Introduction
Chronic kidney disease (CKD) is a disorder in which
the ability of the kidneys to function slowly deteriorates over the
course of >3 months. This complicates the ability of the body to
eliminate waste and maintain fluid and electrolyte balance. CKD
frequently progresses to end-stage renal disease (ESRD), which
requires dialysis or kidney transplantation (1-3).
The increased global prevalence of chronic kidney
disease is largely due to the convergence of demographic and
epidemiological trends. CKD is growing more prevalent in numerous
parts of the world. The primary reasons for this include an
increase in the number of elderly individuals, a higher life
expectancy and an increase in the incidence of non-communicable
diseases, such as diabetes and high blood pressure. More people
with heart disease are living longer lives, using kidney-damaging
medications, and developing inflammatory and metabolic diseases.
This has led to an increase in the number of cases with CKD,
particularly in low- and middle-income nations (1-4).
Hepatitis C virus (HCV) infection presents a
distinct risk to individuals with CKD, particularly those receiving
hemodialysis, due to markedly higher prevalence rates compared to
the general population (5).
Researchers indicate that CKD and HCV infection are becoming
increasingly linked. Patients with CKD are more likely to become
infected with HCV as their immune systems are out of balance, they
have vascular access created or replaced more than once, and they
are often surrounded by healthcare workers, particularly when they
undergo hemodialysis. Chronic HCV infection, conversely, may
expedite the advancement of CKD and exacerbate cardiovascular
outcomes by eliciting prolonged systemic inflammation, endothelial
dysfunction and metabolic disturbance (6,7).
Platelets become active and release platelet factor
4 (PF4; also known as CXCL4) from their alpha granules. Regulated
upon activation, normal T-cell expressed and secreted (RANTES; also
known as CCL5), is a CC chemokine produced by T-cells, platelets
and other cells that reside in tissues (8,9). PF4
and RANTES are chemokines that are critical in controlling the
movement and activation of immune cells. PF4 mainly changes how
monocytes, macrophages, neutrophils, natural killer cells and
T-lymphocytes differentiate, move and become active. Depending on
the amount of PF4 present and the type of cell, it can be involved
in both pro-inflammatory and anti-inflammatory processes (9). When platelets are stimulated, they
produce PF4, which can interact with other chemokines, such as
RANTES. This facilitates the movement of white blood cells, such as
monocytes, to sites of inflammation (10). RANTES is a strong chemoattractant
for memory T-cells, eosinophils, monocytes and other immune cells.
It is essential for delivering white blood cells to inflamed
regions, enhancing immunological responses, and regulating both
innate and adaptive immunity (8).
PF4 and RANTES are key chemicals that link platelet activation to
immune cell recruitment and the spread of inflammatory responses in
both healthy and diseased states (8-10).
Although there is strong evidence linking CKD and
HCV infection to systemic inflammation and poor cardiovascular
outcomes, the platelet-mediated immuno-inflammatory processes that
enhance this connection are not yet fully understood. The majority
of prior research has concentrated on conventional inflammatory
markers or platelet counts, with insufficient emphasis on
platelet-derived chemokines that directly facilitate immune cell
recruitment and vascular inflammation (6,7,10,11).
Data on the behavior of PF4 and RANTES in patients with CKD
co-infected with HCV are limited (12-14),
particularly in populations undergoing hemodialysis, where platelet
activation and viral infection occur concurrently. Addressing this
gap has therapeutic significance, as identifying
platelet-associated inflammatory markers may improve risk
classification and shed light on the mechanisms driving the
increased thrombo-inflammatory load in this vulnerable population.
Therefore, the present study aimed to investigate serum PF4 and
RANTES levels in patients with CKD with and without HCV infection,
highlighting their potential role in the immunopathogenesis of CKD
and HCV co-infection.
Patients and methods
Study population
A case control study was conducted on serum samples
from 90 patients with CKD and positive for HCV, 90 patients with
CKD and negative for HCV and 90 apparently healthy individuals as
the control group. Samples were collected from the Dialysis Center
Laboratory at Al-Imamain Al-Kadhimain Medical City in Baghdad, Iraq
after obtaining official permission from the hospital. The study
samples were collected from patients and healthy controls between
November, 2024 and May, 2025. Patients with HCV were classified
based on the results of tests performed on patients in the dialysis
center laboratory. All patients underwent HCV testing as part of
routine checkups at dialysis centers. The standard diagnosis for
HCV at these centers is the presence of antibodies to the HCV
(anti-HCV antibodies) in the serum of patients, which is determined
using enzyme linked immunosorbent assay (ELISA) technology. All
patients with CKD had stage 5 disease (ESRD) and were undergoing
regular hemodialysis. The inclusion criteria for the patients were
adult patients (≥18 years) with ESRD on maintenance hemodialysis,
while the exclusion criteria included those with acute infections,
autoimmune diseases, malignancy and recent blood transfusion.
