Introduction
Several polyphenolic compounds are known as cancer
chemopreventive agents (1,2). Flavonoids are a class of natural
polyphenolic compounds, ubiquitously occurring and widely consumed
secondary metabolites of plants and have profound pharmacological
properties (3–7). They are reported to have antiviral
(8), antiparasitic (9) and anticancer (10–12)
activities. Flavonoids suppress cancer cell proliferation (13), arrest cell cycle progression
(14), and induce apoptosis
(15). Luteolin
(3′,4′,5,7-tetrahydroxyflavone) is an important member of the
>4,000 different flavonoids family and is present in various
fruits and vegetables. Luteolin is reported to have
anti-inflammatory and/or anti-allergic activities (6,16),
antibacterial (17) and
antineoplastic activities (18).
It is reported that luteolin of artichoke leaf extract has
antioxidant activity on reactive oxygen species in human leucocytes
(19) and β-glucosidase-dependent
liberation of luteolin from artichoke extracts inhibits hepatic
cholesterol biosynthesis (20).
Luteolin also inhibits the growth of a variety of cancer cells
including esophageal squamous carcinoma cells (21) and pancreatic (22), gastric (23) and prostate (24) cancer.
How luteolin exhibits its antitumor effects is not
fully understood, but various mechanisms including its ability to
bind and suppress topoisomerases I and II (25), inhibit cytochrome P450 (CYP) 1
family enzymes (26) and protein
kinase C (27), regulate cell
cycles (28), induce proapoptotic
Fas (29), and increase Bax/Bcl-xL
ratio (30) have been implicated.
However, detailed mechanism of luteolin-induced apoptosis and
chemosensitization in hepatoma cell lines as cancer chemopreventive
agents has been infrequently studied up to date. Hepatocellular
carcinoma (HCC) is the fifth most common cancer with the highest
incidence of adult malignancy evident in areas in which hepatitis B
virus is endemic (31) and is the
second commonest fatal cancer in Southeast Asia. Most patients
usually present in the advanced stage when operation is no longer
feasible.
Luteolin (3′,4′,5,7-tetrahydoxyflavone), isolated
from Ixeris sonchifolia Hance, has been reported to possess
an antiproliferative effect via G1 cell cycle arrest on HepG2 human
HCC cells (32). We also
previously reported that luteolin down-regulated expression of
cyclin-dependent kinase (CDK) 4 and upregulated p53 and CDK
inhibitor p21WAF1/CIP1, leading to growth inhibition
(32). In this study we
investigated in detail the p53 contributed apoptotic mechanism,
which luteolin-induced in HepG2 cells, we checked the
antiproliferative effect and chemopreventive machineries of
luteolin on HepG2 and Hep3B cells.
Materials and methods
Compounds
Flavonoid compounds, luteolin
(3′,4′,5,7-tetra-hydoxyflavone) and apigenin
(4′,5,7-trihydroxyflavone) were purchased from Sigma-Aldrich Co.
LLC (St. Louis, MO, USA). Each compound was dissolved in dimethyl
sulfoxide (DMSO, Sigma-Aldrich Co. LLC) to 20–200 μg/ml stock
solutions for further experiments.
Cell lines and treatment with
compounds
Human HCC cell line HepG2 and Hep3B, and human
hepatocyte-derived Chang liver cells were obtained from American
Type Culture Collection (Manassas, VA, USA). HepG2 and Hep3B were
maintained in minimum essential medium (MEM, Invitrogen Life
Technologies, Carlsbad, CA, USA) with 10% fetal bovine serum (FBS,
Invitrogen Life Technologies) and antibiotics (100 U/ml of
penicillin and 100 μg/ml streptomycin, Invitrogen Life
Technologies). Chang liver cells were maintained with RPMI-1640
(Invitrogen Life Technologies) supplemented with 10% FBS with
antibiotics in humidified atmosphere of 37°C, 5%
CO2.
MTT assay
Each cell type (0.96×104 cells/well) was
seeded in Falcon 96-well plate for MTT assay, which measures
mitochondrial activity in viable cells. This method is based on the
conversion of the
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT,
Sigma-Aldrich Co. LLC) to MTT-formazan crystals by mitochondrial
enzyme. Cells were grown overnight, and the media were replaced
with fresh media, treated with each compound at various
concentrations, and incubated for 48 h. Control groups were treated
with DMSO, equal to the highest percentage (<0.1%) of solvent
used in experimental conditions for growth inhibition and MTT
assay. After 48 h the media were replaced with serum-free media.
MTT was freshly prepared at 5 mg/ml in phosphate-buffered saline
(PBS, Sigma-Aldrich Co. LLC) and passed through a 0.2-μm pore-size
filter. An aliquot of 100 μl of MTT stock solution was added to
each well, and the plate was incubated at 37°C for 4 h in
humidified 5% CO2 incubator. After 4 h media were
removed. Ethanol-DMSO (1:1 mixture solution) (200 μl) was added per
well in order to solubilize the formazan. In addition, after 10 min
the optical density of each well was measured with a
spectrophotometer equipped with a 560-nm filter. Proliferation rate
was calculated from 4 wells using percentage of control.
Fluorescence activated cell sorting
(FACS) analysis
The treated cells were detached using trypsin/EDTA
(Invitrogen Life Technologies), washed with PBS and fixed in 75%
ethanol at 4°C for 30 min. Prior to analyses, cells were washed
again with PBS, resuspended in cold PI solution (PI in PBS, 50
μg/ml) and incubated at room temperature in the dark for 30 min.
Before analysis cell suspensions were filtered with 40-μm pore
nylon mesh for removing debris. Flow cytometry analyses were
performed on a FACScan (Becton-Dickinson, San Jose, CA, USA).
Western blot analysis
Cells were harvested and washed twice in PBS at 4°C.
Total cell lysates were lysed in lysis buffer [40 mM Tris (pH 8.0),
120 mM NaCl, 0.5% NP-40, 0.1 mM sodium orthovanadate, 2 μg/ml
aprotinin]. The supernatant was collected and protein
concentrations were then measured with protein assay reagents
(Pierce, Rockford, IL, USA). Equal amount of proteins were boiled
for 3 min and chilled on ice, subjected to 10–12.5% SDS-PAGE, and
electrophoretically transferred to a nitrocellulose membrane. The
blotting membrane was blocked with PBS/0.1% Tween-20 containing 10%
skim milk for 1 h. Antibodies specific for p21WAF1/CIP1,
p53, p27KIP1, B-cell lymphoma 2 (Bcl-2), cyclin E, CDK2,
caspase-3, Fas, Fas-ligand (FasL), c-Myc, Bcl-2 associated X
protein (Bax), poly(ADP-ribose) polymerase (PARP), and Smad 4 were
purchased from Santa Cruz Biotechnology Inc. (Dallas, TX, USA).
