Introduction
Enteroviruses are a group of small single-strand,
positive-sense RNA viruses in the Enterovirus genus of the
Picornaviridae family (1-3). Infection with certain enteroviruses,
including enterovirus 71 (EV71) and coxsackievirus B, lead to
severe diseases including aseptic meningitis, brainstem
encephalitis, myocarditis and pancreatitis (4,5).
Enterovirus infections are particularly common among children <5
years old, and it is one of the major causative pathogens of hand,
foot and mouth disease (HFMD), which affects millions of children
in the Asia-Pacific region (6,7).
In 2015, there were up to 2 million patients with HFMD, with 129
mortalities reported in China. The clinical presentation of HFMD is
characterized by buccal ulcerative lesions with pain, and skin
rashes on hands and feet without pain (8). A small number of children may
exhibit central nervous system and respiratory system damage,
causing aseptic meningitis, encephalitis, acute flaccid paralysis,
neurogenic pulmonary edema and myocarditis. Critically ill children
progress rapidly, and are at risk of mortality (9). In recent years, there have been a
number of treatments for EV infection, including vaccines,
antivirals and interferon (10,11). Unfortunately, the efficacy of
these treatments is not satisfactory.
The primary active ingredient of Polygonum
cuspidatum is polydatin and resveratrol (RES) (12). Polydatin has exhibited
antitussive, hepatoprotective and anti-shock effects, and the
ability to inhibit platelet aggregation, among other effects
(13). RES is the most valuable
and promising ingredient in P. cuspidatum, and it has
antibacterial, anti-inflammatory, anti-allergic, anti-thrombotic,
anti-oxidant, anti-cancer and anti-mutation effects (14-16). In addition, RES also has antiviral
effects against certain viruses, including influenza virus,
hepatitis C virus, respiratory syncytial virus, Epstein-Barr virus,
African swine fever virus, enterovirus and duck enteritis virus
(17-18).
However, the solubility of RES is poor in aqueous
solution, and it is easily degraded (12,14). Therefore, RES has a low
bioavailability, which results in a limited curative effect.
Nanoparticle (NP) technology may improve the solubility and
therapeutic effects of hydrophobic drugs (19-21). In the present study, in order to
effectively inhibit the replication of EV71 virus, RES-loaded
nanoparticles (RES-NPs) were used, and the protective mechanism of
RES-NPs against rhabdosarcoma (RD) cells infected with EV71 was
investigated.
Materials and methods
Materials
RD cells were purchased from Nanjing Kangxi
Biotechnology Co., Ltd. Dulbecco's modified Eagle's medium (DMEM),
fetal bovine serum (FBS) and collagenase type II were purchased
from Gibco; Thermo Fisher Scientific, Inc. The polyvinylidene
fluoride membranes and enhanced chemiluminescence (ECL) western
blot detection system were purchased from Pierce; Thermo Fisher
Scientific, Inc. The malondial-dehyde (MDA) assay and bicinchoninic
acid (BCA) kits was purchased from Nanjing Jiancheng Bioengineering
Institute. The superoxide dismutase (SOD) assay kit was purchased
from Dojindo Molecular Technologies, Inc. The reactive oxygen
species (ROS) assay kit was obtained from Applygen Technologies,
Inc. Rabbit polyclonal antibodies against GAPDH, protein kinase
R-like endoplasmic reticulum-resident kinase (PERK),
phosphorylated-PERK (p-PERK), eukaryotic initiation factor 2α
(eIF2α), phosphorylated-eIF2α (p-eIF2α), activating transcription
factor 4 (ATF4), glucose regulated protein 78 (GRP78),
transcription factor CCAAT/enhancer binding protein
(C/EBP)-homologous protein (CHOP) and light chain 3A/B (LC3-I/II)
were purchased from Cell Signaling Technology, Inc. Major capsid
protein VP1 (VP1), LC3B (LC3-II), horseradish peroxidase-linked
rabbit anti-mouse IgG and Cy3-labeled goat anti-rabbit IgG were
purchased from Abcam. MTT, RES, apocynin, 4-phenylbutyric acid
(4-PBA) and 3-methyladenine (3-MA) reagents were purchased from
Sigma-Aldrich; Merck KGaA.
Synthesis of RES-NPs
The synthesis of copolymer monomethoxy poly
(ethylene glycol)-b-poly (D,L-lactide) (PEG-PDLLA) was performed as
described previously (22). In
detail, 4 ml RES (1 mg) and PEG-PDLLA (9 mg) solution in
tetrahydrofuran (THF) was slowly added into 10 ml distilled water
at room temperature and stirred; the mixture was dialyzed to remove
THF. RES-NPs were obtained for subsequent use.
Characterization of RES-NPs
The morphology of RES-NPs was observed using
transmission electron microscopy (TEM) as follows: Following
dropping of the sample liquid on a carbon coated copper grid, it
was dyed with 1% uranyl acetate. The size distribution of NPs was
determined by dynamic light scattering (DLS) with a vertically
polarized He-Ne laser (DAWN EOS; Wyatt Technology Corporation).
Zeta potential measurement was performed using Zetasizer Nano-ZS
(Malvern Instruments, Ltd.).
In vitro release
A total of 2 ml PBS (pH 7.4, 1.0 wt % Tween-80)
solution and RES-NPs were added to the dialysis bag (Molecular
weight cutoff=3.5 kDa), and the dialysis bag was placed in 8 ml
release medium and oscillated at 37°C. At intervals of 2 h, 1 ml
buffer solution was removed from the dialysis bag for UV
visualization, and 1 ml fresh buffer solution was added to the
dialysis bag. The calibration curve of RES was obtained based on
the absorbance of RES at 306 nm. Finally, the cumulative release
weight and relative percentage of RES were obtained.
Cytotoxicity test
Cell suspensions were seeded in 96-well plates
(1×105 cells per well, 100 µl per well). The
culture solution containing RES (200 µg/ml) or RES-NPs (RES
concentration, 200 µg/ml) was added to each well, and the
culture plate was placed in an incubator (37°C and 5%
CO2) for 48 h. Following incubation, the supernatant was
discarded and 20 µl MTT solution (5 mg/ml in PBS) was added
to each well. The culture plate was placed in the incubator (37°C
and 5% CO2) and incubated for 4 h prior to removal of
the supernatant. Dimethyl sulfoxide (150 µl) was added to
each of the 96-well plates, and the absorbance at 570 nm was
measured by a microplate reader (Bio-Rad Laboratories, Inc.).
Finally, the 50% cytotoxic concentration (CC50) was
calculated.
Virus isolation and propagation
A total of 4 ml EV71 medium (MOI=2) was prepared as
a virus inoculum. Following culture for 24 h, RD cells were washed
once with PBS, diluted to a density of 1×106 cells, and
incubated with the prepared EV71 medium for 1.5 h at 37°C. To
remove unbound EV71, the cells were washed with fresh medium. Cell
supernatants were collected at 0, 1, 3, 6, 12, 16, 18, 20 and 24 h
after infection, virus titers were determined by cytopathic effect
(CPE), and the median tissue culture infectious dose (TCID50) per
ml was calculated. The results were observed and recorded daily for
5-7 days; CPE was observed under a light microscope (magnification,
×100) and categorized according to the following criteria: i) −: no
cytopathic changes; ii) ±: 1-25% of the cells had pathological
changes; iii) ++: 26-50% of the cells had pathological changes; iv)
+++: 51-75% of the cells had pathological changes; and v) +++:
76-100% of the cells had pathological changes. The number of
positive holes in each row was counted and the virus TCID50 was
calculated using the Reed-Muench method (23) or Karber method (24). If there was no CPE in the negative
control group and the cells grew well, the lowest dilution was 100%
positive and the highest dilution was 100% negative, the test would
be effective.
Antiviral activity assay
Following culture of the RD cells in 96-well plates
for 24 h, 30 µl EV71 (MOI=2) medium and diluted RES-NPs
solution were added and incubated for 1.5 h at 37°C. In order to
remove the virus that was not absorbed, the medium was aspirated.
The cells were washed with PBS and RES (200 µg/ml) or
RES-NPs (RES concentration, 200 µg/ml of each group (doses
were ≤ toxic dose) were added. The CPE of each group was observed
using a light microscope (magnification, ×100) and subjected to MTT
measurement as aforementioned, and finally the half-maximal
inhibitory dose value (IC50) was calculated.
Time course analysis of RES-NPs on EV71
replication
RD cells (3×105 cells/well) were
inoculated in a 96-well plate for 24 h, and the control, RES (200
µg/ml) or RES-NPs (RES concentration=200 µg/ml)
groups were infected with EV71 (MOI=2) for 1.5 h. Cell supernatants
were then collected at different assay times (0, 1, 3, 6, 12, 16,
18, 20 and 24 h) post-infection, and EV71 titers were determined by
calculating the TCID50 values.
Measurement of ROS levels
Dihydroethidium [DHE; Weigras Biotechnology
(Beijing) Co., Ltd], a ROS-level indicative fluorescence probe
(λex=535 nm, λem=610 nm), was used to detect
intracellular superoxide anions. Briefly, RD cells were seeded
in 6-well plates (2×104 cells/well) and
cultured with complete medium for 24 h. Cells were then divided
into different groups and exposed to culture medium (control) or
EV71 or EV71 + RES-NPs (RES concentration=200 µg/ml),
respectively, for 24 h at 37°C. Subsequently, cells were washed
with cold PBS and additionally incubated with fresh medium
containing 10 µM DHE at 37°C in the dark for 20 min to stain
the nuclei. The harvested cells were resuspended in PBS at a
density of 2×107 cells/ml, transferred to a
light-shielded 96-well plate (100 µl cell suspension per
well), followed by determination of DHE intensity using a
fluorescence microplate reader (Bio-Rad Laboratories, Inc.).
Fluorescence images of the cells were captured using an electronic
camera (Olympus Corporation).
Determination of major biochemical
parameters
MDA and SOD levels were measured using MDA and SOD
activity kits, respectively. Briefly, RD cells that were cultured
with complete medium for 24 h were divided into different groups,
and these groups were treated with culture medium (control) or EV71
or EV71 + RES-NPs (RES concentration=200 µg/ml) for 24 h at
37°C. The harvested cells were lysed and the supernatant was
collected by centrifugation at 12,000 × g for 10 min at 4°C.
