Introduction
Stroke is a multi-factorial polygenic disease and a
major cause of death and adult disability (1). Administration of bone marrow stem
cells has been demonstrated to protect ischemic rat brain by
facilitating the recovery of neurological functions, reducing
lesion size, and improving functional outcomes (2). Transplanted mesenchymal stem cells
(MSCs) have also been demonstrated to promote the repair of damaged
brain tissue by differentiating into cells of neuronal or glial
lineage, regulating the immune response, and by the release of
trophic factors that stimulate endogenous repair processes, such as
neurogenesis, angiogenesis, and synaptogenesis (3). However, the application of MSCs can
be complicated by patient-specific factors, such as age (4). Stem cell function generally declines
with age, accompanied by reductions in cell number and in the
proliferation and differentiation potential of MSCs, with
subsequent impairments in their therapeutic effects in ischemic
diseases (5,6). Age is known to be the principal
non-modifiable risk factor for stroke (7), and it is therefore essential to
identify effective methods for rejuvenating senescent MSCs derived
from older donors, in order to improve their therapeutic efficacy
for patients with ischemic stroke.
Adiponectin (APN) is an adipocytokine with a
collagenous domain and a C-terminal globular domain, which is
predominantly secreted by adipose tissues (8). APN has been demonstrated to protect
against ischemic injury (9).
AmongC1q and tumor necrosis factor-related protein (CTRP) family
members, CTRP9 displays the highest amino acid identity to APN.
CRTP9 is also highly expressed in brain tissue (10), and acts as an adipocytokine with
beneficial effects on glucose metabolism, cellular survival,
oxidant response regulation, and inhibition of endoplasmic
reticulum stress, all of which are related to the cellular
senescence process (11–13). However, the effect of exogenous
CTRP9 on cellular senescence in MSCs, which adversely affects the
function of engrafted stem cells, has never been studied.
AMP-activated protein kinase (AMPK) is an
evolutionarily conserved kinase that serves a central role in
maintaining the cellular metabolic balance (14). AMPK initiates biological changes
aimed at restoring cellular energy balance under conditions of
energetic stress, which are related to cellular senescence
(15,16). In addition, AMPK activation has
also been demonstrated to increase the lifespan and to the improve
metabolism (17), while
activation of the AMPK/mammalian target of rapamycin (mTOR) pathway
attenuates age-related dementia (18). As an important target of CTRP9,
AMPK exerts a cytoprotective effect by regulating the oxidant
response and inhibiting endoplasmic reticulum stress (11,19). However, the involvement of AMPK in
CTRP9-induced rejuvenation of aged MSCs remains unclear.
Induction of the oxidative response is implicated in
cellular aging (4), and
maintaining a delicate oxidative/antioxidative balance has been
demonstrated to be critical for MSC maintenance and DNA integrity,
as well as for preventing reactive oxygen species (ROS)-mediated
cellular senescence (20). MSCs
are normally quiescent with an inherently low metabolic rate, and
generate low levels of ROS; however, ROS levels accumulate with
age, leading to ROS-induced oxidative free radical damage and
impaired cellular function, associated with reduced efficacy for
treating ischemic diseases (21,22). CTRP9 is an effective antioxidant
factor that has been reported to ameliorate endothelial dysfunction
via a peroxisome proliferator-activated receptor γ coactivator
(PGC)-1α/AMPK-mediated antioxidant effect (19). These results suggested that this
antioxidant capacity of CTRP9 may influence MSC aging.
The present study aimed to investigate the effect of
CTRP9 on MSC senescence and the role of the PGC-1α/AMPK signaling
pathway in CTRP9-induced rejuvenation in relation to age-related
cellular impairments in MSCs.
Materials and methods
Reagents
X-tremeGENE HP DNA transfection reagent was
purchased from Roche Diagnostics (Basel, Switzerland). Rabbit
monoclonal antibodies against PGC-1α (1:1,000; cat. no. 2178), AMPK
(1:1,000; cat. no. 5832), phospho-AMPK (1:1,000; cat. no. 50081),
and β-actin (1:1,000; cat. no. 4970) were obtained from Cell
Signaling Technology, Inc. (Danvers, MA, USA). Vascular endothelial
growth factor (VEGF, cat. no. RAB0509), basic fibroblast growth
factor (bFGF, cat. no. RAB0184), hepatocyte growth factor (HGF,
cat. no. RAB0214), and insulin-like growth factor (IGF, cat. no.
RAB0229) ELISA kits were purchased from Sigma-Aldrich (Merck KGaA,
Darmstadt, Germany). Horseradish peroxidase-conjugated anti-rabbit
secondary antibodies (1:1,000; sc-2357) were from Santa Cruz
Biotechnology, Inc. (Dallas, TX, USA). Small interfering RNAs
(siRNAs) targeting PGC-1α and AMPK transcripts were purchased from
Thermo Fisher Scientific, Inc. (Waltham, MA, USA). Superoxide
Dismutase (SOD) Activity Colorimetric assay and Lipid Peroxidation
(malondialdehyde; MDA) assay kits were purchased from Abcam
(Cambridge, UK). Human recombinant CTRP9 was obtained from Aviscera
Bioscience Inc. (Santa Clara, CA, USA).
Animals
The young (8-weeks-old, n=12) and aged
(18-months-old, n=12) male C57BL/6 mice were purchased from the
Laboratory Animal Center of Wenzhou Medical University (Wenzhou,
China). All procedures were approved by the Laboratory Animal
Ethics Committee of Wenzhou Medical University. The mice were
housed under a 12 h light/dark cycle at 21±2°C and 30–70% humidity.
Food and water were provided ad libitum.
Cell culture and cell treatment
Bone marrow MSCs were isolated using a standard
protocol, as described previously (23). Briefly, bone marrow was isolated
from the femurs and tibias of mice by flushing with PBS. Adherent
MSCs were propagated and maintained at 37°C and 5% CO2
in high glucose DMEM (HyClone; GE Healthcare Life Sciences, Logan,
UT, USA) supplemented with 10% fetal bovine serum (HyClone; GE
Healthcare Life Sciences) and 1% penicillin/streptomycin (Beyotime
Institute of Biotechnology, Jiangsu, China).
For CTRP9 treatment, cells were cultured with medium
containing 2 µg/ml of recombinant CTRP9 and incubated at 37°C for
various periods, as described previously (13).
Cell proliferation assay
MSCs were plated at 1×104 cells/well in
96-well plates and cell proliferation was determined using a Cell
Counting Kit-8 (CCK-8; HaiGene Technology, Harbin, China),
according to the manufacturer's protocol. The absorbance of each
well at 450 nm was recorded.
