Introduction
Ovarian cancer is the most lethal gynecological
cancer in industrialized countries and is the fifth leading cause
of cancer deaths in women. Ovarian carcinoma becomes often
resistant to conventional systemic therapies even after the first
treatments and the prognosis for patients with advanced ovarian
cancer remains poor (1).
The standard first-line treatment for ovarian cancer
is a combination of paclitaxel and a Pt derivative drug such as
cisplatin (cDDP) or carboplatin alone (2).
cDDP and its derivatives have been widely used to
treat a variety of solid tumors including testicular, ovarian, head
and cervical carcinoma (3).
However, the appearance of resistant cells in the tumor limits its
therapeutic effectiveness, thus cDDP is usually administered in
combination with other drugs (4),
lowering of drug dosages and consequently decrease side-effects,
reducing the opportunity for the development of drug resistance by
cancer cells (5).
Acquired resistance to cDDP and its derivatives is a
multi-factorial process involving many mechanisms, among these
enhanced DNA synthesis and repair is the most common feature of
resistance in almost all resistant cell lines studied. In this
context, the overexpression of folate cycle enzymes dihydrofolate
reductase (DHFR) and thymidylate synthase (TS) played an important
role in a large survey of cDDP resistance human ovarian carcinoma
cell lines (6). Therefore, the
discovery of drugs aimed at impairing the activity of these
enzymes, thus circumventing cDDP-resistance, is of great
interest.
However, even chemotherapy based on combination of
drugs is associated with significant side-effects, highlighting the
need for therapeutic strategies that target tumor cells without
compromising normal tissue function (7). Thus, the development of novel
systemic agents from natural products with low toxicity and few
side-effects is being actively pursued (8,9).
In this regard, cDDP has been recently combined with
berberine, obtaining enhanced apoptotic cell death through a
mitochondria/caspase-mediated pathway in HeLa cells (10).
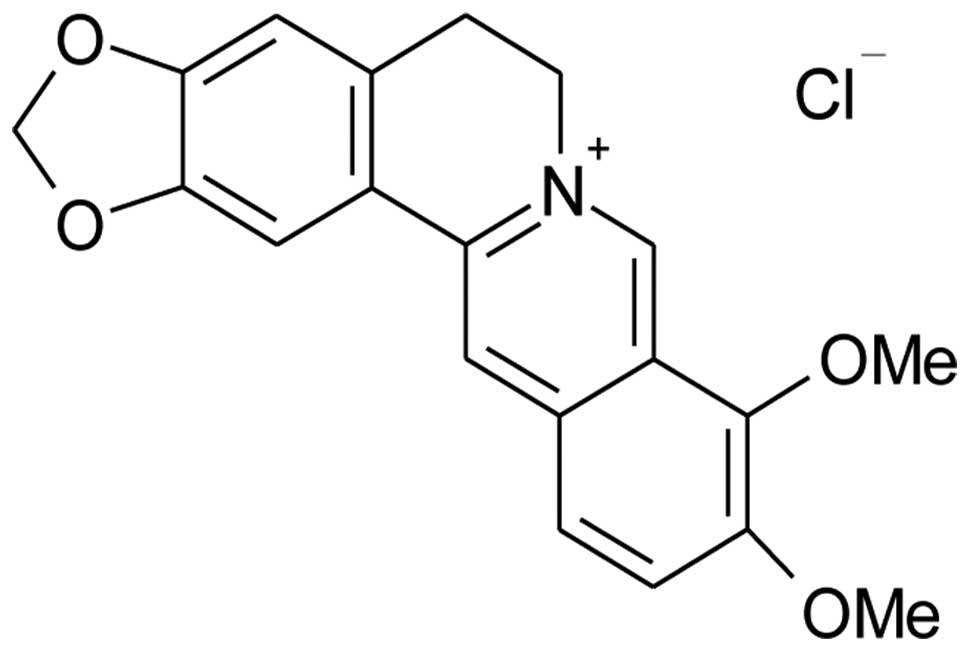
Berberine is an isoquinoline alkaloid (Fig. 1) isolated from a variety of Chinese
herbs, including Coptidis rhizoma, Phellodendron
chinense schneid and Phellodendron amurense and other
important medicinal plant species such as Berberis aristata
and Berberis aquifolium and has diverse pharmacological
actions such as antibacterial (11,12)
and anti-inflammatory (13). The
antineoplastic activities of berberine have been demonstrated
against a variety of human cancer cells both in vitro and
in vivo through suppression of tumor cell proliferation, or
induction of tumor cell apoptosis (14,15).
The pharmacologic effects depend on inhibition of DNA and protein
synthesis, arrest of cell cycle progression (16,17).
Besides, it has been reported that berberine has dual topoisomerase
I and II poisoning activity and binds to double helical DNA with
high affinity. Other potential targets of berberine hydrochloride
include reactive oxygen species generation, mitochondrial function,
RNA binding, the estrogen receptor, matrix metalloproteinase
regulation and p53 activation (18–20).
Nevertheless, it is relatively nontoxic to humans (21).
We have recently reported (22), that the binding at the AT-rich
sequences of the minor groove of double-strand DNA of the natural
antibiotic distamycin (23,24)
was accompanied by the downregulation of both dihydrofolate
reductase (DHFR) and thymidylate synthase (TS) in
cisplatin-sensitive and -resistant cell lines.
TS catalyzes the reductive methylation of dUMP by
CH2H4PteGlu, generating dTMP and
dihydrofolate (25). DHFR
catalyzes the reduction of folate and 7,8-dihydrofolate (DHF) to
5,6,7,8-tetrahydrofolate (THF), utilizing NADPH as cofactor. Both
reactions are essential steps in the biosynthesis of nucleotidic
bases of DNA and thus important targets for chemotherapy (26).
Increasing body of evidence has suggested intimate
interrelationships between folate cycle and polyamine pathways,
since the polyamine key catabolic enzyme spermidine/spermine
N1-acetyl transferase (SSAT; EC 2.3.1.57) was identified as being
one of the most affected genes in studies based on folate cycle
inhibitors such as 5-FU (27–29).
The polyamine pathway is an attractive target in the development of
anticancer strategies (30), since
the naturally occurring polyamines putrescine (Put), spermidine
(Spd) and spermine (Spm) are essential for cell growth and
differentiation and present elevated concentrations in cancer cells
(31). In this regard, we have
also reported that combinations of novel folate cycle inhibitors
with quinoxaline structure and drugs that specifically target
polyamine metabolism, such as diethyl-derivatives of norspermine
(DENSPM) or spermine (BESpm), have synergistic effect in killing
cisplatin-sensitive and drug-resistant daughter human ovarian cell
lines (29). Besides, we have
previously indicated the limited induction of SSAT activity as an
important determinant of the reduced response to BESpm in the
cDDP-resistant human ovarian cancer C13* cells (32).
In the present study, we investigated the effects of
berberine on cDDP-sensitive human ovarian cancer cells and, for the
first time, on the resistant counterparts and examined the
functional role of folate cycle enzymes and polyamine metabolism in
berberine-induced cell growth inhibition. Besides, in this study
the relationship between berberine-induced cell death and TS, DHFR
and SSAT expression was explored, as well as the nature of the
interaction between berberine with either cDDP or a polyamine
analogue against ovarian carcinoma cell models.
Materials and methods
Chemical reagents
Berberine was provided by Dr P. Lombardi,
Naxospharma (Milan, Italy) and was freshly dissolved in DMSO prior
to addition to cell cultures. Cells treated with vehicle only
[DMSO, maximum concentration, 0.5% (v/v) in media] served as
control. [5-3H]dUMP (20 Ci/mmol), was purchased from
Moravek Biochemicals (Brea, CA, USA). BESpm was kindly supplied by
Hoechst Marion Roussel Inc. (Cincinnati, OH, USA).
1-[14C]acetyl coenzyme (1.89 GBq/mmol) was purchased
from Perkin-Elmer Italia (Milan, Italy). All other chemicals were
purchased from Sigma-Aldrich S.r.l. (Milan, Italy), except when
otherwise indicated.
Cell lines
The 2008 cell line was established from a patient
with advanced cystadenocarcinoma of the ovary. The cDDP-resistant
C13* subline, which is ∼15-fold resistant to cDDP, was
derived from the parent 2008 cell line by monthly exposure to cDDP,
followed by chronic exposure to step-wise increases in cDDP
concentration (33). The cell
lines were grown in monolayers in RPMI-1640 medium containing 10%
heat-inactivated foetal bovine serum and 50 μg/ml gentamycin
sulphate. Vero cells, established from kidney cells of the African
green monkey (Cercopithecus aethiops) and obtained from the
Istituto Zooprofilattico (Brescia, Italy), were chosen as a control
cell line (34). All cell media
and serum were purchased from Lonza (Verviers, Belgium). Cultures
were equilibrated with humidified 5% CO2 in air at 37°C.
All studies were performed in mycoplasma-negative cells, as
routinely determined using the MycoAlert Mycoplasma detection kit
(Lonza, Walkersville, MD, USA). Protein content in the assays was
estimated using the Lowry method (35), unless otherwise indicated.
