Introduction
The nuclear lamins were classified as type V
intermediate filament proteins that are critically important for
the structure of the nucleus and are located between the inner
nuclear membrane and peripheral heterochromatin (1). Based on sequence homologies, as well
as biochemical properties, lamins were subdivided into types: A and
B. In mammals, lamin A and C have been rated into A-type but lamin
B1 and B2 have been classified as B-type lamins (2). Lamins, together with lamin-binding
proteins, make up the nuclear matrix and are involved in many
different functions, including nuclear stability, transcription,
DNA replications and genome repair. Interactions of lamins with
chromatin regulate gene expression, responsible for all such
cellular processes as proliferation, differentiation and
carcinogenesis (3,4).
The role of lamins in cancer development and
progression is still unclear. However, overexpression of LMNB1 was
shown in prostate, liver and pancreatic cancers, whereas
downregulation in gastric and in several subtypes of lung cancer
(2,3,5).
Reduced expression of LMNB1 has been also observed in colon cancer,
which is the most common group of malignant tumors throughout the
world (4). Application of
cytostatics plays an important role in colon cancer therapy, but
different sensitivity to chemotherapeutics and development of drug
resistance is frequent thus encouraging scientists to look for new
therapeutic goal (6). Except for
intermediate filaments (lamin B1), other elements of cytoskeleton
could play an important role in the limiting of cancer cell
proliferation.
Recently, the existence of actin in the nucleus has
been accepted and it was shown as an additional element of multiple
nuclear complexes which can actively enter and exit the nuclei
(7,8). Nuclear actin is involved in chromatin
remodeling, transcription and RNA splicing. Furthermore, together
with actin-binding proteins, it forms the functional bridge between
nuclear components and cytoplasm. A hypothesis states that one
interconnected network connect the elements of the nucleus via the
nuclear membrane to the cytoskeleton, cell adhesion molecules and
the extracellular matrix (9). The
recent identification of Actin-Binding site 1 (AB-1) within lamins
A and B prompted us to test whether disability in the structure of
nuclear lamina may have an impact on tumor cell adhesion and
migration. One of the proteins participating in all these processes
is β-catenin, which abnormally high expression in colorectal cancer
has been detected. There is a multifunctional protein which can be
located in cell membrane and function as a structural element of
cadherin/catenin complex to modulate cell-cell adhesion and
migration or detected in nucleus acts as an effector of the Wnt
signaling pathway (10-12). It is known that actin is linked
with cadherin/β-catenin by α-catenin. Additionally, lamin A and B
bind directly to actin and thus it is assumed that the expression
of LMNB1 has an influence on adhesion and migration of colon cancer
cells.
The aim of the present study is to show dependence
between the expression of lamin B1 and β-catenin as well as the
influence of LMNB1 overexpression on cytoskeleton (F-actin, tubulin
and lamin A/C) organization, cell death, cell cycle and migration
of LoVo cells after treatment with 5-FU. Here, we show that
upregulation of LMNB1 expression induced dose-dependent cell death
mainly by mitotic catastrophe. Additionally, overexpression of this
nuclear protein caused the inhibition of colon cancer cells
migration which is connected with lower expression of lamin A/C and
changes in expression of actin and β-catenin.
Materials and methods
Cell culture and treatment
LoVo, a human colon cancer cell line was purchased
from the American Type Culture Collection (CCL-229; ATCC, Manassas,
VA, USA). The cells were cultured in tissue culture flasks or
12-well plates (BD Biosciences, Franklin Lakes, NJ, USA) and grown
as a monolayer at 37°C in a humidified CO2 incubator (5%
CO2) in Dulbecco's modified eagle's medium (DMEM; Lonza,
Verviers, Belgium). The medium was supplemented with 10% fetal
bovine serum (FBS; Sigma-Aldrich, St. Louis, MO, USA) and 50
μg/ml gentamycin (Sigma-Aldrich). LoVo cells were treated
for 24 h with 0.01, 0.05, 0.1, 1, 2.5, 5 and 10 mM concentration of
5-fluorouracil (5-FU; Sigma-Aldrich) for MTT assay and 0.1, 1 and 5
mM for other experiments. The control cells were grown under the
same conditions but without the addition of cytostatic. The LoVo
culture was tested for mycoplasma based on rapid uptake of the DAPI
(Sigma-Aldrich) by cellular DNA. The tests were negative. All in
vitro studies were performed on less than 5 passage cells.
MTT assay
To determine the effect of 5-FU on cell viability
the colorimetric MTT metabolic activity assay was done. The cells
were cultured in 12-well plates and 24 h later were treated with
5-FU at 0.01, 0.05, 0.1, 1, 2.5, 5 and 10 mM doses for another 24
h. The stock solution was prepared by dissolving thiazolyl blue
tetrazolium bromide (MTT; Sigma-Aldrich) in 5 mg/ml
phosphate-buffered saline (PBS). After the cytostatic treatment,
the cells were washed with PBS and incubated for 3 h with MTT
solution which was mixed with medium without phenol red (Lonza) in
the ratio 1:9. The absorbance was measured at 570 nm using
spectrophotometer (Spectra Academy; K-MAC, Daejeon, Korea).
Cell transfection by nucleofection
For the nucleofection, the LoVo cells were cultured
to 80–90% confluency in DMEM with FBS and gentamycin. After
trypsinization, the cells were transfected with using the SE Cell
Line 4D-Nucleofector™ X kit and 2 μg human the cDNA of LMNB1
cloned into pCMV6-XL4 expression plasmid vector (NM_005573;
OriGene, Rockville, MD, USA) according to the manufacturer's
instructions. For determining the unspecific effect of the
upregulation of LMNB1 overexpression, the cells were transfected
with pCMV6-XL4 control plasmid vector (OriGene). After transfection
and growth in the medium for 72 h, the cells were used for further
experiments. The efficiency of transfection was confirmed by
western blot analysis.
Western blot analysis
Semi-quantitative analysis of post-translational
expression of lamin B1 was performed by using western blot
analysis. After transfection, the LoVo cells were lysed with RIPA
buffer (Sigma-Aldrich). Next, BCA protein assay kit (Thermo Fisher
Scientific, Pierce Rockford, IL, USA) to the normalization of
protein concentration was used and 15 μg of total protein
per lane was separated by 4–12% NuPAGE Bis-Tris gel (Novex/Life
Technologies, Carlsbad, CA, USA). Proteins were transferred from
the gel onto the nitrocellulose membrane using iBlot dry western
blotting system (Invitrogen/Life Technologies). Estimation of
protein molecular mass, pre-stained molecular weight marker was
used (Thermo Fisher Scientific). Then, the membrane was processed
using WesternBreeze Chromogenic Western Blot Immunodetection kit
(Invitrogen/Life Technologies) by BenchPro 4100 card processing
station (Invitrogen/Life Technologies) according to the
manufacturer's instructions. First, the membranes were incubated
with primary mouse monoclonal anti-lamin B1 (Invitrogen/Thermo
Fisher Scientific) or mouse monoclonal anti-GADPH (Sigma-Aldrich)
antibodies for 2 h at room temperature (RT). Next, the membranes
were washed and incubated with a ready-to-use solution of alkaline
phosphatase-conjugated anti-species IgG for 1 h in RT. The protein
bands were visualized using a ready-to-use solution of BCIP/NBT
substrate for alkaline phosphatase. The last step of this method
was scanning of membrane and densitometry analysis of the bands
using the Quantity One Basic software (ver. 3.6.5; Bio-Rad
Laboratories, Hercules, CA, USA).
Cell death analysis
The procedure was performed according to the
manufacturer's protocol by using Tali™ apoptosis assay kit/Annexin
V Alexa Fluor® 488 and propidium iodide (PI)
(Invitrogen/Life Technologies). After trypsinization, cells were
centrifuged and resuspended in 100 μl Annexin binding
buffer. Next, to each 100 μl of sample, 5 μl of
Annexin V Alexa Fluor 488 was added. After 20 min of incubation in
the dark, the cells were centrifuged, resuspended in Annexin
binding buffer. Subsequently, 1 μl of PI to each sample was
added and the cells were incubated at room temperature in the dark
for 3 min. The data were analyzed using the FCs express Research
Edition Software (ver4.03; De Novo Software, Los Angeles, CA, USA).
The results revealed that the viable cells were Annexin V Alexa
Fluor 488 and PI-negative; apoptotic cells were Annexin V Alexa
Fluor 488-positive and PI-negative and Annexin V Alexa Fluor 488
and PI-positive; whereas necrotic cells were Annexin V Alexa Fluor
488-negative and PI-positive.
Cell cycle analysis
For cell cycle analysis, the Tali Cell Cycle kit
(Invitrogen/Life Technologies) was used according to the
manufacturer's instructions. First, the treated cells were fixed in
ice-cold 70% ethanol at 4°C and left at −25°C overnight. After the
washing and centrifugation, the cells were resuspended in the Tali
Cell Cycle Solution and incubated in the dark for 30 min. The data
were determined using Tali image-based cytometer (Invitrogen/Life
Technologies) and the percentage of cells in each phase of the cell
cycle was designated using the FCS Express Research Edition
software (version 4.03; De Novo Software).
Fluorescent staining
The LoVo cells with and without overexpression of
LMNB1 after treatment with 0.1, 1 and 5 mM 5-FU were grown on glass
coverslips. For β-tubulin labeling, after the 24 h incubation with
cytostatic, the LoVo cells were prefixed for 10 min with
bifunctional protein crosslinking reagent DTSP (1 mM
3,30-dithiodipropionic acid; Sigma-Aldrich) which was diluted 1:50
in Hanks' balanced salt solution (HBSS; Sigma-Aldrich). Next, the
cells were pre-extracted with TSB [(0.5% Triton X-100 (Serva
Electrophoresis GmbH, Heidelberg, Germany) in MTSB with the
addition of DTSP (dilution 1:50) (microtubule stabilizing buffer: 1
mM EGTA, 10 mM PIPES, 4% poly(ethylene glycol); Sigma-Aldrich) for
10 min and rinsed with TSB (5 min)]. Then, the cells were fixed
with 4% paraformaldehyde (Serva Electrophoresis GmbH) in MTSB for
15 min, washed with PBS (3×5 min) and incubated with 1% bovine
serum albumin (BSA; Sigma-Aldrich) diluted in TBS (Tris-buffered
saline) for 15 min. Tubulin was labeled using a mouse monoclonal
antibody against β-tubulin (Sigma-Aldrich) and goat anti-mouse
antibody TRITC (Sigma-Aldrich).
In the case of other immunofluorescence reactions,
the cells were fixed with 4% paraformaldehyde in PBs, pH 7.4 (15
min, room temperature), incubated with 0 25% Triton X-100 (Serva
Electrophoresis GmbH) for 5 min than blocked in 1% (w/v) BSA/PBS
and double stained for proteins and F-actin, using antibodies and
phalloidin conjugates in the following arrangement: i) rabbit
anti-β-catenin (Sigma-Aldrich), anti-rabbit antibody-Alexa Fluor
488 (Invitrogen/Life Technologies), phalloidin-TRITC
(Sigma-Aldrich); ii) mouse anti-lamin A/C (Sigma-Aldrich),
anti-mouse TRITC (Sigma-Aldrich), Alexa Fluore-488 phalloidin
(Invitrogen/Life Technologies); and iii) mouse anti-lamin B1
(Invitrogen/Life Technologie), anti-mouse TRITC
(Sigma-Aldrich).
Cell nuclei were stained with DAPI (Sigma-Aldrich).
The slides were mounted in Aqua-Poly/Mount (Polysciences,
Warrington, PA, USA) and examined using C1 laser-scanning confocal
microscope system (Nikon, Tokyo, Japan) or Nikon Eclipse E800
fluorescence microscope and NIS-Elements 4.0 software (Nikon).
The measurement of fluorescence intensity of lamin
B1 in LoVo cells with and without overexpression of LMNB1 was
performed on confocal images. The fluorescence intensity
measurement was executed using the ImageJ software (Ver.1.51j8,
National Institute of Health, Bethesda, MD, USA).
