Introduction
A wide variety of replacement grafts have been
attempted in the pursuit of achieving the elusive goal of bone
regeneration in osseous defects. These materials are indicated for
osseous regeneration in the management of periodontal and
peri-implant defects, as are procedures such as socket
preservation, ridge augmentation and guided bone regeneration.
Biocompatibility and cell viability are critical properties for
biomaterials that come in proximity to biological tissues, and
represent the capability of a substance to associate with cells in
a living system without leading to destruction by unfavourable
reactions (1). It is directly
related to cytotoxic effects, or the capacity of the material to
induce damage to a living system (2).
There are two types of materials that induce bone
formation: Grafts and bone substitutes. Both can promote bone
regeneration (3). On the basis of
their source, these materials with applications in regenerative
procedures are autogenous when procured from the same individual,
homogenous or allogenic when they are acquired from individuals of
the same species, and xenogenic when they are obtained from
different species. Alloplastic materials are synthetically
manufactured (4).
Alloplasts are prospective bone substitutes, and
their outstanding versatility allows them to be manufactured via
novel techniques to obtain porous structures that mimic cancellous
bone or resemble the fibrillar portion of the extracellular matrix
(5,6).
Synthetic materials also provide the advantage of
incorporating components with anti-inflammatory and antibiotic
effects, thereby reducing the possibility of complications or
biological reactions. For example, bone morphogenic protein-9 is
used to achieve an osteoinductive effect.
Numerous commercially available osseous grafts
currently utilise synthetic materials combined with natural
products processed in the laboratory to create biomimetic scaffolds
that simulate the extracellular bone matrix (7). Animal bones, fish bones and scales,
avian eggshells and the exoskeletons of marine organisms have been
used to develop xenogenic materials (8).
Eggshells have been used for various reconstructive
surgeries. The mineral composition and high biological activity of
these materials render them potentially functional for biological
and pharmaceutical applications (9). Surface modifications of eggshell
grafts have been attempted to enhance their properties (10). The use of a material derived from
eggshells may also have several other benefits due to its
obtainability and biodegradability (11,12).
The incorporation of collagen into these scaffolds
could improve the efficacy of the biomaterial by contributing to an
osteopromoting quality (13).
Moreover, it has been reported that hydroxyapatite-collagen
scaffolds can have a profound effect on mechanical properties, such
as strength, stiffness and pore size (14). Nevertheless, further investigations
are required to substantiate the demand for collagen to increase
the biocompatibility and stability of the biomaterial without
influencing creeping substitution.
Owing to their extensive array of applications,
several collagen sources have been investigated. However, the use
of collagen, which is mammal-based, has been limited by diseases,
such as bovine spongiform encephalopathy and other spiritual
constraints (15).
Marine-based collagen is a potential replacement for
mammal-based collagen due to its amenable chemical properties, ease
of extraction, biocompatibility, low risk of disease transmission
and environmental contamination, and few religious and ethical
concerns. It has a high collagen content and excellent absorption
properties (15).
Glycerol is a non-toxic, biodegradable liquid that
the FDA considers ‘Generally Recognised as Safe’ (GRAS) (16). Hence, it is referred to as an inert
carrier and provides improved handling characteristics. Glycerol
also provides the advantages of excellent graft containment and
flexibility.
Although, there is limited research on HPA-Coll
composites (14), the literature
on composites derived from hydroxyapatite of egg shell origin and
fish collagen is very limited. All the more glycerine has been used
as a binder here.
The immune response induced by a biomaterial can
influence the healing of biological tissues. Hence, it is a
critical property to consider when selecting a regenerative
material as a primary anti-inflammatory approach can enhance tissue
healing (17).
Before the regenerative capacity of a material can
be assessed, its biocompatibility needs to be established to ensure
that it does not induce adverse tissue reactions. The present study
aimed to examine the biological behaviour of egg shell-derived
hydroxyapatite (EHPA), a composite graft prepared using EHPA and
fish collagen (EHPA/Coll), and a composite material with glycerol
(EHPA/Coll/ Gly) in vitro and in vivo to demonstrate
the cell viability, biocompatibility and inflammatory tissue
reactions of these materials in Wistar rats.
Materials and methods
Eggshells and fish collagen
Domestic chicken eggshells of an indigenous breed of
hen (Aseel) were obtained from a local poultry farm (Kurupseval
farm, Kerala, India). Hydroxyapatite was synthesised from the egg
shells via the chemical precipitation method (18). The steps involved cleaning
eggshells after removing their membranes, followed by drying,
grinding and sintering at high temperature (900˚C in a muffle
furnace), resulting in formation of calcium oxide and further
calcium hydroxide (following a reaction with atmospheric moisture).
Furthermore, the product was reacted with a phosphate source
(ammonium phosphate) to give rise to hydroxyapatite.
Fish collagen was extracted at the Central Institute
of Fisheries Technology, Kochi, India, via a
laboratory-standardised protocol from Rohu fish (Labeo
rohita) and supplied. The scales were subjected to cleaning and
pre-treatment to remove debris, followed by demineralisation (using
0.4M HCl) and further treatment with 0.4 M acetic acid. This was
followed by the removal of insoluble portions by filtration and the
collection of soluble collagen by salting out the pooled filtrate
with NaCl. Centrifugation was performed for 1 h at 8,000 x g and
4˚C, and the collagen was collected as a pellet. Furthermore, the
collagen was re-dissolved in 0.5 M acetic acid, after which the
salting-in and salting-out processes were repeated twice. The salt
was removed from the final suspension via dialysis. The purified
collagen was collected by centrifugation for1 h at 8,000 x g and
4˚C. and freeze-dried. A temperature of 4˚C was maintained
throughout the entire extraction process.
Hydroxyapatite was modified with fish collagen at a
ratio of 60:40 at 80˚C, a vacuum level of 0.02 mbar, and a
bench-top lyophiliser (Labconco) was used to obtain a sample volume
of 5 ml and a flask size of 15 ml. The ratio of 60:40 was selected
based on the preliminary optimisation trials where different ratios
of hydroxyapatite to collagen 70:30,50:50 and 60:40 were tested.
