Introduction
The skeleton provides a mechanical structure for
supporting the human torso, protects internal organs, stores
minerals and facilitates movement. Bone has a number of important
roles, including making red and white blood cells, and generating
and secreting various active substances capable of modulating
biological function in tissues and organs (1–3). Bone
tissue, an endocrine organ, may directly or indirectly modulate
skeletal muscle metabolism (4). In
recent decades, osteoporosis has become one of the major diseases
in the elderly population. Worldwide, millions of people suffer
from osteoporosis and are at a high risk of fractures due to low
bone mass (5). Numerous cytological
studies on the prevention and treatment of osteoporosis have been
conducted in the course of drug development (6). Osteoblasts and osteoclasts are
responsible for forming, destroying, remodeling and repairing the
organic and inorganic matrix that constitutes bones (6–9). Bone
metabolism, which is associated with normal growth, fractures and
osteoporosis due to postmenopausal and age-related osteopenia, is
highly regulated and balanced between bone formation via
osteoblasts and bone resorption via osteoclasts (10). Investigations on the proliferation
and differentiation of osteoblasts are crucial to studies on the
formation of bone and its constant remodeling.
Clinical observations indicate that osteoporosis is
often diagnosed in older patients (age, >50 years) or
post-menopausal women and the practice of hormone replacement
therapy improves bone density in osteoporosis patients (11–16). The
effects of both estrogen and androgen on the maintenance of bone
mass have been widely studied in the past few decades (17). It is known that estrogen has positive
effects on bone formation and the inhibition of bone resorption.
Current estimates state that approximately 20% of Americans with
osteopenia or osteoporosis are male (18), which suggests that men were better
protected against osteoporosis and osteoporotic fractures compared
with women. Some evidence suggests that older men have a risk of
osteoporosis when their androgen levels decrease, and androgenic
steroids have the proven ability to promote osteogenesis (19). On the other hand, muscle and bone are
in constant interaction. With aging, there is a progressive decline
in muscle mass, known as sarcopenia, as well as in bone mass, known
as osteopenia/osteoporosis (19).
The above studies have demonstrated that androgen plays a
significant role in the maintenance of bone mass and muscle, with
benefits in the treatment of osteoporosis.
However, the anabolic steroids involved in hormone
replacement therapy may cause severe side effects, including
changes in mood and libido, increase of aggression, and
pathological cardiovascular symptoms (20,21). The
intake of steroids with strong androgenic pharmacological actions,
including testosterone (17β-hydroxy-androst-4-en-3-one; T) and
dihydrotestosterone (17β-hydroxy-5α-androstan-3-one; DHT), induce
virilescence in women (22).
Therefore, other related steroids should be considered as
short-term substitutes or supplements to attenuate these adverse
effects in the treatment of osteoporosis.
Steroids with weak androgenic effect and the
precursors of androgenic hormones may be considered as candidates.
Pregnenolone (3β-hydroxy-5-pregnen-20-one; Preg) is an endogenous
progestin and an essential precursor for all other steroid hormones
(23). Androstenedione
(androst-4-ene-3,17-dione; AD) is an endogenous androgen and an
intermediate in the biosynthesis of T. It is also the precursor of
certain androgens and estrogens (24). Etiocholanolone
(3α-hydroxy-5β-androstan-17-one; Etio) is an endogenous
5β-androstane steroid, which is one of the major excreted
metabolites of testosterone (25).
Androsterone (3α-hydroxy-5α-androstan-17-one; An), an endogenous
steroid hormone and putative pheromone (26), is a weak androgen with one-seventh of
the androgenic effect of T (27).
Unlike the naturally occurring steroids above, nandrolone
(17β-hydroxy-19-nor-4-androsten-3-one; NA) is a synthetic
anabolic-androgenic steroid derived from T. A previous study has
described that the positive effects of NA include reducing time to
bone consolidation (28). The
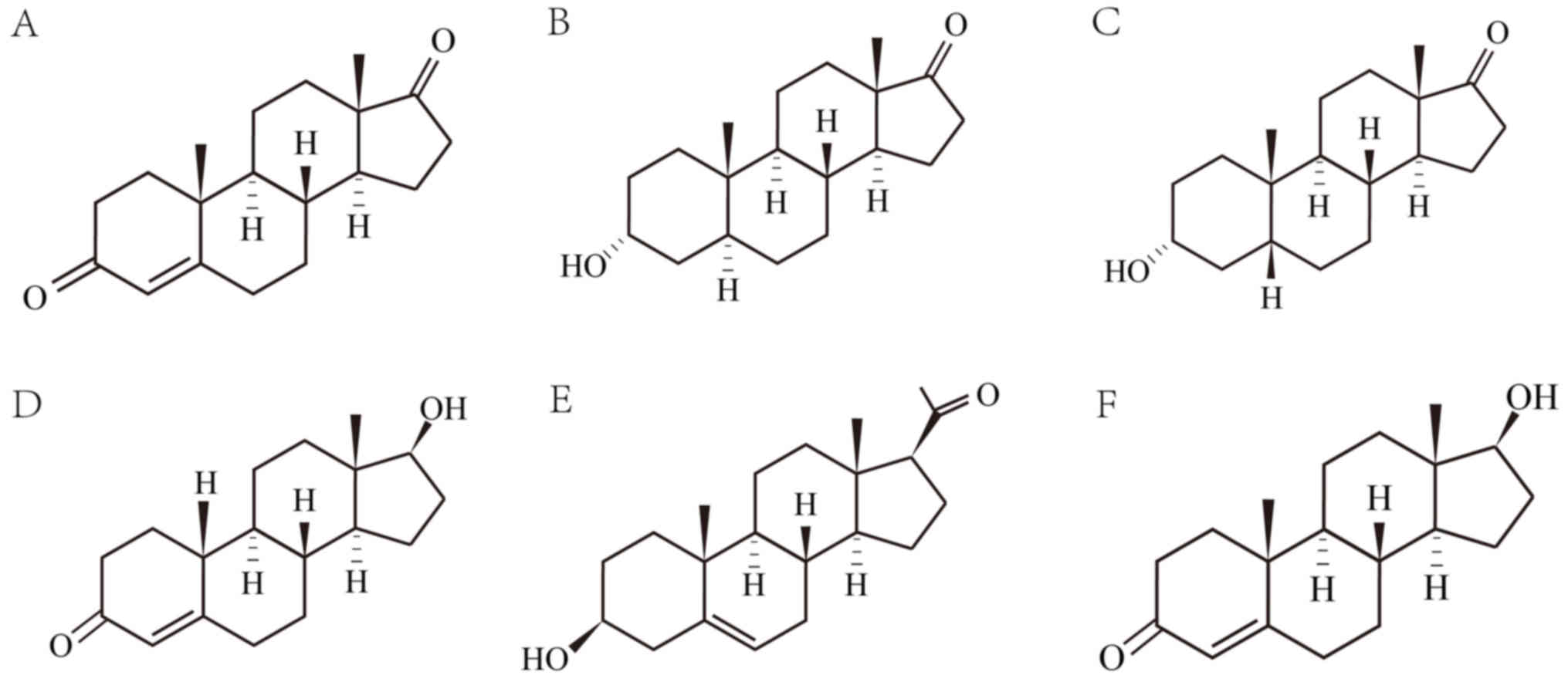
chemical structures of these steroids are illustrated in Fig. 1.
To the best of the authors' knowledge, the effects
on osteoblasts of progestin and the aforementioned androgens have
not been reported. T, the primary male sex hormone, can be
converted to estrogen in vivo, which was considered to be
the mechanism for the effectiveness of T in bone regeneration in a
previous study (29). Although the
stimulatory effect of T incorporated into polymer-bioceramic on
osteoblast proliferation has been described previously (30), the independent effect of T on
osteoblast proliferation was determined in this study.
The aim of the present study was to reveal the
proliferation and differentiation effects of Preg, AD, Etio, An and
NA on human osteoblasts and to explore the potential application of
Preg, AD, Etio and An in treating osteoporosis.
Materials and methods
Materials
Phosphate-buffered saline, Dulbecco's modified
Eagle's medium (DMEM) with low glucose,
trypsine-ethylenediaminetetraacetic acid (EDTA) and
penicillin/streptomycin were obtained from Gibco (Thermo Fisher
Scientific, Inc., Waltham, MA, USA). Fetal bovine serum (FBS) was
purchased from Biological Industries (Kibbutz Beit Haemek, Israel).
