Evaluation of the effect of various main elements on the PrPSc detection by real-time quaking-induced conversion assay
- Authors:
- Published online on: September 7, 2018 https://doi.org/10.3892/ijmm.2018.3867
- Pages: 3231-3237
-
Copyright: © Xiao et al. This is an open access article distributed under the terms of Creative Commons Attribution License.
Abstract
Introduction
Prion disease, or transmissible spongiform encephalopathy, refers to a group of fatal neurodegenerative disorders reported in humans and animals (1). Prion diseases in humans include Creutzfeldt-Jakob disease (CJD), fatal familial insomnia and Gerstmann-Sträussler-Scheinker syndrome. The most common form of human prion disease is sporadic CJD (sCJD), with a worldwide incidence of ~1 case per million individuals annually (2,3). Typically, sCJD presents with rapidly progressing ataxia, dementia and myoclonus. The clinical duration of the disease is usually <2 years, with the majority of patients succumbing to the disease within 6 months.
A definitive diagnosis of sCJD requires neuropathological or immunochemical detection of the prion protein (PrPSc) (4). PrPSc is partially protease-resistant and can induce its normal cellular isoform (PrPC), to undergo a conformational change. This occurs in a self-propagating manner through a seeded aggregation process, resulting in accumulation of PrPSc throughout the brain tissues, together with accompanying spongiform alterations, neuronal loss and gliosis (1,5,6). Other than brain biopsy, there is currently no other disease-specific pre-mortem diagnostic test for sCJD (7). Diagnosis of probable sCJD is based on abnormal findings as determined by clinical examinations and laboratory tests. Abnormal test results include periodic sharp wave complexes on electroencephalogram, and altered signals on brain magnetic resonance image and/or positive detection of 14-3-3 protein in the cerebrospinal fluid (CSF) (8). Postmortem examinations are rarely performed in China due to cultural traditions (9). Therefore, methods that permit the accurate diagnosis of sCJD are required.
Numerous studies have attempted to identify biomarkers in CSF samples for the diagnosis of human prion diseases. Proteins that have showed diagnostic values include 14-3-3, tau, S100 and neuron specific enolase (10-13). However, only a positive test result for 14-3-3 protein in the CSF using western blotting is included in the diagnostic criteria for sCJD (14,15). The detection of PrPSc in the CSF of patients with sCJD and other types of human prion diseases is difficult with routine testing methods, even using the sensitive protein-misfolding cyclic amplification (PMCA) technique.
Recently, a novel technique known as real-time quaking-induced conversion (RT-QuIC) has been developed that is based on amyloid fibril formation, a characteristic feature of prion proteins (16-18). A number of studies have identified that RT-QuIC has good sensitivity and specificity for the diagnosis of sCJD using human CSF samples (16,17). In the present study, a number of experimental variables that may influence RT-QuIC assay were evaluated. Using the identified optimal conditions, the capacity of the RT-QuIC assay to detect PrPSc in the brain homogenates of 263K-infected hamsters and in the CSF samples of probable sCJD patients was also evaluated.
Materials and methods
Ethics statement
The present study was approved by the Ethical Committee of the National Institute for Viral Disease Control and Prevention (Beijing, China) under the protocol 2009ZX10004-101, including the use of brain samples of hamsters infected with the scrapie strain 263K and CSF samples from probable sCJD patients and non-CJD patients. Informed consent was obtained from all patients prior to participation.
Expression and purification of the hamster recombinant PrP (rPrP) protein
Hamster rPrPC protein (residues 23-231; GenBank accession no. K02234) was prepared according to the method described by Wilham et al (18) with a few modifications. Briefly, the DNA sequence of hamster PrPC was ligated into the pRSET vector (cat. no. V35120; Thermo Fisher Scientific, Inc., Waltham, MA, USA), and then the recombinant plasmid was transformed into BL21 (DE3)pLysS competent cells (cat. no. C1500; Beijing Solarbio Science & Technology Co., Ltd., Beijing, China). We expressed rPrPC in 1 liter terrific broth medium (cat. no. 71491; Novagen; Merck KGaA, Darmstadt, Germany). Next, rPrPC protein was denatured with guanidine-HCL and purified by chromatography using Ni-NTA Superflow resin (cat. no. 30430; Qiagen, Hilden, Germany) in an XK 16/40 column (GE Healthcare Life Sciences, Little Chalfont, UK) at a flow rate of 1.9 ml/min. The concentration of rPrPC was adjusted to ~500 µg/ml as determined by the bicinchoninic acid reagent (cat. no. 71285; EMD Millipore, Billerica, MA, USA). The purity of the prepared rPrPC was evaluated by 15% SDS-PAGE and western blot analysis. Briefly, proteins were transferred onto a nitrocellulose filter membrane (cat. no. 10600001; GE Healthcare Life Sciences), which was then blocked with 5% skim milk in tris-buffered saline with 0.1% Tween-20 containing 0.1% Tween-20 for 1 h followed by incubation with the primary antibodies against 3F4 (cat. no. MAB1562-K; EMD Millipore) and β-actin (HX1827, Beijing Huaxing Bochuang Gene Technology, Co., Ltd., Beijing, China) overnight at 4°C. Subsequently, a horseradish peroxidase-conjugated goat anti-mouse IgG secondary antibody (cat. no. 31430; Thermo Fisher Scientific, Inc.) was incubated at 37°C for 2 h. Both primary and secondary antibodies were usedat a dilution of 1:5,000. The results were scanned with Bio-Rad Molecular Imager (Bio-Rad Laboratories, Inc., Hercules, California, USA) and analyzed by ImageJ software (National Institutes of Health, Bethesda, MD, USA).
