Introduction
Multilocular cystic renal cell carcinoma (MCRCC) was
first reported by Perlmann in 1928 and named lymphangiohemangioma
(1). MCRCC is a rare entity,
comprising <2% of all renal carcinomas and it mainly occurs in
patients aged 30–80 years (1).
Histologically, MCRCC consists of multiple cysts containing old and
fresh hemorrhagic material, with clear cells in the septa
separating the cysts, which may make it indistinguishable from
clear-cell carcinoma (2). Although
MCRCC has non-specific symptoms, it is a tumor of low grade, with
low malignant potential; it is curable by surgical resection,
either simple nephrectomy or nephron-sparing surgery, and it has a
favourable prognosis compared with other types of renal cancer,
such as clear-cell and tubulocystic cancer (2). In this study, we present the case of a
28-year-old male patient, who, to the best of our knowledge, is the
youngest MCRCC patient reported to date. This study was approved by
the Ethics Committee of Peking University Shenzhen Hospital
(Shenhen, China) and written informed consent was obtained from the
patient for the publication of the case details.
Case report
A 28-year-old male patient was admitted to the
Department of Urology of Peking University Shenzhen Hospital
(Shenzen, China) on May 28, 2015, with the chief complaint of
urinary irritant symptoms for 1 year. The patient was a non-smoker,
of normal built and normal nutritional status. There was no
abdominal pain, no changes in appetite and/or weight variation, and
no cardiovascular or respiratory symptoms; he had not undergone any
surgery and there was no family history of disease. The patient had
a pulse rate of ~84 beats per min, a temperature of 36.3°C, a blood
pressure of 110/73 mmHg and a respiratory rate of 20 breaths per