Preg, AD, Etio, An, NA and T were from Sigma-Aldrich (Merck KGaA,
Darmstadt, Germany).
Cell culture and drug
intervention
Human fetal osteoblasts (hFOB1.19; ATCC CRL-11372;
American Type Culture Collection, Manassas, VA, USA) were thawed
and sub-cultured in low-glucose DMEM supplemented with 10% FBS and
1% penicillin/streptomycin in a humidified incubator at 37°C with
5% CO2. The medium was changed every 2 days. The cells
were trypsinized with 0.25% trypsin-EDTA and seeded. The culture
medium was then changed to the treatment medium, made with
low-glucose DMEM, 10% FBS, 1% penicillin-streptomycin, 50 mg/ml
ascorbic acid, 0.01 mol/l glycerol-2-phosphate, 100 nmol/l
dexamethasone and supplemented with drugs (Preg, AD, Etio, An, NA
and T). These drugs were added at various concentrations. The
non-drug group (0 mol/l) was used as a blank control. The
measurement for each concentration was conducted six times, and the
mean value was used in analysis.
Cell metabolic activity was assessed with an MTS
assay, as described below. Cells were seeded in tissue culture
plates at a density of 1×104 cells/well and cultured
with the treatment medium for 24 h. Each drug was added at
concentrations of 0, 10−10, 10−8,
10−6 and 10−5 mol/l.
For the measurements of alkaline phosphatase (ALP)
and osteocalcin, the cells were seeded in tissue culture plates
(5×104 cells/cm2) and cultured with the
treatment medium for 5 days. The treatment medium was changed every
2 days. Preg, AD, Etio, An, NA and T were added at a concentration
of 10−10 mol/l.
Cell proliferation assessment
Cell proliferation was assessed using an MTS assay
(CellTiter 96™ AQueous Assay; Promega Corporation,
Madison, WI, USA), following cell culture for 24 h. The MTS and
