Introduction
Cardiovascular diseases (CVD) are the leading cause
of mortality among patients undergoing hemodialysis (HD) (1), with the cardiovascular (CV) mortality
rate being 10-20-fold higher than that in the general population
following stratification for age, race and sex (2). In the general population, vascular
calcification is associated with increased CV mortality (3). Compared to the general population,
vascular calcification is prevalent in patients undergoing HD and
is associated with a higher risk of CV-related mortality (4-6).
Fetuin-A and matrix Gla protein (MGP) are two
proteins that provide a defense against extraosseous calcification.
Fetuin-A is produced in the liver and functions as a calcification
inhibitor by forming soluble colloidal spheres of fetuin-A, calcium
and phosphate, preventing the crystallization of hydroxyapatite
(7). Fetuin-A-deficient mice on a
mineral/vitamin D-rich diet develop calcification in small vessels
of the heart, lungs, kidneys and skin (8). Matrix Gla protein is secreted by
chondrocytes and vascular smooth muscle cells. It binds to newly
formed hydroxyapatite crystals, hindering their accumulation within
the arterial wall. Additionally, MGP inhibits the binding of bone
morphogenetic protein-2 to its receptor, preventing vascular smooth
muscle cells from differentiating into osteoblasts (9). MGP-deficient mice undergo spontaneous
and extensive calcification of cartilage and blood vessels, leading
to death within 2 months of birth due to blood vessel rupture
(10). For MGP to become active,
it must undergo the carboxylation of certain γ-glutamate residues,
followed by the phosphorylation of specific serine residues, in a
process that depends on vitamin K. Consequently, dephosphorylated
uncarboxylated MGP (dp-ucMGP) represents its inactive form and
elevated levels of dp-ucMGP indicate a reduced capacity to inhibit
vascular calcification (11,12).
In line with the increased incidence of vascular calcification in
patients undergoing HD, serum fetuin-A levels are lower in this
population (13,14), whereas dp-ucMGP levels are higher
(15,16). However, the data do not univocally
support a role for serum fetuin-A or dp-ucMGP levels as markers of
CV risk or established CVD.
In a community-based survey, low fetuin-A levels
were shown to be associated with aortic arch calcification
(17). Another study detected a
trend towards lower levels in patients with peripheral artery
disease (18). In another study,
among community-dwelling individuals, lower fetuin-A levels were
independently associated with greater severity of coronary artery
calcification, but not with peripheral artery disease or carotid
intima-media thickness (19).
Conversely, in the Multi-Ethnic Study of Atherosclerosis, fetuin-A
was not associated with the risk of CVD (20). In another cohort of patients who
underwent carotid or lower extremity endarterectomy, fetuin-A was
not associated with the coronary artery calcification score
(21). In a previous study, an
analysis of seven prospective studies found that fetuin-A genetic
variants associated with reduced fetuin-A levels were not related
to the risk of coronary heart disease (CHD) (22). Additionally, in another study
involving 1,049 patients with CHD, fetuin-A levels failed to
predict secondary CHD events over a 6-year follow-up period
(23). Notably, in another study,
fetuin-A was found to be higher in patients with CHD than in those
without (24).
A similar discrepancy is observed in studies
involving patients undergoing HD. Studies have shown that lower
fetuin-A levels are associated with a higher CV mortality (13,14,25-27),
an increased carotid intima-media thickness (13,26,27),
elevated carotid-to-femoral pulse wave velocity (28), higher coronary artery calcification
scores (25) and stroke (29). Conversely, in one study involving
104 patients undergoing HD, fetuin-A levels did not differ between
those with or without coronary artery or abdominal aortic
calcification (30). In another
cohort of 220 patients undergoing HD, serum fetuin-A was not
associated with carotid intima-media thickness, the degrees of
carotid atherosclerotic plaques or calcification, left ventricular
ejection fraction, prevalent CVD and all-cause or CV mortality
(31). Similarly, in another
cohort of 143 patients undergoing HD, no difference in fetuin-A
levels was observed between those with a carotid intima-media
thickness ≥0.8 mm, plaque or stenosis ≥50% and those without
(32).
Similarly, for fetuin-A, a discrepancy relative to
dp-ucMGP has been observed. In a population-based study with a
15.5-year follow-up period, higher dp-ucMGP levels were associated
with an increased incidence of CVD and all-cause mortality
(33). That study did not find a
significant link between dp-ucMGP levels and the risk of myocardial
infarction or sudden cardiac death. Nonetheless, elevated levels of
dp-ucMGP were associated with an increased risk of other CVDs
(33). One possible explanation
for this is that myocardial infarction often results from the
rupture of non-calcified atherosclerotic plaques (34). In another cohort of patients who
experienced myocardial infarction, coronary revascularization, or
first ischemic stroke, those in the highest quartile of dp-ucMGP
had a higher risk of all-cause and CV 5-year mortality (35). In the Multi-Ethnic Study of
Atherosclerosis, a positive association was detected between
dp-ucMGP and the incidence and progression of coronary arteries and
thoracic aorta calcification (36). Interestingly, in patients with
acute coronary syndrome, those with non-ST-elevation myocardial
infarction (NSTEMI) had higher levels of dp-ucMGP than those with
ST-elevation myocardial infarction (STEMI), suggesting a potential
link between dp-ucMGP and coronary artery calcification burden and
more stable atherosclerotic plaques (37). Conversely, in the Health, Aging,
and Body Composition Study, no association was detected between
dp-ucMGP and incident CVD (38).
Mendelian randomization studies have demonstrated that lower
genetically predicted dp-ucMGP levels are associated with a reduced
risk of CHD (39,40). Notably, in the Multi-Ethnic Study
of Atherosclerosis, elevated dp-ucMGP was associated with an
increased risk of incident CVD, CHD and all-cause mortality, but
only in the youngest age quartile (41).
A similar discrepancy has been observed in studies
involving patients undergoing HD. In these patients, high dp-uc MGP
levels have been linked to increased vascular calcification
(15,16,42).
In a previous study, in a cohort of 141 patients with end-stage
kidney disease (ESKD) undergoing living donor kidney
transplantation, elevated dp-ucMGP levels were associated with the
coronary artery calcification score and medial vascular
calcification in the epigastric artery (43). However, in another cohort of 493
patients with ESKD from the same center with a median follow-up
period of 42 months, elevated dp-ucMGP levels were associated with
increased all-cause mortality, but they were not identified as an
independent risk factor of coronary artery or aortic valve
calcification (44). Conversely,
another study demonstrated that, among 391 patients on incident HD,
neither dp-ucMGP nor fetuin-A were associated with coronary artery
calcification, pulse wave velocity or mortality risk (45). Similarly, among participants in the
Chronic Renal Insufficiency Cohort with mild-to-moderate chronic
kidney disease, dp-ucMGP was not associated with either coronary
artery calcification score or pulse wave velocity (46). Moreover, in the same cohort,
elevated dp-ucMGP levels were associated with increased all-cause
mortality, but not with more atherosclerotic CV events (47). Of note, in a study on 198 patients
undergoing HD, low dp-ucMGP levels were associated with a higher
risk of all-cause mortality and CV mortality (48).
In this complex landscape, the present study
investigated the association between fetuin-A or dp-ucMGP and
established CHD in patients undergoing HD. Additionally, the
present study explored other confounding factors that could impact
these associations, underscoring the need for a better
understanding of these interactions to improve patient
outcomes.
Patients and methods
Patients
A total of 126 patients undergoing HD participated
in the study. The mean age of the patients was 65.94±11.85 years,
comprising 89 males and 37 females. The cause of ESKD was diabetic
nephropathy (n=36), primary glomerulonephritis (n=24), hypertension
(n=21), autosomal dominant polycystic kidney disease (n=11),
secondary focal segmental glomerulosclerosis (n=6), cardiorenal
syndrome (n=4), vasculitis (n=4), obstructive nephropathy (n=2),
analgesic nephropathy (n=1) and unknown causes (n=17).
In total, 50 patients had diabetes mellitus, and 40
had a history of CHD. CHD was ascertained by coronary artery
angiography performed for angina symptoms or following a myocardial
infarction. A total of 110 patients underwent recent transthoracic
echocardiography. Among these, 38 patients were identified with
heart failure with preserved ejection fraction (HFpEF), while 26
patients were diagnosed with heart failure with reduced ejection
fraction (HFrEF). Statin therapy was administered to 82 patients,
and the majority received antihypertensive medications. The
attending nephrologists determined the need for phosphate binders,
vitamin D analogues and calcimimetics. All patients had received HD
for at least 6 months prior to inclusion. Each patient underwent
4-h HD sessions three times per week, using polysulfone dialyzers
and bicarbonate-based dialysate with calcium concentrations of 1.25
or 1.5 mmol/l.
