Introduction
Invasive lobular carcinoma (ILC) is the second most
commonly occurring type of breast cancer, which comprises up to
~15% of breast cancer cases, with positive results for progesterone
receptor (PR) and estrogen receptor (ER), and typically has a good
prognosis and is low-grade (1).
There are multiple histological subtypes of ILC; however, they are
defined by a lack of cohesive growth due to the inactivation of the
cell-adhesion protein, E-cadherin (2,3). One
of these subtypes is pleomorphic lobular carcinoma (PLC), which is
clinically significant, although rare. This phenotype is associated
with a poorer prognosis and is more aggressive, resulting from
distinct histopathological characteristics. It may lack the
expression of PR and ER, and exhibit human epidermal growth factor
receptor 2 (HER2)/neu amplification, in contrast to ILC (4).
A post-menopausal status and an older age have been
linked to PLC, and it represents ~15% of ILC cases and <1% of
all breast cancers (4). It
exhibits distinct cytological characteristics that differ from
those of ILC, including higher-grade cytological features, a larger
tumor size, an increased likelihood of developing distant
metastases, the invasion of the lymphovascular system and a more
advanced stage at diagnosis. Distinct clinical behavior is observed
in lobular carcinomas, which tend to metastasize to serosal
surfaces, including the gastrointestinal tract, peritoneum, uterus
and ovaries (5). Breast cancer
fatalities are linked to their distant metastases, and classifying
breast cancer into molecular and histologic subtypes is recognized
for its essential prognostic and predictive significance (6).
The present study describes a rare case of invasive
PLC metastasis to the peritoneum and ovaries of a patient and also
performs a brief review of the literature.
Case report
Patient information
A 51-year-old woman presented to Smart Health Tower
(Sulaymaniyah, Iraq) in May, 2025 with a right breast mass that had
been noticed three weeks before presentation. She had a family
history of breast cancer involving her maternal aunt, maternal
uncle and paternal cousin. Her medical history was significant for
kidney disease, for which she had undergone three surgical
procedures for renal stone removal, as well as a prior diagnostic
laparoscopy for a peritoneal mass and a left thyroid lobectomy. Her
obstetric history revealed gravida 6, para 6, with no history of
abortion and a cumulative lactation period of 12 years.
Clinical findings
Upon a clinical examination, palpable masses were
detected in the right breast, including one in the lower inner
quadrant at the 4-5 o'clock position and another in the
retroareolar region at the 10 o'clock position. Both masses were
irregularly shaped, hard in consistency, and not fixed to the
underlying structures. They caused bulging and mild tethering of
the areola. Additionally, palpable right axillary lymph nodes were
noted, raising concerns about possible nodal involvement.
Diagnostic assessment
A right breast ultrasound revealed multiple
heterogeneous, irregular and hypoechoic masses mixed with fat
echogenicity. A lesion measuring 27x19 mm was identified in the
lower inner quadrant, while another mass at the 9 o'clock position,
located at an anterior-middle depth, measuring 40x14 mm and
extending to the nipple base. Additionally, multiple smaller
adjacent masses measuring <10 mm were observed in the upper
central part of the breast. The evaluation of the right axilla
revealed >10 abnormal, matted lymph nodes across all axillary
levels, with the largest node in level I, measuring 15 mm. No
supraclavicular lymphadenopathy was detected. The findings were
classified as BIRADS-5.
An abdominal ultrasound revealed bilateral renal
staghorn stones, suggesting a possible underlying metabolic
disorder, while the remainder of the examination did not reveal any
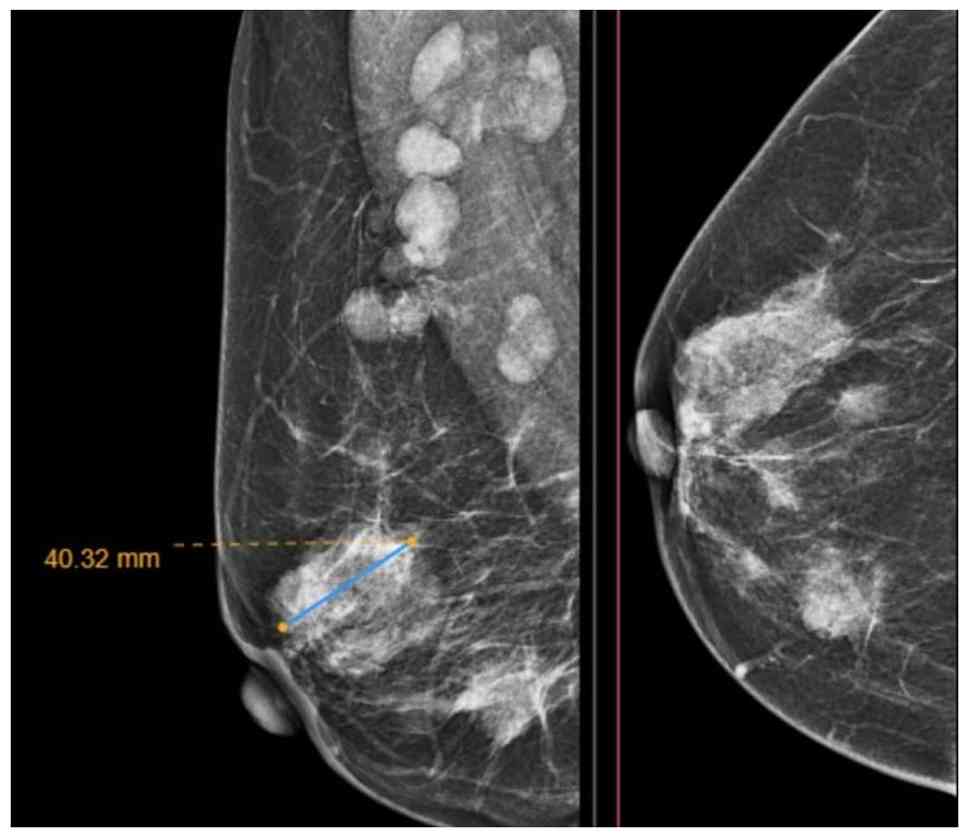
notable findings. A breast mammography revealed multiple hyperdense
masses with irregular margins throughout the right breast, with the
largest lesion in the central region, measuring 40x32 mm. No
microcalcifications were observed (Fig. 1). Right axillary lymph node
involvement was also noted, with a small number of matted abnormal
lymph nodes identified. The findings were classified as BIRADS-5.
