Introduction
For chemotherapy against gynecological cancer
centering on ovarian cancer, platinum and taxane preparations are
widely used. However, neurotoxicity, especially peripheral
neuropathy appearing as an adverse reaction to a taxane
preparation, paclitaxel, is a problem that remains to be solved.
Several patients develop intractable nervous symptoms persisting
for months after receiving the paclitaxel treatment, and this is
one of the factors that cause deterioration of patient quality of
life (QOL). Recently, it was reported that Goshajinkigan, a
traditional Japanese herbal medicine, is useful for coping with
chemotherapy-induced peripheral neuropathy (1).
However, in general, it is difficult to objectively
evaluate the severity of peripheral neuropathy, and very few
reports have referred to the objective electrophysiological
evaluation of neuropathy. We tried to undertake objective
evaluations of neuropathy, including the determination of current
perception thresholds (CPT) in gynecological patients who underwent
chemotherapy including paclitaxel, and developed peripheral
neuropathy. Such patients were randomly assigned to two groups
receiving and not receiving the Goshajinkigan treatment, and the
efficacy of Goshajinkigan in alleviating peripheral neuropathy was
investigated.
Patients and methods
Patients
This study was conducted under the approval of the
Institutional Review Board (IRB) of each study center. The subjects
were patients with ovarian or endometrial cancer who met all of the
following inclusion criteria: i) histological diagnosis of ovarian
or endometrial cancer, ii) having at least one cycle of paclitaxel
and carboplatin combination therapy (TC therapy) conducted as the
first chemotherapy and a National Cancer Institute Common
Terminology Criteria for Adverse Events v3.0 (NCI-CTCAE) peripheral
neuropathy grade of ≥1, iii) age of ≥20 and ≤70 years, and iv)
having provided written consent to participate in this study. The
exclusion criteria were as follows: i) previous use of
Goshajinkigan or vitamin B12 within the past 4 weeks, ii) any
schedule of using another anticancer agent during the study period,
and iii) presence of severe peripheral neuropathy at the initiation
of this study.
Methods
This study was conducted as a parallel group
randomized controlled trial by central registration, and the
registered patients were randomly assigned to the Goshajinkigan
non-treatment group (control group, Group A) and the Goshajinkigan
treatment group (Group B). In Group B, Tsumura Goshajinkigan
Extract Granules® 7.5 g/day (t.i.d.) and vitamin B12
(Methycobal®) at 1,500 μg/day (t.i.d.) were
administered, and in Group A, only vitamin B12 was administered.
The registered patients received a maximum of 6 cycles of TC
therapy (paclitaxel 175–180 mg/m2, i.v. on Day 1;
carboplatin AUC 5–6, i.v. on Day 1; each cycle, 21 days).
Evaluation items
Patients were observed for 6 weeks, and they
underwent CPT determination of the bilateral forefingers, visual
analogue scale (VAS) determination for numbness, grade
classification of motor and sensory neuropathy according to CTCAE
and examination with the subjective neuropathy symptom
questionnaire using modified functional assessment of cancer
therapy-taxane (FACT-Taxane) (2)
before the study treatment, after 3 weeks, and after 6 weeks of the
study treatment to compare the changes in neuropathy symptoms
between the two groups (Tables I
and II).
 | Table IEvaluation items. |
Table I
Evaluation items.
| Evaluation
item | Range |
|---|
Evaluation of
current perception threshold (CPT)
(evaluation of forefinger) | 0–999 (100 = 1
mAmp) |
| Grade of numbness
on visual analogue scale (VAS) | 0–10 |
| Motor and sensory
neuropathy grade (NCI-CTCAE v3.0) | Grade 0 (no
symptom) - Grade 5 |
| Subjective
neuropathy symptom questionnaire examination (FACT-Taxane) | 0–64 |
 | Table IISubjective neuropathy symptom
questionnaire using modified FACT-Taxane. |
Table II
Subjective neuropathy symptom
questionnaire using modified FACT-Taxane.
