Introduction
Thyroid cancer incidence is rapidly increasing in
the USA, and its estimated annual diagnosis and mortality rates in
2017 were 56,870 and 2,010 cases, respectively. Thyroid cancer is
typically classified as papillary, follicular and anaplastic
carcinoma (1). Amongst these
classifications, anaplastic thyroid cancer (ATC) accounts for 1–2%
of all thyroid tumors. ATC is characterized by aggressive and local
invasion, and common distant metastasis (2). Currently, the available therapies
for ATC include chemotherapy, radiotherapy and surgery; however,
effective targeted treatments have yet to be developed. ATC remains
one of the most fatal types of cancer, with a mean survival time of
6 months (3). Therefore, an
enhanced understanding of the molecular mechanisms underlying the
carcinogenesis and progression of ATC would contribute to the
development of novel diagnostic markers and therapeutic
targets.
Gambogic acid (GA), the main active ingredient of
gamboge, is a brownish orange dry resin secreted from the plant
Garcinia hanburyi that is widely distributed in nature. GA
has been reported to inhibit the growth of different types of
cancer, including lung, colorectal, prostate and breast cancer,
hepatocellular carcinoma, multiple myeloma and leukemia (4–10).
The possible mechanisms underlying the antitumor effect of GA are
associated with the induction of apoptosis, inhibition of
telomerase, interruption of nuclear factor-κB signaling pathway and
enhancement of reactive oxygen species accumulation (5,9,11).
However, the potential effect and underlying molecular mechanisms
of GA in ATC remain poorly understood.
Bromodomain-containing protein 4 (BRD4) is an
epigenome reader and a member of the bromodomain and extra-terminal
domain (BET) family of proteins, which are composed of two
bromodomains in tandem and an extra-terminal domain. BRD4 has been
reported to promote cell cycle progression and to regulate cell
growth and transcription (12).
Furthermore, it participates in tumor growth and proliferation in
various tumors, including in lymphoblastic leukemia, glioblastoma,
neuroblastoma, malignant peripheral nerve sheath tumors, melanoma,
lung adenocarcinoma and papillary thyroid cancer (13–21). However, the role of BRD4 in ATC
has yet to be described in detail.
In the present study, the antiproliferative effect
of GA in ATC cells by inducing cell apoptosis was initially
confirmed. In addition, it was demonstrated that BRD4 was a
potential target of GA, and that BRD4 silencing suppressed tumor
growth in vitro and in vivo. Furthermore, in
vitro and in vivo experiments indicated the critical
role of BRD4 in the antiproliferative effects of GA on ATC
cells.
Materials and methods
Cell culture and tissue collection
The human normal thyroid Nthy-ori 3-1 cell line and
two ATC cell lines (SW1736 and KAT-18) were obtained from the
American Type Culture Collection (Manassas, VA, USA). The cell
lines were authenticated using short-tandem repeat profiling
performing by BMR Genomics (Padova, Italia). The cells were
maintained in Dulbecco's modified Eagle's medium (HyClone; GE
Healthcare Life Sciences, Beijing, China) with 10% fetal bovine
serum (HyClone; GE Healthcare Life Sciences) in a humidified
atmosphere with 5% CO2 and 95% air at 37°C. For the Cell
Counting Kit-8 (CCK-8) assay, SW1736 and KAT-18 cells were cultured
with increasing doses of GA (Key Laboratory of Carcinogenesis and
Intervention, China Pharmaceutical University, Nanjing, China; 0,
2, 4 and 6 µg/ml) for 48 h or treated with 2 µg/ml GA
for the indicated time points (0, 24, 48 and 72 h) at 37°C. For the
colony formation assay and flow cytometry, SW1736 and KAT-18 cells
were cultured with increasing doses of GA (0, 2, 4 and 6
µg/ml) for 48 h at 37°C. To discover the association between
BRD4 and GA, ATC cells were then divided into four groups, treated
with BRD4 overexpression plasmid with dimethyl sulfoxide (DMSO),
BRD4 overexpression plasmid with 2 µg/ml GA, control vector
with DMSO, and control vector with 2 µg/ml GA.
Furthermore, human ATC specimens and their adjacent
normal thyroid tissues (10 pairs) were collected from 10 patients
(3 males and 7 females; age range, 51–70 years; median age, 62) who
underwent surgery at the Weifang People's Hospital (Weifang, China)
between January 2017 and October 2017. All ATC specimens were
confirmed by pathological diagnosis according to the World Health
Organization criteria (22). The
present study was approved by the Ethics Committee of Weifang
People's Hospital (Weifang, China). Written informed consent was
obtained from each patient who participated in this study.
Cell transfection
shRNA plasmids for BRD4, which were designed against
the BRD4 gene and constructed in Phblv-u6-puro vectors, were
purchased from Hanbio Biotechnology Co., Ltd. (Shanghai, China). A
non-target scrambled oligonucleotide functioned as the negative
control. To generate stable BRD4 knockdown cells, SW1736 and KAT-18
cells were grown in six-well plates until they reached 40%
confluence. The medium was replaced with 1 ml Dulbecco's modified
Eagle's medium (HyClone; GE Healthcare Life Sciences, Beijing,
China) supplemented with 100 µl viral supernatant
(1×108 UT/ml) and 8 µg/ml polybrene (Hanbio
Biotechnology Co., Ltd.) for 24 h. The SW1736 and KAT-18 cells were
further cultured in Dulbecco's modified Eagle's medium (HyClone; GE
Healthcare Life Sciences, Beijing, China) with 10% fetal bovine
serum (HyClone; GE Healthcare Life Sciences) containing puromycin
(Hanbio Biotechnology Co., Ltd.) at 5 and 3 µg/ml at 37°C
for three passages, respectively. Individual puromycin-resistant
colonies were isolated during drug screening. The shRNA sequences
used in the present study were as follows: shBRD4,
5′-GatccGCCTGGAGATGACATAGTCTTATTCAAGAGATAAGACTATGTCATCTCCAGGTTTTTTc-3′;
and shControl,
5′-GatccTTCTCCGAACGTGTCACGTAATTCAAGAGATTACGTGACACGTTCGGAGAATTTTTTg-3′.
A non-target scrambled oligonucleotide functioned as the negative
control and presented no homology to any human transcripts.
Furthermore, BRD4 overexpression was examined using BRD4
overexpression plasmid and control vectors that were obtained from
Santa Cruz Biotechnology, Inc. (Dallas, TX, USA). Cell transfection
was conducted according to the manufacturer's protocol.
Cell viability assay
Cell viability was examined using a CCK-8 assay.
Briefly, SW1736 and KAT-18 cells were plated into a 96-well plate
(3,000 cells/well). After 0, 24, 48 and 72 h of incubation, 10
µl CCK-8 solution (Beyotime Institute of Biotechnology,
Haimen, China) was added into each well at 37°C. Following 3 h of
culturing, the absorbance of each well was determined using a
Multiskan MK3 device (Thermo Fisher Scientific, Inc., Waltham, MA,
USA) at a wavelength of 450 nm.
Colony formation assay
SW1736 and KAT-18 cells were cultured in a 6-well
plate at a density of 300 cells/well. After 7 days, the cells were
washed with phosphate-buffered saline (PBS), fixed with
paraformaldehyde and stained with crystal violet. Subsequently, the
number of the colonies that had migrated through the pores was
quantified by randomly counting 10 independent visual fields using
the images.