Antibody against c-Jun was obtained from BD Biosciences Pharmingen
(San Diego, CA, USA). Monoclonal antibody to β-actin (Sigma-Aldrich
Co. LLC) was used as an internal control. Horseradish peroxidase
(HRP)-labeled donkey anti-rabbit immunoglobulin and HRP-labeled
donkey anti-goat immunoglobulin were purchased from Santa Cruz
Biotechnology. The HRP-labeled sheep anti-mouse immunoglobulin was
from GE Healthcare Life Sciences (Piscataway, NJ, USA). The
proteins were visualized with the enhanced chemiluminescence (ECL)
detection system (GE Healthcare Life Sciences).
Reverse transcription-polymerase chain
reaction (RT-PCR) analysis
Total RNA was prepared using RNAzol (Teltest,
Finewood, TX, USA). The reverse transcriptase (RT) reaction was
carried out with SuperScript II reverse transcriptase (Life
Technologies). The RT reaction mixture containing 1 μg total RNA,
100 pmol oligo (dT)18, 0.1 mM dNTP mixture, 40 U RNasin
ribonuclease inhibitor (Progmega, Madison, WI, USA), 5×
First-strand buffer and 200 U SuperScript II reverse transcriptase
(Life Technologies). The synthesis of cDNA was performed at 42°C
for 1 h, followed by 15 min of heating at 70°C for inactivating
enzyme. A final volume of 20 μl of reaction containing 1 μl of
template cDNA from RT reaction, 20 pmol of sense and antisense
primers, 2 μl of 10× PCR buffer, 0.5 mM of dNTP mixtur, and 1 U of
AmpliTaq polymerase (PE Biosystem, Waltham, MA, USA) was carried
out on a GeneAmp PCR System 2400 (PE Biosystem). The primer
sequence is as follows (33);
transforming growth factor β1 (TGF-β1) sense
5′-GCCCTGGACACCAACTATTGCT-3′, TGF-β1 antisense
5′-AGGCTCCAAATGTAGGGGCAGG-3′. PCR reaction performed was denature
at 94°C for 40 sec, annealing at 64°C for 45 sec, and extension at
72°C for 45 sec, and 35 cycles were used for amplification. The
amplified PCR products were electrophoresed on 2.5% agarose gels
and visualized by ethidium bromide staining.
Results
Luteolin inhibits the growth of human
hepatocellular cells
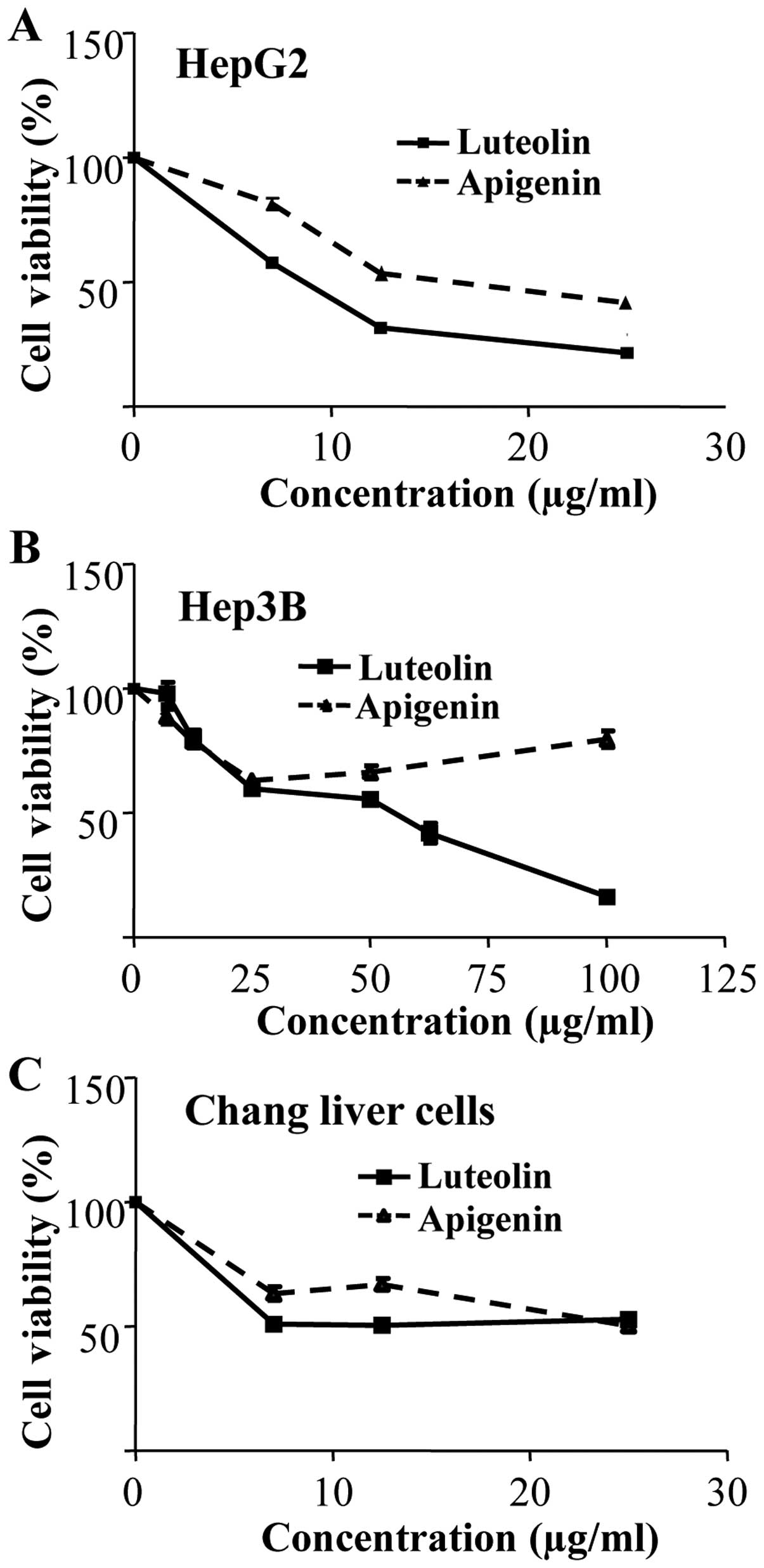
To examine the growth inhibitory potency of luteolin
on hepatocellular cells, cell proliferation was determined by MTT
assay. We also employed apigenin to examine cell proliferation
since luteolin and apigenin were identified as active components in
Ixeris sonchifolia Hance, and luteolin showed a significant
potent effect when comparing their ability on growth inhibition
(32). Luteolin was more potent
than apigenin in all the cell lines tested in the present study
(Fig. 1). In addition, HepG2 was
the most sensitive to luteolin (Fig.
1A). IC50 values of luteolin on HepG2 and Hep3B
cells were ~9 and 55 μg/ml, respectively (Fig. 1A and B). On Chang liver cells, no
significant antiproliferative effect was observed in either
luteolin or apigenin treatment (Fig.
1C).
