Introduction
Helicobacter pylori (H. pylori) is a
spiral-shaped, Gram-negative bacterium that colonizes the human
stomach. Patients infected by H. pylori usually develop
asymptomatic chronic gastritis; there are no obvious clinical
symptoms in the majority of H. pylori cases (1). H. pylori is considered a
class I carcinogen that has been linked to gastric cancer by the
World Health Organization (2). A
recent study demonstrated that seroposi-tivity to H. pylori
proteins is associated with an increased risk of biliary tract
cancers (3). The current
therapeutic regimen for eradicating H. pylori utilizes
proton-pump inhibitors in combination with several antibiotics,
such as clarithromycin, amoxicillin and metronidazole. Although
this regimen has a promising rate of eradication, up to 35% of
patients fail to respond to this treatment (4). Flurazolidone- and rifabutin-based
therapies have also been used in the treatment of H. pylori.
However, the limitations of and intolerance to these drugs make
this therapeutic option less favorable (5). Other antibiotics have also been used
in the treatment of H. pylori, including ciprofloxacin,
fluoroquinolone and streptomycin (6,7).
The rates of eradication in some studies have reached up to 80%;
however, none have achieved a 90–95% eradication rate. Failures are
associated particularly with discontinued treatment, primarily due
to poor patient compliance and increased bacterial resistance
(8).
Lactoferrin is a glycoprotein with multiple
functions, and it is widely distributed in mucosal secretions, such
as saliva, tears and seminal fluid (9). Lactoferrin has bacteriostatic,
antiviral and antifungal properties (10,11). It has been reported that the
bacteriostatic activity of lactoferrin is probably due to the
sequestering of iron, which is essential for microorganism growth
(12). Lactoferrin can also bind
the lipopolysacchride of Gram-negative bacteria and disorganize and
destabilize the bacterial surface, which in turn increases the
ability of antibiotics to permeate the bacteria (13).
Clinical studies have demonstrated that lactoferrin
increases the effectiveness of standard triple therapy for H.
pylori (14–16). However, another study indicated
that lactoferrin was ineffective in the treatment of H.
pylori infection in humans (17). Currently, lactoferrin used in the
treatment of H. pylori is mainly extracted from bovine milk,
or recombinant human lactoferrin (rhLf) is extracted from bacteria,
yeast or rice. In the present study, we examined the effectiveness
of a novel rhLf derived from goat milk in the treatment of H.
pylori in vitro and in vivo.
Materials and methods
Animal experiments
BABL/c mice (8–10 weeks of age) were obtained from
the Animal Center, Kunming Medial University (Yunnan, China). The
animals were housed at 21–22°C, with a 12-h light/dark cycle of
12:12 h. The protocol of the animal experiments was approved by the
Animal Care and Use Committee of Kunming Medical University. All
animal experiments complied with the Guide for the Care and Use of
Laboratory Animals published by the US National Institute of Health
(NIH Publication no. 8523, revised 1985).
The mice received an intragastric administration of
0.1 ml of a suspension of H. pylori ATCC 43504 (a gift from
Professor Lu Hong, Shanghai Jiao Tong University School of
Medicine, Shanghai, China) in brain-heart infusion broth (BHI
broth; 108 CFU/ml) once daily for 4 consecutive days. A
total of 192 mice was divided into 4 groups as follows: mice in
group A wre treated intragastrically with saline; mice in group B
were treated intragastrically with 1.5 g of rhLf derived from the
milk of transgenic goats (Shanghai Jielong Bioengineering Co.,
Ltd., Shanghai, China); mice in group C were treated
intragastrically with the standard triple regimen [clarithromycin
(Goldensun Pharmaceutical Co., Ltd., Xiamen, China), amoxicillin
(Baker Norton Pharmaceutical Co., Ltd., Kunming, China) and a
proton pump inhibitor (omeprazole; Harbin Pharmaceutical Co., Ltd.,
Harbin, China)]; and mice in group D were treated intragastrically
with the triple therapy regimen plus 1.5 g of rhLf daily. Following
7 days of treatment, the mice were sacrificed by cervical
dislocation, and a part of the stomach tissue of each mouse was
immediately removed and quickly frozen in liquid nitrogen. The
remaining parts of the stomach tissue were fixed in 10% formalin,
dehydrated in graded ethyl alcohol and embedded in paraffin.
Histological examination
The paraffin-embedded sections were examined for the
presence of H. pylori by silver staining. The bacteria were
characterized as a spiral rod of 3.0–0.5 mm, located adjacent to
the gastric epithelium. The sections were also stained with
hematoxylin and eosin (H&E), and were scored for their degrees
of inflammation based on the intensity of neutrophilic and/or
lymphocytic infiltration.
Bacterial culture
H. pylori ATCC 43504 was inoculated onto a
Columbia Agar base (CAB; Qingdao Hope Bio-Technology Co., Ltd.,
Qingdao, China) supplemented with 10% (v/v) fetal bovine serum
(Thermo Fisher Scientific, Waltham, MA, USA), 1% (v/v) mixed
antibiotics [trimethoprim, 5 g/l; polymyxin B sulfate, 3.85 g/l;
amphotericin B, 10 g/l; and vancomycin, 10 g/l (all obtained from
Solarbio Co., Beijing, China)] in a jar with an AnaeroPack Campylo
sachet (Mitsubishi Gas Chemical Co., Tokyo, Japan) at 37°C for 3–5
days. For liquid culture, the plate-grown bacteria were inoculated
into liquid BHI broth (Qingdao Hope Bio-Technology) supplemented
with 10% (v/v) fetal bovine serum and 1% (v/v) mixed antibiotics,
with an initial optical density at 600 nm (OD600) of 0.1.
