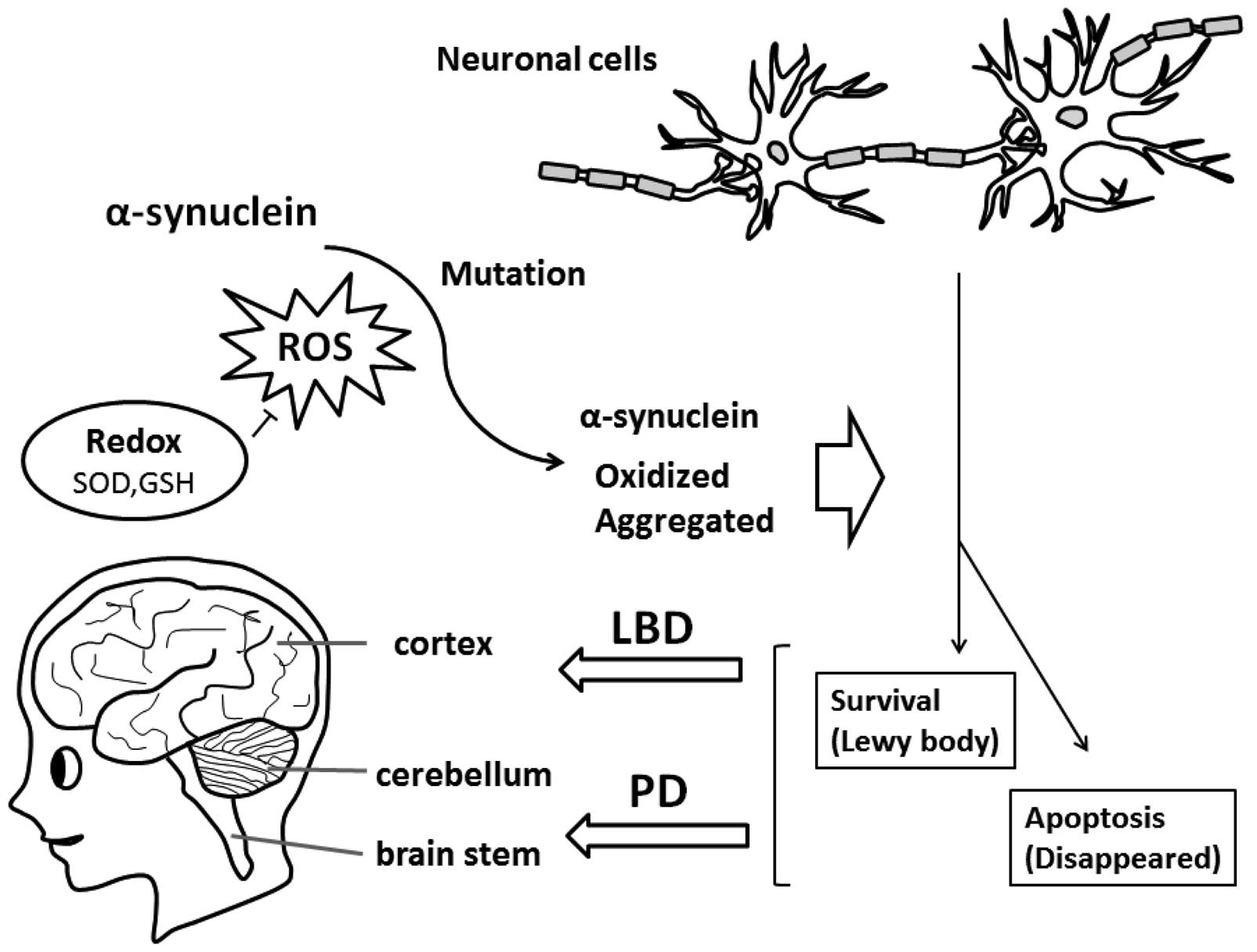
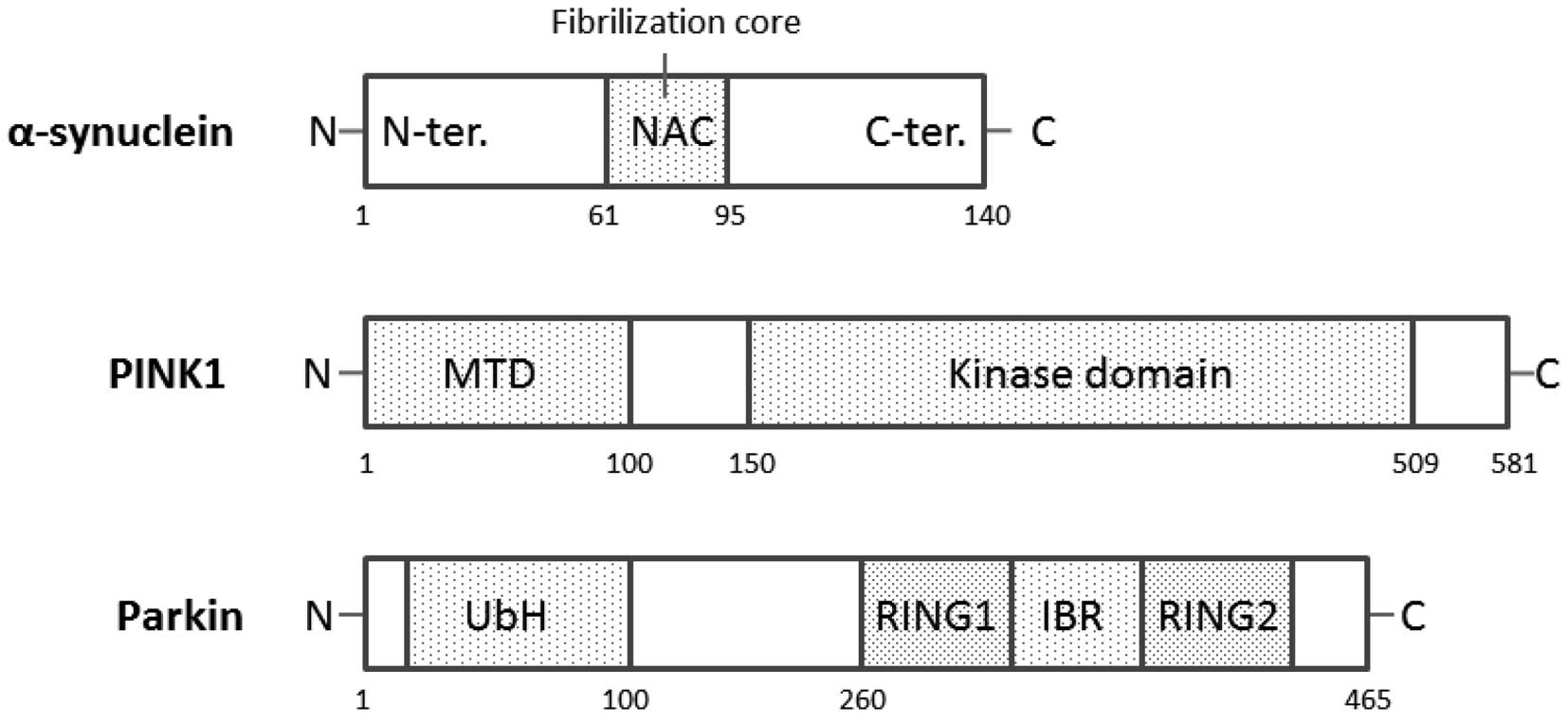
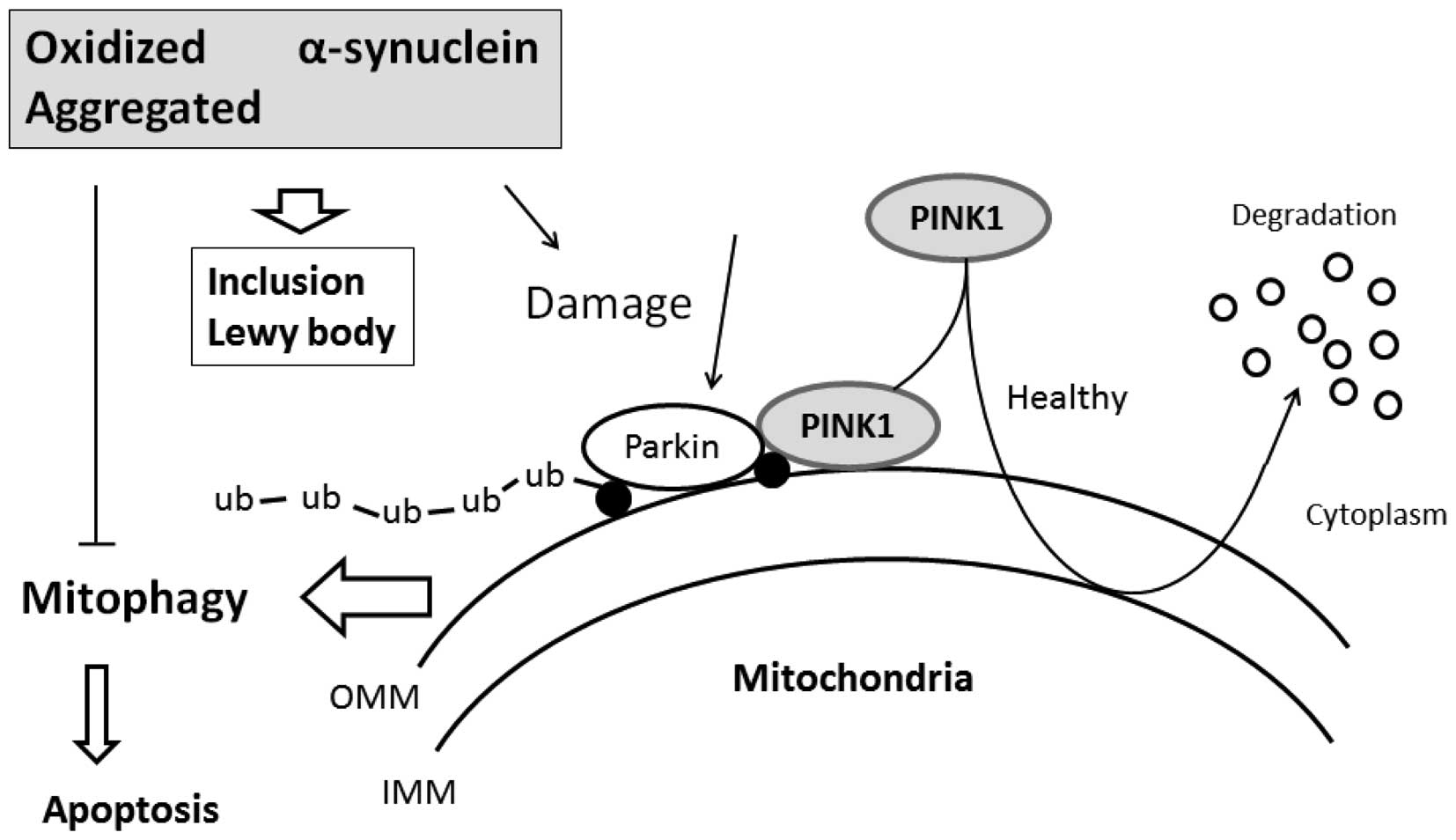
|
1
|
Overk CR and Masliah E: Pathogenesis of
synaptic degeneration in Alzheimer’s disease and Lewy body disease.
Biochem Pharmacol. 88:508–516. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Walker LC and LeVine H III: Corruption and
spread of pathogenic proteins in neurodegenerative diseases. J Biol
Chem. 287:33109–33115. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Schwarz S, Froelich L and Burns A:
Pharmacological treatment of dementia. Curr Opin Psychiatry.