Cell growth assay
Cell growth was determined using a modified crystal
violet assay (36). On selected
days, the tissue culture medium was removed and the cell monolayer
fixed with methanol and stained with 0.2% crystal violet solution
in 20% methanol for at least 30 min. After being washed several
times with distilled water to remove excess dye, the cells were
left to dry. The incorporated dye was solubilised in acidified
isopropanol (1 N HCl:2-propanol, 1:10). After appropriate dilution,
absorbance was determined spectrophotometrically at 540 nm. The
extracted dye was proportional to the cell number. The percentage
of cytotoxicity was calculated by comparing the absorbance of
cultures exposed to the drug to unexposed (control) cultures.
TS catalytic assay
Cells used for the enzyme assay were harvested by
trypsinisation in an exponential growth phase, washed with PBS and
used or stored at −20°C. Cell pellets were thawed by the addition
of ice-cold lysis buffer (200 mM Tris-HCl, pH 7.4, 20 mM
2-mercaptoethanol, 100 mM NaF and 1% Triton X-100), sonicated
(three × 5 sec) and subsequently centrifuged at 14,000 × g for 15
min at 4°C. The supernatant was used for enzyme assays. The TS
catalytic assay was conducted according to a previously reported
method (37); the assay determined
the catalytic activity of TS by measuring the amounts of
3H released during the TS catalyzed conversion of
[5-3H]dUMP to dTMP. Briefly, the assay consisted of the
enzymes in assay buffer (lysis buffer without Triton X-100) and 650
μM 5,10-methylenetetrahydrofolate in a final volume of 50
μl. The reaction was started by adding [5-3H]dUMP
(1 μM final concentration, specific activity 5 mCi/mol),
followed by incubation at 37°C for 60 min and stopped by adding 50
μl of ice-cold 35% trichloroacetic acid. Residual
[5-3H]dUMP was removed by adding 250 μl of 10%
neutral activated charcoal. The charcoal was removed by
centrifugation at 14,000 x g for 15 min at 4°C and a 150-μl
sample of the supernatant was assayed for tritium radioactivity by
liquid scintillation counting in the liquid scintillation analyzer
Tri-Carb 2100 (Packard). For each cell line, the linearity of
[5-3H]dUMP conversion with respect to amount of protein
and time was established.
DHFR catalytic assay
DHFR activity, measured as folate reductase, was
determined by the [3H]-folate reductase assay, as
previously described (38).
Briefly, cells were harvested by trypsinization in an exponential
growth phase, washed with PBS buffer and resuspended in 60 mM
sodium citrate, pH 7.2 containing 10 mM 2-mercaptoethanol. Cell
lysate was prepared by freeze thawing three times. The insoluble
debris was removed by centrifugation at 14,000 × g for 15 min at
4°C. The supernatant was used immediately for enzyme assay after
the determination of protein concentration by the method of
Bradford, using the Bio-Rad reagent with bovine serum albumin as a
standard (39).
[3H]-folate (25 pmol) was preincubated with 1.5
μmol of dithionite for 10 min at 37°C in a total volume of
250 μl. Following this incubation 60 nmol of NADPH and the
enzyme preparation were added together with an appropriate amount
of sodium citrate buffer pH 7.2 to reach a final volume of 500
μl. The reaction was performed for 60 min at 37°C and
stopped by adding, in sequence, 200 μl of a solution 0.027 M
folic acid, 100 μl of 0.1 N HCl and 200 μl of 0.3 N
zinc sulfate. The yellow folate precipitate was pelletted by
centrifugation at 16,000 x g for 45 min and the tritium
radioactivity of the supernatant was measured by adding 500
μl to 10 ml of Emulsifier Scintillation Plus cocktail
(Perkin-Elmer Italia) in the liquid scintillator analyzer Tri-Carb
2100 (Packard). For each cell line, linearity of
[3H]-folate conversion with respect to the amount of
protein and time was established.
Western blotting
Cells were harvested, washed twice in ice-cold 1X
PBS and resuspended in a buffer consisting of 20 mM Tris-HCl (pH
7.4), 150 mM NaCl, 1 mM EDTA (pH 8.0), 1% Triton X-100 and 0.1%
SDS. Cells were lysed by freeze-thaw three times followed by
sonication using three 2- to -3-sec bursts. The insoluble debris
was removed by centrifugation at 15,000 x g for 30 min. Protein
concentrations were determined using the Lowry method (35). Each protein sample (25 μg)
was resolved by SDS-PAGE (12%). The gels were electroblotted onto
hydrophobic polyvinylidene difluoride membranes (Hybond™-P PVDF, GE
Healthcare Bio-Science, Uppsala, Sweden). Antibody staining was
performed with a chemiluminescence detection system (ECL Plus
Western Blotting Detection Reagent, GE Healthcare Bio-Science),
using a 1:500 dilution of the mouse anti-human TS (TS106)
monoclonal primary antibody (Invitrogen S.r.l., Milan, Italy),
1:1,000 dilutions of the mouse anti-human DHFR monoclonal antibody
(Tebu-Bio, Milan, Italy) and 1:1,000 of mouse anti-human β-tubulin
antibody (Sigma-Aldrich) in conjunction with a 1:3,000 dilution of
horseradish peroxidase-conjugated sheep anti-mouse secondary
antibody (GE Healthcare Bio-Science). Quantification of signal
intensity was performed by densitometry on a GS-800 calibrated
densitometer (Bio-Rad) and analysed by using Quantity One software
(Bio-Rad, CA, USA).
Real-time reverse transcription-PCR
analysis
Total RNA was extracted from the cultured cells
using TRI reagent (Sigma-Aldrich). Reverse transcription was
performed with 2 μg of total RNA using random primers
(Promega, Milan, Italy) and M-MLV reverse transcriptase (Promega).
Real-time RT-PCR was performed with 10 ng of cDNA using Power
SYBR® Green PCR Master Mix [Applied Biosystems, Monza
(MI), Italy] and an ABI-PRISM 7900 HT Sequence Detection System
(SDS, Applied Biosystems), followed by a dissociation curve
analysis and subsequent agarose gel electrophoresis to confirm
amplification. The following primer sets were used: TS [NCBI
reference sequence: NM_001071.1], forward: 5′-CAG
ATTATTCAGGACAGGGAGTT-3′, reverse: 5′-CATCAGAG GAAGATCTCTTGGATT-3′;
DHFR [NCBI reference sequence: NM_000791.3], forward: 5′-TGCACAAATG
GGGACGA-3′, reverse: 5′-GGAAATATCTGAATTCAT TCCTGAG-3′; and GAPDH
[NCBI reference sequence: NM_002046.3], forward:
5′-CAAGGTCATCCATGACAA CTTTG-3′, reverse:
5′-GGGCCATCCACAGTCTTCTG-3′. SSAT primer sequences were, forward:
5′-TTATAGAGGCT TTGGCATAGGA-3′, reverse: 5′-TCATTGCAACCTGGCT
TAGA-3′. The amount of target, normalised to an endogenous
reference (glyceraldehydes-3-phosphate dehydrogenase, GAPDH) and
relative to a calibrator (2008 cell line or untreated sample), was
given by 2−ΔΔCt calculation (40). All experiments were carried out
three times in triplicate; amplification plots were analysed using
the ABI-PRISM 7900 HT SDS version 2.1 software (Applied
Biosystems).
[3H]folic acid uptake
studies
[3H]folic acid uptake studies were
performed according to previously published methods (41) with minor modifications. Briefly,
one day after seeding, cells were treated for 72 h and then
incubated for 10 min at 37°C with 50 nM of [3H]folic
acid, after a 20-min acidic treatment with stripping buffer
(acetate pH 4.0) to remove endogenous folates bound to folate
receptor (FR) at the cell surface. At the end of each experiment,
cells were washed with 3X 1 ml of ice-cold PBS pH 7.4, to arrest
the reaction. The cells were then solubilised with 0.3 ml 0.1%
(v/v) Triton X-100 in 1% NaOH and placed at 37°C overnight. An
aliquot was transferred to scintillation vials containing 5 ml of
scintillation cocktail. Radioactivity associated with the cells was
quantified using a scintillation counter (TriCarb 2100, Packard,
USA) and the protein content of each sample measured by the method
of Lowry using bovine serum albumin as the standard.
Assay of SSAT activity
SSAT activity was measured as previously described
(42). The cells were harvested,
washed twice in PBS and suspended in a buffer containing 10 mM
tris(hydroxymethyl)aminomethane (pH 7.2) and 1 mM dithiothreitol.
This suspension was freeze-thawed twice, then cytosolic aliquots
were incubated in 100 mM tris(hydroxymethyl)aminomethane (pH 8.0),
3 mM Spd and 0.5 nmol 1-[14C]acetyl coenzyme A in a
final volume of 50 μl for 10 min at 30°C. The reaction was
stopped by adding 10 μl of 1 M NH2-OH HCl and
boiling in water for 3 min. The resulting samples were spotted onto
P-81 phosphocellulose discs and radioactivity measured by
scintillation counting. The amount of cytosol added to the final
reaction mixture was adjusted to maintain the enzyme/substrate
concentrations within the linear range. Enzyme activity is
expressed as pmol [14C]acetylspermidine formed/min/mg
protein.
