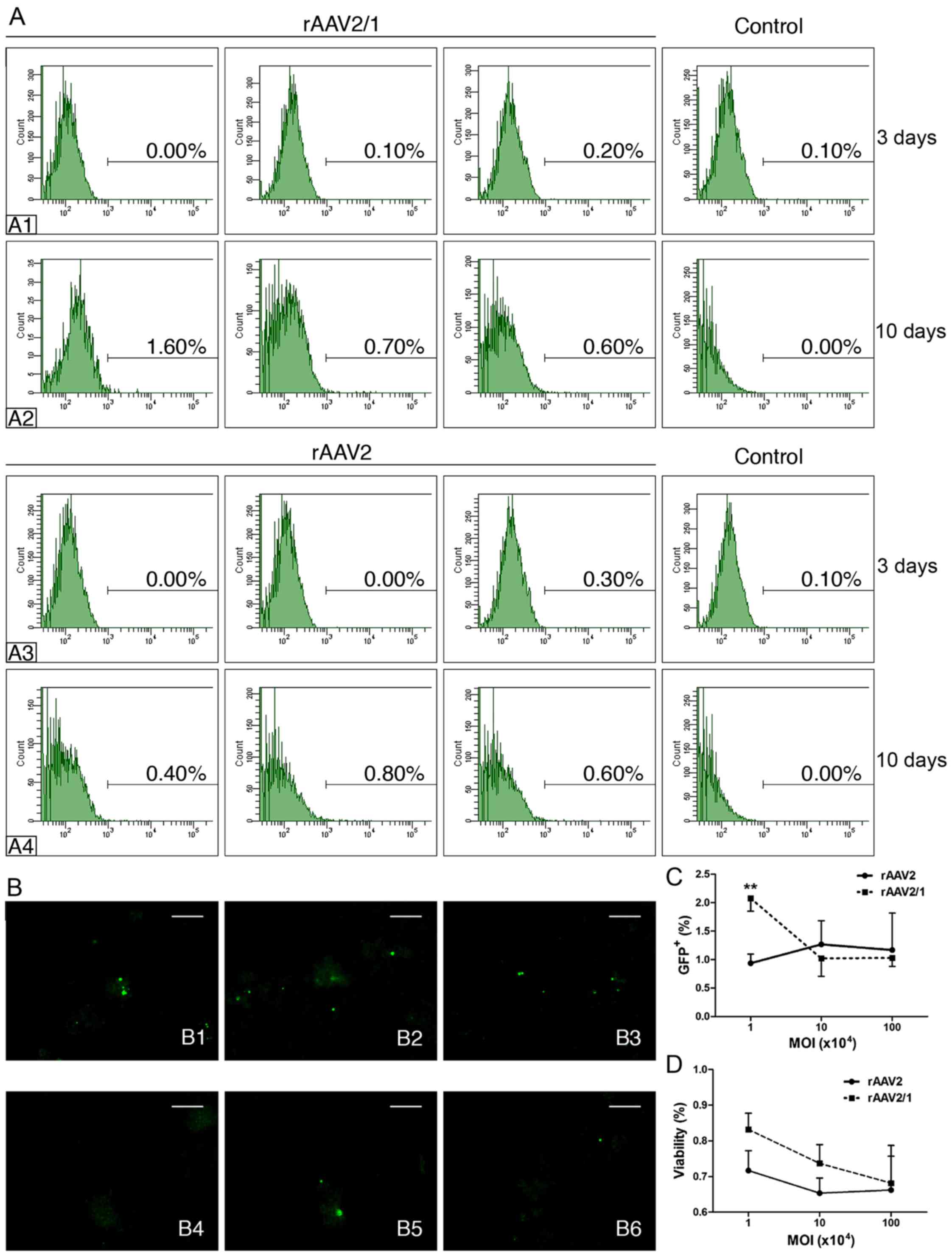
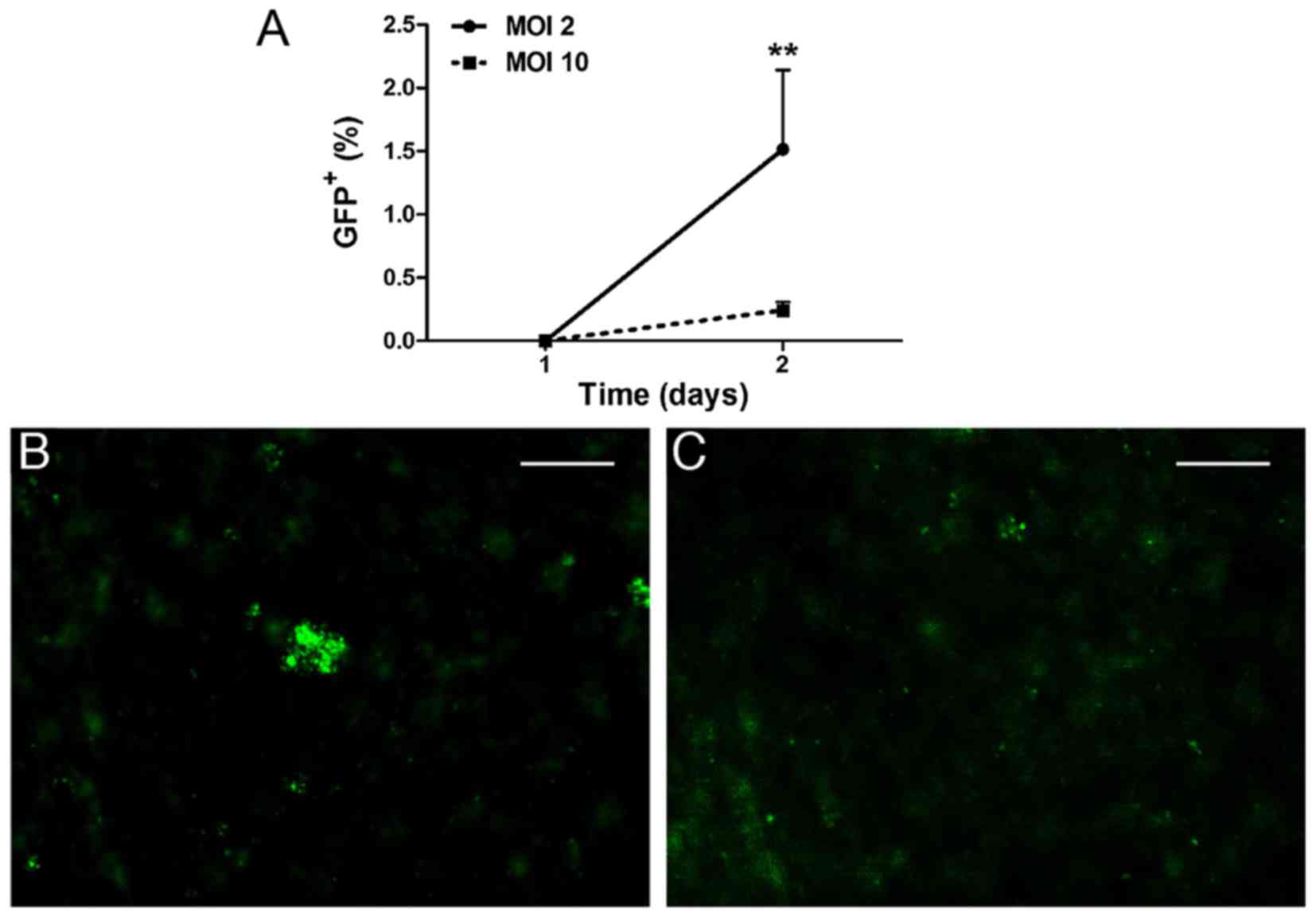