Luteolin induces G1 phase arrest in HepG2
cells and apoptosis in Hep3B cells
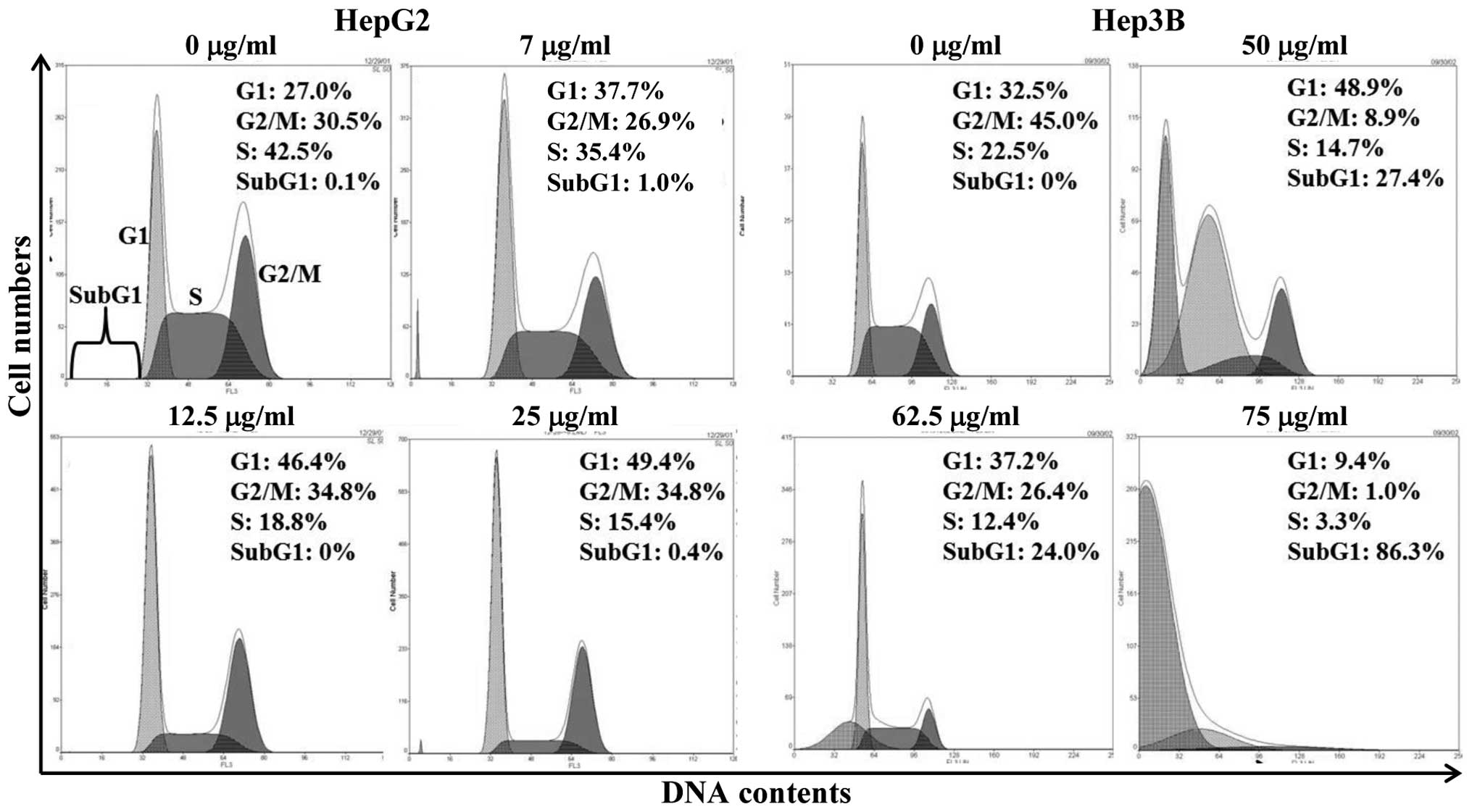
To determine whether luteolin’s growth inhibitory
effect was caused by specifically perturbing cell cycle-related
events, a set of experiment was performed to measure DNA content
and the cell cycle distribution by flow cytometry analysis after
staining with PI. Fig. 2 shows the
relative percentages of HepG2 and Hep3B cells in each phase of the
cell cycle, following a 12-h treatment with varying luteolin
concentrations. In HepG2 cells, luteolin induced the accumulation
of G1 phase of cell cycle in a concentration-dependent manner
(Fig. 2, left panel). However,
Hep3B cells showed increase of subG1 population at the same
time-point (Fig. 2, right panel).
These results suggest that the growth inhibition of HepG2 cells was
the result of a G1 phase arrest and that of Hep3B cells occurred
mainly through apoptosis.
Luteolin modulates cell cycle regulatory
proteins
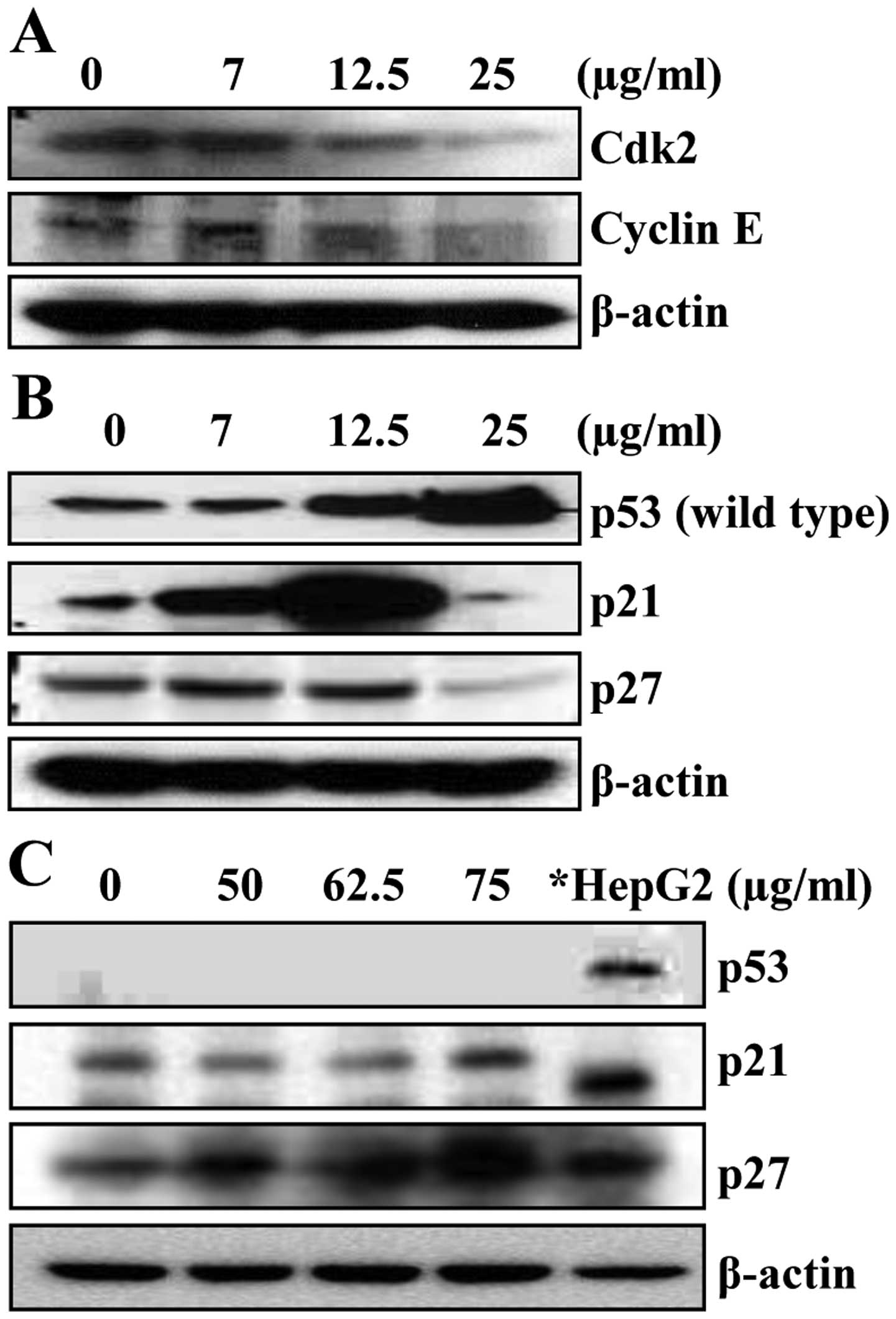
Since luteolin arrested HepG2 cells in the G1 phase
of the cell cycle, we determined the expression levels of cell
cycle regulating factors involved in G1 boundary, such as cyclin E
and Cdk2, by western blot analysis. As shown in Fig. 3A, the protein levels of cdk2 and
cyclin E were decreased in a concentration-dependent manner. These
data indicate that the growth inhibitory effect of luteolin in
HepG2 cells are caused by downregulating cdk2 and cyclin E
expression.
