Introduction
Breast cancer is a leading cause of cancer-related
deaths in women worldwide (1). In
2005, microRNA (miRNA) deregulation was first described in breast
cancer (2). Subsequently, a
growing number of studies reported altered expression of miRNAs was
connected with breast cancer (3).
miRNAs are a class of small conserved non-coding RNAs that play a
significant role in many physiological and pathological processes,
including cell proliferation, angiogenesis, metastasis, and
epigenetic changes (4–8). miRNAs can bind the 3′-UTRs of target
mRNAs and thereby post-transcriptionally regulate gene expression
via translational inhibition or target mRNA degradation (9–13).
In a variety of tumor types, miRNA expression was different between
tumor and normal tissues, as well as between metastatic and
non-metastatic tumors (14,15).
Based on miRNA manipulations, the development of new therapies is
urgently needed.
miR-365 is located on 16p13.12. A previous study
confirmed that miR-365 was downregulated in colon cancer tissues,
was involved in tumor progression and poor survival, and regulated
cancer cell behavior by targeting cyclin D1 and Bcl-2 (16). miR-365 was also overexpressed both
in cells and cutaneous squamous cell carcinoma tissues, suggesting
that miR-365 may act as an oncogene both in vitro and in
vivo (17). Moreover, recent
studies indicated that miR-365 was upregulated in breast cancer
(18). However, its function
remains unclear.
In this study, we investigated miR-365 expression
levels in breast cancer tissues, and evaluated its effect on cell
growth, cell invasion, and cell cycle in breast cancer cells.
Furthermore, we verified the target relationship between miR-365
and ADAMTS-1.
Materials and methods
Breast cancer specimens
Breast cancer specimens and adjacent normal breast
tissues (64 cases) were obtained from the Tumor Hospital Affiliated
to Zhengzhou University from July 2013 to September 2014. All
samples were immediately snap-frozen in liquid nitrogen, and no
patient had received chemotherapy or radiotherapy before surgery.
Clinical data from these samples are listed in Table I. The Ethics Committee of Zhengzhou
University approved this study, and samples were obtained with
informed consent from all patients.
 | Table IClinicopathological characteristics
and miR-365 and ADAMTS-1 expression levels in 64 breast cancer
patients. |
Table I
Clinicopathological characteristics
and miR-365 and ADAMTS-1 expression levels in 64 breast cancer
patients.
| | miR-365 expression
(2−ΔΔCt) | ADAMTS-1 mRNA
expression (2−ΔΔCt) |
|---|
| |
|
|
|---|
| Clinicopathological
factor | n | Mean ± SD | P-value | Mean ± SD | P-value |
|---|
| Age (years) |
| ≤60 | 40 | 2.801±1.031 | 0.373 | 0.463±0.363 | 0.159 |
| >60 | 24 | 2.571±0.920 | | 0.637±0.615 | |
| Lymph node
metastasis |
| Positive | 30 | 2.899±0.909 | 0.161 | 0.476±0.367 | 0.414 |
| Negative | 34 | 2.551±0.966 | | 0.575±0.558 | |
| Tumor size
(cm) |
| ≤4 | 42 | 2.819±1.059 | 0.242 | 0.501±0.497 | 0.532 |
| >4 | 22 | 2.513±0.827 | | 0.581±0.441 | |
| TNM stage |
| I | 5 | 3.936±0.587 | 0.001 | 0.232±0.065 | 0.000 |
| II | 28 | 2.941±0.825 | | 0.294±0.152 | |
| III | 24 | 2.431±0.936 | | 0.699±0.507 | |
| IV | 7 | 1.905±0.932 | | 1.091±0.559 | |
| Estrogen
receptor |
| Positive | 30 | 2.797±0.986 | 0.535 | 0.454±0.385 | 0.247 |
| Negative | 34 | 2.641±1.001 | | 0.593±0.542 | |
| Progesterone
receptor |
| Positive | 28 | 2.786±1.021 | 0.611 | 0.477±0.338 | 0.455 |
| Negative | 36 | 2.658±0.975 | | 0.568±0.563 | |
Cell culture
MDA-MB-231 and MCF-7 breast cancer cell lines were
purchased from American Type Culture Collection (ATCC). Cells were
cultured in DMEM (Gibco, USA) supplemented with 10% fetal bovine
serum (FBS, Gibco), penicillin (100 U/ml), and streptomycin (100
μg/ml). Cells were maintained at 37°C in a humidified chamber with
5% CO2.
RNA extraction and qRT-PCR
Total RNA was isolated from tissues and cells using
TRIzol reagent (Invitrogen, USA) according to the manufacturer’s
instructions. For quantitative detection of miR-365, qRT-PCR
analyses were performed using the Two Step Stemaim-it miR qRT-PCR
Quantitation kit (Novland, China). We quantified U6 small nuclear
RNA (U6 snRNA) as an endogenous control to normalize miRNA levels.
SYBR Premix Ex Taq™ (Takara, Japan) was used to quantify ADAMTS-1
mRNA expression, and GADPH served as an endogenous control. Each
sample was performed in triplicate on the ABI 7500 Fast
thermocycler. Relative expression levels were analyzed using the
2−ΔΔCt or 2−ΔCt method.
miRNA transfection
The miR-365 mimics, miR-365 inhibitor, and negative
controls (NC) were synthesized by GenePharma (Shanghai, China), and
were transfected into MDA-MB-231 and MCF-7 cells (at 100 nM) using
Lipofectamine™ 2000 (Invitrogen) according to the manufacturer’s
instructions. We assessed transfection efficiencies using qRT-PCR
at 24 h post-transfection.
Cell growth assay
Twenty-four hours post-transfection, MDA-MB-231 and
MCF-7 cells were seeded into 96-well plates (4,000 cells/well).
