Introduction
Elevated intraocular pressure (IOP) is a major cause
for glaucoma. Aqueous humour outflow mostly occurs via the
conventional outflow pathway, i.e. the trabecular meshwork (TM) and
Schlemm's canal. Increased resistance in the TM to aqueous humour
outflow leads to elevation of IOP in patients with primary
open-angle glaucoma (POAG) (1,2). It is
considered that increases in the resistance of aqueous humour
outflow through the conventional pathway in TM may be caused by
overexpression of matricellular proteins and substantial
enhancement of the fibrillar extracellular matrix (ECM) and
cell-cell interactions (3–12). However, at present, no effective
treatment for POAG targeting this conventional pathway is available
(13).
In addition, TM cells possess smooth muscle
cell-like properties. TM cell contraction and relaxation affects
the intercellular space, the state of cell-cell junctions and
cell-ECM interactions, and consequently modulates aqueous outflow
through the TM tissue. The actomyosin system, composed of actin
microfilaments and associated proteins, is presented in essentially
all cells, as well as highly organized in TM cells (10). The actomyosin system has a critical
role in regulating cellular morphology, volume, contractility,
adhesion to adjacent cells and ECM production. Such modifications
modulate the hydrodynamics of the aqueous humour outflow pathway at
the cellular level, which is supported by the findings that
actin-disrupting agents or inhibitors of specific protein kinases
regulate these parameters directly or indirectly (14–16).
It is well known that statins prevent the
progression of coronary artery disease, stroke and neuronal cell
death after ischemic injury (17–21).
Statins indirectly inhibit the synthesis of Rho guanine
triphosphatase (GTPase) to affect the organization of the actin
cytoskeleton (22), affect aqueous
outflow associated with a reduction in membrane-bound Rho GTPase
levels, a decrease in myosin light-chain (MLC) phosphorylation and
changes in TM cell shape (23). Rho
GTPase has an important role in actin cytoskeletal organization and
cell adhesion via modulation of MLC phosphorylation.
(24–27) Activation of the Rho/Rho-associated
protein kinase (ROCK) pathway results in TM contraction. By
contrast, inhibition of the Rho/ROCK pathway causes relaxation of
TM with a subsequent increase in outflow facility and reduction in
IOP (28).
Statins also reduce the risk of POAG in
hyperlipidemia patients (29), of
note, they were reported to lower the frequency of glaucoma
progression via a mechanism that has remained elusive (30). Thus, it is of great importance to
understand the underlying mechanism of the protective action of
statins on glaucoma. It is thought that the effect of statins to
improve glaucoma progression may be secondary to their effects on
blood lipids to reduce IOP (31).
Atorvastatin is the most frequently used statin for lipid control
and may also have applications in other conditions (32–34).
However, the role of atorvastatin in modifying IOP and aqueous
humour outflow has remained to be fully elucidated. The aim of the
present study was to determine the effect of atorvastatin on
pathways regulating aqueous outflow and TM cells.
Materials and methods
Reagents and apparatus
Atorvastatin, purchased from Sigma-Aldrich (Merck
KGaA, Darmstadt, Germany), was dissolved in DMSO (Sigma-Aldrich;
Merck KGaA) at 0.05 mol/l as a stock. The perfusion pump system was
purchased from Harvard Bioscience (Holliston, MA, USA). Fluorescein
isothiocyanate (FITC)-phalloidin (catalogue number: 40735ES75) was
purchased from Yeasen (Shanghai, China). Anti-vinculin antibody
(catalogue number: BM4051) and anti-β-catenin antibody (catalogue
number: BM3905) were purchased from Boster Biological Technology
(Wuhan, China). High-glucose Dulbecco's modified Eagle's medium
(DMEM) and fetal bovine serum (FBS) were obtained from Gibco
(Thermo Fisher Scientific, Inc., Waltham, MA, USA). A Cell Counting
Kit-8 (CCK-8) was obtained from Dojindo Molecular Technologies,
Inc. (Kumomoto, Japan). Oligonucleotide primers for
reverse-transcription quantitative polymerase chain reaction
(RT-qPCR) were designed and synthesized by BioTNT Corp. (Shanghai,
China).
Perfusion study
A total of 90 fresh enucleated porcine eyes from 4
to 5-month-old pigs were obtained from Jiading Town Abattoir
(Shanghai, China) within 4 h after death for perfusion and TM-cell
isolation. Atorvastatin stock solution was diluted with Dulbecco's
PBS containing 5.5 mM glucose (GPBS). The eyes were randomly
divided into five treatment groups (n=6 each): Atorvastatin (17,
50, 100 and 200 µM) containing dimethyl sulfoxide (DMSO) (0.38%),
plus GPBS containing the same concentration of DMSO as the control
group. Animal procedures were in compliance with the Statement for
the Use of Animals in Ophthalmic and Vision Research by the US
Association for Research in Vision and Ophthalmology. The protocol
was approved by the Institutional Animal Use and Care Committee in
Fudan University.
Perfusion of enucleated porcine eyes was performed
as previously reported (35). Fresh
porcine eyes were cleaned off extraocular tissue and the posterior
segment was submerged to the limbus in PBS at 34°C. A 21-gauge
infusion needle was inserted through the peripheral cornea into the
anterior chamber. This needle was carefully threaded through the
pupil and the needle tip was positioned within the posterior
chamber to prevent deepening of the anterior chamber that would
have otherwise led to an artificial increase in outflow facility.
The infusion needle was connected to polyethylene tubing, which was
connected via a 3-way valve to i) a drug (or control) solution
reservoir elevated relative to the eye to generate a 15-mmHg
hydrostatic pressure, and ii) a syringe containing perfusion
solution (drug or GPBS) on a perfusion pump, and simultaneously a
pressure sensor in parallel. The IOP in the system as monitored by
the pressure sensor was recorded using a computer. The perfusion
rate was regulated by adjusting the perfusion pump to maintain a
constant pressure of 15 mmHg. The computer also recorded the
perfusion rate (F in µl/min). In addition, a second needle was
inserted intracamerally into the anterior chamber and connected to
a fluid collection chamber. During baseline perfusion, connections
to the (drug or control solution) perfusion pump and the second
(collection) needle were closed. All eyes were perfused for 30 min
at a constant pressure of 15 mmHg and the initial baseline aqueous
outflow facility (C1) was recorded. The C value was
obtained by dividing the perfusion flow rate by the corresponding
intraocular pressure (C1=F/IOP at baseline) as described
previously (3). F and IOP were
constantly measured at 10 Hz. C was calculated by averaging the
values over a 10-sec window and electronically recorded every 10
sec by LabView version 7.1 (National Instruments, Austin, TX, USA).
