | | |
|
<177 | 37.92 | >0.05 | 30.99 | >0.05 |
|
≥177 | 36.12 | | 28.83 | |
| Albumin level
(g/l) | | | | |
|
≥35 | 38.85 | >0.05 | 31.31 | >0.05 |
|
<35 | 35.50 | | 30.01 | |
Multivariate analysis demonstrated that a high bone
marrow plasma cell percentage (≥40%), low platelet count
(<150x109/l), high NLR (≥2.1) and a high calcium
level (≥2.75 mmol/l) were independent adverse prognostic factors
for both OS and PFS in patients with MM (Table IV). In the present study, ISS
stage, β2M ≥5.5 mg/l, elevated LDH level and ECOG PS ≥2 were only
significant for OS, not for PFS; therefore, they were not used in
the multivariate analysis.
 | Table IVPrognostic factors used in
multivariate survival analysis. |
Table IV
Prognostic factors used in
multivariate survival analysis.
| A, OS |
|---|
| Factor | β | B | SE | Wald | P-value (Cox
analysis) | HR | 95% CI |
|---|
| High bone marrow
plasma cell percentage (≥40%) | 0.065 | 0.642 | 0.312 | 4.243 | 0.039 | 1.900 | 1.032-3.501 |
| Low platelet count
(<150x109/l) | 0.039 | 0.609 | 0.310 | 3.871 | 0.049 | 1.839 | 1.002-3.373 |
| High NLR
(≥2.1) | 0.141 | 0.958 | 0.326 | 8.652 | 0.003 | 2.605 | 1.376-4.931 |
| High calcium level
(≥2.75 mmol/l) | 0.212 | 0.980 | 0.328 | 8.959 | 0.003 | 2.665 | 1.403-5.065 |
| B, PFS |
| Factor | β | B | SE | Wald | P-value (Cox
analysis) | HR | 95% CI |
| High bone marrow
plasma cell percentage (≥40%) | 0.149 | 0.543 | 0.270 | 4.040 | 0.044 | 1.721 | 1.014-2.922 |
| Low platelet count
(<150 x109/l) | 0.099 | 0.625 | 0.280 | 4.982 | 0.026 | 1.867 | 1.079-3.231 |
| High NLR
(≥2.1) | 0.186 | 0.844 | 0.273 | 9.590 | 0.002 | 2.326 | 1.363-3.970 |
| High calcium level
(≥2.75 mmol/l) | 0.207 | 0.856 | 0.296 | 8.358 | 0.004 | 2.354 | 1.318-4.207 |
In the OS analysis, the HRs for high bone marrow
plasma cell percentage (≥40%), low platelet count
(<150x109/l), high NLR (≥2.1) and a high calcium
level (≥2.75 mmol/l) were 1.900, 1.839, 2.605 and 2.665,
respectively, with corresponding P-values of 0.039, 0.049, 0.003
and 0.003. The absolute value of standardized coefficient (β)
values for OS were 0.065, 0.039, 0.141 and 0.212, respectively.
These findings indicated that the prognostic impact of a high NLR
(≥2.1) on OS was greater than that of high bone marrow plasma cell
percentage (≥40%) and low platelet count
(<150x109/l), but lower than that of high calcium
levels (≥2.75 mmol/l; Table
IV).
In addition, the HRs for a high bone marrow plasma
cell percentage (≥40%), low platelet count
(<150x109/l), high NLR (≥2.1) and a high calcium
level (≥2.75 mmol/l) in the PFS analysis were 1.721, 1.876, 2.326
and 2.354, respectively, with corresponding P-values of 0.044,
0.026, 0.002 and 0.004. The absolute values of standardized
coefficient β (for PFS) were 0.149, 0.099, 0.186 and 0.207,
respectively. These results further demonstrated that the
prognostic value of a high NLR (≥2.1) on PFS exceeded that of high
bone marrow plasma cell percentage (≥40%) and a low platelet count
(<150x109/l); however, the values were lower than
those of high calcium levels (≥2.75 mmol/l; Table IV).
Discussion
Kelkitli et al (16) initially documented an association
between NLR and survival rates in patients with MM. Since then,
several studies have supported this finding (17-27),
although a small number have reported contradictory results
(32,33). These studies were based on the
hypothesis that an imbalance in immune responses within the bone
marrow microenvironment in MM supports plasma cell proliferation.