Cell apoptosis analysis
Cell apoptosis was assessed using flow cytometry
with staining of the cells using an Annexin V/propidium iodide (PI)
kit (Nanjing KeyGen Biotech Co, Ltd., Nanjing, China) according to
the manufacturer's protocol. Briefly, cells seeded in 24-well
plates were exposed to 0, 2, 4 and 6 µg/ml of GA for 48 h,
then cells were collected and washed twice in ice cold PBS. The
washed cells (2×105/well) were resuspended in 100
µl binding buffer (included in the kit) and stained with 5
µl Annexin V and 5 µl PI for 15 min at room
temperature. Following incubation for 15 min in the dark at room
temperature, flow cytometry was performed. A flow cytometer
(Cytomics FC 500 MPL; Beckman Coulter, Inc., Brea, CA, USA) was
utilized to evaluate the apoptotic levels in each sample according
to the manufacturer's protocol. Data were analyzed using ModFit LT
3.0 (Verity Software House, Inc., Topsham, ME, USA).
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Total RNA was extracted from tissues and cells by
using TRIzol reagent (Invitrogen; Thermo Fisher Scientific, Inc.)
according to the manufacturer's protocol. The concentration of
total RNA was determined by spectrophotometry (TECAN
Infinite® F200PRO microplate reader; Tecan Group, Ltd.,
Mannedorf, Switzerland). For mRNA expression analysis, RNA was
reverse transcribed into cDNA using a PrimeScript RT reagent kit
with gDNA Eraser (Takara Biotechnology Co., Ltd., Dalian, China).
qPCR was then conducted using SYBR Premix Ex Taq (Takara
Biotechnology Co., Ltd.). The qPCR conditions were applied for
detecting mRNAs: 95°C for 30 sec, followed by 40 cycles of 95°C for
30 sec, 60°C for 30 sec and 72°C for 30 sec using the following
primers: BRD4 forward, 5′-CATGGACATGAGCACAATCA-3′, and reverse,
5′-TCATGGTCAGGAGGGTTGTA-3′; β-actin forward,
5′-GATCATTGCTCCTCCTGAGC-3′, and reverse,
5′-ACTCCTGCTTGCTGATCCAC-3′. β-actin served as the internal control.
The specificity of amplification was examined using melting curve
analysis and electrophoresis in 1% agarose gel. The relative
expression level of the target gene mRNA was calculated as the
inverse log of ΔΔCq and normalized to the internal control
(23).
Western blot analysis
SW1736 and KAT-18 cells were lysed to extract the
total protein by using RIPA lysis buffer (Promega Corporation,
Madison, WI, USA) according the manufacturer's instructions.
Protein concentration was determined using a BCA Protein Assay kit
(Thermo Fisher Scientific, Inc.) according the manufacturer's
protocol. Next, the same amount of protein (40 µg) from each
cell line was subjected to 12% SDS-PAGE and transferred onto
polyvinylidene difluoride membranes. The membranes were then
respectively incubated with various diluted primary antibodies
against BRD4 (1:200; cat. no. ab75898; Abcam, Cambridge, MA, USA),
B-cell lymphoma 2 (Bcl-2; 1:500; cat. no. sc7382; Santa Cruz
Biotechnology, Inc.), Bcl-2-associated X protein (Bax; 1:500; cat.
no. sc7480; Santa Cruz Biotechnology, Inc.), cleaved caspase-3
(1:500; cat. no. #9661; Cell Signaling Technology, Inc., Beverly,
MA, USA), cleaved poly(ADP-ribose) polymerase (PARP; 1:500; cat.
no. #5625; Cell Signaling Technology, Inc.) or β-actin (1:800; cat.
no. AA128; Beyotime Institute of Biotechnology, Nantong, China).
Following incubation overnight at 4°C, horseradish
peroxidase-conjugated goat anti-rabbit secondary antibody (1:2,000;
cat. no. A0216; Beyotime Institute of Biotechnology) was added and
incubated at room temperature for 2 h. Specific bands were
visualized with an enhanced chemiluminescence reagent (KeyGen
Biotech Co., Ltd.) on an autoradiographic film. For quantitative
assay, images were analyzed using ImageJ software (version 1.48u;
National Institutes of Health, Bethesda, MD, USA).
Animal studies
All animal studies were approved by the Animal Care
and Welfare Committee of the Weifang People's Hospital and
conducted in strict accordance with the guidelines of the National
Animal Welfare Law of China. Female BALB/c nude mice (4-week-old;
n=36; female; weight range, 20–25 g) were purchased from the
Laboratory Animal Center of Yangzhou University (Yangzhou, China)
and maintained in a specific-pathogen-free environment with a
constant humidity (45–50%) and a constant temperature (25–27°C)
under a 12 h light/dark cycle with ad libitum access to food
and water. All mice were acclimatized for 1 week prior to the
experiments. In order to identify the function of BRD4 in
vivo, the mice (n=6/group) were subcutaneously injected into
the right flanks with 2×106 shControl or shBRD4 SW1736
cells. A total of 3 weeks later, the mice were euthanized with the
dislocation of cervical vertebra. In order to identify the
association between BRD4 and GA in vivo, the mice were
subcutaneously injected with 2×106 SW1736 cells
transfected with BRD4 plasmid (n=12/group) and control plasmid
(n=12/group). Following injection, the mice were divided into 4
groups (n=6/group) (BRD4 plasmid group injected with BRD4 plasmid +
4 mg/kg GA; control plasmid group injected with control plasmid + 4
mg/kg GA) and immediately received GA treatment or 0.9% saline
solution. The intravenous treatments were performed once every 3
days for a total of 21 days. The tumor volume (mm3) was
calculated using the following formula: Volume = 0.5 × length ×
width2. After 3 weeks, the mice were euthanized, and the
tumors were isolated, weighed, photographed and analyzed by
immunohistochemistry.
Immunohistochemistry
Paraffin-embedded archival specimens were cut into
sections 4-µm thick and baked at 65°C for 30 min. Sections
were then dewaxed in xylene and rehydrated via graded alcohol
solutions (100% ethanol, 5 min; 95% ethanol, 5 min; 70% ethanol, 5
min;), blocked in methanol containing 3% hydrogen peroxide for 10
min at room temperature, and then incubated with an BRD4 antibody
(1:200; cat. no. ab75898; Abcam) anti-Ki67 antibody (1:200; cat.
no. sc-23900; Santa Cruz Biotechnology, Inc.) at 4°C overnight.
Following rinsing with PBS solution, biotinylated goat anti-rabbit
serum IgG (1:2,000; cat. no. ab64256; Abcam) was used as a
secondary antibody for 1 h at room temperature and the streptavidin
peroxidase complex reagent was applied for 1 h at room temperature.
Finally, the sections were incubated in a 3,3′-diaminobenzidine
solution (DAB kit; Beyotime Institute of Biotechnology) at room
temperature for 10 min and then counterstained with hematoxylin for
3 min at room temperature. Ten randomly selected visual fields per
section were examined under a light microscope (magnification,
×200) to evaluate the BRD4 and Ki67 expression.
Statistical analysis
Data are expressed as the mean ± standard error.
Statistical analysis was performed using SPSS version 13.0 software
(SPSS, Inc., Chicago, IL, USA). Statistical significance was
determined through unpaired Student's t-test or one-way analysis of
variance. P<0.05 was considered as an indicator of a
statistically significant difference.
