The WNT family of secreted glycoproteins consists of
WNT1 (INT1), WNT2, WNT2B (WNT13), WNT3 (INT4), WNT3A, WNT4, WNT5A,
WNT5B, WNT6, WNT7A, WNT7B, WNT8A, WNT8B, WNT9A (WNT14), WNT9B
(WNT14B), WNT10A, WNT10B, WNT11 and WNT16 (1). WNT signals are transduced through
the Frizzled family comprising seven-transmembrane receptors (FZD1,
FZD2, FZD3, FZD4, FZD5, FZD6, FZD7, FZD8, FZD9 and FZD10) and
single-transmembrane co-receptors (LRP5, LRP6, ROR1 and ROR2) to
initiate the canonical and non-canonical signaling cascades
(2,3).
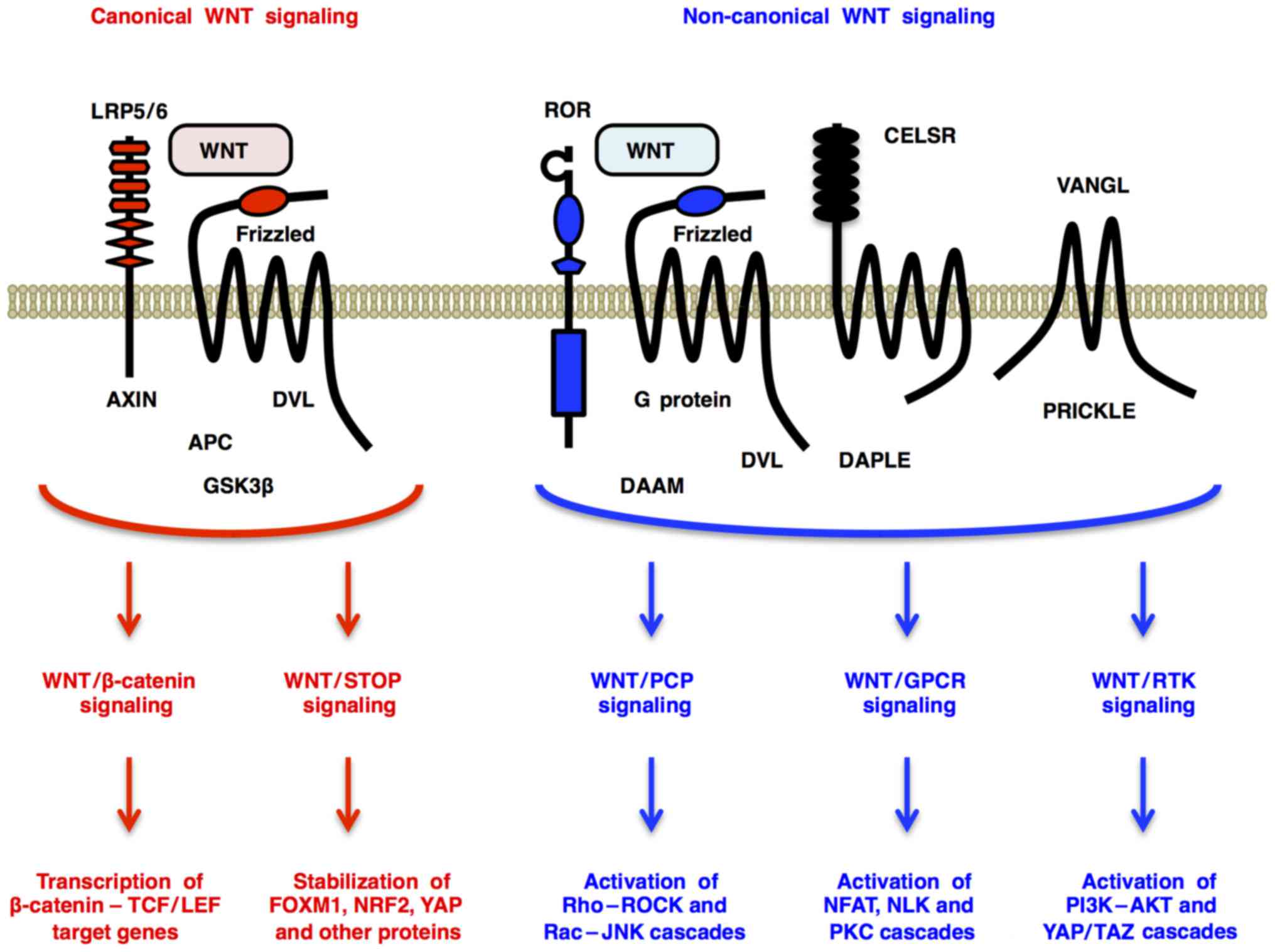
Canonical WNT signaling through Frizzled and LRP5/6
receptors promotes β-catenin-dependent transcription of TCF/LEF
target genes (WNT/β-catenin signaling) (4) and β-catenin-independent
de-repression of FOXM1, NRF2 (NFE2L2), YAP and other proteins
[WNT/stabilization of proteins (STOP) signaling] (5,6)
(Fig. 1). By contrast,
non-canonical WNT signaling through Frizzled or ROR receptors
activates Dishevelled-dependent Rho-ROCK and Rac-JNK cascades
[WNT/planar cell polarity (PCP) signaling] (7); G protein-dependent calcineurin-NFAT,
CAMK2-NLK and PKC cascades [WNT/G protein-coupled receptor (GPCR)
signaling] (2); and receptor
tyrosine kinase (RTK)-dependent PI3K-AKT (8) and YAP/TAZ (9) cascades (WNT/RTK signaling) (Fig. 1). WNT signals regulate
self-renewal, metabolism, survival, proliferation and
epithelial-to-mesenchymal transition (EMT) of target cells
(10–13), and crosstalk with FGF, Hedgehog,
Notch and transforming growth factor-β (TGF-β) signals (14–16). As the intracellular and
intercellular WNT signaling networks orchestrate embryogenesis and
homeostasis, genetic alterations in WNT signaling molecules are
involved in the pathogenesis of various types of human cancers and
noncancerous diseases (Fig.
2).
Next-generation sequencing that produces huge
amounts of genomic, epigenomic and transcriptomic data (17–20) and cell-based technologies, such as
induced pluripotent stem cells (iPSCs) (21–23), direct reprogramming to somatic
stem/progenitor cells (24) and
CRISPR/Cas9-mediated genome editing (25,26), have been elucidating the
mechanistic involvement of the WNT signaling cascades in human
pathophysiology and opening up new therapeutics avenues for human
diseases.
We carried out the Human WNTome and Post-WNTome
Projects to construct a platform of medical WNT research in the
late 1990s and early 2000s (1,2,7 and
references therein). Despite amazing progress in basic studies of
WNT signaling and genetics, there is still a huge gap that must be
addressed before WNT-targeted therapy for patients can be applied.
A mechanistic understanding of the pathogenesis of WNT-related
diseases is necessary to address the gap between basic research and
clinical application. Here, human genetics and genomics of
WNT-related diseases will be reviewed (Table I), and then, clinical application
of WNT signaling-targeted therapy using small-molecule compounds,
human/humanized monoclonal antibodies (mAb) and chimeric antigen
receptor-modified T cells (CAR-T) will be discussed.
In the adult intestine, WNT2B and WNT3 are secreted
from pericryptal cells and Paneth cells, respectively, and
transduce canonical WNT signaling through FZD7 for the maintenance
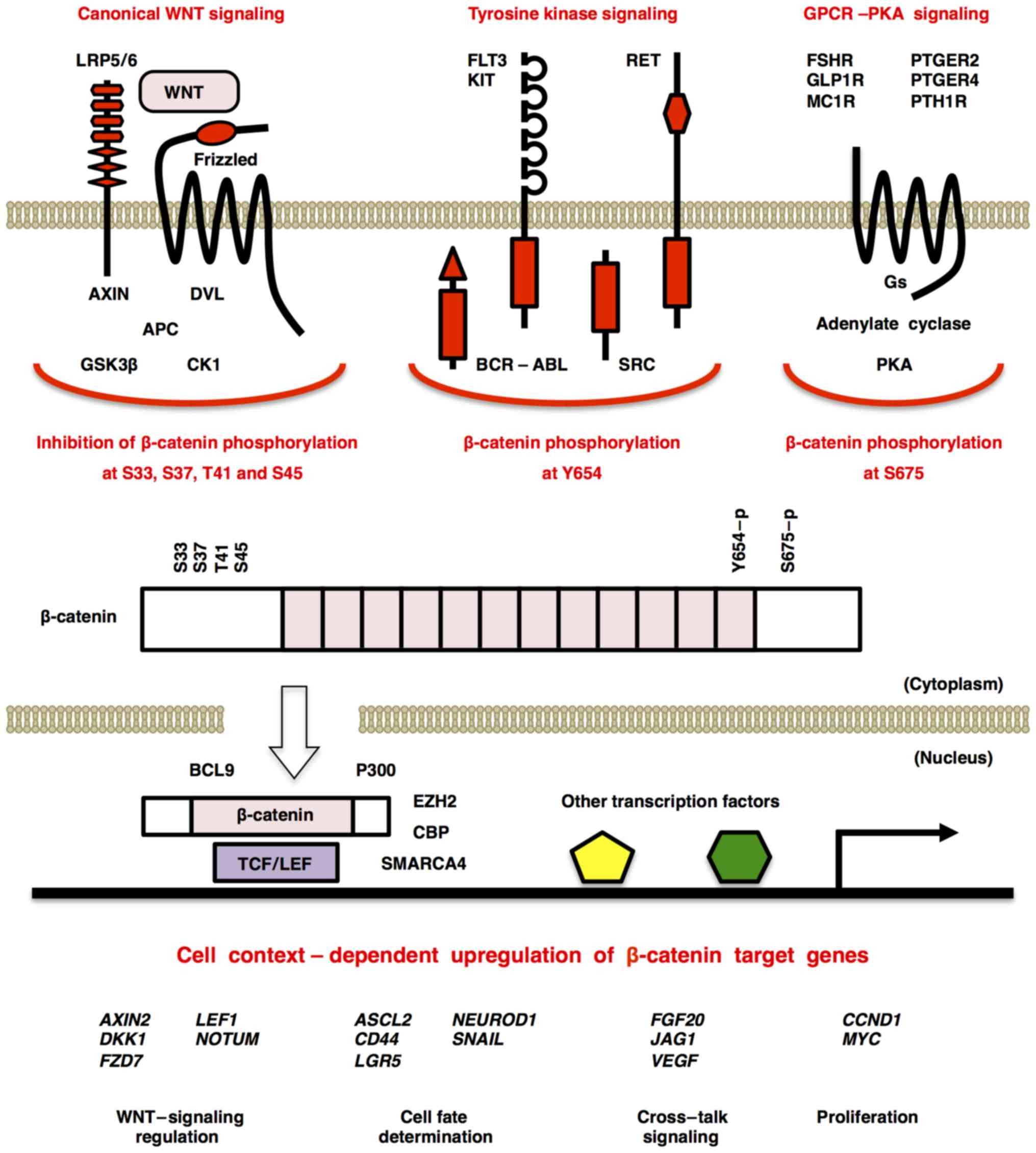
of crypt base columnar (CBC) stem cells (40,41). Binding of canonical WNTs to the
FZD and LRP5/6 receptors induces formation of the
FZD-Dishevelled-AXIN-LRP5/6 complex and release of β-catenin from
its degradation complex consisting of APC, AXIN, casein kinase 1
(CK1) and glycogen synthase kinase 3β (GSK3β), which results in
nuclear translocation of stabilized β-catenin and subsequent
transcriptional activation of TCF/LEF target genes, such as
AXIN2, cyclin D1 (CCND1), FZD7 and c-Myc
(MYC) (Fig. 3).
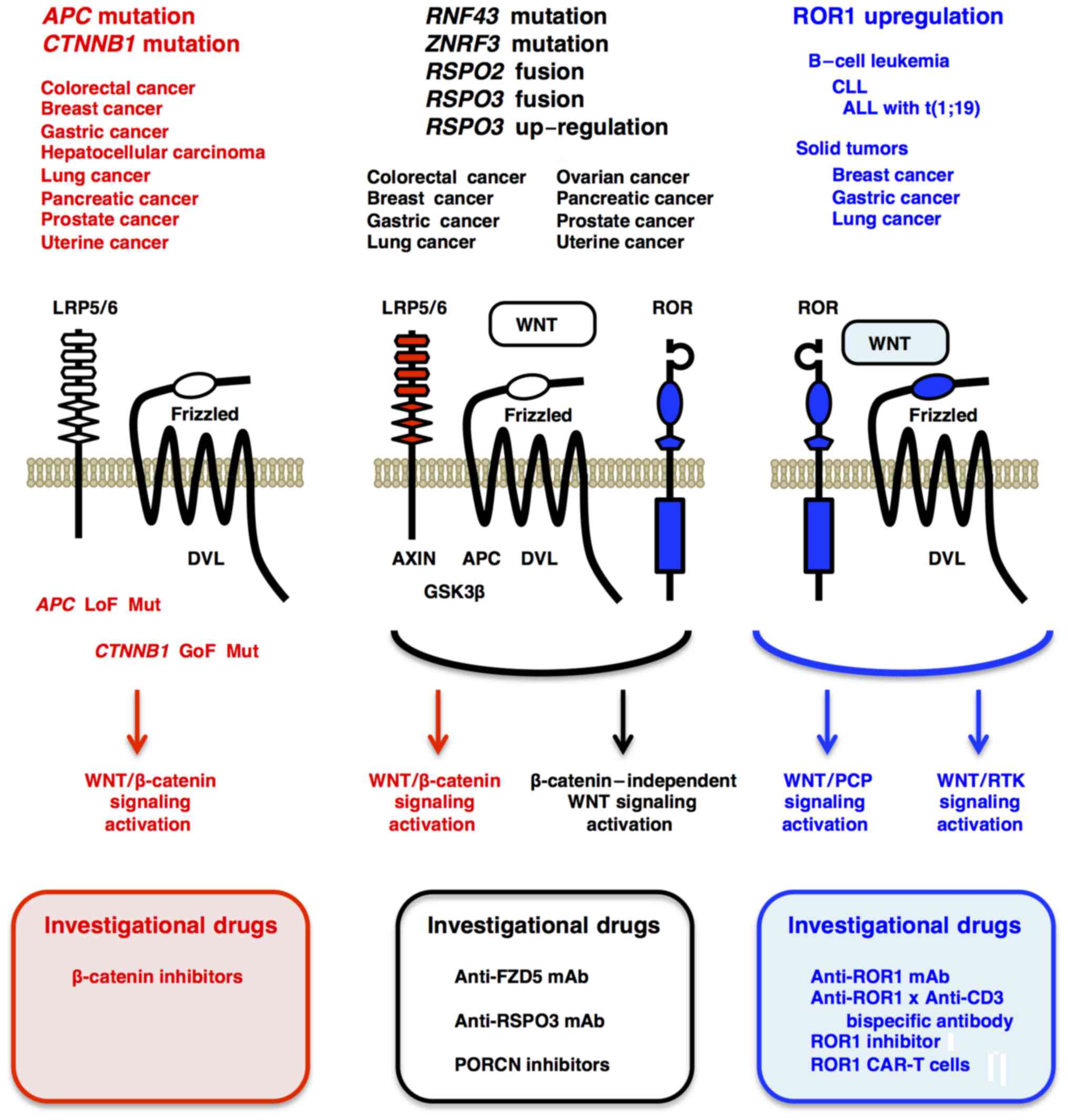
Gain-of-function mutations in the CTNNB1 gene, as well as
loss-of-function mutations in the APC and AXIN2
genes, activate the canonical WNT/β-catenin signaling cascade that
regulates self-renewal, survival, proliferation and differentiation
of tumor cells.