phenazine methosulfate (PMS) solution were warmed and mixed at a
ratio of MTS:PMS=20:1. Then, 200 µl of the mixed solution and 1 ml
culture medium were added to each well, and the cells were
incubated for 2 h at 37°C. The absorbance of the supernatant was
measured using a spectrophotometer at a wavelength of 490 nm and
the optical density (OD) values recorded.
ALP activity assay
An ALP assay was performed following 5 days of cell
culture with the drugs at a concentration of 10−10
mol/l. The activity of ALP, an exo-enzyme used as the marker of the
osteoblastic phenotype, was measured using an Alkaline Phosphatase
Assay kit (Anaspec, Inc., Fremont, CA, USA) according to the
manufacturer's protocol. Briefly, the cells were lysed in 600 µl
lysis buffer provided in the kit under continuous scratching with a
pipette tip. The lysate was then centrifuged for 15 min at 10,000 ×
g at 4°C. The supernatant was added to a non-tissue culture treated
plate and p-Nitrophenyl Phosphate Disodium (Pnpp) ALP substrate
solution was added to each well. The plate was incubated at 37°C
for 1 h. The stop solution provided with the kit was added to each
well following incubation and the absorbance of the solution was
measured using a spectrophotometer at a wavelength of 405 nm. The
values of ALP activity (U/l) were recorded.
Osteocalcin secretion
Osteocalcin, also called bone
γ-carboxyglutamic-acid-containing protein (BGP), is one of the most
abundant proteins in bone and is produced exclusively by
osteoblasts; it has been used as a serum marker of osteoblastic
bone formation (31). A BGP assay
was performed on cells cultured with the drugs at a concentration
of 10−10 mol/l for 5 days. A total of 25 µl culture
