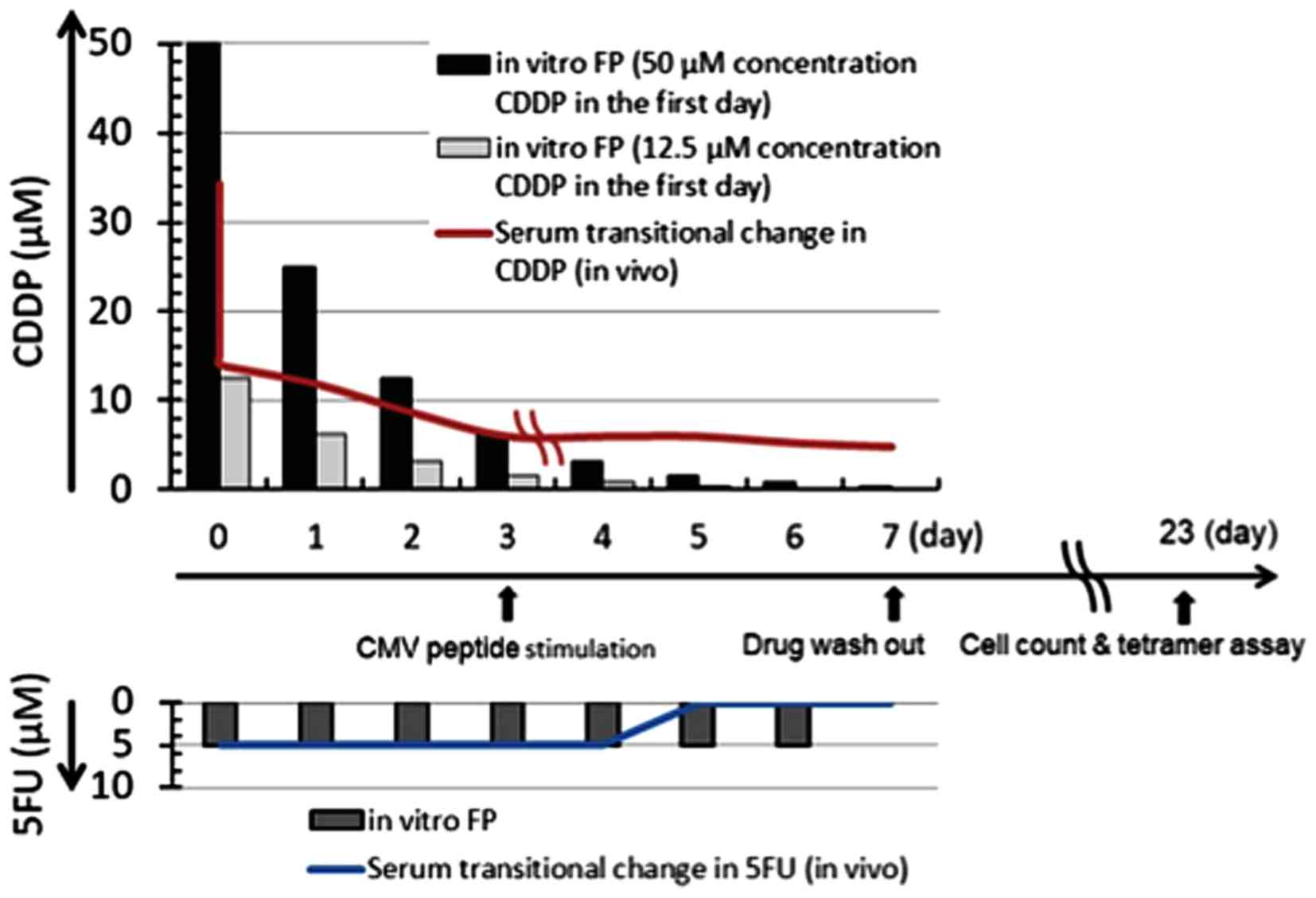
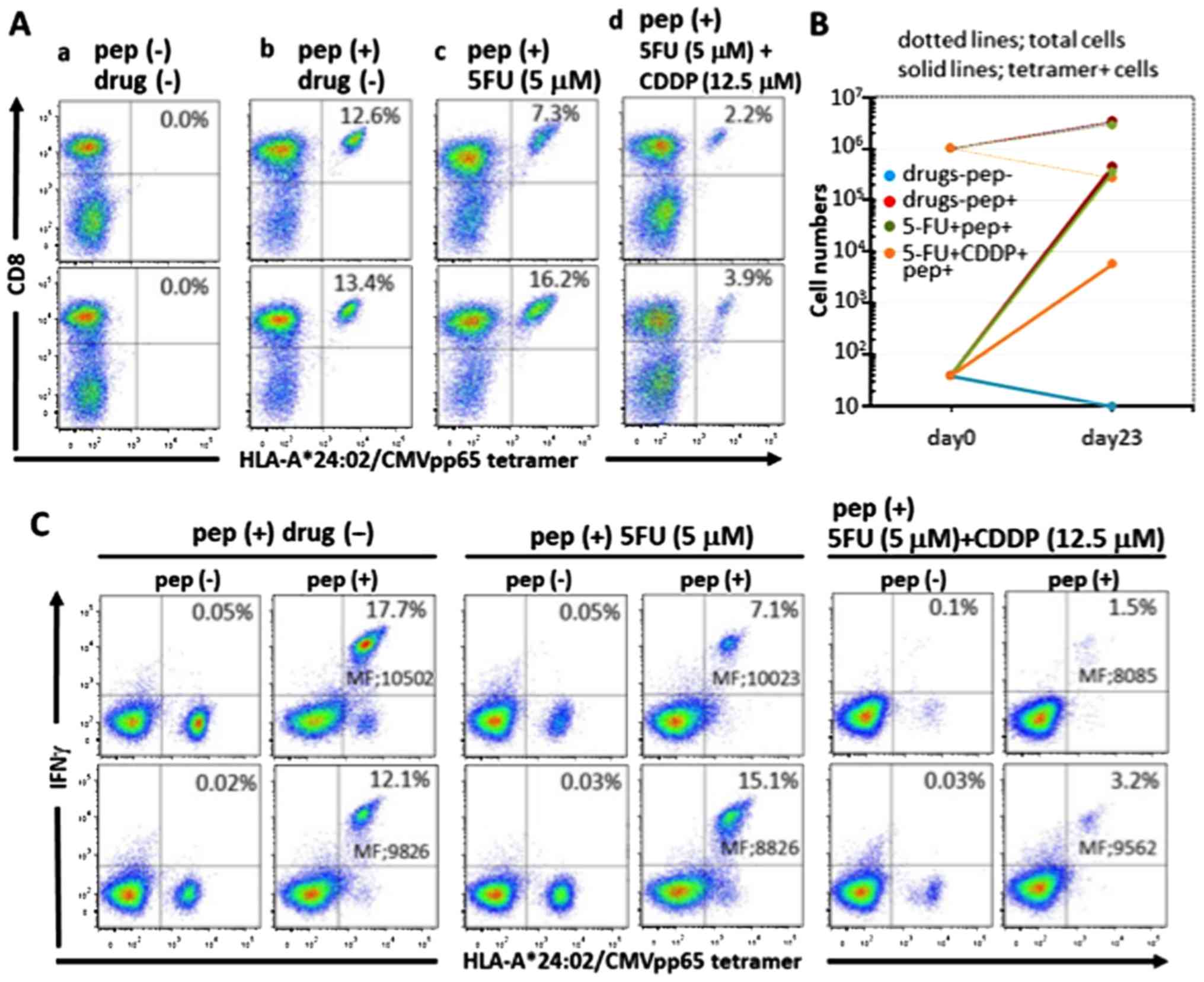
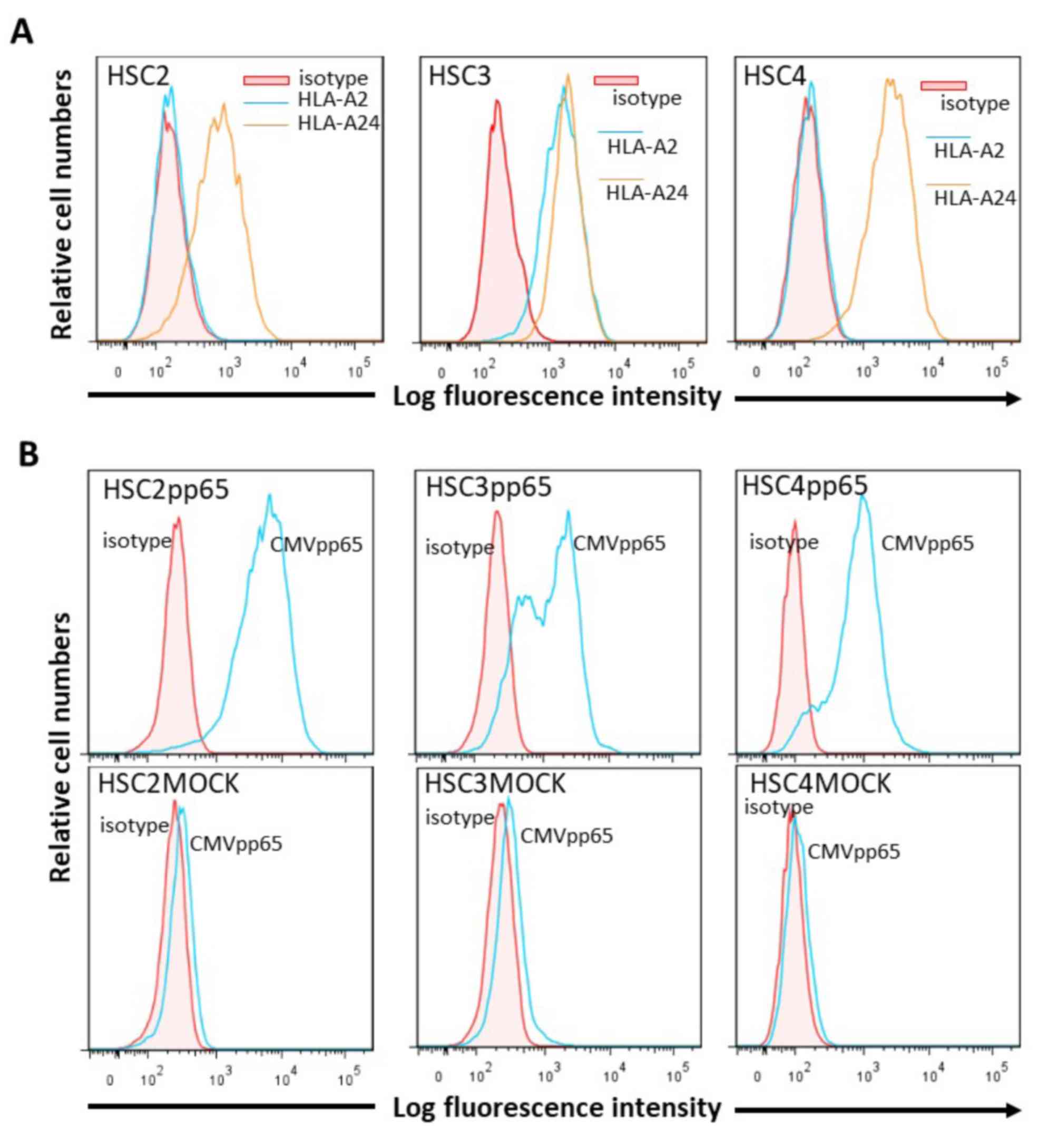
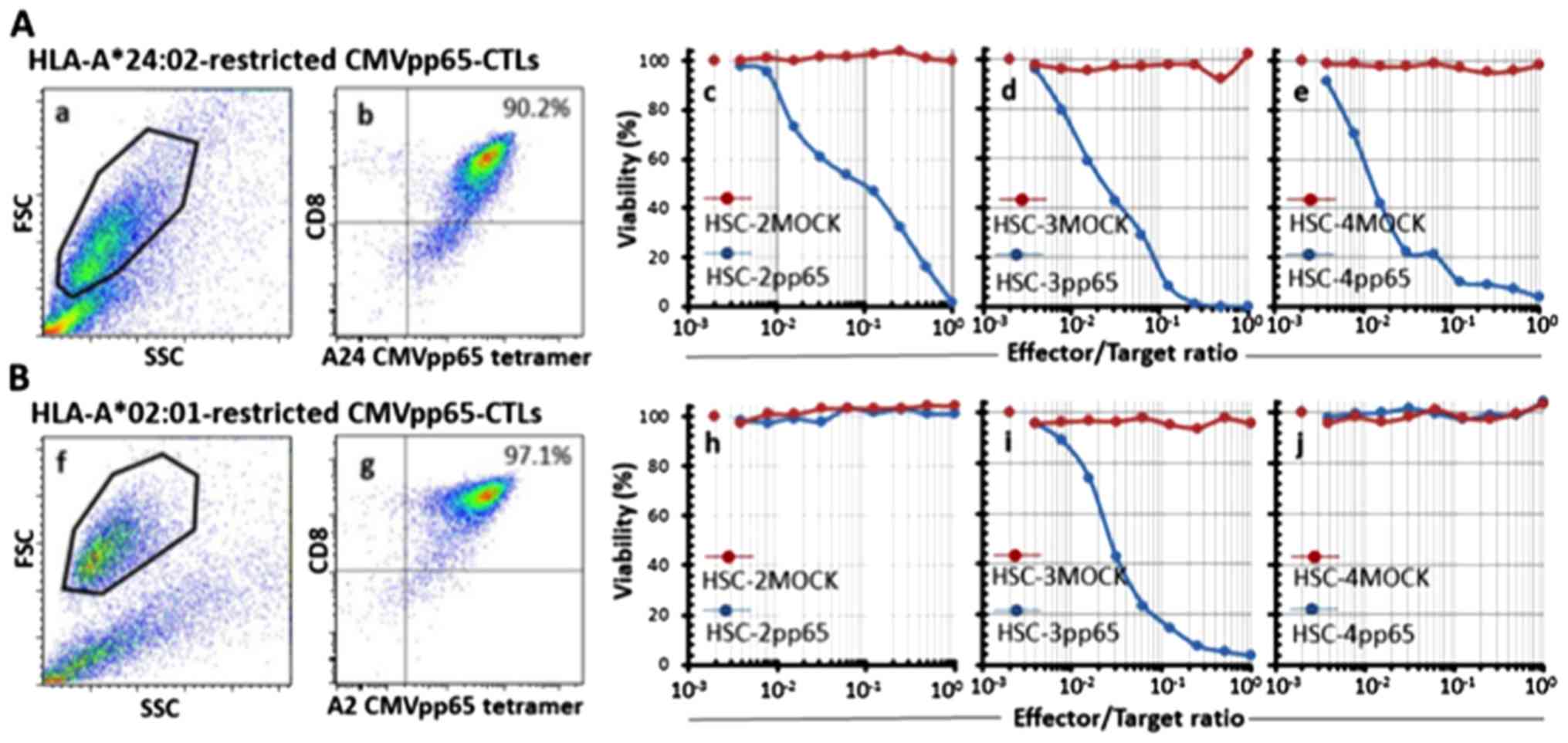
|
1
|
Chaturvedi AK, Anderson WF,
Lortet-Tieulent J, Curado MP, Ferlay J, Franceschi S, Rosenberg PS,
Bray F and Gillison ML: Worldwide trends in incidence rates for
oral cavity and oropharyngeal cancers. J Clin Oncol. 31:4550–4559.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Hori M, Matsuda T, Shibata A, Katanoda K,
