1. Introduction
Periodontitis is a prevalent inflammatory condition
involving the supporting structures of the teeth, gingiva,
periodontal ligament, cementum and alveolar bone. It occurs
primarily due to the accumulation of dental plaque and bacterial
biofilm that trigger the host mediated inflammatory reactions,
thereby causing periodontal destruction. This process eventually
results in tooth mobility and eventual tooth loss if left untreated
(1,2).
Several methods have been employed over a period of
time to accomplish periodontal regeneration. Using barrier
membranes to prevent the rapid migration of epithelial and
connective tissue cells into the defect area, guided tissue
regeneration (GTR) and guided bone regeneration (GBR) are two
well-established clinical modalities (1) that enable slower-growing periodontal
ligament cells, cementoblasts and osteoblasts to repopulate the
site and promote regeneration (3).
Conventional GTR/GBR methods frequently produce only partial and
unpredictable regeneration, despite their success in improving
clinical outcomes. The coronal regions are often populated by a
long junctional epithelium or non-functional connective tissue, and
regeneration is often restricted to the apical portion of the
defect (4).
The majority of regenerative materials available
today are unable to actively direct or instruct cell behavior in a
manner that replicates the intricate structure of the native
periodontium. True regeneration is therefore still difficult to
achieve in standard clinical practice (5,6).
In recent years, 3D printing and biomimetics have
emerged as promising approaches for periodontal regeneration
(7). The combination of biomimetic
principles and additive manufacturing signifies a paradigm shift
toward a more potent, biologically relevant treatment, as
traditional regenerative methods struggle with predictability and
integration. With a focus on their revolutionary role in
next-generation regenerative dentistry, the present review attempts
to summarize and discuss the present uses, underlying mechanisms
and future prospects of 3D printing and biomimetics in implant and
periodontal therapy (4).
2. Convergence of biomimetics and 3D
printing
Biomimetics aims to replicate the natural healing
and developmental processes of tissues. It refers to the design and
development of materials and systems that mimic biological
structures and functions. With the use of bioactive compounds, stem
cell-based treatments and intelligent scaffolds that direct
tissue-specific regeneration, biomimetic approaches in periodontics
seek to replicate the intricate architecture of the periodontium
(8). These methods draw
inspiration from biological concepts, such as the collagen fiber
orientation of the periodontal ligament (PDL), the slow
mineralization of cementum, and the coordinated cellular and
vascular networks necessary for functional regeneration (9,10).
3D printing or additive manufacturing complements
biomimetic approaches by making it possible to precisely fabricate
patient-specific scaffolds with customized geometries, porosities
and compositions on the basis of digital imaging modalities such as
cone-beam computed tomography. Unlike traditional scaffold
fabrication techniques, 3D printing enables the layer-by-layer
spatial arrangement of various materials and cell types, closely
resembling the hierarchical and heterogeneous structure of the
periodontium (4).
Thus, the combination of biomimetic principles and
3D printing technologies offers not only biological and structural
fidelity to natural tissues, but also personalized, functional
regeneration that overcomes the drawbacks of conventional treatment
modalities, including limited tissue specificity and incomplete
integration. It also provides promising periodontal therapies that
are more clinically relevant, predictable and effective (4).