T-cells play a crucial role in immune surveillance; therefore,
increased T-cell infiltration within the bone marrow
microenvironment at tumor sites is considered a favorable
prognostic indicator. By contrast, interactions between malignant
plasma cells and the bone marrow microenvironment enable immune
evasion. Plasma cells secrete chemokines, such as IL-6, which
stimulate neutrophil mobilization and recruitment. Neutrophils can
suppress T-cell function, thereby exerting an adverse effect on
survival. Consequently, an elevated NLR provides indirect evidence
of impaired T-cell recruitment to tumor sites and suppression of
antitumor immune activity, creating a microenvironment conducive to
plasma cell proliferation and dissemination (17,18,36,37).
Although Zhou et al (32) and Avagyan et al (33) reported no association between a
high NLR and survival rates in MM, numerous other studies have
suggested that NLR may serve as a prognostic biomarker (16-27).
Notably, the two studies (32,33)
reporting negative findings were conducted with relatively small
sample sizes of 76 and 54 patients, respectively, which may have
limited their statistical power. Collectively, these studies
reinforce the hypothesis that immune dysregulation in MM markedly
affects patient survival.
In the present study, a high NLR was defined using a
cut-off value of 2.1, whereas previously reported cut-off values
ranged from 1.72 to 4 (16-27).
To facilitate clinical application, the standardization of the NLR
cut-off value is required (38).
In addition, several influential studies, such as those by Abdallah
et al (28), Kumar et
al (29,31) and Ferri et al (30) did not include NLR alongside other
prognostic factors in MM, such as low platelet count and high bone
marrow plasma cell percentage. As a result, the prognostic value of
NLR may have been underestimated relative to these variables. By
contrast, the present study demonstrated that the HR and absolute
value of standardized coefficient β associated with a high NLR
exceeded those of low platelet count and high bone marrow plasma
cell percentage, through Cox proportional hazards analysis for both
OS and PFS. In the OS analysis, the HRs for high NLR, low platelet
count and high bone marrow plasma cell percentage were 2.605, 1.839
and 1.900, respectively, with corresponding absolute values of
standardized coefficient (β) for OS of 0.141, 0.039 and 0.065. In
the PFS analysis, the HRs were 2.326, 1.876 and 1.721,
respectively, with absolute values of standardized coefficient β of
0.186. 0.099 and 0.149. These findings indicated that a high NLR
had a greater prognostic significance than a low platelet count and
high bone marrow plasma cell percentage in patients with MM.
The association between thrombopoiesis and MM is
complex. On one hand, malignant plasma cells secrete cytokines,
such as IL-6 and VEGF, which promote thrombocytosis. On the other
hand, monoclonal immunoglobulins secreted by plasma cells exert
inhibitory effects on platelet production. The balance between
these opposing mechanisms complicates interpretation of the
association between platelet count and disease progression,
although clinical evidence consistently indicates that low platelet
count is a poor prognostic factor in MM (37-39).
Bone marrow plasma cell percentage reflects disease burden;
however, the findings of the present study suggest that its
prognostic value is inferior to that of NLR. Overall, MM
progression results from dynamic interactions between malignant
plasma cells and the bone marrow microenvironment, in which
inflammatory mediators play a pivotal role. Inflammatory cytokines,
such as IL-6, TNFα and other mediators, promote cell adhesion,
angiogenesis and may impair treatment efficacy, rendering
inflammatory markers, such as NLR, valuable indicators of poor
prognosis.
The results of the present study also demonstrated
that a high NLR had a lower prognostic impact than elevated calcium
levels in MM. For OS, the HRs were 2.605 compared with 2.665, and
the absolute values of standardized coefficients β were 0.141
compared with 0.212 for high NLR and high calcium, respectively.
For PFS, the HRs were 2.326 compared with 2.354, and the absolute
values of standardized coefficients β were 0.186 compared with
0.207, respectively. In MM, plasma cell-derived cytokines stimulate
osteoclast activation, leading to bone resorption and calcium
release. Concurrent renal impairment reduces calcium clearance,
contributing to hypercalcemia (40). Hypercalcemia is therefore directly
associated with MM progression, whereas NLR represents an indirect
marker reflecting immune dysregulation, the mechanisms of which
require further clarification.