25:542–550. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Di Giovanni S, Eleuteri S, Paleologou KE,
Yin G, Zweckstetter M, Carrupt PA and Lashuel HA: Entacapone and
tolcapone, two catechol O-methyltransferase inhibitors, block
fibril formation of alpha-synuclein and beta-amyloid and protect
against amyloid-induced toxicity. J Biol Chem. 285:14941–14954.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Crews L, Tsigelny I, Hashimoto M and
Masliah E: Role of synucleins in Alzheimer’s disease. Neurotox Res.
16:306–317. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Goldstein DS, Holmes C, Kopin IJ and
Sharabi Y: Intra-neuronal vesicular uptake of catecholamines is
decreased in patients with Lewy body diseases. J Clin Invest.
121:3320–3330. 2011. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
Onofrj M, Bonanni L, Manzoli L and Thomas
A: Cohort study on somatoform disorders in Parkinson disease and
dementia with Lewy bodies. Neurology. 74:1598–1606. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Paleologou KE and El-Agnaf OM: α-synuclein
aggregation and modulating factors. Subcell Biochem. 65:109–164.
2012. View Article : Google Scholar
|
|
9
|
Cheng F, Li X, Li Y, Wang C, Wang T, Liu
G, Baskys A, Uéda K, Chan P and Yu S: α-Synuclein promotes
clathrin-mediated NMDA receptor endocytosis and attenuates
NMDA-induced dopaminergic cell death. J Neurochem. 119:815–825.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Song JX, Lu JH, Liu LF, Chen LL,
Durairajan SS, Yue Z, Zhang HQ and Li M: HMGB1 is involved in
autophagy inhibition caused by SNCA/α-synuclein overexpression: a
process modulated by the natural autophagy inducer corynoxine B.
Autophagy. 10:144–154. 2014. View Article : Google Scholar
|
|
11
|
Settembre C, Fraldi A, Jahreiss L,
Spampanato C, Venturi C, Medina D, de Pablo R, Tacchetti C,
Rubinsztein DC and Ballabio A: A block of autophagy in lysosomal
storage disorders. Hum Mol Genet. 17:119–129. 2008. View Article : Google Scholar
|
|
12
|
Todde V, Veenhuis M and van der Klei IJ:
Autophagy: principles and significance in health and disease.
Biochim Biophys Acta. 1792:3–13. 2009. View Article : Google Scholar
|
|
13
|
Pan T, Kondo S, Le W and Jankovic J: The
role of autophagy-lysosome pathway in neurodegeneration associated
with Parkinson’s disease. Brain. 131:1969–1978. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Matus S, Valenzuela V and Hetz C: A new
method to measure autophagy flux in the nervous system. Autophagy.
10:710–714. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Giordano S, Darley-Usmar V and Zhang J:
Autophagy as an essential cellular antioxidant pathway in
neurodegenerative disease. Redox Biol. 2:82–90. 2013. View Article : Google Scholar
|
|
16
|
Spencer B, Potkar R, Trejo M, Rockenstein
E, Patrick C, Gindi R, Adame A, Wyss-Coray T and Masliah E: Beclin
1 gene transfer activates autophagy and ameliorates the
neurodegenerative pathology in alpha-synuclein models of
Parkinson’s and Lewy body diseases. J Neurosci. 29:13578–13588.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Isella V, Rucci F, Traficante D, Mapelli
C, Ferri F and Appollonio IM: The applause sign in cortical and
cortical-subcortical dementia. J Neurol. 260:1099–1103. 2013.
View Article : Google Scholar
|
|
18
|
Kahle PJ, Neumann M, Ozmen L, Müller V,
Odoy S, Okamoto N, Jacobsen H, Iwatsubo T, Trojanowski JQ,
Takahashi H, Wakabayashi K, Bogdanovic N, Riederer P, Kretzschmar
HA and Haass C: Selective insolubility of alpha-synuclein in human
Lewy body diseases is recapitulated in a transgenic mouse model. Am
J Patholx. 159:2215–2225. 2013. View Article : Google Scholar
|
|
19
|
Robinson PA: Protein stability and
aggregation in Parkinson’s disease. Biochem J. 413:1–13. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Olanow CW, Perl DP, DeMartino GN and
McNaught KS: Lewy-body formation is an aggresome-related process: a
hypothesis. Lancet Neurol. 3:496–503. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Braak H, Müller CM, Rüb U, Ackermann H,
Bratzke H, de Vos RA and Del Tredici K: Pathology associated with
sporadic Parkinson’s disease - where does it end? J Neural Transm
Suppl. 70:89–97. 2006.
