Introduction
Worldwide, breast cancer is the most prevalent
malignant tumor and the leading cause of cancer-related mortality
among women (1,2). Although there is evidence of an
association with estrogen-receptor (ER)-positive tumors, the
association between weight and breast cancer risk is often greater
for ER-positive breast malignancies (3,4).
According to the Iraqi Cancer Registry (ICR) in 2023, breast cancer
affects 20.5% of all patients with cancer (https://storage.moh.gov.iq/2024/11/24/2024_11_24_12127028949_4299728097670824.pdf).
Arachidonic acid and other fatty acids are converted by the
cyclooxygenase (COX)-2 pathway into a variety of prostaglandins,
prostacyclins and thromboxanes (5). Prostaglandin (PG) E2
(PGE2) stimulates E-type prostanoid receptor activation,
which in turn enhances cellular proliferation, promotes
angiogenesis, inhibits apoptosis, stimulates invasion and motility,
and suppresses immunological responses (6). PGE2 and its receptors play
a prominent role in driving cancer progression (7). PGs belong to the eicosanoid family
and are produced by the majority of cells in the human body
(8). COX-2 overexpression induces
cancer by promoting angiogenesis, reducing tumor immunity and
promoting PGE2 production (6). Different functions are performed by
COX isoforms in healthy and pathological circumstances. In the
majority of tissues, COX-1 is typically produced constantly,
regulating physiological processes, including vascular homeostasis
or cellular responses to hormone stimulation. When growth factors
or inflammation are stimulated, COX-2 is primarily expressed, and
this frequently results in an increased synthesis of
PGE2 in tumor or inflammatory tissue (9). The inducible enzyme COX-2 is linked
to inflammatory illnesses and cancer. Furthermore, it may also
promote angiogenesis, tumor tissue invasion and resistance to
apoptosis (10). Up to 40% of the
expression of COX-2 enzyme in human breast cancer is connected with
numerous factors, including age, tumor size, a high grade, a
negative hormone receptor status, a high proliferation rate and the
presence of human EGFR-2 oncogene amplification (11).
The aim of the present study was to examine the
association of the COX-2 and PGE2 concentrations in the
sera and urine of women with breast cancer, as well as to evaluate
the levels of these parameters to determine their potential use in
predicting disease occurrence. Furthermore, the present study aimed
to evaluate whether the urine test for COX-2 and PGE2
can be used as an alternative to the sera test.
Patients and methods
Patient selection
The present study included 88 women that were
classified into three groups, as follows: Group 1 included 29
patients with breast cancer prior to surgery and without
pharmaceutical treatment, group 2 was comprised of 29 first-degree
relatives, and group 3 included 30 healthy subjects as the
controls; the age of the participants ranged from 25 to 57 years.
All patients and first-degree relatives attended the Oncology
Teaching Hospital in Medical City (Baghdad, Iraq) during the time
of September, 2024 to April, 2025. All women with breast cancer had
invasive ductal carcinoma and did not suffer from any other
malignant diseases; 45% of them had a family history of tumors, and
55% of them had no family history. All patients were ER-positive,
human epidermal growth factor receptor 2 (HER2)-negative, and had
grade II, and stage I and II disease. All the patients were
non-smokers, not pregnant, and had not taken any anti-inflammatory
or non-steroidal anti-inflammatory drugs. First-degree relatives of
the patients with breast cancer were included as a distinct study
group to represent individuals with familial predisposition and a
potentially increased risk of developing breast cancer. This group
was included in an aim to explore whether COX-2 levels demonstrate
differential patterns across healthy controls, individuals with
familial risk and patients with a confirmed breast cancer. This
group was included for exploratory and hypothesis-generating
purposes and was not intended to support screening or clinical risk
stratification. The research Ethics Committee of Mustansiriyah
University (Baghdad, Iraq) reviewed and approved the present study
(approval no. BCSMU/0924/0042C). Ethics approval was obtained from
the author's institution as the governing document. This approval
was presented to the administrations of Oncology Teaching Hospital
in Medical City. As these are affiliated teaching hospitals with
established inter-institutional collaboration protocols, they
granted administrative site permission for sample collection based
on the university's central ethics clearance. Every patient who
took part in the research provided written, informed consent. It
was made clear to the participants that participation was
completely voluntary and that there would be no consequences if
participants opted to withdraw from the study at any moment.
Samples
A total of 5 ml fasting blood and 10 ml midstream
random spot urine in the morning was collected after the study
subjects gave their agreement and completed the face to face
questionnaire, which included (full name, age, weight, hight,
family history of cancer, any other diseases, any medications,
marriage status, number of children, smoking status, and the use of
any contraceptives). At room temperature, blood was allowed to
stand for 30 min to allow complete clotting. Urine samples were
collected from all the women participating in the study into
disposable screw cup containers for the estimation of the study
parameters. The collected specimens were centrifuged at 4,000 rpm
for 10 min. The creatinine concentration was measured in the urine
samples immediately. The sera and the second part of the urine was
separated into small aliquots and stored at -20˚C until use to
measure the COX-2 and PGE2 concentrations.
Calculation of body mass index
(BMI)
BMI was calculated by dividing the weight by the
square of the height, as follows: BMI=weight (kg)/height
(m2).