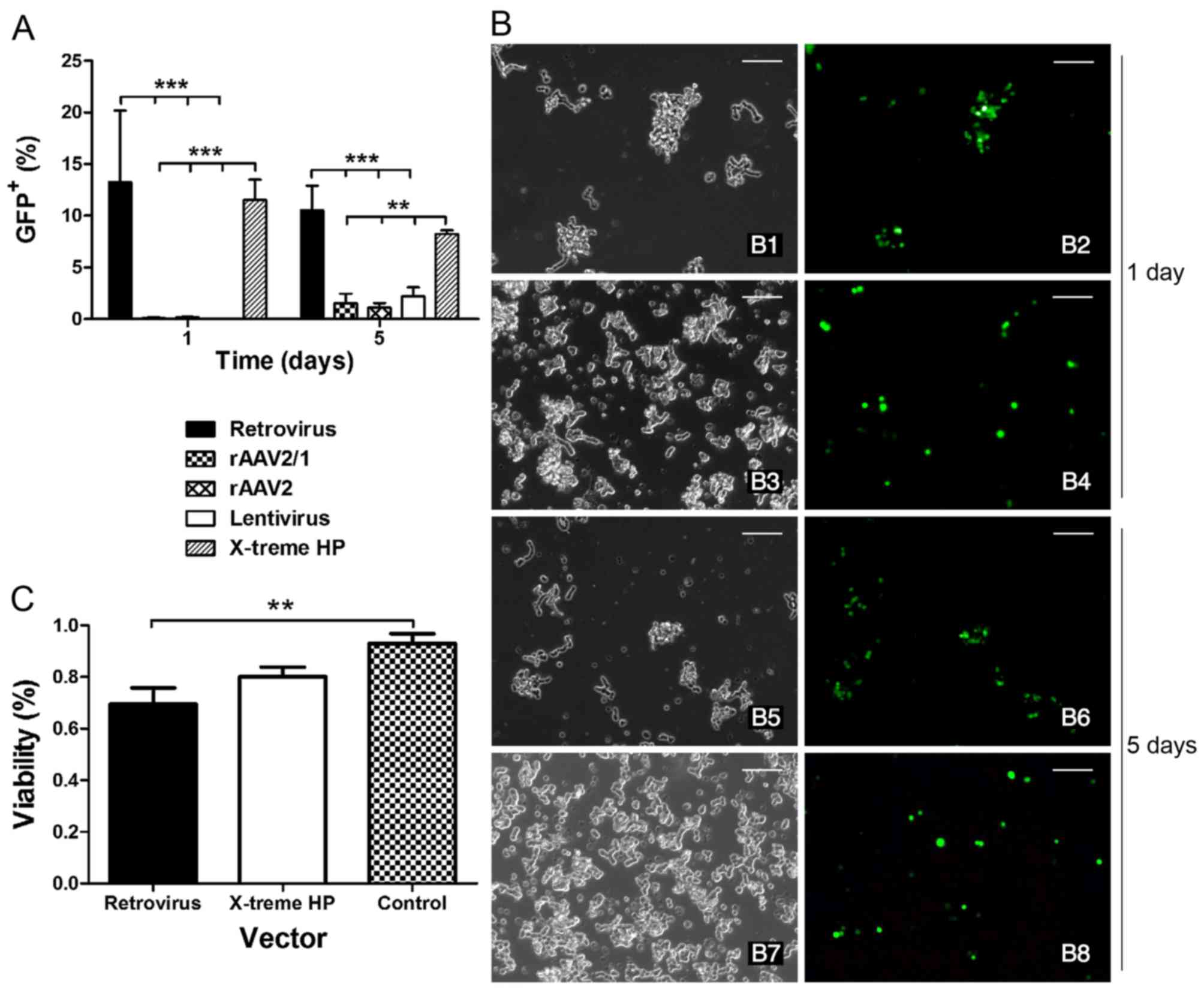