| Upper limb
symptoms |
| Hand numbness
or tingling pain |
| Other
uncomfortable hand sensation |
| Bilateral hand
swelling |
| Sore
fingertips |
| Trouble in
buttoning |
| Difficulty in
feeling the shape of a small object grasped in the hand |
| Lower limb
symptoms |
| Foot numbness
or tingling pain |
| Other
uncomfortable foot sensation |
| Bilateral foot
swelling |
| Difficulty in
walking |
| Other symptoms |
| Joint pain or
muscle convulsion |
| Whole body
swelling |
| Feeling of
whole body weakness |
| Worsened
hearing acuity |
| Noise in the
ear |
| Very concerned
about unusual appearance of hands and nails |
CPT test
The CPT examination has been reported to be useful
for the detection, screening, diagnosis and management of diseases
of peripheral neuropathy (3–5). The
principle and method of CPT determination are shown below. The
nerve diameter differs depending on the nerve type, and each nerve
has a specific frequency suitable for depolarization depending on
its diameter. It is therefore possible to undertake selective
quantitative evaluation of both large fibers and small fibers by
stimulating at different frequencies. The sine-wave current is
gradually increased from a low level (0–9.99 mA) at three different
frequencies of 5, 250 and 2,000 Hz at the region of measurement.
The minimum current perceived is the CPT of each subject. The CPT
at 2,000 Hz corresponds to a large myelinated fiber (Aβ), and the
CPT at 250 Hz corresponds to a small myelinated fiber (Aδ), while
the CPT at 5 Hz corresponds to an unmyelinated nerve (C) (6).
CPT was measured using the Neurometer NervScan
NS3000®. CPT range and within-site CPT ratio analyses of
the bilateral second fingers controlled by the median nerves were
performed.
The CPT Range Analysis compares raw CPT measures to
the normative ranges. CPT values below the minimum CPT normative
range qualify as hyperesthesia and indicate that the nerve fibers
are suffering from inflammation or are under regeneration, and
values above the maximum CPT normative range indicate hypoesthesia
associated with loss of function or neuropathy.
The within-site CPT ratio analysis is an analytical
method for measureing the ratio within the measurement region
(2,000 Hz/5 Hz; 2,000 Hz/250 Hz; 250 Hz/5 Hz). Ratios outside the
healthy ranges indicate very mild sensory abnormalities. The
normative data are displayed in Table
III.
 | Table IIINormative data for median nerve (100
CPT = 1 mAmp). |
Table III
Normative data for median nerve (100
CPT = 1 mAmp).
| Min | Max | Mean | SD |
|---|
| Ranges (Hz) | | | | |
| 2,000 | 120 | 398 | 226 | 80 |
| 250 | 22 | 180 | 81 | 42 |
| 5 | 16 | 101 | 46 | 27 |
| Within-site
ratio | | | | |
| 2 kHz/5 Hz | 2.03 | 14.7 | 6.2 | 4.2 |
| 2 kHz/250 Hz | 1.53 | 5.80 | 3.2 | 2.1 |
| 250 Hz/5 Hz | 0.83 | 4.38 | 2.0 | 1.1 |
Statistical analysis
Statistical analysis was performed using statistical
analysis software, SAS release 9.13 (SAS Institute Japan). The VAS
value, FACT-Taxane score, CTCAE neuropathy grade and therapeutic
effect on CPT (2,000, 250 or 5 Hz) were evaluated at each
time-point in each group employing Wilcoxon’s signed rank test.
Wilcoxon’s rank sum test was employed for the comparison of the
frequencies of the abnormal values of the above evaluation items
and the CPT test between Group A and B. Since this was an
exploratory study, multiplicity was not considered in any test. A
value of p<0.05 was regarded as significant for all data.
Results
In the period from March 8, 2007, to March 31, 2009,
31 patients were registered at the four study centers (Iwate
Medical University Hospital, Tottori University Hospital, Kitasato
University Hospital and Keio University Hospital). All the patients
met the inclusion criteria, although 2 patients dropped out of this
study since they developed deep vein thrombosis in the lower limb
during the TC therapy. Therefore, 29 valid patients (Group A, 15
patients; Group B, 14 patients) were included in the analysis. The
patient background factors did not differ between the two groups
(Table IV).
 | Table IVPatient background. |
Table IV
Patient background.
