|
1.
|
Kudo S, Lambert R, Allen JI, et al:
Nonpolypoid neoplastic lesions of the colorectal mucosa.
Gastrointest Endosc. 68:S3–S47. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
2.
|
Smith JJ, Deane NG, Dhawan P and Beauchamp
RD: Regulation of metastasis in colorectal adenocarcinoma: A
collision between development and tumor biology. Surgery.
144:353–366. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3.
|
Kinzler KW and Vogelstein B: Lessons from
hereditary colorectal cancer. Cell. 87:159–170. 1996. View Article : Google Scholar : PubMed/NCBI
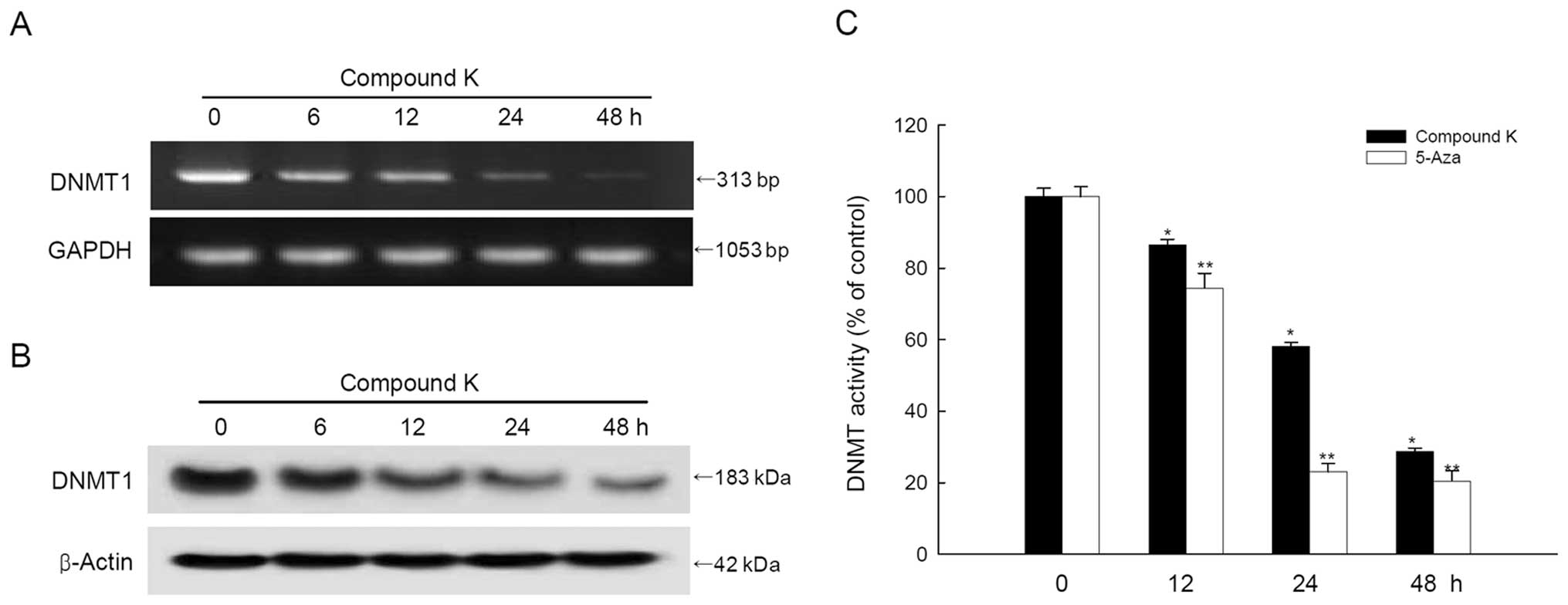
|
|
4.
|
Grady WM and Carethers JM: Genomic and
epigenetic instability in colorectal cancer pathogenesis.
Gastroenterology. 135:1079–1099. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5.
