Introduction
Lipomas are benign tumors composed of mature fat
cells (1). It is the most common
soft tissue tumor in adults with peak incidence between 40–60
years. They may appear at any site but with a broad distinction
between subcutaneous (superficial) and deep-seated lesions.
Although most lipomas are easily diagnosed, those occurring deep
down (e.g., intramuscular lipoma, perineural lipoma) may be
confused with liposarcomas. Though mitoses are rarely seen in
histologic sections of lipomas, karyotypes can easily be obtained
from short-term cultured tumor cells. In 1986, the first
characteristic acquired chromosome aberration of lipomas was
described, the translocation t(3;12)(q27~28;q13~15) (2,3).
Since then, according to the Mitelman Database of Chromosome
Aberrations and Gene Fusions in Cancer (http://cgap.nci.nih.gov/Chromosomes/Mitelman
database updated on February 13, 2015), 476 lipomas with chromosome
aberrations have been reported, with involvement of chromosome
bands 12q13~15 being seen in more than 300 of them. Recombination
may take place with a wide variety of partners, but the by far most
common is the translocation t(3;12)(q27~28;q14~15) which has been
reported in 53 of the aberrant karyotypes registered in the
Mitelman Database. Other recurrently involved chromosome segments
found recombined with 12q13~15 in lipomas are 1p36 and 1p32~34
(each found in 29 cases), 2p22~24 (in 6 cases), 2q35~37 (10 cases),
5q33 (16 cases), 9p21~22 (7 cases) and 12p11~13 and 13q12~14 (8
cases each). The chromosome rearrangements often target the
HMGA2 gene in 12q14.3 with breakpoints occurring both within
and outside the locus; the essential outcome appears to be
deregulation of HMGA2 with truncation of the gene as the
critical event (4 and refs.
therein). The most frequent translocation, t(3;12)(q27~28;q13~15),
generates an HMGA2-LPP fusion gene coding for a
transcription factor containing the AT-hook domain of HMGA2
and C-terminal LIM domains of LPP (5,6).
HMGA2 has also been reported to form fusion genes with
PPAP2 (at 1p32), ACKR3 (at 2q37), EBF1 (at
5q33), NFIB (at 9p22) and LHFP (at 13q12) (7–11).
In the present study, we describe a novel recurrent chromosome
translocation, t(12;18)(q14~15;q12~21) and its molecular
consequences in 12 benign fat cell tumors.
Materials and methods
Ethics statement
The study was approved by the regional ethics
committee (Regional komité for medisinsk forskning-setikk Sør-Øst,
Norge, http://helseforskning.etikkom.no) and written informed
consent was obtained from the patients.
Patients
Table I shows the
patients' gender, age, diagnosis and the location of the tumor. All
tumors were surgically removed.
 | Table IClinical, cytogenetic and molecular
data on the 12 benign fat cell tumors. |
Table I
Clinical, cytogenetic and molecular
data on the 12 benign fat cell tumors.
| Case no. | Gender/ age
(years) | Diagnosis | Location | Karyotype | Cq, Ex1-Ex2 of
HMGA2 | Cq, Ex4-Ex5 of
HMGA2 | Cq of
ACTB | HMGA2
fusion |
|---|
| 1 | M/58 | Lipoma | Intramuscular, left
thigh |
46,XY,t(12;18)(q14~q15;q12~q21)[12]/46,XY[3] | 30.97 | 28.23 | 23.41 | Not done |
| 2 | F/44 | Lipoma | Intramuscular,
right elbow |
46,XX,t(12;18)(q14~q15;q12~q21)[16] | 29.76 | 27.58 | 25.27 | Not done |
| 3 | M/54 | Lipoma | Intramuscular,
right deltoid |
46,XY,t(12;18)(q14~q15;q12~q21)[12]/46,XY[3] | 28.58 | 36.84 | 24.33 |
HMGA2-sequence from 18q12.3 |
| 4 | F/34 | Lipoma | Intramuscular,
right deltoid |
46,XX,t(12;18)(q14~q15;q12~q21)[14] | 30.10 | 39.69 | 24.33 |
HMGA2-SETBP1 |
| 5 | M/38 | Lipoma | Left thoracic
wall |
46,XY,t(12;18)(q14~q15;q12~q21)[15] | 28.73 | 38.84 | 22.45 |
HMGA2-GRIP1 |
| 6 | F/28 | Lipoma | Intramuscular, left
thigh |
46,XX,t(12;18)(q14~q15;q12~q21)[14]/46,XX[1] | 32.57 | 30 | 22.96 | Not done |
| 7 | F/61 | Lipoma | Intramuscular,
right splenius capitis muscle |
46,XX,t(12;18)(q14~q15;q12~q21)[12]/46,XX[3] | 26.28 | 39.36 | 23.67 |
HMGA2-SETBP1 |
| 8 | M/55 |
Osteochon-drolipoma | Intramuscular,
subscapularis muscle |
46,XY,t(12;18)(q14~q15;q12~q21)[15] | 30.04 | 35.64 | 24.97 |
HMGA2-SETBP1 |
| 9 | M/55 | Lipoma | Intramuscular,
right infraspinatus muscle |
46,XY,t(2;18;12)(q37;q12~q21;q14~15)[9] | Not done | Not done | Not done | Not done |
| 10 | M/15 | Lipoma | Foot, right
intrametatarsal |
46,XY,t(12;18)(q14~q15;q12~q21)[15] | Not done | Not done | Not done | Not done |
| 11 | M/64 | Lipoma | Right groin |
46,XY,t(8;9)(p21;q22),t(12;18)(q14~q15;q12~q21)[10] | Not done | Not done | Not done | Not done |
| 12 | M/56 | Lipoma | Intramuscular, left
deltoid |
46,XY,t(12;18)(q14~q15;q12~q21)[5]/46,XY[5] | Not done | Not done | Not done | Not done |
Chromosome banding analysis
Samples from the operation specimens were
mechanically and enzymatically disaggregated and short-term
cultured as described elsewhere (12). The cultures were harvested and the
chromosomes G-banded using Wright stain. The subsequent cytogenetic
analysis and karyotype description followed the recommendations of
the ISCN (13).