Results
Function of GA on the viability,
proliferation and apoptosis of ATC cells
To illustrate the function of GA on ATC cell
viability, a CCK-8 assay was conducted. SW1736 and KAT-18 cells
were cultured with increasing doses of GA (0, 2, 4 and 6
µg/ml) for 48 h. As shown in Fig. 1A, GA therapy for 48 h
significantly inhibited the ATC cell viability in a dose-dependent
manner. A CCK-8 assay was also performed on SW1736 and KAT-18 cells
treated with 2 µg/ml GA for the indicated time points (0,
24, 48 and 72 h). The results demonstrated that GA treatment
decreased the viability of the two ATC cell lines in a
time-dependent manner (Fig.
1B).
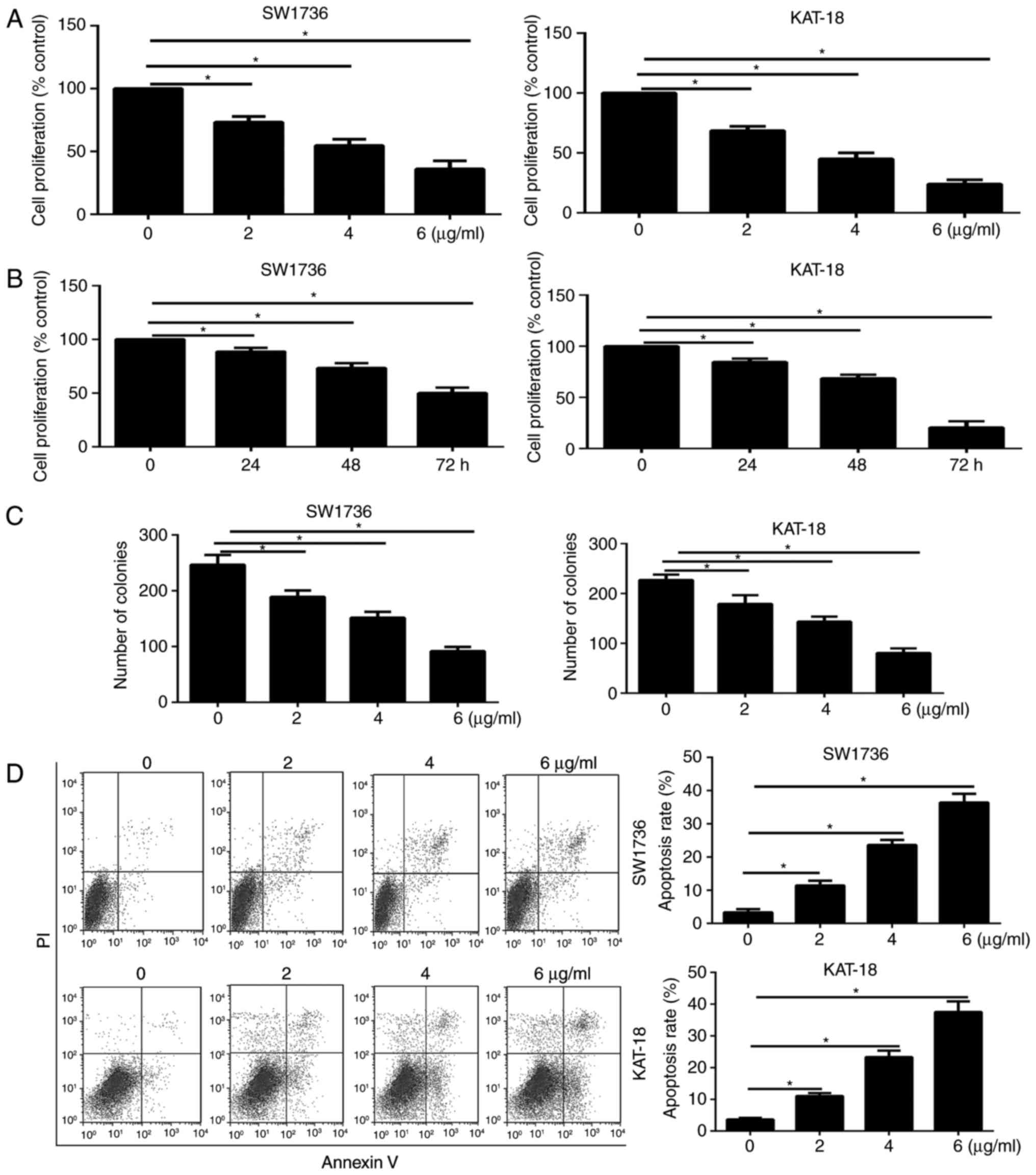 | Figure 1Effect of GA on the viability,
proliferation and apoptosis of ATC cells. (A) SW1736 and KAT-18
cells were treated with (A) DMSO or GA (2, 4 and 6 µg/ml)
for 48 h, and (B) 2 µg/ml GA for 0, 24, 48 and 72 h.
Following incubation, a cell counting kit-8 assay was performed to
determine cell viability. The relative cell viability was defined
as the percentage of cells treated with GA compared with the DMSO,
(C) GA inhibited the colony formation of ATC cells treated with
increasing doses of GA (0, 2, 4 and 6 µg/ml) for 7 days and
then stained with crystal violet. (D) GA increased the apoptosis
rate of ATC cells treated with increasing doses of GA (0, 2, 4 and
6 µg/ml) for 48 h. Cell apoptosis was evaluated by Annexin
V-FITC/propidium iodide staining, followed by flow cytometric
analysis. *P<0.05 vs. corresponding control group.
GA, gambogic acid; ATC, anaplastic thyroid cancer; DMSO, dimethyl
sulfoxide; OD, optical density. |
In order to investigate the function of GA on ATC
cell proliferation, a colony formation assay was then performed. As
shown in Fig. 1C, treatment of
ATC cells with GA at different concentrations for 7 days
significantly decreased the number of colonies in the cultures. The
function of GA on ATC cell apoptosis was also examined by flow
cytometry, and the results revealed that treatment of SW1736 and
KAT-18 cells with GA for 48 h significantly enhanced the apoptosis
rate (Fig. 1D) in a
dose-dependent manner.