Cell proliferation was assessed at 0, 24, 48, 72, and 96 h as
follows: 10 μl Cell Counting Kit-8 (CCK-8) solution (Dojindo,
Japan) was added to each well, and the cells were incubated for an
additional 3 h at 37°C. The absorbance value of OD450
was measured daily and used to estimate the number of viable cells.
Each experiment was repeated three times.
Cell cycle assay
We determined cell cycle distribution using flow
cytometer analyses. Forty-eight hours after transfection, cells
from each group were harvested by trypsinization, washed with cold
PBS, and fixed overnight at 4°C in 70% ethanol. Next, cells were
digested for 2–3 h at 37°C using RNase (0.1 g/l). After propidium
iodide (PI) staining in the dark for 30 min at 4°C, cells were
analyzed using a FACScan® flow cytometer (BD
Biosciences, USA).
Transwell assay
Transfected MDA-MB-231 cells (0.2 ml,
2×105 cells/ml) were seeded in the upper chamber of the
transwell (Costar, USA). The lower chamber was filled with DMEM
plus 10% FBS. Each group was performed in triplicate. Cells were
incubated at 37°C in a humidified atmosphere containing 5%
CO2 for 24 h. Following incubation, non-invading cells
were removed from the upper surface of the membrane and the
Matrigel. Cells on the bottom surface of the membrane were fixed
with 4% paraformaldehyde for 10 min and stained with 0.1% crystal
violet for 30 min at 37°C. The number of invasive cells in three
random fields was counted using an inverted microscope (x200
magnification).
Dual-luciferase assay
The 3′-UTR of ADAMTS-1, which contains a putative
target region for miR-365, was PCR amplified from genomic DNA. The
ADAMTS-1 3′-UTR mutant construct was generated by overlap extension
PCR. Fragments were inserted between the XbaI and
XhoI sites in the pmirGLO Dual-Luciferase miRNA Target
Expression vector (Promega, USA) to generate the recombinant
vectors pmirGLO-ADAMTS-1-wt and pmirGLO-ADAMTS-1-mut. Both
insertions were verified by sequencing (Sangon, China). In
MDA-MB-231 cells, co-transfection of the reporter vectors and miRNA
(miR-365 mimics, miR-365 inhibitor or NC) was performed using
Lipofectamine 2000. Twenty-four hours after transfection,
luciferase and renilla signals were measured using the Dual
Luciferase Reporter Assay kit (Promega) according to the
manufacturer’s protocol.
Western blotting
Total proteins were extracted using RIPA buffer
according to the manufacturer’s instructions. Lysates were
centrifuged at 12,000 × g for 30 min at 4°C, and the supernatants
were obtained. Protein concentrations were determined using a BCA
Protein Assay kit (Beyotime, China). Equal amounts of total protein
(50 μg) were boiled for 5 min, separated by SDS-PAGE, and
transferred to PVDF membranes. After blocking for 1 h at room
temperature with 5% non-fat dry milk, membranes were incubated at