MTT assay
Cell viability was determined by MTT assay. Briefly,
300 µl of MTT reagent was added to each well 2 h prior to
harvesting. The supernatant was then removed and the cells were
incubated with 400 µl of dimethyl sulfoxide for 10 min. Absorbance
at 540 nm was recorded using an ELISA plate reader.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
The expression levels of several genes were analyzed
by RT-qPCR. RNA was extracted from cells using TRIzol reagent
(Thermo Fisher Scientific, Inc.) and reverse transcribed using a
First Strand cDNA Synthesis kit (Roche Diagnostics). qPCR was
performed using Fast Start Universal SYBR Master reagent
(Sigma-Aldrich; Merck KGaA). The samples were subjected to 40
cycles of amplification at 95°C for 15 sec followed by 63°C for 20
sec and 71°C for 25 sec by using specific primers. The
quantification number of cycles (Cq) was set within the exponential
phase of the PCR. The ΔCq value for each target gene was calculated
by subtracting the Cq value for the GAPDH gene (internal control).
Relative fold changes in mRNA expression were calculated using the
formula 2−ΔΔCq (24).
The primer pairs used to detect the mRNA levels of target genes are
listed in Table I.
 | Table ISequences of primers used in the
study. |
Table I
Sequences of primers used in the
study.
| Gene | Primer | Sequence
(′5-3′) |
|---|
| IL-6 | Forward |
TCTATACCACTTCACAAGTCGGA |
| Reverse |
GAATTGCCATTGCACAACTCTTT |
| IL-10 | Forward |
GCCAACGAAGATCCTCCCCCGTAC |
| Reverse |
TAAGAGCAGGCAGCATAGCAGTGC |
| PGC1-α | Forward |
GGAACTGCAGGCCTAACTCC |
| Reverse |
TTGGAGCTGTTTTCTGGTGC |
| AMPK | Forward |
CTCTATGCTTTGCTGTGTGG |
| Reverse |
GGTCCTGGTGGTTTCTGTTG |
| Telomere
length | Forward |
TGAAAGTAGAGGATTGCCACTG |
| Reverse |
AGCCAGAACAGGAACGTAGC |
| GAPDH | Forward |
AATCTCCACTTTGCCACTGC |
| Reverse |
ATGGTGAAGGTCGGTGTGA |
Western blot analysis
MSCs were lysed with ice-cold lysis buffer (Beyotime
Institute of Biotechnology) to obtain total protein. Expression
levels of PGC-1α, AMPK, phospho-AMPK and β-actin were evaluated by
western blotting. Cellular extracts were prepared according to the
manufacturer's instructions. Protein samples were quantified and
separated by SDS-PAGE. Western blot assays were performed as
described previously (22).
ELISA
Total concentrations of VEGF, bFGF, HGF and IGF
proteins secreted by MSCs were assessed by ELISA, according to the
manufacturer's protocol, as described previously (23). The absorbance of each well was
quantified at 450 nm. Samples were analyzed in triplicate.
siRNA knockdown
MSCs were transfected using X-tremeGENE HP DNA
Transfection Reagent, according to the manufacturer's protocol.
Briefly, MSCs at 80% confluence were transfected with siRNA for 6 h
at37°C. The transfection reagent-siRNA mixture was then replaced
with fresh growth medium and the cells were harvested for further
experiments at 72 h post-transfection. The siRNA sequences were:
siRNA-PGC-1α, AAGACGGATTGCCCTCATTTG; siRNA-AMPK,
CCAGGUCAUCAGUACACCAUCUGAU; and siRNA-NT (non-targeting control),
TTCTCCGAACGTGTCACGT.
Relative telomere length measurement
Relative telomere length quantification in U87 cells
was performed using a qPCR approach, as previously described
(25). GAPDH was used as the
normalizing gene. The primer pairs used to detect the telomere
length are listed in Table I.
Relative telomerase activity measurement
(RTA)
Telomerase activity of whole cell lysate was
measured by a Telo TAGGG telomerase PCR ELISA PLUS kit (Roche
Diagnostics GmbH, Penzberg, Germany). Cell lysates were centrifuged
at 12,000 × g (20 min at 4°C) and 3 µl of cell extract was used for
each telomeric repeat PCR amplification reaction and 3 µl of
inactivated cell lysate was used for Telomeric Repeat Amplification
Protocol (TRAP) reaction, according to the manufacturer's
recommendations. The amount of TRAP products was determined with
ELISA by measurement of absorbance at 450 nm using a microplate
reader, as previously reported (26).
Mitochondrial membrane potential
Cells were grown in a 96-well microtiter plate at
37°C for 1 day in complete culture medium to a density of
1×104 cells per well. The cells were then washed with
PBS and incubated at 37°C for 15 min with 5 µg/ml JC-1. After two
wash cycles with PBS, the time-dependent JC-1 fluorescence was
recorded using an ELISA plate reader. The fluorescent probe was
excited at 490 nm and emission was measured at 530 and 590 nm.
SOD activity
SOD activity in cells was determined using a
colorimetric assay kit (Abcam), according to the manufacturer's
protocol. Briefly, protein was isolated from MSC susing lysis
buffer, and SOD activity was measured in 10 µg of total protein
extract. Absorbance was measured at 450 nm.
ROS measurement
Levels of intracellular ROS were determined using
2,7-dichlorodihydrofluorescein diacetate (Beyotime Institute of
Biotechnology), following the manufacturer's instructions. The
fluorescence intensity of the cells was measured using a
fluorescence spectrophotometer, with excitation and emission
wavelengths of 488 and 525 nm, respectively.
Lipid peroxidation assays
Lipid peroxidation was monitored using an assay kit
(Abcam) to measure the formation of MDA, according to the
manufacturer's protocol. Briefly, MSCs (1×106 cells)
were homogenized on ice in 300 µl of MDA lysis buffer (with 3 µl of
100X butylated hydroxytoluene), then centrifuged (13,000 × g, 10
min) to remove insoluble material. The supernatant (200 µl) was
added to 600 µl of thiobarbituric acid and incubated at 95°C for 60
min. The samples were cooled to room temperature in an ice bath for
10 min, and the absorbance at 532 nm was measured
spectrophotometrically.
Statistical analysis
Data were expressed as mean ± standard deviation.
Differences among groups were assessed by one-way analysis of
variance followed by a Tukey's b test, and comparisons between two
groups were evaluated with Student's t-tests, using SPSS package
v19.0 (IBM Corp., Armonk, NY, USA). P<0.05 was considered to
indicate a statistically significant difference.
