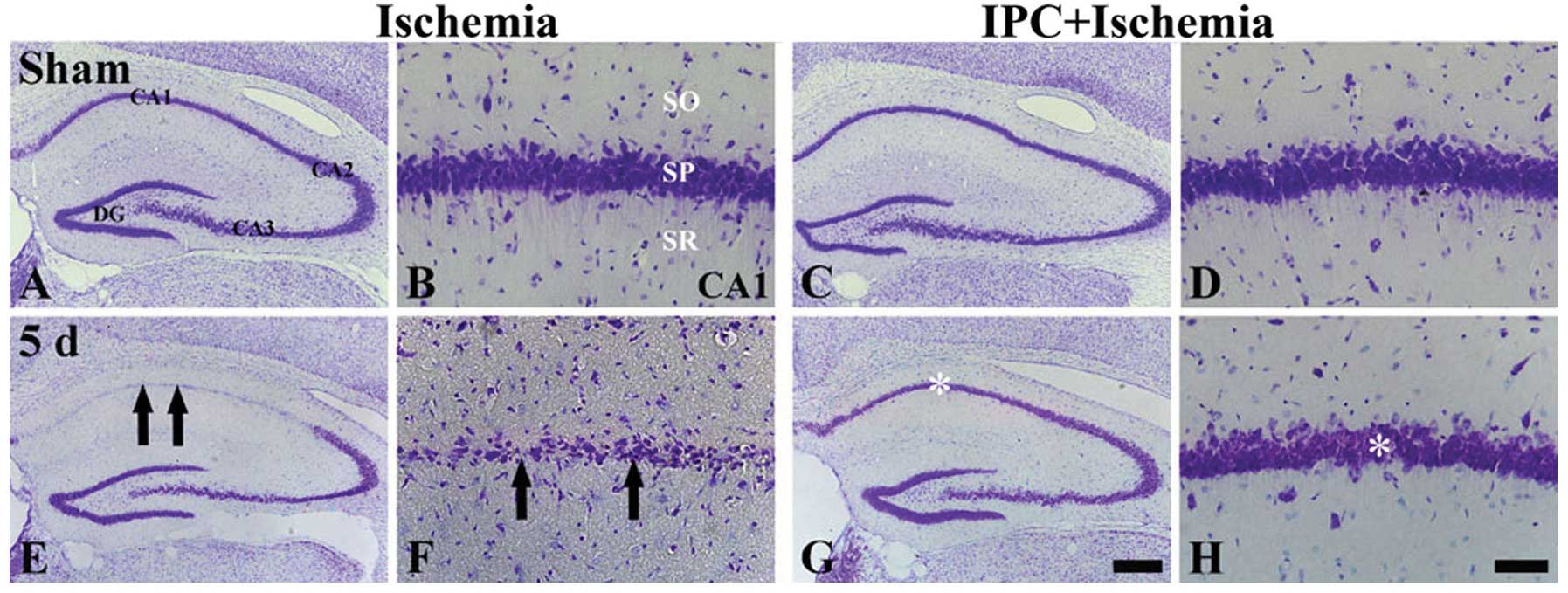
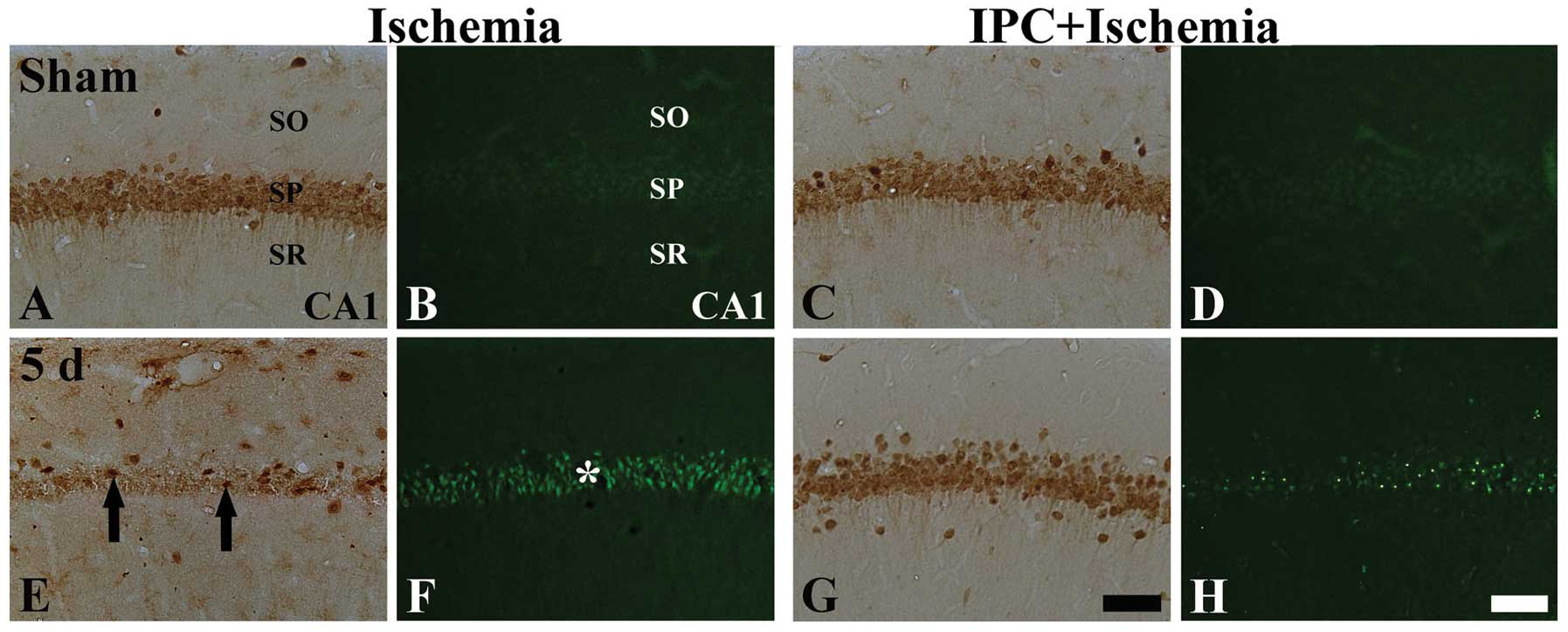
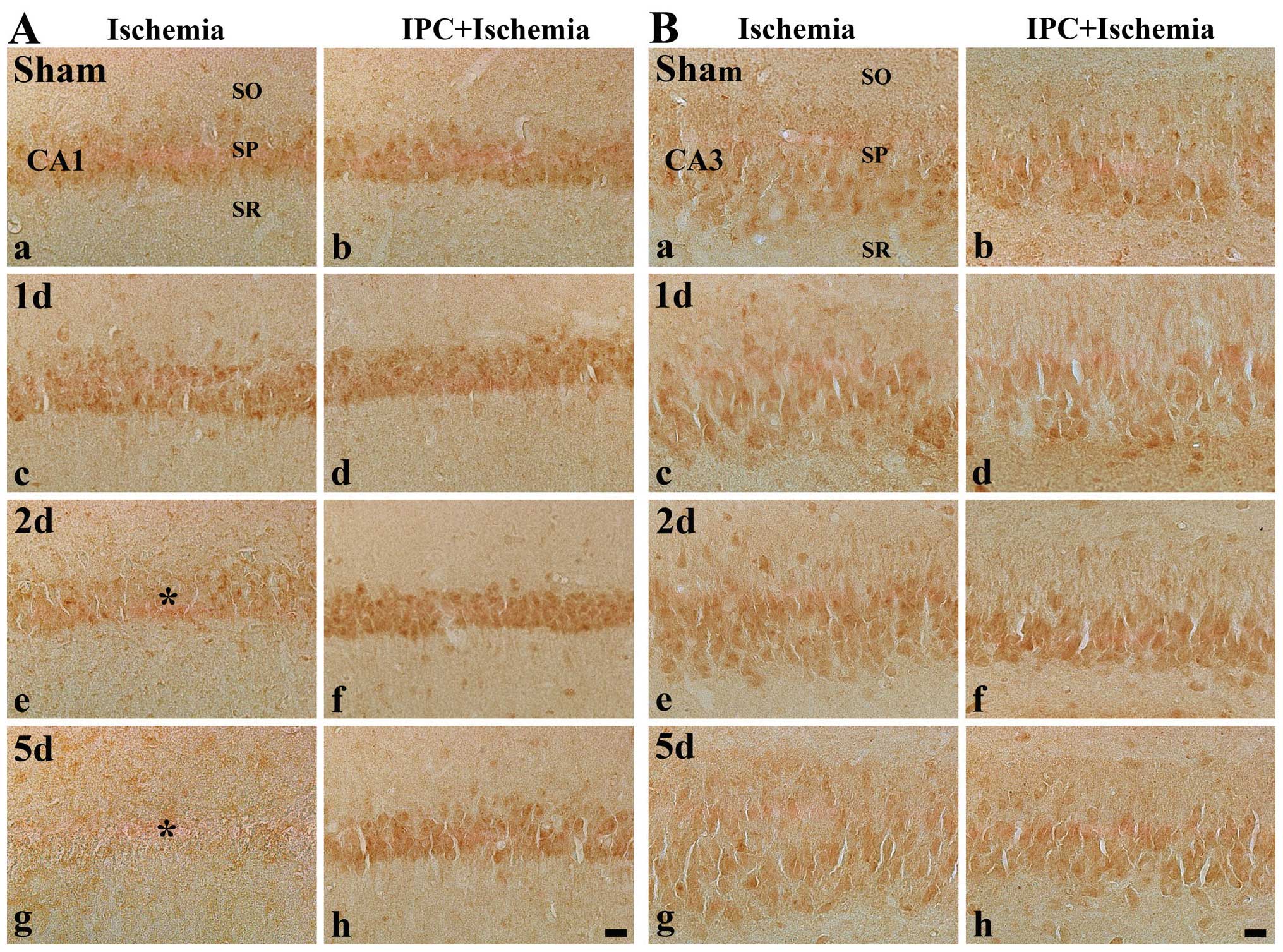
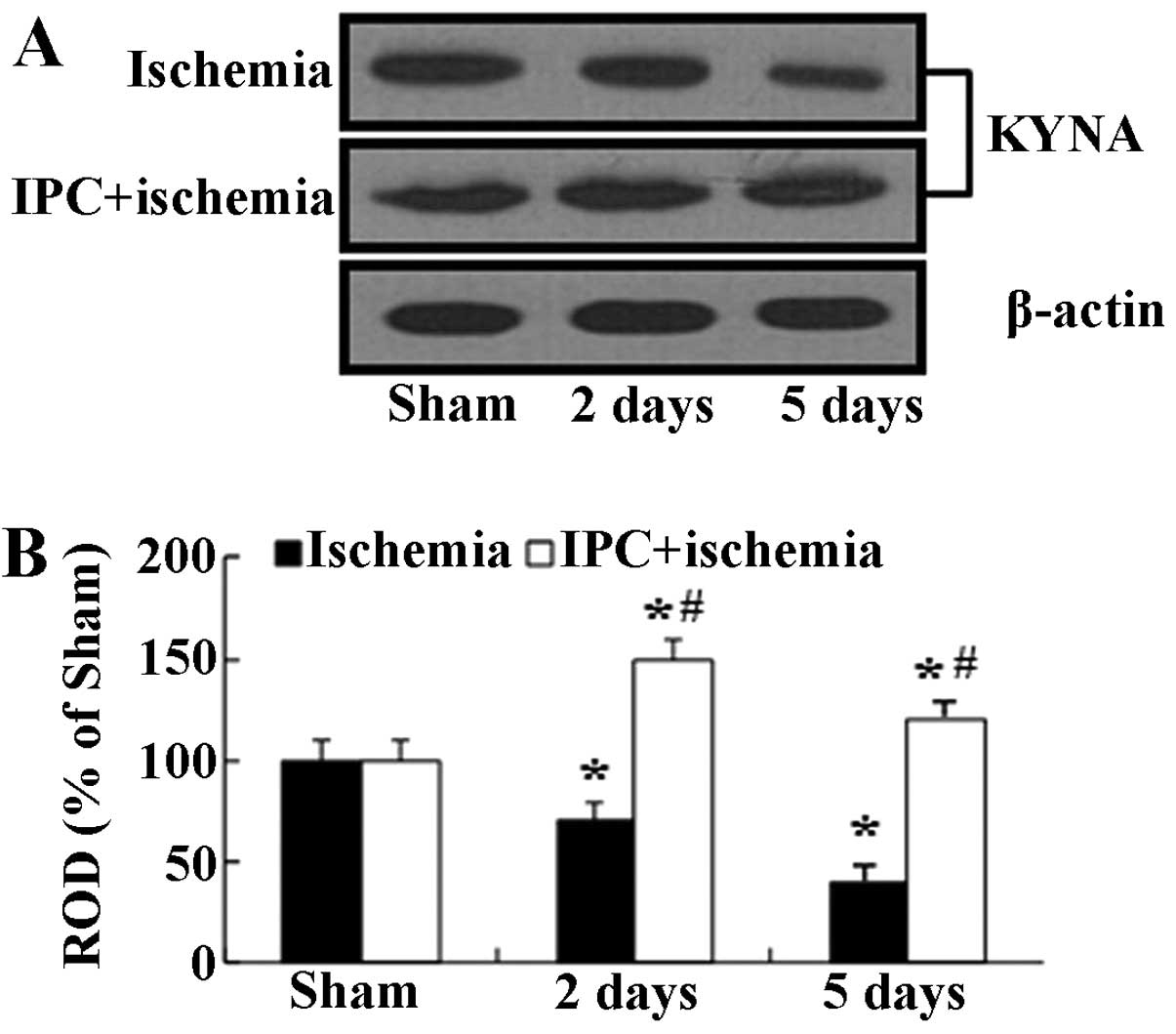
|
1
|
Kirino T: Delayed neuronal death in the
gerbil hippocampus following ischemia. Brain Res. 239:57–69. 1982.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Lin CS, Polsky K, Nadler JV and Crain BJ:
Selective neocortical and thalamic cell death in the gerbil after
transient ischemia. Neuroscience. 35:289–299. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Schmidt-Kastner R and Freund TF: Selective
vulnerability of the hippocampus in brain ischemia. Neuroscience.