3. 3D printing techniques in dentistry
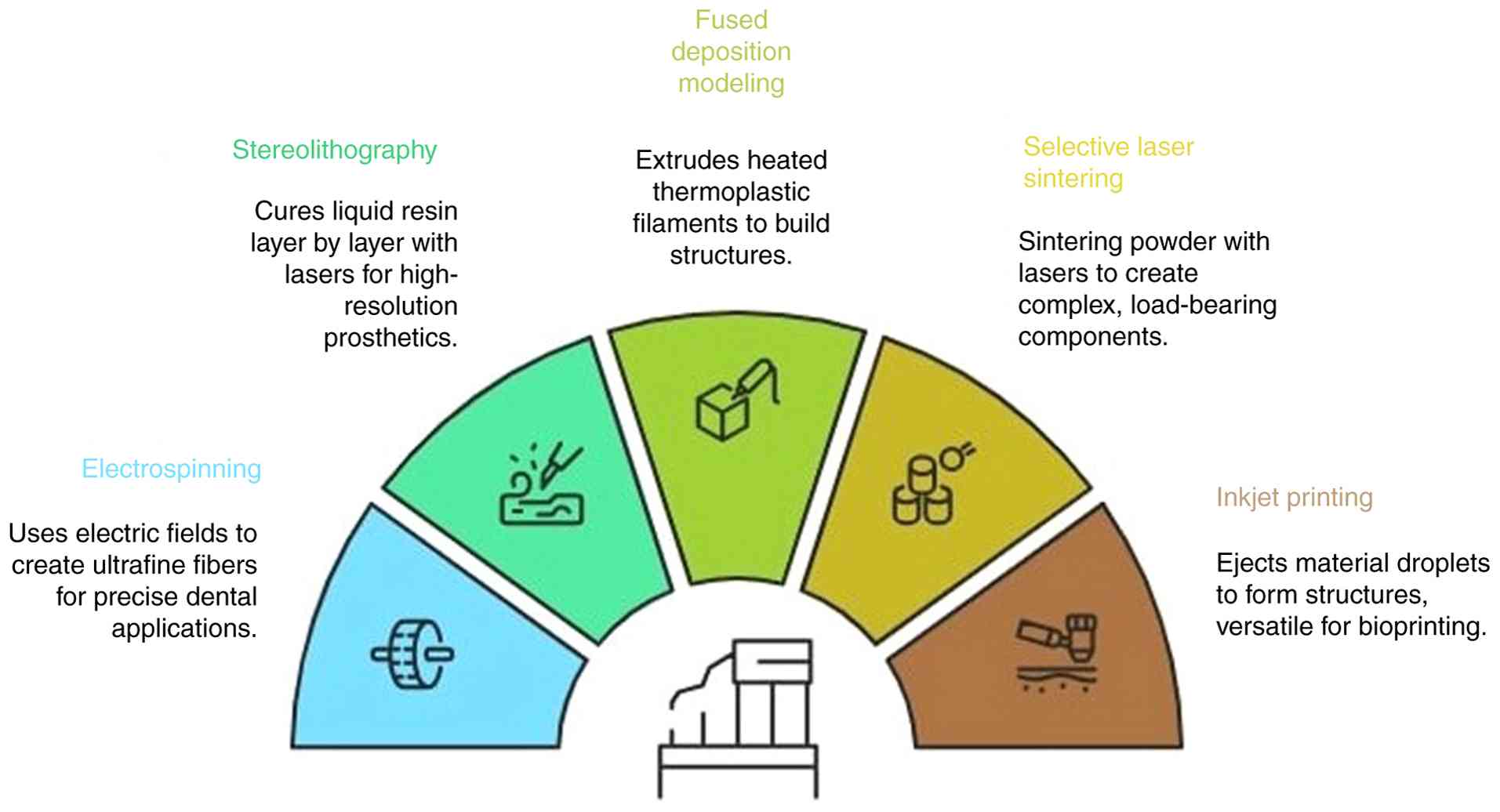
3D printing includes several different techniques,
all of which are appropriate for use in dentistry (Fig. 1). Electrospinning, which uses an
electric field to pull a charged polymer solution into ultrafine
fibers, is one of the most promising methods. At the needle tip, a
Taylor cone is formed, and fibers are then sent in the direction of
a collector plate. Applications, such as antibiotic-eluting
nanofiber scaffolds for regenerative endodontics can benefit from
the ability of electrospinning to precisely control fiber diameter,
porosity and drug loading (5).
The first 3D printing method was created in 1984 and
was termed stereolithography (SLA). Layer by layer, the liquid
photopolymer resin is cured using a laser. High resolution and
rapid speeds render modern SLA printers perfect for detailed
prosthetics and surgical guides (11,12).
Thermoplastic filaments are heated and then extruded
layer by layer through a nozzle in fused deposition modelling, also
known as fused filament fabrication. Despite being widely used and
reasonably priced, it has trouble with unsupported overhangs and
frequently requires dual-nozzle printers and dissolvable support
structures (13).
Using a powder bed fusion process known as selective
laser sintering, a laser is used to sinter powdered material into
solid layers. There is no need for extra support as the structure
is supported by the powder bed itself during printing. Complex,
load-bearing components can benefit from this technique (11).
Finally, inkjet printing creates structures by
ejecting small material droplets onto a substrate. Fast printing
and material versatility, including bioprinting applications, are
made possible by this technique's multi-nozzle heads (13).
A key limitation in current periodontal tissue
engineering is the inability to reproduce the intricate
hierarchical architecture of the periodontium. Conventional
layer-by-layer fabrication techniques generally produce isotropic
scaffolds that fail to replicate the anisotropic orientation of
fibers and mineral phases required for physiological load transfer.