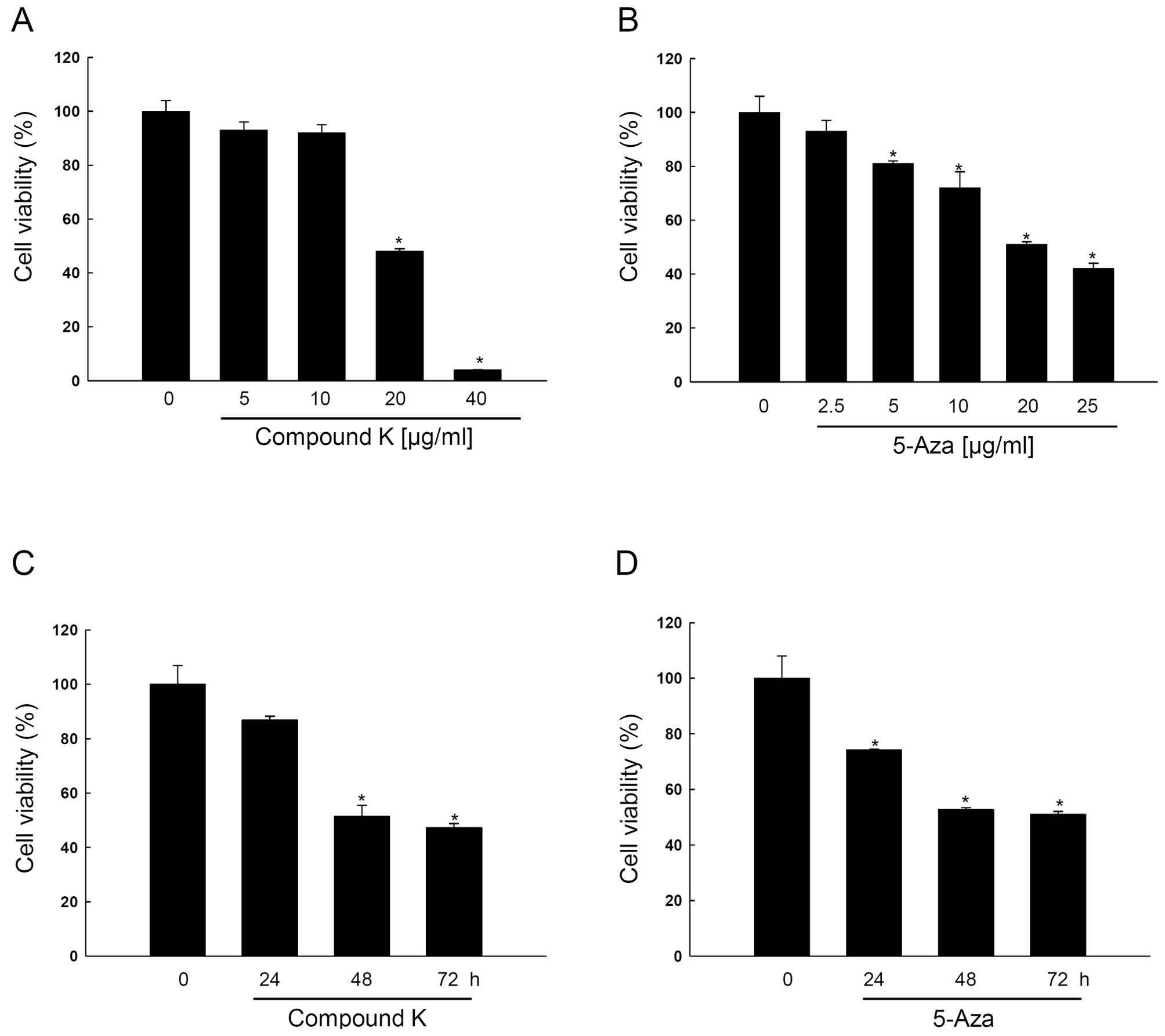
|
Wong JJ, Hawkins NJ and Ward RL:
Colorectal cancer: a model for epigenetic tumorigenesis. Gut.