Total RNA isolation and cDNA
synthesis
Tumor tissue adjacent to that used for cytogenetic
analysis and histologic examination had been frozen and stored at
−80°C from eight tumors (cases 1–8). Total RNA was extracted using
miRNeasy kit, TissueLyser II homogenizer and Qiacube according to
the manufacturer's recommendations (Qiagen Nordic, Stockholm,
Sweden). For cDNA synthesis, 400–500 ng of total RNA were
reverse-transcribed in a 20 μl reaction volume using iScript
Advanced cDNA Synthesis kit for RT-qPCR according to the
manufacturer's instructions (Bio-Rad Laboratories, Oslo, Norway).
cDNA equivalent to 10 ng/μl of total RNA was used as template in
subsequent real-time PCR assays. The Human Universal Reference
Total RNA was used as control (Clontech Laboratories, Takara Bio
Group, Saint-Germainen-Laye, France). According to the company's
information, it is a mixture of total RNAs from a collection of
adult human tissues, chosen to represent a broad range of expressed
genes. Both male and female donors are represented.
Real-time PCR
Real-time PCR was carried out to determine the
expression level of HMGA2. The TaqMan gene expression assays
(Applied Biosystems, Foster City, CA, USA) Hs00171569_m1
(HMGA2 exons 1–2) and Hs00971725_m1 (HMGA2 exons 4–5) were
used. The ACTB gene, assay Hs99999903_m1, was used as
endogenous control. The 20 μl reaction volume contained 1× TaqMan
Universal Master Mix II with UNG, lX of the 20× TaqMan Gene
Expression Mix, and 1 μl cDNA (10 ng equivalent of RNA). Four
replicates of each sample and the endogenous control were
performed. Real-time PCR was run on a CFX96 Touch™ Real-Time PCR
Detection System (Bio-Rad Laboratories). The thermal cycling
included an initial step at 50°C for 2 min, followed by 10 min at
95°C, 40 cycles of 15 sec at 95°C and 1 min at 60°C. The data were
analyzed using Bio-Rad CFX Manager software (Bio-Rad
Laboratories).
3′-Rapid amplification cDNA ends
(3′-RACE)
For 3′-RACE, 100 ng of total RNA were
reverse-transcribed in a 20 μl reaction volume with the A3RNV-RACE
primer (5′-ATC GTT GAG ACT CGT ACC AGC AGA GTC ACG AGA GAG ACT ACA
CGG TAC TGG TTT TTT TTT TTT TTT-3′) using iScript Select cDNA
Synthesis kit according to the manufacturer's instructions (Bio-Rad
Laboratories). One microliter was used as template and amplified
using the outer primer combination HMGA2-846F1 (5′-CCA CTT CAG CCC
AGG GAC AAC CT-3′) and A3R-1New (5′-TCG TTG AGA CTC GTA CCA GCA GAG
TCA C-3′). One microliter of the amplified products was used as
template in nested PCR with the primers HMGA2-982F1 (5′-CAA GAG TCC
CTC TAA AGC AGC TCA-3′) and A3R-3 (5′-CGA GAG AGA CTA CAC GGT ACT
GGT-3′). For both PCRs, the 25 μl reaction volume contained 12.5 μl
of Premix Taq (Takara Bio), template, and 0.4 μM of each of the
forward and reverse primers. PCR cycling consisted of an initial
step of denaturation at 94°C for 30 sec followed by 35 cycles of 7
sec at 98°C, 30 sec at 55°C, 90 sec at 72°C, and a final extension
for 5 min at 72°C.
A total of 3 μl of the PCR products were stained
with GelRed (Biotium, Hayward, CA, USA), analyzed by
electrophoresis through 1.0% agarose gel and photographed. The rest
of the amplified fragments were purified using the Thermo
Scientific GeneJET PCR purification kit (Thermo Fisher Scientific,
Oslo, Norway) and direct sequencing was performed using the light
run sequencing service of GATC Biotech (http://www.gatc-biotech.com/en/sanger-services/lightrun-sequencing.html).
The BLAST (http://blast.ncbi.nlm.nih.gov/Blast.cgi) and BLAT
(http://genome.ucsc.edu/cgi-bin/hgBlat) programs were
used for computer analysis of sequence data.
Reverse transcriptase PCR (RT-PCR)
To verify the results obtained by 3′-RACE, i.e., the
presence of HMGA2-chimeric transcript (see below), PCRs were
performed using the forward primer HMGA2-846F1 and the reverse
primer SETBP1-5390R1 (5′-GCA GCG TGA GGT CAG GAG AGT GC-3′) for
cases 4 and 7. The primers HMGA2-846F1 and SETBP1-5325F1 (5′-GGC
GCT TCA GTA CGG CTG GAT C-3′) were used for case 8. For case 3, the
primer HMGA2-846F1 and the reverse primer 18q21-Rev1 (5′-GCA TTG
GCA GTC CCC TTG CAT T -3′) were used. For case 5, the primer
HMGA2-846F1 and the reverse primer GRIP-intrR1 (5′-TTA AGG TGT GGC
CTT TAG GCG TGA C-3′) were used. The 25 μl PCR volumes contained
12.5 μl of Premix Taq (Takara Bio), 1 μl of diluted cDNA (10 ng
equivalent of RNA), and 0.4 μM of each of the forward and reverse
primers. The PCRs were run on a C-1000 Thermal cycler (Bio-Rad
Laboratories). The PCR conditions were: an initial denaturation at
94°C for 30 sec followed by 35 cycles of 7 sec at 98°C, 120 sec at
68°C, and a final extension for 5 min at 68°C.