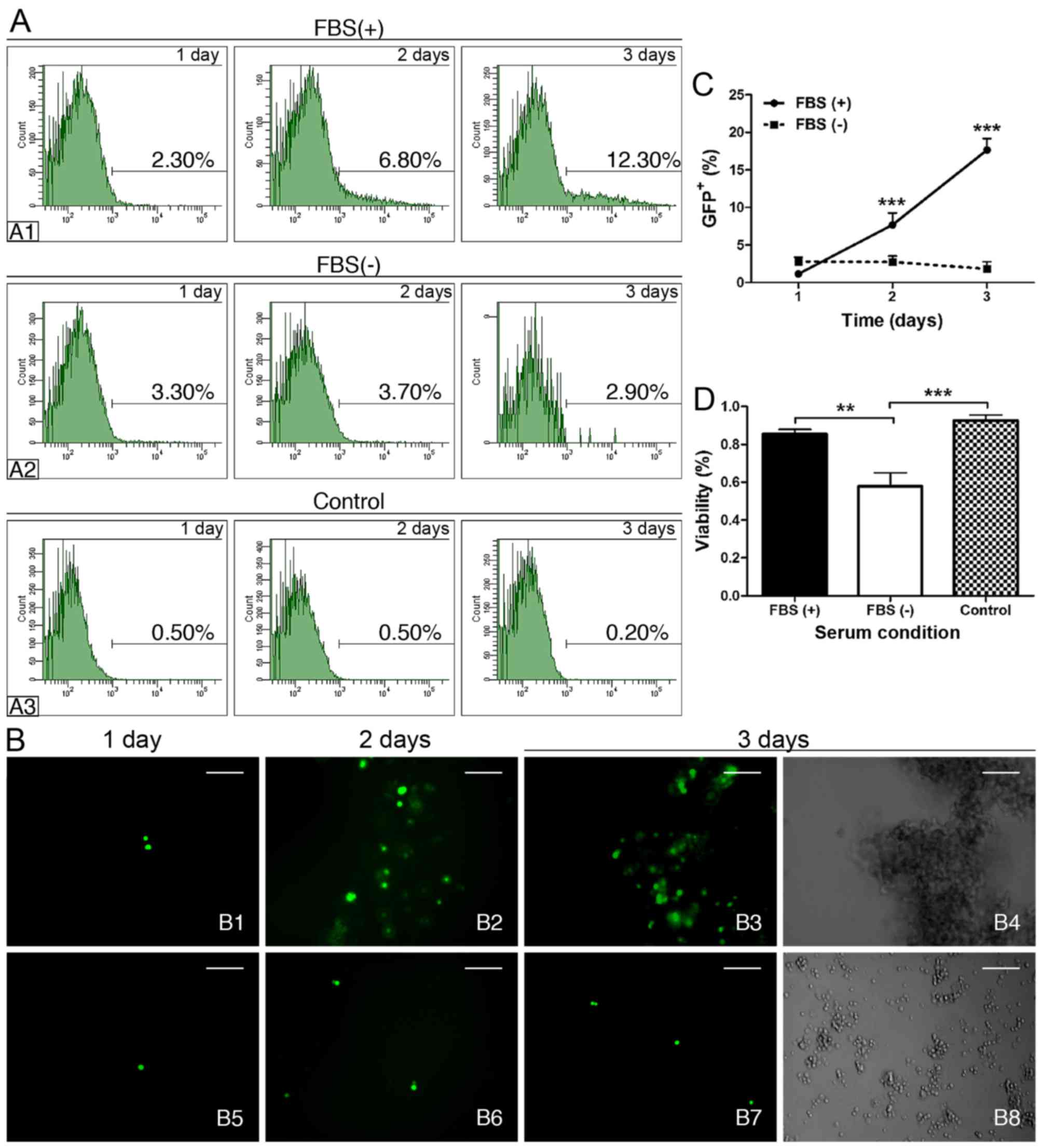
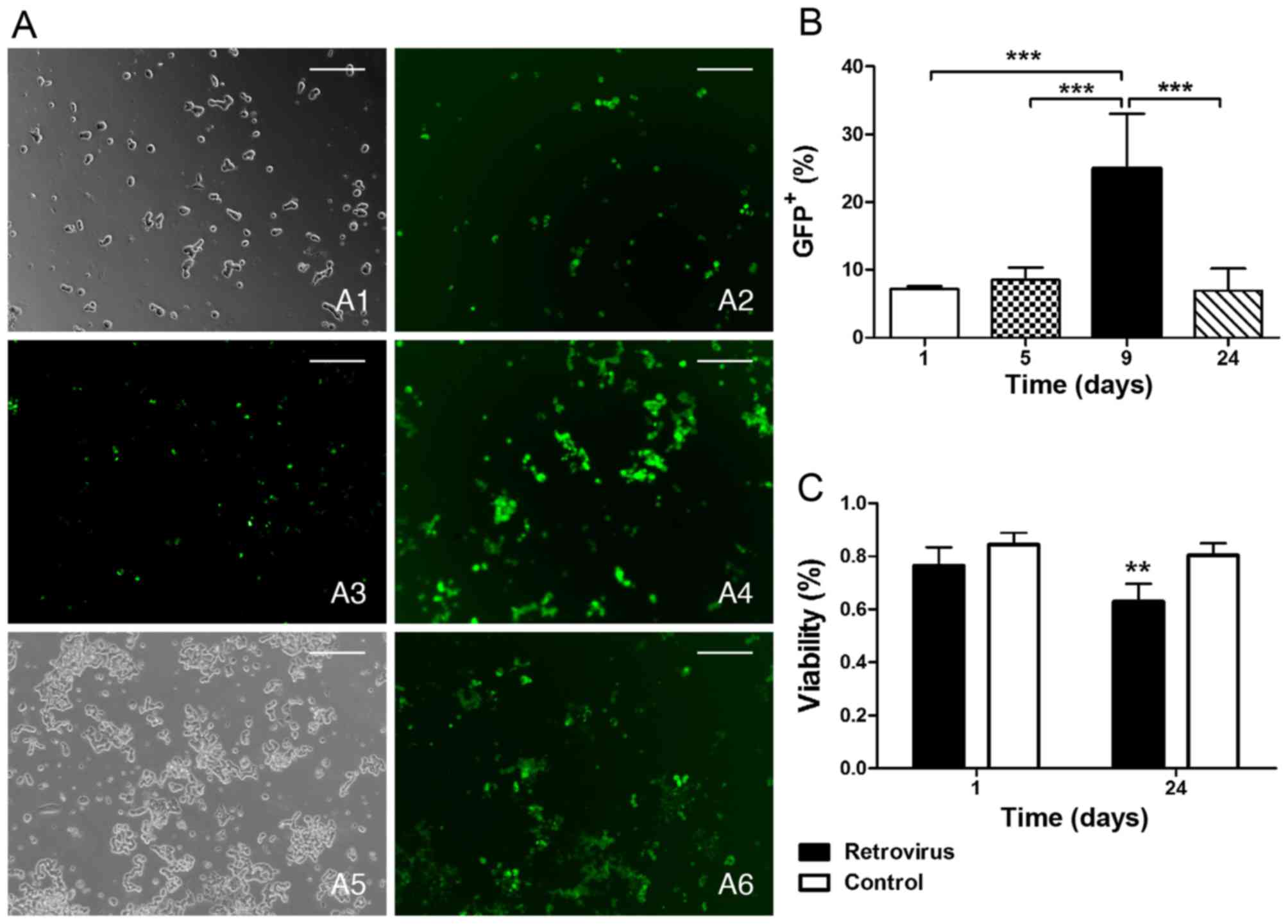