Sera and urine marker analysis
With the use of enzyme-linked immunosorbent assay
(ELISA), the serum and urinary COX-2 concentrations were measured
using the Human PTGS2/COX-2 ELISA kit [cat. no. ELK2096, Elk
(Wuhan) Biotechnology Co., Ltd.]. The assay has a reported lower
limit of detection of 0.125 ng/ml and a detection range of 0.32-20
ng/ml. The manufacturer states that the coefficients of variation
within and between assays are <8 and 10%, respectively. The
PGE2 levels were quantified using the Human
PGE2 ELISA kit [cat. no. ELK9315, Elk (Wuhan)
Biotechnology Co., Ltd.]. This competitive ELISA has a reported
sensitivity of 0.97 pg/ml and a detection range of 3.13-200 pg/ml,
with high specificity and minimal cross-reactivity. At a
wave-length of 450 nm, the optical density (OD) was determined on a
96-well microplate reader and the COX-2 and PGE2
concentrations are expressed in pg/ml. Urine creatinine
concentrations were measured by colorimetric assay using the
automatic Roche/Hitachi cobas c111 System and the concentrations
are expressed in mg/dl. The COX-2 and PGE2
concentrations in urine were divided by the level of creatinine in
urine for the purpose of providing a representative sample,
enabling us to analyzed these parameters without the need for
collecting urine for 24 h.
Statistical analysis
The data were analyzed using SPSS software version
25 (IBM Corp.). Data are presented as the mean ± SD, and analyzed
using one-way ANOVA, followed by Tukey's honest significant
difference (HSD) test for post hoc pairwise comparisons to control
type I error. Pearson's correlation analysis was performed to
correlations among all parameters (12). To assess the accuracy of COX-2 and
PGE2 parameters, receiver operating characteristic (ROC)
analysis was performed and the area under the curve (AUC) was
calculated (13). Cut-off values
for serum and urinary COX-2 and PGE2 were determined
using ROC curve analysis and the Youden index, based on the study
cohort. No internal or external validation procedures were applied.
A value of P<0.05 was considered to indicate a statistically
significant difference.
Results
In total, 45% of the patients in the present study
some had a family history of tumors, with the remainder having no
family history. Some physical features of the study groups are
presented in Table I. The
difference in age and BMI between these groups was not
statistically significant (P>0.05). The post hoc comparison
indicated that all three groups were statistically similar in terms
of age and BMI.
 | Table IAge and BMI among all the study
groups. |
Table I
Age and BMI among all the study
groups.
| Parameter | Breast cancer
(n=29) | First-degree
relatives (n=29) | Control (n=30) | P-valuea | P-valueb | P-valuec | P-valued |
|---|
| Age (years) | 43.62±8.36 | 40.35±10.51 | 40.96±7.01 | 0.365 | 0.964 | 0.471 | 0.334 |
| BMI (kg/m2) | 28.68±3.66 | 26.99±3.12 | 27.07±2.67 | 0.147 | 0.996 | 0.138 | 0.090 |
The data presented in Table II revealed a significant
difference in the serum levels of COX-2 among the groups, with the
breast cancer group manifesting the highest levels (40.21±7.66),
followed by the first-degree relatives (30.91±2.63) and the control
group (21.97±5.74). This finding was substantiated by a P-value of
0.001 underscoring the statistical significance. The post hoc
analysis labels further demonstrated the distinction among all
three groups.
 | Table IICOX-2 enzyme level in sera and the
urine ratio of COX-2 by the level of creatinine in the urine of
patients with breast cancer, first-degree relatives and healthy
controls. |
Table II
COX-2 enzyme level in sera and the
urine ratio of COX-2 by the level of creatinine in the urine of
patients with breast cancer, first-degree relatives and healthy
controls.
| Parameter | Breast cancer
(n=29) | First-degree
relatives (n=29) | Control (n=30) |
P-valuea |
P-valueb |
P-valuec |
P-valued |
|---|
| COX-2 sera
(pg/ml) | 40.21±7.66 | 30.91±2.63 | 21.97±5.74 |
0.030* | 0.001 | 0.001 | 0.001 |
| COX-2 urine
(pg/ml) | 20.55±3.94 | 16.87±1.06 | 39.14±1.77 | 0.001 | 0.001 | 0.001 | 0.001 |
| COX-2/creatinine in
urine (x109) | 2.0±0.92 | 1.62±0.53 | 3.58±1.81 | 0.516 | 0.001 | 0.001 | 0.001 |
Notably, the COX-2 urine levels portrayed an
opposite trend to those of the serum levels. The control group
displayed markedly higher levels (39.14±1.77) as compared to the
breast cancer group (20.55±3.94) and first-degree relatives
(16.87±1.06). The difference was statistically significant
(P<0.001). The post hoc analysis indicated there were
significant differences between the groups (Table II).
The ratio of COX-2 in urine by the level of
creatinine in urine is demonstrated in Table II. The results revealed that there
was a highly significant difference (P<0.001) between the breast
cancer and control groups, and no significant differences were
observed between the breast cancer first-degree relatives group
(P=0.516).