The viability of cells cell exposed to the materials (50 µg/ml) was
found to be 121.199±3.336, 128.31±4.090 and 128.58±3.926. All the
three ratios were found to be non-cytotoxic. Hence the ratio which
mimics the composition of human bone was selected. The procedure
was carried out for 24 h.
The modified material, which was in powder form, was
converted to an injectable consistency by incorporating glycerol
via a mortar and pestle (10 mg of the composite material with 6 ml
glycerol).
Cytotoxicity analysis. Cell line and
culture conditions
L929 murine fibroblasts (NCCS, Pune, India) were
used for the study. They were cultured in Dulbecco's Modified
Eagle's medium (DMEM Hi-media) supplemented with 10% FBS, 1%
glutamine and 1% antibiotic-antimycotic solution (HiMedia
Laboratories, LLC). A humidified atmosphere was maintained
throughout the experiment, and the cells were maintained at 37˚C
and 5% CO2.
Assessment of cell viability by MTT assay.
The effects of the test compounds (EHPA, EHPA/Coll and
EHPA/Coll/Gly) on the cells was assessed using methyl thiazolyl
tetrazolium (MTT) assay, as previously described by Mosmann
(19). The seeding of the cells
(L929 cell line) was performed in a a 96-well microtiter plate at a
density of 5,000 cells/well. The cells were allowed to attach
overnight at 37˚C and 5% CO2. Following adherence,
various concentrations (0, 12.5, 25, 50 and 100 µg/ml) of the
samples were added to the wells followed by incubation for 24 h at
37˚C and 5% CO2. The medium was decanted after 24 h of
incubation. MTT reagent (HiMedia Laboratories, LLC) 1 mg/ml was
added to the wells which were subsequently incubated at 37˚C for 4
h. The MTT solution was removed from the wells, and the formazan
crystals formed were solubilised, and the absorbance was recorded
at 570 nm using a multimode microplate reader (FluoSTAR Omega, BMG
Labtech). The percentage of viable cells in the sample was
calculated with respect to that of the untreated cell control
cells, as previously described (19).
In vivo biocompatibility
The experiment was conducted on 6 Wistar male albino
rats. The animals were 12-15 weeks old and weighed ~250 g. Ethical
clearance was obtained from the Institutional Animal Ethics
Committee (YU/IAEC/25/2022). Animals were supplied by the Animal
House at Yenepoya Research Centre. Pre-operatively, a 10-day
preparation time was used for standard rat health tests. All animal
welfare considerations were taken care. Animals were provided with
special housing conditions such as adequate ventilation, humidity
60-65%, temperature of 22˚C, appropriate cage size, ad
libitum access to food and water. 6 animals were anaesthetised
via an intramuscular injection (80 ml/kg) of a combination of
ketamine hydrochloride and xylazine (ketamine at 80 mg/kg body
weight and xylazine at 10 mg/kg) for 20-30 min. Once the
appropriate level of sedation was obtained, through a 1-cm
incision, three subcutaneous pockets were created in the
paravertebral region using a BP blade (no. 15). Subdermal
implantations were performed using the three materials, namely,
EHPA, composite graft (EHPA/Collagen), composite graft with
glycerine (EHPA/Coll/Gly), in the left, middle and right
subcutaneous pouches, respectively in all 6 animals. Following
implantation, the wounds were sutured using four-zero silk sutures.
Additionally, 0.1% gentamicin was applied to the wound, and
diclofenac 75 mg was injected intramuscularly. Following
anaesthesia using an intramuscular injection (80 ml/kg) of
combination of ketamine hydrochloride and xylazine (ketamine at 80
mg/kg body weight and xylazine at 10 mg/kg), The wound was sutured
with 4-0 silk sutures followed by the application of 0.1%
gentamycin and injection of 75 mg diclofenac. The animals were
observed twice daily for a period of 3 months. All animal welfare
considerations were used to minimize suffering and distress. No
death of any animal was reported during this period. Tissue
processing procedures were carried out using a tissue processor
(Leica Microsystems). The recovered tissue samples (after 14 days)
around the implanted sites were procured and stored in buffered
formalin for 48 h at room temperature. The tissues were then
embedded in paraffin blocks. The samples were cut to a thickness of
6 microns using a microtome (Leica RM 2245).
For histological analyses, the samples were stained
using haematoxylin and eosin (Nice Chemicals Private Limited)
following deparaffinisation using Xylene for 5 min followed by
hydration through graded alcohols. Nuclear staining was performed
using Meyer's haematoxylin (2 min) followed by rinsing with water.
Cytoplasmic staining was performed with eosin (2 min) at room
temperature followed by dehydration, clearing and mounting. The
slides were examined under microscope (Olympus CX41 Research
Microscope) and microphotographs were obtained. The slides were
evaluated by two pathologists who were unaware of the materials
used in the experiment. Representative areas with the highest
density of inflammatory cells were selected. The inflammatory
responses were graded by counting the number of inflammatory cells
at high power in the field showing maximum inflammatory cell
infiltrate as previously described (20) (Table
I).
 | Table IGrading of the inflammatory
response. |
Table I
Grading of the inflammatory
response.
| Grade of
inflammation | Inflammatory
response |
|---|
| Grade 0 | Absence of
inflammatory cells |
| Grade 1 | Mild inflammation, an
average of <25 inflammatory cells per high power field |
| Grade 2 | Moderate
inflammation, an average of 25-124 inflammatory cells per high
power field |
| Grade 3 | Severe inflammation,
an average of ≥125 inflammatory cells per high power field |
Statistical analysis
The cell viability assays were performed in
triplicate and the data are presented as the mean and standard
deviation. The statistical analysis was performed using Statistical
Package for the Social Sciences 18.0 (SPSS, Inc.) The
Kruskal-Wallis test was used to compare the mean inflammatory
scores between the groups followed by Dunn's multiple comparison
test. A P-value <0.05 was considered to indicate a statistically
significant difference. Spearman's rank correlation analysis was
used to assess difference between the inflammatory scores within
the same animal.