Luteolin induced p21WAF1/CIP1
and G1 arrest in a p53-independent manner
Because it has been reported that p53, a tumor
suppressor, regulates a DNA damage-triggered G1 checkpoint by
upregulation of CDK inhibitor p21WAF1/CIP1 (34), we examined the expression patterns
of p53 and p21WAF1/CIP1 by luteolin-treatment. As shown
in Fig. 3B, HepG2 cells treated
with luteolin increased the expression of p53 in a
concentration-dependent manner. Unlike p53, the concentrations of
luteolin ranging from 7 to 12.5 μg/ml markedly increased the
protein level of p21WAF1/CIP1; however, this induction
of p21WAF1/CIP1 was decreased at 25 μg/ml luteolin
(Fig. 3B). This result is
consistent with our previous study which was performed by using
luteolin from Ixeris sonchifolia Hance (32). Because p27KIP1, another
CDK inhibitor, is reported to arrest cells only at the G1 phase
(35,36), we next determined the effect of
luteolin on p27KIP1 expression. HepG2 cells treated with
luteolin showed similar expression pattern of p27KIP1
compared with that of p21WAF1/CIP1 although there was no
prominent increase at 12.5 μg/ml luteolin (Fig. 3B). These results suggest that other
factors besides p53 may be involved in luteolin-induced G1 arrest
as well as growth inhibition in HepG2 cells.
To further confirm our hypothesis that other
mechanisms are involved in luteolin-induced HCC cell growth
inhibition, we examined the effect of luteolin on p53,
p21WAF1/CIP1, and p27KIP1 expressions using
P53-deleted HCC Hep3B cells. As expected, no p53 expression was
observed in Hep3B cells (Fig. 3C).
However, treatment of Hep3B cells with luteolin resulted in a
concentration-dependent increase in the expression of
p21WAF1/CIP1 and p27KIP1 (Fig. 3C). These results from HepG2 and
Hep3B cells suggest that the mechanism(s) other than p53 may be
involved in the upregulation of p21WAF1/CIP1 by
luteolin.
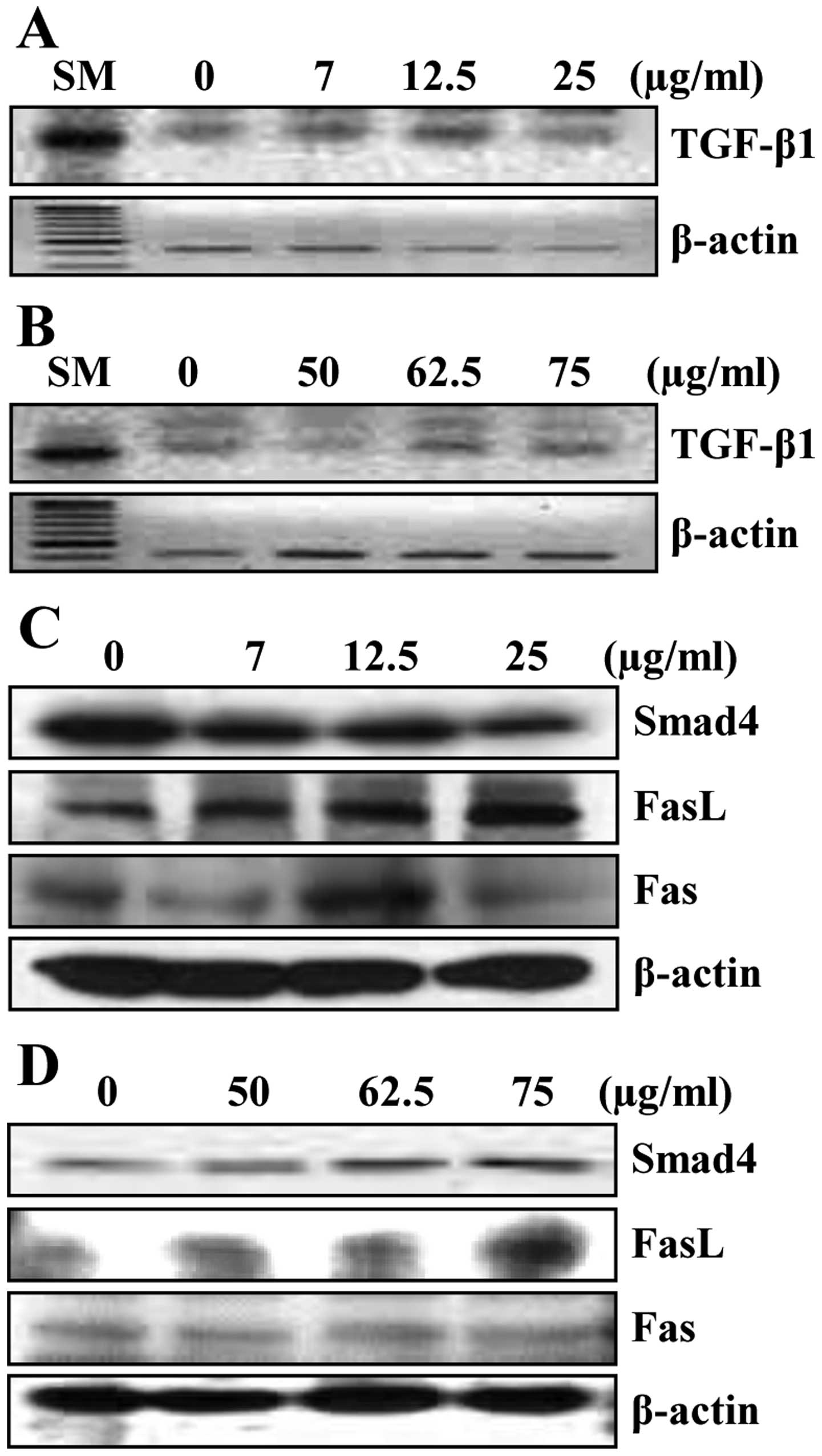
Luteolin modulated the expression of
TGF-β1 and Fas/FasL in HCC cells
Transforming growth factor β1 (TGF-β1) is an
essential regulator of cellular processes including proliferation,
differentiation, migration, cell survival and angiogenesis. TGF-β1
has been reported to exert its function via specific receptors and
intracellular Smad transcription factors. Phosphorylation of
receptor-activated Smads, such as Smad2 or Smad3, leads to
formation of complexes with the common mediator Smad (Smad4), which
are imported to the nucleus, induce cycle-dependent kinase
inhibitors, and then lead to G1 arrest (37). Therefore, TGF-β1 is known as an
upstream G1 arrest signal (34,36).