Although the ultrasound examinations was performed at the
institution, the images were not available because they had not
been digitally archived in the radiology database at the time of
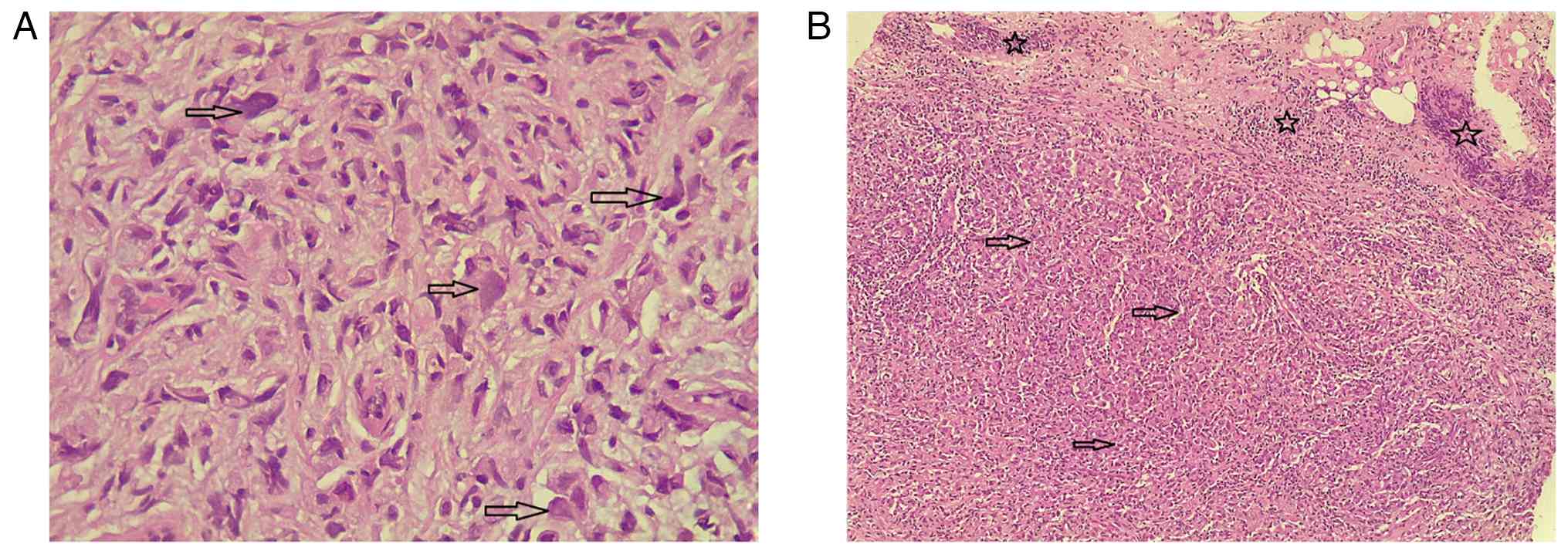
the examination. A core biopsy of the right breast mass and right
axillary lymph node confirmed invasive PLC, grade II (moderately
differentiated). The right axillary lymph node biopsy also revealed
metastatic carcinoma (Fig. 2).
Immunohistochemical analyses were performed on formalin-fixed,
paraffin-embedded tissue. Sections at a thickness of 4-6 µm were
prepared from paraffin blocks, mounted on charged slides and
incubated overnight at 60˚C. Heat-induced antigen retrieval was
carried out using the Dako PT Link system (Agilent Technologies,
Inc.) at 100˚C for 5-10 min, employing either citrate buffer (pH
6.0) or Tris-EDTA buffer (pH 9.0) according to the antibody used.
Following antigen retrieval, the slides were rinsed for 15 min at
room temperature in Tris-buffered saline containing 0.05% Tween-20
(0.05 mol/l Tris/HCl, 0.15 mol/l NaCl; pH 7.6). The tissue sections
were then demarcated using a hydrophobic pen, and endogenous
peroxidase activity was quenched with 3% hydrogen peroxide. Primary
antibodies directed against ER, PR, HER2, Ki-67 and E-cadherin were
applied using the following reagents: ER (rabbit monoclonal, clone
SP1; cat. no. RM-9101-S; Thermo Fisher Scientific, Inc.; dilution
1:100), PR (mouse monoclonal, clone PgR636; cat. no. M3569; Agilent
Technologies, Inc.; dilution 1:100), HER2 (rabbit monoclonal, clone
4B5; cat. no. 790-2991; Roche Diagnostics; ready-to-use), Ki-67
(mouse monoclonal, clone MIB-1; cat. no. M7240; Agilent
Technologies, Inc.; dilution 1:200) and E-cadherin (mouse
monoclonal, clone NCH-38; cat. no. M3612; Agilent Technologies,
Inc.; dilution 1:100). All antibodies were diluted in the
manufacturer's antibody diluent and incubated for 80 min at room
temperature (20-25˚C). Detection was achieved using a horseradish
peroxidase-linked secondary antibody (EnVision™ FLEX HRP polymer
detection system; cat. no. SM802; Agilent Technologies, Inc.;
ready-to-use, no dilution) and 3,3'-diaminobenzidine (DAB)
chromogen, each incubated for 15 min at room temperature (20-25˚C).
Counterstaining was performed with Gill II hematoxylin for 30 sec
at room temperature, after which the slides were dehydrated and
coverslipped. Immunostaining results demonstrated strong nuclear ER
expression with an Allred score of 8 (3 + 5) and PR positivity with
an Allred score of 4 (2 + 2). HER2 staining was negative (score 1),
and the Ki-67 labeling index was approximately 22%. The loss of
E-cadherin expression in tumor cells was observed, consistent with
ILC.