4°C overnight with the primary antibodies (1:900 dilution,
polyclonal rabbit anti-ADAMTS-1 and polyclonal rabbit anti-GAPDH,
Santa Cruz, USA), followed by incubation with the horseradish
peroxidase-linked secondary antibody (1:2,000 dilution, goat
anti-rabbit IgG, Santa Cruz) for 1 h at 37°C. All blots were
visualized using an enhanced chemiluminescence (ECL) system
(Amersham Pharmacia Biotech, NJ, USA).
Statistical analyses
Statistical analyses were performed using SPSS 17.0
software. All experiments were performed a minimum of three times,
and results are expressed as the mean ± standard deviation (SD).
One-way analyses of variance (ANOVA) were used to compare
differences between experimental groups. Differences were
considered statistically significant at P-values <0.05. Asterisk
in the figures indicate the statistical significantce.
Results
miR-365 is upregulated and ADAMTS-1 is
downregulated in breast cancer
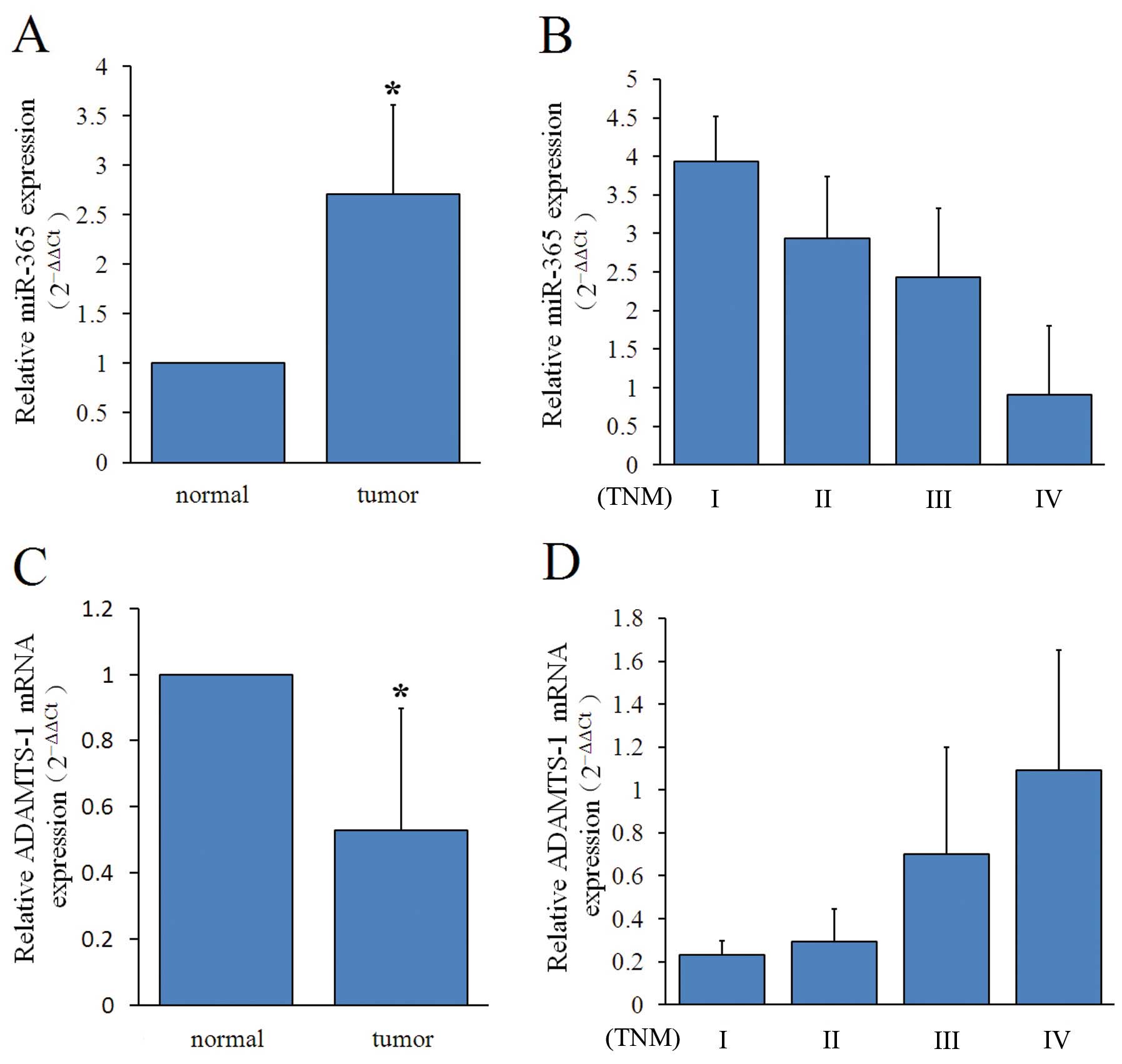
To evaluate miR-365 expression in a cohort of breast
cancer patient samples (n=64), we used qRT-PCR. We found that
miR-365 expression levels were significantly higher in breast
cancer tissues than in adjacent non-tumor tissues (P<0.05,
Table I and Fig. 1A). Statistical analyses showed that
miR-365 expression in breast cancer was associated with clinical
stage (P<0.05, Table 1 and
Fig. 1B). There were no
differences between miR-365 expression and age, tumor size, lymph
node metastasis, or estrogen receptor and progesterone receptor
positivity (P>0.05, Table
I).
Using qRT-PCR assay, we found ADAMTS-1 mRNA
expression in breast cancer varied, with the expression levels
ranging from 0.09 to 2.68. The mean ADAMTS-1 expression value was
significantly lower in breast cancer tissues than in adjacent
non-tumor tissues (P<0.05, Table
I and Fig. 1C). Increased
ADAMTS-1 expression in breast cancer was associated with clinical
stage (P<0.05, Table I and
Fig. 1D). However, we observed no
statistically differences between ADAMTS-1 expression and age,
tumor size, lymph node metastasis, or estrogen receptor and
progesterone receptor positivity (P>0.05, Table I).
miR-365 downregulation inhibits
proliferation in vitro
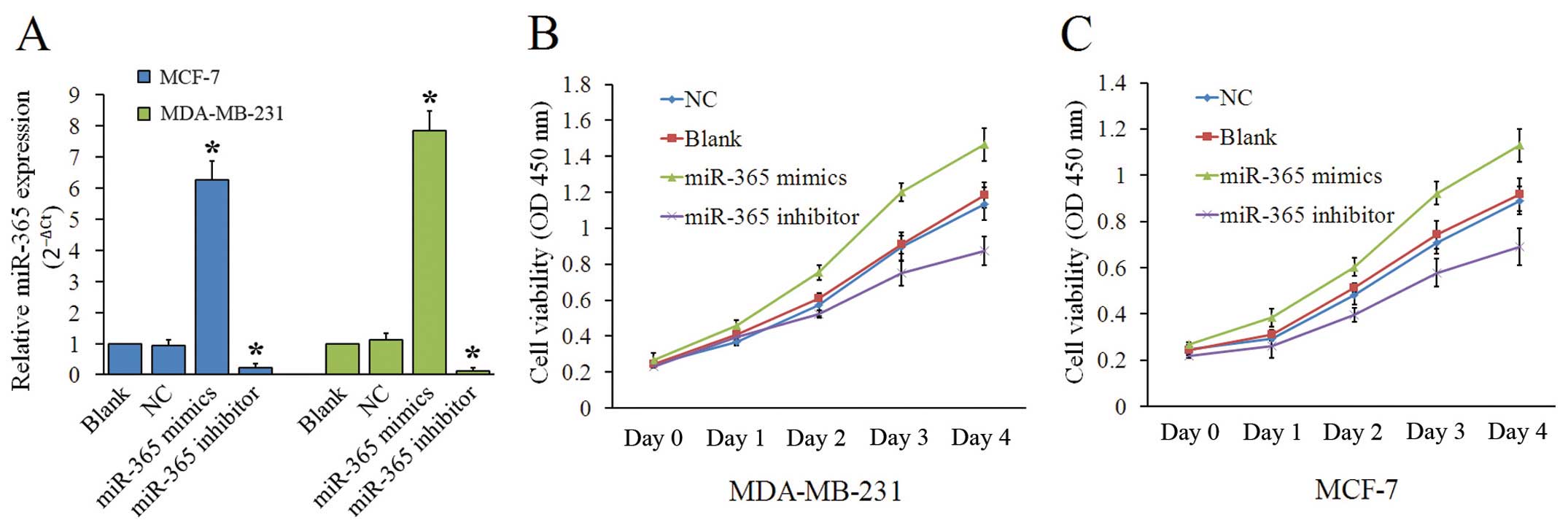
We used qRT-PCR and found that miR-365 expression in
cells transfected with miR-365 mimics was significantly higher than
the Blank group or NC group. miR-365 expression levels in cells
transfected with the miR-365 inhibitor were decreased compared to
the Blank group or NC group (P<0.05, Fig. 2A). In CCK-8 assay, we measured
OD450 values of cells transfected with NC, miR-365
mimics, and the miR-365 inhibitor. As shown in Fig. 2B and C, our data indicate that
miR-365 downregulation reduced proliferation of MDA-MB-231 and
MCF-7 cells.