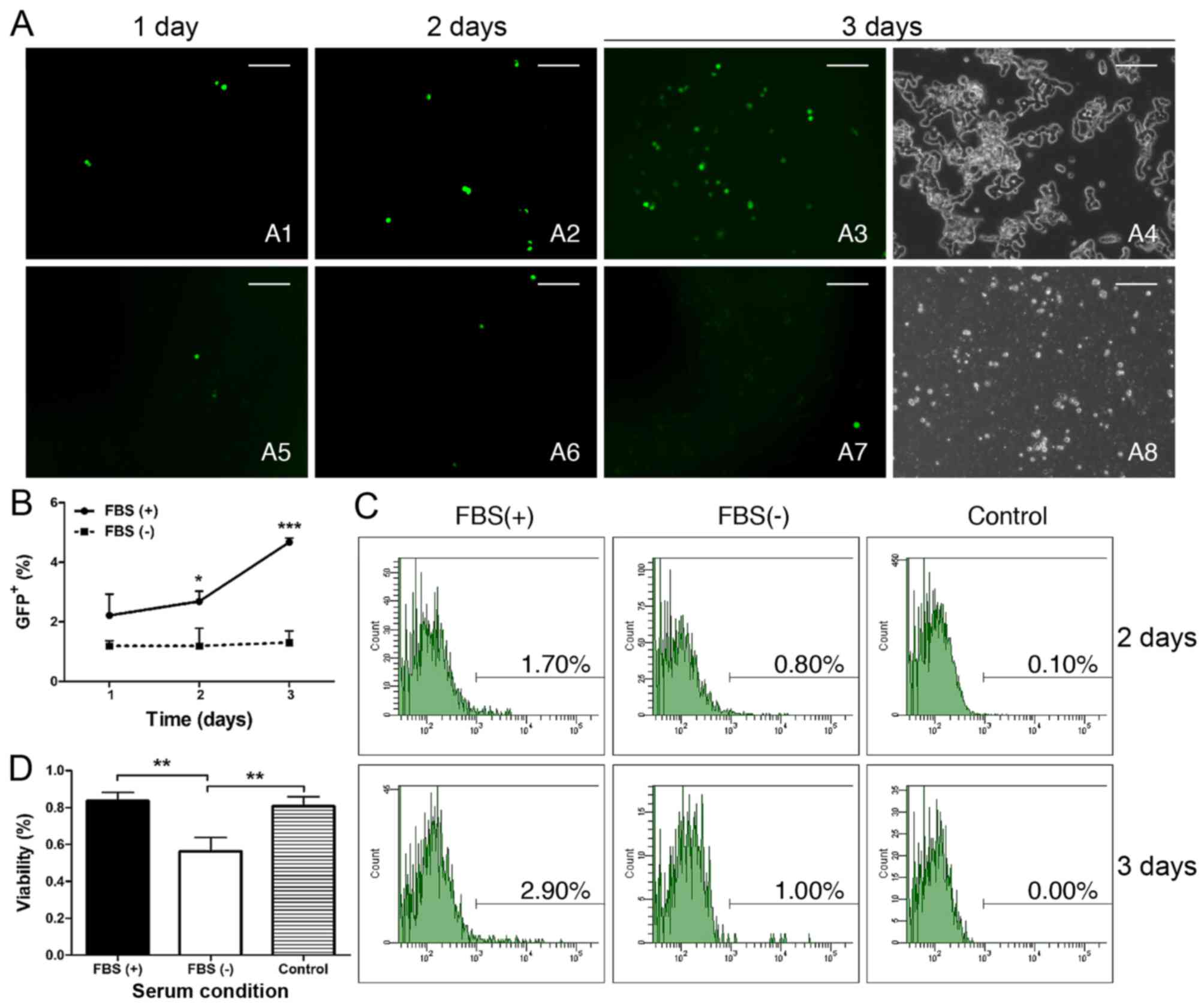