Preparation of brain homogenate (BH) samples
A total of 10 3-week-old female Syrian golden hamsters (45.8±1.3 g; cat. no. 501; Beijing Vital River Laboratory Animal Technology Co., Ltd., Beijing, China) were intracerebrally inoculated with 5 µl hamster-adapted scrapie agents 263K (19,20). Animals were housed withfree access to food and water under a humidity between 40-70%, temperature between 20-26°C and a 12 h light/dark cycle. The incubation time of the 263K-infected hamsters was 70.5±4.93 days (21,22). Animals were sacrificed using ether and exsanguinated, and then the brains were surgically removed from each hamster. Subsequently, 10% BHs were prepared in lysis buffer (100 mM NaCl, 10 mM EDTA, 0.5% Nonidet P-40, 0.5% sodium deoxycholate, 10 mM Tris, pH 7.5) according to a previously described protocol (21,22). In order to set up a PrPSc panel for RT-QuIC, serially diluted (from 10−3 to 10−9) Sc-BH samples were prepared. Proteinase K-resistant PrP signals were detectable in Sc-BH samples of 10−3 using routine western blotting.
CSF samples
A total of 70 CSF samples from probable sCJD (63.3±8.8 years old, male/female ratio: 1.12) and 48 CSF samples from non-CJD patients (55.1±14.5 years old, male/female ratio: 1.28) were included in the current study. Informed consent was obtained from all patients prior to participation. All the samples were collected at the CJD surveillance network of hospitals without any additional treatments and sent to the CJD Surveillance Center during Jan to Dec in 2015. The diagnosis of probable sCJD was conducted according to the diagnostic criteria for CJD issued by the World Health Organization and the surveillance document for CJD issued by Chinese Center for Disease Control and Prevention (Beijing, China) (9). Non-CJD cases included patients whose clinical manifestations and examinations did not fulfill the diagnostic criteria for human prion disease even after follow-up, or patients who had other diagnoses. The samples were collected from the CSF Bank in the China CJD Surveillance Center attached to China CDC (Beijing, China), and 200 µl from each CSF sample was obtained. All enrolled CSF samples were free of blood contamination. Routine CSF biochemistry assays of those specimens, including cell count, glucose level and total protein, were all within the normal ranges.
Evaluation of the effect of various main elements on the reactivity of RT-QuIC
To identify any factors that affect the RT-QuIC assay for detecting PrPSc, a set of variables were independently evaluated. The basic elements in the working buffer were 1X PBS, 85-340 mM NaCl, 0.5-2 mM EDTA, 10-80 µM thioflavin T (ThT) and 5-20 µg rPrPC. These concentrations were selected based on published data and our previous experiments (16,23). In addition, 10−5 diluted BH from 263K-infected hamster at the terminal stage (Sc-BH) and BH from age-matched normal hamsters (Nor-BH) were also prepared as the positive and negative controls, respectively.
RT-QuIC assay
RT-QuIC was conducted in a black 96-well, optical-bottomed plate (Nunc 265301; Thermo Fisher Scientific, Inc.) on a BMG FLUOstar microplate reader (BMG LABTECH GmbH, Ortenberg, Germany). Next, 1 µl diluted BH or 5-30 µl CSF sample was mixed with 1X PBS, 170 mM NaCl, 1 mM EDTA, 0.01 mM ThT, 0.001% SDS and 10 µg rPrPC in a final volume of 100 µl. Each sample was assayed in triplicated. Each reaction contained the following control groups: Blank (reaction buffer only), positive (Sc-BH) and negative (Nor-BH). The working conditions were as follow: Temperature, 50°C; vibration speed, 1.996 × g; vibration/incubation time, 90/30 sec; total reaction time, 90 h. ThT fluorescence (excitation wavelength, 450 nm; emission wavelength, 480 nm) was automatically measured every 30 min and expressed as relative fluorescence units (rfu). The cutoff value was set as the mean value of the negative controls plus 3 times the standard deviation. A sample was considered to be positive when ≥2 wells revealed positive reaction curves.
Results
Determination of appropriate rPrPC concentration
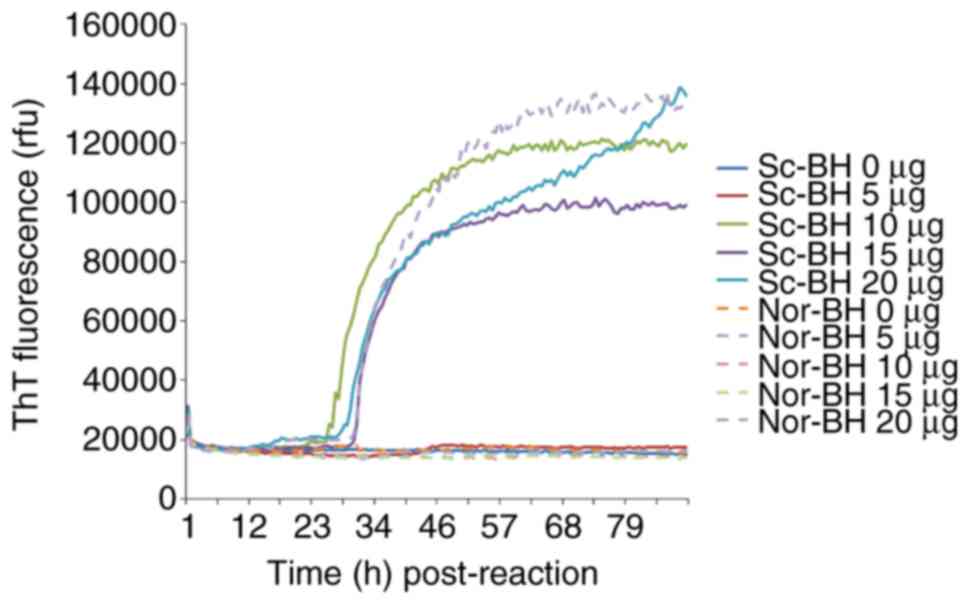
Different amounts (5, 10, 15 and 20 µg) of purified rPrPC, serving as the substrate, were subjected to RT-QuIC. Using Sc-BH as the seed, which induces the fibrillation of rPrPC, increases in the ThT fluorescence curves were observed in the reactions of 10, 15 and 20 µg rPrPC, which began to increase at ~30 h post-reaction, while only a slight elevation of the ThT value was detected in the 5 µg rPrPC reaction (Fig. 1). However, a positive increase of ThT value was also recorded in part of duplication of 20 µg rPrPC using Nor-BH as the seed, indicating a false positive reaction. Based on these data, 10 µg rPrPC was suggested to be the optimal working amount as the substrate in RT-QuIC.
