1. Introduction
Influenza is an acute viral respiratory infection
that occurs annually and is primarily caused by influenza A and B
viruses. It continues to impose a substantial global public health
burden, affecting millions of individuals annually and contributing
to significant morbidity and mortality worldwide. According to the
World Health Organization (WHO), influenza is responsible for
~290,000 to 650,000 respiratory-related deaths globally each year
(1,2). The impact of influenza varies
considerably from year to year, largely due to rapid viral
evolution that results in frequent antigenic changes. This
continuous viral evolution, combined with heterogeneity in
population immunity, contributes to unpredictable and sometimes
highly variable severity of seasonal influenza outbreaks (3,4).
Vaccination remains the primary strategy for the
prevention of seasonal influenza. Influenza vaccines are designed
to elicit immune responses against the strains most likely to
predominate during a given influenza season. These vaccines
primarily target the viral surface glycoproteins hemagglutinin (HA)
and neuraminidase (NA), which are essential for viral entry into
host cells and subsequent propagation within the respiratory tract.
Vaccine composition is determined based on predictions of
circulating seasonal strains generated through coordinated
surveillance by the WHO and national health authorities (5,6).
However, the effectiveness of influenza vaccines demonstrates
considerable inter-seasonal variability, typically ranging between
40 and 60% (7,8). This variability is attributable to
factors, such as antigenic mismatch, ongoing viral evolution and
heterogeneity in host immune responses. The rapid pace of genetic
alterations in influenza viruses represents one of the principal
barriers to achieving consistently high vaccine effectiveness.
Influenza viruses are subject to continuous genetic variation
through antigenic drift, characterized by the accumulation of point
mutations in the HA and NA genes. These mutations alter surface
proteins, enabling the virus to evade immunity acquired through
prior infection or vaccination (9,10).
Consequently, vaccinated individuals may remain susceptible to
infection when circulating strains differ substantially from
vaccine strains. Antigenic drift becomes particularly problematic
during seasons in which vaccine strains are poorly matched to
circulating variants. Evidence indicates that vaccine-virus
mismatch significantly reduces vaccine effectiveness and increases
the likelihood of breakthrough infections (11,12).
For example, during the 2014-2015 influenza season, a substantial
genetic mismatch between the vaccine and circulating HA type 3 and
NA type 2 (H3N2) strains resulted in markedly reduced vaccine
effectiveness (13). In addition
to the antigenic drift, influenza viruses may undergo an antigenic
shift, a process involving major genetic reassortment that can
generate novel viral subtypes. Such shifts may lead to pandemic
strains capable of evading existing seasonal vaccines and spreading
rapidly within populations. Pandemic strains can evade seasonal
vaccines; however, the method through which they function indicates
how the virus can spread rapidly within populations despite this
(14,15). Annual influenza vaccines are
formulated based on strain predictions issued by the WHO and
national surveillance systems. As vaccine production requires
substantial lead time, these predictions are typically made 6 to 9
months prior to the influenza season. This interval increases the
risk of mismatch between vaccine strains and circulating viruses
due to ongoing antigenic drift (16,17).
Furthermore, the majority of influenza vaccines contain inactivated
or attenuated forms of only three or four viral strains, thereby
limiting the breadth of protection (18,19).
Although quadrivalent vaccines have expanded coverage to include an
additional influenza B lineage, protection remains restricted in
scope. Ongoing research is focused on the development of broader or
universal influenza vaccines capable of conferring cross-strain
protection (20,21); however, such vaccines are not yet
available to the public. Vaccine effectiveness is also strongly
influenced by host-related factors. Age, health status and prior
influenza exposure can substantially influence vaccine-induced
immune responses. Certain populations, particularly older adults
and immunocompromised individuals, may exhibit a reduced
responsiveness to vaccination (22,23).
The phenomenon known as ‘original antigenic sin’, or immune
imprinting, may bias immune responses toward previously encountered
strains rather than newly emerging variants (24). The present review therefore
discusses the roles of viral evolution, vaccine design, host immune
responses and epidemiological determinants in explaining the
persistence of influenza infections despite widespread
vaccination.
The present review aimed to: i) Examine the factors
that limit influenza vaccine effectiveness; ii) synthesize recent
evidence explaining breakthrough infections; and iii) discuss
emerging strategies to enhance vaccine performance and global
influenza control.
2. Literature search
The present study was conducted as a structured
narrative review to synthesize current evidence on influenza virus
evolution, vaccine effectiveness, host-related determinants of
vaccine response, and emerging strategies in influenza vaccine
design and prediction. The present review aimed to integrate
findings from virology, immunology, epidemiology and public-health
research to provide a comprehensive and critical overview of
factors influencing the global effectiveness of influenza
vaccination. Although not designed as a formal systematic review,
the study followed a transparent and reproducible search approach
to ensure scholarly rigor and breadth of coverage.
A comprehensive literature search was performed
using the PubMed/MEDLINE, Scopus, Web of Science and Google Scholar
databases to identify relevant publications between January, 2000
and February, 2025. Earlier landmark studies were included where
necessary to provide historical context. Search terms combined
Medical Subject Headings and keywords related to influenza
vaccination and viral evolution, including ‘influenza’, ‘influenza
vaccine’, ‘vaccine effectiveness’, ‘antigenic drift’, ‘antigenic
shift’, ‘immune response’, ‘immunosenescence’, ‘vaccine hesitancy’,
‘universal influenza vaccine’, ‘mRNA vaccine’, ‘genomic
surveillance’, and ‘artificial intelligence’. Boolean operators
were applied to refine results, and reference lists of relevant
articles were screened manually to identify additional sources.
Studies were included if they addressed influenza
vaccine effectiveness, immune responses, viral evolution, vaccine
technologies, or public-health determinants of vaccine uptake.
Clinical, epidemiological, laboratory and review studies published
in the English language were considered. Articles lacking
sufficient methodological detail or unrelated to influenza
vaccination were excluded. Titles and abstracts were screened for
relevance, followed by full-text review of eligible studies.
Evidence was synthesized narratively due to the heterogeneity of
study designs and outcomes, with emphasis on key themes and
emerging trends in vaccine performance, immunological mechanisms,
and future vaccine development. Priority was given to peer-reviewed
studies, systematic reviews and reports from major public-health
organizations to ensure reliability and scientific validity.
3. Influenza virus evolution and vaccine
mismatch
Influenza virus evolution is primarily driven by two
mechanisms: Antigenic drift and antigenic shift. These processes
result in continuous alterations of viral surface proteins,
frequently leading to vaccine mismatch when circulating strains
differ from those selected for vaccine production (9). Such mismatches reduce vaccine
effectiveness because viral evolution remains unpredictable while
vaccine production timelines are fixed. Consequently, robust global
surveillance and regular updates to vaccine formulations are
essential to mitigate this challenge. As illustrated in Fig. 1, influenza viruses evolve through
gradual mutations (antigenic drift) and major genetic reassortment
events (antigenic shift). These mechanisms necessitate frequent
vaccine reformulation and, in some cases, facilitate zoonotic
transmission events that increase pandemic risk (9).
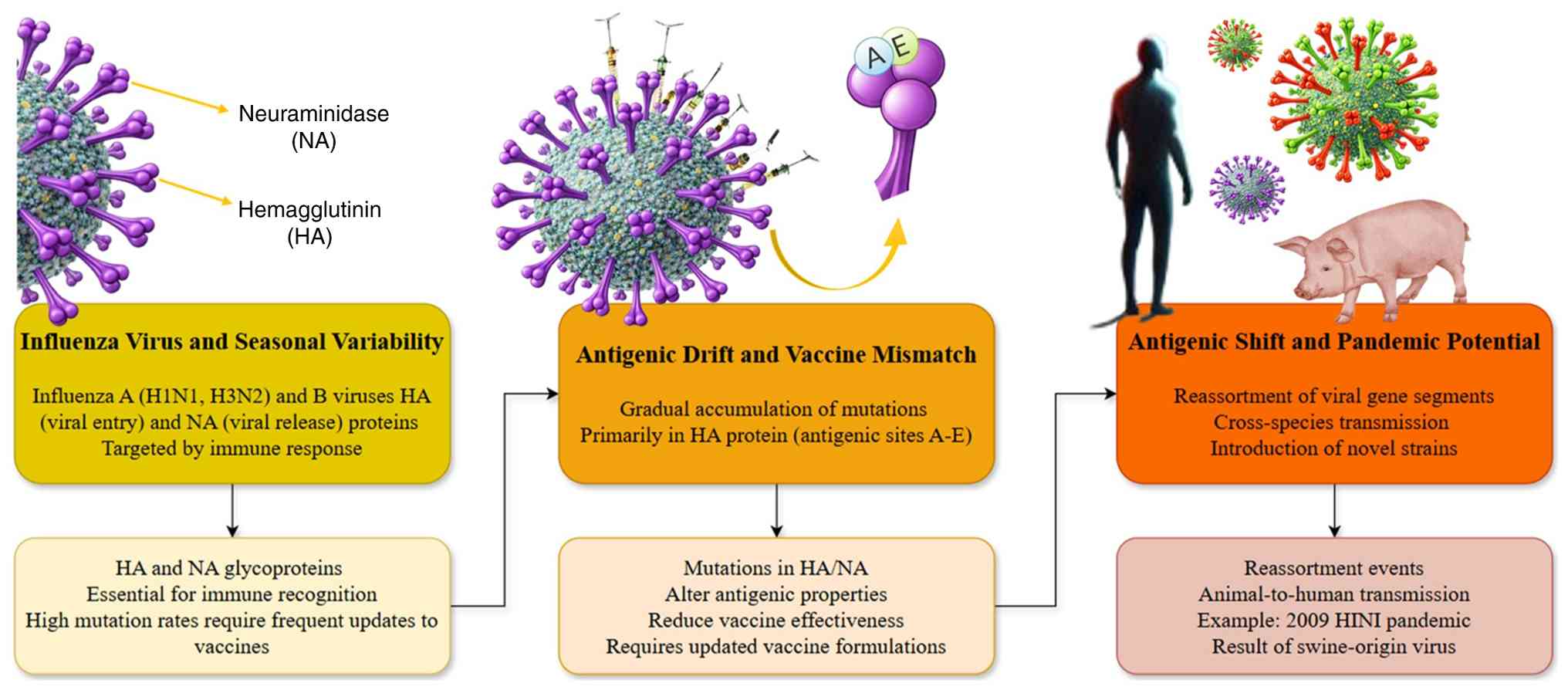 | Figure 1Influenza virus variability and
pandemic potential: antigenic drift and shift. The image
illustrates the evolution of the influenza viruses through
antigenic drift and antigenic shift. (Left panel) Influenza virus
and seasonal variability: The image depicts influenza A (H1N1 and
H3N2) and B viruses, highlighting their key proteins, HA and NA,
which are targeted by the immune response. These proteins have high
mutation rates, necessitating frequent updates to vaccines. (Middle
panel) Antigenic drift and vaccine mismatch: This process involves
gradual mutations in the HA protein, particularly in antigenic
sites A-E, which can lead to mismatches between circulating strains
and vaccine strains, reducing vaccine effectiveness. (Right panel)
Antigenic shift and pandemic potential: Antigenic shift occurs when
the reassortment of viral genes leads to the emergence of novel
strains with the potential for cross-species transmission, such as
the swine-origin virus from the 2009 H1N1 pandemic. This process
increases the risk of pandemics. NA, neuraminidase; HA,
hemagglutinin. |
Influenza virus and seasonal
variability
Seasonal variation in influenza viruses is
sufficiently pronounced to require annual reformulation of vaccines
(25). Influenza A and B viruses
possess segmented RNA genomes encoding multiple proteins essential
for viral replication and immune evasion (26). The viral envelope is characterized
by surface glycoproteins, HA and NA, which are central to viral
infectivity (27). These
glycoproteins form the basis of the influenza subtype
classification system; for example, ‘H1N1’ denotes a virus with HA
subtype 1 and NA subtype 1. HA mediates viral entry through
receptor binding, whereas neuraminidase facilitates viral release
by cleaving sialic acid residues. As illustrated in Fig. 1, HA comprises two subunits: The HA1
globular head responsible for receptor binding and the HA2 stalk
region that mediates membrane fusion (28,29).
