Introduction
microRNAs (miRNA/miRs) are endogenously expressed,
single-stranded RNAs of approximately 22 nucleotides in length that
serve pivotal roles in regulating the expression of target genes
post-transcriptionally. miRNAs are involved in critical cellular
processes including cell development, proliferation,
differentiation and apoptosis (1).
Previous results have demonstrated that miRNAs are frequently
abnormally expressed in various cancers, and function as oncogenes
or tumor suppressors in tumorigenesis and tumor progression
(2,3).
For instance, the oncogenic miR-21 is commonly overexpressed in
gastric cancer and inhibits the tumor suppressors phosphatase and
tensin homolog and programmed cell death protein 4 to promote
gastric cancer growth and invasion (4).
More recently, studies have demonstrated that
miR-124 was downregulated in various cancers, including breast
(5), colon (6), prostate (7), stomach (8), liver (9),
ovarian (10) and brain (11) cancers. For instance, miR-124 was
decreased in breast cancer cell lines and patient specimens;
additionally, it was indicated to attenuate cell growth and
migration by targeting flotillin-1 (5). miR-124 has also been reported to
regulate invasion and metastasis in hepatocellular carcinoma
through regulation of rho associated coiled-coil containing protein
kinase 2 and enhancer of zeste homolog 2 (EZH2) (9). Furthermore, Kang et al (7), documented that miR-124 may suppress
growth and cell invasion in prostate cancer through the proprotein
convertase subtilisin/kexin type 6 (also known as PACE4) pathway.
These data suggest that miR-124 may act as a tumor suppressor. Our
group previously demonstrated that miR-124 suppressed gastric
cancer cell proliferation and induced apoptosis by directly
targeting EZH2 (8). Xia et al
(12) reported that miR-124 inhibited
cell proliferation through downregulation of sphingosine kinase 1
in gastric cancer. To date, however, the biological impacts of
miR-124 on gastric cancer cell metastasis and the corresponding
molecular mechanisms have seldom been investigated.
In the present study, the expression of miR-124 in
gastric cancer and its association with clinical parameters were
investigated. Furthermore, the association of miR-124 with overall
survival (OS) and disease-free survival (DFS) in patients with
gastric cancer was evaluated. Functional studies were also
performed to investigate the function of miR-124 in gastric cancer
cells in vitro and in vivo. Additionally, the
potential targeting action of miR-124-3p against Ras-related C3
botulinum toxin substrate 1 (Rac1) and specificity protein 1 (SP1)
was assessed. These experiments aimed to indicate the underlying
mechanism for the regulation of gastric cancer by miR-124-3p via
targeting of Rac1 and SP1.
Materials and methods
Cell culture
The gastric cancer cell lines SGC-7901 and MKN-28
were obtained from the American Type Culture Collection (Manassas,
VA, USA). The MKN-28 cell line is a derivative of the MKN-74
gastric tubular adenocarcinoma cell line (13). The cell lines used in the present
experiments were reauthenticated by Beijing Microread Genetics Co.,
Ltd. (Beijing, China) via short tandem repeat profiles analysis
every 6 months following resuscitation. The cells were maintained
at 37°C in an atmosphere of 5% CO2 in RPMI-1640 medium
supplemented with 10% fetal bovine serum, 10,00 U/ml penicillin and
10,000 µg/ml streptomycin (Gibco; Thermo Fisher Scientific, Inc.,
Waltham, MA, USA).
Clinical samples
All tissue samples used in the present study were
collected from the Hunan Provincial Tumor Hospital (Changsha,
China). Written informed consent was obtained from all study
participants, and all procedures involving human participants were
approved by the Ethics Committee of the University of South China
Health Authority (Hengyang, China). The collection and use of
tissues followed procedures in accordance with the ethical
standards in the Declaration of Helsinki. The tissue microarrays
(TMAs) consisted of 121 cases of gastric carcinoma tissues
diagnosed by histopathological diagnosis. Specimens were obtained
during surgery, fixed in 10% formalin for 24 h at room temperature,
embedded in paraffin and cut into sections (4-µm thickness), and
stored in the Hunan Provincial Tumor Hospital. The TMAs were used
for immunohistochemistry (IHC) and in situ hybridization
(ISH) analyses. The sample data, including age, sex, histological
grade, tumor size, invasion depth (T stage) and lymph node
metastasis, were obtained from clinical and pathological records.
Furthermore, following reverse transcription-quantitative
polymerase chain reaction (RT-qPCR), the clinical samples were
divided into low expression and high expression groups based on
miR-124-3p expression scores greater or less than 2 (14).
RNA isolation and RT-qPCR
Total RNA from the gastric cancer cell lines was
extracted using TRIzol reagent (Invitrogen; Thermo Fisher
Scientific, Inc., Waltham, MA, USA), and 1 µg of total RNA was
reverse transcribed using a Reverse Transcription System kit
(Promega Corporation, Madison, WI, USA). RT-qPCR was performed
using a Power SYBR-Green PCR Master Mix (Thermo Fisher Scientific,
Inc.), with human β-actin amplified as an internal control. For
miRNA analysis, reverse transcription and RT-qPCR were performed
using an all-in-one miRNA qRT-PCR Detection kit (GeneCopoeia, Inc.,
Rockville, MD, USA) and U6 small nuclear RNA was used as an
endogenous control. For the miRNA and mRNA amplifications, the PCR
cycling conditions were as follows: One cycle at 95°C for 3 min,
followed by 40 cycles at 95°C for 12 sec and 62°C for 35 sec, and
finally 1 cycle at 62-95°C for 15 sec. The relative fold-changes in
expression with respect to the endogenous controls were calculated
by the 2−ΔΔCq method (15). The primers for SP1, Rac1, U6 and
β-actin, synthesized by Invitrogen (Thermo Fisher Scientific,
Inc.), were as follows: For SP1, forward,
5′-TGCCTCCACTTCCTCGATTT-3′ and reverse, 5′-TCTGGTGGGCAGTATGTTGT-3′;
for U6, forward, 5′-UUCUCCGAACGUGUCACGUTT-3′ and reverse,
5′-ACGUGACACGUUCGGAGAATT-3′; and for β-actin, forward,
5′-AGCGAGCATCCCCCAAAGTT-3′; and reverse,
5′-GGGCACGAAGGCTCATCATT-3′.