The cell cycle was affected following
miR-365 downregulation in vitro
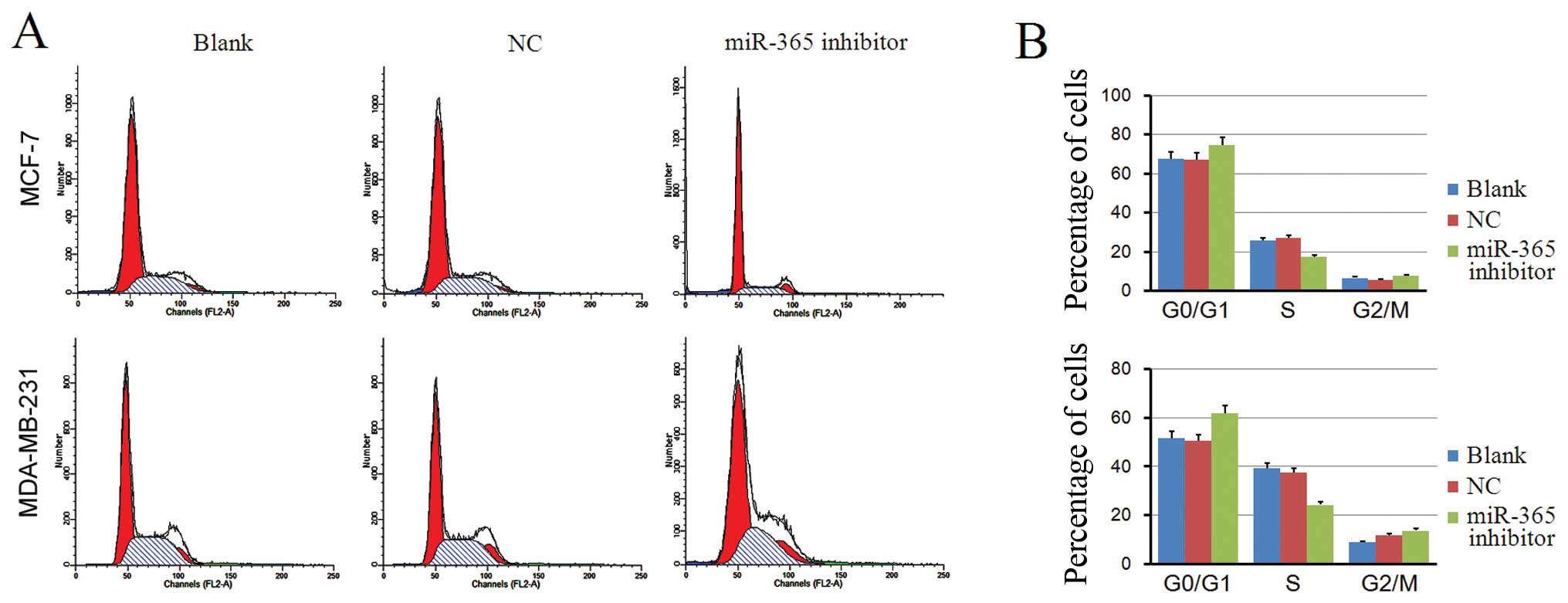
Cell cycle arrest was successfully induced 48 h
post-transfection (Fig. 3). Flow
cytometry histograms show non-transfected cells and cells
transfected with scrambled miR-365 or the miR-365 inhibitor
(Fig. 3A). Cells transfected with
miR-365 inhibitor had a significant increase in the percentage of
G0/G1 cells compared to other groups, suggesting that miR-365
downregulation induced G0/G1 cell cycle arrest (P<0.05, Fig. 3B). These results suggest that
miR-365 downregulation inhibits cell cycle progression in
MDA-MB-231 and MCF-7 cells.