Protein content in the supernatant was detected using a BCA kit. In
addition, 100 µl supernatant was placed into a centrifuge
tube, and the MDA testing solution (200 µl) was added.
Following mixing, the mixture was boiled for 15 min and cooled to
room temperature, followed by centrifugation at 1,000 × g for 10
min at 4°C. A total of 200 µl prepared supernatant was added
to 96-well plate, and the absorbance was measured at 532 nm using a
microplate reader (Bio-Rad Laboratories, Inc.).
SOD was detected based on its ability to inhibit
super-oxide-mediated reduction. A SOD assay kit (cat. no. S311;
Dojindo Molecular Technologies, Inc.) was used to detect its
activity. The harvested cells were homogenized, and the obtained
homogenate was centrifuged at 12,000 × g for 10 min at 4°C. The
protein content in supernatant was determined using the BCA kit. A
mixture of 20 µl supernatant and 160 µl nitro-blue
tetrazolium chloride/enzymatic working solution was added to a
centrifuge tube, and mixed at 4°C for 5 min. Then, 20 µl
reaction-initiating working solution was added and incubated at
37°C for 30 min. The resultant mixture was detected for its
absorbance at 560 nm using the aforementioned microplate
reader.
Western blot analysis
The cells in each group were cultured in a 37°C, 5%
CO2 incubator for 24 h and the supernatant was
discarded. The cells were lysed with 1X RIPA lysis buffer (Cell
Signaling Technology, Inc.). The cells were then collected in a
centrifuge tube and centrifuged at 12,000 × g for 15 min at 4°C.
The lysates were clarified, and the supernatants fractions were
isolated. Protein concentrations in cells were determined using a
bicinchoninic acid protein assay. In total, 30-50 µg of
protein was loaded and separated by SDS-PAGE on 8-12% gels and then
transferred to a PVDF membrane. After blocking the membrane with 5%
non-fat milk for 1 h at room temperature, the following primary
antibodies were used for western blotting: VP1 (cat. no. ab53977;
1:10,000), PERK (cat. no. 3192; 1:1,000), p-PERK (cat. no. 3179;
1:1,000), eIF2α (cat. no. 9722; 1:1,000), p-eIF2α (cat. no. 3398;
1:1,000), ATF4 (cat. no. 11815; 1:1,000), GRP78 (cat. no. 3183;
1:1000), CHOP (cat. no. 2895; 1:1,000), LC3-I/II (cat. no. 4108;
1:1,000) and GAPDH (cat. no. 5174; 1:1,000). Primary antibodies
were incubated with membranes overnight at 4°C. The membrane was
incubated with horseradish peroxidase-linked rabbit anti-mouse IgG
(cat. no. ab6728; 1:2,000) for 2 h at room temperature. Finally,
the blots were visualized using a chemiluminescence system (Pierce;
Thermo Fisher Scientific, Inc.). GeneTools software 4.1 (Syngene)
was used to quantify the immunoblots.
Immunofluorescence analysis
The cells in each group were cultured in a 37°C, 5%
CO2 incubator for 24 h, the cells were fixed with 4%
paraformaldehyde for 20 min at room temperature and blocked with 1%
bovine serum albumin (cat. no. B2064; Sigma-Aldrich; Merck KGaA)
for 30 min at room temperature. After washing the cells with PBS,
the cells were incubated with primary antibodies against LC3-II
(cat. no. ab48394; 1:400) for 24 h at 4°C. After washing with PBS,
the cells were incubated with Cy3-labeled goat anti-rabbit IgG
(cat. no. ab150077; 1:200) for 60 min at room temperature. DAPI (5
mg/ml) was used to stain all cell nuclei at room temperature for 10
min. A fluorescence microscope (magnification, ×400; Olympus
Corporation) was used to observe the morphology of the cells and
the intensity of the fluorescent signals.
Detection of inflammatory cytokines
RD cells infected with EV71 (MOI=2) were centrifuged
at 1,500 × g for 10 min at 4°C, and the supernatant was collected
at 0, 3, 6, 12 and 24 h after infection. The concentration of
interleukin (IL)-6, IL-8 and tumor necrosis factor-α (TNF-α) in the
supernatant was measured by Milliplex magnetic beads (EMD
Millipore) and the FLEXMAP 3D platform (Luminex Corporation). The
resulting data was analyzed by MILLIPLEX Analyst v5.1 (VigeneTech,
Inc.).
Statistical analysis
All experiments were performed a minimum of 3 times,
and the data were analyzed using GraphPad Prism v5 software
(GraphPad Software, Inc.). All data are presented as the mean ±
standard deviation, and were analyzed using one-way analysis of
variance followed by Tukey's post-hoc test. P<0.05 was
considered to indicate a statistically significant difference.
Results
Preparation of RES-NPs
The THF solution containing RES and PEG-PDLLA was
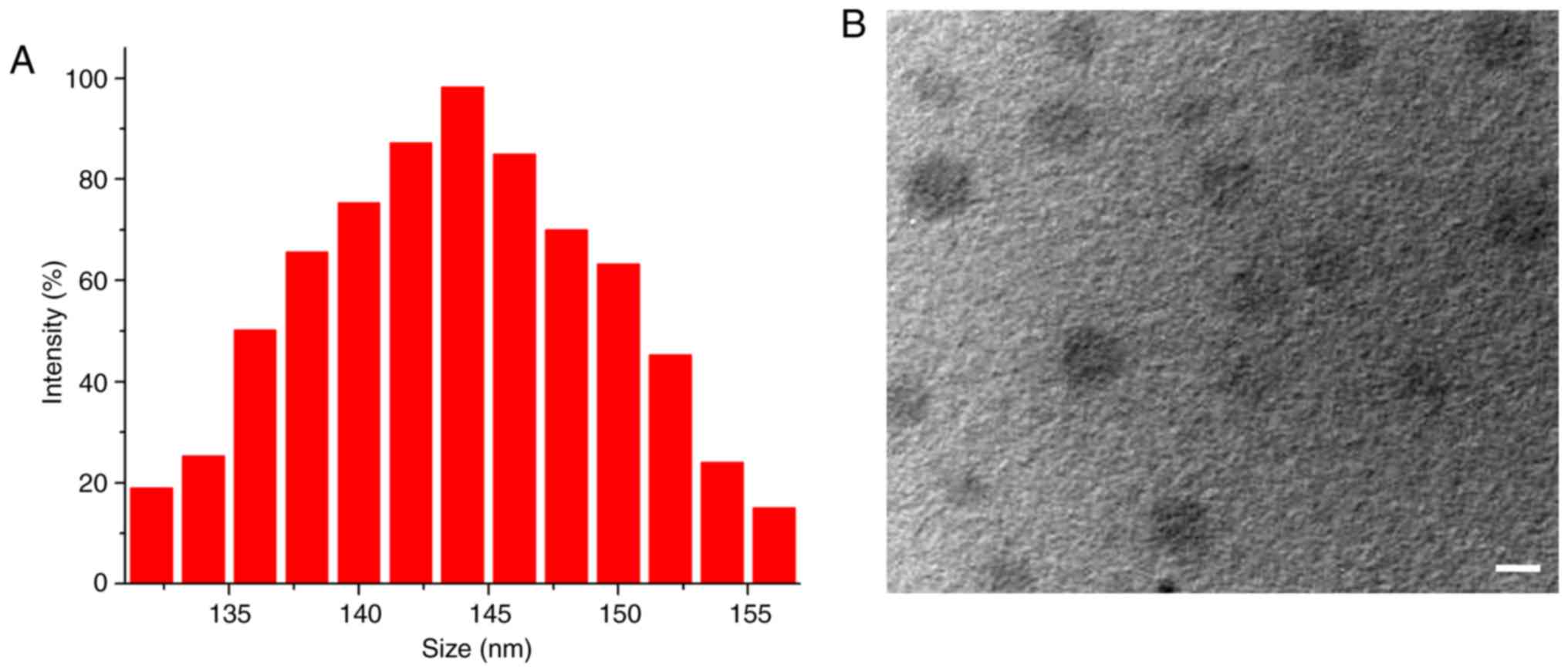
dropped into water, and ideal RES-NPs were obtained. Fig. 1A illustrates the Gaussian
distribution of RES-NP size. The particle size and zeta potential
of the obtained RES-NPs were 144.7 nm (polydispersity index=0.31)
and 0.47±0.012 mV, respectively. TEM data indicated that RES-NPs
were spherical nanoparticles with diameters of ~145 nm (Fig. 1B). All these data indicate that
RES-NPs had small sizes and nearly neutral surface charge,
suggesting that they have potential for practical applications.
Release profile of RES-NPs
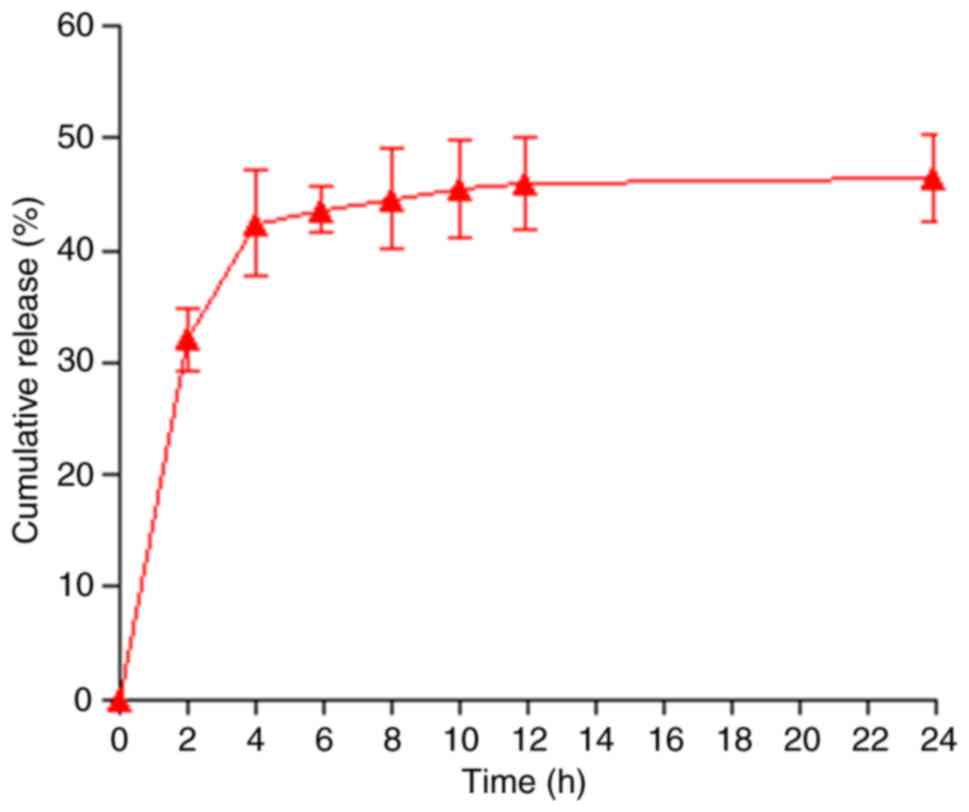
In order to calculate the cumulative release rate of
RES, the released RES were collected at different time points. As
demonstrated in Fig. 2, the
release of RES was rapid during the first 4 h, with a cumulative
release rate of up to 40%, but after 4 h, RES release became slow,
due to the strong hydrophobicity of RES (25).
