Introduction
A ‘paradoxical response’ to cancer treatment is the
emergence of unexpected new lesions and/or the progression of
existing lesions, despite appropriate and effective therapy. This
term is commonly used in association with anti-tuberculosis
treatment, where it describes a phenomenon observed in some
patients with pulmonary and pleural tuberculosis, tuberculous
lymphadenitis and intracranial tuberculoma (1–5).
‘Pseudo-progression’ is another term used to describe a phenomenon
in which lymphocytes activated by an immune checkpoint inhibitor
accumulate in a tumor to expand its shadow, appearing as
enlargement of the primary lesion or development of a new
metastatic lesion. A proportion of patients treated with nivolumab
exhibit pseudo-progression, in which transient disease progression
is followed by a response to an immune checkpoint inhibitor
(6–10). Cancer patients may respond
differently to chemotherapy by exhibiting a response,
deterioration, or occasionally simultaneous occurrence of both.
These different responses may be attributed to the heterogeneity of
the cancer. However, differences in the transient responses to
epidermal growth factor receptor-tyrosine kinase inhibitor
(EGFR-TKI) treatment are rare and, should such a phenomenon be
observed, it should not affect the evaluation of the therapeutic
effect or be considered as an indication for discontinuation of
treatment. We herein describe a case with a transient increase in
the amount of carcinomatous pleural fluid as a paradoxical response
to osimertinib therapy for T790M-mutated lung adenocarcinoma,
despite the primary lesion and pulmonary metastases responding well
to this treatment. The aim of the present study was to report this
phenomenon in order to raise physicians’ awareness of this rare
occurrence.
Case report
A 58-year-old male patient was referred to the Mito
Medical Center in January 2014 after experiencing dyspnea on
exertion for 1 month. The patient had been diagnosed 27 months
prior with adenocarcinoma in the upper lobe of the right lung. An
EGFR exon 19 deletion was identified. Due to multiple metastases in
both lungs, platinum-based chemotherapy and gefitinib therapy were
administered. As third-line therapy, the patient received nivolumab
therapy, but 3 months after the initiation of nivolumab therapy, a
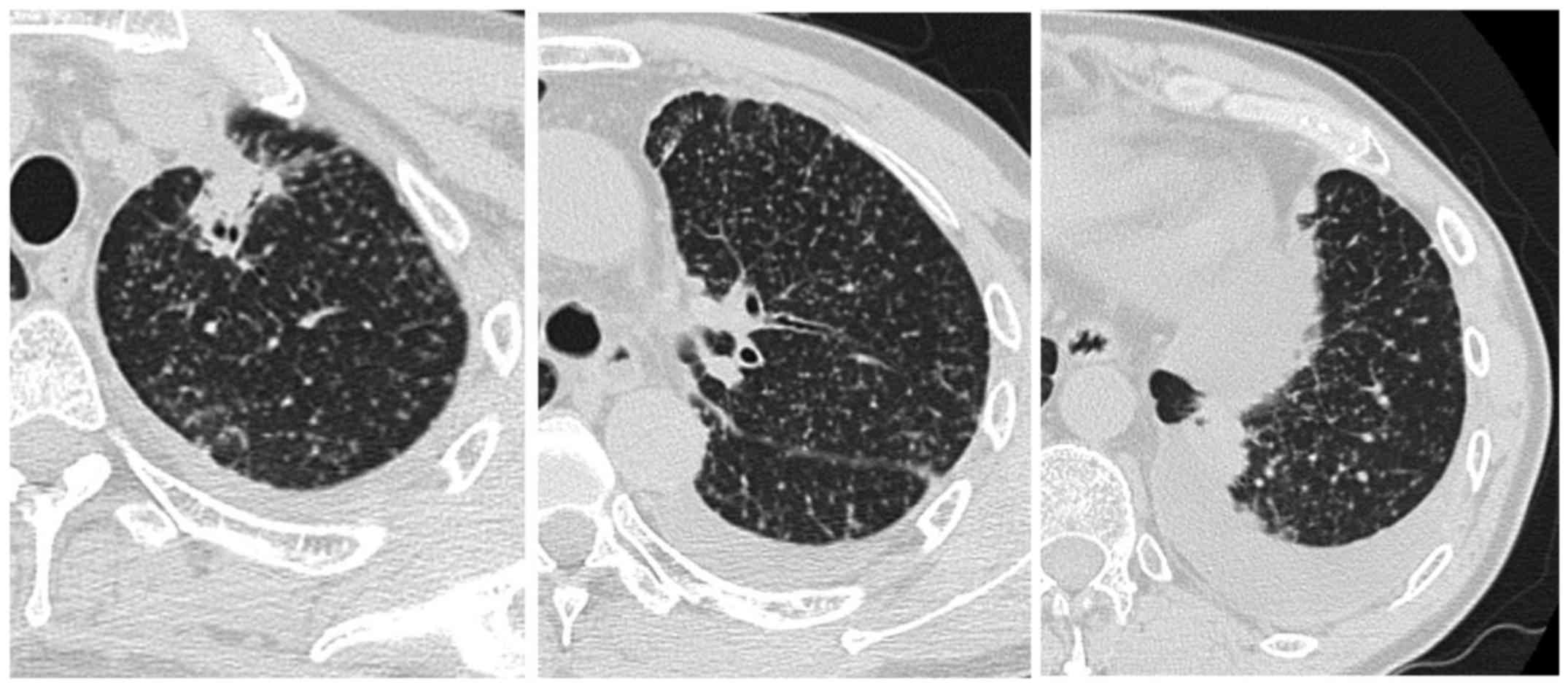
follow-up chest computed tomography (CT) scan revealed regrowth of
the primary pulmonary lesion in the left upper lobe and appearance
of miliary pulmonary metastases and bilateral pleural fluid
(Fig. 1). The serum carcinoembryonic
antigen (CEA) level increased from 252.4 to 340.2 ng/ml in 1 month.