RNF43 and ZNRF3 are transmembrane-type E3-ubiquitin
ligases that downregulate cell-surface FZD receptors through
ubiquitylation and attenuate canonical and non-canonical WNT
signaling, whereas RSPO2 and RSPO3 are RNF43/ZNRF3 ligands that
de-repress FZD receptors from RNF43/ZNRF3-mediated degradation and
enhance WNT signaling (3,42). Loss-of-function mutations in the
RNF43 and ZNRF3 genes, as well as
EIF3E-RSPO2 and PTPRK-RSPO3 fusions,
potentiate the WNT/β-catenin signaling cascade and
β-catenin-independent WNT signaling cascades (Fig. 4).
WNT/β-catenin signals are also necessary for adult
neurogenesis or neuronal plasticity and synaptic maintenance
(53). As WNT/β-catenin signaling
induces the expression of the NeuroD1 transcription factor to
promote neurogenesis in the hippocampus and olfactory bulb,
Dickkopf-related protein 1 (Dkk1) upregulation in the hippocampus
of SAMP8 mice is associated with decreased canonical WNT signaling
and neuronal loss (54) and Wnt3
downregulation in the olfactory bulb of streptozotocin-induced
diabetic rats is associated with impaired odor discrimination,
cognitive dysfunction and increased anxiety (55). Dkk1 induction in the hippocampus
of iDkk1 transgenic mice causes synaptic loss and memory defects
through canonical WNT/β-catenin signaling inhibition and
non-canonical WNT/RhoA-ROCK signaling activation, whereas Dkk1
repression reverts the Alzheimer's disease-like phenotypes in the
iDkk1 transgenic mice (56).
WNT/β-catenin signaling also induces expression of the REST
silencing factor to protect neurons from oxidative stress and
aggregated misfolded protein in aging brains; however, neuronal
nuclear REST is lost in patients with Alzheimer's disease,
frontotemporal dementia and Lewy-body dementia (57). By contrast, impaired canonical
WNT/β-catenin signaling is involved in the pathogenesis of bipolar
disorder through defective resilience to chronic stress (58). WNT7B downregulation in
CXCR4+ neural progenitor cells derived from
bipolar-disease iPSCs is associated with a reduced proliferation
potential, and canonical WNT/β-catenin signaling activation using
GSK3 inhibitor (CHIR99021) restores the proliferation deficits
(59), which explains the
rationale why another GSK3 inhibitor, lithium, is utilized for the
treatment of patients with bipolar disorder. Together, these facts
indicate that impaired WNT/β-catenin signaling is involved in the
pathogenesis of neuropsychiatric diseases, such as Alzheimer's
disease and bipolar disorder.
Bone homeostasis is maintained by mesenchymal stem
cells that generate osteoblasts, osteoblast-derived osteocytes and
other types of mesenchymal cells, as well as hematopoietic stem
cells that give rise to monocytes, monocyte-derived osteoclasts and
other types of blood cells. Canonical WNT/β-catenin signaling
through Frizzled and LRP5/6 receptors promotes RUNX2-dependent
osteoblastic differentiation of mesenchymal stem or progenitor
cells (60,61). Canonical WNT signaling in
osteoblast-lineage cells upregulates BMP2, and then BMP2 signaling
through BMPR1A upregulates WNT7A/10B to synergistically potentiate
osteoblastogenesis and bone formation (62,63). BMP2 signaling in
osteoblast-lineage cells also upregulates the canonical WNT
inhibitors DKK1 and sclerostin (SOST) to turn off canonical WNT
signaling for the fine-tuning of bone mass (64,65). By contrast, parathyroid hormone
(PTH) signaling through PTH1R in osteoblast-lineage cells
downregulates SOST to promote bone formation and upregulates RANK
ligand (RANKL) to induce osteoclastic differentiation of osteoclast
progenitors (66). Non-canonical
WNT5A signaling through ROR2 in osteoclast progenitors upregulates
the RANK receptor to promote RANKL-induced osteoclastogenesis and
bone resorption (67). WNT
signaling cascades crosstalk with BMP, cytokine and PTH signaling
cascades in a context-dependent manner to precisely control the
balance of bone formation and resorption.
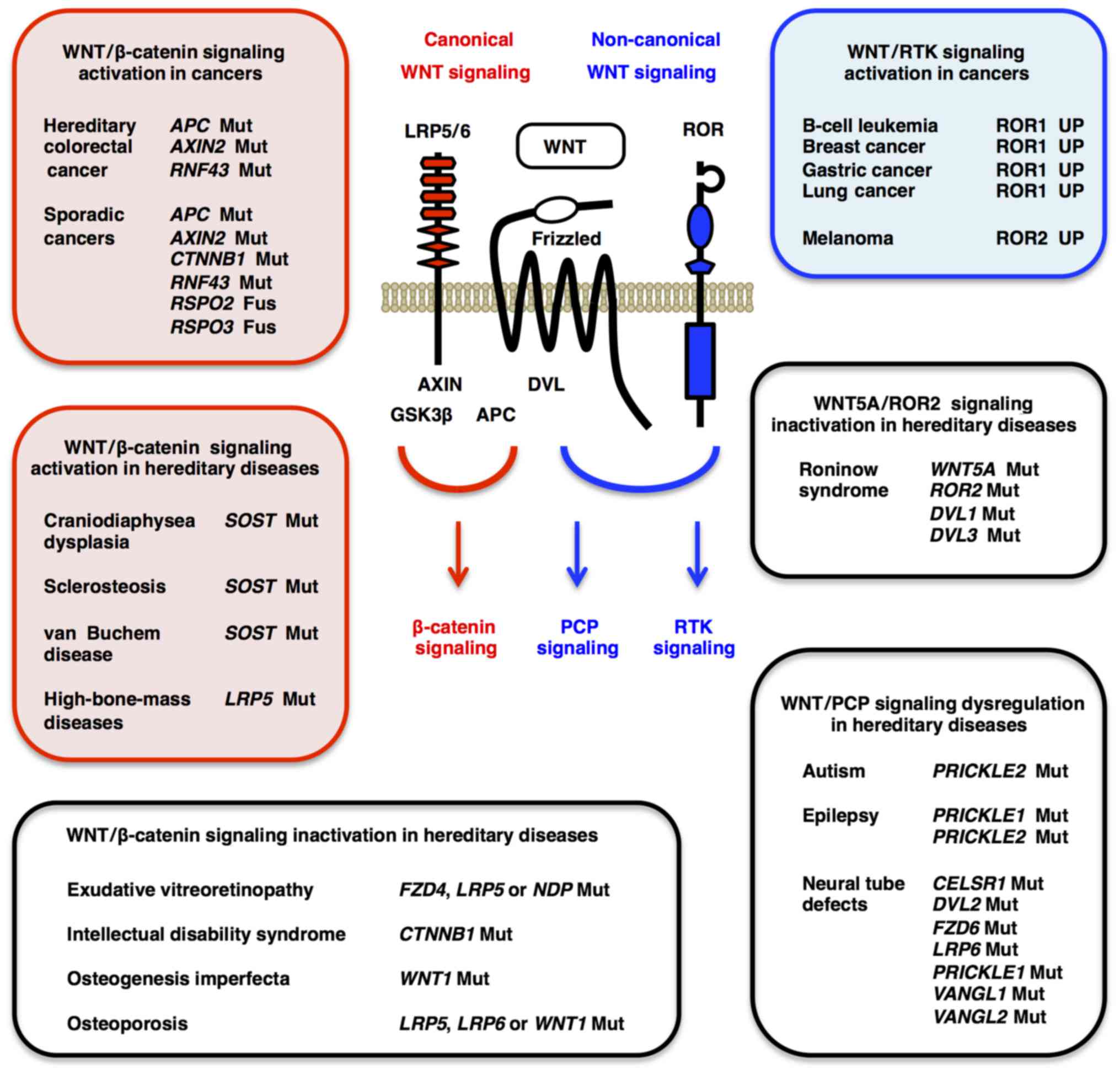
Aberrant canonical WNT signaling activation gives
rise to bone-formation phenotypes (Fig. 2). Loss-of-function mutation or
deletion in the SOST gene occurs in patients with sclerosing
skeletal dysplasias, such as craniodiaphyseal dysplasia (68), sclerosteosis (69) and van Buchem disease (70). Heterozygous mutations in the
N-terminal signal peptide of SOST (V21M/L) are detected in patients
with craniodiaphyseal dysplasia, the most severe form of
SOST-defective disease, which is characterized by massive
hyperostosis with leonine face and craniofacial foraminal stenosis.
Homozygous missense mutation (Q24X) and enhancer deletion in the
SOST gene are detected in patients with sclerosteosis and
van Buchem disease, respectively, which are characterized by
gigantism, facial palsy and hearing loss. Sclerosteosis is a severe
form of SOST-defective disease frequently presenting with
syndactyly, whereas van Buchem disease is a mild form of
SOST-defective disease without syndactyly. By contrast, LRP5
mutations in the first β-propeller domain (for example, D111Y,
G171R, A214T and A242T) have been reported in patients with
high-bone-mass diseases, such as van Buchem disease type 2,
endosteal hyperostosis and osteopetrosis type 1 (71). LRP5 mutations in the first
β-propeller domain are gain-of-function mutations, as SOST and DKK1
bind to the first β-propeller domain of LRP5 to inhibit canonical
WNT signaling (64,65). Loss-of-function SOST mutations and
gain-of-function LRP5 mutations cause bone-formation phenotypes in
patients with sclerosing skeletal dysplasias and high-bone-mass
diseases, respectively.
Defects in canonical WNT signaling and/or aberrant
activation of non-canonical WNT signaling cause bone-resorption
phenotypes (Fig. 2). Osteoporosis
is characterized by low bone mineral density (BMD), deteriorated
bone quality and susceptibility to fracture, whereas osteogenesis
imperfecta is a prenatal-onset osteoporotic disease characterized
by brittle bones (72,73). Homozygous loss-of-function
mutations in the LRP5 gene (such as R428X, E485X, D490fs and
D718X) have been detected in patients with
osteoporosis-pseudoglioma syndrome, which is characterized by
osteoporosis and eye phenotypes (exudative vitreoretinopathy and
susceptibility to blindness) (74). Heterozygous loss-of-function
mutation in the LRP6 gene (R611C) was found in patients with
familial osteoporosis and early-onset coronary artery disease
(75). Heterozygous
loss-of-function WNT1 mutation (C218G) occurs in patients with
early-onset osteoporosis, and homozygous loss-of-function WNT1
mutation (S295X) occurs in patients with osteogenesis imperfecta
(73). By contrast, homozygous
loss-of-function SFRP4 mutations (V161fs, D167fs and R232X)
give rise to Pyle disease, which is characterized by limb
malformation, cortical-bone thinning and fracture, through enhanced
non-canonical WNT5A signaling and osteoclastogenesis (76). In addition to the rare mutations
mentioned above, BMD-associated single nucleotide polymorphisms
(SNPs) in the CTNNB1, LRP5, SOST, WNT4
and WNT16 loci are also associated with slightly increased
fracture risk (77). As rare
mutations and common variations in the canonical WNT/β-catenin
signaling molecules are involved in the pathogenesis of
osteoporosis, pro-WNT/β-catenin signaling therapy is a rational
option for the treatment of patients with osteoporosis.
Vascular development and homeostasis are coordinated
by a network of VEGF, FGF, Notch, angiopoietin (ANGPT), WNT and
other signaling cascades (78,79). Endothelial cells are involved in
the maintenance of blood and lymphatic vessels as well as the
support of somatic stem cells, such as gastric stem cells,
hematopoietic stem cells, liver stem cells, mesenchymal stem cells
and neural stem cells (80,81). VEGF signaling through VEGFR2 and
FGF2 signaling through FGFR1/2 directly promote proliferation and
migration of endothelial tip cells during angiogenic sprouting
(82–84), and then, DLL and JAG signaling
through Notch directly promote stabilization and elongation of
endothelial stalk cells (85–87). ANGPT1 signaling through TIE2 in
endothelial cells promotes vascular maturation and stability,
whereas ANGPT2 signaling through TIE2 promotes vascular
de-stabilization through ANGPT1 signaling inhibition (88). Aberrant canonical WNT/β-catenin
signaling activation in cancer cells induces VEGF upregulation
(89), which leads to unstable
and leaky tumor angiogenesis. By contrast, non-canonical Wnt5a/PCP
signaling downregulates Cskn1 and Bax to promote endothelial
proliferation and survival, respectively, and upregulates Tie2 to
promote vascular maturation and stability (90). Canonical and non-canonical WNT
signaling cascades are directly or indirectly involved in vascular
pathophysiology.
Familial exudative vitreoretinopathy is a hereditary
disorder that is characterized by partial vascular agenesis,
neovascularization and exudation in the retina and susceptibility
to blindness owing to retinal detachment (91). We cloned and characterized the
human FZD4 gene in 1999 (92), and since then germline mutations
in the FZD4 gene (such as C45Y, Y58C, W226X and W496X) have
been reported in patients with exudative vitreoretinopathy
(93–95) (Fig.
2). C45Y and Y58C FZD4 are missense mutations in the
Frizzled-like domain that abolish NDP binding to FZD4, and W226X
and W496X FZD4 are loss-of-function truncation mutations.
NDP and LRP5 mutations have also been reported in
patients with exudative vitreoretinopathy (96,97). Loss-of-function LRP5
mutations occur in patients with osteoporosis-pseudoglioma syndrome
and present with similar eye phenotypes (74), and a loss-of-function
CTNNB1 mutation occurs in a patient with intellectual
disability syndrome complicated with exudative vitreoretinopathy
(51) as mentioned above. NDP is
a secreted protein that binds to the extracellular Frizzled-like
domain of FZD4 and activates the β-catenin signaling cascade
through FZD4 and LRP5 receptors similar to canonical WNT ligands.
Loss-of-function mutations in the NDP, FZD4,
LRP5 and CTNNB1 genes in patients with exudative
vitreoretinopathy indicate involvement of the NDP/β-catenin
signaling defect in the pathogenesis of exudative
vitreoretinopathy.
Ndp and Wnt7a/b are required for vascular
development in the mouse retina and central nervous system,
respectively (97,98), and lithium chloride treatment that
stabilizes β-catenin through GSK3 inhibition upregulates the Vegf
level to ameliorate retinal vascular phenotypes in an Lrp5
knockout mouse model of familial exudative vitreoretinopathy
(99). By contrast, Fzd4
signaling is required for retinal vascular stabilization and
maturation (100), and WNT5A
induces dissociation of Gα12/13 from FZD4 to promote
p115RhoGEF-mediated activation of the RHO signaling cascade in
endothelial cells (101). As
canonical WNT or NDP signaling to the β-catenin cascade can promote
angiogenic sprouting indirectly through transcriptional
upregulation of VEGF and FGF family ligands and non-canonical WNT
signaling through FZD4 can promote retinal vascular stability and
maturation, fine-tuning of the canonical and non-canonical WNT
signaling cascade may be necessary for the treatment of patients
with familial exudative vitreoretinopathy.
PCP is defined as cellular polarity within the
epithelial plane perpendicular to the cellular apico-basal axis
(7). The Drosophila PCP
pathway coordinates orientation of sensory bristles and hairs and
the rotation pattern of ommatidia (102,103), whereas the vertebrate PCP
pathway regulates orientation of sensory hair cells in the inner
ear, collective cell movements during embryogenesis (convergent
extension movements during gastrulation and neural tube closure
during neurulation) (104–107), directional movements of neural
crest cells and tumor invasion (108–111). The PCP pathway is categorized as
the Frizzled-Flamingo-dependent core PCP branch and
Fat-Dachsous-dependent alternative or parallel PCP branch (112,113).