A detailed investigation of the serum and urine
levels of PGE2 across the three different groups was
performed. The results are summarized in Table III. The serum levels of
PGE2 across the breast cancer, control and first-degree
relatives groups displayed mean values of (14.41±7.24, 18.88±8.23
and 15.99±11.82 pg/ml, respectively. Despite these numerical
variations, statistical analysis yielded a P-value of 0.17,
indicating that these differences were not statistically
significant.
 | Table IIIComparison of the sera and urine
level of PGE2 and the ratio of PGE2 by the
level of creatinine in urine in all study groups. |
Table III
Comparison of the sera and urine
level of PGE2 and the ratio of PGE2 by the
level of creatinine in urine in all study groups.
| Parameter | Breast cancer
(n=29) | First-degree
relatives (n=29) | Control (n=30) |
P-valuea |
P-valueb |
P-valuec |
P-valued |
|---|
| PGE2
sera (pg/ml) | 14.41±7.24 | 15.99±11.82 | 18.88±8.23 | 0.806 | 0.495 | 0.153 | 0.173 |
| PGE2
urine (pg/ml) | 52.57±16.10 | 48.75±15.59 | 56.02±16.83 | 0.678 | 0.250 | 0.698 | 0.281 |
|
PGE2/creatinine in urine
(x109) | 5.14±2.56 | 4.69±2.31 | 4.98±2.95 | 0.816 | 0.921 | 0.969 | 0.830 |
Similarly, the urinary levels of PGE2