min. Physical examination revealed no positive signs.
Laboratory examination revealed a white blood cell
count of 9.59×109/l and a haemoglobin level of 157 g/l.
The serum glucose level was 6.13 mmol/l, the blood urea nitrogen
was 7.03 mmol/l and the creatinine level was 64.4 µmol/l. The blood
coagulation parameters and liver function were within the normal
range. The chest X-ray revealed no abnormalities. Urinalysis was
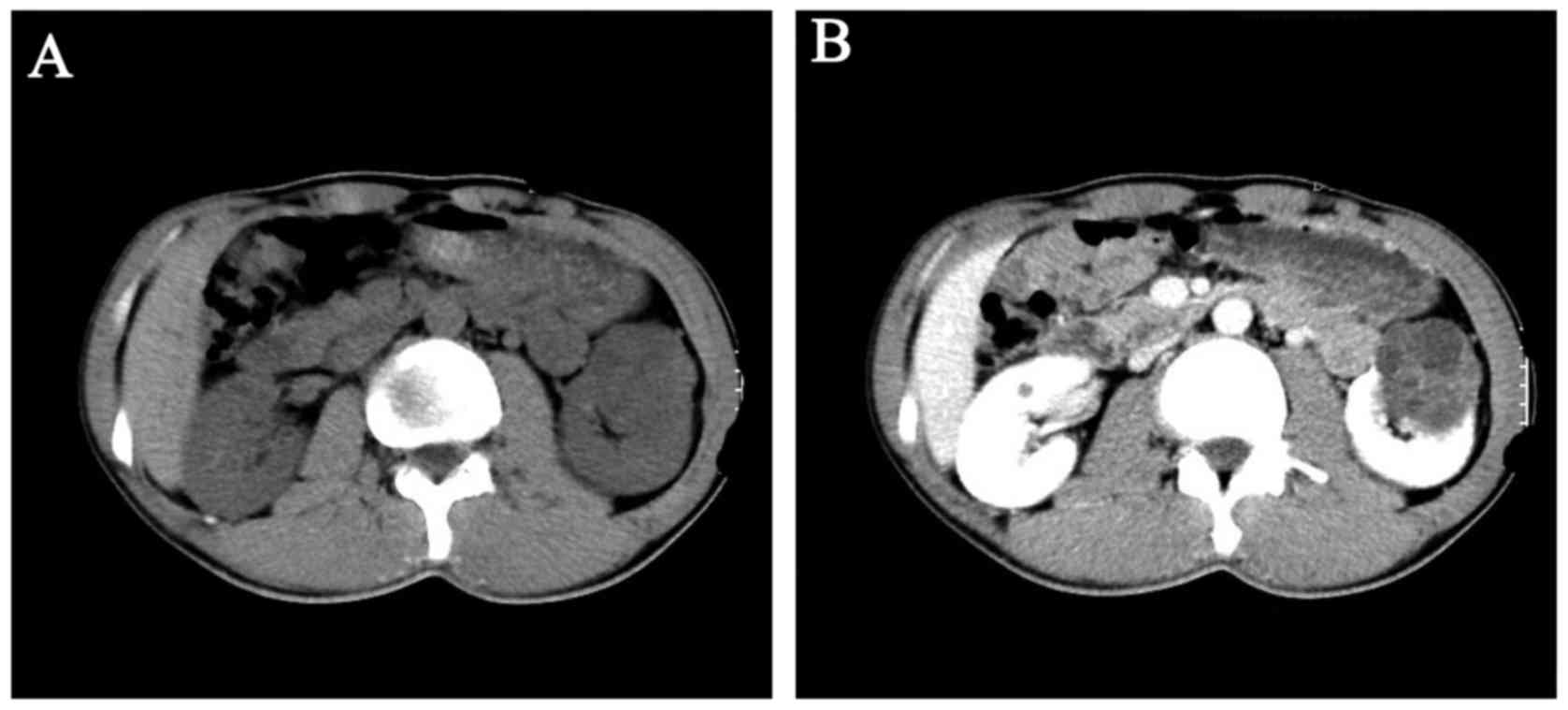
also normal. A 4.2×4.9-cm round isodense mass (24–41 HU) in the
lower pole of the left kidney was identified by a non-contrast CT
scan (Fig. 1A), presenting as a
heterogeneously enhanced mass (39–64 HU) on contrast-enhanced CT
(Fig. 1B).
The patient underwent left radical nephrectomy on
June 5, 2015. Invasion of the surrounding tissue was not observed
intraoperatively, whereas several cysts of different sizes were
identified. Grossly, the mass was well-encapsulated. The
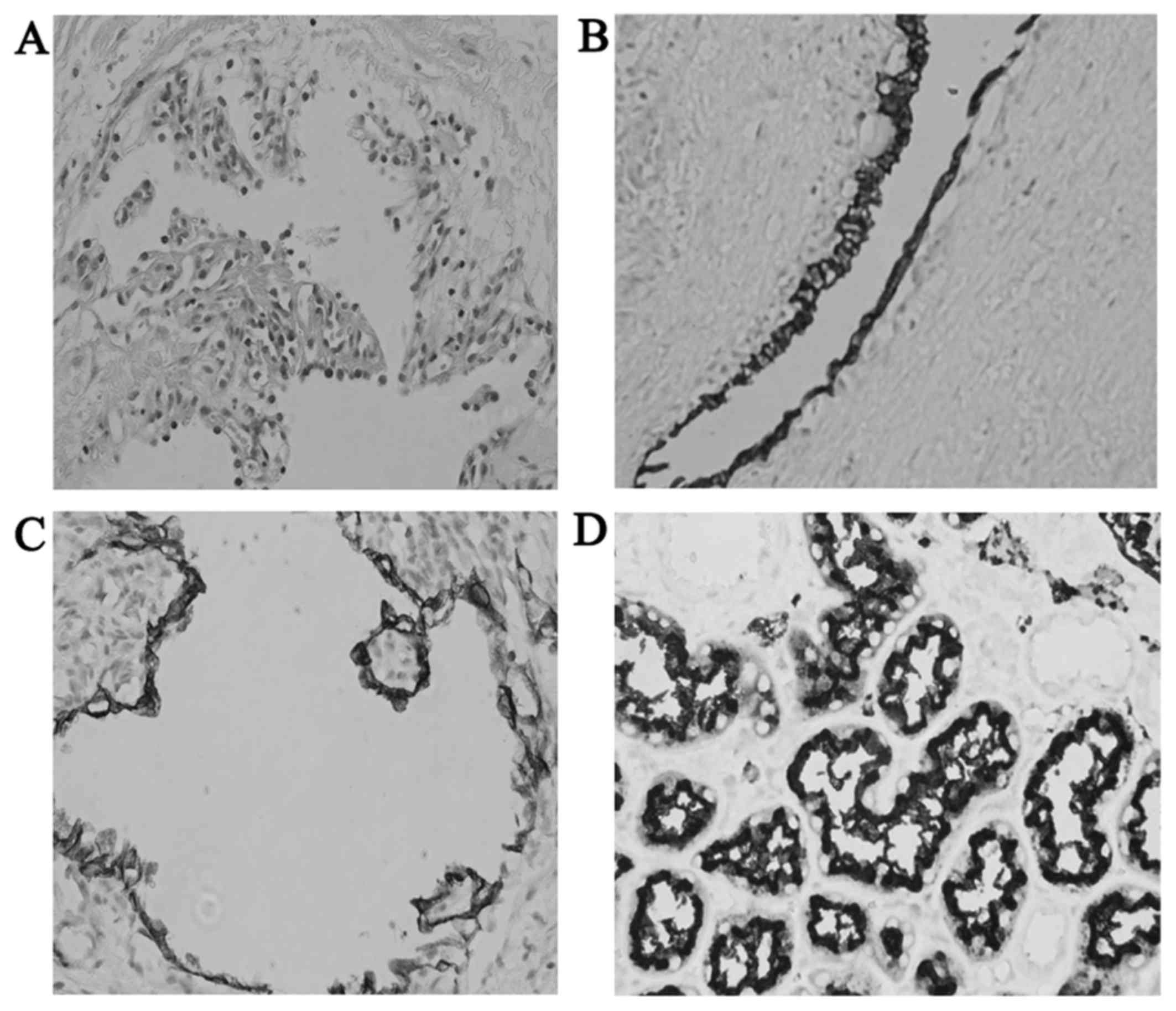
pathologist observed that the clear tumor cells adhered to the
inner wall of the cyst and their nuclei were significantly
hyperchromatic (Fig. 2A). Papillary
growth was identified partially and the cysts were totally
separated by fibrous septae. The immunohistochemical staining
examination (Fig. 2B-D) revealed
that the tumor cells were positive for cytokeratin (CK), CK7 and
epithelial membrane antigen. The final diagnosis was confirmed to
be MCRCC, according to the reference standards of the 2004 World
Health Organization classification (https://www.iarc.fr/en/publications/pdfs-online/pat-gen/bb7/BB7.pdf)
and the recommendations of Ebin and Bonsib (3). Magnetic resonance imaging and sentinel
lymph node biopsy revealed no other metastatic lesions.