The measurement of fluorescence intensity of
junctional protein (β-catenin) in LoVo cells with and without
overexpression of LMNB1 after treatment with 0.1, 1 and 5 mM 5-FU
was performed on confocal images acquired at the brightest signals
at cell-cell interaction areas. The fluorescence intensity
measurement of nuclear F-actin and β-catenin was executed using
Nikon EZ-C1 software (Ver 3.90, Gold; Nikon).
Wound healing assay
The LoVo cells with and without overexpression of
LMNB1 after treatment with 0.1, 1 and 5 mM 5-FU were grown in
6-well plates. The cell monolayer was then scratched using a
sterile pipette tip. For imaging of cell migration during wound
healing assay live cell imaging microscope (Carl Zeiss, Oberkochen,
Germany) was used.
Analysis of publicly available
datasets
To analyze LMNB1 mRNA expression in colorectal
adenocarcinoma, we obtain data from TCGA data Portal Open-Access
directory via the cBio Portal (13). Overall survival analysis was then
performed using the GraphPad Prism 6 (GraphPad Software, Inc., La
Jolla, CA, USA).
Statistical analysis
The data are shown as mean ± SEM. A two-way ANOVA
analysis was performed for wound healing, fluorescence intensity,
cell death and cell cycle data between cells transfected with
control vector and plasmid with cloned cDNA of LMNB1. A
Kruskal-Wallis test was used to evaluate the differences in mean
values between cells treated with cytostatic in comparison to the
control in a group of cells with normal expression of LMNB1 and
transfected with plasmid with cloned cDNA of LMNB1 independently. A
one sample t-test compares the mean with a hypothetical value, thus
it was used for the analysis of MTT data. The differences between
the groups were considered significant at P<0.05. GraphPad Prism
6.0 software was used for statistical analyses. Statistically
significant differences between cells treated with cytostatic in
comparison to the control in a group of cells with normal
expression of LMNB1 are marked by *. In turn, statistically
significant differences within cells with overexpression of LMNB1
are presented in the figures as # and the differences between the
cells transfected with control vector and plasmid with cloned cDNA
of LMNB1 are shown with $ symbol.
Results
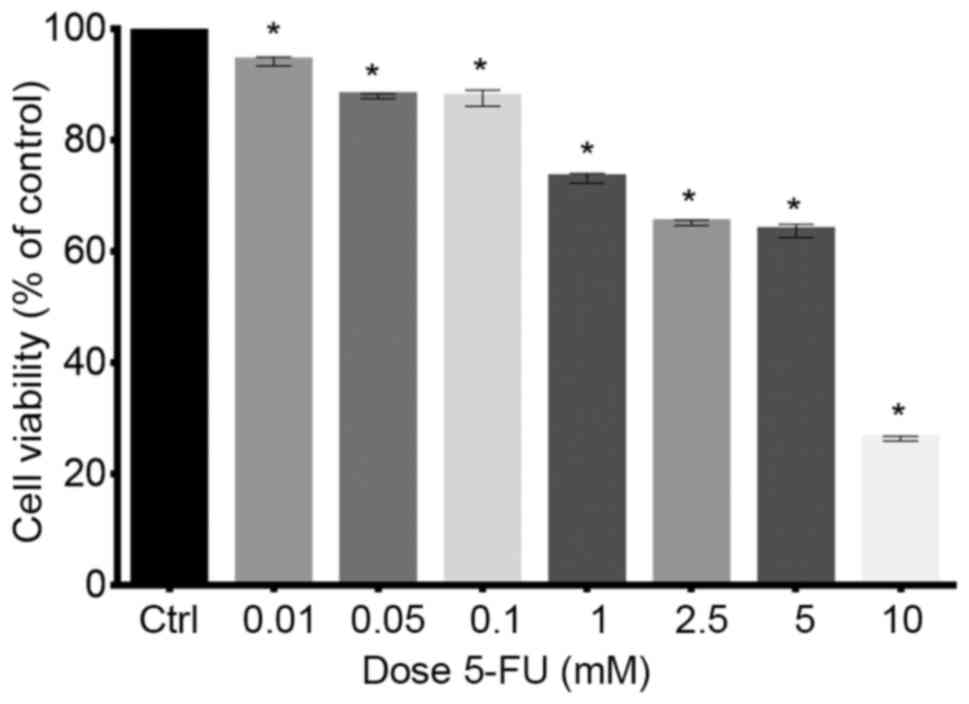
The effect of 5-FU on the viability of
LoVo cells
MTT analysis was performed to select appropriate
doses of the cytostatic drug. This experiment indicated that 5-FU
exhibited a cytostatic effect on the growth of LoVo cell line and
cell viability was decreased in a dose-dependent manner (Fig. 1). Twenty-four hours of treatment of
cells with 0.01, 0.05 and 0.1 mM doses of 5-FU decreased viability
to 94.06, 87.8 and 87.5%, respectively. Following treatment of LoVo
cells with the higher doses (1, 2.5, 5 and 10 mM) survival rate was
even lower and amounted to 73.1, 65.1, 63.6 and 26.3%,
respectively. The results were statistically significant in
comparison to the control (P<0.05) (Fig. 1). On the basis of results obtained
from MTT analysis, for further experiments, 5-FU was used in
concentrations of 0.1, 1 and 5 mM.
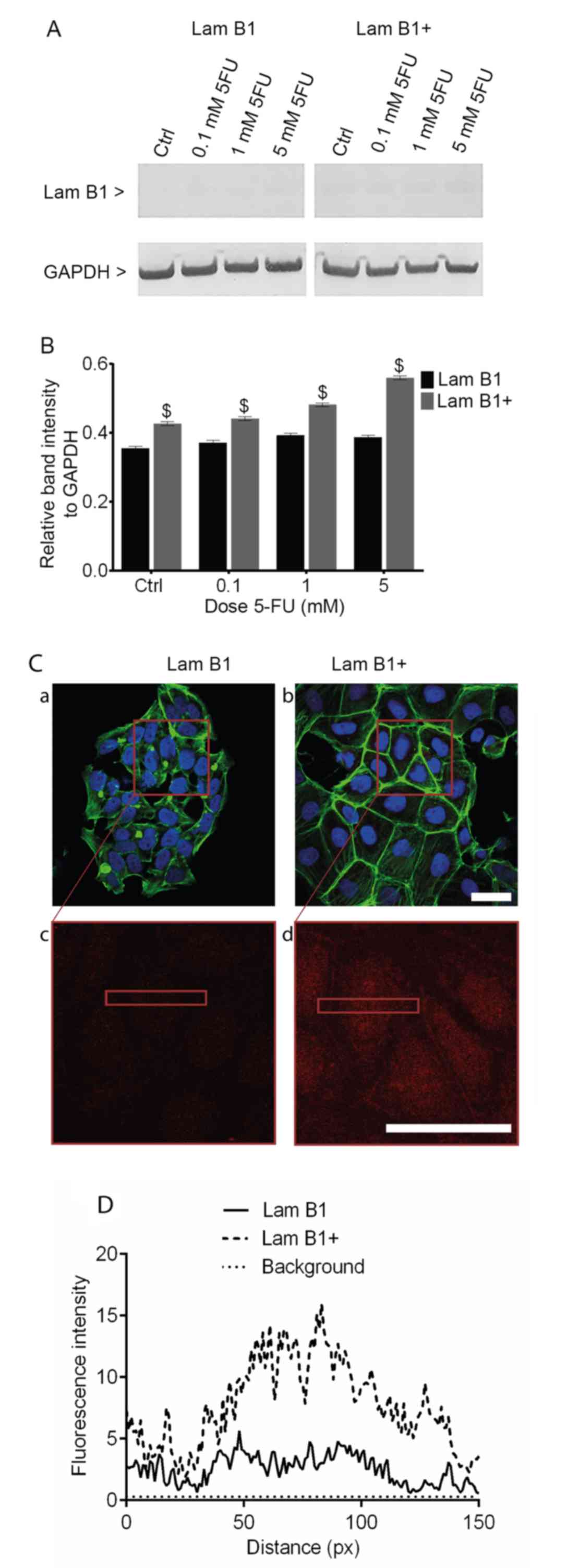
Western blot analysis of LMNB1
expression
The upregulation of LMNB1 expression in LoVo cells
transfected with pCMV6-XL4 expression plasmid vector with cloned
cDNA of LMNB1 was examined by using the western blotting method
(Fig. 2A). The densitometric
analysis confirmed an increase in the post-translational expression
of LMNB1 in the control and cells exposed to 0.1, 1 and 5 mM 5-FU,
as compared to cells transfected with control vector (Fig. 2B). Analysis of the relative to
GADPH band intensity showed 1.17-, 1.18-, 1.21- and 1.41-fold
increase in the post-translational expression of LMNB1 in the
control and cells treated with 5-FU at doses 0.1, 1 and 5 mM,
respectively (Fig. 2B).
Additionally, Fig. 2C shows an
increase of lamin B1 fluorescence intensity in cells transfected
with pCMV6-XL4 expression plasmid vector with cloned cDNA of LMNB1
(Fig. 2C).
The effect of LMNB1 upregulation on cell
death
The type of 5-FU-induced cell death was analyzed by
dual staining with Annexin V and PI. In cells without upregulated
expression of LMNB1, statistically significant differences in the
percentage of live cells were observed following treatment with 1
and 5 mM 5-FU, as compared to the untreated control. The percentage
of live cells in control cells and after treatment with 0.1 mM 5-FU
was similar and amounted to 92.9 and 93%, respectively. However,
when the 1 and 5 mM doses were used, the fraction of live cells was
significantly decreased in comparison to the control to 84 and
78.8% (Fig. 3A). After
upregulation of LMNB1 statistically significant decrease in the
percentage of live cells was observed following their treatment
with 5-FU. The mean values for live cells after 0.1, 1 and 5 mM
5-FU treatment were 76.4, 76.2 and 71.5%, respectively (Fig. 3B). A statistically significant
decrease was also observed in the mean percentage of live cells
between transfected cells with and without altered expression of
LMNB1 after treatment with the same doses of 5-FU (Fig. 3C). In the LoVo cells without
upregulated LMNB1 expression, a statistically significant increase
from 4.5 to 9.8 and to 10.8%, in the percentage of apoptotic cells
was observed following treatment with 1 and 5 mM 5-FU, respectively
(Fig. 3D). Furthermore, much
higher percentage of apoptotic cells was observed in cells with
upregulated LMNB1 expression (Fig.
3E). As shown in Fig. 3E, the
population of apoptotic cells increased from 6.7 to 12, 13.1 and to
24.2% in the control and after treatment with 0.1, 1 and 5 mM 5-FU,
respectively. Moreover, statistically significant differences were
noted between cells with and without elevated expression of LMNB1
after incubation with 0.1 and 5 mM 5-FU (Fig. 3F). Moreover, a statistically
significant increase in the percentage of necrotic cells was
observed in cells without overexpressed LMNB1 after treatment with
1 and 5 mM (to 6.2 and 10.3%, respectively) (Fig. 3G). Statistically significant
increase in the percentage of necrosis was observed only after
treatment of LoVo cells with upregulated expression of LMNB1 with 1
mM 5-FU (Fig. 3H). Comparison
between the cells with normal and overexpressed expression of LMNB1
showed a statistically significant difference in the percentage of
necrotic cells only after their treatment with 5 mM 5-FU (Fig. 3I).