Results
Pathology and cytogenetics
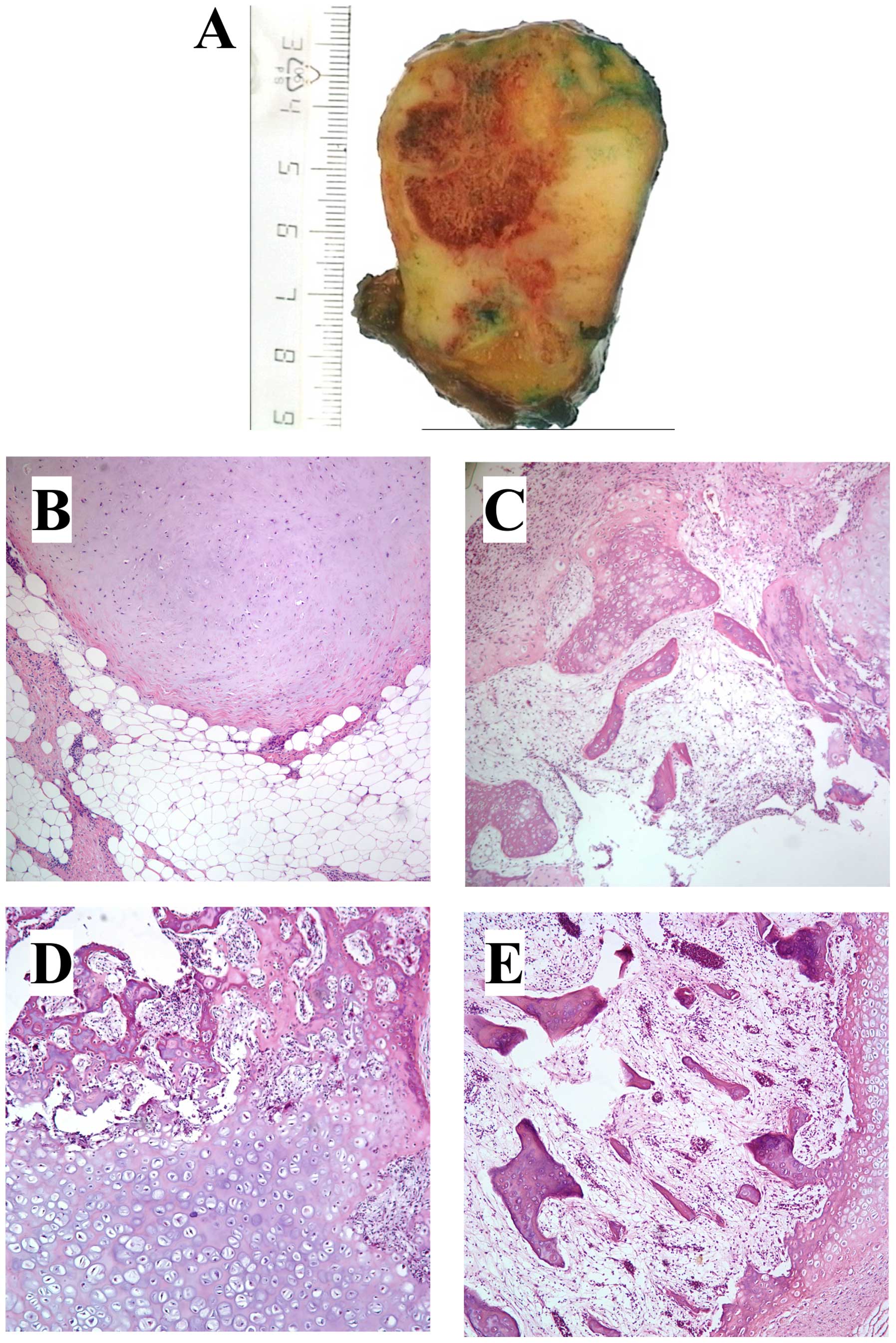
All tumors were ordinary lipomas except case 8,
which was an osteochondrolipoma (Table
I and Fig. 1), a rare variant
of lipoma with metaplastic components of cartilage and bone within
the fatty tissue. In this tumor, microscopic examination showed
lobules of mature fat separated by strands of fibrous tissue and
areas with hyaline cartilage. Other areas showed bony trabeculae
with bone marrow between them (Fig.
1). There was no cellular atypia. Reactive changes with
prominent lymphoid infiltrates in the bone marrow were seen, and in
the fatty tissue and fibrous septa there were mature lymphocytes
and some plasma cells (Fig.
1).