Determination of appropriate NaCl concentration
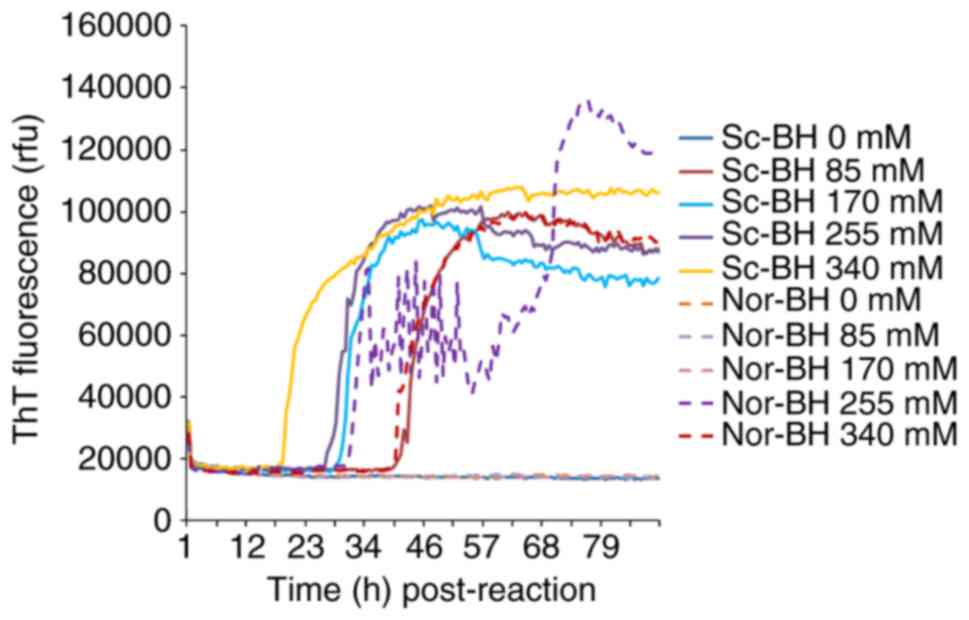
Different concentrations of NaCl (0, 85, 170, 255 and 340 mM) were examined. In the condition of 10−5 dilutedSc-BH, four preparations containing NaCl demonstrated positive reaction. The RT-QuIC reactivity was revealed to be NaCl dose-dependent. Along with the increase in the amount of NaCl, the positive conversion time post-reaction was decreased and the peak values of ThT values increased. However, increased ThT curves were also observed in the preparations of negative control containing 255 and 340 mM NaCl. This suggested that a high concentration of NaCl was able to increase the reactivity of RT-QuIC, as well as increase the possibility of false positive (Fig. 2). Thus, the NaCl concentration of 170 mM was selected for subsequent experiments.
Determination of appropriate EDTA concentration
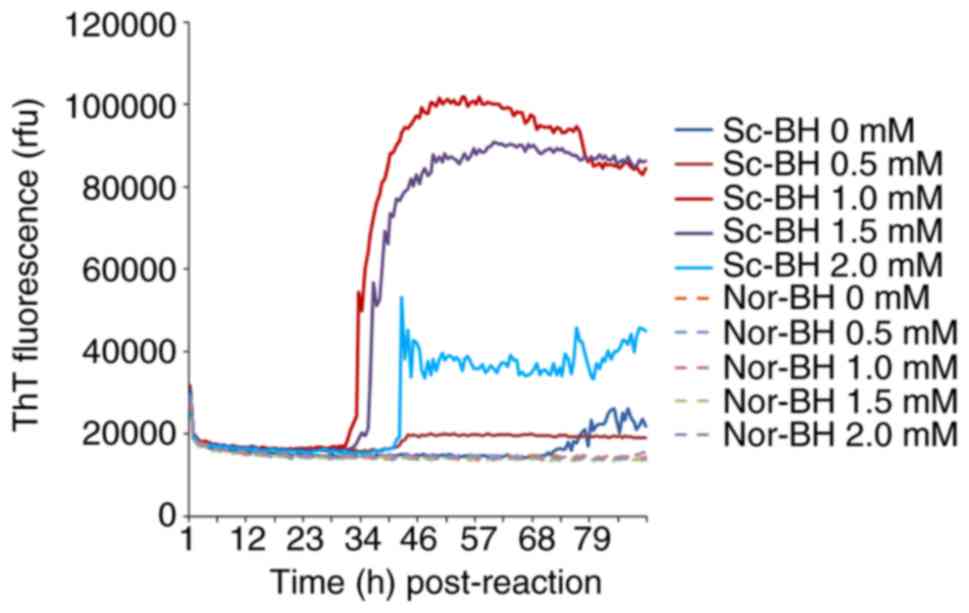
Different concentration of EDTA (0, 0.5, 1, 1.5 and 2 mM) were also tested in the RT-QuIC assay containing 10−5 diluted Sc-BH. After 34 h, the reaction curve with 1 mM EDTA began to increase, followed by the curves of the 1.5 and 2 mM EDTA reactions. At a relatively late stage, the reactions with 0 and 0.5 mM EDTA exhibited weak positive results. These data illustrated that certain concentrations of EDTA (particularly 1 mM) help the reactivity of RT-QuIC assay, whereas high concentrations of EDTA inhibit the assay (Fig. 3).