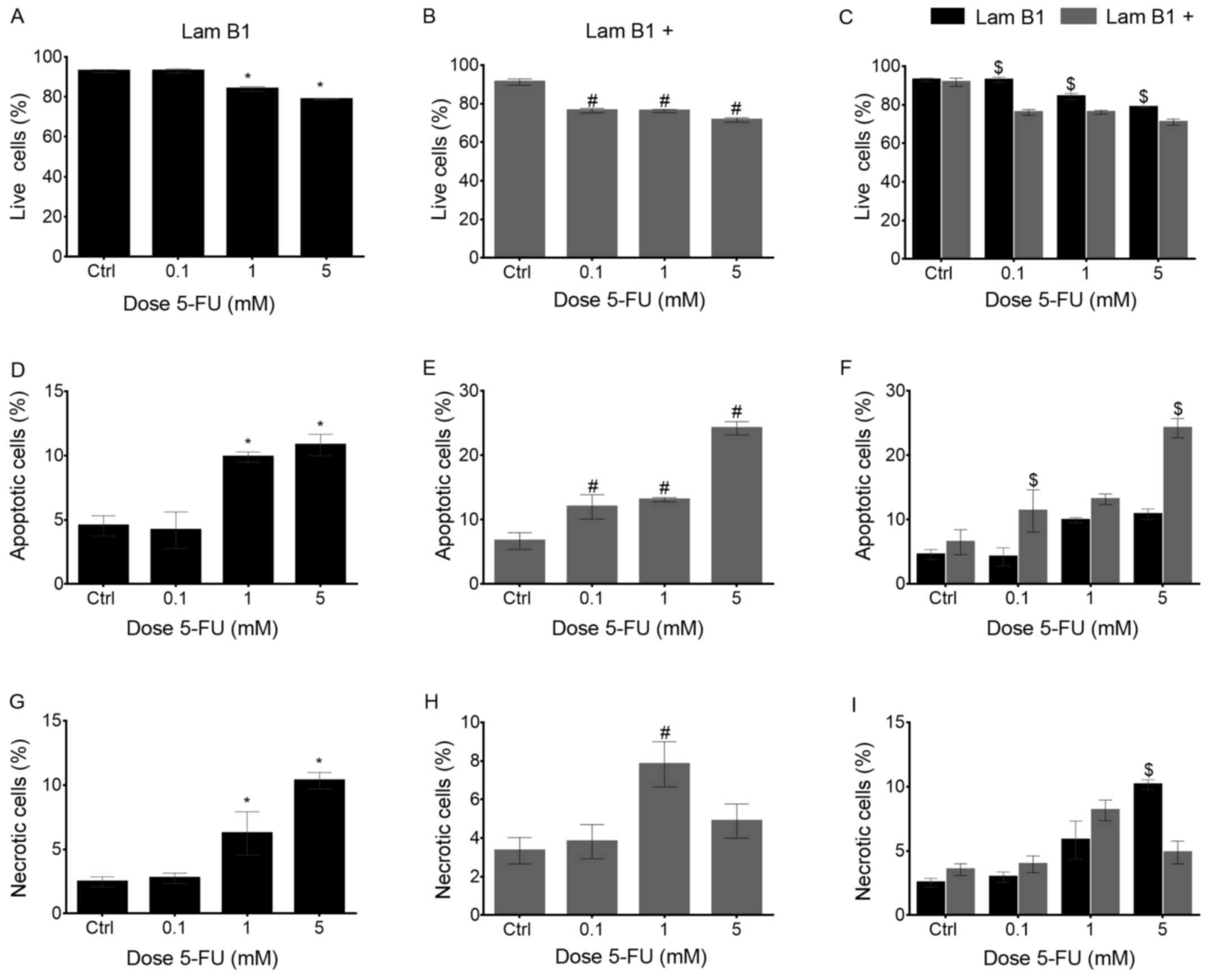 | Figure 3The quantitative analysis of viable,
apoptosis and necrosis in LoVo cells with and without
overexpression of LMNB1 after treatment with 5-FU. (A) Percentage
of viable cells without upregulated LMNB1 in control cells and
treated with 0.1, 1 and 5 mM 5-FU. (B) Percentage of viable cells
with upregulated LMNB1 in control cells and treated with 0.1, 1 and
5 mM 5-FU. (C) Comparison of percentage of viable cells without
upregulated LMNB1 and with upregulated LMNB1 in control cells and
treated with 0.1, 1 and 5 mM 5-FU. (D) Percentage of apoptotic
cells without upregulated LMNB1 in control cells and treated with
0.1, 1 and 5 mM 5-FU. (E) Percentage of apoptotic cells with
upregulated LMNB1 in control cells and treated with 0.1, 1 and 5 mM
5-FU. (F) Comparison of percentage of apoptotic cells without
upregulated LMNB1 and with upregulated LMNB1 in control cells and
treated with 0.1, 1 and 5 mM 5-FU. (G) Percentage of necrotic cells
without upregulated LMNB1 in control cells and treated with 0.1, 1
and 5 mM 5-FU. (H) Percentage of necrotic cells with upregulated
LMNB1 in control cells and treated with 0.1, 1 and 5 mM 5-FU. (I)
Comparison of percentage of necrotic cells without upregulated
LMNB1 and with upregulated LMNB1 in control cells and treated with
0.1, 1 and 5 mM 5-FU. *Statistically significant
differences between cells treated with 5-FU in comparison to the
control in a group of the cell without upregulated LMNB1,
P<0.05. #Statistically significant differences within
cells with upregulated LMNB1, P<0.05. $Statistically
significant differences between cells without upregulated LMNB and
with upregulated LMNB1, P<0.05. |
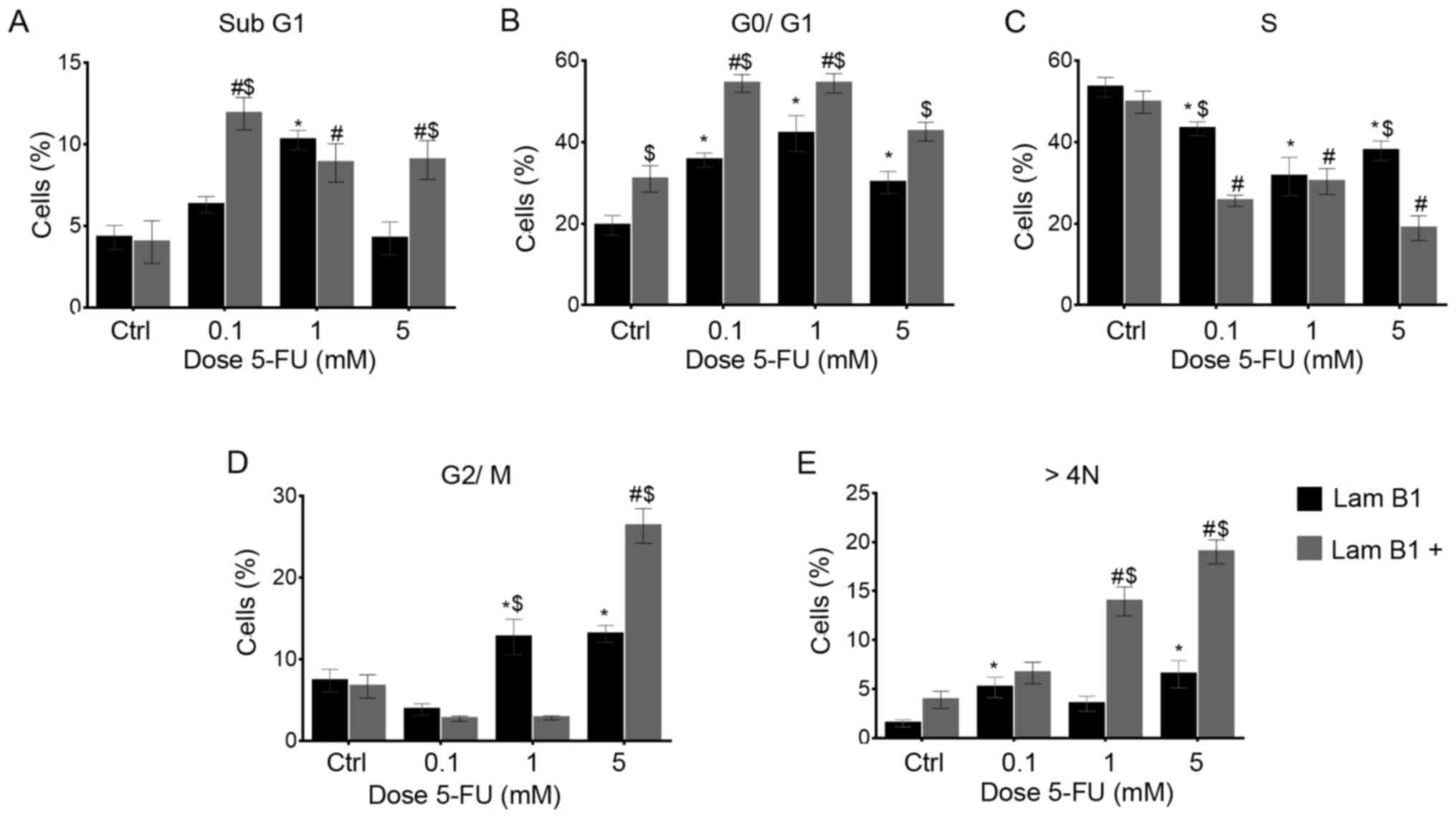
The effect of LMNB1 upregulation on cell
cycle distribution
The cell cycle was analyzed using image-based
cytometer. In the LoVo cells with normal expression of LMNB1, a
statistically significant increase from 19.6 to 35.5 and to 42.1%
in the mean percentage of cells with DNA content corresponding to
G0/G1 phase was observed following treatment with 0.1 and 1 mM
5-FU, respectively (Fig. 4B). The
population of cells in S-phase was 53.4, 43.2, 31.5 and 38.7% in
the control and cells treated with 0.1, 1 and 5 mM doses of 5-FU,
respectively (Fig. 4C). After
treatment with 1 and 5 mM 5-FU, a statistically significant
increase in the number of cells in the G2/M phase was noted
(Fig. 4D). Moreover, there was a
significant increase in the percentage of polyploid cells (from 1.5
in the control to 5.1 and 6.5% after treatment with 0.1 and 5 mM
5-FU, respectively) (Fig. 4E).
Additionally, an increase the percentage of the cell with sub-G1
DNA was noted in cells treated with 0.1 and 1 mM of 5-FU (from 4.2
to 6.5 and 10.3%, respectively) (Fig.
4A). The upregulation of LMNB1 resulted in a statistically
significant increase of cells in the G0/G1-phase after treatment
with 0.1 and 1 mM 5-FU (from 30.9 to 54.3 and 54.4%, respectively)
(Fig. 4B). However, a
statistically significant decrease in S-phase was observed (from
49.7 in the control to 25.6, 30.3 and 18.9% after treatment with
0.1, 1 and 5 mM 5-FU, respectively) (Fig. 4C). Moreover, 1 and 5 mM of 5-FU
induced an increase of polyploidy (Fig. 4E). The increased sub-G1 phase of
cell cycle was observed after treatment with each dose of 5-FU
(Fig. 4A). Furthermore, in control
cells and after 5-FU significant results between cells with and
without upregulated expression of LMNB1 were seen (Fig. 4). Specifically, a statistically
significant increase of sub-G1 was noted after treatment of cells
with 0.1 and 5 mM of 5-FU (Fig.
4A). Differently, reversed results were observed for S-phase
cells (Fig. 4C). Statistically
significant differences were also observed in populations of cells
in G0/G1 (Fig. 4B) and with DNA
content >4N (Fig. 4E).
Fluorescence analysis of proteins
The cytoskeleton is a very important cellular
compartment which undergoes reorganization during the many cell
processes. Simon et al (14) showed that tail domains of type A
and B can directly bind to actin filaments. Additionally, these
authors suggested that particularly lamin A might influence on the
nuclear actin concentration and thereby impact transcription,
chromatin remodeling and actin polymerizable. Furthermore, the
normal shape of nucleus and regularity of nuclear processes are
dependent on interactions between microtubules and lamin networks
(15). Although, lamins are the
nuclear proteins but play also an important role in cell migration
by the link with β-catenin, which undergo nucleocytoplasmic
distribution and participate in both Wnt signaling pathway and
cell-cell adhesion (16). It
turned out also, that lamins may be involved in cell movement
through actin filaments and linker of nucleoskeleton and
cytoskeleton complex (17).