Cell viability assay
SGC-7901 and MKN-28 cells were transfected with 50
nM of miR-124 mimics (miR-124: 5′-UAAGGCACGCGGUGAAUGCCAA-3′),
miR-124 inhibitors (miR-124-LNA: 5′-UUGGCAUUCACCGCGUGCCUUA-3′) or
their scrambled oligonucleotide controls (miR-ctr:
5′-UUCUCCGAACGUGUCACGUTT-3′; and miR-LNA:
5′-CAGUACUUUUGUGUAGUACAA-3′, respectively; Shanghai Jima Industrial
Co., Ltd., Shanghai, China) using Lipofectamine 2000 (Invitrogen;
Thermo Fisher Scientific, Inc.). After 6 h, the transfected cells
were seeded in 96-well plates at a density of 1,000 cells/well. An
MTS assay (Promega Corporation) was performed according to the
manufacturer's instructions after 0 and 2 days of incubation at
37°C. Absorbance values were examined at 490 nm using a Spectra Max
250 spectrophotometer (Molecular Devices, LLC, Sunnyvale, CA,
USA).
IHC and ISH analysis
For baseline staining, the sections were washed with
distilled water, and the cell nuclei were stained with hematoxylin.
The sections were then rinsed in running tap water, differentiated
with 0.3% acid alcohol, and rinsed again in running tap water.
After rinsing in Scott's tap water substitute followed by tap
water, the sections were stained with eosin for 2 min. Finally, the
sections were dehydrated, cleared and mounted.
IHC and ISH were performed according
to standard procedures (14)
For IHC, randomly selected human gastric cancer TMA
sections from all patients were incubated with anti-Rac1 (sc-95;
1:500) and anti-SP1 (sc-17824; 1:500; Santa Cruz Biotechnology,
Inc., Dallas, TX, USA) antibodies at 4°C overnight, then with
horseradish peroxidase (HRP)-conjugated rabbit anti-mouse (BA1058;
1:5,000) and HRP-goat anti-rabbit (BA1058; 1:5,000; Boster
Biological Technology, Pleasanton, CA, USA) antibodies at room
temperature for 1 h. miR-124-3p miRCURY LNA™ custom detection
probes (Exiqon A/S, Vedbaek, Denmark) were used for ISH; the 5′-3′
sequences (enhanced with LNA) were UAAGGCACGCGGUGAAUGCC, with a
digoxigenin label at both the 5′ and 3′ ends. Hybridization,
washing and scanning was performed according to the manufacturer's
instructions. The slides were analyzed under a light microscope
(Nikon Eclipse 80i; Nikon Corporation, Tokyo, Japan) at ×400
magnification by two independent pathologists.
Western blot analysis
Western blot analysis was performed using standard
procedures. Briefly, total protein from the gastric cancer cell
lines was extracted using radioimmunoprecipitation assay lysis
buffer with a proteinase inhibitor (P0013B; Beyotime Institute of
Biotechnology, Haimen, China). The protein concentrations in the
lysates were measured with a Protein BCA Assay kit (Bio-Rad
Laboratories, Inc., Hercules, CA, USA). Subsequently, 30 µg protein
per lane was separated by 10% sodium dodecyl sulfate polyacrylamide
gel electrophoresis and transferred to PVDF (polyvinylidene
difluoride) membranes. To block nonspecific binding, the membranes
were incubated with 5% skimmed milk powder in phosphate-buffered
saline at room temperature for 1 h. The membranes were then
incubated with primary antibodies overnight at 4°C, followed by
horseradish peroxidase-labeled secondary antibodies at room
temperature for 1 h, and protein bands were detected by
electrochemiluminescence (ECL) with ECL western blotting detection
reagents (New England BioLabs, Inc., Ipswich, MA, USA). The
antibodies and their dilutions were the same as those used for the
IHC analysis. β-actin (4967S; 1:500; Cell Signaling Technology,
Inc., Danvers, MA, USA) was used as a protein-loading control.
RNA interference assay
SP1 short hairpin (sh)RNA (HSH054522-CH1), Rac1
shRNA (HSH016063-CH1; GeneCopoeia, Inc.) and control shRNA
(sc-108080; Santa Cruz Biotechnology, Inc.) lentiviral particles
were used for knockdown experiments. SGC-7901 cells were incubated
with the lentiviral vectors (40 µg/l) and 5 mg/ml polybrene
(Shanghai Yi Sheng Biotechnology Co., Ltd., Shanghai, China) with
Lipofectamine 2000 overnight. After 48 h, the cells were harvested
for use in cell viability and expression assays.
SP1 and Rac1 vector construction
SP1 and Rac1-expressing vectors were constructed.
Full-length SP1 and Rac1 cDNAs were purchased from GeneCopeia, Inc.
and were individually subcloned into the eukaryotic expression
vector pcDNA3.1(+) (V79020; Invitrogen; Thermo Fisher Scientific,
Inc.). pcDNA3.1 (+) was used as a negative control. These vectors
(40 nM) were transfected into cells using Lipofectamine 2000 for
overexpression experiments. At 24 h after transfection, the cells
were harvested and analyzed for cell viability.
Luciferase assays
The sequence of the 3′ untranslated region (UTRs) of
the SP1 gene was obtained from the TargetScan database version 7.1
(http://www.targetscan.org/vert_71/).
The 3′UTR was amplified by PCR (14)
from the genomic DNA of the SGC-7901 cell line and inserted into a
pGL3 control vector (Promega Corporation) using the XBA1 site
immediately downstream from the stop codon of luciferase. The SP1
primer sets used for PCR were as follows: Forward
5′-CCTTCAGGGATTTCCAACTG-3′ and reverse, 5′-GTCCAAAAGGCATCAGGGTA-3′.
A mutant insert was also generated in which the first four
nucleotides of the miR-124 binding site the SP1 gene (AUGT-GCAC;
predicted by TargetScan) were mutated using a QIAGEN XL-site
directed Mutagenesis kit (Qiagen, Inc. Valencia, CA, USA). SGC-7901
and MKN-28 cells were cotransfected by nucleoporation (Amaxa
Nucleofector™; Lonza Group, Ltd., Basel, Switzerland) (16) with 5 µg firefly luciferase reporter
vector (Promega Corporation) and 0.5 μg control vector containing
Renilla luciferase (pRL-TK; Promega Corporation). For each
nucleoporation, 50 nM of the miR-124-3p mimic, miR-124-LNA
(inhibitor), miR-ctr or miR-LNA was used. Firefly and Renilla
luciferase activities were measured consecutively using a dual
luciferase assay (Promega Corporation) at 48 h after
transfection.