Results
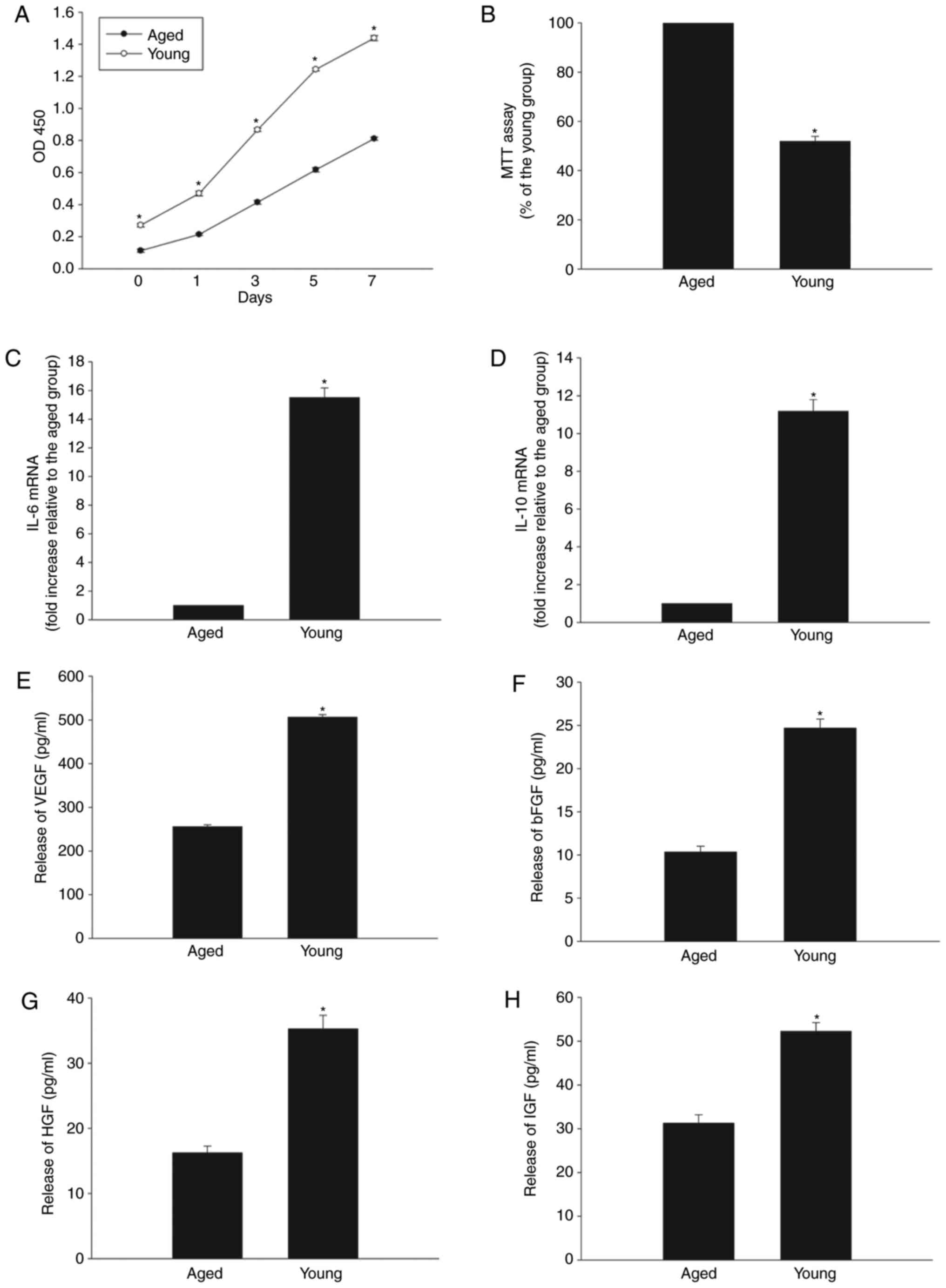
MSCs display degenerative properties
during aging
MSC properties were compared between cells derived
from young (8-weeks-old) and aged (18-months-old) male C57BL/6
mice. Cell proliferation rates measured by CCK-8 assay were
significantly lower in aged MSCs after 1, 3, 5 and 7 days of
culture, compared with the young MSCs (Fig. 1A). MTT assay also demonstrated
impaired cellular viability in the aged group (Fig. 1B). The mRNA expression levels of
IL-10 and IL-6 were reduced in aged MSCs compared with young MSCs
(Fig. 1C and D). Finally, the
levels of secreted VEGF, bFGF, HGF and IGF proteins were
significantly decreased in aged compared with young MSCs, as
evaluated by ELISA (Fig.
1E–H).
CTRP9 restores the degenerative
properties of aged MSCs
The reduced proliferative ability of aged MSCs was
significantly reversed by addition in the culture of human
recombinant CTRP9, according to results from the CCK-8 assay
(Fig. 2A), while cell viability
was increased by CTRP9 at the given doses (2 µg/ml; Fig. 2B). CTRP9 treatment also increased
the immunoregulatory abilities of MSCs accompanied by increased
IL-10 and IL-6 mRNA expression (Fig.
2C and D), and promoted the paracrine abilities of aged MSCs,
as evidenced by increased secretion of VEGF, bFGF, HGF and IGF
(Fig. 2E–H). Furthermore,
incubation of MSCs with CTRP9 increased the senescence-impaired
telomere length (Fig. 2I) and
telomerase activity (Fig.
2J).
