Introduction
Tailgut cysts (TGCs), also known as retrorectal
cystic hamartomas, are rare congenital cystic lesions originating
from remnants of the embryonic hindgut (1,2).
They are typically located in the retrorectal or presacral space, a
potential anatomical area bounded anteriorly by the fascia propria
of the rectum and mesorectum, posteriorly by the presacral fascia,
inferiorly by the levator ani muscle and superiorly by the
peritoneal reflection (1). The
disease was first recognized in the literature as developmental
anomalies arising from the incomplete involution of the postcloacal
gut during embryogenesis (3,4).
Normally, the tailgut undergoes regression by the 6th week of
gestation; failure of this process results in the formation of
these cystic lesions (3). A number
of patients remain asymptomatic, and lesions may be discovered
incidentally (5,6). However, symptomatic cases can present
with nonspecific complaints, including lower abdominal or perineal
pain, constipation, dyschezia, urinary dysfunction, or neurological
symptoms (2,4,7).
This anomaly predominantly affects middle-aged women, with reported
female-to-male ratios ranging from 3:1 to as high as 9:1, although
malignant transformation appears to occur more frequently in males
(1,3,4).
Despite the female predominance, the overall incidence is extremely
low, estimated at ~1 in 40,000 individuals, highlighting the rarity
of this condition (6,8). The diagnosis of TGCs remains
challenging due to their rarity, nonspecific symptoms and anatomic
location, which may mimic or be associated with other anorectal or
pelvic pathologies (6). In
addition, given the potential for neoplastic transformation, early
recognition and management are deemed necessary (2,5). The
present case report describes a case of TGC associated with a
complex perianal fistula in a middle-aged patient.
Case report
Patient information
In July 2024, a 41-year-old female patient presented
to Smart Health Tower (Sulaymaniyah, Iraq) with severe anal pain
during defecation, without radiating to other regions. The patient
denied bleeding per rectum, urinary symptoms (including dysuria,
frequency, or urgency) and any signs of genitourinary disease, and
reported regular bowel habits. She had no history of anal or
perineal discharge, local trauma, systemic symptoms such as fever,
chills, weight loss, or anorexia, nor any chronic illnesses,
including diabetes mellitus, inflammatory bowel disease, or
malignancy. The family history for gastrointestinal, colorectal, or
congenital conditions was negative. Her menstrual and gynecologic
history was normal, with regular cycles and no reported
complications. She had undergone three prior cesarean sections and
a laminectomy ~2 months prior to presentation. There was no history
of immunosuppressive therapy, radiation, or previous pelvic
interventions.
Clinical findings
Upon a physical examination, the patient appeared
comfortable, afebrile and hemodynamically stable. The abdomen was
soft, with no rebound tenderness, guarding, palpable masses,
organomegaly, or ascites. Bowel sounds were normal and audible. A
perineal inspection identified a small external wound at the 6
o'clock position relative to the anal verge, with no active
discharge or signs of local cellulitis. There were no signs of
perianal abscess, skin breakdown in the anal verge, or external
hemorrhoids. A digital rectal examination revealed a normal resting
tone with pinpoint tenderness in the presacral region and localized
tenderness along the posterior rectal wall. There was a deep, soft,
ill-defined fullness palpable in the presacral region, while no
palpable masses, secondary tracts, abscess cavities, or signs of
purulent discharge were noted. The overlying rectal mucosa was
smooth, and there was no intraluminal bulge or mass effect. A
neurological examination revealed normal lower limb strength and
reflexes. Anal reflex and voluntary contraction were intact.
Diagnostic assessments
Blood investigations revealed a normal complete
blood count. Stool calprotectin was measured at 20.2 µg/g (normal
range, <50 µg/g), excluding active inflammatory bowel disease.
