Introduction
Glutamine may be a conditionally essential amino
acid in patients with catabolic disease as it has been demonstrated
that circulating plasma glutamine concentrations drop during
critical illness and following major surgery (1). This drop may be due to glutamine's
central role in nitrogen transport within the body, with glutamine
contributing to both precursors for nucleic acid synthesis, as well
as antioxidant defenses through the production of glutathione
(2). In addition, glutamine is the
preferred fuel for rapidly dividing cells, such an enterocytes in
the small intestine and immune cells, such as lymphocytes,
monocytes and macrophages. For some patients, the synthesis and
release of glutamine from skeletal muscle is insufficient to meet
demands, and a deficiency in glutamine may lead to small intestine
mucosal injury followed by increased wall permeability and
bacterial translocation (3). These
negative effects, along with immunosuppression, may all contribute
to an increased probability of secondary infection risk, which may
impede the recovery time of the patient, or worse, increase
in-patient mortality rates.
More than a dozen clinical studies have suggested
that the provision of parenteral or enteral glutamine
supplementation in both critically ill and surgical patients may
improve nitrogen balance, constitutive protein levels and improve
immune function, while decreasing infection rates, the length of
stay in hospital and mortality rates (4). However, there is much discrepancy
regarding comparing single randomized controlled trials on
glutamine supplementation's effects on clinical endpoints such as
infection rates, length of stay in hospital and mortality (5,6). This
discrepancy may be due to differences in study design, selected
patient populations, severity of the disease, nutritional status of
the patient and/or the differences in glutamine supplementation
relative to different forms (free or dipeptide form), doses and
methods of administration (parenteral or enteral).
Given the inconsistency of the existing literature
and the insufficient statistical power due to small sample sizes, a
pooling of information from individual trials may provide a more
precise and accurate estimate of glutamine's role in reducing the
rates of infectious complications and mortality. To achieve this
result, many investigators have turned to performing a powerful
statistical method known as meta-analysis. Meta-analyses are
fundamental to provide the highest level of evidence to best inform
health care decision-making. Therefore, the purpose and objective
of the present paper was to summarize the evidence from previously
published meta-analyses regarding the effectiveness of glutamine's
role as a therapeutic dietary agent to reduce the occurrence of
infectious complications, length of stay in hospital and
mortality.
Materials and methods
An umbrella review was selected for the current
study. An umbrella review provides a summary of existing published
meta-analyses and systematic reviews and determines whether authors
addressing similar review questions independently observe similar
results and arrive at similar conclusions (7).
A systematic literature search of PubMed, Cochrane
Library and CINAHL from January 1st, 1980 to December 31st, 2016
was conducted using the following search strategy: ‘Glutamine AND
(meta-analysis OR systematic review)’. Only English language
publications were retrieved, which provided quantitative
statistical analysis of pooled treatment effects on the relative
risks and odds ratios of infectious complications, length of stay
in hospital and mortality of critically ill and surgical patients.
Meta-analyses or systematic reviews that did not present study
specific summary data using a minimum of four randomized controlled
trials or which were focused on infants and children were
excluded.
For the published papers that were accepted into the
review, the following information was extracted and entered into a
spreadsheet: Number of publications included in the meta-analysis,
number of total subjects, glutamine dose and method of
administration, pooled treatment effects on the relative risks and
odds ratios of infectious complications, length of stay in hospital
and mortality. In addition, papers were assessed for their
disclosure of quality assessment, statistical heterogeneity
(Cochrane's Q test and I2 statistic) and publication
bias (visual inspection of funnel plots and Egger's or Begg's
regression test). As this is a descriptive summary review of
meta-analyses, no statistical analyses were performed.