Sobue T and Nishimoto H; Japan Cancer Surveillance Research Group:
Cancer incidence and incidence rates in Japan in 2009: A study of
32 population-based cancer registries for the Monitoring of Cancer
Incidence in Japan (MCIJ) project. Jpn J Clin Oncol. 45:884–891.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Gibson MK, Li Y, Murphy B, Hussain MH,
DeConti RC, Ensley J and Forastiere AA; Eastern Cooperative
Oncology Group: Randomized phase III evaluation of cisplatin plus
fluorouracil versus cisplatin plus paclitaxel in advanced head and
neck cancer (E1395): An intergroup trial of the Eastern Cooperative
Oncology Group. J Clin Oncol. 23:3562–3567. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Forastiere AA, Goepfert H, Maor M, Pajak
TF, Weber R, Morrison W, Glisson B, Trotti A, Ridge JA, Chao C, et
al: Concurrent chemotherapy and radiotherapy for organ preservation
in advanced laryngeal cancer. N Engl J Med. 349:2091–2098. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Adelstein DJ, Li Y, Adams GL, Wagner H Jr,
Kish JA, Ensley JF, Schuller DE and Forastiere AA: An intergroup
phase III comparison of standard radiation therapy and two
schedules of concurrent chemoradiotherapy in patients with
unresectable squamous cell head and neck cancer. J Clin Oncol.