Patients with active infection, autoimmune disease,
malignancy, liver pathology, or who have received cytotoxic,
immunosuppressive, or corticosteroid therapy within the previous 6
months were excluded from the study. Patients receiving vitamin K
antagonists were also excluded. Further clinical and demographic
features are presented in Table
I.
 | Table IPatient characteristics. |
Table I
Patient characteristics.
| | No. of
patients | Mean | SD |
|---|
| Age (years) | 126 | 65.94 | 11.85 |
| Duration
(months) | 126 | 56.21 | 50.04 |
| Males/females | 89/37 | | |
| Diabetes mellitus
(yes/no) | 50/76 | | |
| Coronary heart
disease (yes/no) | 40/86 | | |
| HFrEF/HFpEF/No
HF | 26/38/46 | | |
| Systolic blood
pressure (mmHg) | 126 | 132.12 | 21.41 |
| Diastolic blood
pressure (mmHg) | 126 | 65.57 | 13.32 |
| White blood cell
count (c/µl) | 126 | 6,884.81 | 2,286.36 |
| Neutrophils
(c/µl) | 126 | 4,589.67 | 1,726.59 |
| Lymphocytes
(c/µl) | 126 | 1,681.77 | 606.01 |
| Hemoglobin
(g/dl) | 126 | 11.77 | 0.84 |
| Platelets
(c/µl) | 126 | 203.99 | 58.97 |
| Creatinine
(mg/dl) | 126 | 6.38 | 2.04 |
| Urea (mg/dl) | 126 | 130.35 | 27.48 |
| Urea reduction rate
(%) | 126 | 67.53 | 7.60 |
| Residual diuresis
(ml) | 126 | 321.43 | 422.30 |
| Body mass index
(Kg/m2) | 126 | 26.90 | 5.71 |
| Albumin (g/dl) | 126 | 3.63 | 0.10 |
| Cholesterol
(mg/dl) | 126 | 130.93 | 42.42 |
| Triglycerides
(mg/dl) | 126 | 130.33 | 68.77 |
| Calcium
(mg/dl) | 126 | 9.12 | 0.46 |
| Phosphorus
(mg/dl) | 126 | 5.34 | 1.01 |
| Parathyroid hormone
(pg/ml) | 126 | 347.73 | 287.29 |
| Alkaline
phosphatase (U/l) | 126 | 205.80 | 105.30 |
| SGOT (U/l) | 126 | 12.88 | 7.31 |
| SGPT (U/l) | 126 | 11.72 | 9.16 |
| Ferritin
(ng/ml) | 126 | 153.47 | 161.76 |
| TSAT (%) | 126 | 19.01 | 12.12 |
| CRP (mg/dl) | 126 | 1.24 | 0.87 |
| CRP >1 mg/dl
(yes/no) | 63/63 | | |
| Fetuin-A
(mg/ml) | 126 | 1.73 | 1.22 |
| Dp-uc-matrix Gla
protein (pg/ml) | 126 | 4,743.50 | 2,556.75 |
A control group consisting of 24 healthy individuals
(mean age, 64.42±6.49 years; 15 males and 9 females) was included
following a review of medical records and a physical
examination.
Written informed consent was obtained from each
participant, and the study protocol received approval from the
Ethics Committee of the Faculty of Medicine, University of
Thessaly, Larissa, Greece (approval no. 558/10-2-2017).
Patient samples and analyses
Blood samples were drawn at the onset of the second
hemodialysis session of the week, and serum was preserved at
-80˚C.
Serum fetuin-A concentrations were measured using
the Human Fetuin-A ELISA kit (cat. no. CSB-E12882h, Cusabio
Technology, Wuhan, China), which has a sensitivity of 3.9 ng/ml.
Serum dp-ucMGP levels were assessed using the human dp-ucMGP ELISA
kit (cat. no. EH4755, Wuhan Fine Biotech Co., Ltd.), which has a
sensitivity of 46.875 pg/ml. All other parameters were part of
routine laboratory assessments performed concurrently with serum
collection for the measurements of the factors described above.
Statistical analysis
Statistical analysis was performed using the IBM
SPSS Statistics version 29 (IBM Corp.). Since the one-sample
Kolmogorov-Smirnov test revealed that serum fetuin-A and dp-ucMGP
levels were not normally distributed, non-parametric methods were
applied. Specifically, for group comparisons, the Mann-Whitney U
test was performed, with data presented as the median
(interquartile range). Furthermore, Spearman's rank correlation
coefficient was performed to evaluate correlations between
continuous variables, while the Chi-squared test was used to
examined associations between categorical variables. In addition,
receiver operating characteristic (ROC) curve analysis was
performed, and the optimal cut-off was identified by maximizing
Youden's index. Finally, binary logistic regression was used to
assess the independent effects of variables on outcomes. A value of
P<0.05 was considered to indicate a statistically significant
difference.
Results
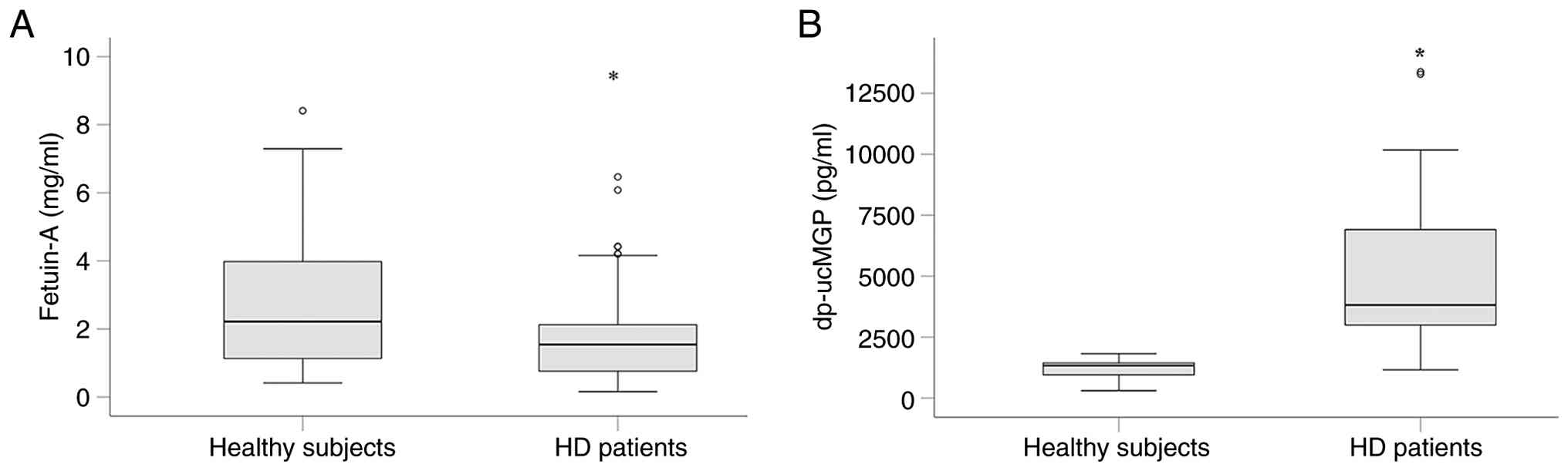
Serum fetuin-A and dp-ucMGP levels in patients
undergoing HD and healthy volunteers. Serum fetuin-A levels
were significantly lower in patients undergoing HD than in healthy
volunteers [1.54 (0.76-2.13) vs. 2.22 (1.423-3.980) mg/ml,
respectively; P=0.02, Mann-Whitney U test] (Fig. 1A).