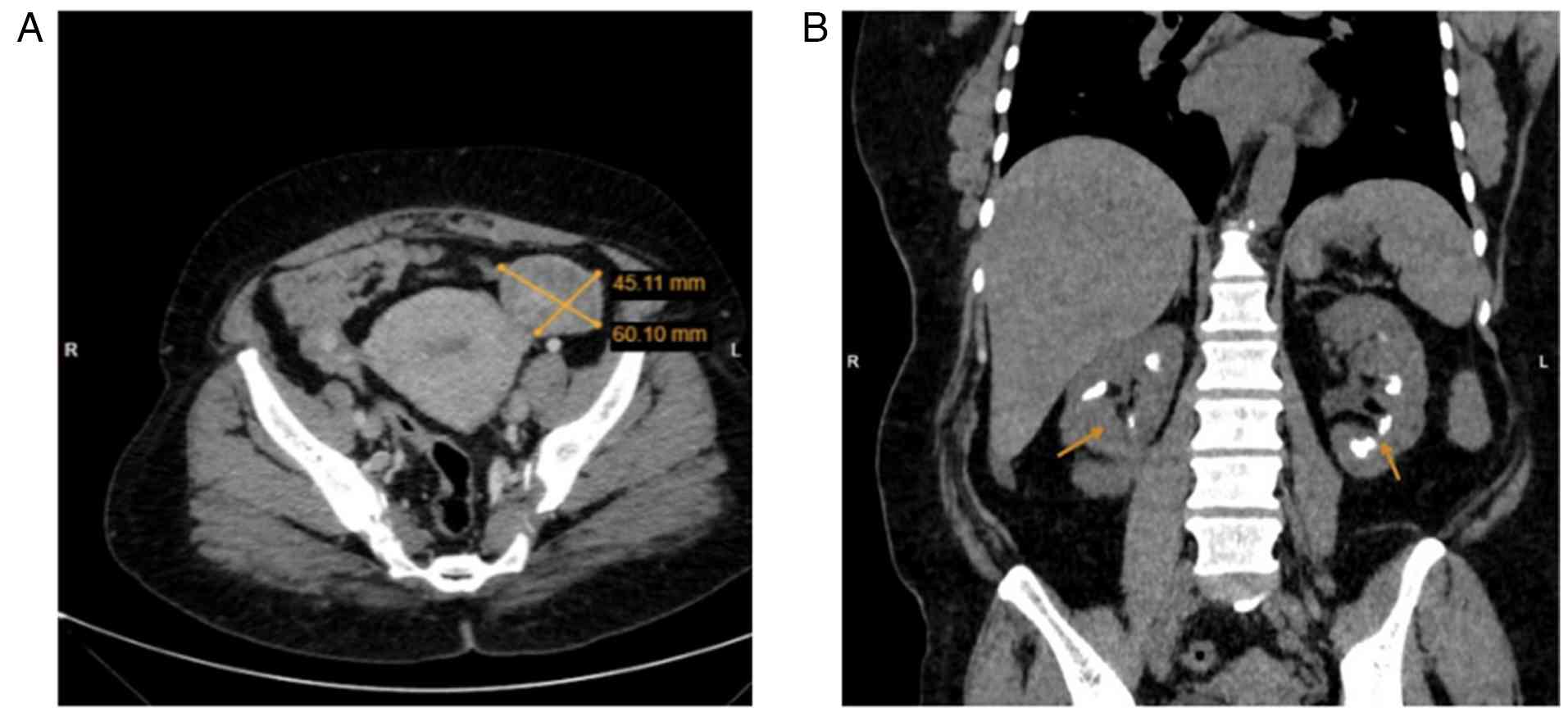
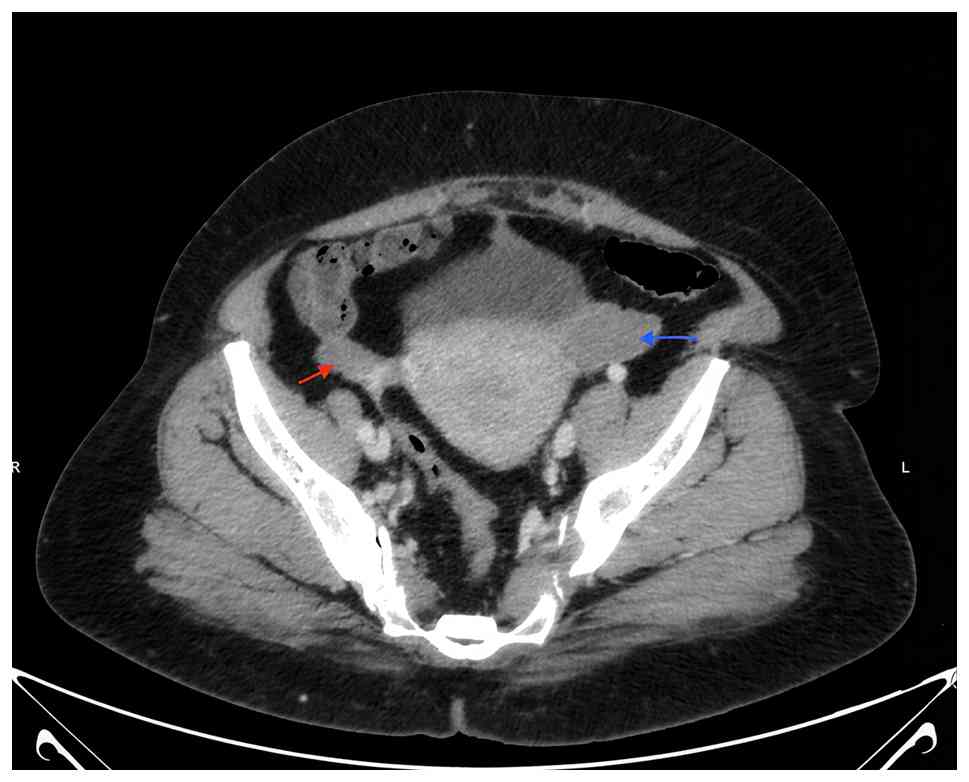
A contrast-enhanced computed tomography (CT) scan of
the chest, abdomen, and pelvis revealed two right breast masses,
the largest measuring 4 cm, with no evidence of chest wall
invasion. Pathological right axillary lymph nodes were present
across all levels, with the largest measuring 1.6 cm, while no
enlarged internal mammary lymph nodes were detected. Additionally,
bilateral adnexal masses were identified, raising the suspicion of
primary ovarian cancer or metastatic disease with associated
peritoneal deposits. Multiple bilateral renal calcifications were
noted in the medullary region, suggesting medullary
nephrocalcinosis (Fig. 3). To
further evaluate the ovarian and peritoneal lesions, a diagnostic
laparoscopy was performed, and biopsies were taken. A
histopathological analysis was then performed. Sections (5-µm-thick
tissue sections were fixed in 10% neutral-buffered formalin at room
temperature for 24 h and subsequently embedded in paraffin. The
sections were then stained with hematoxylin and eosin (Bio Optica
Co.) for 1-2 min at room temperature and examined under a light
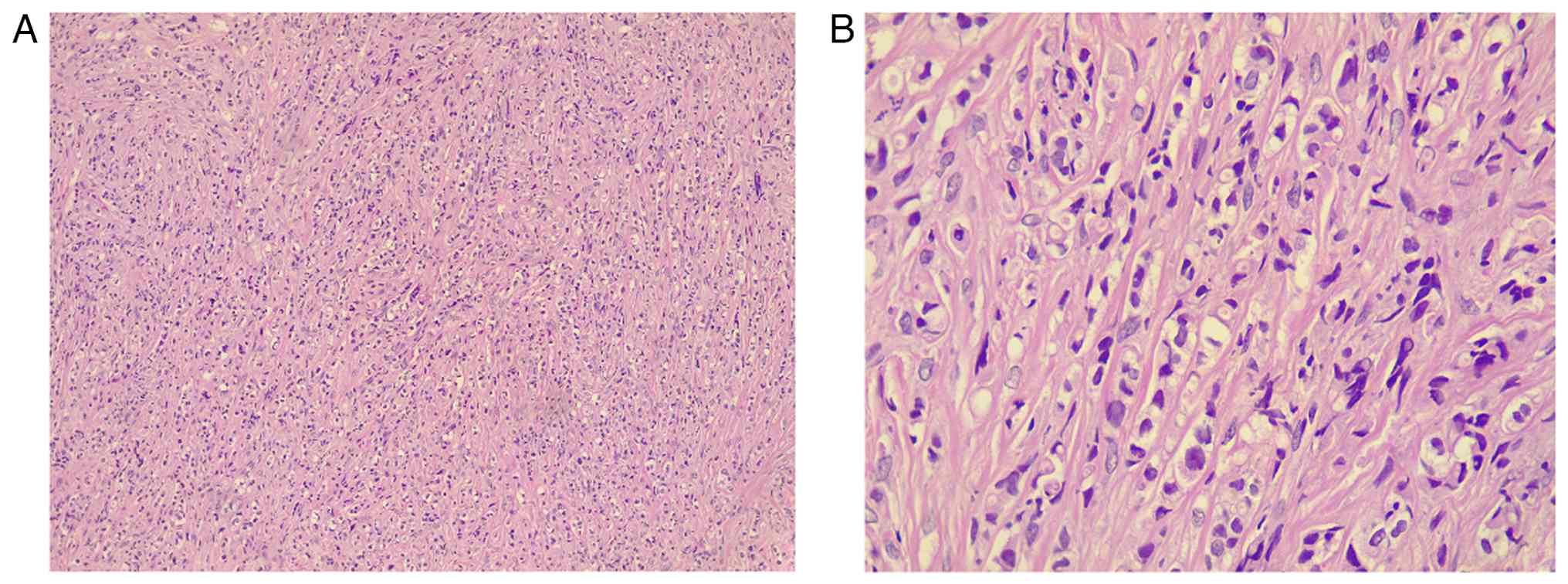
microscope (Leica Microsystems GmbH). The histopathological
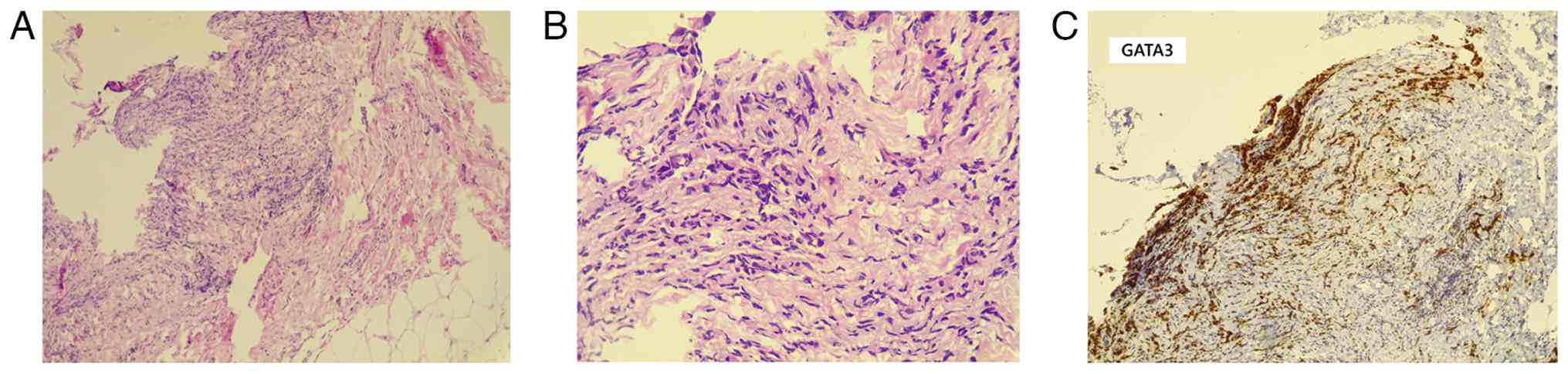
examination revealed extensive infiltration by dyscohesive sheets
and cords of malignant epithelial cells embedded within a
desmoplastic stroma. The tumor cells exhibited marked nuclear
pleomorphism, hyperchromatic nuclei, prominent nucleoli, and
frequent mitotic figures. Occasional intracytoplasmic vacuoles
imparted a signet-ring-like appearance. The peritoneal deposits
exhibited infiltrative epithelial cells associated with stromal
desmoplasia and focal crush artifact, supporting metastatic
involvement (Fig. 4). The findings
of IHC were consistent with metastatic PLC of the breast. The
cytological analysis of the peritoneal fluid was positive for
malignancy, confirming metastatic disease. IHC staining of the
ovarian and peritoneal tumor cells revealed positivity for
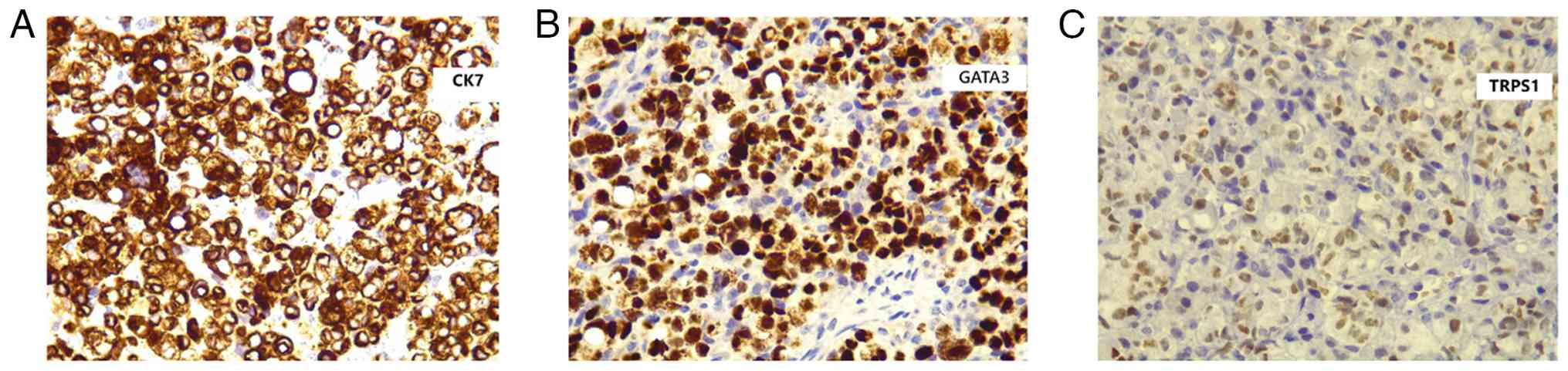
cytokeratin 7 (CK7), GATA-binding protein 3 (GATA3) and
trichorhinophalangeal syndrome type 1 (TRPS1), further supporting a
