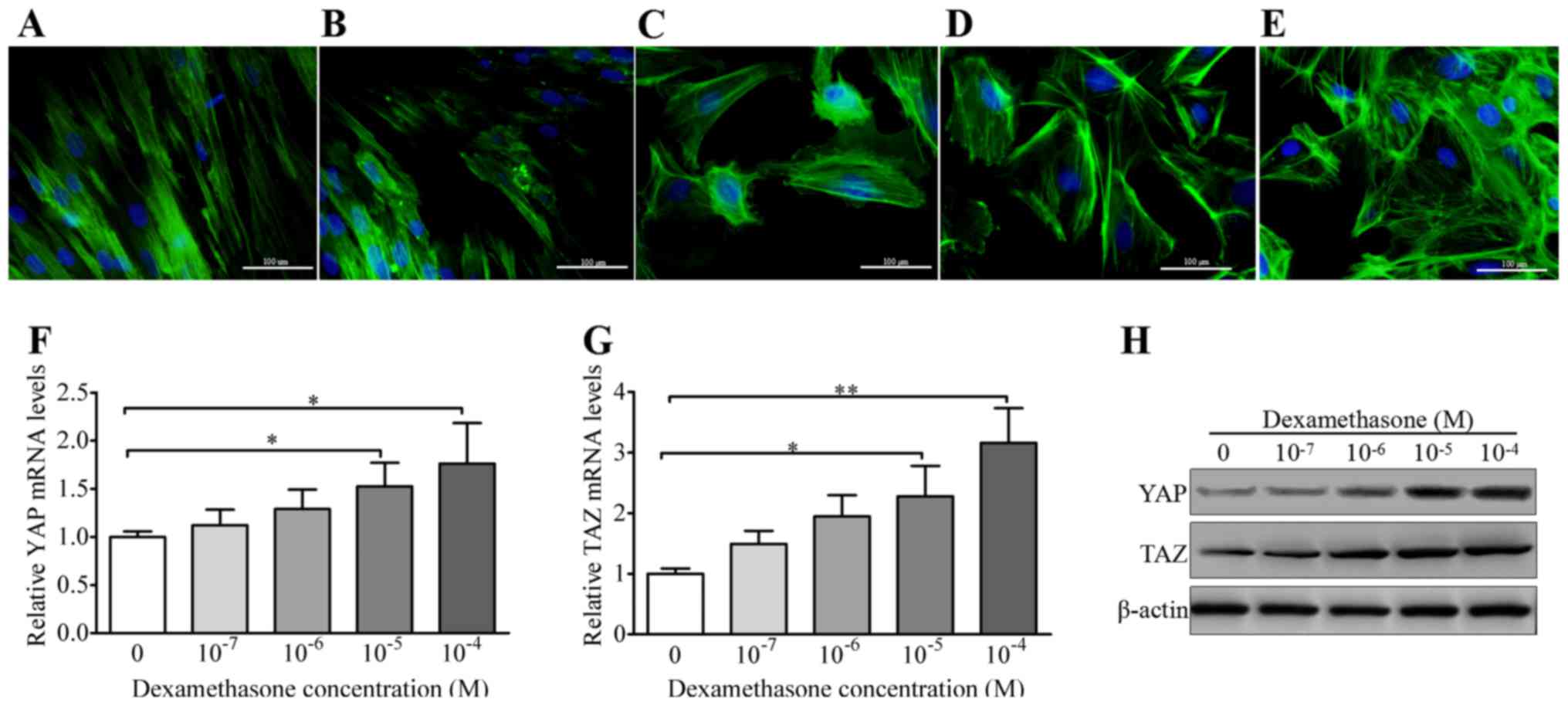
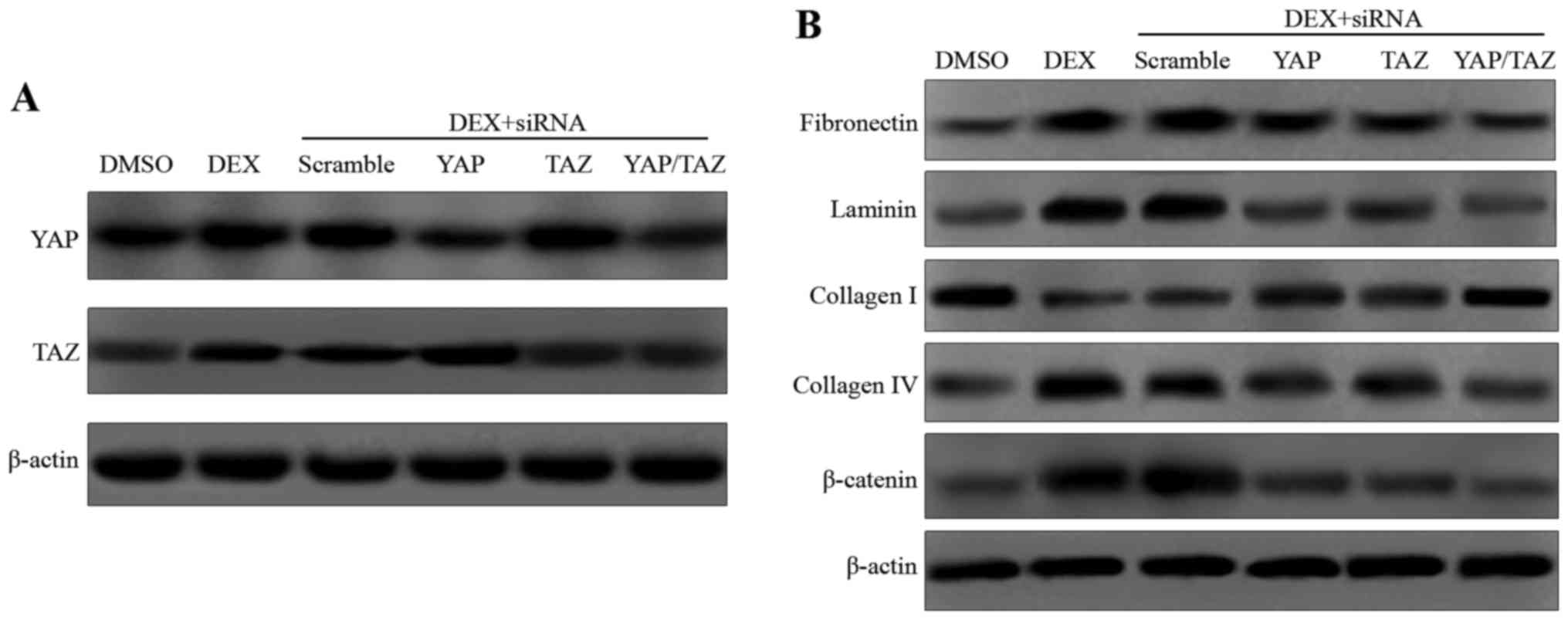