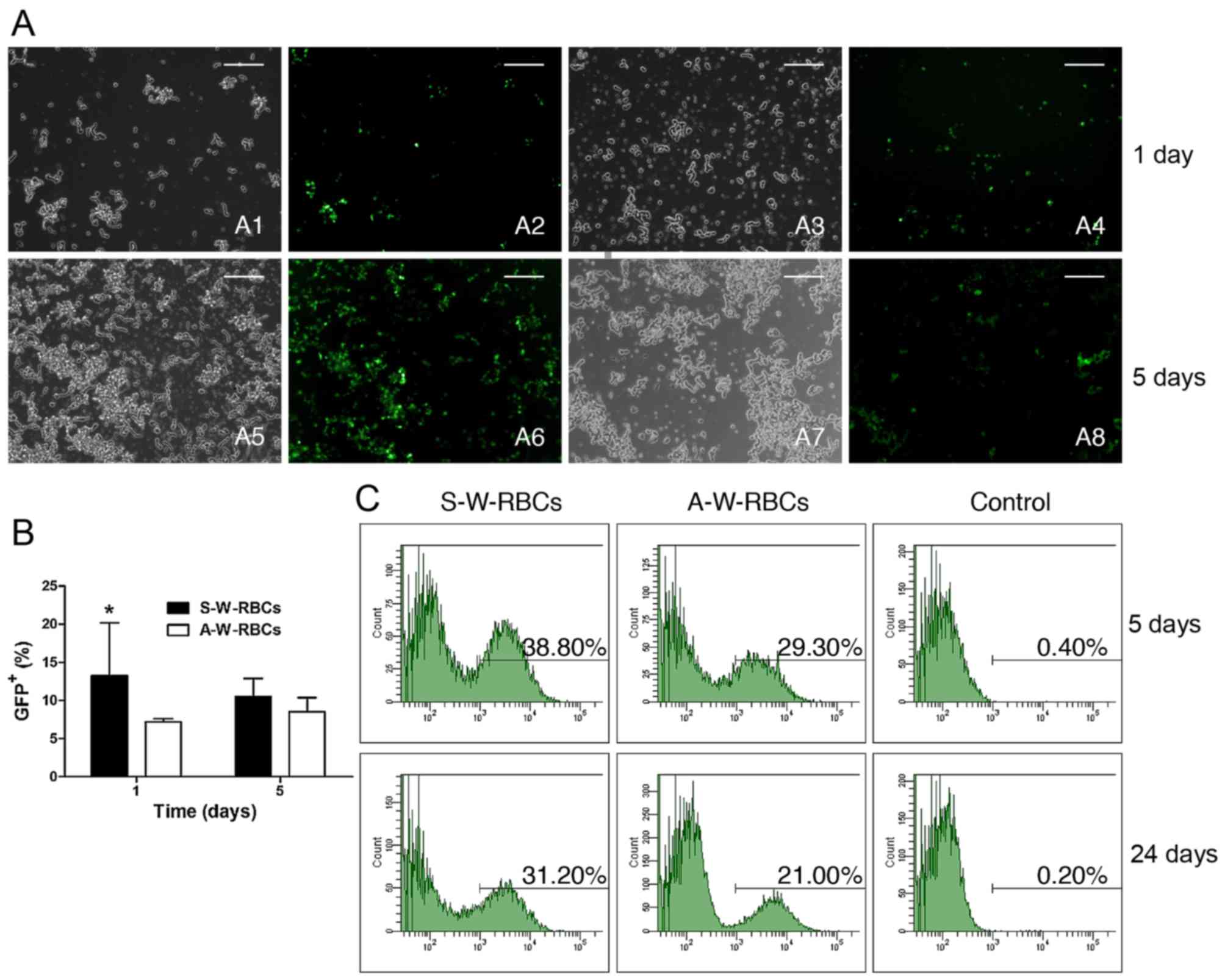
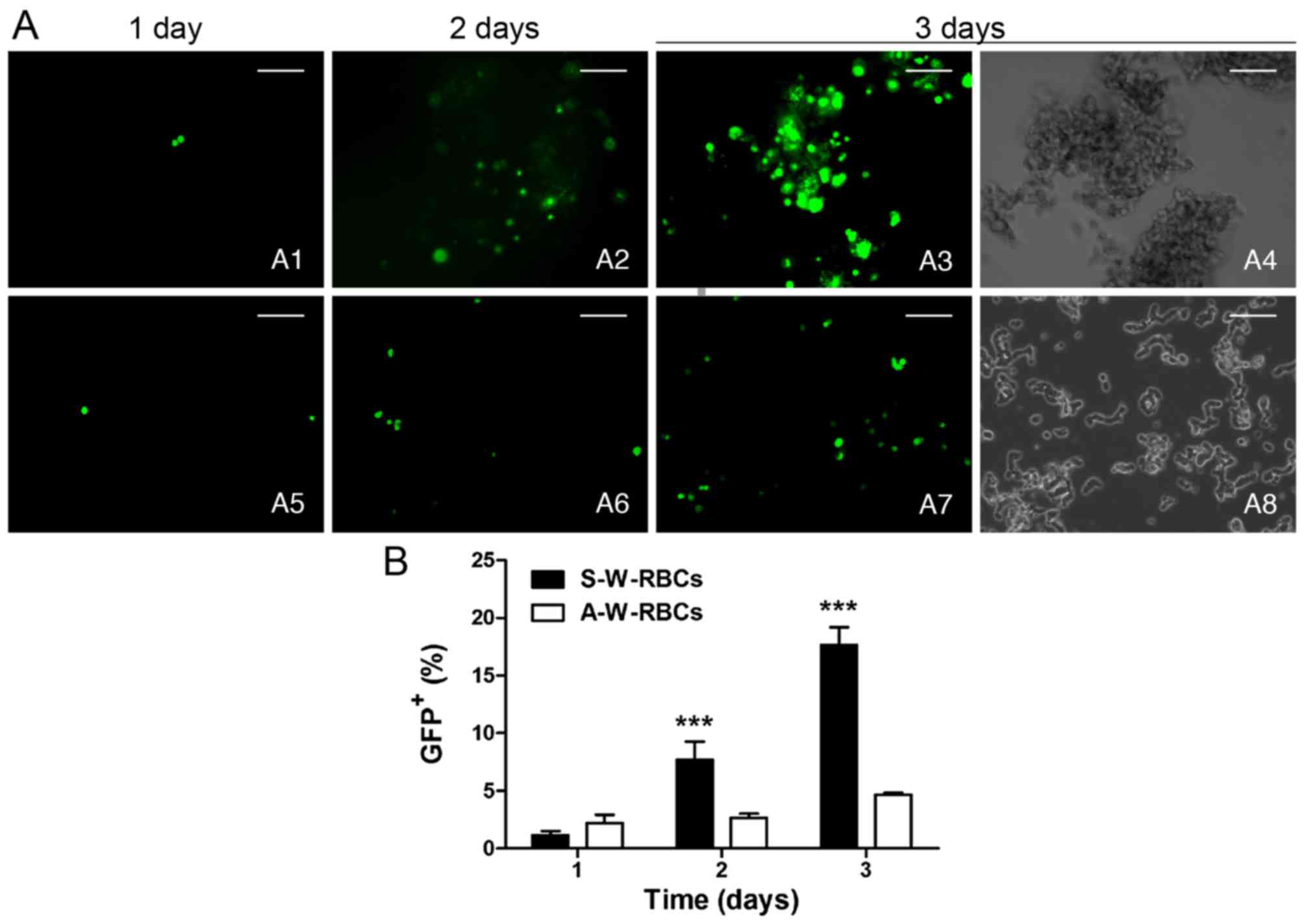
|
1
|
Bishop JO and Madson EC: Retinoblastoma.