Determination of appropriate ThT final concentration
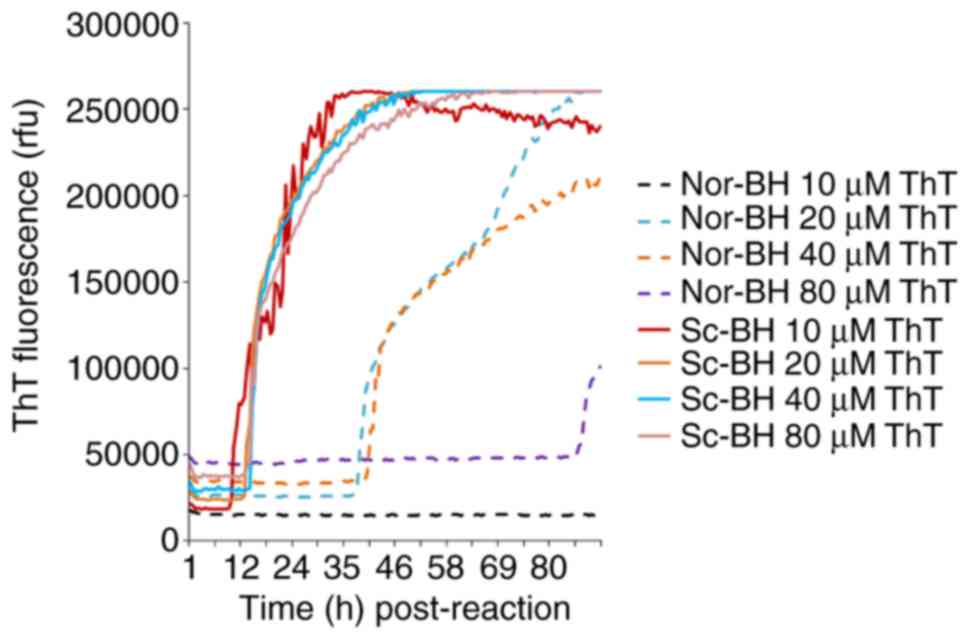
Four different final concentrations of ThT (10, 20, 40 and 80 µM) were also examined in the RT-QuIC assay containing 10−5 diluted Sc-BH. All four preparations of PrPSc exhibited positive curves at approximately 9-12 h post-reaction, and reached similarly high ThT fluorescence values after 30 h of reaction (Fig. 4). However, increased curves were also observed in the negative controls containing 20, 40 and 80 µM ThT, although these emerged with relatively long lag phases (Fig. 4). These findings suggest that an increase in ThT concentration does not enhance the sensitivity of RT-QuIC assay and provides false positive results.
Determination of appropriate SDS concentration
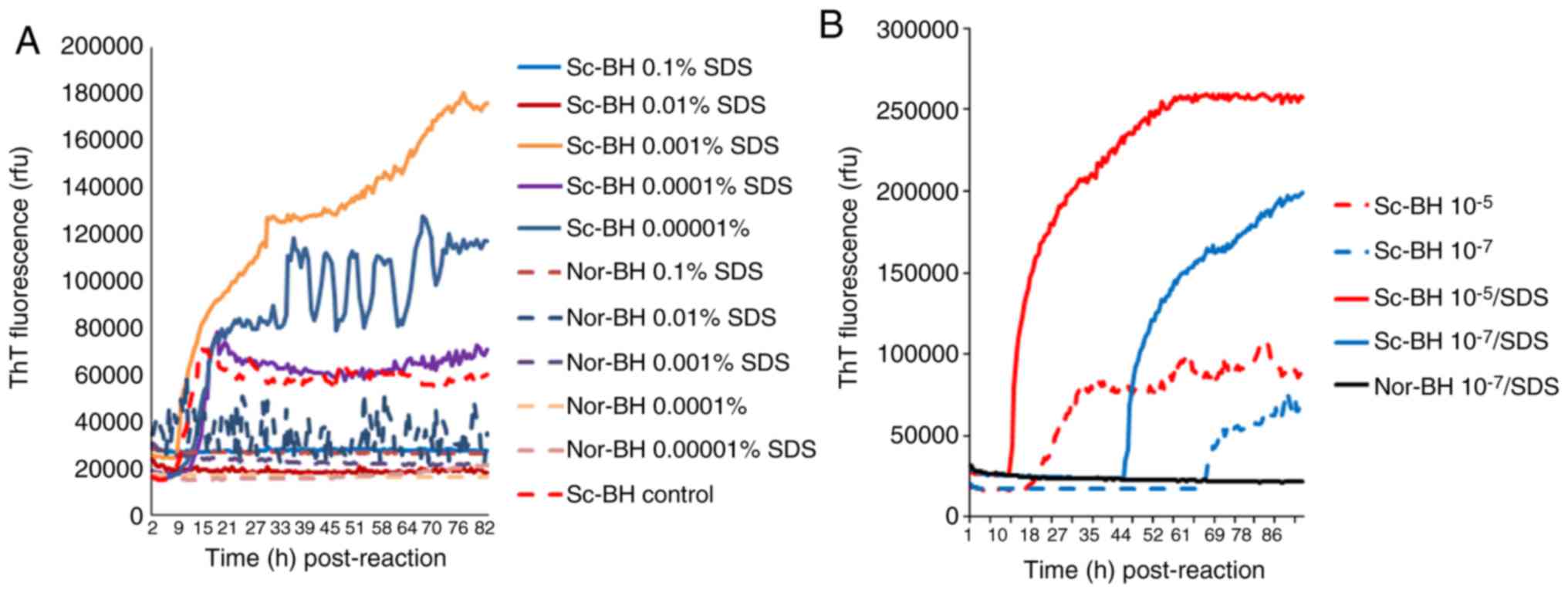
Different concentrations of SDS were added to reactions containing 10−5 diluted Sc-BH, resulting in final concentrations of SDS between 0.1 and 0.00001%. As shown in Fig. 5A, positive reaction curves were observed in the preparations of 0.001, 0.0001 and 0.00001% SDS, among which the reaction using 0.001% SDS exhibited a markedly shorter lag phase and higher ThT value. Positive reactivities were markedly inhibited in the presences of 0.1 and 0.01% SDS, and a false weak positive reaction was observed in the preparation of Nor-BH containing 0.01% of SDS. Furthermore, the effect of 0.001% SDS on the reactivity of different amounts of PrPSc was evaluated. In the conditions of 263K BH diluted for 10−5 and 10−7 times, the presence of 0.001% SDS caused a evidently quicker increase and higher ThT fluorescence values (Fig. 5B).