| Group A (not
administered Goshajinkigan; n=14) | Group B
(administered Goshajinkigan; n=15) | Wilcoxon’s rank sum
test |
|---|
| Age (years) | 59.7 | 55.6 | |
| Performance
status | | | |
| 0 | 9 | 9 | |
| 1 | 5 | 4 | |
| 2 | 0 | 2 | |
| Disease | | | |
| Ovarian
cancer | 12 | 12 | |
| Endometrial
cancer | 2 | 2 | |
| Multiple
cancer | 0 | 1 | |
| Vas score | 3.1±2.2 | 2.5±1.6 | p=0.827 |
| CTCAE neuropathy
grade | | | |
| Motor | 0.6±0.7 | 0.5±0.7 | p=0.760 |
| Sensory | 1.3±0.5 | 1.1±0.4 | p=0.404 |
| FACT-Taxane | 8.5±5.5 | 8.3±8.1 | p=0.896 |
| CPT range (5
Hz) | | | |
| Right | 113±68.3 | 98±46.6 | p=0.535 |
| Left | 120±62.6 | 121±117.9 | p=0.451 |
| CPT range (250
Hz) | | | |
| Right | 156±73.3 | 168±92.1 | p=0.947 |
| Left | 167±63.4 | 163±105.1 | p=0.385 |
| CPT range (2
kHz) | | | |
| Right | 346±140.8 | 375±127.2 | p=0.537 |
| Left | 366±138.8 | 341±133.6 | p=0.354 |
There were no significant differences between Group
A and Group B in terms of changes in VAS score, CTCAE neuropathy
grade, FACT-Taxane and CPT ranges in the period from before the
study treatment to Week 6 of study treatment (Table V). However, when limited to the
grade of sensory neuropathy, symptoms of Grade ≥3 were noted in
Group A at Week 3 or later (Week 6, 14.3%), but not in Group B
(Table VI).
 | Table VChanges in the outcome. |
Table V
Changes in the outcome.
| Group A (not
administered Goshajinkigan; n=14)
| Group B
(administered Goshajinkigan; n=15)
| Wilcoxon’s rank sum
testa |
|---|
| 0 weeks | 3 weeks | 6 weeks | 0 weeks | 3 weeks | 6 weeks |
|---|
| VAS | 3.1±2.2 | 2.9±3.3 | 3.7±3.4 | 2.5±1.6 | 3.8±2.7 | 3.4±2.7 | p=0.827 |
| CTCAE grade | | | | | | | |
| Motor
neuropathy | 0.6±0.7 | 0.6±1.0 | 0.8±1.1 | 0.5±0.7 | 0.6±0.7 | 0.8±0.9 | p=0.760 |
| Sensory
neuropathy | 1.3±0.5 | 1.0±1.0 | 1.3±1.0 | 1.1±0.4 | 1.3±0.5 | 1.4±0.5 | p=0.404 |
| FACT-Taxane | 8.5±5.5 | 6.8±6.9 | 8.9±8.4 | 8.3±8.1 | 9.9±7.1 | 8.2±7.0 | p=0.896 |
| CPT range (5
Hz) | | | | | | | |
| Right | 113±68.3 | 119±59.6 | 137±171.1 | 98±46.6 | 110±94.4 | 95±41.1 | p=0.793 |
| Left | 120±62.6 | 141±84.4 | 154±147.6 | 121±117.9 | 112±87.2 | 119±66.3 | p=0.948 |
| CPT range (250
Hz) | | | | | | | |
| Right | 156±73.3 | 141±67.6 | 140±80.7 | 168±92.1 | 151±122.9 | 148±30.6 | p=0.982 |
| Left | 167±63.4 | 164±76.7 | 159±127.0 | 163±105.1 | 169±93.0 | 164±57.8 | p=0.444 |
| CPT range (2,000
Hz) | | | | | | | |
| Right | 346±140.8 | 360±63.3 | 340±96.6 | 375±127.2 | 347±103.5 | 347±109.7 | p=0.611 |
| Left | 366±138.8 | 393±147.7 | 355±155.5 | 341±133.6 | 334±90.6 | 358±106.3 | p=0.743 |
 | Table VIChanges in CTCAE (sensory neuropathy)
grade. |
Table VI
Changes in CTCAE (sensory neuropathy)
grade.