Whether luteolin affects the expression of TGF-β1 and Smad4 in HCC
cells was examined. The mRNA level of TGF-β1 was gradually
increased by luteolin treatment in HepG2 cells (Fig. 4A). Treatment with various
concentrations of luteolin also induced the mRNA level of TGF-β1 in
Hep3B cells (Fig. 4B).
We next tested the effect of luteolin on Smad4
expression in HCC cells. Results show that Smad4 expression level
in HepG2 cells slightly increased at 12.5 μg/ml and decreased at 25
μg/ml concentration of luteolin (Fig.
4C). In Hep3B cells, luteolin treatment increased Smad4
expression concentration-dependently (Fig. 4D).
Because TGF-β1 signaling is reported to activate
Fas-mediated apoptotic pathways (38), we investigated whether luteolin
affects the expression of Fas and its ligand FasL in HCC cells.
Fig. 4C shows that luteolin
upregulated FasL in a concentration-dependent manner, but Fas
increased up to 12.5 μg/ml, then decreased at 25 μg/ml in HepG2
cells. We also observed similar expression pattern of FasL in Hep3B
cells although Fas was not significantly altered by luteolin
(Fig. 4D).
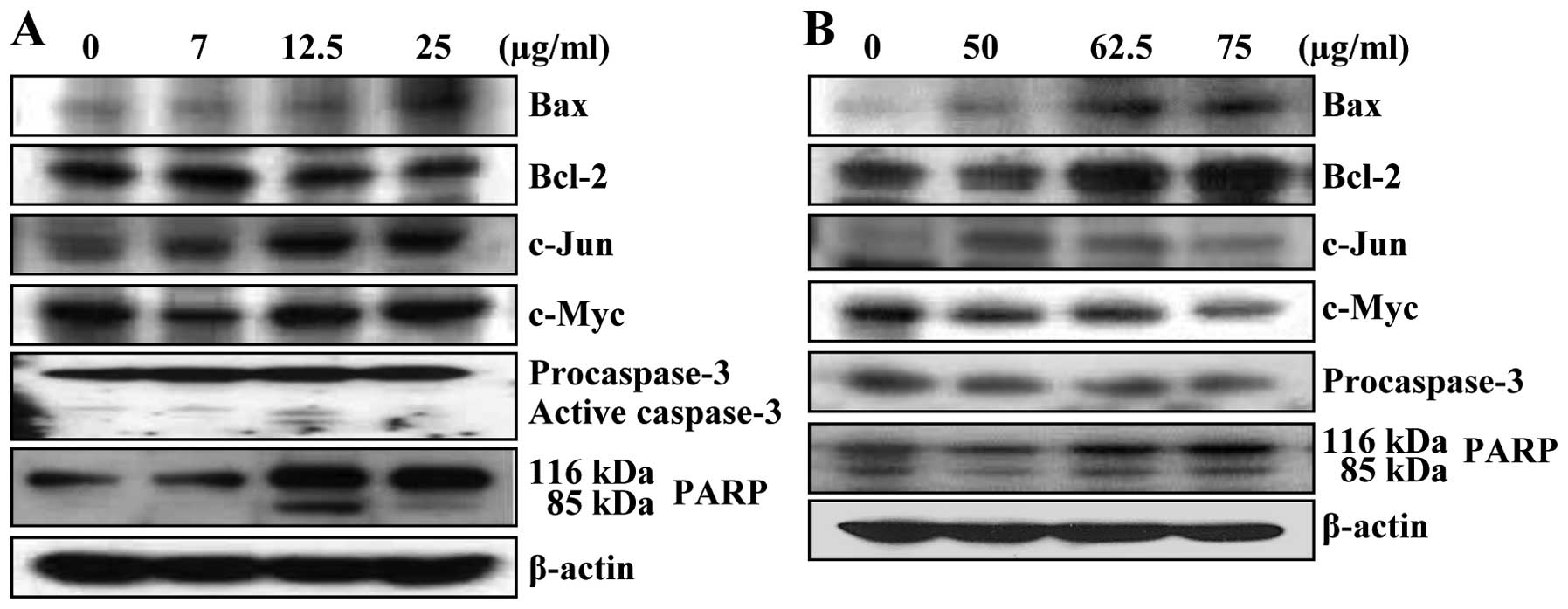
Luteolin triggered apoptosis through
proapoptotic Bax
To confirm the contribution of Fas/FasL and p53 on
luteolin-induced apoptosis, we investigated whether the expression
of apoptotic protein Bax, Bcl-2, caspase-3 and PARP, were modulated
by luteolin. Luteolin treatment increased Bax and slightly
decreased Bcl-2 expression in a concentration-dependent manner in
HepG2 cells (Fig. 5A). Luteolin
also increased Bax expression in Hep3B cells (Fig. 5B). In contrast, Bcl-2 level was
increased simultaneously in Hep3B cells by luteolin (Fig. 5B). To elucidate the mechanism of
Bcl-2 upregulation by luteolin in Hep3B cells, we determined the
effect of luteolin on oncogene expression, such as c-Jun and c-Myc.
An increase of c-Myc and c-Jun expression was observed in HepG2
cells (Fig. 5A); however, the
reduction of these proteins was found in Hep3B cells (Fig. 5B).
Finally, the pro-caspase-3 level and PARP cleavage
were measured in luteolin-treated HCC cells. In the presence of
lutein, the increase of cleavage forms of caspase-3 and PARP were
observed in HepG2 cells (Fig. 5A).