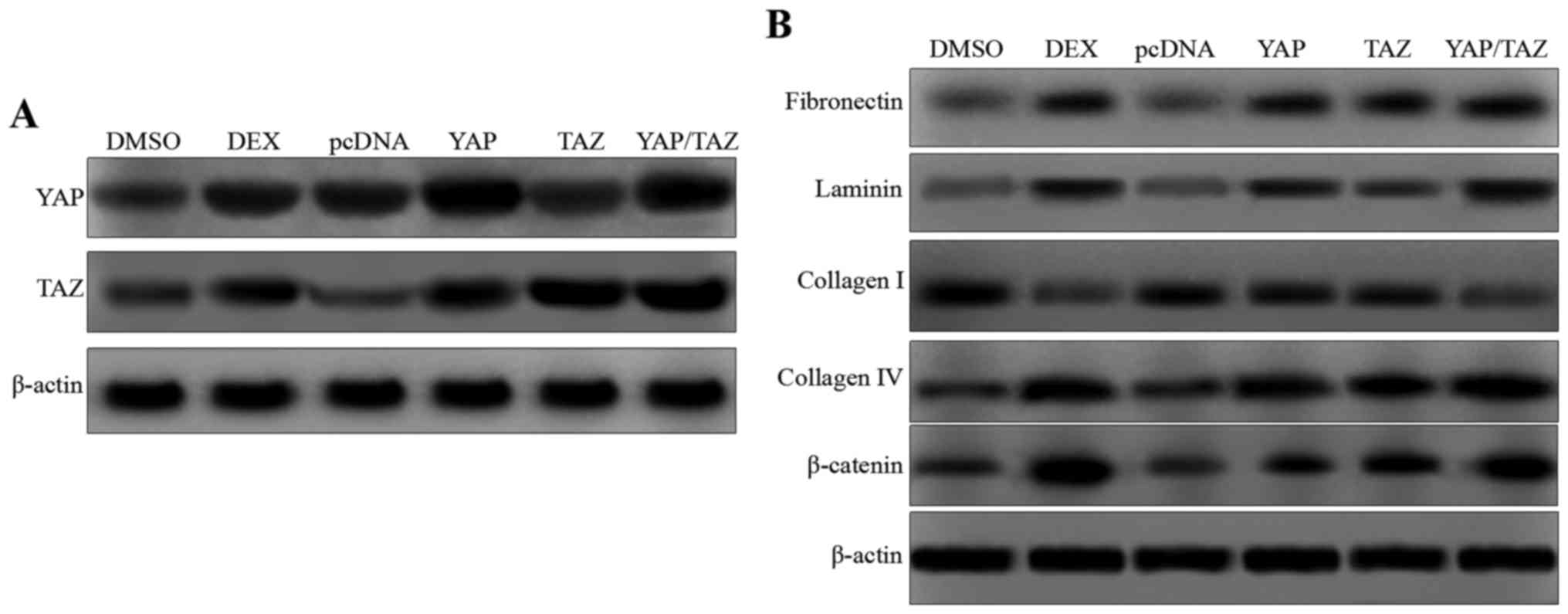
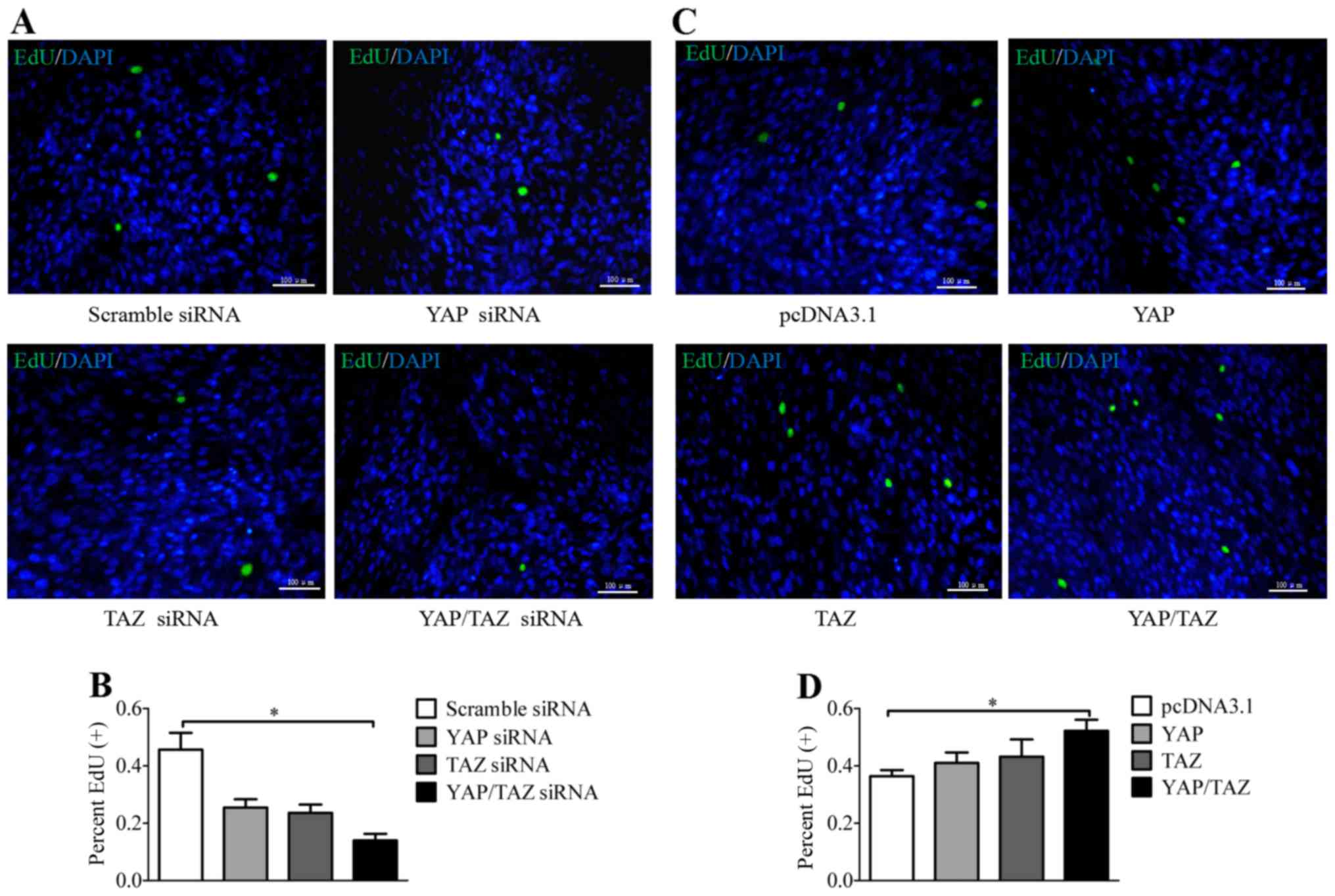
|
1
|
Quigley HA and Broman AT: The number of
people with glaucoma worldwide in 2010–2020. Br J Ophthalmol.