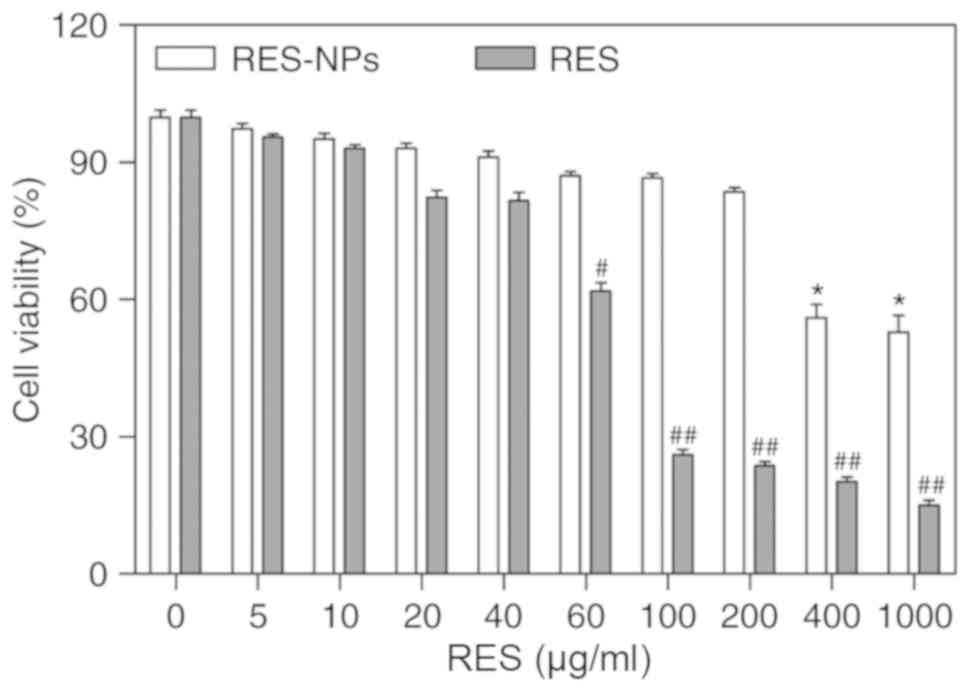
Effect of RES-NPs on viability of RD
cells
To understand the toxicity of RES-NPs and RES on RD
cells, different concentrations of RES-NPs and RES were added to RD
cells and incubated for 48 h to detect cell viability. As indicated
in Fig. 3, RES-NPs and RES were
not significantly toxic to RD cells at 200 and 40 µg/ml,
respectively. However, when the concentration of RES-NPs and RES
was >200 and 40 µg/ml, respectively, the toxicity
increased markedly. The CC50 of RES-NPs and RES were
618.54 and 82.77 µg/ml, respectively.
Antiviral effect of RES-NPs
To understand the association between RES-NPs
treatment time and antiviral efficacy, 2 assays were performed. The
first assay involved the addition of RES-NPs and EV71 to RD cells
concomitantly, and the virus titer was detected after 48 h. The
second assay involved the addition of RES-NPs following EV71
infection of RD cells for 1.5 h, and the virus titer was detected
after 48 h of RES-NPs treatment. As indicated in Fig. 4A, RES-NPs significantly inhibited
the replication of EV71 in a dose-dependent manner in both groups,
and there was no significant difference between the two groups.
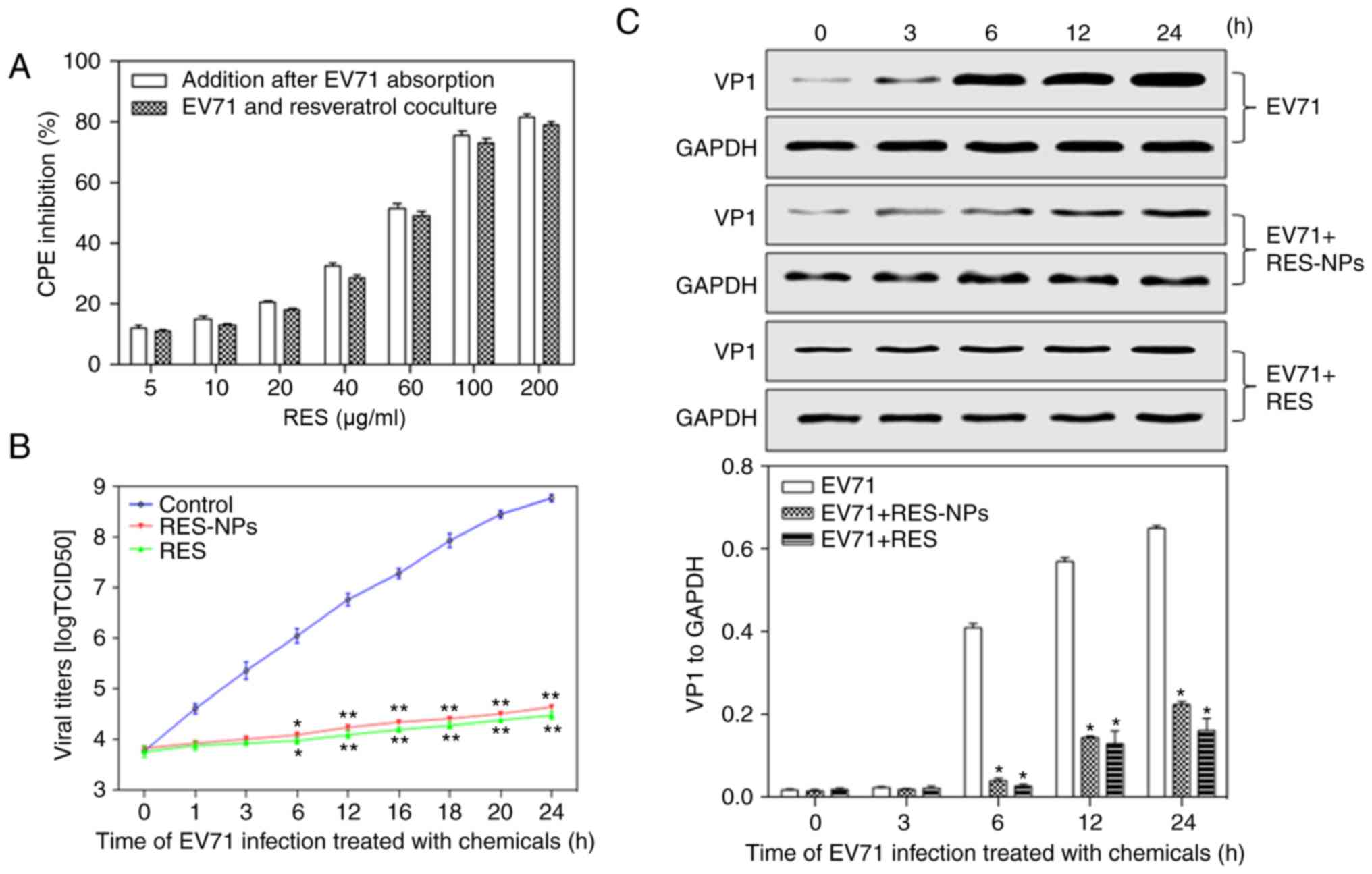 | Figure 4Antiviral effects of RES-NPs on EV71
replication in RD cells. (A) Various doses of RES-NPs were added
with EV71 concomitantly, or following EV71 absorption for 1.5 h.
(B) RES (200 µg/ml) or RES-NPs (RES concentration=200
µg/ml) were added following EV71 absorption for 1.5 h. The
culture supernatants were collected at 0, 1, 3, 6, 12, 16, 18, 20
and 24 h. Data from three repeat experiments are presented as the
mean ± standard deviation. *P<0.05 and **P<0.01
vs. control group. (C) Inhibition of RES or RES-NPs on the
expression of VP1 protein. At 0, 3, 6, 12 and 24 h, RD cell lysates
were subjected to 10% SDS-PAGE and transferred to polyvinylidene
fluoride membranes to determine the expression level of VP1. Data
from three repeat experiments are presented as the mean ± standard
deviation. VP1 levels were normalized with GAPDH.
*P<0.05 vs. EV71. RES, resveratrol; RES-NPs,
resveratrol-loaded nanoparticles; EV71, enterovirus 71; RD,
rhabdosarcoma; VP1, major capsid protein VP1; CPE, cytopathic
effect. |
To additionally understand the time-dependent
antiviral effect of RES or RES-NPs, EV71 was added to RD cells and
incubated for 1.5 h, then added RES (200 µg/ml) or RES-NPs
(RES concentration=200 µg/ml) and incubated for different
times. As demonstrated in Fig.