Determination of appropriate CSF amount
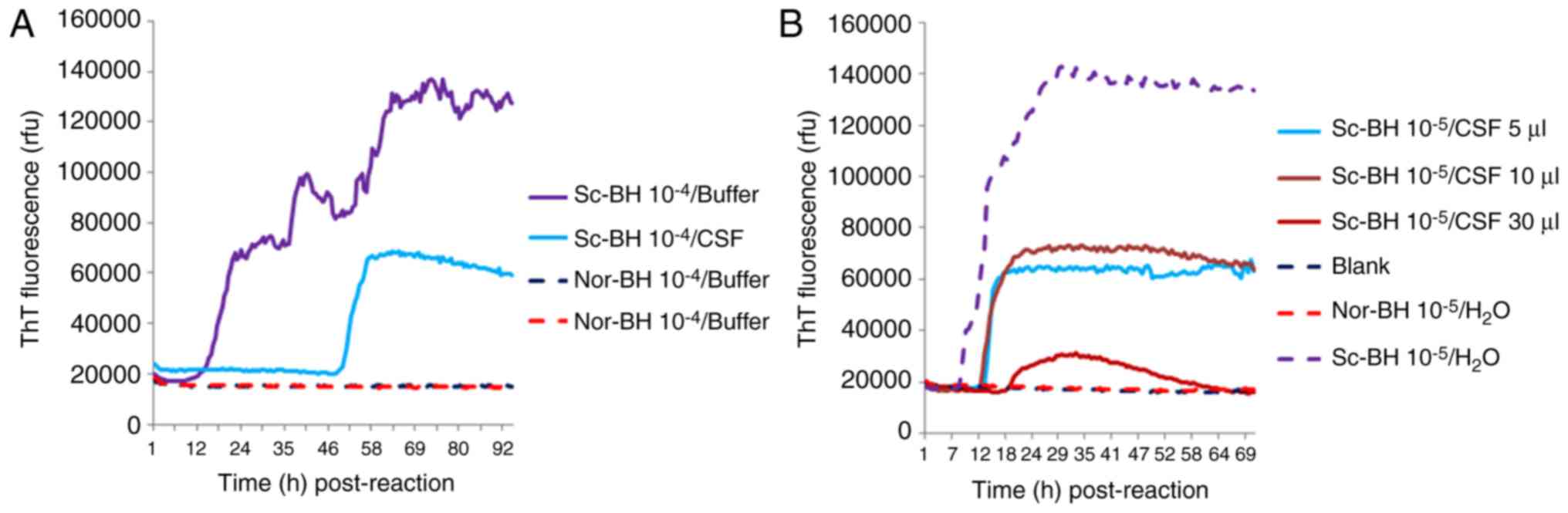
To test the potential influence of human CSF on the reactivity in RT-QuIC assays, 10−5 times diluted Sc-BH was added into 30 µl CSF collected from a non-CJD patient with major indexes in the CSF biochemistry within the normal range. Compared with the same dilution of Sc-BH only, the lag time and fluorescence peak in the reaction of Sc-BH in CSF were evidently longer and lower, respectively (Fig. 6A). Furthermore, 10−5 times diluted Sc-BH was separately mixed with 5, 10 and 30 µl CSF samples. The RT-QuIC assay results demonstrated that the reactions with 5 and 10 µl CSF displayed similar positive reactivities with a much shorter lag phase and higher fluorescence peak in comparison with that of 30 µl CSF (Fig. 6B). Thus, it appears that certain unknown components of human CSF may hamper the RT-QuIC assay; therefore, using relatively large amount of tested CSF in RT-QuIC does not benefit the detecting ability for PrPSc.
Final experimental conditions
Based on the aforementioned data, the experimental conditions of RT-QuIC were set-up, and these involved the final concentrations of 1X PBS, 170 mM NaCl, 1 mM EDTA, 0.01 mM ThT and 0.001% SDS with 10 µg rPrPC and 10 µl CSF in a total reaction volume of 100 µl. The positive control was 10−5 diluted BH collected from 263K-infected hamsters and the negative control was the same dilution of BH obtained from normal hamsters.
Evaluation of the capacity of the optimized RT-QuIC assay in the detection of PrPSc in brain tissues and CSF samples
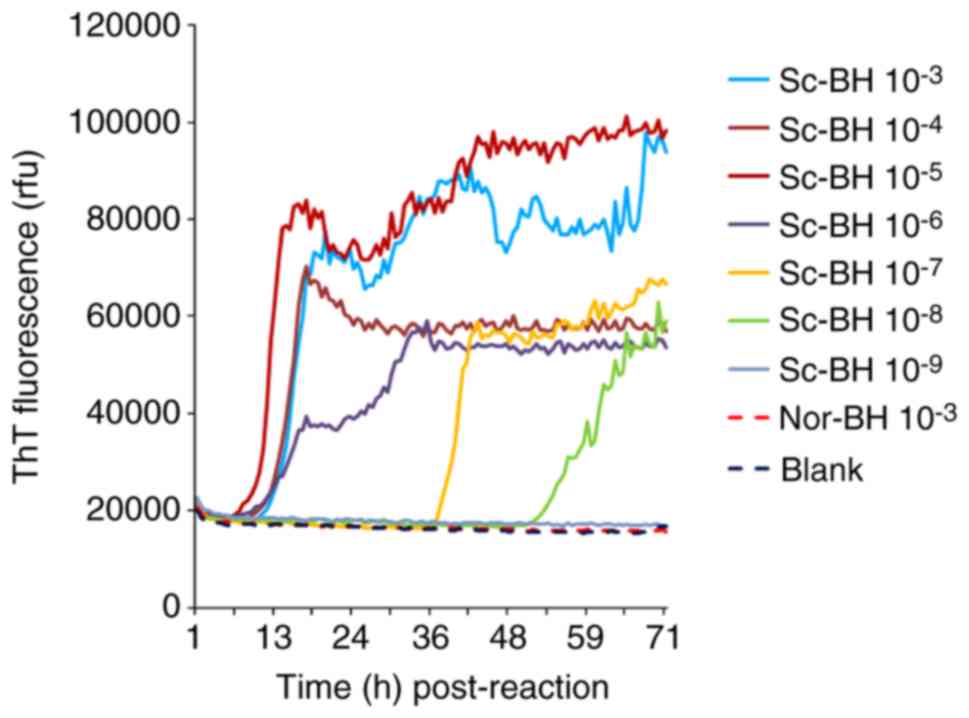
The serially diluted Sc-BHs (10−3 to 10−9) were separately subjected to the optimized RT-QuIC reactions. As shown in Fig. 7, the ThT fluorescence values of the preparations of blank control and Nor-BH were almost unchanged at 90 h post-reaction. By contrast, the ThT values of the reactions containing relatively high amounts of Sc-BH (between 10−3 and 10−6) increased markedly after lag phases of approximately 9-12 h post-reaction. The preparations of 10−7 and 10−8 diluted PrPSc BHs exhibited increased ThT values at 37 and 52 h post-reaction, respectively. No positive result was observed in the reaction of 10−9 diluted PrPSc BH. Furthermore, the ThT fluorescence in the preparations containing high amounts of PrPSc was evidently higher in comparison with that of reactions containing low amounts of PrPSc. The PrPSc detection threshold of the RT-QuIC assay reported in the present study was observed to be 10−8 diluted BH of 263K-infected hamsters.