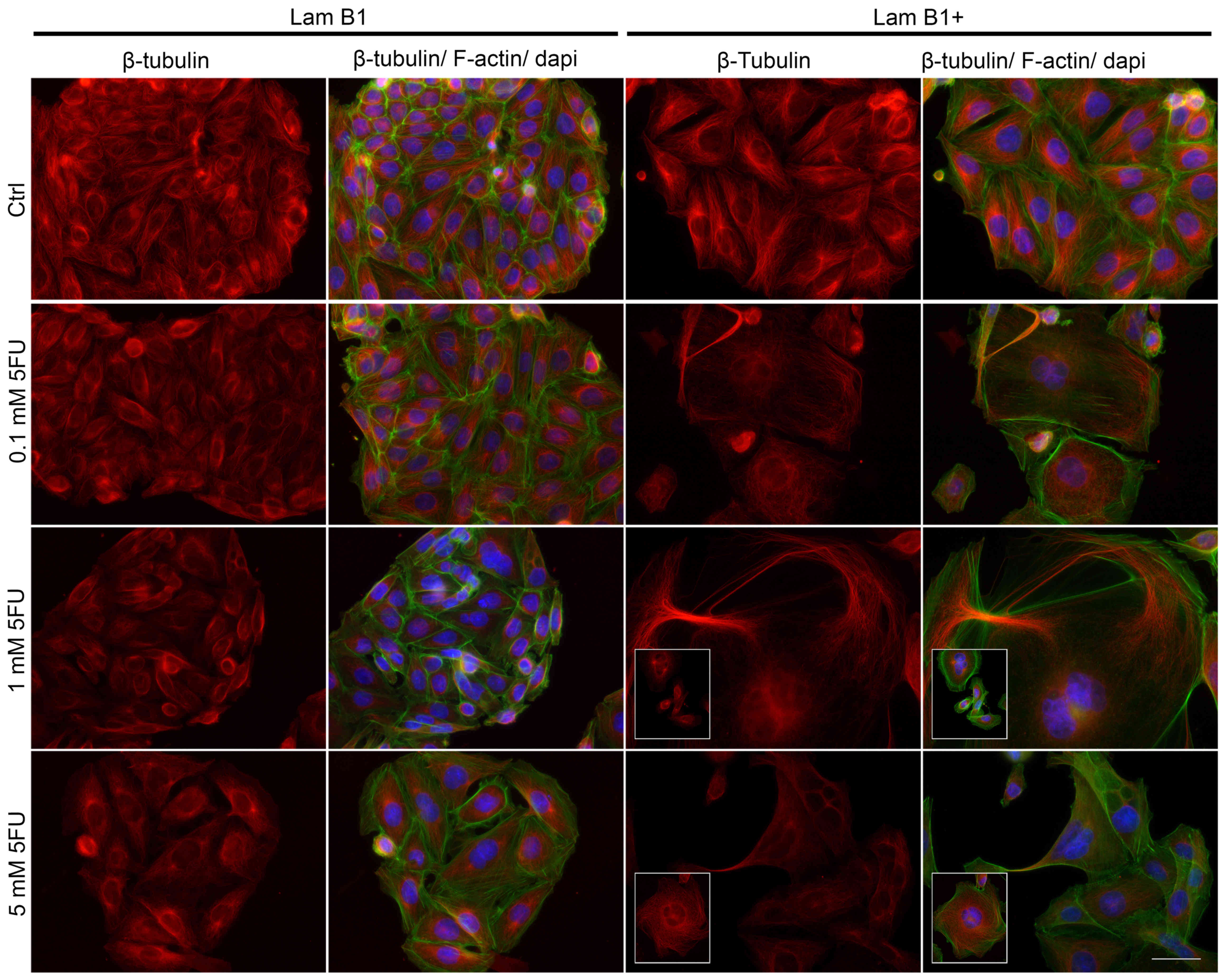
The fluorescence staining of β-tubulin showed
5-FU-induced changes in the organization of microtubules and
mitotic spindle morphology (Fig.
5). In the control cells without upregulated LMNB1, β-tubulin
was organized in a regular and dense network of long tubules, which
radiated from the microtubule-organizing centers (MTOCs) (Fig. 5). Together with 5-FU doses and
especially after 5 mM 5-FU, microtubules formed a less dense
network consisting of shorter fibers, as compared to the control
(Fig. 5). Moreover, in shrunken
cells, a significantly higher fluorescence of tubulin was noted
(Fig. 5). The morphology of LMNB1
upregulated cells underwent changes due to cytoskeletal
reorganization (Fig. 5), and these
changes escalated following treatment with 5-FU. In control cells
with induced overexpression of LMNB1, the organization of
microtubules was similar to control cells without upregulated LMNB1
(Fig. 5). However, after exposure
to 5-FU, giant and multinucleated cells with a phenotype resembling
mitotic catastrophe were observed (Fig. 5). In these cells, microtubule
network was well-developed, but fibers were shorter and in some
cases thicker and/or less regular (Fig. 5). 5-FU induced also changes in the
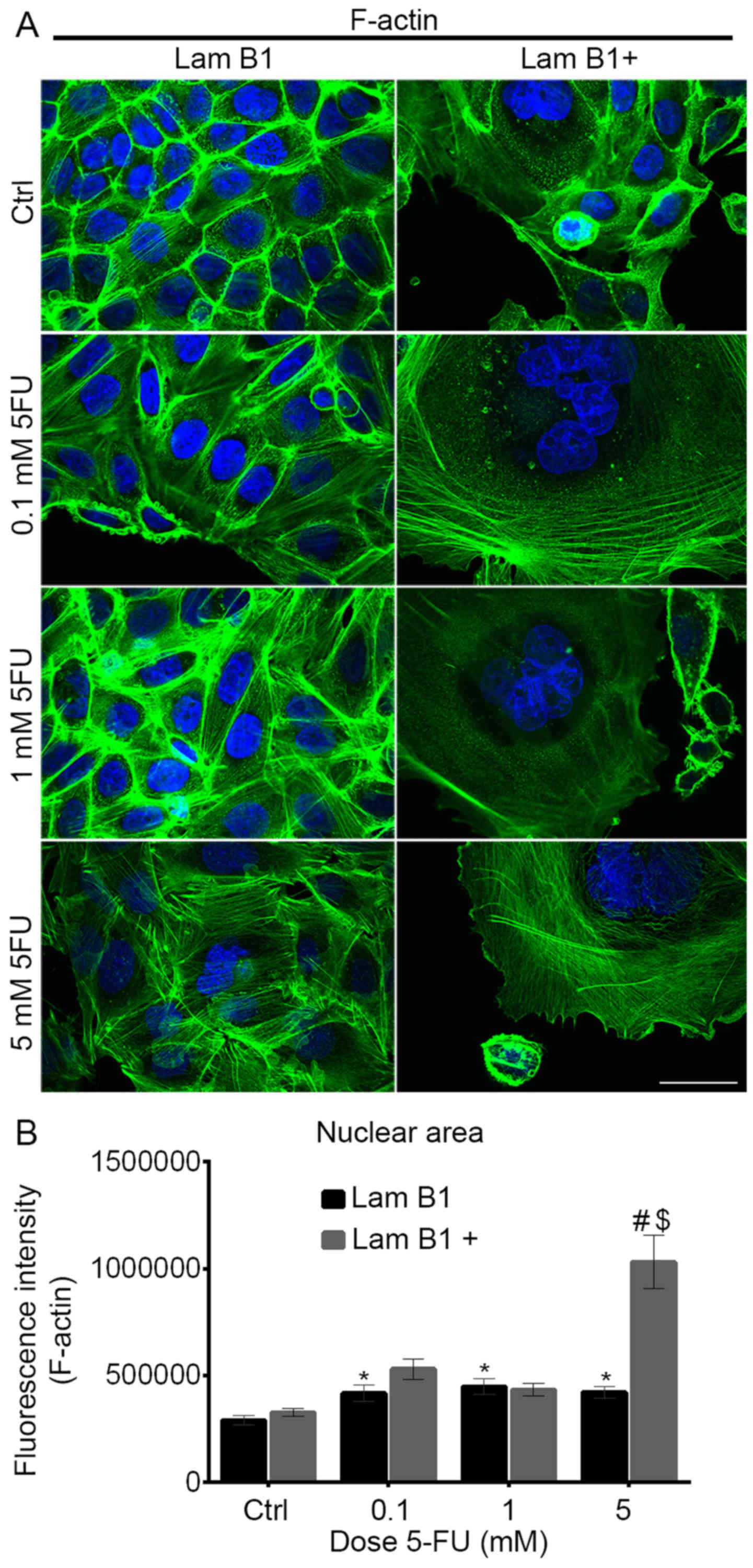
organization of actin cytoskeleton in cells with upregulated
expression of LMNB1 (Figs. 5 and
6A). Control cells without
upregulated LMNB1 were characterized as highly-developed F-actin
with long stress fibers in the cytoplasm and cortically, also in
regions of cell-cell junctions (Figs.
5 and 6A). After treatment
with 5-FU, in smaller cells, they underwent accumulation in the
cortical region, but significant changes in microfilament structure
were not observed (Figs. 5 and
6A). In turn, after upregulation
of LMNB1 and treatment with 5-FU reorganization of actin filaments
occurred (Figs. 5 and 6A). They exhibit a diffuse network of
F-actin with short actin fibers and/or small, punctate
accumulations within the cytoplasm or in the cortical region of
cells (Figs. 5 and 6A). In enlarged multinucleated cells,
F-actin was located mainly in the form of small granules and/or in
the form of short fibers, moreover, they were accumulated in the
cortical area of cells (Figs. 5
and 6A). Analysis of intensity of
nuclear F-actin fluorescence showed a statistically significant
increase in fluorescence intensity after treatment of cells with
normal expression of LMNB1 with 0.1, 1 and 5 mM of 5-FU in
comparison to the control cells (Fig.
6B). Statistically significant increase of nuclear actin
fluorescence was observed also in cells with overexpressed LMNB1
following treatment with 5 mM of 5-FU, as compared to the control
and between cells with and without upregulated LMNB1 (Fig. 6B).
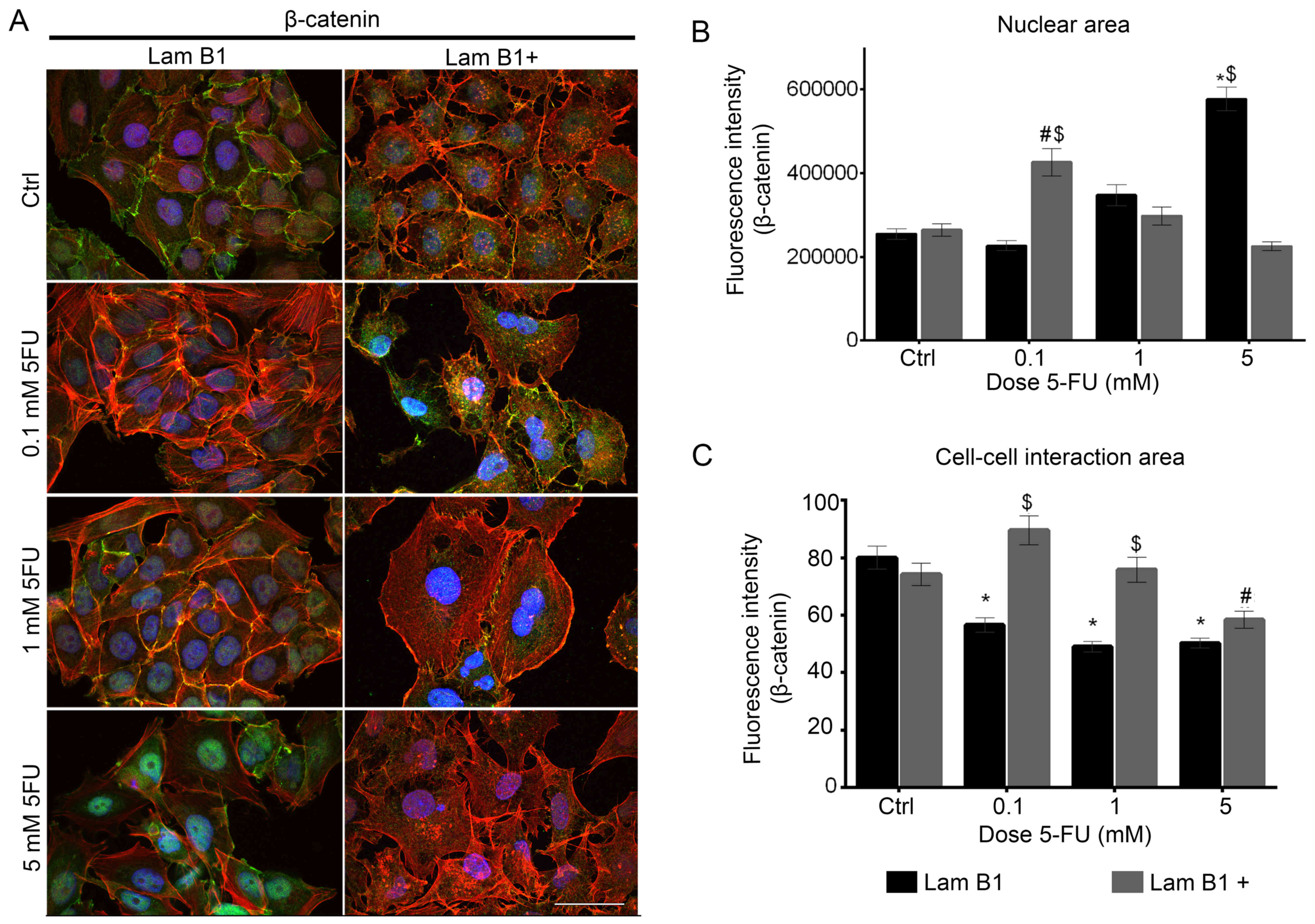
The effect of LMNB1 upregulation and 5-FU on
fluorescence of β-catenin was also investigated (Fig. 7A). Fluorescence double staining of
β-catenin and F-actin in LoVo cells transfected with control
plasmid showed the dose-dependent decrease of fluorescence
β-catenin at the cell periphery area (Fig. 7A). In control cells and cells after
treatment with 0.1 and 1 mM of 5-FU colocalization of β-catenin and
actin filaments was seen. Furthermore, the β-catenin was presented
as a small punctate accumulation in the cytoplasm and in the
nuclear area. Additionally, after treatment of these cells with 5
mM of 5-FU, an increase in accumulation of nuclear β-catenin was
observed (Fig. 7A). After
overexpression of LMNB1 and treatment with 0.1 and 1 mM of 5-FU,
statistically significant increase of β-catenin fluorescence
intensity in cell-cell interaction area was observed, as a
comparison to the cells transfected with control plasmid and
treated with the same dose of 5-FU (Fig. 7B). On the other hand, the
fluorescence intensity of nuclear β-catenin was radically increased
in LoVo cells transfected with control plasmid and after treated
with 5 mM of 5-FU, but in the cells with upregulated LMNB1
expression the nuclear intensity of β-catenin increased following
treatment with the lowest dose of 5-FU and reduced following
treatment with higher concentrations of 5-FU (Fig. 7B). The fluorescence intensity of
nuclear β-catenin in the cells with upregulated LMNB1 and treated
with 5 mM of 5-FU was lower than in the control (Fig. 7B). In control cells with
upregulated expression of LMNB1, β-catenin was located in cell-cell
interaction area and colocalized with cortical actin filaments.