medium was analyzed using a BGP ELISA kit (cat. no. JL19437-48T;
Shanghai Jianglai Bio-Technology Co., Ltd., China) according to the
manufacturer's protocol. The BGP concentration (µg/l) was
recorded.
Statistical analysis
A one-way analysis of variance and Tukey's post-hoc
test were used to assess significant differences. Data are
presented as the mean ± standard deviation. SPSS version 20 (IBM
Corp., Armonk, NY, USA) was used for statistical analysis.
P<0.05 was considered to indicate a statistically significant
difference.
Results
Cell proliferation
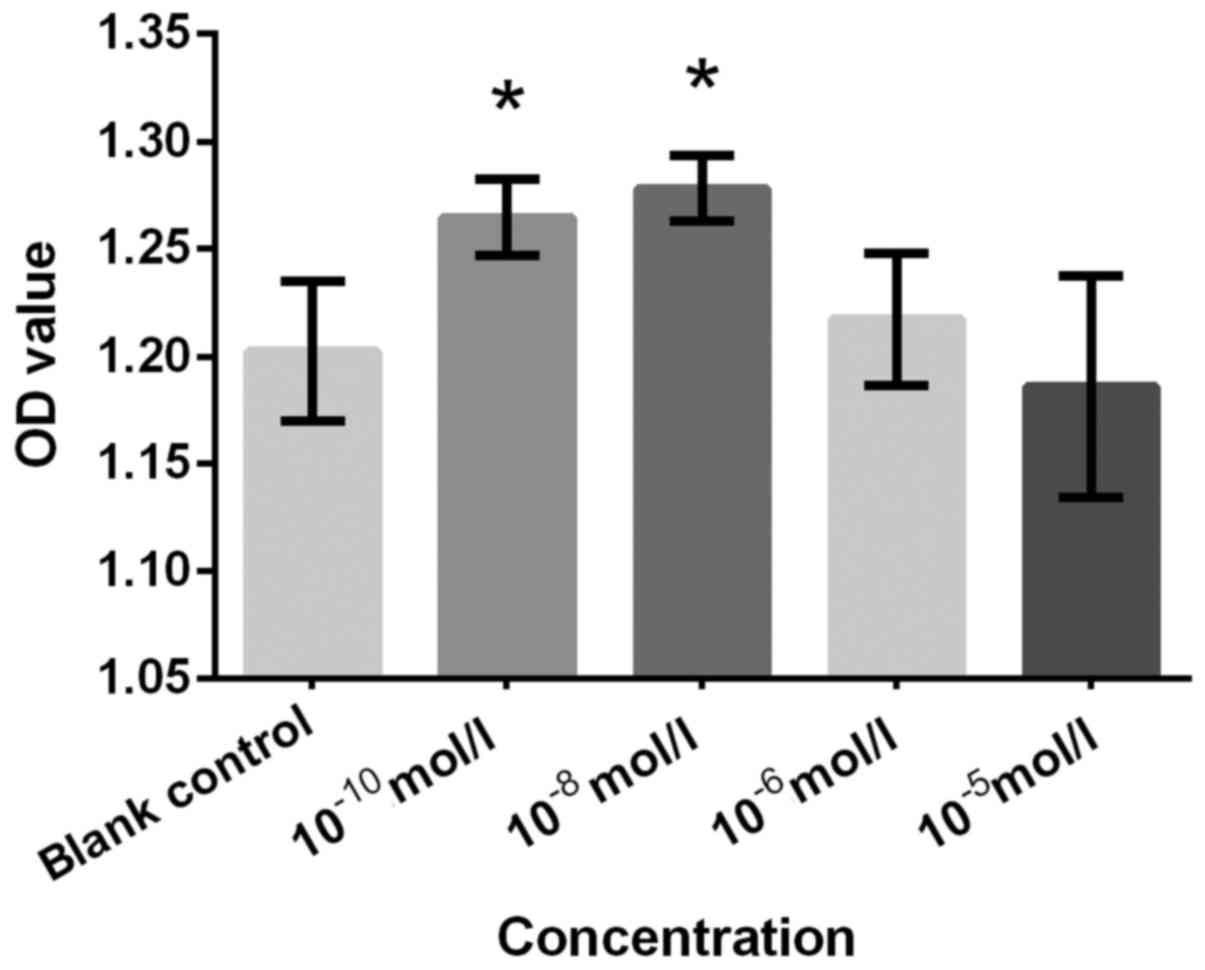
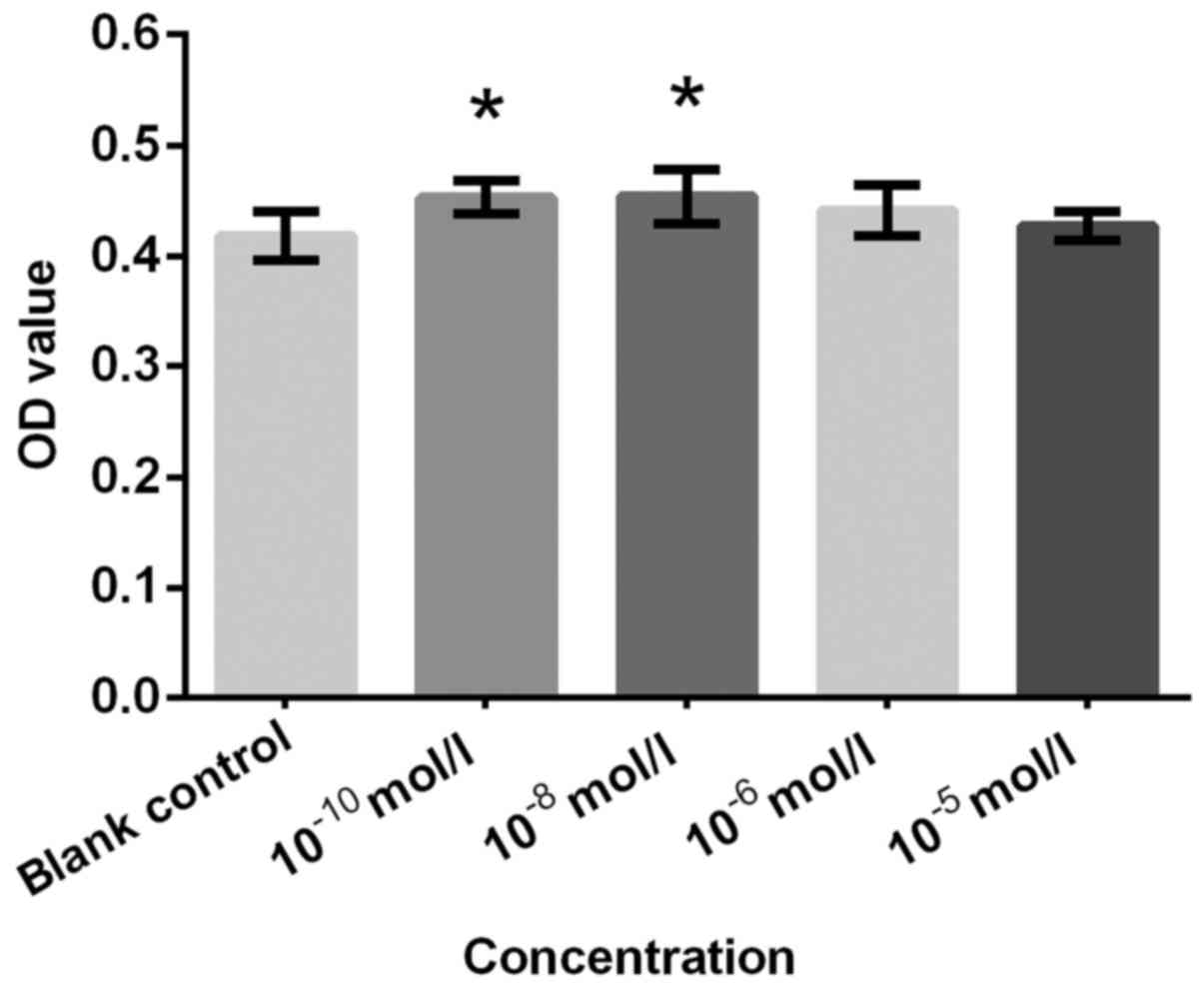
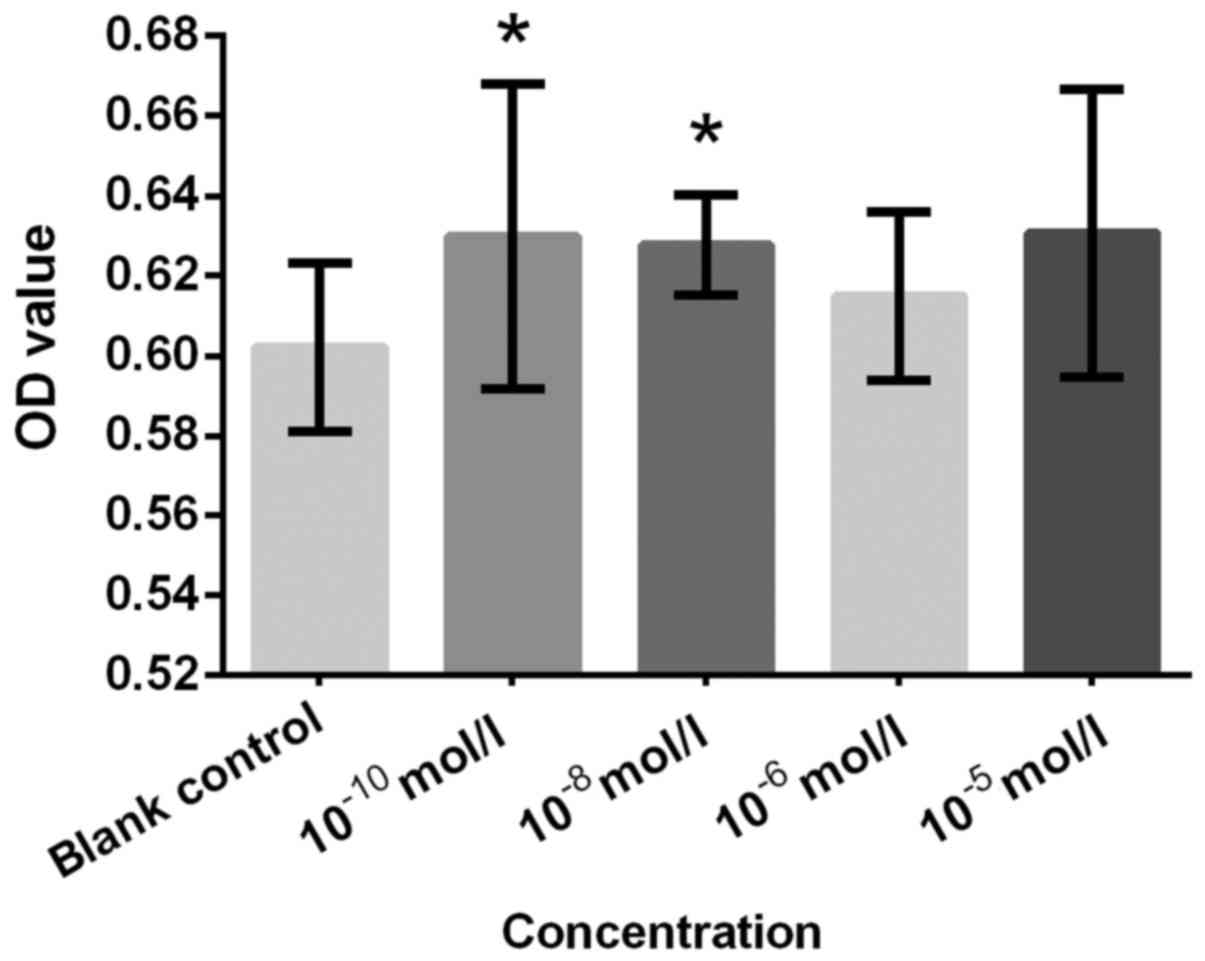
The proliferation effects of cells treated with
drugs at concentrations of 0, 10−10, 10−8,
10−6 and 10−5 mol/l were analyzed using an
MTS assay. The measured OD values for Preg, AD, Etio, An, NA and T
with various concentrations were plotted and are presented in
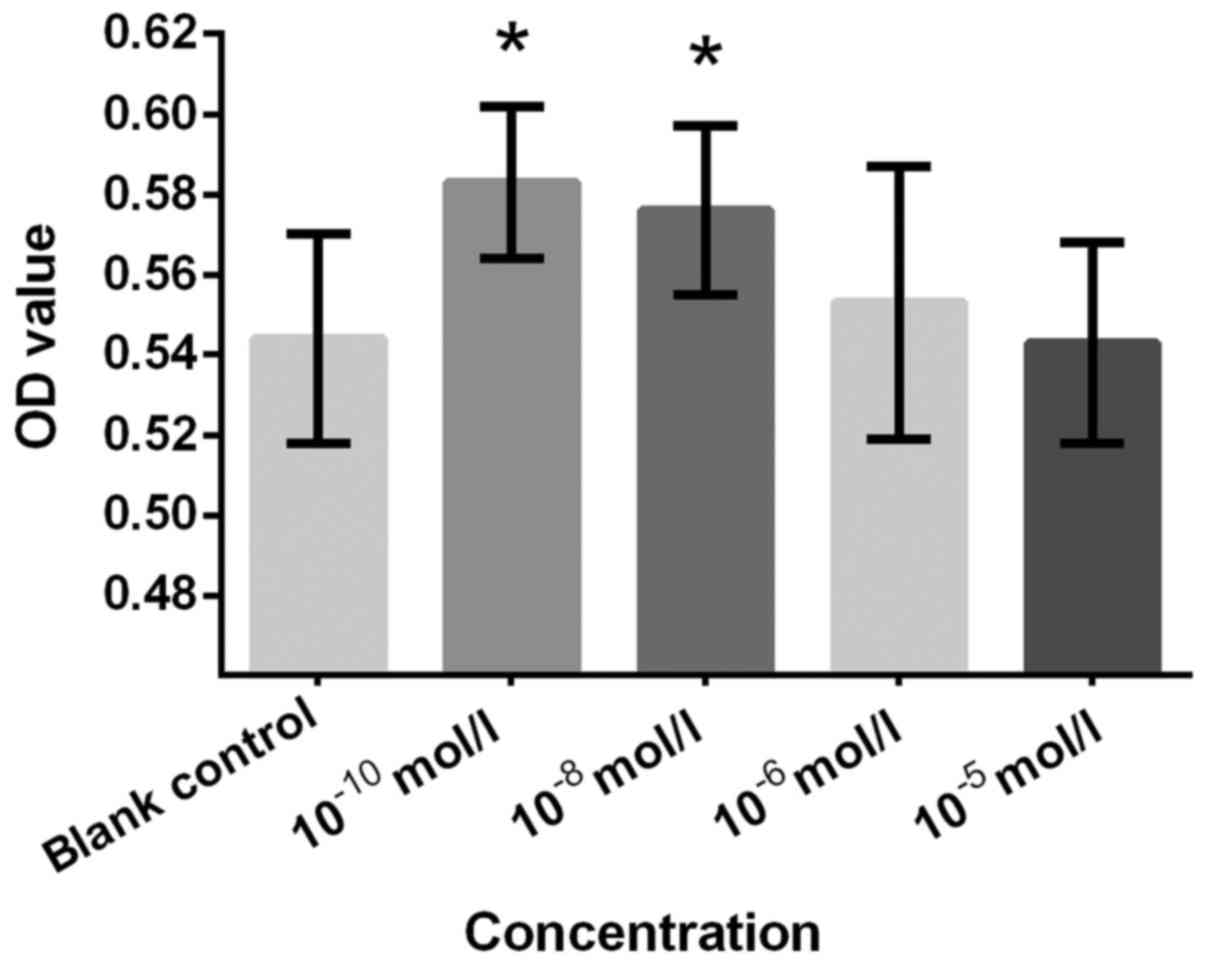
Figs. 2–7, respectively. Cells treated with
10−10 and 10−8 mol/l Preg exhibited
significantly increased proliferation rate compared with the blank
control group (Fig. 2). This finding
was consistent with the effective concentrations for AD and Etio,
which also induced a significant increase in proliferation at
10−10 and 10−8 mol/l compared with the
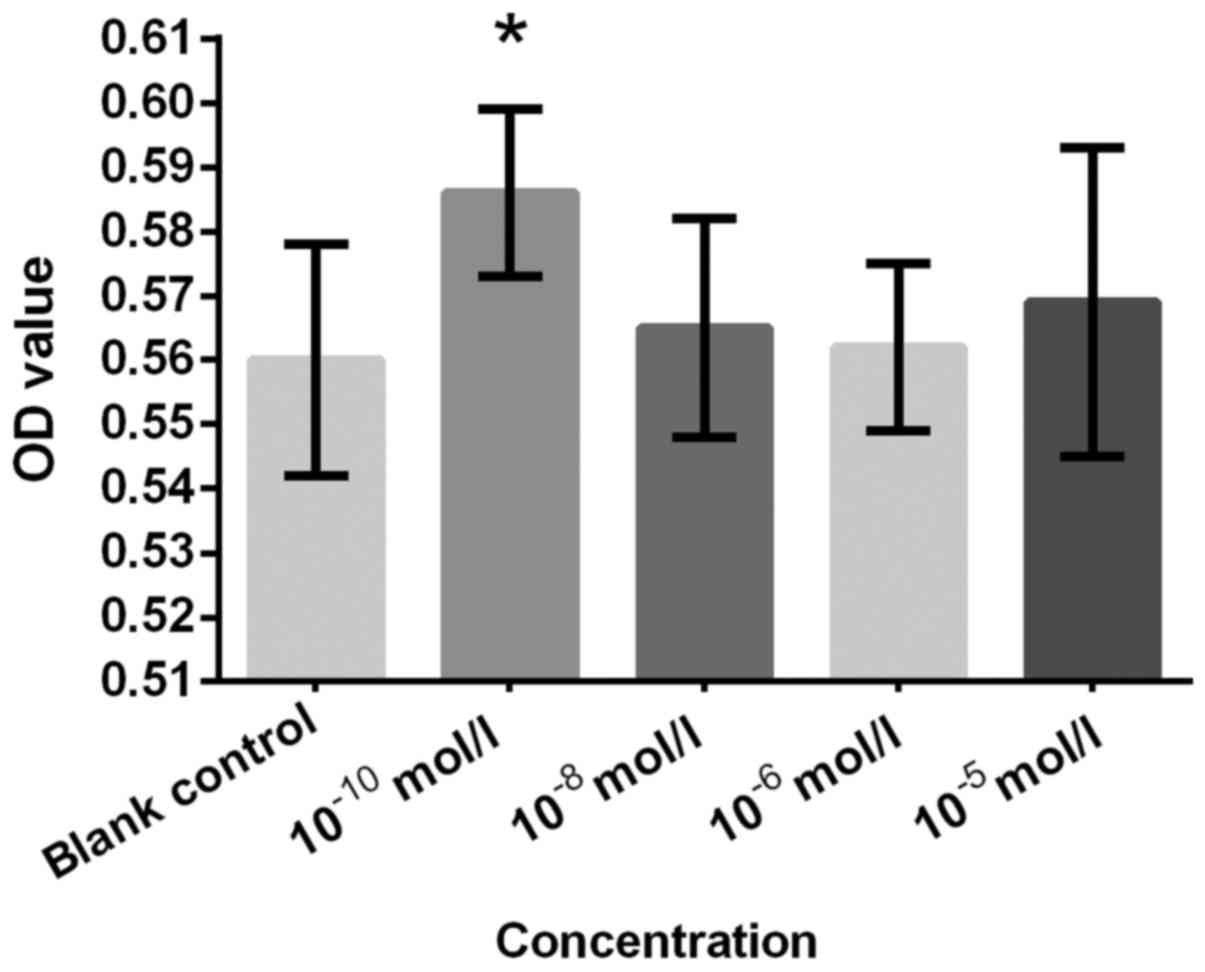
control (Figs. 3 and 4). An significantly increased cell
proliferation at 10−10 mol/l (Fig. 5). T was not as effective as expected
for cell proliferation at these concentrations. Nevertheless, the
results of T still indicated that proliferation was significantly
increased in the 10−10 and 10−8 mol/l groups
compared with the control group (Fig.
