Introduction
Osteosarcoma is the most common primary malignancy
arising from the bone. It occurs most commonly in young people and
affects more males than females (1). There is a predilection for the
metaphyseal region of tubular long bones. It is a cancer that
usually affects the large bones of the arm or leg with 65% of cases
occurring around the knee (2). The
5-year survival rate for patients with osteosarcoma is at an
average of 65% (3). Current
treatment strategies include chemotherapy, radical resection and
irradiation followed by extensive rehabilitation. Although younger
patients with localized osteosarcoma benefit from this standard
treatment (4), aggressive
osteosarcomas respond poorly to conventional cytotoxic chemotherapy
thus necessitating the search for novel therapeutical approaches.
The most accepted compounds for chemoprevention in humans are
naturally occurring dietary substances. Various studies have
demonstrated that green tea catechins inhibit carcinogenesis and
growth of established cancers at various organ sites (5,6).
Many of the chemopreventive effects of green tea are mediated by
its catechins, with (−)-epigallocatechin-3-gallate (EGCG) being the
most abundant and powerful catechin in cancer prevention and
treatment (7). The chemopreventive
actions of EGCG comprise anti-oxidant activities, cell signalling
modulation, apoptosis induction, cell cycle arrest as well as
inhibition of matrix metalloproteinases (MMPs),
urokinase-plasminogen activator, telomerase, DNA methyltransferase
and proteasome (8).
Epidemiological studies indicate that inflammation
serves as a potential risk factor for the development of cancer. It
is generally accepted that up to 25% of human malignancies are
related to chronic inflammation and to viral and bacterial
infections (9). Chronic
inflammation increases the risk of cancer including bone neoplasm,
promotes tumor progression and supports metastatic spread (10,11).
The connection between tumorigenesis and inflammation is mediated
via intrinsic and extrinsic pathways (12). The intrinsic pathway is activated
by various genetic alterations finally producing transformed cells
which can secrete inflammatory mediators and thus can generate an
inflammatory microenvironment. The extrinsic pathway is driven by
inflammation or infections further increasing the risk for cancer
development. Both pathways converge in tumor cells and induce the
activation of several transcription factors including NF-κB, STAT-3
and HIF-1 culminating in the formation of pro-inflammatory factors
such as chemokines, cytokines and PGHS-2. These molecules recruit
and activate various leukocyte populations into the tumor
microenvironment. This concerted action of tumor and micromilieu
results in a more pronounced generation of inflammatory mediators
driving a tumor-promoting amplification loop.
Several genetic and chromosomal abnormalities as
part of the intrinsic pathway have been found in osteosarcoma
patients including chromosomal amplification and loss of
heterozygosity, associated with poor prognosis (13,14).
Additionally, mutations in the tumor suppressor proteins p53 and Rb
have been implicated in the oncogenesis of osteosarcoma, enhancing
the risk and thus contributing to its poorer prognosis (15,16).
Among the inflammatory mediators present in the
tumor microenvironment, IL-1 acts as a crucial player in
inflammation-associated carcinogenesis (17,18).
Increased levels of IL-1 have been identified in several human
tumor entities such as melanoma, head and neck, colon, lung and
breast cancer. In an animal model of human osteosarcoma increased
IL-1 levels have also been reported (19). Overall, patients harbouring
IL-1-positive tumors have markedly worse prognosis (20). IL-1 is produced directly by cancer
cells or by cells of the mircroenvironment. Depending on its
subcellular location, different IL-1 isoforms mediate different
functions. Membrane-bound IL-1α found on malignant cells induces
antitumor immune responses, whereas intracellular precursors of
IL-1α control homeostatic functions. In contrast, low
concentrations of secreted IL-1β downregulate inflammatory
responses and immune mechanisms, whereas high concentrations
promote inflammation-associated tissue damage and tumor
invasiveness (21). IL-1 can
stimulate other cell types to produce pro-angiogenic and
pro-metastatic mediators and thus plays an important role in
inflammation-associated carcinogenesis (17,18).
In this context, increased serum levels of tumor-promoting
cytokines, including IL-6, IL-8 and VEGF, have been reported in
osteosarcoma patients correlating with poor overall survival
(22).
IL-1α and IL-1β exert identical agonist actions by
binding to the IL-1 receptor type I (IL-1RI). A third ligand, the
naturally occurring IL-1 receptor antagonist (IL-1Ra), also binds
to IL-1RI without leading to its activation and competitively
inhibits its activation by IL-1. IL-1RI ligation leads to the
activation of intracellular signalling cascades including NF-κB
(23,24) which provides a mechanistic link
between inflammation and cancer and is a major factor controlling
the ability of both, preneoplastic and malignant cells, to resist
apoptosis-based tumor surveillance mechanisms. NF-κB might also
regulate tumor angiogenesis and invasiveness (25), and may contribute to the
characteristic chemoresistance of tumor cells (26).
Since inflammatory- as well as angiogenesis- and
invasiveness-promoting factors are crucially involved in the
pathogenesis of osteosarcoma, there is a strong medical need to
reduce these external factors as a preventive or therapeutical
measure. Therefore, in this study anti-inflammatory IL-1Ra was
tested either alone or in combination with the green tea-derived
catechin EGCG on the production of IL-1-induced tumorigenic factors
in U-2 OS human osteosarcoma cells. A combined treatment resulted
in a more pronounced inhibition of tumorigenic factors rendering
the combined administration of EGCG and IL-1Ra a promising approach
as an adjuvant therapy in patients with osteosarcoma.
