Introduction
Dry eye disease (DED) is currently defined as a
multifactorial disease of the ocular surface characterized by a
loss of tear film homeostasis, accompanied by inflammation,
hyperosmolarity and neurosensory abnormalities (1). According to the Tear Film and Ocular
Surface Society Dry Eye Workshop II (TFOS DEWS II) consensus, the
key pathophysiological event in DED is the disruption of tear film
stability, followed by a cascade of epithelial damage and
sensitization of corneal afferent nerve pathways (1,2).
Epidemiological studies have demonstrated that the prevalence of
DED varies from 5 to 50%, depending on the diagnostic criteria
used, as well as the geographic and demographic characteristics of
the population (3,4). The pathogenesis of DED involves a
sophisticated network of interconnected etiological factors.
Central to this condition is the disruption of ocular homeostasis,
primarily driven by tear film instability and hyperosmolarity.
These elements function as catalysts for the induction and chronic
propagation of inflammatory responses within the ocular surface
tissues. Consequently, this inflammatory milieu may contribute to
the development of neurosensory abnormalities and subsequent
disruption of visual function (5).
Increasing data indicate an association between DED
and systemic diseases, namely metabolic, autoimmune, endocrine and
chronic inflammatory conditions, that goes beyond viewing DED as
solely a local ophthalmological pathology (3,4).
These observations have contributed to the formation of the concept
of DED as a ‘systemic phenotype’, reflecting the overall metabolic
and immune state of the organism (3,4).
Traditionally viewed as an age-related ophthalmological
inconvenience or one linked to contact lens use, DED warrants a
paradigm shift based on the findings of the Lifelines study
(6). Some researchers have
identified DED associations with comorbidities across nearly all
bodily systems, including musculoskeletal, psychiatric, and notably
gastrointestinal and hepatic systems (6,7). In
recent years, specific attention has been given to interorgan
interactions, particularly the formation of the ‘liver-eye axis’.
The liver serves as the central organ for lipid, vitamin, bile acid
and hormone metabolism, as well as a key immunological filter
regulating the systemic inflammatory response (7,8).
Liver dysfunction is accompanied by the development of chronic
systemic inflammation, oxidative stress and an altered
neuroendocrine regulation, which can potentially exert direct and
indirect effects on tear film homeostasis and the ocular surface
condition (7,9).
Despite the existence of individual clinical and
review publications dedicated to the ophthalmic manifestations of
chronic liver diseases, the majority of these consider ocular
surface pathology primarily as a secondary complication of already
diagnosed liver dysfunction (7,8). The
present scoping review proposes an alternative, clinically oriented
perspective, in which ophthalmic symptoms particularly atypical or
refractory DED are viewed as a potential entry point for detecting
subclinical liver pathology. Unlike previously published works, the
present scoping review systematizes the pathophysiological
mechanisms of the ‘liver-ocular surface’ axis from the standpoint
of DED phenotyping, with an emphasis on the ophthalmologist's
diagnostic vigilance, thereby expanding the clinical interpretation
of ocular surface homeostasis disruptions and justifying an
interdisciplinary approach to early detection of chronic liver
diseases.
Data and methods
Literature search methods
The present scoping review was conducted in
accordance with the Preferred Reporting Items for Systematic
Reviews and Meta-Analyses extension for Scoping Reviews
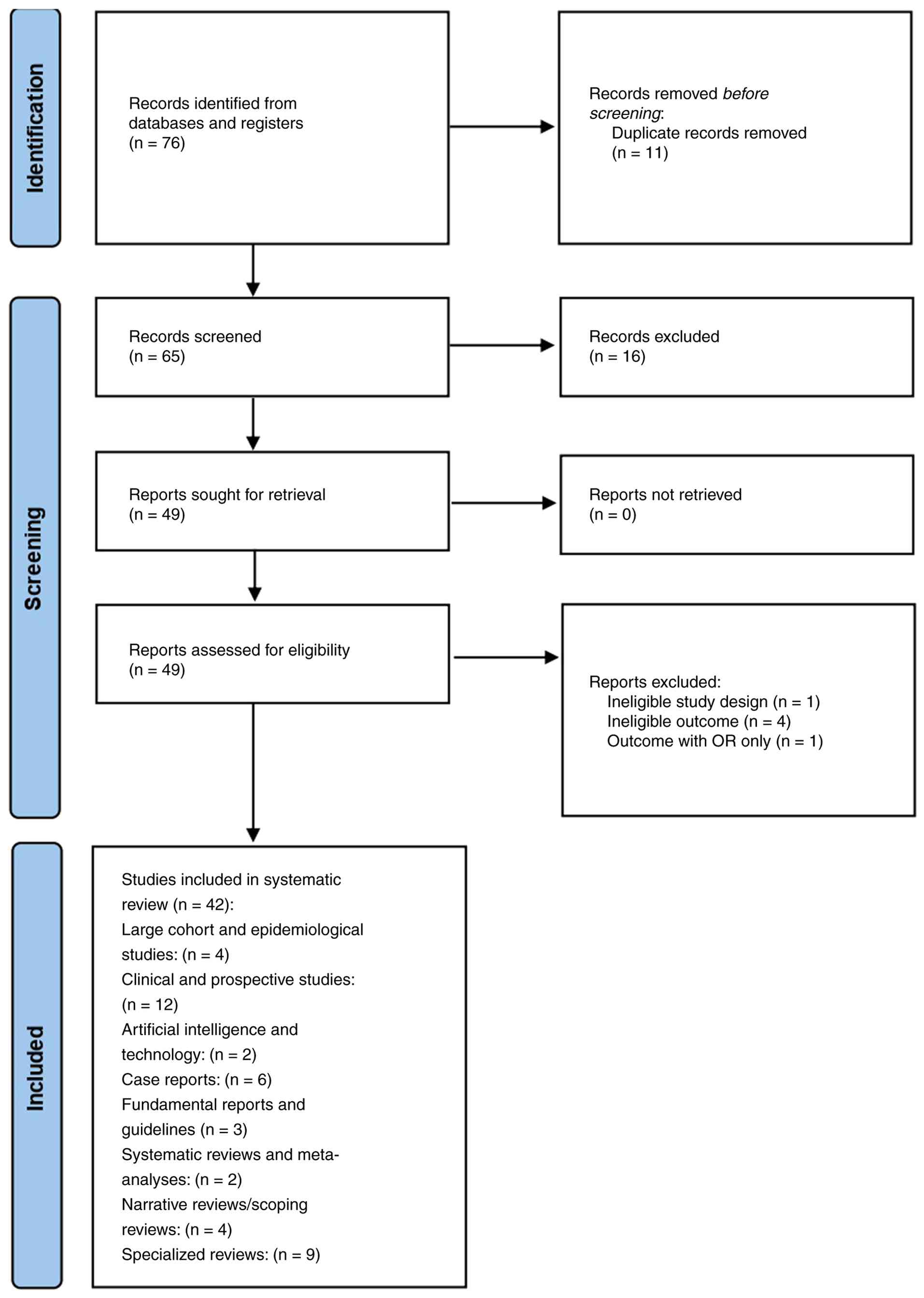
(PRISMA-ScR) guidelines. As detailed in the PRISMA 2020 flow
diagram (Fig. 1), a total of 76
records were initially identified from databases and registers.
Prior to screening, 11 duplicate records were removed. Of the
remaining 65 records that were screened for relevance, 16 were
excluded. Subsequently, all 49 reports sought for retrieval were
successfully retrieved. These 49 reports were then assessed for
eligibility, which resulted in the exclusion of six reports: One
for ineligible study design, four for ineligible outcomes, and one
due to reporting outcomes with odds ratios (OR) only. Consequently,
a total of 42 studies were included in the present systematic
review, encompassing a range of designs, including clinical and
prospective studies (n=12), specialized reviews (n=9), case reports
(n=6), and large cohort/epidemiological studies (n=4). This
methodological approach was selected to systematically map and
synthesize heterogeneous data regarding the association between
chronic liver diseases and ocular surface pathology. Furthermore,
it facilitates a conceptual analysis of clinically-oriented models,
including the ‘ophthalmology-first’ scenario, where ophthalmic
manifestations may precede the diagnosis of underlying hepatic
pathology.