The patient was discharged on the sixth
postoperative day and he had a good recovery. He has since been
routinely followed up and remains alive without evidence of
recurrence.
Discussion
MCRCC was reported in the 2004 WHO classification
and known to have a favourable prognosis (4,5). MCRCC
is a rare type of clear cell RCC, accounting for 2% of all kidney
cancers (2) and its prognosis is
relatively better compared with that of other malignant kidney
tumors (6). The age of patients at
diagnosis is >30 years, with the mean age >55 years (6,7). The
morbidity increases gradually with advancing age. It was previously
reported that the incidence in men is higher compared with that in
women, with a ratio of 3:1 (7).
Microscopically, MCRCC, which is pathologically characterized by a
single layer of low-grade neoplastic clear cells in its cyst wall,
is frequently located in the upper or lower pole of the kidney. The
typical clinical characteristics (gross hematuria, flank pain and a
palpable mass) closely resemble those of RCC; thus, MCRCC may be
misdiagnosed as conventional RCC by less clinically experienced
doctors (6). The pathogenesis of
MCRCC remains unknown at present, with certain scholars suggesting
that intrinsic multilocular cystic growth and papillary cystic
adenocarcinoma are strongly associated with the occurrence of this
disease. From the perspective of molecular biology, deletions in
chromosome 3p may play a significant role in development and
progression; these changes have been reported in 80% of MCRCCs and
are closely associated with a worse result (8). Another cancer suppressor gene, referred
to as the Von Hippel-Lindau gene, which is located in the 3p25
chromosomal area, has a major effect on carcinogenesis (9).