90:262–267. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Gordon MO, Beiser JA, Brandt JD, Heuer DK,
Higginbotham EJ, Johnson CA, Keltner JL, Miller JP, Parrish RK 2nd,
Wilson MR, et al: The Ocular Hypertension Treatment Study: baseline
factors that predict the onset of primary open-angle glaucoma. Arch
Ophthalmol. 120:714–720. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Johnson M: 'What controls aqueous humour
outflow resistance?'. Exp Eye Res. 82:545–557. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Gottanka J, Johnson DH, Martus P and
Lütjen-Drecoll E: Severity of optic nerve damage in eyes with POAG
is correlated with changes in the trabecular meshwork. J Glaucoma.
6:123–132. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Last JA, Pan T, Ding Y, Reilly CM, Keller
K, Acott TS, Fautsch MP, Murphy CJ and Russell P: Elastic modulus
determination of normal and glaucomatous human trabecular meshwork.
Invest Ophthalmol Vis Sci. 52:2147–2152. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kersey JP and Broadway DC:
Corticosteroid-induced glaucoma: a review of the literature. Eye
(Lond). 20:407–416. 2006. View Article : Google Scholar
|
|
7
|
Stokes J, Walker BR, Campbell JC, Seckl
JR, O'Brien C and Andrew R: Altered peripheral sensitivity to
glucocorticoids in primary open-angle glaucoma. Invest Ophthalmol
Vis Sci. 44:5163–5167. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhuo YH, He Y, Leung KW, Hou F, Li YQ,
Chai F and Ge J: Dexamethasone disrupts intercellular junction
formation and cytoskeleton organization in human trabecular
meshwork cells. Mol Vis. 16:61–71. 2010.PubMed/NCBI
|
|
9
|
Knepper PA, Collins JA and Frederick R:
Effects of dexamethasone, progesterone, and testosterone on IOP and
GAGs in the rabbit eye. Invest Ophthalmol Vis Sci. 26:1093–1100.
1985.PubMed/NCBI
|
|
10
|
Clark AF, Brotchie D, Read AT, Hellberg P,
English-Wright S, Pang IH, Ethier CR and Grierson I: Dexamethasone
alters F-actin architecture and promotes cross-linked actin network
formation in human trabecular meshwork tissue. Cell Motil
Cytoskeleton. 60:83–95. 2005. View
Article : Google Scholar
|
|
11
|
Clark AF, Wilson K, McCartney MD, Miggans
ST, Kunkle M and Howe W: Glucocorticoid-induced formation of
cross-linked actin networks in cultured human trabecular meshwork
cells. Invest Ophthalmol Vis Sci. 35:281–294. 1994.PubMed/NCBI
|
|
12
|
Kango-Singh M and Singh A: Regulation of
organ size: insights from the Drosophila Hippo signaling pathway.
Dev Dyn. 238:1627–1637. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Saucedo LJ and Edgar BA: Filling out the
Hippo pathway. Nat Rev Mol Cell Biol. 8:613–621. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Buttitta LA and Edgar BA: How size is
controlled: from Hippos to Yorkies. Nat Cell Biol. 9:1225–1227.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Pan D: Hippo signaling in organ size
control. Genes Dev. 21:886–897. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhao B, Lei QY and Guan KL: The Hippo-YAP
pathway: new connections between regulation of organ size and
cancer. Curr Opin Cell Biol. 20:638–646. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yu FX and Guan KL: The Hippo pathway:
regulators and regulations. Genes Dev. 27:355–371. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wang K, Degerny C, Xu M and Yang XJ: YAP,
TAZ, and Yorkie: a conserved family of signal-responsive
transcriptional coregulators in animal development and human
disease. Biochem Cell Biol. 87:77–91. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Raghunathan VK, Morgan JT, Dreier B,
Reilly CM, Thomasy SM, Wood JA, Ly I, Tuyen BC, Hughbanks M, Murphy
CJ, et al: Role of substratum stiffness in modulating genes
associated with extracellular matrix and mechanotransducers YAP and
TAZ. Invest Ophthalmol Vis Sci. 54:378–386. 2013. View Article : Google Scholar :
|
|
20
|
Dupont S, Morsut L, Aragona M, Enzo E,
Giulitti S, Cordenonsi M, Zanconato F, Le Digabel J, Forcato M,
Bicciato S, et al: Role of YAP/TAZ in mechanotransduction. Nature.