4B, RES or RES-NPs significantly inhibited EV71 replication
during incubation for 6-24 h. On the basis of this result, the
effect of RES or RES-NPs on VP1 synthesis was additionally
analyzed. The results indicated that VP1 was significantly
decreased in the presence of RES (200 µg/ml) or RES-NPs (RES
concentration=200 µg/ml) at 6, 12 and 24 h, suggesting that
RES or RES-NPs may effectively inhibit viral replication in
EV71-infected RD cells (Fig. 4C).
These results suggested that there was no significant difference in
the antiviral efficacy of RES-NPs (200 µg/ml) and RES (200
µg/ml) within 24 h of stimulation. The reason was that,
although the cumulative release of RES released by RES-NPs was only
~40% at 24 h of stimulation, the sustained release of NPs
maintained a high level of local RES cellular concentration and
prolonged the effective drug action time. Therefore, the antiviral
effect was able to reach the levels observed for the small molecule
RES.
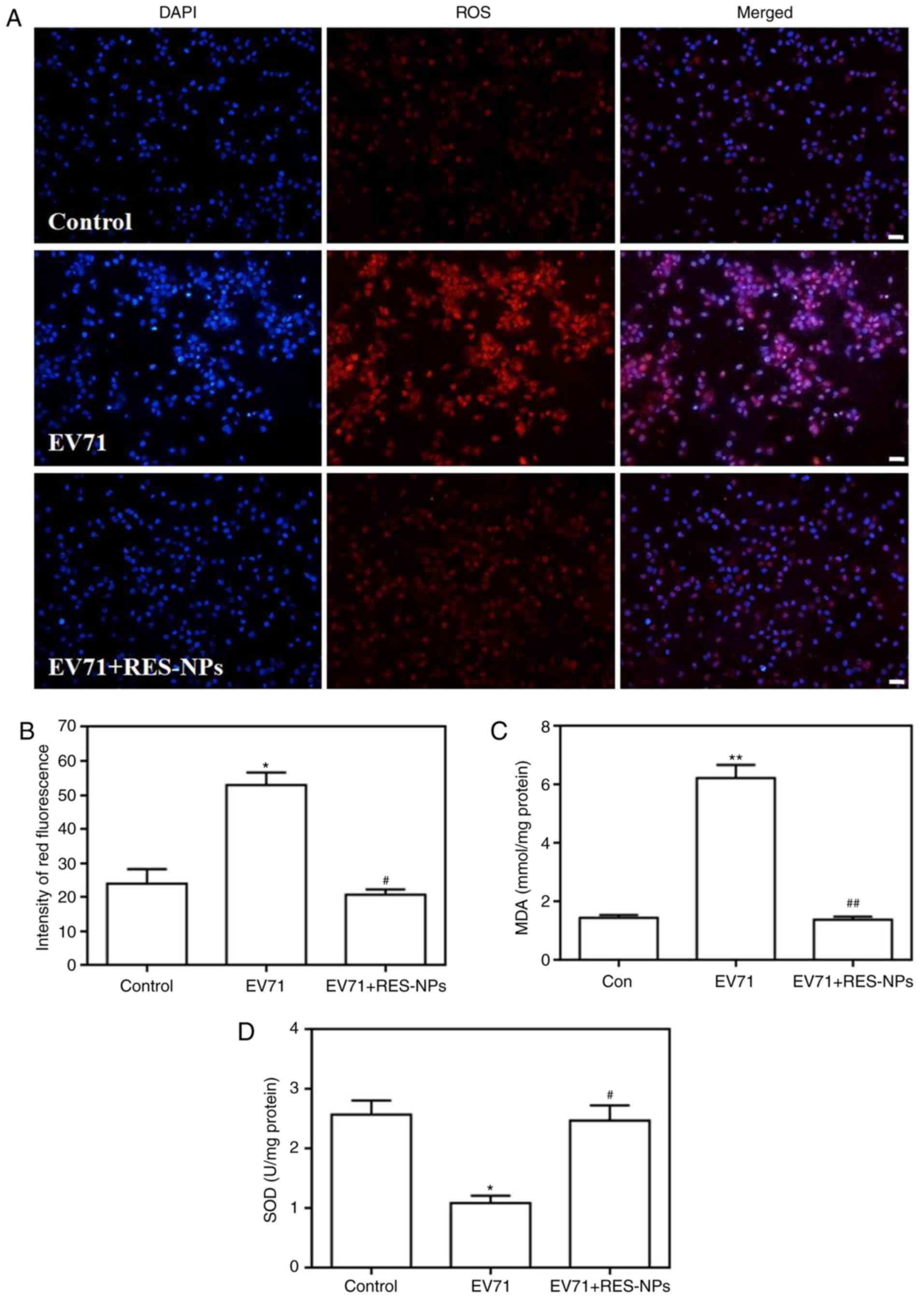
Effect of RES-NPs on EV71-induced ROS
level, MDA content and SOD activity in RD cells
EV71-induced ROS levels were first detected by using
DHE as a fluorescent probe, and the results are presented in
Fig. 5. The images denote that
the RD cells treated with EV71 alone exhibited brighter red
fluorescence signals compared with the control group, suggesting
that EV71 infection may lead to an increase in intracellular ROS
levels. Conversely, the red fluorescence corresponding to the cells
treated with the combination of EV71 and RES-NPs exhibited largely
decreased brightness when compared with those cells treated by EV71
only, demonstrating that RES-NPs may significantly prevent
intracellular ROS production. DHE fluorescence intensity was
quantified, and the data suggested that RES-NPs may completely
inhibit the EV71-induced increase in ROS levels (Fig. 5B).
The data presented in Fig. 5C and D demonstrate that the EV71
infection resulted in a marked increase in the level of MDA, while
leading to a significant decrease in SOD activity; these changes
are markedly associated with the rise of intracellular oxidative
stress levels. In contrast to these observations, RES-NPs (RES
concentration=200 µg/ml) markedly inhibited the increase of
MDA levels and regulated the SOD activity, ensuring that these
indexes remained at normal levels.
Effect of RES-NPs on EV71-induced
endoplasmic reticulum stress (ERS) upregulation in RD cells
The endoplasmic reticulum (ER) is an organelle
responsible for the synthesis and folding of secreted proteins and
transmembrane proteins (26).
When the ER is in a resting state, GRP78 binds to baroreceptors and
maintains the ER homeostasis (26). Viral infection may lead to the
accumulation of misfolded proteins in the ER, which triggers ERS
(27). ERS may contribute to
autophagy via activation of GRP78, PERK, eIF2α, ATF4 and CHOP. In
the present study, EV71 infection upregulated the expression of
p-PERK, p-eIF2α and ATF4 in RD cells at 6, 12 and 24 h (Fig. 6A), but a significant decrease in
their expression in the presence of RES-NPs (RES concentration=200
µg/ml) was observed (Fig.
6B). Concomitantly, RES-NPs failed to inhibit the expression of
p-PERK, p-eIF2α and ATF4 when pretreated with apocynin, an
oxidative stress inhibitor (Fig.
6C). In addition, EV71 infection upregulated the expression of
GRP78 and CHOP in RD cells at 6, 12 and 24 h (Fig. 6D), but a significant decrease in
their expression in the presence of RES-NPs (RES concentration=200
µg/ml) was observed (Fig.
6E). Furthermore, RES-NPs failed to inhibit the expression of
GRP78 and CHOP when pretreated with apocynin (Fig. 6F). These data indicated that the
RES-NPs inhibited the EV71-induced activation of the oxidative
stress-mediated ERS signal pathway.