Literature search strategy
A comprehensive literature search was conducted
across the PubMed/MEDLINE, Scopus and Web of Science international
databases (Table I). The present
scoping review included articles published between 2000 and 2025, a
period reflecting the contemporary evolution of the concept of DED
as a systemic phenotype and the emerging understanding of the
‘liver-ocular surface’ axis. The search strategy employed
combinations of keywords and Medical Subject Headings (MeSH) terms,
including: ‘dry eye disease’, ‘ocular surface’, ‘cornea’, ‘liver
disease’, ‘chronic liver disease’, ‘non-alcoholic fatty liver
disease (NAFLD)’, ‘metabolic dysfunction-associated steatotic liver
disease (MASLD)’, ‘cholestasis’, ‘cirrhosis’, ‘vitamin A
deficiency’, ‘meibomian gland dysfunction’ and ‘systemic
inflammation’. In addition, the reference lists of key reviews and
original studies were manually screened to identify relevant
publications that may have been missed during the primary database
search.
 | Table IDetailed literature search
strategy. |
Table I
Detailed literature search
strategy.
| Database | Search query
(search strings) | Filters/limits |
|---|
| PubMed/MEDLINE | (‘Dry Eye
Syndromes’[Mesh] OR ‘Keratoconjunctivitis Sicca’ OR ‘Meibomian
Gland Dysfunction’ OR ‘Ocular Surface’) AND (‘Liver Diseases’[Mesh]
OR ‘Chronic Liver Disease’ OR ‘NAFLD’ OR ‘MASLD’ OR ‘Liver
Cirrhosis’ OR ‘Cholestasis’) | Years: 2000-2025;
Language: English; Species: Humans. |
| Scopus | TITLE-ABS-KEY
[(‘dry eye’ OR ‘ocular surface’ OR ‘meibomian gland’) AND (‘liver
disease’ OR ‘cirrhosis’ OR ‘hepatitis’ OR ‘steatotic liver’)] | Document type:
Article, Review; Language: English. |
| Web of Science | TS=((‘dry eye’ OR
‘keratoconjunctivitis sicca’ OR ‘ocular surface’) AND (‘chronic
liver disease’ OR ‘NAFLD’ OR ‘MASLD’ OR ‘cirrhosis’)) | Timespan:
2000-2025; Language: English. |
Inclusion and exclusion criteria
The present scoping review included peer-reviewed
publications that met the following eligibility criteria: The
inclusion criteria were the following: i) Original clinical
studies, systematic and narrative reviews and scoping reviews; ii)
studies focused on the epidemiology, pathogenesis, or clinical
manifestations of DED and/or ocular surface pathology in patients
with chronic liver diseases; iii) publications in ophthalmology,
hepatology and multidisciplinary scientific journals; iv) articles
published in the English language. The following exclusion criteria
were used: i) Isolated case reports without an analytical
component; ii) experimental studies lacking clinical
interpretation; iii) non-peer-reviewed sources (textbooks, clinical
websites, expert blogs), unless used exclusively to illustrate
well-established clinical context.
Data selection and analysis
process
The selection of publications was conducted in
stages, beginning with the screening of titles and abstracts,
followed by a full-text analysis of articles meeting the inclusion
criteria. Data extracted from the selected publications included
the type of liver disease, ophthalmic manifestations, proposed
pathogenetic mechanisms and clinical phenotypes of DED. Given the
significant heterogeneity in study designs, patient populations,
types of chronic liver diseases and reported ophthalmic outcomes, a
formal systematic review with a meta-analysis was not planned. The
primary objective of the analysis was the qualitative integration
of data and the development of a conceptual model reflecting the
pathophysiological and clinical interconnections of the
‘liver-ocular surface’ axis.
Results
Data synthesis approach
Data synthesis in the present scoping review was
performed narratively, with a focus on integrating
pathophysiological and clinical evidence derived from heterogeneous
sources. The analysis aimed to identify and systematize the key
mechanisms through which chronic liver diseases impact the ocular
surface, including impaired vitamin A metabolism, persistent
systemic inflammation, immune dysregulation, alterations in lipid
metabolism may contribute to meibomian gland dysfunction and
neurosensory disorders (Table
II).
 | Table IISummary of the included studies on
the liver-ocular surface axis. |
Table II
Summary of the included studies on
the liver-ocular surface axis.
|
Category/pathogenetic axis | Study types | Primary findings
and ocular manifestations | (Refs.) |
|---|
| General overview
and epidemiology | Population-based
cohorts, scoping reviews | Prevalence of DED
in liver disease; identification of risk factors and systemic
comorbidities. | (6-8,33,37) |
| Vitamin A and
metabolic axis | Case reports,
clinical reviews | Link between
hepatic Vitamin A storage (Ito cells) and xerophthalmia,
keratomalacia, and goblet cell loss. | (10-13,27) |
|
Immune-mediated/viral hepatitis | Prospective
studies, Multicenter trials | Association of HCV
with Sjögren's-like syndrome and immune-complex-mediated
inflammation. | (18,19,21,22,24-26) |
| Autoimmune liver
diseases (PBC, AIH) | Case-control
studies, clinical reviews | Overlap between
primary biliary cholangitis (PBC) and severe aqueous-deficient DED;
uveitis in AIH. | (27-29,31,32) |
| Dyslipidemia and
meibomian gland dysfunction | Meta-analyses,
cross-sectional studies | Correlation between
serum lipid profiles (cholesterol, triglycerides) and MGD
severity/tear film instability. | (34-36,38-41) |
| Advanced cirrhosis
and neurosensory changes | Prospective pilot
studies, clinical trials | Changes in corneal
endothelial density, microcirculation, and evoked potentials in
decompensated cirrhosis. | (9,14-16) |
Particular attention was given to characterizing the
typical clinical phenotypes of dry eye disease associated with
various types of chronic liver disease, emphasizing their
pathogenetic features, clinical course, and potential resistance to
standard ophthalmic therapies. This phenotypic analysis facilitated
the correlation of ocular manifestations with specific systemic
mechanisms and stages of hepatic dysfunction. Furthermore, the
clinical significance of the identified ophthalmic findings was
analyzed in the context of early detection of subclinical hepatic
pathology. Within this framework, clinical scenarios were evaluated
where atypical or severe DED could serve as a primary indicator of
systemic imbalance, justifying a broader multidisciplinary
evaluation of the patient. To enhance clarity and clinical utility,
the results were structured using tables and conceptual models that
summarize the interconnections between the type of hepatic
pathology, pathophysiological mechanisms, and ophthalmic
phenotypes. This approach was selected to facilitate the
translation of the findings of the review into practical ophthalmic
and multidisciplinary clinical settings.
Discussion
Mechanistic insights into the
‘liver-ocular surface’ axis: Disruption of Vitamin A
Metabolism
The liver serves as the primary reservoir for
Vitamin A, sequestering 70-80% of total body stores within hepatic
stellate cells (Ito cells) as retinal esters. Systemic retinoid
availability is subsequently regulated through the hepatic
synthesis of retinol-binding protein 4 (RBP4) (7,8). In
the retina, vitamin A plays a pivotal role in phototransduction,
the conversion of light into electrical signals. When hepatic
functions are impaired, the storage and transport mechanisms of
vitamin A are compromised, which may contribute to localized
deficiency of retinoid within the eye (10).