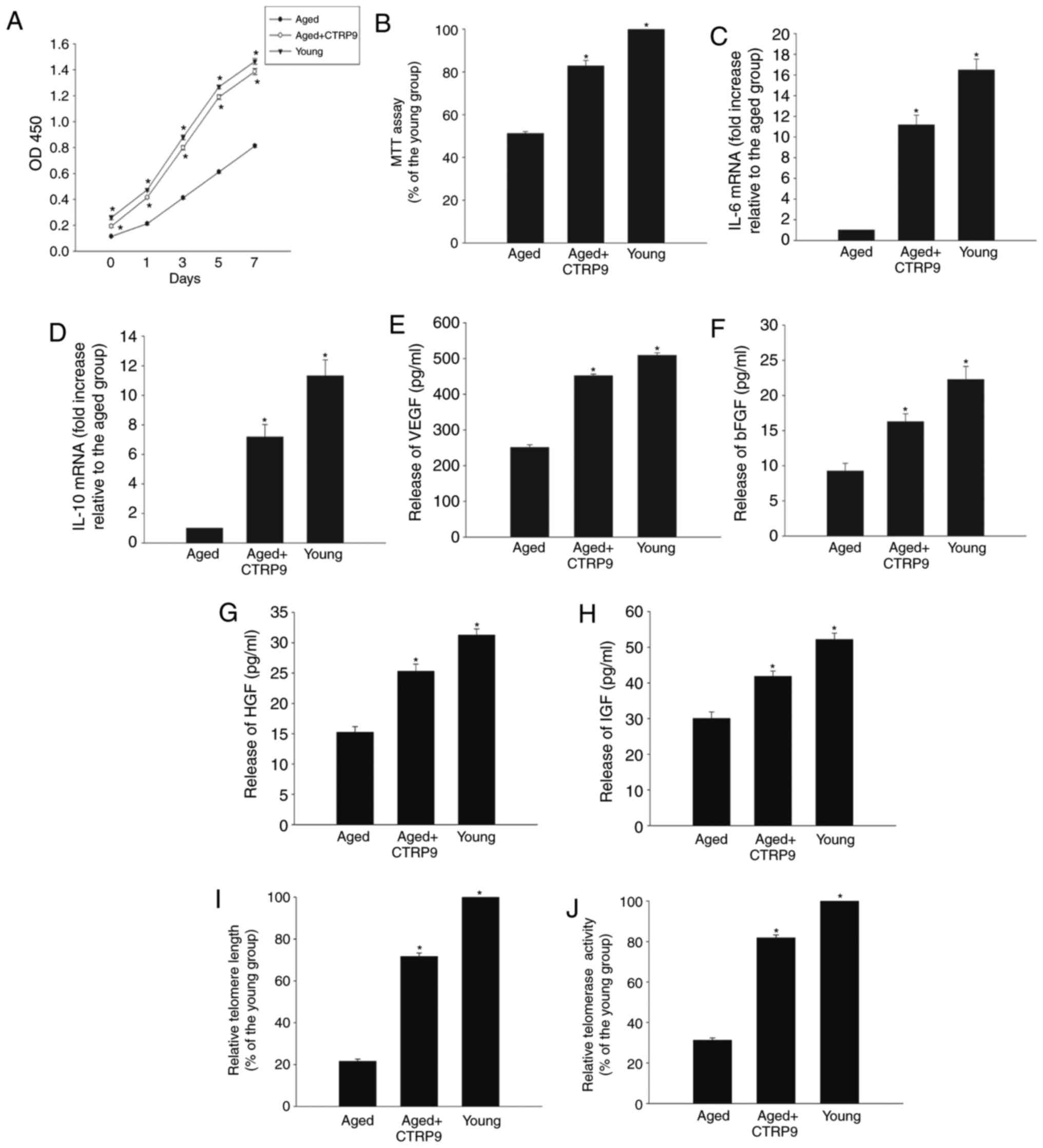 | Figure 2CTRP9 treatment restores the
degenerative properties of aged MSCs. (A) Proliferation of young
and aged MSCs, and aged MSCs treated with CTRP9 (2 µg/ml),
determined withCCK-8 proliferation assay. (B) Viability of young
and aged MSCs, and aged MSCs treated with CTRP9 analyzed with MTT
assay. (C and D) IL-6 and IL-10 mRNA expression levels in young and
aged MSCs, and aged MSCs treated with CTRP9. (E–H) Secretion of
VEGF, bFGF, HGF and IGF into the culture medium of young and aged
MSCs, and aged MSCs treated with CTRP9, as analyzed by ELISA. (I)
Telomere length and (J) relative telomerase activity in young and
aged MSCs, and aged MSCs treated with CTRP9. Data represent means ±
standard deviation from three independent experiments.
*P<0.05 vs. aged. CTRP9, C1q and tumor necrosis
factor-related protein 9; MSCs, mesenchymal stem cells; IL,
interleukin; VEGF, vascular endothelial growth factor; bFGF, basic
fibroblast growth factor; HGF, hepatocyte growth factor; IGF,
insulin-like growth factor. |
The PGC-1α/AMPK signaling pathway is
involved in the rejuvenating effect of CTRP9
Cellular senescence impeded PGC-1α protein
expression, but this effect was significantly reversed by
CTRP9treatment (Fig. 3A and B).
CTRP9 also abrogated the age-mediated suppression of AMPK
phosphorylation (Fig. 3C and D),
suggesting a critical role for CTRP9 in activation of the
PGC-1α/AMPK signaling pathway. Inhibition of PGC-1α or AMPK by
transient transfection with specific siRNAs (Fig. 3E and F) abolished the ameliorating
effects of CTRP9 on age-related decreases in cellular proliferation
(Fig. 4A) and viability (Fig. 4B), and on the decreased
immunoregulatory (Fig. 4C and D)
and paracrine abilities (Fig.
4E–H), accompanied by shortened telomere length (Fig. 4I) and impaired telomerase activity
(Fig. 4J).
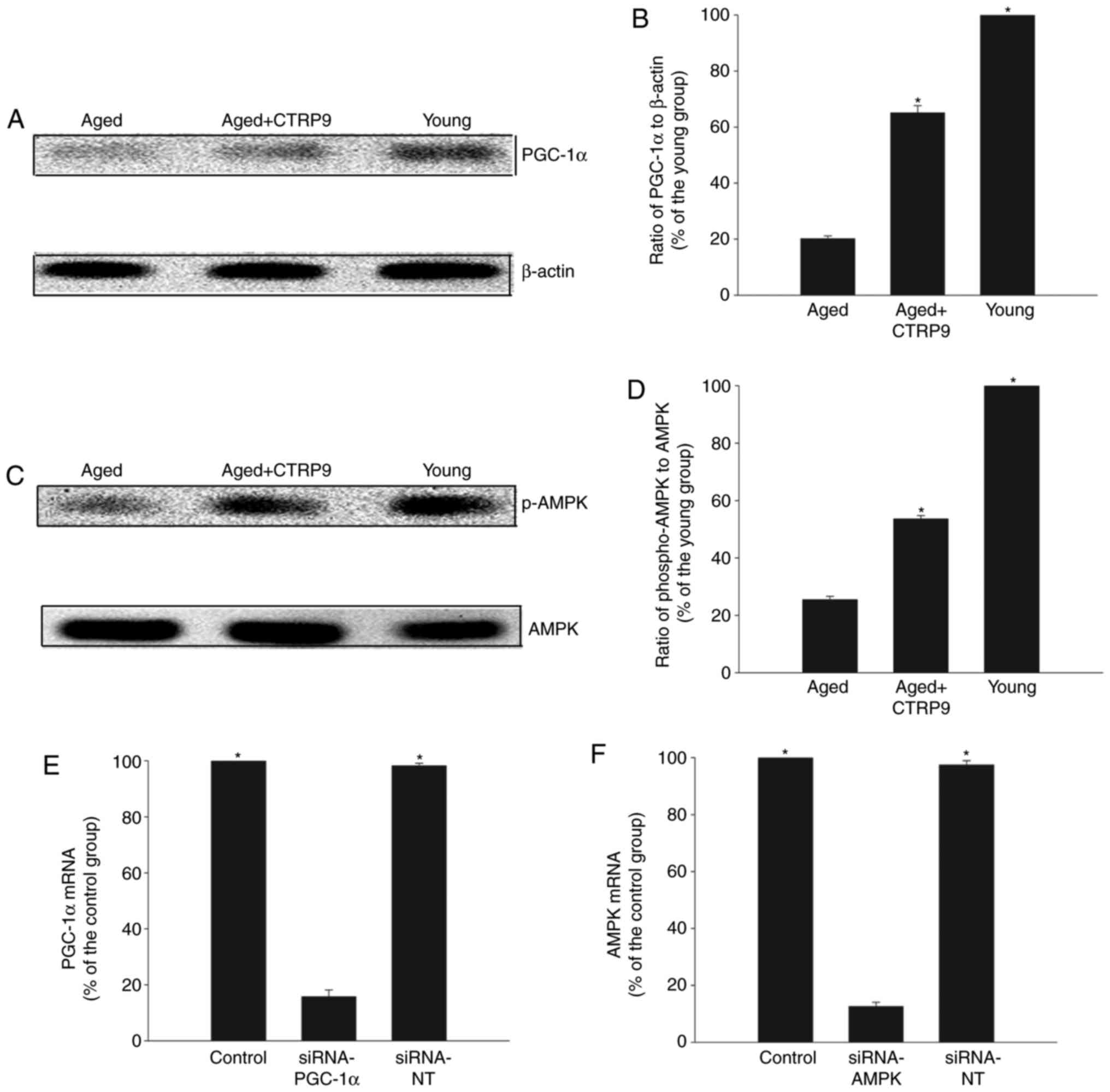 | Figure 3PGC-1α/AMPK signaling pathway
activation. (A) Representative blots and (B) quantification of
western blot analysis of PGC-1α and β-actin protein expression
levels in young MSCs, aged MSCs, and aged MSCs treated with CTRP9
(2 µg/ml). (C) Representative blots and (D) quantification of
western blot analysis of AMPK and p-AMPK protein expression levels
in young MSCs, aged MSCs, and aged MSCs treated with CTRP9. Data
represent mean ± standard deviation from three independent
experiments. *P<0.05 vs. aged. (E) PGC-1α mRNA levels
in MSCs transfected with siRNA-PGC-1α or siRNA-NT.