| CTCAE sensory
neuropathy | Before study
treatment (no.) | After 3 weeks of
study treatment (no.) | After 6 weeks of
study treatment (no.) |
|---|
| Group A (not
administered Goshajinkigan) | | | |
| No symptom | 0 | 5 | 3 |
| Grade 1 | 10 | 5 | 6 |
| Grade 2 | 4 | 3 | 3 |
| Grade 3 | 0 | 1 | 2 |
| Grade 4 | 0 | 0 | 0 |
| Grade 5 | 0 | 0 | 0 |
| Group B
(administered Goshajinkigan) | | | |
| No symptom | 0 | 0 | 0 |
| Grade 1 | 13 | 11 | 9 |
| Grade 2 | 2 | 4 | 6 |
| Grade 3 | 0 | 0 | 0 |
| Grade 4 | 0 | 0 | 0 |
| Grade 5 | 0 | 0 | 0 |
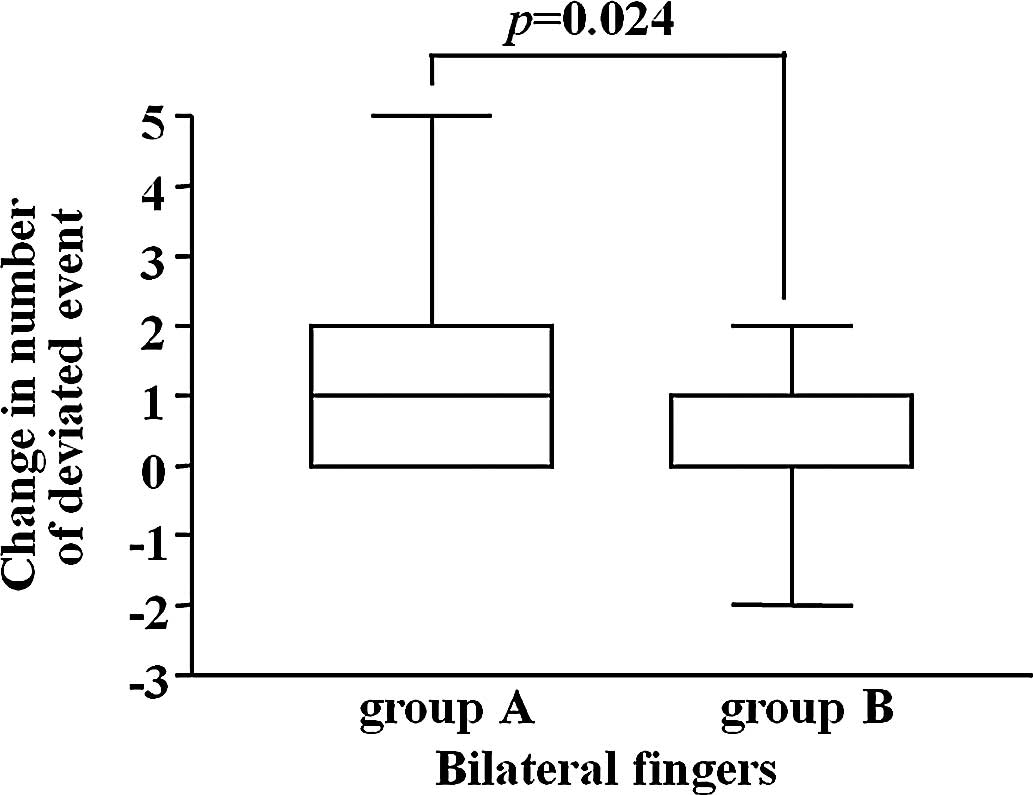
The change in the frequency of abnormal CPT range at
6 weeks of administration from that before treatment was compared
between the groups employing Wilcoxon’s rank sum test, but no
significant difference was observed. However, regarding the change
in the frequency of abnormal CPT ratio, the frequency was
significantly lower in Group B than in Group A (Fig. 1).
Discussion
Paclitaxel, irinotecan hydrochloride and liposomal
doxorubicin, among others, have specific non-hematological
toxicities, inducing the deterioration of patient QOL. Therefore,
it is important to decrease such toxicities. For neurotoxicity in
particular, peripheral neuropathy is known as an adverse reaction
to paclitaxel (7). Vasey et
al (8) reported that the
incidence of peripheral neuropathy in TC therapy was as high as 78%
for sensory disorder and 16% for motor disorder. Recently, it was
reported that Goshajinkigan, a traditional Japanese herbal
medicine, is effective for chemotherapy-induced neurotoxicity.
However, there are few reports on the electrophysiological
evaluation of the severity of peripheral neuropathy.
In this study, we evaluated the peripheral
neuropathy-alleviating effects of Goshajinkigan administered to
patients with gynecological malignancy undergoing TC therapy in a
parallel group randomized controlled trial. There were no
significant differences between the presence and absence of
Goshajinkigan treatment after 3 and 6 weeks of the study treatment
in VAS evaluation for numbness, subjective neuropathy symptom
questionnaire using FACT-Taxane and neuropathy grade according to
CTCAE. Since an anticancer drug induces not only neuropathy, but
also various complications, such as gastrointestinal symptoms,
infections and mental symptoms, it is difficult to avoid biases in
the cases of VAS and FACT-Taxane. This was considered to be the
cause of the failure to obtain significant differences. However, in
CTCAE for sensory neuropathy, an event of Grade 3 was noted in 2
patients (14.3%) of the Goshajinkigan non-treatment group, while no
event of Grade 3 was noted in the Goshajinkigan treatment group.
