Introduction
Sulpforaphane [SFN;
1-isotiocyanato-4-(methylsulfinyl) butane] was first isolated from
broccoli sprouts and at high concentrations is present in plants
belonging to Cruciferae family (1). It is believed to be a potent
chemopreventive agent and was proved to activate phase II
detoxification enzymes including glutathione S-transferase, epoxide
hydrolase, quinone reductase and UDP-glucuronosyltransferase, but
also to suppress phase I drug metabolism enzymes, which may
contribute to simultaneous enhanced cellular excretion and
inhibition of carcinogens (2).
Chemotherapeutic and anticancerogenic capacities of the agent were
demonstrated in vitro by inhibition of cancer cell
proliferation as well as in vivo in a broad range of animal
cancer models, including rodent carcinogenesis models of lung,
mammary gland, stomach, colon and bladder cancers (3–9).
However, the mechanisms of sulforaphane anticancer actions are not
fully understood at present.
The p21WAF1/CIP1 protein belongs to the
Cip/Kip family of Cdk (cyclin-dependent kinase) inhibitors,
participating in cell cycle control via inhibition of the G1
cyclin-Cdk complexes and maintenance of retinoblastoma tumor
suppressor protein (Rb) in its hypophosphorylated state (10–13).
This tumor suppressor mediates p53 activity in the execution of G1
and G2 cell cycle arrest (14–16).
Alterations of p21 and its function may contribute to an
unfavorable prognosis in primary solid tumors and to development of
a drug-resistance phenotype (17–19).
Loss of functional p21 along with p53 may result in unrestrained
cell cycle progression to the S phase despite the presence of DNA
damage, leading to genomic instability and carcinogenesis.
Cyclin D1 is a protein involved in the G1/S cell
cycle progression, via its participation in complexes with
cyclin-dependent kinases cdk4, cdk6 and, as a consequence Rb
phosphorylation and inhibition of its function (20–22).
In this context, the role of cyclin D1 seems to be opposite to p21
and the protein was defined as a protooncogene. In fact, increased
levels of cyclin D1 and/or its gene amplification were reported in
many neoplasms and may be related to cancer cell-specific
alterations of the cell cycle and neoplastic transformation
(23).
Our previous study suggested that cyclin D1
expression in the A549 non-small cell lung carcinoma cell line may
constitute an important determinant of the treatment efficiency
(24). Other studies also
emphasize the role of this protein, as its downregulation via siRNA
approach resulted in inhibition of proliferation, invasion and
metastasis of A549 cells in vitro and in vivo
(25).
The objective of this study was to evaluate cyclin
D1 and p21 expression in the A549 cells treated with sulforaphane
as well as to investigate a potential involvement of cyclin D1 in
the therapeutic outcome of SFN treatment by using cyclin
D1-targeting siRNA. Our studies show a significant role of cyclin
D1 in cell death induction in A549 SFN-treated cells.
Materials and methods
Cell culture and SFN treatment
The human non-small cell lung carcinoma cell line
A549 was kindly provided by Dr P. Kopiński (Department of Gene
Therapy, Ludwik Rydygier Collegium Medicum in Bydgoszcz, Nicolaus
Copernicus University in Toruń, Poland) and by Dr Rajagopal Ramesh
(Department of Pathology, Stanton L. Young Biomedical Research
Center, University of Oklahoma Health Sciences Center, Oklahoma
City, OK, USA). The cells were cultured in monolayers at 37°C in a
humidified CO2 incubator (5% CO2) in DMEM
(Gibco) supplemented with 10% fetal bovine serum (FBS; Gibco) and
50 μg/ml of gentamycin (Sigma-Aldrich). Twenty-four hours after
seeding, the cells were treated with sulforaphane (Sigma-Aldrich)
(30, 60 and 90 μM) for 24 h, and the following experimental
procedures were performed.
siRNA transfection
For the nucleofection of A549 cells a total of
1×106 cells were transfected using SE Cell Line
4D-Nucleofector X kit according to the manufacturer's instructions.
Briefly, the cells were suspended in 100 μl SE Nucleofector
solution containing 3 pmol siRNA against human CCND1 (corresponding
to sequence: 5′-AAG GCC AGT ATG ATT TAT AAA-3′; Qiagen, Hilden,
Germany). Then, the mixture was transferred into transfection
cuvettes. The electroporation was done using 4D-Nucleofector device
(Lonza, Verviers, Belgium) under the program CM-130. As a negative
control, the commercially designed AllStars negative control siRNA
(Qiagen) was used. After transfection, the cells were grown in
medium without antibiotics for 72 h and then they were used for
further experiments.
Videomicroscopy
Seven thousand (7,000) cells suspended in 2 ml of
CDMEM were seeded onto 25-mm cover glasses in cell culture dishes
and incubated for 24 h to allow the cells to spread and form
adhesions to the substratum. Then cells on the 25-mm cover glasses
were transferred to a perfusion filming chamber and were then
placed on the stage of a Zeiss phase-contrast microscope fitted
with camera. The digital images of cells were recorded every thirty
seconds on the computer drive using Scion software to obtain a time
lapse recordings of individual cells. Cells were filmed in the
presence of CDMEM alone for 25 min (50 frames) to observe normal
cell behavior. After 25 min, CDMEM was changed to CDMEM containing
sulforaphane at concentrations 30, 60 and 90 μM and recorded for an
additional 350 frames. Videos were then analyzed qualitatively to
determine the effects of sulforaphane on the cells.
Transmission electron microscopy
(TEM)
Conventional electron microscopy was used to
visualize A549 cell morphology at the ultrastructural level. The
cells were washed with sodium cacodylate buffer and fixed in 3.6%
glutaraldehyde (pH 7.2, Merck) (30 min, RT). After washing in 0.1 M
sodium cacodylate buffer (pH 7.4; Sigma-Aldrich), the cells were
postfixed in 1% buffered OsO4 for 1 h, dehydrated in
ethanol (30–90%) and acetone (90–100%) and embedded in Epon E812
(Roth). Semithin sections were stained with 1% toluidine blue and
used for targeting the cells. Ultrathin sections (40-nm thick) were
double-stained with uranyl acetate (Chemapol) and lead citrate (BDH
England). The material was examined using a transmission electron
microscope JEM 100 CX (Jeol, Tokyo, Japan) operating at 60 kV cell
images were recorded on IMAGO - EM23 film (NDT System, Warsaw,
Poland).
Trypan blue dye exclusion test
Trypan blue dye exclusion method was used to
determine the number of dead and viable cells. The cells were
trypsinized and 10 μl of cell suspension was added to an equal
volume of diluted trypan blue stain (Sigma-Aldrich). The number of
stained cells and total number of cells were determined using
Bürker counting chamber.
Annexin V/propidium iodide (PI) binding
assay
To assess the mode of cell death, the
Tali® Apoptosis kit - Annexin V Alexa Fluor®
488 and propidium iodide (Invitrogen) was used according to the
manufacturer's instructions. In short, after the SFN treatment, the
cells were collected from 6-well plates using trypsin-EDTA
solution, centrifuged at 300 × g for 8 min, resuspended in ABB
(Annexin V binding buffer) and incubated with Annexin V Alexa Fluor
488 at room temperature in the dark for 20 min. Following the
centrifugation at 300 × g for 5 min, the cells were again
resuspended in ABB and incubated with propidium iodide at room
temperature in the dark for 5 min. The cells were examined using a
Tali® Image-Based Cytometer (Invitrogen). The data were
analyzed by FCS Express Research Edition software (version 4.03; De
Novo Software, New Jersey, NJ, USA) and expressed as the percentage
of cells in each population (viable Annexin
V−/PI−; early apoptotic Annexin
V+/PI−; late apoptotic Annexin
V+/PI+; necrotic Annexin
V−/PI+).
DNA content analysis
For DNA content analysis, the Tali® Cell
Cycle kit (Invitrogen) was used according to the manufacturer's
instructions. Briefly, the cells were harvested from 6-well plates
by trypsinization, rinsed with PBS, fixed in ice-cold 70% ethanol
at 4°C, and left at −25°C overnight. The cells were then
centrifuged at 1,000 × g for 5 min at 4°C and washed with PBS.
After centrifugation at 500 × g for 10 min at 4°C, the cells were
resuspended in Tali® Cell Cycle Solution containing
propidium iodide (PI), RNase A, and Triton® X-100.
Following 30-min incubation at RT in the dark, the cells were
analyzed using Tali® Image-Based Cytometer (Invitrogen)
and the percentage of cells in each phase of cell cycle was
determined using FCS Express Research Edition software (version
4.03; De Novo Software, NJ, USA).
Flow cytometric analysis of cyclin D1 and
p21 expression
Cells grown in 6-well plates were harvested, washed
with PBS, centrifuged (5 min, 300 × g) and fixed with 1%
methanol-free, ultra pure formaldehyde (Polysciences, Inc.). After
incubation on ice (15 min in the dark) and subsequent
centrifugation (5 min, 300 × g), the cells in pellets were
permeabilized by the addition of 1 ml of ice-cold 50% (v/v)
methanol (JT Baker) for 30 min on ice, washed twice with cold PBS
and resuspended in 0.5% bovine serum albumin (BSA; Sigma-Aldrich).
For intracellular staining, the cell suspensions were transferred
into flow cytometric tubes containing 20 μl of FITC conjugated
mouse anti-human cyclin D1 (BD Pharmingen) or Alexa Fluor 488
conjugated mouse monoclonal anti-human p21 (Santa Cruz
Biotechnology, Inc.), and 200 μl of 0.5% BSA. Following a 45-min
incubation (4°C, in the dark) and washing with PBS, the cells were
centrifuged (5 min, 500 × g) to wash off excess antibody and
resuspended in 200 μl of PBS for flow cytometric analysis on
FACScan (Becton-Dickinson). CellQuest software (Becton-Dickinson)
was used to calculate the percentage of cyclin D1-positive cells,
and their mean fluorescence intensity.