Synergy analysis
The nature of the interaction between berberine and
cisplatin or the polyamine analogue, combined simultaneously at a
fixed ratio based on the different cell sensitivity to each drug,
was determined using median-effect analysis (43), with the CalcuSyn ver. 2.0 software
(Biosoft, Cambridge, UK), which calculates a non-exclusive case
combination index (CI) for every fraction affected (FA), a measure
of the drug interaction effects. CI values of <1 or >1
indicated synergy and antagonism, respectively, whereas a CI value
of 1 indicated additive effects of the drugs. Growth inhibition was
assayed by the crystal violet dye assay to determine the
dose-response curves for each agent alone and in combination at a
fixed ratio based on their IC50 values. Computer
analysis of the dose-response curves was used to calculate the
combination index (CI) at increasing levels of cell kill.
Statistical analysis
All values report the mean ± SEM, unless otherwise
indicated. Statistical significance was estimated by two-tailed
Student’s t-test performed using Microsoft Excel software; a
difference was considered significant at P<0.05 or
P<0.01.
Results
Effect of berberine on the growth of
cDDP-sensitive and -resistant cell lines
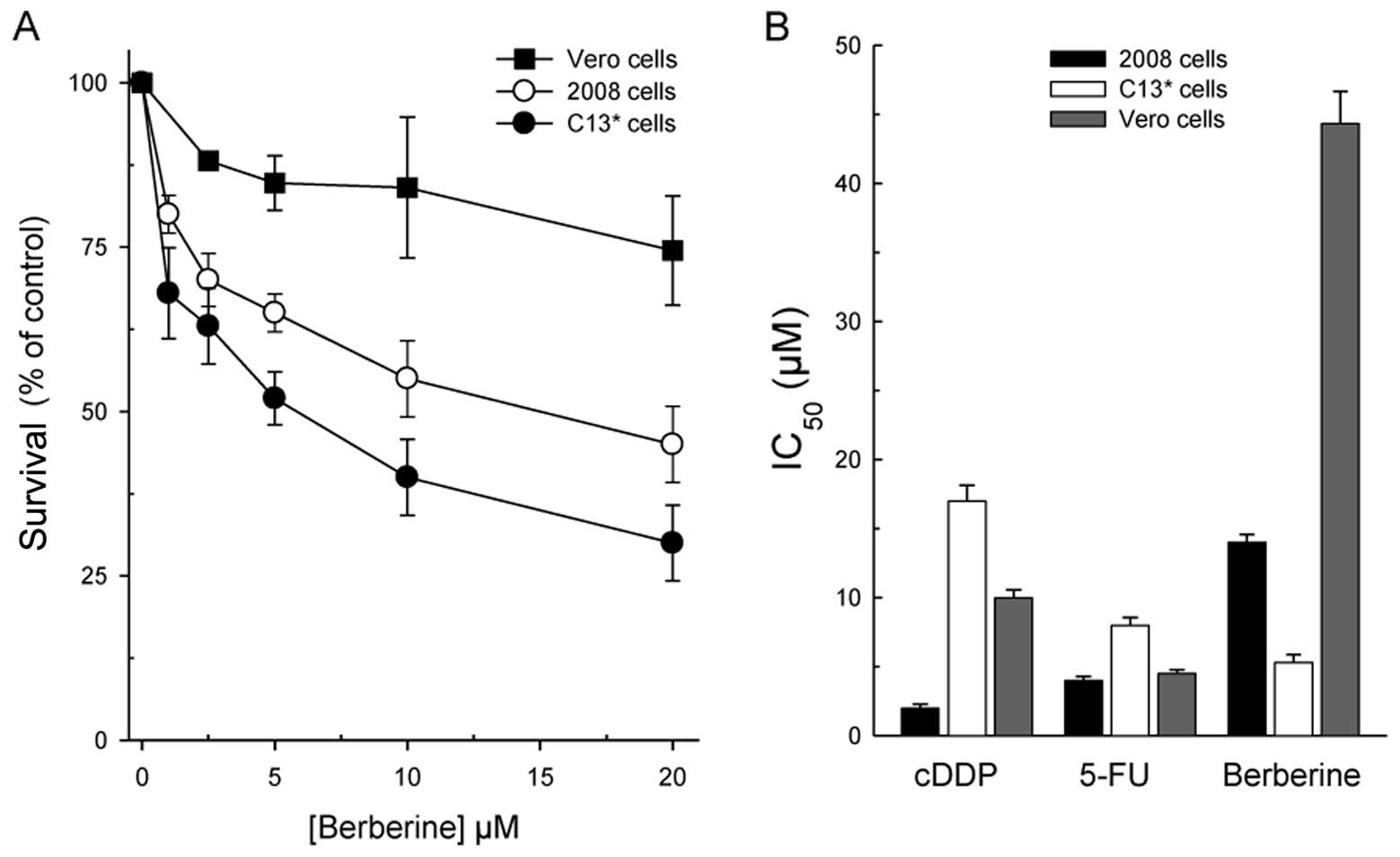
The dose-response curves of berberine in the
cDDP-sensitive and -resistant cell lines (2008 and C13*)
and Vero cells, depicted in Fig.
2A reveal a collateral sensitivity of the cDDP-resistant line
(IC50 = 5.3±0.6 μM versus IC50 =
14.5±1.2 μM in 2008 cDDP-sensitive cells) to the drug.
Collateral sensitivity is a phenomenon which describes a cell
population that is resistant to certain drugs and is more sensitive
to others. Berberine is also quite selective since it was much less
cytotoxic against the non-tumorigenic Vero lineage. This feature
along with the comparison between the efficacy of berberine and
that of the two well-known anticancer drugs, cDDP and 5-FU, towards
the three cell lines is displayed in Fig. 2B. As is evident, berberine is the
least effective of the three drugs against the parental line, but
the most active towards the resistant one. Noteworthy, the 50%
growth reduction of Vero cells was not reached even with 40
μM berberine, but already with 10 μM cDDP and 4.8
μM 5-FU, indicating that berberine is more selective than
the traditional chemotherapeutic compounds.
Modulation of TS and DHFR expression by
berberine
We have previously reported that the cDDP-resistant
human ovarian cancer cell line C13* showed collateral
sensitivity towards two quinoxalinic compounds, new molecules
designed as folate cycle enzyme inhibitors and structurally
unrelated to the classical pteridine-like compounds and that this
effect was related to folate cycle enzyme impairment (44). The resemblance of berberine effects
with those of the two quinoxalines prompted us to evaluate and
compare the effects of berberine on TS and DHFR expression in this
acquired cDDP-resistant line and its parental line 2008.
It is interesting to note that, unlike traditional
folate cycle inhibitors such as 5-FU, the cytotoxicity of berberine
was accompanied by a greater inhibition of TS and DHFR expression
in cell extracts from resistant cells than from sensitive ones.
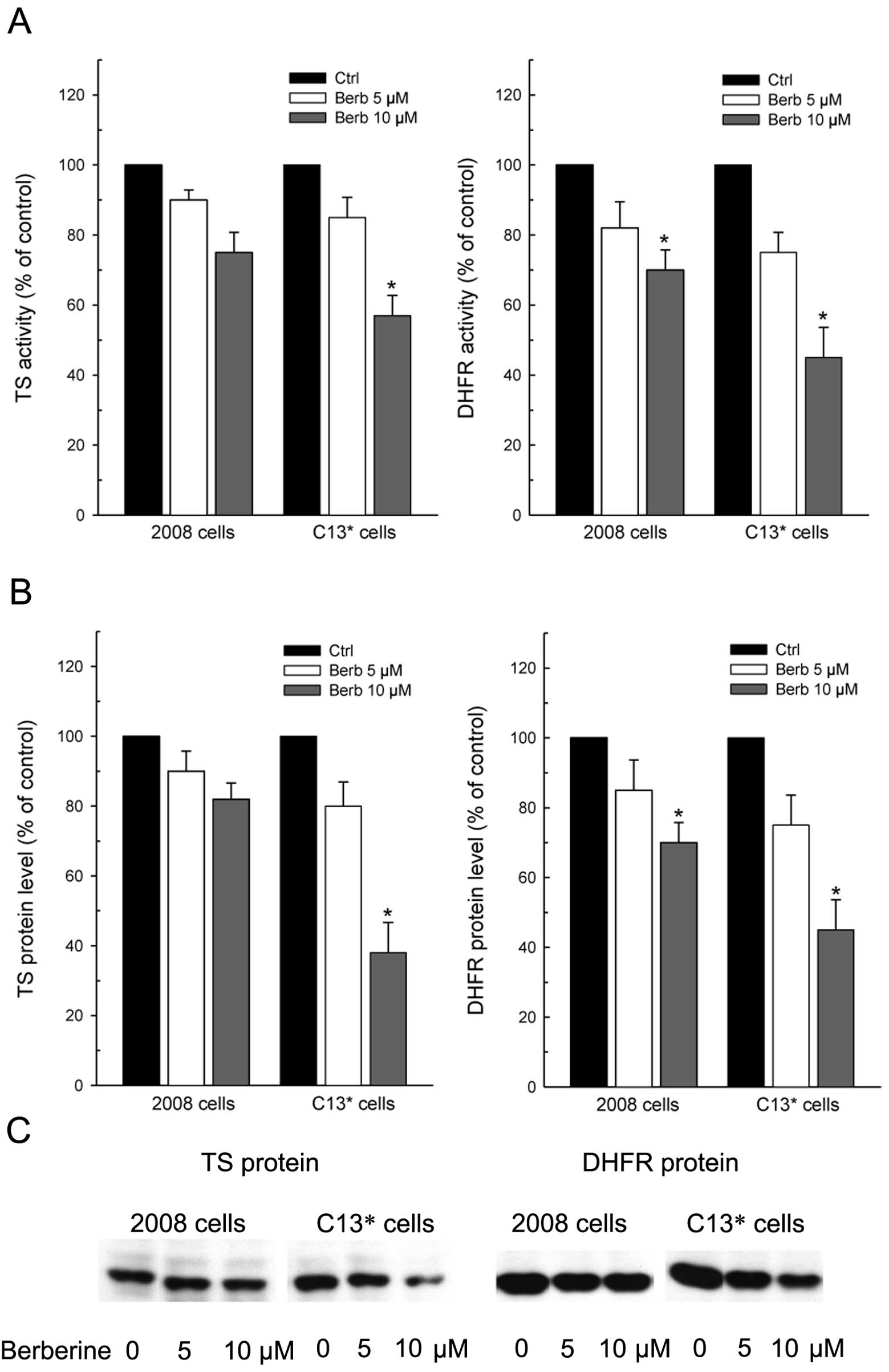
Therefore, the data reported in Fig.