Magnetic-assisted 3D printing has emerged as a promising strategy
to address this challenge. In this approach, an external magnetic
field is applied during direct ink writing to precisely regulate
the spatial orientation of magnetically responsive particles, such
as hydroxyapatite (HAp) nanorods, incorporated within the bioink.
The magnetic field induces the directional alignment of these
nanorods, enabling the fabrication of anisotropic microstructures
that resemble the organized mineral arrangement observed in natural
dental tissues. Such controlled alignment has been explored to
replicate hierarchical dental interfaces, particularly the
enamel-dentin junction, where anisotropic mineral orientation
contributes to a gradual mechanical gradient between tissues. This
biomimetic architecture can improve stress distribution and enhance
crack-deflection and fracture-resistance properties, thereby better
mimicking the functional performance of natural dental interfaces
(14).
4. Scaffold biomaterials used in periodontal
regeneration
The success of periodontal regeneration largely
depends on the selection of suitable scaffold materials to provide
suitable environment for cell differentiation. Various scaffold
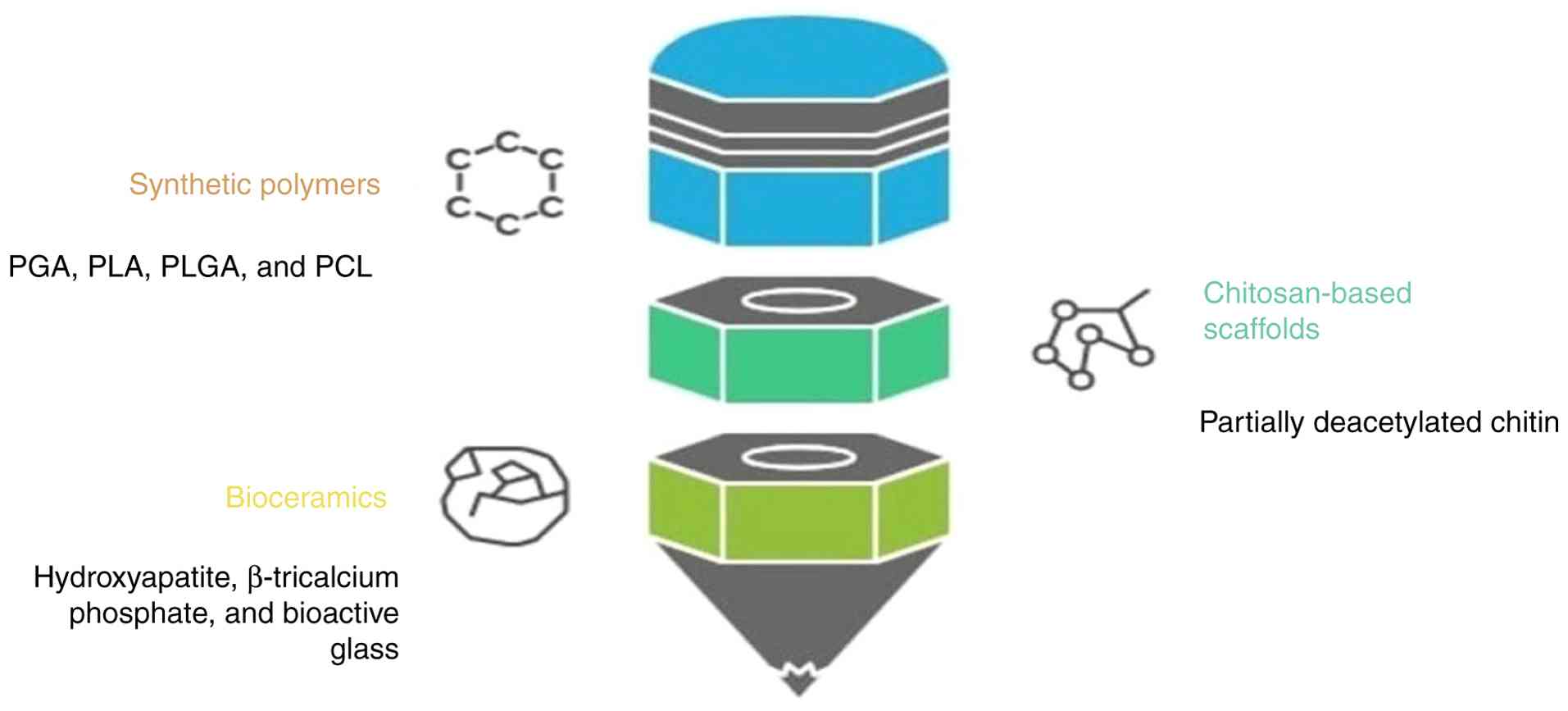
materials available for periodontal regeneration (Fig. 2).