Mouse xenograft model
A gastric cancer model in male BALC/c mice (Beijing
Vital River Laboratory Animal Technology Co., Ltd., Beijing, China)
was established. A total of 12 mice (6 weeks old; 20-22 g) were
used, which were housed under controlled conditions at 25°C, 60%
humidity and a 12-h light/dark cycle, with food and water available
ad libitum. A total of 5×105 SGC-7901 cells were
inoculated subcutaneously into the dorsal flanks of the mice. After
10 days of tumor growth, the tumors were infected with the miR-124
mimic, miR-124-LNA or their controls (n=3 mice per group). Tumor
size was measured every 4 days. The maximum tumor size permitted
was 1.2 cm3. The mice were sacrificed and necropsies
were performed after 28 days, and the tumors were isolated and
weighed. Additionally, the tumor volumes were calculated according
to the formula: AxB2/2 (A: the largest diameter, B: the
diameter perpendicular to A). All of the animal procedures were
performed in accordance with the Animal Research: Reporting of
In Vivo Experiments guidelines and National Institutes of
Health (NIH) Guide for the Care and Use of Laboratory Animals (NIH
Publications No. 8023, revised 1978). Ethical approval was obtained
for the animal experiments from the Ethics Committee of the
University of South China Health Authority. All possible steps were
taken to avoid animal suffering at each stage of the
experiment.
Statistical analysis
Comparisons between groups were performed with
Student's t-test and χ2 test. OS and DFS curves were
plotted for the patients according to the Kaplan-Meier method, with
the log-rank test used for comparison. Survival was recorded from
the day of the surgery in which specimens were extracted.
Differences were considered statistically significant at P<0.05.
The statistical analyses were performed using SPSS 16.0 software
(SPSS, Inc., Chicago, IL, USA).
Results
miR-124-3p affects cell viability and
plate colony formation in vitro and tumor growth in vivo in gastric
cancer cells
Previous studies demonstrated that miR-124-3p was
downregulated in gastric cancer tissues and cell lines,
particularly in SGC-7901 cells, while it was relatively highly
expressed in MKN-28 cells. Therefore, the present study transfected
SGC-7901 cells with miR-124-3p mimics (miR-124) and MKN-28 cells
with miR-124-3p inhibitor (miR-124-LNA) to investigate the changes
in cell viability and colony formation abilities in vitro
and tumor growth abilities in nude mice.
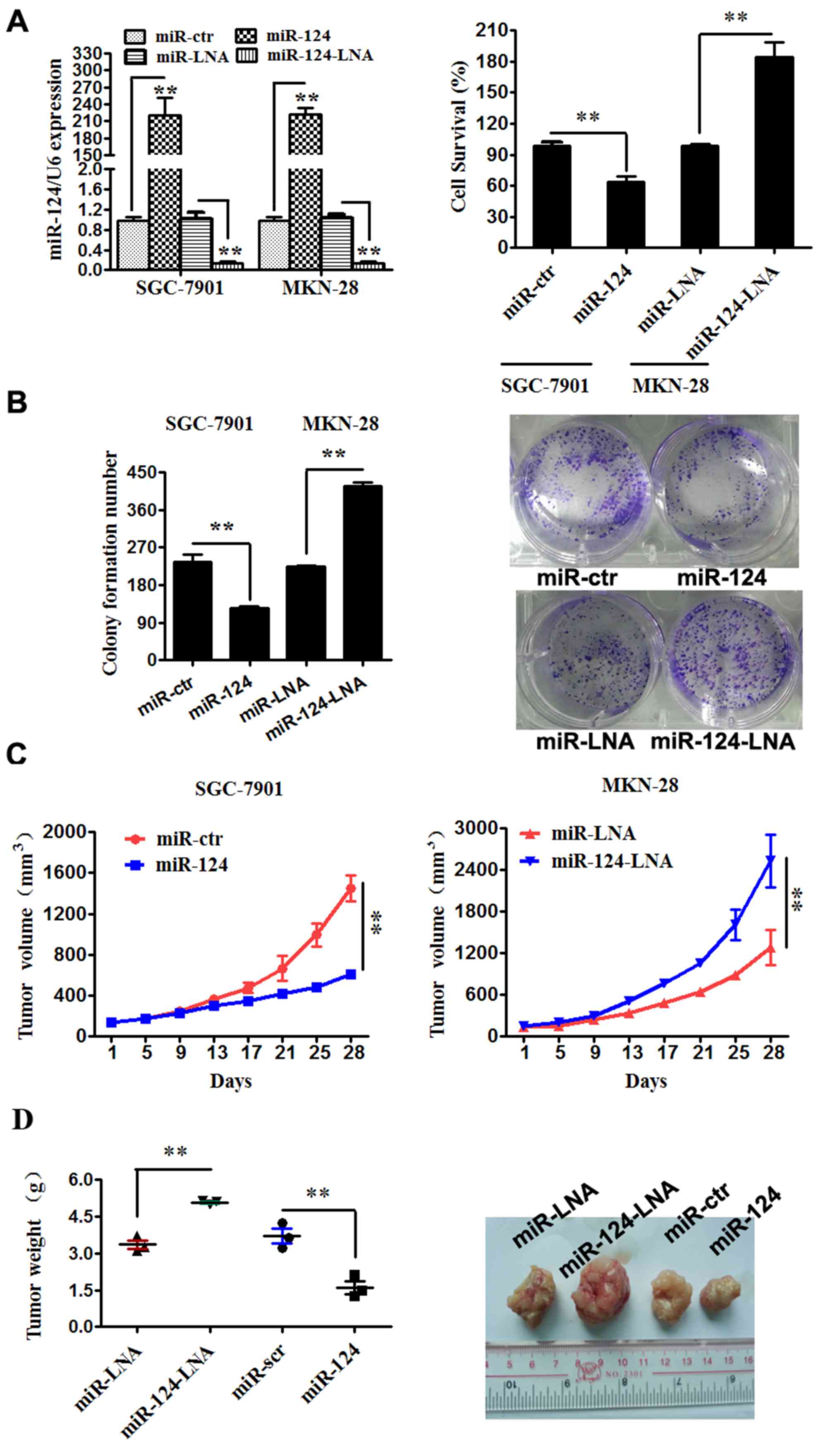
Firstly, SGC-7901 and MKN-28 cells were respectively
transfected with miR-124-3p mimics, miR-124-3p inhibitor and the
corresponding controls, and the transfection was determined to be
successful (Fig. 1A). The results of
subsequent assays indicated that miR-124-3p significantly
suppressed SGC-7901 cell viability (P<0.01; Fig. 1A), plate colony formation (P<0.01;
Fig. 1B) and tumor growth in nude
mice (P<0.01; Fig. 1C and D). By
contrast, miR-124-3p inhibitor promoted MKN-28 cell proliferation
(P<0.01; Fig. 1A), plate colony
formation (P<0.01; Fig. 1B) and
tumor growth in nude mice (P<0.01; Fig. 1C and D).
miR-124-3p is downregulated and
correlates with advanced clinical stage, lymph node metastases and
poor clinical outcomes in gastric cancer
ISH was performed to evaluate miR-124-3p levels in
121 gastric tumors with a TMA. The clinical samples were divided
into low expression and high expression groups based on miR-124-3p
expression scores greater or less than 2 (14). The results indicated that miR-124-3p
was markedly downregulated in stomach tumors, with low miR-124-3p
expression determined in 57.9% (70/121) of the specimens.