On the contrary, compared to the healthy subjects,
the serum dp-ucMGP level was significantly higher in patients
undergoing HD [1334.35 (920.8-1449.6) vs. 3892.38 (2958.9-6983.4)
pg/ml, respectively, P<0.001, Mann-Whitney U test] (Fig. 1B)
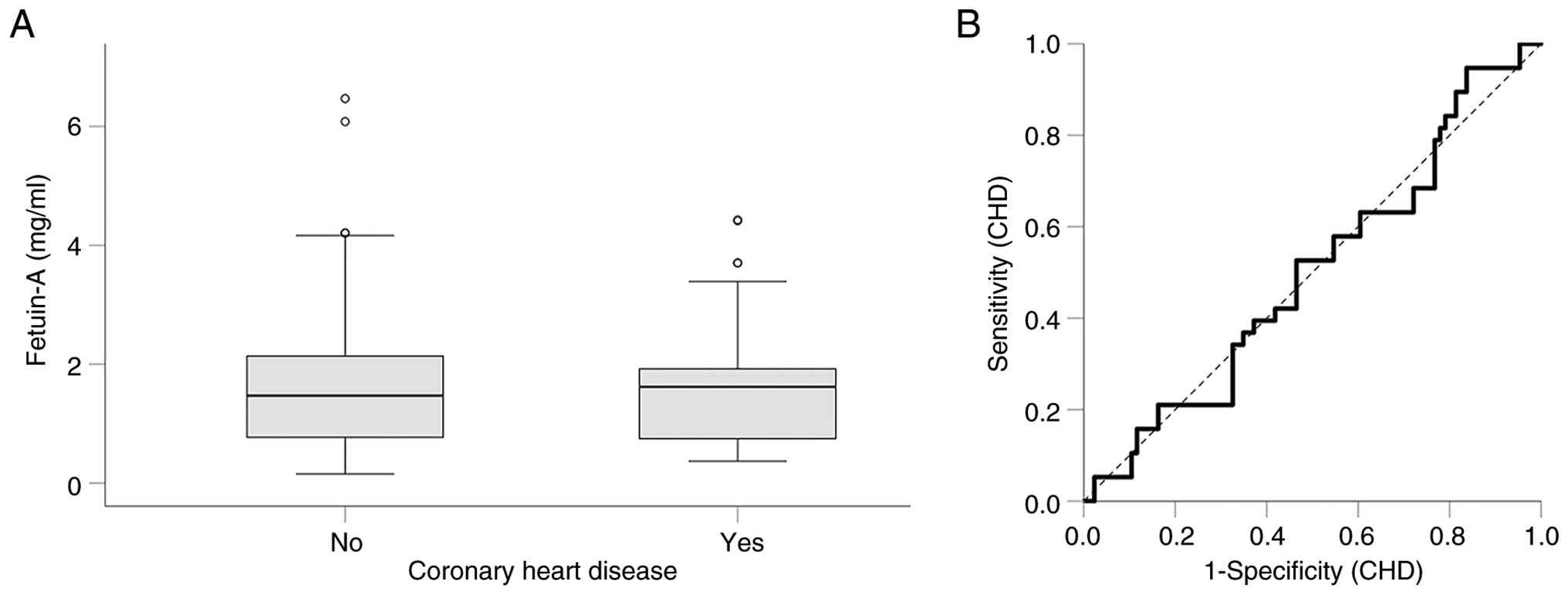
Serum fetuin-A levels in patients
undergoing HD
Serum fetuin-A levels did not differ between
patients undergoing HD with or without established CHD [1.62
(0.74-1.93) vs. 1.47 (0.77-2.15) mg/ml, respectively, P=0.987,
Mann-Whitney U test] (Fig. 2A).
Thus, ROC curve analysis revealed that in patients undergoing HD,
fetuin-A is not a reliable marker of CHD [area under the curve
(AUC), 0.499; 95% confidence interval (CI), 0.390-0.608; P=0.987]
(Fig. 2B).
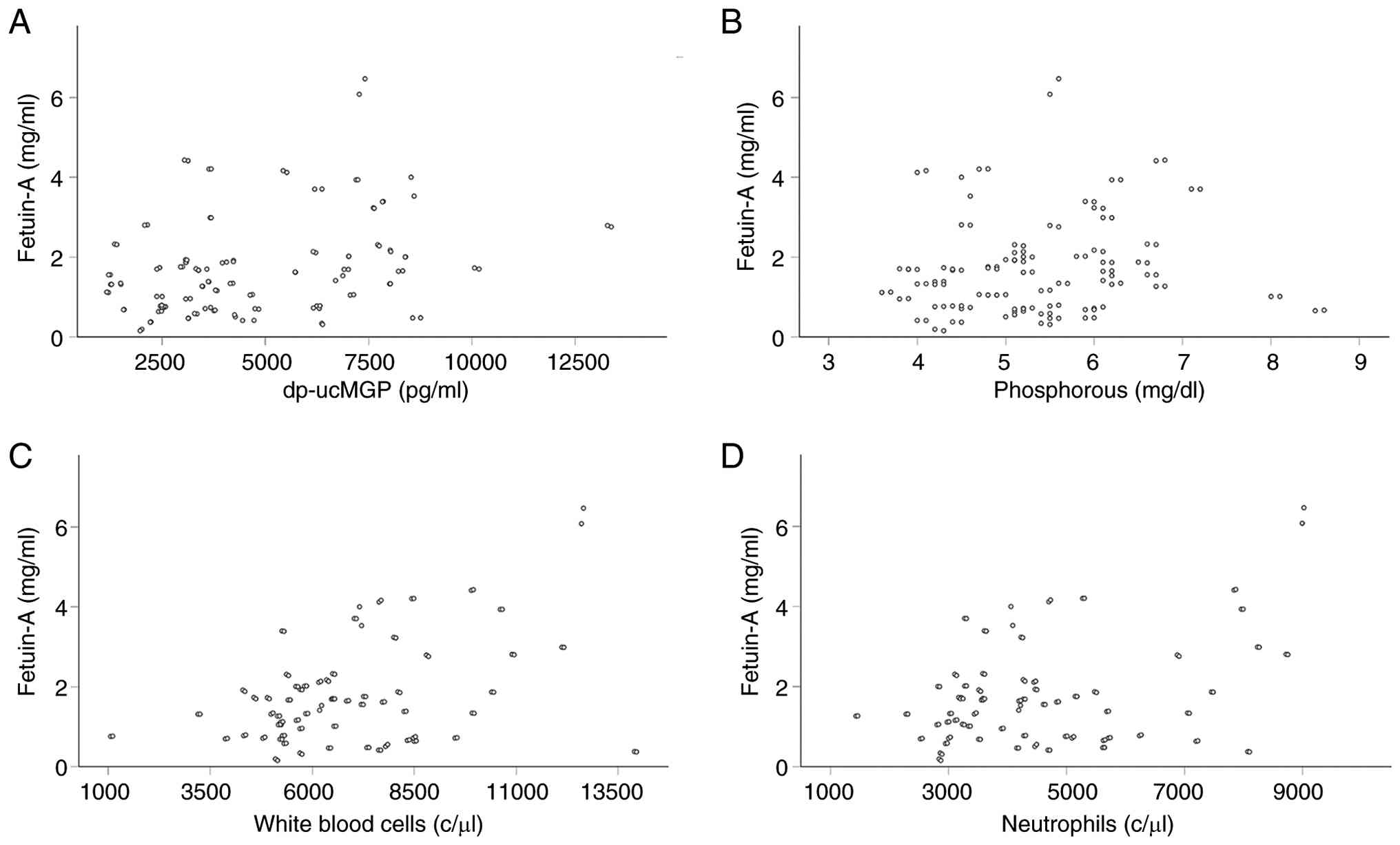
Notably, among the factors depicted in Table I, serum fetuin-A levels exhibited
positive correlations with serum dp-ucMGP levels (Rho=0.311,
P<0.001) (Fig. 3A), and
phosphorus (Rho=0.184, P=0.042) (Fig.