After 30 min, the anterior chamber content was replaced with 5 ml
atorvastatin or control solution. Solution exchange was performed
by stopping the perfusion pump and opening the connections to the
drug (or control) solution reservoir and the second (collection)
needle to allow the solution in the drug reservoir to flow into the
eye by gravity. After exchange of 5 ml solution, connections to the
reservoir containing the drug and the second (collection) needle
were closed again and the perfusion pump restarted. A stable
outflow facility was then obtained and recorded as C2.
Total time of the perfusion was controlled within 2 h. The
difference value (C2-C1) was reported as ΔC.
The percentage change from the baseline was recorded as
(ΔC/C1).
Cell culture
Primary porcine TM cells were pooled from fresh
porcine eyes (n=20) via the tissue adherence method (36). TM cells were identified by
immunofluorescence analysis of the presence of fibronectin, laminin
and vimentin. TM cells were cultured in high-glucose DMEM
supplemented with 10% FBS, 10,000 U/ml streptomycin and 10,000 U/ml
penicillin. Cells were maintained at 37°C in a humidified
atmosphere containing 5% CO2 and cells of the passages
3–5 were used in the present study.
Atorvastatin treatment and
cytoskeletal staining
Fresh solutions of 10, 25, 50, 100 and 200 µM
atorvastatin were prepared in culture medium, whereas the control
group was treated with vehicle only. The final concentration of
DMSO was kept below 0.5% in the culture medium.
TM cells were cultured to confluence on 2%
gelatin-coated glass coverslips and treated with atorvastatin
(10–200 µM) or vehicle for 24 h. Cellular morphological changes
were recorded with a phase-contrast microscope (ECLIPSE Ni-U;
Nikon, Tokyo, Japan). Following drug treatment for 24 h, the cells
were fixed with 4% paraformaldehyde and permeabilized with 0.3%
Triton X-100 in PBS, and subsequently blocked in 10% bovine serum
albumin (Sigma-Aldrich; Merck KGaA) for 1 h. The cells were
labelled with FITC-phalloidin (1:200 dilution) and primary antibody
against vinculin or β-catenin (both 1:100 dilution) for 2 h at room
temperature. After incubation with tetramethylrhodamine-labeled
secondary antibody (Jackson ImmunoResearch Labs; West Grove, PA,
USA; catalogue number: 111-025-003, 1:100 dilution) for 1 h at room
temperature, coverslips were stained with DAPI for 1 min and
observed with a fluorescence microscope (Nikon).
Cell viability
CCK-8 was used to evaluate the effect of
atorvastatin on the viability of TM cells. At 6, 12, 24 and 48 h
after treatment with atorvastatin (50–200 µM) or vehicle, 10 µl
CCK-8 solution was added to 100 µl TM cells per well
(~3×103 cells) in 96-well plates, cells were maintained
at 37°C in a humidified atmosphere containing 5% CO2 for
2 h. Optical density (OD) at 450 nm was then measured using a
microplate reader (BioTek, Winooski, VT, USA). The OD value was
used for quantification of cell proliferation at the different
dosages of atorvastatin as well as different time-points, following
the manufacturer's protocol.
RT-qPCR
To determine the effect of atorvastatin on the
expression of vinculin and β-catenin, total RNA from atorvastatin
(50–200 µM) or vehicle-treated 0–48 h TM cells was isolated using
TRIzol reagent (Invitrogen; Thermo Fisher Scientific, Inc.) and
reverse-transcribed to complementary (c)DNA using a First-Strand
cDNA synthesis kit (BioTNT, Shanghai, China; catalogue number:
A2010B0B01) according to the manufacturer's protocol. The cDNA was
then amplified by PCR using sequence-specific forward and reverse
oligonucleotide primers: Vinculin, forward
5′-ACCAGGCTCCCAAGACCCAT-3′ and reverse 5′-CAGGCGAGTCAGCAGCAACA-3′;
β-catenin, forward 5′-GAGGACAAGCCCCAGGATTA-3′ and reverse
5′-AGCAGTCTCATTCCAAGCCA-3′; and GAPDH, forward
5′-CGGAGTCAACGGATTTGGTCGTAT-3′ and reverse
5′-AGCCTTCTCCATGGTGGTGAAGAC-3′. A real time qPCR kit (BioTNT,
Shanghai, China; catalogue number: A2010A001) was used according to
the manufacturer's protocol to create the PCR reaction mixtures
composed of buffers, dNTP, heat activated Taq DNA polymerase
mixtures, MgCl2 solution and premixed SYBR GREEN dyes.
Amplification was performed at 95°C, 5 min; 95°C, 5 sec, 60°C, 30
sec for 40 cycles. mRNA expression was analyzed on the ABI 7500
qRT-PCR System (Applied Biosystems; Thermo Fisher Scientific, Inc.)
(37).
Western blot analysis
Lysates of TM cells treated with atorvastatin
(50–200 µM) or vehicle for 0–48 h were prepared using
radioimmunoprecipitation assay buffer (Beyotime Institute of
Biotechnology, Shanghai, China) containing phenylmethylsulfonyl
fluoride, a protease inhibitor. Protein concentration was
determined using the BCA assay (Yeasen, Shanghai, China; catalogue
number: 20201ES76) according to the manufacturer's protocol using
bovine serum albumin as a standard. Protein extracts (20 µg
protein/lane) were separated by SDS-PAGE (10% acrylamide) and
transferred onto a polyvinylidene fluoride membrane (EMD Millipore;
Billerica, MA, USA). The membrane was then probed overnight using
antibodies specifically directed against vinculin (Boster
Biological Technology, Ltd., Wuhan, China; BM4051, 1:100 dilution),
β-catenin (Boster Biological Technology, Ltd., BM3905, 1:100
dilution) and GAPDH (Cell Signaling Technology, Inc., Danvers, MA,
USA; catalogue number: 2118; 1:1,000 dilution) at 4°C, followed by
incubation with horseradish peroxidase-conjugated goat anti-rabbit
secondary antibody (Solarbio, Beijing, China; catalogue number:
SE134; 1:1,000 dilution) at room temperature for 1 h. Specific
bands were visualized by Odyssey infrared imaging system (LI-COR,
Inc., Lincoln, NE, USA). The signals were analyzed using ImageJ
software (version 1.48v; National Institutes of Health, Bethesda,
MD, USA). The band densities of each sample were normalized to the
respective GAPDH band. All results were repeated three times.