breast origin for the malignancy (Figs. 5 and 6). IHC was performed on formalin-fixed,
paraffin-embedded tissue sections (4 µm thickness). The sections
were deparaffinized in xylene and rehydrated through graded ethanol
to distilled water. Heat-induced epitope retrieval was carried out
using EnVision™ FLEX Target Retrieval Solution, High pH (cat. no.
K8004, Agilent Technologies, Inc.) in a pressure-based retrieval
system according to the manufacturer's instructions. Endogenous
peroxidase activity was blocked using EnVision™ FLEX
Peroxidase-Blocking Reagent (cat. no. SM801, Agilent Technologies,
Inc.). The primary antibodies were applied at the following working
dilutions: CK7 (mouse monoclonal, clone OV-TL 12/30; cat. no.
M7018, Agilent Technologies, Inc.) at 1:200, GATA3 (mouse
monoclonal, clone L50-823; cat. no. 386M-16, Cell Marque™ Tissue
Diagnostics) at 1:100, and TRPS1 (rabbit monoclonal, clone
EPR16171; cat. no. ab209664, Abcam) at 1:250. All primary
antibodies were diluted in antibody diluent supplied by the
manufacturer and incubated according to standard laboratory
protocols. Antibodies were incubated at room temperature for 30-60
min according to the manufacturer's recommendations.
Immunodetection was performed using the EnVision™ FLEX detection
system (cat. no. SM802, Agilent Technologies, Inc.), which employs
a dextran-based polymer secondary antibody conjugated to
horseradish peroxidase (HRP). This system provides species-specific
secondary antibodies against mouse and rabbit immunoglobulins and
was used ready-to-use (no dilution required), in accordance with
the manufacturer's instructions. All immunohistochemical staining
procedures, including primary antibody incubation and polymer-based
secondary antibody incubation, were carried out at room temperature
(20-25˚C). Primary antibodies were incubated for 30-60 min at room
temperature (20-25˚C), as recommended by the respective
manufacturers. The EnVision™ FLEX HRP-labeled polymer secondary
antibody was applied for 20 min at room temperature. Visualization
with 3,3'-diaminobenzidine (DAB) chromogen (cat. no. DM827, Agilent
Technologies, Inc.) was performed for 5-10 min, with microscopic
monitoring to ensure optimal signal development. Nuclear
counterstaining was performed using hematoxylin solution (cat. no.