21:92–98. 2003. View Article : Google Scholar
|
|
6
|
Cooper JS, Pajak TF, Forastiere AA, Jacobs
J, Campbell BH, Saxman SB, Kish JA, Kim HE, Cmelak AJ, Rotman M, et
al Radiation Therapy Oncology Group 9501/Intergroup: Postoperative
concurrent radiotherapy and chemotherapy for high-risk
squamous-cell carcinoma of the head and neck. N Engl J Med.
350:1937–1944. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Vermorken JB, Mesia R, Rivera F, Remenar
E, Kawecki A, Rottey S, Erfan J, Zabolotnyy D, Kienzer HR, Cupissol
D, et al: Platinum-based chemotherapy plus cetuximab in head and
neck cancer. N Engl J Med. 359:1116–1127. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Laramore GE, Scott CB, al-Sarraf M,
Haselow RE, Ervin TJ, Wheeler R, Jacobs JR, Schuller DE, Gahbauer
RA, Schwade JG, et al: Adjuvant chemotherapy for resectable
squamous cell carcinomas of the head and neck: Report on Intergroup
Study 0034. Int J Radiat Oncol Biol Phys. 23:705–713. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Bernier J, Domenge C, Ozsahin M,
Matuszewska K, Lefèbvre JL, Greiner RH, Giralt J, Maingon P,
Rolland F, Bolla M, et al European Organization for Research and
Treatment of Cancer Trial 22931: Postoperative irradiation with or
without concomitant chemotherapy for locally advanced head and neck
cancer. N Engl J Med. 350:1945–1952. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Brockstein B, Haraf DJ, Rademaker AW, Kies
MS, Stenson KM, Rosen F, Mittal BB, Pelzer H, Fung BB, Witt ME, et
al: Patterns of failure, prognostic factors and survival in
locoregionally advanced head and neck cancer treated with
concomitant chemoradiotherapy: A 9-year, 337-patient,
multi-institutional experience. Ann Oncol. 15:1179–1186. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
de Biasi AR, Villena-Vargas J and
Adusumilli PS: Cisplatin-induced antitumor immunomodulation: A
review of preclinical and clinical evidence. Clin Cancer Res.
20:5384–5391. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hato SV, Khong A, de Vries IJ and
Lesterhuis WJ: Molecular pathways: The immunogenic effects of
platinum-based chemotherapeutics. Clin Cancer Res. 20:2831–2837.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Apetoh L, Ladoire S, Coukos G and
Ghiringhelli F: Combining immunotherapy and anticancer agents: The
right path to achieve cancer cure? Ann Oncol. 26:1813–1823. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Alizadeh D and Larmonier N:
Chemotherapeutic targeting of cancer-induced immunosuppressive
cells. Cancer Res. 74:2663–2668. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Correale P, Aquino A, Giuliani A,
Pellegrini M, Micheli L, Cusi MG, Nencini C, Petrioli R, Prete SP,
De Vecchis L, et al: Treatment of colon and breast carcinoma cells
with 5-fluorouracil enhances expression of carcinoembryonic antigen
and susceptibility to HLA-A(*)02.01 restricted,
CEA-peptide-specific cytotoxic T cells in vitro. Int J Cancer.
104:437–445. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Correale P, Del Vecchio MT, Di Genova G,
Savellini GG, La Placa M, Terrosi C, Vestri M, Urso R, Lemonnier F,
Aquino A, et al: 5-fluorouracil-based chemotherapy enhances the
antitumor activity of a thymidylate synthase-directed polyepitopic
peptide vaccine. J Natl Cancer Inst. 97:1437–1445. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Bergmann-Leitner ES and Abrams SI:
Treatment of human colon carcinoma cell lines with anti-neoplastic
agents enhances their lytic sensitivity to antigen-specific
CD8+ cytotoxic T lymphocytes. Cancer Immunol Immunother.