In chronic liver diseases, particularly cholestatic
and cirrhotic forms, both the storage and mobilization mechanisms
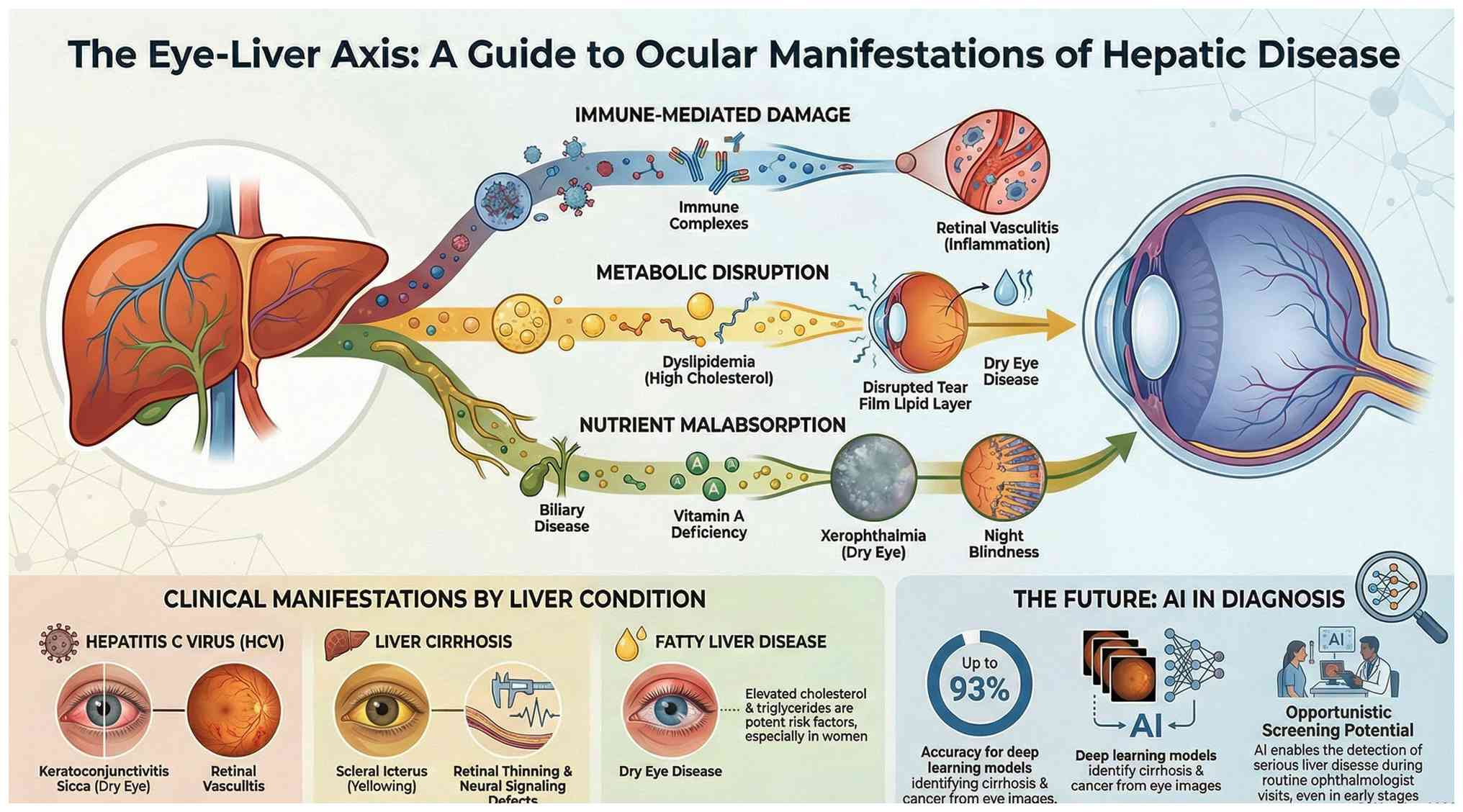
of vitamin A are compromised (Fig.
2). This results in a functional retinoid deficiency that
persists, regardless of adequate dietary intake. This also results
in the development of a functional retinoid deficiency, which
persists even in the presence of adequate dietary intake (8,9).
The pathogenesis of vitamin A deficiency in liver
disease is multifactorial: i) Loss of storage capacity: The
progression of fibrosis activates hepatic stellate cells, leading
to a diminished capacity for retinoid storage (7). ii) Impaired absorption: Cholestasis
reduces the intestinal absorption of fat-soluble vitamins due to
bile acid deficiency (8). iii)
Transport failure: In decompensated cirrhosis, reduced synthesis of
transport proteins (RBP4 and transthyretin) impairs retinoid
delivery to peripheral tissues, including the ocular surface
(9).
At the cellular level, itamin A deficiency disrupts
the differentiation of corneal and conjunctival epithelial cells,
resulting in decreased goblet cell density and reduced mucin
expression (1,3,8).
This mucin-deficient state compromises tear film stability,
rendering the corneal epithelium vulnerable to osmotic and
mechanical stress, which may contribute to the initiation of the
inflammatory cascade characteristic of DED (11). Clinically, these changes manifest
as severe epitheliopathy, filamentary keratitis, and recurrent
corneal erosions (8,9). Prolonged and severe deficiency may
progress to xerophthalmia, featuring squamous metaplasia and
colliquative stromal necrosis. These manifestations are most
pronounced in patients with primary biliary cholangitis and
decompensated cirrhosis, where cholestasis and systemic nutritional
deficits create an unfavorable environment for ocular surface
homeostasis (8,9).
Systemic inflammation and immune
dysregulation
Chronic liver diseases are accompanied by the
persistent activation of innate immunity, characterized by elevated
systemic levels of pro-inflammatory cytokines, including tumor
necrosis factor-α (TNF-α), interleukin (IL)-6 and IL-1β, as well as
the disruption of immune tolerance mechanisms (7,9).
These mediators originate from activated Kupffer cells, monocytes,
and other innate immunity effector cells, sustaining a state of
chronic low-grade inflammation typical of cirrhosis, MASLD and
cholestatic liver diseases (7).
Pro-inflammatory cytokines exert both direct and
indirect effects on ocular surface structures. At the level of
lacrimal glands, they promote acinar cell apoptosis, reduce
secretion of the aqueous component of the tear film, and alter its
protein composition (1,9). Systemic inflammation disrupts the
differentiation and the lipid metabolism of meibocytes in the
meibomian glands, which may contribute to altered meibum quality
and the formation of an unstable lipid layer in the tear film
(1,2). The cytokine-mediated activation of
corneal and conjunctival epithelial cells simultaneously enhances
the expression of adhesion molecules and pro-inflammatory
mediators, forming a self-sustaining inflammatory cascade
characteristic of DED (1,3).
An additional pathogenetic link involves the
dysregulation of the adaptive immune response. In chronic liver
diseases, an imbalance occurs between regulatory T-cells and
effector Th1/Th17 populations, promoting autoimmune activation and
the loss of peripheral tolerance (7,8).
These mechanisms hold particular clinical significance when liver
pathology combines with autoimmune diseases, particularly secondary
Sjögren's syndrome. In secondary Sjögren's syndrome associated with
autoimmune and cholestasis liver diseases (including primary
biliary cholangitis), the lymphocytic infiltration of the lacrimal
glands develops, which may contribute to the progressive
destruction of their secretory apparatus (8,9).
Evidence suggests that these mechanisms may be associated with the
development of an aqueous-deficient DED phenotype. This
manifestation, marked by reduced tear production and inflammation,
appears to be more resistant to standard ophthalmic interventions,
although further prospective studies are required to confirm this
therapeutic resistance (1,9). In clinical practice, such patients
often exhibit refractory DED, necessitating immunomodulatory and
interdisciplinary treatment approaches. Thus, systemic inflammation
and immune dysregulation in chronic liver diseases form the
pathogenetic basis of the inflammatory DED phenotype, exacerbated
by autoimmune comorbidities, underscoring the need to assess
systemic status in patients with severe or atypical ocular surface
disease courses.
Dyslipidemia and meibomian gland
dysfunction
The liver plays a key role in regulating lipid
metabolism, ensuring the synthesis, modification, and clearance of
lipoproteins, fatty acids and cholesterol, while also controlling
systemic lipid homeostasis (7). In
MASLD and other metabolic hepatopathies, a characteristic
dyslipidemia profile develops, including elevated triglyceride
levels, altered low-density to high-density lipoprotein ratios, and
the accumulation of atherogenic and pro-inflammatory lipid
fractions (7,9). These systemic changes affect not only
the vascular bed, but also the lipid composition of secretions from
exocrine glands, including the meibomian glands of the eyelids.
Meibomian glands are specialized sebaceous glands
that secrete a complex mixture of non-polar and polar lipids
(meibum), forming the outer lipid layer of the tear film.
Alterations in systemic lipid metabolism in MASLD may contribute to
disrupted meibocyte differentiation, changes in meibum qualitative
composition, and an increased proportion of saturated and
pro-inflammatory lipids (1,2).
These changes impair the fluidity and spreading of the lipid layer,
reducing its ability to prevent evaporation of the tear film's
aqueous component (1).
An additional pathogenetic factor is the systemic
chronic inflammation characteristic of MASLD, in which
pro-inflammatory cytokines and lipotoxic metabolites exacerbate
meibomian gland dysfunction, contributing to obstruction of the
excretory ducts and formation of subclinical meibomian gland
dysfunction (7). As a result, tear
film instability develops with accelerated evaporation, increased
osmolarity, and secondary activation of the inflammatory cascade on
the ocular surface (1,3).
Thus, in metabolic liver pathology, DED often
manifests as an evaporative phenotype associated with meibomian
gland dysfunction, even in the absence of pronounced local risk
factors such as ophthalmic surgery, contact lens correction, or
adverse environmental conditions (2,7).