Annual vaccine composition is determined through
WHO-coordinated surveillance and may require modifications each
year. The HA head contains five principal antigenic sites (A-E),
which are the primary targets of neutralizing antibodies (30-32).
Mutations within these antigenic sites, and within key positions in
the stalk region, enable the virus to evade immune recognition,
posing substantial challenges for vaccine development (33,34).
Influenza B viruses exhibit less genetic diversity than influenza A
viruses and are categorized into two lineages: Yamagata and
Victoria. Current quadrivalent vaccines include antigens from both
influenza B lineages alongside H1N1 and H3N2 strains to broaden
protection (35). The WHO monitors
global influenza evolution through the Global Influenza
Surveillance and Response System (GISRS), established in
1952(36). This system provides
biannual recommendations for vaccine composition in both the
Northern and Southern Hemispheres (37). Historically, the emergence of novel
influenza subtypes underscores the importance of continuous
surveillance and rapid vaccine adaptation. For example, the A/H2N2
virus responsible for the 1957 Asian influenza pandemic emerged
following earlier H1N1 circulation, illustrating the dynamic nature
of influenza evolution (38,39).
Antigenic drift and vaccine
mismatch
Antigenic drift refers to the gradual accumulation
of amino acid substitutions in the HA and NA surface glycoproteins,
particularly within antigenic epitopes (40). This continuous process enables
influenza viruses to alter their antigenicity, thereby evading
pre-existing host immune responses. Beneficial mutations allow
dominant seasonal variants to emerge and replace previously
circulating strains (41,42). Although detailed evolutionary
tracking is complex due to the large volume of viral samples,
modern genomic technologies now enable high-resolution phylogenetic
analysis of viral diversification (43).
In addition to point mutations, genetic reassortment
substantially contributes to viral diversification, particularly
when multiple influenza subtypes co-circulate. Reassortment between
related strains increases genomic variability and enhances viral
adaptability (44,45). Intra-subtype reassortment can
transfer a drifted HA gene onto a genetically advantageous viral
background, as observed during the severe epidemics in the
1946-1947 and 2003-2004 seasons (46,47).
Similarly, the emergence of reassortant H1N2 viruses during the
2001-2002 season, which predominated in 41 countries, highlighted
the global impact of such events (48,49).
Surveillance programs rely on hemagglutination inhibition assays
and viral RNA sequencing to monitor antigenic drift in HA and NA
(50). Surveillance programs rely
on hemagglutination inhibition assays and viral RNA sequencing to
monitor these incremental mutations in HA and NA. Greater attention
to neuraminidase antigenic drift, assessed through neuraminidase
inhibition assays, may improve vaccine-virus matching and enhance
vaccine effectiveness (21,51).
Antigenic shift and pandemic
potential
In contrast to antigenic drift, antigenic shift
involves abrupt and substantial changes in the antigenic properties
of influenza A viruses. Such shifts frequently result in the
emergence of antigenically novel viruses capable of causing
pandemics (52). Pandemic
influenza viruses often acquire HA and NA genes from animal
reservoirs that are antigenically distinct from circulating human
strains (53,54). These shifts may lead to the
replacement of previously circulating strains, as observed during
the 1957 H2N2 pandemic, which displaced the earlier H1N1 lineage
(55).
The 2009 H1N1 pandemic virus provides a contemporary
example, having emerged through reassortment among avian, human,
and swine influenza viruses (56,57).
As illustrated in Fig. 2,
antigenic shift generates entirely new viral subtypes, whereas
antigenic drift represents gradual genetic evolution that enables
pandemic strains to persist as seasonal variants. The figure
emphasizes the role of these processes in generating new viral
subtypes and sustaining their circulation in humans. A comparison
of the influenza A and B viruses is presented in Table I, highlighting the greater
variability of influenza A, driven by both drift and shift, and its
capacity to cause pandemics, in contrast to influenza B, which
undergoes antigenic drift only. Its two lineages, Victoria and
Yamagata, are included in quadrivalent vaccines for broader
protection.
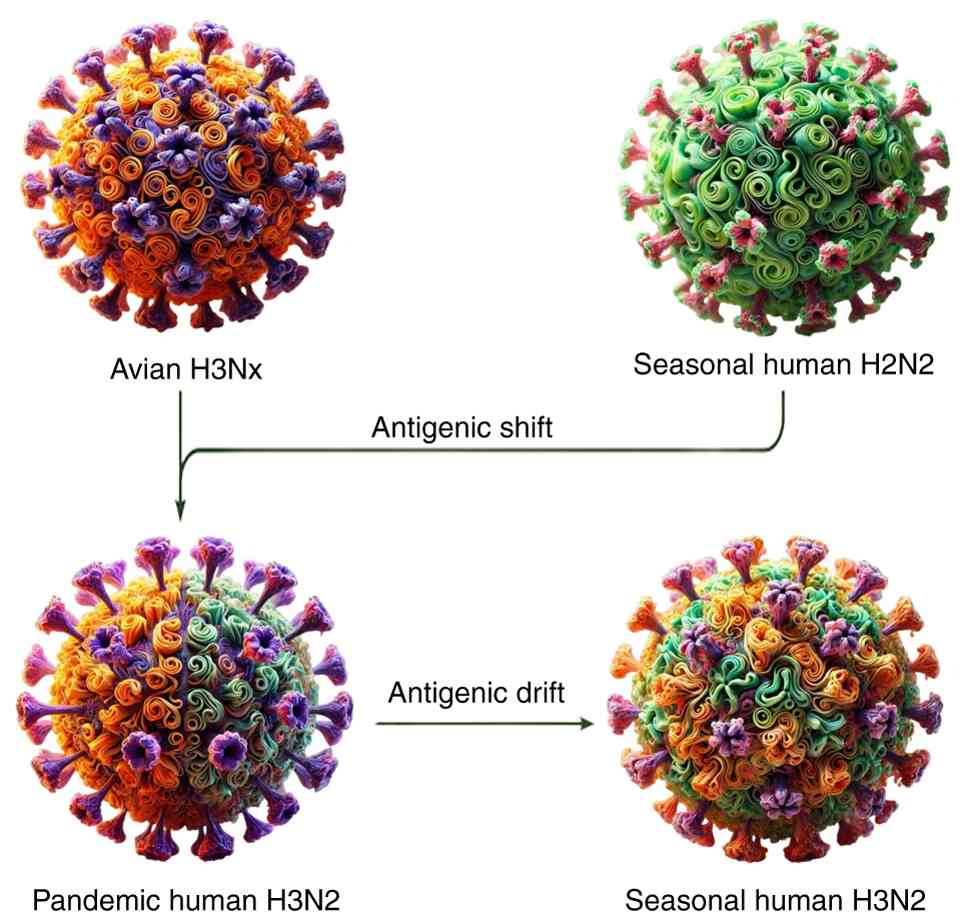 | Figure 2Genetic changes in influenza viruses:
Antigenic drift and antigenic shift. The image illustrates the
genetic changes in influenza A viruses through antigenic shift and
antigenic drift. Avian H3Nx is a subtype of influenza A found in
birds, characterized by the H3 hemagglutinin protein and a variety
of neuraminidase (N) subtypes. It circulates in wild and domestic
birds and occasionally crosses species, posing a potential risk to
humans and other animals (160).
Seasonal Human H2N2 refers to a subtype that caused the 1957 Asian
flu pandemic. This strain, with H2 HA and N2 NA, circulated
seasonally until it was replaced by H3N2 in 1968 (161,162). Pandemic human H3N2 emerged in
1968, causing the Hong Kong flu pandemic, and resulted from a
reassortment event between avian and human influenza viruses. It
continues to circulate seasonally. Seasonal human H3N2 evolved from
the 1968 pandemic strain and continues to cause seasonal flu
infections, especially affecting vulnerable populations (163,164). NA, neuraminidase; HA,
hemagglutinin. |
 | Table IComparison of influenza A and B. |
Table I
Comparison of influenza A and B.
| Feature/aspect | Influenza A
(Refs.) | Influenza B
(Refs.) |
|---|
| Subtypes/lineages
Mechanism of change | H1N1, H3N2 (main
subtypes) (165) | Victoria lineage,
Yamagata lineage (165) |
| | - Antigenic drift:
Gradual mutations in HA and NA proteins (166,167) | - Antigenic drift
only (166,167) |
| | - Antigenic shift:
Genetic reassortment between strains (pandemics) (166,167) | - Lower variability
compared to influenza A (165) |
| Pandemic
potential | High due to
antigenic shift (166,167) Examples: | Low Antigenic shift
does not occur; no history of pandemics (166,167) |
| | - 1918 H1N1
Pandemic (Spanish flu) (166) | |
| | - 1957 H2N2
Pandemic (Asian flu) (166) | |
| | - 1968 H3N2
Pandemic (Hong Kong flu) (166) | |
| | - 2009 H1N1
Pandemic (swine flu) (166) | |
| Seasonal
impact | Causes major
seasonal epidemics with significant mortality and morbidity
(165) | Causes seasonal
outbreaks, generally milder than Influenza A (168) |
| Vaccine
inclusion | Annual vaccines
include: | Included in
quadrivalent vaccines: |
| | - H1N1 (2009
pandemic strain) | - Victoria
lineage |
| | - H3N2 subtype
(169,170) | - Yamagata lineage
(169-171) |
| Surveillance | WHO's GISRS
monitors circulating strains globally (168,173) | Part of GISRS
monitoring; included to ensure broader protection in vaccines |
| | - Biannual updates
for northern and southern hemispheres (169,172,173) | |
| Examples of
reassortment | Genetic
reassortment events drive pandemics and diversity (166,167) | Not applicable due
to lack of antigenic shift |
| | Example: 2009 H1N1
(swine-origin virus combining swine, human, and avian segments)
(167) | |
| Historical
examples | - 1918 Spanish flu
(H1N1) (166) | No historical
pandemics; seasonal infections only (166,167) |
| | - 1957 Asian flu
(H2N2) (166) | |
| | - 1968 Hong Kong
flu (H3N2) (166) | |
| | - 2009 swine flu
pandemic (H1N1) (166) | |
4. Host factors influencing vaccine
response
Age-related immune response
differences
Despite increasing vaccination coverage over recent
decades, influenza-related mortality among older adults remains
substantial. Age represents a critical determinant of vaccine
responsiveness, particularly in older populations. Reduced vaccine
effectiveness contributes to heightened susceptibility to
infectious diseases among older adults, largely due to
immunosenescence, the age-related decline in immune function
(58). Immunosenescence
substantially increases susceptibility to infection and disease
severity in elderly populations. Age-associated immune
deterioration leads to impaired responses to both pathogens and
vaccination (59). Both the
quantity and functional quality of the T-cell compartment decline
with age, correlating with diminished vaccine-induced immune
responses in older adults. Effective vaccine-induced recall
immunity requires balanced T-cell responses that support both
memory formation and high-affinity antibody production. With
advancing age, T-cell responses tend to favor transient effector
activity rather than durable memory or follicular helper T-cell
responses. Consequently, the protective efficacy of vaccine-induced
antibodies is often reduced in older adults (60,61).
Declining immune integrity leads to reduced resistance to
infectious diseases and diminished vaccine effectiveness.