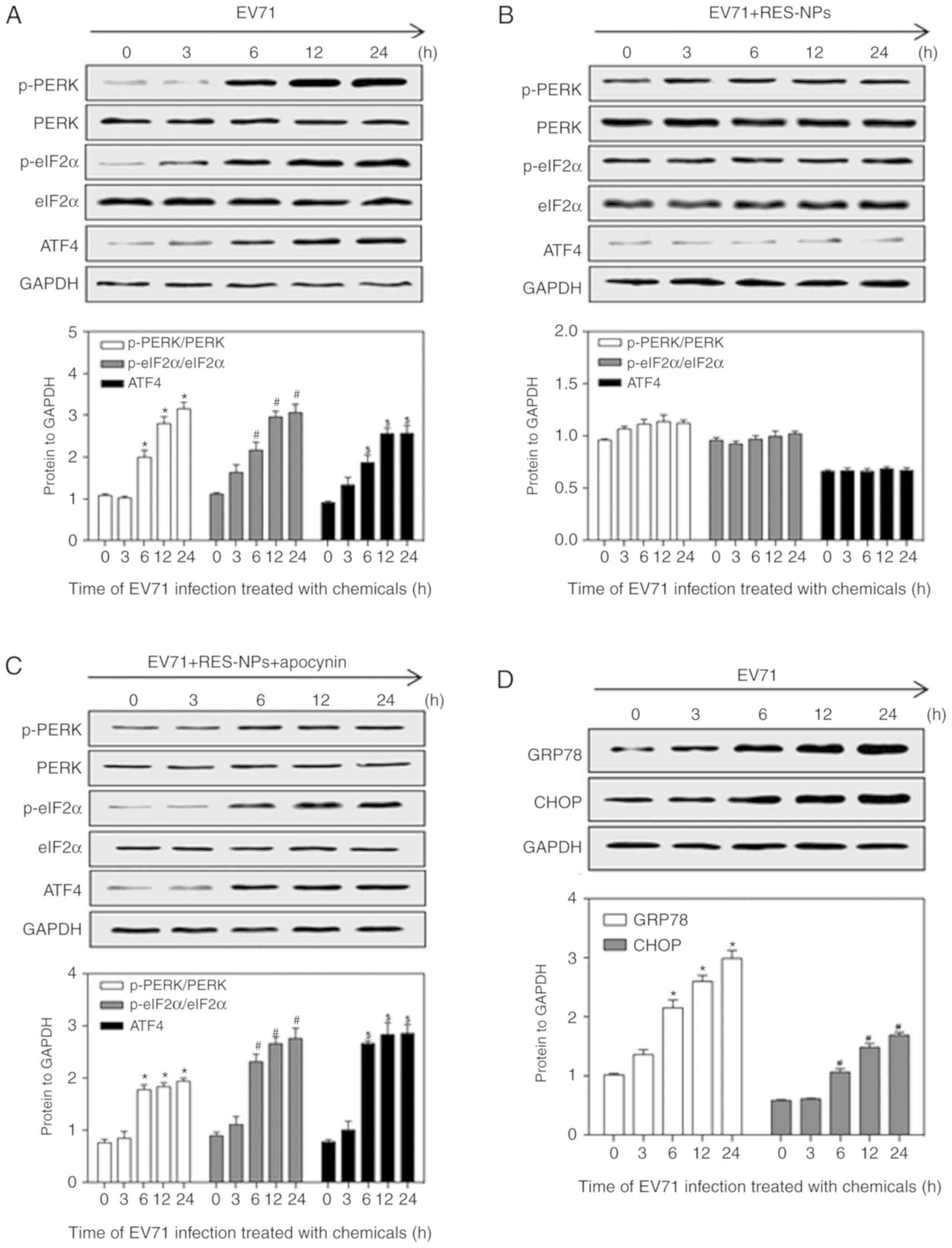 | Figure 6RES-NPs alters the expression levels
of the endoplasmic reticulum stress-associated proteins p-PERK,
PERK, p-eIF2α, eIF2α, ATF4, GRP78 and CHOP in EV71-infected RD
cells. The levels of p-PERK, PERK, p-eIF2α, eIF2α and ATF4 were
measured in RD cells pretreated with (A) culture medium, (B)
RES-NPs (RES concentration, 200 µg/ml) alone or (C) RES-NPs
(RES concentration, 200 µg/ml) + apocynin (300 µM)
for 0, 3, 6, 12 and 24 h using a western blot assay following
protein extraction. p-PERK and p-eIF2α levels were normalized to
PERK and eIF2α, and then normalized to GAPDH. ATF4 levels were
normalized to GAPDH. The levels of GRP78 and CHOP were measured in
RD cells pretreated with (D) culture medium for 0, 3, 6, 12 and 24
h using a western blotting assay following protein extraction. Data
from three repeat experiments are presented as the mean ± standard
deviation. *P<0.05 vs. 0 h p-PERK/PERK group;
#P<0.05 vs. 0 h p-eIF2α/eIF2α group;
$P<0.05 vs. 0 h ATF4 group. The levels of GRP78 and
CHOP were measured in RD cells pretreated with (E) RES-NPs (RES
concentration, 200 µg/ml) alone or (F) RES-NPs (RES
concentration, 200 µg/ml) + apocynin (300 µM) for 0,
3, 6, 12 and 24 h using a western blotting assay following protein
extraction. Data from three repeat experiments are presented as the
mean ± standard deviation. *P<0.05 vs. 0 h
p-PERK/PERK group; #P<0.05 vs. 0 h p-eIF2α/eIF2α
group; $P<0.05 vs. 0 h ATF4 group. RES, resveratrol; RES-NPs,
resveratrol-loaded nanoparticles; PERK, PKR-like endoplasmic
reticulum-resident kinase; p-PERK, phosphorylated PERK; eIF2α,
eukaryotic initiation factor 2α; p-eIF2α, phosphorylated eIF2α;
ATF4, activating transcription factor 4; GRP78, glucose regulated
protein 78, CHOP, transcription factor CCAAT/enhancer binding
protein-homologous protein; EV71, enterovirus 71; RD,
rhabdosarcoma. |
Effect of RES-NPs on EV71-induced
autophagy upregulation in RD cells
Autophagy is a type of programmed cell death through
which the body degrades old proteins and senescent organelles, to
achieve their own metabolic needs and organelle renewal (28-30). Under normal conditions, the level
of autophagy is low. However, in the case of pathogen infection,
autophagy may be rapidly upregulated (31). In the present study, EV71
infection increased the ratio of LC3-II/cytosolic LC3 (LC3-I) in RD
cells at 6, 12 and 24 h (Fig.
7A), but they revealed a significant reduction in the presence
of RES-NPs (RES concentration=200 µg/ml) (Fig. 7B). Furthermore, RES-NPs failed to
inhibit the ratio of LC3-II/LC3-I when pretreated with the 4-PBA
(eIF2α-mediated ERS pathway inhibitor) (Fig. 7C). Concomitantly, red
fluorescent-labeled LC3-II antibody was used to detect the level of
autophagy and DAPI was used to localize the cells. Fig. 7D demonstrated that the RD cells
infected with EV71 alone exhibited brighter red fluorescence
compared with the control group, signifying that the EV71 infection
may lead to an increase in intracellular LC3-II expression.
Conversely, the red fluorescence signals corresponding to the cells
treated with the combination of EV71 and RES-NPs were markedly
decreased when compared with those cells treated with EV71 only,
demonstrating that the use of RES-NPs may significantly prevent
intracellular LC3-II expression. These red fluorescence intensity
data were quantified, and suggested that RES-NPs may almost
completely inhibit the EV71-induced increase in LC3-II expression
(Fig. 7E). These data indicated
that the RES-NPs inhibited the EV71-induced activation of the
ERS-mediated autophagy signaling pathway.
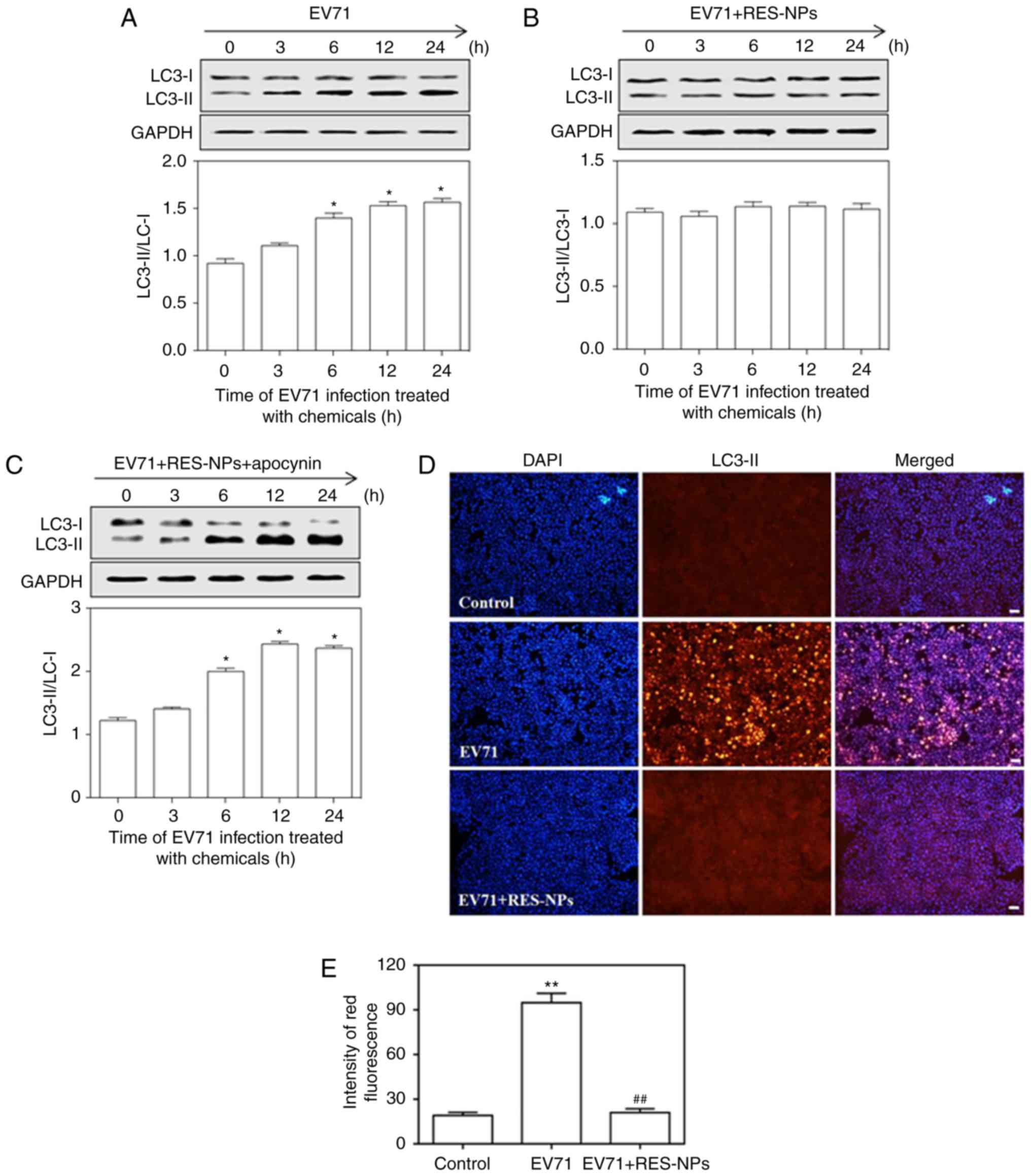 | Figure 7RES-NPs alters the expression levels
of autophagy-associated protein LC3-I in EV71-infected RD cells.
The RD cells were pretreated with (A) culture medium, (B) RES-NPs
(RES concentration, 200 µg/ml) alone or (C) RES-NPs (RES
concentration, 200 µg/ml) + 4-PBA (5 mM) for 0, 3, 6, 12 and
24 h. The expression levels of LC3-I and LC3-II was measured using
a western blotting assay following protein extraction. LC3-I and
LC3-II levels were normalized to GAPDH, and the LC3-II/LC3-I ratio
was quantitatively analyzed. Data from three repeat experiments are
represented as the mean ± standard deviation. *P<0.05
vs. 0 h group. (D) Fluorescence images of RD cells treated with
EV71 alone and the combination of EV71 and RES-NPs (RES
concentration, 200 µg/ml) for 24 h. Scale bar, 50 µm.
(E) Fluorescence intensity of LC3-II expression was quantified.