Additional, granules of β-catenin in the cytoplasm were stained
(Fig. 7A). Moreover, following
treatment with the lowest dose of 5-FU, β-catenin was presented
similarly to results obtained in control cells (Fig. 7A). The incubation of LoVo cells
with upregulated LMNB1 with 1 and 5 mM of 5-FU resulted in changes
in β-catenin distribution. This protein was located in the
periphery of the cells and on the nucleus area, but the intensity
of fluorescence was lower as compared to the control and cells
treated with 0.1 mM of 5-FU (Fig.
7A).
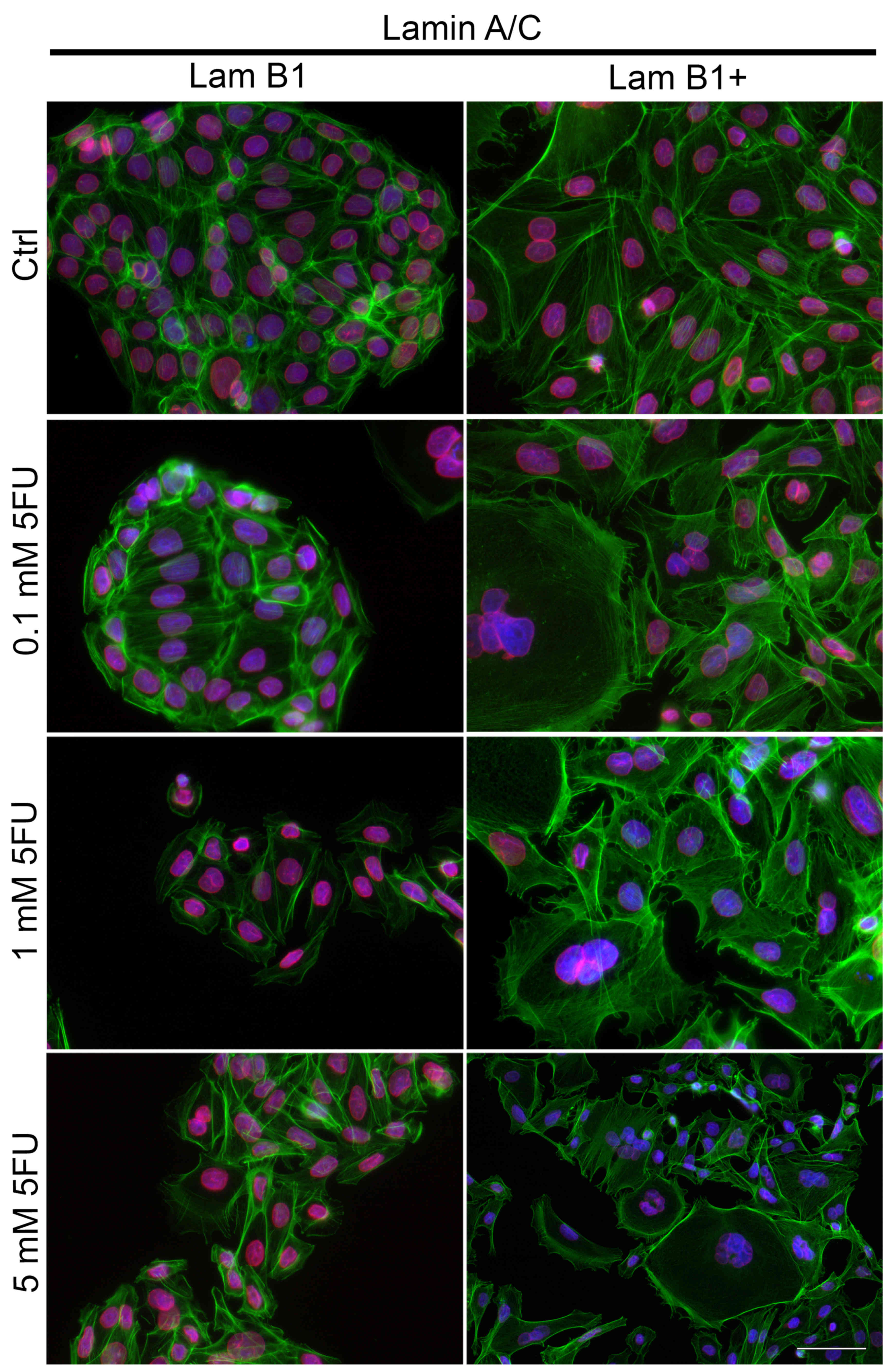
The effect of overexpression of LMNB on fluorescence
staining of lamin A/C also was studied (Fig. 8). In control cells without
upregulated LMNB1 expression and after their treatment with 5-FU,
lamin A/C was located both in the nucleus matrix and in the nuclear
membrane (Fig. 8). Based on the
microscopic analysis high fluorescence intensity at the nuclear
area was observed (Fig. 8).
However, in the cells with overexpressed LMB1, the fluorescence
intensity of lamin A/C was decreased as compared to cells
transfected with control plasmid and localized mainly at the
periphery of the cell nucleus, especially after treatment with 5-FU
(Fig. 8). Additional,
dose-dependent decrease of nuclear lamin A/C fluorescence at the
nuclear area was noted (Fig.
8).
The effect of LMNB1 cDNA transfection on
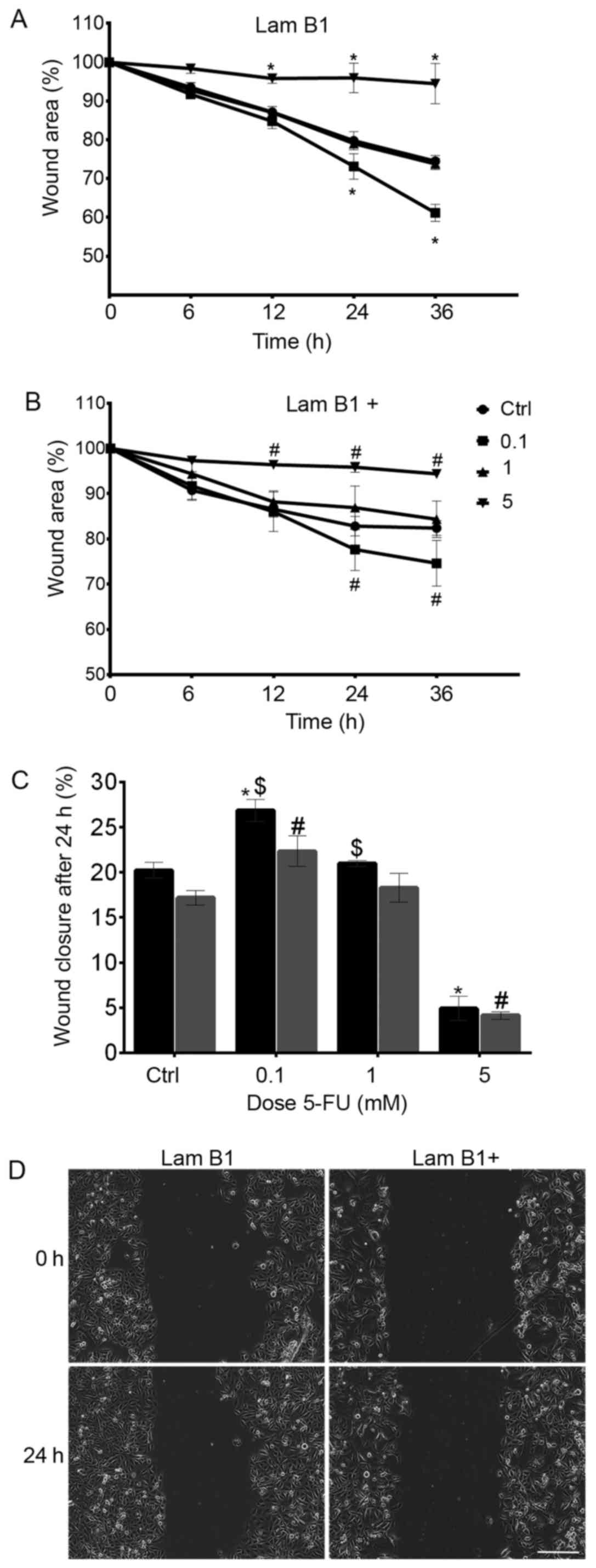
migration of LoVo cells
Wound healing assay is a commonly accepted and
well-developed method to measure cell migration in vitro.
Live-cell imaging system was used to record the process of wound
closure and cell migration. Documentation was done every 10 min and
lasted 36 h. In all cases of the study scratch in the monolayer
were still observed (Fig. 9).
After 24-h incubation with a cytostatic, the inhibition of cell
migration in LMNB1 upregulated cells in comparison to cells without
overexpression of LMNB1 was observed (Fig. 9). As shown in Fig. 9C, the wound area at 24 h after
treatment with 5-FU was 20.22% in cells transfected with control
plasmid. Moreover, following the treatment with 0.1, 1 and 5 mM
5-FU, the percentage reduction in wound size was observed and
amounted to 26.8, 21 and 4.9%, respectively. Furthermore, the
results were statistically significant in cells treated with 0.1
and 5 mM of 5-FU as a comparison to the control (Fig. 9C). Additionally, the cell mobility
measured as wound area was less in cells with overexpressed LMNB1
as compared to cells transfected with control plasmid and amounted
to 17.2, 22.3% (0.1 mM of 5-FU), 18.3% (1 mM 5-FU), 4.1% (5 mM
5-FU) in the control and cells treated with 0.1, 1 and 5 mM 5-FU,
respectively (Fig. 9C). In cells
transfected with control plasmid and treated with 5 mM dose of
cytostatic, in 12, 24 and 36 h migration test, a statistically
significant decrease in cell migration was observed, as compared to
the control. In turn, in these cells incubated with 0.1 mM of 5-FU
after 24 and 36 h of increase of cells migration was noted
(Fig. 9A). The same dependence was
observed in cells with overexpressed LMNB1 (Fig. 9B). As shown in Fig. 9D, the inhibition of migration of
cells with overexpression of LMNB1 following treatment with 0.1 mM
5-FU was noted (Fig. 9D).