RT-qPCR analysis of cyclin D1 and p21
expression
To determine the expression level of p21 and cyclin
D1, SYBR green-based and probe-based RT-qPCR assays, respectively,
were performed using LightCycler 2.0 Instrument (Roche Applied
Science) and LightCycler Software Version 4.0. In the case of SYBR
green-based assays, total RNA from the A549 cells was prepared
using the Total RNA kit (A&A Biotechnology; Gdynia, Poland)
according to the manufacturer's protocol. Then, the reverse
transcription and qPCR reactions were carried out in a single 20-μl
LightCycler capillary (Roche Applied Science) as a one-step RT-qPCR
with TranScriba reverse transcriptase and Master Mix SYBR
(TranScriba-qPCR Master Mix SYBR kit; A&A Biotechnology) and a
pair of gene-specific primers (forward;
5′-GCATGACAGATTTCTACCACTCC-3′; reverse 5′-AAGATGTAGAGCGGGCCTTT-3′),
as specified by the manufacturer. The total reaction mixture (20
μl) contained 60 ng of RNA and 0.2 μM of each primer in addition to
the TranScriba-qPCR Master Mix SYBR kit components. One cycle of
reverse transcription was carried out for 10 min at 50°C, one cycle
of denaturation for 3 min at 95°C, and 43 cycles of denaturation
for 15 sec at 95°C, followed by annealing and elongation for 30 sec
at 57°C.
In turn, a two-step real-time RT-qPCR was performed
to assess the expression level of cyclin D1. The preparation of
cell lysate was done with RealTime ready Cell Lysis kit (Roche
Applied Science) according to the manufacturer's instructions. The
resulting lysate was directly reverse transcribed using
Transcriptor Universal cDNA Master (Roche Applied Science)
following the manufacturer's instructions in a final volume of 20
μl (2 μl of the total lysate/cDNA reaction). Reverse transcription
was carried out for 10 min at 55°C, followed by incubation at 85°C
for 5 min. Synthesized cDNA was stored at −20°C until further use.
RT-qPCRs were conducted with the LightCycler TaqMan Master kit
(Roche Applied Science) in LightCycler capillaries in a 20-μl
reaction volume. PCR reaction mixture contained: 5 μl of cDNA, 1×
TaqMan Master mix, 400 nM of each primer for the examined gene
(forward 5′-TGTCCTACTACCGCCTCACA-3′; reverse
5′-CAGGGCTTCGATCTGCTC-3′), 400 nM Primer mix for the reference gene
(Human PBGD Gene assay; Roche Applied Science, cat. no. 05 046 149
001), 200 nM of each hydrolysis TaqMan probe for the examined (#16;
Roche Applied Science, cat. no. 04 686 896 001) and reference
(Human PBGD Gene assay; Roche Applied Science, cat. no. 05 046 149
001) genes and PCR-grade H2O to adjust to the final
reaction volume. Thermocycling conditions consisted of 10 min of
initial denaturation at 95°C, followed by 45 cycles of denaturation
at 95°C for 10 sec, annealing at 60°C for 30 sec and extension at
72°C for 1 sec with a single fluorescence acquisition step at the
end of extension, followed by final cooling at 40°C for 30 sec.
Relative mRNA expression levels of p21 and cyclin D1 were
quantified using the ΔΔCt method (2−ΔΔCt method)
(26) and the results were
normalized to the expression of the housekeeping gene
glyceraldehyde-3-phosphate dehydrogenase (GAPDH) or porphobilinogen
deaminase (PBGD), respectively, and presented as a fold difference
relative to a calibrator sample (untreated A549 cells).
Cyclin D1 and p21 immunofluorescence
A549 cells growing on coverslips were briefly washed
with PBS, fixed in 4% paraformaldehyde (15 min, RT) and then washed
with PBS (3×5 min). After that, the cells were incubated in
permeabilization solution (0.1% Triton X-100 in PBS) and blocked
with 1% BSA. After permeabilization, the cells were incubated with
mouse monoclonal anti cyclin D1 antibody (Sigma-Aldrich) or mouse
monoclonal anti-p21 antibody (Santa Cruz Biotechnology Inc),
respectively (60 min, RT), washed three times with PBS and
incubated with Alexa Fluor 488 goat anti-mouse IgG (Invitrogen,
Molecular Probes) (60 min, in the dark). Nuclear staining was
performed with DAPI (Sigma-Aldrich). After incubation, the cells
were washed with PBS and then mounted on microscope slides in Aqua
Poly/Mount (Polysciences, Inc.). Both cyclin D1 or p21 and DAPI
staining were examined using a Nikon Eclipse E800 fluorescence
microscope (Nikon; Tokyo, Japan) and NIS-Elements 4.0 software
(Nikon).
Statistical analysis
The analysis was performed using statistical
software (GraphPad Prism, San Diego, CA, USA). The data were
compared by the nonparametric Mann-Whitney U test and the changes
were considered statistically significant at the level of
p<0.05.
Results
Cell morphology
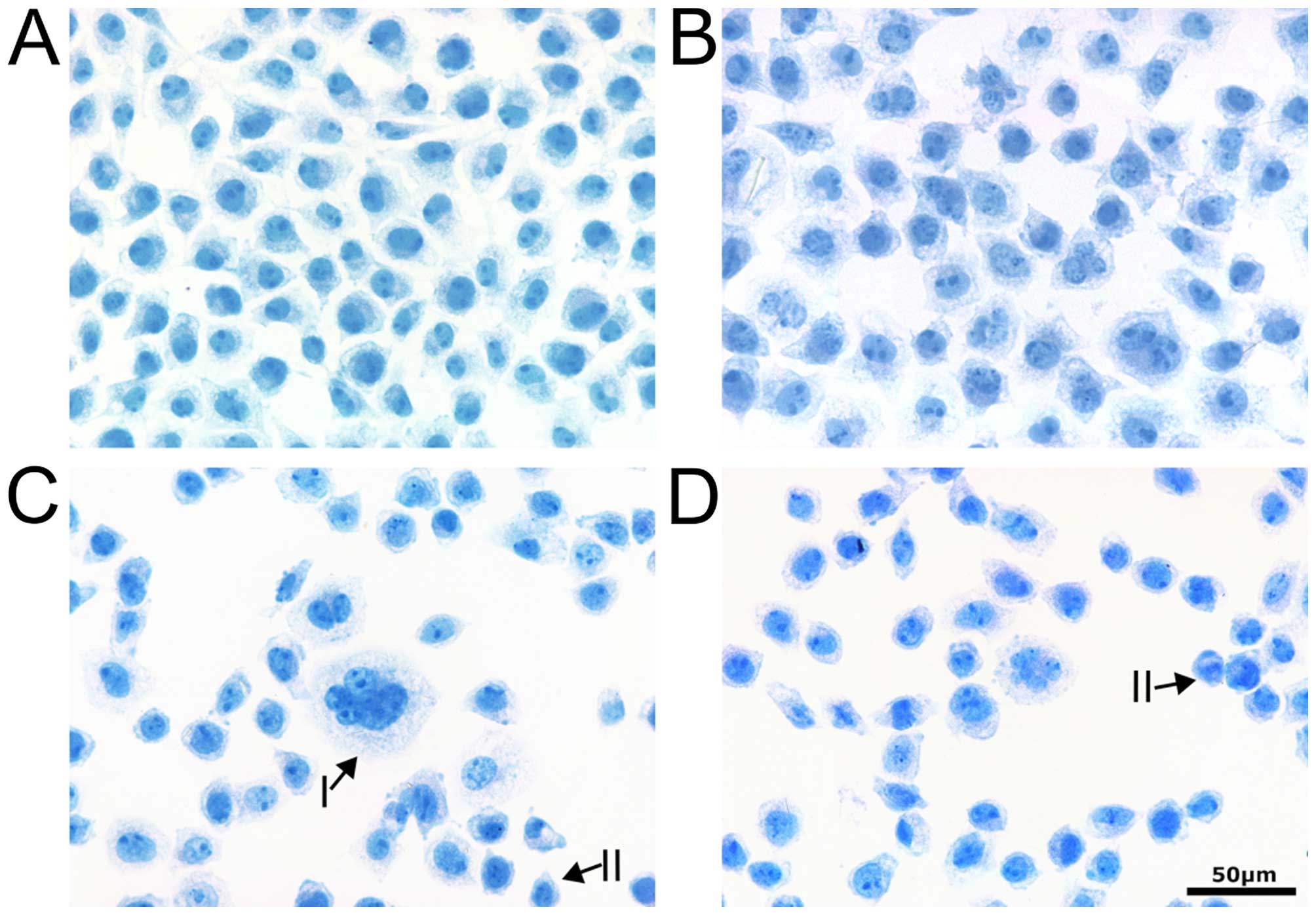
Hematoxylin staining
Control cells and the cells treated with 30 μM SFN
were regular in shape, displayed visible oval nucleus and nucleoli.
Incubation with higher doses of sulforaphane resulted in
cytoplasmic vacuolization and appearance of enlarged cells with
micronuclei, most probably displaying features of mitotic
catastrophe. Some of the cells were shrunken, with condensed
chromatin in the nucleus, suggesting early apoptosis (Fig. 1).