Effect of rhLf on the growth of H.
pylori
After the H. pylori bacteria were allowed to
grow for 18–20 h in the BHI liquid medium, the bacterial cultures
were diluted in 10 ml of BHI liquid medium with rhLf (0–0.5 mg/ml)
or desferrioxamine (DFO; Chengdu Best Reagent Co., Ltd., Chengdu,
China) (0–50 µM), to an initial OD600 of 0.1. The H.
pylori broth cultures were then incubated at 37°C. Every 8 h,
over a 64-h time period, a 200-µl sample of each bacterial
culture was isolated and the OD600 measured. The effect of ferric
ammonium citrate (FAC; Chengdu Best Reagent Co., Ltd.) (0–50
µM) in the presence of 0.5 mg/ml rhLf on bacterial growth
was examined following the same procedure.
Reverse transcription-quantitatve PCR
(RT-qPCR)
Total RNA from the H. pylori cultures was
isolated using TRIzol reagent (Takara, Dalian, China). cDNA was
generated by the reverse transcription of total RNA using a
PrimeScript™ RT reagent kit with a gDNA Eraser (Takara).
Quantitative PCR (qPCR) was conducted using SYBR®Premix
Ex Taq™ II (Takara) on a LightCycler® 480 system (Roche
Applied Science, Penzberg, Germany). 16S rRNA was used as an
internal control. The primers used for PCR were as follows:
cytotoxin-associated gene A (CagA) forward, 5′-GGGCGTG
TTTGATGAGTCCT-3′ and reverse, 5′-TGT ATG TCG GTG GTG GTA GTG-3′;
vacuolating cytotoxin A (VacA) forward, 5′-CCA ACT TAC CCA
CAA ACA CC-3′ and reverse, 5′-TAG CCA ATT CAA ACA CGC TC-3′; urease
(Ure) forward, 5′-TTC TTC TGC CTG GAG TGA TAG T-3′ and
reverse, 5′-TTC TTC TGC CTG GAG TGA TAG T-3′; and 16S rRNA forward,
5′-TGT GGG AGA GGT AGG TGG AA-3′ and reverse, 5′-CAT CGT TTA GGG
CGT GGA CT-3′.
Western blot analysis
The bacteria were collected and washed with PBS.
Total proteins were extracted using a radioimmu-noprecipitation
assay (RIPA) lysis buffer (Beyotime Institute of Biotechnology,
Beijing, China). The lysates (25 µg) of total protein were
loaded and run on a 10% polyacrylamide gel and transferred onto a
polyvinylidene fluoride (PVDF) membrane.
The primary antibodies used were anti-CagA
(sc-25766, rabbit polyclonal antibody; 1:3,000 dilution; Santa Cruz
Biotechnology, Santa Cruz, CA, USA), anti-VacA (sc-25790, rabbit
polyclonal antibody; 1:3,000 dilution; Santa Cruz Biotechnology),
anti-Ure (sc-21016, rabbit polyclonal antibody; 1:3,000 dilution;
Santa Cruz Biotechnology), and anti-GAPDH antibodies (GA1R, mouse
monoclonal antibody; 1:5,000 dilution; Thermo Fisher Scientific,
Waltham, MA, USA). The secondary antibody was a horseradish
peroxidase-coupled anti-rabbit or mouse IgG (1:5,000 dilution;
Santa Cruz Biotechnology). The membrane was exposed to Kodak X-Omat
film (Kodak, Xiamen, China), and the film was then developed.
Assays for tumor necrosis factor α
(TNF-α) and interleukin-8 (IL-8)
After the stomach tissues were homogenized in liquid
nitrogen, the homogenate was lysed on ice for 60 min in lysis
buffer (BioTeke, Beijing, China). The protein concentration was
determined in the supernatant by a Bradford protein assay kit
(BioTeke) using bovine serum albumin as the standard. The levels of
TNF-α and IL-8 in the supernatant were measured using commercially
available ELISA kits (Xinbosheng Biotechnology Co., Ltd., Shenzhen,
China). The values of TNF-α and IL-8 in the stomach tissues were
normalized to the protein concentration.
Statistical analysis
The statistical significance of the differences in
gene and protein expression and in the levels of inflammatory
factors were assessed by one-way analysis of variance (ANOVA),
followed by a Student-Newman-Keuls test. The differences in
bacterial eradication and inflammation were analyzed using the
Wilcoxon rank-sum test. Data were analyzed using SPSS 11.0 software
(SPSS, Inc., Chicago, IL, USA).