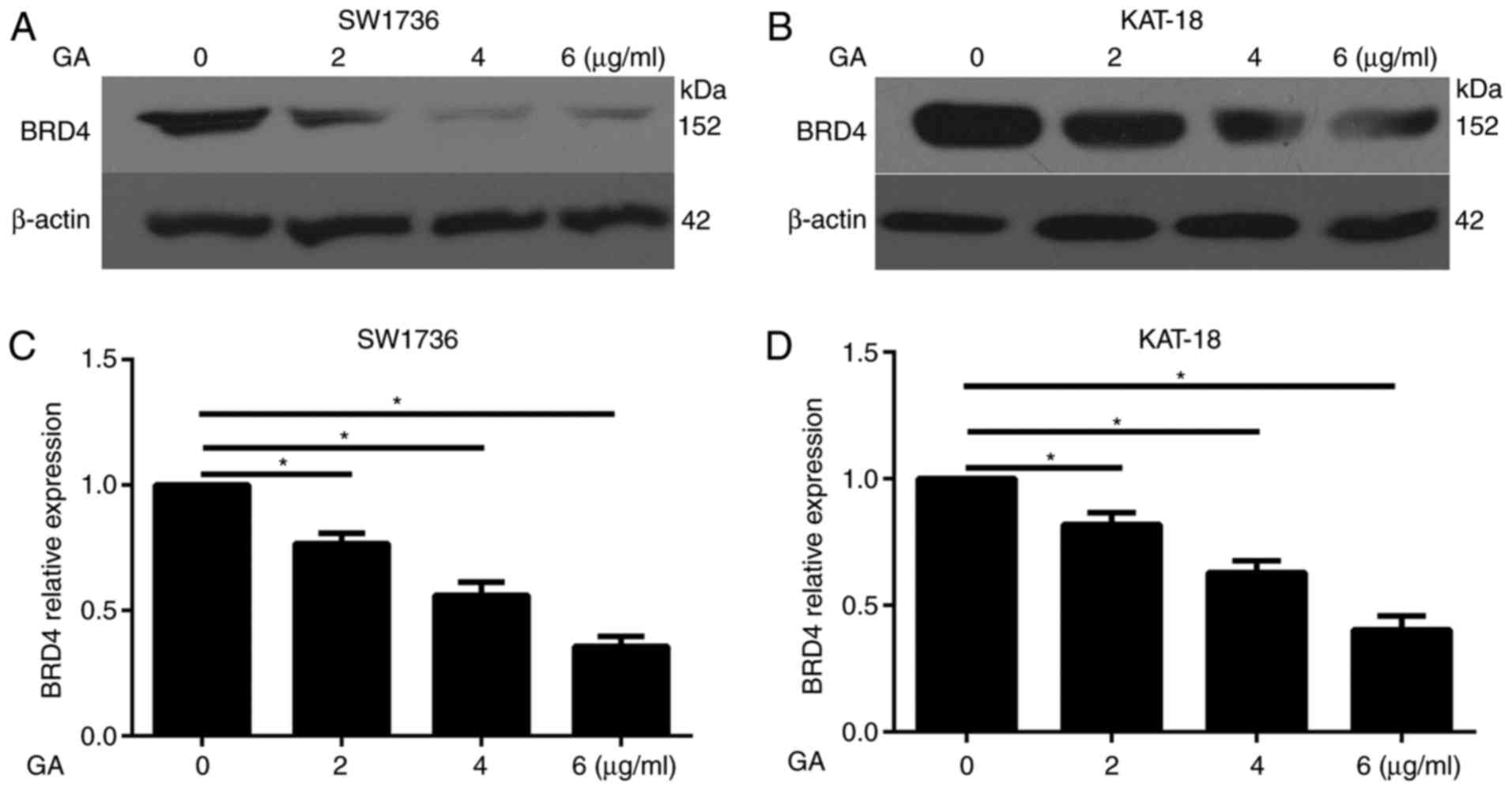
Expression of BRD4 is decreased by GA
treatment in ATC cells
Previous studies have demonstrated that BRD4 served
as an oncogene in in papillary thyroid cancer (24,25). In the present study, we attempted
to illustrate the association of GA and BRD4. The western blot
assay demonstrated that GA treatment (2, 4 and 6 µg/ml)
evidently decreased the protein expression of BRD4 in SW1736
(Fig. 2A) and KAT-18 (Fig. 2B) cells. Furthermore, the RT-qPCR
assay illustrated that GA treatment significantly decreased the
mRNA expression of BRD4 in the two cell lines in a dose-dependent
manner (Fig. 2C and D).
Therefore, BRD4 may serve a critical role in the antitumor effect
of GA in ATC.
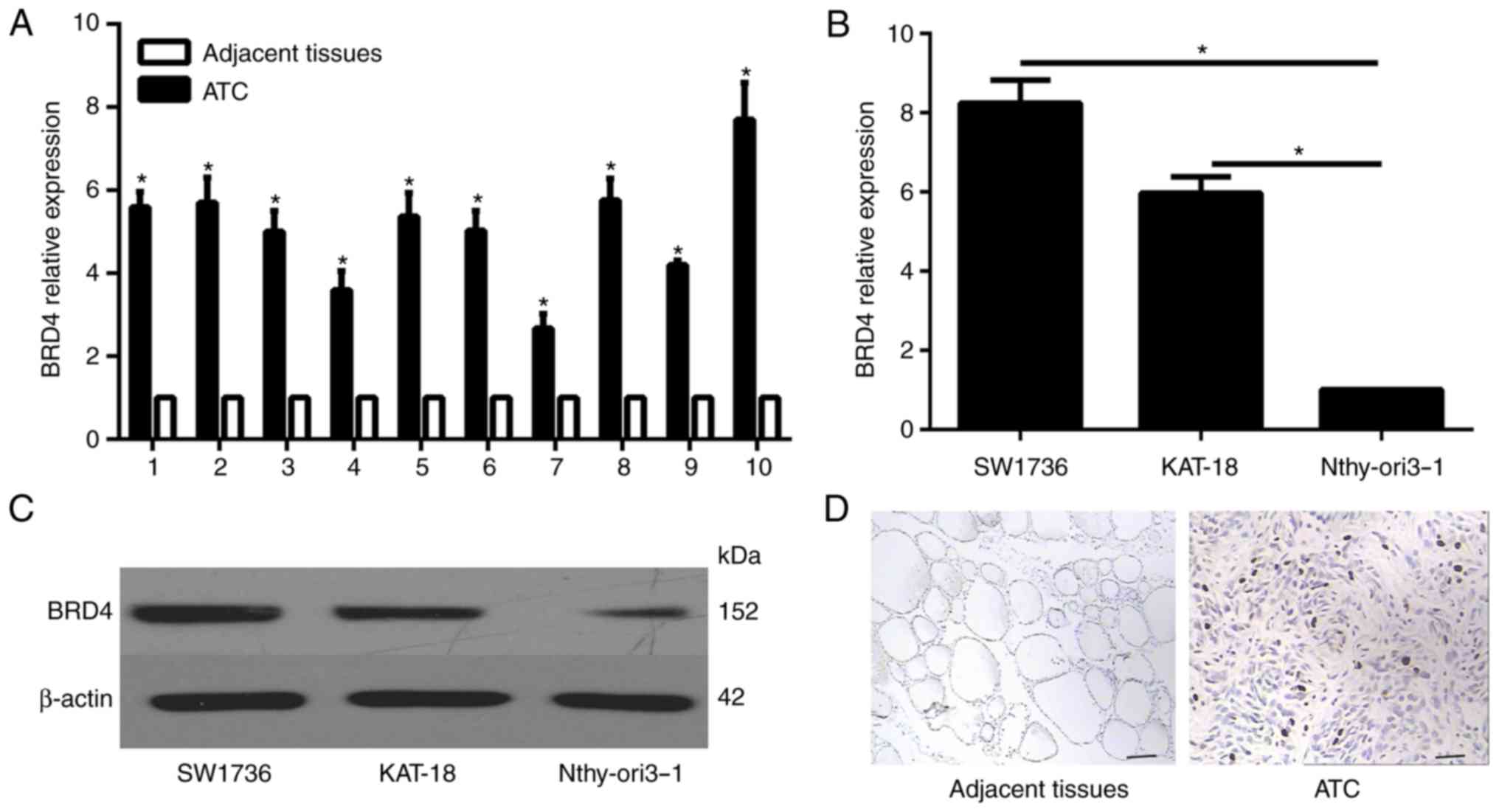
Increased BRD4 expression in ATC tissues
and cells
To determine the expression profile of BRD4 in ATC
tissues, RT-qPCR was performed in 10 ATC specimens and their
corresponding non-neoplastic thyroid tissues. Compared with the
non-neoplastic thyroid tissues, the expression level of BRD4 was
significantly enhanced in the ATC specimens (Fig 3A). Furthermore, the mRNA and
protein expression levels in normal thyroid and ATC cells were
examined by RT-qPCR and western blot analysis, respectively. The
results revealed that the mRNA and protein expression levels of
BRD4 were significantly lower in the human thyroid Nthy-ori 3-1
cell line in comparison with those in the ATC cell lines (Fig. 3B and C). In addition,
immunohistochemical analysis was conducted in the patient
specimens, and the results indicated that BRD4 was evidently
overexpressed in ATC tissues as compared with that in the adjacent
normal tissues (Fig. 3D).