56:140–148. 2007. View Article : Google Scholar : PubMed/NCBI
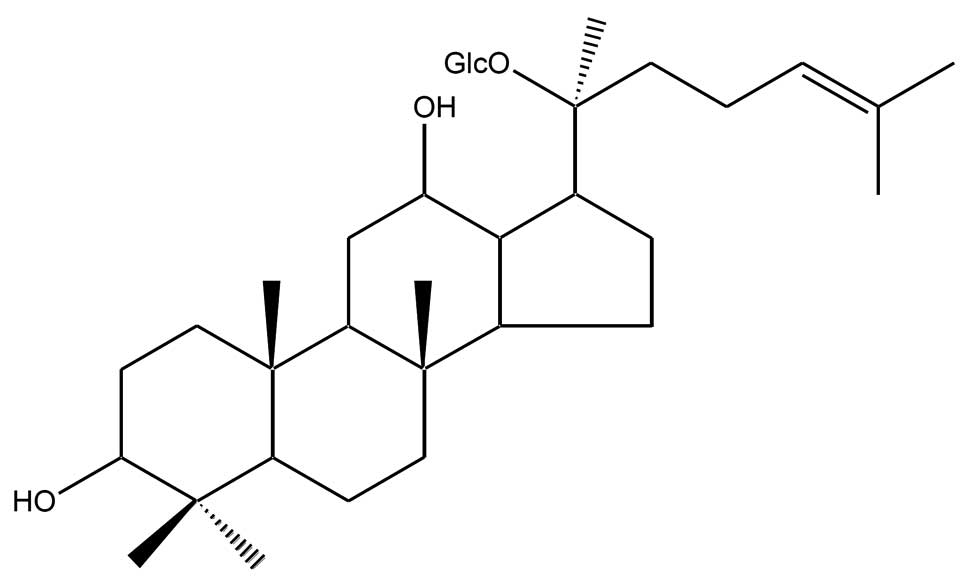
|
|
6.
|
Matos E and Brandani A: Review on meat
consumption and cancer in South America. Mutat Res.
506–507:243–249. 2002.PubMed/NCBI
|
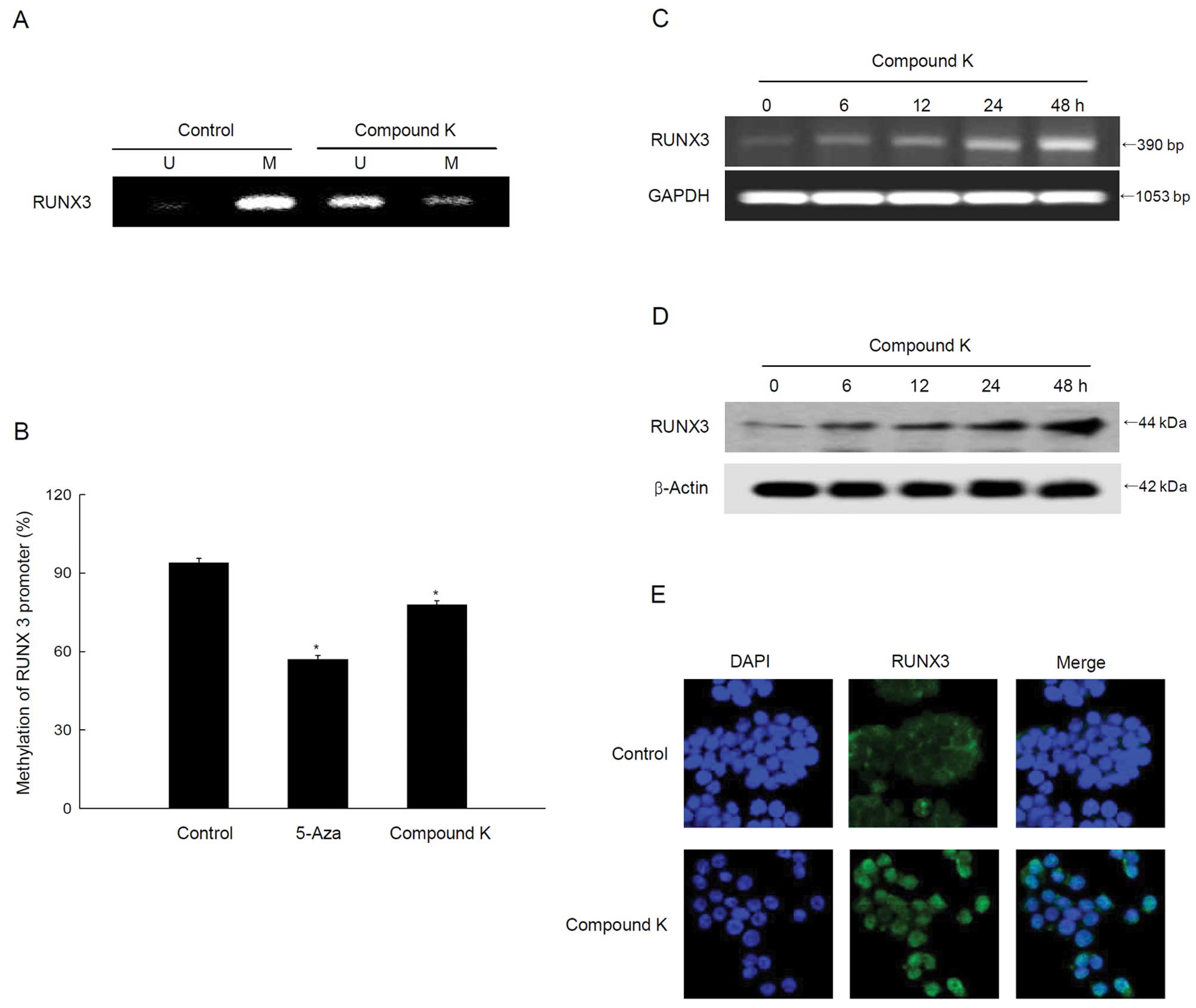
|
7.