Subsequently, the potential clinicopathological implications of
altered miR-124-3p expression were determined. It was observed that
miR-124-3p expression was inversely associated with histological
grade, TNM stage and lymph node metastasis (P=0.037, P=0.005 and
P=0.002, respectively). However, no significant associations
between miR-124-3p expression and age, gender or T stage were
identified (Table I).
 | Table I.Analysis of the association between
miR-124 expression and clinicopathological parameters in primary
gastric cancer. |
Table I.
Analysis of the association between
miR-124 expression and clinicopathological parameters in primary
gastric cancer.
|
|
| miR-124
expression |
|---|
|
|
|
|
|---|
| Variable | Cases, n | Low | High | P-value |
|---|
| Age, years |
|
|
|
|
|
<60 | 69 | 40 | 29 | 1.000 |
|
≥60 | 52 | 30 | 22 |
|
| Gender |
|
|
|
|
|
Male | 65 | 41 | 24 | 0.268 |
|
Female | 56 | 29 | 27 |
|
| Histological grade
(differentiation) |
|
|
|
|
| Well
and moderate | 31 | 23 | 8 | 0.037 |
| Poor
and other | 90 | 47 | 43 |
|
| T stage |
|
|
|
|
|
T1-T2 | 66 | 34 | 32 | 0.142 |
|
T3-T4 | 55 | 36 | 19 |
|
| TNM stage |
|
|
|
|
|
I–II | 46 | 19 | 27 | 0.005 |
|
III–IV | 75 | 51 | 24 |
|
| Lymph node
metastasis |
|
|
|
|
|
Present | 88 | 59 | 29 | 0.002 |
|
Absent | 33 | 11 | 22 |
|
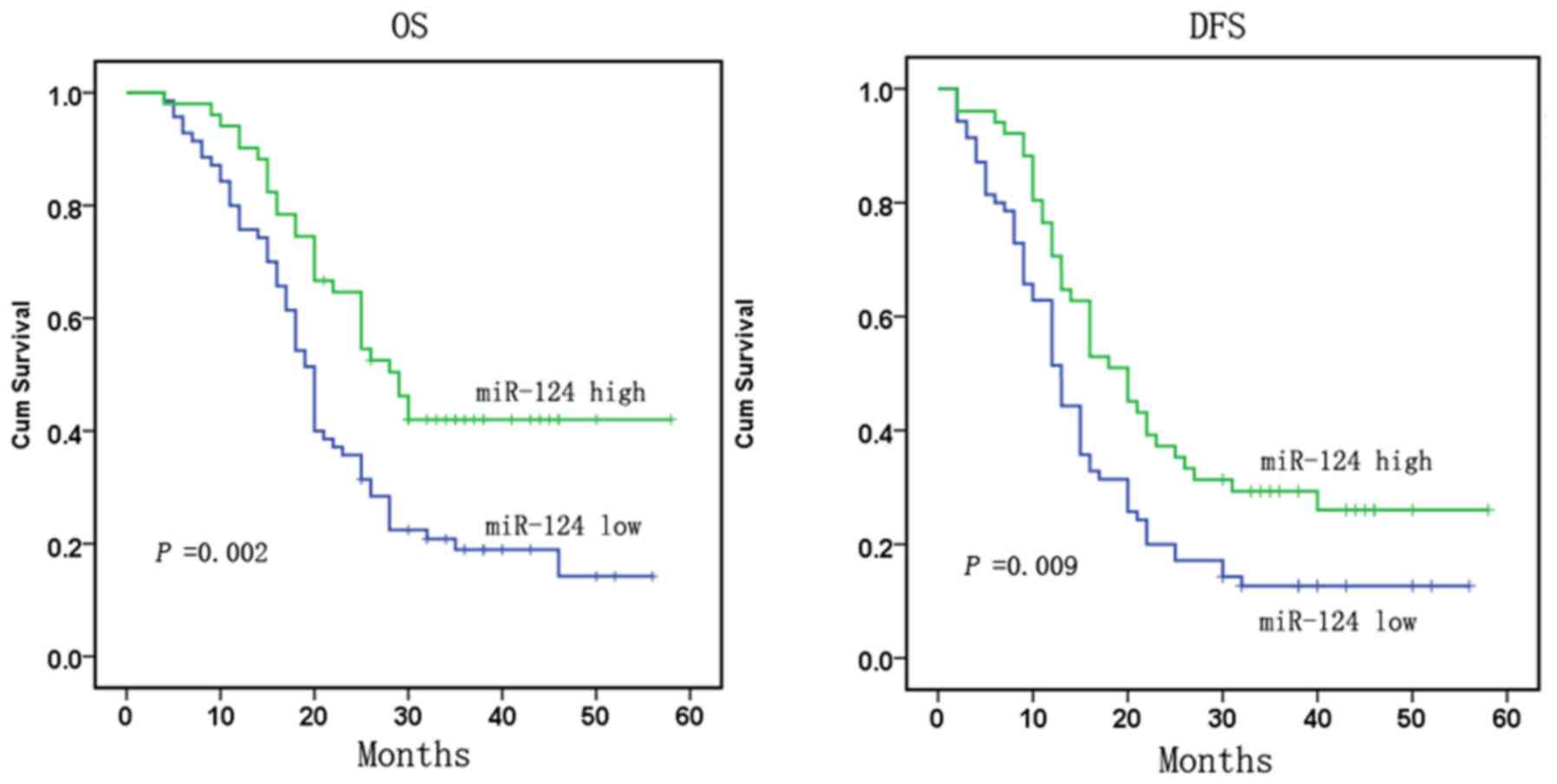
To analyze the significance of
miR-124-3p in terms of clinical prognosis, Kaplan-Meier survival
analysis was performed based on patient OS and DFS rates
The results demonstrated that patients with low
miR-124-3p expression had lower mean OS and DFS rates compared with
patients with high miR-124-3p expression (P=0.002 for OS and
P=0.009 for DFS; Fig. 2).