Results
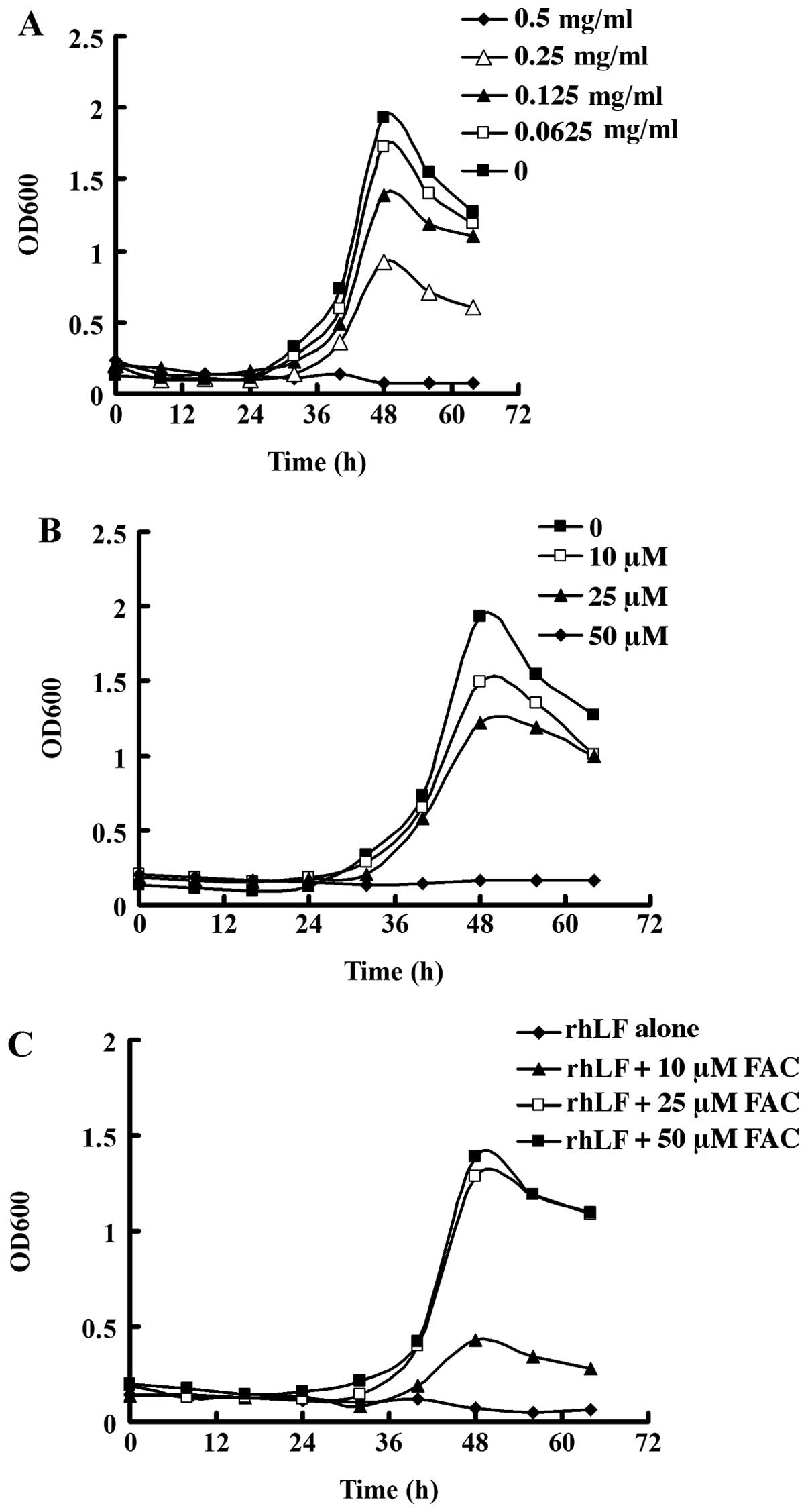
rhLf inhibits the growth of H. pylori in
vitro
It has been shown that rhLf obtained from bacteria,
yeast, or rice is an effective adjuvant for use in the treatment of
H. pylori (18,22). In order to determine the efficacy
of rhLf derived from goat milk in the treatment of H.
pylori, we first examined the effect of rhLf on the growth of
H. pylori. As shown in Fig.
1A, treatment with rhLf suppressed the growth of H.
pylori in a concentration-dependent manner. The growth of H.
pylori was almost completely inhibited by rhLf at a
concentration of 0.5 mg/ml. By contrast, H. pylori in the
control group, which was not treated with rhLf, reached a growth
peak after 48 h.
Lactoferrin has been shown to exhibit bacteriostatic
activities, probably due to its ability to chelate iron (10). We found that similar to rhLf, DFO,
a well-known chelating agent, also effectively suppressed the
growth of H. pylori in a dose-dependent manner (Fig. 1B). By contrast, the addition of
FAC significantly restored the growth of H. pylori (Fig. 1C). However, the growth of H.
pylori was not restored to the same level as the control group
even with a saturated iron supplement, suggesting that other
mechanisms, aside from the chelation of iron, also participate in
the bacteriostatic activity of rhLF.