|
|
22
|
Luk KC, Hyde EG, Trojanowski JQ and Lee
VM: Sensitive fluorescence polarization technique for rapid
screening of alpha-synuclein oligomerization/fibrillization
inhibitors. Biochemistry. 46:12522–12529. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ghosh D, Mondal M, Mohite GM, Singh PK,
Ranjan P, Anoop A, Ghosh S, Jha NN, Kumar A and Maji SK: The
Parkinson’s disease-associated H50Q mutation accelerates
α-Synuclein aggregation in vitro. Biochemistry. 52:6925–6927. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Chai YJ, Kim D, Park J, Zhao H, Lee SJ and
Chang S: The secreted oligomeric form of α-synuclein affects
multiple steps of membrane trafficking. FEBS Lett. 587:452–459.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Campbell BC, McLean CA, Culvenor JG, Gai
WP, Blumbergs PC, Jäkälä P, Beyreuther K, Masters CL and Li QX: The
solubility of alpha-synuclein in multiple system atrophy differs
from that of dementia with Lewy bodies and Parkinson’s disease. J
Neurochem. 76:87–96. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wakabayashi K, Yoshimoto M, Fukushima T,
Koide R, Horikawa Y, Morita T and Takahashi H: Widespread
occurrence of alpha-synuclein/NACP-immunoreactive neuronal
inclusions in juvenile and adult-onset Hallervorden-Spatz disease
with Lewy bodies. Neuropathol Appl Neurobiol. 25:363–368. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Sharon R, Goldberg MS, Bar-Josef I,
Betensky RA, Shen J and Selkoe DJ: alpha-synuclein occurs in
lipid-rich high molecular weight complexes, binds fatty acids, and
shows homology to the fatty acid-binding proteins. Proc Natl Acad
Sci USA. 98:9110–9115. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Dryanovski DI, Guzman JN, Xie Z, Galteri
DJ, Volpicelli-Daley LA, Lee VM, Miller RJ, Schumacker PT and
Surmeier DJ: Calcium entry and α-synuclein inclusions elevate
dendritic mitochondrial oxidant stress in dopaminergic neurons. J
Neurosci. 33:10154–10164. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Müller SK, Bender A, Laub C, Högen T,
Schlaudraff F, Liss B, Klopstock T and Elstner M: Lewy body
pathology is associated with mitochondrial DNA damage in
Parkinson’s disease. Neurobiol Aging. 34:2231–2233. 2013.
View Article : Google Scholar
|
|
30
|
Wilkaniec A, Strosznajder JB and Adamczyk
A: Toxicity of extracellular secreted alpha-synuclein: its role in
nitrosative stress and neurodegeneration. Neurochem Int.
62:776–783. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Xilouri M, Brekk OR and Stefanis L:
α-Synuclein and protein degradation systems: a reciprocal
relationship. Mol Neurobiol. 47:537–551. 2013. View Article : Google Scholar
|
|
32
|
Chinta SJ, Mallajosyula JK, Rane A and
Andersen JK: Mitochondrial α-synuclein accumulation impairs complex
I function in dopaminergic neurons and results in increased
mitophagy in vivo. Neurosci Lett. 486:235–239. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Gandhi S, Muqit MM, Stanyer L, Healy DG,
Abou-Sleiman PM, Hargreaves I, Heales S, Ganguly M, Parsons L, Lees
AJ, Latchman DS, Holton JL, Wood NW and Revesz T: PINK1 protein in
normal human brain and Parkinson’s disease. Brain. 129:1720–1731.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Hong L, Ko HW, Gwag BJ, Joe E, Lee S, Kim
YT and Suh YH: The cDNA cloning and ontogeny of mouse
alpha-synuclein. Neuroreport. 9:1239–1243. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
El-Agnaf OM, Jakes R, Curran MD, Middleton
D, Ingenito R, Bianchi E, Pessi A, Neill D and Wallace A:
Aggregates from mutant and wild-type alpha-synuclein proteins and
NAC peptide induce apoptotic cell death in human neuroblastoma
cells by formation of beta-sheet and amyloid-like filaments. FEBS
Lett. 440:71–75. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Jensen PH, Hojrup P, Hager H, Nielsen MS,
Jacobsen L, Olesen OF, Gliemann J and Jakes R: Binding of Abeta to
alpha- and beta-synucleins: identification of segments in
alpha-synuclein/NAC precursor that bind Abeta and NAC. Biochem J.