Collectively these results suggest that miR-124-3p may serve
critical roles in the carcinogenesis and progression of gastric
cancer.
SP1 and Rac1 are direct targets of
miR-124-3p
To investigate the molecular mechanism of miR-124-3p
in gastric cancer, the TargetScan algorithm (http://www.targetscan.org/vert_71/) was used to search
for putative protein-coding gene targets of miR-124, which
indicated that SP1 and Rac1 are putative targets of miR-124-3p.
Rac1 has previously been identified as a target of miR-124
(17). To ascertain whether SP1 is a
direct target of miR-124-3p, luciferase reporter assays were
conducted. The full-length SP1 3′-UTR was cloned downstream of the
firefly luciferase gene and cotransfected with miR-124-3p mimics,
miR-124 inhibitor or their negative controls, and luciferase
activity was measured at 48 h post-transfection. SGC-7901 cells
cotransfected with SP1 reporter constructs and miR-124-3p exhibited
an approximate 44% reduction in luciferase activity with respect to
those cotransfected with the scrambled oligonucleotide control
(P<0.05). Conversely, miR-124-LNA increased luciferase
responsiveness compared with its control (P<0.05). In turn,
mutation of the putative miR-124-3p sites in the 3′-UTR of SP1
reduced luciferase responsiveness to miR-124 (Fig. 3A).
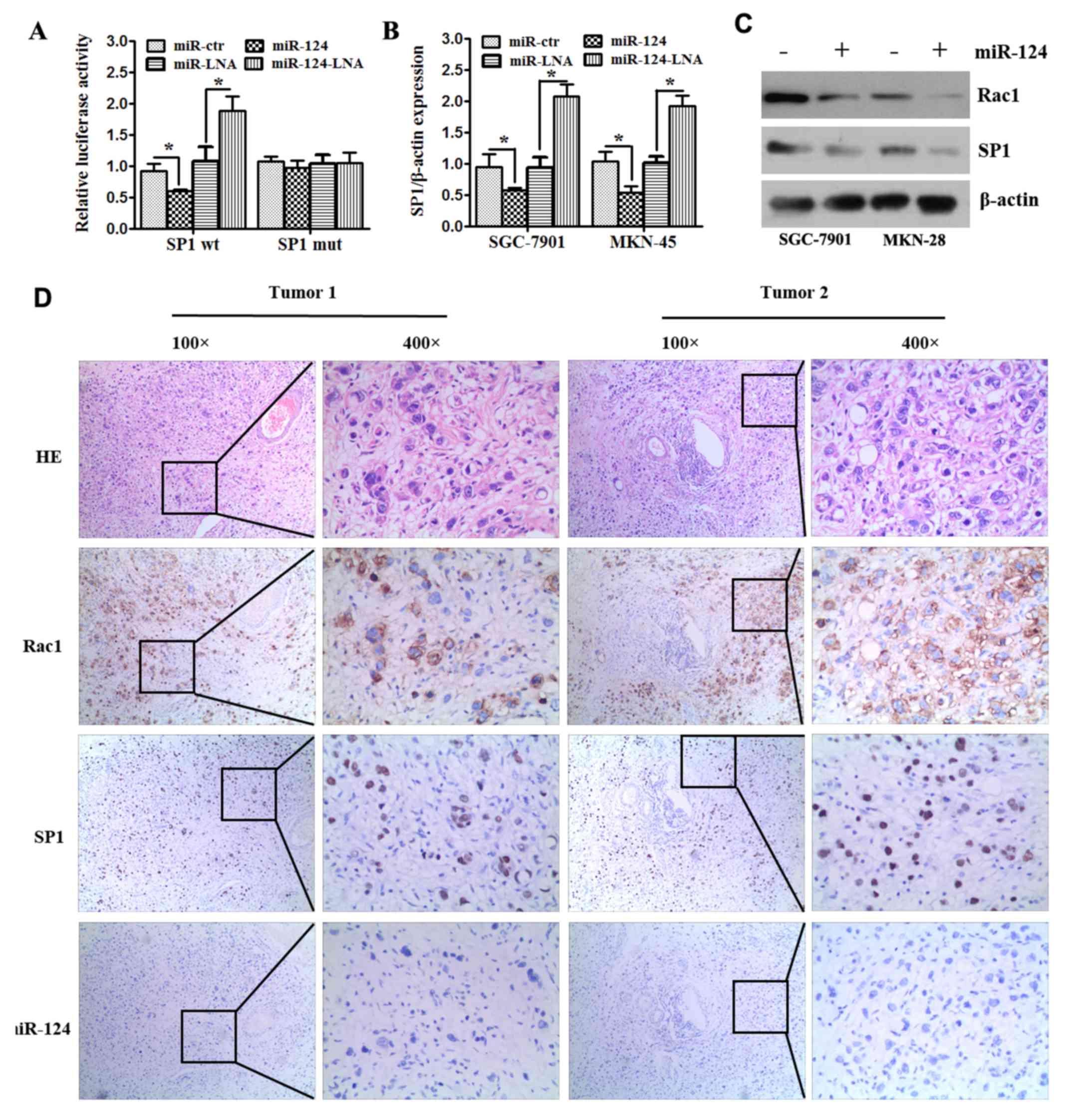 | Figure 3.SP1 and Rac1 are direct targets of
miR-124-3p. (A) Luciferase assay of SGC-7901 and MKN-28 cells
cotransfected with miR-124 mimics, miR-124 inhibitor or
corresponding controls and a luciferase reporter containing SP1
3′-untranslated region (SP1-wt) or mutant constructs in which the
first four nucleotides of the miR-124 binding site were mutated
(SP1-mut). (B) SGC-7901 and MKN-28 cells were transfected with
miR-124 mimics, miR-124 inhibitor or controls; miR-124
overexpression inhibited the mRNA expression of SP1. All data are
presented as means ± SEM. *P<0.05. (C) SGC-7901 and MKN-28 cells
were transfected with miR-124 or scramble; miR-124 overexpression
inhibited the protein expression of SP1 and Rac1. (D) Gastric
cancer specimens were analyzed by immunohistochemistry and in
situ hybridization staining. Representative images of
miR-124-3p, SP1 and Rac1 expression and H&E staining are shown
(magnification, ×100, ×400). SP1, specificity protein 1; Rac1,
Ras-related C3 botulinum toxin substrate 1; miR, microRNA; -wt,
wild type; -mut; mutant; -LNA, inhibitor; -ctr, control; H&E,
hematoxylin and eosin. |
To confirm SP1 and Rac1 as targets of miR-124,
miR-124-3p mimics, miR-124 inhibitor or their scramble controls
were transfected into SGC-7901 and MKN-28 cells, and RT-qPCR and
western blot analysis were performed to detect the expression of
SP1 and Rac1. Following transfection with miR-124 mimics, the
results indicated a significant reduction in the mRNA levels of SP1
in SGC-7901 and MKN-28 cells (P<0.05; Fig. 3B), and a marked reduction in the
protein levels of SP1 and Rac1 in SGC-7901 and MKN-28 cells
(Fig. 3C). By contrast, following
transfection with miR-124 inhibitor, the results indicated a
significant increase in the mRNA levels of SP1 in SGC-7901 and
MKN-28 cells (P<0.05; Fig. 3B).