The effect of LMNB1 expression on
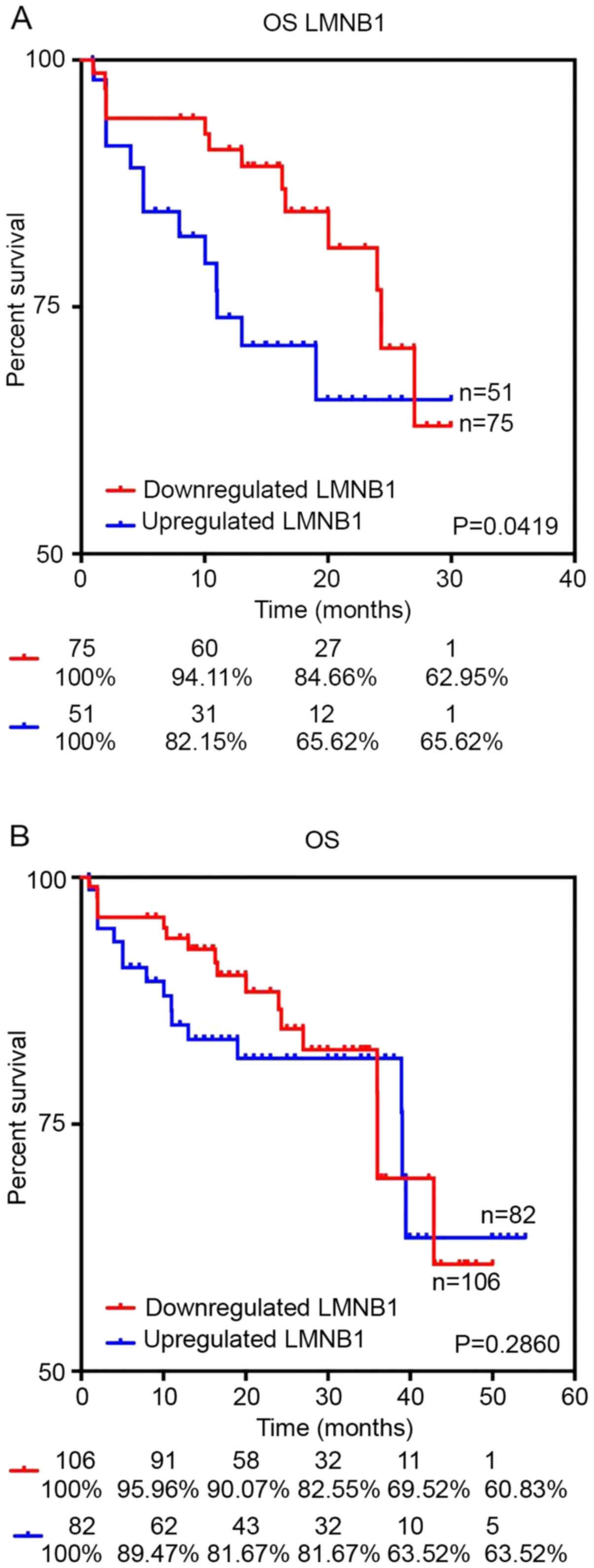
survival of patients with colorectal adenocarcinoma
Using the data obtained from TCGA data Portal
Open-Access directory via cBioPortal database we elucidated
prognostic value of LMNB1 mRNA expression in patients with
colorectal adenocarcinoma. The results showed that there are no
significant differences between patients with low and high
expression of LMNB1 in long perspective. However, patients with
upregulated LMNB1 were characterized by lower survival rates within
the first 30 months (P=0.0419) (Fig.
10).
Discussion
Carcinogenesis, as a multistep process in which many
components could be modified, is a target of scientists all over
the world. In this study, we presented the role of lamin B1 in the
regulation of pathogenesis and progression of colorectal cancer,
which is the most common malignancy and a major cause of
cancer-related deaths (4). Many
authors have shown that expression of LMNB1 in colon cancer is
decreased, and can be related to high aggressive of this tumor
(4,18). Chemotherapeutic treatment of
colorectal cancer is based on 5-fluorouracil (5-FU) which is an
anticancer drug interfering with nucleoside metabolism through
incorporation into RNA and DNA. This leads to cell death (6,19,20).
After treatment of LoVo cells without altered expression of LMNB1
with 5-FU, we observed induction of apoptosis. Similar, apoptotic
effect was presented for these types of cells by Das et al
(21) and Wang et al
(22,23). As it was shown by Mhaidat et
al (24) in colorectal cancer,
5-fluorouracil induced apoptosis mediated by protein kinase C-δ and
by activation of caspase-9. Moreover, 5-FU can modulate some
members of the Bcl-2 protein family and induces cell death
(25). Additionally, it is
possible to increase the apoptotic potential by combination of 5-FU
with other substances. An example of such research are studies
presented by Wang et al (23), in which they observed enhanced
apoptotic effect of 5-FU by SM-1 in LoVo cells. SM-1 is a novel,
proapoptotic agent, which directly activates caspase-3 and induces
the cell death process on the higher level. Furthermore, Milczarek
et al (26) presented that
vitamin D analogs enhance the anticancer effect of 5-FU in mouse
colon cancer model. Thus, the intensity of cytotoxic effect can be
enhanced by manipulation of gene expression, which results in
direct or indirect impact on proliferation, death or migration of
cells. It was presented by Chen et al (27) that after silencing HIF-1α, the
level of apoptotic cells increased after the treatment of cells
with adriamycin, irinotecan, vincristine and 5-FU. Similarly, Li
et al (28) indicated that
human colorectal cancer HCT116 cells after downregulation of
caveolin-1 are more sensitive to 5-FU. In the present study, we
also observed a slight increase in the percentage of apoptotic
cells following treatment with all doses of 5-FU in cells with
upregulated LMNB1, as compared to the control. Except apoptosis, a
small percentage of necrotic and high number of mitotic catastrophe
cells were observed. Necrosis was present also in colorectal cancer
cells by Hamam et al (29)
but they suggested that the higher level of necrotic and apoptotic
cells was observed after treatment of cells with a combination of
5-FU and CUDC-907, than with 5-FU alone. In recent years, many
studies presented that autophagy is linked to 5-FU resistance in
colon cancer cells and inhibition of this process could be
promising apoptotic effect of 5-FU (30,31).
This dependence is connected with the regulation of apoptotic
proteins Bcl-2 and Bcl-xL by 5-FU, which influence the
autophagy-promoting Beclin1VPS34 complex (32,33).
It is also presented that p38MAPK signaling pathway has a very
important role in controlling the balance between autophagy and
induced by 5-FU apoptosis (34).
Necroptosis is another type of cell death, which is indirect
induced by 5-FU (35). In turn,
overexpression of LMNB1 induced by β-Asarone increased cellular
senescence cell population and inhibited colon cancer formation
(36). In the present study, we
observed mitotic catastrophe. After upregulation of LMNB1 in LoVo
cells, a dose-dependent increase of cells with DNA content >4N
were noticed, what testifies of polyploidy cells.
Fluorescence microscopic analysis confirmed the
results. Induction of mitotic catastrophe was also shown by
Yoshikawa et al (37) in
colorectal carcinoma cells (SW480 and COLO320DM) after treatment
with 5-FU. Aforementioned scientists observed a dual effect of
5-FU, which is dependent on the dose of the cytostatic. At the
lower dose (100 ng/ml) the cells were arrested in G2/M phase and
mitotic catastrophe was observed. Following treatment with a higher
dose of 5-FU, the cells underwent apoptosis. In our previous study,
we also presented that induction of cell death with doxorubicin in
CHO AA8 cells is dose-dependent. On the other hand, down-regulation
of cofilin-1 caused lack of apoptosis and following treatment with
doxorubicin only mitotic catastrophe was observed (38). Changes in cell cycle involved not
only a dose-dependent increase of cells with DNA content >4N but
also in G0/G1, s and G2/M phases. Similarly to Chen et al
(39), after treatment of control
cells with 0.1 and 1 mM of 5-FU accumulation in G0/G1 and
significantly decreased the population of S-phase cells was shown.
It is known that in comparison with sensitive cancer cells, the
higher population of 5-FU-resistant cancer cells were arrested in
G0/G1 cell cycle phase (40) but
we observed the increase of cell population in G0/G1 following
treatment of cells with upregulated LMNB1 with 0.1 mM of 5-FU, but
during their incubation with 1 and 5 mM of 5-FU, the population of
cells in this phase decreased in the interest of sub-G1 and >4N,
and in the case of 5 mM to G2/M also. Nie et al (41) after downregulation of Rac1 showed
arrested LoVo cells in G0/G1 phase and induced apoptosis. The
sub-V1 peak has been identified as the apoptotic fraction and in
our study the sub-G1 population square with the analysis of
apoptotic cells presented by dual staining with Annexin V and PI.
Differences were observed following treatment with 5 mM of 5-FU in
control cells and the cells with upregulation of LMNB1, but low
level of the sub-G1 peak did not conform to the lack of apoptosis
(42). Apoptosis has been marked
as DNA degradation into oligonucleosomal-length fragments, but in
the literature, there are many examples where particularly for
apoptosis internucleosomal DNA degradation and fragmentation is not
observed. In this case identified apoptotic cells based on cellular
DNA content is worse way than Annexin V/PI assay or TUNEL analysis
(43–45).
As mentioned above, after treatment with the highest
dose of 5-FU, we observed a dramatic increase in >4N cell
fractions. Moreover, immunofluorescent staining showed increased
number of cells with visible hallmarks of mitotic catastrophe. 5-FU
alone has limited potential to induce endoreduplication alone but
it has been proven that can act synergistically with other drugs.
Colorectal cancer cell lines treated with 5-FU with dual Histone
deacetylase and Phosphatidylinositide 3-Kinase Inhibitor (CUDC-907)
exhibited enhanced cell polyploidization (29). The lamin B1 plays a crucial role in
the maintenance of nuclear structure and function. The decrease in
LMNB1 expression results in disturbed chromatin condensation,
improper heterochromatin localization and altered gene expression
and splicing. It is possible that overexpressed LMNB1 drives to
endoreduplication and allows cells to start another s phase without
mitotic division (46). The
presence of polyploidy coincides with the appearance of cells which
show morphological aspects of mitotic catastrophe. The role of
mitotic catastrophe is still discussed but the most interesting
aspect of this phenomenon possibly is its pro-survival character.
Mitotic catastrophe is commonly observed in p53 mutated tumor cells
which avoid cell cycle checkpoints. Studies showed that polyploid
giant cells have the ability to chromatid exchange and probably the
production of a fully repaired chromosome. Moreover, homologous
chromosomes from the giant cell can be relocated and paired which
is the step to depolyploidization (47,48).
Silencing of LMNB1 affects DNA damage and repair pathway.
Disregulated proteins such as BRCA1 and RAD51 after downregulation
of LMNB1 caused non-efficient DNA repair via both non-homologous
end joining and homologous repair (49). Additionally, depletion of lamin B1
prevented induction of Rad51 expression after IR radiation.
Proteasome inhibitor MG132 repealed this effect which suggests that
lamin B1 stabilizes RAD51 by preventing proteasome-mediated
degradation in cells with IR-induced DNA damage. Moreover, cells
with depleted levels of LMNB1 were characterized by higher
radiosensitivity and lower survival rates (50). We hypothesize that overexpression
of LMNB1 drives to enhanced ability of DNA repair which in turn
allows these 'catastrophic cells' to return to the normal cell
cycle avoiding cell death and provides survival.
Cytoskeletal components (microtubules,
microfilaments and intermediate filaments), especially their
organization and changes, are strictly connected with the state of
cells. The cytoskeleton is also involved in many cellular processes
such as mitosis, proliferation, migration and cell death (51,52).
In the control cells transfected with control plasmid, microtubules
were present as a regular, dense network of long microtubules,
which radiated from the microtubule-organizing centers (MTOCs).