Results
Cytotoxicity of the materials
All three materials, EHPA, EHPA/Coll and
EHPA/Coll/Gly, were found to be non-cytotoxic after 14 days. When
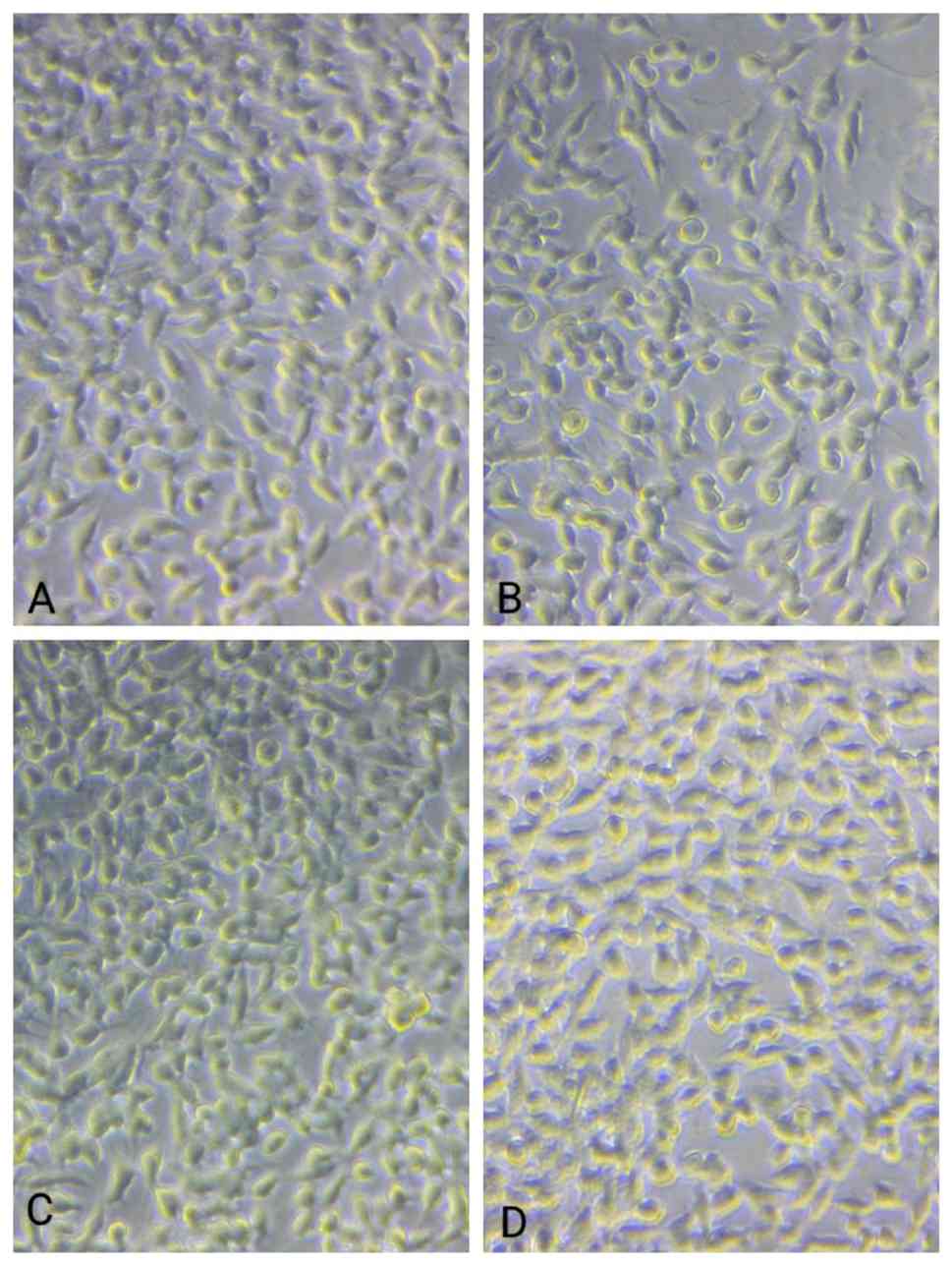
the cells were examined under a phase contrast microscope, no
morphological alterations were detected (Fig. 1).
The effects of the materials at various
concentrations on cell viability are demonstrated in Table II. Although all the materials
promoted cell growth, the EHPA/Coll material resulted in greater
cell viability than did pure EHPA or EHPA/Coll/Gly at
concentrations of 12.5, 25 and 50 µg/ml. However, at a
concentration of 100 µg/ml, EHPA promoted the increased growth of
murine fibroblasts.
 | Table IIEffect of the materials at various
concentrations on the viability of mouse fibroblasts. |
Table II
Effect of the materials at various
concentrations on the viability of mouse fibroblasts.