05-M06002, Bio Optica S.p.A.) for ~1-2 min at room temperature,
followed by rinsing in running tap water, dehydration through
graded alcohols, clearing in xylene and mounting. The patient was
diagnosed with primary invasive PLC of the right breast, with
metastatic involvement of the right axillary lymph nodes, bilateral
ovaries and peritoneum. Based on clinical, radiological and
histopathological findings, the disease was staged as cT2N3M1
according to the TNM classification system.
Therapeutic intervention
A multidisciplinary team evaluated the case of the
patient, and referral to medical oncology was recommended for
definitive management. She subsequently commenced systemic therapy
and received five cycles of palbociclib (125 mg orally once daily)
in combination with letrozole (2.5 mg orally once daily) and
goserelin (Zoladex® 3.6 mg administered subcutaneously
every 28 days) as part of her treatment regimen. The selection of
systemic therapy was guided by the presumed hormone
receptor-positive and HER2-negative profile.
Follow-up and outcomes
The patient undergoes regular follow-ups to monitor
her treatment response and disease progression. A CT scan after the
third cycle revealed a partial response (Fig. 7). Thus far, she remains stable,
with no reported complications or adverse effects from therapy. Her
condition is being closely monitored to ensure optimal
management.
Discussion
Breast cancer metastatic sites usually include the
bones, liver, brain and lungs, while metastases to the female
genital tract are rare. When it occurs, the ovary is the most
frequently affected site (75.8%), followed by the vagina (13.4%),
the uterine corpus (4.7%), the cervix (3.4%), the vulva (2%) and
the fallopian tubes (0.7%), respectively (6). Numerous patients remain asymptomatic,
underscoring the importance of routine gynecologic check-ups.
Distinguishing metastatic disease in the ovary and uterus from
primary ovarian or uterine cancer can be particularly challenging
(6). A focused literature search
was performed using Google Scholar, limited to English-language
publications and case reports describing ovarian or peritoneal
metastases originating from breast cancer. In total, 6 reported
cases of ovarian metastases and 1 case of peritoneal metastasis
were identified and reviewed (Table
I) (6,7-10).