3 explain, at least partly, the collateral sensitivity showed
by C13* cells to berberine, since both TS and DHFR
activities were decreased more in C13* than in 2008
cells by increasing drug concentrations (Fig. 3A). TS activity was reduced at 10
μM berberine by 30 and 45% of the controls in 2008 and
C13* cells, respectively; whereas, the residual DHFR
activity after treatment was 70 and 45% of the respective
controls.
This differential decrease of the enzyme activities
reflected well the levels of both TS and DHFR proteins, which were
both more downregulated in cDDP-resistant than in the sensitive
cells (Fig. 3B and C).
Notably, again unlike 5-FU (44), berberine reduced the level of TS
monomeric form without inducing ternary complex formation, which is
considered a mechanism of resistance to 5-FU (45). Data from densitometric scanning
indicated that residual TS protein levels were ∼80 and 40% of
control after treatment with the higher concentration of berberine
in cDDP-sensitive and in -resistant cells, respectively. Even DHFR
protein was reduced by 30 and 55% in 2008 and C13* cells
when compared to the respective controls.
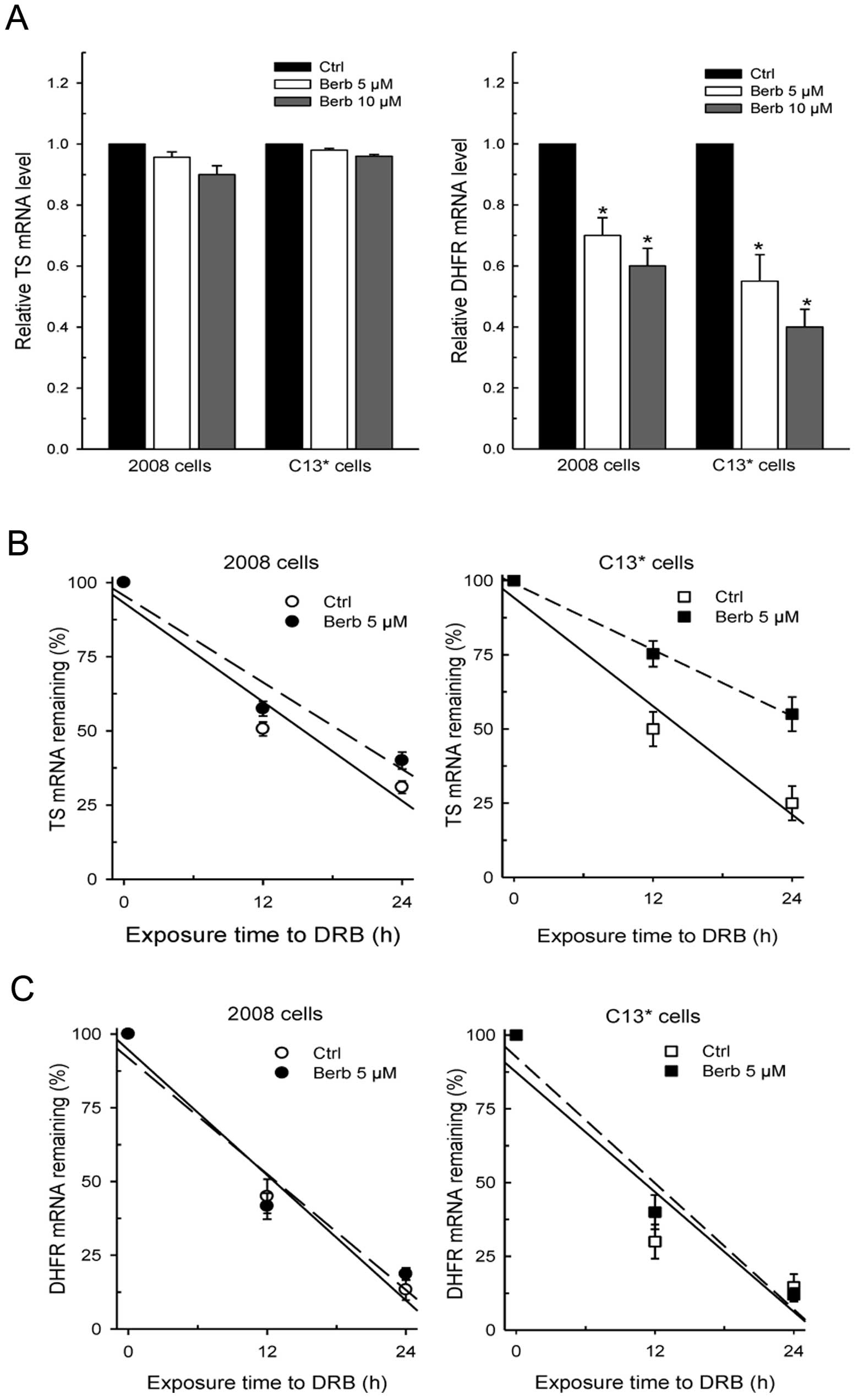
Surprisingly, RT-PCR analysis revealed that only
DHFR protein levels correlated with the respective decrease of DHFR
mRNA amount, again more pronounced in the resistant line. In
particular, the remaining DHFR mRNAs in 2008 cells were 70–80% of
controls after treatment, while being 50–40% of control
C13* cells. On the contrary, TS transcript was not
significantly affected by drug exposure in either cell line
(Fig. 4A).
To clarify whether the failure in impairing TS mRNA
levels and the observed small difference between sensitive and
resistant lines was also due to an effect of the drug on the
turn-over of TS mRNA transcripts, we compared the half-life of TS
and DHFR messengers in the two cell lines pretreated with berberine
5 μM for 72 h and then co-treated with the transcription
inhibitor dichlorobenzimidazole riboside (DRB) to shut off
transcription. The rates of mRNA decay were then monitored over a
24-h period. The data reported in Fig.
4B and C indicate that there is no difference in both basal TS
mRNA and DHFR mRNA stability between sensitive and resistant cells.
However, as is evident from the regression plots of Fig. 4B, treatment with berberine caused a
remarkable slowing of the TS mRNA degradation rate in
C13* cells, which turned over at a rate almost twice
lower than that in sensitive cells. As a result, TS mRNA stability
increased to a lesser extent in 2008 cells from a half-life of 14.5
h in controls to 18.3 h in berberine-treated cells, but to a much
greater extent in the resistant line, from a half-life of 14.2 to
23.8 h, respectively. On the other hand, the rate of DHFR mRNA
degradation was not affected by berberine in either cell line as
the percentage of the remaining messengers was similar to the
controls after drug exposure (Fig.
4C).
Noteworthy, in these conditions, berberine affected
the cellular uptake of folic acid from culture medium (Table I). In particular, 10 μM
berberine reduced the accumulation of 50 nM [3H]folic
acid by ∼13.3 and 27.6% in 2008 and C13* cells,
respectively (from 163.3±4 to 141.4±7 fmoles/mg of protein in 2008
cells and from 182.8±9 to 132.9±11 fmoles/mg of protein in
C13* cells; n=3). Again, this differential effect
correlates with the collateral sensitivity to berberine showed by
the latter line.
 | Table I.Effect of berberine and BESpm on the
uptake of [3H]folic acid in 2008 and C13*
cells. |
Table I.