3B). No correlations were detected between fetuin-A and the
nutritional markers, body mass index (BMI; Rho=0.072, P=0.426) and
serum albumin (Rho=-0.001, P=0.992) (data not shown). Fetuin-A was
also not correlated with the urea reduction ratio (Rho=0.082,
P=0.364) (data not shown). Although a positive correlation was
detected with the white blood cell count (Rho=0.280, P=0.002)
(Fig. 3C) and neutrophils
(Rho=0.226, P=0.012) (Fig. 3D), no
correlation was detected with serum C-reactive protein (CRP;
Rho=0.037, P=0.680) (data not shown). In addition, using a serum
CRP cut-off of 1 mg/dl to detect inflammation did not reveal that
inflammation affects serum fetuin-A levels. Its levels were 1.62
(0.78-2.02) mg/ml in those with a CRP level <1 mg/ml, and 1.39
(0.75-2.28) mg/ml in those with a CRP level >1mg/dl (P=0.658,
Mann-Whitney U test) (Table
II).
 | Table IIDifferences in serum fetuin-A (mg/ml)
between different groups. |
Table II
Differences in serum fetuin-A (mg/ml)
between different groups.
| | No. of
patients | Yes | No | P-value |
|---|
| Male sex | 90 | 1.54
(0.73-2.21) | 1.52
(1.05-2.12) | 0.783 |
| Diabetes
mellitus | 50 | 1.55
(0.75-2.42) | 1.47
(0.76-2.04) | 0.625 |
| Coronary heart
disease | 40 | 1.62
(0.74-1.93) | 1.47
(0.77-2.15) | 0.987 |
| HFpEF | 38 | 1.55
(1.06-2.15) | 1.68
(0.73-2.80) | 0.921 |
| HFrEF | 26 | 1.30
(0.72-1.85) | 1.67
(0.96-2.15) | 0.325 |
| CRP >1
mg/dl | 63 | 1.39
(0.75-2.28) | 1.62
(0.78-2.02) | 0.658 |
Sex did not affect fetuin-A levels, which were 1.54
(0.73-2.21) mg/ml in males and 1.52 (1.05-2.12) mg/ml in females
(P=0.783, Mann-Whitney U test). Also, fetuin-A levels did not
differ in patients with or without diabetes mellitus [1.55
(0.75-2.42) vs. 1.47 (0.76-2.04) mg/ml, respectively, P=0.625,
Mann-Whitney U test], HFpEF [1.55 (1.06-2.15) vs. 1.68 (0.73-2.80)
mg/ml, respectively, P=0.921, Mann-Whitney U test], or HFrEF [1.30
(0.72-1.85) vs. 1.67 (0.96-2.15) mg/ml, respectively, P=0.325,
Mann-Whitney U test] (Table
II).
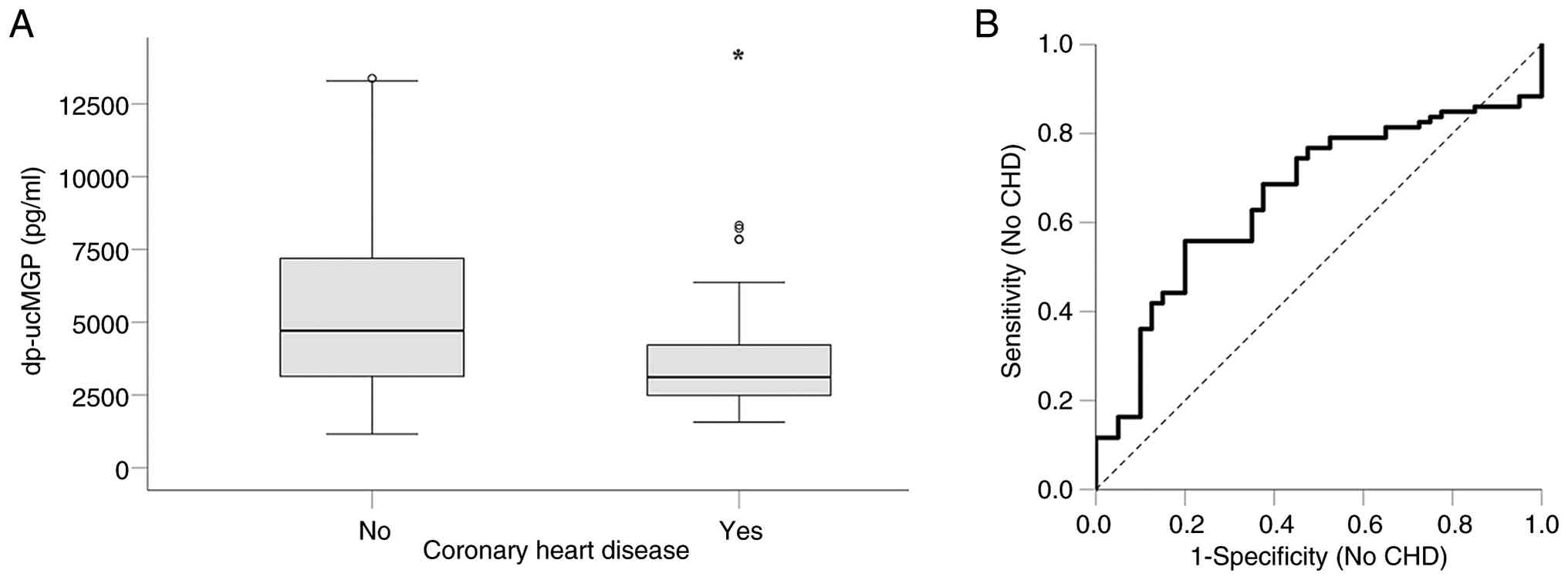
Serum dp-ucMGP levels in patients
undergoing HD
Serum dp-ucMGP levels were significantly lower in
patients undergoing HD with established CHD. They were 3,112.62
(2,487.32-4,221.92) pg/ml in those with CHD and 4,704.96
(3,135.95-7,207.95) pg/ml in those without CHD (P=0.004,
Mann-Whitney U test) (Fig. 4A).
ROC curve analysis revealed an AUC of 0.659 (95% CI, 0.560-0.757;
P=0.004) (Fig. 4B). Below the
optimal cut-off point of 4,241 pg/ml, serum dp-ucMGP exhibited a
specificity for detecting CHD of 80.00% (95% CI, 65.24-89.50%) and
a sensitivity of 55.81% (95% CI, 45.29-65.84%). Thus, serum
dp-ucMGP may be characterized as a moderately significant marker of
CHD in patients undergoing HD.
From the factors depicted in Table I, serum dp-ucMGP levels were
correlated negatively with age (Rho=-0.302, P<0.001) (Fig. 5A), and positively with BMI
(Rho=0.230, P=0.010) (Fig. 5B),
creatinine (Rho-0.187, P=0.037) (Fig.
5C), calcium (Rho=0.398, P<0.001) (Fig. 5D), intact parathyroid hormone
(iPTH; Rho=0.192, P=0.031) (Fig.
5E) and fetuin-A (Rho=0.311, P<0.001) (Fig. 5F). Notably, dp-ucMGP was not
correlated with serum albumin (Rho=0.139, P=0.121) or the urea
reduction ratio (Rho=-0.074, P=0.409) (data not shown). Although no
correlation was detected with CRP (Rho=-0.153, P=0.087), when a CRP
cut-off point of 1 mg/dl was set for defining inflammation, those
with inflammation had lower dp-ucMGP levels [3,631.09
(2,492.57-6,218.71) pg/ml] than those without inflammation
[4,684.97 (3,181.79-7,197.20) pg/ml] (P=0.032, Mann-Whitney U test)
(Table III).