Review of the current status. Surv Ophthalmol. 19:342–366.
1975.PubMed/NCBI
|
|
2
|
Shields CL, Meadows AT, Leahey AM and
Shields JA: Continuing challenges in the management of
retinoblastoma with chemotherapy. Retina. 24:849–862. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Garcia D and Quintyn JC: Treatment of
retinoblastoma by radiation therapy. Sixty-six years later. J Fr
Ophtalmol. 36:87–88. 2013.In French. View Article : Google Scholar
|
|
4
|
Kleinerman RA, Tucker MA, Abramson DH,
Seddon JM, Tarone RE and Fraumeni JF Jr: Risk of soft tissue
sarcomas by individual subtype in survivors of hereditary
retinoblastoma. J Natl Cancer Inst. 99:24–31. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Mouw KW, Sethi RV, Yeap BY, MacDonald SM,
Chen YL, Tarbell NJ, Yock TI, Munzenrider JE, Adams J, Grabowski E,
et al: Proton radiation therapy for the treatment of
retinoblastoma. Int J Radiat Oncol Biol Phys. 90:863–869. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chévez-Barrios P, Chintagumpala M, Mieler
W, Paysse E, Boniuk M, Kozinetz C, Hurwitz MY and Hurwitz RL:
Response of retinoblastoma with vitreous tumor seeding to
adenovirus-mediated delivery of thymidine kinase followed by
ganciclovir. J Clin Oncol. 23:7927–7935. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hurwitz MY, Marcus KT, Chévez-Barrios P,
Louie K, Aguilar-Cordova E and Hurwitz RL: Suicide gene therapy for
treatment of retinoblastoma in a murine model. Hum Gene Ther.
10:441–448. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Nichols KE, Walther S, Chao E, Shields C
and Ganguly A: Recent advances in retinoblastoma genetic research.
Curr Opin Ophthalmol. 20:351–355. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Giacinti C and Giordano A: RB and cell
cycle progression. Oncogene. 25:5220–5227. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Jia RB, Zhang P, Zhou YX, Song X, Liu HY,
Wang LZ, Luo M, Lu J, Ge SF and Fan XQ: VEGF-targeted RNA
interference suppresses angiogenesis and tumor growth of
retinoblastoma. Ophthalmic Res. 39:108–115. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Jia RB, Fan XQ, Wang XL, Zhang XQ, Zhang P
and Lu J: Inhibition of VEGF expression by plasmid-based RNA
interference in the retinoblastoma cells. Zhonghua Yan Ke Za Zhi.
43:493–498. 2007.In Chinese. PubMed/NCBI
|
|
12
|
Chau KY and Ono SJ: Gene transfer into
retinoblastoma cells. Biotechniques. 26:444–446. 1999.PubMed/NCBI
|
|
13
|
Adachi M, Brooks SE, Stein MR, Franklin BE
and Caccavo FA: Destruction of human retinoblastoma after treatment
by the E variant of encephalomyocarditis virus. J Neurooncol.
77:233–240. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Di Polo A and Farber DB: Rod
photoreceptor-specific gene expression in human retinoblastoma
cells. Proc Natl Acad Sci USA. 92:4016–4020. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Herman MM, Perentes E, Katsetos CD, Darcel
F, Frankfurter A, Collins VP, Donoso LA, Eng LF, Marangos PJ,
Wiechmann AF, et al: Neuroblastic differentiation potential of the
human retinoblastoma cell lines Y-79 and WERI-Rb1 maintained in an
organ culture system. An immunohistochemical, electron microscopic,
and biochemical study. Am J Pathol. 134:115–132. 1989.PubMed/NCBI
|
|
16
|
Muncaster MM, Cohen BL, Phillips RA and
Gallie BL: Failure of RB1 to reverse the malignant phenotype of
human tumor cell lines. Cancer Res. 52:654–661. 1992.PubMed/NCBI
|
|
17
|
Daya S and Berns KI: Gene therapy using
adeno-associated virus vectors. Clin Microbiol Rev. 21:583–593.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Al-Saikhan FI: The gene therapy revolution
in ophthalmology. Saudi J Ophthalmol. 27:107–111. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ali RR, Reichel MB, Thrasher AJ, Levinsky
RJ, Kinnon C, Kanuga N, Hunt DM and Bhattacharya SS: Gene transfer
into the mouse retina mediated by an adeno-associated viral vector.
Hum Mol Genet. 5:591–594. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Flannery JG, Zolotukhin S, Vaquero MI,
LaVail MM, Muzyczka N and Hauswirth WW: Efficient
photoreceptor-targeted gene expression in vivo by recombinant
adeno-associated virus. Proc Natl Acad Sci USA. 94:6916–6921. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Maguire AM, Simonelli F, Pierce EA, Pugh
EN Jr, Mingozzi F, Bennicelli J, Banfi S, Marshall KA, Testa F,
Surace EM, et al: Safety and efficacy of gene transfer for Leber's
congenital amaurosis. N Engl J Med. 358:2240–2248. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Maclachlan TK, Lukason M, Collins M,
Munger R, Isenberger E, Rogers C, Malatos S, Dufresne E, Morris J,
Calcedo R, et al: Preclinical safety evaluation of AAV2-sFLT01- a
gene therapy for age-related macular degeneration. Mol Ther.