Immunosenescence also increases vulnerability to age-associated
chronic conditions, including cardiovascular disease and autoimmune
disorders (62,63). Age-related reductions in antibody
responses to influenza vaccination are closely associated with
alterations in T-cell function (64,65).
Primary lymphoid organs, including the thymus and bone marrow,
undergo involution with age, resulting in reduced naïve lymphocyte
production. Ageing is also associated with decreased phagocytic
activity and reduced numbers of T- and B-cell progenitors. Chronic
antigenic stimulation and increased synthesis of proinflammatory
cytokines occur concurrently. Thymic involution significantly
reduces the output of naïve T cells, limiting immune adaptability
in older adults (66). Thymic
activity declines progressively from early life, accompanied by
structural changes in thymic epithelial cells (67,68).
As a result, the number of naïve T cells entering peripheral
circulation decreases with age, reducing immune repertoire
diversity (69). Although
influenza poses a risk across all age groups, children and older
adults remain particularly vulnerable (70). Children may exhibit less mature
immune responses, including reduced memory T-cell formation and
antibody production compared with adults (71). Vaccinated infants often demonstrate
lower vaccine-induced protection, while unvaccinated infants remain
at greatest risk of severe influenza-related morbidity (72). Neonates possess relatively immature
immune systems, characterized by reduced antigen-presenting cell
activity and cytokine production compared with older children and
adults (73).
Impact of comorbidities
In addition to immunosenescence, comorbid conditions
can further impair immune responses to influenza vaccination
(23,74). Comorbidities exert a substantial
influence on vaccine-induced immune responses and overall vaccine
effectiveness (75,76). Conditions, such as chronic kidney
disease, cardiovascular disease, and diabetes mellitus are
associated with reduced vaccine immunogenicity (77,78).
These conditions can impair macrophage and lymphocyte function,
thereby weakening post-vaccination immune responses (78).
Acute infections, including pneumococcal disease and
seasonal influenza, are associated with increased morbidity and
mortality rates in individuals with cardiovascular disease
(79). Evidence suggests that
individuals with cardiovascular disease should receive influenza
vaccination as part of comprehensive secondary prevention
strategies (80). Cardiovascular
disease-related immune alterations may influence responses to
vaccination and inflammatory stimuli. Influenza vaccination may
elicit variable inflammatory responses in individuals with coronary
artery disease (81). Influenza
vaccination is generally safe in individuals with cardiovascular
disease and typically induces protective antibody levels comparable
to those in healthy populations. However, individuals with
congestive heart failure may exhibit lower post-vaccination
antibody titers despite achieving similar seroconversion rates
(80,82).
Obesity represents a key modifier of immune
responses to influenza vaccination. Some studies suggest that
individuals with overweight initially demonstrate higher
serological responses following vaccination compared with
individuals of normal weight (79). However, obese individuals may
exhibit impaired CD8+ T-cell activation and reduced metabolic
responses following vaccination (83). Although individuals with obesity
may be more likely to receive influenza vaccination, immune
responses may be less robust and less durable (84), however, many overweighed persons
show decreased immune responses (innate and adaptive) to the
vaccine and thus make the eventuality of vaccination less efficient
(83). Obesity has also been shown
to be associated with a diminished immune response to the influenza
vaccine. Experimental models demonstrate that obesity is associated
with reduced neutralizing antibody production and impaired viral
clearance following vaccination (85).
Previous exposure and immunity
imprinting
Influenza-related morbidity and mortality remain
substantial despite repeated seasonal vaccination programs. The
effects of repeated and imprinting seasonal influenza vaccinations
were examined to assess the processes and immunological variables
influencing influenza vaccine responses (86). Pre-existing immunity resulting from
prior infection or vaccination can influence responses to
subsequent influenza vaccination (87,88),
which influences both the quantity and quality of antibodies
generated later in life in response to influenza viruses. Antibody
responses later in life are often shaped by early childhood
exposure to influenza viruses. This phenomenon, currently commonly
termed immune imprinting, reflects both beneficial and detrimental
effects of immune history on vaccine effectiveness (24). Immune imprinting may preferentially
stimulate memory B-cell responses to previously encountered strains
while limiting responses to novel variants (23,24).
Repeated annual vaccination may also influence
immune responses to subsequent influenza exposure. Some
epidemiological studies suggest reduced vaccine effectiveness among
individuals vaccinated across multiple consecutive seasons
(89). However, other studies
report sustained or comparable protection with repeated
vaccination, indicating mixed evidence (90,91).
Vaccine immunogenicity remains a central determinant
of vaccine effectiveness (92).
Repeated immunization may alter the immunogenicity of a vaccine,
according to previous research (93). As previously demonstrated, those
who had received an influenza vaccine the year before were far less
likely to experience seroconversion than those who did not
(94). Repetition of the vaccine
may reduce its immunogenicity, which may be one reason. Based on
research conducted by Sherman et al (86), the immune response was found to
have a lower fold change after many seasonal influenza
vaccinations. Using three different vaccine platforms, researchers
found that repeated immunizations had a negative impact on antibody
binding, antibody affinity maturation, and hemagglutination
inhibition responses to H1N1, H3N2 and B strains (93). Previous findings suggest that
repeated vaccination may reduce antibody-secreting cell responses
and impair effector B-cell function following subsequent
vaccination (95). Proposed to
explain the detrimental consequences of several vaccinations is the
phenomena of antigenic imprinting.
5. Immunological and molecular
mechanisms
Humoral vs. cellular immunity
Influenza vaccines primarily target the
hemagglutinin and neuraminidase glycoproteins and function
predominantly by eliciting humoral immune responses against these
viral surface antigens. Vaccine-induced antibodies play a central
role in preventing severe disease by blocking viral entry and
enhancing immune clearance through mechanisms such as phagocytosis,
complement activation, and antibody-dependent cellular cytotoxicity
(96). However, influenza viruses
can evade antibody-mediated immunity through rapid mutation,
particularly within the receptor-binding region of hemagglutinin.
Consequently, vaccine formulations must be updated annually to
match circulating strains. Previous studies have indicated that
inactivated influenza vaccines demonstrate reduced effectiveness
when circulating viral strains differ antigenically from vaccine
strains (96,97). By contrast, T-cell-mediated
cellular immunity provides an additional layer of protection
against influenza infection. CD8+ T-cells contribute to
viral clearance by eliminating infected cells, while
CD4+ T-cells support antibody production through
cytokine-mediated activation of B-cells (98,99).
Individuals with impaired humoral immunity, including older adults
and immunocompromised patients, rely heavily on cellular immune
responses for protection (99).
Following natural infection, influenza-specific CD8+
T-cell responses often serve as key correlates of protection due to
their breadth and durability (96,98).
Current influenza vaccines, however, often elicit relatively weak
T-cell responses, limiting the development of robust cellular
immunity in a number of recipients (100). Although humoral immunity is
frequently prioritized due to the ease of measuring antibody
titers, cellular immunity plays an equally critical role in
cross-strain protection (97,98).
Cellular immune responses target conserved viral epitopes,
potentially providing broader and longer-lasting protection against
diverse influenza strains (101).
In summary, while current influenza vaccines predominantly
stimulate humoral immunity, insufficient activation of cellular
immunity may limit overall vaccine effectiveness. Future vaccine
strategies should aim to induce balanced humoral and cellular
responses to enhance durability and breadth of protection (96,97).
Immune evasion by the influenza
virus
Influenza A viruses employ multiple strategies to
evade detection and clearance by the host immune system (102-104).
A defining feature of these viruses is their extensive genetic and
antigenic diversity, driven primarily by antigenic drift and
antigenic shift. These mechanisms alter viral antigens, reducing
recognition by both innate and adaptive immune responses (101-103,105,106).
Antigenic drift involves gradual accumulation of point mutations in
genes encoding surface glycoproteins such as HA, resulting in
reduced antibody binding while maintaining viral fitness. Antigenic
shift involves the reassortment of gene segments between different
influenza strains, producing novel viruses with distinct antigenic
properties (101,102,104).
The glycosylation of viral surface proteins
represents another major immune-evasion mechanism. This process
involves the addition of oligosaccharide chains to viral proteins,
altering antigenic structure and immune recognition. Glycosylation
is also crucial since it may affect HA and NA glycoprotein
properties. These proteins are also involved in viral entry and
escape. The increased glycosylation of HA, particularly within the
globular head region, can mask neutralizing epitopes and reduce
antibody binding (102,107). Recent H3N2 isolates demonstrate
progressively increased HA glycosylation, suggesting selective
pressure favoring enhanced immune evasion (102,107). Thus, by hiding critical regions
from the immune system, glycosylation protects viruses from immune
evasion.
The innate immune system constitutes the first line
of defense against influenza infection, with interferon signaling
playing a central role. Influenza viruses have evolved mechanisms
to suppress interferon responses, primarily through the activity of
the non-structural protein 1 (NS1). NS1 inhibits interferon
production and disrupts host antiviral pathways, thereby enhancing
viral replication and persistence. NS1 also blocks host gene
expression due to pre-messenger ribonucleic acid (mRNA) processing
and export of host mRNAs to the nucleus. Additionally, influenza
viruses can inhibit protein kinase R and other antiviral signaling
pathways, further weakening host defenses (105). At the adaptive immune level, the
influenza A virus can avoid the human immune response by antigen
drift and antigen shift (105,108). Mutations within viral epitopes
recognized by CD8+ T-cells also facilitate escape from
cellular immune responses, contributing to sustained transmission
(101).
Hypotheses on vaccine-induced immune
modulation
Emerging research on vaccine-induced immune
modulation highlights the importance of adjuvants and delivery
systems in shaping immune memory, durability of protection, and
responses to variant strains. Adjuvants can be defined as agents
that, administered together with a vaccine, enhance the immune
response. They improve immunogenicity and may enhance protection
against antigenically variable viruses, such as influenza (109,110). Adjuvants range from simple
mineral salts to complex lipid-based or particulate systems. The
use of adjuvants in vaccination dates back to early observations
that aluminum salts enhance antibody production when combined with
antigens. Adjuvants are divided into provisors and delivery means.
Adjuvants function by activating innate immune pathways, including
Toll-like receptors and other pattern-recognition receptors,
thereby promoting adaptive immune responses. Delivery systems
improve antigen stability and facilitate targeting to
antigen-presenting cells and lymphoid tissues (109).
The adjuvants of a delivery system incorporate the
antigen in a carrier, which has a dual function of containing the
antigen while also triggering a localized proinflammatory response.
This response recruits immune cells to the site of injection, thus
enhancing immune activation and memory consolidation. This
combination of antigen and adjuvant activates pattern recognition
receptor pathways, such as pathogen-associated molecular patterns
that are negatively associated with self and positively associated
with the production of numerous pro-inflammatory cytokines and
chemokines, which in turn activate cells of the innate immune
system (111).
Different delivery platforms, including emulsions,
nanoparticles and virus-like particles, can substantially influence
the magnitude and durability of immune responses. As an adjuvant,
aluminum salts are frequently used to enhance immunological
response, as well as aid in the retention of the antigen at the
injection site, leading to prolonged immune activation. Emulsions
like MF59 and AS03 increase the avidity of antigen for immune cells
by enhancing their presentation and consequently, the activity of
the immune cells, leading to more robust and durable memory of the
immune system. The ‘modeling’ of viruses is crucial in virosomes
and virus-like particles which help to amplify immune responses and
increase protection against the subsequent infections (111).
As a result of antigenic drift, the surface proteins
of the influenza virus are constantly modified. This dilutes the
matching correlation between the virus prevalent in circulation and
the strain used in the vaccine. This mismatch can diminish the
effectiveness of the vaccine in preventing infection (110,112,113). The inclusion of some adjuvants
and delivery systems into the vaccines has the potential to improve
management of memory immune response, which can aid in controlling
different strains of viruses and more generously reduce the chances
of breakthrough infections. Thus, providing longer protection.