This DED phenotype may remain underrecognized in clinical practice,
as classical signs of ocular surface inflammation often combine
with moderate subjective symptoms, underscoring the importance of
considering the patient's systemic metabolic status when evaluating
is associated with of tear film instability.
Neurosensory impairments
According to the neurosensory concept of dry eye
syndrome, chronic inflammation and tear film hyperosmolarity may
contribute to structural and functional damage of corneal afferent
nerve endings, the disruption of their excitability, and altered
central processing of sensory signals (1,2).
These processes are accompanied by a reduction in the density of
the subbasal nerve plexus, altered expression of neurotrophic
factors, and the dysregulation of the ‘cornea-lacrimal gland’
reflex arc, which exacerbates tear film instability and perpetuates
the vicious cycle of DED (1). In
chronic liver diseases, neurosensory impairments may be exacerbated
by systemic neurometabolic mechanisms. Liver dysfunction is
accompanied by the accumulation of neurotoxic metabolites
(including ammonia and aromatic amino acids), the disruption of
trace element metabolism, and altered neurotransmitter balance,
which underlie hepatic encephalopathy, including its subclinical
forms (7,9). Even in the absence of overt
neurological symptoms, these changes can reduce the sensitivity of
peripheral nerve endings, including corneal afferents, and disrupt
sensory integration (7).
An additional factor is systemic inflammation
characteristic of chronic liver diseases, in which pro-inflammatory
cytokines (TNF-α and IL-6) exert neurotoxic effects, contributing
to dysfunction of peripheral and central sensory pathways (7). Collectively, these mechanisms may
contribute to reduced corneal sensitivity and altered perception of
painful and discomforting stimuli from the ocular surface (1,9).
Clinically, these pathogenetic processes manifest as
DED phenotypes where pronounced corneal epithelial changes and
severe tear film instability combine with minimal or atypical
subjective symptoms (1,2). Such a dissonance between objective
findings and patient complaints represents an important diagnostic
‘red flag’ for the ophthalmologist and may indicate the presence of
systemic neurometabolic disorders, including previously undiagnosed
liver dysfunction (7). Recognizing
this phenotype holds particular significance within the
‘ophthalmology-first’ concept, as it enables viewing reduced
corneal sensitivity and neurosensory dysfunction as potential
indicators of subclinical liver damage.
Clinical phenotypes of ocular
manifestations in liver diseases
Ocular manifestations frequently accompany various
metabolic disorders. Despite comprehensive biochemical, molecular
and metabolic insights into many inherited metabolic disorders,
their pathogenesis (particularly the link between systemic
metabolic dysfunction and ocular pathology) remains poorly
understood. Eye abnormalities may arise from direct toxicity of
aberrant metabolites, accumulation due to synthetic pathway
defects, or impaired energy metabolism (12). Chronic ocular surface desiccation
may contribute to a spectrum of debilitating symptoms, ranging from
foreign body sensation and blurred vision to sight-threatening
complications such as epithelial erosions, corneal ulceration, and
secondary infections (Table
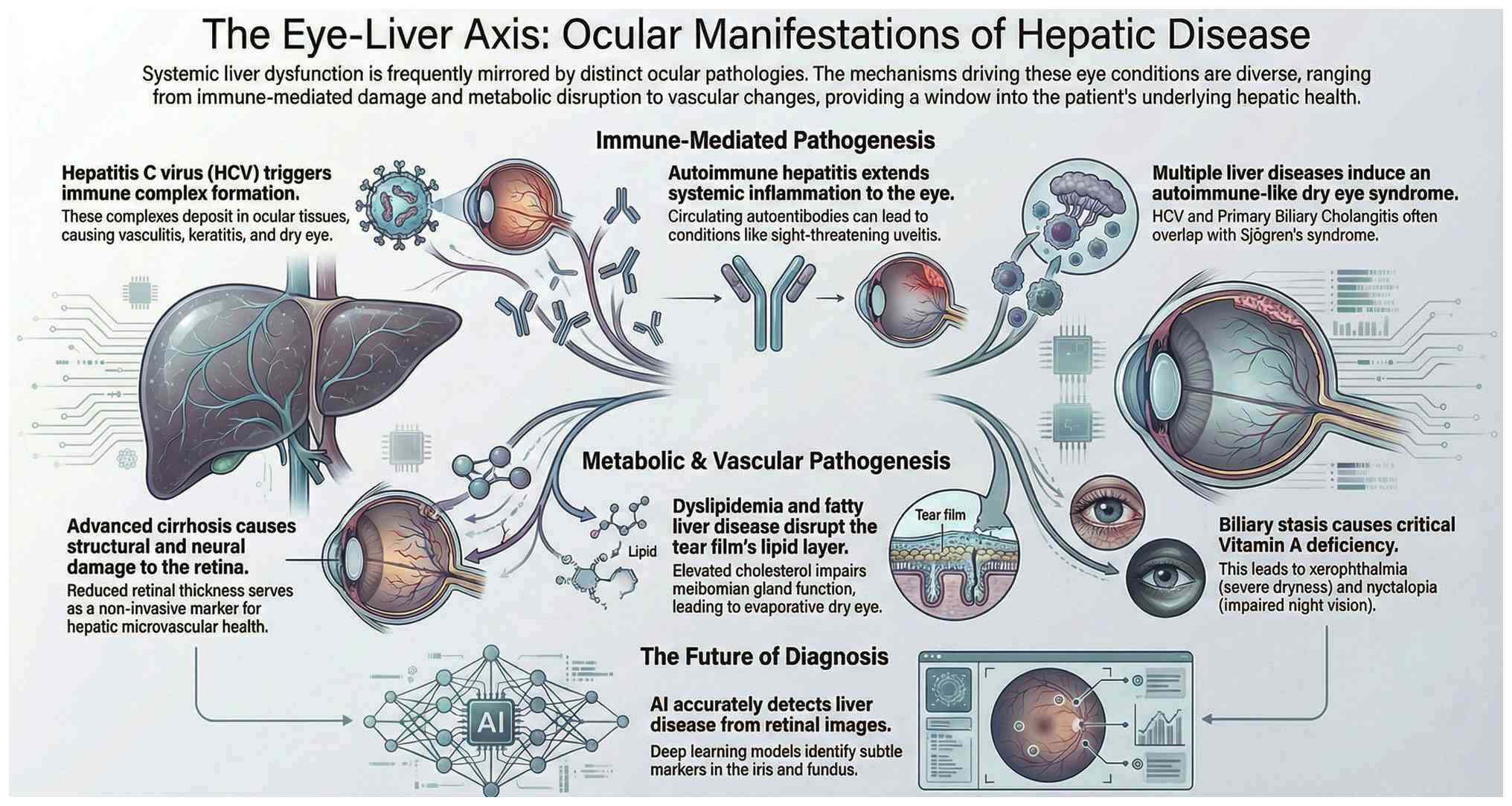
III). Consequently, the presence of keratoconjunctivitis sicca
necessitates a comprehensive systemic evaluation to rule out
Sjögren's syndrome and chronic liver disease (Fig. 3). While conservative management
with lubricants may provide symptomatic relief, persistent or
severe cases require urgent referral to an ophthalmologist for
specialized intervention (13).
 | Table IIIAssociation between hepatic
pathology, ocular manifestations and pathophysiological
mechanisms. |
Table III
Association between hepatic
pathology, ocular manifestations and pathophysiological
mechanisms.