7). These results suggested that Preg, AD, Etio and An should
be considered functional steroids in certain situations. It was
also suggested that Etio and An would be more effective at
~10−10 and ~10−8 mol/l. Preg and AD exhibited
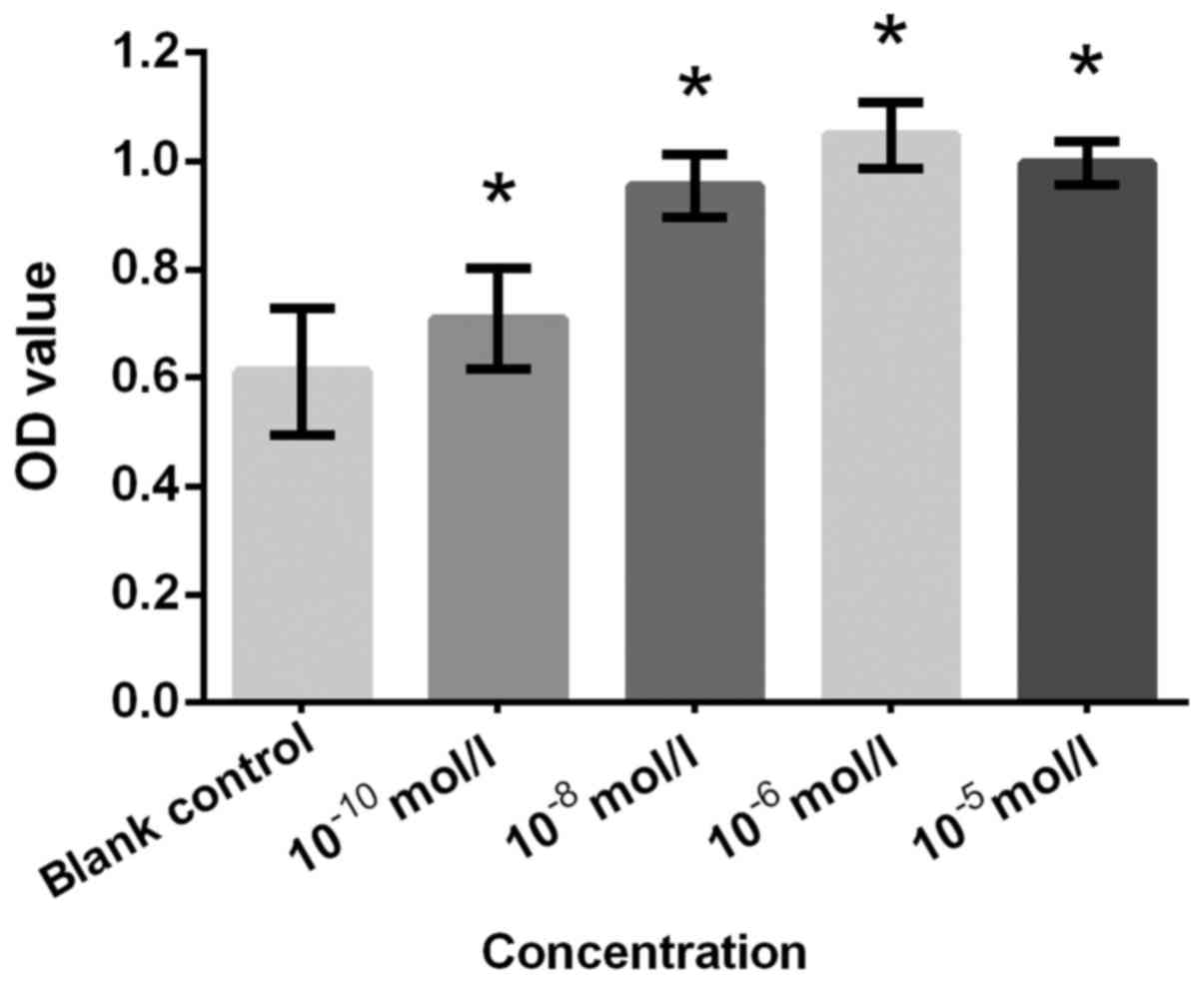
a similar result. NA at concentrations of 10−10,
10−8, 10−6 and 10−5 mol/l
demonstrated significantly increased proliferation compared with
the blank control (all P<0.05; Fig.
6). T plays a key role in the physiological process for males
and females, and is essential for health (22) and for the prevention of osteoporosis
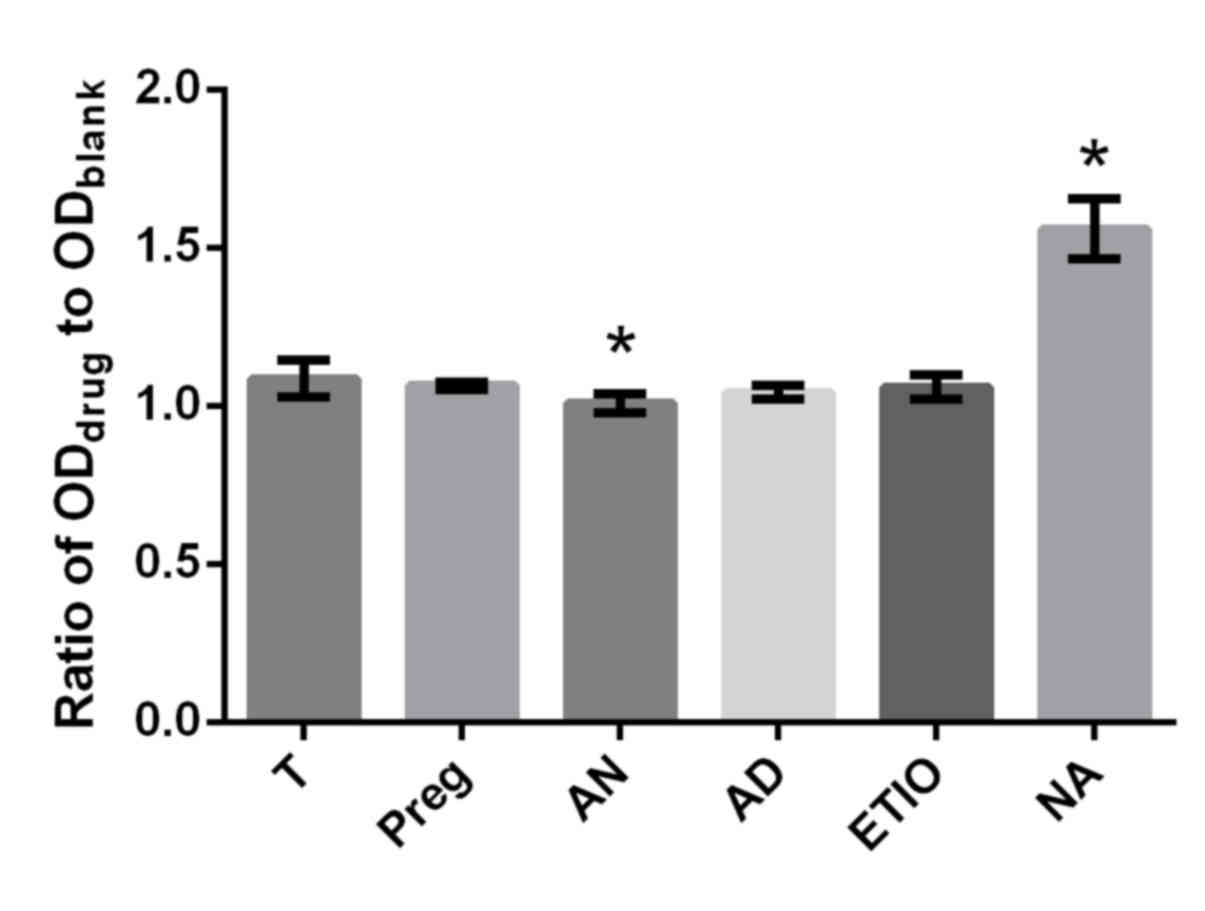
(32). The differences in effect on
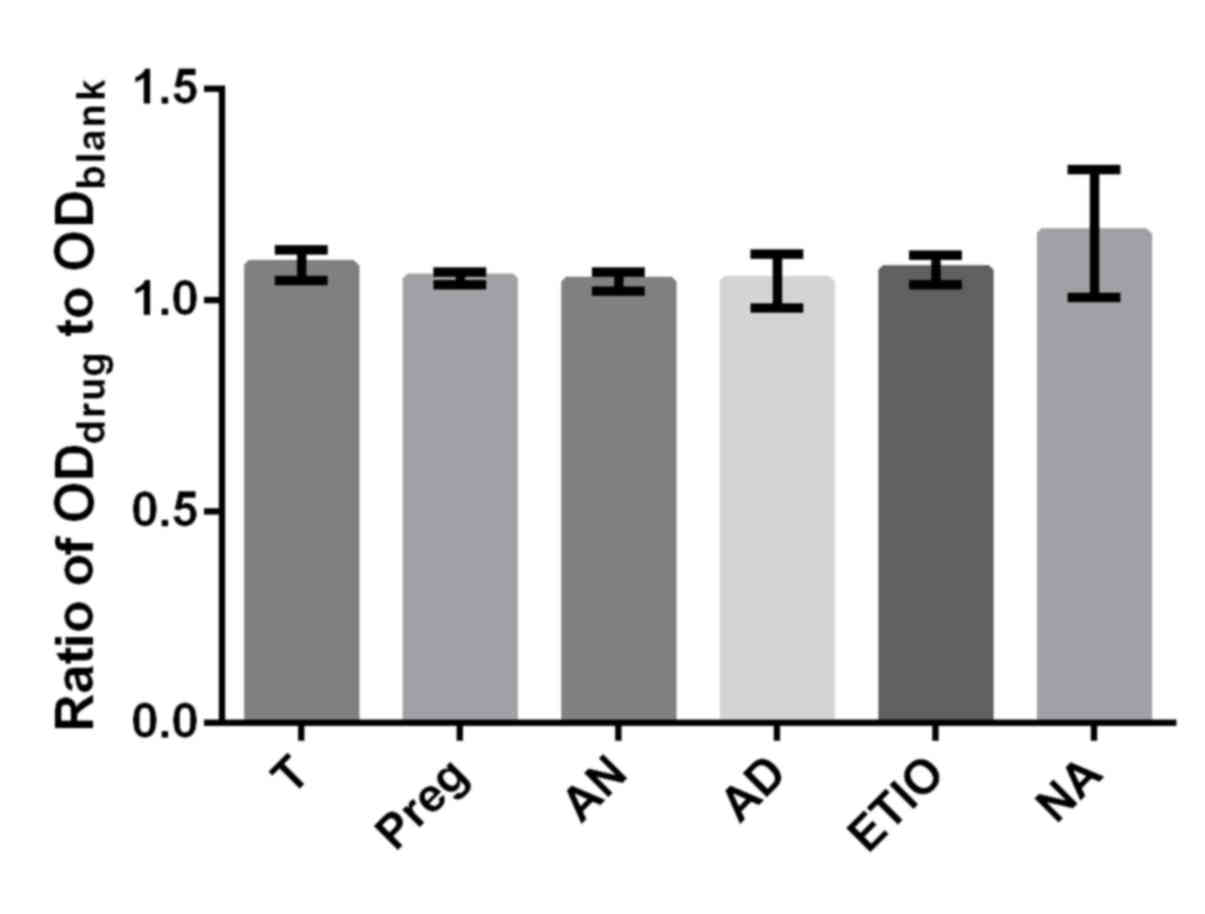
proliferation rate between T and the respective steroid (Preg, AD,
Etio An and NA) at concentrations of 10−10 and
10−8 mol/l were inspected in which the ratio of
ODdrug to ODblank was utilized as a
parameter. No obvious differences in proliferation efficiency
between T and the respective steroid (Preg, AD, Etio, An and NA) at
the concentration of 10−10 mol/l were observed in the
present study (Fig. 8). However, An
significantly decreased cell proliferation and NA significantly