Reversibility of drug-induced
effects
To evaluate whether the atorvastatin-induced effect
is reversible, the medium of TM cells treated with atorvastatin
(100 µM) for 48 h was replaced with culture medium after washing
with PBS. The reversibility of cellular morphological changes was
observed using a phase-contrast microscope and images were
captured. At 24 h after drug washout, cells were fixed and stained
for F-actin, vinculin and β-catenin using the same procedures
described in Atorvastatin treatment and cytoskeletal staining
section.
Statistical analysis
Data were analyzed using STATA version 12.0 software
(StataCorp LLC, College Station, TX, USA). Values are expressed as
the mean ± standard error. One-way analysis of variance followed by
Bonferroni's multiple comparisons test was used for statistical
comparisons of multiple groups. P<0.05 was considered to
indicate a statistically significant difference.
Results
Effects of atorvastatin on aqueous
outflow facility in enucleated porcine eyes
As expected, there was no significant difference
baseline outflow facility (C1) values among all
treatment groups (Fig. 1A). There
was no significant difference in outflow facility between the
control and 17 µM atorvastatin-treated group. Concentrations of
atorvastatin of ≥50 µM significantly increased the outflow
facility. C2 value in the 50, 100 and 200 µM groups was
significantly increased compared with C1 value in each
group (6.96-, 9.59- and 6.99-fold increase, respectively; Fig. 1A). There was no significant
difference among the three higher-concentration groups (Fig. 1B).
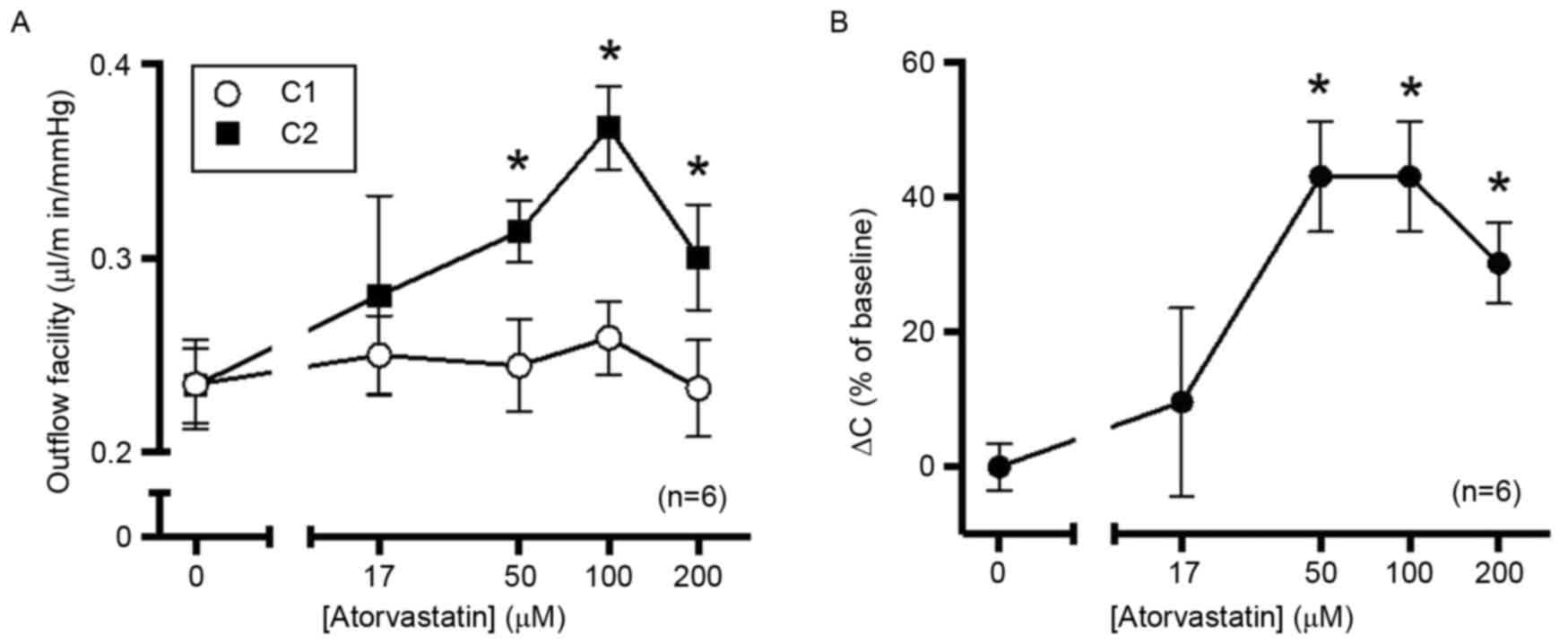 | Figure 1.Atorvastatin increases outflow in
enucleated porcine eyes. After establishing the initial baseline
outflow facility, eyes were perfused with 17, 50, 100 or 200 µM
atorvastatin dissolved in GPBS containing DMSO (0.38%).
Contralateral control eyes were perfused with GPBS containing DMSO
alone, with outflow facility measured at 10 Hz. Time of perfusion
was controlled within 2 h to avoid the ‘washout’ effect. (A)
Baseline outflow facility values were not significantly different
among all groups. Compared with the control group, 50, 100 and 200
µM atorvastatin significantly increased the outflow facility, while
17 µM atorvastatin did not have any significant effect. (B) The
percentage change from baseline exhibited and a typical
dose-response association. *P<0.05 vs. 0 µM atorvastatin.