Materials and methods
Cell culture and stimulation
The human osteosarcoma cell line U-2 OS (27) was purchased from the American Type
Culture Collection (#HTB-96; ATCC, Manassas, VA, USA) and cultured
in McCoy’s 5a medium with L-glutamine supplemented with 10%
heat-inactivated fetal calf serum (all from PAA Laboratories,
Pasching, Austria) in the presence of 100 IU/ml penicillin and 100
μg/ml streptomycin (Gibco-BRL, Grand Island, NY, USA). The
cells were maintained under standard cell culture conditions at
37°C in a 5% CO2 humidified atmosphere. For stimulation
experiments, U-2 OS cells were seeded in 96-well microtiter plates
at a density of 5×103 cells/well, and after a recovery
phase of at least 16 h, stimulated with or without 0.5 ng/ml of
recombinant human IL-1β (PAN Biotech GmbH, Aidenbach, Germany) in
the presence or absence of human recombinant IL-1Ra (R&D
Systems GmbH, Wiesbaden, Germany) and
(−)-epigallocatechin-3-gallate (EGCG, purity ≥95%) purchased from
Sigma-Aldrich Chemie GmbH (Steinheim, Germany) in a time- and
dose-dependent manner as indicated in the figure legends. EGCG and
IL-1Ra treatment started 1 h prior to IL-1 stimulation. All
treatments were conducted in the presence of 0.05 nM
2-mercaptoethanol (Sigma-Aldrich Chemie GmbH) to stabilize EGCG and
quench EGCG-derived ROS.
Metabolic response and cell
viability
The effect of EGCG and IL-1Ra on the metabolic
response was assessed by using MTT assay as described by Mosmann
(28). This assay was based on the
ability of viable cells only to reduce the conversion of the water
soluble MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium
bromide] to an insoluble formazan. After solubilisation, the
concentration was determined spectrophotometrically using the
Titertek Multiscan microplate reader (Flow Laboratories,
Meckenheim, Germany) at 550 nm. The assay is suitable for
determining the metabolic activity/number of viable cells in
proliferation, cytotoxicity or chemosensitivity assays.
Determination of caspase-3 activity
Caspase-3 activity was measured after a 24-h
incubation period with or without IL-1β in the presence or absence
of 50 μM EGCG and increasing concentrations (1–50 ng/ml) of
IL-1Ra. Harvested cells were lyzed with caspase lysis buffer (10 mM
Tris-HCl, 10 mM sodium phosphate buffer, pH 7.5, 130 mM NaCl, 1%
Triton X-100, 10 mm Na2P2O7) and
then incubated with 25 μg/ml of the fluorogenic caspase-3
substrate N-acetyl-DEVD-7-amido-4-trifluoromethyl coumarin (Becton
Dickinson GmbH, Heidelberg, Germany) in 20 mM HEPES (pH 7.5), 10%
glycerol and 2 mM dithiothreitol at 37°C for 2 h in the dark. The
release of the fluorogenic AFC moiety as a measure of caspase
activity was analyzed fluorimetrically using the
Infinite® 200 PRO microplate reader (Tecan Group, Ltd.,
Männedorf, Switzerland) at an excitation/emission wavelength of
390/510 nm. Relative caspase activities were normalized to the
protein content as determined by Bradford dye-binding assay
(29) and compared to the
untreated control whose response was set as one. Etoposide
(Sigma-Aldrich Chemie GmbH) at a concentration of 50 μM was
used as a positive control.
Cytokine and metalloproteinase
assays
Concentrations of human IL-6 and IL-8 in the culture
supernatants were determined by commercially available ELISA kits
(Eli-Pair; Diaclone, Besançon, France). Production of total MMP-2
was quantified by immunoassay using the corresponding
Quantikine® ELISA kit purchased from R&D Systems
GmbH according to the manufacturer’s instructions. Secretion of
VEGF into the supernatants was evaluated using the
RayBio® Human VEGF EIA kit (RayBiotech, Inc., Norcross,
GA, USA). All data were normalized to the metabolic response
obtained by MTT assay.
Data adjustment and statistics
In order to compensate for inter-experiment
variations, data were adjusted by setting the value for the
untreated sample (VEGF, MMP-2) or IL-1-treated sample without
inhibitor (IL-6, IL-8) as 100%. Statistical differences between
mean values were analyzed using the two-tailed non-parametric
Wilcoxon-Mann-Whitney test. A P-value <0.05 was considered to be
statistically significant.
Results
In this study, the effect of IL-1Ra was tested alone
and in combination with EGCG on the expression of IL-1-induced
tumorigenic factors in U-2 OS human osteosarcoma cells. This
approach acts as a model system for tumor-associated inflammation,
playing a key role in carcinogenesis. In order to prevent
autoxidation of EGCG and quench formation of EGCG-derived ROS, all
experiments were conducted in the presence of
2-mercaptoethanol.