Results
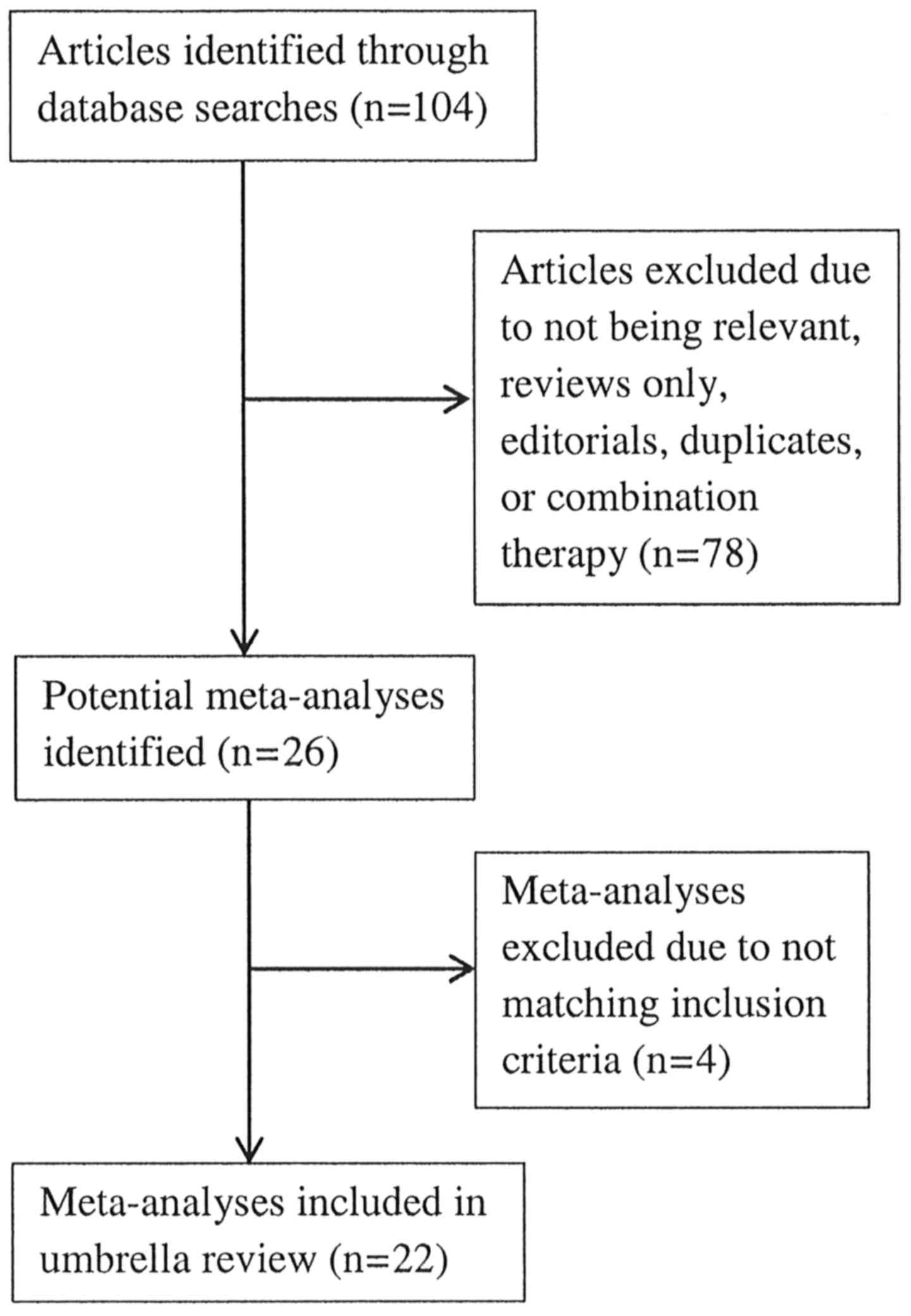
The initial search strategy identified 106 articles
and, following careful review, 26 meta-analyses were retrieved for
inclusion into this umbrella review. There were four meta-analyses
that were excluded because two were not published in English
(8,9),
one fell outside the selection criteria, as it was focused on
preterm infants (10) and one lacked
the necessary methodology and presentation of a well-constructed
meta-analysis (11). A flow chart of
the meta-analyses selection process is summarized in Fig. 1, and Tables
I, II and III provide the detailed analysis of the 22
meta-analyses entered into the umbrella review (12–33).
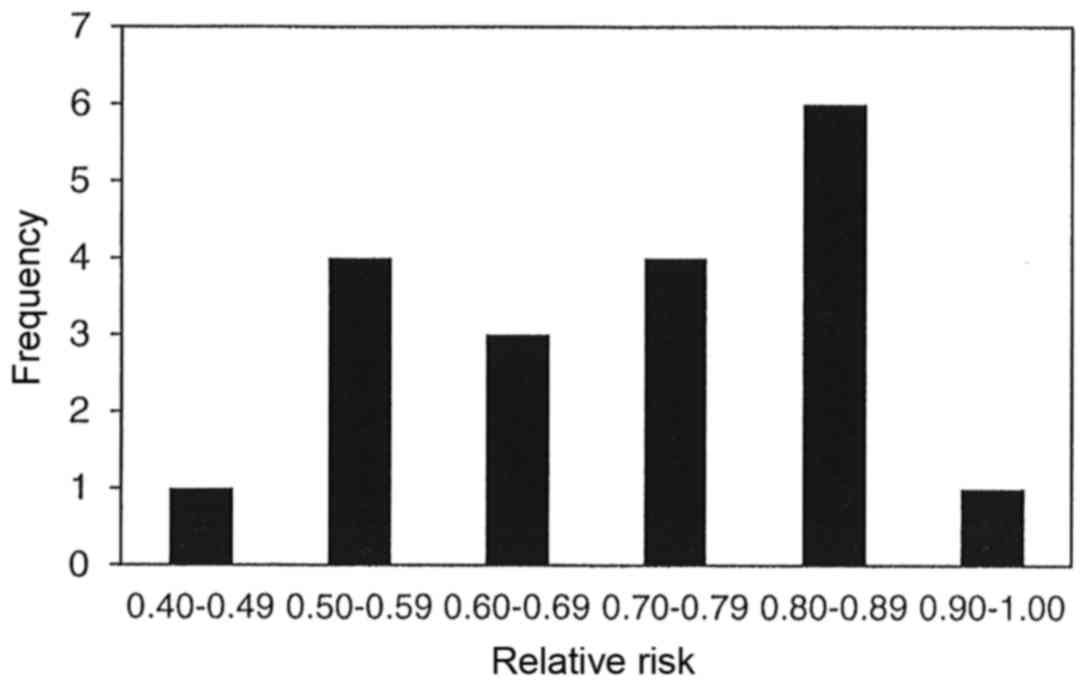
The results of 19 meta-analyses in Table I indicate that parenteral and/or
enteral intakes of 0.3 to 0.45 g/kg/day can provide statistically
significantly reductions in the incidence of infectious
complications for 15 of the 19 meta-analyses with relative risks of
all 19 ranging between 0.42 and 0.93. Statistically significant
heterogeneity was only observed in two of the 19 meta-analyses,
however publication bias was observed in four of the nine
meta-analyses that chose to use a funnel plot analysis and/or Egger
or Begg's regression tests. Fig. 2
displays the bar graph that represents the frequency of the
meta-analyses relative rates for the incidence of infectious
complications.