BRD4 enhances the viability and
proliferation of ATC cells in vitro
BRD4 was silenced in the SW1736 and KAT-18 cells in
order to analyze the influence of this protein on the biological
behavior of ATC cells. Western blot analysis was used to determine
the expression level of BRD4, demonstrating that the shRNA
transfection successfully downregulated BRD4 in the cells (Fig. 4A and B). Subsequently, the
viability of the SW1736 and KAT-18 cells was analyzed via a CCK-8
assay, and BRD4 silencing was observed to significantly decrease
the viability of these cell lines (Fig. 4C and D). Furthermore, the colony
formation assay demonstrated that the BRD4 knockdown significantly
inhibited the proliferation of ATC cells (Fig. 4E and F), while the rate of
apoptosis in these cells was markedly enhanced (Fig. 4G and H). All these data revealed
that BRD4 is involved in the growth of ATC cells.
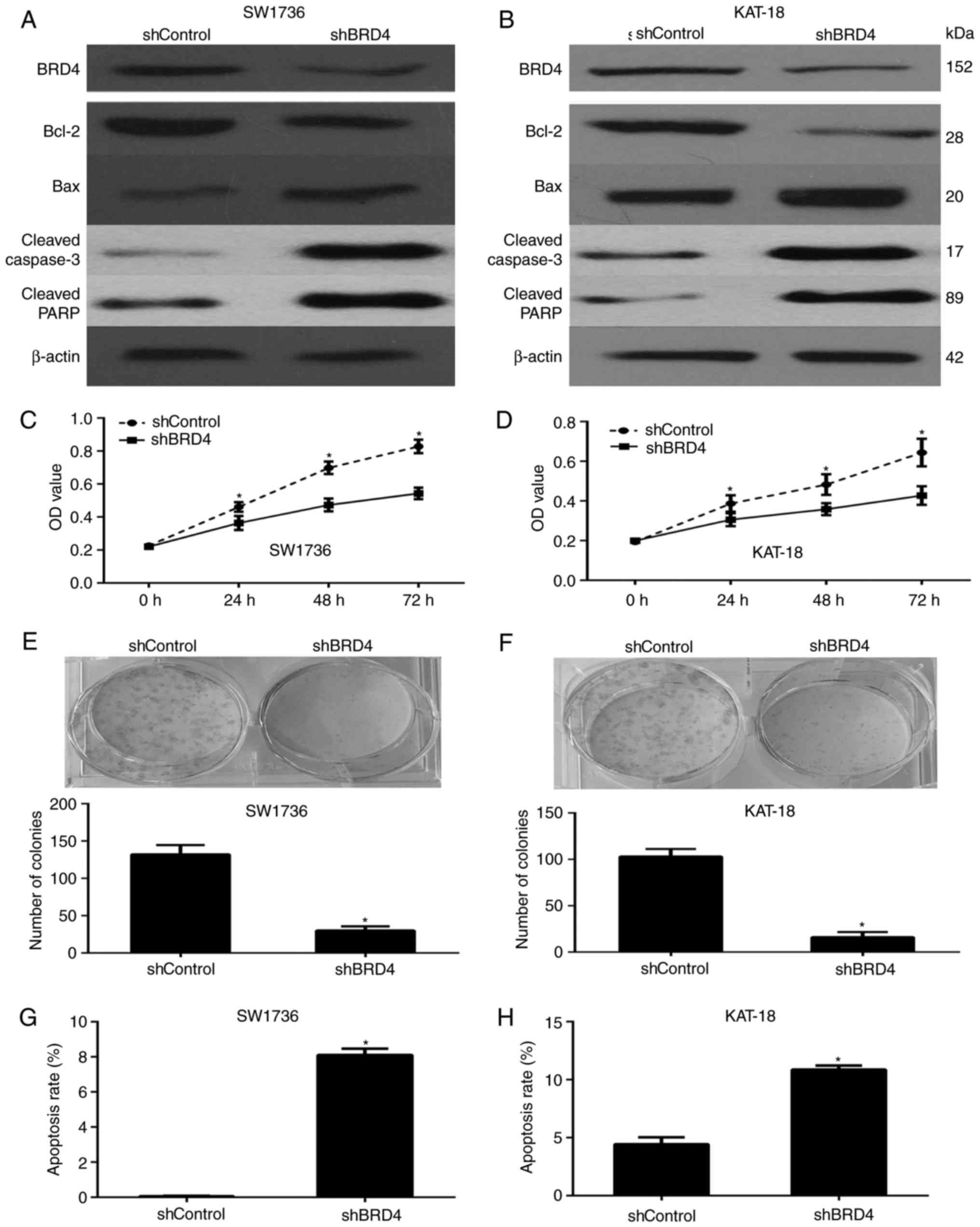 | Figure 4BRD4 expression enhances the
proliferation and decreases the apoptosis in ATC cells. Protein
expression levels of BRD4, Bcl-2, Bax, cleaved caspase-3 and
cleaved PARP in the shControl- or shBRD4-transfected (A) SW1736 and
(B) KAT-18 cells was determined via western blot analysis. The cell
viability of (C) SW1736 and (D) KAT-18 cells was examined via a
cell counting kit-8 assay, demonstrating that BRD4 silencing
significantly inhibited ATC cell viability. BRD4 silencing in (E)
SW1736 and (F) KAT-18 cells reduced the colony-forming efficiency.
Annexin V-FITC/propidium iodide staining in (G) SW1736 and (H)
KAT-18 cells transfected with shBRD4 demonstrated a higher
apoptosis rate, as assessed via flow cytometry.
*P<0.05. BRD4, bromodomain-containing protein 4;
Bcl-2, B-cell lymphoma 2; Bax, Bcl-2-associated X protein; PARP,
poly(ADP-ribose) polymerase; ATC, anaplastic thyroid cancer. |
Western blotting was also performed to analyze the
expression of apoptosis-associated proteins. In accordance with the
aforementioned functional experiments, BRD4 silencing significantly
decreased the expression of the anti-apoptotic factor Bcl-2, while
it enhanced the expression levels of the pro-apoptotic factor Bax,
cleaved caspase-3 and cleaved PARP in the SW1736 and KAT-18 cell
lines (Fig. 4A and B).