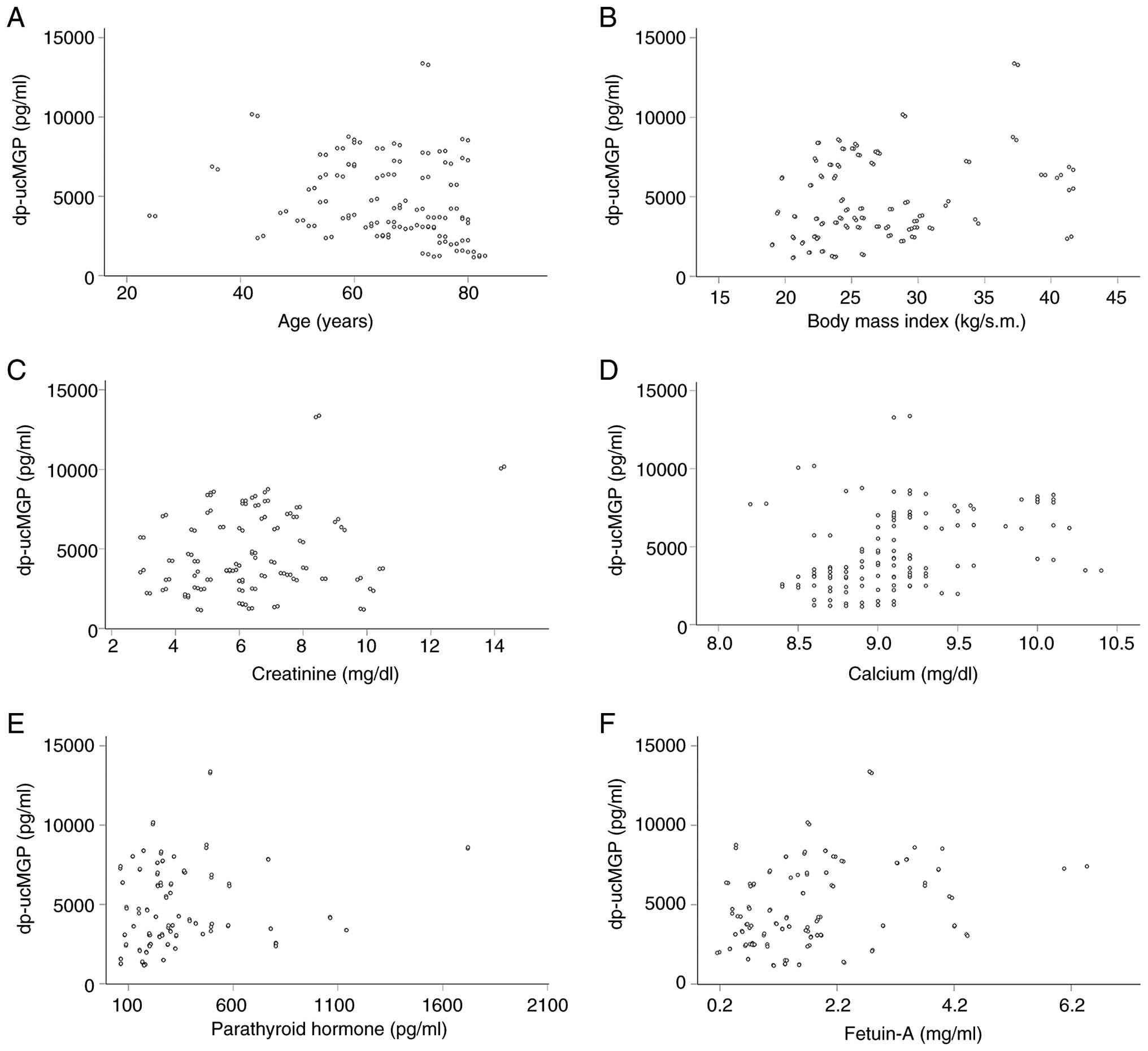 | Figure 5Scatterplots of statistically
significant correlations between dp-ucMGP and the evaluated
factors. Serum dp-ucMGP levels significantly correlated with (A)
age (Rho=-0.302, P<0.001), (B) body mass index (Rho=0.230,
P=0.010), (C) creatinine (Rho=0.187, P=0.037), (D) calcium
(Rho=0.398, P<0.001), (E) parathyroid hormone (Rho=0.192,
P=0.031), and (F) fetuin-A (Rho=0.311, P<0.001). dp-ucMGP,
uncarboxylated matrix Gla protein. |
 | Table IIIDifferences in serum dp-ucMGP (pg/ml)
between different groups. |
Table III
Differences in serum dp-ucMGP (pg/ml)
between different groups.
| | No. of
patients | Yes | No | P-value |
|---|
| Male sex | 90 | 3,766.63
(3,056.58-6,910.13) | 4,451.02
(2,094.04-6,983.42) | 0.750 |
| Diabetes
mellitus | 50 | 3,512.70
(2,490.82-4,943.77) | 4,736.58
(3,088.39-7,377.55) | 0.006 |
| Coronary heart
disease | 40 | 3,112.62
(2,487.32-4,221.92) | 4,704.96
(3,135.95-7,207.95) | 0.004 |
| HFpEF | 38 | 4,451.02
(3,054.16-7,207.95) | 3,810.37
(3,328.87-7,840.34) | 0.571 |
| HFrEF | 26 | 3,094.49
(2,474.14-4,357.54) | 4,008.10
(3,076.50-7,707.95) | 0.028 |
| CRP >1
mg/dl | 63 | 3,631.09
(2,492.57-6,218.71) | 4,684.97
(3,181.79-7,197.20) | 0.032 |
Sex did not affect serum dp-ucMGP levels. They were
3,766.63 (3,056.58-6,910.13) pg/ml in males and 4,451.02
(2,094.04-6,983.42) pg/ml in females (P=0.750, Mann-Whitney U
test). However, dp-ucMGP levels were lower in patients with
diabetes mellitus. They were 3,512.70 (2,490.82-4,943.77) pg/ml in
those with diabetes mellitus and 4,736.58 (3,088.39-7,377.55) pg/ml
in those without diabetes mellitus (P=0.006, Mann-Whitney U test).
The levels of dp-ucMGP did not differ in those with HFpEF [4,451.02
(3,054.16-7,207.95) pg/ml] or without HFpEF [3,810.37
(3,328.87-7,840.34) pg/ml] (P=0.571, Mann-Whitney U test). However,
they were significantly lower in patients with HFrEF [3,094.49
(2,474.14-4,357.54) pg/ml] than in those without HFrEF [4,008.10
(3,076.50-7,707.95) pg/ml)] (P=0.028, Mann-Whitney U test)
(Table III). Notably, CHD was
associated with HFrEF (Chi-squared test, 35.267, P<0.001)
(Table IV), indicating that, in
many cases of HFrEF, CHD may be responsible.
 | Table IVAssociations of CHD with sex,
diabetes, HFrEF and CRP. |
Table IV
Associations of CHD with sex,
diabetes, HFrEF and CRP.
| Variable | Category | CHD (no. of
patients | No CHD (no. of
patients) | χ2 | P-value |
|---|
| Sex | Male | 38 | 52 | 15.955 | <0.001 |
| | Female | 2 | 34 | | |
| Diabetes
mellitus | Yes | 24 | 26 | 10.107 | 0.001 |
| | No | 16 | 60 | | |
| HFrEF | Yes | 22 | 4 | 35.267 | <0.001 |
| | No | 18 | 68 | | |
| CRP >1
mg/dl | Yes | 21 | 42 | 0.147 | 0.702 |
| | No | 19 | 44 | | |
Of note, the male sex was associated with CHD
(Chi-squared test, 15.955, P<0.001), as was diabetes mellitus
(Chi-squared test, 10.107, P=0.001) (Table IV). As regards the other variables
that were associated with serum dp-ucMGP levels, age (P=0.182,
Mann-Whitney U test), BMI (P=0.317, Mann-Whitney U test), calcium
(P=0.825, Mann-Whitney U test), iPTH (P=0.992, Mann-Whitney U
test), creatinine (P=0.067, Mann-Whitney U test) and CRP (P=0.902,
Mann-Whitney U test) did not differ between those with CHD and
those without CHD (Table V). When
the CRP cut-off of 1 mg/dl was set for defining inflammation, no
association was detected between inflammation and CHD (Chi-squared
test, 0.147, P=0.702) (Table
IV).
 | Table VDifferences in factors correlated
with dp-ucMGP in patients with or without CHD. |
Table V
Differences in factors correlated
with dp-ucMGP in patients with or without CHD.
| | CHD | No CHD | P-value |
|---|
| Age (years) | 71.5 (63-77) | 67.0 (59-74) | 0.182 |
| Body mass index
(kg/m2) | 25.30
(22.25-27.80) | 25.54
(23.43-29.77) | 0.317 |
| Creatinine
(mg/dl) | 6.0
(3.77-7.03) | 6.4 (5.1-7.7) | 0.067 |
| Calcium
(mg/dl) | 9.05
(8.7-10.0) | 9.10 (8.8-9.3) | 0.825 |
| Parathyroid hormone
(pg/ml) | 258.7
(199.9-391.9) | 267.3
(175.0-455.0) | 0.992 |
| Fetuin-A
(mg/ml) | 1.62
(0.745-1.93) | 1.47
(0.773-2.145) | 0.987 |
| CRP (mg/dl) | 1.02
(0.79-1.61) | 0.945
(0.78-1.60) | 0.902 |
Among the variables that may causally influence
dp-ucMGP levels, only diabetes mellitus exhibited a causal
association with CHD. Binary logistic regression analysis with CHD
as the outcome variable and 1,000 pg (1 ng) increments of dp-ucMGP
as the predictor variable revealed that for every dp-ucMGP increase
of 1,000 pg/ml, the risk for CHD decreased by 23.4% [odds ratio
(OR), 0.776; 95% CI, 0.649-0.928; P=0.005] (Table VI). Binary logistic regression
analysis with CHD as the outcome variable and 1,000 pg increments
of dp-ucMGP and diabetes mellitus as predictor variables was
performed using the enter method. The overall model was
statistically significant (Chi-squared test, 15.850; P<0.001),
indicating that the predictors reliably distinguished between
individuals with and without CHD. The model explained 16.6% of the
variance (Nagelkerke R2=0.166) and correctly classified
73.8% of cases. Diabetes mellitus almost triplicates the
possibility for CHD (OR, 2.909; 95% CI, 1.295-6.534; P=0.01), and
for every dp-ucMGP increase of 1,000 pg/ml, the risk for CHD
decreased by 18.8% (OR, 0.812; 95% CI, 0.677-0.973; P=0.024). Thus,
dp-ucMGP levels predicted CHD independently of diabetes mellitus
(Table VI).