among the three groups do not exhibit a statistically significant
difference (P-value=0.28). Table
III also presents the ratio of the PGE2 level in
urine by the level of creatinine in urine; the results revealed
that there were no significant differences among the three
groups.
Pearson's correlation analysis (Table IV) provided valuable insight into
the correlations between multiple variables in the context of sera
in all the study groups. According to the results presented in
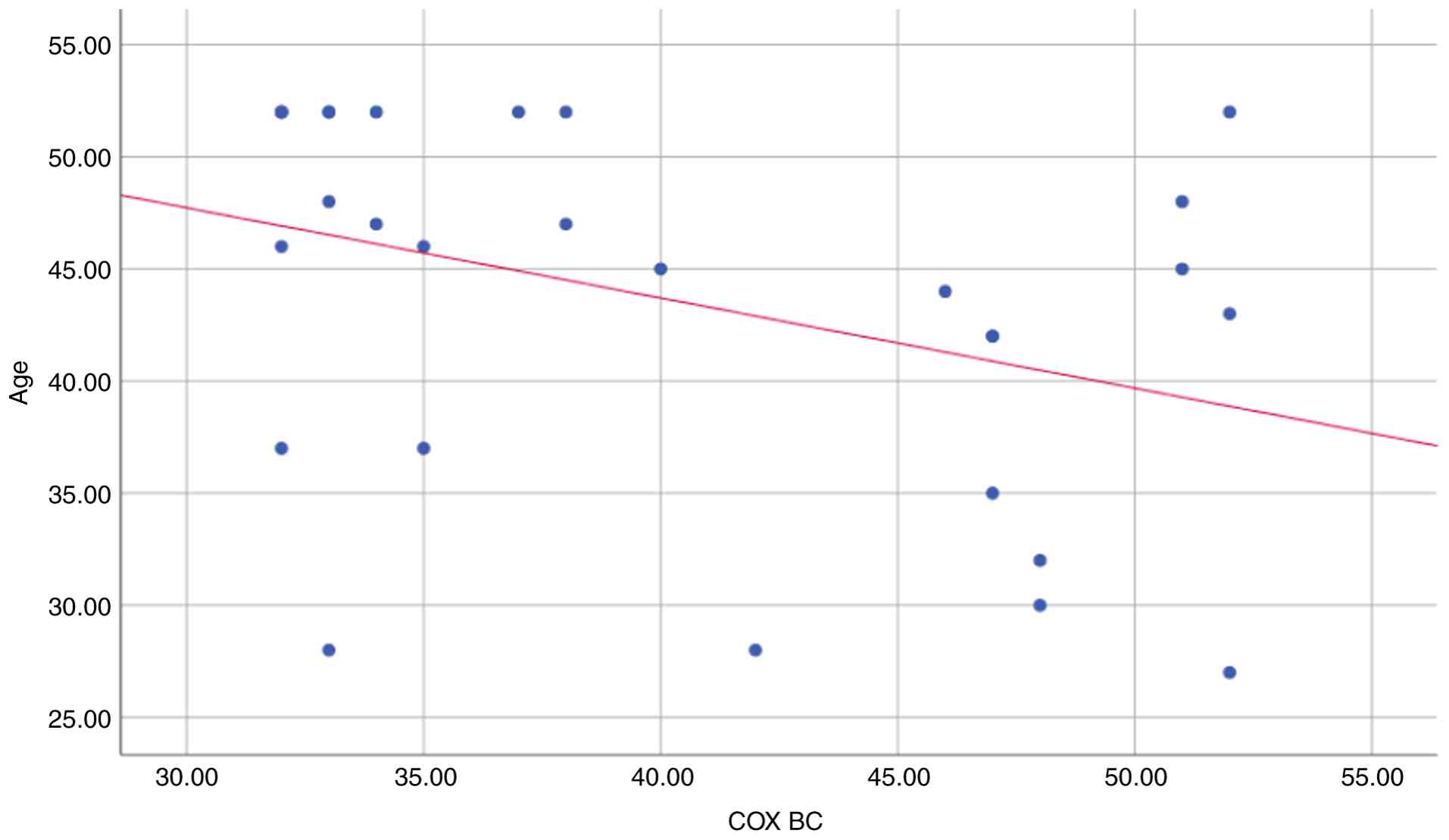
Table IV and Fig. 1, age and serum COX-2 in the breast
cancer group exhibited a significant negative correlation
(r=-0.368, P=0.049). Furthermore, there was no correlation observed
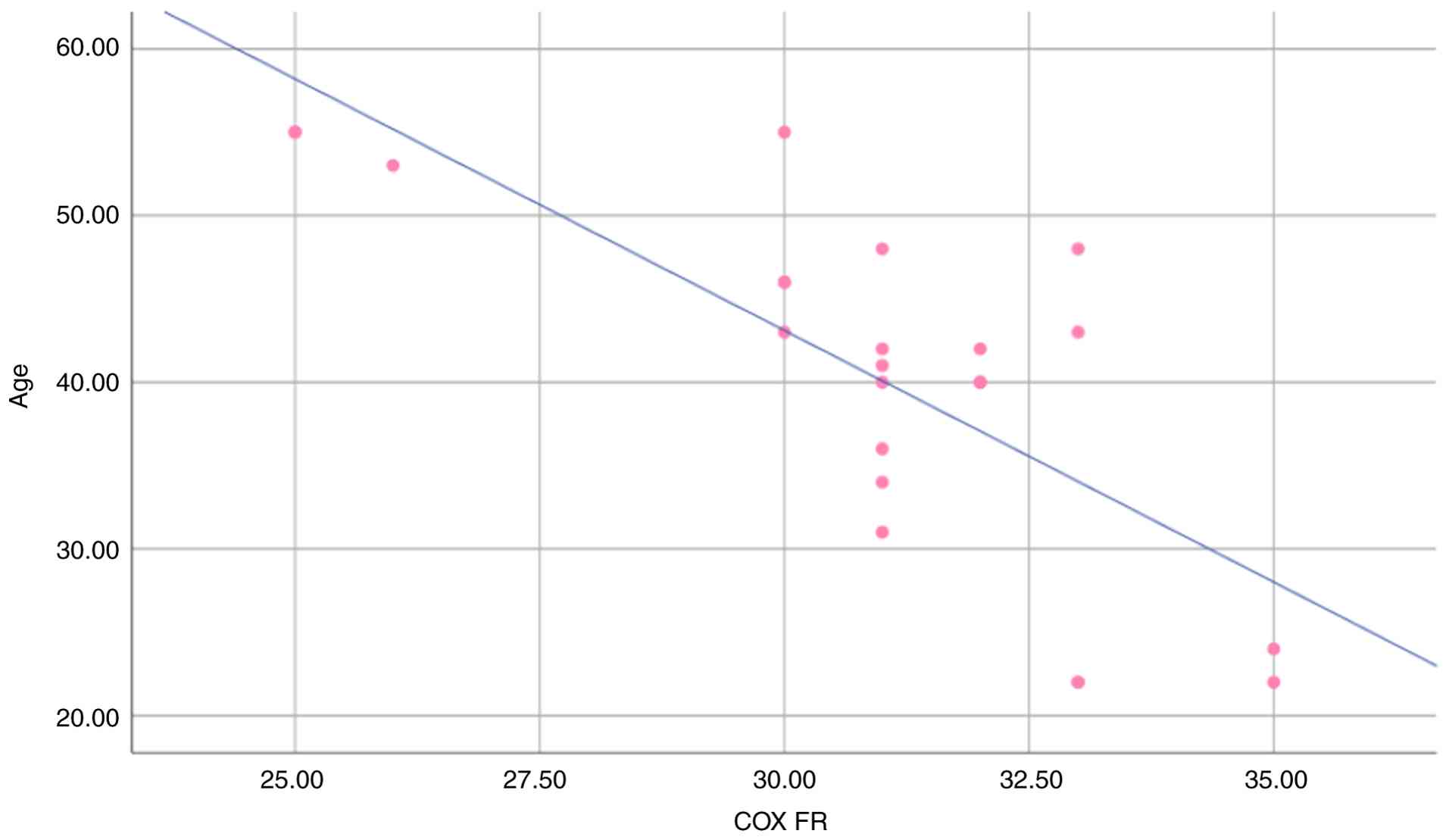
between other parameters. In first-degree relatives group, there
was a strong negative correlation between age and serum COX-2
(r=-0.755, P=0.001), as demonstrated in Table IV and Fig. 2. In the control group, there was no
correlation among all parameters.
 | Table IVCorrelations among the levels of all
studied parameters in serum in all study groups. |
Table IV
Correlations among the levels of all
studied parameters in serum in all study groups.