Collectively these results indicate that miR-124-3p downregulates
SP1, and likely Rac1, by targeting their mRNA 3′UTR. Additionally,
when investigating the clinical relevance of miR-124-3p and its
target genes SP1 and Rac1 in gastric cancer tissues by IHC, it was
observed that miR-124-3p expression was negatively associated with
SP1 and Rac1 expression in randomly selected gastric cancer
sections (Fig. 3D).
miR-124-3p represses carcinogenesis in
gastric cancer by targeting SP1 and Rac1
When investigating the functional effect of
miR-124-3p and SP1/Rac1 on gastric cancer cells, the previous
assays indicated that upregulation of miR-124-3p inhibited the
viability of SGC-7901 and MKN-28 cells through functional
downregulation of SP1 or/and Rac1 expression. Knockdown and rescue
experiments were subsequently performed, in which SGC-7901 and
MKN-28 cells were transfected with sh-ctr, sh-SP1, sh-Rac1, vector,
SP1 vector or Rac1 vector, and the effects on miR-124 expression
confirmed by RT-qPCR (Fig. 4A). As
depicted in Fig. 4B, the rate of cell
survival was considerably lower in cells transfected with
miR-124-3p mimics and SP1 and/or Rac1 vector compared with
respective controls. By contrast, downregulation of miR-124-3p by
miR-124-LNA or upregulation of SP1 and/or Rac1 by overexpression
vectors significantly increased cell survival rate compared with
the controls (Fig. 4B and C;
P<0.05). These results indicated that transfection of miR-124-3p
and knockdown of SP1 and/or Rac1 significantly suppressed the
growth of gastric cancer cells in vitro.
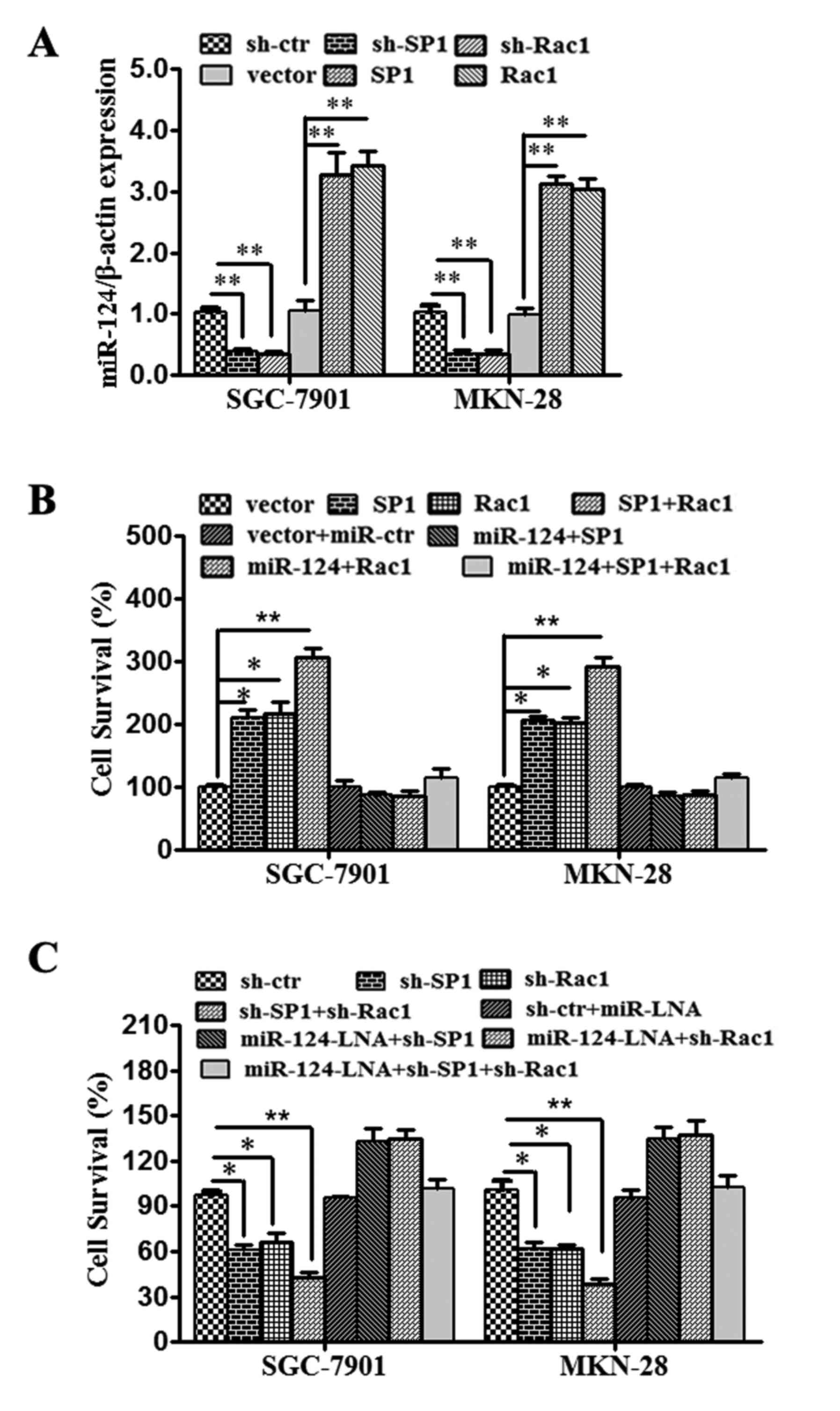 | Figure 4.miR-124-3p represses carcinogenesis
in gastric cancer by targeting SP1 and Rac1. (A) SGC-7901 and
MKN-28 cells were respectively transfected with sh-ctr, sh-SP1,
sh-Rac1, vector, SP1 vector or Rac1 vector; all the transfections
were successful. (B) SGC-7901 and MKN-28 cells were respectively
transfected with vector, SP1, Rac1, Sp1+Rac1, vector+miR-ctr,
miR-124+SP1, miR-124+Rac1 or miR-124+SP1+Rac1, and the viability of
the cells was analyzed. (C) SGC-7901 and MKN-28 cells were
respectively transfected with sh-ctr, sh-SP1, sh-Rac1,
sh-Sp1+sh-Rac1, sh-ctr+miR-LNA, miR-124-LNA+sh-SP1,
miR-124-LNA+sh-Rac1 or miR-124-LNA+sh-SP1+sh-Rac1, and the
viability of the cells was analyzed. All data are presented as
means ± SEM. **P<0.01, *P<0.05. SP1, specificity protein 1;
Rac1, Ras-related C3 botulinum toxin substrate 1; miR, microRNA;
sh-, short hairpin; -LNA, inhibitor; -ctr, control. |
Discussion
miR-124-3p commonly serves integral roles in cell
growth, apoptosis, invasion and metastasis, and serves as a tumor
suppressor in various cancers (5,8,9). In the present study, the downregulation
of miR-124-3p was indicated to be a frequent event in gastric
cancer. Furthermore, low-level expression of miR-124-3p was
significantly associated with clinical stage and lymph node
metastases. Accordingly, Kaplan-Meier survival analysis indicated
that patients whose primary tumors exhibited low-level expression
of miR-124-3p had shorter OS and RFS rates. In previous studies by
our group, it was observed that miRs-200b, −200c and −26a may be
prognostic indicators in gastric cancer (14,18). Here,
miR-124-3p was indicated as a useful prognostic marker for
predicting survival and relapse in patients with gastric
cancer.
While the initial results indicated that miR-124-3p
acted as a tumor suppressor in gastric cancer, the underlying
molecular mechanisms were unknown. Therefore, Rac1 and SP1 were
assessed are potential downstream target genes of miR-124.
miR-124-3p was determined to bind to complementary sites in the
3′-UTRs of Rac1 and SP1, and induce a significant reduction in the
expression levels of Rac1 and SP1. Rac1 is an established GTPase
that belongs to the RAS superfamily of small GTP-binding proteins