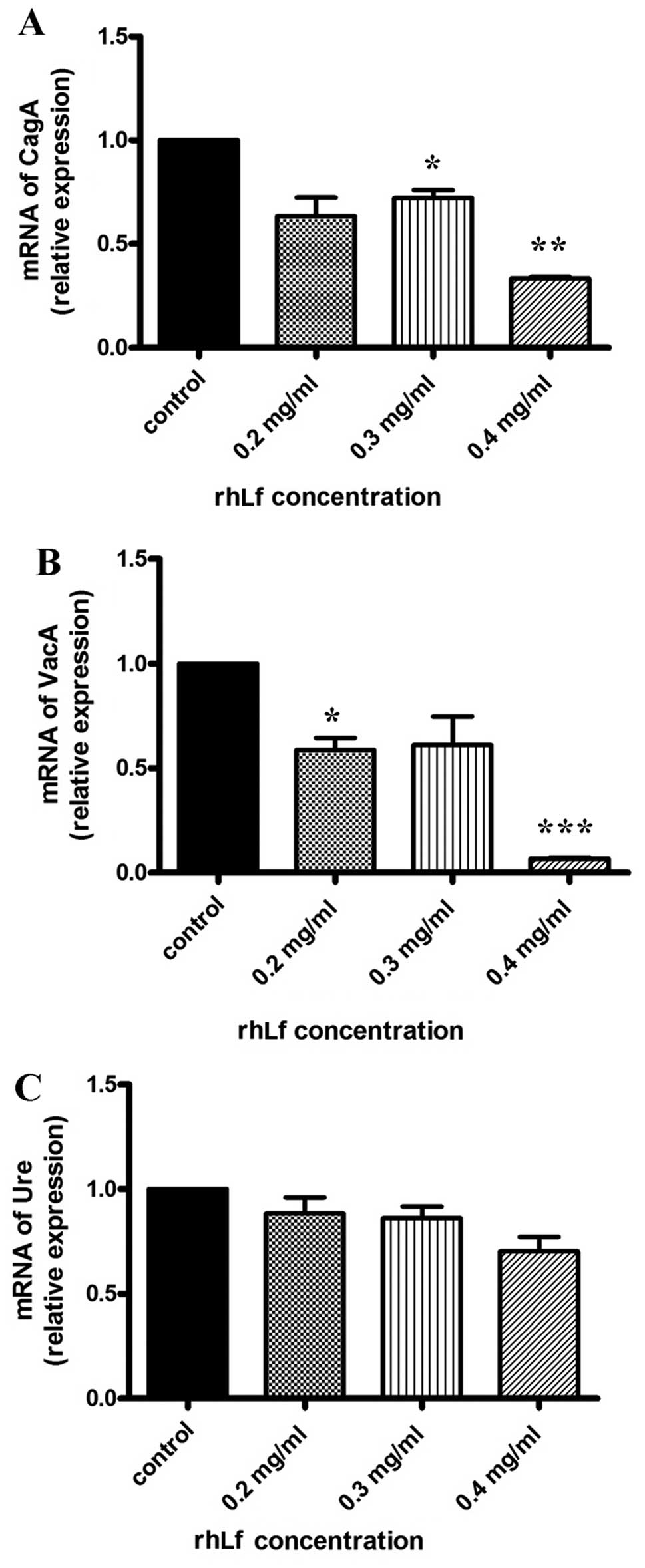
rhLf inhibits the expression of certain
virulence factors in H. pylori
We then determined whether rhLf affects the
expression of 3 major virulence factors (CagA, VacA
and Ure) by RT-qPCR. As shown in Fig. 2, a significant decrease in the
mRNA levels of 2 of these virulence factors (CagA and
VacA) following treatment with 0.4 mg/ml of rhLf was noted.
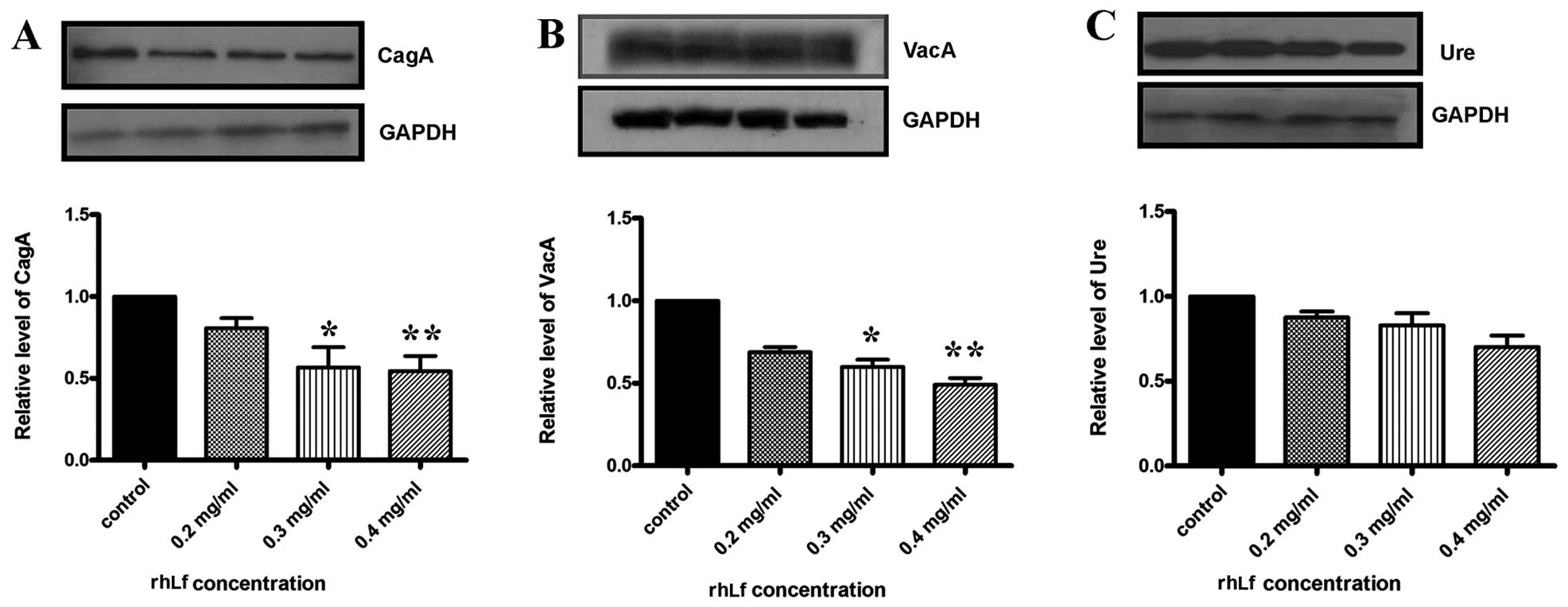
