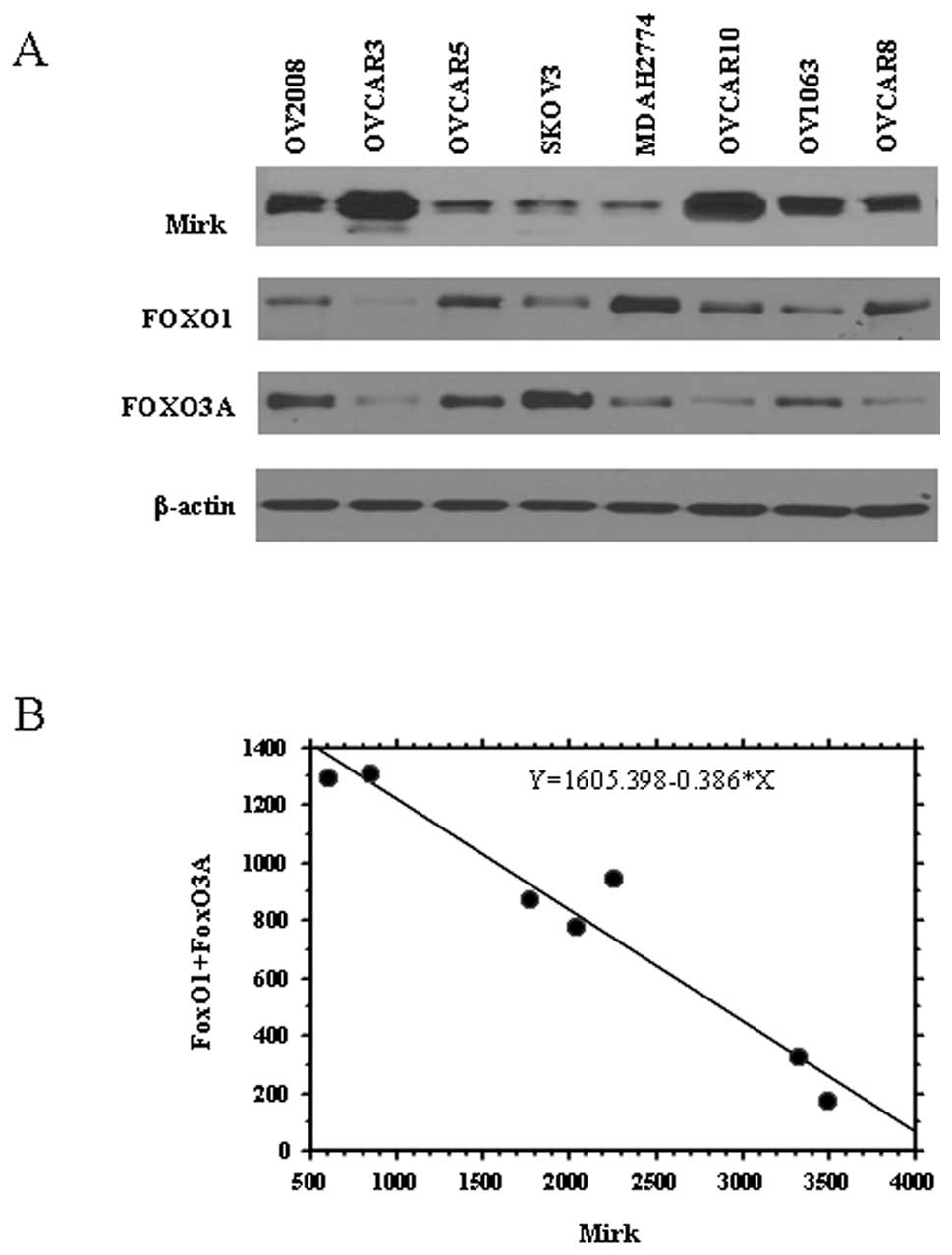
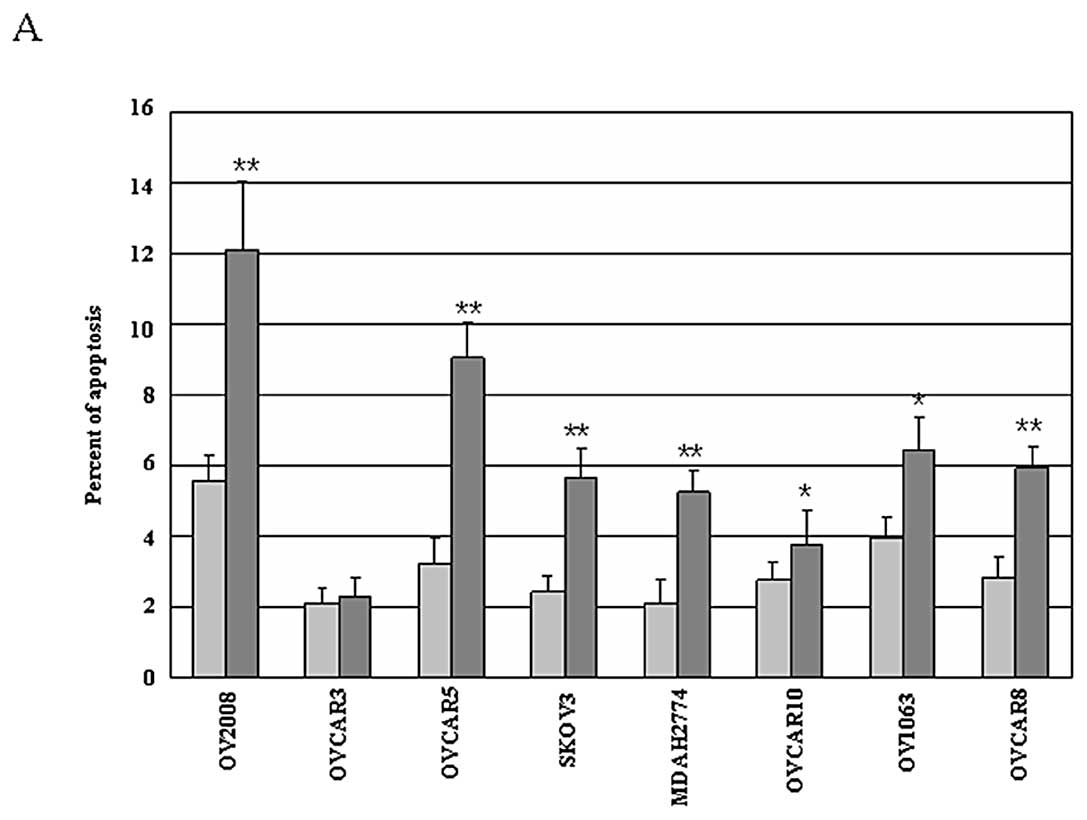
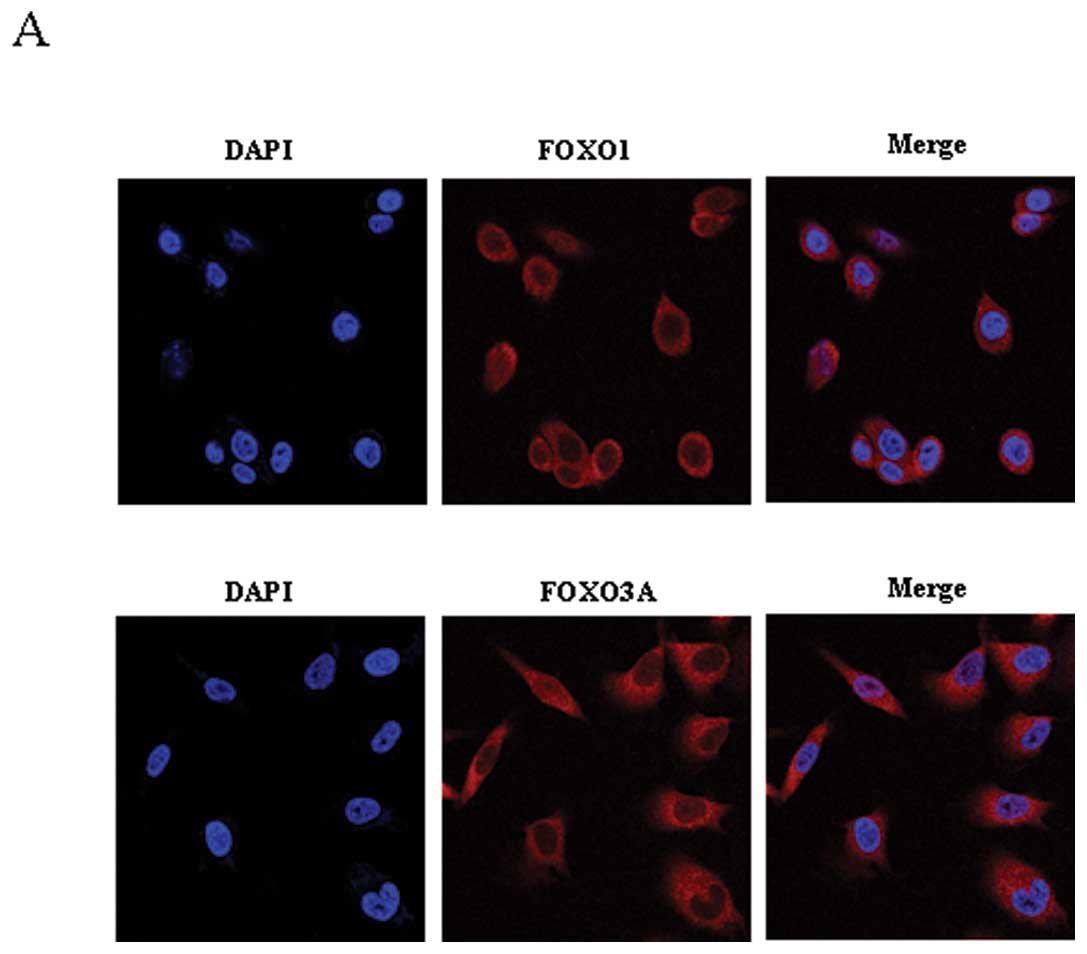
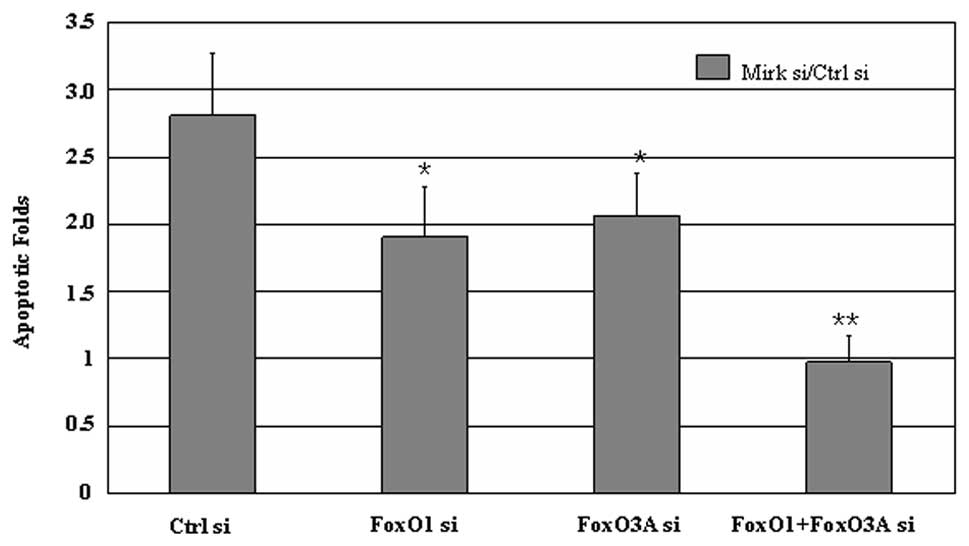
|
1
|
Ozols RF, Bookman MA, Connolly DC, et al:
Focus on epithelial ovarian cancer. Cancer Cell. 5:19–24. 2004.
|
|
2
|
Becker W and Joost HG: Structural and
functional characteristics of Dyrk, a novel subfamily of protein
kinases with dual specificity. Prog Nucleic Acid Res Mol Biol.
62:1–17. 1999.
|
|
3
|
Lee K, Deng X and Friedman E: Mirk protein
kinase is a mitogen-activated protein kinase substrate that