Neural tube defects, including anencephaly,
craniorachischisis and myelomeningocele (open spina bifida), are
the second most common birth defects in humans, and they occur in
~1/1,000 established pregnancies (121). As the neural tube is generated
through orchestrated extension, upward bending and fusion of the
neural plate during embryogenesis, failure of the collective
movement of neural crest precursors results in neural tube defects
(122). Environmental factors,
such as teratogenic chemicals, and no less than 200 genetic factors
are involved in the susceptibility to neural tube defects (123). Mutations in the WNT signaling
related genes, such as CELSR1, DVL2, FZD6,
LRP6, PRICKLE1, VANGL1 and VANGL2,
occur in patients with neural tube defects (115). CELSR1, DVL2, FZD6, PRICKLE1,
VANGL1 and VANGL2 are core PCP components that are involved in
non-canonical WNT signaling cascades, whereas LRP6 is a canonical
WNT receptor (Table I). LRP6
mutants (Y306H, Y373C and V1386L) repress Wnt3a-induced
TCF/LEF-dependent transcription but potentiate Wnt5a-induced
JNK-dependent transcription (116). In addition, a patient with
intellectual disability syndrome caused by a loss-of-function
CTNNB1 mutation presented with exudative vitreoretinopathy
and neural tube defect as mentioned above (51). Mutations in the core PCP signaling
molecules, as well as loss-of-function mutations in the canonical
WNT/β-catenin signaling molecules, give rise to neural tube
defects.
Robinow syndrome is a hereditary disorder that
presents with common features, such as brachydactyly, frontal
bossing, genital hypoplasia, hemivertebra, hypertelorism and
mesomelic limb shortening (125). In addition to DVL1 and
DVL3 mutations in patients with the autosomal dominant form
of Robinow syndrome (120),
WNT5A and ROR2 mutations occur in patients with
autosomal dominant and autosomal recessive forms of Robinow
syndrome, respectively (126,127). By contrast, autosomal dominant
ROR2 mutations occur in patients with brachydactyly type B1
(128). As WNT5A signaling
through the ROR2 receptor activates DVL1/3-mediated RHO-ROCK and
RAC1-JNK signaling cascades to regulate cell polarity and
directional migration (129–132), loss-of-function mutations in the
WNT5A, ROR2, DVL1 and DVL3 genes give
rise to Robinow syndrome through impaired non-canonical WNT
signaling (Fig. 2). However,
osteosclerotic phenotypes in a subset of patients with Robinow
syndrome (133) suggest
reciprocal WNT/β-catenin signaling activation in the bone, and
Robinow syndrome-like phenotypes in mice with null and hypomorphic
Prickle1 alleles (134)
suggest the involvement of core PCP components other than DVLs in
Robinow syndrome. Signaling mechanisms and Robinow syndrome genes
should be further investigated.
WNT/PCP or WNT5A/ROR/Frizzled signaling promotes
invasion, survival and therapeutic resistance of human cancers
(135–141), although WNT5A or non-canonical
WNT/Ca2+ signaling is context-dependently involved in
tumor suppression (142–144). ROR1 is preferentially
upregulated in B-cell leukemia, such as chronic lymphocytic
leukemia (CLL) (145) and
t(1;19) acute lymphoblastic leukemia (ALL) (146). WNT5A-dependent oligomerization
of ROR1 and ROR2 on CLL cells induces recruitment of the guanine
exchange factors ARHGEF1, ARHGEF2 and ARHGEF6 and subsequent
activation of RhoA and Rac1 to promote chemotaxis and
proliferation, respectively (147). ROR1 is also upregulated in
breast cancer, gastric cancer and lung cancer, and ROR1
phosphorylation by MET and SRC promote tumor proliferation and
invasion (148–150). ROR1 interacts with TCL1A (TCL1)
to activate AKT in a mouse model of CLL (151); ROR1 interacts with HER3 and
LLGL2 in breast cancer cells to inhibit STK4 (MST1) through K59
methylation, which leads to transcriptional upregulation of
YAP/TAZ-target genes (150); and
ROR1 interacts with caveolae components in lung cancer cells to
promote survival and resistance to EGFR inhibitors through MET- or
IGF1R-dependent PI3K-AKT signaling activation (152). ROR1 upregulation in B-cell
leukemias and solid tumors promote malignant phenotypes through
ROR1 phosphorylation and activation of WNT/PCP and WNT/RTK
signaling cascades (Fig. 4). By
contrast, ROR2 is upregulated in invasive melanoma (153), and WNT5A/ROR2 signaling induces
recruitment and activation of SRC to promote metastasis (154). WNT5A induces de-palmitoylation
of MCAM adhesion molecules and subsequently polarizes localization
of MCAM and CD44 to promote directional movement and invasion of
melanoma cells (110). These
facts clearly indicate that the WNT/PCP and WNT/RTK signaling
cascades, as well as WNT/β-catenin signaling cascade, drive human
carcinogenesis (Fig. 4).
Development of therapeutics that inhibit the
WNT/β-catenin signaling cascade is a topic of great interest in the
field of clinical oncology and medicinal chemistry (172–175). By contrast, as aberrant
activation and inhibition of WNT signaling cascades are involved in
the pathogenesis of cancer and non-cancerous diseases (Table I), therapeutics that inhibit or
potentiate canonical or non-canonical WNT signaling cascades are
necessary for the future implementation of genome-based medicine
for human diseases. WNT-targeted therapy will be discussed in this
section with emphases on PORCN, RSPO3, WNT ligands, FZD receptors,
ROR1 receptor, tankyrase and β-catenin as targets for anti-WNT
signaling therapy (Table II) and
DKK1, SOST and GSK3β as targets for pro-WNT signaling therapy
(Table III).
PORCN is an endogenous WNT palmitoleoylase that
promotes secretion of WNT family proteins and their interaction
with FZD receptors (164),
whereas NOTUM is an endogenous WNT de-palmitoleoylase that
represses WNT-FZD interaction (176). Small-molecule inhibitors for
PORCN and NOTUM are applicable to anti- and pro-WNT signaling
therapies, respectively. ETC-159 (ETC-1922159) (177), IWP-2 (178), LGK974 (WNT974) (179) and WNT-C59 (180) are representative PORCN
inhibitors that attenuate WNT signaling for in vivo
treatment of colorectal cancer with RSPO translocations and
pancreatic cancer with RNF43 mutations (181–183) (Fig. 4) as well as non-cancerous
diseases, such as cardiac fibrosis (184) and kidney fibrosis (185). By contrast, OMP-131R10 is an
anti-RSPO3 mAb that neutralizes RSPO3 to attenuate canonical WNT
signaling through ubiquitylation-mediated FZD degradation (186). OMP-131R10 inhibits tumor growth
in patient-derived xenograft models of colorectal cancers with
RSPO3 fusion or non-small cell lung cancers and ovarian
cancers with RSPO3 upregulation (Fig.
4). ETC-159, LGK974 and OMP-131R10 are in clinical trials for
the treatment of cancer patients (ClinicalTrials.gov; https://clinical-trials.gov) (Table II).
ROR1 is a rational target of cancer therapeutics as
ROR1 is upregulated in subsets of B-cell leukemia, breast cancer,
gastric cancer and lung cancer but undetectable in most adult
tissues except immature B-cells (Fig.
4). In addition, ROR1 is involved in tumor proliferation,
invasion and therapeutic resistance as mentioned above (145–152). ROR1, ROR2, NTRK1, NTRK2, NTRK3,
MUSK, DDR1 and DDR2 constitute the ROR/NTRK subfamily among the
RTKs, whereas small-molecule inhibitors and mAbs are established
approaches to target RTKs (213,214). ROR1 is predicted to be a
pseudokinase that lacks intrinsic tyrosine kinase activity
(215), but ROR1 is
phosphorylated by other tyrosine kinases, such as MET and SRC, and
activates downstream signaling cascades (148,149). KAN 0439834 is a small-molecule
ROR1 inhibitor that dephosphorylates ROR1 in B-cell leukemia,
breast cancer and lung cancer and induces a cytotoxic effect on
ROR1-expressing tumor cells (216). Cirmtuzumab (UC-961) is a
humanized anti-ROR1 mAb that inhibits WNT5A-induced ROR1 signaling
through ROR1 dephosphorylation and represses in vivo growth
of ROR1-expressing CLL cells (147,217). ROR1-CD3-DART and APVO425 (ES425)
are bispecific antibodies consisting of anti-ROR1 and anti-CD3 mAbs
that redirect cytotoxic T cells to ROR1-expressing tumor cells
(218,219). ROR1 CAR-T cells were also
developed for cancer therapy, and the effectiveness and safety of
ROR1 CAR-T cells have been demonstrated in rodent as well as
non-human primate model experiments (220). Cirmtuzumab is in clinical trials
for the treatment of cancer patients (Table II).
SOST and DKK1 are endogenous canonical WNT
antagonists that induce direct inhibition of osteoblastogenesis as
well as indirect promotion of osteoclastogenesis, and are involved
in the pathogenesis of osteoporosis and cancer-associated
osteolysis, respectively (64,221,222). As SOST and DKK1 are rational
targets of pro-WNT signaling therapy for human diseases, anti-SOST
mAbs (romosozumab, blosozumab and BPS804) (223–225), anti-DKK1 mAbs (BHQ880, DKN-01
and PF-04840082) (226–228) and a bispecific antibody against
SOST and DKK1 (Hetero-DS) (229)
have been developed. Romosozumab, blosozumab and BPS804 are in
clinical trials for female postmenopausal patients with decreased
BMD, whereas BHQ880 and DKN-01 are in clinical trials for patients
with multiple myeloma and other solid tumors, such as
cholangiocarcinoma, esophageal cancer and gastric cancer (Table III).
Tankyrases (TNKS1/PARP5A and TNKS2/PARP5B), PARP1,
PARP2, TIPARP (PARP7) and other PARPs are ADP-ribosyl transferases
belonging to the PARP family (230–232), and ADP-ribosyl transferase
inhibitors, such as olaparib, have been developed for cancer
therapy (233–235). Tankyrases promote degradation of
AXIN1 and AXIN2 through poly-ADP-ribosylation, and tankyrase
inhibitors induce AXIN stabilization for canonical WNT/β-catenin
signaling inhibition (236–238). AZ1366 (239), G007-LK (240), NVP-TNKS656 (241,242) and XAV939 (243,244) are investigational tankyrase
inhibitors that can block canonical WNT/β-catenin signaling in
model animal experiments to repress tumorigenesis (239–244), control neuropathic pain
(245) and promote cardiac
reprogramming from cardiac fibroblasts (24). As tankyrase inhibitors induce a
variety of effects, such as canonical WNT/β-catenin signaling
inhibition, YAP signaling inhibition, PI3K signaling inhibition and
telomere shortening through defective poly-ADP-ribosylation of
AXIN, AMOT, PTEN and TERF1, respectively (236–238), the tankyrase inhibitors
mentioned above are not in clinical trials at present (Table II).
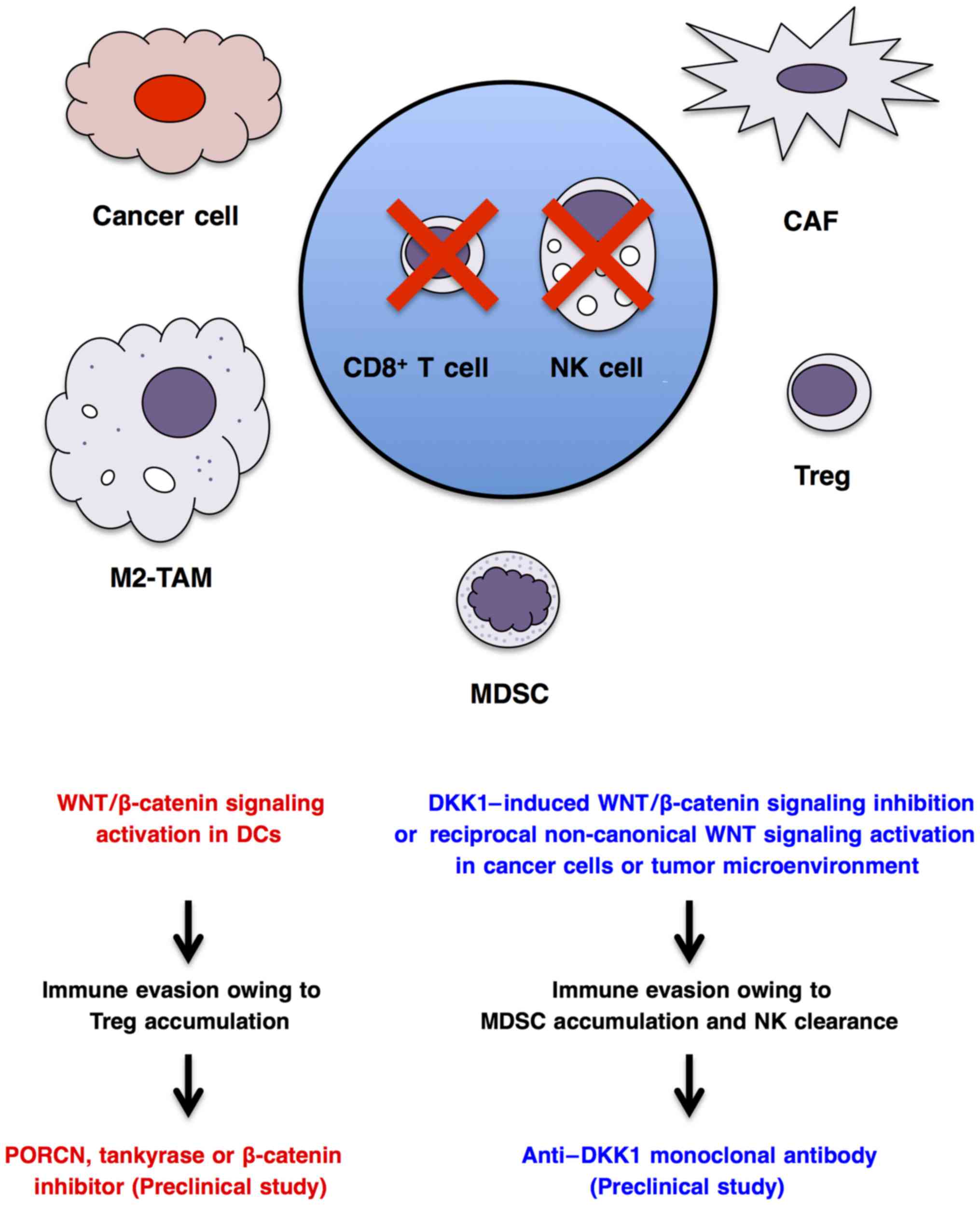
Cancer cells interact with immune cells and stromal
cells to regulate antitumor immunity, angiogenesis and metabolism
in the tumor microenvironment (78,213,263,264). WNT/β-catenin signaling
activation in cancer cells indirectly regulates immunity through
transcriptional regulation of CCL4 chemo-kine or ULBP ligands for
dendritic cells and natural killer cells, respectively (265,266), whereas canonical or
non-canonical WNT signaling activation in dendritic cells (267), macrophages (268), myeloid-derived suppressor cells
(269) and T lymphocytes
(270) directly regulates their
functions and antitumor immunity. WNT/β-catenin signaling
activation in dendritic cells can enhance immune evasion through
accumulation of regulatory T cells (271–273), and anti-WNT signaling therapy
using a PORCN inhibitor, tankyrase inhibitor or β-catenin inhibitor
may be applicable for the treatment of immune evasion (Fig. 5). By contrast, WNT/β-catenin
signaling inhibition in cancer cells or tumor microenvironment
owing to DKK1 upregulation can also lead to immune evasion through
the accumulation of myeloid-derived suppressor cells and clearance
of natural killer and cytotoxic T cells (266,269,274), and pro-WNT signaling therapy
using an anti-DKK1 mAb may be applicable for the treatment of
immune evasion in cancer patients with DKK1 upregulation (Fig. 5). As WNT signaling cascades in
cancer cells, stromal cells and immune cells regulate immune
tolerance and antitumor immunity in a cell context-dependent
manner, comprehensive understanding of WNT-dependent dynamic immune
regulation based on precise immune monitoring is necessary before
prescription of anti- or pro-WNT signaling therapeutics for cancer
patients with immune evasion.