This suggests that the progression of neuropathy can be delayed by
the use of Goshajinkigan.
It is generally difficult to evaluate the severity
of peripheral neuropathy objectively. The CPT-measuring method has
gradually prevailed since it was developed by Katims et al
(9) in 1986, showing a high
prevalence rate in the United States. It has been reported that
this method can be used to make detailed neurological evaluations
of diabetic peripheral neuropathy, carpal tunnel syndrome and
alcoholic peripheral neuropathy (3–5).
No significant difference was noted in the changes
in the CPT range between the groups with and without Goshajinkigan
treatment, but the frequency of abnormal CPT ratio was
significantly lower in the Goshajinkigan-treatment group. The
progression of TC therapy-induced neurotoxicity over the 6-week
observation period was not so marked so that it was not reflected
in the CPT range, showing no significant difference. By contrast,
the CPT ratio showed a significant difference as it may have
reflected very mild sensory abnormalities, suggesting that
Goshajinkigan inhibited the very early progression of
neurotoxicity.
For the prevention and treatment of peripheral
neuropathy induced by anticancer agents, various animal experiments
and clinical studies were conducted using, in addition to NSAIDs
and steroids, antihistaminic drugs, NGF (10,11),
IGF (12), GDNF (13), amifostine (14,15),
glutathione (16), α-lipoic acid
(17), gabapentin (18) and carbamazepine (19), among others, but there are no
established methods.
Goshajinkigan is a kampo drug prepared by adding
Goshitsu (Achyranthes root, anti-allergic effect) and
Shazenshi (Plantago seed, diuretic effect and
interferon-inducing effect) to Hachimijiogan, which is composed of
the following mixed crude drugs: Rehmanma root, Cornus
fruit, Dioscorea rhizome, Alisma rhizome,
Poria sclerotium, Moutan bark, Cinnamon bark
and processed Aconite root, and was originally considered to
be effective against ‘numbness’ due to diabetic peripheral
neuropathy. Goshajinkigan is also effective for lumbar canal
stenosis, lumbar spondylosis deformans and arteriosclerosis
obliterans in the elderly. Similar to the pharmacological effect of
Goshajinkigan, it is considered that the analgesic effect is
exerted by the suppression of pain-transmitting substance release
by κ-opioid receptor stimulation mediated by dynorphin, an
endogenous opioid substance released by ShujiBushi (processed
Aconite root) (20). It is
also considered that the analgesic effect is exerted through the
improvement of peripheral nocireceptor sensitivity, vasodilation
and peripheral circulation by the promotion of NO production due to
the effects of Takusha (Alisma rhizome) and Sanyaku
(Dioscorea rhizome) mediated by bradykinin B2 receptor and
muscarinic acetylcholine receptor (21).
It is expected that Goshajinkigan may become a
first-line therapy against the neurotoxicity of anticancer drugs
not only by alleviating the subjective symptoms of neuropathy, but
also by repairing the nerves. It is essential to delay the
progression of neuropathy in current cancer chemotherapy, which
should be performed with maintenance of QOL.
References
|
1.
|
T KonoH MishimaM ShimadaS MoritaJ
SakamotoPreventive effect of goshajinkigan on peripheral
neurotoxicity of FOLFOX therapy: a placebo-controlled double-blind
randomized phase II study (the GONE Study)Jpn J Clin
Oncol39847849200910.1093/jjco/hyp10019734172
|
|
2.
|
D CellaA PetermanS HudgensK WebsterMA
SocinskiMeasuring the side effects of taxane therapy in oncology:
the functional assesment of cancer therapy-taxane
(FACT-taxane)Cancer15822831200310.1002/cncr.1157812910528
|
|
3.
|
K TakekumaF AndoN NiinoH
ShimokawaPrevalence of hyperesthesia detected by current perception
threshold test in subjects with glucose metabolic impairments in a
communityIntern
Med4111241129200210.2169/internalmedicine.41.112412521200
|
|
4.
|
M OishiY MochizukiY SuzukiK OgawaT
NaganumaY NishijoT MizutaniCurrent perception threshold and
sympathetic skin response in diabetic and alcoholic
polyneuropathiesIntern
Med41819822200210.2169/internalmedicine.41.81912413002
|
|
5.