Similarly, PARP cleavage was detected in Hep3B cells in a
concentration-dependent manner and pro-form of caspase-3 decreased
slightly (Fig. 5B).
Discussion
In the present report, we investigated how luteolin
induces cell death on HCC cell line HepG2 and Hep3B cells. Luteolin
elicited G1 cell cycle arrest in HepG2 and direct apoptosis on
Hep3B cells. Interestingly in the comparative experiment between
HepG2 and Hep3B cells with various concentration of luteolin, we
found that luteolin induced cell cycle arrest on HepG2 cells by
orchestration of three signaling pathways; TGF-β, p53 and
Fas/FasL.
It has been reported that p27KIP1 arrests
cells only at the Gl phase compared to p21WAF/CIP1 which
regulates G1 and G2 phases (35,36).
In the present study, the expression of p27KIP1 on HepG2
was not remarkably changed in comparison with the vehicle-treated
control, and decreased at high concentration of 25 μg/ml.
Interestingly the level of p21WAF/CIP1 dramatically
increased up to 12.5 μg/ml and decreased at 25 μg/ml. This fact
suggests that p21WAF/CIP1 may be a key factor in the G1
cell cycle arrest of HepG2 rather than G2/M arrest around at the
IC50 and we observed increased amount of cells treated
with 12.5 μg/ml luteolin under G2/M phase for 24 h.
Smad4 has been reported to form heteromeric
complexes with Smad2 and Smad3, and these complexes are
translocated to the nucleus, bind to DNA in sequence specific
manner, and regulate gene transcription (37). These complexes induce
cycle-dependent kinase inhibitors p16INK4,
p15INK4, p27KIP1 and p21WAF1/CIP1
to increase, finally leading to G1 arrest (39,40).
In addition, it is ascertain whether TGF-β is the upstream signal
of Smad4 (41), so we investigated
mRNA expression of TGF-β1 to confirm that TGF-β1 might be
associated with this luteolin-induced G1 phase cell cycle arrest.
We also found that the expression pattern of TGF-β1 by luteolin
treatment was similar to the protein expression pattern of Smad4,
which suggest that one possible pathway of luteolin-induced G1
phase arrest may due to TGF-β1 signaling. Polyak et al
(35) reported that TGF-β1 induces
arrest of the cell cycle in G1. Our results in the present study
are relevant to this previous report and suggest that TGF-β1 plays
a role in luteolin-mediated G1 arrest via regulation of
p21WAF/CIP1 and p27KIP1.
Furthermore, our results demonstrated that p53 might
not be crucial for luteolin-induced apoptosis in HCC cells. The
results from the comparative study between HepG2 and Hep3B cells
suggested that luteolin was able to induce cell death through
p53-independent pathways. This was in agreement with previous
observations by others in esophageal squamous cell carcinoma and
prostate cancer cells (42,43).
One of the possible mechanisms for the
luteolin-induced apoptotic cell death is through Fas/FasL pathway.
In our observations, luteolin-induced Fas expression was
accompanied by p53-induced caspase-3 activation and PARP cleavage,
which are hallmarks of apoptosis. In addition, luteolin treatment
decreased Bcl-2, a negative regulator of Fas-induced apoptosis.
Bcl-2 provides a true survival advantage after many diverse
stimuli, including chemotherapeutic agents, γ-radiation and growth
factor deprivation (44). It was
reported that Bcl-2 exerts the ability to counter apoptosis
elicited by Myc under suboptimal growth conditions, and Myc has the
ability to override the retardation of cell-cycle entry by Bcl-2
(45,46). The contrary effect between Myc and
Bcl-2 to apoptosis, and the increased Bcl-2 protein did not allow
Hep3B cells to enter the cell cycle arrest. Also it is reported
that overexpressed Bcl-2 resulted in a significant rise in
p21WAF/CIP1 in endometrial carcinoma cells (47), from this viewpoint, it might be
possible that increased Bcl-2 impinges on Hep3B cells increasing
p21WAF1/CIP1 expression compared to HepG2 cells and
cannot play a role for cell survival. Our results demonstrated that
luteolin-triggered apoptosis in Hep3B cells might result from
continuous increase of TGF-β1 and Fas protein, and despite the
increase of Fas/FasL, Bcl-2 expression increased
concentration-dependently, this seems to be caused by the decrease
of myc protein.
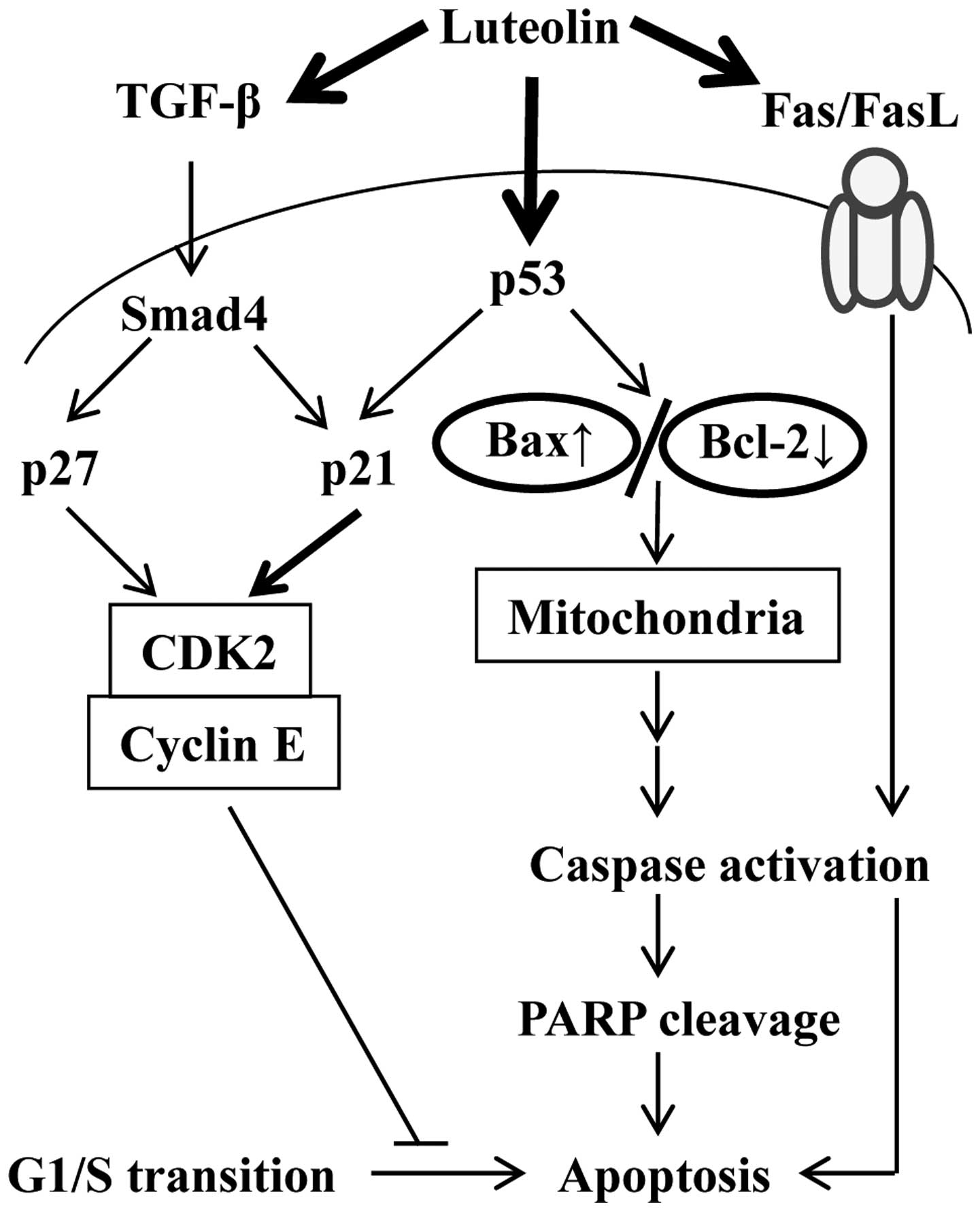
Taken together, our study demonstrates that luteolin
induced G1 phase arrest via TGF-β1, Fas/FasL, and p53 signaling
pathway on HepG2 cells (Fig. 6),
and the strength of these signals is changeable according to the
administered concentration of luteolin. Also,
p21WAF/CIP1 might be a key protein in the G1 cell cycle
arrest of HepG2 at the IC50, and the IC50
value of luteolin on HepG2 of this study correspond to our previous
report (48). On p53 deleted Hep3B
cells, luteolin elicited apoptosis directly via TGF-β1 and Fas/FasL
signaling pathways. On the basis of these results, further studies
are required in animals and in patients to explore the potential of
luteolin as an anticancer agent for liver cancer patients.