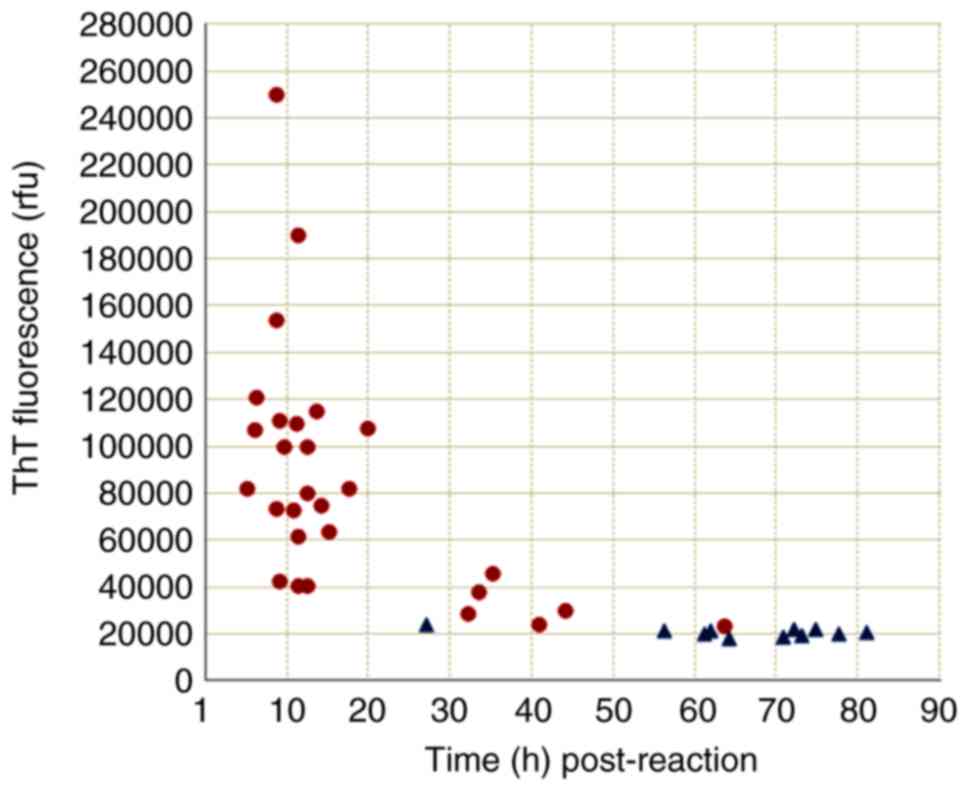
To access the efficacy of the optimized RT-QuIC assay in human sCJD CSF samples, CSF samples from 70 patients that fulfilled the diagnostic criteria for probable sCJD and 48 patients that were classified as non-CJD were assayed. In total, 10 or 30 µl CSF sample from each patient was separately tested. Using 30 µl CSF, 11 (15.71%) samples in the probable sCJD group tested positive, while all tested samples in the non-CJD group were negative. Positive conversion times varied between 8 and 28 h post-reaction, while the peak ThT value varied between 44,000 and 61,000 rfu. Using 10 µl CSF, 40 (57.14%) samples of probable sCJD tested positive, whereas 11 samples of non-CJD also exhibited weakly positive results. Further analysis of the positive reaction patterns in the RT-QuIC assay revealed notably different profiles between the groups of probable sCJD (28 samples) and non-CJD (11 samples). As shown in Fig. 8, the majority of the samples from the probable sCJD group tested positive within 50 h post-reaction (27/28 samples), and exhibited peak ThT values of >25,000 rfu (26/28 samples). By contrast, only 1 case of non-CJD tested positive within 50 h post-reaction, and the peak ThT values of all non-CJD case were <25,000 rfu. These data indicate that applying the CSF RT-QuIC assay with the aforementioned experimental conditions was able to detect substantially more probable sCJD cases, with a significantly shorter lag phase (<50 h post-reaction) and higher ThT fluorescence values (>25,000 rfu).
Discussion
The results of RT-QuIC, which is considered to be a sensitive assay, can be affected by a variety of factors. Under a specific working temperature and vibration speed, the effect of certain variables on the RT-QuIC detection capacity was evaluated in the present study. Similarly to a previous study (12), a type of E. coli-expressed full-length wild-type hamster rPrP was used as substrate in the RT-QuIC assay reported in the current study. In a certain range of rPrPC concentrations, the sensitivity of RT-QuIC exhibits a positive association with the amount of the input rPrPC, however, false positive results are also easily inducible. PrP protein is capable of spontaneous fibrillation in vitro when incubated at certain conditions, for it was reported that agitation alone induces de novo conversion of recombinant prion proteins to b-sheet rich fibrils (24). Using PMCA, a wild-type mouse rPrP protein can be converted into a pathogenic isoform that has the biochemical characteristics of PrPSc in vitro, as well as the typical infectivity on experimental rodents (25). Therefore, careful consideration of the rPrPC amount and of the normal negative control is important, particularly when using different batches of purified rPrPC protein.