|
Nkondjock A and Ghadirian P: Associated
nutritional risk of breast and colon cancers: a population-based
case control study in Montreal, Canada. Cancer Lett. 223:85–91.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
8.
|
Fearon ER and Vogelstein B: A genetic
model for colorectal tumorigenesis. Cell. 61:759–767. 1990.
View Article : Google Scholar : PubMed/NCBI
|
|
9.
|
Grady WM: Genomic instability and colon
cancer. Cancer Metastasis Rev. 23:11–27. 2004. View Article : Google Scholar
|
|
10.
|
Herman JG and Baylin SB: Gene silencing in
cancer in association with promoter hypermethylation. N Engl J Med.
349:2042–2054. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
11.
|
Levanon D, Brenner O, Negreanu V, et al:
Spatial and temporal expression pattern of Runx3 (Aml2) and Runx1
(Aml1) indicates non-redundant functions during mouse
embryogenesis. Mech Dev. 109:413–417. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
12.
|
Woolf E, Xiao C, Fainaru O, et al: Runx3
and Runx1 are required for CD8 T cell development during
thymopoiesis. Proc Natl Acad Sci USA. 100:7731–7736. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
13.
|
Li QL, Kim HR, Kim WJ, et al:
Transcriptional silencing of the RUNX3 gene by CpG hypermethylation
is associated with lung cancer. Biochem Biophys Res Commun.
314:223–228. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
14.
|
Chuang LS and Ito Y: RUNX3 is
multifunctional in carcinogenesis of multiple solid tumors.
Oncogene. 29:2605–2615. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15.
|
Ragnarsson G, Eiriksdottir G,
Johannsdottir JT, Jonasson JG, Egilsson V and Ingvarsson S: Loss of
heterozygosity at chromosome 1p in different solid human tumours:
association with survival. Br J Cancer. 79:1468–1474. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
16.
|
Tanaka K, Yanoshita R, Konishi M, et al:
Suppression of tumourigenicity in human colon carcinoma cells by
introduction of normal chromosome 1p36 region. Oncogene.
8:2253–2258. 1993.PubMed/NCBI
|
|
17.
|
Li QL, Ito K, Sakakura C, et al: Causal
relationship between the loss of RUNX3 expression and gastric
cancer. Cell. 109:113–124. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
18.
|
Goel A, Arnold CN, Tassone P, et al:
Epigenetic inactivation of RUNX3 in microsatellite unstable
sporadic colon cancers. Int J Cancer. 112:754–759. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
19.
|
Jair KW, Bachman KE, Suzuki H, et al: De
novo CpG island methylation in human cancer cells. Cancer Res.
66:682–692. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
20.
|
Rhee I, Bachman KE, Park BH, et al: DNMT1
and DNMT3b cooperate to silence genes in human cancer cells.
Nature. 416:552–556. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
21.
|
Lu R, Wang X, Chen ZF, Sun DF, Tian XQ and
Fang JY: Inhibition of the extracellular signal-regulated
kinase/mitogenactivated protein kinase pathway decreases DNA
methylation in colon cancer cells. J Biol Chem. 282:12249–12259.
2007. View Article : Google Scholar
|
|
22.
|
Santi DV, Norment A and Garrett CE:
Covalent bond formation between a DNA-cytosine methyltransferase
and DNA containing 5-azacytosine. Proc Natl Acad Sci USA.