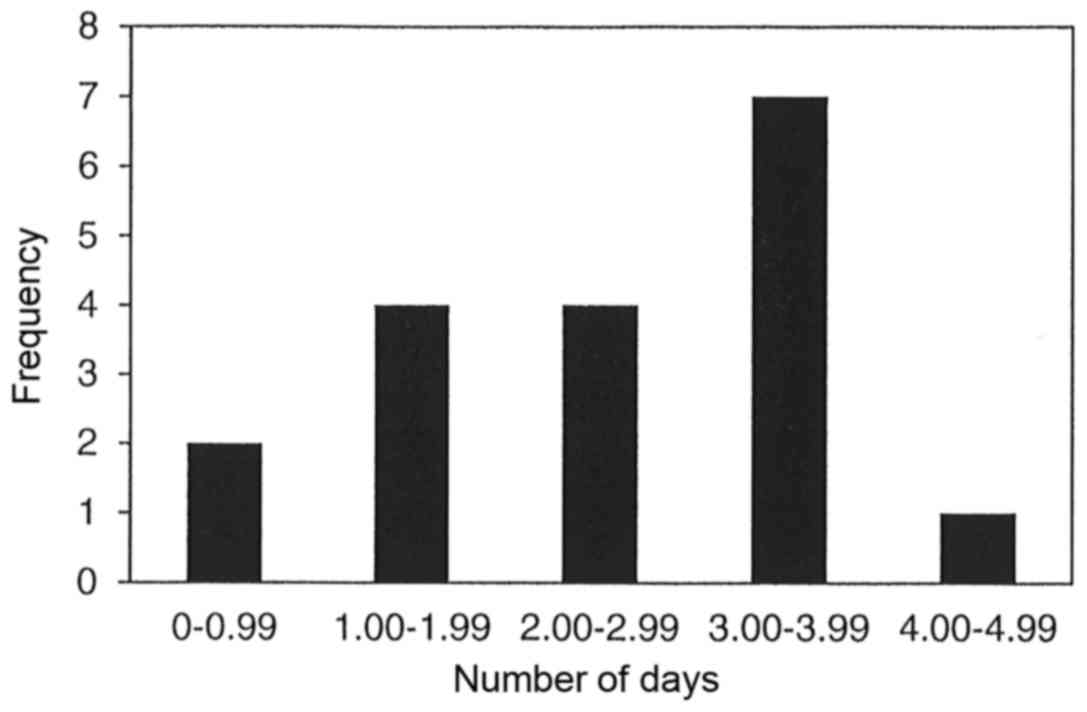
The results of 18 meta-analyses in Table II demonstrate that parenteral and/or
enteral intakes of 0.3 to 0.45 g/kg/day can provide statistically
significantly reductions in the length of stay in hospital for 12
of the 18 meta-analyses. The range for the reduction in length of
stay in hospital for all 12 meta-analyses was between 0.19 to 4.73
days. However, statistically significant heterogeneity was only
observed in 16 of the 18 meta-analyses. Publication bias was
observed in only one of the nine meta-analyses, which chose to use
either funnel plot analysis and/or Egger or Begg's regression test.
Fig. 3 displays the bar graph that
represents the frequency of the meta-analyses average reduction in
length of stay in hospital.