|
A NishimuraT OguraH HaseA MakinodanT HojoY
KatsumiK YagiY MikamiT KuboA correlative electrophysiologic study
of nerve fiber involvement in carpal tunnel syndrome using current
perception thresholdsClin
Neurophysiol11519211924200410.1016/j.clinph.2004.03.022
|
|
6.
|
American Association of Electrodiagnostic
MedicineTechnology review: the neurometer® current
perception threshold (CPT)Muscle
Nerve22523531199910.1002/(SICI)1097-4598(199904)22:4%3C523::AID-MUS16%3E3.0.CO;2-1
|
|
7.
|
K NodaM IkedaR KudoPhase II study of
paclitaxel (BMS-181339) in patients with ovarian cancer by 3-hour
intravenous infusionGan To Kagaku Ryoho2331732519968712825
|
|
8.
|
PA VaseyGC JaysonA GordonPhase III
randomized trial of docetaxel-carboplatin versus
paclitaxel-carboplatin as first-line chemotherapy for ovarian
carcinomaJ Natl Cancer
Inst9616821691200410.1093/jnci/djh32315547181
|
|
9.
|
JJ KatimsDM LongLK NgTranscutaneous nerve
stimulation. Frequency and waveform specificity in humansAppl
Neurophysiol49869119863490223
|
|
10.
|
SC ApfelJC ArezzoL LipsonJA KesslerNerve
growth factor prevents experimental cisplatin neuropathyAnn
Neurol317680199210.1002/ana.4103101141543351
|
|
11.
|
K HayakawaT ItohH NiwaT MutohG SobueNGF
prevention of neurotoxicity induced by cisplatin, vincristine and
taxol depends on toxicity of each drug and NGF treatment schedule:
in vitro study of adult rat sympathetic ganglion explantsBrain
Res794313319199810.1016/S0006-8993(98)00305-9
|
|
12.
|
PC ContrerasJL VaughtJA GrunerC BrosnanC
StefflerJC ArezzoME LewisJA KesslerSC ApfelInsulin-like growth
factor-I prevents development of a vincristine neuropathy in
miceBrain Res7742026199710.1016/S0006-8993(97)81682-49452187
|
|
13.
|
TJ BoucherK OkuseDL BennettJB MunsonJN
WoodSB McMahonPotent analgesic effects of GDNF in neuropathic pain
statesScience290124127200010.1126/science.290.5489.12411021795
|
|
14.
|
G SelvaggiCP BelaniCarboplatin and
paclitaxel in non-small cell lung cancer: the role of
amifostineSemin Oncol265160199910348261
|
|
15.
|
AS PlantingG CatimelPH de MulderA de
GraeffF HöppenerJ VerweijW OsterJB VermorkenRandomized study of a
short course of weekly cisplatin with or without amifostine in
advanced head and neck cancerAnn
Oncol10693700199910.1023/A:100835350591610442192
|
|
16.
|
S CascinuL CordellaE del FerroM FronzoniG
CatalanoNeuroprotective effect of reduced glutathione on
cisplatin-based chemotherapy in advanced gastric cancer: a
randomized double-blind placebo-controlled trialJ Clin
Oncol1326321995
|
|
17.
|
D ZieglerM ReljanovicH MehnertFA
GriesAlpha-lipoic acid in the treatment of diabetic polyneuropathy
in Germany: current evidence from clinical trialsExp Clin
Endocrinol Diabetes107421430199910.1055/s-0029-121213210595592
|
|
18.
|
RD RaoJC MichalakJA SloanEfficacy of
gabapentin in the management of chemotherapy-induced peripheral
neuropathy: a phase 3 randomized, double-blind, placebo-controlled,
crossover trial (N00C3)Cancer11021102118200710.1002/cncr.23008
|
|
19.
|
F EckelR SchmelzH AdelsbergerJ ErdmannS
QuasthoffC LerschPrevention of oxaliplatin-induced neuropathy by
carbamazepine. A pilot studyDtsch Med
Wochenschr1277882200210.1055/s-2002-1959411797144
|
|
20.
|
Y SuzukiK GotoA IshigeY KomatsuJ
KameiAntinociceptive effect of Gosha-jinki-gan, a Kampo medicine,
in streptozotocin-induced diabetic miceJpn J
Pharmacol79169175199910.1254/jjp.79.16910202852
|
|
21.
|
Y SuzukiK GotoA IshigeY KomatsuJ
KameiAntinociceptive mechanism of Gosha-jinki-gan in
streptozotocin-induced diabetic animals: role of nitric oxide in
the peripheryJpn J
Pharmacol79387391199910.1254/jjp.79.38710230868
|