Trans-perineal ultrasonography identified a single external opening
at the 6 o'clock position at the anal verge, from which a tract
extended into the intersphincteric plane. A multiloculated,
thin-walled cystic lesion, measuring 25x16x10 mm, was identified
posterior to the anal canal. The absence of direct communication
with the rectal or anal canal lumen suggested an extra-luminal
origin. A magnetic resonance imaging (MRI) was conducted and
confirmed the presence of a multiloculated cystic lesion, measuring
30x16x15 mm, situated in the presacral region, anterior to the
distal rectum, and extending towards the anorectal junction. The
cyst was thin-walled and lacked solid components, with no
connection to the rectal lumen (Fig.
1). Furthermore, the MRI revealed an associated complex
perianal fistula, classified as grade II according to the St.
James's Hospital Classification. An ileo-colonoscopy revealed
patchy areas of colitis, and multiple biopsies were obtained. A
histopathological analysis was performed on 5-µm-thick
paraffin-embedded tissue sections. The sections were fixed with 10%
neutral buffered formalin at room temperature for 24 h and then
stained with hematoxylin and eosin (H&E; Bio Optica Co.) for
1-2 min at room temperature. The sections were then examined under
a light microscope (Leica Microsystems GmbH). The histopathological
examination revealed no notable abnormalities, effectively ruling
out inflammatory or neoplastic conditions (data not shown).
Therapeutic intervention
Under the jackknife position, the patient underwent
a fistulectomy. The external opening was identified, and the
fistulous tract was meticulously followed using electrocautery,
ensuring controlled dissection in the intersphincteric area. This
dissection led to the identification of the underlying
multiloculated cystic lesion. The cyst and its associated fistulous
tract were removed by en bloc excision, ensuring the complete
removal of both the pathological cyst and the communicating
fistula. Following hemostasis, the surgical defect was
intentionally left open to heal by secondary intention. The
procedure was completed without any intraoperative complications. A
histopathological examination of the cyst was performed on
5-µm-thick paraffin-embedded tissue sections. The sections were