50:445–455. 2001. View Article : Google Scholar
|
|
18
|
Yang S and Haluska FG: Treatment of
melanoma with 5-fluorouracil or dacarbazine in vitro sensitizes
cells to antigen-specific CTL lysis through perforin/granzyme- and
Fas-mediated pathways. J Immunol. 172:4599–4608. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Iwase M, Watanabe H, Kondo G, Ohashi M and
Nagumo M: Enhanced susceptibility of oral squamous cell carcinoma
cell lines to FAS-mediated apoptosis by cisplatin and
5-fluorouracil. Int J Cancer. 106:619–625. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Panaretakis T, Kepp O, Brockmeier U,
Tesniere A, Bjorklund AC, Chapman DC, Durchschlag M, Joza N,
Pierron G, van Endert P, et al: Mechanisms of pre-apoptotic
calreticulin exposure in immunogenic cell death. EMBO J.
28:578–590. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Elliott MR, Chekeni FB, Trampont PC,
Lazarowski ER, Kadl A, Walk SF, Park D, Woodson RI, Ostankovich M,
Sharma P, et al: Nucleotides released by apoptotic cells act as a
find-me signal to promote phagocytic clearance. Nature.
461:282–286. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Apetoh L, Ghiringhelli F, Tesniere A,
Obeid M, Ortiz C, Criollo A, Mignot G, Maiuri MC, Ullrich E,
Saulnier P, et al: Toll-like receptor 4-dependent contribution of
the immune system to anticancer chemotherapy and radiotherapy. Nat
Med. 13:1050–1059. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ramakrishnan R, Assudani D, Nagaraj S,
Hunter T, Cho HI, Antonia S, Altiok S, Celis E and Gabrilovich DI:
Chemotherapy enhances tumor cell susceptibility to CTL-mediated
killing during cancer immunotherapy in mice. J Clin Invest.
120:1111–1124. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Suzuki S, Ishida T, Yoshikawa K and Ueda
R: Current status of immunotherapy. Jpn J Clin Oncol. 46:191–203.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Ohtani T, Yamada Y, Furuhashi A, Ohmura Y,
Nakamura S, Kato H, Yoshikawa K and Kazaoka Y: Activated cytotoxic
T-lymphocyte immunotherapy is effective for advanced oral and
maxillofacial cancers. Int J Oncol. 45:2051–2057. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Mescher MF: Molecular interactions in the
activation of effector and precursor cytotoxic T lymphocytes.
Immunol Rev. 146:177–210. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Halle S, Halle O and Förster R: Mechanisms
and dynamics of T cell-mediated cytotoxicity in vivo. Trends
Immunol. 38:432–443. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Klenerman P and Oxenius A: T cell
responses to cytomegalovirus. Nat Rev Immunol. 16:367–377. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Watanabe K, Suzuki S, Kamei M, Toji S,
Kawase T, Takahashi T, Kuzushima K and Akatsuka Y: CD137-guided
isolation and expansion of antigen-specific CD8 cells for potential
use in adoptive immunotherapy. Int J Hematol. 88:311–320. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Ishiyama M, Miyazono Y, Sasamoto K, Ohkura
Y and Ueno K: A highly water-soluble disulfonated tetrazolium salt
as a chromogenic indicator for NADH as well as cell viability.
Talanta. 44:1299–1305. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kuzushima K, Hayashi N, Kimura H and
Tsurumi T: Efficient identification of HLA-A*2402-restricted
cytomegalovirus-specific CD8(+) T-cell epitopes by a computer
algorithm and an enzyme-linked immunospot assay. Blood.
98:1872–1881. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wills MR, Carmichael AJ, Mynard K, Jin X,
Weekes MP, Plachter B and Sissons JG: The human cytotoxic
T-lymphocyte (CTL) response to cytomegalovirus is dominated by
structural protein pp65: Frequency, specificity, and T-cell
receptor usage of pp65-specific CTL. J Virol. 70:7569–7579.