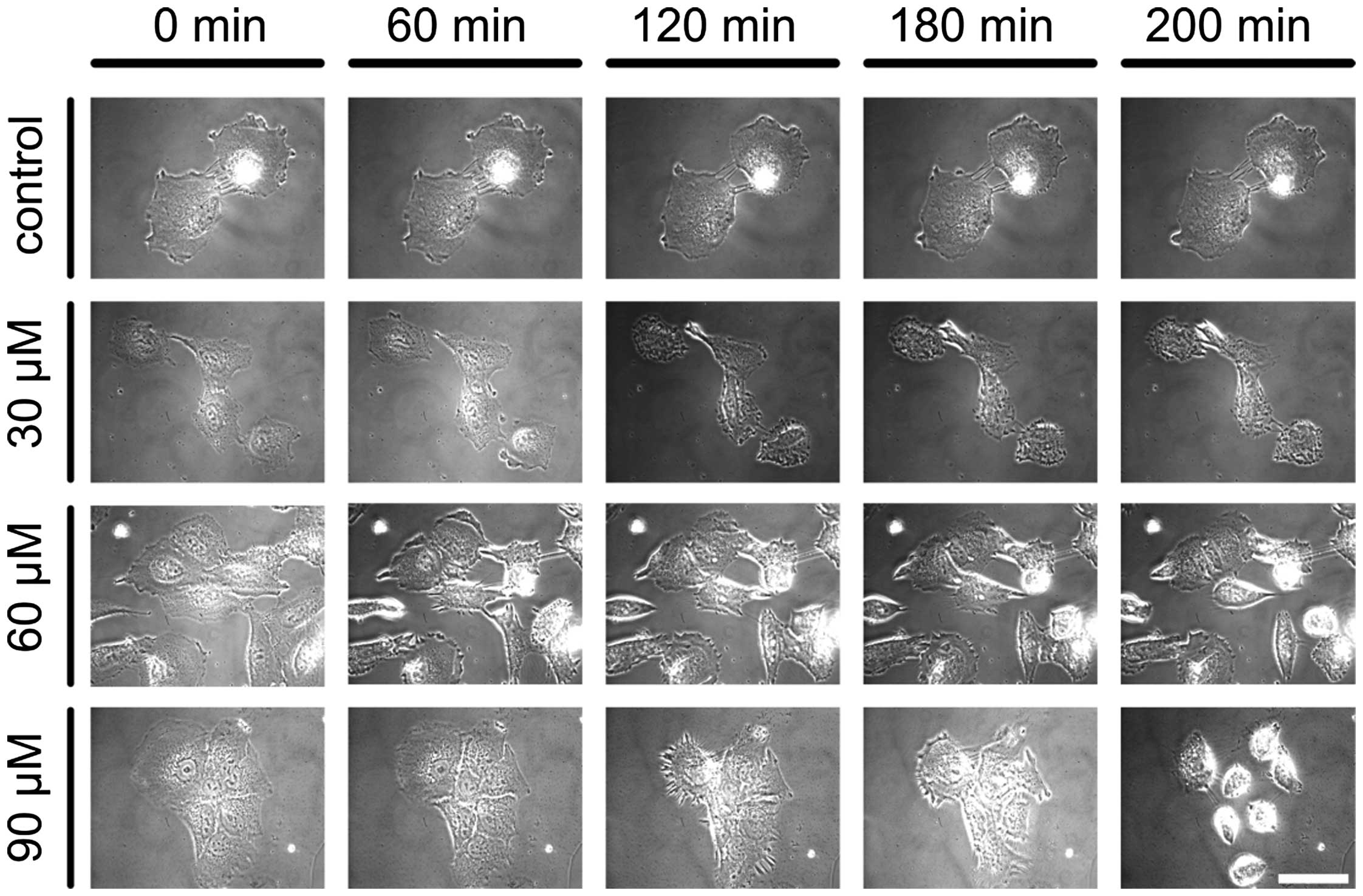
Videomicroscopy
In order to observe cell response to sulforaphane
treatment, representative groups of cells were recorded by
videomicroscopy. Cells were observed for 25 min before introduction
of SFN to the cell environment. In control conditions (cell culture
medium) cells were actively protruding and withdrawing cell edges
around cell periphery for the entire time of recording (200 min for
control and 25 min for SFN-treated cells). After replacing the
growth medium with medium containing sulforophane, we observed the
dose- and time-dependent cell response to SFN. Cells exposed to 30
μM SFN developed short microspikes and densities around cell
periphery and on cell dorsal surface. Observed cells were gradually
shrinking as time of SFN exposure progressed. Cell response was
stronger and occurred faster in higher concentrations of SFN. At
both 60 and 90 μM SFN, cells developed longer microspikes and were
withdrawing and retracting from cell edges all over the cell
periphery. Eventually cells rounded up at about three hours of SFN
treatment and were completely rounded and detached from each other
at 90 μM sulforaphane (Fig.
2).
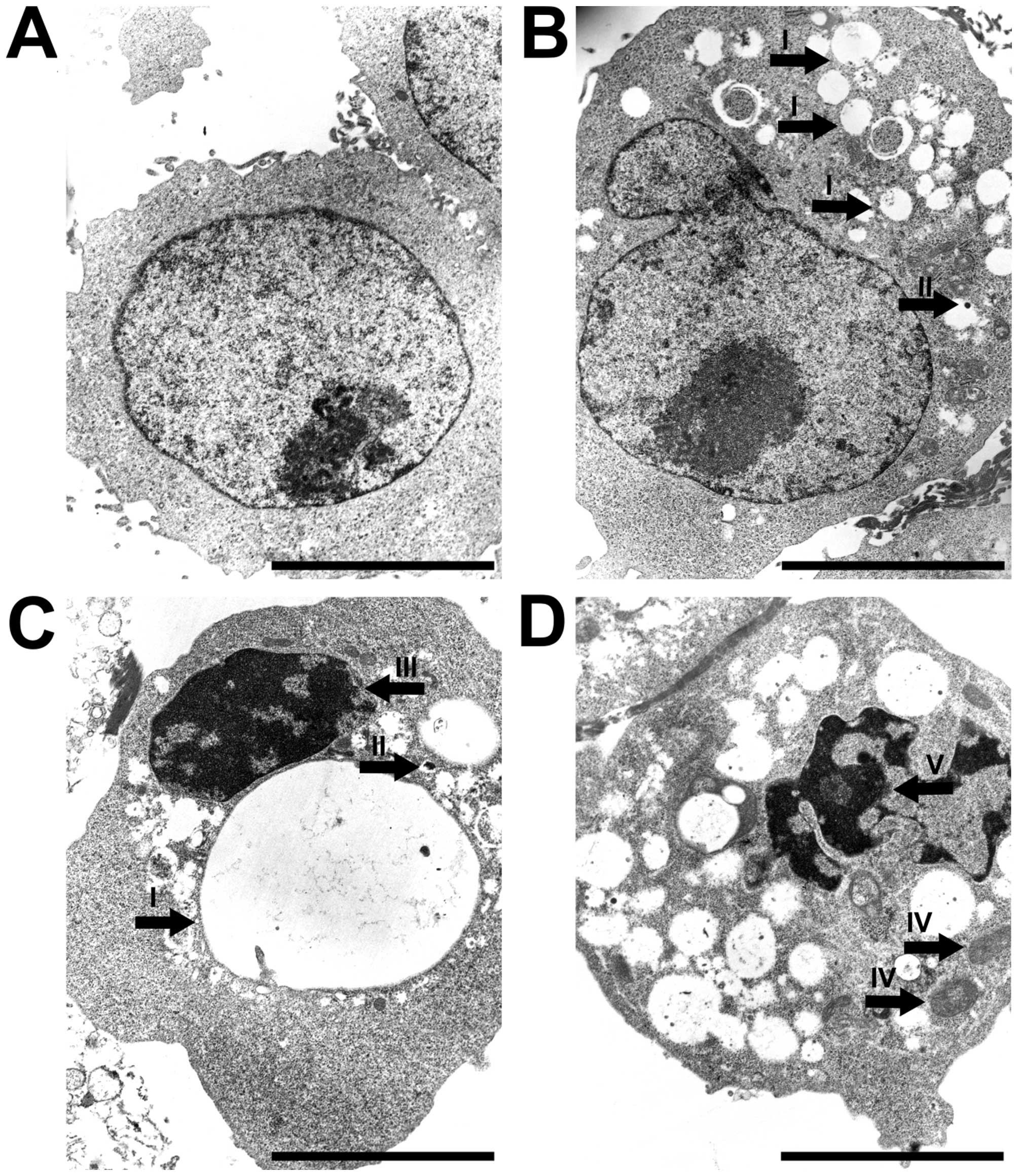
Transmission electron microscopy
Transmission electron microscopy analysis revealed
the presence of ultrastructural alterations in A549 cells, which
appeared as a result of incubation with sulforaphane. The control
cells displayed regular oval shape with prominent nucleus, distinct
nucleolus and maintained the integrity of cellular membranes
(Fig. 3A). The cells treated with
30 μM sulforaphane featured significant cytoplasmic vacuolization
as compared to control. Changes in the nuclear morphology were also
evident (Fig. 3B). Appearance of
large vacuoles and lysosome-like structures, as well as swollen
mitochondria were induced by 60 μM sulforaphane. The cell nucleus
at this concentration was shrunken, with unrecognizable nucleolus
(Fig. 3C). At the highest
sulforaphane concentration, i.e., 90 μM, the cells manifested
irregularities in shape and prominent vacuolization, often
comprising the whole cytoplasmic area. Other typical features
included chromatin condensation and disappearance of nucleoli
(Fig. 3D).
Cell death/survival
Trypan blue dye exclusion test
A dose-dependent decrease in the number of viable
A549 cells was found as a result of sulforaphane treatment. Along
with increasing SFN concentrations, 30, 60 and 90 μM, statistically
significant differences were observed in the cell survival, with
the respective median values reaching 98.4, 88 and 77.4% of the
population.