WNT signaling molecules are dysregulated in human
diseases, such as cancer, bone diseases, cardiovascular diseases,
neuropsychiatric diseases and other PCP-related diseases.
Therapeutics targeting PORCN, RSPO3, FZD receptors, ROR1, β-catenin
and DKK1 are in clinical trials for cancer patients, and
SOST-targeting therapeutics are in clinical trials for osteoporotic
patients. Fine-tuning of WNT-targeting therapeutics is necessary
for the optimization of their clinical efficacy and safety, as WNT
signals regulate a variety of pathophysiological conditions in a
context-dependent manner. WNT-targeting therapeutics have also been
applied as in vitro stem-cell processing reagents for
regenerative medicine.
This study was financially supported in part by a
Grant-in-Aid for the knowledge base project from M. Katoh's
Fund.
|
1
|
Katoh M: WNT and FGF gene clusters
(Review). Int J Oncol. 21:1269–1273. 2002.PubMed/NCBI
|
|
2
|
Katoh M and Katoh M: WNT signaling pathway
and stem cell signaling network. Clin Cancer Res. 13:4042–4045.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Niehrs C: The complex world of WNT
receptor signalling. Nat Rev Mol Cell Biol. 13:767–779. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Yang K, Wang X, Zhang H, Wang Z, Nan G, Li
Y, Zhang F, Mohammed MK, Haydon RC, Luu HH, et al: The evolving
roles of canonical WNT signaling in stem cells and tumorigenesis:
Implications in targeted cancer therapies. Lab Invest. 96:116–136.
2016. View Article : Google Scholar :
|
|
5
|
Acebron SP and Niehrs C:
β-catenin-independent roles of Wnt/LRP6 signaling. Trends Cell
Biol. 26:956–967. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Rada P, Rojo AI, Offergeld A, Feng GJ,
Velasco-Martín JP, González-Sancho JM, Valverde ÁM, Dale T,
Regadera J and Cuadrado A: WNT-3A regulates an Axin1/NRF2 complex
that regulates antioxidant metabolism in hepatocytes. Antioxid
Redox Signal. 22:555–571. 2015. View Article : Google Scholar :
|
|
7
|
Katoh M: WNT/PCP signaling pathway and
human cancer (Review). Oncol Rep. 14:1583–1588. 2005.PubMed/NCBI
|
|
8
|
Zhang S, Chen L, Cui B, Chuang HY, Yu J,
Wang-Rodriguez J, Tang L, Chen G, Basak GW and Kipps TJ: ROR1 is
expressed in human breast cancer and associated with enhanced
tumor-cell growth. PLoS One. 7:e311272012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhuo W and Kang Y: Lnc-ing ROR1-HER3 and
Hippo signalling in metastasis. Nat Cell Biol. 19:81–83. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Medema JP: Cancer stem cells: The
challenges ahead. Nat Cell Biol. 15:338–344. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Holland JD, Klaus A, Garratt AN and
Birchmeier W: Wnt signaling in stem and cancer stem cells. Curr
Opin Cell Biol. 25:254–264. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Lamb R, Bonuccelli G, Ozsvári B,
Peiris-Pagès M, Fiorillo M, Smith DL, Bevilacqua G, Mazzanti CM,
McDonnell LA, Naccarato AG, et al: Mitochondrial mass, a new
metabolic biomarker for stem-like cancer cells: Understanding
WNT/FGF-driven anabolic signaling. Oncotarget. 6:30453–30471. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tam WL and Weinberg RA: The epigenetics of
epithelial-mesenchymal plasticity in cancer. Nat Med. 19:1438–1449.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ranganathan P, Weaver KL and Capobianco
AJ: Notch signalling in solid tumours: A little bit of everything
but not all the time. Nat Rev Cancer. 11:338–351. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Gonzalez DM and Medici D: Signaling
mechanisms of the epithelial-mesenchymal transition. Sci Signal.
7:re82014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Katoh M and Nakagama H: FGF receptors:
Cancer biology and therapeutics. Med Res Rev. 34:280–300. 2014.
View Article : Google Scholar
|
|
17
|
Yu M, Ting DT, Stott SL, Wittner BS,
Ozsolak F, Paul S, Ciciliano JC, Smas ME, Winokur D, Gilman AJ, et
al: RNA sequencing of pancreatic circulating tumour cells
implicates WNT signalling in metastasis. Nature. 487:510–513. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Bozdag S, Li A, Riddick G, Kotliarov Y,
Baysan M, Iwamoto FM, Cam MC, Kotliarova S and Fine HA:
Age-specific signatures of glioblastoma at the genomic, genetic,
and epigenetic levels. PLoS One. 8:e629822013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Miyamoto DT, Zheng Y, Wittner BS, Lee RJ,
Zhu H, Broderick KT, Desai R, Fox DB, Brannigan BW, Trautwein J, et
al: RNA-Seq of single prostate CTCs implicates noncanonical Wnt
signaling in antiandrogen resistance. Science. 349:1351–1356. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Gu Z, Churchman M, Roberts K, Li Y, Liu Y,
Harvey RC, McCastlain K, Reshmi SC, Payne-Turner D, Iacobucci I, et
al: Genomic analyses identify recurrent MEF2D fusions in acute
lymphoblastic leukaemia. Nat Commun. 7:133312016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Pettinato G, Ramanathan R, Fisher RA,
Mangino MJ, Zhang N and Wen X: Scalable differentiation of human
iPSCs in a multicellular spheroid-based 3D culture into
hepatocyte-like cells through direct Wnt/β-catenin pathway
inhibition. Sci Rep. 6:328882016. View Article : Google Scholar
|
|
22
|
Motono M, Ioroi Y, Ogura T and Takahashi
J: WNT-C59, a small-molecule WNT inhibitor, efficiently induces
anterior cortex that includes cortical motor neurons from human
pluripotent stem cells. Stem Cells Transl Med. 5:552–560. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Matsuno K, Mae SI, Okada C, Nakamura M,
Watanabe A, Toyoda T, Uchida E and Osafune K: Redefining definitive
endoderm subtypes by robust induction of human induced pluripotent
stem cells. Differentiation. 92:281–290. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Mohamed TM, Stone NR, Berry EC, Radzinsky
E, Huang Y, Pratt K, Ang YS, Yu P, Wang H, Tang S, et al: Chemical
enhancement of in vitro and in vivo direct cardiac reprogramming.
Circulation. 135:978–995. 2017. View Article : Google Scholar
|
|
25
|
Tao L, Zhang J, Meraner P, Tovaglieri A,
Wu X, Gerhard R, Zhang X, Stallcup WB, Miao J, He X, et al:
Frizzled proteins are colonic epithelial receptors for C. difficile
toxin B. Nature. 538:350–355. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Collery RF, Volberding PJ, Bostrom JR,
Link BA and Besharse JC: Loss of zebrafish mfrp causes
nanophthalmia, hyperopia, and accumulation of subretinal
macrophages. Invest Ophthalmol Vis Sci. 57:6805–6814. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kinzler KW and Vogelstein B: Lessons from
hereditary colorectal cancer. Cell. 87:159–170. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Lammi L, Arte S, Somer M, Jarvinen H,
Lahermo P, Thesleff I, Pirinen S and Nieminen P: Mutations in AXIN2
cause familial tooth agenesis and predispose to colorectal cancer.
Am J Hum Genet. 74:1043–1050. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
29
|
Gala MK, Mizukami Y, Le LP, Moriichi K,
Austin T, Yamamoto M, Lauwers GY, Bardeesy N and Chung DC: Germline
mutations in oncogene-induced senescence pathways are associated
with multiple sessile serrated adenomas. Gastroenterology.
146:520–529. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Muzny DM, Bainbridge MN, Chang K, Dinh HH,
Drummond JA, Fowler G, Kovar CL, Lewis LR, Morgan MB, Newsham IF,
et al Cancer Genome Atlas Network: Comprehensive molecular
characterization of human colon and rectal cancer. Nature.
487:330–337. 2012. View Article : Google Scholar
|
|
31
|
Giannakis M, Mu XJ, Shukla SA, Qian ZR,
Cohen O, Nishihara R, Bahl S, Cao Y, Amin-Mansour A, Yamauchi M, et
al: Genomic correlates of immune-cell infiltrates in colorectal
carcinoma. Cell Rep. 15:857–865. 2016. View Article : Google Scholar :
|
|
32
|
Seshagiri S, Stawiski EW, Durinck S,
Modrusan Z, Storm EE, Conboy CB, Chaudhuri S, Guan Y, Janakiraman
V, Jaiswal BS, et al: Recurrent R-spondin fusions in colon cancer.
Nature. 488:660–664. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Ciriello G, Gatza ML, Beck AH, Wilkerson
MD, Rhie SK, Pastore A, Zhang H, McLellan M, Yau C, Kandoth C, et
al TCGA Research Network: Comprehensive molecular portraits of
invasive lobular breast cancer. Cell. 163:506–519. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Bass AJ, Thorsson V, Shmulevich I,
Reynolds SM, Miller M, Bernard B, Hinoue T, Laird PW, Curtis C,
Shen H, et al Cancer Genome Atlas Research Network: Comprehensive
molecular characterization of gastric adenocarcinoma. Nature.
513:202–209. 2014. View Article : Google Scholar :
|
|
35
|
Guichard C, Amaddeo G, Imbeaud S, Ladeiro
Y, Pelletier L, Maad IB, Calderaro J, Bioulac-Sage P, Letexier M,
Degos F, et al: Integrated analysis of somatic mutations and focal
copy-number changes identifies key genes and pathways in
hepatocellular carcinoma. Nat Genet. 44:694–698. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Campbell JD, Alexandrov A, Kim J, Wala J,
Berger AH, Pedamallu CS, Shukla SA, Guo G, Brooks AN, Murray BA, et
al Cancer Genome Atlas Research Network: Distinct patterns of
somatic genome alterations in lung adenocarcinomas and squamous
cell carcinomas. Nat Genet. 48:607–616. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Bailey P, Chang DK, Nones K, Johns AL,
Patch AM, Gingras MC, Miller DK, Christ AN, Bruxner TJ, Quinn MC,
et al Australian Pancreatic Cancer Genome Initiative: Genomic
analyses identify molecular subtypes of pancreatic cancer. Nature.
531:47–52. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Robinson D, Van Allen EM, Wu YM, Schultz
N, Lonigro RJ, Mosquera JM, Montgomery B, Taplin ME, Pritchard CC,
Attard G, et al: Integrative clinical genomics of advanced prostate
cancer. Cell. 161:1215–1228. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Kandoth C, Schultz N, Cherniack AD, Akbani
R, Liu Y, Shen H, Robertson AG, Pashtan I, Shen R, Benz CC, et al
Cancer Genome Atlas Research Network: Integrated genomic
characterization of endometrial carcinoma. Nature. 497:67–73. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Barker N: Adult intestinal stem cells:
Critical drivers of epithelial homeostasis and regeneration. Nat
Rev Mol Cell Biol. 15:19–33. 2014. View Article : Google Scholar
|
|
41
|
Flanagan DJ, Phesse TJ, Barker N, Schwab
RH, Amin N, Malaterre J, Stange DE, Nowell CJ, Currie SA, Saw JT,
et al: Frizzled7 functions as a Wnt receptor in intestinal
epithelial Lgr5(+) stem cells. Stem Cell Reports. 4:759–767. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Jiang X and Cong F: Novel regulation of
Wnt signaling at the proximal membrane level. Trends Biochem Sci.
41:773–783. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Valenta T, Hausmann G and Basler K: The
many faces and functions of β-catenin. EMBO J. 31:2714–2736. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Katoh M: Mutation spectra of histone
methyltransferases with canonical SET domains and EZH2-targeted
therapy. Epigenomics. 8:285–305. 2016. View Article : Google Scholar
|
|
45
|
Wang Z, Liu P, Inuzuka H and Wei W: Roles
of F-box proteins in cancer. Nat Rev Cancer. 14:233–247. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Kajiguchi T, Katsumi A, Tanizaki R, Kiyoi
H and Naoe T: Y654 of β-catenin is essential for FLT3/ITD-related
tyrosine phosphorylation and nuclear localization of β-catenin. Eur
J Haematol. 88:314–320. 2012. View Article : Google Scholar
|
|
47
|
Jin B, Ding K and Pan J: Ponatinib induces
apoptosis in imatinib-resistant human mast cells by
dephosphorylating mutant D816V KIT and silencing β-catenin
signaling. Mol Cancer Ther. 13:1217–1230. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Fernández-Sánchez ME, Barbier S, Whitehead
J, Béalle G, Michel A, Latorre-Ossa H, Rey C, Fouassier L, Claperon
A, Brullé L, et al: Mechanical induction of the tumorigenic
β-catenin pathway by tumour growth pressure. Nature. 523:92–95.
2015. View Article : Google Scholar
|
|
49
|
van Veelen W, Le NH, Helvensteijn W,
Blonden L, Theeuwes M, Bakker ER, Franken PF, van Gurp L, Meijlink
F, van der Valk MA, et al: β-catenin tyrosine 654 phosphorylation
increases Wnt signalling and intestinal tumorigenesis. Gut.
60:1204–1212. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Kuechler A, Willemsen MH, Albrecht B,
Bacino CA, Bartholomew DW, van Bokhoven H, van den Boogaard MJ,
Bramswig N, Büttner C, Cremer K, et al: De novo mutations in
β-catenin (CTNNB1) appear to be a frequent cause of intellectual
disability: Expanding the mutational and clinical spectrum. Hum
Genet. 134:97–109. 2015. View Article : Google Scholar
|
|
51
|
Dixon MW, Stem MS, Schuette JL, Keegan CE
and Besirli CG: CTNNB1 mutation associated with familial exudative
vitreoretinopathy (FEVR) phenotype. Ophthalmic Genet. 37:468–470.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Lui JH, Hansen DV and Kriegstein AR:
Development and evolution of the human neocortex. Cell. 146:18–36.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Inestrosa NC and Arenas E: Emerging roles
of Wnts in the adult nervous system. Nat Rev Neurosci. 11:77–86.
2010. View Article : Google Scholar
|
|
54
|
Bayod S, Felice P, Andrés P, Rosa P,
Camins A, Pallàs M and Canudas AM: Downregulation of canonical Wnt
signaling in hippocampus of SAMP8 mice. Neurobiol Aging.
36:720–729. 2015. View Article : Google Scholar
|
|
55
|
Wakabayashi T, Hidaka R, Fujimaki S,
Asashima M and Kuwabara T: Diabetes impairs Wnt3 protein-induced
neurogenesis in olfactory bulbs via glutamate transporter 1
inhibition. J Biol Chem. 291:15196–15211. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Marzo A, Galli S, Lopes D, McLeod F,
Podpolny M, Segovia-Roldan M, Ciani L, Purro S, Cacucci F, Gibb A,
et al: Reversal of synapse degeneration by restoring Wnt signaling
in the adult hippocampus. Curr Biol. 26:2551–2561. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Lu T, Aron L, Zullo J, Pan Y, Kim H, Chen
Y, Yang TH, Kim HM, Drake D, Liu XS, et al: REST and stress
resistance in ageing and Alzheimer's disease. Nature. 507:448–454.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Dias C, Feng J, Sun H, Shao NY,
Mazei-Robison MS, Damez-Werno D, Scobie K, Bagot R, LaBonté B,
Ribeiro E, et al: β-catenin mediates stress resilience through
Dicer1/microRNA regulation. Nature. 516:51–55. 2014.PubMed/NCBI
|
|
59
|
Madison JM, Zhou F, Nigam A, Hussain A,
Barker DD, Nehme R, van der Ven K, Hsu J, Wolf P, Fleishman M, et
al: Characterization of bipolar disorder patient-specific induced
pluripotent stem cells from a family reveals neurodevelopmental and
mRNA expression abnormalities. Mol Psychiatry. 20:703–717. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Karantalis V and Hare JM: Use of
mesenchymal stem cells for therapy of cardiac disease. Circ Res.