323:539–546. 1997.PubMed/NCBI
|
|
37
|
Schapira AH and Gegg M: Mitochondrial
contribution to Parkinson’s disease pathogenesis. Parkinsons Dis.
2011:1591602011.
|
|
38
|
Liu F, Hindupur J, Nguyen JL, Ruf KJ, Zhu
J, Schieler JL, Bonham CC, Wood KV, Davisson VJ and Rochet JC:
Methionine sulfoxide reductase A protects dopaminergic cells from
Parkinson’s disease-related insults. Free Radic Biol Med.
45:242–255. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Dias V, Junn E and Mouradian MM: The role
of oxidative stress in Parkinson’s disease. J Parkinsons Dis.
3:461–491. 2013.
|
|
40
|
Weber TA and Reichert AS: Impaired quality
control of mitochondria: aging from a new perspective. Exp
Gerontol. 45:503–511. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Israeli E and Sharon R: Beta-synuclein
occurs in vivo in lipid-associated oligomers and forms
hetero-oligomers with alpha-synuclein. J Neurochem. 108:465–474.
2009. View Article : Google Scholar
|
|
42
|
Valente EM, Abou-Sleiman PM, Caputo V,
Muqit MM, Harvey K, Gispert S, Ali Z, Del Turco D, Bentivoglio AR,
Healy DG, Albanese A, Nussbaum R, González-Maldonado R, Deller T,
Salvi S, Cortelli P, Gilks WP, Latchman DS, Harvey RJ, Dallapiccola
B, Auburger G and Wood NW: Hereditary early-onset Parkinson’s
disease caused by mutations in PINK1. Science. 304:1158–1160. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Blackinton JG, Anvret A, Beilina A, Olson
L, Cookson MR and Galter D: Expression of PINK1 mRNA in human and
rodent brain and in Parkinson’s disease. Brain Res. 1184:10–16.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Weihofen A, Thomas KJ, Ostaszewski BL,
Cookson MR and Selkoe DJ: Pink1 forms a multiprotein complex with
Miro and Milton, linking Pink1 function to mitochondrial
trafficking. Biochemistry. 48:2045–2052. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Jin SM, Lazarou M, Wang C, Kane LA,
Narendra DP and Youle RJ: Mitochondrial membrane potential
regulates PINK1 import and proteolytic destabilization by PARL. J
Cell Biol. 191:933–942. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Beilina A, Van Der Brug M, Ahmad R,
Kesavapany S, Miller DW, Petsko GA and Cookson MR: Mutations in
PTEN-induced putative kinase 1 associated with recessive
parkinsonism have differential effects on protein stability. Proc
Natl Acad Sci USA. 102:5703–5708. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Pridgeon JW, Olzmann JA, Chin LS and Li L:
PINK1 protects against oxidative stress by phosphorylating
mitochondrial chaperone TRAP1. PLoS Biol. 5:e1722007. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Michiorri S, Gelmetti V, Giarda E,
Lombardi F, Romano F, Marongiu R, Nerini-Molteni S, Sale P, Vago R,
Arena G, Torosantucci L, Cassina L, Russo MA, Dallapiccola B,
Valente EM and Casari G: The Parkinson-associated protein PINK1
interacts with Beclin1 and promotes autophagy. Cell Death Differ.