fixed with 10% neutral buffered formalin at room temperature for 24
h and then stained with hematoxylin and eosin (H&E; Bio Optica
Co.) for 1-2 min at room temperature. The sections were then
examined under a light microscope (Leica Microsystems GmbH). This
confirmed a TGC (Fig. 2).
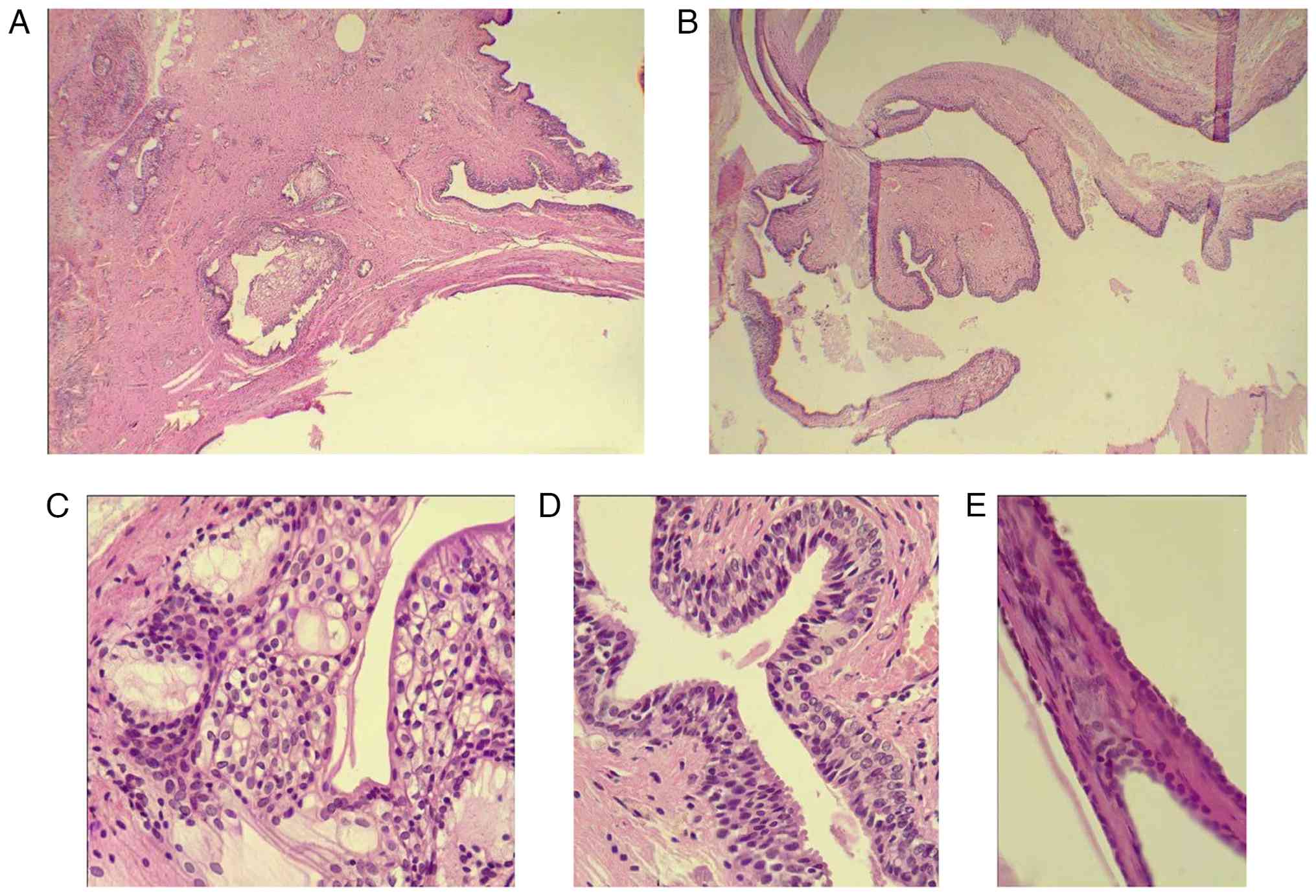 | Figure 2(A) The lesion is cystic and contains
numerous broad fibrous papillae that are lined by epithelium. There
is debris in the lumen of the cyst. (B) There are various types of
epithelium in the lining and wall of the cyst, including
transitional, stratified squamous, mucinous columnar, and flat to
low cuboidal epithelium. (C) The transitional epithelium is
stratified and composed of tall cells with indistinct cytoplasmic
borders, a moderate amount of lightly eosinophilic cytoplasm, and
oval nuclei with fine chromatin. It differs from the transitional
epithelium of the urothelial tract and does not belong to an
otherwise specific category of epithelium in the body. (D) The
stratified squamous epithelium is non-keratinizing and is composed
of cells with distinct cytoplasmic borders, a moderate amount of
clear to lightly eosinophilic cytoplasm, and round nuclei with fine
chromatin. The mucinous glands are lined by tall columnar cells
with abundant cytoplasmic mucin and small, round nuclei with fine
chromatin. (E) The flat to low cuboidal epithelium lines some areas
of the cyst and is composed of cells with minimal eosinophilic
cytoplasm and oval nuclei with fine chromatin. Images illustrate
hematoxylin and eosin staining; (A and B) original magnification,
x40; (C-E) original magnification, x400. |
Follow-up and outcome
The postoperative course was uneventful. Standard
analgesic therapy was administered, and routine wound care was
provided to facilitate healing. The perineal wound exhibited
healthy granulation tissue formation and progressive contraction,
achieving complete closure within 6 weeks of the procedure. During
a follow-up period exceeding 12 months, there was no evidence of
recurrence of either the fistulous disease or the cystic lesion,
and the patient remained asymptomatic.
Discussion
Tumors and cystic lesions in the presacral or
retrorectal space are exceedingly rare (5,9).