mediates survival of colon cancer cells. Cancer Res. 60:3631–3637.
2000.
|
|
4
|
Mercer SE, Ewton DZ, Shah S, Naqvi A and
Friedman E: Mirk/Dyrk1b mediates cell survival in
rhabdomyosarcomas. Cancer Res. 66:5143–5150. 2006.
|
|
5
|
Yang C, Ji D, Weinstein EJ, et al: The
kinase Mirk is a potential therapeutic target in osteosarcoma.
Carcinogenesis. 31:552–558. 2010.
|
|
6
|
Deng X, Ewton DZ, Li S, Naqvi A, Mercer
SE, Landas S and Friedman E: The kinase Mirk/Dyrk1B mediates cell
survival in pancreatic ductal adenocarcinoma. Cancer Res.
66:4149–4158. 2006.
|
|
7
|
MacKeigan JP, Murphy LO and Blenis J:
Sensitized RNAi screen of human kinases and phosphatases identifies
new regulators of apoptosis and chemoresistance. Nat Cell Biol.
7:591–600. 2005.
|
|
8
|
Gao J, Zheng Z, Rawal B, Schell MJ, Bepler
G and Haura EB: Mirk/Dyrk1B, a novel therapeutic target, mediates
cell survival in non-small cell lung cancer cells. Cancer Biol
Ther. 8:1671–1679. 2009.
|
|
9
|
Kops GJ, De Ruiter ND, De Vries-Smits AM,
Powell DR, Bos JL and Burgering BM: Direct control of the Forkhead
transcription factor AFX by protein kinase B. Nature. 398:630–634.