81:6993–6997. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
23.
|
Gabbara S and Bhagwat AS: The mechanism of
inhibition of DNA (cytosine-5-)-methyltransferases by 5-azacytosine
is likely to involve methyl transfer to the inhibitor. Biochem J.
307:87–92. 1995.PubMed/NCBI
|
|
24.
|
Mack GS: Epigenetic cancer therapy makes
headway. J Natl Cancer Inst. 98:1443–1444. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
25.
|
Beisler JA: Isolation, characterization,
and properties of a labile hydrolysis product of the antitumor
nucleoside, 5-azacytidine. J Med Chem. 21:204–208. 1978. View Article : Google Scholar : PubMed/NCBI
|
|
26.
|
Constantinides PG, Jones PA and Gevers W:
Functional striated muscle cells from non-myoblast precursors
following 5-azacytidine treatment. Nature. 267:364–366. 1977.
View Article : Google Scholar : PubMed/NCBI
|
|
27.
|
Lyko F and Brown R: DNA methyltransferase
inhibitors and the development of epigenetic cancer therapies. J
Natl Cancer Inst. 97:1498–1506. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
28.
|
Gaudet F, Hodgson JG, Eden A, et al:
Induction of tumors in mice by genomic hypomethylation. Science.
300:489–492. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
29.
|
Howard G, Eiges R, Gaudet F, Jaenisch R
and Eden A: Activation and transposition of endogenous retroviral
elements in hypomethylation induced tumors in mice. Oncogene.
27:404–408. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30.
|
Deng T and Zhang Y: 5-Aza-2′-deoxycytidine
reactivates expression of RUNX3 by deletion of DNA
methyltransferases leading to caspase independent apoptosis in
colorectal cancer Lovo cells. Biomed Pharmacother. 63:492–500.
2009.
|
|
31.
|
Hong SY, Cho JY and Seo DW: Ginsenoside
Rp1 inhibits proliferation and migration of human lung cancer
cells. Biomol Ther. 19:411–418. 2011. View Article : Google Scholar
|
|
32.
|
Park JW, Lee JC, Ann S, et al: A fermented
ginseng extract, BST204, inhibits proliferation and motility of
human colon cancer cells. Biomol Ther. 19:211–217. 2011. View Article : Google Scholar
|
|
33.
|
Seo EY and Kim WK: Red ginseng extract
reduced metastasis of colon cancer cells in vitro and in vivo. J
Ginseng Res. 35:315–324. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
34.
|
Akao T, Kanaoka M and Kobashi K:
Appearance of compound K, a major metabolite of ginsenoside Rb1 by
intestinal bacteria, in rat plasma after oral
administration-measurement of compound K by enzyme immunoassay.
Biol Pharm Bull. 21:245–249. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
35.
|
Hasegawa H, Sung JH and Huh JH: Ginseng
intestinal bacterial metabolite IH901 as a new anti-metastatic
agent. Arch Pharm Res. 20:539–544. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
36.
|
Quan LH, Cheng LQ, Kim HB, et al:
Bioconversion of ginsenoside Rd into compound K by Lactobacillus
pentosus DC101 isolated from Kimchi. J Gins Res. 34:288–295.
2010. View Article : Google Scholar
|
|
37.
|
Kang KA, Kim YW, Kim SU, et al: G1 phase
arrest of the cell cycle by a ginseng metabolite, compound K, in
U937 human monocytic leukamia cells. Arch Pharm Res. 28:685–690.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
38.
|
Kang KA, Lim HK, Kim SU, et al: Induction
of apoptosis by ginseng saponin metabolite in U937 human monocytic
leukemia cells. J Food Biochem. 29:27–40. 2005. View Article : Google Scholar
|
|
39.
|
Chae S, Kang KA, Chang WY, et al: Effect
of compound K, a metabolite of ginseng saponin, combined with
gamma-ray radiation in human lung cancer cells in vitro and in
vivo. J Agric Food Chem. 57:5777–5782. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
40.
|
Kim AD, Kang KA, Zhang R, et al: Ginseng
saponin metabolite induces apoptosis in MCF-7 breast cancer cells
through the modulation of AMP-activated protein kinase. Environ
Toxicol Pharmacol. 30:134–140. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
41.