Thoracentesis was performed to obtain cytological specimens.
Cytology of the pleural fluid revealed adenocarcinoma cells. A
T790M mutation, together with a deletion within exon 19 of the EGFR
gene, were identified. Osimertinib (80 mg once daily) was therefore
prescribed as fourth-line treatment for the adenocarcinoma. A chest
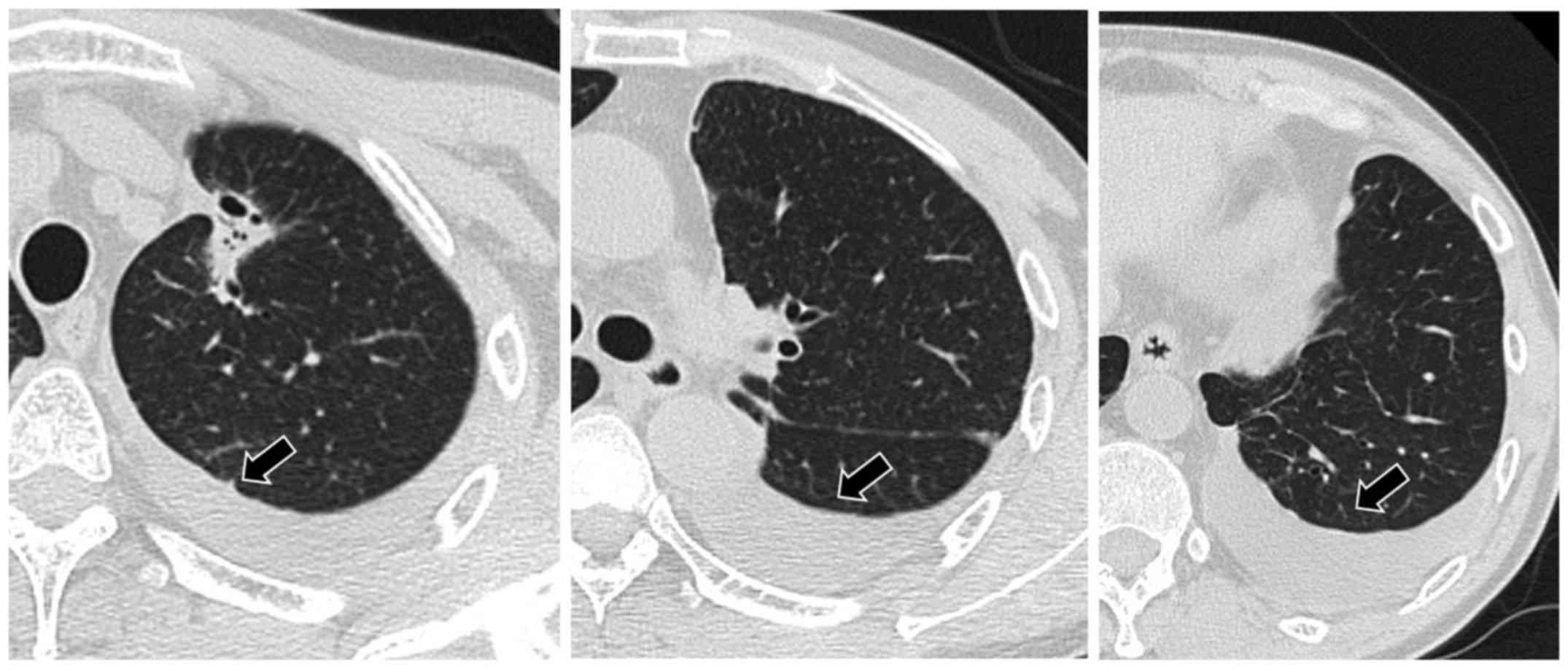
CT scan 1 month after the initiation of osimertinib treatment
revealed shrinkage of the primary lesion and disappearance of
pulmonary metastases, but an increase in the amount of pleural
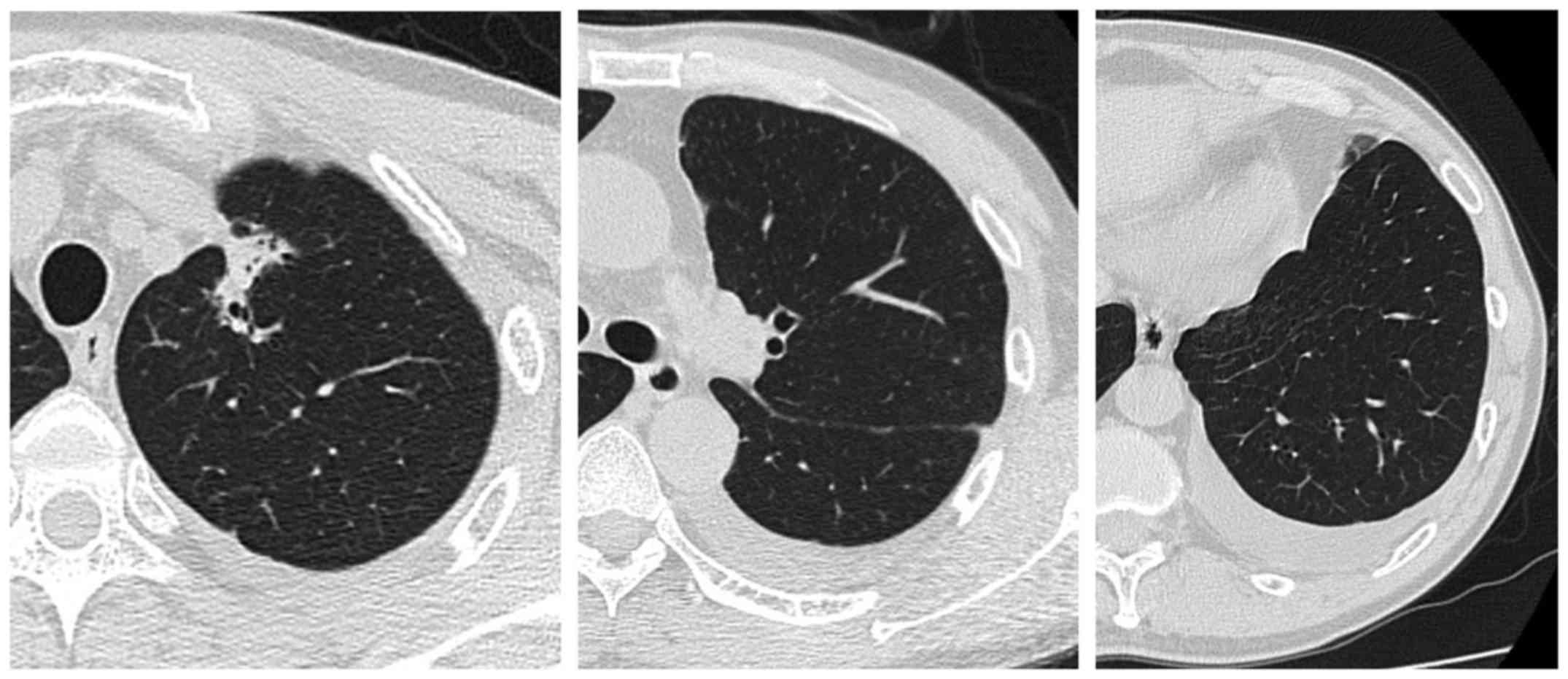
fluid (Fig. 2). A chest CT scan 1
month later revealed a decrease in the amount of the pleural fluid
and tumor shrinkage (Fig. 3). In
addition, the serum CEA level had decreased to 20.2 ng/ml. The
patient currently remains on osimertinib treatment, without major
adverse events or disease recurrence (last follow-up visit, June
2017).
Discussion
Pleural fluid is commonly found in a disseminated
form in lung adenocarcinoma patients with carcinomatous pleuritis.
However, to the best of our knowledge, an increase in the amount of
pleural fluid as a transient paradoxical response to osimertinib
therapy in a patient with EGFR-mutated lung cancer has never been
reported to date. A paradoxical aggravation response, which is a
phenomenon that appears despite effective chemotherapy for
tuberculosis, is generally considered immune-mediated (1–5). Active
tuberculosis leads to an immunosuppressed state via an altered
cell-mediated response (1–5). When active tuberculosis is controlled
by appropriate treatment, enhanced focal immune responses
(immunological rebound) mobilize lymphocytes and macrophages at the
site of lesions (2).