(19). Previous studies have reported
that Rac1 expression was frequently increased in colon, breast and
gastric cancers, medulloblastoma and lung cancer (20–24).
Results also support that Rac1 is implicated in cytoskeletal
rearrangements and in the regulation of multiple cancer-associated
cellular phenotypes, including cell growth, invasion and
metastasis, as well as angiogenesis (25–27). Vader
et al (28) demonstrated that
silencing of Rac1 in vascular endothelial cells inhibited vascular
endothelial growth factor-mediated tube formation as well as
endothelial cell migration, invasion and proliferation in
vitro. In addition, Yoshida et al (25) observed that selective inhibition of
Rac1 activity by the inhibitor NSC23766 suppressed cell growth and
induced apoptosis in different breast cancer cell lines without
being toxic to normal mammary epithelial cells. SP1, a zinc finger
transcription factor, has been reported as upregulated in cervical,
lung, gastric, oral squamous and colon cancers, and has been
implicated in cell proliferation, migration, apoptosis and
angiogenesis through regulation of a variety of cancer-associated
genes (29–33). In the present study, Rac1 and SP1 were
upregulated and negatively associated with miR-124-3p levels in
gastric cancer tissues. Furthermore, Rac1 and SP1 overexpression
rescued the inhibitory effect of miR-124 on cell survival.
Collectively these results demonstrated that miR-124-3p may inhibit
gastric cancer growth by targeting Rac1 and SP1.
Overall, the present study identified the potential
role of miR-124-3p in gastric cancer progression and as an
independent predictor of OS and RFS. The data further indicated
that miR-124-3p suppressed gastric cancer growth by regulating Rac1
and SP1. These findings suggest miR-124-3p may be employed as a
novel prognostic biomarker, as an indicator of treatment strategy
and/or as a potential therapeutic target in gastric cancer.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Science and
Technology Department of Hunan Province, China (grant no.
2018JJ6071), the Education Department of Hunan Province, China
(grant no. 16C1426) and the Science and Technology Department of
Hengyang, China (grant no. 2015KJ16).
Availability of data and materials
All data generated or analyzed in this study are
included in this article.
Authors' contributions
FL and LX designed the study. FL, HH and JZ
performed experiments and collected and analyzed data. FL, LT, XA
and ZZ wrote, reviewed and revised manuscript. All authors read and
approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that there are no conflicts of
interest regarding the publication of this study.
References
|
1
|
He L and Hannon GJ: MicroRNAs: Small RNAs
with a big role in gene regulation. Nat Rev Genet. 5:522–531. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Slack FJ and Weidhaas JB: MicroRNA in
cancer prognosis. N Engl J Med. 359:2720–2722. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Cheng CJ and Slack FJ: The duality of
oncomiR addiction in the maintenance and treatment of cancer.
Cancer J. 18:232–237. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Li L, Zhou L, Li Y, Lin S and Tomuleasa C:
MicroRNA-21 stimulates gastric cancer growth and invasion by
inhibiting the tumor suppressor effects of programmed cell death
protein 4 and phosphatase and tensin homolog. J BUON. 19:228–236.
2014.PubMed/NCBI
|
|
5
|
Li L, Luo J, Wang B, Wang D, Xie X, Yuan
L, Guo J, Xi S, Gao J, Lin X, et al: MicroRNA-124 targets
flotillin-1 to regulate proliferation and migration in breast
cancer. Mol Cancer. 12:1632013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Park SY, Kim H, Yoon S, Bae JA, Choi SY,
Jung YD and Kim KK: KITENIN-targeting microRNA-124 suppresses
colorectal cancer cell motility and tumorigenesis. Mol Ther.