The results were further confirmed by western blot analysis
(Fig. 3). The protein levels of
CagA and VacA were suppressed following treatment with 0.4 mg/ml of
rhLf (Fig. 3A and B). By
contrast, rhLf did not affect the expression of Ure (Fig. 3C). These results suggest that
lactoferrin suppresses the expression of 2 major pathogenic
virulence factors of H. pylori.
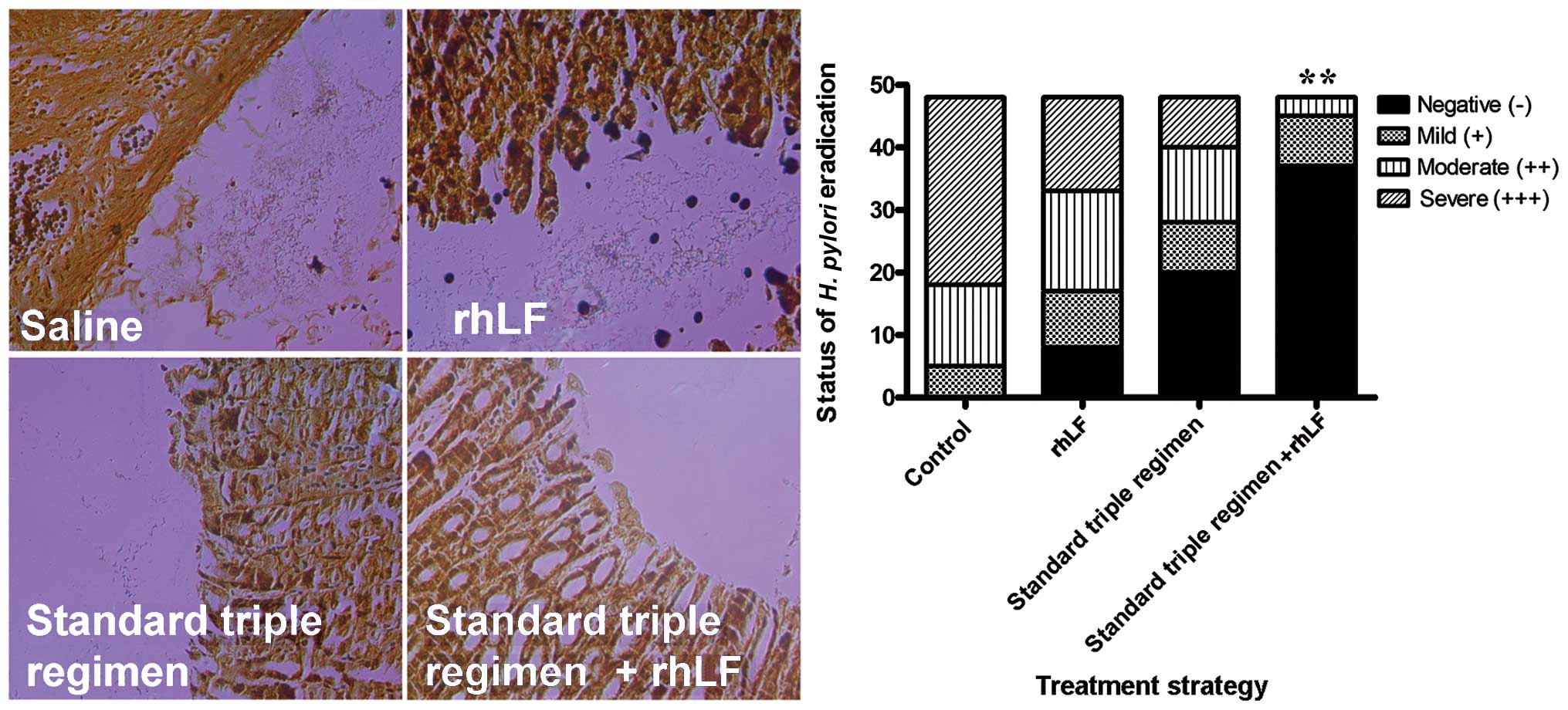
Effects of rhLf on the eradication of H.