| Hepatic
pathology | Ocular
manifestations | Pathophysiological
mechanisms | (Refs.) |
|---|
| Hepatitis C
virus |
Keratoconjunctivitis sicca (Dry Eye
syndrome), retinal vasculitis, ischemic retinopathy, ‘cotton-wool’
spots. | Deposition of
circulating immune complexes, systemic immune activation, and
cryoglobulinemia. | (19,21,22,24,26) |
| Autoimmune
hepatitis | Uveitis,
episcleritis, decreased basal tear secretion (Sjögren's-like
syndrome). | Loss of systemic
self-tolerance; autoantibody-mediated attack on exocrine glands and
the uveal tract. | (18,29,31,32) |
| Liver
cirrhosis | Eyelid
xanthelasmas, conjunctival icterus, fundus vascular remodeling,
delayed visual evoked potentials. | Dyslipidemia,
portal hypertension, and accumulation of systemic neurotoxins
(e.g., ammonia). | (9,14-16) |
| Liver cancer | Iris morphological
shifts, retinal microvascular alterations (detected via AI-driven
models). | Neoplastic toxemia,
systemic metabolic shifts, and altered paraneoplastic
angiogenesis. | (7,17) |
| Minimal hepatic
encephalopathy | Impaired contrast
sensitivity, subclinical neurophysiological deficits (mfVEP
latency). | Astrocyte swelling
due to hyperammonemia; disruption of neurotransmission in the
visual cortex. | (16) |
| Non-alcoholic fatty
liver disease/metabolic dysfunction-associated steatotic liver
disease | Early-stage iris
pigmentation changes, subtle retinal microvascular anomalies. | Chronic low-grade
systemic inflammation and increased oxidative stress. | (7,34,35,38) |
| Cholestatic
disorders (e.g., primary biliary cholangitis) | Xerophthalmia,
nyctalopia (night blindness), Bitot's spots. | Malabsorption of
bile-dependent fat-soluble vitamins (specifically vitamin A
deficiency). | (8,11,27,28) |
Liver cirrhosis and ocular
manifestations
Due to the observed correlations between ocular
morphometry and liver function, chorioretinal parameters can be
utilized as non-invasive indicators of hepatic microvascular
health. Specifically, significant declines in macular volume and
retinal thickness have been associated with advancing cirrhosis
across diverse etiologies (14).
In patients with cirrhosis, regardless of the underlying etiology,
whether viral, alcoholic, or related to non-alcoholic
steatohepatitis, the clinical appearance of scleral icterus serves
as a critical indicator of advanced hepatic dysfunction or an
acute-on-chronic failure (15).
Detecting minimal hepatic encephalopathy in patients with
compensated cirrhosis is vital for preventing further neurological
deterioration. Research indicates that visual electrophysiology can
reveal subtle abnormalities in neural signaling before they
manifest as global cognitive impairment. These findings suggest
that patients with cirrhosis, even those without overt
encephalopathy, may harbor underlying visual-cortical dysfunction.
Using the visual system as a diagnostic proxy allows for a more
objective and early identification of patients at risk,
significantly outperforming traditional associative learning or
visual retention tests (16).
Viral hepatitis and ocular
manifestations
While chronic viral and autoimmune hepatitis are
associated with various ocular findings, these manifestations are
typically non-specific or relatively rare (17). By contrast, hepatitis B virus lacks
distinctive ophthalmic features, with ocular involvement generally
limited to complications arising from secondary cirrhosis (18). Hepatitis C virus (HCV) has been
specifically linked to dry eye syndrome (19), particularly in patients with
concurrent autoimmune disorders. Furthermore, during the era of
interferon-based therapies, a transient form of retinopathy
characterized by the presence of cotton wool spots were frequently
observed as a treatment-related side-effect (20).
The ophthalmic spectrum of HCV infection is diverse,
encompassing conditions, such as retinal vasculitis,
keratoconjunctivitis sicca, DED, keratitis, scleritis and various
retinopathies. The pathogenesis of these manifestations is
primarily attributed to a heightened immune response against HCV
antigens, may contribute to the formation and systemic deposition
of circulating immune complexes. Furthermore, the persistent
presence of HCV can act as a warrant consideration for broader
autoimmune dysregulation, precipitating secondary ocular
pathologies that mirror other systemic autoimmune syndromes
(21). Current evidence points
toward a possible relationship between ocular surface pathology and
systemic factors such as immune complex deposition or HCV-related
autoimmunity. However, given the cross-sectional nature of
available data, these processes should be viewed as contributing
factors rather than confirmed direct association. Clinical
observations support this, demonstrating retinal changes such as
soft exudates and peripheral white dots. Moreover, specular
microscopy reveals that advanced cirrhosis negatively impacts the
ocular surface, specifically resulting in a diminished corneal
epithelial cell count (21).
In patients with HCV infection, microvascular
alterations within the lacrimal gland are linked to glandular
dysfunction. This condition is characterized by increased tear film
osmolarity and a notable reduction in corneal sensitivity.
Clinically, patients with HCV frequently report subjective
symptoms, such as itching and burning sensations. Comparative
analyses have demonstrated that tear break-up time values are
significantly lower in HCV-positive individuals than in healthy
controls, reflecting the higher prevalence and severity of dry eye
symptoms in this population (22).
While pathognomonic ocular indicators of HCV
infection remain unidentified, various syndromes have been
documented in the literature. The most frequent clinical
presentations include ischemic retinopathy, primarily driven by
HCV-induced vasculitis and keratoconjunctivitis sicca. Furthermore,
although the ocular region (specifically the conjunctiva, lacrimal
gland and orbital soft tissues) is a prevalent site for extra-nodal
lymphomas, the overall incidence of HCV-associated ocular adnexal
lymphoma remains low, with only limited studies investigating this
association (23). In a
large-scale prospective study, Cacoub et al (24) evaluated 321 patients with chronic
HCV infection and found that extrahepatic manifestations were
present in 38% of the cohort. Notably, 10% of these patients
presented with xerophthalmia (dry eye) and 12% with xerostomia (dry
mouth), underscoring the significant prevalence of sicca syndrome
in this population (24). HCV
infection frequently overlaps with the clinical presentation of
Sjögren's syndrome, a chronic autoimmune condition primarily
affecting the salivary and lacrimal glands. The resulting dryness
of the oral and ocular mucosae in patients with HCV underscores the
role of the virus in driving systemic autoimmunity, necessitating a
careful differential diagnosis between idiopathic Sjögren's and
HCV-induced sicca syndrome (25).
Consequently, as HCV infection advances, routine screening for
ocular abnormalities becomes imperative. Manifestations, such as
ocular surface damage and DED demonstrate a positive association
with the progression of hepatic fibrosis, suggesting that the
severity of liver scarring may serve as a clinical predictor for
the worsening of sicca symptoms (26).
Primary biliary cholangitis and ocular
manifestations
Chronic biliary stasis in primary biliary
cholangitis frequently results in the malabsorption of lipophilic
nutrients. The subsequent deficiency in vitamin A is a critical
factor in the development of xerophthalmia and impaired dark
adaptation (nyctalopia). When combined with the progression to
cirrhosis and portal hypertension, these metabolic disturbances
exacerbate the damage to the ocular surface and visual function
(27). In the context of primary
biliary cholangitis (PBC), chronic cholestasis-induced
hyperlipidemia often manifests dermatologically as eyelid
xanthelasmas. Although benign, these lesions are strong clinical
indicators of biliary pathology when observed alongside systemic
symptoms like pruritus. Additionally, the high prevalence of dry
eye symptoms (sicca syndrome) in patients with PBC highlights a
frequent autoimmune overlap with Sjögren's syndrome, necessitating
a multidisciplinary diagnostic approach (28).
Autoimmune hepatitis and ocular
manifestations
Characterized by high titers of circulating
autoantibodies, autoimmune hepatitis is associated with significant
extrahepatic involvement, including various ophthalmic conditions.
This association necessitates regular ophthalmological surveillance
for patients diagnosed with autoimmune hepatitis to ensure the
early detection of immune-mediated ocular surface and intraocular
diseases (29,30). The clinical data provided by
Citirik et al (31)
highlight that patients with autoimmune liver disease frequently
exhibit impaired basal tear production and diminished tear film
break-up time. These objective indicators of ocular surface
instability, coupled with reported dry eye symptoms, underscore the
necessity for systematic ophthalmic assessment in the management of
autoimmune hepatitis (31). While
comprehensive literature regarding the ophthalmic manifestations of
autoimmune hepatitis remains limited, a growing body of evidence
from clinical case reports suggests a significant association
between the two (31,32). These preliminary findings indicate
that autoimmune hepatitis may present with a broader spectrum of
ocular involvement than previously recognized, necessitating
further prospective studies to establish definitive prevalence and
pathophysiology. Furthermore, Romanelli et al (32) documented a significant clinical
association between autoimmune hepatitis and uveitis. This suggests
that the systemic inflammatory process characteristic of autoimmune
hepatitis can extend to the uveal tract, potentially resulting in
sight-threatening intraocular inflammation that requires
coordinated immunosuppressive management (32).