Detailed information regarding CKD etiology and
comorbidities was not uniformly available. The age of the patients
with CKD ranged from 30 to 85 years, with a median age of 50 years.
PF4 and RANTES levels were measured for all samples using ELISA.
The manufacturer of the kits used in the present study was
Elabscience (cat. no. E-EL-H6184 for PF4 and cat. no. E-EL-H6179
for RANTES).
The present study was approved by the Ethics
Committee of Ibn Sina University of Medical and Pharmaceutical
Sciences, Baghdad, Iraq (Approval no. ISU14.10.24). Written
informed consent was obtained from all participants prior to sample
collection.
Statistical analysis
Data analysis was performed using the Statistical
Package for Social Sciences (SPSS) software, version 26 (IBM Corp.)
and Microsoft Excel 2019 (Microsoft Corp.). The normality of data
distribution was assessed using the Shapiro-Wilk test. Parametric
tests, including the one-way ANOVA with the LSD post hoc test were
used for normally distributed variables. The Chi-squared test was
used to assess associations between categorical variables. A
P-value <0.05 was considered to indicate a statistically
significant difference.
Results
The sex distribution among three groups (control,
CKD without HCV and CKD with HCV) is demonstrated in Table I. There was a statistically
significant difference between the groups (P=0.005). The highest
proportion of females was found in the control group (43.1%),
whereas males were more frequent in the CKD without HCV group and
CKD with HCV group (each 37.9%).
 | Table IDistribution of sex across the study
groups. |
Table I
Distribution of sex across the study
groups.
| | Groups | |
|---|
| Sex | Controls (%) | CKD negative C
(%) | CKD positive C
(%) | Total (%) | Chi-squared test
P-value |
|---|
| Male | 34 (24.3) | 53 (37.9) | 53 (37.9) | 140 (51.9) | 0.005 |
| Female | 56 (43.1) | 37 (28.5) | 37 (28.5) | 130 (48.1) | |