pylori in vivo
In the present study, the infected mice were divided
into 4 groups: those treated with saline, rhLf, the standard triple
therapy regimen, or the standard triple therapy regimen plus rhLf
(quadruple treatment). To determine the efficiency of rhLf in the
eradication of H. pylori in vivo, we performed
semi-qualitative analysis of the distribution of H. pylori
in the stomach tissues; silver staining was applied to the
formalin-embedded tissue sections. As shown in Fig. 4, the number of H. pylori
bacteria was significantly higher in the rhLf group than in the
group which received the standard triple therapy regimen. However,
quadruple treatment exerted a profound protective effect against
the bacteria, as demonstrated by the complete absence of H.
pylori bacteria in the silver-stained histological
sections.
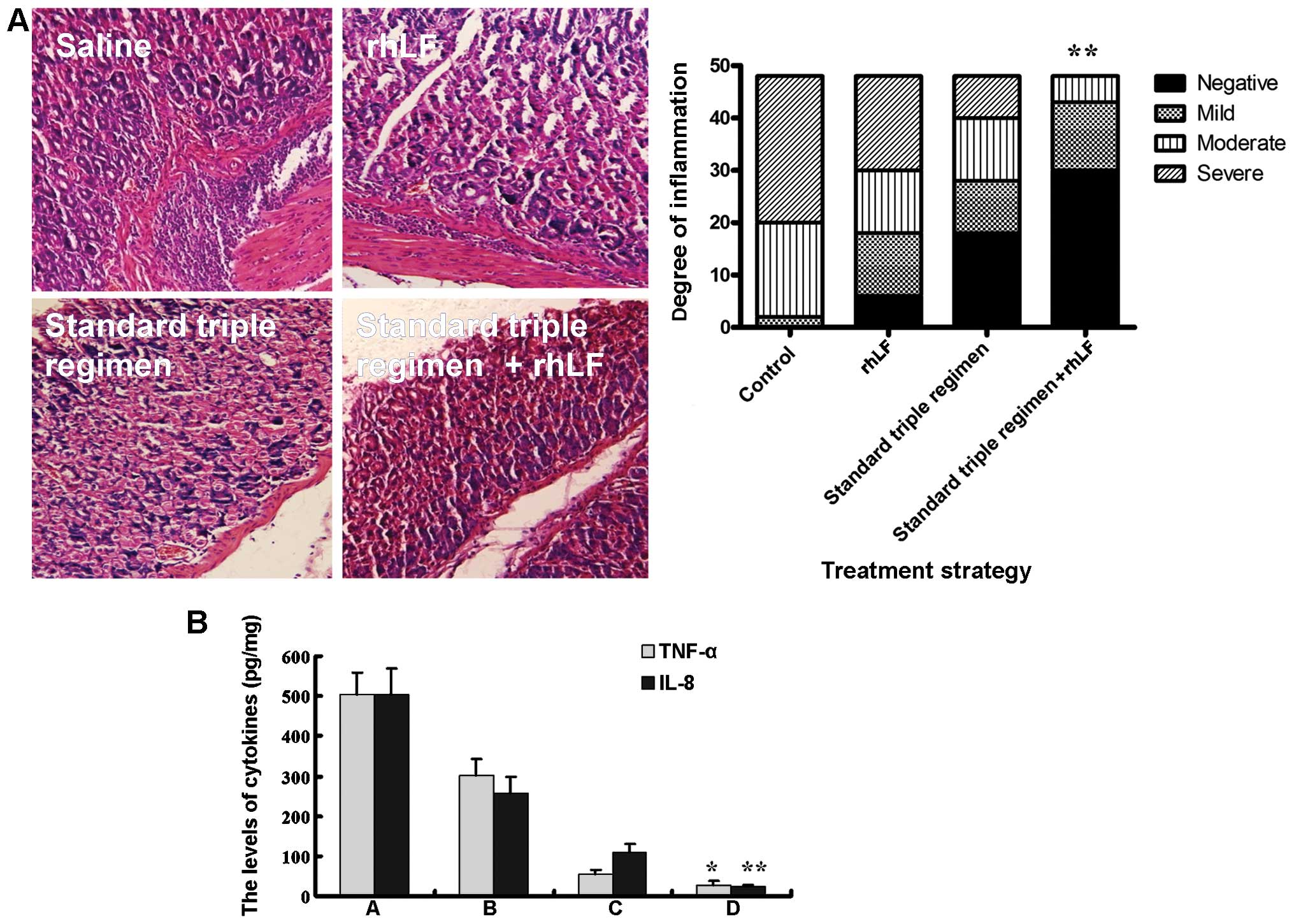
Effects of rhLf on the inflammatory
response in mice infected with H. pylori
To determine the extent of inflammation, the stomach
tissues from the mice in the 4 groups were harvested and stained
with H&E. The group treated with rhLf alone exhibited a similar
degree of gastric inflammation to the group treated with saline
(control group), and the standard triple therapy regimen
significantly reduced inflammation compared with these 2 groups.