116:1413–1430. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Atashi F, Modarressi A and Pepper MS: The
role of reactive oxygen species in mesenchymal stem cell adipogenic
and osteogenic differentiation: A review. Stem Cells Dev.
24:1150–1163. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Chen Y and Alman BA: Wnt pathway, an
essential role in bone regeneration. J Cell Biochem. 106:353–362.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Zhang R, Oyajobi BO, Harris SE, Chen D,
Tsao C, Deng HW and Zhao M: Wnt/β-catenin signaling activates bone
morphogenetic protein 2 expression in osteoblasts. Bone.
52:145–156. 2013. View Article : Google Scholar
|
|
64
|
Ke HZ, Richards WG, Li X and Ominsky MS:
Sclerostin and Dickkopf-1 as therapeutic targets in bone diseases.
Endocr Rev. 33:747–783. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Boudin E, Fijalkowski I, Piters E and Van
Hul W: The role of extracellular modulators of canonical Wnt
signaling in bone metabolism and diseases. Semin Arthritis Rheum.
43:220–240. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Silva BC and Bilezikian JP: Parathyroid
hormone: Anabolic and catabolic actions on the skeleton. Curr Opin
Pharmacol. 22:41–50. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Maeda K, Kobayashi Y, Udagawa N, Uehara S,
Ishihara A, Mizoguchi T, Kikuchi Y, Takada I, Kato S, Kani S, et
al: Wnt5a-Ror2 signaling between osteoblast-lineage cells and
osteoclast precursors enhances osteoclastogenesis. Nat Med.
18:405–412. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Kim SJ, Bieganski T, Sohn YB, Kozlowski K,
Semënov M, Okamoto N, Kim CH, Ko AR, Ahn GH, Choi YL, et al:
Identification of signal peptide domain SOST mutations in autosomal
dominant craniodiaphyseal dysplasia. Hum Genet. 129:497–502. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Brunkow ME, Gardner JC, Van Ness J, Paeper
BW, Kovacevich BR, Proll S, Skonier JE, Zhao L, Sabo PJ, Fu Y, et
al: Bone dysplasia sclerosteosis results from loss of the SOST gene
product, a novel cystine knot-containing protein. Am J Hum Genet.
68:577–589. 2001. View
Article : Google Scholar : PubMed/NCBI
|
|
70
|
Balemans W, Patel N, Ebeling M, Van Hul E,
Wuyts W, Lacza C, Dioszegi M, Dikkers FG, Hildering P, Willems PJ,
et al: Identification of a 52 kb deletion downstream of the SOST
gene in patients with van Buchem disease. J Med Genet. 39:91–97.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Van Wesenbeeck L, Cleiren E, Gram J, Beals
RK, Bénichou O, Scopelliti D, Key L, Renton T, Bartels C, Gong Y,
et al: Six novel missense mutations in the LDL receptor-related
protein 5 (LRP5) gene in different conditions with an increased
bone density. Am J Hum Genet. 72:763–771. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Canalis E: Wnt signalling in osteoporosis:
Mechanisms and novel therapeutic approaches. Nat Rev Endocrinol.
9:575–583. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Laine CM, Joeng KS, Campeau PM, Kiviranta
R, Tarkkonen K, Grover M, Lu JT, Pekkinen M, Wessman M, Heino TJ,
et al: WNT1 mutations in early-onset osteoporosis and osteogenesis
imperfecta. N Engl J Med. 368:1809–1816. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Gong Y, Slee RB, Fukai N, Rawadi G,
Roman-Roman S, Reginato AM, Wang H, Cundy T, Glorieux FH, Lev D, et
al Osteoporosis-Pseudoglioma Syndrome Collaborative Group: LDL
receptor-related protein 5 (LRP5) affects bone accrual and eye
development. Cell. 107:513–523. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Mani A, Radhakrishnan J, Wang H, Mani A,
Mani MA, Nelson-Williams C, Carew KS, Mane S, Najmabadi H, Wu D, et
al: LRP6 mutation in a family with early coronary disease and
metabolic risk factors. Science. 315:1278–1282. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Simsek Kiper PO, Saito H, Gori F, Unger S,
Hesse E, Yamana K, Kiviranta R, Solban N, Liu J, Brommage R, et al:
Cortical-bone fragility: Insights from sFRP4 deficiency in Pyle's
disease. N Engl J Med. 374:2553–2562. 2016. View Article : Google Scholar
|
|
77
|
Estrada K, Styrkarsdottir U, Evangelou E,
Hsu YH, Duncan EL, Ntzani EE, Oei L, Albagha OM, Amin N, Kemp JP,
et al: Genome-wide meta-analysis identifies 56 bone mineral density
loci and reveals 14 loci associated with risk of fracture. Nat
Genet. 44:491–501. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Carmeliet P and Jain RK: Molecular
mechanisms and clinical applications of angiogenesis. Nature.
473:298–307. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Katoh M: Therapeutics targeting
angiogenesis: Genetics and epigenetics, extracellular miRNAs and
signaling networks (Review). Int J Mol Med. 32:763–767.
2013.PubMed/NCBI
|
|
80
|
Hayakawa Y, Ariyama H, Stancikova J,
Sakitani K, Asfaha S, Renz BW, Dubeykovskaya ZA, Shibata W, Wang H,
Westphalen CB, et al: Mist1 expressing gastric stem cells maintain
the normal and neoplastic gastric epithelium and are supported by a
perivascular stem cell niche. Cancer Cell. 28:800–814. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Rafii S, Butler JM and Ding BS: Angiocrine
functions of organ-specific endothelial cells. Nature. 529:316–325.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Goel HL and Mercurio AM: VEGF targets the
tumour cell. Nat Rev Cancer. 13:871–882. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Ferrara N and Adamis AP: Ten years of
anti-vascular endothelial growth factor therapy. Nat Rev Drug
Discov. 15:385–403. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Katoh M: Therapeutics targeting FGF
signaling network in human diseases. Trends Pharmacol Sci.
37:1081–1096. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Zhou W, Wang G and Guo S: Regulation of
angiogenesis via Notch signaling in breast cancer and cancer stem
cells. Biochim Biophys Acta. 1836:304–320. 2013.PubMed/NCBI
|
|
86
|
Zhang P, Yan X, Chen Y, Yang Z and Han H:
Notch signaling in blood vessels: From morphogenesis to
homeostasis. Sci China Life Sci. 57:774–780. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Rostama B, Peterson SM, Vary CP and Liaw
L: Notch signal integration in the vasculature during remodeling.
Vascul Pharmacol. 63:97–104. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Hilbert T and Klaschik S: The
angiopoietin/TIE receptor system: Focusing its role for
ischemia-reperfusion injury. Cytokine Growth Factor Rev.
26:281–291. 2015. View Article : Google Scholar
|
|
89
|
Zhang X, Gaspard JP and Chung DC:
Regulation of vascular endothelial growth factor by the Wnt and
K-ras pathways in colonic neoplasia. Cancer Res. 61:6050–6054.
2001.PubMed/NCBI
|
|
90
|
Korn C, Scholz B, Hu J, Srivastava K,
Wojtarowicz J, Arnsperger T, Adams RH, Boutros M, Augustin HG and
Augustin I: Endothelial cell-derived non-canonical Wnt ligands
control vascular pruning in angiogenesis. Development.
141:1757–1766. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Gilmour DF: Familial exudative
vitreoretinopathy and related retinopathies. Eye (Lond). 29:1–14.
2015. View Article : Google Scholar
|
|
92
|
Kirikoshi H, Sagara N, Koike J, Tanaka K,
Sekihara H, Hirai M and Katoh M: Molecular cloning and
characterization of human Frizzled-4 on chromosome 11q14-q21.
Biochem Biophys Res Commun. 264:955–961. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Zhang K, Harada Y, Wei X, Shukla D,
Rajendran A, Tawansy K, Bedell M, Lim S, Shaw PX, He X, et al: An
essential role of the cysteine-rich domain of FZD4 in Norrin/Wnt
signaling and familial exudative vitreoretinopathy. J Biol Chem.
286:10210–10215. 2011. View Article : Google Scholar :
|
|
94
|
Musada GR, Syed H, Jalali S, Chakrabarti S
and Kaur I: Mutation spectrum of the FZD-4, TSPAN12 and ZNF408
genes in Indian FEVR patients. BMC Ophthalmol. 16:902016.
View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Tang M, Ding X, Li J, Hu A, Yuan M, Yang
Y, Zhan Z, Li Z and Lu L: Novel mutations in FZD4 and
phenotype-genotype correlation in Chinese patients with familial
exudative vitreoretinopathy. Mol Vis. 22:917–932. 2016.PubMed/NCBI
|
|
96
|
Fei P, Zhang Q, Huang L, Xu Y, Zhu X, Tai
Z, Gong B, Ma S, Yao Q, Li J, et al: Identification of two novel
LRP5 mutations in families with familial exudative
vitreoretinopathy. Mol Vis. 20:395–409. 2014.PubMed/NCBI
|
|
97
|
Ye X, Wang Y and Nathans J: The
Norrin/Frizzled4 signaling pathway in retinal vascular development
and disease. Trends Mol Med. 16:417–425. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Stenman JM, Rajagopal J, Carroll TJ,
Ishibashi M, McMahon J and McMahon AP: Canonical Wnt signaling
regulates organ-specific assembly and differentiation of CNS
vasculature. Science. 322:1247–1250. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Wang Z, Liu CH, Sun Y, Gong Y, Favazza TL,
Morss PC, Saba NJ, Fredrick TW, He X, Akula JD, et al:
Pharmacologic activation of Wnt signaling by lithium normalizes
retinal vasculature in a murine model of familial exudative
vitreoretinopathy. Am J Pathol. 186:2588–2600. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Birdsey GM, Shah AV, Dufton N, Reynolds
LE, Osuna Almagro L, Yang Y, Aspalter IM, Khan ST, Mason JC, Dejana
E, et al: The endothelial transcription factor ERG promotes
vascular stability and growth through Wnt/β-catenin signaling. Dev
Cell. 32:82–96. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Arthofer E, Hot B, Petersen J, Strakova K,
Jäger S, Grundmann M, Kostenis E, Gutkind JS and Schulte G: WNT
stimulation dissociates a Frizzled 4 inactive-state complex with
Gα12/13. Mol Pharmacol. 90:447–459. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Adler PN: The frizzled/stan pathway and
planar cell polarity in the Drosophila wing. Curr Top Dev Biol.
101:1–31. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Yang Y and Mlodzik M: Wnt-Frizzled/planar
cell polarity signaling: Cellular orientation by facing the wind
(Wnt). Annu Rev Cell Dev Biol. 31:623–646. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Gao B, Song H, Bishop K, Elliot G, Garrett
L, English MA, Andre P, Robinson J, Sood R, Minami Y, et al: Wnt
signaling gradients establish planar cell polarity by inducing
Vangl2 phosphorylation through Ror2. Dev Cell. 20:163–176. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Nishimura T, Honda H and Takeichi M:
Planar cell polarity links axes of spatial dynamics in neural-tube
closure. Cell. 149:1084–1097. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Pan X, Sittaramane V, Gurung S and
Chandrasekhar A: Structural and temporal requirements of Wnt/PCP
protein Vangl2 function for convergence and extension movements and
facial branchiomotor neuron migration in zebrafish. Mech Dev.
131:1–14. 2014. View Article : Google Scholar :
|
|
107
|
Gödde NJ, Pearson HB, Smith LK and Humbert
PO: Dissecting the role of polarity regulators in cancer through
the use of mouse models. Exp Cell Res. 328:249–257. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Cantrell VA and Jessen JR: The planar cell
polarity protein Van Gogh-Like 2 regulates tumor cell migration and
matrix metalloproteinase-dependent invasion. Cancer Lett.
287:54–61. 2010. View Article : Google Scholar
|
|
109
|
O'Connell MP, Fiori JL, Xu M, Carter AD,
Frank BP, Camilli TC, French AD, Dissanayake SK, Indig FE, Bernier
M, et al: The orphan tyrosine kinase receptor, ROR2, mediates Wnt5A
signaling in metastatic melanoma. Oncogene. 29:34–44. 2010.
View Article : Google Scholar :
|
|
110
|
Wang W, Runkle KB, Terkowski SM, Ekaireb
RI and Witze ES: Protein depalmitoylation is induced by Wnt5a and
promotes polarized cell behavior. J Biol Chem. 290:15707–15716.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Webster MR, Kugel CH III and Weeraratna
AT: The Wnts of change: How Wnts regulate phenotype switching in
melanoma. Biochim Biophys Acta. 1856:244–251. 2015.PubMed/NCBI
|
|
112
|
Katoh M: Function and cancer genomics of
FAT family genes (Review). Int J Oncol. 41:1913–1918.
2012.PubMed/NCBI
|
|
113
|
Matis M and Axelrod JD: Regulation of PCP
by the Fat signaling pathway. Genes Dev. 27:2207–2220. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Katoh M and Katoh M: Identification and
characterization of human PRICKLE1 and PRICKLE2 genes as well as
mouse Prickle1 and Prickle2 genes homologous to Drosophila tissue
polarity gene prickle. Int J Mol Med. 11:249–256. 2003.PubMed/NCBI
|
|
115
|
De Marco P, Merello E, Piatelli G, Cama A,
Kibar Z and Capra V: Planar cell polarity gene mutations contribute
to the etiology of human neural tube defects in our population.
Birth Defects Res A Clin Mol Teratol. 100:633–641. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Allache R, Lachance S, Guyot MC, De Marco
P, Merello E, Justice MJ, Capra V and Kibar Z: Novel mutations in
Lrp6 orthologs in mouse and human neural tube defects affect a
highly dosage-sensitive Wnt non-canonical planar cell polarity
pathway. Hum Mol Genet. 23:1687–1699. 2014. View Article : Google Scholar :
|
|
117
|
Bassuk AG, Wallace RH, Buhr A, Buller AR,
Afawi Z, Shimojo M, Miyata S, Chen S, Gonzalez-Alegre P, Griesbach
HL, et al: A homozygous mutation in human PRICKLE1 causes an
autosomal-recessive progressive myoclonus epilepsy-ataxia syndrome.
Am J Hum Genet. 83:572–581. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Tao H, Manak JR, Sowers L, Mei X, Kiyonari
H, Abe T, Dahdaleh NS, Yang T, Wu S, Chen S, et al: Mutations in
prickle orthologs cause seizures in flies, mice, and humans. Am J
Hum Genet. 88:138–149. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Sowers LP, Loo L, Wu Y, Campbell E, Ulrich
JD, Wu S, Paemka L, Wassink T, Meyer K, Bing X, et al: Disruption
of the non-canonical Wnt gene PRICKLE2 leads to autism-like
behaviors with evidence for hippocampal synaptic dysfunction. Mol
Psychiatry. 18:1077–1089. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
White JJ, Mazzeu JF, Hoischen A, Bayram Y,
Withers M, Gezdirici A, Kimonis V, Steehouwer M, Jhangiani SN,
Muzny DM, et al Baylor-Hopkins Center for Mendelian Genomics: DVL3
alleles resulting in a -1 frameshift of the last exon mediate
autosomal-dominant Robinow syndrome. Am J Hum Genet. 98:553–561.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Copp AJ and Greene ND: Genetics and
development of neural tube defects. J Pathol. 220:217–230.