C1, outflow facility at baseline; C2, outflow
facility after treatment; DMSO, dimethyl sulfoxide; GPBS, PBS
containing 5.5 mM glucose; ΔC, change of outflow. |
Effects of atorvastatin on cell
morphology
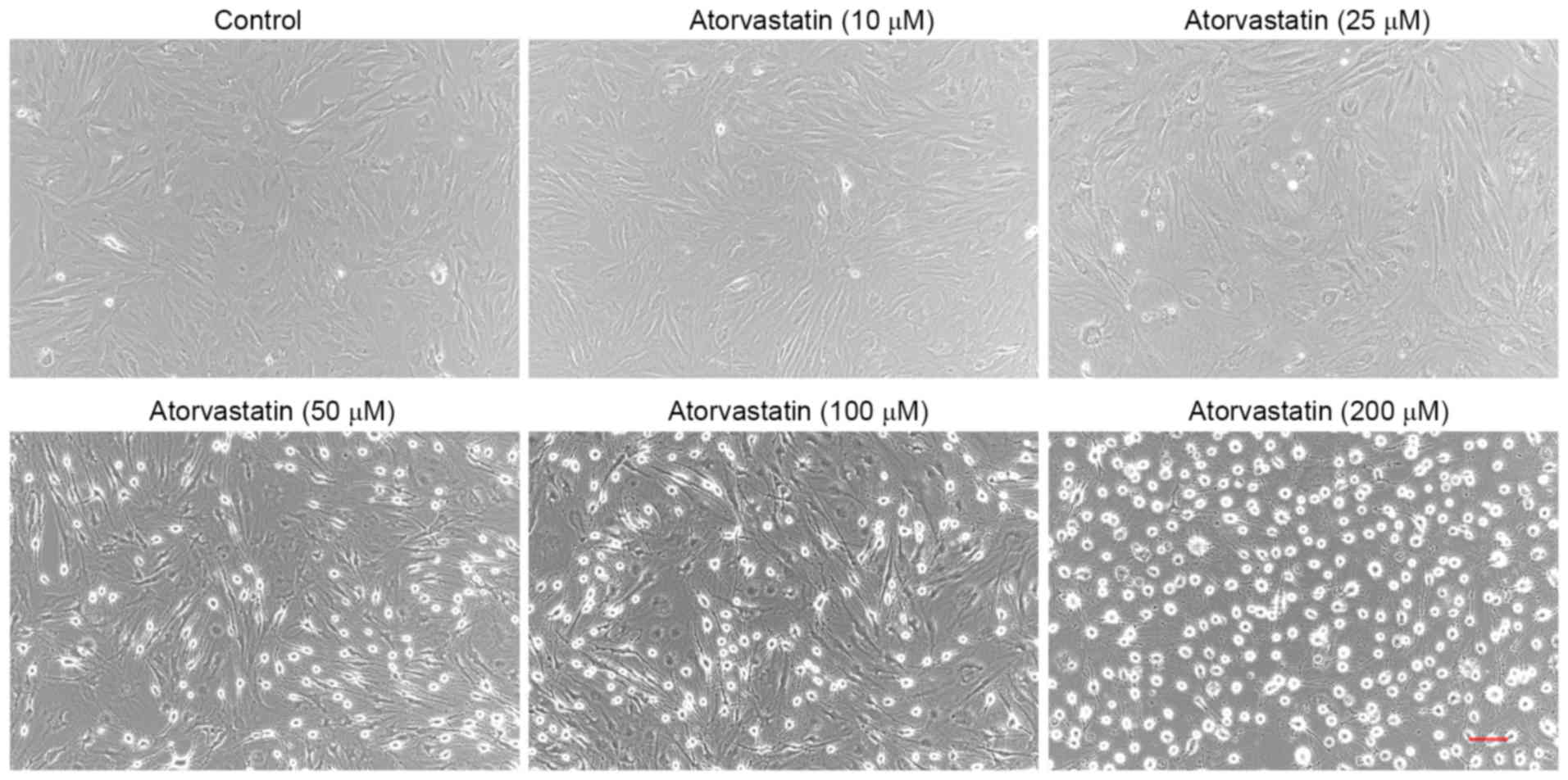
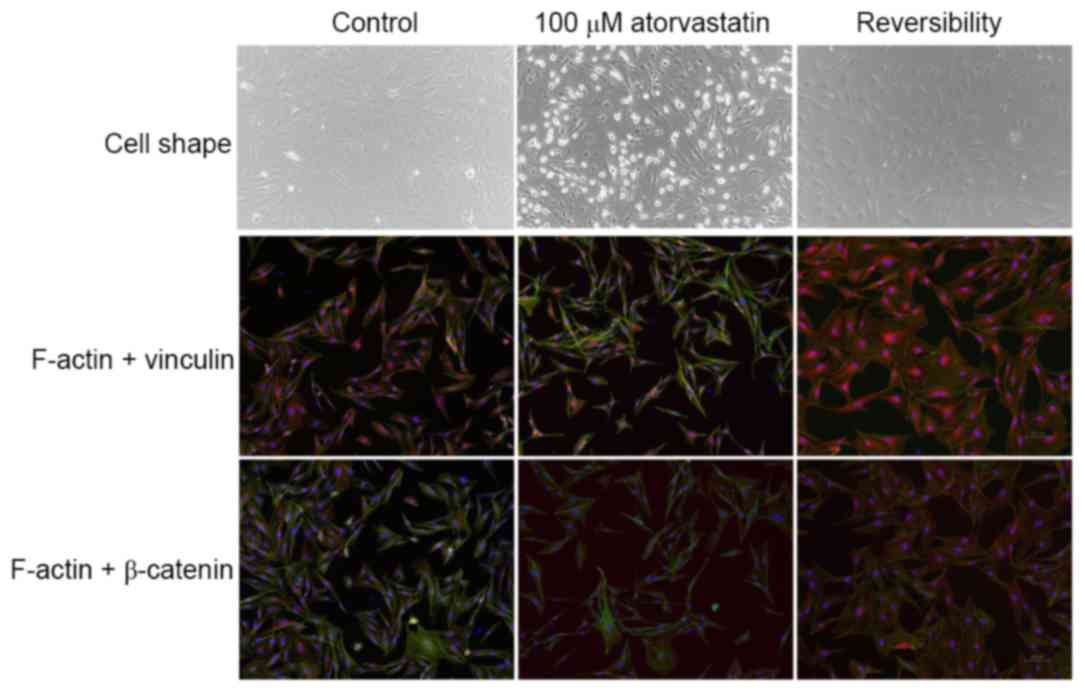
Treatment of confluent TM cells with atorvastatin
(10–200 µM) for 24 h led to certain morphological changes. Compared
with the control group, no obvious morphological changes were
detected in TM cells treated with 10 µM atorvastatin. However,
cells treated with 325 µM atorvastatin exhibited
progressive cell rounding and separation among cells, while the
cells did not detach from the culture dish (Fig. 2). Similar results were observed in
three independent experiments.