40:599–636. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Abe K, Aoki M, Kawagoe J, et al: Ischemic
delayed neuronal death. A mitochondrial hypothesis. Stroke.
26:1478–1489. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Imon H, Mitani A, Andou Y, Arai T and
Kataoka K: Delayed neuronal death is induced without postischemic
hyperexcitability: continuous multiple-unit recording from ischemic
CA1 neurons. J Cereb Blood Flow Metab. 11:819–823. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Shelat PB, Coulibaly AP, Wang Q, Sun AY,
Sun GY and Simonyi A: Ischemia-induced increase in RGS7 mRNA
expression in gerbil hippocampus. Neurosci Lett. 403:157–161. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Vollenweider F, Bendfeldt K, Maetzler W,
Otten U and Nitsch C: GABA(B) receptor expression and cellular
localization in gerbil hippocampus after transient global ischemia.
Neurosci Lett. 395:118–123. 2006. View Article : Google Scholar
|
|
8
|
Hou ST and MacManus JP: Molecular
mechanisms of cerebral ischemia-induced neuronal death. Int Rev
Cytol. 221:93–148. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ientile R, Caccamo D, Marciano MC, et al:
Transglutaminase activity and transglutaminase mRNA transcripts in
gerbil brain ischemia. Neurosci Lett. 363:173–177. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yu S and Cai J: Effects of aniracetam on
extracellular levels of transmitter amino acids in the hippocampus
of the conscious gerbils: an intracranial microdialysis study.
Neurosci Lett. 339:187–190. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Schmued LC and Hopkins KJ: Fluoro-Jade B:
a high affinity fluorescent marker for the localization of neuronal
degeneration. Brain Res. 874:123–130. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Lehotsky J, Burda J, Danielisova V,
Gottlieb M, Kaplan P and Saniova B: Ischemic tolerance: the
mechanisms of neuroprotective strategy. Anat Rec (Hoboken).