| | Cell viability,
% |
|---|
| Concentration,
µg/ml | EHPA | EHPA/Coll | EHPA/Coll/Glyc |
|---|
| 12.5 | 91.01±3.904 | 116.33±3.202 | 79.76±1.769 |
| 25 | 96.03±2.766 | 123.15±3.741 | 113.49±3.487 |
| 50 | 116.38±3.093 | 128.58±3.926 | 121.01±2.998 |
| 100 | 122.82±2.868 | 117.59±2.648 | 106.4±2.597 |
Skin reactions following the placement
of the materials
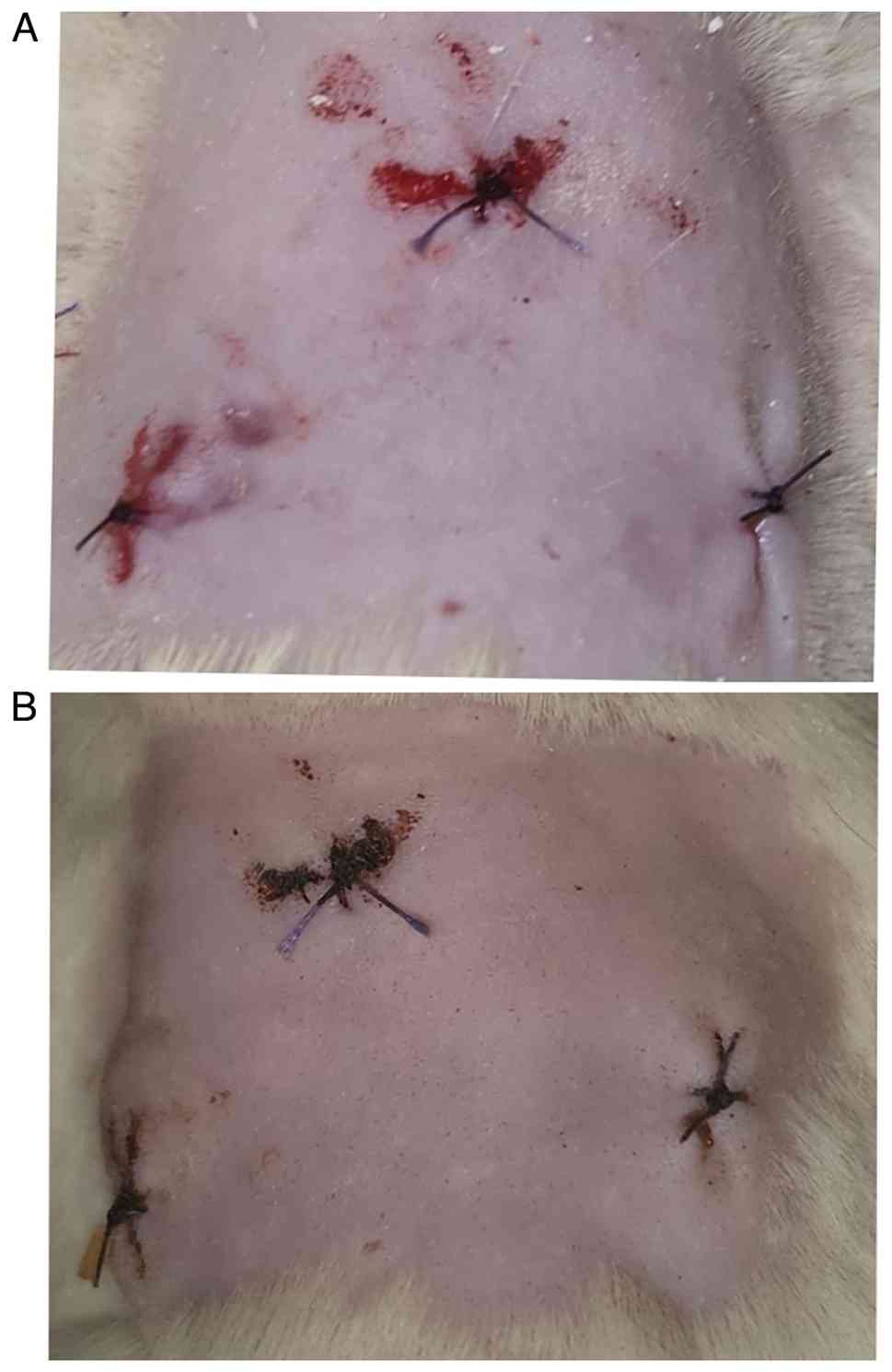
There were no adverse skin reactions noted
throughout a period of 14 days after the materials were placed in
the dorsal soft tissue pouches of the animals. Given that the
initial inflammation caused by surgical trauma is a phase of
healing, all sites exhibited a minimal inflammatory response. There
were no notable signs of hypersensitivity reactions (Fig. 2). The number of inflammatory cells
at a high power field exhibiting the maximum inflammatory cell
infiltrate was calculated (Table
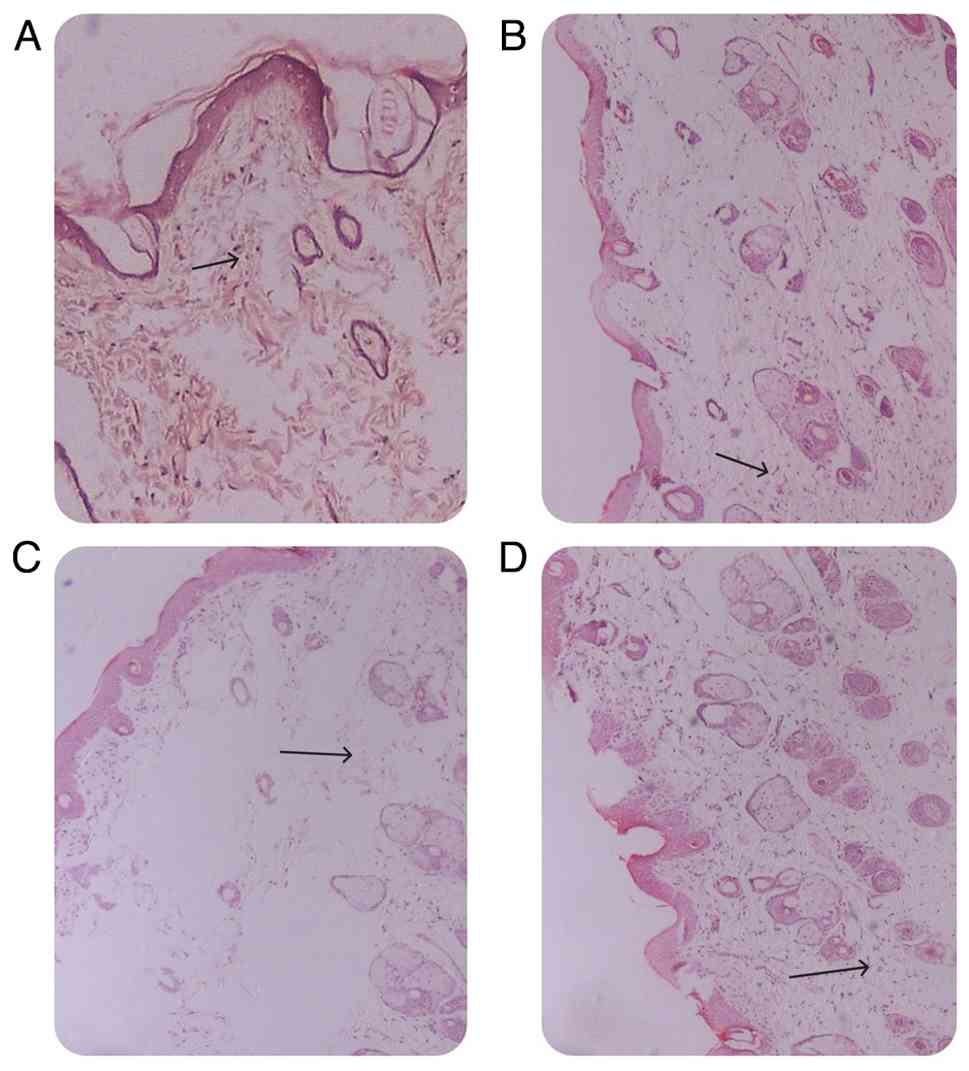
III). At 14 days post-surgery, the initial phagocytic
activities of the cells were noted, followed by the degradation
process and the subsequent reabsorption of the material. In the
implantation area, the particles were surrounded by collagen
fibres, accompanied by a mild inflammatory infiltrate. A sparse
number of multinucleated cells and large blood vessels were also
observed. Minimum numbers of inflammatory cells were noted at all
three sites (Fig. 3).