Indeed, adjuvants or vaccination alone cannot guarantee no
infection will occur. Despite ongoing challenges, seasonal
influenza vaccination remains the most effective strategy for
preventing influenza infection and its complications, particularly
among high-risk populations (96).
6. Epidemiological and behavioral
factors
Vaccine uptake and herd immunity
Influenza is a critical respiratory disease that
substantially contributes to global morbidity and mortality. Timely
and effective vaccination remains the primary strategy for reducing
influenza-related illness and its complications (114). High vaccination coverage
contributes to herd immunity, a key factor in controlling the
spread of infectious diseases at the population level (115). When a sufficiently large
proportion of the population acquires immunity through vaccination
or prior infection, transmission can be significantly reduced.
Although a precise herd immunity threshold for influenza is
difficult to determine due to antigenic variation, a vaccination
coverage of approximately 60-80% may be required to reduce
transmission substantially (116). Emphasizing the societal benefits
of vaccination has been associated with increased uptake. Achieving
high vaccination coverage provides both individual protection and
indirect protection for the broader community (117). It can also be considered a
prosocial act, as it benefits others, by offering both personal and
indirect benefits to unvaccinated individuals, including those who
are too young, immunocompromised, or unable to be vaccinated
(118). Vaccination behaviors are
influenced by individual beliefs, perceived norms and the
availability of accurate information. Public confidence in
vaccination programs is therefore essential for sustaining herd
immunity and improving vaccination coverage (119).
Timing and availability of
vaccination
Influenza affects millions of individuals annually
and can result in severe complications, hospitalization and death,
particularly among high-risk populations (120). In low- and middle-income
countries, early vaccination has been shown to reduce influenza
cases and associated mortality during pandemic simulations
(121). However, the
effectiveness of vaccination programs depends not only on timing,
but also on distribution capacity, accessibility and healthcare
infrastructure. In Europe, multiple manufacturers produce influenza
vaccines, and the availability of specific formulations varies by
region and procurement policies (122). Vaccine availability in a given
location is influenced by licensing, national recommendations,
procurement strategies and healthcare delivery systems (123).
As influenza seasonality varies geographically,
vaccination schedules must be tailored to regional epidemiological
patterns. Regional public health strategies should therefore focus
on optimizing vaccination timing and improving access for local
populations. To broaden the reach of the preventative strategy,
regional protocols may emphasize the need of influenza vaccination
in the local population's native language (124).
Of note, two main vaccination approaches are
commonly used: Seasonal campaigns aligned with influenza peaks and
year-round vaccination strategies in regions without clearly
defined influenza seasons. Campaign-based vaccination programs
often achieve rapid increases in coverage, particularly during
initial implementation. Policymakers should consider local
epidemiology, healthcare infrastructure, and population
characteristics when selecting optimal vaccination strategies
(125).
Vaccine hesitancy and
misinformation
Vaccine hesitancy has been identified by the WHO as
a major global health challenge (125). Vaccine hesitancy has been
observed not only in the general population, but also among
healthcare professionals. Healthcare workers play a crucial role in
vaccine education, administration and the promotion of public
confidence (126). In recent
years, vaccine hesitancy has expanded globally, contributing to the
resurgence of vaccine-preventable diseases (127). Factors contributing to hesitancy
include misinformation, distrust in healthcare systems, and
reliance on non-evidence-based information sources (128). Public concerns regarding vaccine
safety and effectiveness often intensify during outbreaks and
pandemics. This erosion of confidence, commonly termed vaccine
hesitancy, represents a significant public health risk affecting
both vaccinated and unvaccinated populations (129). Influenza vaccines are generally
well tolerated, with the majority of adverse effects being mild and
transient, such as injection-site reactions, low-grade fever and
myalgia (6). Serious adverse
reactions are rare, and extensive post-licensure safety evaluations
have confirmed a favorable safety profile for seasonal influenza
vaccines (130). Nevertheless,
misinformation regarding vaccine safety continues to fuel public
concern and reduce vaccination uptake.
7. Recent advances and future
directions
Toward a universal influenza
vaccine
The capacity of influenza viruses to evade immune
responses through rapid mutation has intensified efforts to develop
a universal influenza vaccine (128). Conventional vaccines provide
limited protection when circulating strains differ antigenically
from predicted strains. Universal vaccine strategies aim to confer
durable protection against multiple influenza subtypes by targeting
conserved viral regions. The HA protein, particularly its conserved
stem region, represents a primary target for universal vaccine
development (131). The HA stem
evolves more slowly than the highly variable globular head, making
it an attractive target for broadly neutralizing antibodies.
Broadly neutralizing antibodies that bind conserved HA stem
epitopes can inhibit viral fusion and provide cross-strain
protection (132). Novel
approaches, including epitope scaffolding and headless HA
constructs, have demonstrated promise in eliciting broadly
neutralizing antibodies (133).
Sequential vaccination strategies have also been
explored to enhance immune responses to conserved viral epitopes.
Incremental exposure to antigenically diverse strains may
strengthen cross-protective immunity against multiple influenza
subtypes (128,134).
Despite these advances, the development of a
universal influenza vaccine remains challenging due to the immune
system's tendency to prioritize responses to variable HA head
epitopes. Redirecting immune responses toward conserved regions
will likely require optimized adjuvants, innovative immunogen
design and combinatorial vaccine strategies (135).
Artificial intelligence (AI) approaches are
increasingly being applied to identify conserved epitopes and
optimize vaccine antigen design. AI-driven models may enhance
vaccine formulation and predict vaccine effectiveness across
diverse populations. Advances in structural biology continue to
reveal novel targets for universal vaccine development (136).
mRNA-based vaccines and adjuvant
innovations
mRNA-based vaccines enable host cells to produce
viral antigens, thereby stimulating targeted immune responses.
Unlike traditional vaccines, mRNA vaccines can be rapidly developed
and adapted to emerging viral strains. This flexibility makes mRNA
platforms particularly well suited for influenza viruses
characterized by rapid antigenic evolution (137,138).
mRNA vaccines have demonstrated the ability to
induce both humoral and cellular immune responses, providing
comprehensive protection. Experimental mRNA vaccines targeting
conserved HA regions have generated broadly neutralizing antibodies
capable of cross-protection against multiple strains (139).
Adjuvants are incorporated into vaccines to enhance
immunogenicity and improve durability of immune responses. Novel
adjuvants are being investigated to improve the magnitude and
persistence of immune responses elicited by mRNA influenza
vaccines. Lipid nanoparticles used for mRNA delivery also function
as adjuvants by promoting cellular uptake and immune activation.
Toll-like receptor agonists represent another class of adjuvants
under investigation for their ability to enhance vaccine-induced
immunity (109,140).
By allowing accurate genomic changes and improved
antigen design, clustered regularly interspaced short palindromic
repeats (CRISPR)-Cas9 technology is emerging as a powerful tool for
identifying conserved genomic regions and optimizing antigen design
in influenza vaccine development. CRISPR-based approaches enable
rapid identification of conserved viral targets and facilitate
iterative vaccine optimization (141).
The integration of mRNA technology, advanced
adjuvants and genome-editing tools provides a promising pathway for
next-generation influenza vaccines. Future research is required
however, to focus on improving durability of protection, expanding
clinical trials across diverse populations and ensuring equitable
global access (142).
Improved surveillance and prediction
models
Advances in genomic sequencing technologies enable
the rapid analysis of influenza virus evolution and mutation
patterns. These technologies facilitate the early detection of
emerging strains and provide real-time insights into antigenic
changes (143,144).
AI models are increasingly used to predict influenza
virus evolution and inform vaccine strain selection.
Machine-learning algorithms analyze genomic, epidemiological and
environmental data to forecast dominant strains for upcoming
influenza seasons (145).
Deep-learning techniques can identify subtle genetic
patterns associated with antigenic variation. In order to forecast
HA and NA alterations that may evade existing vaccinations,
convolutional neural networks have been applied as an example
(146,147).
Integrating AI-based prediction with genomic
surveillance can enhance vaccine strain matching and improve
preparedness. Cloud-based technologies facilitate global
collaboration by allowing genomic data to be shared among
laboratories across the globe. When paired with real-time genetic
data, predictive algorithms help bridge the gap between vaccine
design and virus evolution (148).
CRISPR-Cas diagnostics facilitate the rapid
identification and characterization of influenza viruses, hence
aiding in flu surveillance. A CRISPR-based diagnostic instrument
known as Specific High Sensitivity Enzymatic Reporter UnLOCKing
(SHERLOCK) facilitates real-time monitoring and response strategies
through precise detection of viral alterations (141,149).
Integrating real-time genetic data with
international health networks is essential for influenza
surveillance. Predictive models could be further improved by
extending AI algorithms to incorporate more varied variables, such
as social and environmental elements. Additionally, a promising
path for quick strain identification and vaccine adaption is
provided by integrating CRISPR techniques with AI-driven genomic
surveillance (150,151). Sustained investment in genomic
surveillance, artificial intelligence, and global data-sharing
networks will be essential for improving influenza control and
vaccine effectiveness.
Policy and public health
interventions: Advanced strategies for influenza control
Policy initiatives and public health interventions
remain critical for reducing the global burden of influenza. Recent
developments in encouraging early immunization, overcoming vaccine
reluctance and enhancing accessibility indicate great potential in
reducing the prevalence of the flu.
Timely vaccination campaigns, improved
accessibility, and targeted communication strategies are essential
for preventing outbreaks. To guarantee early and extensive vaccine
delivery, governments and health organizations are using measures
such as ‘flu shot days’ and mobile immunization units. Research
indicates that programs that offer free vaccines or financial
incentives can boost uptake, particularly among underprivileged
groups (152,153).
Vaccine hesitancy continues to pose a major barrier
to achieving high vaccination coverage. Recent public health
campaigns emphasize transparent communication and community
engagement to build trust in vaccination programs. It has been
demonstrated that using digital tools for public awareness, such as
social media, can successfully refute anti-vaccine propaganda.
Personalized reminders and encouragement constitute two behavioral
science-based strategies that are becoming more popular for
promoting vaccination (154,155).
Enhancing the public awareness of influenza risks
and the advantages of vaccinations requires educational programs.
Programs designed for neighborhood organizations, workplaces and
schools to highlight the importance of vaccination in preventing
high-risk persons. These approaches reduce logistical hurdles by
frequently combining education with vaccine availability at the
location of workplaces, schools, or educational institutions
(156,157).
Ensuring equitable access to vaccines, particularly
in underserved and remote communities, remains a key policy
priority. Key steps include setting up immunization centers in
remote and underserved locations and using technology to optimize
vaccine distribution networks. Vaccines are delivered to
disadvantaged and distant communities through collaborations with
non-governmental organizations and local authorities (158,159).
Future strategies should integrate digital
technologies, predictive modeling, and international collaboration
to optimize vaccine distribution and uptake. Incorporating
influenza vaccination into routine immunization programs may
further normalize and increase coverage.
8. Limitations
The present review has several limitations. As a
narrative synthesis, it may be subject to selection bias and does
not include formal meta-analysis. Only English-language
publications were included, and the rapidly evolving nature of
influenza vaccine technologies and AI-based prediction models means
that new evidence may emerge following publication. Nevertheless,
efforts were made to include recent high-quality studies and
authoritative public-health sources to provide a balanced overview
of current knowledge.
9. Conclusion
The prevention of seasonal influenza through
vaccination remains a complex challenge influenced by viral
evolution, host-related factors, vaccine composition and
epidemiological dynamics. Despite advances, such as quadrivalent
vaccine formulations, the overall effectiveness of influenza
vaccination programs remains constrained by the rapid and
continuous evolution of influenza viruses. Antigenic drift remains
one of the most significant threats to vaccine performance, as
structural changes in viral surface proteins, particularly HA and
NA, can reduce the effectiveness of pre-existing immunity.