Acknowledgements
This study was supported by the National Research
Foundation of Korea (NRF) grant funded by the Korea government
(MSIP) (no. 2009-0083538).
References
|
1
|
Chen D and Dou QP: Tea polyphenols and
their roles in cancer prevention and chemotherapy. Int J Mol Sci.
9:1196–1206. 2008. View Article : Google Scholar
|
|
2
|
Berdowska I, Zieliński B, Fecka I,
Kulbacka J, Saczko J and Gamian A: Cytotoxic impact of phenolics
from Lamiaceae species on human breast cancer cells. Food Chem.
141:1313–1321. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Woodman OL and Chan EC: Vascular and
anti-oxidant actions of flavonols and flavones. Clin Exp Pharmacol
Physiol. 31:786–790. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Russo P, Del Bufalo A and Cesario A:
Flavonoids acting on DNA topoisomerases: Recent advances and future
perspectives in cancer therapy. Curr Med Chem. 19:5287–5293. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Guerra-Araiza C, Álvarez-Mejía AL,
Sánchez-Torres S, Farfan-García E, Mondragón-Lozano R,
Pinto-Almazán R and Salgado-Ceballos H: Effect of natural exogenous
antioxidants on aging and on neurodegenerative diseases. Free Radic
Res. 47:451–462. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kritas SK, Saggini A, Varvara G, Murmura
G, Caraffa A, Antinolfi P, Toniato E, Pantalone A, Neri G, Frydas
S, et al: Luteolin inhibits mast cell-mediated allergic
inflammation. J Biol Regul Homeost Agents. 27:955–959. 2013.
|
|
7
|
Sak K: Cytotoxicity of dietary flavonoids
on different human cancer types. Pharmacogn Rev. 8:122–146. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kaul TN, Middleton E Jr and Ogra PL:
Antiviral effect of flavonoids on human viruses. J Med Virol.
15:71–79. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kayser O, Kiderlen AF and Croft SL:
Natural products as anti-parasitic drugs. Parasitol Res. 90(Suppl
2): S55–S62. 2003. View Article : Google Scholar
|
|
10
|
Lin YS, Tsai PH, Kandaswami CC, Cheng CH,
Ke FC, Lee PP, Hwang JJ and Lee MT: Effects of dietary flavonoids,
luteolin, and quercetin on the reversal of epithelial-mesenchymal
transition in A431 epidermal cancer cells. Cancer Sci.
102:1829–1839. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
López-Lázaro M: Flavonoids as anticancer
agents: Structure-activity relationship study. Curr Med Chem
Anticancer Agents. 2:691–714. 2002. View Article : Google Scholar
|
|
12
|
Chahar MK, Sharma N, Dobhal MP and Joshi
YC: Flavonoids: A versatile source of anticancer drugs. Pharmacogn
Rev. 5:1–12. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kawaii S, Tomono Y, Katase E, Ogawa K and
Yano M: Antiproliferative activity of flavonoids on several cancer
cell lines. Biosci Biotechnol Biochem. 63:896–899. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Singh RP and Agarwal R: Natural flavonoids
targeting deregulated cell cycle progression in cancer cells. Curr
Drug Targets. 7:345–354. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ramos S: Effects of dietary flavonoids on
apoptotic pathways related to cancer chemoprevention. J Nutr
Biochem. 18:427–442. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Pandurangan AK, Kumar SA, Dharmalingam P
and Ganapasam S: Luteolin, a bioflavonoid inhibits
azoxymethane-induced colon carcinogenesis: Involvement of iNOS and
COX-2. Pharmacogn Mag. 10(Suppl 2): S306–S310. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Tofighi Z, Molazem M, Doostdar B, Taban P,
Shahverdi AR, Samadi N and Yassa N: Antimicrobial activities of
three medicinal plants and investigation of flavonoids of
tripleurospermum disciforme. Iran J Pharm Res. 14:225–231.
2015.PubMed/NCBI
|
|
18
|
Li YC, Hung CF, Yeh FT, Lin JP and Chung
JG: Luteolin-inhibited
arylamineN-acetyltransferaseactivityandDNA-2-aminofluorene adduct
in human and mouse leukemia cells. Food Chem Toxicol. 39:641–647.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Pérez-García F, Adzet T and Cañigueral S:
Activity of artichoke leaf extract on reactive oxygen species in
human leukocytes. Free Radic Res. 33:661–665. 2000. View Article : Google Scholar
|
|
20
|
Gebhardt R: Inhibition of cholesterol
biosynthesis in HepG2 cells by artichoke extracts is reinforced by
glucosidase pretreatment. Phytother Res. 16:368–372. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wang TT, Wang SK, Huang GL and Sun GJ:
Luteolin induced-growth inhibition and apoptosis of human
esophageal squamous carcinoma cell line Eca109 cells in vitro.
Asian Pac J Cancer Prev. 13:5455–5461. 2012. View Article : Google Scholar
|
|
22
|
Cai X, Lu W, Ye T, Lu M, Wang J, Huo J,
Qian S, Wang X and Cao P: The molecular mechanism of
luteolin-induced apoptosis is potentially related to inhibition of
angiogenesis in human pancreatic carcinoma cells. Oncol Rep.