Data from three repeat experiments are presented as the mean ±
standard deviation. **P<0.01 vs. control;
##P<0.01 vs. EV71. RES, resveratrol; RES-NPs,
resveratrol-loaded nanoparticles; EV71, enterovirus 71; RD,
rhabdosarcoma; PBA, 4-Phenylbutyric acid; LC3,
microtubule-associated proteins 1A/1B-light chain 3. |
Effect of RES-NPs on EV71-induced
inflammatory cytokines secretion and VP1 synthesis in RD cells
Previous data have indicated that the occurrence and
development of HFMD are associated with excessive inflammation and
the immune response in the body (32). Cytokines are a type of
low-molecular-weight protein with a broad range of biological
activities, induced by immunogens and other stimuli (32). They are able to regulate cell
growth and differentiation by regulating their corresponding
receptors, and regulate immune responses (32). It has been identified that
inflammatory cytokines including IL-6, IL-8 and TNF-α were
over-secreted in serum and body fluid of children with HFMD
(33). In the present study,
compared with the control group, EV71-infected cells exhibited
increased secretion levels of IL-6, IL-8 and TNF-α at 6, 12 and 24
h (Fig. 8A-C). The treatment of
RES-NPs (RES concentration=200 µg/ml) suppressed the
secretion of IL-6, IL-8 and TNF-α in EV71-infected RD cells, and no
difference between the control and RES-NPs groups was observed
(Fig. 8A-C). These data suggested
that RES-NPs may effectively inhibit the secretion of inflammatory
cytokines and mitigate the damage of RD cells. Concomitantly,
RES-NPs failed to inhibit the secretion of inflammatory cytokines
when pretreated with apocynin, the oxidative stress inhibitor,
4-PBA, the ERS inhibitor, or 3-MA, the autophagy inhibitor.
Fig. 8D indicated that RES-NPs
may effectively inhibit the synthesis of VP1 and mitigate the virus
replication in EV71-infected RD cells. In addition, RES-NPs failed
to inhibit the synthesis of VP1 when pretreated with the apocynin,
4-PBA or 3-MA (Fig. 8D). These
results suggested that oxidative stress/ERS/autophagy signal
transduction may be involved in the anti-inflammatory effect and
antiviral effect of RES-NPs on EV71-infected RD cells.
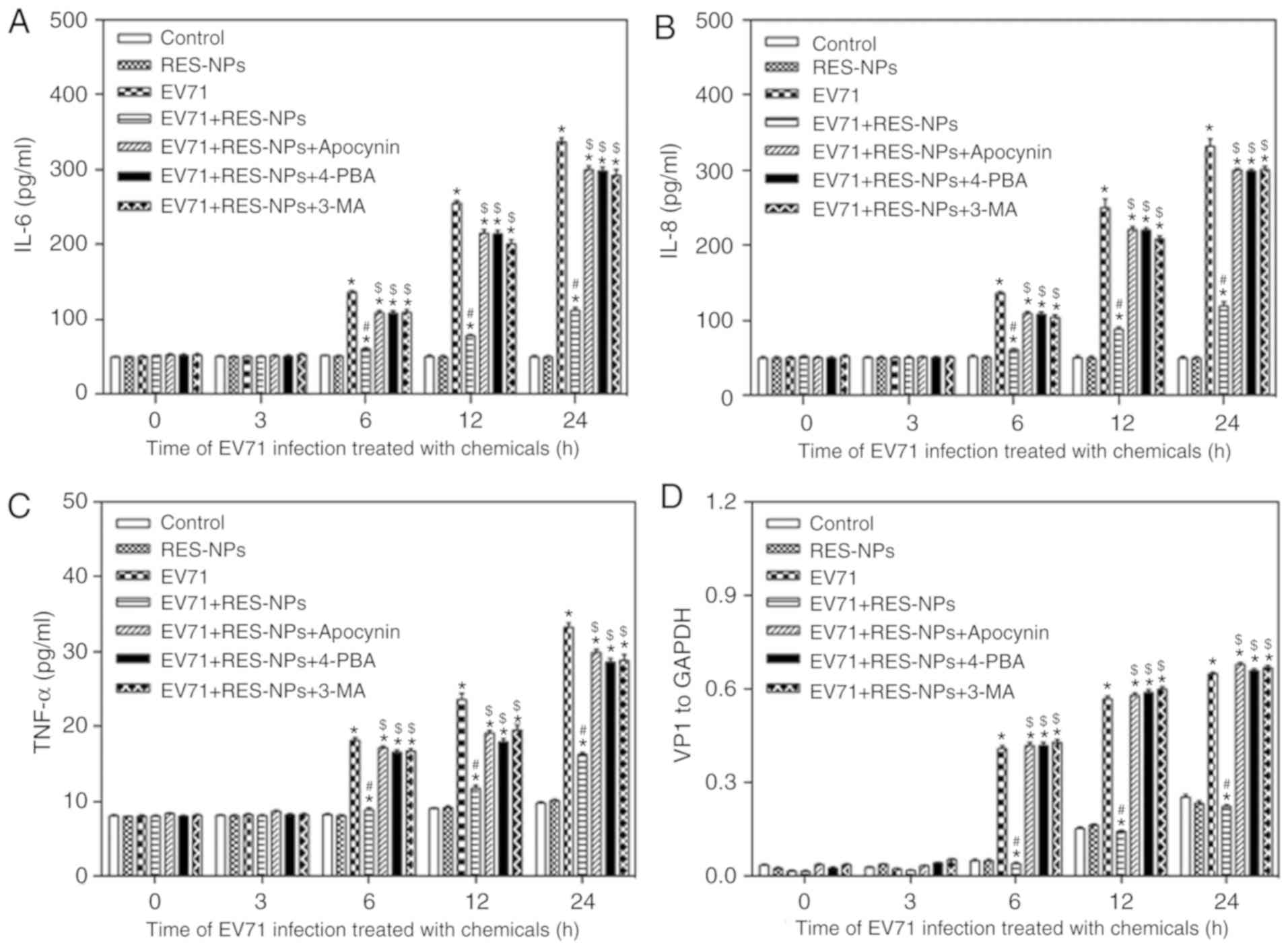 | Figure 8Inhibition of RES-NPs on the several
chemokine releases and expression of VP1 protein in EV71-infected
RD cells at 24 h. Promotion of (A) IL-6, (B) IL-8 and (C) TNF-α
release in RD cells with EV71 infection. The culture supernatants
were harvested at 0, 3, 6, 12 and 24 h after infection to measure
the release of cytokines using the FLEXMAP 3D platform. (D) VP1
levels were measured using a western blotting assay following
protein extraction. VP1 levels were normalized to GAPDH.
Quantitative analysis of the expression of VP1. Data from three
repeat experiments are presented as the mean ± standard deviation.
*P<0.05 vs. control; #P<0.05 vs. EV71;
$P<0.05 vs. EV71 + RES-NPs. IL-6, interleukin-6;
IL-8, interleukin-8; TNF-α, tumor necrosis factor-α; RD,
rhabdosarcoma; EV71, enterovirus 71; RES, resveratrol; RES-NPs,
resveratrol-loaded nanoparticles; Control group, uninfected RD
cells; RES-NPs group, uninfected RD cells treated with RES-NPs;
EV71 group, EV71 infected RD cells; RES-NPs + EV71 group, RD cells
pretreated with RES-NPs (RES concentration, 200 µg/ml) for
1.5 h prior to EV71 infection; VP1, major capsid protein VP1. |
Discussion
EV71 infection may lead to HFMD, aseptic meningitis,
encephalitis and poliomyelitis-like paralysis and other diseases
(34). At present, the
effectiveness of antiviral drugs, vaccines and interferon
treatments in their abilities to completely eradicate the virus is
limited (10,11). It has been observed in clinical
practice that certain traditional Chinese medicines may be
effective in the prevention and treatment of EV71 (35). RES exhibits multiple
pharmacological properties, including anti-inflammatory,
antibacterial, anti-tumor and antifungal effects, lipid metabolism
regulation and cardiovascular protection (14). In addition, RES usually interferes
with the signal transduction pathway in the cell to exert its
extensive anti-viral effects (36,37). As RES is a polyphenolic compound,
it is very polar, with low solubility in water and high solubility
in strong polar solvents including chloroform, methanol, ethanol
and acetone, resulting in low bioavailability. It is unstable to
light and therefore inconvenient for clinical use. NPs use block
copolymers to entrap hydrophobic drugs in hydrophobic cores. NP
systems may increase the solubility and biocompatibility of
hydrophobic drugs. In the present study, an efficient antiviral
effect was obtained by using NPs formulations of RES in EV71
infected-RD cells. However, the antiviral effects and mechanism of
RES-NPs on EV71 have not been widely studied. The present study
confirmed that there was no cytotoxicity of RES-NPs on RD cells at
the dose of 200 µg/ml. Concomitantly, RES-NPs were able to
significantly inhibit CPE caused by EV71, and the inhibitory
activity of RES-NPs on EV71 replication was observed to be
dose-dependent. When RES-NPs (RES concentration=200 µg/ml)
were added simultaneously with or after EV71 absorption for 1.5 h,
the viral titers in cell supernatant decreased, suggesting that
RES-NPs may inhibit EV71 replication in RD cells. These data
suggested that the inhibitory activity of RES-NPs on EV71
replication was time-dependent, but independent of viral attachment
and penetration.
VP1 is an enterovirus-encoded capsid protein that is
exposed on the surface of viral particles and is the major
antigenic determinant of EV71 (38). It is also a specific indicator of
EV71 replication (38). The data
from the present study indicated that the synthesis of VP1 in RD
cells was significantly increased at 6-24 h after EV71 infection,
whereas RES-NPs were able to effectively prevent the synthesis of
VP1 in RD cells infected with EV71. These results suggested that
RES-NPs may exert strong antiviral effects in EV71-infected RD
cells, and the mechanism requires additional exploration.
Rhinovirus and respiratory syncytial virus induce
oxidative stress in cells (39,40). It has been previously identified
that EV71 infection may lead to increased ROS generation (41). MDA is the preferred indicator to
evaluate the ROS content in vivo (42). SOD is an important antioxidant
enzyme that maintains the free radical homeostasis in the human
body (43). The present study
demonstrated that RES-NPs may significantly decrease the production
of ROS and MDA in EV71-infected RD cells. Concomitantly, RES-NPs
may significantly increase the production of SOD in EV71-infected
RD cells.