In a proportion of patients treated with nivolumab,
an IgG4 antibody that targets the programmed cell death protein-1
on the T-cell surface, there is pseudo-progression followed by
transient progression and then a response (6–8). This
immune-related tumor response may result in a temporary increase in
tumor size, regression, or emergence of new lesions, followed by a
response to subsequent treatment (10). The response itself may take longer
compared with that observed using conventional cytotoxic drugs
(10). This pseudo-progression may
be attributed to the inflammation, edema and necrosis that are
associated with immune cell infiltration of the tumor deposits
(9). This immunological response is
important for characterizing the effects of immune checkpoint
inhibitors, and is not observed with conventional cytotoxic drugs
or other targeted therapeutics (8).
The patient presented herein developed an increase in pleural fluid
after 1 month of osimertinib therapy for recurrent T790M-mutated
lung adenocarcinoma. The mechanism underlying this phenomenon has
not been fully elucidated, but it may be hypothesized that rapid
death of cancer cells following successful treatment of pulmonary
and pleural lesions released factors that stimulated the production
of pleural fluid. Another possibility is that increased pleural
fluid is caused by an imbalance between production and drainage. It
was unclear whether there was increased pleural fluid production,
impaired drainage of pleural fluid, or both. There was no evidence
to suggest pleurisy, other than carcinomatous pleuritis. The
pleural fluid was exudative, but there were no findings consistent
with infectious or tuberculous pleural fluid. It is paradoxical
that there was a temporary increase in the pleural fluid containing
T790M-positive cancer cells, despite the prompt response of the
pulmonary metastatic lesions to treatment. Recognizing this
clinically frustrating, but non-malignant paradoxical response, is
meaningful for clinicians, although treatment modification is
unnecessary. It is important that clinicians are aware of this rare
event, whilst simultaneously considering other causes of an
inadequate response to treatment, such as incorrect diagnosis,
inappropriate drug regimen, drug resistance, atypical infectious
disease, adverse effect to therapeutic drugs, or the deterioration
of the disease. An accumulation of knowledge in this aspect of
malignancy and cancer management will ensure continuing improvement
in patient care. Informed consent to the publication of the case
details and associated images was obtained from the patient.
References
|
1
|
Breen RA, Smith CJ, Bettinson H, Dart S,
Bannister B, Johnson MA and Lipman MC: Paradoxical reactions during
tuberculosis treatment in patients with and without HIV
co-infection. Thorax. 59:704–707. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Marshall BG and Chambers MA: Central
nervous system tuberculosis-the paradox of the host immune
response. J Infect. 36:3–4. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Gupta RC, Dixit R, Purohit SD and Saxena
A: Development of pleural effusion in patients during
anti-tuberculosis therapy: Analysis of twenty-nine cases with
review of literature. Indian J Chest Dis Allied Sci. 42:161–166.
2000.PubMed/NCBI
|
|
4
|
Bukharie H: Paradoxical response to
anti-tuberculosis drugs: Resolution with corticosteroid therapy.
Scand J Infect Dis. 32:96–97. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Al-Orainey IO: Diagnosis of latent
tuberculosis: Can we do better? Ann Thorac Med. 4:5–9. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ito K, Hataji O, Katsuta K, Kobayashi T,
Gabazza EC, Yatabe Y, Taguchi O and Yamamoto N: ‘Pseudoprogression’
of pulmonary pleomorphic carcinoma during nivolumab therapy. J
Thorac Oncol. 11:e117–e119. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kolla BC and Patel MR: Recurrent pleural
effusions and cardiac tamponade as possible manifestations of
pseudoprogression associated with nivolumab therapy- a report of
two cases. J Immunother Cancer. 4:802016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Tanizaki J, Hayashi H, Kimura M, Tanaka K,
Takeda M, Shimizu S, Ito A and Nakagawa K: Report of two cases of
pseudoprogression in patients with non-small cell lung cancer
treated with nivolumab-including histological analysis of one case
after tumor regression. Lung Cancer. 102:44–48. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Di Giacomo AM, Danielli R, Guidoboni M,
Calabrò L, Carlucci D, Miracco C, Volterrani L, Mazzei MA, Biagioli
M, Altomonte M and Maio M: Therapeutic efficacy of ipilimumab, an
anti-CTLA-4 monoclonal antibody, in patients with metastatic
melanoma unresponsive to prior systemic treatments: Clinical and
immunological evidence from three patient cases. Cancer Immunol
Immunother. 58:1297–1306. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Hoos A, Parmiani G, Hege K, Sznol M,
Loibner H, Eggermont A, Urba W, Blumenstein B, Sacks N, Keilholz U,
et al: A clinical development paradigm for cancer vaccines and
related biologics. J Immunother. 30:1–15. 2007. View Article : Google Scholar : PubMed/NCBI
|