 | Table VILogistic regression analysis for CHD
with dp-ucMGP alone (model 1) or in combination with diabetes (the
only variable affecting both) (model 2). |
Table VI
Logistic regression analysis for CHD
with dp-ucMGP alone (model 1) or in combination with diabetes (the
only variable affecting both) (model 2).
| | 95% C.I. for
OR |
|---|
| | B | S.E. | Sig. | OR | Lower | Upper |
|---|
| Model 1 | | | | | | |
|
dp-ucMGP
(ng/ml) | -0.254 | 0.091 | 0.005 | 0.776 | 0.649 | 0.928 |
| Model 2 | | | | | | |
|
Diabetes
mellitus | 1.068 | 0.413 | 0.010 | 2.909 | 1.295 | 6.534 |
|
dp-ucMGP
(ng/ml) | -0.208 | 0.092 | 0.024 | 0.812 | 0.677 | 0.973 |
Discussion
Since CVD is the primary cause of mortality in
patients undergoing HD, evaluating CVD pathogenesis and identifying
potential markers for its early detection is critical. The present
study investigated whether fetuin-A, a vascular calcification
inhibitor, and dp-ucMGP, an inactive vascular calcification
inhibitor, are associated with established CHD in a cohort of
stable patients undergoing HD.
As previous studies have shown (13,14),
serum fetuin-A levels were lower in HD patients than in healthy
volunteers. However, its levels did not differ in patients
undergoing HD with or without CHD. The data on the role of fetuin-A
as a risk factor or a marker of established CVD are mixed. Some
studies in the general population have shown that low fetuin-A
levels are associated with an increased risk of CV or established
CVD (17-19),
other studies did not find an association (20-23),
and at least one study demonstrated that fetuin-A was higher in
patients with CHD than in those without (24). Studies involving patients
undergoing HD have also yielded mixed outcomes. Some studies
support the role of fetuin-A as a risk factor or marker of CVD
(13,14,25-29),
while others do not (30-32).
According to the present study, serum fetuin-A cannot serve as a
marker of established CHD in patients undergoing HD. This result
does not entirely contradict studies suggesting that fetuin-A
levels can indicate the risk of CVD. Numerous of those studies
focused on subclinical atherosclerosis or vascular calcification
(13,25-28,30,32),
or they assessed incident CVD rather than established CVD in
general or CHD specifically (13,14,25-27).
In the present study, serum fetuin-A levels
correlated with dp-ucMGP, phosphorus, white blood cell count and
neutrophil levels, but not with CRP. Additionally, factors such as
sex, diabetes mellitus, heart failure and inflammation, as defined
by a CRP cut-off, did not affect fetuin-A levels. Of note, fetuin-A
is considered a negative inflammatory reactant (13,14,26-29).
Notably, not all studies involving patients undergoing HD have
shown a negative association with CRP (31,49).
As regards diabetes mellitus, while some studies have reported an
association with decreased fetuin-A levels (29,31),
others have not (26). Considering
the lack of an association between fetuin-A levels and heart
failure in the present study, in the patients included herein,
heart failure was strongly associated with CHD, implying that, in
the majority of cases, CHD may be the cause of heart failure.
As in previous studies (15,16),
serum dp-ucMGP levels were higher in patients undergoing HD than in
healthy volunteers. In the present study, in patients undergoing
HD, dp-ucMGP levels were significantly lower in those with
established CHD than in those without CHD. ROC curve analysis
revealed an AUC of 0.659 for identifying CHD-free patients,
indicating serum dp-ucMGP as a moderately significant marker of CHD
in patients undergoing HD.
The data on the role of high dp-ucMGP as a risk
factor or a marker of established CVD are mixed. In the general
population, some studies have identified high dp-ucMGP levels as a
risk factor for CVD or a marker of established CVD (33-37,41).
By contrast, others have not found such an association (38), whereas, particularly for CHD,
others have supported the opposite (39,40).
However, even in studies that have supported an association between
high dp-ucMGP levels and CVD, several interesting points emerge. In
one of these, no link was detected between dp-ucMGP levels and the
risk of incident myocardial infarction or sudden cardiac death
(33). In another study, patients
with NSTEMI had higher dp-ucMGP levels than those with STEMI
(37). The results of the last two
studies suggest that high dp-ucMGP levels are associated with
greater coronary artery calcification and, consequently, a lower
risk of atherosclerotic plaque rupture (34). Lastly, one study demonstrated that
elevated dp-ucMGP levels were associated with an increased risk of
incident CVD, CHD and all-cause mortality, but only in the youngest
age quartile (45-to-53-year-olds) (41), i.e., in a younger age group than
the patients undergoing HD in the present study.
In studies involving patients undergoing HD, the
findings on the role of high dp-ucMGP as a CVD risk factor or
marker of established CVD are also inconsistent. Some studies have
shown such an association (15,16,42-44),
while others have failed to detect it (45,46);
notably, in one study, low dp-ucMGP levels were linked to increased
all-cause [hazard ratio (HR), 1.71; 95% CI, 0.92-3.17] and CV
mortality (HR, 1.83; 95% CI, 0.9-3.7) (48). Of note, in one study involving
patients with mild to moderate chronic kidney disease, elevated
dp-ucMGP levels were associated with increased all-cause mortality,
but not with more atherosclerotic CV events (47). Finally, in support of the results
of the present study, studies have failed to confirm CV benefits
despite the potential of vitamin K supplementation to reduce
dp-ucMGP levels in patients with ESKD (49).
In the present study, serum dp-ucMGP levels were
negatively correlated with age and positively correlated with BMI,
creatinine, calcium, iPTH and fetuin-A. Although no correlation was
detected with CRP, when a CRP cut-off of 1 mg/dl was used to define
inflammation, those with inflammation had lower dp-ucMGP levels
than those without. Studies involving patients undergoing HD
conclude with inconsistent results regarding the association
between dp-ucMGP levels and CRP. Some have detected a positive
association (42,44,45),
while others have found no association (50). Sex did not affect serum dp-ucMGP
levels. However, dp-ucMGP levels were lower in patients with
diabetes mellitus. Studies on patients undergoing HD conclude with
varying results regarding the association between dp-ucMGP levels
and diabetes mellitus. Some have detected a positive association
(16,51), while others have found no
association (43,44), or a negative association (38,46).
Finally, dp-uc MGP levels were lower in patients with HFrEF.
However, the strong association between HFrEF and CHD suggests that
CHD may have caused HFrEF in the patients in the present study.
Among the variables that affected dp-ucMGP levels,
only diabetes mellitus was associated with CHD. For every dp-ucMGP
increase of 1 ng/ml, the risk of CHD decreased by 23.4%. Still,
following adjustment for diabetes mellitus, for every dp-ucMGP
increase of 1 ng/ml, the risk for CHD decreased by 18.8%.
The present study has certain limitations. First,
further prospective studies involving a larger patient population
are necessary to verify the findings. The modest discriminatory
capacity of dp-ucMGP for detecting CHD (AUC, 0.659) further
underscores the need for additional research. In addition, the
present study relied on the medical records of patients to
determine the presence of CHD. As a consequence, some cases of
asymptomatic CHD may have been overlooked. Nevertheless, regular
thrice-weekly attendance at the HD unit enables early detection of
CHD, and patients undergo periodic cardiological assessments and
transthoracic echocardiography. Moreover, the results do not
entirely contradict studies suggesting that fetuin-A or dp-ucMGP
levels can indicate the risk of CVD. A number of those studies
focused on subclinical atherosclerosis or vascular calcification
(15,16,42-45),
or they assessed incident CVD rather than established CHD (43-45,47,48).
Finally, we assessed fetuin-A and dp-ucMGP at a single time point
to determine whether they are associated with established CHD.