Cells may affect the homeostasis and function of the
ER in stress conditions, including bacterial or viral infections
(44). A previous research
demonstrated that EV71 infection might cause upregulation and
aberrant redistribution of GRP78 to the cytosol, thereby
facilitating virus replication (45). Under pathological states including
viral infection, ER-localized chaperones, including GRP78, are
activated, and then the unfolded protein response system is
initiated, including the PERK/eIF2α/ATF4 pathway (44). CHOP, also known as growth arrest
and DNA damage-inducing gene 153, is a pro-apoptotic transcription
factor (46) belonging to the
C/EBP transcription factor family that heterodimerizes with other
C/EBPs and may be upregulated by the PERK/eIF2α/ATF4 pathway
(47). In the present study, the
western blot analysis data revealed that exposure to EV71 in RD
cells increased the expression of p-PERK, p-eIF2α, ATF4, GRP78 and
CHOP, indicating that the PERK/eIF2α/ATF4 pathway participated in
EV71-induced virus replication. Following pretreatment with
RES-NPs, the increased expression of p-PERK, p-eIF2α, ATF4, GRP78
and CHOP was significantly inhibited in the EV71-infected RD cells.
Notably, RES-NPs failed to inhibit the expression of p-PERK,
p-eIF2α, ATF4, GRP78 and CHOP when pretreated with apocynin. These
results indicated that RES-NPs may effectively protect the RD cells
against EV71 infection, through inhibiting the activation of the
oxidative stress-mediated ERS pathway and extenuated damage of the
RD cells.
The importance of autophagy in viral infections has
become increasingly apparent and has become an important topic of
study (48). Cell autophagy is
closely associated with the innate immune system of the body. It
may serve roles in the identification and presentation of viral
antigens, activation of natural immune responses and clearance of
the virus. Conversely, the virus may also use the regulation of
autophagy to escape the anti-virus response of the body, and to
maintain their own survival and reproduction (49). There have been a number of studies
on the induction of cell autophagy by viral infection. For example,
membrane cofactor protein is used as a receptor for cell
surface-mediated invasion of various pathogens (50). The successive waves of autophagy
result from distinct molecular pathways and appear to be associated
with anti- and/or pro-measles virus consequences (51). Previous studies have also
identified that viral infection may induce autophagy through the
activation of the ERS signaling pathway (52,53). When autophagy occurs, LC3-I in the
cytoplasm is recruited to the autophagosome to form LC3-II
(49). Therefore, the
LC3-II/LC3-I ratio may be used as an indicator to reflect the level
of autophagy in cells (49). In
the present study, the western blot analysis and immunofluorescence
data revealed that exposure to EV71 in RD cells increased the
LC3-II/LC3-I ratio and LC3-II expression, indicating that the
autophagy pathway participated in EV71 induced virus replication.
Following pretreatment with RES-NPs, the increased LC3-II/LC3-I
ratio and LC3-II expression was significantly inhibited in
EV71-infected RD cells. Notably, RES-NPs failed to inhibit the
LC3-II/LC3-I ratio when pretreated with 4-PBA, the ERS inhibitor.
These data indicated that RES-NPs may effectively protect the host
cells against EV71 infection through inhibition of the activation
of ERS-mediated autophagy pathway and extenuated damage to the RD
cells.
Cytokines serve an important role in the
inflammatory response in HFMD, are involved in the immune and
inflammatory reactions and affect the intensity and duration of the
reaction (33). The investigation
of cytokines has identified that there are >200 types of human
cytokines, including IL-6, IL-8 and TNF-α (54,55). The results from the present study
suggested that the secretion of IL-6, IL-8 and TNF-α significantly
increased in EV71 infected RD cells. Concomitantly, the western
blot analysis results suggested that the expression of VP1 was
significantly increased in EV71-infected RD cells. Following
pretreatment with RES-NPs, VP1 expression levels and the secretion
of IL-6, IL-8 and TNF-α were observed to be decreased in
EV71-infected RD cells. These data indicated that RES-NPs may
effectively inhibit the inflammatory reaction and virus replication
in EV71-infected RD cells. Notably, 3 inhibitors targeting
oxidative stress, ERS or autophagy inhibited the anti-inflammatory
and antiviral effects of RES-NPs on EV71-infected RD cells,
suggesting that the RES-NPs-induced anti-inflammatory and antiviral
effects were closely associated with the oxidative
stress/ERS/autophagy pathway.
In conclusion, the present study demonstrated that
RES-NPs inhibited viral replication and the inflammatory reaction
of EV71-infected RD cells by inhibiting the synthesis of VP1
protein and the secretion of cytokines, and that RES-NPs served
their role through inhibiting the oxida-tive stress-mediated
ERS/autophagy pathway. These results indicate that RES-NPs may be a
feasible alternative strategy for the treatment of EV71 infection.
The underlying mechanism requires additional in vitro and
in vivo studies, in order to examine the oxidative
stress-mediated ERS/autophagy pathway.
Acknowledgments
The authors would like to thank Mr. Ming Liu of
Changchun Institute of Applied Chemistry, Chinese Academy of
Sciences for offering technical guidance for the synthesis of
nanoparticles.
Funding
The present study was supported by a grant from the
Jilin Provincial Science and Technology Department in China (grant
no. 20130206040SF).
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
ND and FW conceived and designed the study. XL, WB,
BW, GX and FW performed the experiments. ND, XL, WB, BW and GX
wrote the paper. FW reviewed and edited the manuscript. All authors
read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Lin JY and Shih SR: Cell and tissue
tropism of enterovirus 71 and other enteroviruses infections. J
Biomed Sci. 21:182014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zhou ZM, Xu Y, Hu CS, Pan QJ and Wei JJ:
Epidemiological features of hand, foot and mouth disease during the
period of 2008-14 in Wenzhou, China. J Trop Pediatr. 63:182–188.
2017.
|
|
3
|
Garmaroudi FS, Marchant D, Hendry R, Luo
H, Yang D, Ye X, Shi J and McManus BM: Coxsackievirus B3
replication and pathogenesis. Future Microbiol. 10:629–653. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Huang YT, Liao JT, Yen LC, Chang YK, Lin
YL and Liao CL: Japanese encephalitis virus replicon-based vaccine
expressing enterovirus-71 epitope confers dual protection from
lethal challenges. J Biomed Sci. 22:742015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Gaaloul I, Riabi S, Harrath R, Hunter T,
Hamda KB, Ghzala AB, Huber S and Aouni M: Coxsackievirus B
detection in cases of myocarditis, myopericarditis, pericarditis
and dilated cardiomy-opathy in hospitalized patients. Mol Med Rep.
10:2811–2818. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Lee KY: Enterovirus 71 infection and
neurological complications. Korean J Pediatr. 59:395–401. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wang Y, Zou G, Xia A, Wang X, Cai J, Gao
Q, Yuan S, He G, Zhang S, Zeng M and Altmeyer R: Enterovirus 71
infection in children with hand, foot, and mouth disease in
Shanghai, China: Epidemiology, clinical feature and diagnosis.
Virol J. 12:832015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Esposito S and Principi N: Hand, foot and
mouth disease: Current knowledge on clinical manifestations,
epidemiology, aetiology and prevention. Eur J Clin Microbiol Infect
Dis. 37:391–398. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yang ZY, Chen XQ, Sun D and Wei D:
Mortality in children with severe hand, foot and mouth disease in
Guangxi, China. Indian Pediatr. 55:137–139. 2018. View Article : Google Scholar
|
|
10
|
Huang X, Zhang X, Wang F, Wei H, Ma H, Sui
M, Lu J, Wang H, Dumler JS, Sheng G and Xu B: Clinical efficacy of
therapy with recombinant human interferon α1b in hand, foot, and
mouth disease with enterovirus 71 infection. PLoS One.
11:e01489072016. View Article : Google Scholar
|
|
11
|
Zhu Z, Ye X, Ku Z, Liu Q, Shen C, Luo H,
Luan H, Zhang C, Tian S, Lim C, et al: Transcutaneous immunization
via rapidly dissolvable microneedles protects against
hand-foot-and-mouth disease caused by enterovirus 71. J Control
Release. 243:291–302. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Jiewei T, Lei W, Xiufeng L, Heming Z,
Xiaoguang L, Haiyan F and Yongqiang T: Microbial transformation of
resveratrol by endophyte Streptomyces sp A12 isolated from
Polygonum cuspidatum. Nat Prod Res. 32:2343–2346. 2018. View Article : Google Scholar
|
|
13
|
Zhao G, Jiang K, Wu H, Qiu C, Deng G and
Peng X: Polydatin reduces staphylococcus aureus lipoteichoic
acid-induced injury by attenuating reactive oxygen species
generation and TLR2-NFκB signaling. J Cell Mol Med. 21:2796–2808.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Heo JR, Kim SM, Hwang KA, Kang JH and Choi
KC: Resveratrol induced reactive oxygen species and endoplasmic
reticulum stress-mediated apoptosis, and cell cycle arrest in the
A375SM malignant melanoma cell line. Int J Mol Med. 42:1427–1435.
2018.PubMed/NCBI
|
|
15
|
Huang YT, Chen YY, Lai YH, Cheng CC, Lin
TC, Su YS, Liu CH and Lai PC: Resveratrol alleviates the
cytotoxicity induced by the radiocontrast agent, ioxitalamate, by
reducing the production of reactive oxygen species in HK-2 human
renal proximal tubule epithelial cells in vitro. Int J Mol Med.
37:83–91. 2016. View Article : Google Scholar :
|
|
16
|
Li K, Li Y, Mi J, Mao L, Han X and Zhao J:
Resveratrol protects against sodium nitroprusside induced nucleus
pulposus cell apoptosis by scavenging ROS. Int J Mol Med.