Owing to the intricate embryologic development of the region, the
spectrum of presacral lesions includes inflammatory conditions such
as fistulas, abscesses and granulomas, as well as neoplastic
lesions. The neoplastic group encompasses osseous and mesenchymal
tumors (including osteoma, osteosarcoma, Ewing sarcoma, lipoma,
liposarcoma, fibrosarcoma, leiomyoma and leiomyosarcoma),
neurogenic tumors (neurofibroma, neurofibrosarcoma, and
ganglioneuroma) and congenital lesions such as teratoma, TGC, and
rectal duplication cyst. Developmental cysts account for more than
half of congenital presacral lesions, and TGCs are among the rarest
congenital types derived from primitive hindgut remnants (4,5,9).
Embryologically, during the 4th week of intrauterine life, the
embryo folds inward to enclose the primitive gut, and the cloacal
membrane (containing endoderm below Hensen's node) becomes
positioned ventrally to enclose the distal portion of the hindgut,
forming the tailgut. Normally, the tailgut regresses by the sixth
week of gestation. Failure of this regression results in a
mucus-secreting cystic lesion known as a TGC (10).
Clinically, TGCs exhibit a broad spectrum of
presentations, ranging from incidental findings to symptomatic
lesions. Symptoms typically result from the mass effect of the
lesion on adjacent structures, leading to lower abdominal pain,
pelvic or sacral discomfort, constipation, rectal fullness,
dysuria, pollakiuria, and urinary retention (6,10).
In some cases, infection or inflammation of the cyst can cause
acute pain or lead to the formation of a sinus tract that drains
externally, often misdiagnosed as a simple abscess or perianal
fistula (11). Furthermore,
malignant transformation, although rare, has been reported in up to
13% of cases, resulting in adenocarcinomas or carcinoid tumors
(3). Due to their deep pelvic
location and non-specific symptoms, retrorectal masses are often
misdiagnosed. Half of all cases are discovered incidentally, and
there is disagreement regarding the effectiveness of digital rectal
examination in their detection (4). When palpable, they are often
described as extrinsic, fluctuant masses during digital rectal
evaluation, raising suspicion for a retrorectal lesion (6). In the present study, a literature
research on Google Scholar was conducted using the key word ‘tail
gut cyst’, and some of the recently published cases were reviewed.
In reviewing 13 reported cases (1-6,8-10,12-15),
there was a marked female predominance (84.6%), and an age range
spanning from the neonatal period to 71 years. The common clinical
presentations included lower abdominal or pelvic pain,
constipation, urinary retention, and perianal swelling, while
approximately a quarter of the cases were incidentally discovered.
In total, 2 cases were initially misdiagnosed as ovarian or dermoid
cysts. Malignant transformation was documented in five cases
(~38.5%), most commonly into adenocarcinoma, followed by squamous
cell carcinoma, with metastases observed in 3 cases (Table I). The patient in the present case
report complained of severe anal pain during defecation without any
associated symptoms or significant medical history. Upon a physical
examination, there was only a small external wound in the anal
verge, without discharge or local cellulitis. A digital rectal
examination revealed a deep, soft, ill-defined fullness palpable in
the presacral region with no palpable masses or signs of
abscess.
 | Table ISummary of 13 recently reported cases
of tailgut cyst identified in the literature. |
Table I
Summary of 13 recently reported cases
of tailgut cyst identified in the literature.