 | Table IIIScoring based on the number of
inflammatory cells per unit area recorded. |
Table III
Scoring based on the number of
inflammatory cells per unit area recorded.
| Sample | Empty control
(group A) | EHPA (group B) | Composite graft
(EHPA + fish collagen, group C) | Composite graft
(EHPA + collagen + glycerine) |
|---|
| Sample 1 | 1 | 2 | 2 | 2 |
| Sample 2 | 1 | 3 | 2 | 1 |
| Sample 3 | 1 | 2 | 2 | 2 |
| Sample 4 | 1 | 2 | 1 | 1 |
| Sample 5 | 0 | 2 | 2 | 1 |
| Sample 6 | 2 | 2 | 1 | 1 |
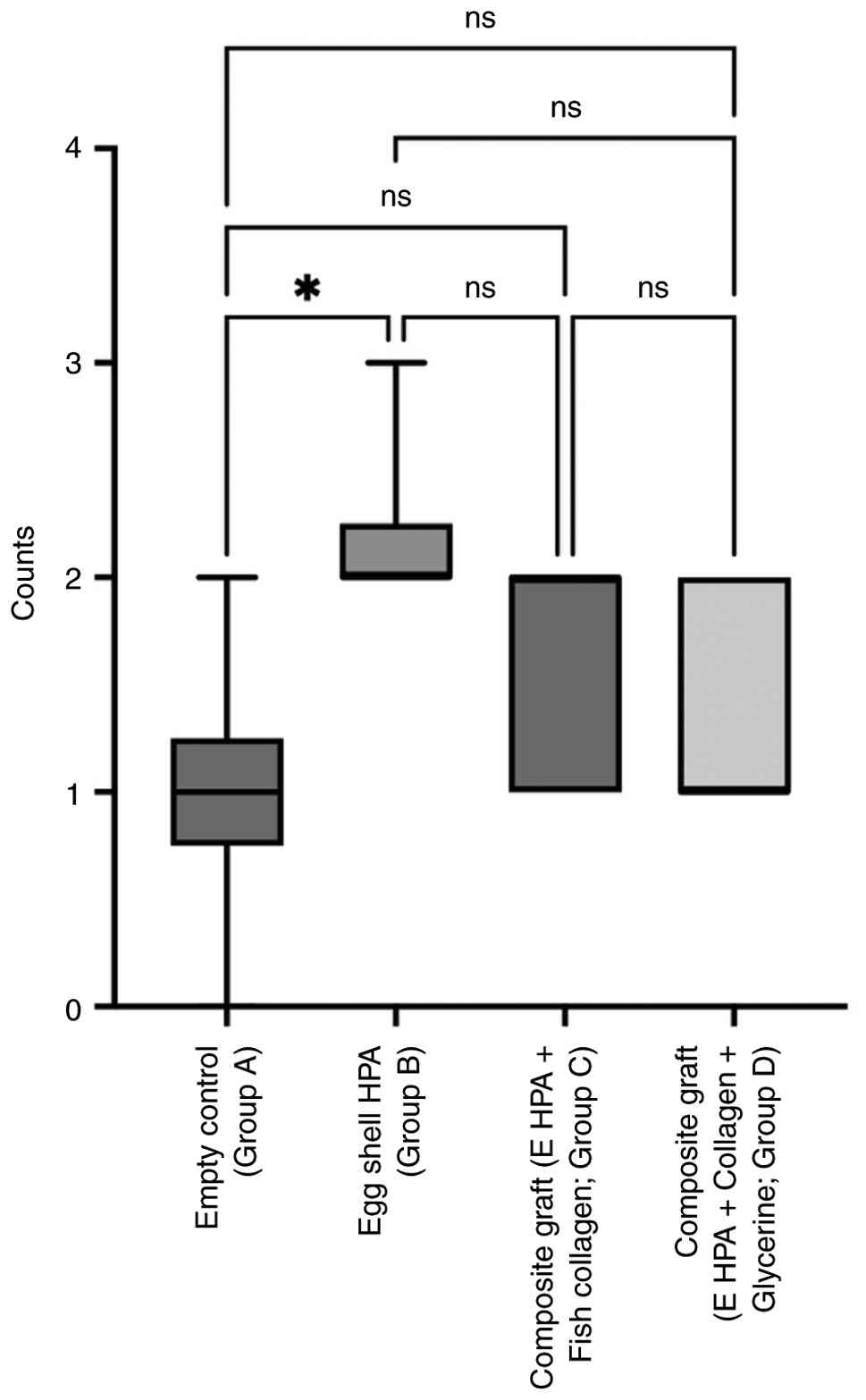
The Kruskal-Wallis was used to compare the mean
inflammatory scores, which revealed that there was no statistically
significant difference in the grade of inflammation (number of
inflammatory cells per unit area) between the groups at the 2nd
week (Fig. 4). Within the same
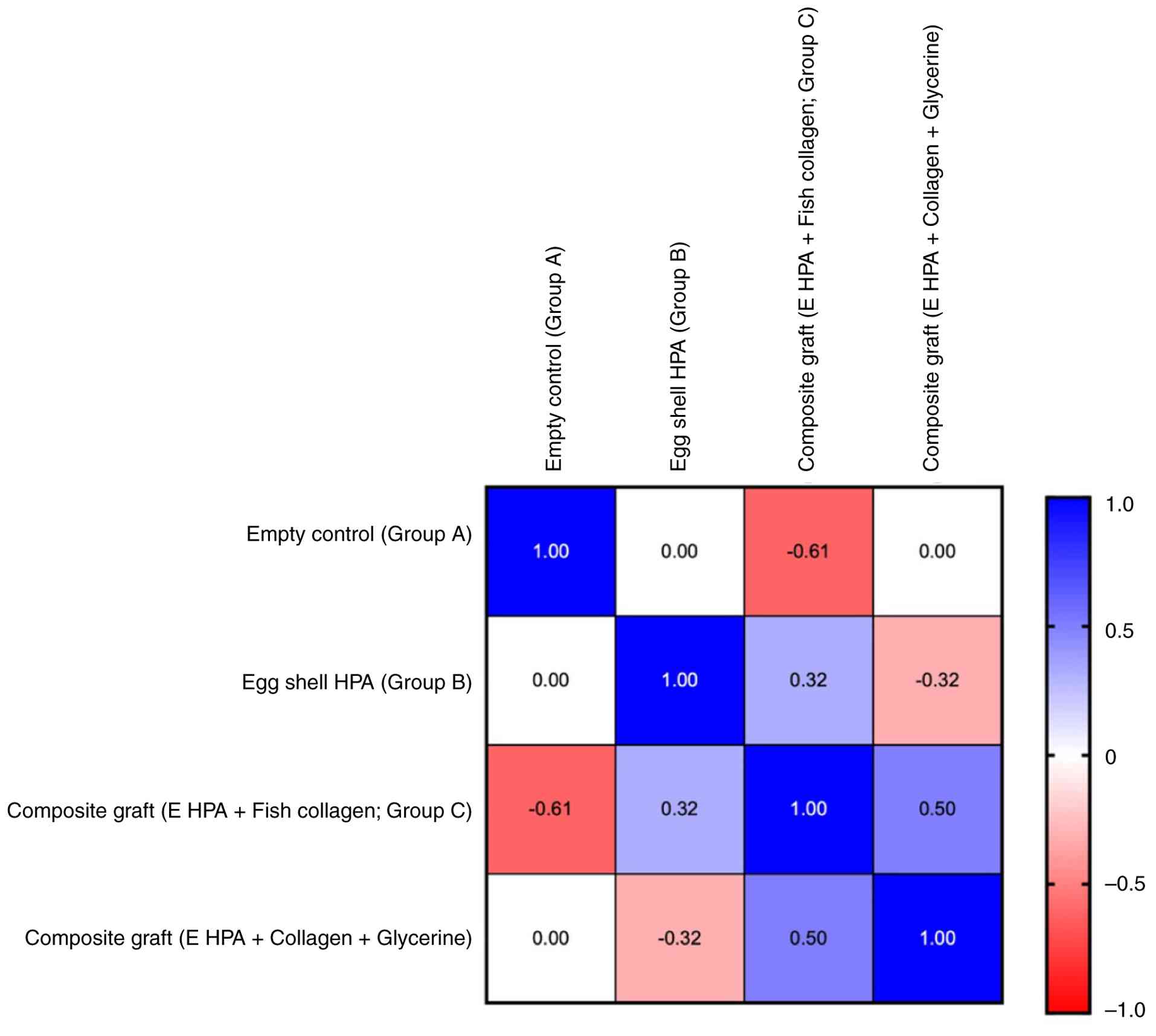
animals, no correlation was found between the inflammatory scores
produced by the different materials (Fig. 5). The animals tolerated the
surgical procedures well. No adverse reactions occurred during the
14-day study period. There was no difference in the mean
inflammatory score between the groups on the 14th day. Further
observations for 3 months period also did not reveal any adverse
reactions.
Discussion
The evaluation of the cytotoxicity and
biocompatibility of regenerative biomaterials is a preliminary and
crucial step before their effectiveness in regenerative medicine
can be assessed. Cytotoxicity refers to the ability of a material
to destroy cells, whereas biocompatibility is the capacity of the
material to perform its function without causing detrimental
effects in the body.
The three bone substitutes derived from eggshells,
eggshell-derived hydroxyapatite, EHPA modified with fish collagen,
and EHPA/collagen/Gly were found to be non-cytotoxic and their use
led to good cell viability, promoting the growth of L929
fibroblasts. No morphological changes were detected in the cells,
and minimal inflammatory infiltration was noted. Similar
observations were noted in other cell lines in other studies, as