increased cell proliferation compared with T at a concentration of
10−8 mol/l (P<0.05; Fig.
9).
ALP activity
The results of the MTS assay indicated that Preg,
AD, Etio, NA and T demonstrated enhancing effects on proliferation
at concentrations of 10−10 and 10−8 mol/l,
while An exhibited an effect at a concentration of 10−10
mol/l. Therefore, an ALP assay was performed with these steroids at
a concentration of 10−10 mol/l. The results indicated
that Preg, AD, Etio, An, NA and T significantly increased ALP
activity compared with the blank control (Fig. 10).
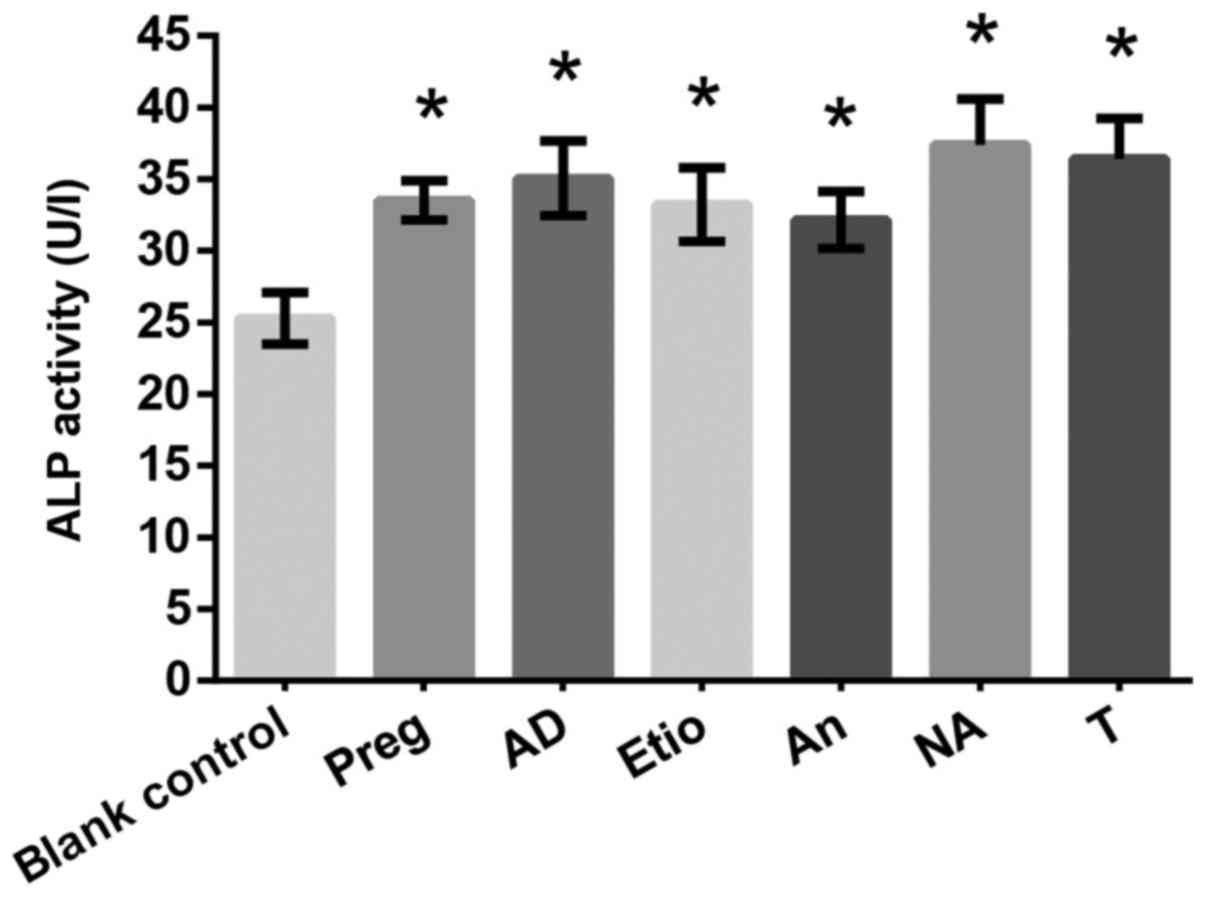 | Figure 10.Effects of drugs (Preg, AD, Etio, An,
NA and T) on alkaline phosphatase activity (U/l). *P<0.05 vs.
blank control. T, testosterone; Preg, pregnenolone; An,
androsterone; AD, androstendione; Etio, etiocholanone; NA,
nandrolone. |
Osteocalcin secretion
Osteoblasts were cultured with the drugs at a
concentration of 10−10 mol/l, and a BGP assay was
performed on day 5. As indicated in Fig. 11, Preg, AD, Etio, An, NA and T
significantly increased BGP secretion significantly at a
concentration of 10−10 mol/l, as compared with the blank
control.