|
Ito K, Lim AC, Salto-Tellez M, et al:
RUNX3 attenuates beta-catenin/T cell factors in intestinal
tumorigenesis. Cancer Cell. 14:226–237. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
42.
|
Ku JL, Kang SB, Shin YK, et al: Promoter
hypermethylation downregulates RUNX3 gene expression in colorectal
cancer cell lines. Oncogene. 23:6736–6742. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
43.
|
Yano T, Ito K, Fukamachi H, et al: The
RUNX3 tumor suppressor upregulates Bim in gastric epithelial cells
undergoing transforming growth factor beta-induced apoptosis. Mol
Cell Biol. 26:4474–4488. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
44.
|
Tong DD, Jiang Y, Li M, et al: RUNX3
inhibits cell proliferation and induces apoptosis by
TGF-beta-dependent and -independent mechanisms in human colon
carcinoma cells. Pathobiology. 76:163–169. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
45.
|
Kodach LL, Jacobs RJ, Heijmans J, et al:
The role of EZH2 and DNA methylation in the silencing of the tumour
suppressor RUNX3 in colorectal cancer. Carcinogenesis.
31:1567–1575. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
46.
|
Lee IK, Kang KA, Lim CM, et al: Compound
K, a metabolite of ginseng saponin, induces mitochondria-dependent
and caspase-dependent apoptosis via the generation of reactive
oxygen species in human colon cancer cells. Int J Mol Sci.
11:4916–4931. 2010. View Article : Google Scholar
|
|
47.
|
Subramaniam MM, Chan JY, Soong R, et al:
RUNX3 inactivation by frequent promoter hypermethylation and
protein mislocalization constitute an early event in breast cancer
progression. Breast Cancer Res Treat. 113:113–121. 2009. View Article : Google Scholar
|
|
48.
|
Ito K, Liu Q, Salto-Tellez M, et al:
RUNX3, a novel tumor suppressor, is frequently inactivated in
gastric cancer by protein mislocalization. Cancer Res.
65:7743–7750. 2005.PubMed/NCBI
|
|
49.
|
Mani S and Herceg Z: DNA demethylating
agents and epigenetic therapy of cancer. Adv Genet. 70:327–240.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
50.
|
Howell PM, Liu Z and Khong HT:
Demethylating agents in the treatment of cancer. Pharmaceuticals.
3:2022–2044. 2010. View Article : Google Scholar
|
|
51.
|
Fang MZ, Wang Y, Ai N, et al: Tea
polyphenol (-)-epigallocatechin-3-gallate inhibits DNA
methyltransferase and reactivates methylation-silenced genes in
cancer cell lines. Cancer Res. 63:7563–7570. 2003.PubMed/NCBI
|
|
52.
|
Fang MZ, Chen D, Sun Y, Jin Z, Christman
JK and Yang CS: Reversal of hypermethylation and reactivation of
p16INK4a, RARbeta, and MGMT genes by genistein and other
isoflavones from soy. Clin Cancer Res. 11:7033–7041. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
53.
|
Fini L, Selgrad M, Fogliano V, et al:
Annurca apple polyphenols have potent demethylating activity and
can reactivate silenced tumor suppressor genes in colorectal cancer
cells. J Nutr. 137:2622–2628. 2007.PubMed/NCBI
|
|
54.
|
Kishikawa S, Murata T, Ugai H, Yamazaki T
and Yokoyama KK: Control elements of Dnmt1 gene are regulated in
cell-cycle dependent manner. Nucleic Acids Res (Suppl). 3:307–308.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
55.
|
Torquati A, O’rear L, Longobardi L, et al:
RUNX3 inhibits cell proliferation and induces apoptosis by
reinstating transforming growth factor beta responsiveness in
esophageal adenocarcinoma cells. Surgery. 136:310–316. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
56.
|
Yamamura Y, Lee WL, Inoue K, Ida H and Ito
Y: RUNX3 cooperates with FoxO3a to induce apoptosis in gastric
cancer cells. J Biol Chem. 281:5267–5276. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
57.
|
Zaidi SK, Sullivan AJ, van Wijnen AJ,
Stein JL, Stein GS and Lian JB: Integration of Runx and Smad
regulatory signals at transcriptionally active subnuclear sites.
Proc Natl Acad Sci USA. 99:8048–8053. 2002. View Article : Google Scholar : PubMed/NCBI
|