| Total | 90 (33.3) | 90 (33.3) | 90 (33.3) | 270 (100.0) | |
As regards age, the results revealed that the
control group had a significantly lower mean age (38.17±15.525
years) compared to both the CKD without HCV group and the CKD with
HCV group (52.19±11.161 years each). The analysis revealed a highly
significant difference among the groups (F=36.104, P=0.0001)
(Table II).
 | Table IIMean age distribution across the study
groups. |
Table II
Mean age distribution across the study
groups.
| Groups | Mean ± SD
deviation | F (P-value) |
|---|
| Control | 38.17±15.525 | 36.104 (0.0001) |
| CKD with negative
C | 52.19±11.161 | |
| CKD with positive
C | 52.19±11.161 | |
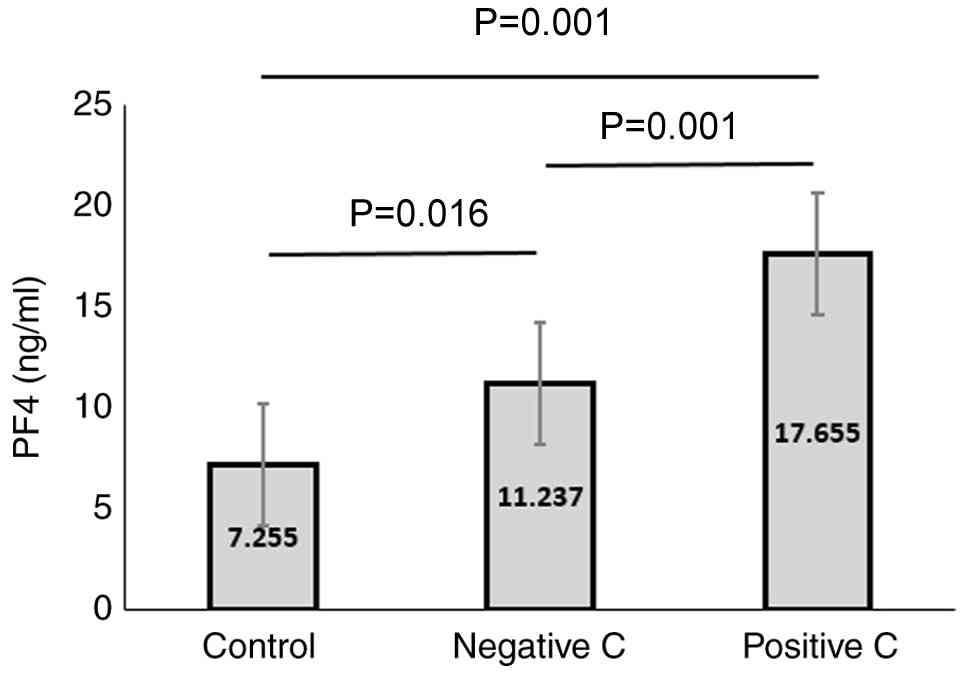
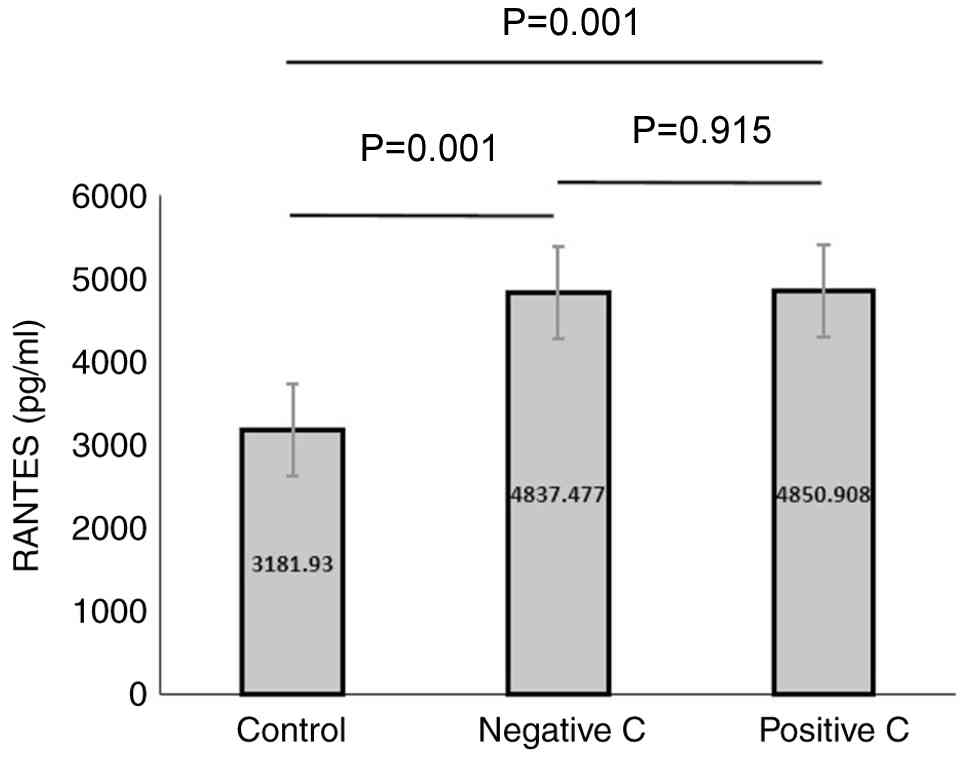
The analysis of PF4 levels revealed statistically
significant differences between all three groups. PF4 levels were
significantly higher in the CKD without HCV group compared to the
control group, and were even higher in the CKD with HCV group.
Additionally, the PF4 levels were significantly higher in the CKD
with HCV group than in CKD without HCV group. A similar was
observed for the RANTES levels (Figs.
1 and 2, and Tables III and IV).
 | Table IIIComparison of mean PF4 and RANTES
levels among the groups. |
Table III
Comparison of mean PF4 and RANTES
levels among the groups.
| Groups | PF4 (ng/ml) | RANTES (pg/ml) |
|---|
| Control | | |
|
Mean | 7.255 | 3181.930 |
|
Std.
deviation | 3.843 | 1461.163 |
| CKD with negative
C | | |
|
Mean | 11.237 | 4837.477 |
|
Std.
deviation | 12.489 | 104.600 |
| CKD with positive
C | | |
|
Mean | 17.655 | 4850.908 |
|
Std.
deviation | 13.92 | 23.629 |
| F (p-value) | 20.391 (0.0001) | 115.860 (0.0001) |
 | Table IVPost hoc multiple comparisons of PF4
and RANTES between the groups. |
Table IV
Post hoc multiple comparisons of PF4
and RANTES between the groups.
| Dependent
variable | Groups | Mean
difference | Sig. |
|---|
| PF4 | Control vs.