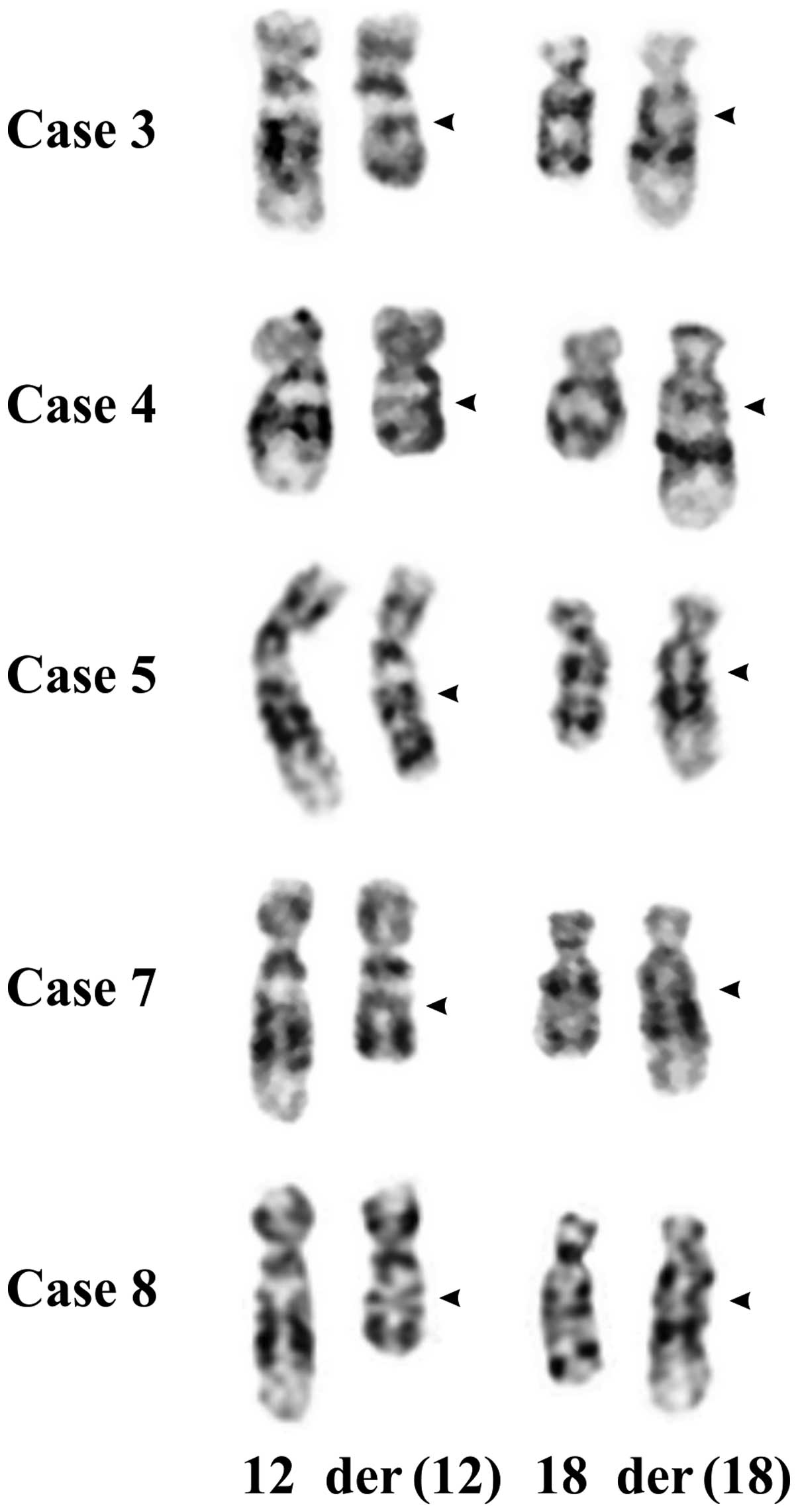
In all 12 cases, 8 males and 4 females (Table I), the tumor cells showed
cytogenetic recombination between chromosome bands 12q14~15 and
18q12~21 (this was how the cases were selected). Nine lipomas and
the osteochondrolipoma (case 8) had a reciprocal
t(12;18)(q14~15;q12~21) as the sole karyotypic aberration, one
lipoma carried a three-way translocation,
t(2;18;12)(q37;q12~21;q14~15), and another tumor had a t(8;9)
(p21;q22) in addition to t(12;18). Partial karyotypes of the
t(12;18)(q14~15;q12~21) are shown in Fig. 2.
Real-time PCR
Real-time PCR was performed with two commercially
available TaqMan assays for the HMGA2 gene, one assay for
exons 1–2 and the other for exons 4–5, in order to find out whether
HMGA2 was rearranged. Because most HMGA2
rearrangements take place in intron 3 of the gene, the result is an
overexpression of exons 1–3 of HMGA2. Thus, the mean
quantification cycle (Cq) was compared between the two assays
(Table I). Similar Cq values for
both assays indicate that both 5′-end (exons 1–2) and 3′-end (exons
4–5) are equally expressed and the HMGA2 locus is most
probably not rearranged. A difference in the Cq values between
assays for exons 1–2 and exons 4–5, with the former having a lower
Cq than the latter, indicates that 5′-end and 3′-end HMGA2
exons are unequally expressed and that HMGA2 is most
probably rearranged. In three lipomas (cases 1, 2 and 6), similar
Cq values between assays for exons 1–2 and exons 4–5 were found
(Table I). In four lipomas and in
the osteochondrolipoma (cases 3–5, 7 and 8), on the other hand, the
Cq values for exons 4–5 were significantly lower than those for
exons 1–2, indicating rearrangement of HMGA2 (Table I).