| Authors, year of
publication | Age/sex |
Presentation/symptoms | Duration of
symptoms | PMSH | Imaging findings | Size of cyst
(cm) |
Misdiagnosis/associated pathology | Malignancy
transformation | Resection status of
the cyst/technique | Outcome and
recurrence | (Refs.) |
|---|
| Malutan et al,
2025 | 30/F | Low abdominal
pain | 3 years | Three surgical
procedures for presumed left ovarian cysts without relieving
symptoms | Presacral tumor with
right pararectal development and mass effect on the rectum | 2.88 | Initially
misdiagnosed as a left ovarian cyst | None | Complete/Open
(Pfannenstiel approach) | Symptom-free and no
recurrence after 1 year of follow-up | (6) |
| Rakia et al,
2025 | 34/M | Asymptomatic
(incidental finding) | N/A | None | Bilobed retrorectal
cysts displacing the rectum | 4 & 7 | None | None |
Complete/laparoscopy | Symptom-free and no
recurrence at the 3-month follow-up | (12) |
| Achugatla et
al, 2025 | Neonate/F | Swelling over
mid-gluteal region above anal opening | Since birth | None | Well-defined
solid-to-cystic lesion in precoccygeal space initially suggestive
of a dermoid cyst | 3.64 | Suspected as dermoid
cyst on imaging | None | Complete/open (Kraske
approach) | Symptom-free and no
recurrence after 1 year of follow-up | (13) |
| Ajredini et
al, 2025 | 63/F | Constipation,
recurrent UTI, difficulty voiding | 3 months | Total hysterectomy
for a large leiomyoma and partial cystectomy | Pericoccygeal
complicated cyst compressing rectum and bladder, later MRI showed a
destructive sacral lesion and L4-L5 vertebral metastasis | 13.8 | None | Transformed into
mucinous adenocarcinoma with metastasis | Complete/Open | Symptoms partially
improved with progressive metastatic disease | (1) |
| Al Jada et al,
2025 | Early 50s/F | Progressive lower
abdominal pain, urinary retention, and tenesmus | 2 months | None | Well-defined
presacral cystic lesion displacing rectum, uterus, and bladder;
follow-up CT/MRI: Recurrent presacral cyst with liver and osseous
metastases | 10.9 | None | Transformed into
mucinous adenocarcinoma with metastases | Incomplete/Open
(exploratory laparotomy) due to intraoperative instability from
left iliac vein injury | Recurrence after 6
months with metastases and succumbed 2 months after commencing
treatment | (3) |
| Kitazawa et
al, 2025 | 40/F | Severe coccygeal pain
exacerbated by sitting | N/A | Uterine polyp treated
with endometrial curettage | Multiloculated cystic
lesion associated with median sacral vein | 1.2 | None | None |
Complete/laparoscopy | Symptom-free | (2) |
| De Crombrugghe et
al, 2024 | 59/M | Bulging and
uncomfortable perianal tumor | N/A | Thrombophlebitis | Large ovoid cystic
lesion in anal canal posterior wall | 3 | None | None | Complete/open | Symptom-free and no
recurrence | (8) |
| Haval et
al, 2024 | 18/F | Intermittent
abdominal pain and constipation | 6 months | None | Multiloculated cystic
mass in presacral/precoccygeal space displacing rectum | 5.4 | None | None | Complete/open
(transabdominal approach) | Symptom-free | (10) |
| Kiosov et
al, 2024 | 45/F | Asymptomatic
(incidental finding) | N/A | None | Oval hypoechoic
inhomogeneous lesion adjacent to posterior rectal wall and
connected to its muscle layer | 2.5 | None | None |
Complete/endoscopy | Symptom-free | (5) |
| Manikandan et
al, 2024 | 23/F | Vaginal discharge
and lower abdominal pain | N/A | Exploratory
laparotomy with marsupialization of cyst | Thick-walled
hypodense lesion with septations and calcifications displacing
rectum | 8.9 | None | Transformed into
moderately differentiated adenocarcinoma | Complete/open
(exploratory laparotomy) | PET-CECT showed
residual disease. chemotherapy was planned but patient lost to
follow-up | (14) |
| Moshtaghian et
al, 2023 | 51/F | Pelvic pain,
constipation, and fecal urgency | N/A | None | Hypodense cystic
lesion in left retrorectal region causing rectal deviation | 2.3 | None | Transformed into
squamous cell carcinoma |
Incomplete/Open | Recurrence and
metastasis; succumbed after 1 year | (9) |
| Russo et al,
2023 | 31/F | Asymptomatic
(incidental finding) | N/A | None | Retrorectal
heterogeneous cystic mass located inferior to gravid uterus | 7.2 | None | None | Complete/open
(sub-umbilical median laparotomy) | Symptom-free | (15) |
| Atiya et al,
2023 | 71/F | Increasing
tailbone/pelvic pain | Several months | Prior mass (unknown
detail) in childhood | Large
multiloculated cyst with septal calcifications | 12.4 | None | Transformed into
adenocarcinoma | Complete/open | Succumbed to
disease | (4) |
The heterogeneous morphology of these cysts
contributes to radiological ambiguity, leading to misdiagnosis in
>50% of cases (4). The
pre-operative diagnosis of TGCs relies heavily on imaging. MRI and
computed tomography (CT) are considered the modalities of choice.