Effect of berberine and BESpm on the
uptake of [3H]folic acid in 2008 and C13*
cells.
| Drugs | 2008 cells
| C13*
cells
|
|---|
| fmoles/mg prot | (%) | fmoles/mg prot | (%) |
|---|
| Ctrl | 163.3±4 | 100 | 182.8±9 | 100 |
| Berberine 10
μM | 141.4±7 | 86.59 | 132.9±11 | 72.70 |
| BESpm 10
μM | 135.2±15 | 82.79 | 139.15±8 | 76.12 |
Modulation of folate cycle enzymes and
SSAT by combining berberine and Spm analogue
We have previously shown that these resistant cells
are less responsive than their cDDP-sensitive counterparts the 2008
cells, to both the spermine analogue N1,N12-bisethylspermine
(BESpm) (46) and the traditional
anti-folates (44), when
administered alone. Therefore, we also evaluated the effects of
berberine combined with the polyamine analogue in the human ovarian
cDDP-resistant C13* cells.
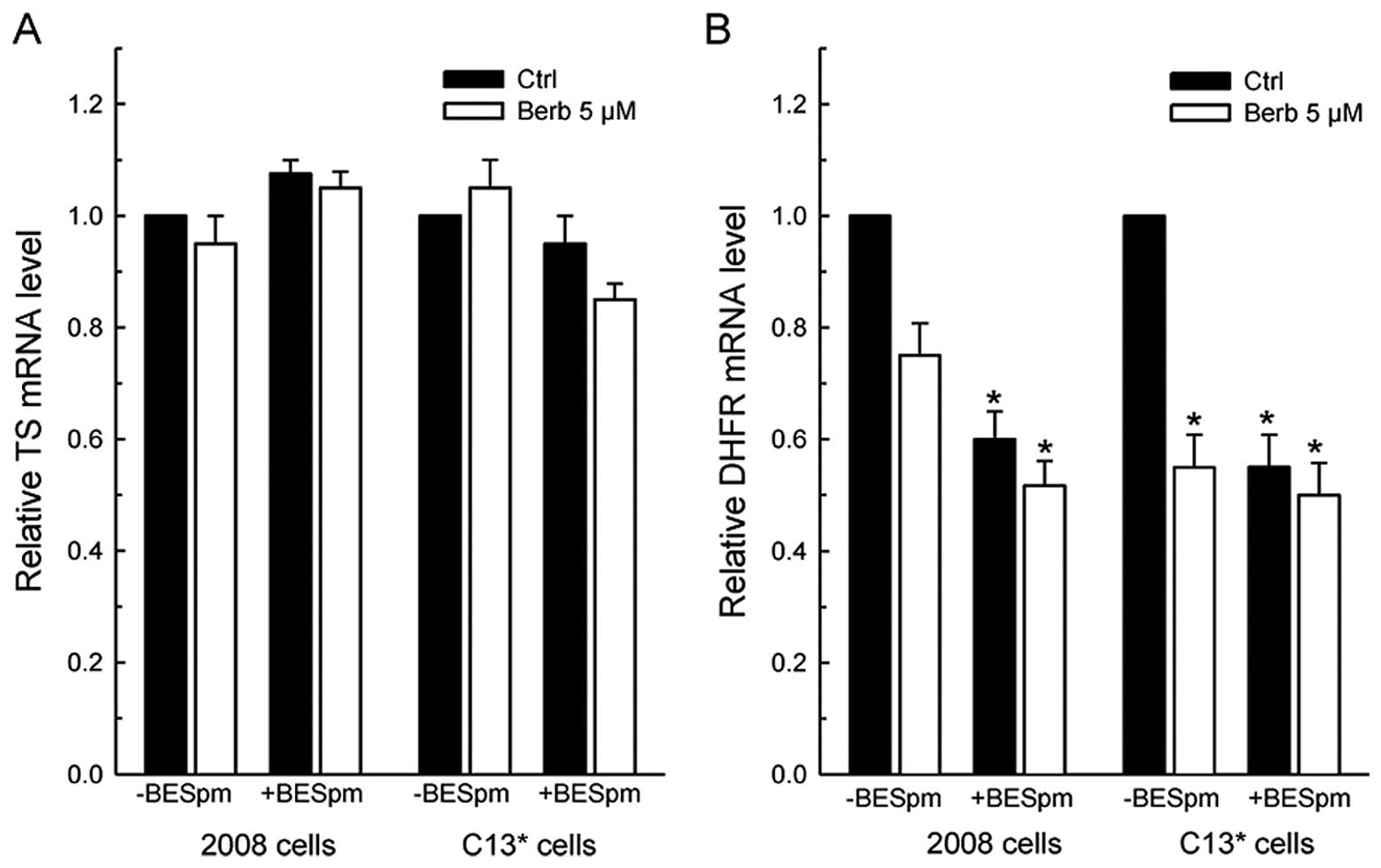
RT-PCR analysis revealed that 10 μM BESpm,
concentration chosen from the previously reported dose-response
studies (32) and berberine did
not significantly affect the TS mRNA levels in either cell line
(Fig. 5A). However, both drugs
decreased the level of DHFR mRNA in the cell lines, again
particularly in resistant cells, but without potentiated effects
when combined (Fig. 5B).
Similarly to berberine, the modulation of folate
cycle enzymes by BESpm seems partly ascribable to the reduced
folate uptake in the cell lines. As reported in Table I, 10 μM BESpm reduced the
accumulation of 50 nM [3H]folic acid by ∼17 and 24% in
2008 and C13* cells, respectively (from 163.3±4 to
135.2±15 fmoles/mg of protein in 2008 cells and from 182.8±9 to
139.15±8 fmoles/mg of protein in C13* cells; n=3).
The effect on SSAT expression was evident. Fig. 6A shows that SSAT mRNA level was
almost tripled and doubled by berberine in 2008 and C13*
cells, respectively, reaching the same induction caused by BESpm.
Notably, berberine alone increased SSAT expression by itself but
even more interestingly, stimulated the SSAT overexpression by the
polyamine analogue. In fact, when combined, berberine increased by
2- and 1.5-fold SSAT mRNA level induced by BESpm in sensitive and
resistant cells, respectively. As a consequence, SSAT activity was
increased by berberine alone in both cell lines, but the drug
over-induced the enzyme activity by the analogue additively in
sensitive cells and synergistically in resistant cells (Fig. 6B).
Therefore, despite SSAT expression being less
inducible by the analogue in the C13* resistant cells
compared to sensitive 2008 cells (46), in the presence of berberine, the
reduced SSAT activity in the resistant line was reverted to
induction level of the enzyme in sensitive cells by the
co-treatment of berberine with Spm analogue.
Effects on cell growth by combining
berberine with cDDP or with BESpm
Previous studies have shown that berberine
potentiated the chemotherapeutic effect of cDDP by enhancing
apoptosis in HeLa cells (10).
Accordingly, we have ascertained the nature of the combination of
berberine with cDDP, combined simultaneously in our cDDP-sensitive
and -resistant cell model, determined by median-effect analysis.
Cells were exposed to each drug alone and to their combinations at
constant ratio, deduced from IC50 values. The survival
data were then plugged into the software CalcuSyn ver. 2.0
(Biosoft), which provides the combination index (CI) for each level
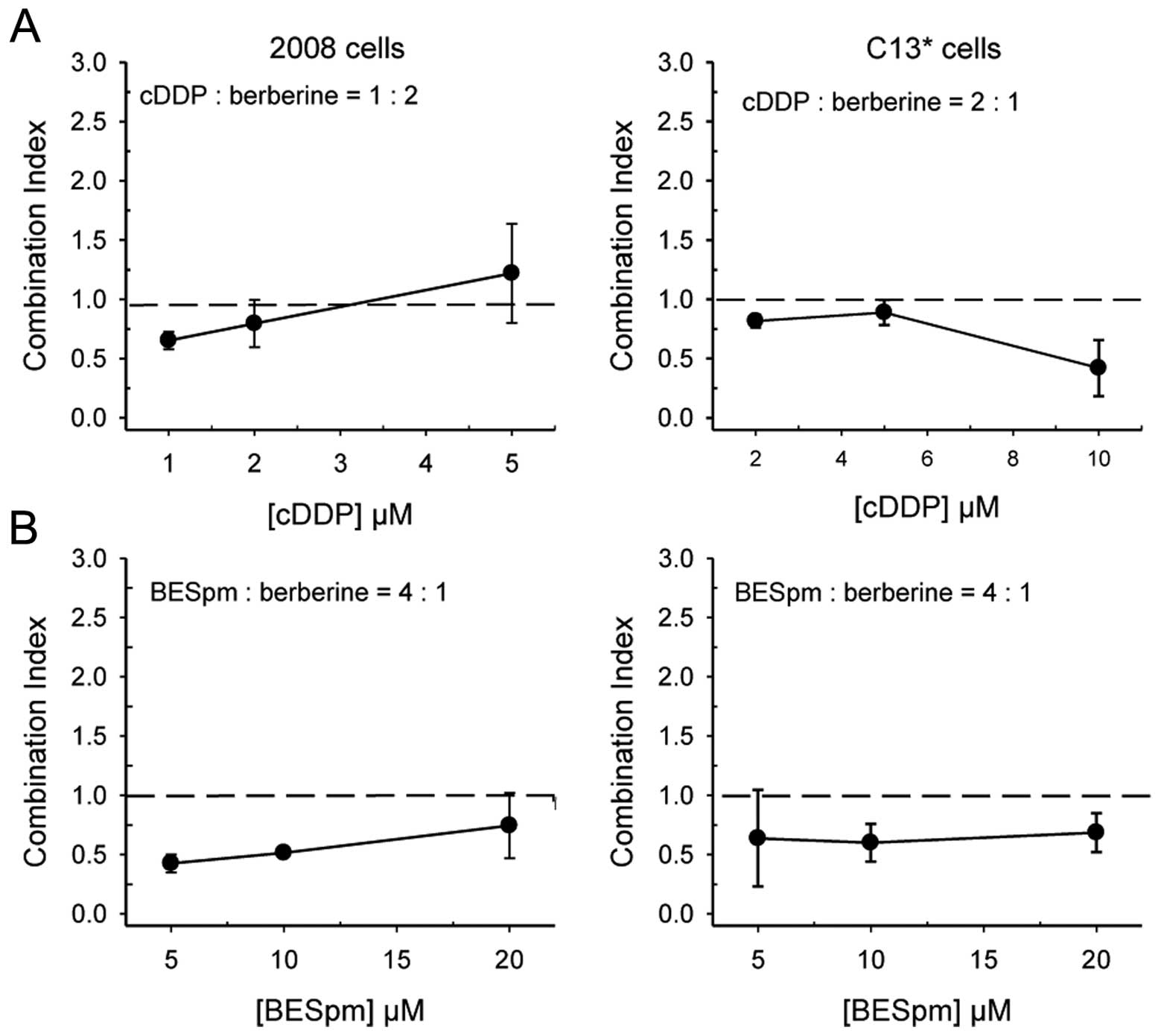
of cell kill. As depicted in Fig.