1996.PubMed/NCBI
|
|
33
|
Dasari S and Tchounwou PB: Cisplatin in
cancer therapy: Molecular mechanisms of action. Eur J Pharmacol.
740:364–378. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Galandrini R, Capuano C and Santoni A:
Activation of lymphocyte cytolytic machinery: Where are we? Front
Immunol. 4:3902013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Shen DW, Pouliot LM, Hall MD and Gottesman
MM: Cisplatin resistance: A cellular self-defense mechanism
resulting from multiple epigenetic and genetic changes. Pharmacol
Rev. 64:706–721. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Geary SM, Lemke CD, Lubaroff DM and Salem
AK: The combination of a low-dose chemotherapeutic agent,
5-fluorouracil, and an adenoviral tumor vaccine has a synergistic
benefit on survival in a tumor model system. PLoS One.
8:e679042013. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Robert C, Thomas L, Bondarenko I, O'Day S,
Weber J, Garbe C, Lebbe C, Baurain JF, Testori A, Grob JJ, et al:
Ipilimumab plus dacarbazine for previously untreated metastatic
melanoma. N Engl J Med. 364:2517–2526. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Hattori T, Mine T, Komatsu N, Yamada A,
Itoh K, Shiozaki H and Okuno K: Immunological evaluation of
personalized peptide vaccination in combination with UFT and UZEL
for metastatic colorectal carcinoma patients. Cancer Immunol
Immunother. 58:1843–1852. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Sato Y, Fujiwara T, Mine T, Shomura H,
Homma S, Maeda Y, Tokunaga N, Ikeda Y, Ishihara Y, Yamada A, et al:
Immunological evaluation of personalized peptide vaccination in
combination with a 5-fluorouracil derivative (TS-1) for advanced
gastric or colorectal carcinoma patients. Cancer Sci. 98:1113–1119.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Masuzawa T, Fujiwara Y, Okada K, Nakamura
A, Takiguchi S, Nakajima K, Miyata H, Yamasaki M, Kurokawa Y, Osawa
R, et al: Phase I/II study of S-1 plus cisplatin combined with
peptide vaccines for human vascular endothelial growth factor
receptor 1 and 2 in patients with advanced gastric cancer. Int J
Oncol. 41:1297–1304. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Iinuma H, Fukushima R, Inaba T, Tamura J,
Inoue T, Ogawa E, Horikawa M, Ikeda Y, Matsutani N, Takeda K, et
al: Phase I clinical study of multiple epitope peptide vaccine
combined with chemoradiation therapy in esophageal cancer patients.
J Transl Med. 12:842014. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Borghaei H, Paz-Ares L, Horn L, Spigel DR,
Steins M, Ready NE, Chow LQ, Vokes EE, Felip E, Holgado E, et al:
Nivolumab versus docetaxel in advanced nonsquamous non-small-cell
lung cancer. N Engl J Med. 373:1627–1639. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Motzer RJ, Escudier B, McDermott DF,
George S, Hammers HJ, Srinivas S, Tykodi SS, Sosman JA, Procopio G,
Plimack ER, et al: CheckMate 025 Investigators: Nivolumab versus
everolimus in advanced renal-cell carcinoma. N Engl J Med.
373:1803–1813. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Sugiyama D, Nishikawa H, Maeda Y, Nishioka
M, Tanemura A, Katayama I, Ezoe S, Kanakura Y, Sato E, Fukumori Y,
et al: Anti-CCR4 mAb selectively depletes effector-type
FoxP3+CD4+ regulatory T cells, evoking
antitumor immune responses in humans. Proc Natl Acad Sci USA.
110:17945–17950. 2013. View Article : Google Scholar
|
|
45
|
Kurose K, Ohue Y, Wada H, Iida S, Ishida
T, Kojima T, Doi T, Suzuki S, Isobe M, Funakoshi T, et al: Phase Ia
Study of FoxP3+ CD4 Treg depletion by infusion of a
humanized anti-CCR4 antibody, KW-0761, in cancer patients. Clin
Cancer Res. 21:4327–4336. 2015. View Article : Google Scholar : PubMed/NCBI
|