follows:
In their study, Gutiérrez-Prieto et al
(21) reported a cell viability of
96% for experimental eggshell-derived hydroxyapatite modified with
silicon and poly lactic-co-glycolic (PLGA); 90% for bovine
bone-PLGA, and 86% for EHPA/PLGA in osteoblast cell lines.
Oladipupo et al (22)
performed an MTT assay on MG63 cells (osteosarcoma cell lines) and
ascertained that, by comparing a sample of EHPA with various
concentrations of ammonium bicarbonate, a sample that contained 0%
ammonium bicarbonate led to a viability of >85%. By contrast, in
EHPA with 40% ammonium bicarbonate, toxicity was detected (22).
In another study, direct-contact assays on
hydroxyapatite-β TCP/agarose disks revealed no cytotoxic effects on
L929 fibroblasts or human osteosarcoma cells (23). The adherence and proliferation of
both cell types on the biomaterial surface, maintaining their
characteristic morphology, were observed. Few transient changes in
several properties, such as cell cycle, size and complexity were
observed. Mild apoptotic changes were induced in Saos-2 osteoblasts
but not in fibroblasts (23).
These findings support the findings of the present study.
Ma et al (24) reported that eggshell-derived
amorphous calcium phosphate (ACP) particles displayed better
biocompatibility than ACP synthesised from synthetic hydroxyapatite
in three-dimensional osteoblastic spheroids. Another study on the
effects of eggshell granulate and calcium carbonate on bovine
osteoblasts revealed that additives (eggshell-derived calcium
carbonate) increased osteoblast activity. The cell cultures treated
with eggshells displayed the most potent effects, whereas for
hyaluronan, a weaker cell activity was detected (25).
Chuysinuan et al (26) conducted a primary cytotoxicity test
on MC3T3 cells and reported that the newly fabricated hydrogel, egg
shell hydroxyapatite incorporated fibroin alginate hydrogel, was
non-cytotoxic. He et al (27) also reported that the biomimetic
collagen composite matrix-hydroxyapatite scaffolds exhibited high
biocompatibility in critical-sized cranial defects in a rat
model.