7A, the combination of cDDP and berberine resulted in mostly
supra-additive effects in 2008 cells and mostly synergistic in
C13* cells, confirming that berberine may facilitate
cDDP activity, even in resistant cells.
We have also ascertained the nature of the
combination of berberine with BESpm, combined simultaneously at a
fixed ratio. As expected from the data of the modulation of the
folate cycle enzymes and of the stimulated induction of SSAT
expression, berberine synergistically increased cell growth
inhibition by BESpm in both cell lines, even in resistant cells.
Fig. 7B shows that berberine and
Spm analogue combination is almost as effective in sensitive 2008
cells as in resistant C13* cells, producing synergistic
cell killing in both lines.
Discussion
The natural isoquinoline berberine has shown
antiproliferative activity against a variety of human cancer cells
(14,15) and it has also been suggested as an
anticancer drug for the treatment of ovarian cancer since it was
shown to inhibit the proliferation of human ovarian tumor SVKO3
cells (47).
We show for the first time, that despite the
cytotoxicity of berberine against a human ovarian cell line it is
lower than that of the traditional anticancer drugs such as cDDP
and 5-FU, the isoquinoline is more effective than the
chemotherapeutic agents against the derived cDDP-resistant
counterpart, C13* cells, which is even more sensitive to
the alkaloid than parental cells, displaying collateral
sensitivity. Besides, the natural compound was also more selective,
showing scant toxicity towards non-tumorigenic Vero lineage.
Even if other mechanisms cannot be ruled out, the
collateral sensitivity seems to correlate, at least partly, with
the higher downregulation of TS and DHFR in C13* cells
compared to 2008 cells. These effects are more relevant since this
resistant line showed elevated expression of the folate cycle
enzymes, thymidylate synthase (TS) and dihydrofolate reductase
(DHFR), due to cisplatin-resistance phenotype (2).
The modulation of folate cycle enzyme expression and
the cytotoxicity may be explained by the ability of berberine to
bind nucleic acids. The binding of berberine to DNA and RNA has
been defined sufficiently efficient and very similar to ethidium
bromide to justify its use in spectrofluorimetric determination of
the nucleic acids (48).
The anticancer activity of berberine appears to
derive from its ability to form strong complexes with
adenine-thymine base pair specific regions of nucleic acids, induce
DNA damage and exert related effects such as telomerase inhibition,
topoisomerase poisoning and inhibition of gene transcription
(49–51). The antiproliferative activity was
also recently ascribed to its ability to bind specifically to
oligonucleotides and to stabilize DNA triplexes or G-quadruplexes
via telomerase and topoisomerase inhibition (52,53).
A non-classical intercalation model for berberine DNA binding has
been confirmed (54). Very
recently, substitution at the 13-position with unprecedented
phenylalkyl groups remarkably enhanced the DNA binding action of
berberine, providing insights into the importance of the
13-position substitution of berberine. This may be useful for the
design and development of berberine structurally related new drugs
with more specific and higher efficacy (55).
It has been reported that DNA intercalating agents
and DNA minor groove-binders also bind to TS mRNA site 1 construct
(56). Moreover, we have also
recently shown that the antibiotic distamycin, which binds at the
relatively narrow AT-rich sequences of the minor groove of
double-strand DNA (23,24), affects the expression of folate
cycle enzymes even at the cellular level; this minor groove binder
is able to modulate TS mRNA in vivo, thus affecting the
translation and enzyme activity (22).
Similarly, berberine was shown to bind
preferentially to AT rich sequences in DNA (57), also present in TS mRNA, thus
accounting for the effect on folate enzymes expression and
consequently on cytotoxicity.
Berberine binding to DNA probably also affects TS
and DHFR gene transcription, but post-transcriptional effects such
as stabilization affects TS mRNA levels rather than DHFR mRNA
levels, suggesting a preferential binding to the former mRNA rather
than the latter. In addition, the post-transcriptional
stabilization of TS mRNA in C13* cells, more than in
2008 cells, may indicate differences in transcript structures
affecting berberine binding and/or differential resistance to mRNA
catabolic enzymes between sensitive and resistant cells. However,
in our experiments, berberine modulated the expression of TS and
DHFR both at transcriptional and post-transcriptional level, since
DHFR mRNA level decreased more in C13* cells than in
2008 cells, whereas TS mRNA levels were almost not affected by
berberine in the cell lines. Nevertheless, the final protein levels
of the two enzymes and their activities were almost equally
affected, but in particular in resistant cells. Therefore, TS
expression appears to be the more differentially modulated by the
alkaloid, as a lack of effect at transcriptional level was
displayed both in sensitive and in resistant cells, along with a
significantly reduced TS mRNA turnover only in C13*
cells. Of note, the impairment of the folate cycle also seems
partly ascribable to a reduction of folic acid accumulation in both
cell lines. In particular, the resistant cells treated with
berberine accumulate less folic acid than sensitive cells when
compared to the respective untreated controls. Folate is a member
of vitamin B family and plays an essential role in cell survival by
participating in the biosynthesis of nucleic and amino acids.
Again, the slightly more compromised accumulation of this parent
compound of purine and pyrimidine bases and amino acids contributes
to explain the reduced tolerance to the isoquinoline alkaloid of
the C13* cells.
DNA binding ligands, including minor groove binders
such as distamycin and intercalators are considered amplifiers of
the DNA damaging effects of anticancer drugs (58). Therefore, as distamycin
synergistically enhanced cDDP cell killing (22), similarly, it is likely that
berberine potentiated the action of cDDP, as evidenced by the
fraction affected of the cells killed in synergy studies, by means
of the downregulation of TS and DHFR expression, thus preventing
the new synthesis of damaged DNA. This mechanism is also supported
by the reduced accumulation of folic acid, particularly in the
resistant cells.
The upregulation of folate cycle enzymes in ovarian
carcinoma cells (6), including
this cDDP-resistant line (44) and
the deficient SSAT expression previously reported (46) represent mechanisms of reduced
sensitivity to better survive the cell inhibition caused by
antifolates such as 5-FU and by polyamine depleting drugs, such as
Spm analogues, but also the drug injury brought about by the
alkylating agents such as cDDP. Therefore, our efforts were aimed
to potentiate cDDP cytotoxicity even in cDDP-resistant cells by
targeting the folate cycle and the polyamine metabolism, which have
been shown to interrelate (27,28).
Our group reported previously a synergistic increase in SSAT mRNA
levels and cell death in response to the combination with novel TS
inhibitors and SSAT inducers (29).
We report now, that the combination of berberine and
the Spm-analogue BESpm modulates folate cycle enzymes, as reflected
in the reduction of DHFR mRNAs in particular; whereas TS mRNAs were
affected only to a lesser extent and in resistant cells, confirming
that interrelations exist between these two metabolic pathways in
this cell model. The data reported suggest that the synergistic
inhibition of cell growth by berberine and BESpm combination
involves DHFR gene downregulation more than TS gene, again mainly
in resistant cells. This effect is accompanied by the induction of
SSAT activity in the cell lines. However, the effect of this drug
combination in sensitive cells seems to be mediated by the
induction of polyamine catabolism rather than by folate cycle
modulation, whereas, in resistant cells both metabolisms are more
affected.
It is almost consolidated evidence that when both
the folate cycle and the polyamine metabolism are concurrently
modulated, cancer cell growth from different tumor types may be
more validly treated. In particular, the combination of classical
or novel inhibitors of TS and DHFR with inducing agents of the key
catabolic enzyme of polyamine catabolism, SSAT, such as DENSpm or
BESpm has proven therapeutic efficacy against tumor cell lines
(27–29).