Negative C |
-3.982556a | 0.016 |
| | Control vs.
Positive C |
-10.400478a | 0.000 |
| | Negative C vs.
Positive C |
-6.417922a | 0.000 |
| RANTES | Control vs.
Negative C |
-1,655.546867a | 0.000 |
| | Control vs.
Positive C |
-1,668.978533a | 0.000 |
| | Negative C vs.
Positive C | -13.431667 | 0.915 |
Discussion
People with CKD have severe immune dysregulation,
which is evidenced by issues with both their innate and adaptive
immune responses, ongoing systemic inflammation, and issues with
their platelets. Uremia causes immune exhaustion, repeated vascular
access and frequent exposure to extracorporeal circulation in
hemodialysis settings, all of which render these patients more
likely to acquire viral infections and maintain ongoing chronic
inflammation. In this case, chemokines that are produced from
platelets are a critical, yet not extensively studied link between
immune system issues and increased inflammation. The present study
opted to investigate PF4 and RANTES (CCL5), since platelets produce
a large quantity of these chemokines when activated. They are also
vital for bringing white blood cells into the body, activating
immune cells and combating infections. PF4 and RANTES are essential
markers for exploring the molecular immunological link between
chronic HCV infection and CKD, due to their participation in
thrombo-inflammatory processes and viral pathogenesis. PF4 and
RANTES are not like other inflammatory markers; they indicate that
platelets are engaging the immune system. This is a procedure that
is becoming increasingly significant in CKD to treat inflammation
and blood vessel issues (4,10,12,15).
Patients with CKD are more prone to become infected
with HCV infection, which increases their risk of mortality and
illness due to complicated immunological and inflammatory pathways
(16,17). Previous studies have found that
patients with HCV infection have worse cardiovascular outcomes and
are more likely to develop CKD (18,19).
Both CKD and viral infections are largely caused by inflammation
and immunological responses, which are mediated by the chemokines
PF4 and RANTES, which are released upon platelet activation
(11,16). Research explicitly examining how
HCV infection affects blood levels of these chemokines in patients
with CKD is still lacking, despite the known roles of PF4 and
RANTES in inflammatory conditions. The majority of the literature
to date has either examined inflammatory markers separately in CKD
or HCV populations or concentrated on the prevalence and clinical
effects of HCV in CKD (11,17,19).
Serum PF4 and RANTES levels in control subjects,
patients with CKD without HCV, and patients with CKD with HCV
infection have not, to date, to the best of our knowledge, been
compared in any previous study. In order to clarify the combined
impact of CKD and HCV on platelet-mediated inflammation and
possibly identify novel biomarkers or therapeutic targets in this
clinically susceptible population, the present study aimed to fill
the gap by assessing these biomarkers across these various
groups.
Known epidemiological patterns of CKD and HCV
infection can account for the observed sex distribution differences
among the three groups in the present study (the control, patients
with CKD without HCV, and patients with CKD with HCV); there was a
higher proportion of females in the control group and a male
predominance in the CKD groups. Males with the prevalence of HCV
infection is greater in CKD, according to several studies (12,14,18,20).
Male predominance in HCV-positive patients, for instance, has been
reported in studies on CKD populations in India and Pakistan. This
is attributed to higher rates of underlying CKD risk factors in
men, such as diabetes and hypertension, as well as potential
increased exposure risk to HCV through behaviors or
healthcare-related factors in men (13,20,21).
In a similar vein, a sizable population-based study conducted in
Taiwan discovered that, in addition to advanced age and other
comorbidities, the male sex was a substantial independent risk
factor for CKD in patients with HCV (22).
In the present study, the larger percentage of women
in the control group may be a result of the demographic makeup of
the general population sample, as well as the distinct risk factor
profiles for HCV infection and CKD. Notably, while the prevalence
of HCV infection tends to be higher in males across CKD
populations, some studies have also documented interactions where
females with HCV may have worse renal outcomes (such as
post-transplant chronic renal failure) (6,23).