On MRI, TGCs are generally hypointense on T1-weighted images and
hyperintense on T2-weighted images. Still, these characteristics
can vary with cyst content. CT scans usually demonstrate
well-defined presacral cystic lesions with central fluid
attenuation, peripheral soft-tissue enhancement, and occasional rim
calcification (3). Transrectal
ultrasound can further define the association of the cyst with the
rectal wall and determine whether the lesion is cystic or solid
(10). Pre-operative MRI and
biopsy have exhibited a sensitivity of 84 and 86%, respectively,
for presacral tumors (1). While a
biopsy may assist in determining the surgical plan and in
identifying malignant transformation, its use is controversial.
Biopsy is discouraged in resectable cases as it is associated with
the risk of infection or malignant seeding along the biopsy tract
(11). However, in unresectable
cases, biopsy may be justified to guide neoadjuvant therapy
(1). In the case presented herein,
trans-perineal ultrasonography identified a single external opening
in the anal verge, from which a tract extended into the
intersphincteric plane. In addition, a multiloculated, thin-walled
cystic lesion was identified posterior to the anal canal, with no
direct communication with the rectal or anal canal lumen,
suggesting an extra-luminal origin. An MRI confirmed a
multiloculated cystic lesion associated with a perianal fistula
without discharge.
Johnson et al (11) described a 16-year-old girl with
persistent pelvic pain who had undergone several surgeries for
presumed recurrent perianal fistulas and abscesses. Upon
re-evaluation, a presacral cystic mass was identified, leading to
the diagnosis of a TGC. The authors of that study highlighted that
TGCs should be considered in patients presenting with recurrent
presacral abscesses or anal fistulas, as a misdiagnosis may result
in delayed recognition and inappropriate management (11). Similarly, Sauer et al
(16) reported the case of a
58-year-old woman initially treated for a presumed presacral
abscess who developed recurrent perianal fistulas despite drainage
procedures. Subsequent abdominoperineal resection revealed an
adenocarcinoma arising from a TGC, emphasizing that persistent or
recurrent fistulas may conceal benign or malignant cystic lesions
(16). In a Korean series of 24
cases, 42% of TGCs were infected, with few presenting as perianal
swelling or discharging sinuses (17). Consistently, Malutan et al
(6) observed that up to half of
TGCs become clinically apparent only after infection or fistula
formation. These findings may explain the case in the present
study, in which a perianal fistula developed without an abscess or
discharge, and the definitive diagnosis could have been easily
overlooked without appropriate diagnostic evaluation.
A histopathological examination remains the gold
standard for confirming the diagnosis. Microscopically, TGCs are
typically multilocular and filled with mucinous material. Their
walls contain irregular bundles of smooth muscle and are lined by a
mixture of epithelial cell types, including keratinizing or
non-keratinizing squamous, columnar, ciliated and transitional
epithelium. These features distinguish TGCs from other cystic
lesions, such as epidermoid cysts or teratomas (7).
Complete surgical excision is the treatment of
choice for both symptomatic and asymptomatic TGCs (3). The surgical approach depends mainly
on the location and extent of the lesion. Lesions above the S3
vertebral level are typically approached anteriorly via a
transabdominal route, while those below are managed through a
posterior route, such as parasacral, transsacral, trans-coccygeal,
or intersphincteric approaches. Combined abdominosacral approaches
are reserved for extensive lesions (6,17).