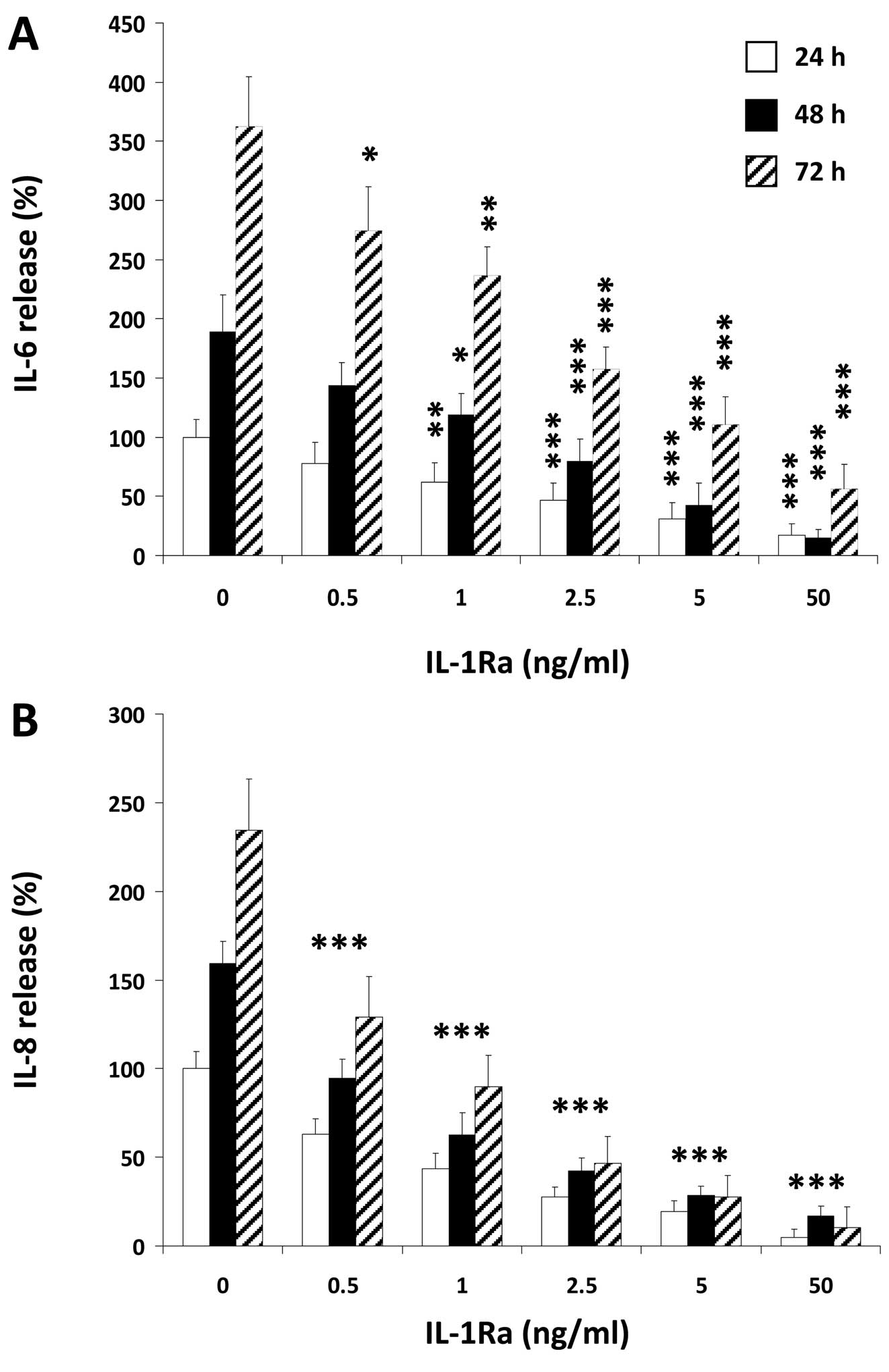
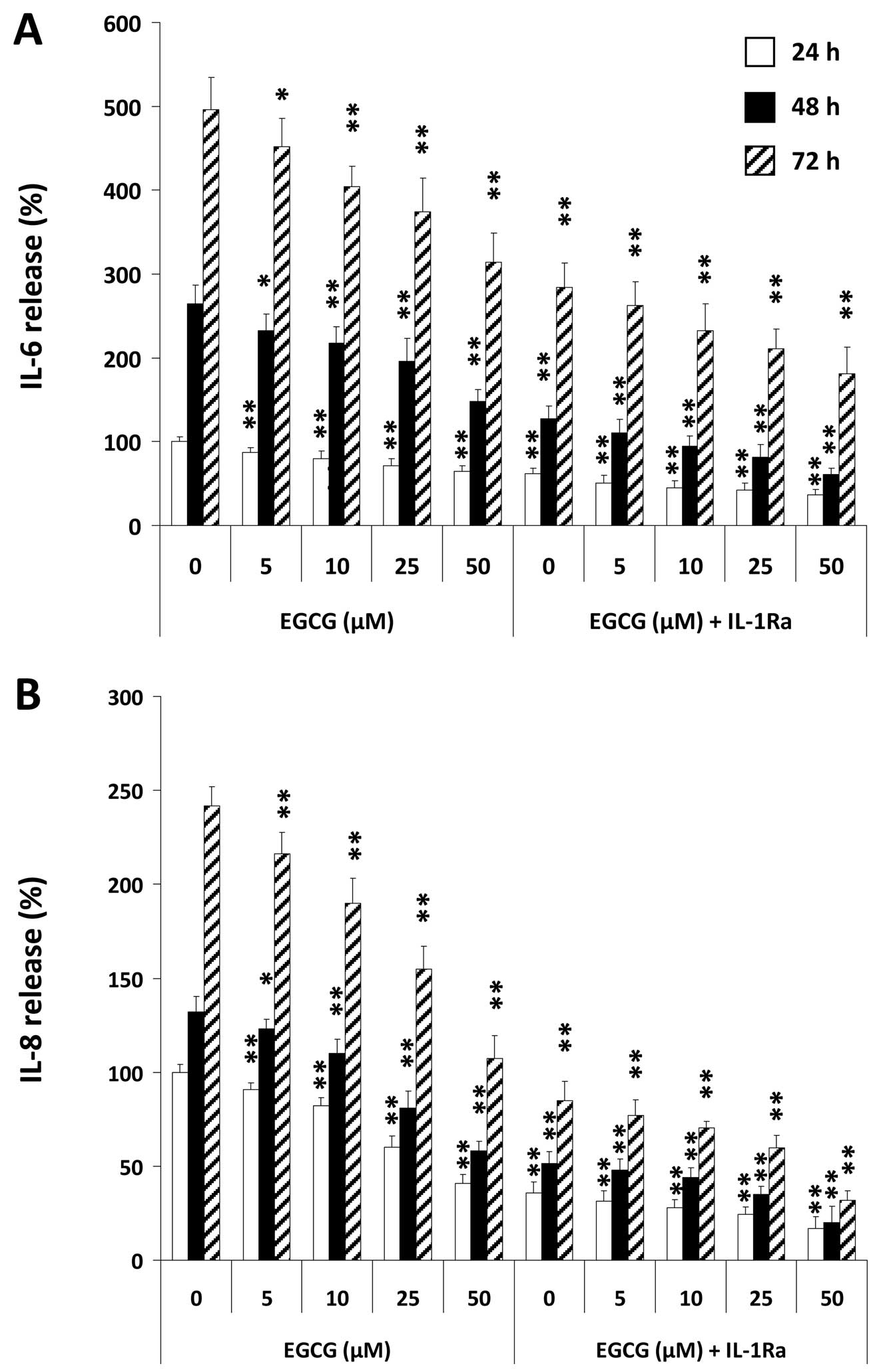
As an example of IL-1-induced tumorigenic factors we
determined the effect of IL-1Ra and EGCG, either alone or in
combination, on secretion of pro-inflammatory IL-6 (Fig. 1A and Fig. 2A) and pro-angiogenic IL-8 (Fig. 1B and Fig. 2B) by U-2 OS cells after stimulation
with IL-1. We found a strong time-dependent IL-6 and IL-8
production by U-2 OS cells after IL-1 stimulation that was blocked
by either IL-1Ra (Fig. 1) or EGCG
(Fig. 2) in a dose-dependent
manner. Since IL-1Ra inhibited the release of these molecules with
an IC50 of ∼1 ng/ml, co-incubation experiments were
conducted with this IL-1Ra concentration. As demonstrated in
Fig. 2, co-treatment of U-2 OS
cells with EGCG and IL-1Ra reduced IL-1-induced cytokine production
in an additive manner. Co-stimulation of U-2 OS cells with 50
μM EGCG and IL-1Ra led to an inhibition of IL-1-induced IL-6
production by ∼2/3 at any timepoint, while IL-1-induced IL-8
release was downregulated at an average of 85%.