miR-365 downregulation inhibits cell
invasion in vitro
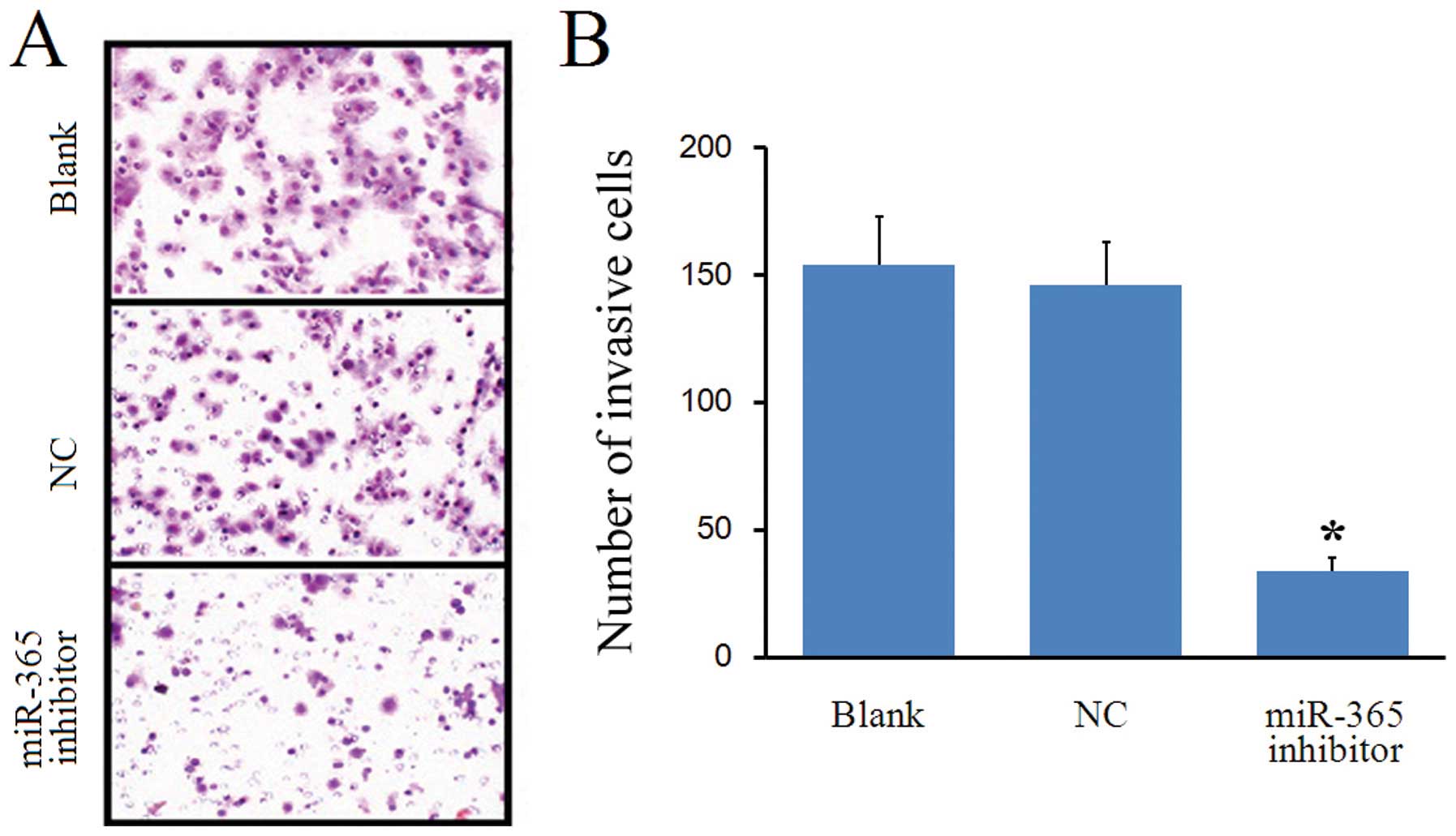
We used transwell assay to investigate the role of
miR-365 in regulating invasion. As shown in Fig. 4, significantly fewer cells invaded
the Matrigel in the miR-365 inhibitor group compared to the Blank
and NC groups (P<0.05). However, no significant differences in
the number of cells that invaded the Matrigel were observed between
the NC group and Blank group (P>0.05). These results indicate
that miR-365 downregulation decreased the invasive capacity of
MDA-MB-231 cells.
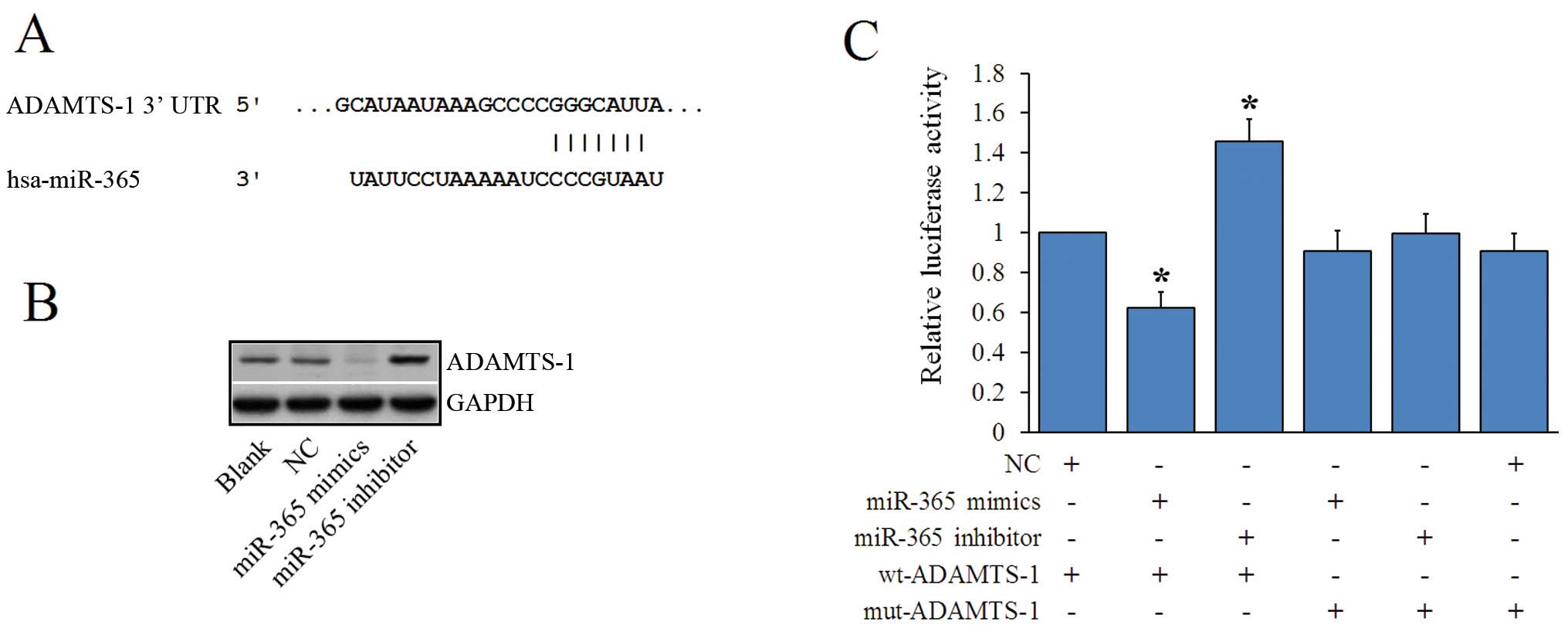
ADAMTS-1 is a direct target of
miR-365
Next, we investigated potential targets of miR-365
using prediction algorithms (TargetScan, PicTar, and miRanda).