Each technique has distinct advantages: The anterior route offers
direct visualization of pelvic structures, such as the ureters and
iliac vessels, whereas the posterior approach provides easier
access to distal lesions but carries a higher risk of nerve injury
(6). Ensuring complete excision
with negative margins is critical, as incomplete resection can lead
to recurrence, infection, or delayed malignant transformation.
Post-operative follow-up is recommended with periodic digital
rectal examinations and imaging analyses, such as CT scans
(3). In addition to open surgery,
endoscopic removal has also been performed in some cases for small,
well-defined lesions adjacent to the rectal wall with no major
vessels in proximity, and acceptable outcomes have been achieved
(5).
A multidisciplinary approach is essential to ensure
an accurate diagnosis and optimal management. Inadequate pre- and
peri-operative work-up may result in recurrence or a missed
opportunity for cure (1). A
previous case report assumed that recurrent lesions decades after
incomplete excision may sometimes be associated with malignant
change (4). This highlights the
necessity for complete primary excision and vigilant long-term
follow-up (4). Among the cases
reviewed herein, mortality occurred in 3 cases due to malignant
transformation. All patients underwent surgical excision, with the
open approach used most frequently (~76.9%). In 2 cases which were
malignant, the excision was incomplete and ended in metastasis and
mortality. Outcomes were excellent among patients with benign
lesions, with no recurrence during follow-up, whereas those with
malignant transformation had a poorer prognosis. The patient in the
present case report underwent the en bloc excision of both the TGC
and its associated fistula without any intra- or post-operative
complications. Over 1-year follow-up, the patient remained
asymptomatic and had no recurrence. The limitation of this report
include the inability to retrieve the histopathological image from
the initial Ileo-colonoscopy biopsy.
In conclusion, TGCs, as a congenital anomaly, may
present with non-specific symptoms and may be associated with or
masked by other perianal conditions, such as fistulas,
necessitating appropriate investigation to avoid misdiagnosis or
inappropriate management.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
SL, OHG, KAA and SHS were involved in the design and
conception of the study, in the literature review, and in managing
the case. SMF was the radiologist who performed the radiological
examination and prepared the related figures. RMA was the
pathologist who performed the histopathological examination of the
case and also critically revised the manuscript. MMA, HOA, KFHH and
FHK were involved in the literature review, in the drafting of the
manuscript, in critical revision, and in the design of the study.
All authors have read and approved the final manuscript. SL and KAA
confirm the authenticity of all the raw data.
Ethics approval and consent to
participate
Written informed consent was obtained from the
patient for participation in the present case report.
Patient consent for publication
Written informed consent was obtained from the
patient for the publication of the present case report and any
accompanying images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ajredini M, Saracoglu C, Kus AA, Cetin SE,
Aytac E and Ozer L: Clinical course of a tailgut cyst transformed
to retrorectal mucinous adenocarcinoma: A case report. Int J Surg
Case Rep. 134(111635)2025.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Kitazawa M, Karasawa S, Nakamura S,
Yamamoto Y and Soejima Y: Laparoscopic excision of a tailgut cyst
with refractory pain: A case report. Cureus.
17(e77644)2025.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Al Jada I, Oweidat M, Khaleel M, Harb O,
Abu Nahla U, Bleibel R, Arafeh M and Hassouneh AWM: Recurrent
presacral tailgut cyst with mucinous adenocarcinoma and metastasis.
Radiol Case Rep. 20:4022–4027. 2025.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Atiya S, Horn A, Wedel W and Lintel N: A
rare case of ruptured tailgut cyst leading to carcinomatosis. Case
Rep Pathol. 2023(1282058)2023.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Kiosov O, Tkachov V and Gulevskyi S:
Endoscopic resection of tailgut cyst. Case Rep Gastrointest Med.