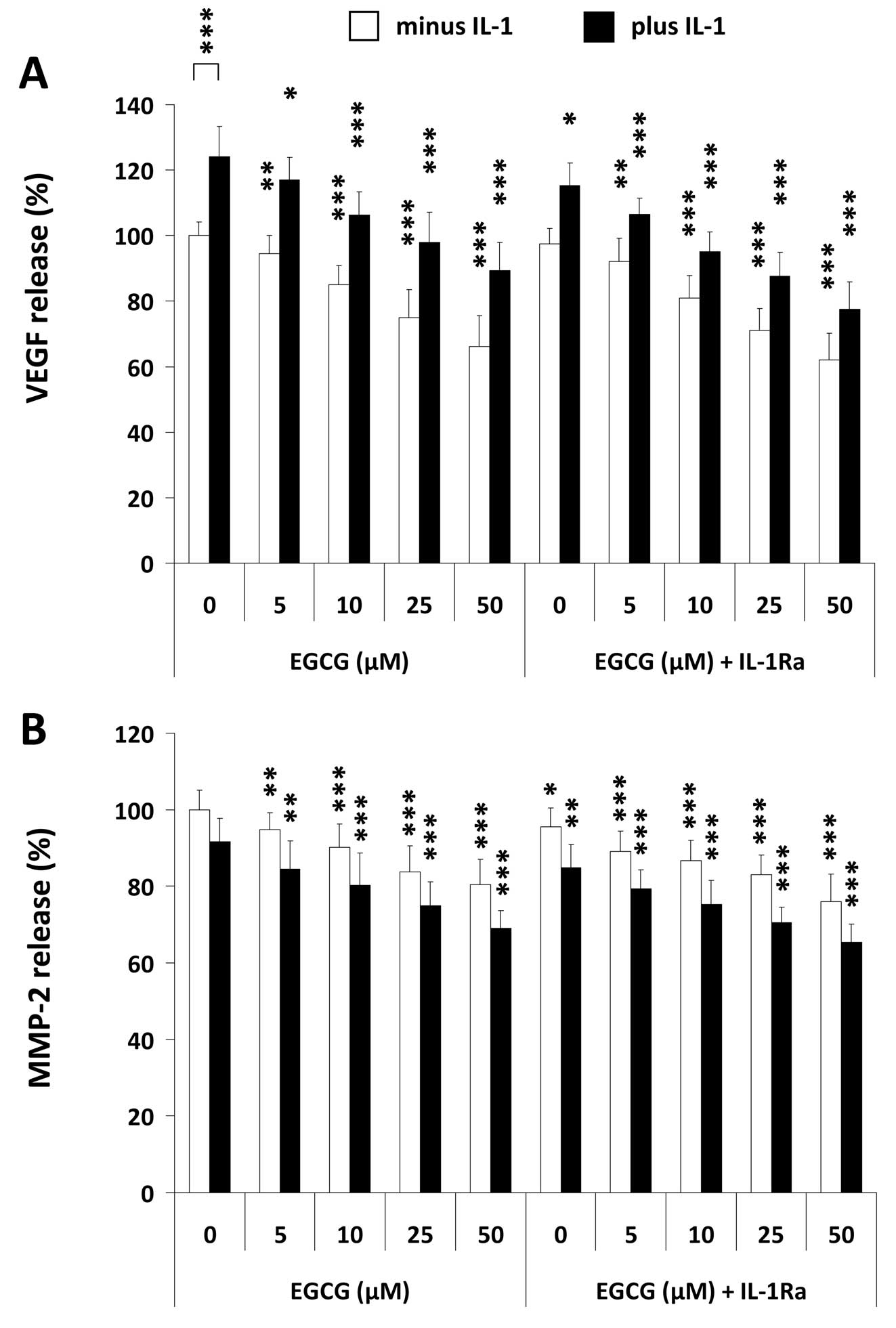
Increased levels of different growth factors have
been previously reported in osteosarcoma including VEGF (30). We therefore tested whether U-2 OS
cells constitutively secrete this pro-angiogenic cytokine and
whether its secretion was altered by IL-1, IL-1Ra and EGCG,
respectively. As demonstrated in Fig.