474:179–183. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Li C, Chen J, Lu B, Shi Z, Wang H, Zhang
B, Zhao K, Qi W, Bao J and Wang Y: Molecular switch role of Akt in
Polygonatum odoratum lectin-induced apoptosis and autophagy in
human non-small cell lung cancer A549 cells. PLoS One.
9:e1015262014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Huang G, Lv J, Li T, Huai G, Li X, Xiang
S, Wang L, Qin Z, Pang J, Zou B, et al: Notoginsenoside R1
ameliorates podocyte injury in rats with diabetic nephropathy by
activating the I3K/Akt signaling pathway. Int J Mol Med.
38:1179–1189. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wang Y, Liu Y, Wang H, Li C, Qi P and Bao
J: Agaricus bisporus lectins mediates islet β-cell proliferation
through regulation of cell cycle proteins. Exp Biol Med (Maywood).
237:287–296. 2012. View Article : Google Scholar
|
|
24
|
Wang Y, Wang H, Liu Y, Li C, Qi P and Bao
J: Antihyperglycemic effect of ginsenoside Rh2 by inducing islet
β-cell regeneration in mice. Horm Metab Res. 44:33–40. 2012.
View Article : Google Scholar
|
|
25
|
Schmittgen TD and Livak KJ: Analyzing
real-time PCR data by the comparative C(T) method. Nat Protoc.
3:1101–1108. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Perkins TW, Alvarado JA, Polansky JR,
Stilwell L, Maglio M and Juster R: Trabecular meshwork cells grown
on filters. Conductivity and cytochalasin effects. Invest
Ophthalmol Vis Sci. 29:1836–1846. 1988.PubMed/NCBI
|
|
27
|
Thomasy SM, Morgan JT, Wood JA, Murphy CJ
and Russell P: Substratum stiffness and latrunculin B modulate the
gene expression of the mechanotransducers YAP and TAZ in human
trabecular meshwork cells. Exp Eye Res. 113:66–73. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Rozsa FW, Reed DM, Scott KM, Pawar H,
Moroi SE, Kijek TG, Krafchak CM, Othman MI, Vollrath D, Elner VM,
et al: Gene expression profile of human trabecular meshwork cells
in response to long-term dexamethasone exposure. Mol Vis.
12:125–141. 2006.PubMed/NCBI
|
|
29
|
Steely HT, Browder SL, Julian MB, Miggans
ST, Wilson KL and Clark AF: The effects of dexamethasone on
fibronectin expression in cultured human trabecular meshwork cells.
Invest Ophthalmol Vis Sci. 33:2242–2250. 1992.PubMed/NCBI
|
|
30
|
Filla MS, Clark R and Peters DM: A
syndecan-4 binding peptide derived from laminin 5 uses a novel PKCε
pathway to induce cross-linked actin network (CLAN) formation in
human trabecular meshwork (HTM) cells. Exp Cell Res. 327:171–182.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Clark AF, Miggans ST, Wilson K, Browder S
and McCartney MD: Cytoskeletal changes in cultured human glaucoma
trabecular meshwork cells. J Glaucoma. 4:183–188. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zhou L, Li Y and Yue BY: Glucocorticoid
effects on extracellular matrix proteins and integrins in bovine
trabecular meshwork cells in relation to glaucoma. Int J Mol Med.