Following treatment with a cytostatic, especially with the highest
dose microtubules, formed a less dense network with shorter fibers
or were depolymerized in apoptotic cells. In our previous study, we
showed significant thickening of microtubule bundles which was
particularly evident in the giant multinucleated cells after 10 and
20 mM of caffeine treatment of H1299 cells. Furthermore, in
shrunken cells β-tubulin underwent depolymerization and the network
of microtubules was not observed (53). Similar observation was presented by
Pawlik et al (54). As
shown, after the exposure of H1299 cells to
phenethylisothiocyanate, the rearrangement of tubulin was observed
(54). Here, actin filaments were
present in the cytoplasm and in the cell-cell interaction areas. In
turn, 5-FU resulted in disorganization of actin cytoskeleton in
cells with upregulated LMNB1. In enlarged multinucleated cells
expanded F-actin cytoskeleton was located mainly in the form of
small granules and/or short fibers within the cytoplasm or in the
cortical region of cells. A well-developed actin cytoskeleton in
cells with the phenotype of mitotic catastrophe was also observed
in other studies (54–56). In apoptotic cells, actin filaments
are located in the whole cytoplasm and accumulated near the plasma
membrane. A number of previous studies presented that actin
polymerization and accumulation at the cell periphery is
indispensable for plasma membrane blebbing and apoptotic bleb
formation (57–61). Thus, actin cytoskeleton, located in
the cortical region in shrunken cells is very important during the
apoptotic process. However, not only cytoplasmic actin is crucial
in this cell death, because F-actin presence in the nucleus of
different cell lines following treatment with cytostatics has been
implicated in chromatin remodeling during both apoptosis and
mitotic catastrophe (38,62,63).
In this study, we observed the high intensity of nuclear F-actin
fluorescence in LoVo cells with overexpression of LMNB1 and
following treatment with 5 mM of 5-FU. This correlated with a
number of cells with mitotic catastrophe phenotype. These
observations are connected with our previous study, where we showed
F-actin in the transcriptionally active regions of the cell nucleus
during active cell death. Additionally, it was presented that
F-actin colocalized with SATB1 protein, which is one of the most
important nuclear proteins involved in chromatin organization
(64–66).
As evidenced by the above study, nuclear F-actin
plays an essential role in cell death, but cortical actin filaments
maintain the shape, polarity of cells and are involved in cell-cell
adhesion (67). Adhesion proteins
form the complexes of inter-cellular junctions by interaction with
the cytoskeleton (67–69). β-catenin is a multi-functional
protein, which is an element of adherent junctions and acts as a
transcriptional activator in Wnt signaling pathway (70,71).
Additionally, β-catenin is indirect, by α-catenin links to actin
filaments (72). Stability of
cell-cell adhesions depends on the proper organization of actin
filaments, which may impact on cell migration and invasion. In the
study presented by Gagat et al (73), the stabilization of F-actin through
overexpression of tropomyosin-1 in EA.hy926 cells increased
junctional β-catenin expression and thereby preserved endothelial
barrier function against oxidative stress conditions. Here, the
fluorescence intensity of β-catenin located in cell-cell
interaction areas in LoVo cells with an elevated level of LMNB1
following treatment with all doses of 5-FU was significantly higher
in comparison to the cells with a naive expression of LMNB1 and the
treatment with the same doses of cytostatic. This result can
suggest that junctions between the cells with overexpressed LMNB1
are stronger, with consequences for the control of cancer cell
dissociation and spread. Tumor invasion and metastasis frequently
coincide with the loss of cell-cell adhesion proteins, mainly
cadherin, but the key to the adhesive activity is the interaction
between these proteins, catenin and actin cytoskeleton (74,75).
On the other hand, in cells transfected with control plasmid, a
dose-dependent increase of nuclear β-catenin was observed. However,
after upregulation of LMNB1 inverse results were observed. This
observation agrees with Han et al (10). They showed that downregulation of
β-catenin inhibited the invasion and migration of LoVo cells.
Additionally, we also present here the slight inhibition of cell
migration in cells with upregulated LMNB1.
Very important in the aspect of the regulation of
migration and tumor invasion is lamin A/C. In the present study, we
observed a high level of fluorescence intensity of lamin A/C in
control cells treated with all doses of 5-FU. In turn, upregulation
of LMNB1 decreased the fluorescence of lamin A/C in all doses of
5-FU. Willis et al (76)
proposed that the high risk of colorectal cancer-related mortality
is connected with an expression of lamin A. Lamin A increases
invasive potential of the tumor due to its interaction with actin
filaments. Dynamics of these interactions caused the loss of cell
adhesion and led to increased cell motility.
Then, using the data obtained from TCGA data Portal
Open-Access directory via cBioPortal we investigated if patients
with overexpression of LMNB1 mRNA were characterized by lower
survival rates. However, there are no significant differences in
long-term overall survival but a significantly lower survival rate
in patients with high expression of LMNB1 was observed in the first
30 months. As we described above overexpression of LMNB1 was
associated with the appearance of the cells with morphological
hallmarks of mitotic catastrophe. Mitotic catastrophe plays a dual
role in the cell cycle. This mechanism eliminates cells which
failed proper mitosis and limits proliferation. Simultaneously,
cells which undergo mitotic catastrophe are more resistant to
cytostatic drugs and are capable of repairing damaged DNA to return
to the normal cell cycle. It is possible that mitotic catastrophe
cells in patients with colorectal cancer may be a reservoir of the
cells responsible for faster disease progression, but further
investigations are necessary to confirm this hypothesis.
In summary, this is the first report which shows the
influence of upregulation of LMNB1 in colon cancer cells. Based on
the current results we may conclude that the overexpression of
LMNB1 in LoVo cells causes induction of mitotic catastrophe by
5-FU, intensification of cell-cell junctions and limits migration
of cells.
Acknowledgments
The present study was co-supported by research tasks
within the framework of statutory activities (Nicolaus Copernicus
University in Toruń, Faculty of Medicine, Collegium Medicum in
Bydgoszcz).
References
|
1
|
Dittmer TA and Misteli T: The lamin
protein family. Genome Biol. 12:2222011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Butin-Israeli V, Adam SA, Goldman AE and
Goldman RD: Nuclear lamin functions and disease. Trends Genet.
28:464–471. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Sakthivel KM and Sehgal P: A Novel role of
lamins from genetic disease to cancer biomarkers. Oncol Rev.
10:3092016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Li L, Du Y, Kong X, Li Z, Jia Z, Cui J,
Gao J, Wang G and Xie K: Lamin B1 is a novel therapeutic target of
betulinic acid in pancreatic cancer. Clin Cancer Res. 19:4651–4661.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Irianto J, Pfeifer CR, Ivanovska IL, Swift
J and Discher DE: Nuclear lamins in cancer. Cell Mol Bioeng.
9:258–267. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Jensen NF, Stenvang J, Beck MK, Hanáková
B, Belling KC, Do KN, Viuff B, Nygård SB, Gupta R, Rasmussen MH, et
al: Establishment and characterization of models of chemotherapy
resistance in colorectal cancer: Towards a predictive signature of
chemoresistance. Mol Oncol. 9:1169–1185. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hofmann WA: Cell and molecular biology of
nuclear actin. Int Rev Cell Mol Biol. 273:219–263. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Falahzadeh K, Banaei-Esfahani A and
Shahhoseini M: The potential roles of actin in the nucleus. Cell J.
17:7–14. 2015.PubMed/NCBI
|
|
9
|
Houben F, Ramaekers FC, Snoeckx LH and
Broers JL: Role of nuclear lamina-cytoskeleton interactions in the
maintenance of cellular strength. Biochim Biophys Acta.
1773:675–686. 2007. View Article : Google Scholar
|
|
10
|
Han J, Gao B, Jin X, Xu Z, Li Z, Sun Y and
Song B: Small interfering RNA-mediated downregulation of
beta-catenin inhibits invasion and migration of colon cancer cells
in vitro. Med Sci Monit. 18:BR273–BR280. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Jamieson C, Sharma M and Henderson BR: Wnt
signaling from membrane to nucleus: B-catenin caught in a loop. Int
J Biochem Cell Biol. 44:847–850. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Serebryannyy LA, Yemelyanov A, Gottardi CJ
and de Lanerolle P: Nuclear α-catenin mediates the DNA damage
response via β-catenin and nuclear actin. J Cell Sci.
130:1717–1729. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Cerami E, Gao J, Dogrusoz U, Gross BE,
Sumer SO, Aksoy BA, Jacobsen A, Byrne CJ, Heuer ML, Larsson E, et
al: The cBio cancer genomics portal: An open platform for exploring
multi-dimensional cancer genomics data. Cancer Discov. 2:401–404.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Simon DN, Zastrow MS and Wilson KL: Direct
actin binding to A- and B-type lamin tails and actin filament
bundling by the lamin A tail. Nucleus. 1:264–272. 2010. View Article : Google Scholar
|
|
15
|
Tariq Z, Zhang H, Chia-Liu A, Shen Y, Gete
Y, Xiong ZM, Tocheny C, Campanello L, Wu D, Losert W, et al: Lamin
A and microtubules collaborate to maintain nuclear morphology.
Nucleus. 8:433–446. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Brembeck FH, Rosário M and Birchmeier W:
Balancing cell adhesion and Wnt signaling, the key role of
beta-catenin. Curr Opin Genet Dev. 16:51–59. 2006. View Article : Google Scholar
|
|
17
|
Houben F, Willems CH, Declercq IL,
Hochstenbach K, Kamps MA, Snoeckx LH, Ramaekers FC and Broers JL:
Disturbed nuclear orientation and cellular migration in A-type
lamin deficient cells. Biochim Biophys Acta. 1793:312–324. 2009.
View Article : Google Scholar
|
|
18
|
Moss SF, Krivosheyev V, de Souza A, Chin
K, Gaetz HP, Chaudhary N, Worman HJ and Holt PR: Decreased and
aberrant nuclear lamin expression in gastrointestinal tract
neoplasms. Gut. 45:723–729. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zhang N, Yin Y, Xu SJ and Chen WS:
5-Fluorouracil: Mechanisms of resistance and reversal strategies.
Molecules. 13:1551–1569. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zhang L, Song R, Gu D, Zhang X, Yu B, Liu
B and Xie J: The role of GLI1 for 5-Fu resistance in colorectal
cancer. Cell Biosci. 7:172017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Das D, Preet R, Mohapatra P, Satapathy SR,
Siddharth S, Tamir T, Jain V, Bharatam PV, Wyatt MD and Kundu CN:
5-Fluorouracil mediated anti-cancer activity in colon cancer cells
is through the induction of Adenomatous Polyposis Coli: Implication
of the long-patch base excision repair pathway. DNA Repair (Amst).
24:15–25. 2014. View Article : Google Scholar
|
|
22
|
Wang Z, Jiang B, Chen L, Di J, Cui M, Liu
M, Ma Y, Yang H, Xing J, Zhang C, et al: GOLPH3 predicts survival
of colorectal cancer patients treated with 5-fluorouracil-based
adjuvant chemotherapy. J Transl Med. 12:152014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wang Y, Yuan S, Li L, Yang D, Xu C, Wang S
and Zhang D: Novel proapoptotic agent SM-1 enhances the inhibitory
effect of 5-fluorouracil on colorectal cancer cells in vitro and in
vivo. Oncol Lett. 13:4762–4768. 2017.PubMed/NCBI
|
|
24
|
Mhaidat NM, Bouklihacene M and Thorne RF:
5-Fluorouracil-induced apoptosis in colorectal cancer cells is
caspase-9-dependent and mediated by activation of protein kinase
C-δ. Oncol Lett. 8:699–704. 2014.PubMed/NCBI
|
|
25
|
Nita Me, Nagawa H, Tominaga O, Tsuno N,
Fujii S, Sasaki S, Fu CG, Takenoue T, Tsuruo T and Muto T:
5-Fluorouracil induces apoptosis in human colon cancer cell lines
with modulation of Bcl-2 family proteins. Br J Cancer. 78:986–992.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Milczarek M, Psurski M, Kutner A and
Wietrzyk J: Vitamin d analogs enhance the anticancer activity of
5-fluorouracil in an in vivo mouse colon cancer model. BMC Cancer.