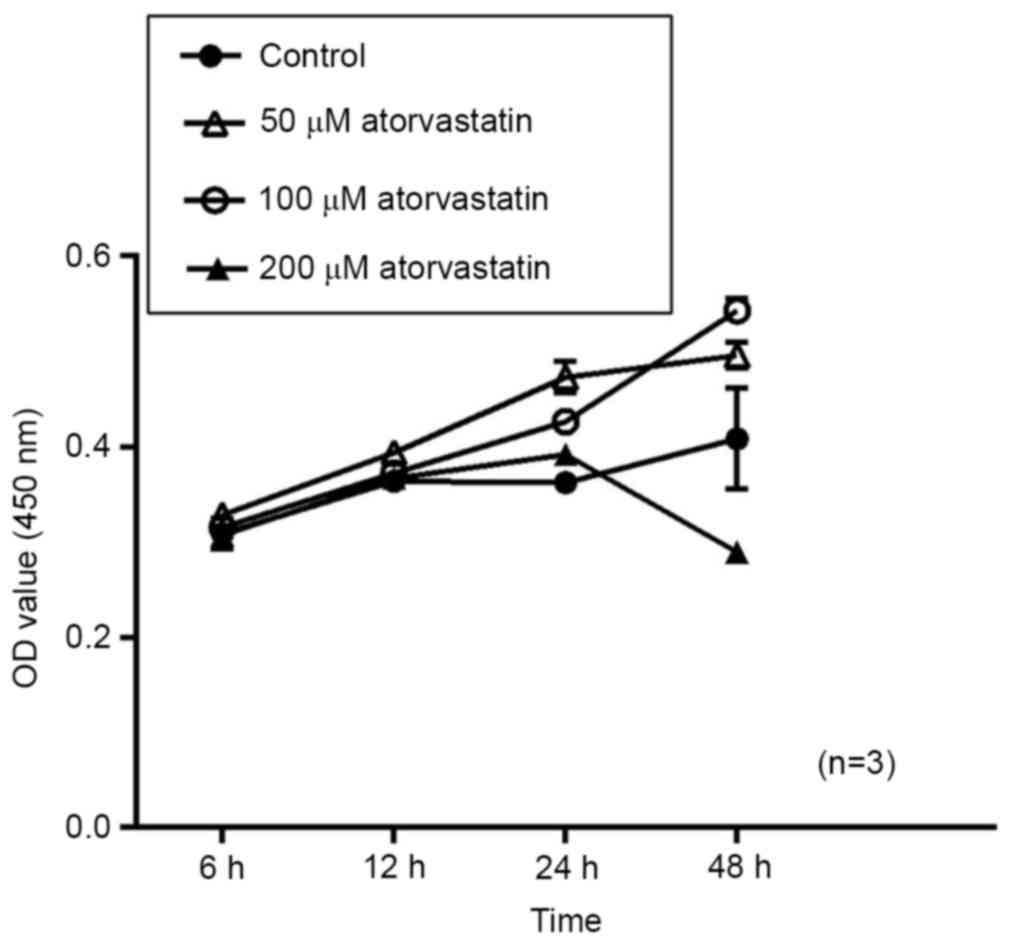
Cell viability
To evaluate the potential effect of atorvastatin on
cell viability, porcine TM cells were treated with 50–200 µM
atorvastatin and subjected to a CCK-8 assay at 6, 12, 24 and 48 h.
Compared with that in the mock-treated group, no significant change
in cell viability was detected after treatment with atorvastatin
(50–100 µM) for 48 h. However, the cell viability indicated a
downtrend in the group treated with 200 µM atorvastatin after 24 h;
although this was not significantly different. (Fig. 3). Therefore, these findings suggest
that high concentrations of atorvastatin may inhibit TM cell
viability. And experimental results may be statistically
significant after extending the intervention time.
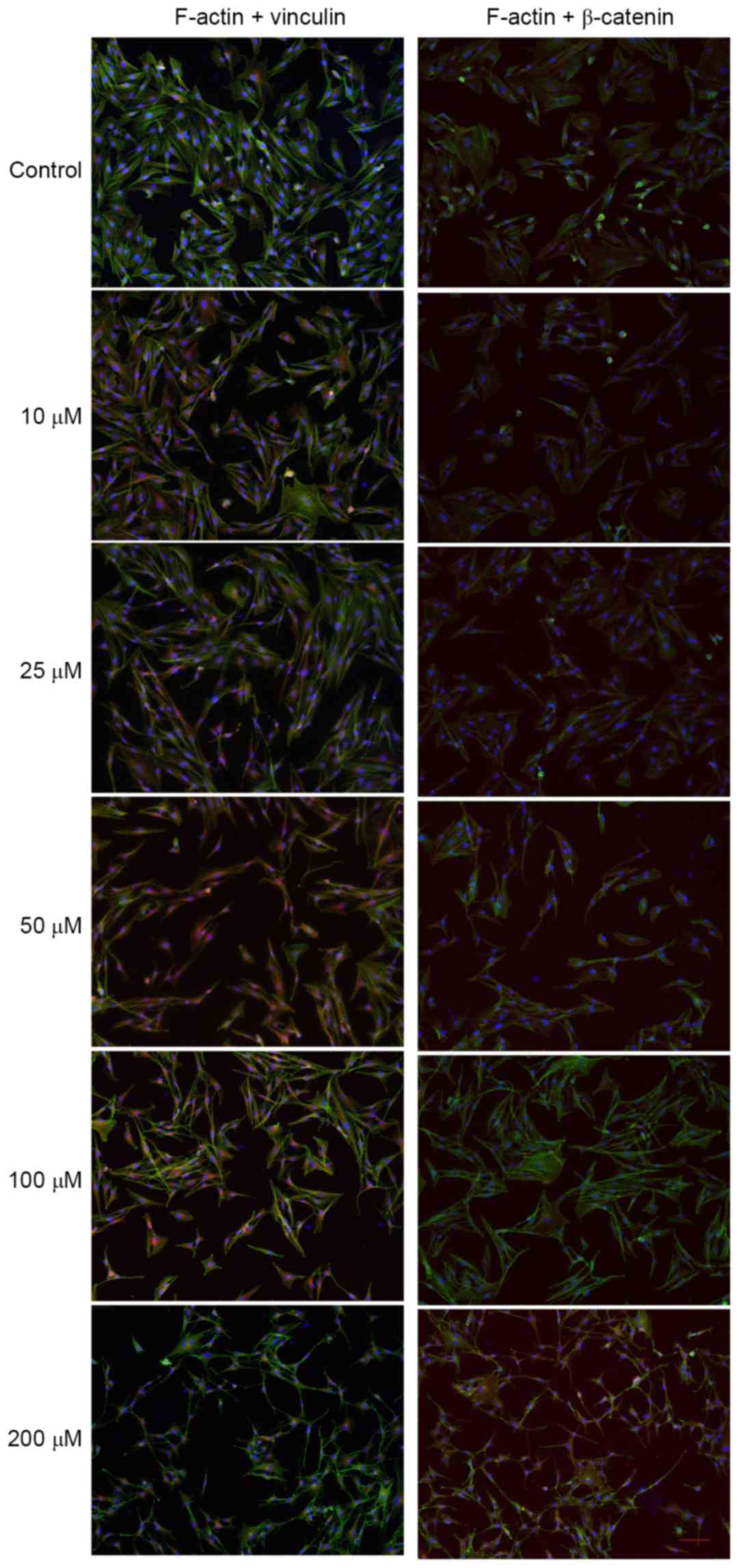
Effects of atorvastatin on
cytoskeletal organization and focal adhesion of TM cells
From Fig. 4, it may
be observed that the distribution of F-actin (green) in the mock-TM
cells was relatively uniform, while vinculin (red) and β-catenin
(red) were mainly distributed around the nuclei. A dose-dependent,
marked reduction of F-actin and changes in the distribution of
vinculin and β-catenin were observed in TM cells following 24 h of
treatment with atorvastatin (10–200 µM). An association between
changes in the actin cytoskeletal organization/distribution of
focal adhesions and morphological changes in TM cells was apparent
(Fig. 4).