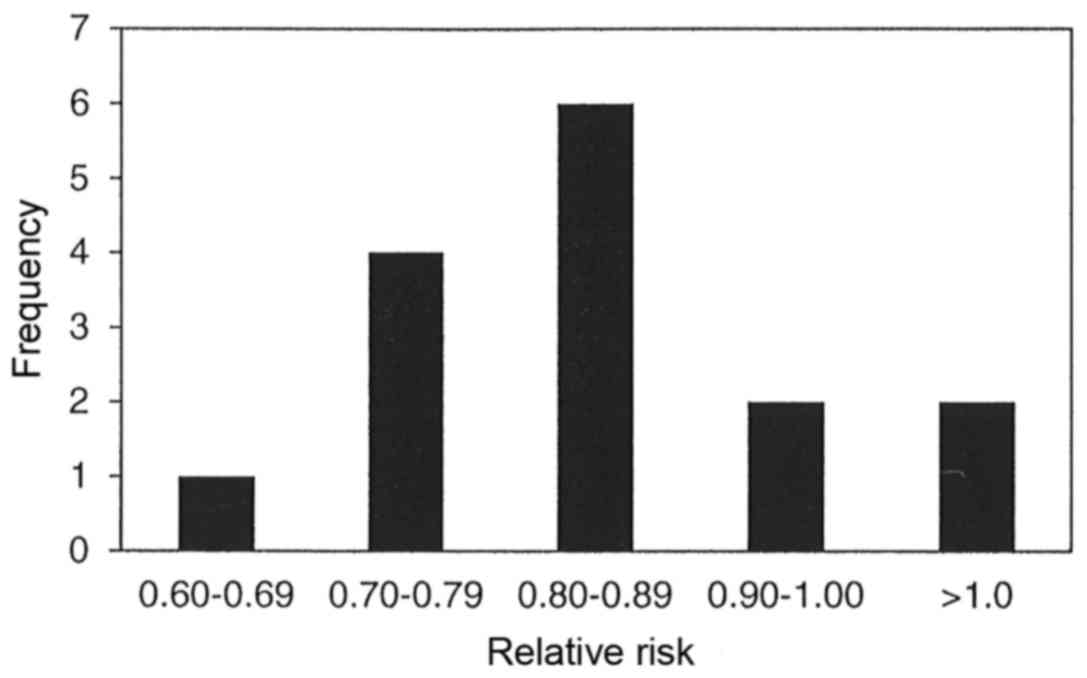
The results of 15 meta-analyses in Table III indicate that parenteral and/or
enteral intakes of 0.3 to 0.42 g/kg/day provided statistically
significant reductions in mortality for only four of the 15
meta-analyses with relative risk for all 15 meta-analyses ranging
between 0.64 to 1.28. Statistically significant heterogeneity was
not observed in any of the 15 meta-analyses, and publication bias
was observed in only one of the 10 meta-analyses, which chose to
use funnel plot analysis and/or Egger or Begg's regression test.
Fig. 4 displays the bar graph that
represents the frequency of the meta-analyses relative rates for
mortality.
Discussion
With regards to the incidence of infectious
complications among critically ill and surgical patients, 15 of the
19 meta-analyses (79%) presented statistically significant
reductions. The rate of infection for all 19 meta-analyses
demonstrated a reduction ranging from 7 to 58%. Just over half of
the meta-analyses (10 of 19) identified reductions of between 10
and 30%, and one-third of the meta-analyses (seven of 19) found
reduction of between 30 and 50%. It should be noted that four of
the nine meta-analyses presented with significant publication bias,
but more troubling is the fact that only nine of the 19
meta-analyses (47%) assessed for publication bias. Publication bias
occurs because small studies with null results tend not to be
published, and this problem is referred to as the ‘file drawer
problem’. As published studies are more likely to report positive
research outcomes than unpublished ones, the significance of the
effect size of the weighted average of the published studies is
overestimated, and this can potentially bias the results of the
meta-analysis. On a positive note, only one of the 15 meta-analyses
that identified statistically significant reductions in infectious
complications presented with statistically significant
heterogeneity.
In regard to the length of stay in hospital, 12 of
the 18 meta-analyses (67%) presented statistically significant
decreases in which the time in hospital decreased between 0.19 to
4.73 days. There were ~two-thirds of the meta-analyses (11 of 18)
that demonstrated a 2 to 4 day reduction in the length of stay in
hospital. However, it should be noted that 16 of the 18
meta-analyses (89%) presented with statistically significant
heterogeneity and this weakens the clinical certainty of this
effect. Ideally the studies combined into any given meta-analysis
should all have used the same experimental protocols; however
increased heterogeneity is inevitable due to the wide variation in
study design. Differences in study design include number of
subjects, age, sex, body mass index, nutritional status (plasma
glutamine concentration at admission), total energy intake, type of
disease or surgery, disease severity, dosage, timing, duration,
chemical form (L-glutamine, alanyl-L-glutamine or
glycyl-L-glutamine) and route of glutamine administration
(parenteral vs. enteral). On a positive note, only one of the nine
meta-analyses presented with statistically significant publication
bias, but again this is problematic as only nine of the 18
meta-analyses (50%) assessed for publication bias.