Fatty liver disease and ocular
manifestations
In a large-scale nationwide survey involving 17,364
Korean adults, researchers identified a significant association
between DED and systemic metabolic health (33). That study revealed that patients
with DED exhibited markedly higher rates of systemic comorbidities;
specifically, dyslipidemia was established as a potent risk factor,
with an adjusted odds ratio of 1.63. These findings suggest that
ocular surface stability is closely intertwined with lipid
metabolism and systemic inflammatory status (33). The association between dyslipidemia
and DED is robustly supported by large-scale epidemiological data.
In addition to nationwide surveys, specific studies involving 5,627
Korean women (34) and 15,294
Korean adults (35) have
consistently demonstrated that elevated serum cholesterol and
triglyceride levels are associated with an increased prevalence of
DED. This suggests that lipid imbalances may compromise the
meibomian gland secretions, may contribute to evaporative tear film
instability.
Research involving adults aged 25-70 years has
identified the female sex as a significant risk factor for DED
(36). The increased prevalence in
this group underscores the necessity for sex-stratified screening,
particularly when assessing extrahepatic manifestations in women
with chronic liver disease or metabolic syndrome (36). A previous retrospective analysis of
306 patients (ages 18-87 years) further corroborated these
findings, demonstrating a significantly elevated incidence of DED
among women >40 years of age and individuals with concurrent
dyslipidemia. These data suggest a critical intersection between
age-related hormonal shifts and lipid metabolic dysfunction, both
of which appear to act as compounding risk factors for ocular
surface deterioration (37).
Another critical limitation concerns the geographic
and ethnic concentration of the current evidence base. A
significant portion of the large-scale epidemiological data linking
dyslipidemia to DED is derived from East Asian populations
(33-35)
(e.g., the Korean National Health and Nutrition Examination Survey)
and specific European cohorts (37) (e.g., the Dutch Lifelines study).
Geographic variations in climate, air quality and dietary habits,
alongside ethnic differences in the genetic susceptibility to
metabolic syndrome and MASLD, may influence the strength of the
liver-ocular surface association. Consequently, the findings
presented herein should be interpreted with caution when applied to
other populations, such as African or Hispanic cohorts, where
longitudinal data on this specific axis remain limited.
Evidence from systematic reviews and meta-analyses
has consolidated the link between dyslipidemia and ocular health,
establishing that elevated levels of total cholesterol, low-density
lipoprotein cholesterol and high-density lipoprotein cholesterol
are associated with an increased risk of DED (38). This association is particularly
pronounced in female populations (39). Furthermore, cross-sectional data
suggest that abnormal total cholesterol, low-density lipoprotein
cholesterol and high-density lipoprotein cholesterol, and
triglyceride profiles contribute to the progression of DED. The
underlying pathophysiology likely involves impaired tear film
stability and meibomian gland dysfunction, where systemic lipid
imbalances disrupt the lipid layer of the tear film (40). Evidence suggests that
lipoprotein-mediated pathways link systemic lipid dysregulation to
DED. While early ophthalmic screening for patients with
dyslipidemia offers a pathway to mitigate ocular morbidity, further
research is required to map the exact mechanistic intersections
between these conditions (41).
Furthermore, the role of lipid-lowering therapies, such as statins,
as a treatment modality for DED remains largely unexplored. Future
clinical investigations are warranted to determine if modulating
systemic lipids can serve as a primary or adjunctive therapy for
alleviating DED.
The ‘ophthalmology-first’ concept
The majority of published studies focus on analyzing
ophthalmic manifestations in patients with an already established
diagnosis of chronic liver disease, where ocular symptoms are
considered one of the systemic complications of the primary process
(7-9).
In such studies, DED, corneal epitheliopathies, and tear film
instability are typically interpreted as consequences of pronounced
metabolic, inflammatory, or autoimmune liver dysfunction. However,
accumulated clinical observations and epidemiological data indicate
that in some patients, atypical or refractory DED course may
precede the detection of liver pathology and serve as the first
clinical manifestation of subclinical liver dysfunction (6,7).
In particular, DED phenotypes have been described
that are characterized by disproportionately severe epithelial
changes despite minimal local risk factors, reduced corneal
sensitivity, and weak or paradoxical subjective symptoms, which do
not fit into classical models of ocular surface disease (1,2).
Such clinical scenarios may reflect systemic pathogenetic
processes; disruptions in vitamin A metabolism, dyslipidemia,
chronic systemic inflammation and neurosensory
dysfunction-associated with the early stages of MASLD, cholestasis
diseases, or other forms of chronic liver disease (7,9).
Evidence suggests that refractory DED and reduced
corneal sensitivity may be associated with extrahepatic
manifestations. Therefore, in cases where local associations are
insufficient to explain the clinical picture, clinicians may
consider a systemic evaluation as part of a comprehensive
diagnostic approach, acknowledging the associative nature of the
current evidence (1,7). Such an approach includes minimal
screening of liver function through assessment of biochemical
markers, metabolic profile and risk factors for chronic
hepatopathies, which is particularly relevant in preoperative
ophthalmologic practice and in managing patients with atypical DED
forms.
The physiological and pathological links between the
liver and eye are profound, with various ocular manifestations
often serving as the first clinical indicators of underlying
hepatic dysfunction. These ophthalmic signs, spanning viral,
congenital and autoimmune etiologies, are instrumental in the early
detection of liver disease. Facilitating such timely diagnosis is
paramount, as it enables the implementation of early intervention
and targeted management strategies, ultimately optimizing patient
prognosis and long-term clinical outcomes (42). The ‘ophthalmology-first’ approach
emphasizes the associative link between the ocular surface and
systemic health. Rather than acting as a definitive diagnostic
tool, atypical ophthalmic manifestations may represent subclinical
signals that warrant consideration for a multidisciplinary workup,
particularly when conventional dry eye therapies yield suboptimal
results (7,8).
Implementation and clinical
contextualization
To operationalize the ‘ophthalmology-first’ concept,
specific clinical scenarios were identified where DED
manifestations may justify further systemic investigation (Table IV). These include the following:
i) Phenotypic discordance: Severe aqueous-deficient DED
disproportionate to the age of a patient, medication profile, or
environmental risk factors; ii) refractory course: DED that remains
non-responsive to optimized Tier 1 and Tier 2 therapies for a
period of 3-6 months in the absence of Sjögren's syndrome; iii)
concurrent neurotrophic signs: Reduced corneal sensitivity or
persistent epithelial defects without traditional local
neurotrophic triggers.
 | Table IVClinical phenotypes and suggested
multidisciplinary diagnostic pathways. |
Table IV
Clinical phenotypes and suggested
multidisciplinary diagnostic pathways.
| Ocular clinical
presentation | Suspected
systemic/hepatic association | Recommended initial
diagnostic action | (Refs.) |
|---|
| Severe
aqueous-deficient DED (disproportionate to local risk factors,
e.g., age, screen time) | Autoimmune liver
diseases (e.g., primary biliary cholangitis, autoimmune
hepatitis) | Referral for
autoimmune markers (ANA, AMA, ASMA) and liver transaminases
(ALT/AST). | (18,27,28,31,32) |
| Refractory MGD and
tear film instability (resistant to standard eyelid hygiene and
lubricants) | Metabolic
dysfunction-associated steatotic liver disease (MASLD/NAFLD) | Comprehensive serum
lipid profile (TC, LDL-C, TG) and hepatic
ultrasound/FibroScan. | (34,38-41) |
| Diminished corneal
sensitivity (in the absence of contact lens wear or ocular
surgery) | Chronic cholestasis
or severe cirrhosis (leading to secondary vitamin A
deficiency) | Serum retinol
(vitamin A) levels and assessment of biliary markers (GGT, alkaline
phosphatase). | (8,10,11,13,27) |
| Persistent corneal
epitheliopathy (slow healing without local neurotrophic
triggers) | Advanced hepatic
fibrosis/systemic metabolic dysregulation | Evaluation of
albumin levels and systemic inflammatory markers (CRP) to assess
liver synthetic function. | (7,9,14,15) |
It should be noted that this screening approach is
recommendation-based and context-dependent. Its implementation
should be tailored to local clinical practice settings, specialist
availability, and patient risk profiles. In appropriate settings,
an ophthalmologist may consider initiating a ‘liver-surface axis’
evaluation by requesting a baseline metabolic and hepatic profile,
focusing on serum cholesterol, triglycerides, and liver
transaminases, particularly in patients >40 years of age with
concurrent dyslipidemia. This proactive approach aims to facilitate
early interdisciplinary dialogue rather than replacing formal
hepatological diagnostic protocols.