41:2485–2492. 2018.PubMed/NCBI
|
|
17
|
Zhang L, Li Y, Gu Z, Wang Y, Shi M, Ji Y,
Sun J, Xu X, Zhang L, Jiang J and Shi W: Resveratrol inhibits
enterovirus 71 replication and pro-inflammatory cytokine secretion
in rhabdosarcoma cells through blocking IKKs/NF-κB signaling
pathway. PLoS One. 10:e01168792015. View Article : Google Scholar
|
|
18
|
Zhao X, Xu J, Song X, Jia R, Yin Z, Cheng
A, Jia R, Zou Y, Li L, Yin L, et al: Antiviral effect of
resveratrol in ducklings infected with virulent duck enteritis
virus. Antiviral Res. 130:93–100. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li K, Liu Y, Zhang S, Xu Y, Jiang J, Yin
F, Hu Y, Han B, Ge S, Zhang L and Wang Y: Folate receptor-targeted
ultrasonic PFOB nanoparticles: Synthesis, characterization and
application in tumor-targeted imaging. Int J Mol Med. 39:1505–1515.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Jiang X, Zhong Y, Zheng L and Zhao J:
Nano-hydroxyapatite/collagen film as a favorable substrate to
maintain the phenotype and promote the growth of chondrocytes
cultured in vitro. Int J Mol Med. 41:2150–2158. 2018.PubMed/NCBI
|
|
21
|
Wu L, Chen M, Mao H, Wang N, Zhang B, Zhao
X, Qian J and Xing C: Albumin-based nanoparticles as
methylprednisolone carriers for targeted delivery towards the
neonatal Fc receptor in glomerular podocytes. Int J Mol Med.
39:851–860. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Xu C, Peng Y, Zhang Q, Xu XP, Kong XM and
Shi WF: USP4 positively regulates RLR-induced NF-κB activation by
targeting TRAF6 for K48-linked deubiquitination and inhibits
enterovirus 71 replication. Sci Rep. 8:134182018. View Article : Google Scholar
|
|
23
|
Hamilton MA, Russo RC and Thurston RV:
Trimmed Spearman-Karber method for estimating median lethal
concentrations in toxicity bioassays. Environ Sci Technol.
11:714–719. 1977. View Article : Google Scholar
|
|
24
|
Reed LJ and Muench H: A simple method of
estimating fifty percent endpoints. Am J Epidemiol. 27:493–497.
1938. View Article : Google Scholar
|
|
25
|
Li J, Zhou Y, Zhang W, Bao C and Xie Z:
Relief of oxidative stress and cardiomyocyte apoptosis by using
curcumin nanoparticles. Collolds Surf B Biointerfaces. 153:174–182.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Song S, Tan J, Miao Y and Zhang Q:
Crosstalk of ER stress-mediated autophagy and ER-phagy: Involvement
of UPR and the core autophagy machinery. J Cell Physiol.
233:3867–3874. 2018. View Article : Google Scholar
|
|
27
|
Fraser JE, Wang C, Chan KW, Vasudevan SG
and Jans DA: Novel dengue virus inhibitor 4-HPR activates ATF4
independent of protein kinase R-like endoplasmic reticulum kinase
and elevates levels of eIF2α phosphorylation in virus infected
cells. Antiviral Res. 130:1–6. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Thomas M, Davis T, Loos B, Sishi B,
Huisamen B, Strijdom H and Engelbrecht AM: Autophagy is essential
for the maintenance of amino acids and ATP levels during acute
amino acid starvation in MDAMB231 cells. Cell Biochem Funct.
36:65–79. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Lekli I, Haines DD, Balla G and Tosaki A:
Autophagy: An adaptive physiological countermeasure to cellular
senescence and ischaemia/reperfusion-associated cardiac
arrhythmias. J Cell Mol Med. 21:1058–1072. 2017. View Article : Google Scholar
|
|
30
|
Schroeder S, Zimmermann A,
Carmona-Gutierrez D, Eisenberg T, Ruckenstuhl C, Andryushkova A,
Pendl T, Harger A and Madeo F: Metabolites in aging and autophagy.
Microb Cell. 1:110–114. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Deretic V and Klionsky DJ: Autophagy and
inflammation: A special review issue. Autophagy. 14:179–180. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Shao P, Wu X, Li H, Wu Z, Yang Z and Yao
H: Clinical significance of inflammatory cytokine and chemokine
expression in hand, foot and mouth disease. Mol Med Rep.
15:2859–2866. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Shang W, Qian S, Fang L, Han Y and Zheng
C: Association study of inflammatory cytokine and chemokine
expression in hand foot and mouth disease. Oncotarget.
8:79425–79432. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Yi EJ, Shin YJ, Kim JH, Kim TG and Chang
SY: Enterovirus 71 infection and vaccines. Clin Exp Vaccine Res.
6:4–14. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wang M, Tao L and Xu H: Chinese herbal
medicines as a source of molecules with anti-enterovirus 71
activity. Chin Med. 11:22016. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Shi Y, Li Y, Huang C, Ying L, Xue J, Wu H,
Chen Z and Yang Z: Resveratrol enhances HBV replication through
activating Sirt1-PGC-1α-PPARα pathway. Sci Rep. 6:247442016.
View Article : Google Scholar
|
|
37
|
Liu T, Zang N, Zhou N, Li W, Xie X, Deng
Y, Ren L, Long X, Li S, Zhou L, et al: Resveratrol inhibits the
TRIF-dependent pathway by upregulating sterile alpha and armadillo
motif protein, contributing to anti-inflammatory effects after
respiratory syncytial virus infection. J Virol. 88:4229–4236. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Li X, Huang Y, Sun M, Ji H, Dou H, Hu J,
Yan Y, Wang X and Chen L: Honeysuckle-encoded microRNA2911 inhibits
enterovirus 71 replication via targeting VP1 gene. Antiviral Res.
152:117–123. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Mihaylova VT, Kong Y, Fedorova O, Sharma
L, Dela Cruz CS, Pyle AM, Iwasaki A and Foxman EF: Regional
differences in airway epithelial cells reveal tradeoff between
defense against oxidative stress and defense against rhinovirus.
Cell Rep. 24:3000–3007.e3. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Griffiths C, Drews SJ and Marchant DJ:
Respiratory syncytial virus: Infection, detection, and new options
for prevention and treatment. Clin Microbiol Rev. 30:277–319. 2017.
View Article : Google Scholar :
|
|
41
|
Cheng ML, Weng SF, Kuo CH and Ho HY:
Enterovirus 71 induces mitochondrial reactive oxygen species
generation that is required for efficient replication. PLoS One.
9:e1132342014. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Guo S, Yao Q, Ke Z, Chen H, Wu J and Liu
C: Resveratrol attenuates high glucose-induced oxidative stress and
cardio-myocyte apoptosis through AMPK. Mol Cell Endocrinol.
412:85–94. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Yang J, Yin HS, Cao YJ, Jiang ZA, Li YJ,
Song MC, Wang YF, Wang ZH, Yang R, Jiang YF, et al: Arctigenin
attenuates ischemia/reperfusion induced ventricular arrhythmias by
decreasing oxidative stress in rats. Cell Physiol Biochem.
49:728–742. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Hu DD, Mai JN, He LY, Li PQ, Chen WX, Yan
JJ, Zhu WD, Deng L, Wei D, Liu DH, et al: Glucocorticoids prevent
entero-virus 71 capsid protein VP1 induced calreticulin surface
exposure by alleviating neuronal ER stress. Neurotox Res.
31:204–217. 2017. View Article : Google Scholar
|
|
45
|
Jheng JR, Wang SC, Jheng CR and Horng JT:
Enterovirus 71 induces dsRNA/PKR-dependent cytoplasmic
redistribution of GRP78/BiP to promote viral replication. Emerg
Microbes Infect. 5:e232016.PubMed/NCBI
|
|
46
|
Hosomi S, Grootjans J, Huang YH, Kaser A
and Blumberg RS: New insights into the regulation of natural-killer
group 2 member D (NKG2D) and NKG2D-ligands: Endoplasmic reticulum
stress and CEA-related cell adhesion molecule 1. Front Immunol.
18:13242018. View Article : Google Scholar
|
|
47
|
Zhong F, Xie J, Zhang D, Han Y and Wang C:
Polypeptide from chlamys farreri suppresses ultraviolet-B
irradiation-induced apoptosis through restoring ER redox
homeostasis, scavenging ROS generation, and suppressing the
PERK-eIF2a-CHOP pathway in HaCaT cells. J Photochem Photobiol B.
151:10–16. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Jung KI, Pyo CW and Choi SY: Influenza A
virus-induced autophagy contributes to enhancement of virus
infectivity by SOD1 downregulation in alveolar epithelial cells.
Biochem Biophys Res Commun. 498:960–966. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Wei Y, Cao XN, Tang XL, Shen LJ, Lin T, He
DW, Wu SD and Wei GH: Urban fine particulate matter (PM2.5)
exposure destroys blood-testis barrier (BTB) integrity through
excessive ROS-mediated autophagy. Toxicol Mech Methods. 28:302–319.
2018. View Article : Google Scholar
|
|
50
|
Richetta C, Grégoire IP, Verlhac P, Azocar
O, Baguet J, Flacher M, Tangy F, Rabourdin-Combe C and Faure M:
Sustained autophagy contributes to measles virus infectivity. PLoS
Pathog. 9:e10035992013. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Rozières A, Viret C and Faure M: Autophagy
in measles virus infection. Viruses. 9:pii: E359. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Yan Y, Liu S, Li M, Zhao Y, Shao X, Hang M
and Bu X: Recombinant Newcastle disease virus expressing human
IFN-λ1 (rL-hIFN-λ1)-induced apoptosis of A549 cells is connected to
endoplasmic reticulum stress pathways. Thorac Cancer. 9:1437–1452.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Dash S, Chava S, Aydin Y, Chandra PK,
Ferraris P, Chen W, Balart LA, Wu T and Garry RF: Hepatitis C virus
infection induces autophagy as a prosurvival mechanism to alleviate
hepatic ER-stress response. Viruses. 8:pii: E150. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Lee JY, Son M, Kang JH and Choi UY: Serum
interleukin-6 levels as an indicator of aseptic meningitis among
children with enterovirus 71-induced hand, foot and mouth disease.
Postgrad Med. 130:258–263. 2018. View Article : Google Scholar
|
|
55
|
Zhu L, Li W, Qi G, Liu N, Sheng L, Shang L
and Qi B: The immune mechanism of intestinal tract Toll-like
receptor in mediating EV71 virus type severe hand-foot-and-mouth
disease and the MAPK pathway. Exp Ther Med. 13:2263–2266. 2017.
View Article : Google Scholar : PubMed/NCBI
|