| Parameter | COX-2 BC group | PGE2 BC
group | COX-2 FR group | PGE2 FR
group | COX-2 control
group | PGE2
control group |
|---|
| Age (years) | | | | | | |
|
r | -0.368 | -0.162 | -0.755b | -0.163 | 0.221 | 0.156 |
|
P-value | 0.049a | 0.402 | 0.001 | 0.456 | 0.250 | 0.418 |
| BMI
(kg/m2) | | | | | | |
|
r | 0.253 | -0.169 | -0.351 | 0.236 | 0.086 | 0.121 |
|
P-value | 0.185 | 0.379 | 0.101 | 0.278 | 0.656 | 0.531 |
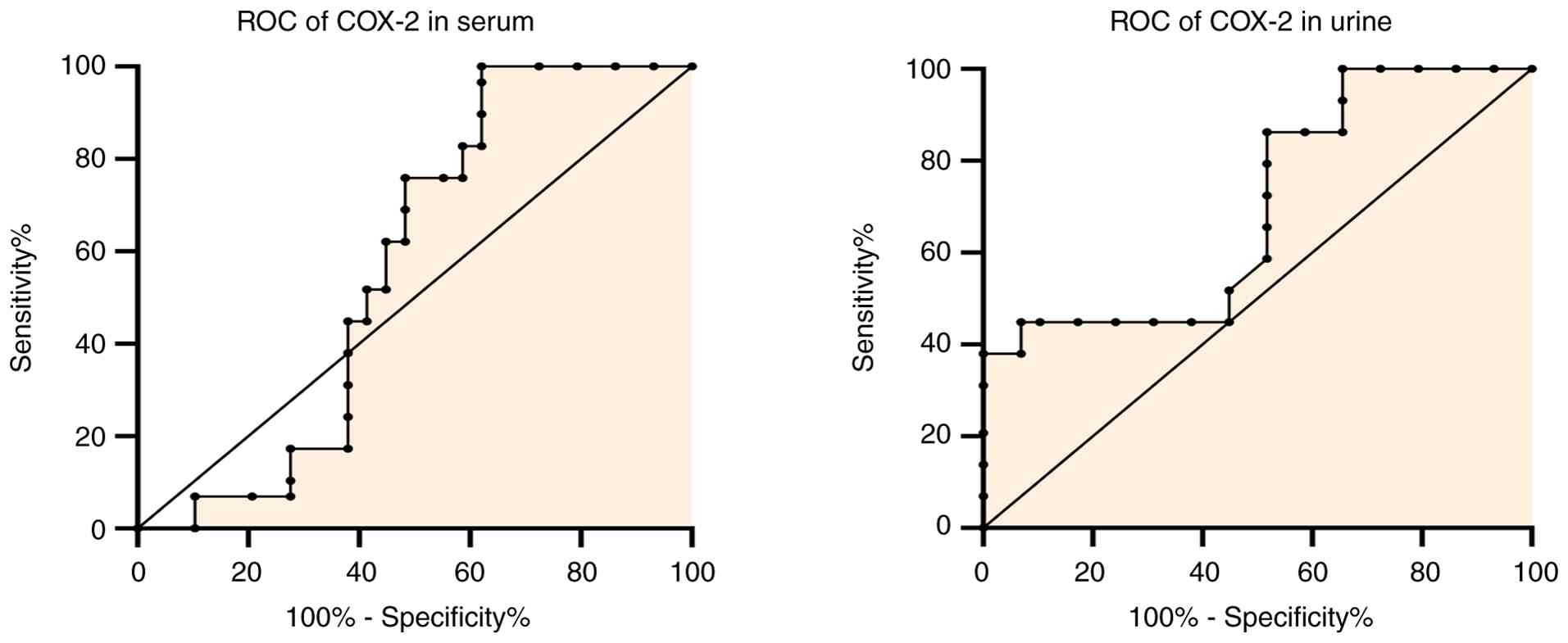
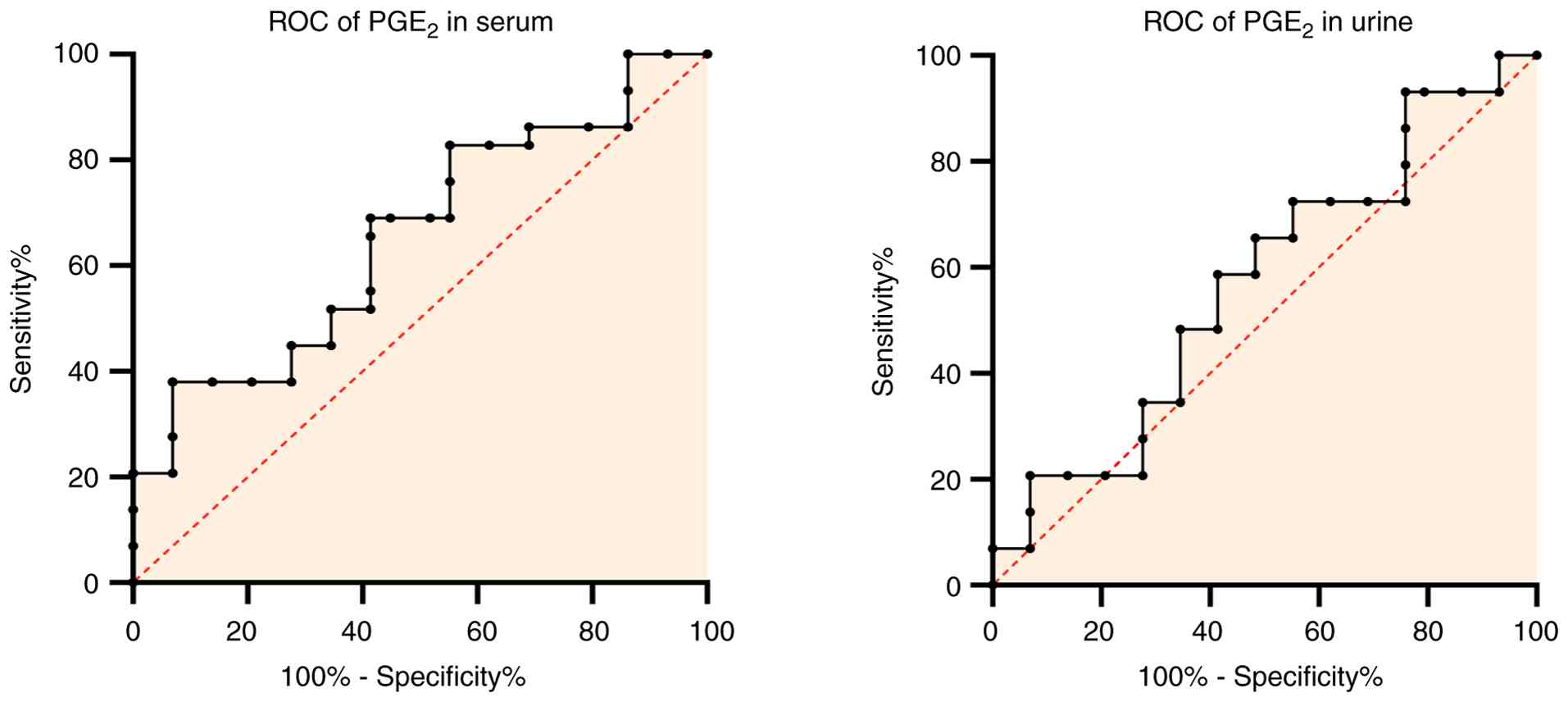
The diagnostic performance of various biomarkers in
differentiating between cases of breast cancer and controls is
illustrated in Figs. 3 and
4. Sera and urine COX-2 exhibited
exceptional diagnostic accuracy, as indicated by a sensitivity and
specificity of 100% and an AUC of 1.00. These findings suggest that
these particular markers are extremely reliable for distinguishing
patients with breast cancer from controls. On the other hand, the
serum and urine PGE2 ROC curve demonstrated a
considerably similar efficacy, with a sensitivity of 38%,
specificity of 93%, and an AUC of 0.66 for serum with a cut-off
value of 9.93, and a sensitivity and specificity of 59%, and an AUC
of 0.57 for urine with a cut-off value of 48.77.