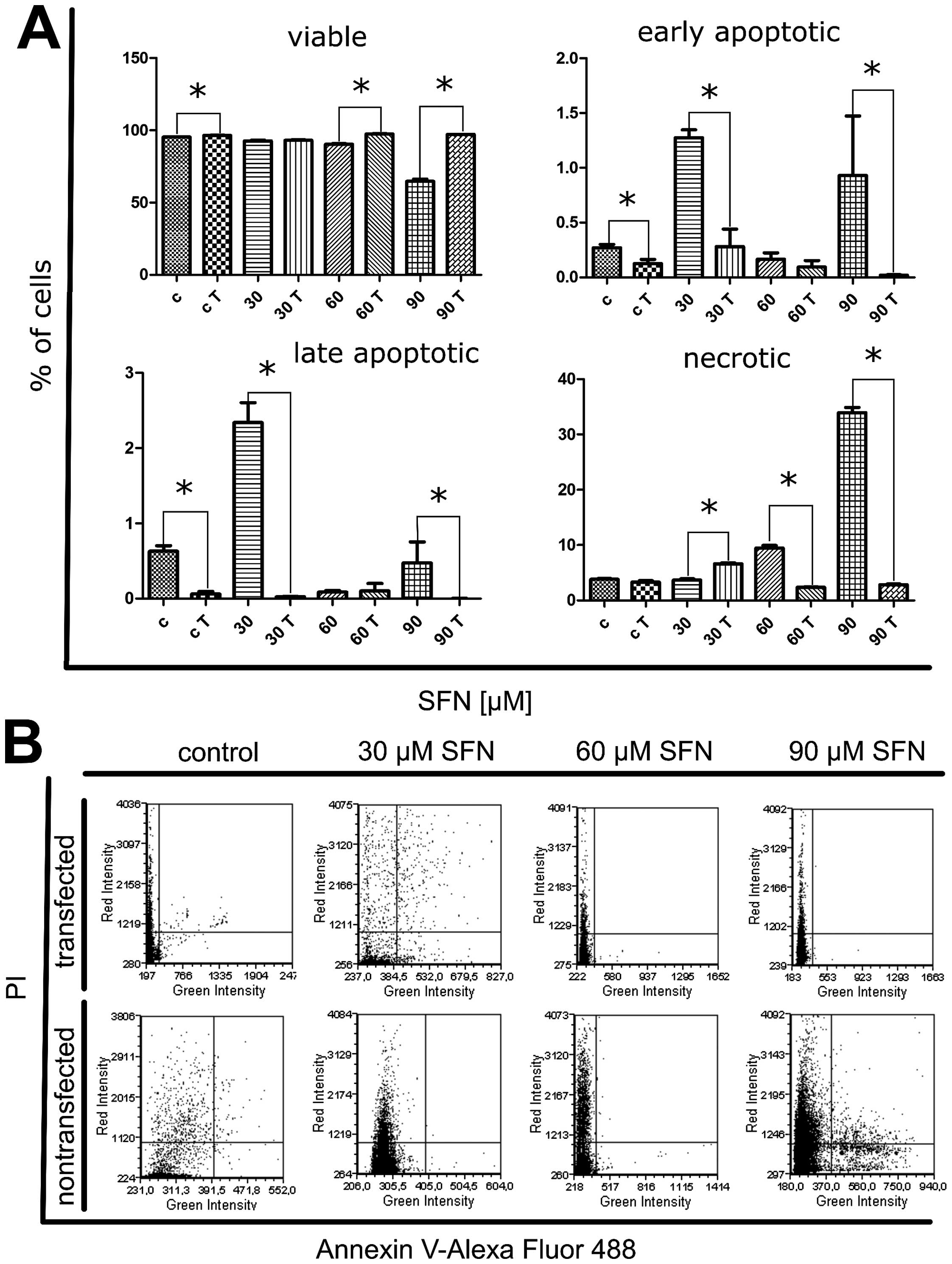
Annexin V assay
After treatment with all the concentrations of
sulforaphane used in this study the percentage of living cells
decreased (Mann-Whitney U, p<0.05) (Fig. 4). In non-transfected cells, the
percentages of early and late apoptosis was increased at 30 μM
sulforaphane as compared to the control cells, which was followed
by a further decrease at higher concentrations. The tendency was
reversed as far as necrosis is concerned, giving increased
percentage of dead cells starting at 60 μM sulforaphane. After
siRNA transfection we found decreased percentage of early and late
apoptotic cells at 30 and 90 μM sulforaphane, as well as necrotic
cells at 60 and 90 μM sulforaphane, as opposed to 30 μM
sulforaphane, at which the percentages of necrotic cells were
higher for the transfected cells (Mann-Whitney U, p<0.05). The
survival of cells was unaffected by transfection, as we obtained
higher percentages of living cells after siRNA procedure not only
at 60 and 90 μM sulforaphane concentrations, but also in control
cells, in which the percentages of early and late apoptotic cells
were significantly reduced following transfection (Fig. 4).
Cell cycle and cell cycle regulatory
proteins
Cell cycle
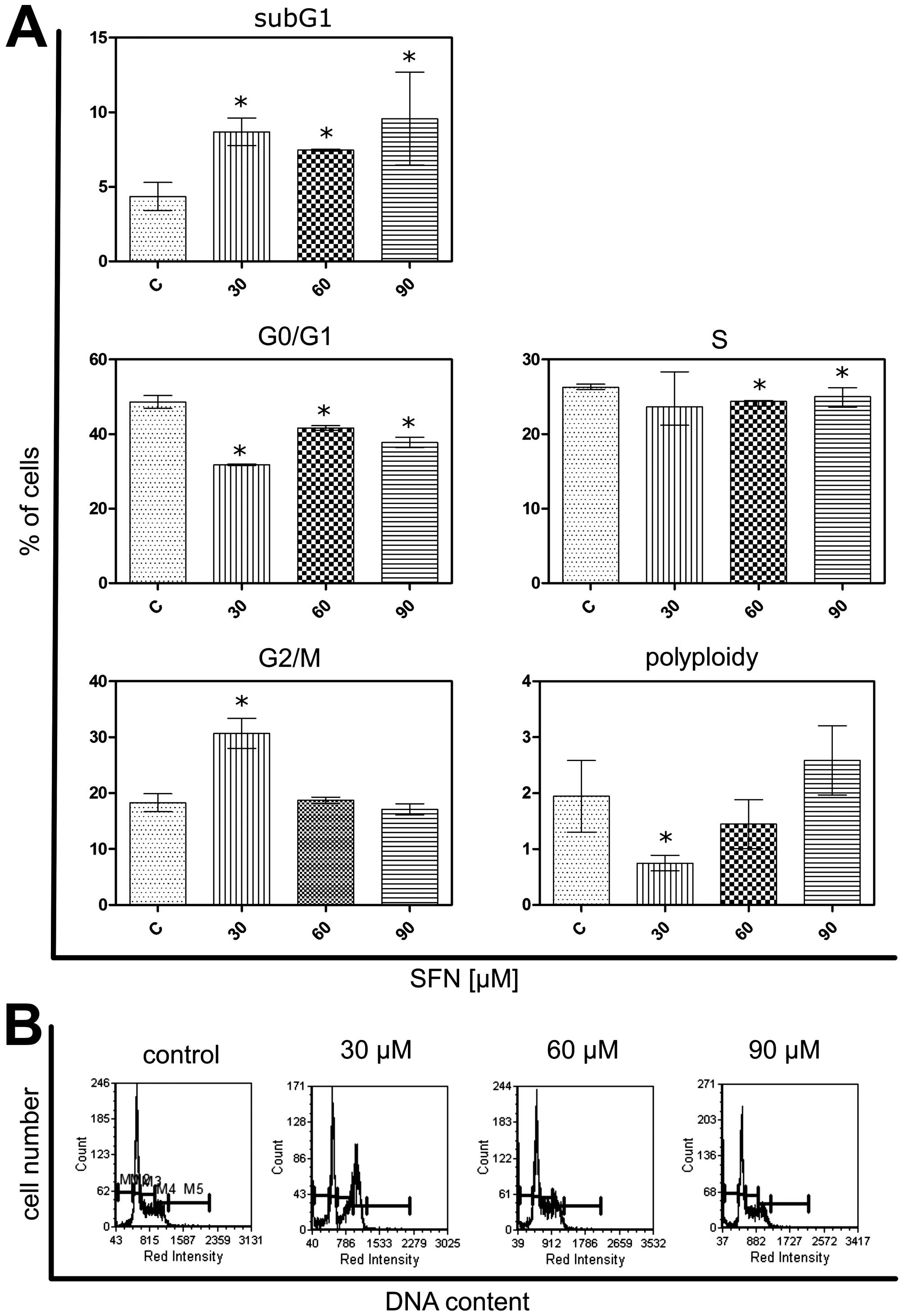
Cell cycle distribution was assessed using the
Talli-based image cytometry. The subG1 fraction increased following
the treatment with all the concentrations of sulforaphane used in
the study, which was parallel with increased percentages of cell
death observed by us. On the contrary, the G0/G1 phase population
was compromised, with the most prominent effect resulting from 30
μM dose of the agent. Interestingly, the same concentration induced
an increased G2/M phase population along with a decreased polyploid
fraction of cells, which implies a functional G2/M arrest of the
cell cycle. The S phase fraction declined in turn as a result of 60
and 90 μM sulforaphane (Fig.
5).