22:1653–1664. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kang S, Zhao Y, Hu K, Xu C, Wang L, Liu J,
Yao A, Zhang H and Cao F: miR-124 exhibits antiproliferative and
antiaggressive effects on prostate cancer cells through PACE4
pathway. Prostate. 74:1095–1106. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Xie L, Zhang Z, Tan Z, He R, Zeng X, Xie
Y, Li S, Tang G, Tang H and He X: MicroRNA-124 inhibits
proliferation and induces apoptosis by directly repressing EZH2 in
gastric cancer. Mol Cell Biochem. 392:153–159. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zheng F, Liao YJ, Cai MY, Liu YH, Liu TH,
Chen SP, Bian XW, Guan XY, Lin MC, Zeng YX, et al: The putative
tumour suppressor microRNA-124 modulates hepatocellular carcinoma
cell aggressiveness by repressing ROCK2 and EZH2. Gut. 61:278–289.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhang H, Wang Q, Zhao Q and Di W: miR-124
inhibits the migration and invasion of ovarian cancer cells by
targeting SphK1. J Ovarian Res. 6:842013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
An L, Liu Y, Wu A and Guan Y: microRNA-124
inhibits migration and invasion by down-regulating ROCK1 in glioma.
PLoS One. 8:e694782013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Xia J, Wu Z, Yu C, He W, Zheng H, He Y,
Jian W, Chen L, Zhang L and Li W: miR-124 inhibits cell
proliferation in gastric cancer through down-regulation of SPHK1. J
Pathol. 227:470–480. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Capes-Davis A, Theodosopoulos G, Atkin I,
Drexler HG, Kohara A, MacLeod RA, Masters JR, Nakamura Y, Reid YA,
Reddel RR, et al: Check your cultures! A list of cross-contaminated
or misidentified cell lines. Int J Cancer. 127:1–8. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Tang H, Deng M, Tang Y and Xie X, Guo J,
Kong Y, Ye F, Su Q and Xie X: miR 200b and miR 200c as prognostic
factors and mediators of gastric cancer cell progression. Clin
Cancer Res. 19:5602–5612. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) Method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Liu A, Yu Q, Peng Z, Huang Y, Diao S,
Cheng J, Wang W and Hong M: miR-200b inhibits CD133+
glioma cells by targeting the AKT pathway. Oncol Lett.
13:4701–4707. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hu CB, Li QL, Hu JF, Zhang Q, Xie JP and
Deng L: miR-124 inhibits growth and invasion of gastric cancer by
targeting ROCK1. Asian Pac J Cancer Prev. 15:6543–6546. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Deng M, Tang HL, Lu XH, Liu MY, Lu XM, Gu
YX, Liu JF and He ZM: miR-26a suppresses tumor growth and
metastasis by targeting FGF9 in gastric cancer. PLoS One.
8:e726622013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Jennings RT and Knaus UG: Rho family and
Rap GTPase activation assays. Methods Mol Biol. 1124:79–88. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zhao H, Dong T, Zhou H, Wang L, Huang A,
Feng B, Quan Y, Jin R, Zhang W, Sun J, et al: miR-320a suppresses
colorectal cancer progression by targeting Rac1. Carcinogenesis.
35:886–895. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Dokmanovic M, Hirsch DS, Shen Y and Wu WJ:
Rac1 contributes to trastuzumab resistance of breast cancer cells:
Rac1 as a potential therapeutic target for the treatment of
trastuzumab-resistant breast cancer. Mol Cancer Ther. 8:1557–1569.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhan H, Liang H, Liu X, Deng J, Wang B and
Hao X: Expression of Rac1, HIF-1α, and VEGF in gastric carcinoma:
Correlation with angiogenesis and prognosis. Onkologie. 36:102–107.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chen B, Gao Y, Jiang T, Ding J, Zeng Y, Xu
R and Jiang X: Inhibition of tumor cell migration and invasion
through knockdown of Rac1 expression in medulloblastoma cells. Cell
Mol Neurobiol. 31:251–257. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Gastonguay A, Berg T, Hauser AD, Schuld N,
Lorimer E and Williams CL: The role of Rac1 in the regulation of
NF-κB activity, cell proliferation, and cell migration in non-small
cell lung carcinoma. Cancer Biol Ther. 13:647–656. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Yoshida T, Zhang Y, Rivera Rosado LA, Chen
J, Khan T, Moon SY and Zhang B: Blockade of Rac1 activity induces
G1 cell cycle arrest or apoptosis in breast cancer cells through
downregulation of cyclin D1, survivin, and X-linked inhibitor of
apoptosis protein. Mol Cancer Ther. 9:1657–1668. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Rathinam R, Berrier A and Alahari SK: Role
of Rho GTPases and their regulators in cancer progression. Front
Biosci. 16:2561–2571. 2011. View
Article : Google Scholar
|
|
27
|
Bid HK, Roberts RD, Manchanda PK and
Houghton PJ: RAC1: An emerging therapeutic option for targeting
cancer angiogenesis and metastasis. Mol Cancer Ther. 12:1925–1934.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Vader P, van der Meel R, Symons MH, Fens
MH, Pieters E, Wilschut KJ, Storm G, Jarzabek M, Gallagher WM,
Schiffelers RM, et al: Examining the role of Rac1 in tumor
angiogenesis and growth: A clinically relevant RNAi-mediated
approach. Angiogenesis. 14:457–466. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhang J, Li S, Yan Q, Chen X, Yang Y, Liu
X and Wan X: Interferon-β induced microRNA-129-5p down-regulates
HPV-18 E6 and E7 viral gene expression by targeting SP1 in cervical
cancer cells. PLoS One. 8:e813662013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yang WB, Chen PH, Hsu T, Fu TF, Su WC,
Liaw H, Chang WC and Hung JJ: Sp1-mediated microRNA-182 expression
regulates lung cancer progression. Oncotarget. 5:740–753. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Qiu T, Zhou X, Wang J, Du Y, Xu J, Huang
Z, Zhu W, Shu Y and Liu P: miR-145, miR-133a and miR-133b inhibit
proliferation, migration, invasion and cell cycle progression via
targeting transcription factor Sp1 in gastric cancer. FEBS Lett.
588:1168–1177. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kim DW, Ko SM, Jeon YJ, Noh YW, Choi NJ,
Cho SD, Moon HS, Cho YS, Shin JC, Park SM, et al:
Anti-proliferative effect of honokiol in oral squamous cancer
through the regulation of specificity protein 1. Int J Oncol.
43:1103–1110. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zhao Y, Zhang W, Guo Z, Ma F, Wu Y, Bai Y,
Gong W, Chen Y, Cheng T, Zhi F, et al: Inhibition of the
transcription factor Sp1 suppresses colon cancer stem cell growth
and induces apoptosis in vitro and in nude mouse xenografts.
Oncol Rep. 30:1782–1792. 2013. View Article : Google Scholar : PubMed/NCBI
|