Herein, the mean age of the control group
(38.17±15.53 years) was significantly lower than that of both CKD
groups (52.19±11.16 years), and the difference was highly
significant (F=36.104, P=0.0001). This is in line with established
epidemiological evidence indicating that the prevalence of HCV
infection and CKD increases with age. According to numerous
studies, CKD primarily affects middle-aged and older individuals,
and its incidence significantly increases beyond the age of 50
years. In elderly CKD populations, HCV infection is also more
prevalent and has a larger clinical impact. For instance,
HCV-seropositive patients were considerably older than HCV-negative
people and had a greater prevalence of CKD, particularly in those
>45 years of age, according to a large population-based study
conducted in Taiwan (22). The
majority of patients with CKD, whether HCV-positive or -negative,
were between the ages of 35 and 50 years, according to another
Indian study, and the prevalence of HCV was highest in these
middle-aged groups (13).
Additionally, this age-associated risk is influenced by
pathophysiological factors, such cumulative kidney injury, extended
exposure to viral infections, or comorbidities (e.g., diabetes and
hypertension) (11,14). Therefore, the greater age in CKD
groups (independent of HCV status) corresponds with the higher risk
and progression of renal disease and chronic infections observed in
later life, whereas the younger age in the controls indicates the
lack of CKD and HCV-related comorbidities.
A progressive increase in platelet activation and
inflammatory chemokine release linked to both CKD and HCV infection
is suggested by the finding that PF4 levels were lowest in healthy
controls, significantly elevated in patients with CKD without HCV,
and highest in the patients with CKD with HCV. These findings are
also reflected by the RANTES levels, and all groups exhibited
statistically significant differences. Cortical roles in
inflammatory and immunological processes are played by the
chemokines, PF4 and RANTES, which are held in platelet alpha
granules and released upon platelet activation. Chronic
inflammation, endothelial dysfunction and an altered hemostasis are
known to activate platelets in CKD, which increases PF4 and RANTES
levels compared to healthy individuals (7,9). The
additional inflammatory and immunological stimulation brought on by
HCV infection is probably the cause of the further rise in PF4 and
RANTES observed in HCV-infected CKD patients. Chronic inflammation
and systemic immunological activation brought on by HCV are known
to aggravate kidney damage and promote platelet activation, both of
which increase the release of chemokines (11,24).
In addition, immune-complex deposition and direct viral effects on
kidney tissue aggravate HCV-related kidney disease by promoting the
release of inflammatory chemokines such as PF4 and RANTES platelets
from immune cells (24). In the
present study, the detailed virological characteristics of HCV
infection, including genotype, viral load, and antiviral treatment
status, were not uniformly available and therefore could not be
analyzed. This may exacerbate the course of CKD by increasing
vascular inflammation. The levels of these chemokines gradually
increased from the controls to those with CKD without HCV and those
with CKD with HCV, which is in line with data form a previous study
demonstrating that persistent HCV infection causes kidney damage
and systemic inflammation, apart from the inflammatory milieu of
CKD alone (25). These
associations are discussed in the context of biological
plausibility and previously published evidence rather than direct
statistical testing in the present study. Inflammation and platelet
activation are central features of both CKD and chronic HCV
infection. Previous studies have consistently reported elevated
levels of inflammatory markers, such as C-reactive protein, IL-6
and TNF-α in CKD and HCV, reflecting persistent immune activation
and endothelial dysfunction. In parallel, platelet-related
abnormalities, including increased platelet activation and release
of platelet-derived mediators, have been described in these
conditions. Within this inflammatory milieu, PF4 and RANTES
represent platelet-derived chemokines that link platelet activation
with immune cell recruitment. The elevated PF4 and RANTES levels
observed in the present study are therefore consistent with the
broader pattern of heightened inflammation and platelet involvement
reported in CKD and HCV, supporting their potential role as
complementary biomarkers of thrombo-inflammatory activity in this
population (11,16).
On the whole, the results of the present study are
consistent with earlier research demonstrating that inflammation
causes platelet-derived chemokines to rise in CKD and that HCV
infection intensifies this systemic inflammatory response, leading
to additional elevations in PF4 and RANTES levels. This lends
credence to the notion that HCV exacerbates inflammatory pathways
and platelet activation in patients with CKD.