Notably, the present study exhibits several
limitations. As a retrospective cohort study, it is subject to
selection and information bias. In addition, due to financial
constraints, comprehensive cytogenetic evaluation using FISH was
not performed, precluding the analysis of the association between
NLR and R-ISS or R2-ISS. Future studies incorporating complete
cytogenetic data are required to enable the more objective
evaluation of the prognostic role of NLR. The association between
NRL and chromosomal abnormalities of multiple myeloma, as detected
using FISH techniques and staging classifications, such as R-ISS or
R2-ISS, is an exciting area of research. Zuo et al (41) demonstrated that a NLR ≥2 was
strongly associated with R-ISS. However, their study also
demonstrated that there were no significant differences between the
value of NLR and the presence of 1q21 amplification, therefore,
there was also no association between NLR and R2-ISS (41). Thus, the association between NLR
and the presence of chromosomal abnormalities, as well as staging
classifications based on chromosomal abnormalities, such as R-ISS
or R2-ISS, is a worth considering. The authors hope to further
develop the study, exploring the association between NLR and
chromosomal abnormalities of multiple myeloma, as well as with
R-ISS or R2-ISS. The present study was conducted in Vietnam, a
developing country, where limited drug availability necessitated
use of multiple treatment regimens, including VTD, VCD and VRD,
resulting in heterogeneous treatment outcomes. Consequently,
adjustment for treatment regimens in prognostic analyses was
limited. Larger prospective studies stratified by treatment
protocol are required to address these issues.
In conclusion, a high NLR is a valuable adverse
prognostic factor in MM and appears to have greater prognostic
significance than a low platelet count and bone marrow plasma cell
percentage. However, further large-scale studies are required to
determine the most appropriate and standardized NLR cut-off value
for routine clinical application.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
MPV conceived the study. NDN, MPV, HV, PTP and TTN
designed the study. HV, PTP, TVOK, HYD, TTMN and THH participated
in data collection and processing. NDN, MPV, HV and PTP
participated in data analysis and interpretation. All authors
participated in the literature search in the writing of the
manuscript. All authors have read and approved the final
manuscript. HV and PTP confirm the authenticity of the raw
data.
Ethics approval and consent to
participate
The study protocol was approved by The Institutional
Review Board of Bach Mai hospital (no. 7219/QĐ-BM: date 31 December
2024). All patients were informed and consented to participate in
this study. Patients, their guardians or family members were called
to request consent.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that have no competing
interests.
References
|
1
|
Abduh MS: An overview of multiple myeloma:
A monoclonal plasma cell malignancy's diagnosis, management, and
treatment modalities. Saudi J Biol Sci. 31(103920)2024.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Forster S, Radpour R and Ochsenbein AF:
Molecular and immunological mechanisms of clonal evolution in
multiple myeloma. Front Immunol. 14(1243997)2023.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Giannakoulas N, Ntanasis-Stathopoulos I
and Terpos E: The role of marrow microenvironment in the growth and
development of malignant plasma cells in multiple myeloma. Int J
Mol Sci. 22(4462)2021.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Wang L, Shi M, Sung AY, Yin CC, Bai Y and
Chen M: Role of the bone marrow microenvironment in multiple
myeloma: Impact of niches on drug resistance mechanisms. Semin
Diagn Pathol. 42(150916)2025.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Pinto V, Bergantim R, Caires HR, Seca H,
Guimarães JE and Vasconcelos MH: Multiple myeloma: Available
therapies and causes of drug resistance. Cancers (Basel).
12(407)2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Nass J and Efferth T: Drug targets and
resistance mechanisms in multiple myeloma. Cancer Drug Resist.
1:87–117. 2018.
|
|
7
|
Corre J, Munshi NC and Avet-Loiseau H:
Risk factors in multiple myeloma: Is it time for a revision? Blood.
137:16–19. 2021.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Wallington-Beddoe CT and Mynott RL:
Prognostic and predictive biomarker developments in multiple
myeloma. J Hematol Oncol. 14(151)2021.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Hanbali A, Hassanein M, Rasheed W, Aljurf
M and Alsharif F: The evolution of prognostic factors in multiple
myeloma. Adv Hematol. 2017(4812637)2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Greipp PR, San Miguel J, Durie BG, Crowley
JJ, Barlogie B, Bladé J, Boccadoro M, Child JA, Avet-Loiseau H,
Kyle RA, et al: International staging system for multiple myeloma.
J Clin Oncol. 23:3412–3420. 2005.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Romano A, Laura Parrinello N, Cerchione C,
Letizia Consoli M, Parisi M, Calafiore V, Martino E, Conticello C,
Di Raimondo F and Alberto Palumbo G: The NLR and LMR ratio in newly
diagnosed MM patients treated upfront with novel agents. Blood
Cancer Journal. 7(649)2017.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Kaddoura M, Binder M, Dingli D, Buadi FK,
Lacy MQ, Gertz MA, Dispenzieri A, Kapoor P, Hwa L, Fonder A, et al:
Impact of achieving a complete response to initial therapy of
multiple myeloma and predictors of subsequent outcome. Am J
Hematol. 97:267–273. 2022.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Palumbo A, Avet-Loiseau H, Oliva S,
Lokhorst HM, Goldschmidt H, Rosinol L, Richardson P, Caltagirone S,
Lahuerta JJ, Facon T, et al: Revised international staging system
for multiple myeloma: A report from international myeloma working
group. J Clin Oncol. 33:2863–2869. 2015.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Rajkumar SV: Multiple myeloma: 2024 update
on diagnosis, risk-stratification, and management. Am J Hematol.