Discussion
Given the increasing prevalence of breast cancer and
the absence of reliable biomarkers for early prediction, the
present study was conducted in an aim to contribute to the
identification of novel markers in serum and urine that may assist
in the early detection of breast cancer. The mean ± SD age of the
patients in the present study was 40.62±12.36 years. As previously
demonstrated, the risk of acquiring breast cancer increases with
age as follows: A 1.5% risk at age 40, 3% at age 50, and >4% at
age 70 years (14). Women who have
had long-term estrogen circulation in their bodies are more
susceptible to developing breast cancer (15). An increased number of menstrual
cycles also increases the risk of developing breast cancer. Women
who menarche early and menopause at a later stage are more likely
to develop breast cancer (16).
BMI is classified as follows: Underweight (<18
kg/m2) and normal weight (18.5-24.9 kg/m2).
While overweight (BMI between 25.0 and 29.9 kg/m2),
class I obesity (BMI of 30.0 to 34.9 kg/m2), class II
obesity (BMI of 35.0 to 39.9 kg/m2) and class III or
severe obesity (BMI of 40 kg/m2) are all examples of
obesity, the medical risk increases gradually as the degree of
obesity increases (17,18). In the present study, the results
summarized in Table I demonstrate
that the BMI the patients in all the groups was >25
kg/m2, indicating that the population in the present
study was overweight. Epidemiological data indicate that obesity
increases the risk of developing breast cancer (19). According to the study by
Fontvieille et al (20)
women >50 years of age with a higher BMI are at a greater risk
of developing cancer compared to those with a lower BMI.
Additionally, it has been found that a higher BMI is linked to more
aggressive biological characteristics of the tumor, such as a
larger size and a higher percentage of lymph node metastasis.
According to the meta-analysis by Smith et al (21), there was a low positive association
between the risk of developing breast cancer and BMI, with an
increase in BMI of 5 kg/m2 being associated with an
increase in the risk of developing breast cancer of 2%.
Nonetheless, among premenopausal women, a higher BMI reduces the
risk of developing breast cancer. Obesity and overweight are
considered to provide protection against premenopausal breast
cancer, with the exception of women having a family history of
breast cancer. Therefore, body fat may be a better measure of
breast cancer risk in postmenopausal women than BMI or body weight.
Body fat distribution may also influence the risk of developing
breast cancer (22). According to
observational data, women with and without a family history of
breast cancer may have a similar link between their BMI and the
risk of developing the disease (23).
The results of the present study demonstrated a
significant increase in serum COX-2 in patients with breast cancer
compared with the first-degree relatives and the controls. These
results are in line with those of the study by Habeeb (24). Moreover, in urine, there were
highly significant differences between the control group and
patients with breast cancer and, first-degree relatives
(P<0.001). Previous research has revealed a correlation between
COX-2 expression and the ER (25),
progesterone receptor and HER-2 positive status (26). The enzyme aromatase, which converts
testosterone to estrogen, is expressed and activated to a greater
extent when PGs are present. Through the activation of the ER and
its target genes, estrogen can promote the development of cancer
cells (27). According to the
study by Jana et al (28),
COX-2 may promote the growth and angiogenesis of breast tumors
through this mechanism. According to that study, COX-2 expression
was detected in invasive and in situ ductal carcinoma, but
not in normal breast tissue (28).
In 2014, in the USA, the overexpression of COX-2 was repeatedly
reported in all stages of breast cancer by molecular biologists
from numerous independent laboratories (29). In 2021, in India, Bhutani et
al (30) examined the spectrum
of COX-2 expression in normal breast tissue, and compared COX-2
expression with histological prognostic factors and hormone
receptor status in ductal carcinoma in situ next to and inside
invasive carcinoma. They found that the reduction of COX-2 may
provide a potential target for the prevention of breast cancer
oncogenesis and as an adjuvant treatment following surgery to
minimize local recurrence (30).
In the present study, the results of ROC curve analysis (Fig. 3) demonstrated that sera and urine
COX-2 had the high diagnostic efficacy in the diagnosis of BC
(AUC=1).
COX-2 expression inhibits GSK3β enzyme activity,
which is a major Wnt component that phosphorylates β-catenin and
stimulates its eventual destruction via proteasome (31,32).
In urine, the reduced concentration of COX-2 may cause the
activation of β-catenin mediated transcriptional signaling and the
disruption of the balance between cell proliferation and
differentiation thus paving the way for tumorigenesis (33).