3A, the osteosarcoma cells constitutively secrete high amounts
of VEGF that can be further enhanced in the presence of IL-1. EGCG
was found to downregulate both, the constitutive as well as the
IL-1-mediated VEGF secretion, in a dose-dependent manner. In
contrast, co-incubation of U-2 OS cells with the agents suppressed
VEGF production in an additive manner (Fig. 3A). IL-1Ra in combination with 50
μM EGCG reduced the IL-1-induced VEGF secretion as well as
the spontaneous production by ∼38%.
Since matrix metalloproteinases (MMPs) play an
important role in the pathogenesis of osteosarcoma (31), we investigated the secretion
profile of invasiveness- and angiogenesis-promoting MMP-2 by U-2 OS
cells in the presence of IL-1Ra and different concentrations of
EGCG, after stimulation with IL-1. It has been shown previously
that MMP 2 secretion can be induced indirectly by IL-1 via IL-6 and
activation of the JAK/STAT pathway (32). Our data clearly identified a strong
spontaneous MMP-2 secretion by U-2 OS cells that was not augmented
by added IL-1 (Fig. 3B). EGCG
alone dose-dependently decreased MMP-2 secretion down to 75% of the
untreated control. Also IL-1Ra single-agent treatment slightly
reduced MMP-2 release. Interestingly, IL-1Ra in combination with
EGCG significantly augmented EGCG-mediated decline in MMP-2
production in any case (P<0.05) (Fig. 3B).
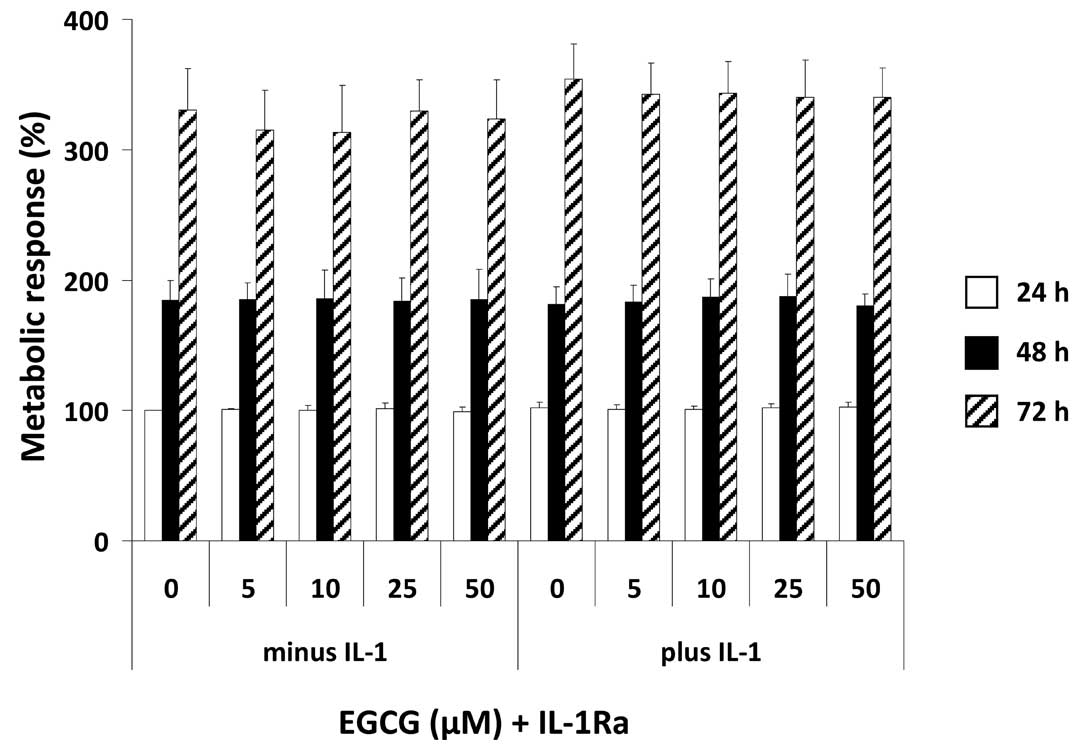
In order to elucidate whether the effects on
mediator release are related to a decline in the metabolic
response, U-2 OS cells were treated with IL-1Ra and increasing
concentrations of EGCG in the presence or absence of IL-1, followed
by determination of the metabolic activity as evidenced by MTT
assay. We found that neither EGCG and IL-1Ra alone nor a
combinatorial treatment had any effect on the metabolic response in
U-2 OS cells (Fig. 4). Moreover,
an effect of IL-1 on the metabolic response of U-2 OS cells could
also not be observed.