99:1802–1824. 2024.PubMed/NCBI View Article : Google Scholar
|
|
15
|
D'Agostino M, Cairns DA, Lahuerta JJ,
Wester R, Bertsch U, Waage A, Zamagni E, Mateos MV, Dall'Olio D,
van de Donk NWCJ, et al: Second revision of the international
staging system (R2-ISS) for overall survival in multiple myeloma: A
European myeloma network (EMN) report within the HARMONY project. J
Clin Oncol. 40:3406–3418. 2022.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Kelkitli E, Atay H, Cilingir F, Güler N,
Terzi Y, Ozatlı D and Turgut M: Predicting survival for multiple
myeloma patients using baseline neutrophil/lymphocyte ratio. Ann
Hematol. 93:841–846. 2014.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Wongrakpanich S, George G, Chaiwatcharayut
W, Biso S, Candelario N, Mittal V, Pomerantz S and Varadi G: The
prognostic significance of neutrophil-to-lymphocyte and
platelet-to-lymphocyte ratios in patients with multiple myeloma. J
Clin Lab Anal. 30:1208–1213. 2016.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Onec B, Okutan H, Albayrak M, Saribacak
Can E, Aslan V, Unver Koluman B, Soyer Kosemehmetoglu O, Albayrak A
and Kos DM: The Predictive role of the neutrophil/lymphocyte ratio
in survival with multiple myeloma: A single center experience. J
Clin Lab Anal. 31(e22032)2017.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Mu S, Ai L, Fan F, Sun C and Hu Y:
Prognostic role of neutrophil-lymphocyte ratio in multiple myeloma:
A dose-response meta-analysis. Onco Targets Ther. 11:499–507.
2018.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Zeng Q, Liu Z, Li Q and Liu T: Prognostic
value of neutrophil to lymphocyte ratio and clinicopathological
characteristics for multiple myeloma: A meta-analysis. Medicine
(Baltimore). 97(e12678)2018.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Szudy-Szczyrek A, Mlak R, Mielnik M,
Szczyrek M, Nowaczyńska A, Homa-Mlak I, Zmorzyński S, Kuśmierczuk
K, Sompor J, Filip A, et al: Prognostic value of pretreatment
neutrophil-to-lymphocyte and platelet-to-lymphocyte ratios in
multiple myeloma patients treated with thalidomide-based regimen.
Ann Hematol. 99:2881–2891. 2020.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Zhang X, Duan J, Wen Z, Xiong H, Chen X,
Liu Y, Liao K and Huang C: Are the derived indexes of peripheral
whole blood cell counts (NLR, PLR, LMR/MLR) clinically significant
prognostic biomarkers in multiple myeloma? A systematic review and
meta-analysis. Front Oncol. 11(766672)2021.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Giri S, Dahal S, Bal S, Godby KN, Richman
J, Olszewski AJ, Williams GR, Brown C, Buford TW, Costa LJ and
Bhatia S: Pre-treatment neutrophil to lymphocyte ratio as a
biomarker of frailty and predictor of survival among older adults
with multiple myeloma. J Geriatr Oncol. 13:486–492. 2022.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Mikulski D, Kościelny K, Nowicki M,
Wawrzyniak E, Kalwas M, Kowalik M, Pryt M, Sęczkowska E, Świątek A,
Wierzbowska A and Fendler W: Neutrophil to lymphocyte ratio (NLR)
impact on the progression-free survival and overall survival of
multiple myeloma patients treated with high-dose chemotherapy and
autologous stem cell transplantation. Leuk Lymphoma. 64:98–106.
2023.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Mehra K, Ramasamy C, Perkit NR, Singh A,
Kumar MS and Seetharaman K: Significance of
neutrophil-to-lymphocyte ratio, platelet-to-lymphocyte ratio and
monocyte-to-lymphocyte ratio in the prognosis of newly diagnosed
multiple myeloma patients. Blood. 142 (Suppl 1)(S6695)2023.
|
|
26
|
Zhang Y, Yao X, Zhang Y, Chen Z, Qin Z,
Cai Y, Xia W and Hu H: Albumin-to-globulin ratio combined with
neutrophil-to-lymphocyte ratio as a prognostic predictor in
multiple myeloma with renal impairment. Blood Lymphat Cancer.