Partially denatured rPrPC protein may help to form fibrils in RT-QuIC assay. In addition, using specific concentrations of PBS, NaCl, EDTA and SDS in the working buffer benefits the induction of earlier and higher positive reactions in the RT-QuIC assay. Meanwhile, excessive concentrations of salt and cleaning surfactants lead to a false positive result or inhibit the reactivity of RT-QuIC. ThT is a commonly used chemical for diagnosis of the amyloid structure; however, it is not perfectly specific for amyloid (26). The spectroscopic change of ThT may differ largely, depending on the particular protein and experimental conditions. In the present study, it was also observed that a high concentration of ThT induced evidently false positive reactions in the negative control sample. Thus, a careful balance of the use of salt, cleaning surfactants and ThT concentrations in the reaction buffer is essential for ensuring the sensitivity and specificity of RT-QuIC assay.
The current study also demonstrated that the amount of human CSF used can inhibit the RT-QuIC assay. Human CSF samples with normal biochemical profiles significantly reduced the ThT fluorescence intensity and prolonged the lag time until a positive reaction was detected. These results suggested that there were certain unknown factors in human CSF that inhibited the PrPSc amyloid formation in the RT-QuIC assay. An improved RT-QuIC assay sensitivity was detected in reactions using a relatively low amount of human CSF (5 and 10 µl) under the experimental conditions of the current study. Further studies identifying and removing any inhibitor(s) in human CSF would improve the sensitivity of CSF RT-QuIC assay for sCJD detection.
Using optimized experimental conditions in the present study, samples containing 10−8 dilution of 263K hamster BHwere successfully detected, which indicated a higher PrPSc detection capacity in comparison with that of routine western blot analysis (10−3) and that of PMCA (10−5) using 10% BH of normal hamsters as a substrate (27). A number of CSF samples from probable CJD and non-CJD patients were also preliminarily screened in the present study. Approximately 60% of the CSF samples from the probable sCJD patient group tested positive. Notably, the majority of these results occurred within 50 h post-reaction (median, 11.85 h), and had high peak ThT fluorescence values (median, 77,500 rfu). Certain CSF samples from the non-CJD patient group also exhibited weakly positive reactions, however, these had markedly longer lag phases (median, 70.70 h post-reaction) and evidently reduced peak ThT values (median, 21,000 rfu). On the basis of these data, it is proposed that <50 h post-reaction and/or >25,000 rfu should be used as the cut-off values for positive test results when applying the CSF RT-QuIC assay with the experimental conditions reported in the current study. Certainly, a larger number of samplesare required to further validate the suitability of these criteria in the clinical diagnosis of sCJD.
In conclusion, the present study evaluated the factors which may affect the RT-QuIC assay, and confirmed that RT-QuIC was capable of detecting traces of PrPSc. Application of this assay using CSF samples in the present study revealed the potential use as a pre-mortem tool for the diagnosis of sCJD.
Funding
This work was supported by Chinese National Natural Science Foundation Grants (grant nos. 81630062, 81301429 and 81572048), National Key Research and Development Plan (grant no. 2016YFC1202700) and SKLID Development Grant (grant nos. 2012SKLID102 and 2015SKLID503).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Authors' contributions
KX designed the study, acquired the data and prepared the manuscript; QS prepared the manuscript; JW, CG and BYZ assisted in the RT-QuIC assay; CC performed statistical analysis; WZ and QS prepared the samples; XPD, who was the corresponding author, designed the study and revised the manuscript. All authors read and approved the final manuscript.
Ethics approval and consent to participate
The present study was approved by the Ethical Committee of the National Institute for Viral Disease Control and Prevention (Beijing, China; protocol 2009ZX10004-101). Informed consent was obtained from all patients prior to participation.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no conflict of interest.
Acknowledgments
The authors would like to thank Dr Shelley Robison for English language editing.
References
|
Prusiner SB: Prions Proc Natl Acad Sci USA. 95:13363–13383. 1998. View Article : Google Scholar | |
|
Ladogana A, Puopolo M, Croes EA, Budka H, Jarius C, Collins S, Klug GM, Sutcliffe T, Giulivi A, Alperovitch A, et al: Mortality from Creutzfeldt-Jakob disease and related disorders in Europe, Australia, and Canada. Neurology. 64:1586–1591. 2005. View Article : Google Scholar : PubMed/NCBI | |
|
Chen C and Dong XP: Epidemiological characteristics of human prion diseases. Infect Dis Poverty. 5:472016. View Article : Google Scholar : PubMed/NCBI | |
|
Budka H, Aguzzi A, Brown P, Brucher JM, Bugiani O, Gullotta F, Haltia M, Hauw JJ, Ironside JW, Jellinger K, et al: Neuropathological diagnostic criteria for Creutzfeldt-Jakob disease (CJD) and other human spongiform encephalopathies (prion diseases). Brain Pathol. 5:459–466. 1995. View Article : Google Scholar : PubMed/NCBI | |
|
Borchelt DR, Scott M, Taraboulos A, Stahl N and Prusiner SB: Scrapie and cellular prion proteins differ in their kinetics of synthesis and topology in cultured cells. J Cell Biol. 110:743–752. 1990. View Article : Google Scholar : PubMed/NCBI | |
|