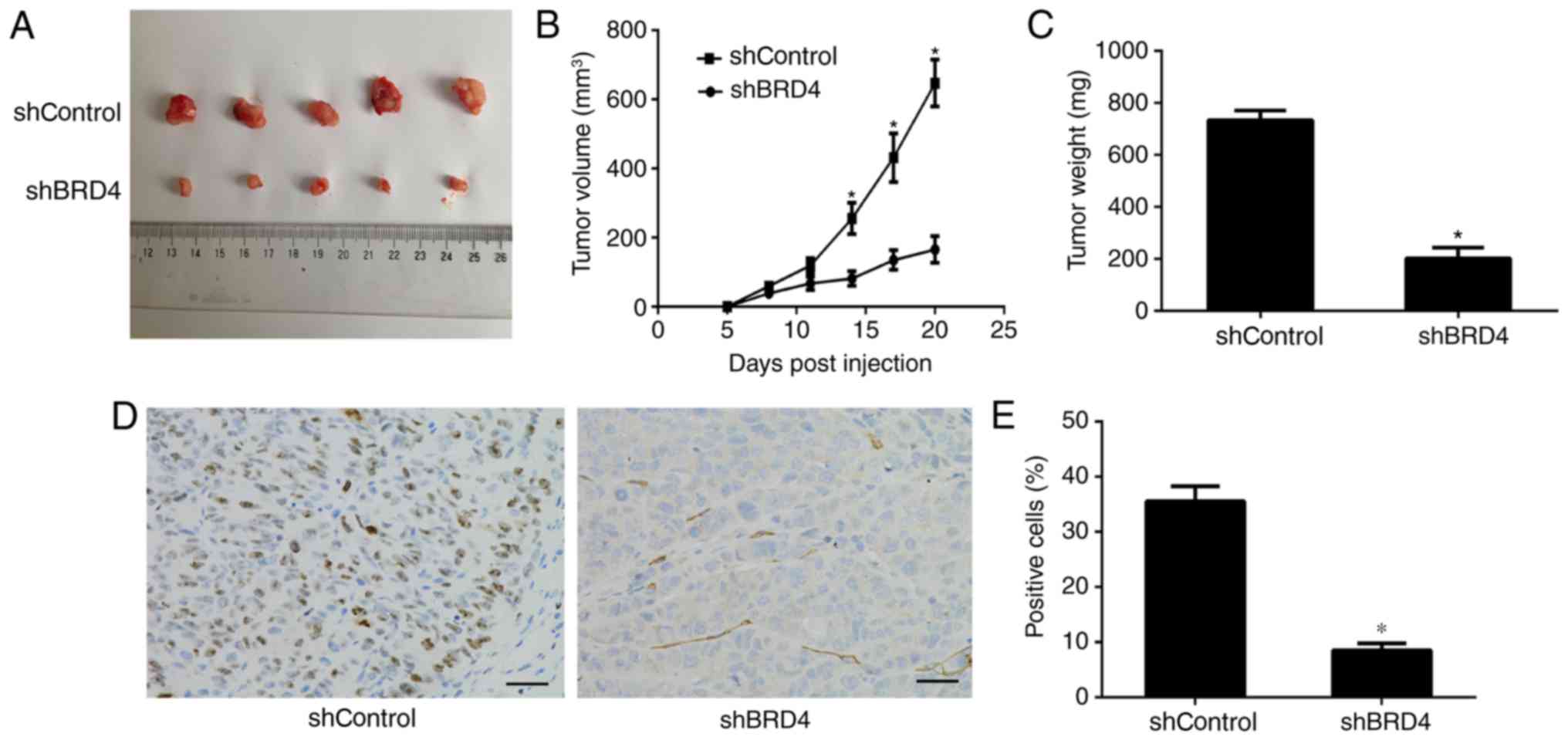
BRD4 is essential for anaplastic thyroid
cancer progression in vivo
To investigate the correlation of BRD4 expression
with the ATC progression in vivo, SW1736 cells transfected
with shBRD4 and shControl were subcutaneously injected into nude
mice (n=6/group). At 3 weeks after injection, the tumors were
removed, and images were captured (Fig. 5A). The tumor volume and weight of
the excised tumors indicated that BRD4 knockdown significantly
delayed the tumor growth (Fig. 5B
and C). Histological analysis of tumor proliferation also
demonstrated that shBRD4 tumors had significantly fewer Ki-67
positive cells compared with those in the shControl tumors
(Fig. 5D and E). These data
illustrated that BRD4 expression enhanced ATC growth in
vivo.
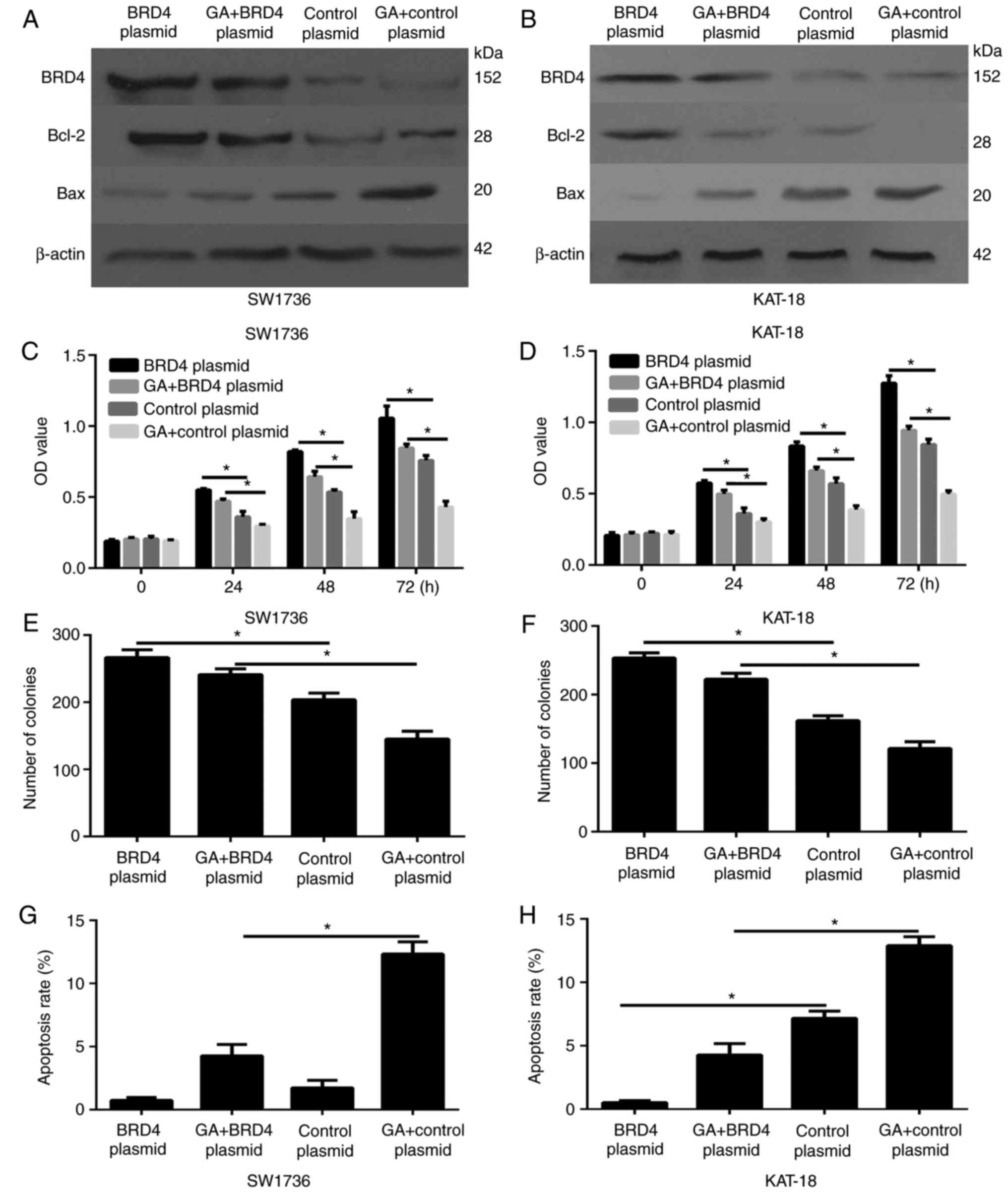
BRD4 is critical for the anti-cell growth
effects of GA on ATC cells in vitro
To further illustrated the correlation of GA and
BRD4, BRD4 expression was overexpressed by transfection with BRD4
overexpression plasmids. As shown in Fig. 6A and B, the BRD4 overexpression
plasmid notably increased the expression of this protein in ATC
cells compared with the control vector group.