Biocompatibility tests have also revealed that human
mesenchymal cells can infiltrate and remain viable after culture on
collagen-hydroxyapatite scaffolds and antibiotic-doped substrates
(ciprofloxacin and gentamycin) (28). Wang et al (29) established that the
collagen/glycerol/pullulan gel exhibited maximum cell attachment
and uniform cell distribution, with an improved morphology
displaying better extension and three-dimensional characteristics,
with sufficiently extended filamentous pseudopods. It was
hypothesized that glycerine and pullulan enhanced the physical
properties of the gel and also were conducive to cell attachment
and proliferation (29). The
present study also revealed that the incorporation of glycerine
does not negatively impact the cell viability of the material.
In the present study, at 1 week post-implantation,
no adverse skin reactions (Fig.
1B) were noted at all three sites. At 2 weeks, the initial
phagocytic activities of the cell were noted, followed by the
degradation process and subsequent reabsorption of the material. In
the implantation area, the particles were surrounded by collagen
fibres, with mild inflammatory infiltrate. A sparse number of
multinucleated cells and large blood vessels were also observed.
The minimum number of inflammatory cells was noted at all three
sites.
In the study by Prohl et al (30), the histological analysis of
inflammatory tissue reactions was compared between a novel material
based on a xenogenic bone substitute combined with hyaluronic acid
and another xenogenic material of similar composition and a sham
operation group. A 2 weeks post-implantation, moderate inflammatory
reactions were observed in all three study groups. No differences
were found between the groups with respect to pro-inflammatory and
anti-inflammatory cells (30).
The study by Markel et al (31) revealed that the HPA product
generated a low inflammatory reaction compared to demineralised
bone matrix. L929 cells, which are mouse fibroblast cell lines, due
to their pro-healing properties, are advantageous in regenerative
medicine. Hence, their use is highly recommended in studies
involving biomaterials, drug delivery and tissue regeneration.
The present study demonstrated that the
incorporation of additives, such as fish collagen and glycerol into
eggshell-derived hydroxyapatite did not have any negative impact on
the cell viability or biocompatibility of the materials. The
materials did not lead to any adverse skin reactions in the Wistar
rats; hence, their potential as bone regenerative materials should
be considered. Further in vivo studies on animal models and
humans are warranted to establish the regenerative potential of the
material.
The present study has certain limitations which
should be mentioned. The present study only evaluated the in
vitro cell viability of the materials and the tissue reactions
following their placement in the subcutaneous pouches of Wistar
rats. Other mechanical properties, such as compressive strength and
injectability, need to be assessed. The regenerative capacity of
the materials needs to be confirmed in vitro and in animal
models before performing any human studies.
In conclusion, in the present study, hydroxyapatite
derived from egg shells was used and then modified with fish
collagen and glycerine. The samples were evaluated for their cell
viability in mouse fibroblasts. All three materials were found to
be non-cytotoxic. The biocompatibility was tested in an animal
model, which established that the materials were biocompatible.
Further studies are required however, to prove their regenerative
properties and their application as bone substitutes.
Acknowledgements
The authors acknowledge the Yenepoya Pharmacy
College and Yenepoya Research Centre for providing technical
support, as well as Dr Ranajit Das, Yenepoya Research Center,
Yenepoya University (Mangalore, India) for his contribution in the
statistical analysis of the data.
Funding
Funding: The present study received financial support from
Yenepoya University (Project no. YU/Seed grant/130-2022).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
SP was involved in the conception of the study, in
acquisition and interpretation of data, and in the drafting of the
manuscript. RKS was involved in the conception of the study, in the
interpretation of data, in manuscript editing, and in the critical
evaluation of the study content. NGT was involved in the conception
of the study, in the interpretation of data, and in the evaluation
of the study content. SS conducted the experiments and was involved
in the acquisition of data. RA was involved in the acquisition of
data, in data interpretation, and in manuscript preparation. BPK
conducted the experiments and was involved in the conception of the
study. PMS was involved in data interpretation and manuscript
revision. SS and RA confirm the authenticity of all the raw data.
The manuscript has been read and approved by all the authors.
Ethics approval and consent to
participate
Ethical approval was obtained from the Institutional
Animal Ethics Committee (YU/IAEC/25/2022).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Bassi APF, Bizelli VF, Consolaro RB and de
Carvalho PSP: Biocompatibility and osteopromotor factor of bovine
integral Bone-A microscopic and histometric analysis. Front. Oral
Maxillofac. Med. 3:1–11. 2021.
|
|
2
|
Hosseinpour S, Gaudin A and Peters OA: A
critical analysis of research methods and experimental models to
study biocompatibility of endodontic materials. Int Endod J. 55
(Suppl 2):S346–S369. 2022.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Zhao R, Yang R, Cooper PR, Khurshid Z,
Shavandi A and Ratnayake J: Bone grafts and substitutes in
dentistry: A review of current trends and developments. Molecules.
26(3007)2021.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Gill S, Prakash M, Forghany M and
Vaderhobli RM: An ethical perspective to using bone grafts in
dentistry. J Am Dent Assoc. 153:88–91. 2020.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Dorj B, Won JE, Purevdorj O, Patel KD, Kim
JH, Lee EJ and Kim HW: A novel therapeutic design of
microporous-Structured biopolymer scaffolds for drug loading and
delivery. Acta Biomater. 10:1238–1250. 2014.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Patel KD, Kim TH, Mandakhbayar N, Singh
RK, Jang JH, Lee JH and Kim HW: Coating biopolymer nanofibers with
carbon nanotubes accelerates tissue healing and bone regeneration
through orchestrated Cell-and Tissue-regulatory responses. Acta
Biomater. 108:97–110. 2020.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Rather HA, Patel R, Yadav UCS and Vasita
R: Dual Drug-delivering Polycaprolactone-collagen scaffold to
induce early osteogenic differentiation and coupled angiogenesis.
Biomed. Mater. 15(45008)2020.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Suh H, Han D, Park J, Lee DH, Lee WS and
Han CD: A Bone replaceable artificial bone substitute:
Osteoinduction by combining with bone inducing agent. Artif Organs.