Synthetic polymers
Second-generation degradable membranes, replace the
non-resorbable membrane (PTFE) and are primarily comprised of
synthetic polymers. They are frequently utilized as scaffold
materials due to their adjustable mechanical strength, degradation
profiles and manufacturing reproducibility. Among these, the most
widely used materials are poly(glycolic acid) (PGA), poly(lactic
acid) (PLA), poly(lactic-co-glycolic acid) (PLGA) and
Polycaprolactone (PCL) (15).
PGA offers high mechanical stability and
biocompatibility. It has demonstrated promising results in both
in vitro and in vivo models for tissue regeneration,
and promotes the growth and production of the extracellular matrix
by periodontal ligament stem cells (16).
Compared with PGA, PLA is more hydrophobic and
breaks down into lactic acid. Its efficacy as a scaffold is
increased when it is combined with natural polymers, such as
chitosan, to enhance cell attachment and degradation behavior
(15).
The benefits of PLA and PGA are combined in PLGA,
which provides properties that can be adjusted by changing the
copolymer ratio. PLGA scaffolds have demonstrated success in
promoting cell adhesion, controlled drug delivery, and periodontal
tissue regeneration, particularly when functionalized with
proteins, such as fibronectin or when combined with hydrogels
(16).
PCL is characterized by high flexibility and a slow
rate of degradation. To improve tissue regeneration and control
inflammation, it has been utilized as a delivery system for
antimicrobial and anti-inflammatory drugs, such as tetracyclines,
ibuprofen and resveratrol. The biological performance and
structural mimicry of composite scaffolds containing PCL and
osteoconductive materials, such as gelatin or calcium phosphate are
enhanced (17).
Chitosan-based scaffolds in
periodontal regeneration
As a naturally occurring biopolymer produced by
partially deacetylating chitin, chitosan has demonstrated immense
promise in periodontal regenerative treatments. It is particularly
well-suited for use in periodontal scaffolding due to its intrinsic
qualities, which include biocompatibility, biodegradability,
antimicrobial activity and mucoadhesiveness. To replicate the
structure of periodontal tissues, recent developments have
investigated layered and composite scaffolds that incorporate
chitosan (18).
Owing to the high cell viability of osteoblasts,
fibroblasts and PDL cells, a tri-layered scaffold comprise of
crosslinked chitosan and genipin has been shown to support the
simultaneous regeneration of bone, gingiva and periodontal
ligament. Similarly, the addition of bioactive glass nanoparticles
to chitosan matrices enhances their mechanical flexibility and
cellular metabolic activity, particularly when they are wet,
suggesting that they may have in vivo functionality
(18,19).
The function of chitosan extends beyond structural
repair. It reduces key clinical indicators of periodontitis,
including plaque buildup, gingival inflammation and attachment
loss, when combined with ascorbate to create a bioadhesive gel with
improved antimicrobial efficacy. The robust attachment of chitosan
to root surfaces promotes tissue stability and decreases the
inflammatory infiltration (20).
Bioceramic-based scaffolds
Bioceramics, such as hydroxyapatite, β-tricalcium
phosphate and bioactive glass have shown immense promise in
periodontal regeneration due to their superior osteoconductive and
osteoinductive qualities. These materials promote alveolar bone
repair in periodontal defects by providing advantageous
biocompatibility, mechanical strength, and biodegradability. The
clinical versatility of these materials has increased due to their
availability in versatile forms, such as injectable systems, pastes
and granules (15).