Vaccine-induced protection is also influenced by host-related
factors, including immunosenescence, comorbidities and immune
imprinting, which contribute to heterogeneous immune responses
across populations. The majority of currently available influenza
vaccines predominantly induce humoral immune responses, with
comparatively limited activation of cellular immunity, which is
essential for broader and longer-lasting protection. Beyond
biological constraints, vaccine uptake is negatively affected by
misinformation, limited access and distrust in public health
systems. Nevertheless, emerging technological innovations,
including mRNA-based vaccines, novel adjuvants, AI-driven
prediction models and universal vaccine strategies, provide
promising opportunities to enhance influenza prevention. Achieving
meaningful reductions in influenza burden will require coordinated
global efforts involving technological innovation, equitable
vaccine distribution, public trust, and sustained collaboration
between governments, healthcare institutions and communities.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
DHMA and DHB conceptualized and designed the study.
All authors (DHMA, DHB, HHK, BRA, SHS, AAA, SKA and RRQ)
contributed to writing, literature review and manuscript revision.
All authors read and approved the final manuscript. Data
authentication is not applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
No authors listed. Prevention and control
of influenza: recommendations of the Advisory Committee on
Immunization Practices (ACIP). MMWR Recomm Rep. 48:1–28.
1999.PubMed/NCBI
|
|
2
|
Mancini P, Brandtner D, Veneri C, Bonanno
Ferraro G, Iaconelli M, Puzelli S, Facchini M, Di Mario G,
Stefanelli P, Lucentini L, et al: Evaluation of trends in influenza
A and B viruses in wastewater and human surveillance data: Insights
from the 2022-2023 season in Italy. Food Environ Virol.
17(6)2024.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Weinberger DM, Harboe ZB, Viboud C, Krause
TG, Miller M, Mølbak K and Konradsen HB: Pneumococcal disease
seasonality: Incidence, severity and the role of influenza
activity. Eur Respir J. 43:833–841. 2014.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Owuor DC, de Laurent ZR, Nyawanda BO,
Emukule GO, Kondor R, Barnes JR, Nokes DJ, Agoti CN and Chaves SS:
Genetic and potential antigenic evolution of influenza A
(H1N1)pdm09 viruses circulating in Kenya during 2009-2018 influenza
seasons. Sci Rep. 13(22342)2023.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Wang X, Kulkarni D, Dozier M, Hartnup K,
Paget J, Campbell H and Nair H: Usher Network for COVID-19 Evidence
Reviews (UNCOVER) group. Influenza vaccination strategies for
2020-21 in the context of COVID-19. J Glob Health.
10(020310)2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Grohskopf LA, Blanton LH, Ferdinands JM,
Chung JR, Broder KR and Talbot HK: Prevention and Control of
Seasonal Influenza with Vaccines: Recommendations of the Advisory
Committee on Immunization Practices — United States, 2023-24
Influenza Season. MMWR Recomm Rep 72 (No. RR-2):1-25, 2023. DOI:
http://dx.doi.org/10.15585/mmwr.rr7202a1.
|
|
7
|
Flannery B, Clippard J, Zimmerman RK,
Nowalk MP, Jackson ML, Jackson LA, Monto AS, Petrie JG, McLean HQ,
Belongia EA, et al: Early estimates of seasonal influenza vaccine
effectiveness-United States, January 2015. MMWR Morb Mortal Wkly
Rep. 64:10–15. 2015.PubMed/NCBI
|
|
8
|
Huddleston J and Bedford T: Timely vaccine
strain selection and genomic surveillance improves evolutionary
forecast accuracy of seasonal influenza A/H3N2. medRxiv: doi:
10.1101/2024.09.11.24313489.
|
|
9
|
Kim H, Webster RG and Webby RJ: Influenza
virus: Dealing with a drifting and shifting pathogen. Viral
Immunol. 31:174–183. 2018.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Mosa AI: Antigenic variability. Front
Immunol. 11(2057)2020.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Tricco AC, Chit A, Soobiah C, Hallett D,
Meier G, Chen MH, Tashkandi M, Bauch CT and Loeb M: Comparing
influenza vaccine efficacy against mismatched and matched strains:
A systematic review and meta-analysis. BMC Med.
11(153)2013.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Trombetta CM, Kistner O, Montomoli E,
Viviani S and Marchi S: Influenza viruses and vaccines: The role of
vaccine effectiveness studies for evaluation of the benefits of
influenza vaccines. Vaccines (Basel). 10(714)2022.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Xie H, Wan XF, Ye Z, Plant EP, Zhao Y, Xu
Y, Li X, Finch C, Zhao N, Kawano T, et al: H3N2 mismatch of 2014-15
Northern hemisphere influenza vaccines and head-to-head comparison
between human and ferret antisera derived antigenic maps. Sci Rep.
5(15279)2015.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Steel J and Lowen AC: Influenza A virus
reassortment. Curr Top Microbiol Immunol. 385:377–401.
2014.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Harrington WN, Kackos CM and Webby RJ: The
evolution and future of influenza pandemic preparedness. Exp Mol
Med. 53:737–749. 2021.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Amorij JP, Huckriede A, Wilschut J,
Frijlink HW and Hinrichs WL: Development of stable influenza
vaccine powder formulations: Challenges and possibilities. Pharm
Res. 25:1256–1273. 2008.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Wolk DM, Lanyado A, Tice AM, SherMohammed
M, Kinar Y, Goren A, Chabris CF, Meyer MN, Shoshan A and Abedi V:
Prediction of influenza complications: development and validation
of a machine learning prediction model to improve and expand the
identification of vaccine-hesitant patients at risk of severe
influenza complications. J Clin Med. 11(4342)2022.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Sparrow E, Wood JG, Chadwick C, Newall AT,
Torvaldsen S, Moen A and Torelli G: Global production capacity of
seasonal and pandemic influenza vaccines in 2019. Vaccine.
39:512–520. 2021.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Carregaro RL, Roscani ANCP, Raimundo ACS,
Ferreira L, Vanni T, Da Graça Salomão M, Probst LF and Viscondi
JYK: Immunogenicity and safety of inactivated quadrivalent
influenza vaccine compared with the trivalent vaccine for influenza
infection: an overview of systematic reviews. BMC Infect Dis.
23(563)2023.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Osterhaus A, Fouchier R and Rimmelzwaan G:
Towards universal influenza vaccines? Philos Trans R Soc Lond B
Biol Sci. 366:2766–2773. 2011.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Wang Y, Tang CY and Wan XF: Antigenic
characterization of influenza and SARS-CoV-2 viruses. Anal Bioanal
Chem. 414:2841–2881. 2022.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Lord JM: The effect of aging of the immune
system on vaccination responses. Hum Vaccin Immunother.
9:1364–1367. 2013.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Dhakal S and Klein SL: Host factors impact
vaccine efficacy: Implications for seasonal and universal influenza
vaccine programs. J Virol. 93:e00797–19. 2019.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Henry C, Palm AK, Krammer F and Wilson PC:
From original antigenic sin to the universal influenza virus
vaccine. Trends Immunol. 39:70–79. 2018.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Nafziger AN and Pratt DS: Seasonal
influenza vaccination and technologies. J Clin Pharmacol.
54:719–731. 2014.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Petrova VN and Russell CA: The evolution
of seasonal influenza viruses. Nat Rev Microbiol. 16:47–60.
2018.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Gaymard A, Le Briand N, Frobert E, Lina B
and Escuret V: Functional balance between neuraminidase and
haemagglutinin in influenza viruses. Clin Microbiol Infect.
22:975–983. 2016.PubMed/NCBI View Article : Google Scholar
|
|
28
|
De Vries E, Du W, Guo H and de Haan CAM:
Influenza A virus hemagglutinin-neuraminidase-receptor balance:
preserving virus motility. Trends Microbiol. 28:57–67.
2020.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Xu R and Wilson IA: Structural
characterization of an early fusion intermediate of influenza virus
hemagglutinin. J Virol. 85:5172–5182. 2011.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Swanson N: Characterization of in vitro
fitness and antigenic drift differences between circulating H1N1
influenza clades (unpublished thesis). Johns Hopkins University,
2024.
|
|
31
|
Qiu Y, Stegalkina S, Zhang J, Boudanova E,
Park A, Zhou Y, Prabakaran P, Pougatcheva S, Ustyugova IV, Vogel
TU, et al: Mapping of a novel H3-specific broadly neutralizing
monoclonal antibody targeting the hemagglutinin globular head
isolated from an elite influenza virus-immunized donor exhibiting
serological breadth. J Virol. 94:e01035–19. 2020.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Tsuchiya E, Sugawara K, Hongo S, Matsuzaki
Y, Muraki Y, Li ZN and Nakamura K: Antigenic structure of the
haemagglutinin of human influenza A/H2N2 virus. J Gen Virol. 82 (Pt
10):2475–2484. 2001.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Wiley DC, Wilson IA and Skehel JJ:
Structural identification of the antibody-binding sites of Hong
Kong influenza haemagglutinin and their involvement in antigenic
variation. Nature. 289:373–378. 1981.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Gerhard W, Yewdell J, Frankel ME and
Webster R: Antigenic structure of influenza virus haemagglutinin
defined by hybridoma antibodies. Nature. 290:713–717.
1981.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Baxter D: Evaluating the case for
trivalent or quadrivalent influenza vaccines. Hum Vaccin
Immunother. 12:2712–2717. 2016.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Daniels RS and McCauley JW: The health of
influenza surveillance and pandemic preparedness in the wake of the
COVID-19 pandemic. J Gen Virol. 104(001822)2023.PubMed/NCBI View Article : Google Scholar
|
|
37
|
World Health Organization (WHO): Sixth
Meeting of National Influenza Centres and Influenza Surveillance in
the Western Pacific and South-East Asia Regions, Hanoi, Viet Nam,
29-31 May 2012: Meeting report. WHO, Geneva, 2012.
|
|
38
|
Sánchez de Prada L: Humoral immune
response after seasonal influenza vaccination (unpublished thesis).
University of Valladolid, 2023.
|
|
39
|
Graham BS: Advances in antiviral vaccine
development. Immunol Rev. 255:230–242. 2013.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Altman MO, Angeletti D and Yewdell JW:
Antibody immunodominance: The key to understanding influenza virus
antigenic drift. Viral Immunol. 31:142–149. 2018.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Tjärnhage E: Exploring the field of
respiratory virus preventions: How to deal with future pandemics
(unpublished thesis). University of Oslo, 2024.
|
|
42
|
Dunning J, Thwaites RS and Openshaw PJM:
Seasonal and pandemic influenza: 100 years of progress, still much
to learn. Mucosal Immunol. 13:566–573. 2020.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Volz EM, Koelle K and Bedford T: Viral
phylodynamics. PLoS Comput Biol. 9(e1002947)2013.PubMed/NCBI View Article : Google Scholar
|
|
44
|
White MC and Lowen AC: Implications of
segment mismatch for influenza A virus evolution. J Gen Virol.
99:3–16. 2018.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Yong OX: Epidemiological and evolutionary
dynamics of influenza B viruses in Kuala Lumpur, Malaysia between
2012 and 2014. MSc thesis, University of Malaya, 2016.
|
|
46
|
Holmes EC, Ghedin E, Miller N, Taylor J,
Bao Y, St George K, Grenfell BT, Salzberg SL, Fraser CM, Lipman DJ
and Taubenberger JK: Whole-genome analysis of human influenza A
virus reveals multiple persistent lineages and reassortment among
recent H3N2 viruses. PLoS Biol. 3(e300)2005.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Nelson MI, Viboud C, Simonsen L, Bennett
RT, Griesemer SB, St George K, Taylor J, Spiro DJ, Sengamalay NA,
Ghedin E, et al: Multiple reassortment events in the evolutionary
history of H1N1 influenza A virus since 1918. PLoS Pathog.