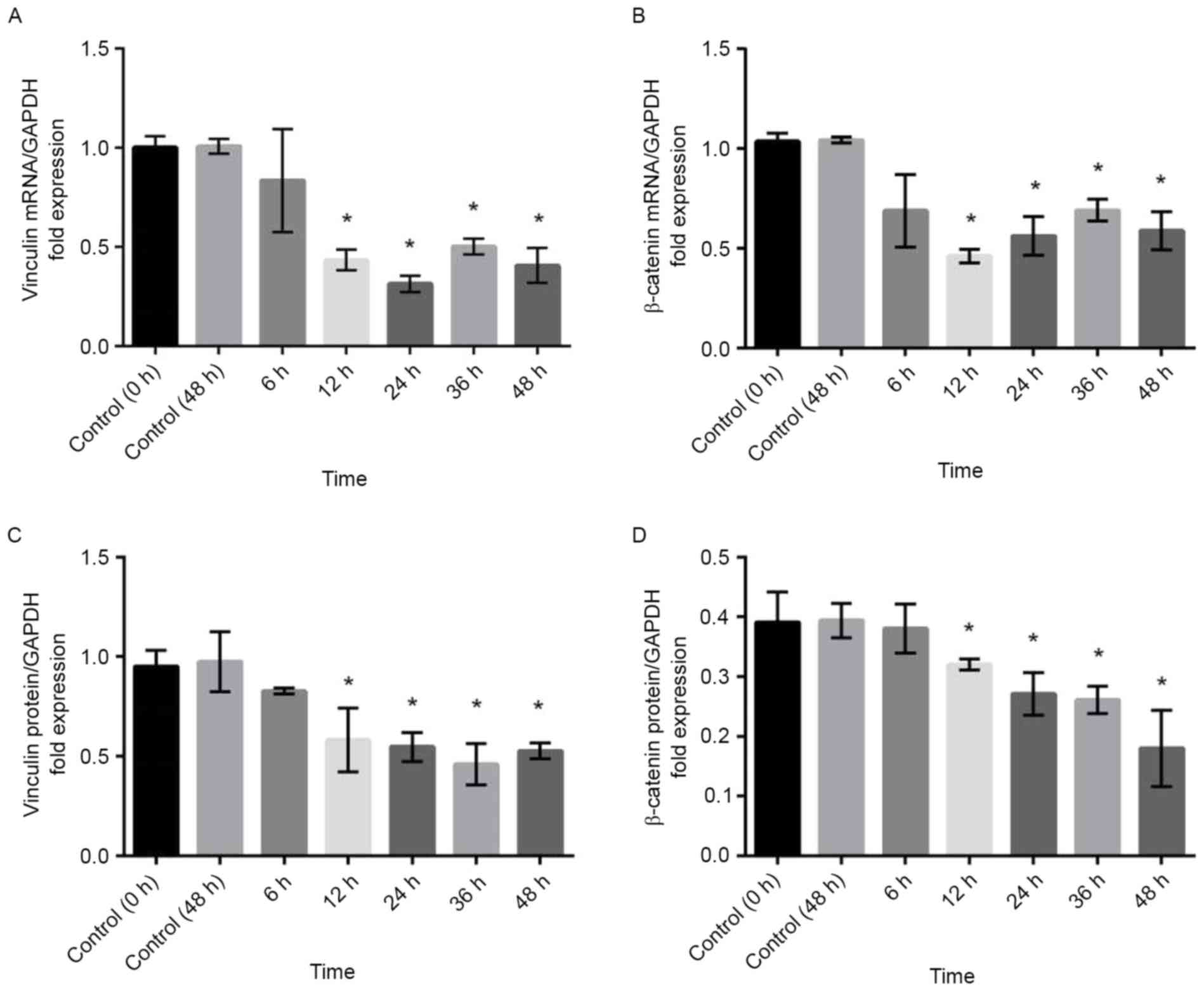
Effects of atorvastatin on expression
of vinculin and β-catenin
To confirm the results obtained by
immunohistochemistry, RT-qPCR and western blot analysis were
performed. Compared with that in control cells, the expression of
vinculin and β-catenin mRNA was significantly decreased in TM cells
treated with 100 µM atorvastatin for durations of ≥12 h (Fig. 5A and B). Similar results were
obtained by western blot analysis, which demonstrated that the
expression of vinculin and β-catenin protein was decreased by
atorvastatin (Fig. 5C and D).
Reversal of atorvastatin-induced
morphological and cytoskeletal changes
Of note, the atorvastatin-induced morphological
changes, modification of F-actin organization and focal adhesions
were reverted to those prior to treatment within 24 h of replacing
the culture supernatant with atorvastatin-free medium (Fig. 6).
Discussion
The present study aimed to determine the effects of
atorvastatin on TM cells and the aqueous humour outflow pathway of
enucleated porcine eyes. The results demonstrated that atorvastatin
increased the aqueous humour outflow facility in the whole-eye
perfusion model, which was also identified to be associated with
changes in TM cell morphology as well as the distribution of actin
cytoskeleton and focal adhesions in vitro.