Bioinformatics analyses predicted the ADAMTS-1 3′-UTR contained
miR-365 binding sites (Fig. 5A).
We analyzed ADAMTS-1 expression levels using western blotting and
found that ADAMTS-1 expression was significantly lower in
MDA-MB-231 cells following transfection with the miR-365 mimics.
Moreover, ADAMTS-1 expression was significantly higher in cells
transfected with the miR-365 inhibitor (Fig. 5B). To examine the target
relationship, we constructed two reporter plasmids,
pmirGLO-ADAMTS-1-wt and pmirGLO-ADAMTS-1-mut. These vectors were
transfected into MDA-MB-231 cells with NC, miR-365 mimics, or the
miR-365 inhibitor. We found that the luciferase activity was
significantly reduced when the miR-365 mimic was co-transfected
with the wt-ADAMTS-1 reporter plasmid. In contrast, the miR-365
inhibitor significantly promoted wt-ADAMTS-1 reporter plasmid
luciferase activity (Fig. 5C).
These data indicate that miR-365 regulated ADAMTS-1 expression by
directly targeting the 3′-UTR of ADAMTS-1.
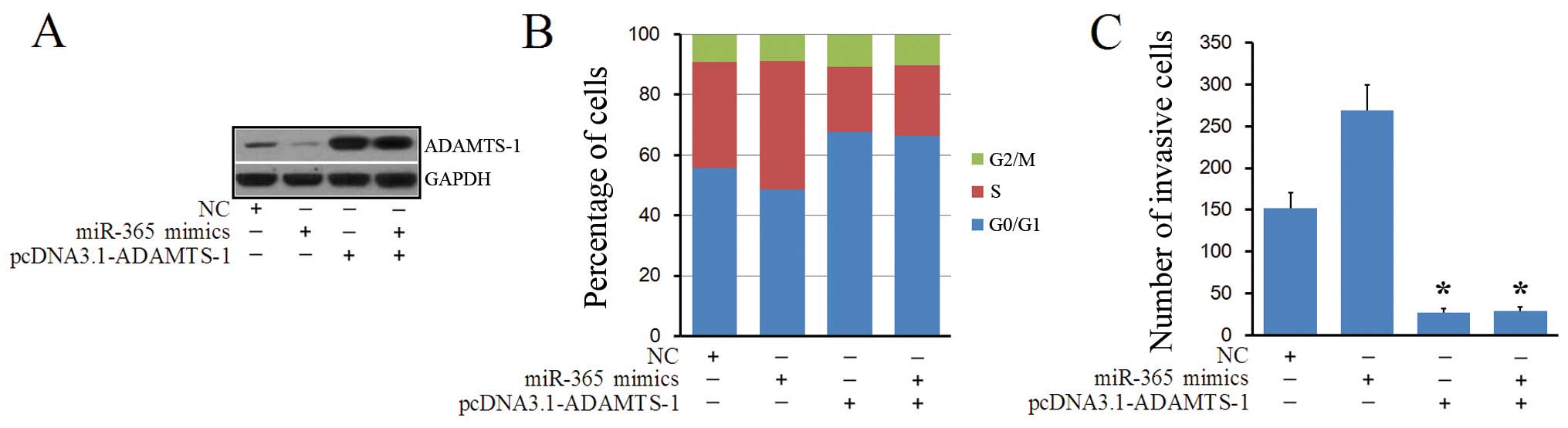
ADAMTS-1 expression restores the
pro-invasion and cell cycle function of miR-365
We constructed an expression vector
(pcDNA3.1-ADAMTS-1) that exogenously expressed ADAMTS-1 lacking the
3′-UTR to further demonstrate the target relationship. Western blot
analyses showed that co-transfection of miR-365 mimics and
pcDNA3.1-ADAMTS-1 increased ADAMTS-1 expression as well as restored
the function of the miR-365 mimics in regulating ADAMTS-1
expression (Fig. 6A). We also
analyzed MDA-MB-231 cells co-transfected with miR-365 mimics and
pcDNA3.1-ADAMTS-1 using flow cytometry and found that the
population of cells in the G0/G1 phase increased, and the number of
cells in the S phase decreased (Fig.
6B). Transwell assay revealed that the invasion accelerating
effect of miR-365 was reversed by transfection with
pcDNA3.1-ADAMTS-1 (Fig. 6C). Taken
together, these findings further suggested that miR-365 regulated
ADAMTS-1 expression through binding to its 3′-UTR.
Discussion
An increasing number of studies have revealed that
dysregulation of specific miRNAs, which function as tumor
suppressor or oncogenes, control breast cancer formation and
progression and are found in tumor biopsies and body fluids
(19–21). mir-15a has recently been found to
participate in breast cancer progression. In vitro, miR-15a
plays a role in potentiating apoptosis, suppressing proliferation,
and mediating cell cycle arrest (22). It was recently found that miR-30c
is dysregulated in breast cancer and its elevated expression
correlated with poor patient survival. Moreover, miR-30c promotes
the invasive phenotype of MDA-MB-231 cells (23). It has been reported that miR-10b
upregulation is associated with brain metastases of breast cancer,
suggesting that miR-10b can serve as a therapeutic target for
anti-metastasis therapy (24).