3′-RACE
3′-RACE on lipomas (cases 3–5 and 7) and the
osteochondrolipoma (case 8) (Table
I) amplified fragments which by Sanger sequence analysis were
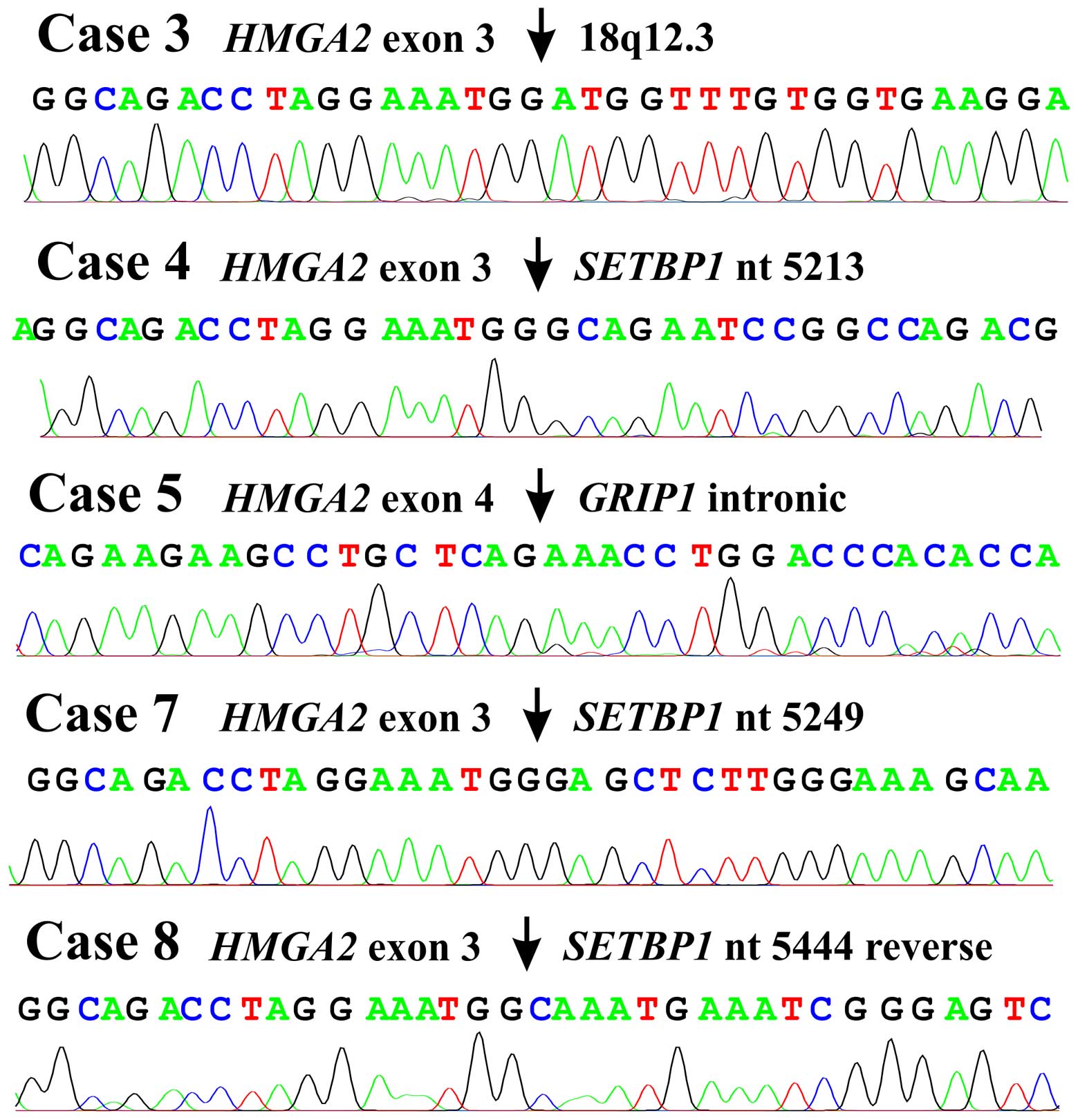
found to be chimeric HMGA2-cDNA fragments. In lipomas 3, 4
and 7 as well as the osteochondrolipoma, exon 3 of HMGA2 was
fused to sequences from 18q12.3 (Fig.
3) which in lipomas 4 and 7 and the osteochondrolipoma
consisted of the 3′-untranslated region of the SETBP1 gene.
In lipoma 3, exon 3 of HMGA2 was fused with a sequence 10
kbp downstream of the SETBP1 gene (Fig. 3). In lipoma 5, exon 4 of
HMGA2 was fused to a sequence 500 Mbp distal to HMGA2
in an intron of GRIP1 in 12q14.3 (Fig. 3).
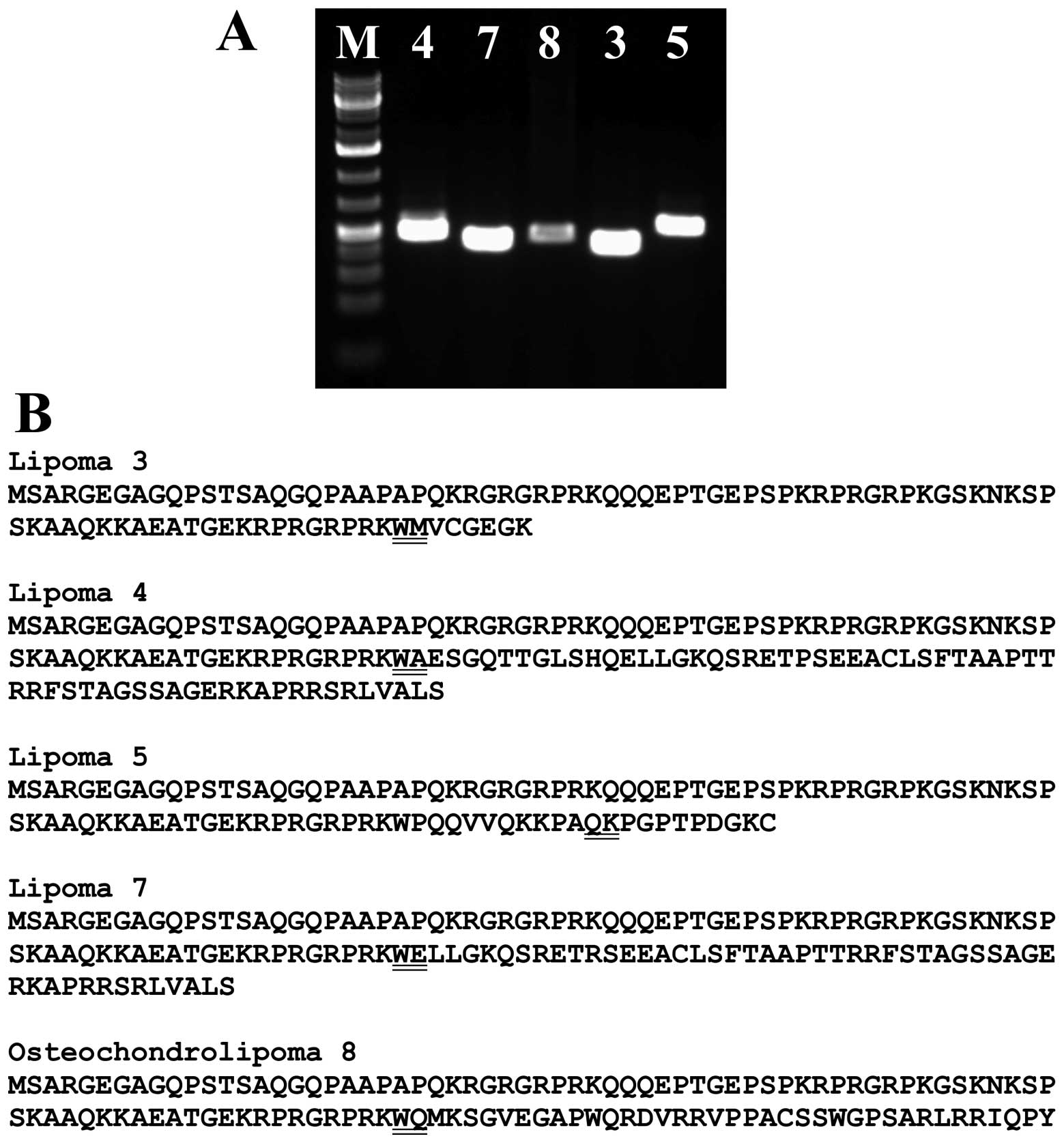
PCR with the primer HMGA2-846F1 and specific reverse
primers amplified a cDNA fragment (Fig. 4A) which by direct sequencing was
shown to display the same fusion point as the 3′-RACE amplified
fragment (Fig. 3).