In the present study, there were no significant
differences in the PGE2 concentration in serum and urine
among the three groups. The relative rates of
15-hydroxyprostaglandin dehydrogenase dependent breakdown and
COX-2/PGE synthase dependent production determine the steady state
cellular levels of PGE2. Ilyan et al (34) examined the association between the
serum levels of PGE2, COX-2, and 25-hydroxyvitamin D in
patients with breast cancer in Indonesia. They demonstrated that
serum PGE2 and vitamin D levels varied according to the
stage of breast cancer; the PGE2 serum level and breast
cancer stage were not significantly associated with serum 25(OH) D
levels (34). Herein, there was no
significant association between COX-2 levels in blood and urine;
therefore, urine cannot be used as a substitute for blood, and vice
versa.
There are certain limitations to the present study,
which should be mentioned. The statistical power may have been
diminished by the comparatively small sample size, particularly for
diagnostic accuracy analyses such as ROC curves. In addition, the
study population was limited to patients with ER-positive,
HER2-negative, grade II, early-stage breast cancer, which
represents a highly selected cohort and limits the generalizability
of the results to other molecular subtypes and disease stages.
In conclusion, the results of the present study
demonstrated an elevation in COX-2 levels during the early stages
of breast cancer, indicating its potential role in disease
progression. The ROC analysis further suggested that this enzyme
may serve as an effective diagnostic biomarker. Moreover, the
measurements of the PGE2 concentration revealed the
absence of severe inflammation among patients, consistent with
their classification in the early stages of the disease.
Acknowledgements
The authors would like to express their gratitude to
Dr Mohammed Abdul Latif from the College of Medicine, Al-Nahrain
University, Baghdad, Iraq for his valuable advice on statistical
analysis. They also extend their sincere thanks to Dr Ali Abdul
Hussein from the College of Medical Technology, University of
Baghdad, Baghdad, Iraq for his assistance in conducting the
ELISA.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
HQM edited the manuscript, and was also involved in
the conceptualization and design of the study. TJT and RSM
collected and analyzed data. MIH oversaw the study, participated in
data curation, and reviewed and edited the report. The final text
has been reviewed and approved by all authors. HQM and TJT confirm
the authenticity of all the raw data.
Ethics approval and consent to
participate
The present study was examined and authorized by the
Research Ethics Committee of Mustansiriyah University, Baghdad,
Iraq (approval no. BCSMU/0924/0042C), and subsequently authorized
by THE Oncology Teaching Hospital/Medical City administration per
standard institutional collaboration procedures. Every patient who
took part in the study provided written, informed consent. It was
made to the participants that their participation was completely
voluntary and that there would be no consequences if they opted to
withdraw from the study at any moment. By using coded IDs instead
of names or medical record numbers, anonymity and confidentiality
were protected. The study upheld high ethical standards in
accordance with international ethical norms, such as the
Declaration of Helsinki.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Bhushan A, Gonsalves A and Menon JU:
Current state of breast cancer diagnosis, treatment, and
theranostics. Pharmaceutics. 13(723)2021.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Al-Ganimi AK and Abd Al-Salam AS:
Incidence of breast cancer among blood groups of women in the holy
Governorate of Karbala. Med J Babylon. 20:338–340. 2023.
|
|
3
|
Clusan L, Ferrière F, Flouriot G and
Pakdel F: A basic review on estrogen receptor signaling pathways in
breast cancer. Int J Mol Sci. 24(6834)2023.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Bakheet MM, Ali HM and Talab TJ:
Evaluation of some proinflammatory cytokines and biochemical
parameters in pre and postmenopausal breast cancer women. Cytokine.
179(156632)2024.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Wang B, Wu L, Chen J, Dong L, Chen C, Wen
Z, Hu J, Fleming I and Wang DW: Metabolism pathways of arachidonic
acids: Mechanisms and potential therapeutic targets. Signal
Transduct Target Ther. 6(94)2021.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Santiso A, Heinemann A and Kargl J:
Prostaglandin E2 in the tumor microenvironment, a convoluted affair
mediated by EP receptors 2 and 4. Pharmacol Rev. 76:388–413.
2024.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Jin K, Qian C, Lin J and Liu B:
Cyclooxygenase-2-prostaglandin E2 pathway: A key player in
tumor-associated immune cells. Front Oncol.
13(1099811)2023.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Jara-Gutiérrez Á and Baladron V: The role
of prostaglandins in different types of cancer. Cells.
10(1487)2021.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Walker OL, Dahn ML, Power Coombs MR and
Marcato P: The prostaglandin E2 pathway and breast cancer stem
cells: Evidence of increased signaling and potential targeting.
Fronti Oncol. 11(791696)2022.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Ye Y, Wang X, Jeschke U and von Schönfeldt
V: COX-2-PGE2-EPs in gynecological cancers. Arch Gynecol Obstet.
301:1365–1375. 2020.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Sahu A, Raza K, Pradhan D, Jain AK and
Verma S: Cyclooxygenase-2 as a therapeutic target against human
breast cancer: A comprehensive review. WIREs Mech Dis.
15(e1596)2023.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Norman G: Likert scales, levels of
measurement and the ‘laws’ of statistics. Adv Health Sci Educ
Theory Pract. 15:625–632. 2010.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Hajian-Tilaki K: Receiver operating
characteristic (ROC) curve analysis for medical diagnostic test
evaluation. Caspian J Intern Med. 4:627–235. 2013.PubMed/NCBI
|
|
14
|
Zhu JW, Charkhchi P, Adekunte S and Akbari
MR: What is known about breast cancer in young women? Cancers.