17:962–974. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Matsuda S, Kitagishi Y and Kobayashi M:
Function and characteristics of PINK1 in mitochondria. Oxid Med
Cell Longev. 2013:6015872013. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Rakovic A, Shurkewitsch K, Seibler P,
Grünewald A, Zanon A, Hagenah J, Krainc D and Klein C: Phosphatase
and tensin homolog (PTEN)-induced putative kinase 1
(PINK1)-dependent ubiquitination of endogenous Parkin attenuates
mitophagy: study in human primary fibroblasts and induced
pluripotent stem cell-derived neurons. J Biol Chem. 288:2223–2237.
2013. View Article : Google Scholar :
|
|
51
|
Chu CT: A pivotal role for PINK1 and
autophagy in mitochondrial quality control: implications for
Parkinson disease. Hum Mol Genet. 19:R28–R37. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Greene AW, Grenier K, Aguileta MA, Muise
S, Farazifard R, Haque ME, McBride HM, Park DS and Fon EA:
Mitochondrial processing peptidase regulates PINK1 processing,
import and Parkin recruitment. EMBO Rep. 13:378–385. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Dexter DT and Jenner P: Parkinson disease:
from pathology to molecular disease mechanisms. Free Radic Biol
Med. 62:132–144. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Murakami T, Moriwaki Y, Kawarabayashi T,
Nagai M, Ohta Y, Deguchi K, Kurata T, Morimoto N, Takehisa Y,
Matsubara E, Ikeda M, Harigaya Y, Shoji M, Takahashi R and Abe K:
PINK1, a gene product of PARK6, accumulates in
alpha-synucleinopathy brains. J Neurol Neurosurg Psychiatry.
78:653–654. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Butler EK, Voigt A, Lutz AK, Toegel JP,
Gerhardt E, Karsten P, Falkenburger B, Reinartz A, Winklhofer KF
and Schulz JB: The mitochondrial chaperone protein TRAP1 mitigates
α-Synuclein toxicity. PLoS Genet. 8:e10024882012. View Article : Google Scholar
|
|
56
|
Bornhorst J, Chakraborty S, Meyer S,
Lohren H, Brinkhaus SG, Knight AL, Caldwell KA, Caldwell GA, Karst
U, Schwerdtle T, Bowman A and Aschner M: The effects of pdr1,
djr1.1 and pink1 loss in manganese-induced toxicity and the role of
α-synuclein in C. elegans. Metallomics. 6:476–490. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Sampaio-Marques B, Felgueiras C, Silva A,
Rodrigues M, Tenreiro S, Franssens V, Reichert AS, Outeiro TF,
Winderickx J and Ludovico P: SNCA (α-synuclein)-induced toxicity in
yeast cells is dependent on sirtuin 2 (Sir2)-mediated mitophagy.
Autophagy. 8:1494–1509. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Todd AM and Staveley BE: Pink1 suppresses
alpha-synuclein-induced phenotypes in a Drosophila model of
Parkinson’s disease. Genome. 51:1040–1046. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Todd AM and Staveley BE: Expression of
Pink1 with α-synuclein in the dopaminergic neurons of Drosophila
leads to increases in both lifespan and healthspan. Genet Mol Res.
11:1497–1502. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Hajjar T, Meng GY, Rajion MA, Vidyadaran
S, Othman F, Farjam AS, Li TA and Ebrahimi M: Omega 3
polyunsaturated fatty acid improves spatial learning and
hippocampal peroxisome proliferator activated receptors (PPARα and
PPARγ) gene expression in rats. BMC Neurosci. 13:1092012.
View Article : Google Scholar
|
|
61
|
Galland L: Diet and inflammation. Nutr
Clin Pract. 25:234–241. 2010. View Article : Google Scholar
|
|
62
|
Eckert GP, Franke C, Nöldner M, Rau O,
Wurglics M, Schubert-Zsilavecz M and Müller WE: Plant derived
omega-3-fatty acids protect mitochondrial function in the brain.