Discussion
We describe here a new recurrent chromosome
translocation, t(12;18)(q14~15;q12~21), in lipomas. According to
the Mitelman Database of Chromosome Aberrations and Gene Fusions in
Cancer (http://cgap.nci.nih.gov/Chromosomes/Mitelman, Database
last updated on February 13, 2015), there are 13 cases with
aberration involving breakpoints on the long arm of chromosome 18
but none of them had t(12;18) (q14~15;q12~21). Whether some of
these cases could nevertheless be cytogenetically similar to the
tumors of our series, is a possibility we cannot test.
Because changes of chromosomal bands 12q13~15 in
lipomas are almost always associated with rearrangement and/or
activation of HMGA2 (14),
we decided to investigate further the possible involvement of
HMGA2 also in the present cases. The real-time PCR and
3′-RACE experiments showed that HMGA2 was expressed as a
chimeric HMGA2 transcript in five cases. In four lipomas,
exons 1–3 of HMGA2 were fused to a sequence of SETBP1
(cases 4, 7 and 8) or an intragenic sequence from 18q12.3 (case 3)
10 kbp distal to SETBP1. In one tumor (case 5), the
translocation t(12;18) resulted in fusion of exons 1–4 of
HMGA2 with an intronic sequence of GRIP1 which also
maps to chromosome band 12q14.3. The ensuing HMGA2 fusion
transcripts code for putative proteins which contain amino acid
residues 1–83 of HMGA2 (accession number NP_003474.1) corresponding
to exons 1–3 of the gene and amino acid residues from the fused
sequences (cases 3, 4, 7 and 8) or, in the tumor of case 5, 94
amino acid residues are translated from exons 1–4 of HMGA2
and joined with residues from a fused sequence from chromosome 12
(Fig. 4B). Thus, the pattern is
similar to that seen in other rearrangements of HMGA2 found
in lipomas, i.e., disruption of the HMGA2 locus leaves
intact exons 1–3 of the gene which encode the AT-hook domains and
separates them from the 3′-terminal part of the gene (15). The biological activity of HMGA2
polypeptides in which the acidic tail has been replaced by a
variable number of seemingly random amino acid residues, has not
been studied in detail. The present data nevertheless add to the
evidence that expression of truncated HMGA2 is involved in
the development of lipomas. Fedele et al (16) showed that the expression of a
truncated form of HMGA2 protein carrying only the three DNA-binding
domains, or the expression of a fusion protein carrying the three
DNA-binding domains of HMGA2 and the LIM domains of the lipoma
preferred partner gene (LPP) protein, caused transformation of
NIH3T3 cells. The acquisition of LPP ectopic sequences did not
increase the transforming ability of the truncated form of HMGA2
(16). Moreover, transgenic mice
expressing a truncated form of the HMGA2 protein develop obesity
and an abnormally high prevalence of lipomas (17,18).
The real-time PCR results indicated that full length
HMGA2 transcript was expressed in lipomas 1, 2 and 6
(Table I). In addition, FISH
showed that the breakpoint was distal to the HMGA2 locus in
lipomas 1 and 2 (data not shown). Similar data have been reported
for lipomas carrying the t(5;12)(q32~33;q14~15) (9,19)
where FISH studies (9) have shown
that the genomic breakpoints usually lie outside the HMGA2
locus. Subsequently, Bartuma et al (19) showed that 4 out of 5 examined
lipomas with t(5;12) had aberrant expression of the entire
HMGA2 gene. Similar findings have also been reported for
uterine leiomyomas with rearrangements involving chromosome band
12q15 (20): A study of 38 uterine
leiomyomas showed that dysregulation of HMGA2 expression,
not the formation of HMGA2 fusion transcripts, was the
principal pathobiological mechanism in these tumors.
The recurrent HMGA2 partner gene,
SETBP1, codes for a protein which contains six regions rich
in proline (P), glutamine (E), serine (S), and threonine (T)
residues (PEST sequences), three nuclear localization signals,
three sequential proline-rich repeats PPLPPPPP at the
carboxyl-terminal end, a region homologous to the SKI oncoprotein,
and a SET binding region (21).
The encoded protein has been shown to interact specifically with
the SET protein both in a yeast two-hybrid system and in human
cells (21). Constitutional
mutations in this gene are associated with Schinzel-Giedion midface
retraction syndrome (22).