In regard to mortality rate, 10 of the 15
meta-analyses (67%) identified a reduction that ranged between 10
and 30%, however, only four of the 15 meta-analyses (27%)
demonstrated statistically significant reductions. More troubling
is the fact that these four meta-analyses represented four of the
six that possessed <10 clinical studies in their make-up (4, 4,
6 and 9 respectively). Meanwhile, nine of the 11 meta-analyses with
non-significant findings were made up of between 10 to 36 clinical
studies, and so there appears to be a bias towards meta-analyses
presenting statistical significance when using <10 clinical
trials. This relationship was not observed in the infectious
complications or length of stay in hospital umbrella reviews.
However, heterogeneity and publication bias was never found to be
an issue, as none of the meta-analyses presented statistically
significant heterogeneity, and only one of the 10 meta-analyses
suggested a potential for publication bias.
The apparent effect of glutamine supplementation to
reduce the incidence of infectious complications in critically ill
and surgical patients is probably due to the fact that glutamine is
the preferred fuel for enterocytes for maintaining gastrointestinal
structure and decreasing gut mucosal atrophy, which could otherwise
lead to increased intestinal permeability and bacterial
translocation (3). In addition,
glutamine positively affects lymphocyte proliferation, cytokine
production, macrophage phagocytosis and the secretion of intestinal
immune globulin IgA, which further reduces bacterial adhesion and
prevents translocation (34).
Therefore, glutamine serves a critical role in the protection of
the intestinal immune barrier and leads to increased resistance to
bacterial translocation and, hence, reduces infectious complication
rates. This fact is supported by a meta-analysis that presented
beneficial changes in the markers of intestinal inflammation and
mucosal permeability with abdominal surgery patients (35). Finally, glutamine supplementation
enhances cellular heat shock protein production (HSP70 and HSP72)
and glutathione expression, which both protect cells and enhance
cell survival. Heat shock proteins are involved in the repair and
removal of damaged proteins, and glutathione reacts directly with
reactive oxygen species in order to prevent oxidative damage
(36). Collectively, both mechanisms
of action reduce the likelihood of cellular apoptosis.
If glutamine supplementation can reduce the
incidence of hospital acquired infectious complications, then, when
averaged over a large number of patients, this can result in a
shorted length of stay in hospital. If a critically ill or surgical
patient does acquire an infectious complication, this will prolong
that patient's length of stay in hospital and require more
resources. Hospital acquired infections are expensive to treat, and
it is estimated that hospital acquired infections can prolong a
patient's length of stay by at least 4 days; this can have a
significant financial impact on hospitalization cost (27). It has been observed that the additional
cost of supplemented glutamine treatment is more than completely
offset by cost savings in hospital care (37).
Low plasma glutamine concentrations in patients
admitted to hospitals are an independent predictor of premature
death, but it is unclear whether the decline of circulating
glutamine contributes to death, or whether it is a simple marker of
disease severity (38). Assuming that
supplemental glutamine's primary mechanism of action is in the
prevention of hospital acquired infections, then it is
understandable why there were no significant changes observed in
mortality rates. To begin with, the mortality rate for patients
admitted to hospital as inpatients is ~2% (39). Furthermore, hospital acquired
infections occur in ~4% of all admitted patients and only ~6% of
those patients with hospital acquired infections die during their
hospitalization (40,41). From these data, it can be estimated
that the mortality rate due to hospital acquired infections for all
patients admitted to hospitals is ~0.25%, which represents only an
eighth of all in-patient mortality rates. Therefore, for the
majority of meta-analyses that did not identify a significant
difference in mortality rates, this is probably due to the fact
that the majority of clinical studies used in these meta-analyses
were too small and too underpowered to detect these small positive
changes in mortality rates.
Although enteral glutamine supplementation should
benefit intestinal enterocyte function and overall gut barrier
structure better than parenteral glutamine supplementation, it has
been suggested that there is a greater treatment effect observed
for parenteral compared with enteral glutamine supplementation
(12,14,16,18). This is thought to be due to the higher
bioavailability of glutamine through parenteral administration,
whereas enteral glutamine is used by the gut, therefore the immune
system outside the gut would not have been affected by enteral
glutamine supplementation. However, only one of the eight
meta-analyses that performed a subgroup analysis on administration
type presented a significant difference between parenteral and
enteral administration, and this meta-analysis found that the
length of stay in hospital was significantly shortened for the
enteral route (4.5 days less for enteral vs. 2.5 days less for
parenteral) (23). To confuse matters
more, the only two meta-analyses on clinical studies that used
enteral glutamine supplementation with critically ill patients
demonstrated completely opposite effects on length of stay in
hospital with one presenting a 4.73 day reduction and the other
with a 0.19 day reduction (it should be noted that these two
findings represent the two extremes from the 18 meta-analyses in
this category) (24,32). Although both of these meta-analyses
examined clinical studies using enteral feeding in critically ill
patients only, there was only one common clinical study used by
both meta-analyses of the combined total of 12 different clinical
studies. The fact that there appears no real difference in outcomes
between enteral and parenteral should make sense, considering that
many of the potential protective mechanisms of glutamine
supplementation given either enterally or parenterally overlap and
are quite similar.