MiR-148a and miR-133a have been identified as biomarkers for breast
cancer detection using a three-stage microRNA analysis (25). Furthermore, recent research found
that miRNA-342-3p regulated sensitivity to the chemotherapy drugs
paclitaxel and cisplatin in a breast cancer cell line (26). Based on microarray analysis data,
miR-365 is highly expressed (18).
However, the effect of miR-365 on cell proliferation, cell cycle,
and cell invasion in breast cancer remained unknown. Here, we
investigated the mechanisms of the oncogene miR-365, and performed
luciferase reporter, western blotting, and restore assays to
identify whether ADAMTS-1 is a direct target of miR-365.
In this study, we found that miR-365 expression is
significantly higher in breast cancer tissues than normal adjacent
tissues. In addition, elevated miR-365 expression was associated
with clinical stage. These results suggest that upregulation of
miR-365 may be related to breast cancer development. Moreover, we
found that miR-365 suppression significantly restricted cell
proliferation and cell invasion, as well as induced G0/G1 cell
cycle arrest in vitro. These data indicate that miR-365 may
function as an oncogene to promote cell proliferation by mediating
G0/G1 cell cycle arrest.
ADAMTS-1 is an active multifunctional
metalloproteinase that participates in a variety of carcinomas
(27–30). However, the role of this enzyme in
cancer development remains controversial. In 1999, Kuno et
al showed that ADAMTS-1 is associated with the extracellular
matrix, which may play a role in the inflammatory process (31). When overexpressed,
catalytically-active ADAMTS-1 increases tumor growth rates and
recruits fibroblastic cells, thus inducing a stromal reaction in
vivo (32). In addition,
increased ADAMTS-1 expression has been reported in pancreatic
tumors with metastatic phenotypes (33). Low ADAMTS-1 expression was observed
in non-small-cell lung cancer, and its dysregulation correlated
with the development and progression of lung carcinoma (30). Recent studies revealed that
ADAMTS-1 expression was decreased in human breast cancers, and
ADAMTS-1 knockdown accelerated cell migration, invasion, and
formation of invadopodia (34).
Consistent with previous reports, we found ADAMTS-1 was
downregulated in breast cancer. Based on our bioinformatics search
of several online databases, we determined that ADAMTS-1 mRNA
contained miR-365 binding sites. We found that miR-365 inhibited
ADAMTS-1 expression in breast cancer cells. Using luciferase
reporter and restore assays, we further confirmed that ADAMTS-1 is
directly regulated by miR-365. Therefore, we propose that miR-365
functions by regulating ADAMTS-1 expression.
In conclusion, our study suggests that upregulated
miR-365 negatively regulates ADAMTS-1 expression by targeting its
3′-UTR. We also found that miR-365 contributes to regulation of
cell proliferation, invasion, and the cell cycle in breast cancer
cells. Therefore, inhibition of miR-365 might be a potential
therapeutic strategy for breast cancer.
Acknowledgements
The authors are grateful to all the staff at the
study centre who contributed to this study. This study was
supported by the National Natural Science Foundation of China
(81272188; 81301726).
References
|
1
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Iorio MV, Ferracin M, Liu CG, Veronese A,
Spizzo R, Sabbioni S, Magri E, Pedriali M, Fabbri M, Campiglio M,
et al: MicroRNA gene expression deregulation in human breast
cancer. Cancer Res. 65:7065–7070. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
O’Day E and Lal A: MicroRNAs and their
target gene networks in breast cancer. Breast Cancer Res.
12:2012010. View
Article : Google Scholar
|
|
4
|
Lee RC, Feinbaum RL and Ambros V: The C.
elegans heterochronic gene lin-4 encodes small RNAs with antisense
complementarity to lin-14. Cell. 75:843–854. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Brennecke J and Cohen SM: Towards a
complete description of the microRNA complement of animal genomes.
Genome Biol. 4:2282003. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Plummer PN, Freeman R, Taft RJ, Vider J,
Sax M, Umer BA, Gao D, Johns C, Mattick JS, Wilton SD, et al:
MicroRNAs regulate tumor angiogenesis modulated by endothelial
progenitor cells. Cancer Res. 73:341–352. 2013. View Article : Google Scholar
|
|
7
|
Li Q, Zhu F and Chen P: miR-7 and miR-218
epigenetically control tumor suppressor genes RASSF1A and Claudin-6
by targeting HoxB3 in breast cancer. Biochem Biophys Res Commun.
424:28–33. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Markou A, Yousef GM, Stathopoulos E,
Georgoulias V and Lianidou E: Prognostic significance of
metastasis-related microRNAs in early breast cancer patients with a
long follow-up. Clin Chem. 60:197–205. 2014. View Article : Google Scholar
|
|
9
|
Bartel DP: MicroRNAs: Genomics,
biogenesis, mechanism, and function. Cell. 116:281–297. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Meister G, Landthaler M, Dorsett Y and
Tuschl T: Sequence-specific inhibition of microRNA- and
siRNA-induced RNA silencing. RNA. 10:544–550. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Krol J, Loedige I and Filipowicz W: The
widespread regulation of microRNA biogenesis, function and decay.
Nat Rev Genet. 11:597–610. 2010.PubMed/NCBI
|
|
12
|
Bartel DP: MicroRNAs: Target recognition
and regulatory functions. Cell. 136:215–233. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Fabian MR, Sonenberg N and Filipowicz W:
Regulation of mRNA translation and stability by microRNAs. Annu Rev
Biochem. 79:351–379. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sachdeva M and Mo YY: MicroRNA-145
suppresses cell invasion and metastasis by directly targeting mucin
1. Cancer Res. 70:378–387. 2010. View Article : Google Scholar :
|
|
15
|
Ma L, Teruya-Feldstein J and Weinberg RA:
Tumour invasion and metastasis initiated by microRNA-10b in breast
cancer. Nature. 449:682–688. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Nie J, Liu L, Zheng W, Chen L, Wu X, Xu Y,
Du X and Han W: microRNA-365, down-regulated in colon cancer,
inhibits cell cycle progression and promotes apoptosis of colon
cancer cells by probably targeting Cyclin D1 and Bcl-2.