13:2942013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chen J, Ding Z, Peng Y, Pan F, Li J, Zou
L, Zhang Y and Liang H: HIF-1α inhibition reverses multidrug
resistance in colon cancer cells via downregulation of
MDR1/P-glycoprotein. PLoS One. 9:e988822014. View Article : Google Scholar
|
|
28
|
Li Z, Wang N, Huang C, Bao Y, Jiang Y and
Zhu G: Down-regulation of caveolin-1 increases the sensitivity of
drug-resistant colorectal cancer HCT116 cells to 5-fluorouracil.
Oncol Lett. 13:483–487. 2017.PubMed/NCBI
|
|
29
|
Hamam R, Ali D, Vishnubalaji R, Alsaaran
ZF, Chalisserry EP, Alfayez M, Aldahmash A and Alajez NM and Alajez
NM: Enhanced efficacy of 5-fluorouracil in combination with a dual
histone deacetylase and phosphatidylinositide 3-kinase inhibitor
(CUDC-907) in colorectal cancer cells. Saudi J Gastroenterol.
23:34–38. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yao CM, Kang KA, Piao MJ, Ryu YS, Fernando
PMDJ, Oh MC, Park JE, Shilnikova K, Na SY, Jeong SU, et al: Reduced
autophagy in 5-fluorouracil resistant colon cancer cells. Biomol
Ther (Seoul). 25:315–320. 2017. View Article : Google Scholar
|
|
31
|
Tang JC, Feng YL, Liang X and Cai XJ:
Autophagy in 5-fluorouracil therapy in gastrointestinal gancer:
Trends and challenges. Chin Med J (Engl). 129:456–463. 2016.
View Article : Google Scholar
|
|
32
|
Shimizu S, Kanaseki T, Mizushima N, Mizuta
T, Arakawa-Kobayashi S, Thompson CB and Tsujimoto Y: Role of Bcl-2
family proteins in a non-apoptotic programmed cell death dependent
on autophagy genes. Nat Cell Biol. 6:1221–1228. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Pattingre S, Tassa A, Qu X, Garuti R,
Liang XH, Mizushima N, Packer M, Schneider MD and Levine B: Bcl-2
antiapoptotic proteins inhibit Beclin 1-dependent autophagy. Cell.
122:927–939. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
de la Cruz Morcillo MA, Valero ML,
Callejas Valera JL, Arias González L, Melgar Rojas P, Galán Moya
EM, García-Gil E, García-Cano J and Sánchez-Prieto R: P38MAPK is a
major determinant of the balance between apoptosis and autophagy
triggered by 5-fluorouracil: implication in resistance. Oncogene.
31:1073–1085. 2012. View Article : Google Scholar
|
|
35
|
Oliver Metzig M, Fuchs D, Tagscherer KE,
Gröne HJ, Schirmacher P and Roth W: Inhibition of caspases primes
colon cancer cells for 5-fluorouracil-induced TNF-α-dependent
necroptosis driven by RIP1 kinase and NF-κB. Oncogene.
35:3399–3409. 2016. View Article : Google Scholar
|
|
36
|
Liu L, Wang J, Shi L, Zhang W, Du X, Wang
Z and Zhang Y: β-Asarone induces senescence in colorectal cancer
cells by inducing lamin B1 expression. Phytomedicine. 20:512–520.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Yoshikawa R, Kusunoki M, Yanagi H, Noda M,
Furuyama JI, Yamamura T and Hashimoto-Tamaoki T: Dual antitumor
effects of 5-fluorouracil on the cell cycle in colorectal carcinoma
cells: A novel target mechanism concept for pharmacokinetic
modulating chemotherapy. Cancer Res. 61:1029–1037. 2001.PubMed/NCBI
|
|
38
|
Grzanka D, Marszałek A, Gagat M, Izdebska
M, Gackowska L and Grzanka A: Doxorubicin-induced F-actin
reorganization in cofilin-1 (nonmuscle) down-regulated CHO AA8
cells. Folia Histochem Cytobiol. 48:377–386. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Chen XX, Lai MD, Zhang YL and Huang Q:
Less cytotoxicity to combination therapy of 5-fluorouracil and
cisplatin than 5-fluorouracil alone in human colon cancer cell
lines. World J Gastroenterol. 8:841–846. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Guo X, Goessl E, Jin G, Collie-Duguid ES,
Cassidy J, Wang W and O'Brien V: Cell cycle perturbation and
acquired 5-fluorouracil chemoresistance. Anticancer Res. 28(1A):
9–14. 2008.PubMed/NCBI
|
|
41
|
Nie F, Zhao SY, Song FX and Li PW: Changes
of cytoskeleton and cell cycle in Lovo cells via deletion of Rac1.
Cancer Biomark. 14:335–342. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Huang X, Halicka HD, Traganos F, Tanaka T,
Kurose A and Darzynkiewicz Z: Cytometric assessment of DNA damage
in relation to cell cycle phase and apoptosis. Cell Prolif.
38:223–243. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Darzynkiewicz Z, Juan G, Li X, Gorczyca W,
Murakami T and Traganos F: Cytometry in cell necrobiology: Analysis
of apoptosis and accidental cell death (necrosis). Cytometry.
27:1–20. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Oberhammer F, Wilson JW, Dive C, Morris
ID, Hickman JA, Wakeling AE, Walker PR and Sikorska M: Apoptotic
death in epithelial cells: Cleavage of DNA to 300 and/or 50 kb
fragments prior to or in the absence of internucleosomal
fragmentation. EMBO J. 12:3679–3684. 1993.PubMed/NCBI
|
|
45
|
Tao D, Wu J, Feng Y, Qin J, Hu J and Gong
J: New method for the analysis of cell cycle-specific apoptosis.
Cytometry A. 57:70–74. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Camps J, Erdos MR and Ried T: The role of
lamin B1 for the maintenance of nuclear structure and function.
Nucleus. 6:8–14. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Horbay R and Stoika R: Giant cell
formation: The way to cell death or cell survival? Cent Eur J Biol.
6:675–684. 2011.
|
|
48
|
Erenpreisa J and Cragg MS: Mitotic death:
A mechanism of survival? A review. Cancer Cell Int. 1:12001.
View Article : Google Scholar
|
|
49
|
Butin-Israeli V, Adam SA, Jain N, Otte GL,
Neems D, Wiesmüller L, Berger SL and Goldman RD: Role of lamin b1
in chromatin instability. Mol Cell Biol. 35:884–898. 2015.
View Article : Google Scholar :
|
|
50
|
Liu NA, Sun J, Kono K, Horikoshi Y, Ikura
T, Tong X, Haraguchi T and Tashiro S: Regulation of homologous
recombi-national repair by lamin B1 in radiation-induced DNA
damage. FASEB J. 29:2514–2525. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Hall A: The cytoskeleton and cancer.
Cancer Metastasis Rev. 28:5–14. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Pollard TD: The cytoskeleton, cellular
motility and the reductionist agenda. Nature. 422:741–745. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Hałas M, Izdebska M,
Klimaszewska-Wiśniewska A, Gagat M, Radciniewska D, Glińska A,
Gizler K, Bielińska E and Grzanka A: Caffeine induces cytoskeletal
changes and cell death in H1299 cells. Cent Eur J Biol. 9:727–738.
2014.
|
|
54
|
Pawlik A, Szczepański MA, Klimaszewska A,
Gackowska L, Zuryn A and Grzanka A: Phenethyl
isothiocyanate-induced cytoskeletal changes and cell death in lung
cancer cells. Food Chem Toxicol. 50:3577–3594. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Grzanka D, Marszałek A, Izdebska M,
Gackowska L, Andrzej Szczepanski M and Grzanka A: Actin
cytoskeleton reorganization correlates with cofilin nuclear
expression and ultrastructural changes in cho aa8 cell line after
apoptosis and mitotic catastrophe induction by doxorubicin.
Ultrastruct Pathol. 35:130–138. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Izdebska M, Gagat M, Grzanka D, Halas M
and Grzanka A: The role of exportin 6 in cytoskeletal-mediated cell
death and cell adhesion in human non-small-cell lung carcinoma
cells following doxorubicin treatment. Folia Histochem Cytobiol.
52:195–205. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Desouza M, Gunning PW and Stehn JR: The
actin cytoskeleton as a sensor and mediator of apoptosis.
Bioarchitecture. 2:75–87. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Grzanka A and Grzanka D: Distribution of
actin in etoposide-induced human leukemia cell line K-562 using
fluorescence and immunoelectron microscopy technique. Pol J Pathol.
53:43–50. 2002.PubMed/NCBI
|
|
59
|
Grzanka A, Grzanka D and Orlikowska M:
Cytoskeletal reorganization during process of apoptosis induced by
cytostatic drugs in K-562 and HL-60 leukemia cell lines. Biochem
Pharmacol. 66:1611–1617. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Grzanka A, Grzanka D, Zuryń A, Grzanka AA
and Safiejko-Mroczka B: Reorganization of actin in K-562 and HL-60
cells treated with taxol. Neoplasma. 53:56–61. 2006.PubMed/NCBI
|
|
61
|
Levee MG, Dabrowska MI, Lelli JL Jr and
Hinshaw DB: Actin polymerization and depolymerization during
apoptosis in HL-60 cells. Am J Physiol. 271:C1981–C1992. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Grzanka D, Grzanka A, Izdebska M,
Gackowska L, Stepien A and Marszalek A: Actin reorganization in CHO
AA8 cells under-going mitotic catastrophe and apoptosis induced by
doxorubicin. Oncol Rep. 23:655–663. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Erenpreisa J, Ivanov A, Wheatley SP,
Kosmacek EA, Ianzini F, Anisimov AP, Mackey M, Davis PJ, Plakhins G
and Illidge TM: Endopolyploidy in irradiated p53-deficient tumour
cell lines: Persistence of cell division activity in giant cells
expressing Aurora-B kinase. Cell Biol Int. 32:1044–1056. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Grzanka D, Gagat M and Izdebska M:
Involvement of the SATB1/F-actin complex in chromatin
reorganization during active cell death. Int J Mol Med.
33:1441–1450. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Grzanka D, Izdebska M,
Klimaszewska-Wisniewska A and Gagat M: The alterations in sATB1 and
nuclear F-actin expression affect apoptotic response of the MCF-7
cells to geldanamycin. Folia Histochem Cytobiol. 53:79–87. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Grzanka D, Kowalczyk AE, Izdebska M,
Klimaszewska-Wisniewska A and Gagat M: The interactions between
sATB1 and F-actin are important for mechanisms of active cell
death. Folia Histochem Cytobiol. 53:152–161. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Hoelzle MK and Svitkina T: The
cytoskeletal mechanisms of cell-cell junction formation in
endothelial cells. Mol Biol Cell. 23:310–323. 2012. View Article : Google Scholar :
|
|
68
|
Braga VM: Cell-cell adhesion and
signalling. Curr Opin Cell Biol. 14:546–556. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Matter K and Balda MS: Signalling to and
from tight junctions. Nat Rev Mol Cell Biol. 4:225–236. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Astudillo P and Larraín J: Wnt signaling
and cell-matrix adhesion. Curr Mol Med. 14:209–220. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Macdonald BT, Tamai K and He X:
Wnt/beta-catenin signaling: Components, mechanisms, and diseases.
Dev Cell. 17:9–26. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Itoh M, Nagafuchi A, Moroi S and Tsukita
S: Involvement of ZO-1 in cadherin-based cell adhesion through its
direct binding to alpha catenin and actin filaments. J Cell Biol.
138:181–192. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Gagat M, Grzanka D, Izdebska M and Grzanka
A: Effect of L-homocysteine on endothelial cell-cell junctions
following F-actin stabilization through tropomyosin-1
overexpression. Int J Mol Med. 32:115–129. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Gooding JM, Yap KL and Ikura M: The
cadherin-catenin complex as a focal point of cell adhesion and
signalling: New insights from three-dimensional structures.
BioEssays. 26:497–511. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Cavallaro U and Christofori G: Cell
adhesion and signalling by cadherins and Ig-CAMs in cancer. Nat Rev
Cancer. 4:118–132. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Willis ND, Cox TR, Rahman-Casañs SF, Smits
K, Przyborski SA, van den Brandt P, van Engeland M, Weijenberg M,
Wilson RG, de Bruïne A, et al: Lamin A/C is a risk biomarker in
colorectal cancer. PLoS One. 3:e29882008. View Article : Google Scholar : PubMed/NCBI
|