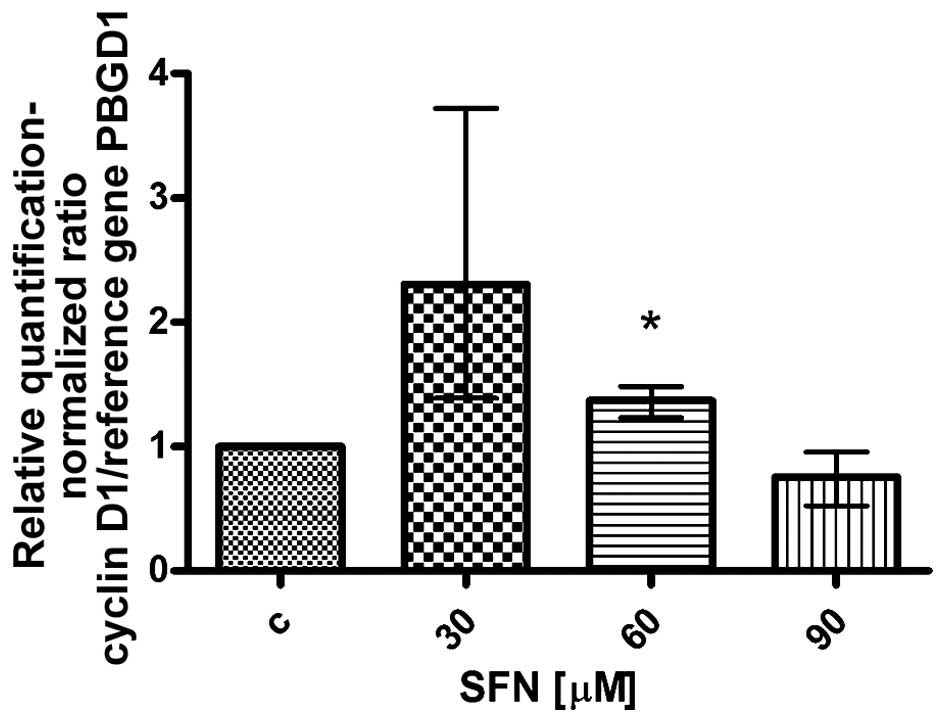
Expression of cyclin D1
Expression of cyclin D1 after treatment with
sulforaphane was changeable. RT-qPCR analysis showed that 30 and 60
μM concentrations of the drug induced an increase and 90 μM
concentration induced a decrease in cyclin D1 mRNA (Mann-Whitney U
test, p<0.05) (Fig. 6), whereas
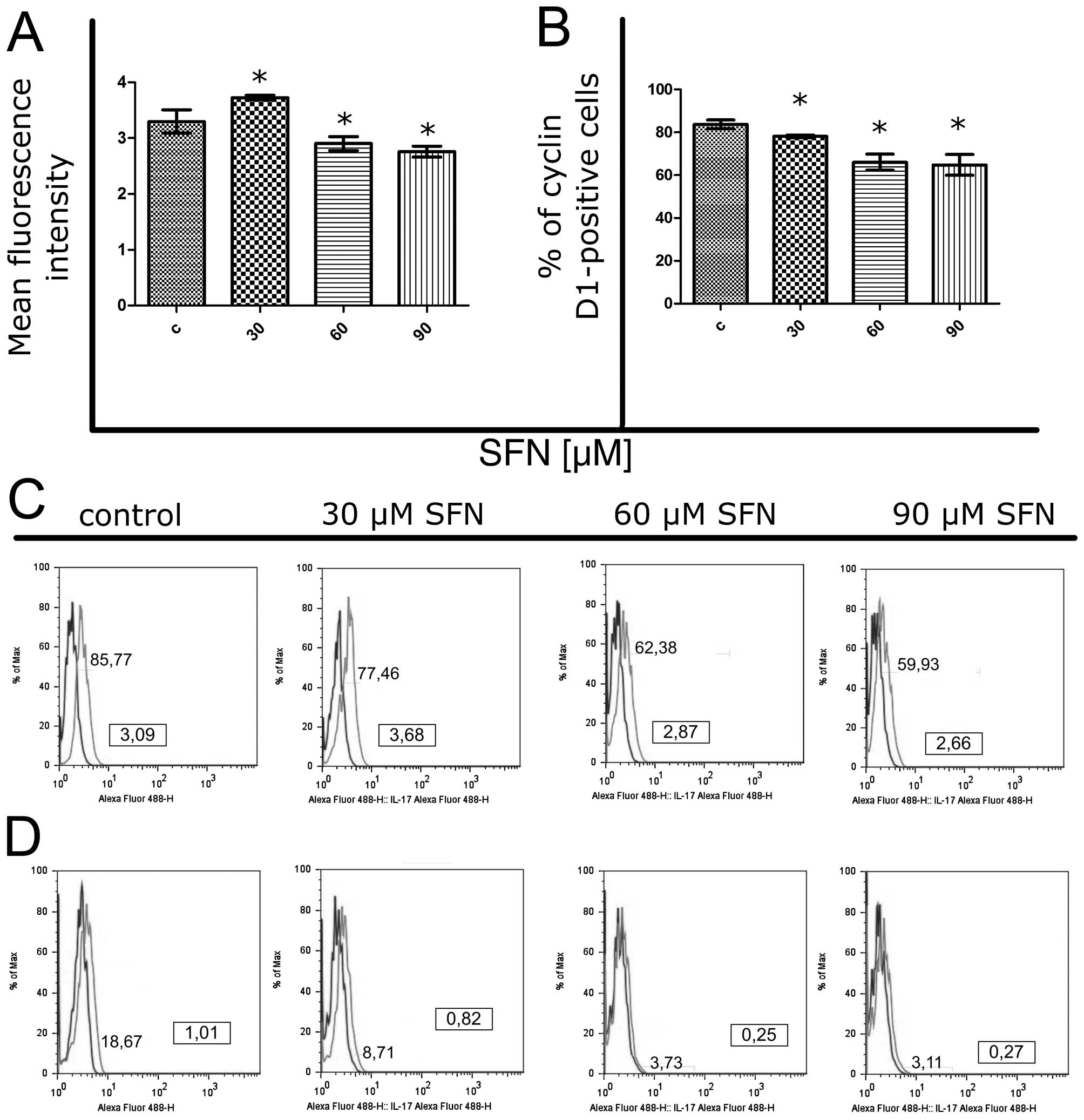
cytometric analysis revealed a slight increase in fluorescence
intensity of cyclin D1 at 30 μM, followed by decreases at 60 and 90
μM sulforaphane. Percentages of cyclin D1-positive cells were
decreased after the treatment as compared to the control cells
(Mann-Whitney U test, p<0.05), displaying a dose-dependent
tendency (Fig. 7).
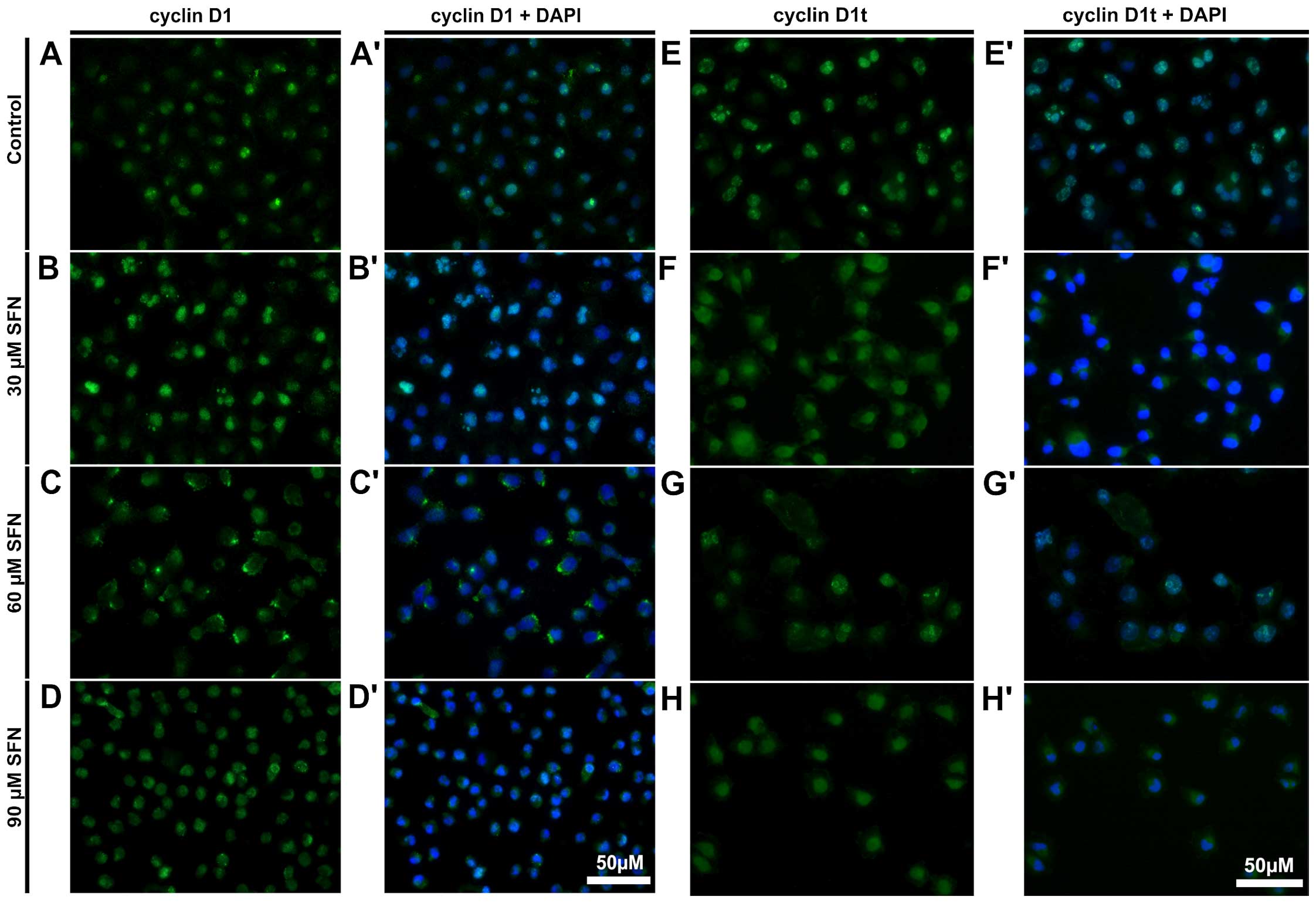
Immunofluorescent labeling of cyclin D1 in
SFN-treated cells showed similar tendency to flow cytometric
results, i.e., the most visible increase in cyclin D1 fluorescence
and nuclear localization was observed at 30 μM sulforaphane,
followed by decreased expression of this protein at higher
concentrations (Fig. 8).