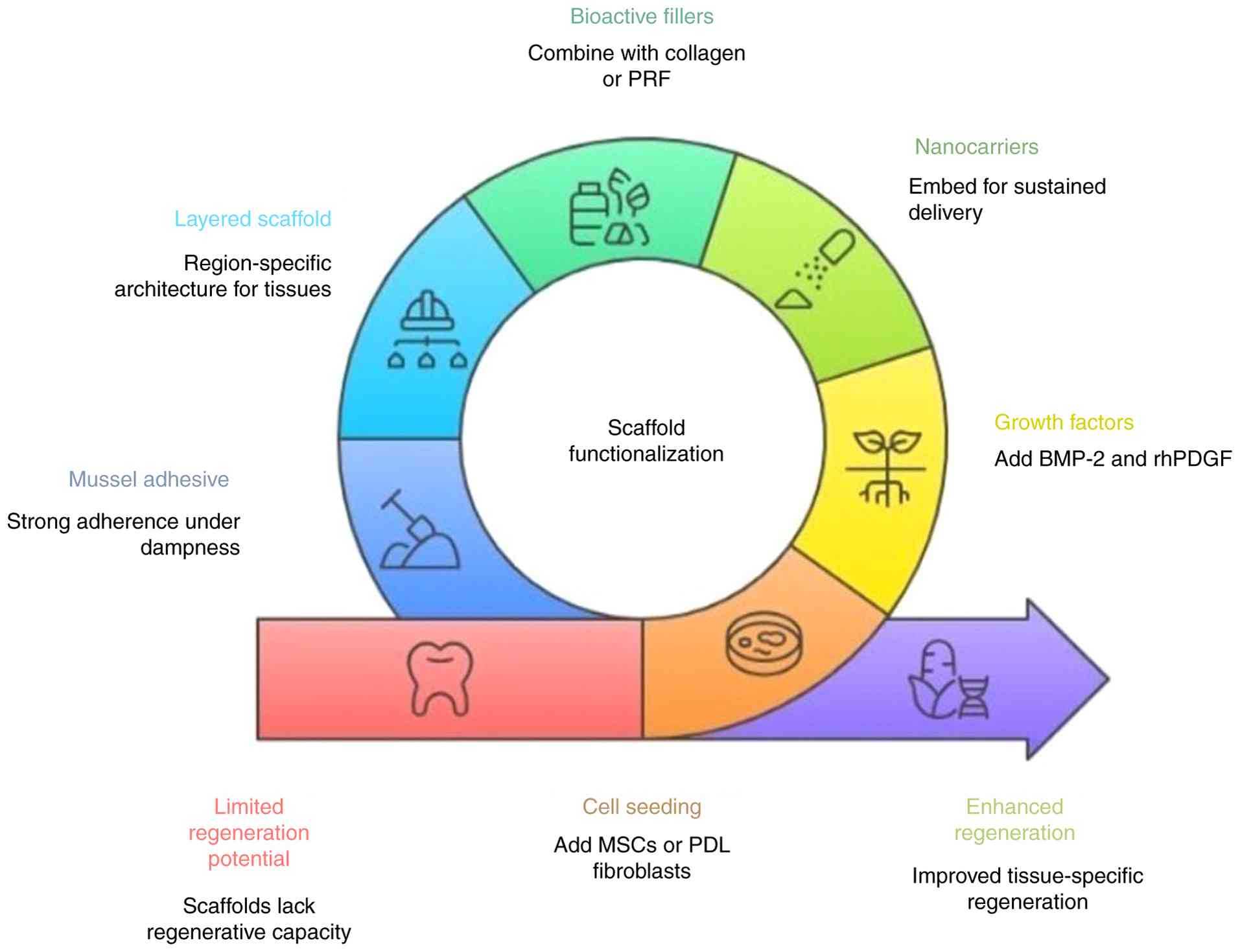
5. Functionalization of 3D printed
scaffolds
To increase the potential of 3D printed scaffolds,
they are functionalized with living cells and bioactive compounds
are added to 3D-printed scaffolds to improve their capacity for
regeneration (Fig. 3). To
encourage tissue-specific regeneration and hasten healing,
mesenchymal stem cells or PDL fibroblasts can be seeded onto these
scaffolds. To promote cellular migration, differentiation and
proliferation, growth factors, such as bone morphogenetic protein-2
and recombinant human platelet-derived growth factor are also
added. Additionally, by embedding nanocarriers in the scaffold
matrix, therapeutic agents can be delivered locally and
sustainably, reducing systemic exposure and guaranteeing prolonged
bioactivity. This overcomes the lack of regenerative potential of
3D printed scaffold materials (21-23).
An essential aspect to consider during 3D
bioprinting is cell viability which is affected by various factors,
such as shear stress, nozzle pressure and individual property of
bioink. These often result in compromised the stem cells. However,
the optimization of bioink can help in overcoming this obstacle.
Gelatin-methacryloyl (GelMA) hydrogels have been shown to reduce
the shear thinning, while also preserving the viability of cells.
Therefore, the careful optimization of bioink properties can
enhance the effectiveness of bioprinting in regenerative
periodontics. In addition, batch standardization and quality
control are essential for clinical translation. Computational
modelling approaches with micro-CT porosity mapping can help ensure
minimal variability in scaffold architecture across production
batches (14).
Role of growth factors
Synthetic polymers are excellent options for tissue
engineering applications due to their favorable mechanical
strength, adjustable degradation kinetics, and adaptability in drug
and gene delivery systems (15).