Pharmacol Res. 61:234–241. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Lee J, Park S, Lee JY, Yeo YK, Kim JS and
Lim J: Improved spatial learning and memory by perilla diet is
correlated with immunoreactivities to neurofilament and α-synuclein
in hilus of dentate gyrus. Proteome Sci. 10:722012. View Article : Google Scholar
|
|
64
|
Pabon MM, Jernberg JN, Morganti J,
Contreras J, Hudson CE, Klein RL and Bickford PC: A
spirulina-enhanced diet provides neuroprotection in an α-synuclein
model of Parkinson’s disease. PLoS One. 7:e452562012. View Article : Google Scholar
|
|
65
|
Villegas I, Sánchez-Fidalgo S and Alarcón
de la Lastra C: New mechanisms and therapeutic potential of
curcumin for colorectal cancer. Mol Nutr Food Res. 52:1040–1061.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Singh PK, Kotia V, Ghosh D, Mohite GM,
Kumar A and Maji SK: Curcumin modulates α-synuclein aggregation and
toxicity. ACS Chem Neurosci. 4:393–407. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Ahmad B and Lapidus LJ: Curcumin prevents
aggregation in α-synuclein by increasing reconfiguration rate. J
Biol Chem. 287:9193–9199. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Das S, Mitrovsky G, Vasanthi HR and Das
DK: Antiaging properties of a grape-derived antioxidant are
regulated by mitochondrial balance of fusion and fission leading to
mitophagy triggered by a signaling network of
Sirt1-Sirt3-Foxo3-PINK1-PARKIN. Oxid Med Cell Longev.
2014:3451052014. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Eid N, Ito Y, Maemura K and Otsuki Y:
Elevated autophagic sequestration of mitochondria and lipid
droplets in steatotic hepatocytes of chronic ethanol-treated rats:
an immunohistochemical and electron microscopic study. J Mol
Histol. 44:311–326. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Kones R: Parkinson’s disease:
mitochondrial molecular pathology, inflammation, statins, and
therapeutic neuroprotective nutrition. Nutr Clin Pract. 25:371–389.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Girish C and Muralidhara: Propensity of
Selaginella delicatula aqueous extract to offset rotenone-induced
oxidative dysfunctions and neurotoxicity in Drosophila
melanogaster: implications for Parkinson’s disease.
Neurotoxicology. 33:444–456. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Chen L, Thiruchelvam MJ, Madura K and
Richfield EK: Proteasome dysfunction in aged human alpha-synuclein
transgenic mice. Neurobiol Dis. 23:120–126. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Liu W, Vives-Bauza C, Acín-Peréz- R,
Yamamoto A, Tan Y, Li Y, Magrané J, Stavarache MA, Shaffer S, Chang
S, Kaplitt MG, Huang XY, Beal MF, Manfredi G and Li C: PINK1 defect
causes mitochondrial dysfunction, proteasomal deficit and
alpha-synuclein aggregation in cell culture models of Parkinson’s
disease. PLoS One. 4:e45972009. View Article : Google Scholar
|
|
74
|
Kamp F, Exner N, Lutz AK, Wender N,
Hegermann J, Brunner B, Nuscher B, Bartels T, Giese A, Beyer K,
Eimer S, Winklhofer KF and Haass C: Inhibition of mitochondrial
fusion by α-synuclein is rescued by PINK1, Parkin and DJ-1. EMBO J.
29:3571–3589. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Wilhelmus MM, Nijland PG, Drukarch B, de
Vries HE and van Horssen J: Involvement and interplay of Parkin,
PINK1, and DJ1 in neurodegenerative and neuroinflammatory
disorders. Free Radic Biol Med. 53:983–992. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Tamura T, Yoshida M, Hashizume Y and Sobue
G: Lewy body-related α-synucleinopathy in the spinal cord of cases
with incidental Lewy body disease. Neuropathology. 32:13–22. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Funabe S, Takao M, Saito Y, Hatsuta H,
Sugiyama M, Ito S, Kanemaru K, Sawabe M, Arai T, Mochizuki H,
Hattori N and Murayama S: Neuropathologic analysis of Lewy-related
α-synucleinopathy in olfactory mucosa. Neuropathology. 33:47–58.
2013. View Article : Google Scholar
|