1:339–346. 1998.PubMed/NCBI
|
|
33
|
Peng J, Feng XY, Ye ZM, Luo Q, Cheng YL,
Wu ZZ, Lei CT and Gong B: Effects of dexamethasone and HA1077 on
actin cytoskeleton and β-catenin in cultured human trabecular
meshwork cells. Int J Ophthalmol. 9:1376–1380. 2016.
|
|
34
|
Lange AW, Sridharan A, Xu Y, Stripp BR,
Perl AK and Whitsett JA: Hippo/Yap signaling controls epithelial
progenitor cell proliferation and differentiation in the embryonic
and adult lung. J Mol Cell Biol. 7:35–47. 2015. View Article : Google Scholar :
|
|
35
|
Morgan JT, Murphy CJ and Russell P: What
do mechanotransduction, Hippo, Wnt, and TGFβ have in common? YAP
and TAZ as key orchestrating molecules in ocular health and
disease. Exp Eye Res. 115:1–12. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Filla MS, Schwinn MK, Sheibani N, Kaufman
PL and Peters DM: Regulation of cross-linked actin network (CLAN)
formation in human trabecular meshwork (HTM) cells by convergence
of distinct beta1 and beta3 integrin pathways. Invest Ophthalmol
Vis Sci. 50:5723–5731. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Rao PV, Deng PF, Kumar J and Epstein DL:
Modulation of aqueous humor outflow facility by the Rho
kinase-specific inhibitor Y-27632. Invest Ophthalmol Vis Sci.
42:1029–1037. 2001.PubMed/NCBI
|
|
38
|
Sit AJ and Liu JH: Pathophysiology of
glaucoma and continuous measurements of intraocular pressure. Mol
Cell Biomech. 6:57–69. 2009.PubMed/NCBI
|
|
39
|
McKee CT, Wood JA, Shah NM, Fischer ME,
Reilly CM, Murphy CJ and Russell P: The effect of biophysical
attributes of the ocular trabecular meshwork associated with
glaucoma on the cell response to therapeutic agents. Biomaterials.
32:2417–2423. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Wood JA, McKee CT, Thomasy SM, Fischer ME,
Shah NM, Murphy CJ and Russell P: Substratum compliance regulates
human trabecular meshwork cell behaviors and response to
latrunculin B. Invest Ophthalmol Vis Sci. 52:9298–9303. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Wood JA, Shah NM, McKee CT, Hughbanks ML,
Liliensiek SJ, Russell P and Murphy CJ: The role of substratum
compliance of hydrogels on vascular endothelial cell behavior.
Biomaterials. 32:5056–5064. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Chew SY and Low WC: Scaffold-based
approach to direct stem cell neural and cardiovascular
differentiation: an analysis of physical and biochemical effects. J
Biomed Mater Res A. 97:355–374. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Han H, Wecker T, Grehn F and Schlunck G:
Elasticity-dependent modulation of TGF-β responses in human
trabecular meshwork cells. Invest Ophthalmol Vis Sci. 52:2889–2896.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Zhao B, Li L, Lei Q and Guan KL: The
Hippo-YAP pathway in organ size control and tumorigenesis: an
updated version. Genes Dev. 24:862–874. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Wordinger RJ and Clark AF: Effects of
glucocorticoids on the trabecular meshwork: towards a better
understanding of glaucoma. Prog Retin Eye Res. 18:629–667. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Morrison DK: The 14-3-3 proteins:
integrators of diverse signaling cues that impact cell fate and
cancer development. Trends Cell Biol. 19:16–23. 2009. View Article : Google Scholar
|
|
47
|
Junglas B, Kuespert S, Seleem AA, Struller
T, Ullmann S, Bösl M, Bosserhoff A, Köstler J, Wagner R, Tamm ER,
et al: Connective tissue growth factor causes glaucoma by modifying
the actin cytoskeleton of the trabecular meshwork. Am J Pathol.
180:2386–2403. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Tovar-Vidales T, Roque R, Clark AF and
Wordinger RJ: Tissue transglutaminase expression and activity in
normal and glaucomatous human trabecular meshwork cells and
tissues. Invest Ophthalmol Vis Sci. 49:622–628. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Mao W, Millar JC, Wang WH, Silverman SM,
Liu Y, Wordinger RJ, Rubin JS, Pang IH and Clark AF: Existence of
the canonical Wnt signaling pathway in the human trabecular
meshwork. Invest Ophthalmol Vis Sci. 53:7043–7051. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Varelas X, Miller BW, Sopko R, Song S,
Gregorieff A, Fellouse FA, Sakuma R, Pawson T, Hunziker W, McNeill
H, et al: The Hippo pathway regulates Wnt/beta-catenin signaling.
Dev Cell. 18:579–591. 2010. View Article : Google Scholar : PubMed/NCBI
|