4(e1000012)2008.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Ellis JS, Alvarez-Aguero A, Gregory V, Lin
YP, Hay A and Zambon MC: Influenza A H1N2 viruses, United Kingdom,
2001-02 influenza season. Emerg Infect Dis. 9:304–310.
2003.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Hsieh YH, Huang HM and Lan YC: On temporal
patterns and circulation of influenza virus strains in Taiwan,
2008-2014: Implications of 2009 pH1N1 pandemic. PLoS One.
11(e0154695)2016.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Xu X, Smith CB, Mungall BA, Lindstrom SE,
Hall HE, Subbarao K, Cox NJ and Klimov A: Intercontinental
circulation of human influenza A (H1N2) reassortant viruses during
the 2001-2002 influenza season. J Infect Dis. 186:1490–1493.
2002.PubMed/NCBI View
Article : Google Scholar
|
|
51
|
Wong SS and Webby RJ: Traditional and new
influenza vaccines. Clin Microbiol Rev. 26:476–492. 2013.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Eichelberger MC and Monto AS:
Neuraminidase, the forgotten surface antigen, emerges as an
influenza vaccine target for broadened protection. J Infect Dis.
219 (Suppl 1):S75–S80. 2019.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Shoham D: The modes of evolutionary
emergence of primal and late pandemic influenza virus strains from
viral reservoir in animals: An interdisciplinary analysis.
Influenza Res Treat. 2011(861792)2011.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Van Reeth K and Vincent AL: Influenza
viruses. In: Diseases of Swine. 10th edition. Wiley-Blackwell,
Hoboken, NJ, pp576-593, 2019.
|
|
55
|
Webster RG: Antigenic variation in
influenza viruses. In: Origin and Evolution of Viruses. Elsevier,
pp377-401, 1999.
|
|
56
|
Gatherer D: The 2009 H1N1 influenza
outbreak in its historical context. J Clin Virol. 45:174–178.
2009.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Bush RM and Cox NJ: Influenza evolution.
In: Infectious Disease and Host-Pathogen Evolution. Dronamraju KR
(ed). Cambridge University Press, pp175-197, 2004.
|
|
58
|
Patriarca PA and Cox NJ: Influenza
pandemic preparedness plan for the United States. J Infect Dis. 176
(Suppl 1):S4–S7. 1997.PubMed/NCBI View
Article : Google Scholar
|
|
59
|
Chen L, Shao C, Li J and Zhu F: Impact of
immunosenescence on vaccine immune responses and countermeasures.
Vaccines (Basel). 12(1289)2024.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Frasca D, Blomberg BB, Garcia D, Keilich
SR and Haynes L: Age-related factors that affect B cell responses
to vaccination in mice and humans. Immunol Rev. 296:142–154.
2020.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Gustafson CE, Kim C, Weyand CM and Goronzy
JJ: Influence of immune aging on vaccine responses. J Allergy Clin
Immunol. 145:1309–1321. 2020.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Dugan HL, Henry C and Wilson PC: Aging and
influenza vaccine-induced immunity. Cell Immunol.
348(103998)2020.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Aiello A, Farzaneh F, Candore G, Caruso C,
Davinelli S, Gambino CM, Ligotti ME, Zareian N and Accardi G:
Immunosenescence and its hallmarks: How to oppose aging
strategically? A review of potential options for therapeutic
intervention. Front Immunol. 10(2247)2019.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Müller L, Andrée M, Moskorsz W, Drexler I,
Walotka L, Grothmann R, Ptok J, Hillebrandt J, Ritchie A, Rabl D,
et al: Age-dependent immune response to the Biontech/Pfizer
BNT162b2 coronavirus disease 2019 vaccination. Clin Infect Dis.
73:2065–2072. 2021.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Mosterín Höpping A, McElhaney J, Fonville
JM, Powers DC, Beyer WEP and Smith DJ: The confounded effects of
age and exposure history in response to influenza vaccination.
Vaccine. 34:540–546. 2016.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Saurwein-Teissl M, Lung TL, Marx F,
Gschoesser C, Asch E, Blasko I, Parson W, Böck G, Schönitzer D,
Trannoy E and Grubeck-Loebenstein B: Lack of antibody production
following immunization in old age: Association with CD8(+)CD28(−) T
cell clonal expansions and imbalance in Th1/Th2 cytokine
production. J Immunol. 168:5893–5899. 2002.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Ciabattini A, Nardini C, Santoro F,
Garagnani P, Franceschi C and Medaglini D: Vaccination in the
elderly: The challenge of immune changes with aging. Semin Immunol.
40:83–94. 2018.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Nikolich-Žugich J: Aging of the T cell
compartment in mice and humans: From no naive expectations to foggy
memories. J Immunol. 193:2622–2629. 2014.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Barbouti A, Vasileiou PVS, Evangelou K,
Vlasis KG, Papoudou-Bai A, Gorgoulis VG and Kanavaros P:
Implications of oxidative stress and cellular senescence in
age-related thymus involution. Oxid Med Cell Longev.
2020(7986071)2020.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Fink PJ: The biology of recent thymic
emigrants. Annu Rev Immunol. 31:31–50. 2013.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Ruf BR and Knuf M: The burden of seasonal
and pandemic influenza in infants and children. Eur J Pediatr.
173:265–276. 2014.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Sakala IG, Eichinger KM and Petrovsky N:
Neonatal vaccine effectiveness and the role of adjuvants. Expert
Rev Clin Immunol. 15:869–878. 2019.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Rudd BD: Neonatal T cells: A
reinterpretation. Annu Rev Immunol. 38:229–247. 2020.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Zhang X, Zhivaki D and Lo-Man R: Unique
aspects of the perinatal immune system. Nat Rev Immunol.
17:495–507. 2017.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Andrew MK, Pott H, Staadegaard L, Paget J,
Chaves SS, Ortiz JR, McCauley J, Bresee J, Nunes MC, Baumeister E,
et al: Age differences in comorbidities, presenting symptoms, and
outcomes of influenza illness requiring hospitalization: A
worldwide perspective from the global influenza hospital
surveillance network. Open Forum Infect Dis.
10(ofad244)2023.PubMed/NCBI View Article : Google Scholar
|
|
76
|
Magyari M and Sorensen PS: Comorbidity in
multiple sclerosis. Front Neurol. 11(851)2020.PubMed/NCBI View Article : Google Scholar
|
|
77
|
Xu Q, Wei H, Wen S, Chen J, Lei Y, Cheng
Y, Huang W, Wang D and Shu Y: Factors affecting the immunogenicity
of influenza vaccines in humans. BMC Infect Dis.
23(211)2023.PubMed/NCBI View Article : Google Scholar
|
|
78
|
Prendecki M, Clarke C, Brown J, Cox A,
Gleeson S, Guckian M, Randell P, Pria AD, Lightstone L, Xu XN, et
al: Effect of previous SARS-CoV-2 infection on humoral and T-cell
responses to single-dose BNT162b2 vaccine. Lancet. 397:1178–1181.
2021.PubMed/NCBI View Article : Google Scholar
|
|
79
|
Kwetkat A and Heppner HJ: Comorbidities in
the elderly and their possible influence on vaccine response.
Interdiscip Top Gerontol Geriatr. 43:73–85. 2020.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Li Y, Cao GY, Jing WZ, Liu J and Liu M:
Global trends and regional differences in incidence and mortality
of cardiovascular disease, 1990-2019: Findings from the 2019 global
burden of disease study. Eur J Prev Cardiol. 30:276–286.
2023.PubMed/NCBI View Article : Google Scholar
|
|
81
|
Frasca D and Blomberg BB: B cell function
and influenza vaccine responses in healthy aging and disease. Curr
Opin Immunol. 29:112–118. 2014.PubMed/NCBI View Article : Google Scholar
|
|
82
|
Werba JP, Veglia F, Amato M, Baldassarre
D, Massironi P, Meroni PL, Riboldi P, Tremoli E and Camera M:
Patients with a history of stable or unstable coronary heart
disease have different acute phase responses to an inflammatory
stimulus. Atherosclerosis. 196:835–840. 2008.PubMed/NCBI View Article : Google Scholar
|
|
83
|
Callahan ST, Wolff M, Hill HR and Edwards
KM: NIAID Vaccine and Treatment Evaluation Unit (VTEU) Pandemic
H1N1 Vaccine Study Group. Impact of body mass index on
immunogenicity of pandemic H1N1 vaccine in children and adults. J
Infect Dis. 210:1270–1274. 2014.PubMed/NCBI View Article : Google Scholar
|
|
84
|
Honce R and Schultz-Cherry S: Impact of
obesity on influenza A virus pathogenesis, immune response, and
evolution. Front Immunol. 10(1071)2019.PubMed/NCBI View Article : Google Scholar
|
|
85
|
Harris JA, Moniz MH, Iott B, Power R and
Griggs JJ: Obesity and the receipt of influenza and pneumococcal
vaccination: A systematic review and meta-analysis. BMC Obes.
3(24)2016.PubMed/NCBI View Article : Google Scholar
|
|
86
|
Sherman AC, Lai L, Bower M, Natrajan MS,
Huerta C, Karmali V, Kleinhens J, Xu Y, Rouphael N and Mulligan MJ:
The effects of imprinting and repeated seasonal influenza
vaccination on adaptive immunity after influenza vaccination.
Vaccines (Basel). 8(663)2020.PubMed/NCBI View Article : Google Scholar
|
|
87
|
Cobey S and Hensley SE: Immune history and
influenza virus susceptibility. Curr Opin Virol. 22:105–111.
2017.PubMed/NCBI View Article : Google Scholar
|
|
88
|
Jang H and Ross TM: Preexisting
influenza-specific immunity and vaccine effectiveness. Expert Rev
Vaccines. 18:1043–1051. 2019.PubMed/NCBI View Article : Google Scholar
|
|
89
|
McLean HQ, Thompson MG, Sundaram ME, Meece
JK, McClure DL, Friedrich TC and Belongia EA: Impact of repeated
vaccination on vaccine effectiveness against influenza A (H3N2) and
B during 8 seasons. Clin Infect Dis. 59:1375–1385. 2014.PubMed/NCBI View Article : Google Scholar
|
|
90
|
Bartoszko JJ, McNamara IF, Aras OAZ,
Hylton DA, Zhang YB, Malhotra D, Hyett SL, Morassut RE, Rudziak P
and Loeb M: Does consecutive influenza vaccination reduce
protection against influenza: A systematic review and
meta-analysis. Vaccine. 36:3434–3444. 2018.PubMed/NCBI View Article : Google Scholar
|
|
91
|
Zelner J, Petrie JG, Trangucci R, Martin
ET and Monto AS: Effects of sequential influenza A (H1N1)pdm09
vaccination on antibody waning. J Infect Dis. 220:12–19.
2019.PubMed/NCBI View Article : Google Scholar
|
|
92
|
Chen JR, Liu YM, Tseng YC and Ma C: Better
influenza vaccines: An industry perspective. J Biomed Sci.
27(33)2020.PubMed/NCBI View Article : Google Scholar
|
|
93
|
Grohskopf LA, Alyanak E, Broder KR,
Blanton LH, Fry AM, Jernigan DB and Atmar RL: Prevention and
control of seasonal influenza with vaccines: Recommendations of the
Advisory Committee on Immunization Practices-United States, 2020-21
influenza season. MMWR Recomm Rep. 69:1–24. 2020.PubMed/NCBI View Article : Google Scholar
|
|
94
|
Rolfes MA, Flannery B, Chung JR,
O'Halloran A, Garg S, Belongia EA, Gaglani M, Zimmerman RK, Jackson
ML, Monto AS, et al: Effects of influenza vaccination in the United
States during the 2017-2018 influenza season. Clin Infect Dis.