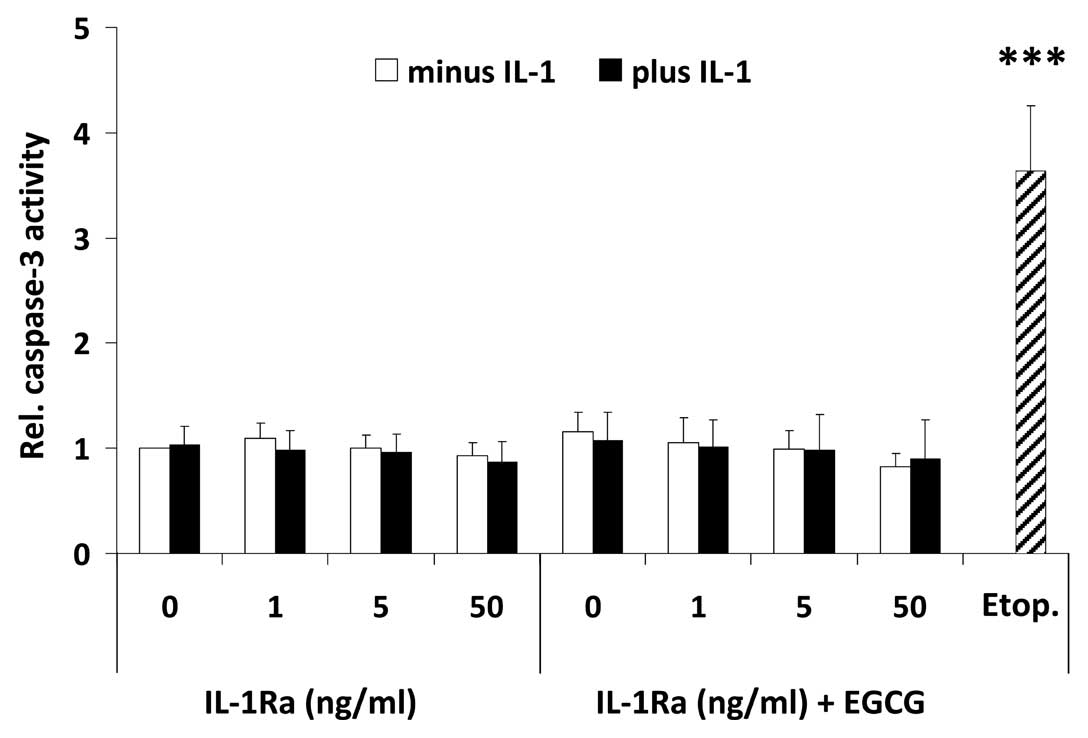
To further shed light on the mechanism which causes
a decrease in mediator release, U-2 OS cells were treated with the
highest dose of EGCG used in this study and increasing
concentrations of IL-1Ra in the presence or absence of IL-1,
followed by fluorometrical determination of caspase-3 activity,
indicative for apoptosis induction. Fig. 5 shows that neither treatment had
any effect on caspase-3 activation, implying that the decline in
mediator release is unrelated to apoptosis induction.
Discussion
The aim of the present study was to shed light on
the combined effects of IL-1Ra and the green tea-derived catechin
EGCG on the expression of IL-1-induced tumorigenic factors in the
human osteosarcoma cell line U-2 OS (27). U-2 OS cells have been characterized
to overexpress the oncoprotein Mdm-2 (33). Aberrant Mdm-2 expression can be
found in a variety of human tumors of diverse tissue origin
including osteosarcoma contributing to the malignant phenotype. In
osteosarcoma, Mdm-2 overexpression occurs with high frequency as a
result of an upregulated MDM2 mRNA expression and
translation (34).
We found that IL-1Ra and EGCG act additively in
down-regulating secretion of pro-inflammatory IL-6 as well as
pro-angiogenic IL-8 and VEGF, thus blocking production of
tumorigenic mediators in the tumor microenvironment. Since IL-1Ra
and EGCG were also found to suppress export of
invasiveness-promoting MMP-2 by U-2 OS cells, targeting the
inflammatory network in U-2 OS cells by IL-1Ra and EGCG can be
considered as a promising approach in the treatment of
osteosarcoma. EGCG and IL-1Ra were used at concentrations not
inducing apoptosis and not affecting the metabolic response of U-2
OS cells. Even after co-incubation with EGCG and IL-1Ra, effects on
cell viability and apoptosis induction did not occur. From these
findings, it can be concluded that a combinatory administration of
IL-1Ra and EGCG reduces the impact of tumorigenic factors by
interfering with intracellular signalling cascades such as NF-κB.
It is well documented that, beside its antioxidant activities, EGCG
targets the key transcription factor in tumorigenesis, NF-κB
(8,35,36).
EGCG plasma concentrations achievable after oral
administration of green tea extracts or catechins cover the lower
micromolar range up to 60 μM (37–39).
Greater oral bioavailability of free catechins in humans can be
achieved when consumed in the absence of food (40). EGCG was found in our study to
significantly reduce release of tumorigenic factors already at a
concentration of 5 μM. We therefore speculate that EGCG will
also interrupt the inflammatory network in vivo; however, in
order to strengthen this assumption, chemoprevention studies in
experimental models of human osteosarcoma will have to be
performed.
IL-1Ra was found in this study to be a potent
inhibitor of IL-1-induced tumorigenic factors in U-2 OS cells. In
an animal model of human osteosarcoma, systemic IL-1Ra (anakinra™)
dose-dependently inhibited different forms of thermal and
osteosarcoma-induced hyperalgesia (19). Because of its collagenase and
prostaglandin-inhibiting properties, anakinra is approved for the
treatment of chronic inflammatory diseases including rheumatoid
arthritis (41) and systemic onset
juvenile idiopathic arthritis (42). It has also been identified to be
powerful in blocking IL-1 effects in numerous pathological settings
(41). With respect to cancer,
anakinra was successfully used in treating the rare
lymphoproliferative disorder Castleman’s disease (43) and myeloma (44). Anakinra and other IL-1-blocking
agents such as canakunimab™ (anti-IL-1β antibody) or rilonacept™
(construct of the two extracellular chains of IL-1RI/IL-1RAcP
complex fused to the Fc segment of IgG) could thus be promising
therapeutics for human metastatic diseases. The latter two agents
have been approved for the treatment of the cryopyrin-associated
periodic syndrome (45,46). As summarized by Dinarello (47), there are two main reasons for the
use of IL-1-blocking agents in the treatment of metastatic
diseases. No organ toxicities or gastrointestinal and
haematological abnormalities were observed and no, unlike for
TNF-blocking agents, opportunistic infections were reported except
rare bacterial and upper airway infections. Due to the safety of
IL-1 blockage, clinical trials are encouraged. An NIH trial of
anakinra in the treatment of cutaneous melanoma is ongoing because
IL-1 plays a pivotal role in angiogenesis by inducing/upregulating
pro-angiogenic IL-8 and VEGF, contributing to the pathogenesis of
e.g. multiple melanoma (47).