A CT scan was the most frequently utilized imaging modality, and
the diagnosis was supported by microscopic examination. Surgical
intervention was undertaken as the primary treatment. One patient
died during the course of follow-up, and another was lost to
follow-up.
 | Table IReview of some breast cancer
metastases cases to the ovaries or peritoneum in literature. |
Table I
Review of some breast cancer
metastases cases to the ovaries or peritoneum in literature.
| First author, year of
publication | No. of patients | Age, years | Type of cancer | Presentation/clinical
findings | Imaging | Histopathological,
Immunohistochemical and cytological examinations | Treatment | Postoperative
care | Outcome | (Refs.) |
|---|
| Dominguez, 2021 | Case 1 | 42 | Invasive lobular
carcinoma. | Abdominal pain,
bloating vomiting and appetite loss. | CT: Revealed ovarian
growth, bilateral, probably malignant. | PFC: Invasive lobular
carcinoma of breast origin. | Exploratory
laparotomy, total abdominal hysterectomy and bilateral
salpingo-oophorectomy | Lost to follow
up. | Lost to
follow-up. | (6) |
| | Case 2 | 43 | Metastatic carcinoma
with mixed lobular and ductal features. | Intraoperatively a
grossly normal cervix, bilateral ovaries and appendix. Uterus:
endometrial polyp, multiple leiomyoma, and thin endometrium | Not mentioned. | HPE: Metastatic
carcinoma, mixed lobular and ductal features, probably breast in
origin on bilateral ovaries. IHC: +ve ER/PR stains. | Total abdominal
hysterectomy & bilateral salpingo-oophorectomy. | Tamoxifen, adjuvant
therapy continued. | Not mentioned. | (6) |
| | Case 3 | 38 | Invasive ductal
carcinoma. | Uterus, bilateral
ovaries and fallopian tubes grossly normal. Multiple implants over
the peritoneum. | CT: Pelvic mass. | IHC: Triple-negative
result of ER,PR/HER2-neu. HPE: metastatic, poorly differentiated,
probably ductal carcinoma, bilateral ovaries. | Exploratory
laparotomy, ascitic fluid drainage, bilateral salpingo-oophorectomy
infracolic omentectomy, and peritoneal biopsy. | Advised adjuvant
chemotherapy but refused treatment. | Not mentioned. | (6) |
| Naito, 2012 | 1 | 54 | Breast cancer with
solid and luminal structures. | Abdominal distention,
high serum carcinoembryonic antigen & carbohydrate antigen
15-3. | PET/CT: Bilateral
ovarian tumors with ascites. | HPE: Solid and
luminal structures. IHC: +ve for ER, PR, CK-7 and GCDFP15.-ve for
HER2 overexpression and CK20. | Bilateral
oophorectomies for definitive ovarian tumors diagnosis. | Aromatase inhibitor
therapy. | Patient in stable
condition. | (7) |
| Sohail, 2021 | 1 | 69 | Invasive lobular
breast carcinoma metastasizing to the gastric wall and
peritoneum. | Black tarry stool,
increased lethargy with early satiety, mild abdominal tenderness,
thickened gastric folds. | CT: Gastric wall
thickening, bilateral hydronephrosis and mild to moderate
ascites. | IHC: +ve for ER, PR
and -ve for human epidermal growth factor receptor-2. | Diagnostic
paracentesis, gastroduodenoscopy, endoscopic ultrasound, wedge
resection and biopsy of stomach and peritoneum. | Patient decided on
palliative care. | Succumbed after a few
weeks in the hospice facility. | (8) |
| Tada, 2022 | 1 | 57 | Malignant phyllodes
tumor. | Abdominal fullness,
bilateral lower leg edema, an enlarged cystic mass occupying the
pelvic cavity and abdomen. | CT: Cystic mass with
a thickened septum and solid component occupying abdominal
cavity. | HPE: proliferating
stromal spindle-shaped cells with irregular nuclei arranged in a
fascicular or sheeted pattern. IHC: +ve for CD10, CD34, and SMA,
-ve for desmin, S-100, AE1/AE3, EMA, STAT6, ER, PR, MDM2, p16 and
IMP3 | Emergent laparotomy,
right oophorectomy & partial omentectomy. | 6 courses of
chemotherapy consisting of doxorubicin and ifosfamide | Patient in stable
condition. | (9) |
| Lin, 2023 | 1 | 53 | Invasive breast
carcinoma. | Abdominal distension,
irregular vaginal bleeding, and chest distress, abnormal bone
metabolism, cancer antigen 125 present. | CDU: A mass in right
adnexal area. | HPE: Left breast
cancer with ovarian and multiple bone metastases. IHC: Ki-67 5%,
+ve for ER, PR, CK, CK7, HER2 and GCDFP15. -ve for CA125, and
p53. | Removed uterus,
bilateral adnexa, bilateral pelvic lymph nodes, abdominal
para-aortic lymph nodes, greater omentum, and appendix. | Fulvestrant and
azolephosphonic after the breast cancer diagnosis. | Patient in stable
condition. | (10) |
Women who have a breast cancer history are up to 3-
to 7-fold more likely to develop primary ovarian cancer than
ovarian metastases. Risk factors contributing to the development of
breast and ovarian cancers include an older age, low parity,
exogenous estrogen exposure, and having a familial history of BRCA1
and BRCA2 gene mutations (11).
Being a type of ILC, PLC exhibits a classic growth pattern but is
distinguished by nuclear atypia and often presents with a
plasmacytoid, histiocytoid, or apocrine morphological appearance
(2). The molecular genetics of PLC
is similar to that of ILC. However, they also exhibit additional
genetic changes, such as HER2 amplification, which may contribute
to their aggressive behavior. The genetic modifications in PLC are
more similar to those of invasive ductal carcinoma than those of
ILC (12).