However, interpreting the results is challenging as the patients
who survive to that time point are not a random sample. Patients
with more calcified arteries may have an improved prognosis, since
more calcified atherosclerotic plaques are less prone to rupture
(34). Thus, patients with CHD
with higher dp-ucMGP levels or lower fetuin-A levels, and possibly
more calcified coronary arteries, are more likely to be alive and
were included in the assessment. This survival imbalance may
influence the observed association between fetuin-A or dp-ucMGP and
CHD. In other words, dp-ucMGP may be a negative marker of prevalent
CHD and, concurrently, a risk factor for incident CHD. Following-up
on the patient cohort for incident CHD will help clarify this
concept.
In conclusion, the present study demonstrates that,
in patients undergoing HD, fetuin-A levels are not associated with
established CHD. Conversely, elevated dp-ucMGP levels indicate
CHD-free patients, though its accuracy is moderate.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
TE designed the study. MD and PM obtained patient
samples and clinical data. AK performed the ELISA measurements. TE,
MD, AK, MT, PM, CP, EL, AB and IS analyzed and interpreted the
results. T.E. wrote the draft manuscript; T.E., M.D., and M.T.
edited the manuscript. TE, AK and MD confirm the authenticity of
all the raw data. All authors drafted the manuscript, critically
revised the manuscript, and agreed to be fully accountable for
ensuring the integrity and accuracy of the work, and have read and
approved the final manuscript.
Ethics approval and consent to
participate
The present study was conducted in accordance with
the Declaration of Helsinki and was approved by the Ethics
Committee of the University of Thessaly, Faculty of Medicine
(Approval no. 558/10-2-2017). Informed consent was obtained from
all subjects involved in the study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Jankowski J, Floege J, Fliser D, Böhm M
and Marx N: Cardiovascular disease in chronic kidney disease.
Circulation. 143:1157–1172. 2021.
|
|
2
|
Foley RN, Parfrey PS and Sarnak MJ:
Clinical epidemiology of cardiovascular disease in chronic renal
disease. Am J Kidney Dis. 32:S112–S119. 1998.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Allison MA, Hsi S, Wassel CL, Morgan C, Ix
JH, Wright CM and Criqui MH: Calcified atherosclerosis in different
vascular beds and the risk of mortality. Arteriosclerosis Thromb
Vasc Biol. 32:140–146. 2012.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Goodman WG, Goldin J, Kuizon BD, Yoon C,
Gales B, Sider D, Wang Y, Chung J, Emerick A and Greaser L:
Coronary-artery calcification in young adults with end-stage renal
disease who are undergoing dialysis. New Engl J Med. 342:1478–1483.
2000.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Liabeuf S, Desjardins L, Diouf M, Temmar
M, Renard C, Choukroun G and Massy ZA: The addition of vascular
calcification scores to traditional risk factors improves
cardiovascular risk assessment in patients with chronic kidney
disease. PLoS One. 10(e0131707)2015.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Hutcheson JD and Goettsch C:
Cardiovascular calcification heterogeneity in chronic kidney
disease. Circul Res. 132:993–1012. 2023.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Heiss A, DuChesne A, Denecke B, Grötzinger
J, Yamamoto K, Renné T and Jahnen-Dechent W: Structural basis of
calcification inhibition by alpha 2-HS glycoprotein/fetuin-A.
Formation of colloidal calciprotein particles. J Biol Chem.
278:13333–13341. 2003.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Schäfer C, Heiss A, Schwarz A, Westenfeld
R, Ketteler M, Floege J, Muller-Esterl W, Schinke T and
Jahnen-Dechent W: The serum protein alpha 2-Heremans-Schmid
glycoprotein/fetuin-A is a systemically acting inhibitor of ectopic
calcification. J Clin Invest. 112:357–366. 2003.PubMed/NCBI View
Article : Google Scholar
|
|
9
|
Iyemere VP, Proudfoot D, Weissberg PL and
Shanahan CM: Vascular smooth muscle cell phenotypic plasticity and
the regulation of vascular calcification. J Intern Med.
260:192–210. 2006.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Luo G, Ducy P, McKee MD, Pinero GJ, Loyer
E, Behringer RR and Karsenty G: Spontaneous calcification of
arteries and cartilage in mice lacking matrix GLA protein. Nature.
386:78–81. 1997.PubMed/NCBI View
Article : Google Scholar
|
|
11
|
Schurgers LJ, Cranenburg ECM and Vermeer
C: Matrix Gla-protein: The calcification inhibitor in need of
vitamin K. Thromb Haemost. 100:593–603. 2017.PubMed/NCBI
|
|
12
|
Kumric M, Borovac JA, Kurir TT, Martinovic
D, Separovic IF, Baric L and Bozic J: Role of matrix gla protein in
the complex network of coronary artery disease: A comprehensive
review. Life (Basel). 11(737)2021.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Stenvinkel P, Wang K, Qureshi AR, Axelsson
J, Pecoits-Filho R, Gao P, Barany P, Lindholm B, Jogestrand T,
Heimbürger O, et al: Low fetuin-A levels are associated with
cardiovascular death: Impact of variations in the gene encoding
fetuin. Kidney Int. 67:2383–2392. 2005.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Ketteler M, Bongartz P, Westenfeld R,
Wildberger JE, Mahnken AH, Böhm R, Metzger T, Wanner C,
Jahnen-Dechent W and Floege J: Association of low fetuin-A (AHSG)
concentrations in serum with cardiovascular mortality in patients
on dialysis: A cross-sectional study. Lancet. 361:827–833.
2003.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Schurgers LJ, Barreto DV, Barreto FC,
Liabeuf S, Renard C, Magdeleyns EJ, Vermeer C, Choukroun G and
Massy ZA: The circulating inactive form of matrix Gla protein is a
surrogate marker for vascular calcification in chronic kidney
disease. Clin J Am Soc Nephrol. 5:568–575. 2010.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Fain ME, Kapuku GK, Paulson WD, Williams
CF, Raed A, Dong Y, Knapen MHJ, Vermeer C and Pollock NK: Inactive
matrix gla protein, arterial stiffness, and endothelial function in
african american hemodialysis patients. Am J Hypertension.
31:735–741. 2018.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Lin YH, Lin MH, Shi CS, Lin YS, Lin CL,
Yang YH, Liao YS, Chen MY, Tsai MH and Lin MS: The impact of
fetuin-A on predicting aortic arch calcification: Secondary
analysis of a community-based survey. Front Cardiovasc Med.
11(1415438)2024.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Jirak P, Mirna M, Wernly B, Paar V, Thieme
M, Betge S, Franz M, Hoppe U, Lauten A, Kammler J, et al: Analysis
of novel cardiovascular biomarkers in patients with peripheral
artery disease. Minerva Med. 109:443–450. 2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Ix JH, Barrett-Connor E, Wassel CL,
Cummins K, Bergstrom J, Daniels LB and Laughlin GA: The
associations of fetuin-A with subclinical cardiovascular disease in
community-dwelling persons. J Am Coll Cardiol. 58:2372–2379.
2011.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Aroner SA, St-Jules DE, Mukamal KJ, Katz
R, Shlipak MG, Criqui MH, Kestenbaum B, Siscovick DS, de Boer IH,
Jenny NS, et al: Fetuin-A, glycemic status, and risk of
cardiovascular disease: The multi-ethnic study of atherosclerosis.
Atherosclerosis. 248:224–229. 2016.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Cahalane RM, Barrett HE, Ross AM,
Mulvihill JJE, Purtill H, Selvarajah L, O'Brien J, Kavanagh EG,
Moloneye MA, Egan SM, et al: On the association between circulating
biomarkers and atherosclerotic calcification in a cohort of
arterial disease participants. Nutr Metab Cardiovasc Dis.
31:1533–1541. 2021.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Laugsand LE, Ix JH, Bartz TM, Djousse L,
Kizer JR, Tracy RP, Dehghan A, Rexrode K, Lopez OL, Rimm EB, et al:
Fetuin-A and risk of coronary heart disease: A Mendelian
randomization analysis and a pooled analysis of AHSG genetic
variants in 7 prospective studies. Atherosclerosis. 243:44–52.
2015.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Roos M, von Eynatten M, Heemann U,
Rothenbacher D, Brenner H and Breitling LP: Serum fetuin-A,
cardiovascular risk factors, and six-year follow-up outcome in
patients with coronary heart disease. Am J Cardiol. 105:1666–1672.