292:2002–2012. 2009. View
Article : Google Scholar
|
|
13
|
Gidday JM: Cerebral preconditioning and
ischaemic tolerance. Nat Rev Neurosci. 7:437–448. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kardesoglu E, Isilak Z, Uz O and Yiginer
O: Ischemic conditioning: a current concept in reducing reperfusion
injury. Chin Med J (Engl). 124:4802011.
|
|
15
|
Swartz KJ, During MJ, Freese A and Beal
MF: Cerebral synthesis and release of kynurenic acid: an endogenous
antagonist of excitatory amino acid receptors. J Neurosci.
10:2965–2973. 1990.PubMed/NCBI
|
|
16
|
Kemp JA, Foster AC, Leeson PD, et al:
7-Chlorokynurenic acid is a selective antagonist at the glycine
modulatory site of the N-methyl-D-aspartate receptor complex. Proc
Natl Acad Sci USA. 85:6547–6550. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kessler M, Terramani T, Lynch G and Baudry
M: A glycine site associated with N-methyl-D-aspartic acid
receptors: characterization and identification of a new class of
antagonists. J Neurochem. 52:1319–1328. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Hilmas C, Pereira EF, Alkondon M,
Rassoulpour A, Schwarcz R and Albuquerque EX: The brain metabolite
kynurenic acid inhibits alpha7 nicotinic receptor activity and
increases non-alpha7 nicotinic receptor expression:
physiopathological implications. J Neurosci. 21:7463–7473.
2001.PubMed/NCBI
|
|
19
|
Moroni F, Cozzi A, Sili M and Mannaioni G:
Kynurenic acid: a metabolite with multiple actions and multiple
targets in brain and periphery. J Neural Transm. 119:133–139. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Sas K, Csete K, Vecsei L and Papp JG:
Effect of systemic administration of L-kynurenine on
corticocerebral blood flow under normal and ischemic conditions of
the brain in conscious rabbits. J Cardiovasc Pharmacol. 42:403–409.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lugo-Huitron R, Blanco-Ayala T,
Ugalde-Muniz P, et al: On the antioxidant properties of kynurenic
acid: free radical scavenging activity and inhibition of oxidative
stress. Neurotoxicol Teratol. 33:538–547. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Salvati P, Ukmar G, Dho L, et al: Brain
concentrations of kynurenic acid after a systemic neuroprotective
dose in the gerbil model of global ischemia. Prog
Neuropsychopharmacol Biol Psychiatry. 23:741–752. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Cozzi A, Carpenedo R and Moroni F:
Kynurenine hydroxylase inhibitors reduce ischemic brain damage:
studies with (m-nitrobenzoyl)-alanine (mNBA) and
3,4-dimethoxy-[-N-4-(nitrophenyl)thiazol-2yl]-benzenesulfonamide
(Ro 61-8048) in models of focal or global brain ischemia. J Cereb
Blood Flow Metab. 19:771–777. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Abo M, Yamauchi H, Suzuki M, Sakuma M and
Urashima M: Facilitated beam-walking recovery during acute phase by
kynurenic acid treatment in a rat model of photochemically induced
thrombosis causing focal cerebral ischemia. Neurosignals.
15:102–110. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Germano IM, Pitts LH, Meldrum BS,
Bartkowski HM and Simon RP: Kynurenate inhibition of cell
excitation decreases stroke size and deficits. Ann Neurol.
22:730–734. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Fukui S, Schwarcz R, Rapoport SI, Takada Y
and Smith QR: Blood-brain barrier transport of kynurenines:
implications for brain synthesis and metabolism. J Neurochem.
56:2007–2017. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Lee CH, Park JH, Cho JH, et al: Changes
and expressions of Redd1 in neurons and glial cells in the gerbil
hippocampus proper following transient global cerebral ischemia. J
Neurol Sci. 344:43–50. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Lee CH, Park JH, Choi JH, Yoo KY, Ryu PD
and Won MH: Heat shock protein 90 and its cochaperone, p23, are
markedly increased in the aged gerbil hippocampus. Exp Gerontol.