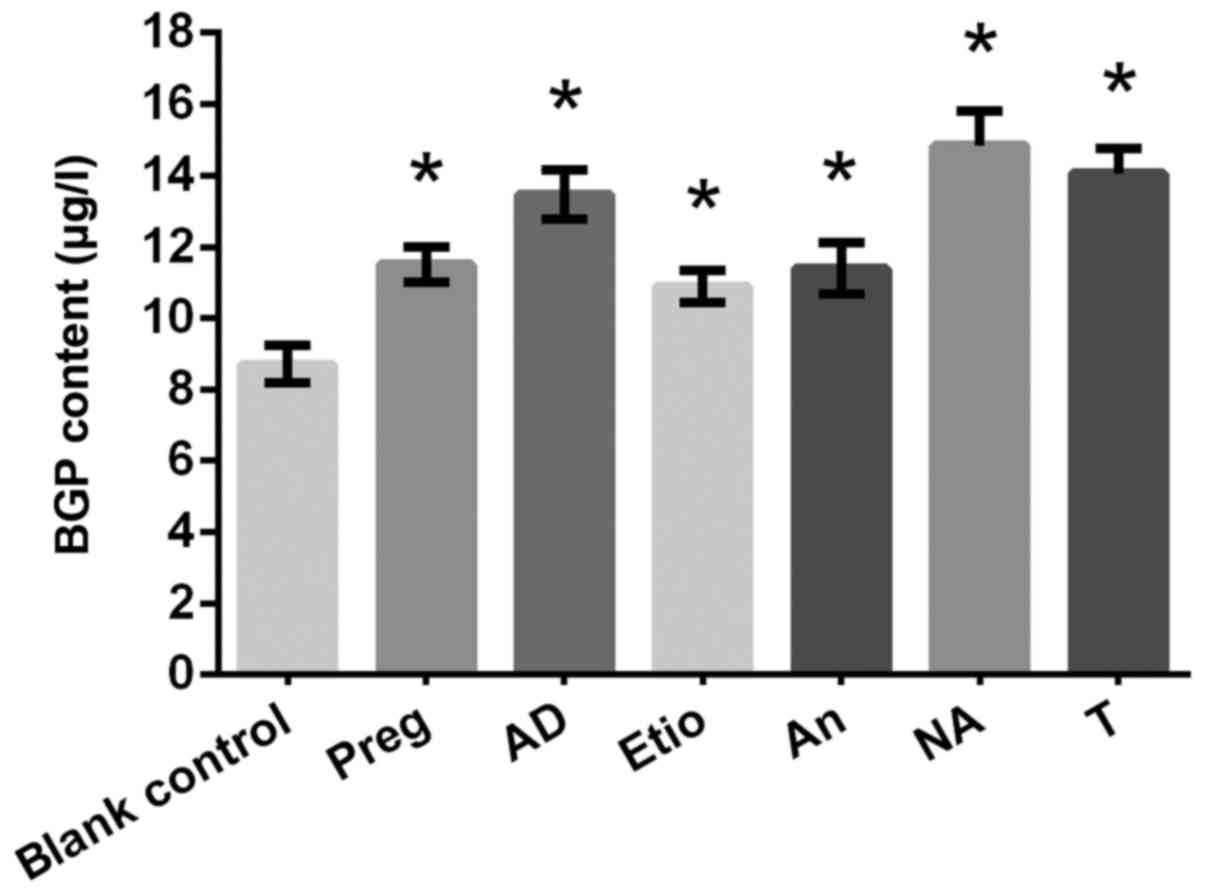 | Figure 11.Effects of drugs (Preg, AD, Etio, An,
NA and T) on the γ-carboxyglutamic-acid-containing protein content
(µg/l). *P<0.05 vs. blank control. T, testosterone; Preg,
pregnenolone; An, androsterone; AD, androstendione; Etio,
etiocholanone; NA, nandrolone. |
Discussion
A deficiency in sex steroids is well-established as
a causative factor for osteoporosis and bone loss, and thus hormone
replacement therapy is widely used in clinical practice (33–35).
Although the role that androgens play in bone regeneration has been
revealed to be via the estrogen receptors following conversion to
estrogen (36,37), certain studies have reported that
androgens may affect bone regeneration independently (17,20).
Among the androgens, T and DHT were the main steroids explored in
these studies (33). The present
study demonstrated that Preg, AD, Etio, and An could improve the
proliferation and differentiation of bone cells in
vitro.
Although the effects of these steroids on bone
health in vivo may not be an exact replication of those
in vitro, their clinically active effects in treating
osteoporosis are still worthy of consideration. Preg is a precursor
of androgens and estrogens, and AD is a precursor of T, DHT and
estrogens. An can be converted into DHT via 3α-hydroxysteroid
dehydrogenase and 17β-hydroxysteroid dehydrogenase, and could be
considered to be a metabolic intermediate in its own right
(38,39). Therefore, androgens and progestin,
and their metabolites, may promote bone regeneration.
The present study demonstrated that NA stimulated
osteoblast proliferation more efficiently compared with T at
concentrations of 10−8 mol/l. Nevertheless, the side
effects of NA in clinical practice cannot be ignored.
Nephrotoxicity of NA remains a multi-factorial and partly
irreversible side effect (40). A
previous study on Wistar female rats treated with NA decanoate
revealed that administration of NA damaged uterine tissue and
fertility (41). Etio and An have
always been considered to be metabolites of androgens that possess
no physiological effect (42,43).
However, to the best of our knowledge, this is the first study to
demonstrate that Etio and An could improve osteoblast
proliferation.
In addition to these active factors, the
extracellular environment may greatly affect bone health. Bone
health depends on a sufficient blood supply, since vascular
networks provide nutrients, oxygen, and progenitor cells that are
essential for bone function (44).
Therefore, the effects of androgens and progestin on blood vessels
need to be clarified in relation to bone maintenance. The
proliferation of endothelial cell vessels is important for forming
and/or renovating the extensive networks of blood vessels for bone
regeneration and fracture healing. Furthermore, a previous study
demonstrated that endothelial cells themselves enhanced bone
formation (45). Certain evidence
supports the beneficial effects of androgen on vascular functions.
One study indicated that androgen stimulates endothelial cell
proliferation via an androgen receptor/vascular endothelial growth
factor/cyclin A-mediated mechanism (46). Additional investigations into the
effect of Preg, AD, Etio, An and NA on these aspects should be
conducted.
In conclusion, the present study demonstrated for
the first time that Preg, AD, Etio, and An improved the
proliferation of osteoblasts in vitro. These steroids also
significantly increased ALP activity and BGP secretion of hFOB
cells. These findings may potentially represent novel therapeutic
strategies for the treatment of osteoporosis. Therapy with these
individual agents, in combination (e.g. estrogen plus the weak
androgen or estrogen plus Preg), or the application of progestogen
and the weak androgen during the ‘drug holiday’ of bisphosphonate
may be more effective and safe treatment strategies.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
XCW designed the study, performed the experiments,
analyzed and interpreted the data and drafted and revised the
manuscript. MQZ participated in the design of the study, and helped
to analyze and interpret the data. All authors have read and
approved the final version of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ellis SL, Grassinger J, Jones A, Borg J,
Camenisch T, Haylock D, Bertoncello I and Nilsson SK: The
relationship between bone, hemopoietic stem cells, and vasculature.