The silencing effect of siRNA transfection on cyclin
D1 expression in A549 cells was confirmed cytometrically (Fig. 7) and microscopically (Fig. 8). Slight, but significantly
attenuated expression was found in control cells and after
treatment with 30 μM sulforaphane.
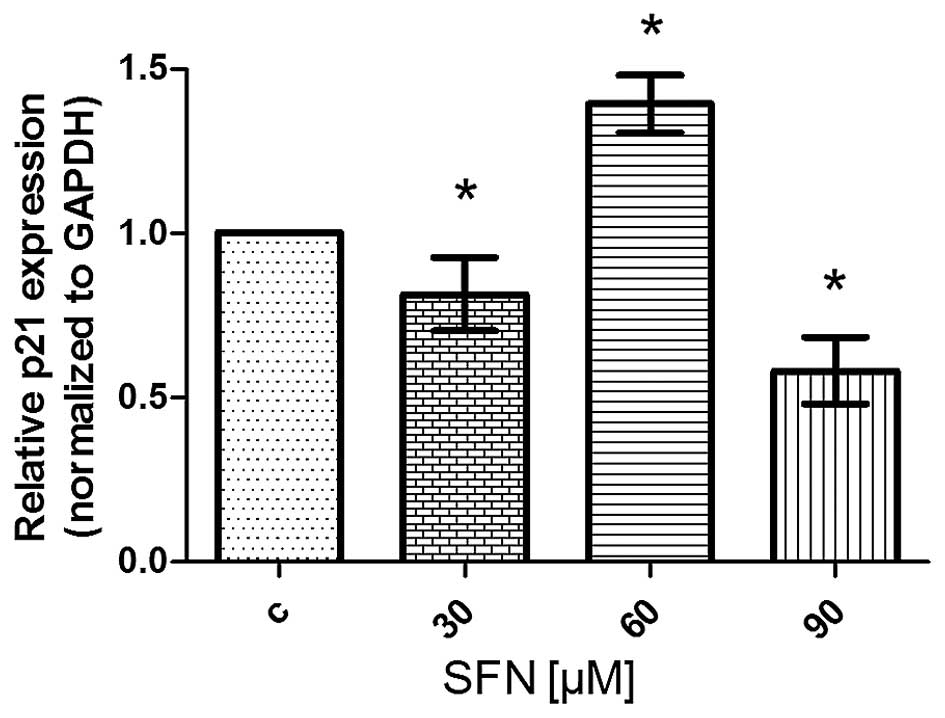
Expression of p21
Sulforaphane induced changes in the expression of
p21. RT-qPCR experiment showed significant increase of p21 mRNA
after treatment with 60 μM sulforaphane, but slightly decreased
levels resulting from the treatment with other doses, i.e., 30 and
90 μM SFN (Fig. 9). Flow
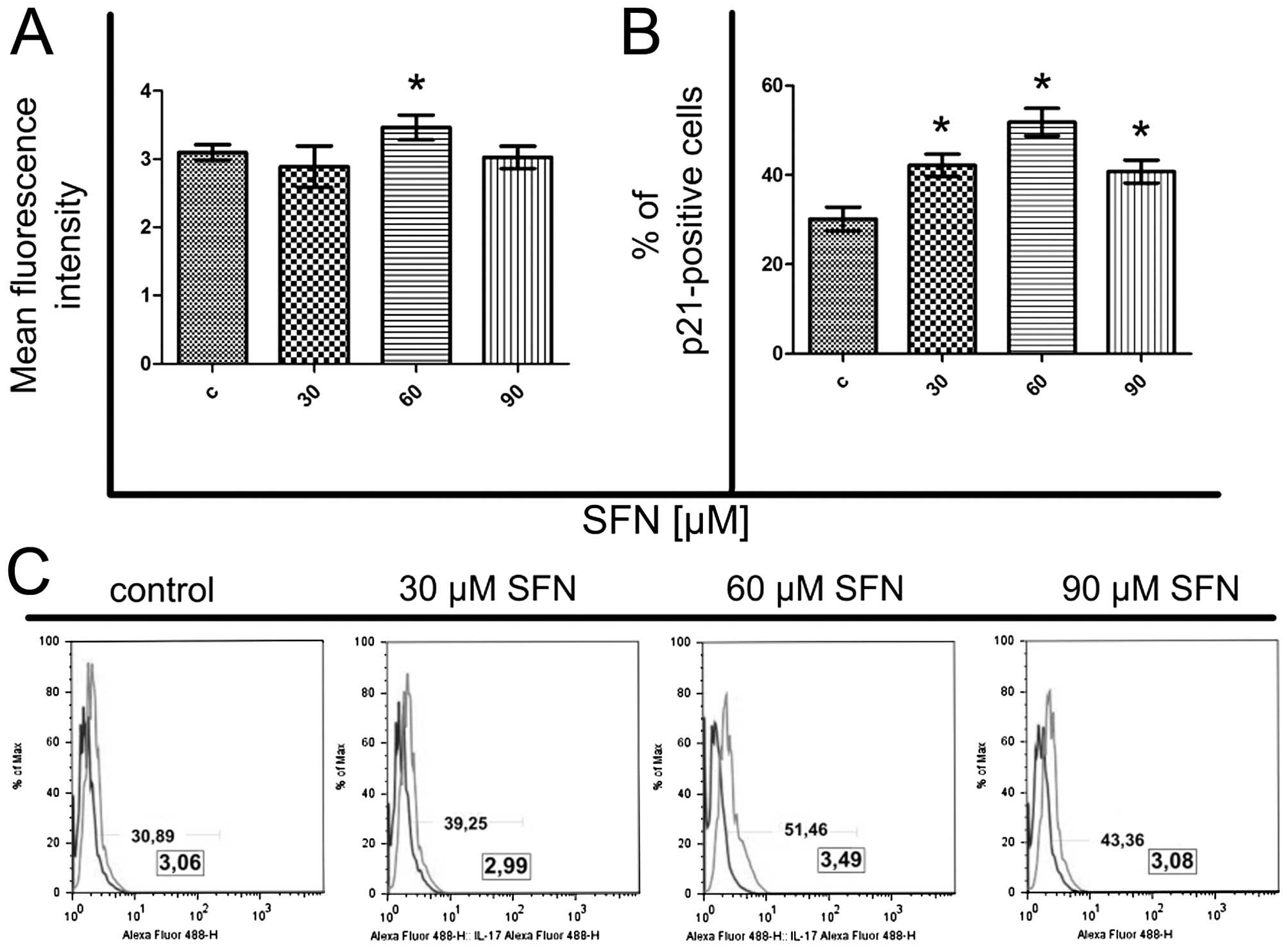
cytometric measurements indicated that fluorescence intensity of
p21 protein was increased at 60 μM SFN. At the same time, the
percentage of p21-positive cells was increased (Mann-Whitney U
test, p<0.05), with the most visible effect at 60 μM SFN
concentration (Fig. 10).
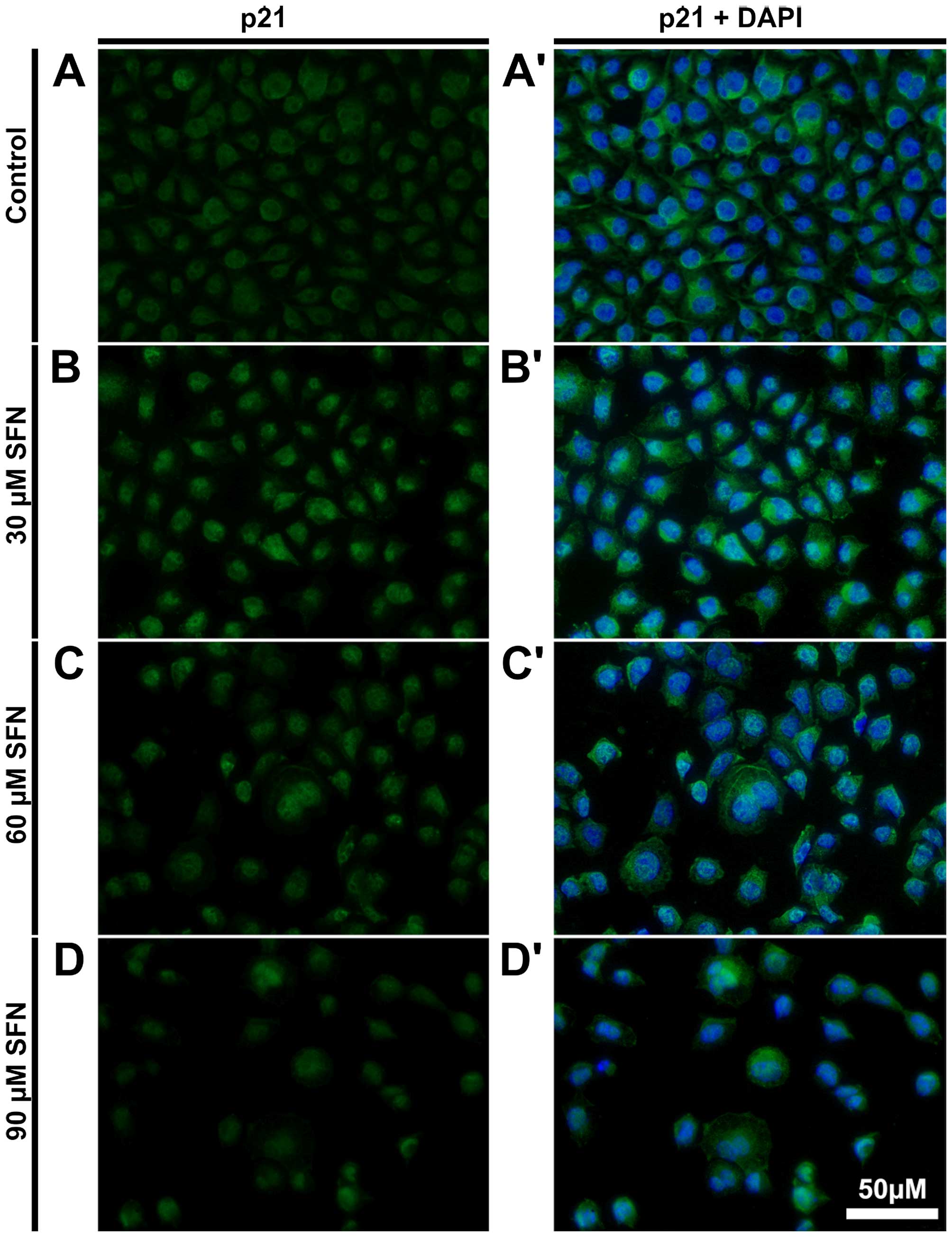
Fluorescence microscopic examination of p21 in A549
control and SFN-treated cells suggested the most significant
induction of this protein at 30 and 60 μM SFN, as the most intense
labeling featured these doses (Fig.