2010.
|
|
122
|
Muñoz-Soriano V, Belacortu Y and Paricio
N: Planar cell polarity signaling in collective cell movements
during morphogenesis and disease. Curr Genomics. 13:609–622. 2012.
View Article : Google Scholar :
|
|
123
|
Wu G, Huang X, Hua Y and Mu D: Roles of
planar cell polarity pathways in the development of neutral tube
defects. J Biomed Sci. 18:662011. View Article : Google Scholar :
|
|
124
|
de la Hoz AB, Maortua H, García-Rives A,
Martínez-González MJ, Ezquerra M and Tejada MI: 3p14 de novo
interstitial microdeletion in a patient with intellectual
disability and autistic features with language impairment: A
comparison with similar cases. Case Rep Genet.
2015:8763482015.PubMed/NCBI
|
|
125
|
Mazzeu JF, Pardono E, Vianna-Morgante AM,
Richieri-Costa A, Ae Kim C, Brunoni D, Martelli L, de Andrade CE,
Colin G and Otto PA: Clinical characterization of autosomal
dominant and recessive variants of Robinow syndrome. Am J Med Genet
A. 143:320–325. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Person AD, Beiraghi S, Sieben CM,
Hermanson S, Neumann AN, Robu ME, Schleiffarth JR, Billington CJ
Jr, van Bokhoven H, Hoogeboom JM, et al: WNT5A mutations in
patients with autosomal dominant Robinow syndrome. Dev Dyn.
239:327–337. 2010.
|
|
127
|
Afzal AR, Rajab A, Fenske CD, Oldridge M,
Elanko N, Ternes-Pereira E, Tüysüz B, Murday VA, Patton MA, Wilkie
AO, et al: Recessive Robinow syndrome, allelic to dominant
brachydactyly type B, is caused by mutation of ROR2. Nat Genet.
25:419–422. 2000. View
Article : Google Scholar : PubMed/NCBI
|
|
128
|
Oldridge M, Fortuna AM, Maringa M,
Propping P, Mansour S, Pollitt C, DeChiara TM, Kimble RB,
Valenzuela DM, Yancopoulos GD, et al: Dominant mutations in ROR2,
encoding an orphan receptor tyrosine kinase, cause brachydactyly
type B. Nat Genet. 24:275–278. 2000. View
Article : Google Scholar : PubMed/NCBI
|
|
129
|
Green JL, Kuntz SG and Sternberg PW: Ror
receptor tyrosine kinases: Orphans no more. Trends Cell Biol.
18:536–544. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Minami Y, Oishi I, Endo M and Nishita M:
Ror-family receptor tyrosine kinases in noncanonical Wnt signaling:
Their implications in developmental morphogenesis and human
diseases. Dev Dyn. 239:1–15. 2010.
|
|
131
|
Petrova IM, Malessy MJ, Verhaagen J,
Fradkin LG and Noordermeer JN: Wnt signaling through the Ror
receptor in the nervous system. Mol Neurobiol. 49:303–315. 2014.
View Article : Google Scholar
|
|
132
|
Debebe Z and Rathmell WK: Ror2 as a
therapeutic target in cancer. Pharmacol Ther. 150:143–148. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Bunn KJ, Lai A, Al-Ani A, Farella M, Craw
S and Robertson SP: An osteosclerotic form of Robinow syndrome. Am
J Med Genet A. 164A:2638–2642. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Liu C, Lin C, Gao C, May-Simera H, Swaroop
A and Li T: Null and hypomorph Prickle1 alleles in mice phenocopy
human Robinow syndrome and disrupt signaling downstream of Wnt5a.
Biol Open. 3:861–870. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Ford CE, Qian Ma SS, Quadir A and Ward RL:
The dual role of the novel Wnt receptor tyrosine kinase, ROR2, in
human carcinogenesis. Int J Cancer. 133:779–787. 2013. View Article : Google Scholar
|
|
136
|
Asad M, Wong MK, Tan TZ, Choolani M, Low
J, Mori S, Virshup D, Thiery JP and Huang RY: FZD7 drives in vitro
aggressiveness in Stem-A subtype of ovarian cancer via regulation
of non-canonical Wnt/PCP pathway. Cell Death Dis. 5:e13462014.
View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Qin L, Yin YT, Zheng FJ, Peng LX, Yang CF,
Bao YN, Liang YY, Li XJ, Xiang YQ, Sun R, et al: WNT5A promotes
stemness characteristics in nasopharyngeal carcinoma cells leading
to metastasis and tumorigenesis. Oncotarget. 6:10239–10252. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Thiele S, Rachner TD, Rauner M and
Hofbauer LC: WNT5A and its receptors in the bone-cancer dialogue. J
Bone Miner Res. 31:1488–1496. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Kumawat K and Gosens R: WNT-5A: Signaling
and functions in health and disease. Cell Mol Life Sci. 73:567–587.
2016. View Article : Google Scholar :
|
|
140
|
Wei H, Wang N, Zhang Y, Wang S, Pang X and
Zhang S: Wnt-11 overexpression promoting the invasion of cervical
cancer cells. Tumour Biol. 37:11789–11798. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Arabzadeh S, Hossein G, Salehi-Dulabi Z
and Zarnani AH: WNT5A-ROR2 is induced by inflammatory mediators and
is involved in the migration of human ovarian cancer cell line
SKOV-3. Cell Mol Biol Lett. 21:92016. View Article : Google Scholar
|
|
142
|
Jiang W, Crossman DK, Mitchell EH, Sohn P,
Crowley MR and Serra R: WNT5A inhibits metastasis and alters
splicing of Cd44 in breast cancer cells. PLoS One. 8:e583292013.
View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Easter SL, Mitchell EH, Baxley SE, Desmond
R, Frost AR and Serra R: Wnt5a suppresses tumor formation and
redirects tumor phenotype in MMTV-Wnt1 tumors. PLoS One.
9:e1132472014. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Wang MT, Holderfield M, Galeas J,
Delrosario R, To MD, Balmain A and McCormick F: K-Ras promotes
tumorigenicity through suppression of non-canonical Wnt signaling.
Cell. 163:1237–1251. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Fukuda T, Chen L, Endo T, Tang L, Lu D,
Castro JE, Widhopf GF II, Rassenti LZ, Cantwell MJ, Prussak CE, et
al: Antisera induced by infusions of autologous Ad-CD154-leukemia B
cells identify ROR1 as an oncofetal antigen and receptor for Wnt5a.
Proc Natl Acad Sci USA. 105:3047–3052. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
146
|
Bicocca VT, Chang BH, Masouleh BK, Muschen
M, Loriaux MM, Druker BJ and Tyner JW: Crosstalk between ROR1 and
the Pre-B cell receptor promotes survival of t(1;19) acute
lymphoblastic leukemia. Cancer Cell. 22:656–667. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Yu J, Chen L, Cui B, Widhopf GF II, Shen
Z, Wu R, Zhang L, Zhang S, Briggs SP and Kipps TJ: Wnt5a induces
ROR1/ROR2 heterooligomerization to enhance leukemia chemotaxis and
proliferation. J Clin Invest. 126:585–598. 2016. View Article : Google Scholar :
|
|
148
|
Gentile A, Lazzari L, Benvenuti S,
Trusolino L and Comoglio PM: The ROR1 pseudokinase diversifies
signaling outputs in MET-addicted cancer cells. Int J Cancer.
135:2305–2316. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Hojjat-Farsangi M, Moshfegh A,
Daneshmanesh AH, Khan AS, Mikaelsson E, Osterborg A and Mellstedt
H: The receptor tyrosine kinase ROR1 - an oncofetal antigen for
targeted cancer therapy. Semin Cancer Biol. 29:21–31. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Li C, Wang S, Xing Z, Lin A, Liang K, Song
J, Hu Q, Yao J, Chen Z, Park PK, et al: A ROR1-HER3-lncRNA
signalling axis modulates the Hippo-YAP pathway to regulate bone
metastasis. Nat Cell Biol. 19:106–119. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Widhopf GF II, Cui B, Ghia EM, Chen L,
Messer K, Shen Z, Briggs SP, Croce CM and Kipps TJ: ROR1 can
interact with TCL1 and enhance leukemogenesis in Eμ-TCL1 transgenic
mice. Proc Natl Acad Sci USA. 111:793–798. 2014. View Article : Google Scholar
|
|
152
|
Yamaguchi T, Lu C, Ida L, Yanagisawa K,
Usukura J, Cheng J, Hotta N, Shimada Y, Isomura H, Suzuki M, et al:
ROR1 sustains caveolae and survival signalling as a scaffold of
cavin-1 and caveolin-1. Nat Commun. 7:100602016. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
O'Connell MP, Marchbank K, Webster MR,
Valiga AA, Kaur A, Vultur A, Li L, Herlyn M, Villanueva J, Liu Q,
et al: Hypoxia induces phenotypic plasticity and therapy resistance
in melanoma via the tyrosine kinase receptors ROR1 and ROR2. Cancer
Discov. 3:1378–1393. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Lai SS, Xue B, Yang Y, Zhao L, Chu CS, Hao
JY and Wen CJ: Ror2-Src signaling in metastasis of mouse melanoma
cells is inhibited by NRAGE. Cancer Genet. 205:552–562. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Niemann S, Zhao C, Pascu F, Stahl U,
Aulepp U, Niswander L, Weber JL and Müller U: Homozygous WNT3
mutation causes tetra-amelia in a large consanguineous family. Am J
Hum Genet. 74:558–563. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
156
|
Woods CG, Stricker S, Seemann P, Stern R,
Cox J, Sherridan E, Roberts E, Springell K, Scott S, Karbani G, et
al: Mutations in WNT7A cause a range of limb malformations,
including Fuhrmann syndrome and Al-Awadi/Raas-Rothschild/Schinzel
phocomelia syndrome. Am J Hum Genet. 79:402–408. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Biason-Lauber A, Konrad D, Navratil F and
Schoenle EJ: A WNT4 mutation associated with Müllerian-duct
regression and virilization in a 46, XX woman. N Engl J Med.
351:792–798. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Mandel H, Shemer R, Borochowitz ZU,
Okopnik M, Knopf C, Indelman M, Drugan A, Tiosano D,
Gershoni-Baruch R, Choder M, et al: SERKAL syndrome: An
autosomal-recessive disorder caused by a loss-of-function mutation
in WNT4. Am J Hum Genet. 82:39–47. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Kirikoshi H, Sekihara H and Katoh M:
WNT10A and WNT6, clustered in human chromosome 2q35 region with
head-to-tail manner, are strongly coexpressed in SW480 cells.
Biochem Biophys Res Commun. 283:798–805. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Adaimy L, Chouery E, Megarbane H, Mroueh
S, Delague V, Nicolas E, Belguith H, de Mazancourt P and Megarbane
A: Mutation in WNT10A is associated with an autosomal recessive
ectodermal dysplasia: The odonto-onycho-dermal dysplasia. Am J Hum
Genet. 81:821–828. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
161
|
van den Boogaard MJ, Créton M, Bronkhorst
Y, van der Hout A, Hennekam E, Lindhout D, Cune M and Ploos van
Amstel HK: Mutations in WNT10A are present in more than half of
isolated hypodontia cases. J Med Genet. 49:327–331. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
162
|
Yu P, Yang W, Han D, Wang X, Guo S, Li J,
Li F, Zhang X, Wong SW, Bai B, et al: Mutations in WNT10B are
identified in individuals with oligodontia. Am J Hum Genet.
99:195–201. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
163
|
Massink MP, Créton MA, Spanevello F,
Fennis WM, Cune MS, Savelberg SM, Nijman IJ, Maurice MM, van den
Boogaard MJ and van Haaften G: H and van Haaften G.
Loss-of-function mutations in the WNT co-receptor LRP6 cause
autosomal-dominant oligodontia. Am J Hum Genet. 97:621–626. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Poulsen A, Ho SY, Wang W, Alam J, Jeyaraj
DA, Ang SH, Tan ES, Lin GR, Cheong VW, Ke Z, et al: Pharmacophore
model for Wnt/Porcupine inhibitors and its use in drug design. J
Chem Inf Model. 55:1435–1448. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
165
|
Grzeschik KH, Bornholdt D, Oeffner F,
König A, del Carmen Boente M, Enders H, Fritz B, Hertl M, Grasshoff
U, Höfling K, et al: Deficiency of PORCN, a regulator of Wnt
signaling, is associated with focal dermal hypoplasia. Nat Genet.
39:833–835. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
166
|
Liu C, Widen SA, Williamson KA, Ratnapriya
R, Gerth-Kahlert C, Rainger J, Alur RP, Strachan E, Manjunath SH,
Balakrishnan A, et al UK10K Consortium: A secreted
WNT-ligand-binding domain of FZD5 generated by a frameshift
mutation causes autosomal dominant coloboma. Hum Mol Genet.
25:1382–1391. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Fröjmark AS, Schuster J, Sobol M,
Entesarian M, Kilander MB, Gabrikova D, Nawaz S, Baig SM, Schulte
G, Klar J, et al: Mutations in Frizzled 6 cause isolated
autosomal-recessive nail dysplasia. Am J Hum Genet. 88:852–860.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Parma P, Radi O, Vidal V, Chaboissier MC,
Dellambra E, Valentini S, Guerra L, Schedl A and Camerino G:
R-spondin1 is essential in sex determination, skin differentiation
and malignancy. Nat Genet. 38:1304–1309. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
169
|
Brüchle NO, Frank J, Frank V, Senderek J,
Akar A, Koc E, Rigopoulos D, van Steensel M, Zerres K and Bergmann
C: RSPO4 is the major gene in autosomal-recessive anonychia and
mutations cluster in the furin-like cysteine-rich domains of the
Wnt signaling ligand R-spondin 4. J Invest Dermatol. 128:791–796.
2008. View Article : Google Scholar
|
|
170
|
Ekici AB, Hilfinger D, Jatzwauk M, Thiel
CT, Wenzel D, Lorenz I, Boltshauser E, Goecke TW, Staatz G,
Morris-Rosendahl DJ, et al: Disturbed Wnt signalling due to a
mutation in CCDC88C causes an autosomal recessive non-syndromic
hydrocephalus with medial diverticulum. Mol Syndromol. 1:99–112.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
171
|
Aznar N, Midde KK, Dunkel Y, Lopez-Sanchez
I, Pavlova Y, Marivin A, Barbazán J, Murray F, Nitsche U, Janssen
KP, et al: Daple is a novel non-receptor GEF required for trimeric
G protein activation in Wnt signaling. eLife. 4:e070912015.
View Article : Google Scholar : PubMed/NCBI
|
|
172
|
Voronkov A and Krauss S: Wnt/β-catenin
signaling and small molecule inhibitors. Curr Pharm Des.
19:634–664. 2013. View Article : Google Scholar
|
|
173
|
Takebe N, Miele L, Harris PJ, Jeong W,
Bando H, Kahn M, Yang SX and Ivy SP: Targeting Notch, Hedgehog, and
Wnt pathways in cancer stem cells: Clinical update. Nat Rev Clin
Oncol. 12:445–464. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
174
|
Tai D, Wells K, Arcaroli J, Vanderbilt C,
Aisner DL, Messersmith WA and Lieu CH: Targeting the WNT signaling
pathway in cancer therapeutics. Oncologist. 20:1189–1198. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
175
|
Pelay-Gimeno M, Glas A, Koch O and
Grossmann TN: Structure-based design of inhibitors of
protein-protein interactions: Mimicking peptide binding epitopes.
Angew Chem Int Ed Engl. 54:8896–8927. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
176
|
Kakugawa S, Langton PF, Zebisch M, Howell
SA, Chang TH, Liu Y, Feizi T, Bineva G, O'Reilly N, Snijders AP, et
al: Notum deacylates Wnt proteins to suppress signalling activity.