Notably, the immunomodulator mifamurtide (liposomal
muramyl tripeptide phosphatidylethanolamine; MEPACT™) which has
been approved in Europe for the treatment of non-metastatic
osteosarcoma in combination with chemotherapy (48), has come under scrutiny because of
studies suggesting an increased risk of serious adverse effects
associated with its use whilst concomitantly lacking any survival
benefit (49). In osteosarcoma
patients, increased plasma concentrations of pro-inflammatory
mediators such as IL-1, TNF and IL-6 are detected after
administration of mifamurtide (50) obviously due to its
macrophage/monocyte activating capacity (51). From this observation and regarding
our own data, one can hypothesize that mifamurtide probably
enhances the impact of tumorigenic stimuli within the tumor
microenvironment thereby forcing a critical amplification mechanism
in tumor-associated inflammation triggered by IL-6 and others. All
these results clearly demonstrate that there is a strong medical
need for the development of new concepts how such inflammatory
activities working in osteosarcoma may be therapeutically targeted
with novel combinations of chemopreventive drugs.
In summary, a therapeutic approach that combines the
IL-1 activity inhibiting effects of IL-1Ra and the anti-angiogenic
and anti-inflammatory activities of EGCG might impair the
development of a malignant phenotype in osteosarcoma cells and
produce a crucial additive antitumoral response compared to IL-1Ra
or EGCG administered in monotherapy.
Acknowledgements
We thank Werner Falk, Department of
Internal Medicine I (University of Regensburg, Germany), for
critical reading of the manuscript and his helpful comments.
References
|
1
|
Longhi A, Pasini A, Cicognani A, et al:
Height as a risk factor for osteosarcoma. J Pediatr Hematol Oncol.
27:314–318. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Vigorita VJ and Ghelman B: Orthopaedic
Pathology. 2nd edition. Lippincott, Williams and Wilkins;
Philadelphia, PA: 2008
|
|
3
|
Bielack SS, Kempf-Bielack B, Delling G, et
al: Prognostic factors in high-grade osteosarcoma of the
extremities or trunk: an analysis of 1,702 patients treated on
neoadjuvant cooperative osteosarcoma study group protocols. J Clin
Oncol. 20:776–790. 2002. View Article : Google Scholar
|
|
4
|
Hegyi M, Semsei AF, Jakab Z, et al: Good
prognosis of localized osteosarcoma in young patients treated with
limb-salvage surgery and chemotherapy. Pediatr Blood Cancer.
57:415–422. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Khan N, Afaq F and Mukhtar H: Cancer
chemoprevention through dietary antioxidants: progress and promise.
Antioxid Redox Signal. 10:475–510. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Shankar S, Ganapathy S, Hingorani SR and
Srivastava RK: EGCG inhibits growth, invasion, angiogenesis and
metastasis of pancreatic cancer. Front Biosci. 13:440–452. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Yang CS, Lambert JD, Ju J, Lu G and Sang
S: Tea and cancer prevention: molecular mechanisms and human
relevance. Toxicol Appl Pharmacol. 224:265–273. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Khan N and Mukhtar H: Multitargeted
therapy of cancer by green tea polyphenols. Cancer Lett.
269:269–280. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hussain SP and Harris CC: Inflammation and
cancer: an ancient link with novel potentials. Int J Cancer.
121:2373–2380. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Multhoff G, Molls M and Radons J: Chronic
inflammation in cancer development. Front Immunol. 2:982012.
View Article : Google Scholar
|
|
11
|
Terpos E and Dimopoulos MA: Interaction
between the skeletal and immune systems in cancer: mechanisms and
clinical implications. Cancer Immunol Immunother. 60:305–317. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mantovani A, Allavena P, Sica A and
Balkwill F: Cancer-related inflammation. Nature. 454:436–444. 2008.
View Article : Google Scholar
|
|
13
|
Smida J, Baumhoer D, Rosemann M, et al:
Genomic alterations and allelic imbalances are strong prognostic
predictors in osteosarcoma. Clin Cancer Res. 16:4256–4267. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ta HT, Dass CR, Choong PF and Dunstan DE:
Osteosarcoma treatment: state of the art. Cancer Metastasis Rev.
28:247–263. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Heinsohn S, Evermann U, Zur SU, Bielack S
and Kabisch H: Determination of the prognostic value of loss of
heterozygosity at the retinoblastoma gene in osteosarcoma. Int J
Oncol. 30:1205–1214. 2007.PubMed/NCBI
|
|
16
|
Longhi A, Benassi MS, Molendini L,
Macchiagodena M, Picci P and Bacci G: Osteosarcoma in blood
relatives. Oncol Rep. 8:131–136. 2001.PubMed/NCBI
|
|
17
|
Lin WW and Karin M: A cytokine-mediated
link between innate immunity, inflammation, and cancer. J Clin
Invest. 117:1175–1183. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
18
|
Voronov E, Carmi Y and Apte RN: Role of
IL-1-mediated inflammation in tumor angiogenesis. Adv Exp Med Biol.
601:265–270. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Baamonde A, Curto-Reyes V, Juarez L, Meana
A, Hidalgo A and Menendez L: Antihyperalgesic effects induced by
the IL-1 receptor antagonist anakinra and increased IL-1beta levels
in inflamed and osteosarcoma-bearing mice. Life Sci. 81:673–682.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lewis AM, Varghese S, Xu H and Alexander
HR: Interleukin-1 and cancer progression: the emerging role of
interleukin-1 receptor antagonist as a novel therapeutic agent in
cancer treatment. J Transl Med. 4:482006. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Apte RN, Krelin Y, Song X, et al: Effects
of micro-environment-and malignant cell-derived interleukin-1 in
carcinogenesis, tumour invasiveness and tumour-host interactions.
Eur J Cancer. 42:751–759. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Rutkowski P, Kaminska J, Kowalska M, Ruka
W and Steffen J: Cytokine and cytokine receptor serum levels in
adult bone sarcoma patients: correlations with local tumor extent
and prognosis. J Surg Oncol. 84:151–159. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Gay NJ, Gangloff M and O’Neill LA: What
the Myddosome structure tells us about the initiation of innate
immunity. Trends Immunol. 32:104–109. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Watters TM, Kenny EF and O’Neill LA:
Structure, function and regulation of the Toll/IL-1 receptor
adaptor proteins. Immunol Cell Biol. 85:411–419. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Karin M: Nuclear factor-kappaB in cancer
development and progression. Nature. 441:431–436. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Singh S and Khar A: Biological effects of
curcumin and its role in cancer chemoprevention and therapy.