*P<0.05 vs. siRNA-PGC-1α. (F) AMPK mRNA levels in
MSCs transfected with siRNA-AMPK or siRNA-NT. *P<0.05
vs. siRNA-AMPK. PGC-1α, peroxisome proliferator-activated receptor
γcoactivator-1α; AMPK, AMP-activated protein kinase; MSCs,
mesenchymal stem cells; CTRP9, C1q and tumor necrosis
factor-related protein 9; p-, phosphorylated; si, small
interfering; NT, non-targeting control. |
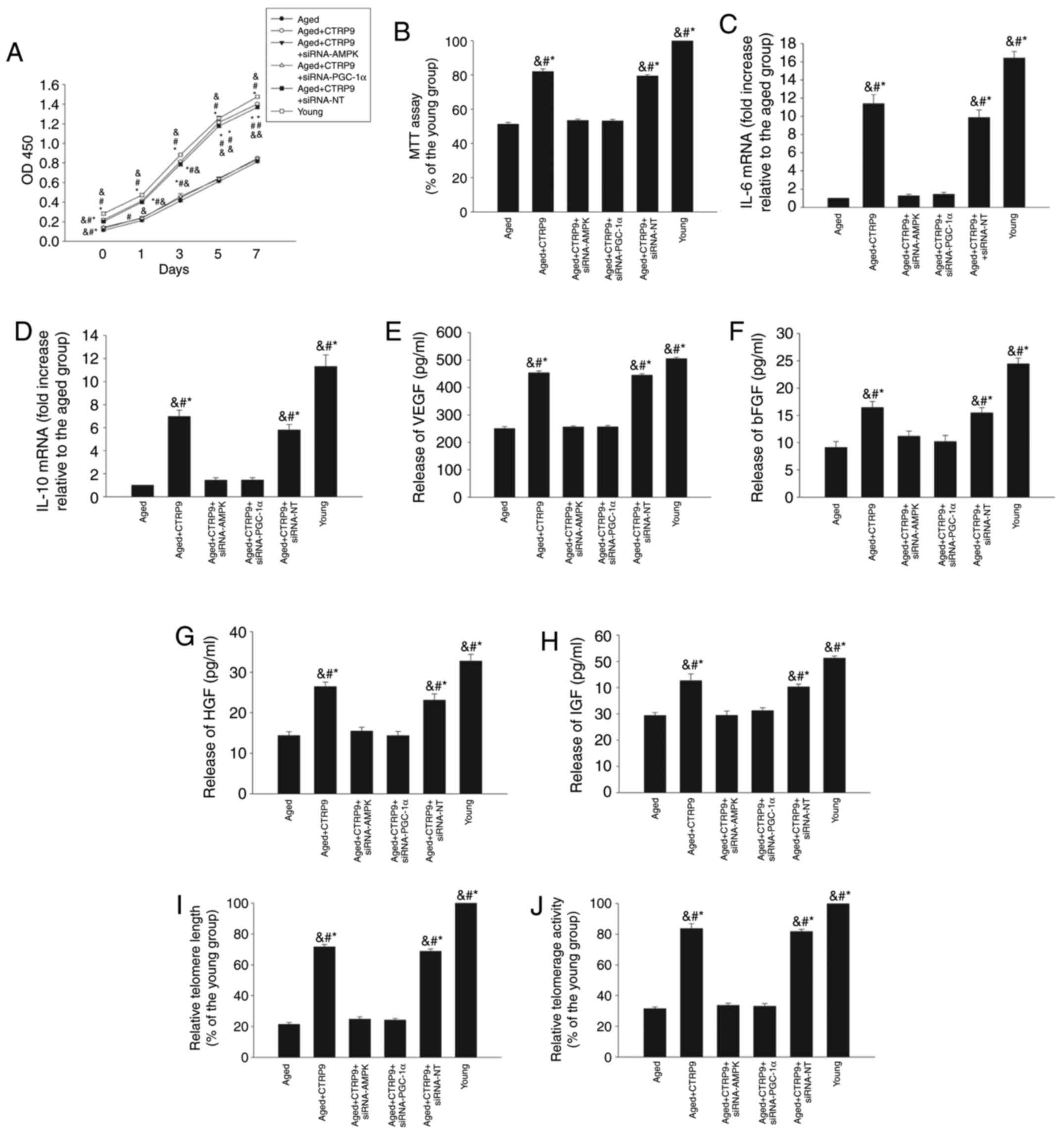 | Figure 4PGC-1α/AMPK signaling pathway is
involved in the rejuvenating effect of CTRP9. Aged MSCs were
treated with CTRP9 (2 µg/ml) or transfected with siRNA-PGC-1α,
siRNA-AMPK, or siRNA-NT in the presence of CTRP9 (2 µg/ml). (A)
Cell proliferation was determined by CCK-8 proliferation assay. (B)
Cell viability was analyzed by MTT assay. (C and D) IL-6 and IL-10
mRNA levels in MSCs were analyzed with RT-qPCR. (E–H) Secretion of
VEGF, bFGF, HGF and IGF into the culture medium was analyzed by
ELISA. (I) Telomere length was analyzed by RT-qPCR. (J) Relative
telomerase activity was measured. Data represent mean ± standard
deviation from three independent experiments. *P<0.05
vs. aged MSCs; #P<0.05 vs. aged+CTRP9+siRNA-PGC-1α
and &P<0.05 vs. aged+CTRP9+siRNA-AMPK. PGC-1α,
peroxisome proliferator-activated receptor γcoactivator-1α; AMPK,
AMP-activated protein kinase; CTRP9, C1q and tumor necrosis
factor-related protein 9; MSCs, mesenchymal stem cells; si, small
interfering; NT, non-targeting control; IL, interleukin; RT-qPCR,
reverse transcription-quantitative polymerase chain reaction; VEGF,
vascular endothelial growth factor; bFGF, basic fibroblast growth
factor; HGF, hepatocyte growth factor; IGF, insulin-like growth
factor. |
CTRP9 exerts a rejuvenating effect via
the antioxidant response
The role of the antioxidant response in
CTRP9-induced rejuvenation of MSCs was further investigated by
examining the mitochondrial transmembrane potential, activation of
SOD, generation of ROS, and lipid peroxidation by MDA assay. Aging
significantly decreased the mitochondrial transmembrane potential
(Fig. 5A) and SOD activation
(Fig. 5B), while it increased ROS
generation (Fig. 5C) and MDA
activation (Fig. 5D). By
contrast, CTRP9 treatment significantly increased the mitochondrial
trans-membrane potential (Fig.