Nature. 519:187–192. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
177
|
Madan B, Ke Z, Harmston N, Ho SY, Frois
AO, Alam J, Jeyaraj DA, Pendharkar V, Ghosh K, Virshup IH, et al:
Wnt addiction of genetically defined cancers reversed by PORCN
inhibition. Oncogene. 35:2197–2207. 2016. View Article : Google Scholar
|
|
178
|
Chen B, Dodge ME, Tang W, Lu J, Ma Z, Fan
CW, Wei S, Hao W, Kilgore J, Williams NS, et al: Small
molecule-mediated disruption of Wnt-dependent signaling in tissue
regeneration and cancer. Nat Chem Biol. 5:100–107. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
179
|
Liu J, Pan S, Hsieh MH, Ng N, Sun F, Wang
T, Kasibhatla S, Schuller AG, Li AG, Cheng D, et al: Targeting
Wnt-driven cancer through the inhibition of Porcupine by LGK974.
Proc Natl Acad Sci USA. 110:20224–20229. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
180
|
Proffitt KD, Madan B, Ke Z, Pendharkar V,
Ding L, Lee MA, Hannoush RN and Virshup DM: Pharmacological
inhibition of the Wnt acyltransferase PORCN prevents growth of
WNT-driven mammary cancer. Cancer Res. 73:502–507. 2013. View Article : Google Scholar
|
|
181
|
van de Wetering M, Francies HE, Francis
JM, Bounova G, Iorio F, Pronk A, van Houdt W, van Gorp J,
Taylor-Weiner A, Kester L, et al: Prospective derivation of a
living organoid biobank of colorectal cancer patients. Cell.
161:933–945. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
182
|
Cheng Y, Phoon YP, Jin X, Chong SY, Ip JC,
Wong BW and Lung ML: Wnt-C59 arrests stemness and suppresses growth
of nasopharyngeal carcinoma in mice by inhibiting the Wnt pathway
in the tumor microenvironment. Oncotarget. 6:14428–14439. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
183
|
Madan B, Ke Z, Lei ZD, Oliver FA, Oshima
M, Lee MA, Rozen S and Virshup DM: NOTUM is a potential
pharmacodynamic biomarker of Wnt pathway inhibition. Oncotarget.
7:12386–12392. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
184
|
Blyszczuk P, Müller-Edenborn B, Valenta T,
Osto E, Stellato M, Behnke S, Glatz K, Basler K, Lüscher TF,
Distler O, et al: Transforming growth factor-β-dependent Wnt
secretion controls myofibroblast formation and myocardial fibrosis
progression in experimental autoimmune myocarditis. Eur Heart J.
38:1413–1425. 2017.
|
|
185
|
Madan B, Patel MB, Zhang J, Bunte RM,
Rudemiller NP, Griffiths R, Virshup DM and Crowley SD: Experimental
inhibition of porcupine-mediated Wnt O-acylation attenuates kidney
fibrosis. Kidney Int. 89:1062–1074. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
186
|
Diamond JR, Eckhardt SG, Bendell JC,
Munster P, Morris VK, Kopetz S, Cattaruzza F, Kapoun AM, Dupont J
and Faoro L: A Phase 1a/b study of OMP-131R10, an anti-RSPO3
antibody, in advanced solid tumors and previously treated
metastatic colorectal cancer (CRC). Presented at: TAT 2016
Conference; Washington DC. 21–23 March, 2016;
|
|
187
|
Katoh M, Hirai M, Sugimura T and Terada M:
Cloning, expression and chromosomal localization of Wnt-13, a novel
member of the Wnt gene family. Oncogene. 13:873–876.
1996.PubMed/NCBI
|
|
188
|
Katoh M, Kirikoshi H, Terasaki H and
Shiokawa K: WNT2B2 mRNA, upregulated in primary gastric cancer, is
a positive regulator of the WNT- β-catenin-TCF signaling pathway.
Biochem Biophys Res Commun. 289:1093–1098. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
189
|
Jiang H, Li F, He C, Wang X, Li Q and Gao
H: Expression of Gli1 and Wnt2B correlates with progression and
clinical outcome of pancreatic cancer. Int J Clin Exp Pathol.
7:4531–4538. 2014.PubMed/NCBI
|
|
190
|
Li G, Liu Y, Su Z, Ren S, Zhu G, Tian Y
and Qiu Y: MicroRNA-324-3p regulates nasopharyngeal carcinoma
radioresistance by directly targeting WNT2B. Eur J Cancer.
49:2596–2607. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
191
|
Li SJ, Yang XN and Qian HY: Antitumor
effects of WNT2B silencing in GLUT1 overexpressing cisplatin
resistant head and neck squamous cell carcinoma. Am J Cancer Res.
5:300–308. 2014.
|
|
192
|
Kobayashi M, Huang CL, Sonobe M, Kikuchi R
and Date H: Ad-shWnt2b vector therapy demonstrates antitumor
activity in orthotopic intrapleural models as monitored with the in
vitro imaging system (IVIS). Anticancer Res. 36:5887–5893. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
193
|
Tokuhara M, Hirai M, Atomi Y, Terada M and
Katoh M: Molecular cloning of human Frizzled-6. Biochem Biophys Res
Commun. 243:622–627. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
194
|
Cantilena S, Pastorino F, Pezzolo A,
Chayka O, Pistoia V, Ponzoni M and Sala A: Frizzled receptor 6
marks rare, highly tumourigenic stem-like cells in mouse and human
neuroblastomas. Oncotarget. 2:976–983. 2011. View Article : Google Scholar
|
|
195
|
Kim BK, Yoo HI, Kim I, Park J and Kim Yoon
S: FZD6 expression is negatively regulated by miR-199a-5p in human
colorectal cancer. BMB Rep. 48:360–366. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
196
|
Corda G, Sala G, Lattanzio R, Iezzi M,
Sallese M, Fragassi G, Lamolinara A, Mirza H, Barcaroli D, Ermler
S, et al: Functional and prognostic significance of the genomic
amplification of frizzled 6 (FZD6) in breast cancer. J Pathol.
241:350–361. 2017. View Article : Google Scholar :
|
|
197
|
Saitoh T, Hirai M and Katoh M: Molecular
cloning and characterization of human Frizzled-5 gene on chromosome
2q33.3-q34 region. Int J Oncol. 19:105–110. 2001.PubMed/NCBI
|
|
198
|
Steinhart Z, Pavlovic Z, Chandrashekhar M,
Hart T, Wang X, Zhang X, Robitaille M, Brown KR, Jaksani S,
Overmeer R, et al: Genome-wide CRISPR screens reveal a Wnt-FZD5
signaling circuit as a druggable vulnerability of RNF43-mutant
pancreatic tumors. Nat Med. 23:60–68. 2017. View Article : Google Scholar
|
|
199
|
Sagara N, Toda G, Hirai M, Terada M and
Katoh M: Molecular cloning, differential expression, and
chromosomal localization of human frizzled-1, frizzled-2, and
frizzled-7. Biochem Biophys Res Commun. 252:117–122. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
200
|
Simmons GE Jr, Pandey S,
Nedeljkovic-Kurepa A, Saxena M, Wang A and Pruitt K: Frizzled 7
expression is positively regulated by SIRT1 and β-catenin in breast
cancer cells. PLoS One. 9:e988612014. View Article : Google Scholar
|
|
201
|
Vincan E, Flanagan DJ, Pouliot N, Brabletz
T and Spaderna S: Variable FZD7 expression in colorectal cancers
indicates regulation by the tumour microenvironment. Dev Dyn.
239:311–317. 2010.
|
|
202
|
Qiu X, Jiao J, Li Y and Tian T:
Overexpression of FZD7 promotes glioma cell proliferation by
upregulating TAZ. Oncotarget. 7:85987–85999. 2016.PubMed/NCBI
|
|
203
|
Song J, Gao L, Yang G, Tang S, Xie H, Wang
Y, Wang J, Zhang Y, Jin J, Gou Y, et al: MiR-199a regulates cell
proliferation and survival by targeting FZD7. PLoS One.
9:e1100742014. View Article : Google Scholar : PubMed/NCBI
|
|
204
|
Koike J, Takagi A, Miwa T, Hirai M, Terada
M and Katoh M: Molecular cloning of Frizzled-10, a novel member of
the Frizzled gene family. Biochem Biophys Res Commun. 262:39–43.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
205
|
Gong C, Qu S, Lv XB, Liu B, Tan W, Nie Y,
Su F, Liu Q, Yao H and Song E: BRMS1L suppresses breast cancer
metastasis by inducing epigenetic silence of FZD10. Nat Commun.
5:54062014. View Article : Google Scholar : PubMed/NCBI
|
|
206
|
Terasaki H, Saitoh T, Shiokawa K and Katoh
M: Frizzled-10, upregulated in primary colorectal cancer, is a
positive regulator of the WNT-β-catenin-TCF signaling pathway. Int
J Mol Med. 9:107–112. 2002.PubMed/NCBI
|
|
207
|
Hanaoka H, Katagiri T, Fukukawa C,
Yoshioka H, Yamamoto S, Iida Y, Higuchi T, Oriuchi N, Paudyal B,
Paudyal P, et al: Radioimmunotherapy of solid tumors targeting a
cell-surface protein, FZD10: Therapeutic efficacy largely depends
on radio-sensitivity. Ann Nucl Med. 23:479–485. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
208
|
Gurney A, Axelrod F, Bond CJ, Cain J,
Chartier C, Donigan L, Fischer M, Chaudhari A, Ji M, Kapoun AM, et
al: Wnt pathway inhibition via the targeting of Frizzled receptors
results in decreased growth and tumorigenicity of human tumors.
Proc Natl Acad Sci USA. 109:11717–11722. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
209
|
Nielsen TO, Poulin NM and Ladanyi M:
Synovial sarcoma: Recent discoveries as a roadmap to new avenues
for therapy. Cancer Discov. 5:124–134. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
210
|
Le PN, McDermott JD and Jimeno A:
Targeting the Wnt pathway in human cancers: Therapeutic targeting
with a focus on OMP-54F28. Pharmacol Ther. 146:1–11. 2015.
View Article : Google Scholar
|
|
211
|
Ferreira Tojais N, Peghaire C, Franzl N,
Larrieu-Lahargue F, Jaspard B, Reynaud A, Moreau C, Couffinhal T,
Duplàa C and Dufourcq P: Frizzled7 controls vascular permeability
through the Wnt-canonical pathway and crosstalk with endothelial
cell junction complexes. Cardiovasc Res. 103:291–303. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
212
|
Phesse T, Flanagan D and Vincan E:
Frizzled7: A promising Achilles' heel for targeting the Wnt
receptor complex to treat cancer. Cancers (Basel). 8. pp. 502016,
View Article : Google Scholar
|
|
213
|
Katoh M: FGFR inhibitors: Effects on
cancer cells, tumor microenvironment and whole-body homeostasis
(Review). Int J Mol Med. 38:3–15. 2016.PubMed/NCBI
|
|
214
|
Shabani M and Hojjat-Farsangi M: Targeting
receptor tyrosine kinases using monoclonal antibodies: The most
specific tools for targeted-based cancer therapy. Curr Drug
Targets. 17:1687–1703. 2016. View Article : Google Scholar
|
|
215
|
Gentile A, Lazzari L, Benvenuti S,
Trusolino L and Comoglio PM: Ror1 is a pseudokinase that is crucial
for Met-driven tumorigenesis. Cancer Res. 71:3132–3141. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
216
|
Mellstedt H, Hojjat-Farsangi M,
Daneshmanesh AH, Moshfegh A, Byström S, Norin M, Olin T, Schultz J,
Vågberg J and Österborg A: First generation of a small chemical
molecule ROR1 RTK tyrosine kinase inhibitor. Ann Oncol. 27(Suppl
6): 15332016. View Article : Google Scholar
|
|
217
|
Khan AS, Hojjat-Farsangi M, Daneshmanesh
AH, Hansson L, Kokhaei P, Österborg A, Mellstedt H and Moshfegh A:
Dishevelled proteins are significantly upregulated in chronic
lymphocytic leukaemia. Tumour Biol. 37:11947–11957. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
218
|
Barat B, Chichili G, Ciccarone V, Tamura
J, Gorlatov S, Spliedt M, Chen F, Koenig S, Moore P, Bonvini E, et
al: Development of a humanized ROR1 × CD3 bispecific DART molecule
for the treatment of solid and liquid tumors. (Abstract).
Proceedings of the 107th Annual Meeting of the American Association
for Cancer Research; Apr 16–20, 2016; New Orleans, LA
AACR: Cancer Res. 76(Suppl 14): Abstract
1489. 2016.
|
|
219
|
Blankenship JW, Misher L, Mitchell D,
Zhang N, Tan P, Hoyos GH, Ravikumar P, Bader R, McMahan CJ, Miller
RE, et al: Anti-ROR1 × anti-CD3 ADAPTIR™ molecule, ES425, redirects
T-cell cytotoxicity and inhibits tumor growth in preclinical models
of triple-negative breast cancer. (Abstract). Proceedings of the
107th Annual Meeting of the American Association for Cancer
Research; Apr 16–20, 2016; New Orleans, LA. Philadelphia
AACR: Cancer Res. 76(Suppl 14): Abstract
4995. 2016.
|
|
220
|
Berger C, Sommermeyer D, Hudecek M, Berger
M, Balakrishnan A, Paszkiewicz PJ, Kosasih PL, Rader C and Riddell
SR: Safety of targeting ROR1 in primates with chimeric antigen
receptor-modified T cells. Cancer Immunol Res. 3:206–216. 2015.
View Article : Google Scholar :
|
|
221
|
Tian E, Zhan F, Walker R, Rasmussen E, Ma
Y, Barlogie B and Shaughnessy JD Jr: The role of the Wnt-signaling
antagonist DKK1 in the development of osteolytic lesions in
multiple myeloma. N Engl J Med. 349:2483–2494. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
222
|
Voorzanger-Rousselot N, Goehrig D, Journe
F, Doriath V, Body JJ, Clézardin P and Garnero P: Increased
Dickkopf-1 expression in breast cancer bone metastases. Br J
Cancer. 97:964–970. 2007.PubMed/NCBI
|
|
223
|
McClung MR, Grauer A, Boonen S, Bolognese
MA, Brown JP, Diez-Perez A, Langdahl BL, Reginster JY, Zanchetta
JR, Wasserman SM, et al: Romosozumab in postmenopausal women with
low bone mineral density. N Engl J Med. 370:412–420. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
224
|
Recker RR, Benson CT, Matsumoto T,
Bolognese MA, Robins DA, Alam J, Chiang AY, Hu L, Krege JH, Sowa H,
et al: A randomized, double-blind phase 2 clinical trial of
blosozumab, a sclerostin antibody, in postmenopausal women with low
bone mineral density. J Bone Miner Res. 30:216–224. 2015.
View Article : Google Scholar
|
|
225
|
Roschger A, Roschger P, Keplingter P,
Klaushofer K, Abdullah S, Kneissel M and Rauch F: Effect of
sclerostin antibody treatment in a mouse model of severe
osteogenesis imperfecta. Bone. 66:182–188. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
226
|
Fulciniti M, Tassone P, Hideshima T,
Vallet S, Nanjappa P, Ettenberg SA, Shen Z, Patel N, Tai YT,
Chauhan D, et al: Anti-DKK1 mAb (BHQ880) as a potential therapeutic
agent for multiple myeloma. Blood. 114:371–379. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
227
|
Bendell JC, Murphy JE, Mahalingam D,
Halmos B, Sirard CA, Landau SB and Ryan DP: A Phase 1 study of
DKN-01, an anti-DKK1 antibody, in combination with paclitaxel (pac)
in patients with DKK1 relapsed or refractory esophageal cancer (EC)
or gastro-esophageal junction tumors (GEJ). J Clin Oncol. 34(Suppl
4S): Abstract 111. 2016. View Article : Google Scholar
|
|
228
|
Betts AM, Clark TH, Yang J, Treadway JL,
Li M, Giovanelli MA, Abdiche Y, Stone DM and Paralkar VM: The
application of target information and preclinical
pharmacokinetic/pharmacodynamic modeling in predicting clinical
doses of a Dickkopf-1 antibody for osteoporosis. J Pharmacol Exp
Ther. 333:2–13. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
229
|
Florio M, Gunasekaran K, Stolina M, Li X,
Liu L, Tipton B, Salimi-Moosavi H, Asuncion FJ, Li C, Sun B, et al:
A bispecific antibody targeting sclerostin and DKK-1 promotes bone
mass accrual and fracture repair. Nat Commun. 7:115052016.