Bioactive substances, such as fibroblast growth
factor-2 (FGF-2) have been effectively transported by PLGA.
Research has demonstrated that PLGA scaffolds containing plasmid
FGF-2 (pFGF-2) release the gene continuously without causing an
initial burst, and retain its bioactivity for as long as 21 days
(24). In contrast to PLGA
scaffolds without gene loading, this controlled delivery stimulates
the growth of human periodontal ligament cells (hPDLCs), increases
the expression of extracellular matrix proteins (such as collagen I
and scleraxis), and facilitates the formation of more ordered
PDL-like tissues in vivo with less root resorption (23).
Furthermore, composite scaffolds that combine
synthetic scaffolds with bioactive fillers, such as collagen or
platelet-rich fibrin, HAp and calcium derived from eggshells have
demonstrated enhanced hPDLC cell adhesion and viability. By
addressing the inherent drawbacks of 3D scaffolds, such as low cell
affinity and hydrophobicity, these changes improve its
biofunctionality (25).
The modification of PLGA scaffolds to achieve the
ideal porosity, structural integrity and spatial distribution of
genetic material or growth factors is possible. Due to these
characteristics, PLGA is a very versatile and promising scaffold
material for tissue engineering in the periodontal and peri-implant
regions (24-26).
Layered scaffolds
By allowing for a region-specific architecture that
replicates the intricate structure of the periodontal apparatus,
layering in 3D-printed scaffolds provides a tactical advantage for
periodontal regeneration. The alveolar bone, PDL and cementum are
three separate, yet functionally related tissues that comprise the
periodontium. Each has specific biological and structural needs.
Layered scaffolds support the different cellular behaviors required
for each tissue type by enabling the incorporation of different
microstructures and pore sizes within a single construct. For
example, electrospun nanofibrous layers mimic the fibrous network
of the PDL, whereas the larger, interconnected pores of the bone
facing layer encourage vascularization and bone ingrowth.
Additionally, layering facilitates the creation of a gradual
interface between these areas, which is very similar to the manner
in which that bone, ligament and cementum naturally transition. To
prevent soft tissue invasion, an upper layer in some designs may
function as a GTR barrier. Therefore, layered 3D-printed scaffolds
provide a biomimetic platform for the regeneration of periodontal
tissue in a spatially and functionally coordinated manner (27,28).
Mussel adhesive proteins (MAPs)
MAPs provide a unique advantage in enhancing
scaffold-based periodontal regeneration. Stable scaffold placement
is made possible by strong adherence under damp conditions, which
eliminates the need for additional fixation techniques. MAPs
support the differentiation of periodontal ligament and
bone-forming cells, enhance extracellular matrix secretion and
improve cell attachment when added to scaffolds or applied as
surface coatings. These biological effects promote improved
integration with surrounding structures, and hasten tissue
regeneration. When combined with biomaterials, such as titanium or
calcium silicate, MAPs also improve the osteogenic potential of
scaffolds, promoting more efficient bone regeneration. Their
compatibility with platelet concentrates, such as PRF, further
promote healing. MAPs facilitate the handling of scaffolds and
membranes, and their position during surgery, increasing their
usability. Overall, the inclusion of MAPs in scaffolds improves
their functional results and exhibits promise for more consistent
and effective periodontal regeneration (29).
6. Barriers to clinical translation
Currently the use of 3D-printed scaffolds for
periodontal regeneration remains largely confined to preclinical
stages, in which small-animal models (e.g., rat calvarial or
fenestration defects) are utilized to demonstrate proof-of-concept
regeneration of PDL, cementum, and alveolar bone. Although these
models validate the ability of multiphasic scaffolds to guide
tissue integration, the clinical translation to humans is limited
at this stage due to the anatomical differences between the animal
models and human periodontium. Challenges, such as vascularization
and antimicrobial functionality for the scaffold still need to be
addressed to ensure its stability in the oral cavity. To bridge
this gap, early phase human trials are warranted to confirm the
safety and effectiveness of bioinks and the resulting clinical
efficacy of the scaffolds to make progress toward personalized
defect specific therapies (30).
7. Future directions
While the field of bioprinting has evolved to
provide more sophisticated scaffolds, challenges remain. The
currently available biomaterials have their own limitations;
thereby, the need to refine the scaffolds to resemble the complex
nature of the periodontium is necessary. Future studies are
required to focus on the use of nanotechnology and layered
scaffolding to advance the bioprinting methods. Since
vascularization is crucial for the survival of the scaffold,
further studies are required to focus on the incorporation of
pre-vascularized networks and to also aim to improve the cellular
response to stimulate the differentiation of the mesenchymal cells.