5A) and SOD activation (Fig.
5B), and greatly reduced ROS generation (Fig. 5C) and MDA activation (Fig. 5D). Furthermore, inhibition of
PGC-1α or AMPK by siRNA abolished the antioxidant effects of CTRP9,
resulting in increased ROS generation and MDA activation, and
decreased mitochondrial membrane potential and SOD activation
(Fig. 5).
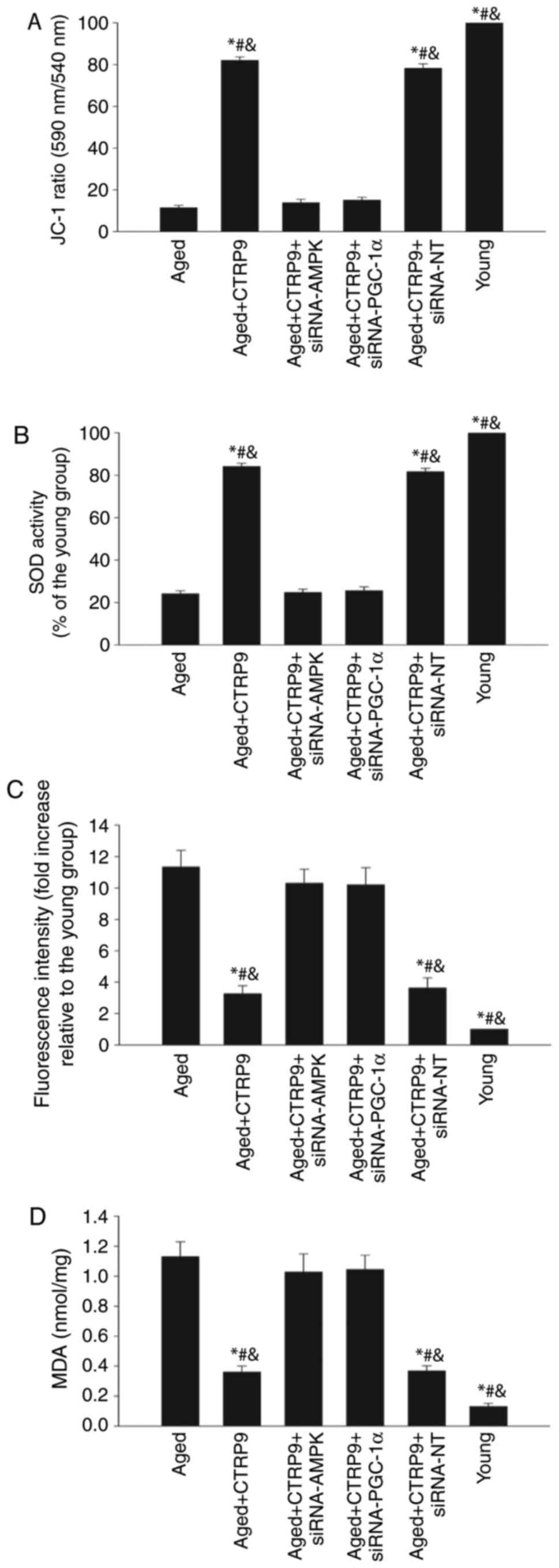 | Figure 5CTRP9 exerts a rejuvenating effect
via the antioxidant response. Aged MSCs were treated with CTRP9 (2
µg/ml) or transfected with siRNA-PGC-1α, siRNA-AMPK or siRNA-NT in
the presence of CTRP9 (2 µg/m). Young MSCs were also analyzed in
parallel. (A) Mitochondrial membrane potential was measured with
the JC-1 dye; (B) SOD activity was evaluated by colorimetric assay.
(C) Intracellular ROS production was analyzed by fluorescence
spectrophotometry. (D) Lipid peroxidation was evaluated by MDA
formation. Data represent mean ± standard deviation from three
independent experiments. *P<0.05 vs. aged MSCs;
#P<0.05 vs. aged+CTRP9+siRNA-PGC-1α and
&P<0.05 vs. aged+CTRP9+siRNA-AMPK. CTRP9, C1q and
tumor necrosis factor-related protein 9; MSCs, mesenchymal stem
cells; si, small interfering; PGC-1α, peroxisome
proliferator-activated receptor γcoactivator-1α; AMPK,
AMP-activated protein kinase; NT, non-targeting control; SOD,
superoxide dismutase; ROS, reactive oxygen species; MDA,
malondialdehyde. |
Discussion
Aging is associated with reduced organ function and
an increased incidence of disease (27), and functional impairment and
mortality have been demonstrated to be significantly increased in
aged stroke patients compared with relatively young patients
(28). Stem cell therapy has
shown great potential for repairing and remodeling the
neurovascular structure following ischemic brain injury (29); however, functional improvement
after stem cell transplantation is impaired by cellular senescence
(6). It is therefore crucial to
be able to rejuvenate aged MSCs in order to improve their efficacy
for the treatment of ischemic stroke. MSC aging driven by both
intrinsic and extrinsic factors is linked to impaired MSC
self-renewal and regeneration (30), as well as decreased
immunoregulatory and angiogenic abilities (31,32), which are important in the
treatment of ischemic stroke. The results of the current study
indicated that MSC proliferation and viability, immune modulation,
and angiogenesis-related paracrine ability were all impaired in
MSCs from aged donors.