25:459–466. 2001.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Dupoirieux L, Pourquier D, Souyris FF,
Surgery M and Prof H, Surgery E and Prof H: Powdered eggshell: A
pilot study on a new bone substitute for use in maxillofacial
surgery. J Cranio Maxillofac Surg. 23:187–190. 1995.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Zhang Y, Xue C, Zhang Y, Zhang Q, Zhang K,
Liu Y, Shan Z, Qiu W, Chen G, Li N, et al: Cocktail effect of ionic
patch driven by triboelectric nanogenerator for diabetic wound
healing. Chin Chem Lett. 35(109196)2024.
|
|
11
|
Opris H, Bran S, Dinu C, Baciut M, Prodan
DA, Mester A and Baciut G: Clinical applications of avian
eggshell-derived hydroxyapatite. Bosn J Basic Med Sci. 20:430–437.
2020.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Park JW, Bae SR, Suh JY, Lee DH, Kim SH,
Kim H and Lee CS: Evaluation of bone healing with eggshell-derived
bone graft substitutes in rat calvaria: A pilot study. J Biomed
Mater Res. 87:203–214. 2008.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Keil C, Gollmer B, Zeidler-Rentzsch I,
Gredes T and Heinemann F: Histological evaluation of extraction
sites grafted with bio-Oss collagen: Randomised controlled trial.
Ann Anat. 237(151722)2021.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Ryan AJ, Gleeson JP, Matsiko A, Thompson
EM and O'Brien FJ: Effect of different hydroxyapatite incorporation
methods on the structural and biological properties of porous
collagen scaffolds for bone repair. J Anat. 227:732–745.
2015.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Furtado M, Chen L, Chen Z, Chen A and Cui
W: Development of fish collagen in tissue regeneration and drug
delivery. Engineered Regeneration. 3:217–231. 2022.
|
|
16
|
Food and Drug Administration. Glycerine.
182(1320)2016.https://www.law.cornell.edu/cfr/text/21/182.1320.
|
|
17
|
Low KL, Tan SH, Zein SH, Roether JA,
Mouriño V and Boccaccini AR: Calcium phosphate-based composites as
injectable bone substitute materials. J Biomed Mater Res B Appl
Biomater. 94:273–286. 2010.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Prathap S, Rajesh KS, Thomas NG,
Venkateshan J, Prathap MS and Surya S: Domestic chicken
Eggshell-Derived bone substitute: Synthesis, characterisation, and
in vitro cell viability. J Pharm Bioallied Sci. 16 (Suppl
4):S3116–S3119. 2024.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Mosmann T: Rapid colorimetric assay for
cellular growth and survival: Application to proliferation and
cytotoxicity assays. J Immunol Methods. 65:55–63. 1983.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Yaltirik M, Ozbahar H, Bilgic B and
Issever H: Reactions of connective tissue to mineral trioxide
aggregate and amalgam. J Endod. 30:95–99. 2004.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Gutiérrez-Prieto SJ, Fonseca LF,
Sequeda-Castañeda LG, Díaz KJ, Castañeda LY, Leyva-Rojas JA,
Salcedo-Reyes JC and Acosta AP: Elaboration and biocompatibility of
an Eggshell-derived hydroxyapatite material modified with Si/PLGA
for bone regeneration in dentistry. Int J Dent.
2019(5949232)2019.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Oladipupo OF, Adekola AH, Ofudje EA,
Al-Ahmary KM, Al-Mhyawi SR, Alshdoukhi IF, Alrahili MR and Alsaiari
AA: Eggshell-derived scaffold of hydroxyapatite-ammonium
bicarbonate nanocomposite: Bioactivity and cytotoxicity studies.
Heliyon. 10(e36493)2024.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Alcaide M, Serrano MC, Pagani R,
Sánchez-Salcedo S, Vallet-Regí M and Portolés MT: Biocompatibility
markers for the study of interactions between osteoblasts and
composite biomaterials. Biomaterials. 30:45–51. 2009.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Ma Q, Rubenis K, Sigurjónsson OE,
Hildebrand T, Standal T, Zemjane S, Locs J, Loca D, Jostein H and
Haugen HJ: Eggshell-derived amorphous calcium phosphate: Synthesis,
characterisation, and biofunctions as bone graft materials in novel
3D osteoblastic spheroids model. Smart Materials Med. 4:522–537.
2023.
|
|
25
|
Neunzehn J, Szuwart T and Wiesmann HP:
Eggshells as natural calcium carbonate source in combination with
hyaluronan as beneficial additives for bone graft materials, an in
vitro study. Head Face Med. 11(12)2015.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Chuysinuan P, Nooeaid P, Thanyacharoen T,
Techasakul S, Pavasant P and Kanjanamekanant K: Injectable
eggshell-derived hydroxyapatite-incorporated fibroin-alginate
composite hydrogel for bone tissue engineering. Int J Biol
Macromol. 193:799–808. 2021.PubMed/NCBI View Article : Google Scholar
|
|
27
|
He H, Wang L, Cai X, Wang Q, Liu P and
Xiao J: Biomimetic collagen composite matrix-hydroxyapatite
scaffold induces bone regeneration in critical size cranial
defects. Materials Design. 236(112510)2023.
|
|
28
|
Filip Ionescu OL, Mocanu AG, Neacşu IA,
Ciocilteu MV, Rău G and Neamţu J: Biocompatibility studies on a
Collagen-hydroxyapatite biomaterial. Curr Health Sci J. 48:217–225.
2022.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Wang X, Komasa S, Tahara Y, Inui S,
Matsumoto M and Maekawa K: Novel injectable
Collagen/Glycerol/Pullulan gel promotes osteogenic differentiation
of mesenchymal stem cells and the repair of rat cranial defects.
Gels. 10(775)2024.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Prohl A, Batinic M, Alkildani S, Hahn M,
Radenkovic M, Najman S, Jung O and Barbeck M: In vivo analysis of
the biocompatibility and bone healing capacity of a novel bone
grafting material combined with hyaluronic acid. Int J Mol Sci.
22(4818)2021.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Markel DC, Guthrie ST, Wu B, Song Z and
Wooley PH: Characterisation of the inflammatory response to four
commercial bone graft substitutes using a murine biocompatibility
model. J Inflamm Res. 5:13–18. 2012.PubMed/NCBI View Article : Google Scholar
|