The present study indicated that no detectable
washout effect was observed within 2 h. Compared with that in the
control group, atorvastatin increased the aqueous humour outflow
facility in enucleated porcine eyes in a dose-dependent manner. The
magnitude of this increase was comparable to the previously
reported 40% facility increase observed in an organ-cultured
porcine eye anterior segment perfused with 100 µM lovastatin for 45
h (23), and was less than the 80%
facility increase observed in porcine eyes after perfusion with
Y-27632 for 3 h (15). Furthermore,
atorvastatin takes effect more rapidly than lovastatin in
increasing aqueous humour outflow. In the present study, the
perfusion time was limited to 2 h to minimize the washout effect,
and to explore the short-term effectiveness of atorvastatin in
improving outflow facility. ‘Washout’ is a phenomenon occurring in
eyes of non-human species referring to a time-dependent increase in
outflow facility (38,39), while it has remained controversial
whether the washout effect is present in porcine eyes (40,41). A
typical washout effect is 6–26% for different durations of
perfusion (15,41–43).
Subsequently, the in vivo observation of the
present study was confirmed in vitro, as atorvastatin
treatment caused significant changes in cellular morphology and the
localization of cell-cell focal adhesions. The actin cytoskeleton
and focal adhesions are important structures for numerous cell
functions. Upon a decrease of cell-cell adhesions and cell-ECM
junctions, the intercellular space, through which the aqueous
humour flows, is expanded (10),
which may increase the outflow facility and decrease IOP. In the
present study, we speculate that atorvastatin treatment decreased
the expression of vinculin and β-catenin at the mRNA and the
protein level, and relaxed the actin cytoskeleton via inhibiting
the synthesis of Rho GTPase. The change of cell morphology and
adhesions regulates the hydrodynamics of aqueous humour outflow.
The present study demonstrated that atorvastatin may affect the
aqueous humour outflow pathway at the cellular level.
The effects of statins on cell morphology typically
require at least 18 h (23). In the
present study, atorvastatin at concentrations of >17 µM
significantly elevated the outflow facility within 2 h in the
whole-eye perfusion system. In addition, atorvastatin at
concentrations of >10 µM affected TM cell morphology within 24
h, suggesting that the statin concentration is an important factor
in the modification of cell morphology and aqueous humour
outflow.
It is acknowledged that the present study had
certain limitations; for instance, changes in outflow in the
single-pressure perfusion system do not differentiate between
pressure-dependent (largely trabecular) or pressure-independent
(uveoscleral) outflow. A multiple-pressure study will be performed
in the future.
In conclusion, atorvastatin, a cholesterol-lowering
drug, elevated aqueous humour outflow facility in a porcine
whole-eye perfusion model within 2 h. The effect was associated
with TM cell relaxation and decreased expression of focal adhesion
proteins in TM cells. It was therefore demonstrated that
atorvastatin decreased the IOP in vivo and in vitro.
It was speculated that the effects of atorvastatin may be
via blocking the Rho/ROCK signalling pathway. The present
results enhanced the current knowledge on the effect of statins
themselves on the morphology of TM cells and the contractile tone
of the aqueous outflow pathway (44). The results may provide evidence that
atorvastatin is a novel therapeutic agent for POAG. The present
study exemplified that novel treatment methods for POAG may be
identified by exploring clinically known drugs. Further animal
experiments and clinical studies may be required to confirm the
potential value of atorvastatin in the treatment of POAG.
Acknowledgements
The present study was sponsored by the Natural
Science Foundation of Shanghai (Grant no. 16ZR1404500).
References
|
1
|
Overby DR, Stamer WD and Johnson M: The
changing paradigm of outflow resistance generation: Towards
synergistic models of the JCT and inner wall endothelium. Exp Eye
Res. 88:656–670. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Fuchshofer R and Tamm ER: Modulation of
extracellular matrix turnover in the trabecular meshwork. Exp Eye
Res. 88:683–688. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Grant WM: Experimental aqueous perfusion
in enucleated human eyes. Arch Ophthalmol. 69:783–801. 1963.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Picht G, Welge-Luessen U, Grehn F and
Lütjen-Drecoll E: Transforming growth factor beta 2 levels in the
aqueous humor in different types of glaucoma and the relation to
filtering bleb development. Graefes Arch Clin Exp Ophthalmol.
239:199–207. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Johnson M: What controls aqueous humour
outflow resistance? Exp Eye Res. 82:1–557. 2006. View Article : Google Scholar
|
|
6
|
Tamm ER and Fuchshofer R: What increases
outflow resistance in primary open-angle glaucoma? Surv Ophthalmol.
52(Suppl 2): S101–S104. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ethier CR, Read AT and Chan DW: Effects of
latrunculin-B on outflow facility and trabecular meshwork structure
in human eyes. Invest Ophthalmol Vis Sci. 47:1991–1998. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bahler CK, Hann CR, Fautsch MP and Johnson
DH: Pharmacologic disruption of Schlemm's canal cells and outflow
facility in anterior segments of human eyes. Invest Ophthalmol Vis
Sci. 45:2246–2254. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hu Y, Gabelt BT and Kaufman PL: Monkey
organ-cultured anterior segments: Technique and response to H-7.
Exp Eye Res. 82:1100–1108. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Tian B, Gabelt BT, Geiger B and Kaufman
PL: The role of the actomyosin system in regulating trabecular
fluid outflow. Exp Eye Res. 88:713–717. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bornstein P: Matricellular proteins: An
overview. J Cell Commun Signal. 3:163–165. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Haddadin RI, Oh DJ, Kang MH, Filippopoulos
T, Gupta M, Hart L, Sage EH and Rhee DJ: SPARC-null mice exhibit
lower intraocular pressures. Invest Ophthalmol Vis Sci.
50:3771–3777. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Clark AF and Pang IH: Advances in glaucoma
therapeutics. Expert Opin Emerg Drugs. 7:141–163. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Cai S, Liu X, Glasser A, Volberg T, Filla
M, Geiger B, Polansky JR and Kaufman PL: Effect of latrunculin-A on
morphology and actin-associated adhesions of cultured human
trabecular meshwork cells. Mol Vis. 6:132–143. 2000.PubMed/NCBI
|
|
15
|
Rao PV, Deng PF, Kumar J and Epstein DL:
Modulation of aqueous humor outflow facility by the Rho
kinase-specific inhibitor Y-27632. Invest Ophthalmol Vis Sci.