Carcinogenesis. 33:220–225. 2012. View Article : Google Scholar
|
|
17
|
Zhou M, Liu W, Ma S, Cao H, Peng X, Guo L,
Zhou X, Zheng L, Guo L, Wan M, et al: A novel onco-miR-365 induces
cutaneous squamous cell carcinoma. Carcinogenesis. 34:1653–1659.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yan LX, Huang XF, Shao Q, Huang MY, Deng
L, Wu QL, Zeng YX and Shao JY: MicroRNA miR-21 overexpression in
human breast cancer is associated with advanced clinical stage,
lymph node metastasis and patient poor prognosis. RNA.
14:2348–2360. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Shah NR and Chen H: MicroRNAs in
pathogenesis of breast cancer: Implications in diagnosis and
treatment. World J Clin Oncol. 5:48–60. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Blondal T, Jensby Nielsen S, Baker A,
Andreasen D, Mouritzen P, Wrang Teilum M and Dahlsveen IK:
Assessing sample and miRNA profile quality in serum and plasma or
other biofluids. Methods. 59:S1–S6. 2013. View Article : Google Scholar
|
|
21
|
Jiang Q, Wang Y, Hao Y, Juan L, Teng M,
Zhang X, Li M, Wang G and Liu Y: miR2Disease: A manually curated
database for microRNA deregulation in human disease. Nucleic Acids
Res. 37:Database. D98–D104. 2009. View Article : Google Scholar :
|
|
22
|
Li P, Xie XB, Chen Q, Pang GL, Luo W, Tu
JC, Zheng F, Liu SM, Han L, Zhang JK, et al: MiRNA-15a mediates
cell cycle arrest and potentiates apoptosis in breast cancer cells
by targeting synuclein-γ. Asian Pac J Cancer Prev. 15:6949–6954.
2014. View Article : Google Scholar
|
|
23
|
Dobson JR, Taipaleenmäki H, Hu YJ, Hong D,
van Wijnen AJ, Stein JL, Stein GS, Lian JB and Pratap J:
hsa-miR-30c promotes the invasive phenotype of metastatic breast
cancer cells by targeting NOV/CCN3. Cancer Cell Int. 14:732014.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ahmad A, Sethi S, Chen W, Ali-Fehmi R,
Mittal S and Sarkar FH: Up-regulation of microRNA-10b is associated
with the development of breast cancer brain metastasis. Am J Transl
Res. 6:384–390. 2014.PubMed/NCBI
|
|
25
|
Shen J, Hu Q, Schrauder M, Yan L, Wang D,
Medico L, Guo Y, Yao S, Zhu Q, Liu B, et al: Circulating miR-148b
and miR-133a as biomarkers for breast cancer detection. Oncotarget.
5:5284–5294. 2014.PubMed/NCBI
|
|
26
|
Ma T, Zhang J, Wu J and Tang J: Effect of
miR-342-3p on chemotherapy sensitivity in triple-negative breast
cancer. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 39:488–495. 2014.(In
Chinese). PubMed/NCBI
|
|
27
|
Llamazares M, Obaya AJ, Moncada-Pazos A,
Heljasvaara R, Espada J, López-Otín C and Cal S: The ADAMTS12
metalloproteinase exhibits anti-tumorigenic properties through
modulation of the Ras-dependent ERK signalling pathway. J Cell Sci.
120:3544–3552. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Porter S, Scott SD, Sassoon EM, Williams
MR, Jones JL, Girling AC, Ball RY and Edwards DR: Dysregulated
expression of adamalysin-thrombospondin genes in human breast
carcinoma. Clin Cancer Res. 10:2429–2440. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Porter S, Span PN, Sweep FC, Tjan-Heijnen
VC, Pennington CJ, Pedersen TX, Johnsen M, Lund LR, Rømer J and
Edwards DR: ADAMTS8 and ADAMTS15 expression predicts survival in
human breast carcinoma. Int J Cancer. 118:1241–1247. 2006.
View Article : Google Scholar
|
|
30
|
Rocks N, Paulissen G, Quesada Calvo F,
Polette M, Gueders M, Munaut C, Foidart JM, Noel A, Birembaut P and
Cataldo D: Expression of a disintegrin and metalloprotease (ADAM
and ADAMTS) enzymes in human non-small-cell lung carcinomas
(NSCLC). Br J Cancer. 94:724–730. 2006.PubMed/NCBI
|
|
31
|
Kuno K, Terashima Y and Matsushima K:
ADAMTS-1 is an active metalloproteinase associated with the
extracellular matrix. J Biol Chem. 274:18821–18826. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Rocks N, Paulissen G, Quesada-Calvo F,
Munaut C, Gonzalez ML, Gueders M, Hacha J, Gilles C, Foidart JM,
Noel A, et al: ADAMTS-1 metalloproteinase promotes tumor
development through the induction of a stromal reaction in vivo.
Cancer Res. 68:9541–9550. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Masui T, Hosotani R, Tsuji S, Miyamoto Y,
Yasuda S, Ida J, Nakajima S, Kawaguchi M, Kobayashi H, Koizumi M,
et al: Expression of METH-1 and METH-2 in pancreatic cancer. Clin
Cancer Res. 7:3437–3443. 2001.PubMed/NCBI
|
|
34
|
Freitas VM, do Amaral JB, Silva TA, Santos
ES, Mangone FR, Pinheiro JJ, Jaeger RG, Nagai MA and
Machado-Santelli GM: Decreased expression of ADAMTS-1 in human
breast tumors stimulates migration and invasion. Mol Cancer.
12:22013. View Article : Google Scholar : PubMed/NCBI
|