2010.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Akin F, Celik O, Altun I, Ayca B, Diker
VO, Satılmıs S and Sahin C: Relationship of fibroblast growth
factor 23 and fetuin-A to coronary atherosclerosis. J Diabetes
Complications. 29:550–555. 2015.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Mohamed ON, Mohamed MRM, Hassan IG,
Alakkad AF, Othman A, Setouhi A and Issa AS: The relationship of
fetuin-A with coronary calcification, carotid atherosclerosis, and
mortality risk in non-dialysis chronic kidney disease. J Lipid
Atheroscler. 13:194–211. 2024.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Pertosa G, Simone S, Ciccone M, Porreca S,
Zaza G, Dalfino G, Memoli B, Procino A, Bonomini M, Sirolli V, et
al: Serum Fetuin A in hemodialysis: A link between derangement of
calcium-phosphorus homeostasis and progression of atherosclerosis?
Am J Kidney Dis. 53:467–474. 2009.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Hermans MMH, Brandenburg V, Ketteler M,
Kooman JP, van der Sande FM, Boeschoten EW, Leunissen KM, Krediet
RT and Dekker FW: Netherlands cooperative study on the adequacy of
Dialysis (NECOSAD). Association of serum fetuin-A levels with
mortality in dialysis patients. Kidney Int. 72:202–207.
2007.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Pateinakis P, Papagianni A, Douma S,
Efstratiadis G and Memmos D: Associations of fetuin-A and
osteoprotegerin with arterial stiffness and early atherosclerosis
in chronic hemodialysis patients. BMC Nephrol.
14(122)2013.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Chen HY, Chiu YL, Hsu SP, Pai MF, Yang JY
and Peng YS: Low serum fetuin A levels and incident stroke in
patients with maintenance haemodialysis. Eur J Clin Invest.
43:387–396. 2013.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Pencak P, Czerwieńska B, Ficek R, Wyskida
K, Kujawa-Szewieczek A, Olszanecka-Glinianowicz M, Więcek A and
Chudek J: Calcification of coronary arteries and abdominal aorta in
relation to traditional and novel risk factors of atherosclerosis
in hemodialysis patients. BMC Nephrol. 14(10)2013.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Collado S, Coll E, Nicolau C, Azqueta M,
Pons M, Cruzado JM, de la Torre B, Deulofeu R, Mojal S, Pascual J
and Cases A: Serum osteoprotegerin in prevalent hemodialysis
patients: Associations with mortality, atherosclerosis and cardiac
function. BMC Nephrol. 18(290)2017.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Ossareh S, Rayatnia M, Vahedi M, Jafari H
and Zebarjadi M: Association of serum fetuin-A with vascular
calcification in hemodialysis patients and its' impact on 3-year
mortality. Iran J Kidney Dis. 14:500–509. 2020.PubMed/NCBI
|
|
33
|
Willeit K, Santer P, Tschiderer L,
Pechlaner R, Vermeer C, Willeit J and Kiechl S: Association of
desphospho-uncarboxylated matrix gla protein with incident
cardiovascular disease and all-cause mortality: Results from the
prospective Bruneck Study. Atherosclerosis. 353:20–27.
2022.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Virmani R, Burke AP, Farb A and Kolodgie
FD: Pathology of the unstable plaque. Prog Cardiovasc Dis.
44:349–356. 2002.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Mayer O, Seidlerová J, Bruthans J,
Filipovský J, Timoracká K, Vaněk J, Cerná L, Wohlfahrt P, Cífková
R, Theuwissen E and Vermeer C: Desphospho-uncarboxylated matrix
Gla-protein is associated with mortality risk in patients with
chronic stable vascular disease. Atherosclerosis. 235:162–168.
2014.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Berlot AA, Fu X, Shea MK, Tracy R, Budoff
M, Kim RS, Naveed M, Booth SL, Kizer JR and Bortnick AE: Matrix Gla
protein and the long-term incidence and progression of coronary
artery and aortic calcification in the multi-ethnic study of
atherosclerosis. Atherosclerosis. 392(117505)2024.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Bilalic A, Kurir TT, Kumric M, Borovac JA,
Matetic A, Supe-Domic D and Bozic J: Circulating levels of
dephosphorylated-uncarboxylated matrix gla protein in patients with
acute coronary syndrome. Molecules. 26(1108)2021.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Shea MK, Booth SL, Weiner DE, Brinkley TE,
Kanaya AM, Murphy RA, Simonsick EM, Wassel CL, Vermeer C and
Kritchevsky SB: Health ABC Study. Circulating vitamin K Is
inversely associated with incident cardiovascular disease risk
among those treated for hypertension in the health, aging, and body
composition study (Health ABC). J Nutr. 147:888–895.
2017.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Liu YP, Gu YM, Thijs L, Knapen MH, Salvi
E, Citterio L, Petit T, Carpini SD, Zhang Z, Jacobs L, et al:
Inactive matrix gla protein is causally related to adverse health
outcomes. Hypertension. 65:463–470. 2015.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Zwakenberg SR, Burgess S, Sluijs I,
Weiderpass E, Beulens JWJ and van der Schouw YT: Circulating
phylloquinone, inactive Matrix Gla protein and coronary heart
disease risk: A two-sample mendelian randomization study. Clin
Nutr. 39:1131–1136. 2020.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Berlot AA, Fu X, Shea MK, Tracy R, Budoff
M, Kim RS, Naveed M, Booth SL, Kizer JR and Bortnick AE: Inactive
matrix gla protein and cardiovascular outcomes: The multi-ethnic
study of atherosclerosis. J Am Heart Assoc.
14(e036459)2025.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Delanaye P, Krzesinski JM, Warling X,
Moonen M, Smelten N, Médart L, Pottel H and Cavalier E:
Dephosphorylated-uncarboxylated Matrix Gla protein concentration is
predictive of vitamin K status and is correlated with vascular
calcification in a cohort of hemodialysis patients. BMC Nephrol.
15(145)2014.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Jaminon AMG, Dai L, Qureshi AR, Evenepoel
P, Ripsweden J, Söderberg M, Witasp A, Olauson H, Schurgers LJ and
Stenvinkel P: Matrix Gla protein is an independent predictor of
both intimal and medial vascular calcification in chronic kidney
disease. Sci Rep. 10(6586)2020.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Dai L, Li L, Erlandsson H, Jaminon AMG,
Qureshi AR, Ripsweden J, Brismar TB, Witasp A, Heimbürger O,
Jørgensen HS, et al: Functional vitamin K insufficiency, vascular
calcification and mortality in advanced chronic kidney disease: A
cohort study. PLoS One. 16(e0247623)2021.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Fitzpatrick J, Kim ED, Sozio SM, Jaar BG,
Estrella MM, Monroy-Trujillo JM and Parekh RS: Calcification
biomarkers, subclinical vascular disease, and mortality among
multiethnic dialysis patients. Kidney Int Rep. 5:1729–1737.
2020.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Shea MK, Wang J, Barger K, Weiner DE,
Townsend RR, Feldman HI, Rosas SE, Chen J, He J, Flack J, et al:
Association of vitamin K status with arterial calcification and
stiffness in chronic kidney disease: The chronic renal
insufficiency cohort. Curr Dev Nutr. 7(100008)2023.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Shea MK, Barger K, Booth SL, Wang J,
Feldman HI, Townsend RR, Chen J, Flack J, He J, Jaar BG, et al:
Vitamin K status, all-cause mortality, and cardiovascular disease
in adults with chronic kidney disease: The chronic renal
insufficiency cohort. Am J Clin Nutr. 115:941–948. 2022.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Schlieper G, Westenfeld R, Krüger T,
Cranenburg EC, Magdeleyns EJ, Brandenburg VM, Djuric Z, Damjanovic
T, Ketteler M, Vermeer C, et al: Circulating nonphosphorylated
carboxylated matrix gla protein predicts survival in ESRD. J Am Soc
Nephrol. 22:387–395. 2011.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Gluba-Brzózka A, Michalska-Kasiczak M,
Franczyk-Skóra B, Nocuń M, Banach M and Rysz J: Markers of
increased cardiovascular risk in patients with chronic kidney
disease. Lipids Health Dis. 13(135)2014.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Mao L, Huang H, Zhou M and Zhou C:
Correlation between circulating dephosphorylated uncarboxylated
matrix Gla protein and vascular calcification in peritoneal
dialysis patients. Int J Artif Organs. 47:885–893. 2024.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Nyvad J, Christensen KL, Andersen G,
Reinhard M, Nørgaard BL, Madsen JS, Nielsen S, Thomsen MB, Jensen
JM, Peters CD and Buus NH: PIVKA-II but not dp-ucMGP is associated
with aortic calcification in chronic kidney disease. BMC Nephrol.
25(426)2024.PubMed/NCBI View Article : Google Scholar
|