19:326–334. 2011. View Article : Google Scholar
|
|
23
|
Lukason M, DuFresne E, Rubin H, Pechan P,
Li Q, Kim I, Kiss S, Flaxel C, Collins M, Miller J, et al:
Inhibition of choroidal neovascularization in a nonhuman primate
model by intravitreal administration of an AAV2 vector expressing a
novel anti-VEGF molecule. Mol Ther. 19:260–265. 2011. View Article : Google Scholar :
|
|
24
|
Auricchio A, Kobinger G, Anand V,
Hildinger M, O'Connor E, Maguire AM, Wilson JM and Bennett J:
Exchange of surface proteins impacts on viral vector cellular
specificity and transduction characteristics: The retina as a
model. Hum Mol Genet. 10:3075–3081. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Pang J, Cheng M, Haire SE, Barker E,
Planelles V and Blanks JC: Efficiency of lentiviral transduction
during development in normal and rd mice. Mol Vis. 12:756–767.
2006.PubMed/NCBI
|
|
26
|
Mátrai J, Chuah MK and VandenDriessche T:
Recent advances in lentiviral vector development and applications.
Mol Ther. 18:477–490. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Miyoshi H, Takahashi M, Gage FH and Verma
IM: Stable and efficient gene transfer into the retina using an
HIV-based lentiviral vector. Proc Natl Acad Sci USA.
94:10319–10323. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Garson K, Gamwell LF, Pitre EM and
Vanderhyden BC: Technical challenges and limitations of current
mouse models of ovarian cancer. J Ovarian Res. 5:392012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Tosetti F, Venè R, Arena G, Morini M,
Minghelli S, Noonan DM and Albini A: N-(4-hydroxyphenyl)retinamide
inhibits retinoblastoma growth through reactive oxygen
species-mediated cell death. Mol Pharmacol. 63:565–573. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Cepko CL: Emerging gene therapies for
retinal degenerations. J Neurosci. 32:6415–6420. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Day TP, Byrne LC, Schaffer DV and Flannery
JG: Advances in AAV vector development for gene therapy in the
retina. Adv Exp Med Biol. 801:687–693. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Janson CG, McPhee SW, Leone P, Freese A
and During MJ: Viral-based gene transfer to the mammalian CNS for
functional genomic studies. Trends Neurosci. 24:706–712. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Xu XL, Fang Y, Lee TC, Forrest D,
Gregory-Evans C, Almeida D, Liu A, Jhanwar SC, Abramson DH and
Cobrinik D: Retinoblastoma has properties of a cone precursor tumor
and depends upon cone-specific MDM2 signaling. Cell. 137:1018–1031.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Koirala A, Conley SM and Naash MI: A
review of therapeutic prospects of non-viral gene therapy in the
retinal pigment epithelium. Biomaterials. 34:7158–7167. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Boylan NJ, Kim AJ, Suk JS, Adstamongkonkul
P, Simons BW, Lai SK, Cooper MJ and Hanes J: Enhancement of airway
gene transfer by DNA nanoparticles using a pH-responsive block
copolymer of polyethylene glycol and poly-L-lysine. Biomaterials.
33:2361–2371. 2012. View Article : Google Scholar :
|
|
36
|
McIntosh JH, Cochrane M, Cobbold S,
Waldmann H, Nathwani SA, Davidoff AM and Nathwani AC: Successful
attenuation of humoral immunity to viral capsid and transgenic
protein following AAV-mediated gene transfer with a non-depleting
CD4 antibody and cyclosporine. Gene Ther. 19:78–85. 2012.
View Article : Google Scholar
|
|
37
|
VandenDriessche T: Muscling through AAV
immunity. Blood. 114:2009–2010. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Harvey AR, Kamphuis W, Eggers R, Symons
NA, Blits B, Niclou S, Boer GJ and Verhaagen J: Intravitreal
injection of adeno-associated viral vectors results in the
transduction of different types of retinal neurons in neonatal and
adult rats: A comparison with lentiviral vectors. Mol Cell
Neurosci. 21:141–157. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Daly G and Chernajovsky Y: Recent
developments in retroviral-mediated gene transduction. Mol Ther.
2:423–434. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Liu Y and Deisseroth A: Tumor vascular
targeting therapy with viral vectors. Blood. 107:3027–3033. 2006.
View Article : Google Scholar
|
|
41
|
Ramamoorth M and Narvekar A: Non viral
vectors in gene therapy- an overview. J Clin Diagn Res.
9:GE01–GE06. 2015.PubMed/NCBI
|
|
42
|
Yin H, Kanasty RL, Eltoukhy AA, Vegas AJ,
Dorkin JR and Anderson DG: Non-viral vectors for gene-based
therapy. Nat Rev Genet. 15:541–555. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Chan CL, Ewert KK, Majzoub RN, Hwu YK,
Liang KS, Leal C and Safinya CR: Optimizing cationic and neutral
lipids for efficient gene delivery at high serum content. J Gene
Med. 16:84–96. 2014. View Article : Google Scholar : PubMed/NCBI
|