CTRP9 is a novel adipokine exclusively expressed in
adipocytes and also highly expressed in the brain (10). CTRP9 has been suggested to act as
a regulator of vascular function, and to exert protective effects
in ischemic diseases (12). The
cytoprotective effects of CTRP9 have been well documented, through
promoting cell proliferation, inhibiting the oxidative response,
and modulating endoplasmic reticulum stress (13,33). The present study demonstrated that
CTRP9 treatment promoted MSC proliferation and viability and
increased their immunoregulatory and angiogenic abilities,
accompanied by elongation of telomere length and recovery of
telomerase activity.
PGC-1α is a transcriptional coactivator and a
fundamental regulator of mitochondrial function (34). Several studies have highlighted an
important protective role for PGC-1α in cellular senescence
(35), and activation of PGC-1α
maintains cellular metabolic homeostasis through regulation of
mitochondrial ROS to exert a rejuvenating effect in senescent cells
(14). Activation of PGC-1α also
enhances the engraftment and angiogenesis of MSCs in ischemia
diseases (36). The present study
demonstrated that cellular senescence inhibited PGC-1α and that
exogenous CTRP9 re-activated PGC-1α in aged MSCs, while PGC-1α
inhibition abolished the CTRP9-mediated rejuvenation. These results
suggested that CTRP9 may attenuate senescence-stimulated MSC
dysfunction through activation of PGC-1α.
AMPK is an energy sensor that is ubiquitously
expressed in MSCs and exerts antisenescence effects (31). The protective effects of APNCTRP9
on cellular protection are expected to be partially mediated by the
activation of AMPK signaling (13). In addition, AMPK activation has
been demonstrated to be responsible for the anti-inflammatory
effect of CTRP9 in cells (37).
The current results demonstrated that CTRP9 prevented the
senescence-mediated suppression of AMPK phosphorylation, while
knockdown of AMPK eradicated the inhibitory effects of CTRP9 on MSC
dysfunction in response to senescence, implying an essential role
for AMPK.
Impaired mitochondrial function and increased
oxidative effects lead to DNA damage and are considered to
contribute to cellular senescence in stem cells (4). Accumulating oxidant stress impairs
the function of aged MSCs via excessive ROS production (23). A recent study revealed that CTRP9
promotes MSC proliferation and migration, and protects against
hydrogen peroxide-induced cellular death through its antioxidative
effect (13). Similarly, the
present results demonstrated that senescence elicited an oxidant
reaction, and this effect was prevented by CTRP9 treatment.
Furthermore, this CTRP9-mediated antioxidant effect was suppressed
by knockdown of PGC-1α or AMPK expression by siRNA.
In conclusion, CTRP9 treatment inhibited the oxidant
effect associated with cell senescence via PGC-1α/AMPK signaling
activation, contributing to rejuvenation of MSCs. CTRP9 may thus
serve as a candidate for rejuvenating MSCs and for potentially
improving the therapeutic efficacy of MSC transplantation in aged
patients with ischemic stroke.
Acknowledgments
Not applicable.
Funding
This study was supported by the Zhejiang Provincial
Natural Science Foundation of China (grant nos. LQ17C060002,
LY16H160053 and LY17H160052), the Medical Scientific Research
foundation of Zhejiang Province of China (grant no. WKJ-ZJ-1525)
and the Wenzhou Science and Technology Plan Project (grant no.
Y20170088).
Availability of data and materials
The analyzed datasets generated during the study are
available from the corresponding author on reasonable request.
Authors' contributions
QL and ZZZ made substantial contributions to the
acquisition of data, CDW, LC and JLL made substantial contributions
to the analysis and interpretation of data. YCW, JDX and ZPS were
the major contributors in writing the manuscript. WMZ and XBC were
involved in conception and design, revising it critically for
important intellectual content. All authors read and approved the
final manuscript.
Ethics approval and consent to
participate
All procedures involving the use of animals were
approved by the Laboratory Animal Ethics Committee of Wenzhou
Medical University.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Iadecola C: The neurovascular unit coming
of age: A journey through neurovascular coupling in health and
disease. Neuron. 96:17–42. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
van Velthoven CT, van de Looij Y,
Kavelaars A, Zijlstra J, van Bel F, Huppi PS, Sizonenko S and
Heijnen CJ: Mesenchymal stem cells restore cortical rewiring after
neonatal ischemia in mice. Ann Neurol. 71:785–796. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
van Velthoven CT, Kavelaars A and Heijnen
CJ: Mesenchymal stem cells as a treatment for neonatal ischemic
brain damage. Pediatr Res. 71:474–481. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Akunuru S and Geiger H: Aging, clonality,
and rejuvenation of hematopoietic stem cells. Trends Mol Med.
22:701–712. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Townsley DM, Dumitriu B and Young NS: Bone
marrow failure and the telomeropathies. Blood. 124:2775–2783. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Liu M, Lei H, Dong P, Fu X, Yang Z, Yang
Y, Ma J, Liu X, Cao Y and Xiao R: Adipose-derived mesenchymal stem
cells from the elderly exhibit decreased migration and
differentiation abilities with senescent properties. Cell
Transplant. 26:1505–1519. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Badan I, Buchhold B, Hamm A, Gratz M,
Walker LC, Platt D, Kessler Ch and Popa-Wagner A: Accelerated glial
reactivity to stroke in aged rats correlates with reduced
functional recovery. J Cereb Blood Flow Metab. 23:845–854. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Goldstein BJ, Scalia RG and Ma XL:
Protective vascular and myocardial effects of adiponectin. Nat Clin
Pract Cardiovasc Med. 6:27–35. 2009. View Article : Google Scholar :
|
|
9
|
Gonon AT, Widegren U, Bulhak A, Salehzadeh
F, Persson J, Sjöquist PO and Pernow J: Adiponectin protects
against myocardial ischaemia-reperfusion injury via AMP-activated
protein kinase, Akt, and nitric oxide. Cardiovasc Res. 78:116–122.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yang G, Qin C, Wang B, Jia J, Yuan X, Sun
C and Li W: Molecular identification and functional analysis of
CTRP9 in Epinepheluscoioides. J Mol Endocrinol. 58:179–191. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bai S, Cheng L, Yang Y, Fan C, Zhao D, Qin
Z, Feng X, Zhao L, Ma J, Wang X, et al: C1q/TNF-related protein 9
protects diabetic rat heart against ischemia reperfusion injury:
Role of endoplasmic reticulum stress. Oxid Med Cell Longev.