11).
Discussion
Sulforaphane was previously shown to trigger
specific cell death mechanisms in different types of cancer cells.
For instance, it was demonstrated that some unique events may be
typical for particular cells (27). Different apoptotic pathways,
depending on a cell line were activated in response to sulforaphane
in various breast cancer cell lines, i.e., in MDA-MB-231 cells
induction of Fas ligand and caspase-8 and -3 cascade and
poly(ADP-ribose) (PARP) was found, whereas in MDA-MB-468, MCF-7 and
T47D cells reduced Bcl-2 expression, cytochrome c release,
caspase-9 and -3 activation and PARP cleavage occurred (27). Contradictory observations appeared
regarding the effects of this agent on prostate cancer cells as
well (28,29). In several prostate cell lines
apoptotic cell death was reported (28), but in PC-3 and LNCaP cells
concomitant autophagy induction was documented, along with its
consequences for cytochrome c release and apoptosis
inhibition (29).
The role of sulforaphane (SFN) and phenethyl
isothiocyanate (PEITC) as well as their molecular targets were
previously investigated in A549 non-small cell lung cancer cells
(30). It was reported that
sulforaphane as compared to PEITC displayed rather weak
protein-binding activity, but was more potent as oxidative damage
inducer in these cells (30).
Cytotoxic effects of combined SFN and tumor necrosis factor-related
apoptosis-inducing ligand (TRAIL) treatment were shown to be
mediated by caspase-3 activation in A549 cells (31). Similarly to previous studies
(30), we have found that
apoptotic cells appeared starting from 30 μM SFN concentration.
However, percentages of early and late apoptotic cells seemed to be
low in comparison with the rate of necrosis induction, especially
at higher doses, i.e., 60 and 90 μM SFN. The major cell death mode
triggered in our experiment was necrosis, which was nevertheless
attenuated after cyclin D1 siRNA transfection. This allows us to
hypothesize that cyclin D1 may be one of the molecular targets for
sulforaphane-induced necrosis in A549 cells. Interestingly, also
the fraction of early and late apoptotic cells in response to SFN
treatment was compromised by transfection, suggesting that cyclin
D1 may be important for different modes of cell death execution in
our study. Another study documented reduced viability of
sulforaphane-treated ovarian cancer cell lines, which was
accompanied by downregulation of cyclin D1, cdk4 and cdk6 (32). In multiple myeloma cell lines with
GFP-cyclin D1 fusion protein expression it was shown that an
intrinsic mitochondrial pathway of apoptosis as well as
ER-dependent pathway were enhanced in response to bortezomib
treatment as compared to cell lines with GFP expression (33). Concomitantly, it was documented
that this effect was mediated by alterations in
apoptosis/inflammation-related gene expression pattern. As also
suggested by previous studies, cyclin D1 overexpression may
contribute to sensitization of myeloma cells to different
anti-myeloma compounds including dexamethasone, melphalan,
bortezomib, and immunomodulatory thalidomide compounds, which was
most probably related to a concomitant induction of the prolonged
S-phase (34). Interestingly, in
B-lymphoid cell lines it was found that constitutive low levels of
cyclin D1 may result in delayed apoptotic response via chaperoning
activity of induced Hsp70 exerted on pro-apoptotic factors
(35). In this study we documented
that silencing of cyclin D1 in A549 cells may result in
significantly reduced necrotic cell death following SFN treatment,
suggesting that this protein plays important role in modulation of
not only apoptotic, but also necrotic cell death mechanisms.
Recently, a role of cyclin D1 in the programmed necrotic cell death
of neurons via the ROCK-p27Kip1-cyclinD1/CDK4-LIMK2-DRP1 has been
documented to be related to mitochondrial dysfunction (36). One of the mechanisms of SFN action
may be ROS generation. We observed abundant swollen mitochondria by
TEM, and believe that similar pathway may be involved in necrosis
induction in A549 cells by SFN.
We have previously shown that high levels of cyclin
D1 may be crucial for polyploid cell appearance/stability after
mitotic failure in A549 and HL-60 cells (24,37).
Furthermore, it has been suggested that one of the
pathways through which SFN influences cyclin D1 may be via
inhibition of NF-κB in mouse prostate cancer model (38). Moreover, downregulation of cyclin
D1 in sulforaphane-treated HT-29 human carcinoma cells was proven
to depend on SFN-induced oxidative stress and may be mediated by
JNK pathway as well (39). In this
study, a decrease of cyclin D1 protein was found resulting from 60
and 90 μM SFN as well. Besides cell death, we observed also cell
cycle arrest in G2/M, which was evident at 30 μM SFN-treated cells
as compared to control cells, but not at higher concentrations,
suggesting cell death induction from this control point of the cell
cycle.
Thus far influence of different synthetic or natural
anticancer compounds on cell cycle regulatory proteins has been
investigated in A549 cells, often showing downregulation of cyclin
D1 and increased expression of p21, accompanying G0/G1 or G2/M cell
cycle arrests and apoptotic cell death (40–45).
Previous studies have shown that growth inhibitory effects of
sulforaphane may also result from the G1 cell cycle arrest, which
correlated with reduced cyclin D1 and p21 induction (39). p21 was shown to be an important
mediator of A549 cells sensitivity to chemotherapeutic drugs
(46). In this study we also
observed p21 induction at 60 μM SFN as revealed by fluorescence
intensity and RT-qPCR analysis, which may be correlated with a
slightly increased percentage of G0/G1 cells at this concentration
as compared with the lowest SFN dose. The percentage of
p21-positive cells was significantly increased by sulforaphane
treatment, which also may have contributed to G2/M cell cycle
arrest. However, in our study this cell cycle checkpoint seemed to
be active only at the lowest dose of SFN, most probably due to
enhanced cell death mechanisms at higher concentrations.
In conclusion our results indicate that the major
cell death mode induced by sulforaphane in A549 cells was necrosis,
which was most probably related to oxidative stress induction.
Differential regulation of cell cycle proteins cyclin D1 and p21
was observed as a result of the treatment, which may be dependent
on the applied concentration of SFN. Cyclin D1 seems to be an
important target for regulation and determination of the outcome of
the treatment, i.e., cell death induction in A549 cells, which was
evident in response to SFN, but also visible in control cells,
albeit to a much lower degree. To the best of our knowledge this is
the first report documenting a substantial contribution of cyclin
D1 to cell death, especially necrosis induction, in SFN-treated
A549 cells.
Acknowledgements
This study was supported by the Nicolaus Copernicus
University in Toruń, Collegium Medicum, Faculty of Medicine (grant
no. 895).
References
|
1
|
Clarke JD, Dashwood RH and Ho E:
Multi-targeted prevention of cancer by sulforaphane. Cancer Lett.
269:291–304. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Brooks JD, Paton VG and Vidanes G: Potent
induction of phase 2 enzymes in human prostate cells by
sulforaphane. Cancer Epidemiol Biomarkers Prev. 10:949–954.
2001.PubMed/NCBI
|
|
3
|
Hecht SS, Kenney PM, Wang M and Upadhyaya
P: Benzyl isothiocyanate: An effective inhibitor of polycyclic
aromatic hydrocarbon tumorigenesis in A/J mouse lung. Cancer Lett.
187:87–94. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhang Y, Kensler TW, Cho CG, Posner GH and
Talalay P: Anticarcinogenic activities of sulforaphane and
structurally related synthetic norbornyl isothiocyanates. Proc Natl
Acad Sci USA. 91:3147–3150. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Singletary K and MacDonald C: Inhibition
of benzo[a]pyrene- and 1,6-dinitropyrene-DNA adduct formation in
human mammary epithelial cells bydibenzoylmethane and sulforaphane.
Cancer Lett. 155:47–54. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Fahey JW, Haristoy X, Dolan PM, Kensler
TW, Scholtus I, Stephenson KK, Talalay P and Lozniewski A:
Sulforaphane inhibits extracellular, intracellular, and
antibiotic-resistant strains of Helicobacter pylori and prevents
benzo[a]pyrene-induced stomach tumors. Proc Natl Acad Sci USA.
99:7610–7615. 2002. View Article : Google Scholar
|
|
7
|
Kassie F, Uhl M, Rabot S, Grasl-Kraupp B,
Verkerk R, Kundi M, Chabicovsky M, Schulte-Hermann R and Knasmüller
S: Chemoprevention of 2-amino-3-methylimidazo[4,5-f]quinoline
(IQ)-induced colonic and hepatic preneoplastic lesions in the F344
rat by cruciferous vegetables administered simultaneously with the
carcinogen. Carcinogenesis. 24:255–261. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Chung FL, Conaway CC, Rao CV and Reddy BS:
Chemoprevention of colonic aberrant crypt foci in Fischer rats by
sulforaphane and phenethyl isothiocyanate. Carcinogenesis.