14:49–62. 2024.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Wang M, Yue X, Ding Y, Cai Z, Xiao H,
Huang H and He J: A nomogram based on circulating inflammatory
factors for predicting prognosis of newly diagnosed multiple
myeloma patients. J Inflamm Res. 18:2077–2090. 2025.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Abdallah NH, Smith AN, Geyer S, Binder M,
Greipp PT, Kapoor P, Dispenzieri A, Gertz MA, Baughn LB, Lacy MQ,
et al: Conditional survival in multiple myeloma and impact of
prognostic factors over time. Blood Cancer J. 13(78)2023.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Kumar S, Leleu X, Weisel K, Popat R,
Craigie S, Patel L, Ramirez AO, Ge W, MA Q, Hampp C and Jagannath
S: PB2090: Systematic literature review of prognostic factors for
relapsed/refractory multiple myeloma. Hemasphere. 7
(Suppl)(e9426734)2023.
|
|
30
|
Ferri GM, Yildirim C, Park J, Do VN,
Brophy MT, Munshi NC, Fillmore NR and Edwards CV: Development of
novel peripheral blood count-based prognostic index for multiple
myeloma. Blood. 144 (Suppl 1)(S6981)2024.
|
|
31
|
Kumar S, Leleu X, Weisel KC, Popat R,
Suero B, Craigie S, Spin P, Patel L, Ramirez AO, Hampp C, et al:
Ranking the importance of prognostic factors for
relapsed/refractory multiple myeloma: international physician panel
consensus following a systematic literature review. Clin Lymphoma
Myeloma Leuk. 25:730–738.e11. 2025.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Zhou X, Wang J, Xia J, Cheng F, Mao J, Zhu
J and Guo H: Evaluation of neutrophil-to-lymphocyte ratio in newly
diagnosed patients receiving borte-zomib-based therapy for multiple
myeloma. Cancer Biomark. 22:43–48. 2018.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Avagyan G, Voskanyan A, Grigoryan A,
Saaryan M, Muradyn L, Avagyan G and Ghahramanyan N: MM-469 The
prognostic value of serum calcium levels, neutrophil to lymphocyte
ratio and platelet count in multiple myeloma: A retrospective
cohort study. Clin Lymphoma Myeloma Leuk. 24 (Suppl
1)(S559)2024.
|
|
34
|
Azam F, Latif MF, Farooq A, Tirmazy SH,
AlShahrani S, Bashir S and Bukhari V: Performance status assessment
by using ECOG (eastern cooperative oncology group) score for cancer
patients by oncology healthcare professionals. Case Rep Oncol.
12:728–736. 2019.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Garderet L, D'Souza A, Jacobs P, van
Biezen A, Schönland S, Kroeger N, Morris C and Hari P: Response
assessment in myeloma: Practical manual on consistent reporting in
an era of dramatic therapeutic advances. Biol Blood Marrow
Transplant. 23:1193–1202. 2017.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Liu S, Shi J, Guo H, Xu F, Wei M, Sun K
and Chen Y: Prognostic significance of the inflammatory index-based
scoring system in patients preliminarily diagnosed with multiple
myeloma in the bortezomib-based chemotherapy era. Cancer Manag Res.
11:9409–9420. 2019.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Kalal AA, Shetty VV, Shetty KP, Arumugam
M, Shetty RA, Kulkarni NV and Shetty DP: Correlation between
platelet-to-lymphocyte ratio and neutrophil-to-lymphocyte ratio
with hematological parameters in multiple myeloma patients. Biomed
Biotechnol Res J. 6:132–137. 2022.
|
|
38
|
Zhaoyun L and Rong F: Predictive role of
immune profiling for survival of multiple myeloma patients. Front
Immunol. 12(663748)2021.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Diaconescu D, Ilinescu AM, Popescu CD,
Soare DB, Ene G and Bumbea H: Prognostic significance of neutrophil
to lymphocyte ratio in multiple myeloma patient. Doc Haematol Rev
Rom Hematol. 3:81–87. 2025.
|
|
40
|
LeBlanc R, Bergstrom DJ, Côté J, Kotb R,
Louzada ML and Sutherland HJ: Management of myeloma manifestations
and complications: The cornerstone of supportive care:
Recommendation of the Canadian myeloma research group (formerly
myeloma Canada research network) consensus guideline consortium.
Clin Lymphoma Myeloma Leuk. 22:e41–e56. 2022.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Zuo H, Zhai L, Liu X, Gao H and Xu P:
Prognostic significance of neutrophil-lymphocyte ratio in multiple
myeloma patients. Transl Cancer Res. 7:88–96. 2018.
|