To the best of our knowledge, elderly patients
presenting with indolent gross hematuria may suffer from urinary
tract tumors. Contrast-enhanced CT is required for such patients to
detect highly vascular renal parenchymal tumors. The CT attenuation
values of benign tumors is generally <15 HU (10). However, patients with enhanced
lesions (>39 HU) are highly suspicious for RCC. We must bear in
mind that CT cannot reliably confirm the diagnosis of MCRCC, and
pathological confirmation is always required.
Since the primary characteristics of MCRCC are low
grade and stage, the outcome is generally favorable. The majority
of previously published cases were stage I and surgical resection
was timely performed. Tumor size was not found to be associated
with patient survival time (5).
Acknowledgements
The present study was supported by Science and
Technology Development Fund Project of Shenzhen (grant nos.
JCYJ20150403091443329 and JCYJ20170307111334308), the fund of
‘San-ming’ Project of Medicine in Shenzhen (SZSM201612066), and the
fund of Guangdong Key Medical Subject.
References
|
1
|
Cheng L and Huang W: Multilocular cystic
clear cell neoplasm of low malignant potential: Evolving concept,
classification and unifying terminology. Zhonghua Bing Li Xue Za
Zhi. 43:721–722. 2014.(In Chinese). PubMed/NCBI
|
|
2
|
Wahal SP and Mardi K: Multilocular cystic
renal cell carcinoma: A rare entity with review of literature. J
Lab Physicians. 6:50–52. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ebin JN and Bonsib SM: Extensively cystic
renal neoplasms: Cystic nephroma, cystic partially differentiated
nephroblastoma, multilocular cystic renal cell carcinoma, and
cystic hamartoma of renal pelvis. Semin Diagn Pathol. 15:2–20.
1998.PubMed/NCBI
|
|
4
|
Williamson SR, MacLennan GT, Lopez-Beltran
A, Montironi R, Tan PH, Martignoni G, Grignon DJ, Eble JN, Idrees
MT, Scarpelli M and Cheng L: Cystic partially regressed clear cell
renal cell carcinoma: A potential mimic of multilocular cystic
renal cell carcinoma. Histopathology. 63:767–779. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tamura Y, Okamura K, Ogura H, Kawada T,
Tsuji H, Takahashi Y, Fukabori Y, Kobayashi M, Imai K and Yamanaka
H: Multilocular cystic renal cell carcinoma: A report of 2 cases.
Hinyokika Kiyo. 36:437–441. 1990.(In Japanese). PubMed/NCBI
|
|
6
|
Ghosh P and Saha K: Multilocular cystic
renal cell carcinoma: A rare, unique entity and diagnostic
challenge. Arch Iran Med. 17:129–132. 2014.PubMed/NCBI
|
|
7
|
Chowdhury AR, Chakraborty D, Bhattacharya
P and Dey RK: Multilocular cystic renal cell carcinoma a diagnostic
dilemma: A case report in a 30-year-old woman. Urol Ann. 5:119–121.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yamaguchi S, Yoshihiro S, Matsuyama H,
Nagao K, Fukunaga K, Matsumoto H, Matsuda K, Oba K and Naito K: The
allelic loss of chromosome 3p25 with c-myc gain is related to the
development of clear-cell renal cell carcinoma. Clin Genet.
63:184–191. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Halat SK and MacLennan GT: Multilocular
cystic renal cell carcinoma. J Urol. 177:3432007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Imura J, Ichikawa K, Takeda J, Tomita S,
Yamamoto H, Nakazono M, Takimoto T, Ueda Y and Fujimori T:
Multilocular cystic renal cell carcinoma: A clinicopathological,
immuno- and lectin histochemical study of nine cases. APMIS.
112:183–191. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Singhai A, Babu S, Verma N and Singh V:
Multilocular cystic renal cell carcinoma: A rare entity. BMJ Case
Rep. 2013:pii: bcr20120084572013. View Article : Google Scholar
|
|
12
|
Deger AN, Capar E, Ucar BI, Deger H and
Tayfur M: Adult multicystic nephroma: Case report and review of the
literature. J Clin Diagn Res. 9:ED22–ED23. 2015.PubMed/NCBI
|
|
13
|
Guo A, Wei L, Song X and Liu A: Adult
wilms tumor with intracaval and intracardiac extension: Report of a
case and review of literature. J Cancer. 2:132–135. 2011.
View Article : Google Scholar : PubMed/NCBI
|