21:2287–2291. 2000. View Article : Google Scholar
|
|
9
|
Verhoeven DT, Verhagen H, Goldbohm RA, van
den Brandt PA and van Poppel G: A review of mechanisms underlying
anti-carcinogenicity by brassica vegetables. Chem Biol Interact.
103:79–129. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Harper JW, Adami GR, Wei N, Keyomarsi K
and Elledge SJ: The p21 Cdk-interacting protein Cip1 is a potent
inhibitor of G1 cyclin-dependent kinases. Cell. 75:805–816. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Xiong Y, Hannon GJ, Zhang H, Casso D,
Kobayashi R and Beach D: p21 is a universal inhibitor of cyclin
kinases. Nature. 366:701–704. 1993. View
Article : Google Scholar : PubMed/NCBI
|
|
12
|
Denicourt C and Dowdy SF: Cip/Kip
proteins: More than just CDKs inhibitors. Genes Dev. 18:851–855.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Gartel AL, Serfas MS and Tyner AL:
p21-negative regulator of the cell cycle. Proc Soc Exp Biol Med.
213:138–149. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Waldman T, Kinzler KW and Vogelstein B:
p21 is necessary for the p53-mediated G1 arrest in human cancer
cells. Cancer Res. 55:5187–5190. 1995.PubMed/NCBI
|
|
15
|
Wu H, Wade M, Krall L, Grisham J, Xiong Y
and Van Dyke T: Targeted in vivo expression of the cyclin-dependent
kinase inhibitor p21 halts hepatocyte cell-cycle progression,
postnatal liver development and regeneration. Genes Dev.
10:245–260. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Bunz F, Dutriaux A, Lengauer C, Waldman T,
Zhou S, Brown JP, Sedivy JM, Kinzler KW and Vogelstein B:
Requirement for p53 and p21 to sustain G2 arrest after DNA damage.
Science. 282:1497–1501. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Sharpless NE and DePinho RA: Telomeres,
stem cells, senescence, and cancer. J Clin Invest. 113:160–168.
2004. View
Article : Google Scholar : PubMed/NCBI
|
|
18
|
Cariou S, Donovan JC, Flanagan WM, Milic
A, Bhattacharya N and Slingerland JM: Down-regulation of
p21WAF1/CIP1 or p27Kip1 abrogates
antiestrogen-mediated cell cycle arrest in human breast cancer
cells. Proc Natl Acad Sci USA. 97:9042–9046. 2000. View Article : Google Scholar
|
|
19
|
Giannakakou P, Robey R, Fojo T and
Blagosklonny MV: Low concentrations of paclitaxel induce cell
type-dependent p53, p21 and G1/G2 arrest instead of mitotic arrest:
Molecular determinants of paclitaxel-induced cytotoxicity.
Oncogene. 20:3806–3813. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Baldin V, Lukas J, Marcote MJ, Pagano M
and Draetta G: Cyclin D1 is a nuclear protein required for cell
cycle progression in G1. Genes Dev. 7:812–821. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Sellers WR and Kaelin WG Jr: Role of the
retinoblastoma protein in the pathogenesis of human cancer. J Clin
Oncol. 15:3301–3312. 1997.PubMed/NCBI
|
|
22
|
Malumbres M, Sotillo R, Santamaría D,
Galán J, Cerezo A, Ortega S, Dubus P and Barbacid M: Mammalian
cells cycle without the D-type cyclin-dependent kinases Cdk4 and
Cdk6. Cell. 118:493–504. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Donnellan R and Chetty R: Cyclin D1 and
human neoplasia. Mol Pathol. 51:1–7. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Litwiniec A, Gackowska L, Helmin-Basa A,
Zuryń A and Grzanka A: Low-dose etoposide-treatment induces
endoreplication and cell death accompanied by cytoskeletal
alterations in A549 cells: Does the response involve senescence?
The possible role of vimentin. Cancer Cell Int. 13:92013.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Huang H, Hu YD, Li N and Zhu Y: Inhibition
of tumor growth and metastasis by non-small cell lung cancer cells
transfected with cyclin D1-targeted siRNA. Oligonucleotides.
19:151–162. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(T)(−Delta Delta C) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
27
|
Pledgie-Tracy A, Sobolewski MD and
Davidson NE: Sulforaphane induces cell type-specific apoptosis in
human breast cancer cell lines. Mol Cancer Ther. 6:1013–1021. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Singh AV, Xiao D, Lew KL, Dhir R and Singh
SV: Sulforaphane induces caspase-mediated apoptosis in cultured
PC-3 human prostate cancer cells and retards growth of PC-3
xenografts in vivo. Carcinogenesis. 25:83–90. 2004. View Article : Google Scholar
|
|
29
|
Herman-Antosiewicz A, Johnson DE and Singh
SV: Sulforaphane causes autophagy to inhibit release of cytochrome
C and apoptosis in human prostate cancer cells. Cancer Res.
66:5828–5835. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Mi L, Wang X, Govind S, Hood BL, Veenstra
TD, Conrads TP, Saha DT, Goldman R and Chung FL: The role of
protein binding in induction of apoptosis by phenethyl
isothiocyanate and sulforaphane in human non-small lung cancer
cells. Cancer Res. 67:6409–6416. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Jin CY, Moon DO, Lee JD, Heo MS, Choi YH,
Lee CM, Park YM and Kim GY: Sulforaphane sensitizes tumor necrosis
factor-related apoptosis-inducing ligand-mediated apoptosis through
downregulation of ERK and Akt in lung adenocarcinoma A549 cells.
Carcinogenesis. 28:1058–1066. 2007. View Article : Google Scholar
|
|
32
|
Chaudhuri D, Orsulic S and Ashok BT:
Antiproliferative activity of sulforaphane in Akt-overexpressing
ovarian cancer cells. Mol Cancer Ther. 6:334–345. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Bustany S, Cahu J, Guardiola P and Sola B:
Cyclin D1 sensitizes myeloma cells to endoplasmic reticulum
stress-mediated apoptosis by activating the unfolded protein
response pathway. BMC Cancer. 15:2622015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kuroda Y, Sakai A, Tsuyama N, Katayama Y,
Munemasa S, Asaoku H, Okikawa Y, Nakaju N, Mizuno M, Ogawa K, et
al: Ectopic cyclin D1 overexpression increases chemosensitivity but
not cell proliferation in multiple myeloma. Int J Oncol.
33:1201–1213. 2008.PubMed/NCBI
|
|
35
|
Roué G, Pichereau V, Lincet H, Colomer D
and Sola B: Cyclin D1 mediates resistance to apoptosis through
upregulation of molecular chaperones and consequent redistribution
of cell death regulators. Oncogene. 27:4909–4920. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kim JE, Ryu HJ, Kim MJ and Kang TC: LIM
kinase-2 induces programmed necrotic neuronal death via dysfunction
of DRP1-mediated mitochondrial fission. Cell Death Differ.
21:1036–1049. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Zuryń A, Litwiniec A,
Klimaszewska-Wiśniewska A, Nowak JM, Gackowska L, Myśliwiec BJ,
Pawlik A and Grzanka A: Expression of cyclin D1 after treatment
with doxorubicin in the HL-60 cell line. Cell Biol Int. 38:857–867.
2014. View Article : Google Scholar
|
|
38
|
Shankar S, Ganapathy S and Srivastava RK:
Sulforaphane enhances the therapeutic potential of TRAIL in
prostate cancer orthotopic model through regulation of apoptosis,
metastasis, and angiogenesis. Clin Cancer Res. 14:6855–6866. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Shen G, Xu C, Chen C, Hebbar V and Kong
AN: p53-independent G1 cell cycle arrest of human colon carcinoma
cells HT-29 by sulforaphane is associated with induction of p21CIP1
and inhibition of expression of cyclin D1. Cancer Chemother
Pharmacol. 57:317–327. 2006. View Article : Google Scholar
|
|
40
|
Yu J, Sun R, Zhao Z and Wang Y:
Auricularia polytricha polysaccharides induce cell cycle arrest and
apoptosis in human lung cancer A549 cells. Int J Biol Macromol.
68:67–71. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Zhang F, Zhang T, Teng ZH, Zhang R, Wang
JB and Mei QB: Sensitization to gamma-irradiation-induced cell
cycle arrest and apoptosis by the histone deacetylase inhibitor
trichostatin A in non-small cell lung cancer (NSCLC) cells. Cancer
Biol Ther. 8:823–831. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Lv XJ, Zhao LJ, Hao YQ, Su ZZ, Li JY, Du
YW and Zhang J: Schisandrin B inhibits the proliferation of human
lung adenocarcinoma A549 cells by inducing cycle arrest and
apoptosis. Int J Clin Exp Med. 8:6926–6936. 2015.PubMed/NCBI
|
|
43
|
Yuan L, Zhang Y, Xia J, Liu B, Zhang Q,
Liu J, Luo L, Peng Z, Song Z and Zhu R: Resveratrol induces cell
cycle arrest via a p53-independent pathway in A549 cells. Mol Med
Rep. 11:2459–2464. 2015.
|
|
44
|
Sikdar S, Mukherjee A and Khuda-Bukhsh AR:
Anti-lung cancer potential of pure esteric-glycoside
condurangogenin A against nonsmall-cell lung cancer cells in vitro
via p21/p53 mediated cell cycle modulation and DNA damage-induced
apoptosis. Pharmacogn Mag. 11(Suppl 1): S73–S85. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Singh N, Nambiar D, Kale RK and Singh RP:
Usnic acid inhibits growth and induces cell cycle arrest and
apoptosis in human lung carcinoma A549 cells. Nutr Cancer. 65(Suppl
1): 36–43. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Liu Z, Sun M, Lu K, Liu J, Zhang M, Wu W,
De W, Wang Z and Wang R: The long noncoding RNA HOTAIR contributes
to cisplatin resistance of human lung adenocarcinoma cells via
downregualtion of p21WAF1/CIP1 expression. PLoS One.
8:e772932013. View Article : Google Scholar
|