Blood. 118:1516–1524. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Felix R, Elford PR, Stoercklé C, Cecchini
M, Wetterwald A, Trechsel U, Fleisch H and Stadler BM: Production
of hemopoietic growth factors by bone tissue and bone cells in
culture. J Bone Miner Res. 3:27–36. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Guntur AR and Rosen CJ: Bone as an
endocrine organ. Endocr Pract. 18:758–762. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Cianferotti L and Brandi ML: Muscle-bone
interactions: Basic and clinical aspects. Endocrine. 45:165–177.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Rendina E, Hembree KD, Davis MR, Marlow D,
Clarke SL, Halloran BP, Lucas EA and Smith BJ: Dried plum's unique
capacity to reverse bone loss and alter bone metabolism in
postmenopausal osteoporosis model. PLoS One. 8:e605692013.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Faienza MF, Ventura A, Marzano F and
Cavallo L: Postmenopausal osteoporosis: The role of immune system
cells. Clin Dev Immunol. 2013:5759362013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Armas LA and Recker RR: Pathophysiology of
osteoporosis: New mechanistic insights. Endocrinol Metab Clin North
Am. 41:475–486. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Rachner TD, Khosla S and Hofbauer LC:
Osteoporosis: Now and the future. Lancet. 377:1276–1287. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Charles JF and Aliprantis AO: Osteoclasts:
More than ‘bone eaters’. Trends Mol Med. 20:449–459. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Clarke BL and Khosla S: Androgens and
bone. Steroids. 74:296–305. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Gallagher JC and Tella SH: Prevention and
treatment of postmenopausal osteoporosis. J Steroid Biochem Mol
Biol. 142:155–170. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Jackson RD and Mysiw WJ: Insights into the
epidemiology of postmenopausal osteoporosis: The women's health
initiative. Semin Reprod Med. 32:454–462. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Bowring CE and Francis RM: National
Osteoporosis Society's position statement on hormone replacement
therapy in the prevention and treatment of osteoporosis. Menopause
Int. 17:63–65. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Tirabassi G, Biagioli A and Balercia G:
Bone benefits of testosterone replacement therapy in male
hypogonadism. Panminerva Med. 56:151–163. 2014.PubMed/NCBI
|
|
15
|
Cartwright B, Robinson J, Seed PT,
Fogelman I and Rymer J: Hormone replacement therapy versus the
combined oral contraceptive pill in premature ovarian failure: A
randomized controlled trial of the effects on bone mineral density.
J Clin Endocrinol Metab. 101:3497–3505. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Jo DG, Lee HS, Joo YM and Seo JT: Effect
of testosterone replacement therapy on bone mineral density in
patients with Klinefelter syndrome. Yonsei Med J. 54:1331–1335.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kasperk CH, Wergedal JE, Farley JR,
Linkhart TA, Turner RT and Baylink DJ: Androgens directly stimulate
proliferation of bone cells in vitro. Endocrinology. 124:1576–1578.
1989. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Drake MT and Khosla S: Male osteoporosis.
Endocrinol Metab Clin North Am. 41:629–641. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Swartz CM and Young MA: Male hypogonadism
and bone fracture. N Engl J Med. 318:9961988. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Nieminen MS, Rämö MP, Viitasalo M,
Heikkilä P, Karjalainen J, Mäntysaari M and Heikkilä J: Serious
cardiovascular side effects of large doses of anabolic steroids in
weight lifters. Eur Heart J. 17:1576–1583. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lamb DR: Anabolic steroids in athletics:
How well do they work and how dangerous are they? Am J Sports Med.
12:31–38. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Bassil N, Alkaade S and Morley JE: The
benefits and risks of testosterone replacement therapy: A review.
Ther Clin Risk Manag. 5:427–448. 2009.PubMed/NCBI
|
|
23
|
Frick KM, Kim J, Tuscher JJ and Fortress
AM: Sex steroid hormones matter for learning and memory: Estrogenic
regulation of hippocampal function in male and female rodents.
Learn Mem. 22:472–493. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Devlin TM: Textbook of biochemistry: With
clinical correlations. 7th. Hoboken; New Jersey: 2010
|
|
25
|
Kaminski RM, Marini H, Kim WJ and Rogawski
MA: Anticonvulsant activity of androsterone and etiocholanolone.
Epilepsia. 46:819–827. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Motofei IG: A dual physiological character
for cerebral mechanisms of sexuality and cognition: Common somatic
peripheral afferents. BJU Int. 108:1634–1639. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Gruyter WD: Concise encyclopedia biology.
Berlin: 1996
|
|
28
|
Marques DRC, Marques D, Ibanez JF, Freitas
IB, Hespanha AC, Monteiro JF, Eggert M and Becker A: Effects of
nandrolone decanoate on time to consolidation of bone defects
resulting from osteotomy for tibial tuberosity advancement. Vet
Comp Orthop Traumatol. 30:351–356. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Nawata H, Tanaka S, Tanaka S, Takayanagi
R, Sakai Y, Yanase T, Ikuyama S and Haji M: Aromatase in bone cell:
Association with osteoporosis in postmenopausal women. J Steroid
Biochem Mol Biol. 53:165–174. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
da Costa KJ, Passos JJ, Gomes AD,
Sinisterra RD, Lanza CR and Cortés ME: Effect of testosterone
incorporation on cell proliferation and differentiation for
polymer-bioceramic composites. J Mater Sci Mater Med. 23:2751–2759.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zoch ML, Clemens TL and Riddle RC: New
insights into the biology of osteocalcin. Bone. 82:42–49. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Tuck SP and Francis RM: Testosterone, bone
and osteoporosis. Front Horm Res. 37:123–132. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Weinstein RS, Jilka RL, Parfitt AM and
Manolagas SC: The effects of androgen deficiency on murine bone
remodeling and bone mineral density are mediated via cells of the
osteoblastic lineage. Endocrinology. 138:4013–4021. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Diamond TH, Higano CS, Smith MR, Guise TA
and Singer FR: Osteoporosis in men with prostate carcinoma
receiving androgen-deprivation therapy: Recommendations for
diagnosis and therapies. Cancer. 100:892–899. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Daniell HW, Dunn SR, Ferguson DW, Lomas G,
Niazi Z and Stratte PT: Progressive osteoporosis during androgen
deprivation therapy for prostate cancer. J Urol. 163:181–186. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Sessa G and Weissmann G: Differential
effecs of etiocholanolone on phospholipid/cholesterol structures
containing either testosterone or estradiol. Biochim Biophys Acta.
150:173–180. 1968. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Novak FJ and Lambert JG: Pregnenolone,
testosterone, and estradiol in the migratory locust
Locustamigratoria; a gas chromatographical-mass spectrometrical
study. Gen Comp Endocrinol. 76:73–82. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Penning TM, Burczynski ME, Jez JM, Hung
CF, Lin HK, Ma H, Moore M, Palackal N and Ratnam K: Human
3alpha-hydroxysteroid dehydrogenase isoforms (AKR1C1-AKR1C4) of the
aldo-keto reductase superfamily: functional plasticity and tissue
distribution reveals roles in the inactivation and formation of
male and female sex hormones. Biochem J. 351:67–77. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Kamrath C, Hochberg Z, Hartmann MF, Remer
T and Wudy SA: Increased activation of the alternative ‘backdoor’
pathway in patients with 21-hydroxylase deficiency: Evidence from
urinary steroid hormone analysis. J Clin Endocrinol Metab.
97:E367–E375. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Tsitsimpikou C, Vasilaki F, Tsarouhas K,
Fragkiadaki P, Tzardi M, Goutzourelas N, Nepka C, Kalogeraki A,
Heretis I, Epitropaki Z, et al: Nephrotoxicity in rabbits after
long-term nandrolone decanoate administration. Toxicol Lett.
259:21–27. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Belardin LB, Simão VA, Leite GA, Chuffa LG
and Camargo IC: Dose-dependent effects and reversibility of the
injuries caused by nandrolone decanoate in uterine tissue and
fertility of rats. Birth Defects Res B Dev Reprod Toxicol.
101:168–177. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
The biological properties of
etiocholanolone, . Combined clinical staff conference at the
National Institutes of Health. Ann Intern Med. 67:1268–1295. 1967.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Kaminski RM, Marini H, Kim WJ and Rogawski
MA: Anticonvulsant activity of androsterone and etiocholanolone.
Epilepsia. 46:819–827. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Roux BM, Akar B, Zhou W, Stojkova K,
Barrera B, Brankov JG and Brey EM: Preformed vascular networks
survive and enhance vascularization in critical sized cranial
defectsTissue Eng Part A. 2018, View Article : Google Scholar : (Epub ahead of print).
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Pirraco RP, Iwata T, Yoshida T, Marques
AP, Yamato M, Reis RL and Okano T: Endothelial cells enhance the in
vivo bone-forming ability of osteogenic cell sheets. Lab Invest.
94:663–673. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Cai J, Hong Y, Weng C, Tan C,
Imperato-McGinley J and Zhu YS: Androgen stimulates endothelial
cell proliferation via an androgen receptor/VEGF/cyclin A-mediated
mechanism. Am J Physiol Heart Circ Physiol. 300:H1210–H1221. 2011.
View Article : Google Scholar : PubMed/NCBI
|