69:1845–1853. 2019.PubMed/NCBI View Article : Google Scholar
|
|
95
|
McElhaney JE, Kuchel GA, Zhou X, Swain SL
and Haynes L: T-cell immunity to influenza in older adults: A
pathophysiological framework for development of more effective
vaccines. Front Immunol. 7(41)2016.PubMed/NCBI View Article : Google Scholar
|
|
96
|
Janssens Y, Joye J, Waerlop G, Clement F,
Leroux-Roels G and Leroux-Roels I: The role of cell-mediated
immunity against influenza and its implications for vaccine
evaluation. Front Immunol. 13(959379)2022.PubMed/NCBI View Article : Google Scholar
|
|
97
|
Tsang TK, Lam KT, Liu Y, Fang VJ, Mu X,
Leung NHL, Peiris JM, Leung GM, Cowling BJ and Tu W: Investigation
of CD4 and CD8 T cell-mediated protection against influenza A virus
in a cohort study. BMC Med. 20(230)2022.PubMed/NCBI View Article : Google Scholar
|
|
98
|
L'Huillier AG, Ferreira VH, Hirzel C,
Nellimarla S, Ku T, Natori Y, Humar A and Kumar D: T-cell responses
following natural influenza infection or vaccination in solid organ
transplant recipients. Sci Rep. 10(10104)2020.PubMed/NCBI View Article : Google Scholar
|
|
99
|
Velasco-Medina AA, García-León ML,
Velázquez-Sámano G and Wong-Chew RM: The cellular and humoral
immune response to influenza vaccination is comparable in asthmatic
and healthy subjects. Hum Vaccin Immunother. 17:98–105.
2021.PubMed/NCBI View Article : Google Scholar
|
|
100
|
Rosendahl Huber SK, Hendriks M, Jacobi
RHJ, van de Kassteele J, Mandersloot-Oskam JC, van Boxtel RAJ,
Wensing AMJ, Rots NY, Luytjes W and van Beek J: Immunogenicity of
influenza vaccines: Evidence for differential effect of secondary
vaccination on humoral and cellular immunity. Front Immunol.
9(3103)2019.PubMed/NCBI View Article : Google Scholar
|
|
101
|
Quiñones-Parra S, Loh L, Brown LE,
Kedzierska K and Valkenburg SA: Universal immunity to influenza
must outwit immune evasion. Front Microbiol. 5(285)2014.PubMed/NCBI View Article : Google Scholar
|
|
102
|
Schmolke M and García-Sastre A: Evasion of
innate and adaptive immune responses by influenza A virus. Cell
Microbiol. 12:873–880. 2010.PubMed/NCBI View Article : Google Scholar
|
|
103
|
Rashid F, Xie Z, Li M, Xie Z, Luo S and
Xie L: Roles and functions of IAV proteins in host immune evasion.
Front Immunol. 14(1323560)2023.PubMed/NCBI View Article : Google Scholar
|
|
104
|
Hale BG, Albrecht RA and García-Sastre A:
Innate immune evasion strategies of influenza viruses. Future
Microbiol. 5:23–41. 2010.PubMed/NCBI View Article : Google Scholar
|
|
105
|
Van de Sandt CE, Kreijtz JH and
Rimmelzwaan GF: Evasion of influenza A viruses from innate and
adaptive immune responses. Viruses. 4:1438–1476. 2012.PubMed/NCBI View Article : Google Scholar
|
|
106
|
Hsu ACY: Influenza virus: A master
tactician in innate immune evasion and novel therapeutic
interventions. Front Immunol. 9(743)2018.PubMed/NCBI View Article : Google Scholar
|
|
107
|
Kim P, Jang YH, Kwon SB, Lee CM, Han G and
Seong BL: Glycosylation of hemagglutinin and neuraminidase of
influenza A virus as signature for ecological spillover and
adaptation among influenza reservoirs. Viruses.
10(183)2018.PubMed/NCBI View Article : Google Scholar
|
|
108
|
Su G, Chen Y, Li X and Shao JW: Virus
versus host: Influenza A virus circumvents the immune responses.
Front Microbiol. 15(1394510)2024.PubMed/NCBI View Article : Google Scholar
|
|
109
|
Zhao T, Cai Y, Jiang Y, He X, Wei Y, Yu Y
and Tian X: Vaccine adjuvants: mechanisms and platforms. Signal
Transduct Target Ther. 8(283)2023.PubMed/NCBI View Article : Google Scholar
|
|
110
|
Shichinohe S and Watanabe T: Advances in
adjuvanted influenza vaccines. Vaccines (Basel).
11(1391)2023.PubMed/NCBI View Article : Google Scholar
|
|
111
|
Facciolà A, Visalli G, Laganà A and Di
Pietro A: An overview of vaccine adjuvants: Current evidence and
future perspectives. Vaccines (Basel). 10(819)2022.PubMed/NCBI View Article : Google Scholar
|
|
112
|
Li Z, Zhao Y, Li Y and Chen X:
Adjuvantation of influenza vaccines to induce cross-protective
immunity. Vaccines (Basel). 9(75)2021.PubMed/NCBI View Article : Google Scholar
|
|
113
|
Chan L, Alizadeh K, Alizadeh K, Fazel F,
Kakish JE, Karimi N, Knapp JP, Mehrani Y, Minott JA, Morovati S, et
al: Review of influenza virus vaccines: the qualitative nature of
immune responses to infection and vaccination is a critical
consideration. Vaccines (Basel). 9(979)2021.PubMed/NCBI View Article : Google Scholar
|
|
114
|
Zanobini P, Bonaccorsi G, Lorini C, Haag
M, McGovern I, Paget J and Caini S: Global patterns of seasonal
influenza activity, duration of activity and virus (sub)type
circulation from 2010 to 2020. Influenza Other Respir Viruses.
16:696–706. 2022.PubMed/NCBI View Article : Google Scholar
|
|
115
|
Yadegari I, Omidi M and Smith SR: The
herd-immunity threshold must be updated for multi-vaccine
strategies and multiple variants. Sci Rep. 11(22970)2021.PubMed/NCBI View Article : Google Scholar
|
|
116
|
Plans-Rubió P: The vaccination coverage
required to establish herd immunity against influenza viruses. Prev
Med. 55:72–77. 2012.PubMed/NCBI View Article : Google Scholar
|
|
117
|
Jung H and Albarracín D: Concerns for
others increase the likelihood of vaccination against influenza and
COVID-19 more in sparsely rather than densely populated areas. Proc
Natl Acad Sci USA. 118(e2007538118)2021.PubMed/NCBI View Article : Google Scholar
|
|
118
|
Böhm R and Betsch C: Prosocial
vaccination. Curr Opin Psychol. 43:307–311. 2022.PubMed/NCBI View Article : Google Scholar
|
|
119
|
Al-Hasan A, Khuntia J and Yim D: Does
seeing what others do through social media influence vaccine uptake
and help in the herd immunity through vaccination? A
cross-sectional analysis. Front Public Health.
9(715931)2021.PubMed/NCBI View Article : Google Scholar
|
|
120
|
Froes F, Timóteo A, Almeida B, Raposo JF,
Oliveira J, Carrageta M, Duque S and Morais A: Influenza
vaccination in older adults and patients with chronic disorders: A
position paper from the Portuguese Society of Pulmonology, the
Portuguese Society of Cardiology, the Portuguese Society of
Diabetology, the Portuguese Society of Infectious Diseases and
Clinical Microbiology, the Portuguese Society of Geriatrics and
Gerontology, and the Study Group of Geriatrics of the Portuguese
Society of Internal Medicine. Pulmonology. 30:422–436.
2024.PubMed/NCBI View Article : Google Scholar
|
|
121
|
Lutz CS, Biggerstaff M, Rolfes MA, Lafond
KE, Azziz-Baumgartner E, Porter RM, Reed C and Bresee JS:
Estimating the number of averted illnesses and deaths as a result
of vaccination against an influenza pandemic in nine low-and
middle-income countries. Vaccine. 39:4219–4230. 2021.PubMed/NCBI View Article : Google Scholar
|
|
122
|
Stuurman AL, Rizzo C and Haag M:
Investigating the procurement system for understanding seasonal
influenza vaccine brand availability in Europe. PLoS One.
16(e0248943)2021.PubMed/NCBI View Article : Google Scholar
|
|
123
|
Dhar R, Ghoshal AG, Guleria R, Sharma S,
Kulkarni T, Swarnakar R, Samaria JK, Chaudhary S, Gaur SN,
Christopher DJ, et al: Clinical practice guidelines 2019: Indian
consensus-based recommendations on influenza vaccination in adults.
Lung India. 37 (Supplement):S4–S18. 2020.PubMed/NCBI View Article : Google Scholar
|
|
124
|
Igboh LS, Roguski K, Marcenac P, Emukule
GO, Charles MD, Tempia S, Herring B, Vandemaele K, Moen A, Olsen
SJ, et al: Timing of seasonal influenza epidemics for 25 countries
in Africa during 2010-19: A retrospective analysis. Lancet Glob
Health. 11:e729–e739. 2023.PubMed/NCBI View Article : Google Scholar
|
|
125
|
Yoon D, Kim JH, Lee H and Shin JY: Updates
on vaccine safety and post-licensure surveillance for adverse
events following immunization in South Korea, 2005-2017. Yonsei Med
J. 61:623–630. 2020.PubMed/NCBI View Article : Google Scholar
|
|
126
|
Cuschieri S and Grech V: A comparative
assessment of attitudes and hesitancy for influenza vis-à-vis
COVID-19 vaccination among healthcare students and professionals in
Malta. Z Gesundh Wiss. 30:2441–2448. 2022.PubMed/NCBI View Article : Google Scholar
|
|
127
|
Wang SY, Pan XJ, Deng X, Zhang HJ and Chen
ZP: An exploratory framework of vaccine hesitancy monitoring and
early warning system in China. Zhonghua Yu Fang Yi Xue Za Zhi.
55:925–930. 2021.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
128
|
Lim CML, Komarasamy TV, Adnan NAAB,
Radhakrishnan AK and Balasubramaniam VR: Recent advances,
approaches and challenges in the development of universal influenza
vaccines. Influenza Other Respir Viruses. 18(e13276)2024.PubMed/NCBI View Article : Google Scholar
|
|
129
|
World Health Organization (WHO): SAGE
working group on vaccine hesitancy: Report. WHO, Geneva, 2024.
https://www.who.int/docs/default-source/immunization/sage/2014/october/sage-working-group-revised-report-vaccine-hesitancy.pdf.
|
|
130
|
Osterholm MT, Kelley NS, Sommer A and
Belongia EA: Efficacy and effectiveness of influenza vaccines: A
systematic review and meta-analysis. Lancet Infect Dis. 12:36–44.
2012.PubMed/NCBI View Article : Google Scholar
|
|
131
|
Bullard BL and Weaver EA: Strategies
targeting hemagglutinin as a universal influenza vaccine. Vaccines
(Basel). 9(257)2021.PubMed/NCBI View Article : Google Scholar
|
|
132
|
Mader K and Dustin LB: Beyond bNAbs: Uses,
risks, and opportunities for therapeutic application of
non-neutralising antibodies in viral infection. Antibodies (Basel).