1999.
|
|
10
|
Nemoto S, Fergusson MM and Finkel T:
Nutrient availability regulates SIRT1 through a forkhead-dependent
pathway. Science. 306:2105–2108. 2004.
|
|
11
|
Fu W, Ma Q, Chen L, et al: MDM2 acts
downstream of p53 as an E3 ligase to promote FOXO ubiquitination
and degradation. J Biol Chem. 284:13987–14000. 2009.
|
|
12
|
Yang Y, Hou H, Haller EM, Nicosia SV and
Bai W: Suppression of FOXO1 activity by FHL2 through SIRT1-mediated
deacetylation. EMBO J. 24:1021–1032. 2005.
|
|
13
|
Brunet A, Bonni A, Zigmond MJ, et al: Akt
promotes cell survival by phosphorylating and inhibiting a Forkhead
transcription factor. Cell. 96:857–868. 1999.
|
|
14
|
Nakae J, Barr V and Accili D: Differential
regulation of gene expression by insulin and IGF-1 receptors
correlates with phosphorylation of a single amino acid residue in
the forkhead transcription factor FKHR. EMBO J. 19:989–996.
2000.
|
|
15
|
Dijkers PF, Medema RH, Lammers JW,
Koenderman L and Coffer PJ: Expression of the pro-apoptotic Bcl-2
family member Bim is regulated by the forkhead transcription factor
FKHR-L1. Curr Biol. 10:1201–1204. 2000.
|
|
16
|
Dijkers PF, Medema RH, Pals C, et al:
Forkhead transcription factor FKHR-L1 modulates cytokine-dependent
transcriptional regulation of p27(KIP1). Mol Cell Biol.
20:9138–9148. 2000.
|
|
17
|
Deng R, Tang J, Xie BF, Feng GK, Huang YH,
Liu ZC and Zhu XF: SYUNZ-16, a newly synthesized alkannin
derivative, induces tumor cells apoptosis and suppresses tumor
growth through inhibition of PKB/AKT kinase activity and blockade
of AKT/FOXO signal pathway. Int J Cancer. 127:220–229. 2010.
|
|
18
|
Skurk C, Maatz H, Kim HS, Yang J, Abid MR,
Aird WC and Walsh K: The Akt-regulated forkhead transcription
factor FOXO3a controls endothelial cell viability through
modulation of the caspase-8 inhibitor FLIP. J Biol Chem.
279:1513–1525. 2004.
|
|
19
|
Tang TT, Dowbenko D, Jackson A, Toney L,
Lewin DA, Dent AL and Lasky LA: The forkhead transcription factor
AFX activates apoptosis by induction of the BCL-6 transcriptional
repressor. J Biol Chem. 277:14255–14265. 2002.
|
|
20
|
Arimoto-Ishida E, Ohmichi M, Mabuchi S, et
al: Inhibition of phosphorylation of a forkhead transcription
factor sensitizes human ovarian cancer cells to cisplatin.
Endocrinology. 145:2014–2022. 2004.
|
|
21
|
Goto T, Takano M, Hirata J and Tsuda H:
The involvement of FOXO1 in cytotoxic stress and drug-resistance
induced by paclitaxel in ovarian cancers. Br J Cancer.
98:1068–1075. 2008.
|
|
22
|
Deng X, Ewton DZ and Friedman E:
Mirk/Dyrk1B maintains the viability of quiescent pancreatic cancer
cells by reducing levels of reactive oxygen species. Cancer Res.
69:3317–3324. 2009.
|
|
23
|
Hu J and Friedman E: Depleting Mirk kinase
increases cisplatin toxicity in ovarian cancer cells. Genes Cancer.
1:803–811. 2010.
|
|
24
|
Chang HS, Lin CH, Yang CH, et al:
Increased expression of Dyrk1a in HPV16 immortalized keratinocytes
enable evasion of apoptosis. Int J Cancer. 120:2377–2385. 2007.
|
|
25
|
Von Groote-Bidlingmaier F, Schmoll D, Orth
HM, Joost HG, Becker W and Barthel A: DYRK1 is a co-activator of
FKHR (FOXO1a)-dependent glucose-6-phosphatase gene expression.
Biochem Biophys Res Commun. 300:764–769. 2003.
|
|
26
|
Hu J, Nakhla H and Friedman E: Transient
arrest in a quiescent state allows ovarian cancer cells to survive
suboptimal growth conditions and is mediated by both Mirk/dyrk1b
and p130/Rb2. Int J Cancer. 129:307–318. 2011.
|
|
27
|
Chakrabarty A, Sanchez V, Kuba MG,
Rinehart C and Arteaga CL: Breast cancer special feature: feedback
upregulation of HER3 (ErbB3) expression and activity attenuates
antitumor effect of PI3K inhibitors. Proc Natl Acad Sci USA.
22:5021–5026. 2011.
|
|
28
|
Thompson FH, Nelson MA, Trent JM, et al:
Amplification of 19q13.1–q13.2 sequences in ovarian cancer. G-band,
FISH, and molecular studies. Cancer Genet Cytogenet. 87:55–62.
1996.
|