46:768–772. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Nakamura H, Katsumata T, Nishiyama Y,
Otori T, Katsura K and Katayama Y: Effect of ischemic
preconditioning on cerebral blood flow after subsequent lethal
ischemia in gerbils. Life Sci. 78:1713–1719. 2006. View Article : Google Scholar
|
|
30
|
Lee CH, Park JH, Yoo KY, et al: Pre- and
post-treatments with escitalopram protect against experimental
ischemic neuronal damage via regulation of BDNF expression and
oxidative stress. Exp Neurol. 229:450–459. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Lee JC, Kim IH, Cho GS, et al: Ischemic
preconditioning-induced neuroprotection against transient cerebral
ischemic damage via attenuating ubiquitin aggregation. J Neurol
Sci. 336:74–82. 2014. View Article : Google Scholar
|
|
32
|
Kitagawa K, Matsumoto M, Kuwabara K, et
al: ’Ischemic tolerance’ phenomenon detected in various brain
regions. Brain Res. 561:203–211. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Dhodda VK, Sailor KA, Bowen KK and
Vemuganti R: Putative endogenous mediators of
preconditioning-induced ischemic tolerance in rat brain identified
by genomic and proteomic analysis. J Neurochem. 89:73–89. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Stenzel-Poore MP, Stevens SL, King JS and
Simon RP: Preconditioning reprograms the response to ischemic
injury and primes the emergence of unique endogenous
neuroprotective phenotypes: a speculative synthesis. Stroke.
38:680–685. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Feng Z, Davis DP, Sasik R, Patel HH,
Drummond JC and Patel PM: Pathway and gene ontology based analysis
of gene expression in a rat model of cerebral ischemic tolerance.
Brain Res. 1177:103–123. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kaminski RM, Zielinska E, Dekundy A, van
Luijtelaar G and Turski W: Deficit of endogenous kynurenic acid in
the frontal cortex of rats with a genetic form of absence epilepsy.
Pol J Pharmacol. 55:741–746. 2003.
|
|
37
|
Stone TW: Kynurenines in the CNS: from
endogenous obscurity to therapeutic importance. Prog Neurobiol.
64:185–218. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Zadori D, Klivenyi P, Szalardy L, Fulop F,
Toldi J and Vecsei L: Mitochondrial disturbances, excitotoxicity,
neuroinflammation and kynurenines: novel therapeutic strategies for
neurodegenerative disorders. J Neurol Sci. 322:187–191. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Saito K, Nowak TS Jr, Markey SP and Heyes
MP: Mechanism of delayed increases in kynurenine pathway metabolism
in damaged brain regions following transient cerebral ischemia. J
Neurochem. 60:180–192. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Saito K, Nowak TS Jr, Suyama K, et al:
Kynurenine pathway enzymes in brain: responses to ischemic brain
injury versus systemic immune activation. J Neurochem.
61:2061–2070. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Stone TW: Development and therapeutic
potential of kynurenic acid and kynurenine derivatives for
neuroprotection. Trends Pharmacol Sci. 21:149–154. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Schwarcz R and Pellicciari R: Manipulation
of brain kynurenines: glial targets, neuronal effects, and clinical
opportunities. J Pharmacol Exp Ther. 303:1–10. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Vamos E, Pardutz A, Klivenyi P, Toldi J
and Vecsei L: The role of kynurenines in disorders of the central
nervous system: possibilities for neuroprotection. J Neurol Sci.
283:21–27. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Zadori D, Klivenyi P, Vamos E, Fulop F,
Toldi J and Vecsei L: Kynurenines in chronic neurodegenerative
disorders: future therapeutic strategies. J Neural Transm.
116:1403–1409. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Lipton SA and Rosenberg PA: Excitatory
amino acids as a final common pathway for neurologic disorders. N
Engl J Med. 330:613–622. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Dirnagl U, Iadecola C and Moskowitz MA:
Pathobiology of ischaemic stroke: an integrated view. Trends
Neurosci. 22:391–397. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Endres M and Dirnagl U: Ischemia and
stroke. Adv Exp Med Biol. 513:455–473. 2002. View Article : Google Scholar
|
|
48
|
Lai TW, Zhang S and Wang YT:
Excitotoxicity and stroke: identifying novel targets for
neuroprotection. Prog Neurobiol. 115:157–188. 2014. View Article : Google Scholar
|
|
49
|
Hoyte L, Barber PA, Buchan AM and Hill MD:
The rise and fall of NMDA antagonists for ischemic stroke. Curr Mol
Med. 4:131–136. 2004. View Article : Google Scholar : PubMed/NCBI
|