Regulatory guidelines and standardization protocols are mandatory
to promote the clinical adoption of 3D printed scaffolds, thereby
offering a personalized patient specific solution for periodontal
regeneration.
8. Conclusion
Conventional approaches, such as GTR/GBR are
currently enhanced by 3D printed scaffolds for improved tissue
healing and regeneration. However, the clinical significance
remains limited due to the challenges faced in the development of
bioinks and maintaining the resorption rate of scaffolds. The field
is still a growing arena undergoing constant development to
improvise clinical outcomes in terms of the controlled release of
bioactive agents and to mimic the dynamic nature of the
periodontium. Although preclinical studies have yielded results,
further studies are required to address the long-term clinical
benefits to provide superior regenerative solution and regulatory
guidelines. Standardization is also essential to translate the
findings into clinical practice. 3D printing and biomimetics stand
as an excellent solution for providing patient specific outcomes in
periodontal regeneration.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
MM and SUN were involved in the concept and design
of the study, in the acquisition, analysis and interpretation of
data from the literature, and in the drafting, reviewing and
editing of the manuscript. ASU and SK were involved in the concept
and design of the study, and in the drafting, reviewing and editing
of the manuscript. All authors have read and approved the final
manuscript. Data authentication is not applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Use of artificial intelligence tools
During the preparation of this work, AI tools were
used to improve the readability and language of the manuscript or
to generate images, and subsequently, the authors revised and
edited the content produced by the AI tools as necessary, taking
full responsibility for the ultimate content of the present
manuscript.
References
|
1
|
Vahdatinia F, Hooshyarfard A, Jamshidi S,
Shojaei S, Patel K, Moeinifard E, Haddadi R, Farhadian M, Gholami L
and Tayebi L: 3D-printed soft membrane for periodontal guided
tissue regeneration. Materials (Basel). 16(1364)2023.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Davidopoulou S, Karakostas P, Batas L,
Barmpalexis P, Assimopoulou A, Angelopoulos C and Tsalikis L:
Multidimensional 3D-printed scaffolds and regeneration of intrabony
periodontal defects: A systematic review. J Funct Biomater.
15(44)2024.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Behfarnia P, Khorasani MM, Birang R and
Abbas FM: Histological and histomorphometric analysis of animal
experimental dehiscence defect treated with three bioabsorbable GTR
collagen membranes. Dent Res J (Isfahan). 9:574–581.
2012.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Lee HS, Byun SH, Cho SW and Yang BE: Past,
present, and future of regeneration therapy in oral and periodontal
tissue: A review. Appl Sci. 9(1046)2019.
|
|
5
|
Raveau S and Jordana F: Tissue engineering
and three-dimensional printing in periodontal regeneration: A
literature review. J Clin Med. 9(4008)2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Zhang Q, Zhou J, Zhi P, Liu L, Liu C, Fang
A and Zhang Q: 3D printing method for bone tissue engineering
scaffold. Med Nov Technol Devices. 17(None)2023.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Gul M, Arif A and Ghafoor R: Role of
three-dimensional printing in periodontal regeneration and repair:
Literature review. J Indian Soc Periodontol. 23:504–510.
2019.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Jiang S, Wang M and He J: A review of
biomimetic scaffolds for bone regeneration: Toward a cell-free
strategy. Bioeng Transl Med. 6(e10206)2021.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Vaquette C, Pilipchuk SP, Bartold PM,
Hutmacher DW, Giannobile WV and Ivanovski S: Tissue engineered
constructs for periodontal regeneration: Current status and future
perspectives. Adv Healthc Mater. 7(e1800457)2018.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Roato I, Masante B, Putame G, Massai D and
Mussano F: Challenges of periodontal tissue engineering: Increasing
biomimicry through 3D printing and controlled dynamic environment.
Nanomaterials (Basel). 12(3878)2022.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Dwivedi R and Mehrotra D: 3D bioprinting
and craniofacial regeneration. J Oral Biol Craniofac Res.
10:650–659. 2020.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Almeida ND, Carneiro CA, de Marco AC,
Porto VC and França R: 3D bioprinting techniques and bioinks for
periodontal tissues regeneration: A literature review. Biomimetics
(Basel). 9(480)2024.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Gudapati H, Dey M and Ozbolat I: A
comprehensive review on droplet-based bioprinting: Past, present
and future. Biomaterials. 102:20–42. 2016.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Hou Y, Li Y, Yang A, Lu X, Han X, Yang Z,
Sun J and Liu Y: Revolutionizing periodontitis treatment: The
promise of GelMA hydrogel. Int J Pharm. 681(125850)2025.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Woo HN, Cho YJ, Tarafder S and Lee CH:
Recent advances in scaffolds for integrated periodontal
regeneration. Bioact Mater. 6:3328–3342. 2021.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Sun X, Xu C, Wu G, Ye Q and Wang C:
Poly(lactic-co-glycolic acid): Applications and future prospects
for periodontal tissue regeneration. Polymers (Basel).