Even though immune-mediated platelet destruction,
bone marrow suppression, the impaired liver production of
thrombopoietin and hypersplenism leading to platelet sequestration
are some of the mechanisms that cause thrombocytopenia (decreased
platelet count) in chronic HCV infection (12,15,26),
the serum levels of platelet-derived chemokines, such as PF4 and
RANTES can still be increased, despite the lower platelet count and
this case can be explained by several factors.
The remaining circulating platelets in chronic
inflammatory states tend to be hyperactivated, despite the fact
that overall platelet counts decline in HCV infection. Despite
thrombocytopenia, activated platelets can increase the serum levels
of PF4 and RANTES by releasing these chemokines from their alpha
granules into the bloodstream (15,27).
HCV infection causes autoimmune reactions and the creation of
immune complexes against platelets, which destroys the platelets,
but also activates them before they are cleared. Ironically, the
immune system’s targeting of platelets may temporarily or in
subsets of platelets boost the release of chemokines (26,27).
Even though chronic HCV infection often causes thrombocytopenia, a
decrease in the number of platelets does not always indicate that
the platelets are less active. In inflammatory conditions, such as
CKD and HCV infection, the residual circulating platelets are
frequently hyperreactive and demonstrate increased degranulation
capability. When these hyperactivated platelets are activated, they
can release numerous platelet-derived chemokines, such as PF4 and
RANTES, from their alpha granules. Furthermore, immune-mediated
platelet destruction and faster platelet turnover may render
younger, more reactive platelets with more secretory potential more
likely to respond. In the case of CKD and HCV co-infection, ongoing
systemic inflammation and endothelial dysfunction may cause
platelets to become even more active, maintaining chemokine levels
high even though there are fewer platelets. This observation
highlights a critical pathophysiological notion: the functionality
of platelets, not just their number, may be a key factor in the
thrombo-inflammatory burden in patients with CKD who are also
infected with another disease. This provides new information
regarding the increased risk of inflammation and heart disease in
patients with CKD (12,15).
Even though liver disease and bone marrow
suppression decrease platelet production, thrombopoietin levels can
occasionally rise in response, encouraging megakaryocytes to create
younger, more reactive platelets that release more PF4 and RANTES
when activated (12,27). Elevated systemic levels of PF4 and
RANTES can be found in serum when platelets in inflammatory
vascular beds or organs (such the kidneys in CKD or the liver in
HCV) become activated and release these chemicals locally (28).
In summary, thrombocytopenia in chronic HCV is
characterized by a lower platelet count mostly as a result of
increased destruction and decreased formation; yet, the remaining
or newly formed platelets are hyperactivated, which results in
increased PF4 and RANTES secretion. This explains why in patients
infected with HCV, PF4 and RANTES levels increase even when their
platelet counts are low.
The findings of elevated PF4 and RANTES levels in
patients with CKD co-infected with HCV raise several important
avenues for future research and potential clinical application.
Given their close association with platelet activation and
inflammation, PF4 and RANTES may serve as candidate biomarkers for
identifying CKD-HCV patients at an increased risk of
thrombo-inflammatory complications, including cardiovascular
events. Further research is required to determine whether
investigating these chemokines over time can help to identify who
is at a higher risk more accurately than traditional inflammatory
indicators. The role of platelet-derived chemokines suggests that
altering the way platelets function or chemokines signal may be
beneficial in treating patients. Some of these treatments could
include improved antiplatelet procedures, tailored
anti-inflammatory medicines, or methods for limiting the number of
times platelets and immune cells come into contact. More
mechanistic and interventional research are needed to determine
whether addressing PF4- and RANTES-mediated pathways can improve
the health of this high-risk group by increasing inflammation.
In conclusion, the present study demonstrates that
patients with CKD and HCV infection have markedly higher levels of
the platelet-derived chemokines, PF4 and RANTES, in their
circulation than those with CKD without HCV. This is due to the
fact that the virus damages platelets, increasing the likelihood of
inflammation. The increased chemokine levels found in the
co-infected group suggests that HCV infection may exacerbate the
pre-existing inflammatory milieu in CKD. These findings shed light
on putative immuno-inflammatory processes that could explain the
increased illness risk in patients with CKD and HCV co-infection.
They also support previous research on how PF4 and RANTES impact
the immune system and produce inflammation via platelets (9,29-32).