Prusiner SB: Novel properties and biology of scrapie prions. Curr Top Microbiol Immunol. 172:233–257. 1991.PubMed/NCBI | |
|
Manix M, Kalakoti P, Henry M, Thakur J, Menger R, Guthikonda B and Nanda A: Creutzfeldt-Jakob disease: Updated diagnostic criteria, treatment algorithm, and the utility of brain biopsy. Neurosurg Focus. 39:E22015. View Article : Google Scholar : PubMed/NCBI | |
|
Zerr I, Kallenberg K, Summers DM, Romero C, Taratuto A, Heinemann U, Breithaupt M, Varges D, Meissner B, Ladogana A, et al: Updated clinical diagnostic criteria for sporadic Creutzfeldt-Jakob disease. Brain. 132:2659–2668. 2009. View Article : Google Scholar : PubMed/NCBI | |
|
Shi Q, Zhou W, Chen C, Gao C, Xiao K, Wang J, Zhang BY, Wang Y, Zhang F and Dong XP: Quality evaluation for the surveillance system of human prion diseases in China based on the data from 2010 to 2016. Prion. 10:484–491. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Hsich G, Kenney K, Gibbs CJ, Lee KH and Harrington MG: The 14-3-3 brain protein in cerebrospinal fluid as a marker for transmissible spongiform encephalopathies. N Engl J Med. 335:924–930. 1996. View Article : Google Scholar : PubMed/NCBI | |
|
Sanchez-Juan P, Sanchez-Valle R, Green A, Ladogana A, Cuadrado-Corrales N, Mitrová E, Stoeck K, Sklaviadis T, Kulczycki J, Hess K, et al: Influence of timing on CSF tests value for Creutzfeldt-Jakob disease diagnosis. J Neurol. 254:901–906. 2007. View Article : Google Scholar : PubMed/NCBI | |
|
Otto M, Wiltfang J, Cepek L, Neumann M, Mollenhauer B, Steinacker P, Ciesielczyk B, Schulz-Schaeffer W, Kretzschmar HA and Poser S: Tau protein and 14-3-3 protein in the differential diagnosis of Creutzfeldt-Jakob disease. Neurology. 58:192–197. 2002. View Article : Google Scholar : PubMed/NCBI | |
|
Beaudry P, Cohen P, Brandel JP, Delasnerie-Lauprêtre N, Richard S, Launay JM and Laplanche JL: 14-3-3 protein, neuron-specific enolase, and S-100 protein in cerebrospinal fluid of patients with Creutzfeldt-Jakob disease. Dement Geriatr Cogn Disord. 10:40–46. 1999. View Article : Google Scholar : PubMed/NCBI | |
|
Collins S, Boyd A, Fletcher A, Gonzales M, McLean CA, Byron K and Masters CL: Creutzfeldt-Jakob disease: Diagnostic utility of 14-3-3 protein immunodetection in cerebrospinal fluid. J Clin Neurosci. 7:203–208. 2000. View Article : Google Scholar : PubMed/NCBI | |
|
Shi Q, Gao C, Zhou W, Zhang BY, Chen JM, Tian C, Jiang HY, Han J, Xiang NJ, Wang XF, et al: Surveillance for Creutzfeldt-Jakob disease in China from 2006 to 2007. BMC Public Health. 8:3602008. View Article : Google Scholar : PubMed/NCBI | |
|
McGuire LI, Peden AH, Orru CD, Wilham JM, Appleford NE, Mallinson G, Andrews M, Head MW, Caughey B, Will RG, et al: Real time quaking-induced conversion analysis of cerebrospinal fluid in sporadic Creutzfeldt-Jakob disease. Ann Neurol. 72:278–285. 2012. View Article : Google Scholar : PubMed/NCBI | |
|
Atarashi R, Satoh K, Sano K, Fuse T, Yamaguchi N, Ishibashi D, Matsubara T, Nakagaki T, Yamanaka H, Shirabe S, et al: Ultrasensitive human prion detection in cerebrospinal fluid by real-time quaking-induced conversion. Nat Med. 17:175–178. 2011. View Article : Google Scholar : PubMed/NCBI | |
|
Wilham JM, Orru CD, Bessen RA, Atarashi R, Sano K, Race B, Meade-White KD, Taubner LM, Timmes A and Caughey B: Rapid end-point quantitation of prion seeding activity with sensitivity comparable to bioassays. PLoS Pathog. 6:e10012172010. View Article : Google Scholar : PubMed/NCBI | |
|
Wang GR, Shi S, Gao C, Zhang BY, Tian C, Dong CF, Zhou RM, Li XL, Chen C and Dong XP: Alternations of tau protein and its phosphorylated profiles in the experimental hamsters infected by scrapie agents 263K and 139A. Bing Du Xue Bao. 25:202–207. 2009.In Chinese. PubMed/NCBI | |
|
Zhang BY, Tian C, Han J, Gao C, Shi Q, Chen JM, Jiang HY, Zhou W and Dong XP: Establishment of a stable PrP(Sc) panel from brain tissues of experimental hamsters with scrapie strain 263K. Biomed Environ Sci. 22:151–156. 2009. View Article : Google Scholar : PubMed/NCBI | |
|
Zhang J, Chen L, Zhang BY, Han J, Xiao XL, Tian HY, Li BL, Gao C, Gao JM, Zhou XB, et al: Comparison study on clinical and neuropathological characteristics of hamsters inoculated with scrapie strain 263K in different challenging pathways. Biomed Environ Sci. 17:65–78. 2004.PubMed/NCBI | |
|
Gao JM, Gao C, Han J, Zhou XB, Xiao XL, Zhang J, Chen L, Zhang BY, Hong T and Dong XP: Dynamic analyses of PrP and PrP(Sc) in brain tissues of golden hamsters infected with scrapie strain 263K revealed various PrP forms. Biomed Environ Sci. 17:8–20. 2004.PubMed/NCBI | |
|
Zanusso G, Monaco S, Pocchiari M and Caughey B: Advanced tests for early and accurate diagnosis of Creutzfeldt-Jakob disease. Nat Rev Neurol. 12:325–333. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Ladner-Keay CL, Griffith BJ and Wishart DS: Shaking alone induces de novo conversion of recombinant prion proteins to beta-sheet rich oligomers and fibrils. PLoS One. 9:e987532014. View Article : Google Scholar | |
|
Yuan Z, Yang L, Chen B, Zhu T, Hassan MF, Yin X, Zhou X and Zhao D: Protein misfolding cyclic amplification induces the conversion of recombinant prion protein to PrP oligomers causing neuronal apoptosis. J Neurochem. 133:722–729. 2015. View Article : Google Scholar : PubMed/NCBI | |
|
Biancalana M and Koide S: Molecular mechanism of Thioflavin-T binding to amyloid fibrils. Biochim Biophys Acta. 1804.1405–1412. 2010. | |
|
Orru CD, Wilham JM, Vascellari S, Hughson AG and Caughey B: New generation QuIC assays for prion seeding activity. Prion. 6:147–152. 2012. View Article : Google Scholar : PubMed/NCBI |