View Article : Google Scholar : PubMed/NCBI
|
|
230
|
Liu C and Yu X: ADP-ribosyltransferases
and poly ADP-ribosylation. Curr Protein Pept Sci. 16:491–501. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
231
|
Katoh M and Katoh M: Identification and
characterization of human TIPARP gene within the CCNL amplicon at
human chromosome 3q25.31. Int J Oncol. 23:541–547. 2003.PubMed/NCBI
|
|
232
|
Roper SJ, Chrysanthou S, Senner CE,
Sienerth A, Gnan S, Murray A, Masutani M, Latos P and Hemberger M:
ADP-ribosyltransferases Parp1 and Parp7 safeguard pluripotency of
ES cells. Nucleic Acids Res. 42:8914–8927. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
233
|
Haince JF, Rouleau M, Hendzel MJ, Masson
JY and Poirier GG: Targeting poly(ADP-ribosyl)ation: A promising
approach in cancer therapy. Trends Mol Med. 11:456–463. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
234
|
Gunderson CC and Moore KN: Olaparib: An
oral PARP-1 and PARP-2 inhibitor with promising activity in ovarian
cancer. Future Oncol. 11:747–757. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
235
|
Nathubhai A, Haikarainen T, Koivunen J,
Murthy S, Koumanov F, Lloyd MD, Holman GD, Pihlajaniemi T, Tosh D,
Lehtiö L, et al: Highly potent and isoform selective dual site
binding tankyrase/Wnt signaling inhibitors that increase cellular
glucose uptake and have antiproliferative activity. J Med Chem.
60:814–820. 2017. View Article : Google Scholar
|
|
236
|
Riffell JL, Lord CJ and Ashworth A:
Tankyrase-targeted therapeutics: Expanding opportunities in the
PARP family. Nat Rev Drug Discov. 11:923–936. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
237
|
Wang W, Li N, Li X, Tran MK, Han X and
Chen J: Tankyrase inhibitors target YAP by stabilizing angiomotin
family proteins. Cell Rep. 13:524–532. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
238
|
Kulak O, Chen H, Holohan B, Wu X, He H,
Borek D, Otwinowski Z, Yamaguchi K, Garofalo LA, Ma Z, et al:
Disruption of Wnt/β-catenin signaling and telomeric shortening are
inextricable consequences of tankyrase inhibition in human cells.
Mol Cell Biol. 35:2425–2435. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
239
|
Scarborough HA, Helfrich BA, Casás-Selves
M, Schuller AG, Grosskurth SE, Kim J, Tan AC, Chan DC, Zhang Z,
Zaberezhnyy V, et al: AZ1366: An inhibitor of tankyrase and the
canonical Wnt pathway that limits the persistence of non-small cell
lung cancer cells following EGFR inhibition. Clin Cancer Res.
23:1531–1541. 2017. View Article : Google Scholar
|
|
240
|
Lau T, Chan E, Callow M, Waaler J, Boggs
J, Blake RA, Magnuson S, Sambrone A, Schutten M, Firestein R, et
al: A novel tankyrase small-molecule inhibitor suppresses APC
mutation-driven colorectal tumor growth. Cancer Res. 73:3132–3144.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
241
|
Shultz MD, Cheung AK, Kirby CA, Firestone
B, Fan J, Chen CH, Chen Z, Chin DN, Dipietro L, Fazal A, et al:
Identification of NVP-TNKS656: The use of structure-efficiency
relationships to generate a highly potent, selective, and orally
active tankyrase inhibitor. J Med Chem. 56:6495–6511. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
242
|
Arqués O, Chicote I, Puig I, Tenbaum SP,
Argilés G, Dienstmann R, Fernández N, Caratù G, Matito J,
Silberschmidt D, et al: Tankyrase inhibition blocks Wnt/β-catenin
pathway and reverts resistance to PI3K and AKT inhibitors in the
treatment of colorectal cancer. Clin Cancer Res. 22:644–656. 2016.
View Article : Google Scholar
|
|
243
|
Huang SM, Mishina YM, Liu S, Cheung A,
Stegmeier F, Michaud GA, Charlat O, Wiellette E, Zhang Y, Wiessner
S, et al: Tankyrase inhibition stabilizes axin and antagonizes Wnt
signalling. Nature. 461:614–620. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
244
|
Casás-Selves M, Kim J, Zhang Z, Helfrich
BA, Gao D, Porter CC, Scarborough HA, Bunn PA Jr, Chan DC, Tan AC,
et al: Tankyrase and the canonical Wnt pathway protect lung cancer
cells from EGFR inhibition. Cancer Res. 72:4154–4164. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
245
|
Feng W, Teng R, Zhao Y, Gao J and Chu H:
Epigenetic modulation of Wnt signaling contributes to neuropathic
pain in rats. Mol Med Rep. 12:4727–4733. 2015.PubMed/NCBI
|
|
246
|
Meijer L, Skaltsounis AL, Magiatis P,
Polychronopoulos P, Knockaert M, Leost M, Ryan XP, Vonica CA,
Brivanlou A, Dajani R, et al: GSK-3-selective inhibitors derived
from Tyrian purple indirubins. Chem Biol. 10:1255–1266. 2003.
View Article : Google Scholar
|
|
247
|
Wu Y, Ai Z, Yao K, Cao L, Du J, Shi X, Guo
Z and Zhang Y: CHIR99021 promotes self-renewal of mouse embryonic
stem cells by modulation of protein-encoding gene and long
intergenic non-coding RNA expression. Exp Cell Res. 319:2684–2699.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
248
|
Atkinson JM, Rank KB, Zeng Y, Capen A,
Yadav V, Manro JR, Engler TA and Chedid M: Activating the
Wnt/β-catenin pathway for the treatment of melanoma: Application of
LY2090314, a novel selective inhibitor of glycogen synthase
kinase-3. PLoS One. 10:e01250282015. View Article : Google Scholar
|
|
249
|
Ding S, Wu TY, Brinker A, Peters EC, Hur
W, Gray NS and Schultz PG: Synthetic small molecules that control
stem cell fate. Proc Natl Acad Sci USA. 100:7632–7637. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
250
|
Lalit PA, Salick MR, Nelson DO, Squirrell
JM, Shafer CM, Patel NG, Saeed I, Schmuck EG, Markandeya YS, Wong
R, et al: Lineage reprogramming of fibroblasts into proliferative
induced cardiac progenitor cells by defined factors. Cell Stem
Cell. 18:354–367. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
251
|
Narcisi R, Arikan OH, Lehmann J, Ten Berge
D and van Osch GJ: Differential effects of small molecule WNT
agonists on the multilineage differentiation capacity of human
mesenchymal stem cells. Tissue Eng Part A. 22:1264–1273. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
252
|
Fiskus W, Sharma S, Saha S, Shah B,
Devaraj SG, Sun B, Horrigan S, Leveque C, Zu Y, Iyer S, et al:
Pre-clinical efficacy of combined therapy with novel β-catenin
antagonist BC2059 and histone deacetylase inhibitor against AML
cells. Leukemia. 29:1267–1278. 2015. View Article : Google Scholar
|
|
253
|
Trautmann M, Sievers E, Aretz S, Kindler
D, Michels S, Friedrichs N, Renner M, Kirfel J, Steiner S, Huss S,
et al: SS18-SSX fusion protein-induced Wnt/β-catenin signaling is a
therapeutic target in synovial sarcoma. Oncogene. 33:5006–5016.
2014. View Article : Google Scholar
|
|
254
|
Jang GB, Hong IS, Kim RJ, Lee SY, Park SJ,
Lee ES, Park JH, Yun CH, Chung JU, Lee KJ, et al: Wnt/β-catenin
small-molecule inhibitor CWP232228 preferentially inhibits the
growth of breast cancer stem-like cells. Cancer Res. 75:1691–1702.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
255
|
Emami KH, Nguyen C, Ma H, Kim DH, Jeong
KW, Eguchi M, Moon RT, Teo JL, Kim HY, Moon SH, et al: A small
molecule inhibitor of β-catenin/CREB-binding protein transcription
[corrected]. Proc Natl Acad Sci USA. 101:12682–12687. 2004.
View Article : Google Scholar
|
|
256
|
Fang L, Zhu Q, Neuenschwander M, Specker
E, Wulf-Goldenberg A, Weis WI, von Kries JP and Birchmeier W: A
small-molecule antagonist of the β-catenin/TCF4 interaction blocks
the self-renewal of cancer stem cells and suppresses tumorigenesis.
Cancer Res. 76:891–901. 2016. View Article : Google Scholar
|
|
257
|
Hwang SY, Deng X, Byun S, Lee C, Lee SJ,
Suh H, Zhang J, Kang Q, Zhang T, Westover KD, et al: Direct
targeting of β-catenin by a small molecule stimulates proteasomal
degradation and suppresses oncogenic Wnt/β-catenin signaling. Cell
Rep. 16:28–36. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
258
|
Sukhdeo K, Mani M, Zhang Y, Dutta J, Yasui
H, Rooney MD, Carrasco DE, Zheng M, He H, Tai YT, et al: Targeting
the β-catenin/TCF transcriptional complex in the treatment of
multiple myeloma. Proc Natl Acad Sci USA. 104:7516–7521. 2007.
View Article : Google Scholar
|
|
259
|
Lenz HJ and Kahn M: Safely targeting
cancer stem cells via selective catenin coactivator antagonism.
Cancer Sci. 105:1087–1092. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
260
|
Takada K, Zhu D, Bird GH, Sukhdeo K, Zhao
JJ, Mani M, Lemieux M, Carrasco DE, Ryan J, Horst D, et al:
Targeted disruption of the BCL9/β-catenin complex inhibits
oncogenic Wnt signaling. Sci Transl Med. 4:148ra1172012. View Article : Google Scholar
|
|
261
|
Henderson WR Jr, Chi EY, Ye X, Nguyen C,
Tien YT, Zhou B, Borok Z, Knight DA and Kahn M: Inhibition of
Wnt/beta-catenin/CREB binding protein (CBP) signaling reverses
pulmonary fibrosis. Proc Natl Acad Sci USA. 107:14309–14314. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
262
|
Zhou L, Li Y, Hao S, Zhou D, Tan RJ, Nie
J, Hou FF, Kahn M and Liu Y: Multiple genes of the
renin-angiotensin system are novel targets of Wnt/β-catenin
signaling. J Am Soc Nephrol. 26:107–120. 2015. View Article : Google Scholar
|
|
263
|
Gajewski TF, Schreiber H and Fu YX: Innate
and adaptive immune cells in the tumor microenvironment. Nat
Immunol. 14:1014–1022. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
264
|
Son B, Lee S, Youn H, Kim E, Kim W and
Youn B: The role of tumor microenvironment in therapeutic
resistance. Oncotarget. 8:3933–3945. 2017.
|
|
265
|
Spranger S, Bao R and Gajewski TF:
Melanoma-intrinsic β-catenin signalling prevents anti-tumour
immunity. Nature. 523:231–235. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
266
|
Malladi S, Macalinao DG, Jin X, He L,
Basnet H, Zou Y, de Stanchina E and Massagué J: Metastatic latency
and immune evasion through autocrine Inhibition of WNT. Cell.
165:45–60. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
267
|
Xu WD, Wang J, Yuan TL, Li YH, Yang H, Liu
Y, Zhao Y and Herrmann M: Interactions between canonical Wnt
signaling pathway and MAPK pathway regulate differentiation,
maturation and function of dendritic cells. Cell Immunol.
310:170–177. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
268
|
Naskar D, Maiti G, Chakraborty A, Roy A,
Chattopadhyay D and Sen M: Wnt5a-Rac1-NF-κB homeostatic circuitry
sustains innate immune functions in macrophages. J Immunol.
192:4386–4397. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
269
|
D'Amico L, Mahajan S, Capietto AH, Yang Z,
Zamani A, Ricci B, Bumpass DB, Meyer M, Su X, Wang-Gillam A, et al:
Dickkopf-related protein 1 (Dkk1) regulates the accumulation and
function of myeloid derived suppressor cells in cancer. J Exp Med.
213:827–840. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
270
|
Staal FJ and Arens R: Wnt signaling as
master regulator of T-lymphocyte responses: Implications for
transplant therapy. Transplantation. 100:2584–2592. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
271
|
Swafford D and Manicassamy S: Wnt
signaling in dendritic cells: Its role in regulation of immunity
and tolerance. Discov Med. 19:303–310. 2015.PubMed/NCBI
|
|
272
|
Holtzhausen A, Zhao F, Evans KS, Tsutsui
M, Orabona C, Tyler DS and Hanks BA: Melanoma-derived Wnt5a
promotes local dendritic-cell expression of IDO and
immunotolerance: Opportunities for pharmacologic enhancement of
immunotherapy. Cancer Immunol Res. 3:1082–1095. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
273
|
Hanks BA: Immune evasion pathways and the
design of dendritic cell-based cancer vaccines. Discov Med.
21:135–142. 2016.PubMed/NCBI
|
|
274
|
Kaur A, Webster MR and Weeraratna AT: In
the Wnt-er of life: Wnt signalling in melanoma and ageing. Br J
Cancer. 115:1273–1279. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
275
|
Law NC, Weck J, Kyriss B, Nilson JH and
Hunzicker-Dunn M: Lhcgr expression in granulosa cells: Roles for
PKA-phosphorylated β-catenin, TCF3, and FOXO1. Mol Endocrinol.
27:1295–1310. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
276
|
Liu Z and Habener JF: Glucagon-like
peptide-1 activation of TCF7L2-dependent Wnt signaling enhances
pancreatic beta cell proliferation. J Biol Chem. 283:8723–8735.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
277
|
Bellei B, Pitisci A, Catricalà C, Larue L
and Picardo M: Wnt/β-catenin signaling is stimulated by
α-melanocyte-stimulating hormone in melanoma and melanocyte cells:
Implication in cell differentiation. Pigment Cell Melanoma Res.
24:309–325. 2011. View Article : Google Scholar
|
|
278
|
Furuyashiki T and Narumiya S: Stress
responses: The contribution of prostaglandin E(2) and its
receptors. Nat Rev Endocrinol. 7:163–175. 2011. View Article : Google Scholar
|
|
279
|
Brudvik KW, Paulsen JE, Aandahl EM, Roald
B and Taskén K: Protein kinase A antagonist inhibits β-catenin
nuclear translocation, c-Myc and COX-2 expression and tumor
promotion in Apc(Min/+) mice. Mol Cancer. 10:1492011. View Article : Google Scholar
|
|
280
|
Jansen SR, Holman R, Hedemann I, Frankes
E, Elzinga CR, Timens W, Gosens R, de Bont ES and Schmidt M:
Prostaglandin E2 promotes MYCN non-amplified
neuroblastoma cell survival via β-catenin stabilization. J Cell Mol
Med. 19:210–226. 2015. View Article : Google Scholar
|
|
281
|
Estus TL, Choudhary S and Pilbeam CC:
Prostaglandin-mediated inhibition of PTH-stimulated β-catenin
signaling in osteoblasts by bone marrow macrophages. Bone.
85:123–130. 2016. View Article : Google Scholar : PubMed/NCBI
|