Accordingly, our results show that the co-treatment
with berberine and BESpm, which caused DHFR downregulation and SSAT
induction, resulted in a synergic cell killing in both lines,
reverting the sensitization of resistant cells to the Spm analogue
to the level of the sensitive counterpart. This evidence,
correlating with the synergic induction of SSAT activity by drug
combination is particularly important in resistant cells, since the
cDDP-resistance phenotype conferred defective basal SSAT expression
in C13* cells in comparison to parental 2008 cells and
thus a reduced sensitivity to the induction of SSAT activity and
cell growth inhibition by the polyamine analogues (29,46).
The consequent polyamine depletion caused by SSAT
induction by berberine and BESpm combination may facilitate DNA
intercalation by berberine, along with decreased DNA aggregation
leading to relaxation of chromatin structure (59). This may favour the interaction and
the effects of the analogue with DNA, partly accounting for the
enhanced cytotoxicity of the combinations. In addition, the
enhanced induction of polyamine catabolism leads to increased
acetylated Spm or Spd concentration and to elevation of reactive
oxygen species (29,60), which correlating with Spm
depletion, may initiate the apoptotic cascade and account for
synergistic interactions.
Moreover, decarboxylated S-adenosylmethionine (SAM)
is an essential precursor in the formation of both spermidine and
spermine. SAM is formed from methionine through the addition of
adenosine. Because 5-methyltetrahydrofolate donates a methyl group
to homocysteine to produce methionine, folate deficiency may
decrease polyamine synthesis (61). Therefore, BESpm may cause polyamine
depletion both by the well-known direct action on the enzymes of
polyamine metabolism and by reducing folate uptake by cells,
particularly by resistant ones.
In conclusion, our data show for the first time that
a cDDP-resistant and polyamine analogue cross-resistant cell line
is more sensitive to berberine than the parental cell line. This
collateral sensitivity is partly attributable to a more pronounced
downregulation of folate cycle enzymes in cDDP-resistant than in
the sensitive cells. These data also show that berberine is more
effective against resistant cells than cDDP or 5-FU and even more
selective than the traditional anticancer drugs. Besides, we
demonstrate that the natural compound berberine enhances the cell
growth inhibition caused by cDDP and confirm the interrelationship
between folate cycle and polyamine metabolism, since SSAT
expression was upregulated by the combination of berberine and an
Spm analogue, accounting for the observed synergistic cell
killing.
Abbreviations:
|
cDDP
|
cisplatin or
cis-diamminedichloroplatinum;
|
|
Put
|
putrescine;
|
|
Spd
|
spermidine;
|
|
Spm
|
spermine;
|
|
SSAT
|
spermidine/spermine
N1-acetyltransferase;
|
|
BESpm
|
N1,N12-bisethylspermine;
|
|
DENSpm
|
N1,N11-diethylnorspermine;
|
|
DHFR
|
dihydrofolate reductase;
|
|
TS
|
thymidylate synthase;
|
|
5-FU
|
5-fluorouracil;
|
|
MTX
|
methotrexate;
|
|
CH2FH4
|
5,10-methylenetetrahydrofolate;
|
|
FH4
|
tetrahydrofolate
|
Acknowledgements
This study was supported by EU LIGHTS
(LIGands to interfere with Human Thymidylate Synthase) project of
the 6th Framework Program, LSHCCT-2006-037852 and AIRC IG 10474 to
M.P. Costi. This study was also supported by Associazione Angela
Serra per la Ricerca sul Cancro, Azienda Ospedaliera Policlinico di
Modena, Modena, Italy.
References
|
1.
|
Ozols RF, Bookman MA, Connolly DC, et al:
Focus on 433 epithelial ovarian cancer. Cancer Cell. 5:19–24. 2004.
View Article : Google Scholar
|
|
2.
|
Muggia F: Platinum compounds 30 years
after the introduction of cisplatin: implications for the treatment
of ovarian cancer. Gynecol Oncol. 112:275–281. 2009.PubMed/NCBI
|
|
3.
|
Jakubowicz-Gil J, Paduch R, Piersual T,
Glowniak K, Gawron A and Kandefer M: The effect of quercetin on
pro-apoptotic activity of cisplatin in HeLa cells. Biochem
Pharmacol. 6:1343–1350. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
4.
|
Maeda H, Hori S, Ohizumi H, Segawa T,
Kakehi Y, Ogawa O and Kakizuka A: Effective treatment of advanced
solid tumors by the combination of arsenic trioxide and
L-buthionine-sulfoximine. Cell Death Differ. 11:737–746. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
5.
|
Umanzor J and Aguiluz M: Concurrent
cisplatin/gemcitabine chemotherapy along with radiotherapy in
locally advanced cervical carcinoma: a phase II trial. Gynecol
Oncol. 100:70–75. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
6.
|
Scanlon KJ and Kashani-Sabet M: Elevated
expression of thymidylate synthase cycle genes in
cisplatin-resistant human ovarian carcinoma A2780 cells. Proc Natl
Acad Sci USA. 85:650–653. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
7.
|
Wysocki PJ: Targeted therapy of
hepatocellular cancer. Expert Opin Investig Drugs. 19:265–274.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
8.
|
Lin CC, Yang JS, Chen JT, et al: Berberine
induces apoptosis in human HSC-3 oral cancer cells via simultaneous
activation of the death receptor-mediated and mitochondrial
pathway. Anticancer Res. 27:3371–3378. 2007.PubMed/NCBI
|
|
9.
|
Mantena SK, Sharma SD and Katiyar SK:
Berberine, a natural product, induces G1-phase cell cycle arrest
and caspase-3-dependent apoptosis in human prostate carcinoma
cells. Mol Cancer Ther. 5:296–308. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10.
|
Yoon MJ, So HS, Cho HJ, et al: Berberine,
a natural product, combined with cisplatin enhanced apoptosis
through a mitochondria/caspase-mediated pathway in HeLa cells. Biol
Pharm Bull. 31:789–795. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11.
|
Yi ZB, Yan Y, Liang YZ, et al: Evaluation
of the antimicrobial mode of berberine by LC/ESI-MS combined with
principal component analysis. J Pharm Biomed Anal. 44:301–304.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
12.
|
Liu JC, Chan P, Chen YJ, et al: The
antihypertensive effect of the berberine derivative
6-protoberberine in spontaneously hypertensive rats. Pharmacology.
59:283–289. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
13.
|
Kuo CL, Chi CW and Liu TY: The
anti-inflammatory potential of berberine in vitro and in vivo.
Cancer Lett. 203:127–137. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
14.
|
Un Y, Xun K, Wang Y, et al: A systematic
review of the anti-cancer properties of berberine, a natural
product from Chinese herbs. Anticancer Drugs. 20:757–769. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
15.
|
Tang J, Feng Y, Tsao S, et al: Berberine
and Coptidis rhizoma as novel antineoplastic agents: a
review of traditional use and biomedical investigations. J
Ethnopharmacol. 126:5172009.
|
|
16.
|
Kong B, Huang S, Wang W, May D, et al:
Arsenic trioxide induces apoptosis in cisplatin-sensitive and
-resistant ovarian cancer cell lines. Int J Gynecol Cancer.
15:872–877. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
17.
|
Lin JP, Yang JS, Lee JH, Hsieh WT and
Chung JG: Berberine induces cell cycle arrest and apoptosis in
human gastric carcinoma SNU-5 cell line. World J Gastroenterol.
12:21–28. 2006.PubMed/NCBI
|
|
18.
|
Halestrap AP, Doran E, Gillespie JP and
O’Toole A: Mitochondria and cell death. Biochem Soc Trans.
28:170–171. 2000.
|
|
19.
|
Duverger V, Sartorius U,
Klein-Bauernschmitt P, Krammer PH and Schlehofer JR: Enhancement of
cisplatin-induced apoptosis by infection with adeno-associated
virus type 2. Int J Cancer. 97:706–712. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
20.
|
Li JJ, Tang Q, Li Y, Hu BR, Ming ZY, Fu Q,
Qian JQ and Xiang JZ: Role of oxidative stress in the apoptosis of
hepatocellular carcinoma induced by combination of arsenic trioxide
and ascorbic acid. Acta Pharmacol Sin. 27:1078–1084. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
21.
|
Jantova S, Cipak L and Letasiova S:
Berberine induces apoptosis through a mitochondrial/caspase pathway
in human promonocytic U937 cells. Toxicol In Vitro. 21:25–31. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
22.
|
Marverti G, Guaitoli G, Ligabue A,
Frassineti C, Monti MG, Lombardi P and Costi MP: Distamycin A and
derivatives as synergic drugs in cisplatin-sensitive and -resistant
ovarian cancer cells. Amino Acids. 42:641–653. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23.
|
Arcamone F, Pencos PG, Orezzi PG,
Nicolella V and Pirelli AM: Structure and synthesis of distamycin
A. Nature. 203:1064–1065. 1964. View Article : Google Scholar : PubMed/NCBI
|
|
24.
|
Abu-Daya A and Fox KR: Interaction of
minor groove binding ligands with long AT tracts. Nucleic Acids
Res. 25:4962–4969. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
25.
|
Carreras CW and Santi DV: The catalytic
mechanism and structure of thymidylate synthase. Annu Rev Biochem.