SETBP1 is otherwise involved in hematologic malignancies
(23–25). The oncogenic function of
SETBP1 was first reported in 2006 when a NUP98-SETBP1
fusion gene was identified in T cell acute lymphoblastic leukemia
carrying a t(11;18)(p15;q12) (24). In 2010, SETBP1 was found to
be overexpressed in a case of AML cytogenetically characterized by
a t(12;18)(p13;q12) targeting ETV6 in the second breakpoint
(23). The same study also showed
that SETBP1 overexpression is a recurrent molecular event in
AML (found in 53 of 192 patients) and is associated with shorter
overall survival, especially in elderly patients (23). Recently, recurrent mutations in
SETBP1, frequently targeting the SKI-homologous domain, have
been identified in several types of myeloid malignancies, including
chronic and acute myeloid leukemias (25 and refs. therein). In the three
HMGA2-SETBP1 fusions described here, the breakpoint in
SETBP1 occurred in the 3′-untranslated region (3′-UTR). This
3′-UTR is 4.8 kbp long, or almost half the size of the 9.9 kbp long
transcript mRNA of SETBP1 (sequence with the accession
number NM_015559 version 2). The function of this 3′-UTR is unknown
but presumably it contains sequences that influence the expression
of SETBP1 (26).
Case 8, which also had a HMGA2-SETBP1 fusion,
was diagnosed as osteochondrolipoma (Fig. 1). This is a very rare type of tumor
on which no prior cytogenetic or molecular genetic information
exists. A search in ‘PubMed' using the term ‘osteochondrolipoma'
yielded 6 articles (27–32). As the name indicates, the tumor is
characterized by the presence of mature fatty tissue together with
cartilage and bone formation with lipocytes, chondrocytes, and
osteoblasts being the predominant cell types (31). The pathogenesis of
osteochondrolipomas is unknown. Lin et al (33) reported that mesenchymal stem cells
(MSCs) may be found in human lipomas and that they have
characteristics similar to those of MSCs derived from adipose
tissue. Thus, lipoma-derived MSCs can differentiate into
adipocytes, osteoblasts, and chondrocytes after induction (33) and could be the source of the
different cell types in these tumors. The present case with a
t(12;18)(q14~15;q12~21) and an HMGA2-SETBP1 fusion identical
to those found in the ordinary lipomas further supports the
association both pathogenetically and otherwise between
osteochondrolipomas and other lipoma subtypes. Worthy of mention is
that the reciprocal translocation t(3;12)(q27~28;q13~15), i.e., the
most common translocation in lipomas, has also been observed in
three cases of osteolipoma (34).
Evidently, the special phenotypes of these lipoma variants cannot
be attributed to the tumor pathogenetic mechanism.
The present study provides yet another example of
the fact that what at the cytogenetic level appears to be similar
is in fact heterogeneous at the molecular level (9,19).
This may reflect the intriguing pathogenetic role of HMGA2,
which seems to be entirely different from the highly specific gene
fusions present in, for example, myxoid liposarcomas (14).
Acknowledgements
The authors thank Hege Kilen Andersen and Kristin
Andersen for technical help. The present study was supported by
grants from the Norwegian Cancer Society and the Norwegian Radium
Hospital Foundation.
References
|
1
|
Fletcher CDM, Bridge JA, Hogendoorn PCW
and Mertens F: WHO Classification of Tumours of Soft Tissue and
Bone. 4th edition. IARC Press; Lyon: 2013
|
|
2
|
Heim S, Mandahl N, Kristoffersson U,
Mitelman F, Rööser B, Rydholm A and Willén H: Reciprocal
translocation t(3;12) (q27;q13) in lipoma. Cancer Genet Cytogenet.
23:301–304. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Turc-Carel C, Dal Cin P, Rao U, Karakousis
C and Sandberg AA: Cytogenetic studies of adipose tissue tumors. I.
A benign lipoma with reciprocal translocation t(3;12)(q28;q14).
Cancer Genet Cytogenet. 23:283–289. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bartuma H, Hallor KH, Panagopoulos I,
Collin A, Rydholm A, Gustafson P, Bauer HC, Brosjö O, Domanski HA,
Mandahl N, et al: Assessment of the clinical and molecular impact
of different cytogenetic subgroups in a series of 272 lipomas with
abnormal karyotype. Genes Chromosomes Cancer. 46:594–606. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Petit MM, Mols R, Schoenmakers EF, Mandahl
N and Van de Ven WJ: LPP, the preferred fusion partner gene of
HMGIC in lipomas, is a novel member of the LIM protein gene family.
Genomics. 36:118–129. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Schoenmakers EF, Wanschura S, Mols R,
Bullerdiek J, Van den Berghe H and Van de Ven WJ: Recurrent
rearrangements in the high mobility group protein gene, HMGI-C, in
benign mesenchymal tumours. Nat Genet. 10:436–444. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Bianchini L, Birtwisle L, Saâda E, Bazin
A, Long E, Roussel JF, Michiels JF, Forest F, Dani C, Myklebost O,
et al: Identification of PPAP2B as a novel recurrent translocation
partner gene of HMGA2 in lipomas. Genes Chromosomes Cancer.
52:580–590. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Broberg K, Zhang M, Strömbeck B, Isaksson
M, Nilsson M, Mertens F, Mandahl N and Panagopoulos I: Fusion of
RDC1 with HMGA2 in lipomas as the result of chromosome aberrations
involving 2q35–37 and 12q13–15. Int J Oncol. 21:321–326.
2002.PubMed/NCBI
|
|
9
|
Nilsson M, Mertens F, Höglund M, Mandahl N
and Panagopoulos I: Truncation and fusion of HMGA2 in lipomas with
rearrangements of 5q32→q33 and 12q14→q15. Cytogenet Genome Res.
112:60–66. 2006. View Article : Google Scholar
|
|
10
|
Nilsson M, Panagopoulos I, Mertens F and
Mandahl N: Fusion of the HMGA2 and NFIB genes in lipoma. Virchows
Arch. 447:855–858. 2005. View Article : Google Scholar
|
|
11
|
Petit MM, Schoenmakers EF, Huysmans C,
Geurts JM, Mandahl N and Van de Ven WJ: LHFP, a novel translocation
partner gene of HMGIC in a lipoma, is a member of a new family of
LHFP-like genes. Genomics. 57:438–441. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mandahl N: Methods in solid tumour
cytogenetics. Human Cytogenetics: Malignancy and Acquired
Abnormalities. Rooney DE: Oxford University Press; New York: pp.