IHC is an essential tool for diagnosing and
differentiating primary ovarian tumors from metastatic ones. For
example, GATA3 can help distinguish between the two in cases with a
history of breast cancer, as its expression is positive in breast
carcinoma, but negative in ovarian carcinoma. However, this
differentiation may not be relevant in the rare case of ovarian
mesonephric carcinoma, as it may produce GATA3. In cases of breast
cancer metastatic to the ovary, CK7 positivity may also be
observed. Furthermore, when the primary breast carcinoma expresses
ERs, ER immunoreactivity can serve as an additional supportive
diagnostic marker, as demonstrated in the present patient (13). The expression of ER and PR is at
lower levels in PLC, and it can exhibit an increase in HER2/neu, in
contrast to other types of ILC, which usually strongly express ER
and PR and are negative for HER2 (2,4). The
expression rates of PR and ERα in ILC are up to 60-70 and 95%,
respectively, and Ki-67 staining results are usually low in ILC
(3).
In another study, 24 patients were retrospectively
reviewed who were pathologically confirmed to have metastases in
ovaries from breast cancer, with data collected from two centers
between 2000 and 2019(11). The
results of that study demonstrated that 80% of the cases had
negative HER2/neu results, and almost 90% were ER- and PR-positive
(11). Another component sought is
gross cystic disease fluid protein 15 (GCDFP-15), which has high
specificity for breast carcinoma, but low sensitivity (14). In a previous case report described
the case of a 53-year-old with abdominal distension, irregular
vaginal bleeding, chest distress and a history of having a left
breast mass for 2-3 years (10).
The patient had an invasive breast carcinoma along with ovarian and
multiple bone metastases; IHC results revealed Ki-67 5%, positive
results for ER, PR, cytokeratin, CK7, HER2 and GCDFP15, and
negative results for CA125 and p53(10).
The lack of E-cadherin expression is commonly used
in IHC to distinguish between lobular and ductal lesions
histologically (1). E-cadherin,
consisting of the transmembrane E-cadherin protein along with α, β,
γ and p120 catenin, is crucial for establishing intercellular tight
junctions (15). It binds to
identical molecules on adjacent cells in a homophilic fashion,
facilitating cell-cell adhesion and maintaining cellular polarity.
The cytoplasmic localization of p120 catenin can serve as a
positive IHC marker for ILC (3).
The pattern of metastasis in ILC is distinct from
that of invasive ductal carcinoma, which generally spreads more
extensively and often affects the plasma membrane layer. While both
ILC and invasive ductal carcinoma commonly metastasize to the bones
and liver, ILC is more likely to spread to the gastrointestinal
tract, peritoneum, retroperitoneum, and genitourinary system, with
the ovaries being a particularly frequent site. Additionally, ILC
often spreads to the central nervous system, including the
meninges, where carcinomatous meningitis is almost exclusively seen
in ILC patients. Rare metastatic sites for ILC include the orbit,
parotid gland, perianal region, and even within the tumor itself.
The loss of E-cadherin, due to a CDH1 mutation, disrupts cell
adhesion, which contributes to the characteristic infiltration
pattern of ILC. This mutation can also activate signaling pathways
such as Rho/ROCK, potentially aiding tumor cell survival and
spread. However, these mechanisms do not fully explain the tendency
of ILC to metastasize to particular sites, suggesting that
additional molecular and environmental factors are at play and need
further investigation (16).
Metastasis from breast cancer to the gastric wall is
uncommon and can be challenging to identify due to its
morphological similarity to primary gastrointestinal cancers
(8). McLemore et al
(17) found an incidence rate of
0.3% of patients with metastatic gastrointestinal tract disease
originating from breast carcinoma among 12,001 cases of metastatic
disease. Sohail et al (8)
reported the case of a 69-year-old woman presenting with abdominal
pain and episodes of black tarry stool. The patient had a history
of stage II-B left breast cancer, which using IHC, was found to be
ER +95%, PR +5%, HER2-negative and Ki-67 70%. A CT scan of the
abdomen revealed moderate to severe thickening of the gastric wall,
bilateral hydronephrosis, and mild to moderate ascites (8). Wedge resection and biopsy were
performed for the stomach and peritoneum, with biopsy results
suggesting the diagnosis of metastatic disease originating from ILC
of the breast, as indicated by GATA3 positivity in the peritoneal
tumor deposit. The IHC results were ER +80%, PR +1% and
HER2-negative, consistent with their diagnosis (8). Breast cancer metastasis to the
stomach clinically presents similarly to that of primary gastric
cancer; dyspepsia, epigastric pain, anorexia, vomiting, early
satiety and bleeding are commonly occurring symptoms. Linitis
plastica is the most frequent pattern of metastasis, characterized
by diffuse infiltration of the submucosa and muscularis propria
(18).
Haque et al (19) analyzed patients with cT1-4N1-3M0
breast cancer who had either ILC or PLC histology from the national
cancer database and had undergone definitive surgical treatment. Of
a total of 115,260 patients, 99.63% had ILC, while only 0.37% had
PLC. Patients with PLC received systemic chemotherapy more often,
whereas hormonal therapy was administered less often. They also
found that worse overall survival was associated with PLC (19). The administration of chemotherapy,
radiation therapy, and hormonal therapy has been linked to improved
overall survival in patients (19).
Given the rarity yet clinical significance of
gynecological metastases from breast cancer, particularly lobular
subtypes such as PLC, patients require vigilant surveillance.
Routine monitoring should include annual pelvic examinations and
transvaginal ultrasound, especially for premenopausal women or
those with lobular histology, family history, or symptoms such as
abdominal distension or irregular bleeding.
The present case report has several limitations that
should be acknowledged. First, the IHC evaluation of the ovarian
and peritoneal metastatic lesions was limited to CK7, GATA3 and
TRPS1. A broader IHC panel commonly used to exclude primary
ovarian, peritoneal, gastrointestinal, or pulmonary malignancies,
such as PAX8, WT1, mammaglobin, GCDFP-15, CK20, p53 and TTF-1, was
not performed. Consequently, the exclusion of non-breast primary
tumors relied on a combination of clinical context, radiological
findings, morphological concordance with the primary breast tumor,
and positivity for breast-associated markers rather than on an
extensive exclusionary immunoprofile. Second, the original breast
ultrasound images were not available for inclusion. Although the
examination was performed at the institution, the images had not
been digitally archived in the radiology database at the time of
the study. Third, as a single-patient case report, the findings
cannot be generalized, and causal inferences regarding disease
behavior or treatment response are limited. Finally, although
histopathological images were provided, quantitative morphometric
analysis and a larger set of representative images were not
available because the pathological evaluation was conducted in a
routine clinical setting without access to digital whole-slide
scanning and validated image-analysis tools. Despite these
limitations, the present case report contributes valuable clinical,
histopathological and diagnostic insights into the rare occurrence
of PLC metastasizing to the ovaries and peritoneum.