2024(5538439)2024.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Malutan AM, Suciu VE, Ignat FL, Diculescu
D, Ciortea R, Boțan EC, Bucuri CE, Roman MP, Nati I, Ormindean C
and Mihu D: Tailgut cyst-gynecologist's pitfall: Literature review
and case report. Diagnostics (Basel). 15(108)2025.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Kobayashi T, Ishida M, Miki H, Yagyu T,
Hatta M, Hamada M, Hirose Y and Sekimoto M: Analysis of the
clinicopathological features of tailgut cyst with emphasis on the
development of neoplastic lesions. Oncol Lett.
27(286)2024.PubMed/NCBI View Article : Google Scholar
|
|
8
|
de Crombrugghe J, Cimpean S, Verset L and
Mehdi A: Tailgut cyst, management of a rare perianal mass: A case
report. AME Case Rep. 9(18)2024.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Moshtaghian M, Shahsiah R, Jafari F and
Aghili M: Malignant transformation of tailgut cyst to squamous cell
carcinoma, a rare case with poor outcome. Clin Case Rep.
11(e6893)2023.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Haval S, Dwivedi D and Nichkaode P:
Presacral tailgut cyst. Ann Afr Med. 23:237–241. 2024.PubMed/NCBI View Article : Google Scholar : (In French,
English).
|
|
11
|
Johnson KN, Young-Fadok TM, Carpentieri D,
Acosta JM and Notrica DM: Case report: misdiagnosis of tailgut cyst
presenting as recurrent perianal fistula with pelvic abscess. J
Pediatr Surg. 48:e33–e36. 2013.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Rakia S, Mohamed Ali M, Hsairi M, Karim S,
Brahim G, Sassi K and Slima MB: Case Report: Tailgut cyst:
Diagnosis and treatment. F1000Research. 14(747)2025.
|
|
13
|
Achugatla S, Karkera P, Pikale H and
Bodhanwala M: Tailgut cyst in a neonate with review of
literature-22nd case in pediatric population. J Indian Assoc
Pediatr Surg. 30:558–559. 2025.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Manikandan M, Kapoor A and Yadav V:
Tailgut cyst adenocarcinoma in a young female. JRCR. 15:136–138.
2024.
|
|
15
|
Russo D, Bartholmot C, Fabre JM and Bardol
T: Neuroendocrine tumor arising in a tailgut cyst in a pregnant
woman: First case report and review of the literature. SODA: Jun 7,
2023 (Epub ahead of print).
|
|
16
|
Sauer J, Wolf HK and Junginger T:
Adenocarcinoma in a tail-gut cyst: A rare cause of recurrent
perianal fistula. Chirurg. 71:712–716. 2000.PubMed/NCBI View Article : Google Scholar : (In German).
|
|
17
|
Sakr A, Kim HS, Han YD, Cho MS, Hur H, Min
BS, Lee KY and Kim NK: Single-center experience of 24 cases of
tailgut cyst. Ann Coloproctol. 35:268–274. 2019.PubMed/NCBI View Article : Google Scholar
|















![(A) Axial section images [images on
the upper panel (left panel, T2-weighted fat-saturated; right
panel, T2-weighted); images on the lower panel (left panel, T1
fat-saturated pre-contrast; right panel, T1 fat-saturated
post-contrast)] illustrating an elongated multiloculated
thin-walled cystic lesion in the presacral space (green arrows),
extending from the posterior aspect of the rectum toward the
sacrococcygeal region while rectum and vertebrae are normal with no
surrounding soft tissue edema. (B) Sagittal plane images (left
panel, T2-weighted fat-saturated; middle panel, T2-weighted; right
panel, T1 fat-saturated post-contrast) illustrating a presacral
space lesion (green arrows), which is above the pelvic diaphragm
and extending toward the anorectal junction.](/article_images/wasj/8/2/wasj-08-02-00447-g00.jpg)