ATC cells were then divided into four groups,
treated with BRD4 overexpression plasmid + DMSO, BRD4
overexpression plasmid + 2 µg/ml GA, control vector + DMSO,
and control vector + 2 µg/ml GA. The CCK-8 (Fig. 6C and D) and colony formation
(Fig. 6E and F) assays revealed
that overexpression of BRD4 was able to partially abrogate the
inhibitory effects of GA on ATC cells. Furthermore, overexpression
of BRD4 partially counteracted the suppression of cell apoptosis
(Fig. 6G and H). Furthermore, the
western blot analysis results demonstrated that BRD4 overexpression
rescued the expression of the antiapoptotic protein Bcl-2, and
counteracted the expression of pro-apoptosis gene Bax, which was
induced by GA (Fig. 6A and B).
Overexpression of BRD4 was demonstrated to counteract the effect of
GA, so BRD4 down-regulation is critical for the antitumor growth
effects of GA. These data manifested that GA inhibited the PTC
progression through the inhibition of BRD4.
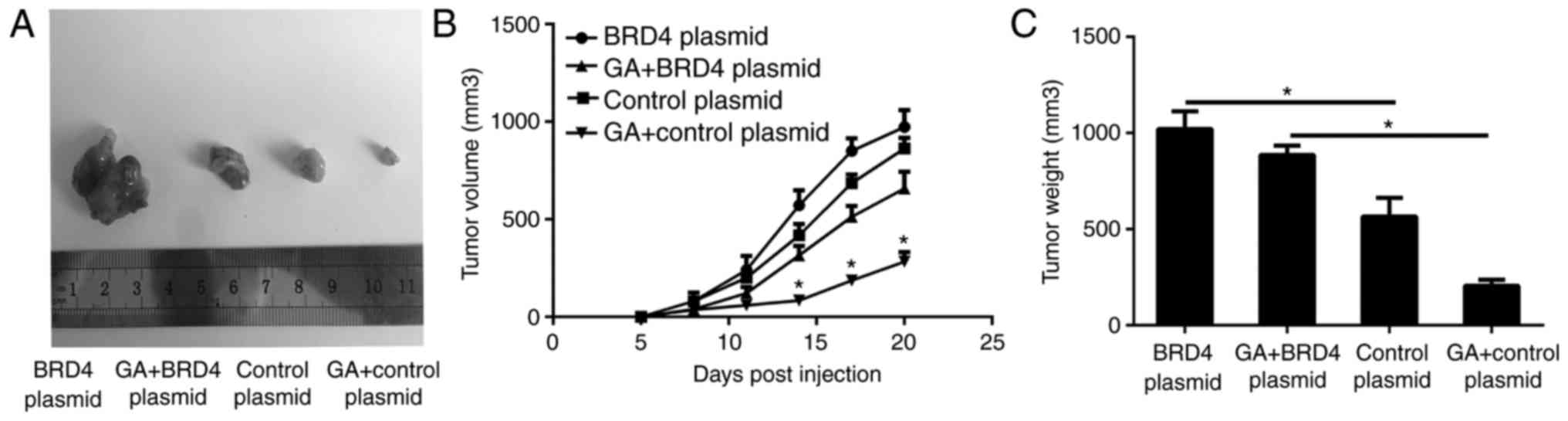
GA suppresses tumor growth through the
inhibition of BRD4 in vivo
Tumor xenografts of SW1736 cells transfected with
BRD4 overexpression and control vector were used to evaluate the
antitumor effect of GA (4 mg/kg) in nude mice in vivo. At 21
days after injection, the tumors were removed and images are shown
in Fig. 7A. The periodic
measurements of the tumor volume (Fig. 7B) and the final weight (Fig. 7C) of the excised tumors
demonstrated that BRD4 overexpression was able to significantly
abrogate the antitumor effects of GA on ATC cells.
Discussion
In vitro and in vivo studies have
demonstrated that GA exerts potent antitumor effects on solid
tumors and hematological malignancies, while may also induce
apoptosis in other types of cancer cells (4–10).
GA has been reported to promote apoptosis and resistance to
metastatic potential in triple negative breast cancer (5). Furthermore, GA may enhance the
efficiency of other chemical drugs in drug resistance tumour types
via different molecular mechanisms. Gambogic acid sensitized
resistant breast cancer cells to doxorubicin through inhibiting
P-glycoprotein and suppressing survivin expression (7). GA as a non-competitive inhibitor of
ATP-binding cassette transporter B1 reversed the multidrug
resistance of human epithelial cancer types by promoting
ATP-binding cassette transporter B1 protein degradation (8). GA sensitized ovarian cancer cells to
doxorubicin through reactive oxygen species-mediated apoptosis
(9). A combination of GA with
cisplatin enhanced the antitumor effects on cisplatin-resistant
lung cancer cells by downregulating multidrug resistance-associated
protein 2 and low density lipoprotein receptors expression
(10). In the present study, it
was observed that BRD4 expression was decreased by GA treatment.
Furthermore, BRD4 silencing enhanced the apoptosis rate and
decreased the proliferation of ATC cell lines.
Consistent with previous findings (5), the current study results confirmed
that GA treatment significantly decreased the viability and
proliferation of SW1736 and KAT-18 cells by increasing the cell
apoptosis rate (Fig. 1). Western
blot analysis and RT-qPCR results also revealed that the expression
level of BRD4 was significantly decreased following GA treatment
(Fig. 2). Furthermore, BRD4 was
significantly upregulated in ATC tissues as compared with that in
normal thyroid tissues (Fig. 3).
These data suggested that BRD4 may be involved in ATC pathogenesis
and may serve as a target for GA.
The epigenetic mechanism is a rapidly progressing
field in oncological research, and the modulation of epigenetic
regulators is considered as an alternative therapeutic strategy for
cancer (13). BRD4 is a member of
the BET family of proteins, which contain two bromodomains in
tandem and an extra-terminal domain (13,14). It has been reported that BRD4, as
a conserved epigenome regulator, regulates the expression of
numerous genes involved in cell growth, cell cycle progression and
inflammation in different types of cancer (13,14). The inhibition of BRD4 in papillary
thyroid cancer cells by JQ1 has been demonstrated to result in cell
cycle arrest at G0/G1 phase, enhance
131I uptake in vitro and suppress tumor growth
in vivo. JQ1 is also able to inhibit the proliferation of
ATC in vitro (24,25). However, the function of BRD4 in
ATC has yet to be fully clarified.
In the present study, silencing BRD4 in ATC cells
was observed to repress their viability, proliferation and
apoptosis (Fig. 4), whereas BRD4
overexpression caused the opposite effects on these cells (Fig. 6). Furthermore, in vivo
results demonstrated that BRD4 was involved in tumor growth
(Figs. 5 and 7) and thus may serve as an antitumor
target in ATC. Furthermore, the current study aimed to explore the
molecular mechanism by which BRD4 functions as an oncogene in ATC.
The results revealed that BRD4 silencing was associated with the
downregulation of the anti-apoptotic protein Bcl-2 and the
upregulation of the pro-apoptotic protein Bax. These data indicated
that BRD4 promoted tumor growth by inhibiting cell apoptosis.