9(189)2017.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Zupančič Š, Baumgartner S, Lavrič Z,
Petelin M and Kristl J: Local delivery of resveratrol using
polycaprolactone nanofibers for treatment of periodontal disease. J
Drug Deliv Sci Technol. 30 (Part B):408–416. 2015.
|
|
18
|
Varoni EM, Vijayakumar S, Canciani E,
Cochis A, De Nardo L, Lodi G, Rimondini L and Cerruti M:
Chitosan-based trilayer scaffold for multitissue periodontal
regeneration. J Dent Res. 97:303–311. 2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Paczkowska-Walendowska M, Koumentakou I,
Lazaridou M, Bikiaris D, Miklaszewski A, Plech T and
Cielecka-Piontek J: 3D-printed chitosan-based scaffolds with
Scutellaria baicalensis extract for dental applications.
Pharmaceutics. 16(359)2024.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Suo L, Wu H, Wang P, Xue Z, Gao J and Shen
J: Improvement of periodontal tissue regeneration using a
3D-printed carbon nanotube/chitosan/sodium alginate composite
scaffold. J Biomed Mater Res B Appl Biomater. 111:73–84.
2023.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Heckmann L, Fiedler J, Mattes T, Dauner M
and Brenner RE: Interactive effects of growth factors and
three-dimensional scaffolds on multipotent mesenchymal stromal
cells. Biotechnol Appl Biochem. 49 (Pt 3):185–194. 2008.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Kim SE, Park JH, Cho YW, Chung H, Jeong
SY, Lee EB and Kwon IC: Porous chitosan scaffold containing
microspheres loaded with transforming growth factor-beta1:
Implications for cartilage tissue engineering. J Control Release.
91:365–374. 2003.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Giannobile WV: Periodontal tissue
engineering by growth factors. Bone. 19 (1 Suppl):S23–S37.
1996.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Jiang L, Ding Z, Xia S, Liu Y, Lei S,
Zhong M and Chen X: Poly(lactic-co-glycolic acid) scaffold loaded
with plasmid DNA encoding fibroblast growth factor-2 promotes
periodontal ligament regeneration of replanted teeth. J Periodontal
Res. 55:488–495. 2020.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Espitia-Quiroz LC, Fernández-Orjuela AL,
Anaya-Sampayo LM, Acosta-Gómez AP, Sequeda-Castañeda LG,
Gutiérrez-Prieto SJ, Roa-Molina NS and García-Robayo DA: Viability
and adhesion of periodontal ligament fibroblasts on a
hydroxyapatite scaffold combined with collagen, polylactic
acid-polyglycolic acid copolymer and platelet-rich fibrin: A
preclinical pilot study. Dent J (Basel). 10(167)2022.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Wang CY, Chiu YC, Lee AK, Lin YA, Lin PY
and Shie MY: Biofabrication of gingival fibroblast cell-laden
collagen/strontium-doped calcium silicate 3D-printed Bi-layered
scaffold for osteoporotic periodontal regeneration. Biomedicines.
9(431)2021.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Abedi N, Rajabi N, Kharaziha M,
Nejatidanesh F and Tayebi L: Layered scaffolds in periodontal
regeneration. J Oral Biol Craniofac Res. 12:782–797.
2022.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Porta M, Tonda-Turo C, Pierantozzi D,
Ciardelli G and Mancuso E: Towards 3D multi-layer scaffolds for
periodontal tissue engineering applications: Addressing
manufacturing and architectural challenges. Polymers (Basel).
12(2233)2020.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Kwan JC, Dondani J, Iyer J, Muaddi HA,
Nguyen TT and Tran SD: Biomimicry and 3D-printing of mussel
adhesive proteins for regeneration of the periodontium-A review.
Biomimetics (Basel). 8(78)2023.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Chen H, Wang Y, Lai Y, Meng C, Ning X, Xu
T, Song G, Zhang Y, Lin Y and Han B: Advances of 3D bioprinting
technology for periodontal tissue regeneration. iScience.
28(112532)2025.PubMed/NCBI View Article : Google Scholar
|