The present study did not examine platelet numbers; however, the
findings support the concept that platelet activation pathways
contribute to systemic inflammation in this population. The present
study addresses a fundamental gap in the understanding of the
interactions between HCV infection, CKD and platelet-derived
inflammatory biomarkers. It indicates that further longitudinal and
mechanistic studies are necessary to ascertain their potential
prognostic and therapeutic effects.
Acknowledgements
The authors would like to thank the staff of the
Dialysis Center Laboratory at Al-Imamain Al-Kadhimain Medical City
in Baghdad, Iraq for their support in sample collection.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
HHL was involved in the conceptualization of the
study, as well as in the study methodology, data collection, and in
the writing of the original draft of the manuscript. AMJA was
involved in data analysis and interpretation, and in the writing,
reviewing and editing of the manuscript. Both authors have read and
approved the final manuscript. HHL and AMJA confirm the
authenticity of all the raw data.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of Ibn Sina University of Medical and Pharmaceutical
Sciences, Baghdad, Iraq (Approval no. ISU14.10.24). Written
informed consent was obtained from all participants prior to sample
collection.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Guo J, Jiao W, Xia S, Xiang X, Zhang Y, Ge
X and Sun Q: The global, regional, and national patterns of change
in the burden of chronic kidney disease from 1990 to 2021. BMC
Nephrol. 26(136)2025.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Francis A, Harhay MN, Ong ACM,
Tummalapalli SL, Ortiz A, Fogo AB, Fliser D, Roy-Chaudhury P,
Fontana M, Nangaku M, et al: Chronic kidney disease and the global
public health agenda: An international consensus. Nat Rev Nephrol.
20:473–485. 2024.PubMed/NCBI View Article : Google Scholar
|
|
3
|
GBD Chronic Kidney Disease Collaboration.
Global, regional, and national burden of chronic kidney disease,
1990-2017: A systematic analysis for the global burden of disease
study 2017. Lancet. 395:709–733. 2020.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Lv JC and Zhang LX: Prevalence and disease
burden of chronic kidney disease. Adv Exp Med Biol. 1165:3–15.
2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Alshwayyat S, Hanifa H, Almasri N, Taimeh
Z, Alshwaiyat Y, Khdair T, Alfawares M, Alsaleh B and Assaf N:
Hepatitis C virus among hemodialysis patients: Burden, risks, and
prevention in resource-limited regions. Int Urol Nephrol.
57:3725–3731. 2025.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Fabrizi F, Donato FM and Messa P:
Association between hepatitis C virus and chronic kidney disease: A
systematic review and meta-analysis. Ann Hepatol. 17:364–391.
2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Hsu CN, Liao WT, Chen WL, Chang-Chien GP,
Lin S and Tain YL: Plasma and urinary platelet factor 4 as
biomarkers for cardiovascular risk in children with chronic kidney
disease. Biomedicines. 11(3318)2023.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Crawford A, Angelosanto JM, Nadwodny KL,
Blackburn SD and Wherry EJ: A role for the chemokine RANTES in
regulating CD8 T cell responses during chronic viral infection.
PLoS Pathog. 7(e1002098)2011.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Ji Y, Zhang Q, Li H, Chen L, Wu Y and Lin
S: Platelet factor 4: A mysterious chemokine in inflammatory
regulation diseases. J Inflamm Res. 18:4481–4495. 2025.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Rayes J, Bourne JH, Brill A and Watson SP:
The dual role of platelet-innate immune cell interactions in
thrombo-inflammation. Res Pract Thromb Haemost. 4:23–35.
2020.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Azmi AN, Tan SS and Mohamed R: Hepatitis C
and kidney disease: An overview and approach to management. World J
Hepatol. 7:78–92. 2015.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Rawi S and Wu GY: Pathogenesis of
thrombocytopenia in chronic HCV infection: A review. J Clin Transl
Hepatol. 8:184–191. 2020.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Gupta A, Sharma S and Nirala A: Prevalence
of hepatitis C infection among patients of chronic kidney disease
in a tertiary care hospital of Western Uttar Pradesh. J Indian Acad
Clin Med. 25:177–180. 2024.
|
|
14
|
Sánchez-Rodríguez CC, Galindo-Contreras
OG, Mendoza-Martínez AM, Sánchez-Nuncio HR and Luna-Domínguez JH:
Chronic kidney disease and hepatitis C virus. Ann Hepatol
[Internet]. 2025;30(S1). Available from: https://www.elsevier.es/es.
|
|
15
|
Dahal S, Upadhyay S, Banjade R, Dhakal P,
Khanal N and Bhatt VR: Thrombocytopenia in patients with chronic
hepatitis C virus infection. Mediterr J Hematol Infect Dis.