Operationalizing the
‘ophthalmology-first’ hypothesis
Given the current evidence, it is suggested that
certain DED phenotypes serve as clinical cues for systemic
investigation. This approach is intended as a hypothesis-generating
framework rather than a formal clinical guideline.
High-suspicion phenotypes. i) The ‘metabolic’
phenotype: Severe meibomian gland dysfunction and tear film
instability in patients with existing dyslipidemia, potentially
signaling MASLD; ii) the ‘autoimmune’ phenotype: Profound aqueous
deficiency (Schirmer <5 mm) in middle-aged women, suggesting a
link to PBC or autoimmune hepatitis; iii) the ‘neuro-nutritional’
phenotype: Reduced corneal sensitivity and persistent epithelial
defects, which may be associated with vitamin A malabsorption in
chronic cholestasis.
Minimalist screening panel. When these
phenotypes are encountered without obvious ocular causes, a
baseline systemic evaluation is justified: i) Liver function tests:
Alanine transaminase, aspartate aminotransferase, gamma-glutamyl
transferase and alkaline phosphatase to assess hepatocyte integrity
and cholestasis; ii) lipid profile: Total cholesterol, low-density
lipoprotein and triglycerides; iii) optional: Serum retinol levels
in the event that nutritional deficiency is suspected.
Priority populations. Screening should be
prioritized in: i) Patients with refractory DED (unresponsive to
standard therapy for >6 months); ii) patients with concurrent
metabolic syndrome (obesity and/or hypertension); iii) female
patients aged 40-70 years presenting with new-onset severe sicca
symptoms.
Current evidence gaps and research
perspectives
The current evidence base is characterized by a
predominance of associative and predominantly cross-sectional
studies (2,3,38),
along with significant heterogeneity in the ophthalmological
protocols and diagnostic criteria for dry eye syndrome used. The
absence of standardized approaches to ocular surface assessment and
the limited number of prospective studies complicate establishing
causal associations between chronic liver diseases and the
development of DED ophthalmic phenotypes. In this regard, promising
directions for future research include prospective cohort projects
with an ophthalmological entry point, as well as the development
and validation of objective bioindicators, including the use of
artificial intelligence technologies for analyzing images of the
ocular surface and anterior eye segment as potential indicators of
systemic pathology.
Methodology note
Only peer-reviewed scientific publications from
ophthalmological, hepatological, and interdisciplinary journals
were included in the evidence section of the present scoping
review, used to form conclusions on the epidemiology, pathogenesis,
and clinical phenotypes of dry eye syndrome in liver diseases. This
approach was selected to ensure methodological rigor and
reproducibility of the findings.
Resource classification
Individual clinical-information and educational
resources (including MSD Manual, NHS, Mayo Clinic, Ophthalmology
Advisor and similar sources) were placed in a non-numbered
‘Additional Clinical-Information Resources’ section and used solely
for illustrating clinical context and describing well-known
clinical syndromes. In accordance with the International Committee
of Medical Journal Editors (ICMJE) recommendations and EQUATOR
Network standards, these resources were not considered sources of
scientific evidence and were not used in forming the key analytical
conclusions of the review.
Limitations
Despite the significant correlations identified in
this review, several limitations should be acknowledged. First, the
majority of the included clinical evidence is derived from
cross-sectional or retrospective studies. While these data
establish a robust association between chronic liver diseases and
ocular surface pathology, their inherent design limits the ability
to confirm a definitive causal relationship or to establish a
precise temporal sequence of events. For instance, although a
strong correlation exists between systemic dyslipidemia and
meibomian gland dysfunction, it remains unclear whether lipid
imbalances serve as the primary driver of ocular surface
deterioration or represent a concurrent manifestation of a broader
metabolic syndrome.
Furthermore, there is a notable lack of large-scale,
prospective longitudinal studies that track the progression of DED
in parallel with the stages of hepatic dysfunction. The
heterogeneity in study populations, diagnostic criteria for DED,
and the varying etiologies of liver disease (viral vs. metabolic
vs. autoimmune) further complicate the generalization of these
findings. Finally, while emerging AI-based models show promise in
identifying hepatobiliary pathologies through ocular bioindicators,
these tools require rigorous external validation in diverse
clinical settings before they can be integrated into standard
diagnostic protocols. Future research should prioritize
longitudinal cohorts to transition from identifying associations to
validating the ‘ophthalmology-first’ concept as a reliable clinical
pathway.
Conclusion
Current Evidence and Mechanistic Insights into
Systemic Associations data suggest that dry eye syndrome and
corneal epithelial diseases may be more than localized
ophthalmological conditions. Disruptions in ocular surface
homeostasis often correlate with systemic metabolic, immune, and
neurosensory processes, within which the liver is hypothesized to
play a significant regulatory role. The ‘liver-ocular surface’ axis
concept serves as a conceptual model to integrate disparate
clinical observations into a framework that may explain various DED
phenotypes observed in patients with chronic liver diseases.
Key pathogenetic mechanisms
The present scoping review identifies several
pathways through which liver dysfunction is associated with ocular
surface changes, including disruptions in vitamin A metabolism,
systemic inflammation, immune dysregulation, and
dyslipidemia-related meibomian gland dysfunction. These pathways
are closely linked to DED clinical phenotypes that frequently
present with atypical or refractory courses, potentially diverging
from traditional localized disease models.
Clinical implications and the
‘ophthalmology-first’ concept
The ‘ophthalmology-first’ concept proposes that
severe or atypical dry eye syndrome, persistent corneal
epitheliopathies, and reduced corneal sensitivity may serve as
potential clinical indicators of undiagnosed hepatic dysfunction.
Rather than acting as definitive diagnostic markers, these findings
suggest the utility of an expanded diagnostic mindset and emphasize
the importance of interdisciplinary collaboration, particularly
when managing patients with DED that is disproportionate to local
risk factors.
Practical significance
Recognizing the associative nature of DED and
chronic liver disease provides a basis for improved risk
stratification and personalized management. Incorporating a
systemic (including hepatic) assessment into examination algorithms
for patients with severe or atypical DED might facilitate the
earlier detection of occult somatic pathology, although these
integrated diagnostic pathways require further clinical
validation.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
AK, AS and DK were involved in the writing,
reviewing and editing of the manuscript, as well as in the writing
and preparation of the original draft of the manuscript, and the
conceptualization of the study. AB, NK and AA supervised the study,
and were also involved in project administration, in the literature
search and in the conceptualization of the study. All authors have
read and approved the final manuscript. AK and AS confirm the
authenticity of all the raw data.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Use of artificial intelligence tools
During the preparation of this work, AI tools were
used to improve the readability and language of the manuscript or
to generate images, and subsequently, the authors revised and
edited the content produced by the AI tools as necessary, taking
full responsibility for the ultimate content of the present
manuscript.
References
|
1
|
Craig JP, Nichols KK, Akpek EK, Caffery B,
Dua HS, Joo CK, Liu Z, Nelson JD, Nichols JJ, Tsubota K and
Stapleton F: TFOS DEWS II definition and classification report.
Ocul Surf. 15:276–283. 2017.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Wolffsohn JS, Arita R, Chalmers R,
Djalilian A, Dogru M, Dumbleton K, Gupta PK, Karpecki P, Lazreg S,
Pult H, et al: TFOS DEWS II diagnostic methodology report. Ocul
Surf. 15:539–574. 2017.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Stapleton F, Alves M, Bunya VY, Jalbert I,
Lekhanont K, Malet F, Na KS, Schaumberg D, Uchino M, Vehof J, et
al: TFOS DEWS II epidemiology report. Ocul Surf. 15:334–365.
2017.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Zemanová M: Dry eye disease. A review.
Cesk Slov Oftalmol. 77:107–119. 2021.PubMed/NCBI View
Article : Google Scholar
|
|
5
|
Harrell CR, Feulner L, Djonov V, Pavlovic
D and Volarevic V: The molecular mechanisms responsible for tear
hyperosmolarity-induced pathological changes in the eyes of dry eye
disease patients. Cells. 12(2755)2023.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Vehof J, Snieder H, Jansonius N and
Hammond CJ: Prevalence and risk factors of dry eye in 79,866
participants of the population-based Lifelines cohort study in the
Netherlands. Ocul Surf. 19:83–93. 2021.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Wu J, Duan C, Yang Y, Wang Z, Tan C, Han C
and Hou X: Insights into the liver-eyes connections, from
epidemiological, mechanical studies to clinical translation. J
Transl Med. 21(712)2023.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Patel R, Nair S, Choudhry H, Jaffry M and
Dastjerdi M: Ocular manifestations of liver disease: An important
diagnostic aid. Int Ophthalmol. 44(177)2024.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Rishi A, Ashish J, Rashmi K and Sushil F:
Ocular Manifestations and Endothelial cell density in Chronic Liver
Disease Patients and its Co-relation with Severity of Liver
Disease. Trop Gastroenterol. 46:71–76. 2025.
|
|
10
|
Sajovic J, Meglič A, Glavač D, Markelj Š,
Hawlina M and Fakin A: The role of vitamin A in retinal diseases.