Four of six meta-analyses that performed subgroup
analysis on high vs. low dose glutamine supplementation observed
that the mortality rate was lower with high dose compared to low
dose glutamine supplementation, but the differences between groups
were never statistically significant (12,14–16,22,30). Of the three meta-analyses that
performed subgroup analyses comparing critically ill vs. surgical
patients, it also appears that there was no significant difference
between these two groups in their response to glutamine
supplementation (12,15,16).
In regard to side effects, only 11 of the 22
meta-analyses (50%) addressed this topic with six of them noting
that no side effects were observed (12–14,19,24,26), and the remaining five meta-analyses all
mentioned the REDOX study that indicated that critically ill
patients with glutamine doses of >0.5 g/kg/day had higher urea
concentrations and higher mortality rates (20–22,32). However, it has been noted that there
were problems with the REDOX study as 30% of the patients in the
glutamine supplementation group were shown to have baseline renal
failure at admission, which is a very common exclusion criteria for
the majority of glutamine supplementation clinical trials (16,21). In the
REDOX post-hoc analysis the authors of the study concluded that
high-dose glutamine supplementation was only associated with
increased mortality in critically ill patients with multi organ
failure (42).
Using mostly Jadad or Cochrane quality assessment
scales, 18 of the 22 meta-analyses obtained quality assessment
scores and the average percentages of high and low quality clinical
studies used in any given meta-analysis was 30.4 and 13.8%,
respectively. Although none of the meta-analyses specified that
clinical studies were excluded from their analysis, more recently
published meta-analyses may have excluded low quality studies from
the outset without specifying that they were excluded. For the
majority of the meta-analyses (11 of the 22), the number of
clinical studies entered for analysis ranged between 10 and 20.
There were five meta-analyses that used <6 clinical studies, and
one meta-analysis published in 2014 used 53 clinical studies, and
these six analyses represent 43% of the 125 unique clinical studies
used by all 22 meta-analyses (20). Of
the 125 unique clinical studies used by the 22 meta-analyses in
this umbrella review, 57 (46%) were used only once. This
potentially suggests that there are many clinical studies of such
poor quality that they may have made the inclusion criteria by one
meta-analysis but excluded by all the others. Or it may simply be
due to the fact that a particular meta-analysis may have had a very
specific focus [acute pancreatitis (18,19), burn
patients (31) or gastrointestinal
tumor patients (29)] and therefore
the clinical studies used by this meta-analysis would have been
specifically selected by this one meta-analysis and no other.
This umbrella review has certain limitations. One
limitation is that only three indexing systems were searched, and
thus it is possible that some meta-analyses were not identified.
Another limitation is that only one author performed the search and
selection of the meta-analyses included in this umbrella review.
And finally, as in all literature reviews, the quality of this
umbrella review is directly related to the quality of the included
meta-analyses, which are dependent upon the quality of the
individual clinical studies used to conduct the meta-analysis.
In conclusion, this umbrella review supports the
statement by the American Society for Parenteral and Enteral
Nutrition that suggests that parenteral glutamine supplementation
should probably be given early and in doses between 0.2 and 0.5
g/kg/day (43). This statement is
supported by the fact that a majority of the meta-analyses reviewed
identified significant positive benefits for glutamine
supplementation to reduce the rate of hospital acquired infectious
complications, and shortening of the length of stay in hospital. In
addition, glutamine supplementation appeared to reduce the rate of
hospital mortality, but for the majority of these meta-analyses
they did not reach statistical significance. However, it must be
appreciated that all these positive results with caution in light
of the fact that there exists statistically significant
heterogeneity in the length of stay in hospital meta-analyses, and
statistically significant publication bias in the infectious
complications meta-analyses. Also, the lack of quality assessment
of published clinical studies is problematic, as clinical studies
of very low quality may have been included in these meta-analyses,
which can therefore potentially bias their overall outcomes.
Despite the problems of heterogeneity, publication bias and quality
assessment, the potential benefits observed in this umbrella review
strongly suggests that parenteral and enteral glutamine
supplementation as a component of nutritional support can be
considered as an approach to improve the outcomes of critically ill
and surgical patients. Finally, in regards to determining if there
is beneficial effect on reducing hospital mortality rates with
glutamine supplementation, more well-designed multicenter
randomized controlled trials using large populations are
required.
References
|
1
|
Wischmeyer PE: Glutamine: Mode of action
in critical illness. Crit Care Med. 35 Suppl 9:S541–S544. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Wischmeyer PE: Glutamine: Role in critical
illness and ongoing clinical trials. Curr Opin Gastroenterol.
24:190–197. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wischmeyer PE: Glutamine: Role in gut
protection in critical illness. Curr Opin Clin Nutr Metab Care.