165–203. 2001
|
|
13
|
Schaffer LG, Slovak ML and Campbell LJ:
ISCN 2009: An International System for Human Cytogenetic
Nomenclature. Karger; Basel: 2009
|
|
14
|
Heim S and Mitelman F: Cancer
Cytogenetics. 3rd Edition. Wiley-Blackwell; New York, NY: 2009
|
|
15
|
Cleynen I and Van de Ven WJ: The HMGA
proteins: A myriad of functions (Review). Int J Oncol. 32:289–305.
2008.PubMed/NCBI
|
|
16
|
Fedele M, Berlingieri MT, Scala S,
Chiariotti L, Viglietto G, Rippel V, Bullerdiek J, Santoro M and
Fusco A: Truncated and chimeric HMGI-C genes induce neoplastic
transformation of NIH3T3 murine fibroblasts. Oncogene. 17:413–418.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Arlotta P, Tai AK, Manfioletti G, Clifford
C, Jay G and Ono SJ: Transgenic mice expressing a truncated form of
the high mobility group I-C protein develop adiposity and an
abnormally high prevalence of lipomas. J Biol Chem.
275:14394–14400. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Battista S, Fidanza V, Fedele M,
Klein-Szanto AJ, Outwater E, Brunner H, Santoro M, Croce CM and
Fusco A: The expression of a truncated HMGI-C gene induces
gigantism associated with lipomatosis. Cancer Res. 59:4793–4797.
1999.PubMed/NCBI
|
|
19
|
Bartuma H, Panagopoulos I, Collin A,
Trombetta D, Domanski HA, Mandahl N and Mertens F: Expression
levels of HMGA2 in adipocytic tumors correlate with morphologic and
cytogenetic subgroups. Mol Cancer. 8:362009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Quade BJ, Weremowicz S, Neskey DM, Vanni
R, Ladd C, Dal Cin P and Morton CC: Fusion transcripts involving
HMGA2 are not a common molecular mechanism in uterine leiomyomata
with rearrangements in 12q15. Cancer Res. 63:1351–1358.
2003.PubMed/NCBI
|
|
21
|
Minakuchi M, Kakazu N, Gorrin-Rivas MJ,
Abe T, Copeland TD, Ueda K and Adachi Y: Identification and
characterization of SEB, a novel protein that binds to the acute
undifferentiated leukemia-associated protein SET. Eur J Biochem.
268:1340–1351. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Hoischen A, van Bon BW, Gilissen C, Arts
P, van Lier B, Steehouwer M, de Vries P, de Reuver R, Wieskamp N,
Mortier G, et al: De novo mutations of SETBP1 cause
Schinzel-Giedion syndrome. Nat Genet. 42:483–485. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Cristóbal I, Blanco FJ, Garcia-Orti L,
Marcotegui N, Vicente C, Rifon J, Novo FJ, Bandres E, Calasanz MJ,
Bernabeu C, et al: SETBP1 overexpression is a novel leukemogenic
mechanism that predicts adverse outcome in elderly patients with
acute myeloid leukemia. Blood. 115:615–625. 2010. View Article : Google Scholar
|
|
24
|
Panagopoulos I, Kerndrup G, Carlsen N,
Strömbeck B, Isaksson M and Johansson B: Fusion of NUP98 and the
SET binding protein 1 (SETBP1) gene in a paediatric acute T cell
lymphoblastic leukaemia with t(11;18)(p15;q12). Br J Haematol.
136:294–296. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Trimarchi T, Ntziachristos P and Aifantis
I: A new player SETs in myeloid malignancy. Nat Genet. 45:846–847.
2013. View
Article : Google Scholar : PubMed/NCBI
|
|
26
|
Matoulkova E, Michalova E, Vojtesek B and
Hrstka R: The role of the 3′ untranslated region in
post-transcriptional regulation of protein expression in mammalian
cells. RNA Biol. 9:563–576. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ahmadi SA, van Landeghem FK, Blechschmidt
C, Lieber K, Haberl EJ and Thomale UW: Intratentorial
osteochondrolipoma in a 9-year-old boy. J Neurosurg Pediatr.
3:386–391. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Gru AA and Santa Cruz DJ:
Osteochondrolipoma: A subcutaneous lipoma with chondroid and bone
differentiation of the chest wall. J Cutan Pathol. 39:461–463.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Gültekin SE, Kahraman S and Karadayı K:
Parosteal osteochondrolıpoma of the mandıble. J Oral Maxillofac
Pathol. 16:280–282. 2012. View Article : Google Scholar
|
|
30
|
Nishio J, Ideta S, Iwasaki H and Naito M:
Scapular osteochondrolipoma: Imaging features with pathological
correlation. Oncol Lett. 6:817–820. 2013.PubMed/NCBI
|
|
31
|
Rau T, Soeder S, Olk A and Aigner T:
Parosteal lipoma of the thigh with cartilaginous and osseous
differentiation: An osteochondrolipoma. Ann Diagn Pathol.
10:279–282. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Soulard R, Nguyen AT, Souraud JB, Oddon
PA, Fouet B and Cathelinaud O: Osteochondrolipoma of the
submandibular region: A case report and review of the literature.
Head Neck Pathol. 6:486–491. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Lin TM, Chang HW, Wang KH, Kao AP, Chang
CC, Wen CH, Lai CS and Lin SD: Isolation and identification of
mesenchymal stem cells from human lipoma tissue. Biochem Biophys
Res Commun. 361:883–889. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Fritchie KJ, Renner JB, Rao KW and Esther
RJ: Osteolipoma: Radiological, pathological, and cytogenetic
analysis of three cases. Skeletal Radiol. 41:237–244. 2012.
View Article : Google Scholar
|