In order to further investigate the association
between GA and BRD4, BRD4 expression was upregulated through
transfection with a BRD4 overexpression plasmid in SW1736 and
KAT-18 cells. The biological effects of GA on ATC cells were
significantly weakened by BRD4 overexpression (Fig. 6), suggesting that GA partially
exerted its anticancer functions by downregulating BRD4. The
apoptosis rate of ATC cells was significantly decreased by GA and
partially rescued by BRD4 overexpression. These results
demonstrated that GA regulated BRD4 and induced apoptosis.
In conclusion, the results of the present study
suggested that BRD4 serves an important role in ATC progression
in vitro and in vivo, and may function as a novel
target for the development of alternative therapeutic approaches
for cancer. Furthermore, BRD4 is essential for the
antiproliferative and pro-apoptotic effects of GA on ATC.
Acknowledgments
Not applicable.
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2017. CA Cancer J Clin. 67:7–30. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Nagaiah G, Hossain A, Mooney CJ,
Parmentier J and Remick SC: Anaplastic thyroid cancer: A review of
epidemiology, pathogenesis, and treatment. J Oncol.
2011:5423582011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
O'Neill JP and Shaha AR: Anaplastic
thyroid cancer. Oral Oncol. 49:702–706. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Qi Q, Lu N, Wang XT, Gu HY, Yang Y, Liu W,
Li C, You QD and Guo QL: Anti-invasive effect of gambogic acid in
MDA-MB-231 human breast carcinoma cells. Biochem Cell Biol.
86:386–395. 2008. View
Article : Google Scholar : PubMed/NCBI
|
|
5
|
Li C, Qi Q, Lu N, Dai Q, Li F, Wang X, You
Q and Guo Q: Gambogic acid promotes apoptosis and resistance to
metastatic potential in MDA-MB-231 human breast carcinoma cells.
Biochem Cell Biol. 90:718–730. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Li D, Song XY, Yue QX, Cui YJ, Liu M, Feng
LX, Wu WY, Jiang BH, Yang M, Qu XB, et al: Proteomic and
bioinformatic analyses of possible target-related proteins of
gambogic acid in human breast carcinoma MDA-MB-231 cells. Chin J
Nat Med. 13:41–51. 2015.PubMed/NCBI
|
|
7
|
Wang S, Wang L, Chen M and Wang Y:
Gambogic acid sensitizes resistant breast cancer cells to
doxorubicin through inhibiting P-glycoprotein and suppressing
survivin expression. Chem Biol Interact. 235:76–84. 2015.
View Article : Google Scholar
|
|
8
|
Wang X, Deng R, Lu Y, Xu Q, Yan M, Ye D
and Chen W: Gambogic acid as a non-competitive inhibitor of
ATP-binding cassette transporter B1 reverses the multidrug
resistance of human epithelial cancers by promoting ATP-binding
cassette transporter B1 protein degradation. Basic Clin Pharmacol
Toxicol. 112:25–33. 2013. View Article : Google Scholar
|
|
9
|
Wang J and Yuan Z: Gambogic acid
sensitizes ovarian cancer cells to doxorubicin through ROS-mediated
apoptosis. Cell Biochem Biophys. 67:199–206. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhang W, Zhou H, Yu Y, Li J, Li H, Jiang
D, Chen Z, Yang D, Xu Z and Yu Z: Combination of gambogic acid with
cisplatin enhances the antitumor effects on cisplatin-resistant
lung cancer cells by downregulating MRP2 and LRP expression. Onco
Targets Ther. 9:3359–3368. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Liu WY, Wu XU, Liao CQ, Shen J and Li J:
Apoptotic effect of gambogic acid in esophageal squamous cell
carcinoma cells via suppression of the NF-κB pathway. Oncol Lett.
11:3681–3685. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yang Z, He N and Zhou Q: Brd4 recruits
P-TEFb to chromosomes at late mitosis to promote G1 gene
expression and cell cycle progression. Mol Cell Biol. 28:967–976.
2008. View Article : Google Scholar
|
|
13
|
Wang YH, Sui YN, Yan K, Wang LS, Wang F
and Zhou JH: BRD4 promotes pancreatic ductal adenocarcinoma cell
proliferation and enhances gemcitabine resistance. Oncol Rep.
33:1699–1706. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wang YH, Sui XM, Sui YN, Zhu QW, Yan K,
Wang LS, Wang F and Zhou JH: BRD4 induces cell migration and
invasion in HCC cells through MMP-2 and MMP-9 activation mediated
by the Sonic hedgehog signaling pathway. Oncol Lett. 10:2227–2232.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Segura MF, Fontanals-Cirera B,
Gaziel-Sovran A, Guijarro MV, Hanniford D, Zhang G, González-Gomez
P, Morante M, Jubierre L, Zhang W, et al: BRD4 sustains melanoma
proliferation and represents a new target for epigenetic therapy.
Cancer Res. 73:6264–6276. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Puissant A, Frumm SM, Alexe G, Bassil CF,
Qi J, Chanthery YH, Nekritz EA, Zeid R, Gustafson WC, Greninger P,
et al: Targeting MYCN in neuroblastoma by BET bromodomain
inhibition. Cancer Discov. 3:308–323. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Cheng Z, Gong Y, Ma Y, Lu K, Lu X, Pierce
LA, Thompson RC, Muller S, Knapp S and Wang J: Inhibition of BET
bromodomain targets genetically diverse glioblastoma. Clin Cancer
Res. 19:1748–1759. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Patel AJ, Liao CP, Chen Z, Liu C, Wang Y
and Le LQ: BET bromodomain inhibition triggers apoptosis of
NF1-associated malignant peripheral nerve sheath tumors through Bim
induction. Cell Rep. 6:81–92. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ott CJ, Kopp N, Bird L, Paranal RM, Qi J,
Bowman T, Rodig SJ, Kung AL, Bradner JE and Weinstock DM: BET
bromodomain inhibition targets both c-Myc and IL7R in high-risk
acute lymphoblastic leukemia. Blood. 120:2843–2852. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lockwood WW, Zejnullahu K, Bradner JE and
Varmus H: Sensitivity of human lung adenocarcinoma cell lines to
targeted inhibition of BET epigenetic signaling proteins. Proc Natl
Acad Sci USA. 109:19408–19413. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Andrieu G, Tran AH, Strissel KJ and Denis
GV: BRD4 regulates breast cancer dissemination through
Jagged1/Notch1 signaling. Cancer Res. 76:6555–6567. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Molinaro E, Romei C, Biagini A, Sabini E,
Agate L, Mazzeo S, Materazzi G, Sellari-Franceschini S, Ribechini
A, Torregrossa L, et al: Anaplastic thyroid carcinoma: From
clinicopathology to genetics and advanced therapies. Nat Rev
Endocrinol. 13:644–660. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2−ΔΔC T method. Methods. 25:402–408.
2001. View Article : Google Scholar
|
|
24
|
Gao X, Wu X, Zhang X, Hua W and Zhang Y,
Maimaiti Y, Gao Z and Zhang Y: Inhibition of BRD4 suppresses tumor
growth and enhances iodine uptake in thyroid cancer. Biochem
Biophys Res Commun. 469:679–685. 2016. View Article : Google Scholar
|
|
25
|
Mio C, Lavarone E, Conzatti K, Baldan F,
Toffoletto B, Puppin C, Filetti S, Durante C, Russo D, Orlacchio A,
et al: MCM5 as a target of BET inhibitors in thyroid cancer cells.
Endocr Relat Cancer. 23:335–347. 2016. View Article : Google Scholar : PubMed/NCBI
|