9(e2017019)2017.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Sohal A, Singh C, Bhalla A, Kalsi H and
Roytman M: Renal manifestations of chronic hepatitis C: A review. J
Clin Med. 13(5536)2024.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Awan AA, Jadoul M and Martin P: Hepatitis
C in chronic kidney disease: An overview of the KDIGO guideline.
Clin Gastroenterol Hepatol. 18:2158–2167. 2020.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Park H, Chen C, Wang W, Henry L, Cook RL
and Nelson DR: Chronic hepatitis C virus (HCV) increases the risk
of chronic kidney disease (CKD) while effective HCV treatment
decreases the incidence of CKD. Hepatology. 67:492–504.
2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Cacoub P, Desbois AC, Isnard-Bagnis C,
Rocatello D and Ferri C: Hepatitis C virus infection and chronic
kidney disease: Time for reappraisal. J Hepatol. 65(1
Suppl):S82–S94. 2016.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Karim R, Mohammad N, Khan MW, Ayaz M, Nabi
A and Khursheed K: Prevalence of hepatitis C virus infection among
chronic kidney disease patients on regular hemodialysis. J Gandhara
Med Dent Sci. 10:21–24. 2023.
|
|
21
|
Zahoor N, Malik A and Zaman M: Prevalence
of HCV among hemodialysis patients at a kidney center, Gujrat: A
gender-based comparative analysis. IJCMCR 2025; 51 [Internet].
2025;51(5):1-5. Available from: doi:
10.46998/IJCMCR.2025.51.001274.
|
|
22
|
Li WC, Lee YY, Chen IC, Wang SH, Hsiao CT
and Loke SS: Age and gender differences in the relationship between
hepatitis C infection and all stages of Chronic kidney disease. J
Viral Hepat. 21:706–715. 2014.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Ip S, Hussaini T, Daulat A, Nalufar P, Erb
SR, Yoshida EM and Marquez V: Interaction of gender and hepatitis C
in risk of chronic renal failure after liver transplantation. Ann
Hepatol. 16:230–235. 2017.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Barsoum RS, William EA and Khalil SS:
Hepatitis C and kidney disease: A narrative review. J Adv Res.
8:113–130. 2017.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Zhang H, Xu H, Wu R, Yu G, Sun H, Lv J,
Wang X, Chi X, Gao X, Kong F, et al: Association of hepatitis C and
B virus infection with CKD and impact of hepatitis C treatment on
CKD. Sci Rep. 9(1910)2019.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Fouad YM: Chronic hepatitis C-associated
thrombocytopenia: Aetiology and management. Trop Gastroenterol.
34:58–67. 2013.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Nurwidda ADP, Setiawan PB, Nusi IA,
Purbayu H, Sugihartono T, Maimunah U, Kholili U, Widodo B, Vidyani
A, Miftahussurur M and Thamrin H: Thrombocytopenia in chronic
hepatitis C. In: Proceedings of Surabaya International Physiology
Seminar (SIPS 2017), pp446-452.
|
|
28
|
Mihăilă RG: A look at platelet count in
chronic hepatitis C infection. EM J Hepatol. 5:97–103. 2017.
|
|
29
|
von Hundelshausen P, Koenen RR, Sack M,
Mause SF, Adriaens W, Proudfoot AEI, Hackeng TM and Weber C:
Heterophilic interactions of platelet factor 4 and RANTES promote
monocyte arrest on endothelium. Blood. 105:924–930. 2005.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Sonmez O and Sonmez M: Role of platelets
in immune system and inflammation. Porto Biomed J. 2:311–314.
2017.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Martí F, Bertran E, Llucià M, Villén E,
Peiró M, Garcia J and Rueda F: Platelet factor 4 induces human
natural killer cells to synthesize and release interleukin-8. J
Leukoc Biol. 72:590–597. 2002.PubMed/NCBI
|
|
32
|
Baltus T, von Hundelshausen P, Mause SF,
Buhre W, Rossaint R and Weber C: Differential and additive effects
of platelet-derived chemokines on monocyte arrest on inflamed
endothelium under flow conditions. J Leukoc Biol. 78:435–441.
2005.PubMed/NCBI View Article : Google Scholar
|