64:721–762. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
26.
|
Costi P and Ferrari S: Update on
antifolate drugs targets. Curr Drug Targets. 2:135–166. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
27.
|
Choi W, Gerner EW, Ramdas L, et al:
Combination of 5-fluorouracil and N1,N11-Diethylnorspermine
markedly activates spermidine/spermine N1-acetyltransferase
expression, depletes polyamines and synergistically induces
apoptosis in colon carcinoma cells. J Biol Chem. 280:3295–3304.
2005. View Article : Google Scholar
|
|
28.
|
Allen WL, McLean EG, Boyer J, et al: The
role of spermidine/spermine N1-acetyltransferase in determining
response to chemotherapeutic agents in colorectal cancer cells. Mol
Cancer Ther. 6:128–137. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
29.
|
Marverti G, Ligabue A, Guerrieri D, et al:
Spermidine/spermine N1-acetyltranferase modulation by novel folate
cycle inhibitors in cisplatin-sensitive and -resistant human
ovarian cancer cell lines. Gynecol Oncol. 117:202–210. 2010.
View Article : Google Scholar
|
|
30.
|
Wallace HM and Niiranen K: Polyamine
analogues - an update. Amino Acids. 33:261–265. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
31.
|
Wallace HM and Fraser AV: Inhibitors of
polyamine metabolism: review article. Amino Acids. 26:353–365.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
32.
|
Marverti G, Monti MG, Pegg AE, et al:
Spermidine/spermine N1-acetyltransferase transient over-expression
restores sensitivity of resistant human ovarian cancer cells to
N1,N12-bis(ethyl)spermine and to cisplatin. Carcinogenesis.
26:1677–1686. 2005. View Article : Google Scholar
|
|
33.
|
Andrews PA, Murphy MP and Howell SB:
Differential potentiation of alkylating and platinating agent
cytotoxicity in human ovarian carcinoma cells by glutathione
depletion. Cancer Res. 45:6250–6253. 1985.PubMed/NCBI
|
|
34.
|
Rossi T, Coppi A, Bruni E, Ruberto A,
Santachiara S and Baggio G: Effects of anti-malarial drugs on MCF-7
and Vero cell replication. Anticancer Res. 27:2555–2560.
2007.PubMed/NCBI
|
|
35.
|
Lowry OH, Rosebrough NJ, Farr AL and
Randall RJ: Protein measurement with the Folic phenol reagent. J
Biol Chem. 193:265–275. 1951.PubMed/NCBI
|
|
36.
|
Kueng W, Siber E and Eppenberger U:
Quantification of cells cultured on 96-well plates. Anal Biochem.
182:16–19. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
37.
|
van Triest B, Pinedo HM, van Hensbergen Y,
et al: Thymidylate synthase level as the main predictive parameter
for sensitivity to 5-fluorouracil, but not for folate-based
thymidilate synthase inhibitors, in 13 nonselected colon cancer
cell lines. Clin Cancer Res. 5:643–654. 1999.
|
|
38.
|
Rothenberg SP, Perwaiz Iqbal M and Da
Costa M: A simplified radioenzymatic assay for dihydrofolate
reductase using [3H]dihydrofolate. Anal Biochem.
103:152–156. 1980.PubMed/NCBI
|
|
39.
|
Bradford MM: A rapid and sensitive method
for quantification of microgram quantities of protein utilising the
principle of protein-dye binding. Anal Biochem. 72:248–254. 1976.
View Article : Google Scholar : PubMed/NCBI
|
|
40.
|
Arocho A, Chen B, Ladanyi M and Pan Q:
Validation of the 2−ΔΔCt calculation as an alternate
method of data analysis for quantitative PCR of BCR-ABL P210
transcripts. Diagn Mol Pathol. 15:56–61. 2006.
|
|
41.
|
Kansara V, Paturi D, Luo S, Gaudana R and
Mitra AK: Folic acid transport via high affinity carrier-mediated
system in human retinoblastoma cells. Int J Pharm. 355:210–219.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
42.
|
Casero RA Jr, Gabrielson EW and Pegg AE:
Immunohistochemical staining of spermidine/spermine
N1-acetyltransferase super-induced in response to treatment with
antitumour polyamine analogues. Cancer Res. 54:3955–3958. 1994.
|
|
43.
|
Chou TC and Talalay P: Quantitative
analysis of dose-effect relationships: the combined effects of
multiple drugs or enzyme inhibitors. Advances in Enzyme Regulation.
Weber G: Pergamon Press; New York, NY: pp. 27–55. 1984, View Article : Google Scholar : PubMed/NCBI
|
|
44.
|
Marverti G, Ligabue A, Paglietti G, et al:
Collateral sensitivity to novel thymidylate synthase inhibitors
correlates with folate cycle enzymes impairment in
cisplatin-resistant human ovarian cancer cells. Eur J Pharmacol.
615:17–26. 2009. View Article : Google Scholar
|
|
45.
|
Chu E and Allegra CJ: The role of
thymidylate synthase as an RNA binding protein. Bioessays.
18:191–198. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
46.
|
Marverti G, Piccinini G, Ghiaroni S,
Barbieri D, Quaglino D and Moruzzi MS: N1,N12-bis(ethyl)spermine
effect on growth of
cis-diamminedichloroplatinum(II)-sensitive and -resistant
human ovarian carcinoma cell lines. Int J Cancer. 78:33–40.
1998.PubMed/NCBI
|
|
47.
|
Orfila L, Rodrıguez M, Colman T, Hasegawa
M, Merentes E and Arvelo F: Structural modification of berberine
alkaloids in relation to cytotoxic activity in vitro. J
Ethnopharmacol. 71:449–456. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
48.
|
Gong GQ, Zong ZX and Song YM:
Spectrofluorometric determination of DNA and RNA with berberine.
Spectrochimica Acta A Mol Biomol Spectrosc. 55:1903–1907. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
49.
|
Debnath D, Suresh Kumar G, Nandi R and
Maiti M: Interaction of berberine chloride with deoxyribonucleic
acids: evidence for base and sequence specificity. Indian J Biochem
Biophys. 26:201–208. 1989.PubMed/NCBI
|
|
50.
|
Bhadra K, Maiti M and Suresh Kumar G:
Berberine-DNA complexation: new insights into the cooperative
binding and energetic aspects. Biochim Biophys Acta.
1780:1054–1061. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
51.
|
Tan W, Li Y, Chen M and Wang Y: Berberine
hydrochloride: anticancer activity and nanoparticulate delivery
system. Int J Nanomedicine. 6:1773–1777. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
52.
|
Maiti M and Kumar GS: Polymorphic nucleic
acid binding of bioactive isoquinoline alkaloids and their role in
cancer. J Nucleic Acids. 2010:1–23. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
53.
|
Bhadra K and Kumar GS: Therapeutic
potential of nucleic acid-binding isoquinoline alkaloids: binding
aspects and implications for drug design. Med Res Rev. 31:821–862.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
54.
|
Ferraroni M, Bazzicalupi C, Bilia AR and
Gratteri P: X-ray diffraction analyses of the natural isoquinoline
alkaloids Berberine and Sanguinarine complexed with double helix
DNA d(CGTACG). Chem Commun. 47:4917–4919. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
55.
|
Bhowmik D, Hossain M, Buzzetti F, D’Auria
R, Lombardi P and Kumar GS: Biophysical studies on the effect of
the 13 position substitution of the anticancer alkaloid berberine
on its DNA binding. J Phys Chem B. 116:2314–2324. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
56.
|
Cho J and Rando RR: Specific binding of
Hoechst 33258 to site 1 thymidylate synthase mRNA. Nucleic Acids
Res. 28:2158–2163. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
57.
|
Mazzini S, Bellucci MC and Mondelli R:
Mode of binding of the cytotoxic alkaloid berberine with the double
helix oligo-nucleotide d(AAGAATTCTT)2. Bioorg Med Chem.
11:505–514. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
58.
|
Hiraku Y, Oikawa S and Kawanishi S:
Distamycin A, a minor groove binder, changes enediyne-induced DNA
cleavage sites and enhances apoptosis. Nucleic Acids Res (Suppl).
2:95–96. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
59.
|
Basu S, Feuerstein BG, Dennis FD, Lubich
WP, Bergeron RJ, Samejima K and Marton LJ: Correlation between the
effects of polyamine analogues on DNA conformation and cell growth.
Cancer Res. 49:5591–5597. 1989.PubMed/NCBI
|
|
60.
|
Agostinelli E, Arancia G, Dalla Vedova L,
Belli F, Marra M, Salvi M and Toninello A: The biological functions
of polyamine oxidation products by amine oxidases: perspectives of
clinical applications. Amino Acids. 27:347–358. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
61.
|
Sun D, Wollin A and Stephen AM: Moderate
folate deficiency influences polyamine synthesis in rats. J Nutr.
132:2632–2637. 2002.PubMed/NCBI
|