In conclusion, breast cancer metastases to the
ovaries and peritoneum are uncommon, yet clinically significant.
IHC is a crucial tool for distinguishing between these metastatic
carcinomas and other carcinomas they may resemble.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
AMS and HAY were major contributors to the
conception of the study, as well as to the literature search for
related studies. SHH, MKA, AAQ and HHF contributed to the
literature search, the acquisition and interpretation of the
patient's data, and manuscript preparation. FHK and KMS contributed
to the design of the study, the literature review, the critical
revision of the manuscript, and the processing of the table. AMS,
LRAP, SOA and RMA assisted in the diagnosis and management of the
patient, and participated in manuscript review. HAY and AMA were
the pathologists who performed the diagnoses. FHK and SHH confirm
the authenticity of all the raw data. All authors have read and
approved the final manuscript.
Ethics approval and consent to
participate
Written informed consent was obtained from the
patient for participation in the present study.
Patient consent for publication
Written informed consent was obtained from the
patient for the publication of the present case report and any
accompanying images.
Competing interests
The authors declare that they have no competing
interests.
Use of artificial intelligence tools
During the preparation of this work, AI tools were
used to improve the readability and language of the manuscript or
to generate images, and subsequently, the authors revised and
edited the content produced by the AI tools as necessary, taking
full responsibility for the ultimate content of the present
manuscript.
References
|
1
|
Riedlinger GM, Joshi S, Hirshfield KM,
Barnard N and Ganesan S: Targetable alterations in invasive
pleomorphic lobular carcinoma of the breast. Breast Cancer Res.
23(7)2021.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Rakha EA, Van Deurzen CH, Paish EC,
Macmillan RD, Ellis IO and Lee AH: Pleomorphic lobular carcinoma of
the breast: Is it a prognostically significant pathological subtype
independent of histological grade? Mod Pathol. 26:496–501.
2013.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Reed AE, Kutasovic JR, Lakhani SR and
Simpson PT: Invasive lobular carcinoma of the breast: Morphology,
biomarkers and'omics. Breast Cancer Res. 17(12)2015.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Al-Baimani K, Bazzarelli A, Clemons M,
Robertson SJ, Addison C and Arnaout A: Invasive pleomorphic lobular
carcinoma of the breast: Pathologic, clinical, and therapeutic
considerations. Clin Breast Cancer. 15:421–425. 2015.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Da Silva L, Parry S, Reid L, Keith P,
Waddell N, Kossai M, Clarke C, Lakhani SR and Simpson PT: Aberrant
expression of E-cadherin in lobular carcinomas of the breast. Am J
Surg Pathol. 32:773–783. 2008.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Dominguez RS and Billod JA: Ovary and
uterus, rare sites of metastases from breast cancer: A case series.
Int J Med Rev Case Rep. 5:79–85. 2021.
|
|
7
|
Naito K, Oura S, Yasuoka H and Okamura Y:
A case of pseudo-meigs' syndrome associated with ovarian metastases
from breast cancer. J Breast Cancer. 15:474–477. 2012.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Sohail A, Khan A, Gross A and Shah H:
Breast carcinoma metastasising to the gastric wall and the
peritoneum: What physicians need to know. BMJ Case Rep.
14(e241467)2021.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Tada Y, Yasunaga M, Tomonobe H, Yamada Y,
Hori E, Okugawa K, Yahata H, Oda Y and Kato K: A case of malignant
phyllodes tumor of the breast metastasizing to the ovary. Int J
Surg Pathol. 30:427–431. 2022.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Lin XY, Zhou XJ, Yang SP, Zheng JX and Li
ZJ: Pseudo-Meigs' syndrome secondary to breast cancer with ovarian
metastasis: A case report and literature review. Front Oncol.
13(1091956)2023.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Cerkauskaite D, Zilinskas K, Varnelis P,
El Oreibi M, Asejev V and Dulskas A: Ovarian metastases from breast
cancer: A report of 24 cases. J Gynecol Obstet Hum Reprod.
50(102075)2021.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Simpson PT, Reis-Filho JS, Lambros MB,
Jones C, Steele D, Mackay A, Iravani M, Fenwick K, Dexter T, Jones
A, et al: Molecular profiling pleomorphic lobular carcinomas of the
breast: Evidence for a common molecular genetic pathway with
classic lobular carcinomas. J Pathol. 215:231–244. 2008.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Katcher AH, Greenman MP, Roychoudhury S
and Goldberg GL: Utilization of immunohistochemistry in gynecologic
tumors: An expert review. Gynecol Oncol Rep.
56(101550)2024.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Fiel MI, Cernaianu G, Burstein DE and
Batheja N: Value of GCDFP-15 (BRST-2) as a specific
immunocytochemical marker for breast carcinoma in cytologic
specimens. Acta Cytol. 40:637–641. 1996.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Butler D and Rosa M: Pleomorphic lobular
carcinoma of the breast: A morphologically and clinically distinct
variant of lobular carcinoma. Arch Pathol Lab Med. 137:1688–1692.
2013.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Yu B, Yan L, Wang H and Yang J and Yang J:
Invasive lobular carcinoma of the breast: Metastatic patterns and
treatment modalities-a review. Front Oncol.
15(1631670)2025.PubMed/NCBI View Article : Google Scholar
|
|
17
|
McLemore EC, Pockaj BA, Reynolds C, Gray
RJ, Hernandez JL, Grant CS and Donohue JH: Breast cancer:
Presentation and intervention in women with gastrointestinal
metastasis and carcinomatosis. Ann Surg Oncol. 12:886–894.
2005.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Jones GE, Strauss DC, Forshaw MJ, Deere H,
Mahedeva U and Mason RC: Breast cancer metastasis to the stomach
may mimic primary gastric cancer: Report of two cases and review of
literature. World J Surg Oncol. 5(75)2007.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Haque W, Arms A, Verma V, Hatch S, Butler
EB and Teh BS: Outcomes of pleomorphic lobular carcinoma versus
invasive lobular carcinoma. Breast. 43:67–73. 2019.PubMed/NCBI View Article : Google Scholar
|