Notably, quadruple treatment reduced inflammation to a greater
extent than the triple therapy regimen (Fig. 5A). Subsequently, we determined the
levels of the inflammatory cytokines TNF-α and IL-8 in the stomach
tissues (Fig. 5B). Unlike the
standard triple therapy regimen, which markedly inhibited the
increase in the TNF-α and IL-8 levels induced by the bacterium,
treatment with rhLf alone resulted in only a slight decrease in the
TNF-α and IL-8 levels (Fig. 5B).
However, both the TNF-α and IL-8 levels markedly decreased in the
group that received the quadruple treatment, compared to the group
treated with the the triple therapy regimen alone. These results
suggest that the addition of rhLf to the triple therapy regimen
further inhibits the inflammatory response.
Discussion
H. pylori is one of the most prevalent
bacteria; it infects the gastric mucosa, and plays an etiological
role in gastritis and peptic ulcer disease. H. pylori
infection is usually asymptomatic and persistent, and thus has a
high rate of prevalence (19).
Previous studies have demonstrated that triple therapy with
amoxicillin, clarithromycin and a proton pump inhibitor for 7 days
led to a pooled eradication rate of <80% (16,20,21). We are currently facing two great
challenges: the emerging multiple drug resistance of H.
pylori, and the very limited number of alternative antibiotics.
As a result, the development of novel treatment strategies is both
necessary and urgent.
The bacteriostatic ability of lactoferrin, as an
iron carrier, has been proven to be effective in the treatment of a
variety of pathogens, including H. pylori (10). In the present study, we examined
the effectiveness of rhLf in the treatment of H. pylori in
vitro and in vivo. rhLf inhibited the growth of H.
pylori in a concentration-dependent manner in vitro,
which is consistent with the results of a previous study (22). Our results confirm that iron
chelation seems to be one of the mechanisms underlying the
inhibition of the growth of H. pylori. Our results
demonstrated that rhLf decreased the expression of 2 major
virulence factors, CagA and VacA. CagA is encoded by
the CagA gene which is located in the cag
pathogenicity island (23).
Following the attachment of H. pylori to host gastric
epithelial cells, CagA is injected into cells through the type IV
secretion system (24). The
presence and injection of CagA triggers host immune responses,
including the secretion of IL-8 and the recruitment of various
inflammatory factors (25). The
other well-studied virulence factor of H. pylori is VacA,
which is internalized into host epithelial cells through
endocytosis (26). VacA causes
massive vacuolation in epithelial cells and disrupts normal cell
function, including endosome and lysosome activity, and
cytoskeleton-dependent functions (27). However, whether the inhibitory
effects of rhLf on the expression of CagA and VacA
are associated with its iron sequestration remains unknown.
In this study, the analysis of the stomach tissues
by silver staining revealed that treatment with rhLf alone failed
to eradicate bacteria in the H. pylori-infected mice.
However, quadruple treatment (triple therapy regimen plus rhLF)
eradicated the bacteria more effectively than the triple therapy
regimen. Indeed, it has been noted previously that treatment with
lactoferrin over a 7-day period does not have an effect on the
elimination of H. pylori (17), whereas the addition of lactoferrin
improves the efficacy of standard triple therapy against H.
pylori infection in human subjects (14–16). Likewise, rhLf alone did not
attenuate the inflammatory response in H. pylori-infected
mice. However, the inflammatory response in mice infected with
H. pylori was markedly reduced by the standard triple
therapy regimen plus rhLf, compared to the triple regimen alone.
Taken together, our results demonstrate the efficacy of rhLf as an
adjuvant in the treatment of H. pylori in mice.
Acknowledgments
The present study was supported by a grant
(2012FB027) from the Joint fund for the Yunnan Department of
Science and Technology, Kunming Medical University.
References
|
1
|
Bytzer P, Dahlerup JF, Eriksen JR, Jarbøl
DE, Rosenstock S and Wildt S; Danish Society for Gastroenterology:
Diagnosis and treatment of Helicobacter pylori infection. Dan Med
Bull. 58:C42712011.PubMed/NCBI
|
|
2
|
Olokoba AB, Obateru OA and Bojuwoye MO:
Helicobacter pylori eradication therapy: A review of current
trends. Niger Med J. 54:1–4. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Murphy G, Michel A, Taylor PR, Albanes D,
Weinstein SJ, Virtamo J, Parisi D, Snyder K, Butt J, McGlynn KA, et
al: Association of seropositivity to Helicobacter species and
biliary tract cancer in the ATBC study. Hepatology. 60:1963–1971.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Leung WK and Graham DY: Rescue therapy for
Helicobacter pylori. Curr Treat Options Gastroenterol. 5:133–138.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Gisbert JP and Pajares JM: Review article:
Helicobacter pylori ‘rescue’ regimen when proton pump
inhibitor-based triple therapies fail. Aliment Pharmacol Ther.
16:1047–1057. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Dresner D, Coyle W, Nemec R, Peterson R,
Duntemann T and Lawson JM: Efficacy of ciprofloxacin in the
eradication of Helicobacter pylori. South Med J. 89:775–778. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sanaka M, Kuyama Y, Yamanaka M and Iwasaki
M: Decrease in serum concentrations of Helicobacter pylori IgG
antibodies during antituberculosis therapy: the possible
eradication by rifampicin and streptomycin. Am J Gastroenterol.