2016:19020252016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhao D, Yang J and Yang L: Insights for
oxidative stress and mTOR signaling in myocardial
ischemia/reperfusion injury under diabetes. Oxid Med Cell Longev.
2017:64374672017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yan W, Guo Y, Tao L, Lau WB, Gan L, Yan Z,
Guo R, Gao E, Wong GW, Koch WL, et al: C1q/tumor necrosis
factor-related protein-9 regulates the fate of implanted
mesenchymal stem cells and mobilizes their protective effects
against ischemic heart injury via multiple novel signaling
pathways. Circulation. 136:2162–2177. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Rabinovitch RC, Samborska B, Faubert B, Ma
EH, Gravel SP, Andrzejewski S, Raissi TC, Pause A, St-Pierre J and
Jones RG: AMPK maintains cellular metabolic homeostasis through
regulation of mitochondrial reactive oxygen species. Cell Rep.
21:1–9. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Oakhill JS, Scott JW and Kemp BE: AMPK
functions as an adenylate charge-regulated protein kinase. Trends
Endocrinol Metab. 23:125–132. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Chandel NS and Tuveson DA: The promise and
perils of antioxidants for cancer patients. N Engl J Med.
371:177–178. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Riera CE, Merkwirth C, Filho CDDM and
Dillin A: Signaling networks determining life span. Annu Rev
Biochem. 85:35–64. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Goldberg J, Currais A, Prior M, Fischer W,
Chiruta C, Ratliff E, Daugherty D, Dargusch R, Finley K,
Esparza-Moltó PB, et al: The mitochondrial ATP synthase is a shared
drug target for aging and dementia. Aging Cell. 17:2018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Sun H, Zhu X, Zhou Y, Cai W and Qiu L:
C1q/TNF-related protein-9 ameliorates Ox-LDL-induced endothelial
dysfunction via PGC-1α/AMPK-mediated antioxidant enzyme induction.
Int J Mol Sci. 18:10972017. View Article : Google Scholar
|
|
20
|
Gharibi B, Farzadi S, Ghuman M and Hughes
FJ: Inhibition of Akt/mTOR attenuates age-related changes in
mesenchymal stem cells. Stem Cells. 32:2256–2266. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Otsu K, Das S, Houser SD, Quadri SK,
Bhattacharya S and Bhattacharya J: Concentration-dependent
inhibition of angiogenesis by mesenchymal stem cells. Blood.
113:4197–4205. 2009. View Article : Google Scholar :
|
|
22
|
Xia W, Xie C, Jiang M and Hou M: Improved
survival of mesenchymal stem cells by macrophage migration
inhibitory factor. Mol Cell Biochem. 404:11–24. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Xia W, Zhuang L, Deng X and Hou M: Long
noncoding RNA-p21 modulates cellular senescence via the
Wnt/β-catenin signaling pathway in mesenchymal stem cells. Mol Med
Rep. 16:7039–7047. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
25
|
Crepin T, Carron C, Roubiou C, Gaugler B,
Gaiffe E, Simula-Faivre D, Ferrand C, Tiberghien P, Chalopin JM,
Moulin B, et al: ATG-induced accelerated immune senescence:
Clinical implications in renal transplant recipients. Am J
Transplant. 15:1028–1038. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Xie Z, Xia W and Hou M: Long intergenic
noncoding RNA-p21 mediates cardiac senescence via the Wnt/β-catenin
signaling pathway in doxorubicin-induced cardiotoxicity. Mol Med
Rep. 17:2695–2704. 2018.
|
|
27
|
Smith BD, Smith GL, Hurria A, Hortobagyi
GN and Buchholz TA: Future of cancer incidence in the United
States: Burdens upon an aging, changing nation. J Clin Oncol.
27:2758–2765. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ye X, Hu J and Cui G: Therapy effects of
bone marrow stromal cells on ischemic stroke. Oxid Med Cell Longev.
2016:76829602016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Gervois P, Wolfs E, Ratajczak J, Dillen Y,
Vangansewinkel T, Hilkens P, Bronckaers A, Lambrichts I and Struys
T: Stem cell-based therapies for ischemic stroke: Preclinical
results and the potential of imaging-assisted evaluation of donor
cell fate and mechanisms of brain regeneration. Med Res Rev.
36:1080–1126. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Li H, Liu P, Xu S, Li Y, Dekker JD, Li B,
Fan Y, Zhang Z, Hong Y, Yang G, et al: FOXP1 controls mesenchymal
stem cell commitment and senescence during skeletal aging. J Clin
Invest. 127:1241–1253. 2017. View
Article : Google Scholar : PubMed/NCBI
|
|
31
|
Xia W, Zhang F, Xie C, Jiang M and Hou M:
Macrophage migration inhibitory factor confers resistance to
senescence through CD74-dependent AMPK-FOXO3a signaling in
mesenchymal stem cells. Stem Cell Res Ther. 6:822015. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Loisel S, Dulong J, Ménard C, Renoud ML,
Meziere N, Isabelle B, Latour M, Bescher N, Pedeux R, Bertheuil N,
et al: Brief report: Proteasomal indoleamine 2,3-dioxygenase
degradation reduces the immunosuppressive potential of clinical
grade-mesenchymal stromal cells undergoing replicative senescence.
Stem Cells. 35:1431–1436. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Liu Q, Zhang H, Lin J, Zhang R, Chen S,
Liu W, Sun M, Du W, Hou J and Yu B: C1q/TNF-related protein 9
inhibits the cholesterol-induced Vascular smooth muscle cell
phenotype switch and cell dysfunction by activating AMP-dependent
kinase. J Cell Mol Med. 21:2823–2836. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Patten IS and Arany Z: PGC-1 coactivators
in the cardiovascular system. Trends Endocrinol Metab. 23:90–97.
2012. View Article : Google Scholar
|
|
35
|
Sahin E, Colla S, Liesa M, Moslehi J,
Müller FL, Guo M, Cooper M, Kotton D, Fabian AJ, Walkey C, et al:
Telomere dysfunction induces metabolic and mitochondrial
compromise. Nature. 470:359–365. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Solis MA, Wei YH, Chang CH, Yu CH, Kuo PL
and Huang LL: Hyaluronan upregulates mitochondrial biogenesis and
reduces adenoside triphosphate production for efficient
mitochondrial function in slow-proliferating human mesenchymal stem
cells. Stem Cells. 34:2512–2524. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Jung CH, Lee MJ, Kang YM, Lee YL, Seol SM,
Yoon HK, Kang SW, Lee WJ and Park JY: C1q/TNF-related protein-9
inhibits cytokine-induced vascular inflammation and leukocyte
adhesiveness via AMP-activated protein kinase activation in
endothelial cells. Mol Cell Endocrinol. 419:235–243. 2015.
View Article : Google Scholar : PubMed/NCBI
|