15(1917)2023.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Bieuville M, Faugère D, Galibert V, Henard
M, Dujon AM, Ujvari B, Pujol P, Roche B and Thomas F: Number of
lifetime menses increases breast cancer occurrence in
postmenopausal women at high familial risk. Front Ecol Evolution.
11(912083)2023.
|
|
16
|
Das U, Soren S and Kar N: Menstrual and
reproductive factors associated with risk of breast cancer among
Indian women: A cross sectional study from National Family Health
Survey, 2019-21. Arch Public Health. 82(55)2024.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Jan A and Weir CB: BMI classification
percentile and cut off points. StatPearls, Treasure Island, FL,
USA, 1-4, 2021.
|
|
18
|
Al-Shimmery AH, Al-Alwany MH, Chabuck ZA,
Al-Mammori RT, Mokif TA, Mahdi ZA, Al-Dahmoshi HO, Al-Khafaji NS,
Al-Hindy HA, Abed SY and Abdulabbas HS: Assessment of tumor
necrosis factor-α, interleukin-17, and vitamin D3 levels on a group
of gastrointestinal tumor patients in Babylon Provence, Iraq. Med J
Babylon. 20:362–367. 2023.
|
|
19
|
Andò S, Gelsomino L, Panza S, Giordano C,
Bonofiglio D, Barone I and Catalano S: Obesity, leptin and breast
cancer: Epidemiological evidence and proposed mechanisms. Cancers
(Basel). 11(62)2019.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Fontvieille E, Viallon V, Recalde M,
Cordova R, Jansana A, Peruchet-Noray L, Lennon H, Heath AK, Aune D,
Christakoudi S, et al: Body mass index and cancer risk among adults
with and without cardiometabolic diseases: Evidence from the EPIC
and UK Biobank prospective cohort studies. BMC Med.
21(418)2023.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Smith SG, Sestak I, Morris MA, Harvie M,
Howell A, Forbes J and Cuzick J: The impact of body mass index on
breast cancer incidence among women at increased risk: An
observational study from the International Breast Intervention
Studies. Breast Cancer Res Treat. 188:215–223. 2021.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Tran TX, Chang Y, Choi HR, Kwon R, Lim GY,
Kim EY, Ryu S and Park B: Adiposity, body composition measures, and
breast cancer risk in Korean premenopausal women. JAMA Netw Open.
7(e245423)2024.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Cao J, Li J, Zhang Z, Qin G, Pang Y, Wu M,
Gu K and Xu H: Interaction between body mass index and family
history of cancer on the risk of female breast cancer. Sci Rep.
14(4927)2024.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Habeeb MH: Expression of Cyclooxygenase-2
and interlukin-6 mrnas in Iraqi patients with breast cancer. Kufa
Med J. 19:116–121. 2023.
|
|
25
|
Denkert C, Winzer KJ, Müller BM, Weichert
W, Pest S, Köbel M, Kristiansen G, Reles A, Siegert A, Guski H and
Hauptmann S: Elevated expression of cyclooxygenase-2 is a negative
prognostic factor for disease free survival and overall survival in
patients with breast carcinoma. Cancer. 97:2978–2987.
2003.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Zeeneldin AA, Mohamed AM, Abdel HA, Taha
FM, Goda IA and Abodeef WT: Survival effects of cyclooxygenase-2
and 12-lipooxygenase in Egyptian women with operable breast cancer.
Indian J Cancer. 46:54–60. 2009.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Molehin D, Filleur S and Pruitt K:
Regulation of aromatase expression: Potential therapeutic insight
into breast cancer treatment. Mol Cell Endocrinol.
531(111321)2021.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Jana D, Sarkar DK, Ganguly S, Saha S, Sa
G, Manna AK, Banerjee A and Mandal S: Role of cyclooxygenase 2
(COX-2) in prognosis of breast cancer. Indian J Surg Oncol.
5:59–65. 2014.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Harris RE, Casto BC and Harris ZM:
Cyclooxygenase-2 and the inflammogenesis of breast cancer. World J
Clin Oncol. 5:677–692. 2014.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Bhutani N, Moga S, Poswal P, Sharma B,
Arora S and Singla S: COX-2 expression in carcinoma of the breast
and surrounding non-neoplastic breast tissue. Arch Br Cancer.
8:29–36. 2021.
|
|
31
|
Nunez F, Bravo S, Cruzat F, Montecino M
and De Ferrari GV: Wnt/β-catenin signaling enhances
cyclooxygenase-2 (COX2) transcriptional activity in gastric cancer
cells. PLoS One. 6(e18562)2011.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Mahmoud HQ, Hamzah MI and Ibrahim MJ:
Evaluation of serum and urine Dickkopf-1 concentrations and their
relation with lipid profile in Iraqi Women with breast carcinoma.
Malaysian J Sci. 43:16–26. 2025.
|
|
33
|
Mahmoud HQ, Hamzah MI and Ibrahim MJ:
Study of β-Catenin levels in sera and urine of Iraqi women with
breast carcinoma. Iraqi J Sci. 66:2719–2727. 2025.
|
|
34
|
Ilyan T, Retnoningrum D, Hendrianingtyas
M, Widyaningrum D and Rachmawati B: Association of
25-hydroxyvitamin D, cyclooxygenase-2 and prostaglandin E2 serum
levels in breast cancer patients. Indonesian Biomed J. 13:426–432.
2021.
|