94:1983–1984. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kusters JG, van Vliet AH and Kuipers EJ:
Pathogenesis of Helicobacter pylori infection. Clin Microbiol Rev.
19:449–490. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Vorland LH: Lactoferrin: a multifunctional
glycoprotein. APMIS. 107:971–981. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Farnaud S and Evans RW: Lactoferrin: a
multifunctional protein with antimicrobial properties. Mol Immunol.
40:395–405. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ikeda M, Nozaki A, Sugiyama K, Tanaka T,
Naganuma A, Tanaka K, Sekihara H, Shimotohno K, Saito M and Kato N:
Characterization of antiviral activity of lactoferrin against
hepatitis C virus infection in human cultured cells. Virus Res.
66:51–63. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ellison RT III, Giehl TJ and LaForce FM:
Damage of the outer membrane of enteric Gram-negative bacteria by
lactoferrin and transferrin. Infect Immun. 56:2774–2781.
1988.PubMed/NCBI
|
|
13
|
Drago-Serrano ME, de la Garza-Amaya M,
Luna JS and Campos-Rodriguez R: Lactoferrin-lipopolysaccharide
(LPS) binding as key to antibacterial and antiendotoxic effects.
Int Immunopharmacol. 12:1–9. 2012. View Article : Google Scholar
|
|
14
|
Di Mario F, Aragona G, Bò ND, Ingegnoli A,
Cavestro GM, Moussa AM, Iori V, Leandro G, Pilotto A and Franzè A:
Use of lactoferrin for Helicobacter pylori eradication. Preliminary
results. J Clin Gastroenterol. 36:396–398. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Sachdeva A and Nagpal J: Meta-analysis:
efficacy of bovine lacto-ferrin in Helicobacter pylori eradication.
Aliment Pharmacol Ther. 29:720–730. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
de Bortoli N, Leonardi G, Ciancia E, Merlo
A, Bellini M, Costa F, Mumolo MG, Ricchiuti A, Cristiani F, Santi
S, et al: Helicobacter pylori eradication: a randomized prospective
study of triple therapy versus triple therapy plus lactoferrin and
probiotics. Am J Gastroenterol. 102:951–956. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Guttner Y, Windsor HM, Viiala CH and
Marshall BJ: Human recombinant lactoferrin is ineffective in the
treatment of human Helicobacter pylori infection. Aliment Pharmacol
Ther. 17:125–129. 2003. View Article : Google Scholar
|
|
18
|
Miehlke S, Reddy R, Osato MS, Ward PP,
Conneely OM and Graham DY: Direct activity of recombinant human
lactoferrin against Helicobacter pylori. J Clin Microbiol.
34:2593–2594. 1996.PubMed/NCBI
|
|
19
|
Peek RM Jr and Blaser MJ: Helicobacter
pylori and gastrointestinal tract adenocarcinomas. Nat Rev Cancer.
2:28–37. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
20
|
Vergara M, Vallve M, Gisbert JP and Calvet
X: Meta-analysis: comparative efficacy of different proton-pump
inhibitors in triple therapy for Helicobacter pylori eradication.
Aliment Pharmacol Ther. 18:647–654. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Altintas E, Sezgin O, Ulu O, Aydin O and
Camdeviren H: Maastricht II treatment scheme and efficacy of
different proton pump inhibitors in eradicating Helicobacter
pylori. World J Gastroenterol. 10:1656–1658. 2004.PubMed/NCBI
|
|
22
|
Dial EJ, Hall LR, Serna H, Romero JJ, Fox
JG and Lichtenberger LM: Antibiotic properties of bovine
lactoferrin on Helicobacter pylori. Dig Dis Sci. 43:2750–2756.
1998. View Article : Google Scholar
|
|
23
|
Censini S, Lange C, Xiang Z, Crabtree JE,
Ghiara P, Borodovsky M, Rappuoli R and Covacci A: cag, a
pathogenicity island of Helicobacter pylori, encodes type
I-specific and disease-associated virulence factors. Proc Natl Acad
Sci USA. 93:14648–14653. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Christie PJ and Vogel JP: Bacterial type
IV secretion: conjugation systems adapted to deliver effector
molecules to host cells. Trends Microbiol. 8:354–360. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Fischer W, Puls J, Buhrdorf R, Gebert B,
Odenbreit S and Haas R: Systematic mutagenesis of the Helicobacter
pylori cag pathogenicity island: essential genes for CagA
translocation in host cells and induction of interleukin-8. Mol
Microbiol. 42:1337–1348. 2001. View Article : Google Scholar
|
|
26
|
Papini E, Satin B, Norais N, de Bernard M,
Telford JL, Rappuoli R and Montecucco C: Selective increase of the
permeability of polarized epithelial cell monolayers by
Helicobacter pylori vacuolating toxin. J Clin Invest. 102:813–820.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Cover TL and Blanke SR: Helicobacter
pylori VacA, a paradigm for toxin multifunctionality. Nat Rev
Microbiol. 3:320–332. 2005. View Article : Google Scholar : PubMed/NCBI
|