9:607–612. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
van Zanten AR: Glutamine and antioxidants:
Status of their use in critical illness. Curr Opin Clin Nutr Metab
Care. 18:179–186. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Cynober L and De Bandt JP: Glutamine in
the intensive care unit. Curr Opin Clin Nutr Metab Care. 17:98–104.
2014.PubMed/NCBI
|
|
6
|
Mundi MS, Shah M and Hurt RT: When is it
appropriate to use glutamine in critical illness? Nutr Clin Pract.
31:445–450. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Aromataris E, Fernandez R, Godfrey CM,
Holly C, Khalil H and Tungpunkom P: Summarizing systematic reviews:
Methodological development, conduct and reporting of an umbrella
review approach. Int J Evid Based Healthc. 13:132–140. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Jiang ZM and Jiang H: The clinical
efficacy of glutamine dipeptides on postoperative patients: An
updated systematic review of randomized controlled trials from
Europe and Asia (1997–2005). Zhonghua Yi Xue Za Zhi. 86:1610–1614.
2006.(In Chinese). PubMed/NCBI
|
|
9
|
Jiang H, Chen W, Hu W, Cai B and Liao RJ:
The impact of glutamine-enhanced enteral nutrition on clinical
outcome of patients with critical illness: A systematic review of
randomized controlled trials. Zhonghua Shao Shang Za Zhi.
25:325–330. 2009.(In Chinese). PubMed/NCBI
|
|
10
|
Moe-Byrne T, Brown JV and McGuire W:
Glutamine supplementation to prevent morbidity and mortality in
preterm infants. Cochrane Database Syst Rev.
4:CD0014572016.PubMed/NCBI
|
|
11
|
Castro-Gutiérrez V and Rada G: Is there a
role for glutamine supplementation in the management of acute
pancreatitis? Medwave. 16(Suppl 3): e65122016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Novak F, Heyland DK, Avenell A, Drover JW
and Su X: Glutamine supplementation in serious illness: A
systematic review of the evidence. Crit Care Med. 30:2022–2029.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Avenell A: Glutamine in critical care:
Current evidence from systematic reviews. Proc Nutr Soc.
65:236–241. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Avenell A: Hot topics in parenteral
nutrition. Current evidence and ongoing trials on the use of
glutamine in critically-ill patients and patients undergoing
surgery. Proc Nutr Soc. 68:261–268. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Bollhalder L, Pfeil AM, Tomonaga Y and
Schwenkglenks M: A systematic literature review and meta-analysis
of randomized clinical trials of parenteral glutamine
supplementation. Clin Nutr. 32:213–223. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Chen QH, Yang Y, He HL, Xie JF, Cai SX,
Liu AR, Wang HL and Qiu HB: The effect of glutamine therapy on
outcomes in critically ill patients: A meta-analysis of randomized
controlled trials. Crit Care. 18:R82014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Crowther M, Avenell A and Culligan DJ:
Systematic review and meta-analyses of studies of glutamine
supplementation in haematopoietic stem cell transplantation. Bone
Marrow Transplant. 44:413–425. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Asrani V, Chang WK, Dong Z, Hardy G,
Windsor JA and Petrov MS: Glutamine supplementation in acute
pancreatitis: A meta-analysis of randomized controlled trials.
Pancreatology. 13:468–474. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zhong X, Liang CP and Gong S: Intravenous
glutamine for severe acute pancreatitis: A meta-analysis. World J
Crit Care Med. 2:4–8. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tao KM, Li XQ, Yang LQ, Yu WF, Lu ZJ, Sun
YM and Wu FX: Glutamine supplementation for critically ill adults.
Cochrane Database Syst Rev. CD0100502014.PubMed/NCBI
|
|
21
|
Wischmeyer PE, Dhaliwal R, McCall M,
Ziegler TR and Heyland DK: Parenteral glutamine supplementation in
critical illness: A systematic review. Crit Care. 18:R762014.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Oldani M, Sandini M, Nespoli L, Coppola S,
Bernasconi DP and Gianotti L: Glutamine supplementation in
intensive care patients: A meta-analysis of randomized clinical
trials. Medicine (Baltimore). 94:e13192015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Yong L, Lu QP, Liu SH and Fan H: Efficacy
of glutamine-enriched nutrition support for patients with severe
acute pancreatitis: A meta-analysis. JPEN J Parenter Enteral Nutr.
40:83–94. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
van Zanten AR, Dhaliwal R, Garrel D and
Heyland DK: Enteral glutamine supplementation in critically ill
patients: A systematic review and meta-analysis. Crit Care.