13(28)2024.PubMed/NCBI View Article : Google Scholar
|
|
133
|
Fu Y, Zhang Z, Sheehan J, Avnir Y,
Ridenour C, Sachnik T, Sun J, Hossain MJ, Chen LM, Zhu Q, et al: A
broadly neutralizing anti-influenza antibody reveals ongoing
capacity of haemagglutinin-specific memory B cells to evolve. Nat
Commun. 7(12780)2016.PubMed/NCBI View Article : Google Scholar
|
|
134
|
Krammer F, Palese P and Steel J: Advances
in universal influenza virus vaccine design and antibody mediated
therapies based on conserved regions of the hemagglutinin. Curr Top
Microbiol Immunol. 386:301–321. 2015.PubMed/NCBI View Article : Google Scholar
|
|
135
|
Aartse A, Mortier D, Mooij P, Hofman S,
van Haaren MM, Corcoran M, Karlsson Hedestam GB, Eggink D,
Claireaux M, Bogers WMJM, et al: Primary antibody response after
influenza virus infection is first dominated by low-mutated HA-stem
antibodies followed by higher-mutated HA-head antibodies. Front
Immunol. 13(1026951)2022.PubMed/NCBI View Article : Google Scholar
|
|
136
|
Qiu X, Duvvuri VR and Bahl J:
Computational approaches and challenges to developing universal
influenza vaccines. Vaccines (Basel). 7(45)2019.PubMed/NCBI View Article : Google Scholar
|
|
137
|
Jackson NAC, Kester KE, Casimiro D,
Gurunathan S and DeRosa F: The promise of mRNA vaccines: A biotech
and industrial perspective. NPJ Vaccines. 5(11)2020.PubMed/NCBI View Article : Google Scholar
|
|
138
|
Petsch B, Schnee M, Vogel AB, Lange E,
Hoffmann B, Voss D, Schlake T, Thess A, Kallen KJ, Stitz L, et al:
Protective efficacy of in vitro synthesized, specific mRNA vaccines
against influenza A virus infection. Nat Biotechnol. 30:1210–1216.
2012.PubMed/NCBI View Article : Google Scholar
|
|
139
|
Park HJ, Bang YJ, Kwon SP, Kwak W, Park
SI, Roh G, Bae SH, Kim JY, Kwak HW, Kim Y, et al: Analyzing immune
responses to varied mRNA and protein vaccine sequences. NPJ
Vaccines. 8(84)2023.PubMed/NCBI View Article : Google Scholar
|
|
140
|
Huang J, Yuen D, Mintern JD and Johnston
AP: Opportunities for innovation: Building on the success of lipid
nanoparticle vaccines. Curr Opin Colloid Interface Sci.
55(101468)2021.PubMed/NCBI View Article : Google Scholar
|
|
141
|
Teng M, Yao Y, Nair V and Luo J: Latest
advances of virology research using CRISPR/Cas9-based gene-editing
technology and its application to vaccine development. Viruses.
13(779)2021.PubMed/NCBI View Article : Google Scholar
|
|
142
|
Al Fayez N, Nassar MS, Alshehri AA,
Alnefaie MK, Almughem FA, Alshehri BY, Alawad AO and Tawfik EA:
Recent advancement in mRNA vaccine development and applications.
Pharmaceutics. 15(1972)2023.PubMed/NCBI View Article : Google Scholar
|
|
143
|
Chauhan RP and Gordon ML: Review of genome
sequencing technologies in molecular characterization of influenza
A viruses in swine. J Vet Diagn Invest. 34:177–189. 2022.PubMed/NCBI View Article : Google Scholar
|
|
144
|
Gong X, Hu M, Chen W, Yang H, Wang B, Yue
J, Jin Y, Liang L and Ren H: Reassortment network of influenza A
virus. Front Microbiol. 12(793500)2021.PubMed/NCBI View Article : Google Scholar
|
|
145
|
Zhao AP, Li S, Cao Z, Hu PJH, Wang J,
Xiang Y, Xie D and Lu X: AI for science: predicting infectious
diseases. J Saf Sci Resil. 5:130–146. 2024.
|
|
146
|
Danchin A: Artificial intelligence-based
prediction of pathogen emergence and evolution in the world of
synthetic biology. Microb Biotechnol. 17(e70014)2024.PubMed/NCBI View Article : Google Scholar
|
|
147
|
Mohamed T, Sayed S, Salah A and Houssein
EH: Deep neural network for virus mutation prediction: A
comprehensive review. In: Integrating Meta-Heuristics and Machine
Learning for Real-World Optimization Problems. Studies in
Computational Intelligence. Houssein EH, Abd Elaziz M, Oliva D and
Abualigah L (eds). Vol 1038. Springer, Cham, 2022.
|
|
148
|
Gong M, Liu L, Sun X, Yang Y, Wang S and
Zhu H: Cloud-based system for effective surveillance and control of
COVID-19: useful experiences from Hubei, China. J Med Internet Res.
22(e18948)2020.PubMed/NCBI View
Article : Google Scholar
|
|
149
|
Mustafa MI and Makhawi AM: SHERLOCK and
DETECTR: CRISPR-Cas systems as potential rapid diagnostic tools for
emerging infectious diseases. J Clin Microbiol. 59:e00745–20.
2021.PubMed/NCBI View Article : Google Scholar
|
|
150
|
Kulkarni A, Tanga S, Karmakar A, Hota A
and Maji B: CRISPR-based precision molecular diagnostics for
disease detection and surveillance. ACS Appl Bio Mater.
6:3927–3945. 2023.PubMed/NCBI View Article : Google Scholar
|
|
151
|
Umar Ibrahim A, Pwavodi PC, Ozsoz M,
Al-Turjman F, Galaya T and Agbo JJ: CRISPR biosensing and AI-driven
tools for detection and prediction of COVID-19. J Exp Theor Artif
Intell. 35:489–505. 2023.
|
|
152
|
Rutherford N: The transductive flu:
Disruptive virality and biosocial immunity (unpublished thesis).
Université d'Ottawa, 2024.
|
|
153
|
Hannings AN, Darley A, Kearney JC,
Upchurch BL and Groft K: Marketing mobile influenza vaccine clinics
on a college campus. J Am Pharm Assoc. 62:551–554.e1.
2022.PubMed/NCBI View Article : Google Scholar
|
|
154
|
Tuckerman J, Kaufman J and Danchin M:
Effective approaches to combat vaccine hesitancy. Pediatr Infect
Dis J. 41:e243–e245. 2022.PubMed/NCBI View Article : Google Scholar
|
|
155
|
Steffens MS, Dunn AG, Leask J and Wiley
KE: Using social media for vaccination promotion: practices and
challenges. Digital Health. 6(2055207620970785)2020.PubMed/NCBI View Article : Google Scholar
|
|
156
|
Benjamin-Chung J, Arnold BF, Kennedy CJ,
Mishra K, Pokpongkiat N, Nguyen A, Jilek W, Holbrook K, Pan E and
Kirley PD: Evaluation of a city-wide school-located influenza
vaccination program in Oakland, California, with respect to
vaccination coverage, school absences, and laboratory-confirmed
influenza: a matched cohort study. PLoS Med.
17(e1003238)2020.PubMed/NCBI View Article : Google Scholar
|
|
157
|
Gualano MR, Santoro PE, Borrelli I, Rossi
MF, Amantea C, Tumminello A, Daniele A, Beccia F and Moscato U:
Employee participation in workplace vaccination campaigns: A
systematic review and meta-analysis. Vaccines.
10(1898)2022.PubMed/NCBI View Article : Google Scholar
|
|
158
|
Hill I, Porter Z, Mayer J, Verdeflor A,
Brown K and Lawrence D: Building the evidence base for advancing
vaccine equity. 2023.
|
|
159
|
Friede M, Palkonyay L, Alfonso C, Pervikov
Y, Torelli G, Wood D and Kieny MP: WHO initiative to increase
global and equitable access to influenza vaccine in the event of a
pandemic: supporting developing country production capacity through
technology transfer. Vaccine. 29:A2–A7. 2011.PubMed/NCBI View Article : Google Scholar
|
|
160
|
World Health Organization (WHO): Influenza
(avian and other zoonotic). WHO, Geneva, 2026. https://www.who.int/news-room/fact-sheets/detail/influenza-%28avian-and-other-zoonotic%29.
Accessed March 5, 2026.
|
|
161
|
Centers for Disease Control and Prevention
(CDC): 1957-1958 pandemic (H2N2 virus). CDC, Atlanta, GA, 2019.
https://archive.cdc.gov/www_cdc_gov/flu/pandemic-resources/1957-1958-pandemic.html.
Accessed March 5, 2026.
|
|
162
|
Centers for Disease Control and Prevention
(CDC): 1968 pandemic (H3N2 virus). CDC, Atlanta, GA, 2019.
https://archive.cdc.gov/www_cdc_gov/flu/pandemic-resources/1968-pandemic.html.
Accessed March 5, 2026.
|
|
163
|
Komadina N, McVernon J, Hall R and Leder
K: A historical perspective of influenza A(H1N2) virus. Emerg
Infect Dis. 20:6–12. 2014.PubMed/NCBI View Article : Google Scholar
|
|
164
|
Jester BJ, Uyeki TM and Jernigan DB: Fifty
years of influenza A(H3N2) following the pandemic of 1968. Am J
Public Health. 110:669–676. 2020.PubMed/NCBI View Article : Google Scholar
|
|
165
|
Centers for Disease Control and Prevention
(CDC): Types of influenza viruses. CDC, Atlanta, GA, 2025.
https://www.cdc.gov/flu/about/viruses-types.html.
Accessed March 5, 2026.
|
|
166
|
Centers for Disease Control and Prevention
(CDC): How flu viruses can change: ‘Drift’ and ‘Shift’. CDC,
Atlanta, GA, 2024. https://www.cdc.gov/flu/php/viruses/change.html.
Accessed March 5, 2026.
|
|
167
|
Centers for Disease Control and Prevention
(CDC): Understanding influenza viruses. CDC, Atlanta, GA, 2024.
https://www.cdc.gov/flu/php/viruses/index.html.
Accessed March 5, 2026.
|
|
168
|
World Health Organization: Global
influenza surveillance and response system (GISRS). https://www.who.int/initiatives/global-influenza-surveillance-and-response-system.
Accessed March 5, 2026.
|
|
169
|
World Health Organization (WHO):
Recommendations for influenza vaccine composition for the 2026-2027
northern hemisphere season. WHO, Geneva, 2026. https://www.who.int/news/item/27-02-2026-recommendations-for-influenza-vaccine-composition-for-the-2026-2027-northern-hemisphere-season.
Accessed March 5, 2026.
|
|
170
|
World Health Organization (WHO):
Recommended composition of influenza virus vaccines for use in the
2026-2027 northern hemisphere influenza season. WHO, Geneva, 2026.
https://www.who.int/publications/m/item/recommended-composition-of-influenza-virus-vaccines-for-use-in-the-2026-2027-northern-hemisphere-influenza-season.
Accessed March 5, 2026.
|
|
171
|
World Health Organization (WHO): Questions
and answers: Recommended composition of influenza virus vaccines
for use in the 2026-2027 northern hemisphere influenza season. WHO,
Geneva, 2026. https://cdn.who.int/media/docs/default-source/vcm-northern-hemisphere-recommendation-2026-2027/b.-27-feb-2026-frequently-asked-questions.pdf.
Accessed March 5, 2026.
|
|
172
|
World Health Organization: Global
influenza virological surveillance (FluNet/GISRS overview).
https://www.who.int/data/gho/data/themes/global-influenza-virological-surveillance.
Accessed March 5, 2026.
|
|
173
|
Hay AJ and McCauley JW: The WHO global
influenza surveillance and response system (GISRS)-A future
perspective. Influenza Other Respir Viruses. 12:551–557.
2018.PubMed/NCBI View Article : Google Scholar
|
