Anticancer Agents Med Chem. 6:259–270. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ponten J and Saksela E: Two established in
vitro cell lines from human mesenchymal tumours. Int J Cancer.
2:434–447. 1967. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Mosmann T: Rapid colorimetric assay for
cellular growth and survival: application to proliferation and
cytotoxicity assays. J Immunol Methods. 65:55–63. 1983. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Bradford MM: A rapid and sensitive method
for the quantitation of microgram quantities of protein utilizing
the principle of protein-dye binding. Anal Biochem. 72:248–254.
1976. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Qu Y, Xu J, Jiang T, et al: Difference in
pre- and postchemotherapy vascular endothelial growth factor levels
as a prognostic indicator in osteosarcoma. J Int Med Res.
39:1474–1482. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Korpi JT, Hagstrom J, Lehtonen N, et al:
Expression of matrix metalloproteinases-2, -8, -13, -26, and tissue
inhibitors of metalloproteinase-1 in human osteosarcoma. Surg
Oncol. 20:e18–e22. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Xie TX, Wei D, Liu M, et al: Stat3
activation regulates the expression of matrix metalloproteinase-2
and tumor invasion and metastasis. Oncogene. 23:3550–3560. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Landers JE, Cassel SL and George DL:
Translational enhancement of mdm2 oncogene expression in human
tumor cells containing a stabilized wild-type p53 protein. Cancer
Res. 57:3562–3568. 1997.PubMed/NCBI
|
|
34
|
Ladanyi M, Cha C, Lewis R, Jhanwar SC,
Huvos AG and Healey JH: MDM2 gene amplification in metastatic
osteosarcoma. Cancer Res. 53:16–18. 1993.PubMed/NCBI
|
|
35
|
Hoffmann J, Junker H, Schmieder A, et al:
EGCG downregulates IL-1RI expression and suppresses IL-1-induced
tumorigenic factors in human pancreatic adenocarcinoma cells.
Biochem Pharmacol. 82:1153–1162. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Lambert JD and Yang CS: Mechanisms of
cancer prevention by tea constituents. J Nutr. 133:S3262–S3267.
2003.PubMed/NCBI
|
|
37
|
Lin JK, Liang YC and Lin-Shiau SY: Cancer
chemoprevention by tea polyphenols through mitotic signal
transduction blockade. Biochem Pharmacol. 58:911–915. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Pisters KM, Newman RA, Coldman B, et al:
Phase I trial of oral green tea extract in adult patients with
solid tumors. J Clin Oncol. 19:1830–1838. 2001.PubMed/NCBI
|
|
39
|
Van Amelsvoort JM, Van Hof KH, Mathot JN,
Mulder TP, Wiersma A and Tijburg LB: Plasma concentrations of
individual tea catechins after a single oral dose in humans.
Xenobiotica. 31:891–901. 2001.PubMed/NCBI
|
|
40
|
Chow HH, Hakim IA, Vining DR, et al:
Effects of dosing condition on the oral bioavailability of green
tea catechins after single-dose administration of Polyphenon E in
healthy individuals. Clin Cancer Res. 11:4627–4633. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Dinarello CA: Biologic basis for
interleukin-1 in disease. Blood. 87:2095–2147. 1996.PubMed/NCBI
|
|
42
|
Hedrich CM, Bruck N, Fiebig B and Gahr M:
Anakinra: A safe and effective first-line treatment in systemic
onset juvenile idiopathic arthritis (SoJIA). Rheumatol Int. Nov
15–2011.(Epub ahead of print).
|
|
43
|
El-Osta H, Janku F and Kurzrock R:
Successful treatment of Castleman’s disease with interleukin-1
receptor antagonist (Anakinra). Mol Cancer Ther. 9:1485–1488.
2010.
|
|
44
|
Lust JA, Lacy MQ, Zeldenrust SR, et al:
Induction of a chronic disease state in patients with smoldering or
indolent multiple myeloma by targeting interleukin 1β-induced
interleukin 6 production and the myeloma proliferative component.
Mayo Clin Proc. 84:114–122. 2009.
|
|
45
|
Hoffman HM, Throne ML, Amar NJ, et al:
Efficacy and safety of rilonacept (interleukin-1 Trap) in patients
with cryopyrin-associated periodic syndromes: results from two
sequential placebo-controlled studies. Arthritis Rheum.
58:2443–2452. 2008. View Article : Google Scholar
|
|
46
|
Lachmann HJ, Kone-Paut I,
Kuemmerle-Deschner JB, et al: Use of canakinumab in the
cryopyrin-associated periodic syndrome. N Engl J Med.
360:2416–2425. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Dinarello CA: Why not treat human cancer
with interleukin-1 blockade? Cancer Metastasis Rev. 29:317–329.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Meyers PA: Muramyl tripeptide
(mifamurtide) for the treatment of osteosarcoma. Expert Rev
Anticancer Ther. 9:1035–1049. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Mifamurtide: osteosarcoma: ineffective and
harmful. Prescrire Int. 20:892011.
|
|
50
|
Kleinerman ES, Jia SF, Griffin J, Seibel
NL, Benjamin RS and Jaffe N: Phase II study of liposomal muramyl
tripeptide in osteosarcoma: the cytokine cascade and monocyte
activation following administration. J Clin Oncol. 10:1310–1316.
1992.PubMed/NCBI
|
|
51
|
Ando K, Mori K, Corradini N, Redini F and
Heymann D: Mifamurtide for the treatment of nonmetastatic
osteosarcoma. Expert Opin Pharmacother. 12:285–292. 2011.
View Article : Google Scholar : PubMed/NCBI
|