19:2942015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Jiang Z, Jiang H and Fürst P: The impact
of glutamine dipeptides on outcomes of surgical patients:
Systematic review of randomized controlled trials from Europe and
Asia. Clin Nutr Suppl. 1:17–23. 2004. View Article : Google Scholar
|
|
26
|
Zheng YM, Li F, Zhang MM and Wu XT:
Glutamine dipeptide for parenteral nutrition in abdominal surgery:
A meta-analysis of randomized controlled trials. World J
Gastroenterol. 12:7537–7541. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang Y, Jiang ZM, Nolan MT, Jiang H, Han
HR, Yu K, Li HL, Jie B and Liang XK: The impact of glutamine
dipeptide-supplemented parenteral nutrition on outcomes of surgical
patients: A meta-analysis of randomized clinical trials. JPEN J
Parenter Enteral Nutr. 34:521–529. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yue C, Tian W, Wang W, Huang Q, Zhao R, Li
Q and Li J: The impact of perioperative glutamine-supplemented
parenteral nutrition on outcomes of patients undergoing abdominal
surgery: A meta-analysis of randomized clinical trials. Am Surg.
79:506–513. 2013.PubMed/NCBI
|
|
29
|
Kang K, Shu XL, Zhang YS, Liu XL and Zhao
J: Effect of glutamine enriched nutrition support on surgical
patients with gastrointestinal tumor: A meta-analysis of randomized
controlled trials. Chinese Med J (Engl). 128:245–251. 2015.
View Article : Google Scholar
|
|
30
|
Sandini M, Nespoli L, Oldani M, Bernasconi
DP and Gianotti L: Effect of glutamine dipeptide supplementation on
primary outcomes for elective major surgery: Systematic review and
meta-analysis. Nutrients. 7:481–499. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Lin JJ, Chung XJ, Yang CY and Lau HL: A
meta-analysis of trials using the intention to treat principle for
glutamine supplementation in critically ill patients with burn.
Burns. 39:565–570. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Mottaghi A, Yeganeh MZ, Golzarand M,
Jambarsang S and Mirmiran P: Efficacy of glutamine-enriched enteral
feeding formulae in critically ill patients: A systematic review
and meta-analysis of randomized controlled trials. Asia Pac J Clin
Nutr. 25:504–512. 2016.PubMed/NCBI
|
|
33
|
Heyland DK, Dhaliwal R, Drover JW,
Gramlich L and Dodek P; Canadian Critical Care Clinical Practice
Guidelines Committee, : Canadian clinical practice guidelines for
nutrition support in mechanically ventilated, critically ill adult
patients. JPEN J Parenter Enteral Nutr. 27:355–373. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Newsholme P: Why is L-glutamine metabolism
important to cells of the immune system in health, postinjury,
surgery or infection? J Nutr. 131(9 Suppl): 2515S–2524S.
2001.PubMed/NCBI
|
|
35
|
Shu XL, Yu TT, Kang K and Zhao J: Effects
of glutamine on markers of intestinal inflammatory response and
mucosal permeability in abdominal surgery patients: A
meta-analysis. Exp Ther Med. 12:3499–3506. 2016.PubMed/NCBI
|
|
36
|
Oliveira GP, Dias CM, Pelosi P and Rocco
PR: Understanding the mechanisms of glutamine action in critically
ill patients. An Acad Bras Cienc. 82:417–430. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Pradelli L, Povero M, Muscaritoli M and
Eandi M: Updated cost-effectiveness analysis of supplemental
glutamine for parenteral nutrition of intensive-care patients. Eur
J Clin Nutr. 69:546–551. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Rodas PC, Rooyackers O, Hebert C, Norberg
Å and Wernerman J: Glutamine and glutathione at ICU admission in
relation to outcome. Clin Sci (Lond). 122:591–597. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Baker GR, Norton PG, Flintoft V, Blais R,
Brown A, Cox J, Etchells E, Ghali WA, Hébert P, Majumdar SR, et al:
The Canadian adverse events study: The incidence of adverse events
among hospital patients in Canada. CMAJ. 170:1678–1686. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Magill SS, Edwards JR, Bamberg W, Beldavs
ZG, Dumyati G, Kainer MA, Lynfield R, Maloney M, McAllister-Hollod
L, Nadle J, et al: Multistate point-prevalence survey of health
care-associated infections. N Engl J Med. 370:1198–1208. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Klevens RM, Edwards JR, Richards CL
Jr..Horan TC, Gaynes RP, Pollock DA and Cardo DM: Estimating health
care-associated infections and deaths in U.S. hospitals, 2002.
Public Health Rep. 122:160–166. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Heyland DK, Elke G, Cook D, Berger MM,
Wischmeyer PE, Albert M, Muscedere J, Jones G and Day AG; Canadian
Critical Care Trials Group, : Glutamine and antioxidants in the
critically ill patient: A post hoc analysis of a large-scale
randomized trial. JPEN J Parenter Enteral Nutr. 39:401–409. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Vanek VW, Matarese LE, Robinson M, Sacks
GS, Young LS and Kochevar M; Novel Nutrient Task Force, Parenteral
Glutamine Workgroup; American Society for Parenteral and Enteral
Nutrition (A.S.P.E.N.) Board of Directors, : A.S.P.E.N. position
paper: Parenteral nutrition glutamine supplementation. Nutr Clin
Pract. 26:479–494. 2011. View Article : Google Scholar : PubMed/NCBI
|