42:1029–1037. 2001.PubMed/NCBI
|
|
16
|
Gabelt BT, Hu Y, Vittitow JL, Rasmussen
CR, Grosheva I, Bershadsky AD, Geiger B, Borrás T and Kaufman PL:
Caldesmon transgene expression disrupts focal adhesions in HTM
cells and increases outflow facility in organ-cultured human and
monkey anterior segments. Exp Eye Res. 82:935–944. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Downs JR, Clearfield M, Weis S, Whitney E,
Shapiro DR, Beere PA, Langendorfer A, Stein EA, Kruyer W and Gotto
AM Jr: Primary prevention of acute coronary events with lovastatin
in men and women with average cholesterol levels: Results of
AFCAPS/TexCAPS. Air force/texas coronary atherosclerosis prevention
study. JAMA. 279:1615–1622. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Endo A: The discovery and development of
HMG-CoA reductase inhibitors. J Lipid Res. 33:1569–1582.
1992.PubMed/NCBI
|
|
19
|
Vaughan CJ and Delanty N: Neuroprotective
properties of statins in cerebral ischemia and stroke. Stroke.
30:1969–1973. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zacco A, Togo J, Spence K, Ellis A, Lloyd
D, Furlong S and Piser T: 3-hydroxy-3-methylglutaryl coenzyme A
reductase inhibitors protect cortical neurons from excitotoxicity.
J Neurosci. 23:11104–11111. 2003.PubMed/NCBI
|
|
21
|
Schmeer C, Kretz A and Isenmann S:
Statin-mediated protective effects in the central nervous system:
General mechanisms and putative role of stress proteins. Restor
Neurol Neurosci. 24:79–95. 2006.PubMed/NCBI
|
|
22
|
Yin H, Gui Y and Zheng XL:
2-methoxyestradiol inhibits atorvastatin-induced rounding of human
vascular smooth muscle cells. J Cell Physiol. 222:556–564.
2010.PubMed/NCBI
|
|
23
|
Song J, Deng PF, Stinnett SS, Epstein DL
and Rao PV: Effects of cholesterol-lowering statins on the aqueous
humor outflow pathway. Invest Ophthalmol Vis Sci. 46:2424–2432.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Burridge K and Wennerberg K: Rho and Rac
take center stage. Cell. 116:167–179. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Etienne-Manneville S and Hall A: Rho
GTPases in cell biology. Nature. 420:629–635. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kaibuchi K, Kuroda S and Amano M:
Regulation of the cytoskeleton and cell adhesion by the Rho family
GTPases in mammalian cells. Annu Rev Biochem. 68:459–486. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Somlyo AP and Somlyo AV: Signal
transduction through the RhoA/Rho-kinase pathway in smooth muscle.
J Muscle Res Cell Motil. 25:613–615. 2004.PubMed/NCBI
|
|
28
|
Wang J, Liu X and Zhong Y:
Rho/Rho-associated kinase pathway in glaucoma (Review). Int J
Oncol. 43:1357–1367. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Stein JD, Newman-Casey PA, Talwar N, Nan
B, Richards JE and Musch DC: The relationship between statin use
and open-angle glaucoma. Ophthalmology. 119:2074–2081. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
De Castro DK, Punjabi OS, Bostrom AG,
Stamper RL, Lietman TM, Ray K and Lin SC: Effect of statin drugs
and aspirin on progression in open-angle glaucoma suspects using
confocal scanning laser ophthalmoscopy. Clin Exp Ophthalmol.
35:506–513. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Openkova YY, Korobeiynikova EN, Rykin VS
and Vinkova GA: The analysis of status of biochemical indicators in
blood serum and lacrimal fluid in patients with primary open-angle
glaucoma. Klin Lab Diagn. 8–11. 2013.(In Russian). PubMed/NCBI
|
|
32
|
DiNicolantonio JJ, Lavie CJ, Serebruany VL
and O'Keefe JH: Statin wars: The heavyweight match-atorvastatin
versus rosuvastatin for the treatment of atherosclerosis, heart
failure, and chronic kidney disease. Postgrad Med. 125:7–16. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Stoekenbroek RM, Boekholdt SM, Fayyad R,
Laskey R, Tikkanen MJ, Pedersen TR and Hovingh GK; Incremental
Decrease in End Points Through Aggressive Lipid Lowering Study
Group, : High-dose atorvastatin is superior to moderate-dose
simvastatin in preventing peripheral arterial disease. Heart.
101:356–362. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Sillesen H, Amarenco P, Hennerici MG,
Callahan A, Goldstein LB, Zivin J, Messig M and Welch KM: Stroke
Prevention by Aggressive Reduction in Cholesterol Levels
Investigators: Atorvastatin reduces the risk of cardiovascular
events in patients with carotid atherosclerosis: A secondary
analysis of the stroke prevention by aggressive reduction in
cholesterol levels (SPARCL) trial. Stroke. 39:3297–3302. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhang Y, Toris CB, Liu Y, Ye W and Gong H:
Morphological and hydrodynamic correlates in monkey eyes with laser
induced glaucoma. Exp Eye Res. 89:748–756. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Polansky JR, Weinreb RN, Baxter JD and
Alvarado J: Human trabecular cells. I. Establishment in tissue
culture and growth characteristics. Invest Ophthalmol Vis Sci.
18:1043–1049. 1979.PubMed/NCBI
|
|
37
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Barany EH and Gassmann HB: The effect of
death on outflow resistance in normal and sympathectomized rabbit
eyes. Invest Ophthalmol. 4:206–210. 1965.PubMed/NCBI
|
|
39
|
Erickson-Lamy K, Schroeder AM, Bassett-Chu
S and Epstein DL: Absence of time-dependent facility increase
(“washout”) in the perfused enucleated human eye. Invest Ophthalmol
Vis Sci. 31:2384–2388. 1990.PubMed/NCBI
|
|
40
|
Vaajanen A, Vapaatalo H and Oksala O: A
modified in vitro method for aqueous humor outflow studies in
enucleated porcine eyes. J Ocul Pharmacol Ther. 23:124–131. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Rao PV, Deng P, Sasaki Y and Epstein DL:
Regulation of myosin light chain phosphorylation in the trabecular
meshwork: Role in aqueous humour outflow facility. Exp Eye Res.
80:197–206. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Khurana RN, Deng PF, Epstein DL and Rao P
Vasantha: The role of protein kinase C in modulation of aqueous
humor outflow facility. Exp Eye Res. 76:39–47. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Epstein DL, Roberts BC and Skinner LL:
Nonsulfhydryl-reactive phenoxyacetic acids increase aqueous humor
outflow facility. Invest Ophthalmol Vis Sci. 38:1526–1534.
1997.PubMed/NCBI
|
|
44
|
Pokrovskaya O, Wallace D and O'Brien C:
The emerging role of statins in glaucoma pathological mechanisms
and therapeutics. Open J Ophthalmol. 4:124–138. 2014. View Article : Google Scholar
|