Int J Mol Sci. 23(1014)2022.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Kopecký A, Benda F and Němčanský J:
Xerosis in patient with vitamin A deficiency-a case report. Cesk
Slov Oftalmol. 73:222–224. 2018.PubMed/NCBI
|
|
12
|
Rajappa M, Goyal A and Kaur J: Inherited
metabolic disorders involving the eye: A clinico-biochemical
perspective. Eye (Lond). 24:507–518. 2010.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Prasad D and Bhriguvanshi A: Ocular
manifestations of liver disease in children: Clinical aspects and
implications. Ann Hepatol. 19:608–613. 2020.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Gifford FJ, Moroni F, Farrah TE,
Hetherington K, MacGillivray TJ, Hayes PC, Dhaun N and Fallowfield
JA: The eye as a non-invasive window to the microcirculation in
liver cirrhosis: A prospective pilot study. J Clin Med.
9(3332)2020.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Fortea JI, Carrera IG, Puente A and Crespo
J: Hepatic cirrhosis. Medicine (Barcelona). 13:297–307. 2020.
|
|
16
|
Sabry RM, Hamad O, Khalil HEM, Mohammed
SI, Eid RA and Hosny H: The role of multifocal visual evoked
potential in detection of minimal hepatic encephalopathy in
patients with compensated liver cirrhosis. BMC Neurol.
25(45)2025.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Parmar UPS, Morya AK, Gupta PC, Arora A
and Verma N: Role of artificial intelligence-based ocular
biomarkers in hepatobiliary diseases: A scoping review. World J
Hepatol. 17(109801)2025.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Alshahrani S, Aljumah AA and Alluhaidan A:
Uveitis and autoimmune hepatitis, a real entity? A case report with
review of the literature. Saudi J Ophthalmol. 35:73–77.
2021.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Jacobi C, Wenkel H, Jacobi A, Korn K,
Cursiefen C and Kruse FE: Hepatitis C and ocular surface disease.
Am J Ophthalmol. 144:705–711. 2007.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Abd El-Badie Mohamed M and Abd-El Azeem
Eed K: Retinopathy associated with interferon therapy in patients
with hepatitis C virus. Clin Ophthalmol. 6:1341–1345.
2012.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Bertrand RHC, Bertrand ALX, Gomes TM and
Ferreira ASP: An eye on hepatitis C: A review. Arq Bras Oftalmol.
82:161–167. 2019.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Iqbal S, Ahmed I and Azam A: Proportion of
dry eye in hepatitis C patients. Pak J Ophthalmol. 34:286–289.
2018.
|
|
23
|
Fang SL, Teo L, Loo CY, Li JW and Santosa
A: Lessons of the month 2: Ocular manifestations and complications
of hepatitis C infection. Clin Med (Lond). 21:e417–e419.
2021.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Cacoub P, Renou C, Rosenthal E, Cohen P,
Loury I, Loustaud-Ratti V, Yamamoto AM, Camproux AC, Hausfater P,
Musset L, et al: Extrahepatic manifestations associated with
hepatitis C virus infection. A prospective multicenter study of 321
patients. The GERMIVIC. Groupe d'Etude et de Recherche en Medecine
Interne et Maladies Infectieuses sur le Virus de l'Hepatite C.
Medicine (Baltimore). 79:47–56. 2000.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Yeh CC, Wang WC, Wu CS, Sung FC, Su CT,
Shieh YH, Chang SN and Su FH: Association of Sjögrens Syndrome in
patients with chronic hepatitis virus infection: A population-based
analysis. PLoS One. 11(e0161958)2016.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Gumus K, Yurci A, Mirza E, Arda H, Oner A,
Topaktas D and Karakucuk S: Evaluation of ocular surface damage and
dry eye status in chronic hepatitis C at different stages of
hepatic fibrosis. Cornea. 28:997–1002. 2009.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Martín LL, Rocha-de-Lossada C,
Marín-Martínez S and Peraza-Nieves JE: Sterile, recurrent, and
bilateral corneal perforation related to primary biliary cirrhosis
complicated by secondary Sjögren syndrome and vitamin A deficiency.
Arq Bras Oftalmol. 84:606–609. 2021.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Selmi C and Gershwin ME: Chronic
autoimmune epithelitis in Sjögren's Syndrome and primary biliary
cholangitis: A comprehensive review. Rheumatol Ther. 4:263–279.
2017.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Efe C, Ozaslan E, Purnak T, Ozbalkan Z and
Altiparmak E: Uveitis in patients with autoimmune hepatitis. Am J
Ophthalmol. 149:684–685; author reply 685. 2010.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Linzay CD, Sharma B and Pandit S:
Autoimmune Hepatitis. In: StatPearls [Internet]. StatPearls
Publishing, Treasure Island, FL, 2026.
|
|
31
|
In: StatPearls [Internet]. Treasure Island
(FL). StatPearls Publishing; 2026. Citirik M, Berker N, Kacar S and
Kekilli M: Ocular findings in patients with autoimmune liver
disease. Ocul Immunol Inflamm. 20:438–442. 2012.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Romanelli RG, La Villa G, Almerigogna F,
Vizzutti F, Di Pietro E, Fedi V, Gentilini P and Laffi G: Uveitis
in autoimmune hepatitis: A case report. World J Gastroenterol.
12:1637–1640. 2006.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Roh HC, Lee JK, Kim M, Oh JH, Chang MW,
Chuck RS and Park CY: Systemic comorbidities of dry eye Syndrome:
The Korean National health and nutrition examination survey V, 2010
to 2012. Cornea. 35:187–192. 2016.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Chun YH, Kim HR, Han K, Park YG, Song HJ
and Na KS: Total cholesterol and lipoprotein composition are
associated with dry eye disease in Korean women. Lipids Health Dis.
12(84)2013.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Park HW and Park JW: The Association
between symptoms of dry eye Syndrome and metabolic outcome in a
general population in Korea. J Korean Med Sci. 31:1121–1126.
2016.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Rathnakumar K, Ramachandran K, Baba D,
Ramesh V, Anebaracy V, Vidhya R, Vinothkumar R, Poovitha R and
Geetha R: Prevalence of dry eye disease and its association with
dyslipidemia. J Basic Clin Physiol Pharmacol. 29:195–199.
2018.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Posa A, Sel S, Dietz R, Sander R, Paulsen
F, Bräuer L and Hammer C: Historical profiling of dry eye
patients-potential trigger factors and comorbidities. Klin Monbl
Augenheilkd. 241:110–118. 2024.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Li Y, Xie L, Song W, Chen S, Cheng Y, Gao
Y, Huang M, Yan X and Yang S: Association between dyslipidaemia and
dry eye disease: A systematic review and meta-analysis. BMJ Open.
13(e069283)2023.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Wang TH, Tsai YJ, Wang YH, Wu CL and Lin
IC: Relationship between dry eye disease and dyslipidemia: A
systematic review. J Clin Med. 12(6631)2023.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Serrano-Morales JM, Álvarez-Santaliestra
N, Sánchez-González MC, Ballesteros-Sánchez A and Sánchez-González
JM: Impact of dyslipidemia on tear film and meibomian gland
dysfunction: A cross-sectional study of the interplay between serum
lipid profile and ocular surface health. J Ophthalmol.
2024(7345270)2024.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Yang Y, Zhang R, Zhang M, Yang Z, Ma Z,
Zhang Y, Wu M, Guo D and Bi H: Association between disorders of
lipid metabolism and oculopathy: An overview. Int J Med Sci.
22:3878–3894. 2025.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Vitiello L, De Bernardo M, Guercio Nuzio
S, Mandato C, Rosa N and Vajro P: Pediatric liver diseases and
ocular changes: What hepatologists and ophthalmologists should know
and share with each other. Dig Liver Dis. 52:1–8. 2020.PubMed/NCBI View Article : Google Scholar
|