28:1353–1361. 2012.PubMed/NCBI
|
|
23
|
Lu J, Li G, He K, Jiang W, Xu C, Li Z,
Wang H, Wang W, Wang H, Teng X, et al: Luteolin exerts a marked
antitumor effect in cMet-overexpressing patient-derived tumor
xenograft models of gastric cancer. J Transl Med. 13:422015.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Chiu FL and Lin JK: Downregulation of
androgen receptor expression by luteolin causes inhibition of cell
proliferation and induction of apoptosis in human prostate cancer
cells and xenografts. Prostate. 68:61–71. 2008. View Article : Google Scholar
|
|
25
|
Chowdhury AR, Sharma S, Mandal S, Goswami
A, Mukhopadhyay S and Majumder HK: Luteolin, an emerging
anti-cancer flavonoid, poisons eukaryotic DNA topoisomerase I.
Biochem J. 366:653–661. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Buening MK, Chang RL, Huang MT, Fortner
JG, Wood AW and Conney AH: Activation and inhibition of
benzo(a)pyrene and aflatoxin B1 metabolism in human liver
microsomes by naturally occurring flavonoids. Cancer Res. 41:67–72.
1981.PubMed/NCBI
|
|
27
|
Ferriola PC, Cody V and Middleton E Jr:
Protein kinase C inhibition by plant flavonoids. Kinetic mechanisms
and structure-activity relationships. Biochem Pharmacol.
38:1617–1624. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ko WG, Kang TH, Lee SJ, Kim YC and Lee BH:
Effects of luteolin on the inhibition of proliferation and
induction of apoptosis in human myeloid leukaemia cells. Phytother
Res. 16:295–298. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
29
|
Selvendiran K, Koga H, Ueno T, Yoshida T,
Maeyama M, Torimura T, Yano H, Kojiro M and Sata M: Luteolin
promotes degradation in signal transducer and activator of
transcription 3 in human hepatoma cells: An implication for the
antitumor potential of flavonoids. Cancer Res. 66:4826–4834. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Chang J, Hsu Y, Kuo P, Kuo Y, Chiang L and
Lin C: Increase of Bax/ Bcl-XL ratio and arrest of cell cycle by
luteolin in immortalized human hepatoma cell line. Life Sci.
76:1883–1893. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Villanueva A, García C, Paules AB, Vicente
M, Megías M, Reyes G, de Villalonga P, Agell N, Lluís F, Bachs O,
et al: Disruption of the antiproliferative TGF-beta signaling
pathways in human pancreatic cancer cells. Oncogene. 17:1969–1978.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Yee SB, Lee JH, Chung HY, Im KS, Bae SJ,
Choi JS and Kim ND: Inhibitory effects of luteolin isolated from
Ixeris sonchifolia Hance on the proliferation of HepG2 human
hepatocellular carcinoma cells. Arch Pharm Res. 26:151–156. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Derynck R, Jarrett JA, Chen EY, Eaton DH,
Bell JR, Assoian RK, Roberts AB, Sporn MB and Goeddel DV: Human
transforming growth factor-beta complementary DNA sequence and
expression in normal and transformed cells. Nature. 316:701–705.
1985. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Hartwell LH and Kastan MB: Cell cycle
control and cancer. Science. 266:1821–1828. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Polyak K, Kato JY, Solomon MJ, Sherr CJ,
Massague J, Roberts JM and Koff A: p27Kip1, a cyclin-Cdk
inhibitor, links transforming growth factor-beta and contact
inhibition to cell cycle arrest. Genes Dev. 8:9–22. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Toyoshima H and Hunter T: p27, a novel
inhibitor of G1 cyclin-Cdk protein kinase activity, is related to
p21. Cell. 78:67–74. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Massagué J: TGF-beta signal transduction.
Annu Rev Biochem. 67:753–791. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Kim SG, Jong HS, Kim TY, Lee JW, Kim NK,
Hong SH and Bang YJ: Transforming growth factor-beta 1 induces
apoptosis through Fas ligand-independent activation of the Fas
death pathway in human gastric SNU-620 carcinoma cells. Mol Biol
Cell. 15:420–434. 2004. View Article : Google Scholar :
|
|
39
|
Depoortere F, Pirson I, Bartek J, Dumont
JE and Roger PP: Transforming growth factor beta(1) selectively
inhibits the cyclic AMP-dependent proliferation of primary thyroid
epithelial cells by preventing the association of cyclin D3-cdk4
with nuclear p27(kip1). Mol Biol Cell. 11:1061–1076. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Park BJ, Park JI, Byun DS, Park JH and Chi
SG: Mitogenic conversion of transforming growth factor-beta1 effect
by oncogenic Ha-Ras-induced activation of the mitogen-activated
protein kinase signaling pathway in human prostate cancer. Cancer
Res. 60:3031–3038. 2000.PubMed/NCBI
|
|
41
|
Nakao A, Imamura T, Souchelnytskyi S,
Kawabata M, Ishisaki A, Oeda E, Tamaki K, Hanai J, Heldin CH,
Miyazono K, et al: TGF-beta receptor-mediated signalling through
Smad2, Smad3 and Smad4. EMBO J. 16:5353–5362. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Zhang Q, Zhao XH and Wang ZJ: Cytotoxicity
of flavones and flavonols to a human esophageal squamous cell
carcinoma cell line (KYSE-510) by induction of G2/M arrest and
apoptosis. Toxicol In Vitro. 23:797–807. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Kobayashi T, Nakata T and Kuzumaki T:
Effect of flavonoids on cell cycle progression in prostate cancer
cells. Cancer Lett. 176:17–23. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Walker A, Taylor ST, Hickman JA and Dive
C: Germinal center-derived signals act with Bcl-2 to decrease
apoptosis and increase clonogenicity of drug-treated human B
lymphoma cells. Cancer Res. 57:1939–1945. 1997.PubMed/NCBI
|
|
45
|
Cory S, Vaux DL, Strasser A, Harris AW and
Adams JM: Insights from Bcl-2 and Myc: Malignancy involves
abrogation of apoptosis as well as sustained proliferation. Cancer
Res. 59(Suppl): S1685–S1692. 1999.
|
|
46
|
Green DR and Evan GI: A matter of life and
death. Cancer Cell. 1:19–30. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Crescenzi E and Palumbo G: Bcl-2 exerts a
pRb-mediated cell cycle inhibitory function in HEC1B endometrial
carcinoma cells. Gynecol Oncol. 81:184–192. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Attoub S, Hassan AH, Vanhoecke B, Iratni
R, Takahashi T, Gaben AM, Bracke M, Awad S, John A, Kamalboor HA,
et al: Inhibition of cell survival, invasion, tumor growth and
histone deacetylase activity by the dietary flavonoid luteolin in
human epithelioid cancer cells. Eur J Pharmacol. 651:18–25. 2011.
View Article : Google Scholar
|